- 1Clinical Neurorehabilitation, Department of Neurology, University of Zurich, Zurich, Switzerland
- 2Rehabilitation Initiative and Technology Center Zurich, RITZ, Zurich, Switzerland
- 3Department of Neurology, Johns Hopkins University, Baltimore, MD, USA
Although the architecture of a dopaminergic (DA) system within the primary motor cortex (M1) was well characterized anatomically, its functional significance remained obscure for a long time. Recent studies in rats revealed that the integrity of DA fibers in M1 is a prerequisite for successful acquisition of motor skills. This essential contribution of DA for motor learning is plausible as it modulates M1 circuitry at multiple levels thereby promoting plastic changes that are required for information storage: at the network level, DA increases cortical excitability and enhances the stability of motor maps. At the cellular level, DA induces the expression of learning-related genes via the transcription factor c-Fos. At the level of synapses, DA is required for the formation of long-term potentiation, a mechanism that likely is a fingerprint of a motor memory trace within M1. DA fibers innervating M1 originate within the midbrain, precisely the ventral tegmental area (VTA) and the medial portion of substantia nigra (SN). Thus, they could be part of the meso-cortico-limbic pathway – a network that provides information about saliency and motivational value of an external stimulus and is commonly referred as “reward system.” However, the behavioral triggers of the release of dopamine in M1 are not yet identified. As alterations in DA transmission within M1 occur under various pathological conditions such as Parkinson disease or ischemic and traumatic brain injury, a deeper understanding of the interaction of VTA/SN and M1 may reveal a deeper insight into a large spectrum of neurological disorders.
Introduction
The primary motor cortex (M1) receives dopaminergic (DA) projections from mesencephalic brainstem nuclei. The integrity of this DA meso-cortical pathway has been recently shown to be a prerequisite of successful motor learning in rats. Apart from providing details on morphology and behavioral experiments, we describe how DA signaling facilitates plastic changes within M1 circuitry at various levels thereby promoting storage of newly acquired movement sequences. In addition to putative behavioral triggers and dynamics of DA release in M1, the assignability of knowledge gained in rodent models to human subjects becomes discussed. Finally, we provide an overview of M1 plasticity in neurological diseases characterized by a DA deficit.
DA in M1 is Necessary for Motor Learning in Rats
Apart from prefrontal region (PFC), sensorimotor areas receive the largest amount of DA innervation within the rodent neocortex (1). In M1, DA terminals are distributed inhomogeneously with a preference for deep cortical layers [layer V/VI (2)]. Regarding postsynaptic elements, D1 receptors are expressed in both, superficial (layers I, II, IIIa) and deep (V and VI) layers (3), whereas D2-receptors are expressed primarily in layer V but 10-fold less than D1 (4). Additionally, co-localization of both receptor subtypes were observed in layer V/VI motor neurons (5). Retrograde tracing identified the ipsilateral DA midbrain nuclei to be the origin of motor-cortical DA innervation: about 73% of DA midbrain neurons projecting to M1 are located in the rostro-lateral ventral tegmental area (VTA, also referred to as region A10) whereas a smaller amount of neurons can be found in the rostro-medial substantia nigra [SN; also referred to as region A9; 12%; (6)].
To define the functional role of this DA system, we assessed the effect of removing DA terminals within M1 on acquisition of a motor skill-learning paradigm (6, 7). Rats were trained in a single-pellet reaching task (8), while selective destruction of DA terminals was performed by injecting 6-hydroxi-dopamine (6-OHDA) either directly in M1 or within the rostral VTA/SN, both contralateral to the preferred paw. DA depletion of M1 resulted in an impaired gain in motor performance between subsequent training sessions (inter-session learning) when compared to controls. This impairment could be resolved by substituting levodopa directly within M1 using osmotic mini-pumps. As the short-term improvement of performance within one session (intra-session learning) was not affected by lesions, DA seems to be required for longer-lasting storage mechanisms underlying motor memory consolidation (9). Inducing 6-OHDA lesions in rats that already learned the task did not influence reaching performance, indicating a role of DA for movement acquisition but not for movement execution. As the effect of 6-OHDA lesions could be mimicked by injection of the D1-receptor antagonist SCH-29930 or the D2-receptor antagonists raclopride and sulpiride, motor learning seems to depend on both receptor subtypes (7). Whereas direct 6-OHDA lesions in M1 resulted only in a gradual but significant impairment of motor skill acquisition, destroying the origin of the motor-cortical DA innervation in VTA/SN completely abolished motor learning. This can be explained by a higher effectiveness of DA depletion caused by brainstem lesions when compared to direct injection of 6-OHDA into M1. Remarkably, 6-OHDA lesions within the midbrain could be applied with sufficient precision to avoid any side effects like motivational deficits or extrapyramidal symptoms that would be expected in case of damaging projections to the PFC or to striatum. In summary, the integrity of DA projection from midbrain to M1 is a prerequisite of successful movement acquisition based on D1- and D2-receptor mediated mechanisms.
How DA May Promote Plasticity in M1
Although extra-cortical brain regions as cerebellum and basal ganglia contribute to motor learning (10), M1 is thought to be the place where motor memory becomes stored (11). This motor memory storage depends on M1 ability to undergo experience-dependent changes, a phenomenon commonly referred to as motor plasticity (12). DA has been shown to modulate M1 circuitry at several levels thereby affecting various processes of motor-learning dependent plasticity.
Level of M1 Network-Physiology
Motor learning induces an enlargement of the motor-cortical representation (motor map) of particular body-parts that became trained, a phenomenon observed in rodents, primates, and humans (13–15). This enlargement is learning specific as it does not occur in response to mere motor activation and its magnitude is proportional to learning success (16, 17). The enlargement of motor representations furthermore depends on intact signaling of modulatory neurotransmitters: preventing cholinergic transmission by destroying basal forebrain structures in rats abolished both, expansion of motor maps and skill acquisition (18). Comparably, intra-motor-cortical injection of the D2-receptor antagonist raclopride induced a collapse of motor representations evoked by epidural electrical microstimulation in rats, whereas blocking D1 receptors with SCH 23390 had no effects (19).
Besides the spatial expansion of motor maps, transcranial magnetic stimulation (TMS) in humans revealed a training-related increment in M1 excitability in response to learning a piano sequence (15). In rats, blocking D2-receptors by intra-cortical injection of raclopride significantly increased stimulation thresholds necessary to evoke motor responses indicating a decreased level of motor-cortical excitability (19). Thus, by stabilizing motor representations and by increasing cortical excitability via D2-receptor dependent processes, DA signaling in M1 seems to be ideally suited to support learning-related changes on M1 network level.
Level of Gene Expression
Motor learning requires protein synthesis within M1 neurons (20) as a prerequisite of subsequent learning-dependent changes including increased spine-turnover and dendritic growth (11, 12). In M1 of rats, expression of the transcription factor c-Fos becomes induced in response to learning an acrobatic skill (21). Because c-Fos expression was highest during skill acquisition and subsequently decreased in the maintenance phase, the learning-specificity of this phenomenon is highly plausible. c-Fos is considered to be not only a marker of recent neuronal activation but also for experience-dependent changes (22) and is known to become induced by DA signaling (23). Electrical stimulation of neurons within the rostro-lateral VTA induces an increased expression of c-Fos within the ipsilateral M1 that can be blocked by intra-cortical injection of the D1- and D2-receptor antagonists SCH 23990 and raclopride (6). Thus, DA efferents from the midbrain are capable to support the expression of learning-relevant proteins in M1.
Level of Synaptic Transmission
In rats, motor skill learning induces a long-lasting increase of synaptic strength in M1 horizontal connections of layer II/III suggesting an association with long-term potentiation (LTP)-like plasticity (24). In line with this assumptions, capacity to induce LTP within these connections was reduced whereas long-term depression (LTD) was increased, suggesting that the learning-induced gain in synaptic strength expended the capacity of LTP-formation (25). Several weeks after skill acquisition, the ability to form LTP was restored while the horizontal connections of layer II/III remained strengthened (26). Bath application of both, the D1-receptor antagonist SCH 29339 and the D2-receptor antagonist raclopride markedly reduced the ability of M1 horizontal connections to form LTP (7), suggesting the necessity of intact DA signaling for long-lasting synaptic plasticity in M1.
DA Outside of M1
As tetanic stimulation of somatosensory cortex (S1) mediates LTP induction in M1 (27), it is assumed that motor-learning dependent synaptic plasticity is mediated by somatosensory input (28, 29). Thus, a close and precise interaction between M1 and S1 is a prerequisite for successful motor learning (28). In S1, intra-cortical injection of both, D1- and D2-receptor antagonists induced an enlargement of somatosensory evoked potential (SEP)-amplitude consistent with an increased cortical excitability (30). Thus, by reducing S1 excitability, DA may serve focusing on relevant (= strong) somatosensory input thereby improving signal-to-noise ratio (31) and improving sensory discrimination (32). However, if improved somatosensory information processing in sensorimotor cortex could also be one mechanism by which DA facilitates motor learning remains to be tested.
What Do Dopaminergic Signals in M1 Encode?
Dopaminergic neurons projecting to M1 are located in the rostro-lateral VTA and, to a lesser extent, in the rostro-medial SN (6). Thus, this projection is part of the meso-cortico-limbic system that provides DA input to cortical (mainly prefrontal cortex) and limbic (e.g., amygdala and hippocampus) structures (33, 34). In general, this system is thought to evaluate environmental stimuli with respect to their value and behavioral significance. Whereas the activity of specific neuronal subgroups is coupled to a particular content like motivational value or saliency (35), the time course of dopamine release may encode if a stimulus is pleasant (“rewarding”) or aversive [“punishing”; (36, 37)]. In rodents, DA projections to PFC critically modulate spatial working memory (38, 39) and attention selection (40). In a computational model that simulates the effect of DA on PFC network (39), low concentrations of DA constitute a “gate closed” state characterized by a boosted inhibitory drive and stereotypical behavior. At higher concentrations, DA induces a “gate open” state characterized by an increased excitability of the network and consecutive explorative behavior. Based on these considerations, one may hypothesize that the DA midbrain-to-M1 projection facilitates the occurrence of plastic changes within the motor-cortical network in response to salient, novel, or appetitive stimuli. However, it is illusive which environmental cues during motor learning are capable of activating midbrain neurons that trigger the release of DA in M1.
In contrast to DA neurons forming the nigro-striatal pathway, meso-cortically projecting neurons are characterized by fast firing (30 Hz), slow dopamine reuptake and reduced if not lacking D2-receptor dependent auto-inhibition (41). This class of DA cells therefore seems to be well suited to provide a sustained release of DA. In addition, voltammetric studies revealed that the clearance of extracellular DA within the amygdala or the PFC is considerably slower when compared to striatum (42, 43). Thus, DA projections to cortical and limbic structures rather seem to influence DA concentrations on a larger time-scale. This tonic form of modulatory neurotransmission would be well suited to support longer-lasting changes required for motor memory consolidation such as LTP-like plasticity. However, the kinetics of DA release and clearance within M1 still remain to be experimentally established.
Can These Findings be Assigned to Humans?
Notable differences in magnitude and organization have to be taken into account between species regarding DA innervation. At first, the midbrain of primates harbors three to seven times more DA neurons when compared to rodents (44). Thus, cortical – and especially motor-cortical – DA innervation is accordingly much more pronounced in primates (45, 46). Furthermore, cortically projecting DA neurons in primates can be found beyond VTA and SN: in owl and rhesus monkeys, 15% of DA efferents toward M1 are located within the retrorubral field [RRF, also referred to as region A8; (2, 47)], a region that completely lacks meso-cortically projecting neurons in rodents (48, 49). Finally, whereas DA terminals are homogenously distributed among cortical layers in primates, deeper layers receive stronger DA innervation in rodents than superficial ones (2, 50). Taken together, presumably as a consequence of neocortical development, DA innervation of cortex underwent a gain in complexity during phylogeny. However, no significant differences in quantity and distribution of DA terminals in M1 could be detected for non-human primates and humans (51).
Despite these pronounced differences between species, DA signaling in humans seems to have similar function for motor learning when compared to the rodent model: in humans, administration of a single dose of levodopa or the D2-receptor agonist cabergoline facilitated the encoding of an elementary motor memory in M1 [influencing the direction of TMS – evoked thumb movement by prior training; (50, 52)]. On the other hand, blocking DA-receptors with haloperidol interfered with motor learning (52). The magnitude of this levodopa-induced improvement in movement acquisition depends on multiple factors like age (50, 53) and genetic variations of DA metabolizing enzymes and DA receptor isoforms (54). Thus, a beneficial effect of external DA stimulation is mainly expectable when the DA system is challenged due to degenerative processes associated with (healthy) aging or genetic polymorphisms related to a low effectiveness of DA transmission.
Although the cited studies in humans are in a good agreement with findings derived from animal research, transfer of knowledge across species is limited due to differences in experimental methodology and the increased complexity of the DA system in higher-order species. For example, intra-cortical injection of the D2-receptor antagonist raclopride reduces M1 excitability in rats (19) whereas administration of DA antagonists in humans has the opposite effect (52, 55, 56). However, as intra-cortical injection in the animal model is expected to induce only local changes, systemic application of drugs will influence several systems thereby causing a different net effect. In this example, blocking striatal DA-receptors may disinhibit excitatory thalamo-cortical projections thereby increasing M1 excitability (57, 58). Furthermore, the effect of exogenous DA stimulation on motor learning in humans depends on various genetic polymorphisms within cleaving enzymes (e.g., catechol-O-methyltransferase), receptor isoforms and reuptake transporters (54). Thus, administration of levodopa may range from beneficial to disadvantageous effects depending on the individual genetic profile. In contrast to humans, genetic profiles in rodent inbred stems are expected to be quite homogenous. Thus, it is not surprising that results in human studies spread around a larger range when compared to animal experiments.
The Role of DA for M1 Plasticity in Neurological Diseases
In Parkinson disease (PD), degeneration of DA neurons projecting to the neocortex occurs early in the course of disease (59, 60) and leads to a 70% reduction of DA fibers within M1 and other frontal cortical areas (61). Apart from the loss of nigro-striatal DA projections causing the classical extrapyramidal symptoms of PD like stiffness, tremor, and bradykinesia (62), PD patients also suffer from motor-learning deficits (63, 64). In line with these observations, capability of M1 to undergo plastic changes in response to TMS paired associative stimulation or theta-burst stimulation protocols is abolished in PD patients off DA medication (65–69). Interestingly, substitution of a single dose of l-DOPA rescued LTP- (and LTD)-like plasticity only in patients on chronic DA medication (65, 67, 68) but not in newly diagnosed PD patients naïve to l-DOPA (66). Thus, restoration of M1 plasticity likely reflects a long-duration effect of l-DOPA treatment, whereas reduction of extrapyramidal symptoms that occurs immediately after l-DOPA substitution depends on a short-duration response (65). In PD patients with a long-lasting course of disease suffering from fluctuations (e.g., wearing off phenomenon) or dyskinesia (e.g., peak of dose dyskinesia) l-DOPA dependent restoration of M1 plasticity is deficient (67) and even dysfunctional effects on plasticity-formation occur in response to l-DOPA administration (65). Taken together, M1 capability for plasticity-formation in PD patients depends on duration of disease (= degree of denervation) and persistent effects of DA treatment. If these phenomena can be explained by the degeneration of DA fibers directly within M1, by indirect effects on M1 circuitry (e.g., changes in striato-thalamo-cortical signaling) or by a sum effect of both is still an open question.
Apart from PD where DA neurons irreversibly degenerate, a state of a functional “DA deficit” may also emerge in case of brain injury (53, 54). After experimental application of traumatic brain lesions in rats, a sustained down-regulation of DA reuptake transporters (DAT) and increased concentrations of the DA synthesizing enzyme tyrosine hydroxylase (TH) within the frontal cortex indicate profound alterations of DA transmission in response to mechanical damage (70, 71). If ischemic brain injury leads to similar widespread changes in DA signaling has not been investigated, yet. However, chronic stroke survivors also profited from l-DOPA administration in acquiring an elementary motor-learning paradigm (72) and levodopa treatment in combination with physiotherapy was superior to physiotherapy alone in hemiparetic stroke patients (73, 74).
Conclusion and Future Research
In rats, meso-cortical DA projections are required for successful motor learning (Table 1: 1–3). By promoting plasticity at the level of learning-related gene expression, synaptic transmission, and network-physiology, DA signaling is well suited to facilitate the storage of novel movement sequences within M1 circuitry. Based on present literature, we propose that mesencephalic DA neurons innervating M1 are activated by novel and salient external stimuli, exerting a tonic and longer-lasting shift in cortical DA concentrations. Thus, the DA meso-cortical pathway is thought to support M1 circuitry to adapt to changes in environmental requirements. However, this hypothesis needs further experimental confirmation: combining telemetric recording of VTA activity and M1 field potentials during a skilled reaching task could reveal the behavioral cue preceding the activation of M1-projecting mesencephalic neurons. Furthermore dynamics of DA signaling within M1 could be assessed using voltammetric measurements.
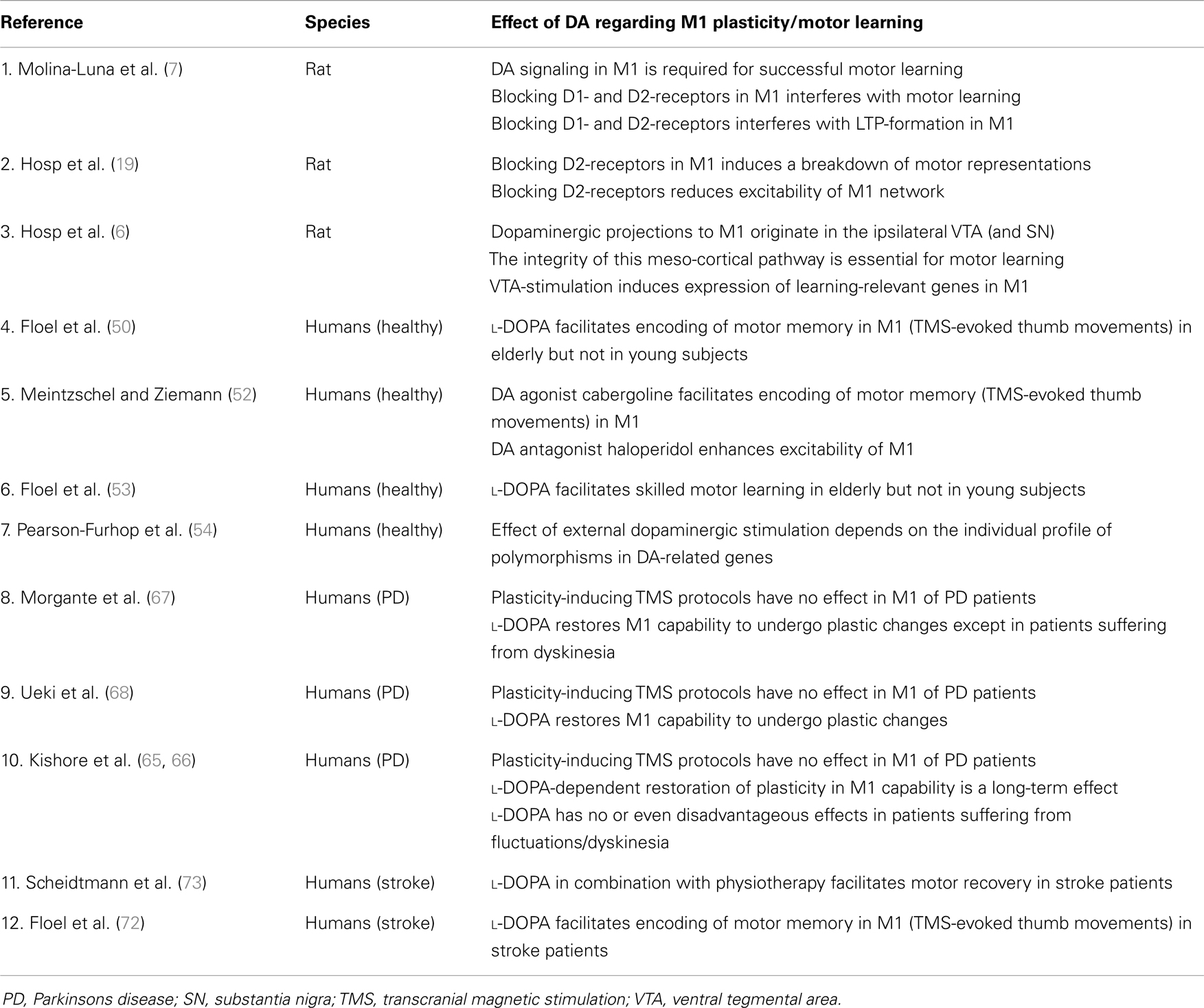
Table 1. Collection of key papers studying the effect of dopaminergic signaling on plasticity in M1 and/or motor learning.
In human subjects, the specific role of the meso-cortical projections toward M1 can be hardly defined, as alterations (systemic administration of drugs or degeneration of DA neurons) usually affect the entire DA system. However, DA signaling seems to influence M1 plasticity and motor learning in a quite similar way when compared to the rodent model: administration of l-DOPA improves the encoding of elementary motor memory in healthy subjects characterized by a low effectiveness of DA signaling (e.g. due to aging process; Table 1: 4–6). In patients suffering from PD, induction of plastic changes using TMS is severely impaired, a phenomenon that can be reversed by administration of l-DOPA at least in the early course of disease (Table 1: 8–10). In future research on humans, special attention should be paid to genetic variability with respect of DA receptor isoforms and DA cleaving or metabolizing enzymes as this molecular profile substantially shapes the effect of exogenous DA stimulation (Table 1: 7).
Apart from PD where l-DOPA substitution is the medication of choice, a DA deficiency requiring intervention may also occur in case of ischemic (or traumatic) brain injury (Table 1: 11–12). To test this hypothesis, changes on key determinants of DA signaling could be assessed in response to experimentally applied ischemic lesions (M1) in rats with respect to peri-infarct cortex and target regions of the DA system remote to the lesion side. Besides the valuable insight into long-term consequences of ischemic stroke on modulatory transmitter systems, these experiments could provide strong support for the application of l-DOPA in stroke survivors highlighting the requirement for further clinical trials.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
1. Berger B, Verney C, Alvarez C, Vigny A, Helle KB. New dopaminergic terminal fields in the motor, visual (area 18b) and retrosplenial cortex in the young and adult rat. Immunocytochemical and catecholamine histochemical analyses. Neuroscience (1985) 15(4):983–98. doi: 10.1016/0306-4522(85)90248-9
2. Berger B, Gaspar P, Verney C. Dopaminergic innervation of the cerebral cortex: unexpected differences between rodents and primates. Trends Neurosci (1991) 14(1):21–7. doi:10.1016/0166-2236(91)90179-X
3. Savasta M, Dubois A, Scatton B. Autoradiographic localization of D1 dopamine receptors in the rat brain with [3H]SCH 23390. Brain Res (1986) 375(2):291–301. doi:10.1016/0006-8993(86)90749-3
4. Ariano MA, Fisher RS, Smyk-Randall E, Sibley DR, Levine MS. D2 dopamine receptor distribution in the rodent CNS using anti-peptide antisera. Brain Res (1993) 609(1–2):71–80. doi:10.1016/0006-8993(93)90857-J
5. Awenowicz PW, Porter LL. Local application of dopamine inhibits pyramidal tract neuron activity in the rodent motor cortex. J Neurophysiol (2002) 88(6):3439–51. doi:10.1152/jn.00078.2002
6. Hosp JA, Pekanovic A, Rioult-Pedotti MS, Luft AR. Dopaminergic projections from midbrain to primary motor cortex mediate motor skill learning. J Neurosci (2011) 31(7):2481–7. doi:10.1523/JNEUROSCI.5411-10.2011
7. Molina-Luna K, Pekanovic A, Rohrich S, Hertler B, Schubring-Giese M, Rioult-Pedotti MS, et al. Dopamine in motor cortex is necessary for skill learning and synaptic plasticity. PLoS One (2009) 4(9):e7082. doi:10.1371/journal.pone.0007082
8. Buitrago MM, Ringer T, Schulz JB, Dichgans J, Luft AR. Characterization of motor skill and instrumental learning time scales in a skilled reaching task in rat. Behav Brain Res (2004) 155(2):249–56. doi:10.1016/j.bbr.2004.04.025
9. Luft AR, Buitrago MM. Stages of motor skill learning. Mol Neurobiol (2005) 32(3):205–16. doi:10.1385/MN:32:3:205
10. Hikosaka O, Nakamura K, Sakai K, Nakahara H. Central mechanisms of motor skill learning. Curr Opin Neurobiol (2002) 12(2):217–22. doi:10.1016/S0959-4388(02)00307-0
11. Monfils MH, Plautz EJ, Kleim JA. In search of the motor engram: motor map plasticity as a mechanism for encoding motor experience. Neuroscientist (2005) 11(5):471–83. doi:10.1177/1073858405278015
12. Hosp JA, Luft AR. Cortical plasticity during motor learning and recovery after ischemic stroke. Neural Plast (2011) 2011:871296. doi:10.1155/2011/871296
13. Kleim JA, Barbay S, Nudo RJ. Functional reorganization of the rat motor cortex following motor skill learning. J Neurophysiol (1998) 80(6):3321–5.
14. Nudo RJ, Milliken GW, Jenkins WM, Merzenich MM. Use-dependent alterations of movement representations in primary motor cortex of adult squirrel monkeys. J Neurosci (1996) 16(2):785–807.
15. Pascual-Leone A, Nguyet D, Cohen LG, Brasil-Neto JP, Cammarota A, Hallett M. Modulation of muscle responses evoked by transcranial magnetic stimulation during the acquisition of new fine motor skills. J Neurophysiol (1995) 74(3):1037–45.
16. Molina-Luna K, Hertler B, Buitrago MM, Luft AR. Motor learning transiently changes cortical somatotopy. Neuroimage (2008) 40(4):1748–54. doi:10.1016/j.neuroimage.2007.11.018
17. Kleim JA, Hogg TM, VandenBerg PM, Cooper NR, Bruneau R, Remple M. Cortical synaptogenesis and motor map reorganization occur during late, but not early, phase of motor skill learning. J Neurosci (2004) 24(3):628–33. doi:10.1523/JNEUROSCI.3440-03.2004
18. Conner JM, Culberson A, Packowski C, Chiba AA, Tuszynski MH. Lesions of the basal forebrain cholinergic system impair task acquisition and abolish cortical plasticity associated with motor skill learning. Neuron (2003) 38(5):819–29. doi:10.1016/S0896-6273(03)00288-5
19. Hosp JA, Molina-Luna K, Hertler B, Atiemo CO, Luft AR. Dopaminergic modulation of motor maps in rat motor cortex: an in vivo study. Neuroscience (2009) 159(2):692–700. doi:10.1016/j.neuroscience.2008.12.056
20. Luft AR, Buitrago MM, Ringer T, Dichgans J, Schulz JB. Motor skill learning depends on protein synthesis in motor cortex after training. J Neurosci (2004) 24(29):6515–20. doi:10.1523/JNEUROSCI.1034-04.2004
21. Kleim JA, Lussnig E, Schwarz ER, Comery TA, Greenough WT. Synaptogenesis and Fos expression in the motor cortex of the adult rat after motor skill learning. J Neurosci (1996) 16(14):4529–35.
22. Rose SP. How chicks make memories: the cellular cascade from c-fos to dendritic remodelling. Trends Neurosci (1991) 14(9):390–7. doi:10.1016/0166-2236(91)90027-R
23. Wang JQ, McGinty JF. The full D1 dopamine receptor agonist SKF-82958 induces neuropeptide mRNA in the normosensitive striatum of rats: regulation of D1/D2 interactions by muscarinic receptors. J Pharmacol Exp Ther (1997) 281(2):972–82.
24. Rioult-Pedotti MS, Friedman D, Hess G, Donoghue JP. Strengthening of horizontal cortical connections following skill learning. Nat Neurosci (1998) 1(3):230–4. doi:10.1038/678
25. Rioult-Pedotti MS, Friedman D, Donoghue JP. Learning-induced LTP in neocortex. Science (2000) 290(5491):533–6. doi:10.1126/science.290.5491.533
26. Rioult-Pedotti MS, Donoghue JP, Dunaevsky A. Plasticity of the synaptic modification range. J Neurophysiol (2007) 98(6):3688–95. doi:10.1152/jn.00164.2007
27. Iriki A, Pavlides C, Keller A, Asanuma H. Long-term potentiation in the motor cortex. Science (1989) 245(4924):1385–7. doi:10.1126/science.2551038
28. Sakamoto T, Arissian K, Asanuma H. Functional role of the sensory cortex in learning motor skills in cats. Brain Res (1989) 503(2):258–64. doi:10.1016/0006-8993(89)91672-7
29. Asanuma H, Pavlides C. Neurobiological basis of motor learning in mammals. Neuroreport (1997) 8(4):i–vi.
30. Hosp JA, Hertler B, Atiemo CO, Luft AR. Dopaminergic modulation of receptive fields in rat sensorimotor cortex. Neuroimage (2011) 54(1):154–60. doi:10.1016/j.neuroimage.2010.07.029
31. Foote SL, Morrison JH. Development of the noradrenergic, serotonergic, and dopaminergic innervation of neocortex. Curr Top Dev Biol (1987) 21:391–423. doi:10.1016/S0070-2153(08)60145-3
32. Lepore F, Ptito M, Jasper HH. Two Hemispheres – One Brain: The Functions of the Corpus Callosum. New York: Alan Liss Co. (1986).
33. Fallon JH. Collateralization of monoamine neurons: mesotelencephalic dopamine projections to caudate, septum, and frontal cortex. J Neurosci (1981) 1(12):1361–8.
34. Swanson LW. The projections of the ventral tegmental area and adjacent regions: a combined fluorescent retrograde tracer and immunofluorescence study in the rat. Brain Res Bull (1982) 9(1–6):321–53. doi:10.1016/0361-9230(82)90145-9
35. Bromberg-Martin ES, Matsumoto M, Hikosaka O. Dopamine in motivational control: rewarding, aversive, and alerting. Neuron (2010) 68(5):815–34. doi:10.1016/j.neuron.2010.11.022
36. Schultz W. Multiple dopamine functions at different time courses. Annu Rev Neurosci (2007) 30:259–88. doi:10.1146/annurev.neuro.28.061604.135722
37. Schultz W. Behavioral dopamine signals. Trends Neurosci (2007) 30(5):203–10. doi:10.1016/j.tins.2007.03.007
38. Floresco SB, Magyar O. Mesocortical dopamine modulation of executive functions: beyond working memory. Psychopharmacology (Berl) (2006) 188(4):567–85. doi:10.1007/s00213-006-0404-5
39. Seamans JK, Yang CR. The principal features and mechanisms of dopamine modulation in the prefrontal cortex. Prog Neurobiol (2004) 74(1):1–58. doi:10.1016/j.pneurobio.2004.10.002
40. Chudasama Y, Robbins TW. Dopaminergic modulation of visual attention and working memory in the rodent prefrontal cortex. Neuropsychopharmacology (2004) 29(9):1628–36. doi:10.1038/sj.npp.1300490
41. Lammel S, Hetzel A, Hackel O, Jones I, Liss B, Roeper J. Unique properties of mesoprefrontal neurons within a dual mesocorticolimbic dopamine system. Neuron (2008) 57(5):760–73. doi:10.1016/j.neuron.2008.01.022
42. Garris PA, Wightman RM. Different kinetics govern dopaminergic transmission in the amygdala, prefrontal cortex, and striatum: an in vivo voltammetric study. J Neurosci (1994) 14(1):442–50.
43. Mundorf ML, Joseph JD, Austin CM, Caron MG, Wightman RM. Catecholamine release and uptake in the mouse prefrontal cortex. J Neurochem (2001) 79(1):130–42. doi:10.1046/j.1471-4159.2001.00554.x
44. German DC, Manaye KF. Midbrain dopaminergic neurons (nuclei A8, A9, and A10): three-dimensional reconstruction in the rat. J Comp Neurol (1993) 331(3):297–309. doi:10.1002/cne.903310302
45. Lewis DA, Campbell MJ, Foote SL, Goldstein M, Morrison JH. The distribution of tyrosine hydroxylase-immunoreactive fibers in primate neocortex is widespread but regionally specific. J Neurosci (1987) 7(1):279–90.
46. Gaspar P, Berger B, Febvret A, Vigny A, Henry JP. Catecholamine innervation of the human cerebral cortex as revealed by comparative immunohistochemistry of tyrosine hydroxylase and dopamine-beta-hydroxylase. J Comp Neurol (1989) 279(2):249–71. doi:10.1002/cne.902790208
47. Williams SM, Goldman-Rakic PS. Widespread origin of the primate mesofrontal dopamine system. Cereb Cortex (1998) 8(4):321–45. doi:10.1093/cercor/8.4.321
48. Loughlin SE, Fallon JH. Substantia nigra and ventral tegmental area projections to cortex: topography and collateralization. Neuroscience (1984) 11(2):425–35. doi:10.1016/0306-4522(84)90034-4
49. Deutch AY, Goldstein M, Baldino F Jr, Roth RH. Telencephalic projections of the A8 dopamine cell group. Ann N Y Acad Sci (1988) 537:27–50. doi:10.1111/j.1749-6632.1988.tb42095.x
50. Floel A, Breitenstein C, Hummel F, Celnik P, Gingert C, Sawaki L, et al. Dopaminergic influences on formation of a motor memory. Ann Neurol (2005) 58(1):121–30. doi:10.1002/ana.20536
51. Raghanti MA, Stimpson CD, Marcinkiewicz JL, Erwin JM, Hof PR, Sherwood CC. Cortical dopaminergic innervation among humans, chimpanzees, and macaque monkeys: a comparative study. Neuroscience (2008) 155(1):203–20. doi:10.1016/j.neuroscience.2008.05.008
52. Meintzschel F, Ziemann U. Modification of practice-dependent plasticity in human motor cortex by neuromodulators. Cereb Cortex (2006) 16(8):1106–15. doi:10.1093/cercor/bhj052
53. Floel A, Vomhof P, Lorenzen A, Roesser N, Breitenstein C, Knecht S. Levodopa improves skilled hand functions in the elderly. Eur J Neurosci (2008) 27(5):1301–7. doi:10.1111/j.1460-9568.2008.06079.x
54. Pearson-Fuhrhop KM, Minton B, Acevedo D, Shahbaba B, Cramer SC. Genetic variation in the human brain dopamine system influences motor learning and its modulation by L-DOPA. PLoS One (2013) 8(4):e61197. doi:10.1371/journal.pone.0061197
55. Ziemann U, Tergau F, Bruns D, Baudewig J, Paulus W. Changes in human motor cortex excitability induced by dopaminergic and anti-dopaminergic drugs. Electroencephalogr Clin Neurophysiol (1997) 105(6):430–7. doi:10.1016/S0924-980X(97)00050-7
56. Boroojerdi B. Pharmacologic influences on TMS effects. J Clin Neurophysiol (2002) 19(4):255–71. doi:10.1097/00004691-200208000-00002
57. DeLong MR. Primate models of movement disorders of basal ganglia origin. Trends Neurosci (1990) 13(7):281–5. doi:10.1016/0166-2236(90)90110-V
58. Berardelli A, Rona S, Inghilleri M, Manfredi M. Cortical inhibition in Parkinson’s disease. A study with paired magnetic stimulation. Brain (1996) 119(Pt 1):71–7. doi:10.1093/brain/119.1.71
59. Moore RY, Whone AL, Brooks DJ. Extrastriatal monoamine neuron function in Parkinson’s disease: an 18F-dopa PET study. Neurobiol Dis (2008) 29(3):381–90. doi:10.1016/j.nbd.2007.09.004
60. McRitchie DA, Cartwright HR, Halliday GM. Specific A10 dopaminergic nuclei in the midbrain degenerate in Parkinson’s disease. Exp Neurol (1997) 144(1):202–13. doi:10.1006/exnr.1997.6418
61. Gaspar P, Duyckaerts C, Alvarez C, Javoy-Agid F, Berger B. Alterations of dopaminergic and noradrenergic innervations in motor cortex in Parkinson’s disease. Ann Neurol (1991) 30(3):365–74. doi:10.1002/ana.410300308
62. Bernheimer H, Birkmayer W, Hornykiewicz O, Jellinger K, Seitelberger F. Brain dopamine and the syndromes of Parkinson and Huntington. Clinical, morphological and neurochemical correlations. J Neurol Sci (1973) 20(4):415–55. doi:10.1016/0022-510X(73)90175-5
63. Frith CD, Bloxham CA, Carpenter KN. Impairments in the learning and performance of a new manual skill in patients with Parkinson’s disease. J Neurol Neurosurg Psychiatry (1986) 49(6):661–8. doi:10.1136/jnnp.49.6.661
64. Verschueren SM, Swinnen SP, Dom R, De Weerdt W. Interlimb coordination in patients with Parkinson’s disease: motor learning deficits and the importance of augmented information feedback. Exp Brain Res (1997) 113(3):497–508. doi:10.1007/PL00005602
65. Kishore A, Popa T, Velayudhan B, Joseph T, Balachandran A, Meunier S. Acute dopamine boost has a negative effect on plasticity of the primary motor cortex in advanced Parkinson’s disease. Brain (2012) 135(Pt 7):2074–88. doi:10.1093/brain/aws124
66. Kishore A, Joseph T, Velayudhan B, Popa T, Meunier S. Early, severe and bilateral loss of LTP and LTD-like plasticity in motor cortex (M1) in de novo Parkinson’s disease. Clin Neurophysiol (2012) 123(4):822–8. doi:10.1016/j.clinph.2011.06.034
67. Morgante F, Espay AJ, Gunraj C, Lang AE, Chen R. Motor cortex plasticity in Parkinson’s disease and levodopa-induced dyskinesias. Brain (2006) 129(Pt 4):1059–69. doi:10.1093/brain/awl031
68. Ueki Y, Mima T, Kotb MA, Sawada H, Saiki H, Ikeda A, et al. Altered plasticity of the human motor cortex in Parkinson’s disease. Ann Neurol (2006) 59(1):60–71. doi:10.1002/ana.20692
69. Suppa A, Marsili L, Belvisi D, Conte A, Iezzi E, Modugno N, et al. Lack of LTP-like plasticity in primary motor cortex in Parkinson’s disease. Exp Neurol (2011) 227(2):296–301. doi:10.1016/j.expneurol.2010.11.020
70. Yan HQ, Kline AE, Ma X, Hooghe-Peters EL, Marion DW, Dixon CE. Tyrosine hydroxylase, but not dopamine beta-hydroxylase, is increased in rat frontal cortex after traumatic brain injury. Neuroreport (2001) 12(11):2323–7. doi:10.1097/00001756-200108080-00009
71. Yan HQ, Kline AE, Ma X, Li Y, Dixon CE. Traumatic brain injury reduces dopamine transporter protein expression in the rat frontal cortex. Neuroreport (2002) 13(15):1899–901. doi:10.1097/00001756-200210280-00013
72. Floel A, Hummel F, Breitenstein C, Knecht S, Cohen LG. Dopaminergic effects on encoding of a motor memory in chronic stroke. Neurology (2005) 65(3):472–4. doi:10.1212/01.wnl.0000172340.56307.5e
73. Scheidtmann K, Fries W, Muller F, Koenig E. Effect of levodopa in combination with physiotherapy on functional motor recovery after stroke: a prospective, randomised, double-blind study. Lancet (2001) 358(9284):787–90. doi:10.1016/S0140-6736(01)05966-9
Keywords: dopamine, motor cortex, motor learning, cortical plasticity, memory
Citation: Hosp JA and Luft AR (2013) Dopaminergic meso-cortical projections to M1: role in motor learning and motor cortex plasticity. Front. Neurol. 4:145. doi: 10.3389/fneur.2013.00145
Received: 13 May 2013; Accepted: 14 September 2013;
Published online: 07 October 2013.
Edited by:
Asha Kishore, Sree Chitra Tirunal Institute for Medical Sciences and Technology, IndiaReviewed by:
Miguel Coelho, University of Lisbon, PortugalFrancesca Morgante, University of Messina, Italy
Copyright: © 2013 Hosp and Luft. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Andreas R. Luft, Department of Neurology, University of Zurich, Frauenklinikstrasse 26, 8091 Zurich, Switzerland e-mail:YW5kcmVhcy5sdWZ0QHVzei5jaA==

