- 1Department of Neurology, The George Washington University, Washington, DC, United States
- 2Department of Neurology, Beijing Friendship Hospital, Capital Medical University, Beijing, China
- 3Department of Neurology, Qilu Hospital of Shandong University, Jinan, China
- 4The Key Laboratory of Geriatrics, Beijing Hospital, Beijing Institute of Geriatrics, Ministry of Health, Beijing, China
- 5Department of Pharmacology, The George Washington University, Washington, DC, United States
- 6Department of Physiology, The George Washington University, Washington, DC, United States
- 7Department of Pathology, Peking Union Medical College Hospital, Chinese Academy of Medical Science, Beijing, China
- 8Department of Neurology, Affiliated Hospital of Qingdao University, Qingdao, China
Biomarkers that assess treatment response for patients with the autoimmune disorder, myasthenia gravis (MG), have not been evaluated to a significant extent. We hypothesized the pro-inflammatory cytokine, osteopontin (OPN), may be associated with variability of response to glucocorticoids (GCs) in patients with MG. A cohort of 250 MG patients treated with standardized protocol of GCs was recruited, and plasma OPN and polymorphisms of its gene, secreted phosphoprotein 1 (SPP1), were evaluated. Mean OPN levels were higher in patients compared to healthy controls. Carriers of rs11728697*T allele (allele definition: one of two or more alternative forms of a gene) were more frequent in the poorly GC responsive group compared to the GC responsive group indicating an association of rs11728697*T allele with GC non-responsiveness. One risk haplotype (AGTACT) was identified associated with GC non-responsiveness compared with GC responsive MG group. Genetic variations of SPP1 were found associated with the response to GC among MG patients.
Introduction
Myasthenia gravis (MG) is a chronic autoimmune disorder caused by antibodies directed against postsynaptic proteins, primarily the skeletal muscle nicotinic acetylcholine receptor (AChR). Oral glucocorticoids (GCs) are the primary therapy (1, 2), but response rates to GCs are highly variable among studies ranging from 5 to 30% (3–7). No clinical or biological markers exist that predict GC responsiveness. Genetic polymorphisms have been identified that are associated with therapeutic response to GCs in other autoimmune and inflammatory conditions (8), and we recently identified a single nucleotide polymorphism (SNP) in the GC receptor gene as an independent factor associated with short-term GC responsiveness among patients with MG (9).
Osteopontin (OPN) is a pro-inflammatory cytokine, and increased circulating levels have been associated with inflammatory muscle diseases and muscular dystrophy (10–13) as well as the onset and progression of Crohn’s disease, myocarditis, uveitis, idiopathic retroperitoneal fibrosis, and rheumatoid arthritis (14–16). Furthermore, SPP1 gene polymorphisms have been identified as being associated with GC responsiveness in boys with Duchenne muscular dystrophy (13); however, a larger validation study published after we began our work did not confirm the association (17). We hypothesized that plasma OPN may be a marker of treatment responsiveness and that genetic variations (polymorphisms) in the secreted phosphoprotein 1 (SPP1) gene, which encodes OPN, are associated with differences in GC responsiveness of patients with MG.
We assessed the relationship among clinical characteristics and plasma OPN level of MG patients. Further, we evaluated the relationship between SNPs of SPP1 gene and response to a standardized 3-month GC treatment protocol with prospectively collected outcome data. As our primary outcome assessment, we used the quantitative MG score (QMGS), which is a validated scale and was recommended by the MG Foundation of America as the primary clinical outcome measure for clinical trials (18). Several studies using the QMGS have shown that a change of 3 or more points to be clinically meaningful (19–21).
Subjects and Methods
Study Population
We assessed the same cohort as in our separate association study of GR gene polymorphisms and response to GCs (9). Three hundred forty-two consecutively identified MG patients who had not received immunosuppressive agents were recruited and followed from Beijing Friendship Hospital, Capital Medical University, and Affiliated Hospital of Qingdao University. The diagnosis of MG was based on a typical clinical history of variable weakness involving ocular, bulbar, limb, or a combination of muscle groups. Fatigable weakness was evident on physical examination. Alternative diagnoses, such as central nervous system disorders, myopathies, and motor neuron disorders were excluded. A positive result in at least one of three was required: (1) increased serum level of anti-acetylcholine receptor antibody (AChRAb); (2) decremental response to low frequency repetitive nerve stimulation; or (3) positive response to the neostigmine test. Muscle specific kinase patients were excluded.
Fifty-two patients were excluded because of a contraindication to GC therapy or refusal to receive GC treatment. The GC treatment was initiated with 0.75–1 mg/kg/day of prednisone or equivalent methylprednisolone. The dosage of GCs was tapered gradually when definite improvement was appreciated or was maintained for 3 months. Patients who had received plasma exchange, intravenous immunoglobulin or immunosuppressants during the study period were excluded. Patients who were excluded for other causes are described in Figure 1. DNA samples from seven patients were depleted from use in our previous study (9). Two hundred fifty patient samples underwent SPP1 genotyping. A subset of 74 MG patients and 50 healthy controls underwent evaluation for plasma for OPN levels. Patients were stratified into subgroups by gender, age of onset (22), clinical presentation at disease onset, AChRAb status, presence of thymoma, disease duration before treatment, and QMGS before treatment (Table 1). Patients were followed monthly for 3 months after treatment initiation and QMGS determined by a physician trained in its performance (18). The control group consisted of 474 healthy individuals age-matched to the study population and seen during the study period at each participating institution. All study participants were northern Han Chinese and non-consanguineous. Change of QMGS was used as a primary efficacy measurement. Improvement of 3 or greater points of the QMGS or a QMGS becoming 0 identified a patient as being responsive to GCs (21, 23). The study was approved by ethical committees of the hospitals, and all participants provided written informed consent.

Figure 1. Enrollment profile. GC, glucocorticoid; IVIg, intravenous immune globulin; SNP, single nucleotide polymorphism.
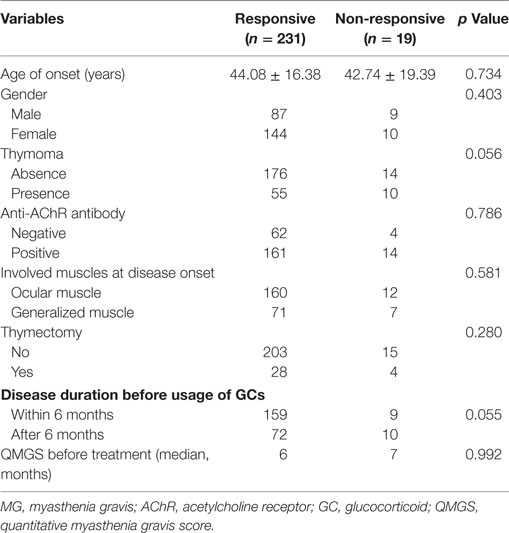
Table 1. Comparison of the demographic and clinical characteristics of patients between responsive and non-responsive patients with MG.
Enzyme-Linked Immunosorbent Assay (ELISA)
Blood samples were collected in ethylenediaminetetraacetic acid from patients prior to the initiation of GC or any other immunotherapy. Plasma was isolated and stored at −80°C until evaluation. The concentration of OPN was determined by the quantitative sandwich ELISA using the Quantikine kit (R&D Systems, Minneapolis, MN, USA). Antibodies against AChR (AChRAb) was detected by using ELISA kit (RSR Limited, Cardiff, UK) (24). The testing was performed according to the instructions of the kit. The results were expressed as inhibition rate of AChR binding, calculated according to the formula in the instructions: Inhibition rate (%) = 100 × (test sample absorbance/negative control absorbance).
SNP Selection and Genotyping
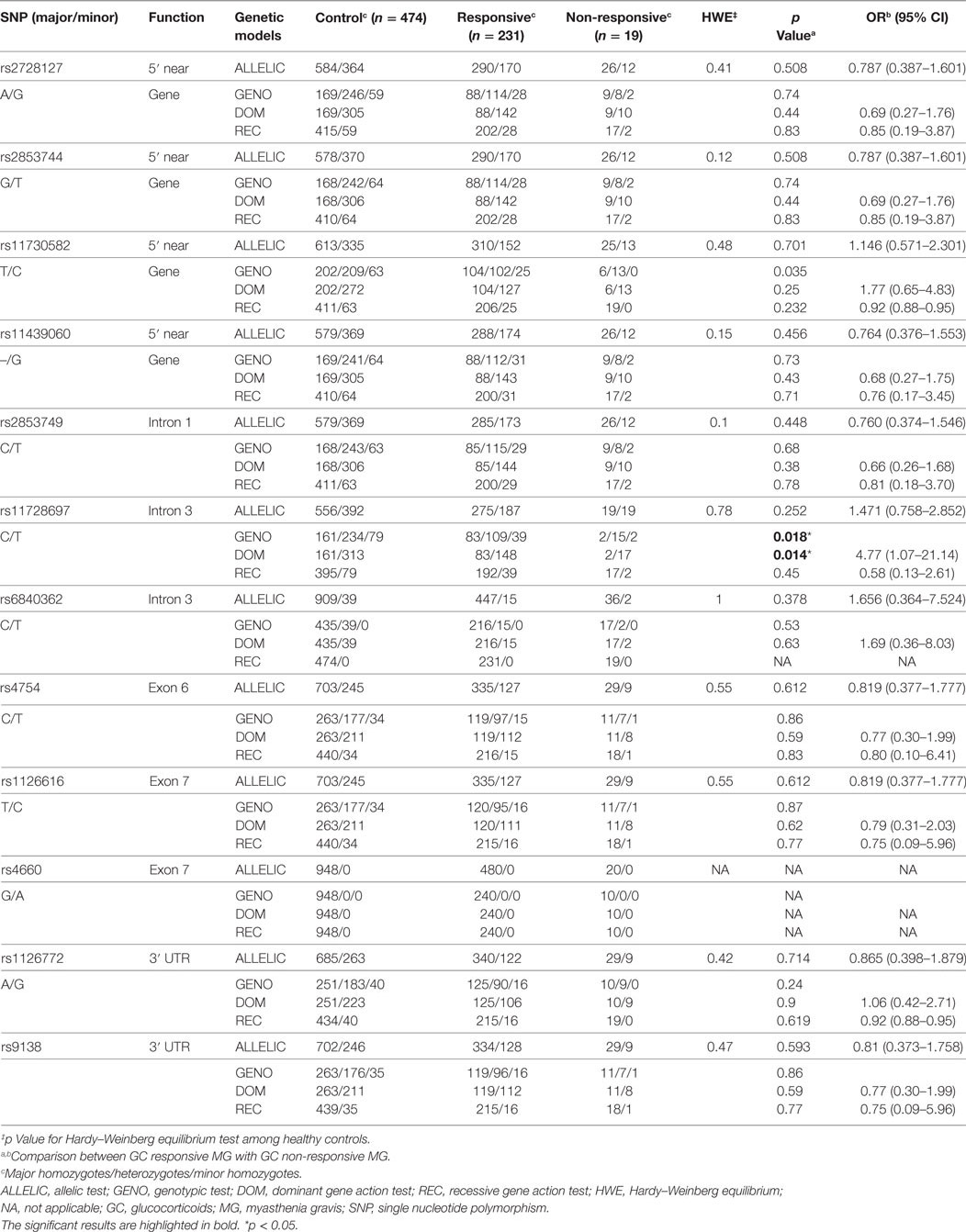
Twelve SNPs (variations in single base pairs in a DNA sequence) were selected based on previous reports and information from NCBI dbSNP and HapMap database [CHB database, HapMap phase version 3, release 27 (2009, February)], in an attempt to cover the majority of the SPP1 gene region by linkage disequilibrium (LD). Among the 12 SNPs, 1 tag SNP (rs2853749) was selected using the HapMap database with the software as previously described, and 11 SNPs (rs2728127, rs2853744, rs11730582, rs11439060, rs11728697, rs6840362, rs4754, rs1126616, rs4660, rs1126772, and rs9138) were previously reported (25–33). Ten of these have functional potential (rs2728127, rs2853744, rs11730582, rs11439060 in the 5′ near gene; rs11728697, rs4754, rs1126616, rs4660 in coding region; rs1126772, rs9138 in 3′-untranslated region). One SNP has been previously investigated (rs6840362), which showed a significant difference allele distribution in European American patients with SLE (30). The location and function of the SNPs are shown in Table 2.
Single nucleotide polymorphism genotyping was performed using a custom-designed SNPscanTM Kit (Genesky Biotechnologies Inc., Shanghai, China). For quality assurance, 3.87% (28/724) of the total samples were randomly repeated. Concordance for duplicate samples was 100% for all assays.
Statistical Analysis
Statistical analysis was performed with SPSS Statistics 13 (SPSS Corporation, Chicago, IL, USA), SHEsis software (Bio-X Life Science Research Center, Shanghai, China), Haploview 4.2 software, and GraphPad Prism 5 (GraphPad Software, Inc., La Jolla, CA, USA). A two-side comparison with p < 0.05 was considered statistically significant. The database, constructed using SQL server, contains data pertaining to the SNPs, clinical features and treatment, and clinical follow-up of patients.
The normality of the data was tested using the method of Kolmogorov and Smirnov. Continuous variables were presented as mean ± SD or median, 25th and 75th percentiles; categorical variables were presented as a percentage. Differences between groups were analyzed with independent sampled t test or the Mann–Whitney U-test for continuous variables and by chi-square test or Fisher exact test for categorical variables. The association between the GC efficacy and MG phenotype and SPP1 genotypes was examined by multivariate regression analysis. In this pilot study, we did not make a correction for multiple comparisons. The Haploview 4.2 software was used to calculate pairwise LD of SNPs and construct haplotype blocks. Haplotypes are defined as genetic variations that are inherited together. Haplotype frequencies were estimated with Partition–Ligation–Combination–Subdivision Expectation Maximization algorithm implemented in SHEsis software.
Functional Annotation and Expression Quantitative Trait Locus (eQTL) Analysis
Functional annotations of SNPs were investigated using RegulomeDB, a database which provides assessment of whether SNPs are located in known or predicted regulatory elements, including regions of DNase I hypersensitivity, binding sites for transcription factors (TFs), and promoter regions that regulate transcription (34).
With the aim of exploring the molecular basis of the observed associations, eQTLs analysis was performed by using published cell-specific eQTL dataset (35).
Results
Patient Characteristics
The study cohort consisted of 250 patients of which 154 (61.6%) were women (Table 1). The onset age ranged from 15 to 80 years, mean 43.98 ± 16.59 years. The disease duration prior to GC therapy ranged from 0.2 to 48 months with a median duration of 4 months (interquartile range of 2–11 months). Among 474 healthy controls, 238 were men, and 236 are women with an age range of 14–78 and median age of 45 years.
Response to GCs
Quantitative MG score ranged from 1 to 35 (median QMGS was 6, interquartile ranged from 4 to 11) at study onset, and after 3 months of treatment, a significant reduction in QMGS was observed ranging from 0 to 29 points (median QMGS was 1, interquartile ranged from 0 to 3, p < 0.0001). The change in QMGS ranged from −2 to 18 (median QMGS was 5, interquartile ranged from 3 to 8).
Two hundred thirty-one patients (92.4%) were considered GC responsive, and 19 (7.6%) were considered GC non-responsive. Clinical characteristics of the patients are summarized in Table 1. No differences were found between a poor response to GC therapy with age of onset, gender, presence of thymoma, AChR antibody status, muscle group involvement at disease onset, thymectomy, disease duration before GC treatment, or QMGS before treatment (p = 0.73, 0.41, 0.78, 0.79, 0.59, 0.28, 0.06, and 0.99, respectively) (Table 1).
OPN Concentrations in MG Patients and Healthy Controls
Prior to initiation of immunotherapy, mean OPN plasma levels were greater among MG patients (68.33 ± 43.03 ng/ml) compared to healthy controls (50.19 ± 38.74 ng/ml; p = 0.013; Figure 2). We performed a subgroup analysis of the patients with the highest OPN levels (mean + 2 SD of controls; 127.67 ng/ml in the study), which identified 8 patients, and compared them to the remaining 66 patients (Figure 3). We found that the MG patients with the highest levels of OPN had lower percentage of positive AChRAb (37.5 vs 85.94%, p = 0.006, Figure 3C). No difference in onset age, gender, presence of thymoma, involved muscle group at disease onset, duration before GCs treatment, QMGS at the sample collection, and change of QMGS after 3 months GC treatment (p = 0.667, 0.227, 0.641, 1.00, 1.00, 0.373, 0.606, respectively, Figures 3A,B,D–H) was found between the two subgroups.
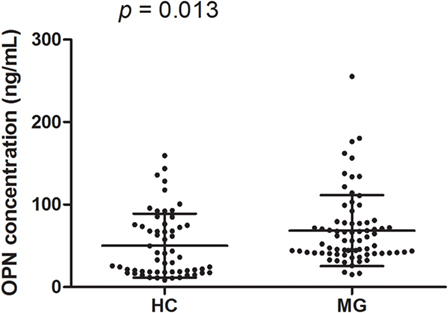
Figure 2. Osteopontin (OPN) levels among myasthenia gravis (MG) patients and healthy controls. Mean OPN levels were higher in MG patients (68.33 ± 43.03 ng/ml) compared to healthy controls (50.19 ± 38.74 ng/ml; p = 0.013).
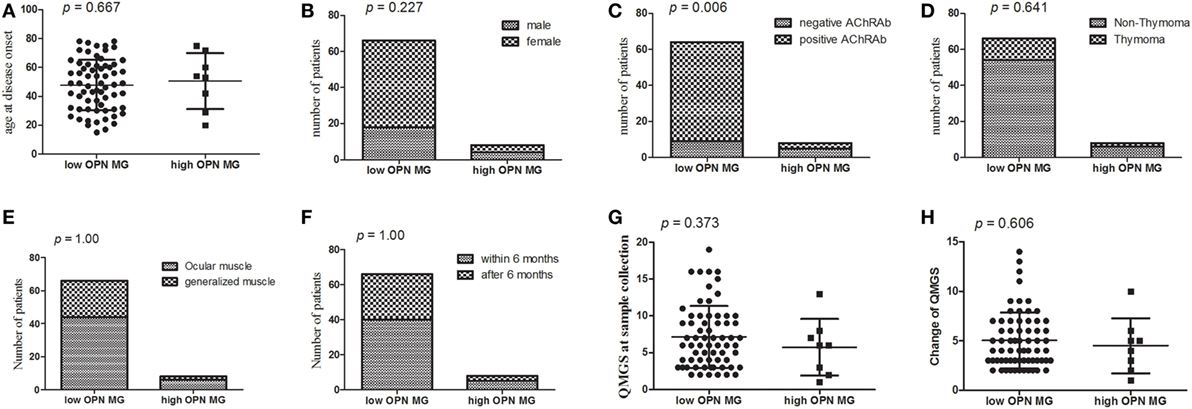
Figure 3. Clinical features of myasthenia gravis (MG) patients with the greatest osteopontin (OPN) levels. The MG patients with the highest levels of OPN had lower percentage of positive acetylcholine receptor (AChR) antibody (37.5 vs 85.94%, p = 0.006, Figure 3C). No difference in age of disease onset, gender, presence of thymoma, involved muscle at disease onset, duration before glucocorticoid (GC) treatment, quantitative MG score (QMGS) at the sample collection, and change of QMGS after 3 months GC treatment (p = 0.667, 0.227, 0.641, 1.00, 1.00, 0.373, 0.606, respectively, Figures 3A,B,D–H).
Association between SPP1 Gene Variation and Response to GC
The success rates of genotyping the 12 SNPs among MG patients and healthy controls were greater than 99.7%. None of the participants had the rs4660 polymorphism. The genotyping data of 12 SNPs of the healthy controls did not deviate from the Hardy–Weinberg equilibrium (HWE, p = 0.1–1, Table 2). General characteristics of 12 SNPs in the SPP1 gene of patients are shown in Table 2. The distribution of genotypes in the GC sensitive and insensitive groups was consistent with the Hardy–Weinberg equilibrium (Table 2).
The rs11728697 T carriers (C/T + T/T genotypes) were more frequent in the GC non-responsive group compared to the GC responsive group (89.5 vs 64.1%; dominant model: p = 0.014; OR = 4.77, 95% CI = 1.07–21.14), indicating association with a poor response to GCs. No statistically significant differences were observed for the remaining SNPs (Table 2). No difference in the frequency of the rs11728697 T carriers was observed between MG patients and healthy control groups (66 vs 66%, dominant model, p = 0.99).
The association between SPP1 gene variation and responses to GCs was further examined by multivariate logistic regression analysis, with GC non-response as the dependent variable, and with onset age, gender, involved muscles at disease onset, AChRAb, presence of thymoma, disease duration before GC treatment, and rs11728697 as independent variables. The rs11728697 T carrier was found as an independent factor for GC non-responsiveness (p = 0.019, OR = 4.76, 95% CI = 1.03–21.99).
SPP1 Haplotypes and GC Efficacy in MG Patients
The LD test among 12 SNPs in the SPP1 gene is shown in Figure 4. According to Haploview, the haplotype block structure of the SPP1 gene consists of two blocks. The first block ranges from rs2728127 to rs11728697 (rs2728127, rs2853744, rs11730582, rs11439060, rs2853749, and rs11728697; D′ ranging from 0.93 to 1.0), the second from rs4754 to rs9138 (rs4754, rs1126616, rs1126772, and rs9138; D′ ranging from 0.99 to 1.0). A total of four common haplotypes were identified across the first block, ranging in frequency from 35.5 to 7.3% in all patients (GTTGTC: 35.5%, AGCACT: 32.6%, AGTACC: 21.5%, AGTACT: 7.3%). One risk haplotype (AGTACT) in the first block was identified (OR = 2.61, 95% CI = 1.09–6.73, p = 0.041) in the GC non-responsive MG group compared with GC responsive MG group (15.8 vs 6.5%) (Table 3). Three common haplotypes were identified across the second block, ranging in frequency from 46.4 to 27.0% in total patient (CTAC: 46.4%, TCAA: 27.0%, CTGC: 26.2%). No risk haplotype was found across the second block. There was no difference of OPN concentration between MG patients with rs11728697 CC (77.34 ± 54.36 ng/ml) and those with rs11728697 CT + TT (61.84 ± 31.68 ng/ml; p = 0.127) (Figure 5).
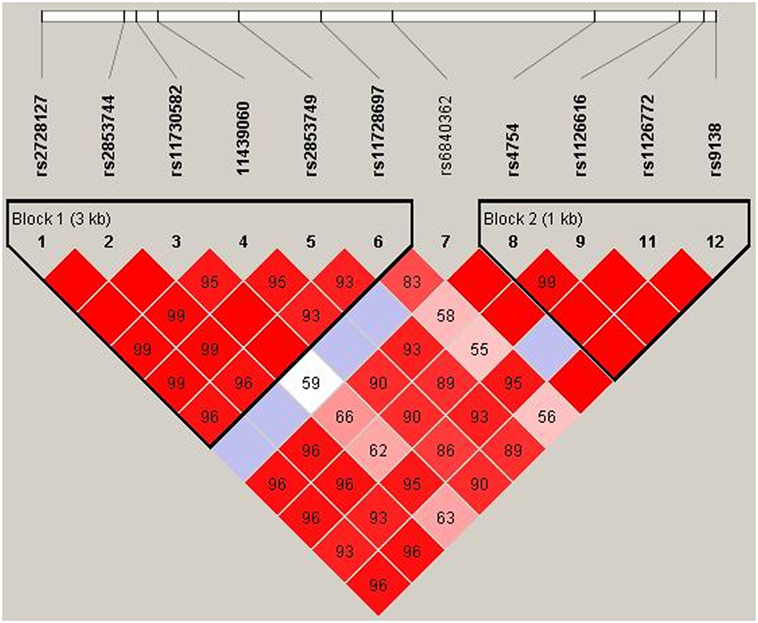
Figure 4. Haplotype block of SPP1 variants. Generated by Haploview (version 4.2). Dark red, strong linkage disequilibrium (LD); light red, weak LD.
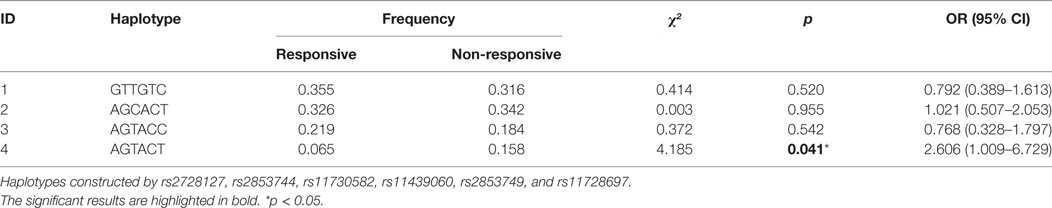
Table 3. Haplotype analysis of the SPP1 gene variants between glucocorticoids responsive and non-responsive subjects.
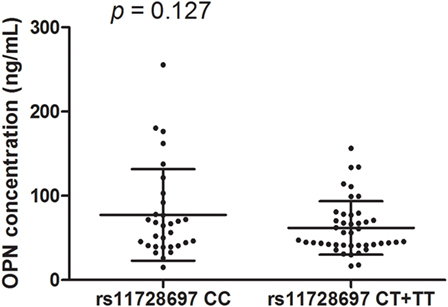
Figure 5. Osteopontin (OPN) concentrations in subgroups with rs11728697 genotypes. No difference in OPN concentrations was found between rs11728697 CC (77.34 ± 54.36 ng/ml) and rs11728697 CT + TT (61.84 ± 31.68 ng/ml).
RegulomeDB Analysis of rs11728697
According to the functional annotation information from the RegulomeDB scoring, rs11728697 was identified as score 1d (http://www.regulomedb.org/snp/chr4/88898940) with evidence for eQTL, TF binding, a matched TF binding motif, and location within DNase-sensitive site. rs11728697 is within the region of the binding site of TF regulatory factor X 3 that was detected by Chip-Seq analysis in K562 cell line (36). rs11728697 was found to be linked with rs12502049 (D′ = 0.958 and R2 = 0.724), which was identified as score 1f with evidence for mapping to a predicted TF binding site and/or within a DNase I sensitivity peak and correlating with gene expression. rs12502049 is known to regulate the expression of SPP1 gene that was detected by eQTL analysis in a lymphoblastoid cell line (37).
Discussion
We identified an SNP in the SPP1 gene (rs11728697), which was associated a poor response to GC treatment among patients with MG. One risk haplotype (AGTACT) containing a mutation at this SNP was identified among the GC non-responsive patients compared with the GC responsive patients. The carrier of the variant does not differ between MG patients and the controls, indicating the association of rs11728697 T with GC response is not determined by its involvement in pathogenesis of MG but associates with GC treatment response. Presently, there is no evidence of a direct interaction between OPN and GC response pathways; however, OPN influences T and B cell function, which provides indirect pathways that could be associated with GC treatment responsiveness (38). Further, rs11728697 is in a location of the SPP1 gene that may bind transcriptional factors offering a mechanism for GC effect.
Previous studies of genetic variations of response to GC therapy in autoimmune or inflammatory disorders are limited. Investigation of GC responsiveness in SLE identified SNP associations in the GC receptor gene (39). We also identified an SNP in the GC receptor gene that was associated with a poor treatment response among patients with MG (9). A study of Duchenne muscular dystrophy found an rs28357094 polymorphism, which lies in the gene promoter of SPP1, to be associated with GC response. In this study, we aimed to explore whether polymorphisms in the gene of an immuno-modulating protein was associated with GC responsiveness in MG.
There is no accepted definition of treatment response for MG therapeutics, which is a limitation of our investigation. As more precise clinical and biological definitions of treatment response are identified, genetic and biomarker studies may identify more robust associations. The QMGS is a validated scale and was recommended by the MG Foundation of America Task Force as the primary clinical outcome measure (19). Several studies have indicated that an improvement of 3 or more points on the QMG scale is clinically meaningful (19–21). Pascuzzi and colleagues treated 116 patients with similar regimens of 60–80 mg daily of prednisone and with prolonged follow-up, 5% were described as unresponsive to treatment (3). Our result is consistent with the findings of this investigation. Others have found higher rates of limited treatment response using less strict definitions from ours. However, their study had a longer observational length, which would indicate that treatment failure may have been contaminated by patients developing GC complications. A retrospective study over 2 years of observation found 13% of patients were unchanged or worse, and in a long-term study of 104 patients, 13 percent had no improvement in MG manifestations with treatment (6, 7). Further, an observational study without a standardized treatment protocol found about one-third of patients have significant disability despite 6 months of treatment, which usually included prednisone (40).
We found that the mean plasma concentration of OPN was significantly increased in MG patients compared with controls, which is consistent with its role as a pro-inflammatory cytokine and is consistent with its elevation in other autoimmune and inflammatory diseases (38). We did not appreciate an association of OPN levels with any clinical parameters or genotypes of this SNP. There are several potential explanations. Circulating cytokine levels have disease-related and diurnal variations, which may not have been accounted for by this study. The investigation may not have been able to distinguish disease-specific variations due to a lack of statistical power. From the present investigation whether circulating OPN levels or mutations in SPP1 may be predictive of treatment response cannot be determined. SPP1 expression is elevated in muscle of animals with passive transfer MG (41). OPN has been found to be involved in progression of endogenous autoreactive germinal centers leading to enhanced antinuclear antibody production in an animal of lupus (42), which points to the multiple mechanisms that are involved in autoantibody production. No difference in the proportion of rs11728697 T carrier was found in association with the plasma level of OPN. This likely relates to several potential factors impacting circulating levels of OPN, including other genetic associations with GC response.
In summary, increased mean plasma levels of OPN are found among MG patients, but these had no relationship to patient demographics or treatment response. We found a mutation at rs11728697 and two haplotypes in the SPP1 gene to be associated with poor response to GC treatment among MG patients. In particular, subgroup analyses did not find associations with clinical characteristics of patients, but conclusions are limited because of small sample sizes. A larger validation study will be required to confirm these observations. For MG and other autoimmune disorders, it is critical to move toward identification of markers that predict treatment response with the intent of providing individual treatment (43).
Ethics Statement
The study was approved by ethical committees of Beijing Friendship Hospital, Capital Medical University, and Affiliated Hospital of Qingdao University, and all participants provided informed consent.
Author Contributions
YX, H-FL, and HK conceptualized and designed the study, interpreted the data, and wrote the manuscript. LS designed the genotyping experiment and performed bioinformatics analysis. LK contributed to discussion. H-FL, SW, XZ, XG, and YL diagnosed and treated patients for this study. YX, LS, and YH performed statistical analysis. YM maintained the database and determined the pathologic diagnosis for the study.
Conflict of Interest Statement
YX received research support from Beijing Natural Science Foundation. H-FL received research support from National Natural Science Foundation of China and Shandong Provincial Natural Science Foundation. YM received research support from National Natural Science Foundation of China. HK receives grant support from the Myasthenia Gravis Foundation of Illinois and the Muscular Dystrophy Association, fees for serving on data and safety monitoring boards from Genentech and Novartis, consulting fees from Alnylam Pharmaceuticals, UCB, Rubius Therapeutics, RA Pharmaceuticals, and Momenta Pharmaceuticals, receiving study medication from Genentech, and holding a patent related to technology to focus a complement inhibitor on the neuromuscular junction for the treatment of myasthenia gravis (U.S. patent no. 8,961,981). LK receives grant support from the Muscular Dystrophy Association. All other authors report no disclosures.
Acknowledgments
Participants in this study were enrolled and followed from Beijing Friendship Hospital, Capital Medical University, and Affiliated Hospital of Qingdao University. The authors thank Hong-jun Hao (Department of Neurology, Peking University First Hospital) for anti-AChR Ab testing.
Funding
The present study was supported by the National Natural Science Foundation of China (No. 81070963 to H-FL, No. 81400068 to YM), Beijing Natural Science Foundation (No. 7142042 to YX), and Shandong Provincial Natural Science Foundation (No. ZR2010HM019 to H-FL.). The Meta A. Neumann endowment provided additional support (HK).
Abbreviations
AChR, acetylcholine receptor; GC, glucocorticoid; ELISA, enzyme-linked immunosorbent assay; eQTLs, expression quantitative trait loci; IVIG, intravenous immunoglobulin; LD, linkage disequilibrium; MG, myasthenia gravis; OPN, osteopontin; PE, plasma exchange; QMGS, quantitative MG score; SNP, single nucleotide polymorphism; SPP1, secreted phosphoprotein 1.
References
1. Meriggioli MN, Sanders DB. Autoimmune myasthenia gravis: emerging clinical and biological heterogeneity. Lancet Neurol (2009) 8(5):475–90. doi: 10.1016/S1474-4422(09)70063-8
2. Richman DP, Agius MA. Treatment of autoimmune myasthenia gravis. Neurology (2003) 61(12):1652–61. doi:10.1212/01.WNL.0000098887.24618.A0
3. Pascuzzi RM, Coslett HB, Johns TR. Long-term corticosteroid treatment of myasthenia gravis: report of 116 patients. Ann Neurol (1984) 15(3):291–8. doi:10.1002/ana.410150316
4. Skeie GO, Apostolski S, Evoli A, Gilhus NE, Illa I, Harms L, et al. Guidelines for treatment of autoimmune neuromuscular transmission disorders. Eur J Neurol (2010) 17(7):893–902. doi:10.1111/j.1468-1331.2010.03019.x
5. Sghirlanzoni A, Peluchetti D, Mantegazza R, Fiacchino F, Cornelio F. Myasthenia gravis: prolonged treatment with steroids. Neurology (1984) 34(2):170–4. doi:10.1212/WNL.34.2.170
6. Cosi V, Citterio A, Lombardi M, Piccolo G, Romani A, Erbetta A. Effectiveness of steroid treatment in myasthenia gravis: a retrospective study. Acta Neurol Scand (1991) 84(1):33–9. doi:10.1111/j.1600-0404.1991.tb04899.x
7. Evoli A, Batocchi AP, Palmisani MT, Lo Monaco M, Tonali P. Long-term results of corticosteroid therapy in patients with myasthenia gravis. Eur Neurol (1992) 32(1):37–43. doi:10.1159/000116785
8. Ramamoorthy S, Cidlowski JA. Corticosteroids: mechanisms of action in health and disease. Rheum Dis Clin North Am (2016) 42(1):15–31. doi:10.1016/j.rdc.2015.08.002
9. Xie Y, Meng Y, Li HF, Hong Y, Sun L, Zhu X, et al. GR gene polymorphism is associated with inter-subject variability in response to glucocorticoids in patients with myasthenia gravis. Eur J Neurol (2016) 23(8):1372–9. doi:10.1111/ene.13040
10. Kyriakides T, Pegoraro E, Hoffman EP, Piva L, Cagnin S, Lanfranchi G, et al. SPP1 genotype is a determinant of disease severity in Duchenne muscular dystrophy: predicting the severity of Duchenne muscular dystrophy: implications for treatment. Neurology (2011) 77(20):1858. doi:10.1212/WNL.0b013e318239b9ae author reply 9
11. Glas J, Seiderer J, Bayrle C, Wetzke M, Fries C, Tillack C, et al. The role of osteopontin (OPN/SPP1) haplotypes in the susceptibility to Crohn’s disease. PLoS One (2011) 6(12):e29309. doi:10.1371/journal.pone.0029309
12. Shin T, Ahn M, Kim H, Kim HM, Matsumoto Y. Increased expression of osteopontin in the heart tissue of Lewis rats with experimental autoimmune myocarditis. J Vet Med Sci (2006) 68(4):379–82. doi:10.1292/jvms.68.379
13. Hikita ST, Vistica BP, Jones HR, Keswani JR, Watson MM, Ericson VR, et al. Osteopontin is proinflammatory in experimental autoimmune uveitis. Invest Ophthalmol Vis Sci (2006) 47(10):4435–43. doi:10.1167/iovs.06-0064
14. Binder M, Christoph S, Sehnert B, Uhl M, Peter HH, Voll RE, et al. Elevated serum osteopontin levels in idiopathic retroperitoneal fibrosis. Clin Exp Rheumatol (2012) 30(5):772–5.
15. Song JJ, Hwang I, Cho KH, Garcia MA, Kim AJ, Wang TH, et al. Plasma carboxypeptidase B downregulates inflammatory responses in autoimmune arthritis. J Clin Invest (2011) 121(9):3517–27. doi:10.1172/JCI46387
16. Wang KX, Denhardt DT. Osteopontin: role in immune regulation and stress responses. Cytokine Growth Factor Rev (2008) 19(5–6):333–45. doi:10.1016/j.cytogfr.2008.08.001
17. van den Bergen JC, Hiller M, Böhringer S, Vijfhuizen L, Ginjaar HB, Chaouch A, et al. Validation of genetic modifiers for Duchenne muscular dystrophy: a multicentre study assessing SPP1 and LTBP4 variants. J Neurol Neurosurg Psychiatry (2015) 86(10):1060–5. doi:10.1136/jnnp-2014-308409
18. Jaretzki A III, Barohn RJ, Ernstoff RM, Kaminski HJ, Keesey JC, Penn AS, et al. Myasthenia gravis: recommendations for clinical research standards. Task Force of the Medical Scientific Advisory Board of the Myasthenia Gravis Foundation of America. Neurology (2000) 55(1):16–23. doi:10.1212/WNL.55.1.16
19. Howard JF Jr., Barohn RJ, Cutter GR, Freimer M, Juel VC, Mozaffar T, et al. A randomized, double-blind, placebo-controlled phase II study of eculizumab in patients with refractory generalized myasthenia gravis. Muscle Nerve (2013) 48(1):76–84. doi:10.1002/mus.23839
20. Wolfe GI, Barohn RJ, Sanders DB, McDermott MP, Muscle Study G. Comparison of outcome measures from a trial of mycophenolate mofetil in myasthenia gravis. Muscle Nerve (2008) 38(5):1429–33. doi:10.1002/mus.21142
21. Bedlack RS, Simel DL, Bosworth H, Samsa G, Tucker-Lipscomb B, Sanders DB. Quantitative myasthenia gravis score: assessment of responsiveness and longitudinal validity. Neurology (2005) 64(11):1968–70. doi:10.1212/01.WNL.0000163988.28892.79
22. Aarli JA. Myasthenia gravis in the elderly: is it different? Ann N Y Acad Sci (2008) 1132:238–43. doi:10.1196/annals.1405.040
23. Barohn RJ, McIntire D, Herbelin L, Wolfe GI, Nations S, Bryan WW. Reliability testing of the quantitative myasthenia gravis score. Ann N Y Acad Sci (1998) 841:769–72. doi:10.1111/j.1749-6632.1998.tb11015.x
24. Hewer R, Matthews I, Chen S, McGrath V, Evans M, Roberts E, et al. A sensitive non-isotopic assay for acetylcholine receptor autoantibodies. Clin Chim Acta (2006) 364(1–2):159–66. doi:10.1016/j.cccn.2005.05.035
25. Han B, Kang HM, Seo MS, Zaitlen N, Eskin E. Efficient association study design via power-optimized tag SNP selection. Ann Hum Genet (2008) 72(6):834–47. doi:10.1111/j.1469-1809.2008.00469.x
26. Ramchandani D, Weber GF. An osteopontin promoter polymorphism is associated with aggressiveness in breast cancer. Oncol Rep (2013) 30(4):1860–8. doi:10.3892/or.2013.2632
27. Wang J, Nong L, Wei Y, Qin S, Zhou Y, Tang Y. Association of osteopontin polymorphisms with nasopharyngeal carcinoma risk. Hum Immunol (2014) 75(1):76–80. doi:10.1016/j.humimm.2013.09.014
28. Gazal S, Sacre K, Allanore Y, Teruel M, Goodall AH, The CARDIOGENICS Consortiumet al. Identification of secreted phosphoprotein 1 gene as a new rheumatoid arthritis susceptibility gene. Ann Rheum Dis (2015) 74(3):e19. doi:10.1136/annrheumdis-2013-204581
29. Wang JL, Nong LG, Tang YJ, Wei YS, Yang FL, Wang CF. Correlation between OPN gene polymorphisms and the risk of nasopharyngeal carcinoma. Med Oncol (2014) 31(7):20. doi:10.1007/s12032-014-0020-x
30. Han S, Guthridge JM, Harley IT, Sestak AL, Kim-Howard X, Kaufman KM, et al. Osteopontin and systemic lupus erythematosus association: a probable gene-gender interaction. PLoS One (2008) 3(3):e0001757. doi:10.1371/journal.pone.0001757
31. Arjomandi M, Galanter JM, Choudhry S, Eng C, Hu D, Beckman K, et al. Polymorphism in osteopontin gene (SPP1) is associated with asthma and related phenotypes in a Puerto Rican population. Pediatr Allergy Immunol Pulmonol (2011) 24(4):207–14. doi:10.1089/ped.2011.0095
32. Juge PA, van Steenbergen HW, Constantin A, Tobon GJ, Schaeverbeke T, Gazal S, et al. SPP1 rs9138 variant contributes to the severity of radiological damage in anti-citrullinated protein autoantibody-negative rheumatoid arthritis. Ann Rheum Dis (2014) 73(10):1840–3. doi:10.1136/annrheumdis-2014-205539
33. Mi Y, Ren K, Dai F, Zhu L, Feng N. OPN gene polymorphisms, rs17524488 GG/G, rs11730582 T/C, and rs9138 C/A, and cancer risk in a Chinese population. Sci Rep (2015) 5:14164. doi:10.1038/srep14164
34. Xie D, Boyle AP, Wu L, Zhai J, Kawli T, Snyder M. Dynamic trans-acting factor colocalization in human cells. Cell (2013) 155(3):713–24. doi:10.1016/j.cell.2013.09.043
35. Fairfax BP, Makino S, Radhakrishnan J, Plant K, Leslie S, Dilthey A, et al. Genetics of gene expression in primary immune cells identifies cell type-specific master regulators and roles of HLA alleles. Nat Genet (2012) 44(5):502–10. doi:10.1038/ng.2205
36. Jolma A, Kivioja T, Toivonen J, Cheng L, Wei G, Enge M, et al. Multiplexed massively parallel SELEX for characterization of human transcription factor binding specificities. Genome Res (2010) 20(6):861–73. doi:10.1101/gr.100552.109
37. Veyrieras JB, Kudaravalli S, Kim SY, Dermitzakis ET, Gilad Y, Stephens M, et al. High-resolution mapping of expression-QTLs yields insight into human gene regulation. PLoS Genet (2008) 4(10):e1000214. doi:10.1371/journal.pgen.1000214
38. Rittling SR, Singh R. Osteopontin in immune-mediated diseases. J Dent Res (2015) 94(12):1638–45. doi:10.1177/0022034515605270
39. Zou YF, Xu JH, Wang F, Liu S, Tao JH, Cai J, et al. Association study of glucocorticoid receptor genetic polymorphisms with efficacy of glucocorticoids in systemic lupus erythematosus: a prospective cohort study. Autoimmunity (2013) 46(8):531–6. doi:10.3109/08916934.2013.830714
40. Farrugia ME, Carmichael C, Cupka BJ, Warder J, Brennan KM, Burns TM. The modified Rankin scale to assess disability in myasthenia gravis: comparing with other tools. Muscle Nerve (2014) 50(4):501–7. doi:10.1002/mus.24214
41. Zhou Y, Kaminski HJ, Gong B, Cheng G, Feuerman JM, Kusner L. RNA expression analysis of passive transfer myasthenia supports extraocular muscle as a unique immunological environment. Invest Ophthalmol Vis Sci (2014) 55(7):4348–59. doi:10.1167/iovs.14-14422
42. Sakamoto K, Fukushima Y, Ito K, Matsuda M, Nagata S, Minato N, et al. Osteopontin in spontaneous germinal centers inhibits apoptotic cell engulfment and promotes anti-nuclear antibody production in Lupus-prone mice. J Immunol (2016) 197(6):2177–86. doi:10.4049/jimmunol.1600987
Keywords: myasthenia gravis, glucocorticoid, osteopontin, secreted phosphoprotein 1, quantitative myasthenia gravis score
Citation: Xie Y, Li H-F, Sun L, Kusner LL, Wang S, Meng Y, Zhang X, Hong Y, Gao X, Li Y and Kaminski HJ (2017) The Role of Osteopontin and Its Gene on Glucocorticoid Response in Myasthenia Gravis. Front. Neurol. 8:230. doi: 10.3389/fneur.2017.00230
Received: 13 February 2017; Accepted: 11 May 2017;
Published: 31 May 2017
Edited by:
Ghazala Hayat, Saint Louis University, United StatesReviewed by:
Janice C. Wong, Brigham and Women’s Hospital/Massachusetts General Hospital, United StatesHoward Sander, New York University School of Medicine, United States
A. Arturo Leis, Methodist Rehabilitation Center, United States
Copyright: © 2017 Xie, Li, Sun, Kusner, Wang, Meng, Zhang, Hong, Gao, Li and Kaminski. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Hai-Feng Li, ZHJsaGZAMTYzLmNvbQ==;
Henry J. Kaminski, aGthbWluc2tpQG1mYS5nd3UuZWR1
†These authors have contributed equally to this work.
 Yanchen Xie1,2†
Yanchen Xie1,2† Linda L. Kusner
Linda L. Kusner Henry J. Kaminski
Henry J. Kaminski