- 1Neuro-Ophthalmology and Neurocognition, Hôpital Neurologique Pierre Wertheimer, Hospices Civils de Lyon, Bron, France
- 2Lyon I University, Lyon, France
- 3ImpAct Team, CRNL INSERM U1028 CNRS UMR5292, Bron, France
- 4SynatAc Team, Institut NeuroMyogène INSERM U1217/UMR CRS 5310, Lyon, France
Hypertrophic degeneration of the inferior olive is mainly observed in patients developing palatal tremor (PT) or oculopalatal tremor (OPT). This syndrome manifests as a synchronous tremor of the palate (PT) and/or eyes (OPT) that may also involve other muscles from the branchial arches. It is associated with hypertrophic inferior olivary degeneration that is characterized by enlarged and vacuolated neurons, increased number and size of astrocytes, severe fibrillary gliosis, and demyelination. It appears on MRI as an increased T2/FLAIR signal intensity and enlargement of the inferior olive. There are two main conditions in which hypertrophic degeneration of the inferior olive occurs. The most frequent, studied, and reported condition is the development of PT/OPT and hypertrophic degeneration of the inferior olive in the weeks or months following a structural brainstem or cerebellar lesion. This “symptomatic” condition requires a destructive lesion in the Guillain–Mollaret pathway, which spans from the contralateral dentate nucleus via the brachium conjunctivum and the ipsilateral central tegmental tract innervating the inferior olive. The most frequent etiologies of destructive lesion are stroke (hemorrhagic more often than ischemic), brain trauma, brainstem tumors, and surgical or gamma knife treatment of brainstem cavernoma. The most accepted explanation for this symptomatic PT/OPT is that denervated olivary neurons released from inhibitory inputs enlarge and develop sustained synchronized oscillations. The cerebellum then modulates/accentuates this signal resulting in abnormal motor output in the branchial arches. In a second condition, PT/OPT and progressive cerebellar ataxia occurs in patients without structural brainstem or cerebellar lesion, other than cerebellar atrophy. This syndrome of progressive ataxia and palatal tremor may be sporadic or familial. In the familial form, where hypertrophic degeneration of the inferior olive may not occur (or not reported), the main reported etiologies are Alexander disease, polymerase gamma mutation, and spinocerebellar ataxia type 20. Whether or not these are associated with specific degeneration of the dentato–olivary pathway remain to be determined. The most symptomatic consequence of OPT is eye oscillations. Therapeutic trials suggest gabapentin or memantine as valuable drugs to treat eye oscillations in OPT.
Introduction
The terminology and the nosology of hypertrophic inferior olive degeneration and palatal tremor (PT) or oculopalatal tremor (OPT) has evolved over time and needs some clarification. Unilateral or bilateral hypertrophic olivary degeneration (HOD) in the medulla oblongata was first anatomically described in late nineteenth century (1). At the same time, literature focused on the observation of rhythmic PT (2) using different terms such as palatal nystagmus, palatal myoclonus, or palatal myorhythmia. It was finally classified among tremors in 1990 (3). PT is often associated with synchronous eye oscillations and such cases are termed OPT. It can also be associated with synchronous movements of the larynx, pharynx, diaphragm, and facial muscles. PT or OPT has been described in association with the anatomical observation of HOD (4). HOD was later demonstrated on MRI, where it appears as an increased T2/FLAIR signal intensity and enlargement of the inferior olive (5–7). This unique degeneration of the inferior olive most frequently develops weeks or months (8, 9) secondary to a lesion within the dentato–olivary pathway (10), originally referred to as the Guillain–Mollaret triangle (11). The lesion is most often a hemorrhagic stroke.
In 1990, Deuschl et al. suggested differentiating symptomatic PT, developing secondary to brainstem or cerebellar lesions, from essential PT (EPT) for which there is no evidence of a structural lesion (12). Patients with EPT usually have objective ear click, which is less frequent (8%) in the symptomatic form. Involvement of the tensor veli palatini muscle in EPT and of the levator veli palatini muscle in symptomatic PT might explain this clinical difference (3). However, those with symptomatic PT may also experience ear click; to distinguish forms it is of note that EPT patients neither show involvement of eye and other muscles nor evidence of structural abnormalities of the inferior olive (13). Furthermore, the etiology of EPT is heterogeneous with a considerable proportion of psychogenic cases (14) and may disappear over time (15). EPT is therefore a different disease without HOD and does not concern this review; below, PT refers to the symptomatic form.
Later on, Sperling and Herrmann (6) and then Samuel et al. (13) described a syndrome of progressive ataxia and palatal tremor (PAPT). Some of them disclose OPT. In these cases, ataxia progresses and is not the result of a monophasic illness. Sporadic and familial forms of PAPT are described. There is no visible structural causative lesion on the dentato–olivary pathway, but HOD on MRI is present in most cases. Although a specific lesion of the dendato–olivary pathway is not yet identified, PAPT could be considered as a subgroup of symptomatic PT or OPT and will therefore be described in this review.
Clinical Features of PT and OPT
The first observations of synchronous rhythmical movement of the eye and palate were published 150 years ago (2). Since then, different publications have reported the clinical features of this abnormal palatal and eye movement (11, 16, 17).
Symptomatic PT is characterized by involuntary movements of the soft palate and pharynx, due to rhythmic contraction of the levator veli palatine (8, 16) (Video S1 in Supplementary Material). The movements are most commonly bilateral and symmetrical (18). In this case, the soft palate is contracted superiorly and posteriorly along with the uvula with synchronous closing of the pharynx (8). Sometimes the movement can be unilateral, the palate and uvula then being drawn to one side (17, 19). The movements are continuous, the rhythm being most frequently between 100 and 160/min (or 1.5–3 Hz) and persist during sleep (3). Patients with symptomatic PT very rarely complain of ear click (3, 18).
Oculopalatal tremor refers to the synchronous combination of PT and pendular nystagmus. Pendular nystagmus is found to be present in 30% of symptomatic PT (3), probably less frequently in case of PAPT [4 out of 28 cases in Samuel et al. (13)]. In series of patients with pendular nystagmus, up to 18% of those with HOD do not develop PT (20, 21). Patients have mainly vertical pendular oscillations of the eyes with varied combinations of torsional and horizontal components (21–24) (Video S2 in Supplementary Material). The nystagmus can sometimes take the form of convergent–divergent nystagmus (20, 25). This pendular nystagmus is of quite large mean amplitude (8°), high peak velocity (16°/s), and demonstrates irregularity (24) (Figure 1). It is most frequently asymmetric and dissociated in direction in the two eyes (24). While PT is mostly asymptomatic, patients with OPT complain of disturbing oscillopsia, decreased visual acuity, with deterioration of vision-specific health-related quality of life (24, 26). Other than the observed synchrony, attempts have been made to relate characteristics of the nystagmus to the associated palatal movements (22) and to the side of HOD, but the randomness of the directions, waveforms, as well as disconjugacy of nystagmus could just reflect randomly formed couplings in inferior olivary neurons (27). Furthermore, the other associated ocular motor deficit secondary to the brainstem lesion may contribute to disconjugacy of the nystagmus (28).
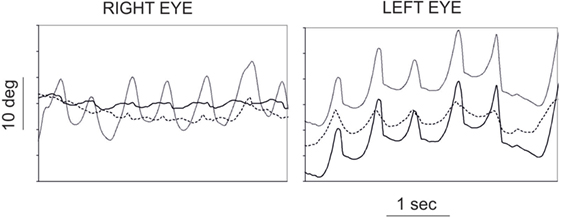
Figure 1. Eye position (in degrees) traces according to time (in seconds) for right (left panel) and left (right panel) eye in an oculopalatal tremor patient. Continuous line: horizontal position, discontinuous line: vertical position, and dotted gray line: torsional position. Adapted from Ref. (24).
Other synchronous movements can be associated with palatal myoclonus, most frequently involving muscles of the gill arches: the face, the tongue, the floor of the mouth, the pharynx, the larynx, and the diaphragm (29) (Videos S1 and S2 in Supplementary Material). In some rare cases, skeletal muscle tremor, mainly of the upper limbs, may be associated (30–32). Some cases of OPT, secondary to lesion of the dentato–olivary pathway, present with focal or generalized dystonia, constituting a variant of OPT (33).
Etiologies
Symptomatic PT and OPT
According to the earliest described cases (2, 4, 11, 16, 17), the most common form of PT/OPT is secondary to a monophasic structural lesion of the brainstem or the cerebellum. The topography of the lesion involves the dentato–olivary pathway, part of the Guillain–Mollaret triangle (11), i.e., the pathway coming from the contralateral dentate nucleus, through the contralateral brachium conjunctivum crossing the midline, turning around the ipsilateral red nucleus, and descending in the ipsilateral central tegmental tract to the inferior olive (Figure 2). Central tegmental tract lesions are the most frequent and seem to be more specifically associated with OPT compared to lesions of dentate nuclei/brachium conjunctivum where only PT is observed (21, 24, 34). In these cases of symptomatic PT or OPT, the condition develops at least 1 month and up to 8 years (median between 10 and 11 months) after the occurrence of the presumed anatomical lesion (9, 13). Symptomatic PT becomes increasingly intense, reaching a peak between 5 and 24 months after lesion (35). Once established, PT or OPT persists for life, with the exception of a few patients in whom PT or OPT is reported to have disappeared completely after many years, although MRI show persistent signal change in the inferior olivary nucleus (21) (Video S3 in Supplementary Material).
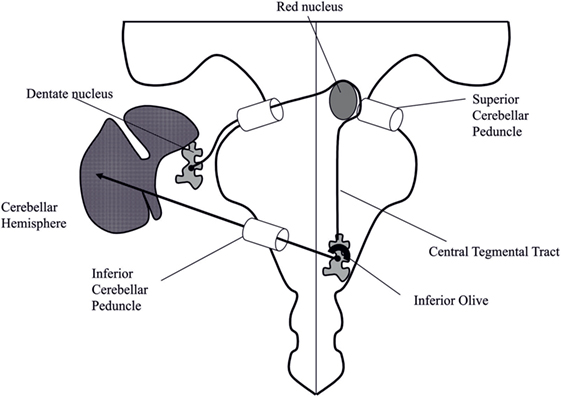
Figure 2. Schematic representation of the Guillain–Mollaret triangle. The pathway coming from the contralateral dentate nucleus, through the contralateral brachium conjunctivum crosses the midline, turns around the ipsilateral red nucleus, and descends in the ipsilateral central tegmental tract to the inferior olive. Adapted from Ref. (36).
The most frequent etiology of structural brainstem or cerebellar lesion is vascular and more often hemorrhagic than ischemic (11, 37). Other etiologies include brain trauma, brainstem tumors, surgical or gamma knife removal of brainstem cavernoma (38), multiple sclerosis (MS), and a broad range of other unspecific lesions [see Table 3 in Samuel et al. (13)]. It is assumed that, to be causative, this primary lesion has to be destructive, a condition that is most easily satisfied by vascular, neurosurgical, or gamma knife lesions (19).
The MS cases might be further discussed. MS has been identified as a common cause of OPT, from 3 (12) to 10% (13). Mostly historical articles report cases of OPT or PT secondary to MS (39, 40). However, in MS, pendular nystagmus is much more often observed without OPT (24, 41, 42). This pendular nystagmus is of small amplitude (1°), low mean peak velocity (6°/s), high mean frequency (4–6 Hz), and is highly regular, like a sine wave (24, 41). Although confused with OPT (42), there is neither an associated PT nor HOD on MRI (24). It may be added that in historical neuropathological cases, almost all lesions are vascular in nature (11). The relatively high proportion of reported MS in PT and OPT might therefore have been overestimated. Indeed, a review of historical cases finds that they do not meet the current clinical criteria for diagnosis of MS or OPT, and neuroimaging or pathology was lacking (40, 43). Some cases seem to correspond to pendular nystagmus associated with MS, other to brainstem hemorrhage, sporadic, or familial PAPT. A notable exception is the report of two patients with clinical, biological, and MRI criteria for MS, developing OPT associated with HOD on MRI (44, 45). Although the second case was complex with history of posterior fossa tumor and radiation therapy (45), these are the only convincing observations of OPT in MS.
According to the topography of the structural lesion, other neurological manifestations may be observed in association with OPT. Patients frequently present contralateral hemiplegia, contralateral hemi-hypoesthesia or spinothalamic syndrome, ipsilateral facial palsy, ipsilateral kinetic cerebellar syndrome (24). In the case of unilateral cerebellar signs, pendular nystagmus is more pronounced in the eye on the affected side (3). Patients also frequently have a deficit in the horizontal eye movement, including fascicular abducens nerve palsy, internuclear ophthalmoplegia, one and a half syndrome, nuclear abducens syndrome (nuclear VI), or horizontal saccadic palsy (24, 41). Central vestibular manifestations have also been reported in association to OPT (46). These manifestations usually result from the primary lesion and present as a monophasic event.
Delayed and progressive worsening of extremity and gait cerebellar ataxia associated with OPT, secondary to identified structural etiologies (stroke; cavernoma; tumor and radiation therapy; subarachnoid hemorrhage; brain trauma) has also been reported (36, 47, 48). The mechanisms of OPT with delayed ataxia following brainstem lesion is not understood, although it seems to occur with larger and bilateral acute brainstem lesions (47). Hemosiderin deposition has been suggested (48), but it cannot explain the cases observed in brainstem tumors and radiotherapy. Delayed ataxia or movement disorder following a monophasic structural lesion without OPT has been reported (49, 50), which could suggest that not all progressive disorders arise from primary neurodegenerative processes (13).
Progressive Ataxia and PT
In 1985, Sperling and Herrmann (6) suggested to distinguish a syndrome associating PT, HOD, and progressive cerebellar ataxia. This entity was reported again (51), and the syndrome of progressive ataxia and palatal tremor (PAPT) was more precisely defined by Samuel et al. (13). The authors suggested differentiating sporadic PAPT from familial forms of PAPT. None of the patients have structural brainstem or cerebellar lesion, but cerebellar ataxia and cerebellar atrophy on MRI progress over years. These cases might correspond to the “degenerative” etiology suggested in older reports [see review in Samuel et al. (13)].
Sporadic PAPT
In sporadic PAPT, other than gait, trunk and limb ataxia, dysarthria, non-specific cerebellar ocular motor dysfunction is observed, such as gaze-evoked nystagmus, jerk vertical nystagmus, hypermetric saccades, and saccadic pursuit (13). All reported patients present PT. Four out of 28 patients reviewed in Samuel et al. (13) have OPT and 2 internuclear ophthalmoplegia, which indicate brainstem involvement. Patients often complain of poor vision due to oscillopsia or diplopia. Hearing loss seems to be quite frequently associated with sporadic PAPT (in four out of six patients). Other neurological manifestations are not specific. The cerebellar ataxia may precede or follow the occurrence of PT (52). Almost all patients show abnormal bilateral signal and/or HOD on MRI. There is no single theory unifying etiologies of sporadic PAPT, although some of them might be due to polymerase gamma (POLG) mutation (53, 54).
In older reports, PT and HOD has also been reported in other degenerative neurological disorders such as pathologically proven progressive supranuclear palsy (55, 56) and other undetermined neurodegenerative diseases (57). The nosology of these cases, presenting with progressive neurological deficit, other than cerebellar ataxia, needs to be clarified.
Familial PAPT
Familial PAPT is more complex than sporadic PAPT and may include a variety of etiologies. They are associated with marked brainstem and cervical cord atrophy with corticospinal tract findings, and the olivary MRI abnormalities may be lacking (13). Three main known etiologies may be considered: Alexander disease, POLG mutation, and spinocerebellar ataxia type 20 (SCA20).
Alexander disease is one of the most reported known etiologies of familial progressive neurological disorder associated with PT (57–61). Alexander disease is a leukodystrophy, that is pathologically characterized by the presence of Rosenthal fibers, and that is caused by mutations in the gene encoding glial fibrillary acidic protein on chromosome 17q21 (62) and present as a progressive neurological disorder that can occur in an infantile, juvenile, or adult form (59). It usually results from de novo mutations, with autosomal dominant inheritance in future generations (59). In juvenile and adult forms, the patients exhibit palatal myoclonus, spastic tetraparesia, mild cerebellar dysfunction, and associated ocular motor abnormalities (60). There is no description of HOD in large series of adult-onset Alexander disease (63), but one recent case with a phenotype of PAPT presented inferior olive hypertrophia (64). In only one case, associated “ocular myoclonus” was described (60).
Recent observations of PT or OPT with HOD, or HOD without clinical manifestations of PT or OPT have been reported in association with POLG mutation (53, 54, 65). Mutations of the mitochondrial DNA (mtDNA) encoded by the POLG gene are an important cause of pediatric and adult-onset mitochondrial disease. In adults, they are associated with multiple mtDNA deletions leading to a wide spectrum of dominant and recessive progressive neurological disorders, often described as syndromes, such as progressive external ophthalmoplegia, Alpers syndrome, sensory ataxic neuropathy, dysarthria and ophthalmoparesis (65, 66). POLG mutation should also be considered in patients with PAPT or progressive ataxia with inferior olive hypersignal (54), even in sporadic cases, and even without other frequently associated neurological signs such as sensory neuronopathy associated with weakness of ocular, pharyngeal, axial, and/or limb muscles (66).
Autosomal dominant SCA20 is a rare spinocerebellar ataxia characterized by a slowly progressive ataxia and dysarthria; two-thirds of those affected also display PT (“myoclonus”) with increased inferior olivary T2 signal (67). In these patients, CT scan shows dentate calcification, without concomitant pallidal calcification. The locus of genetic mutation overlaps that of spinocerebellar ataxia type 5 on chromosome 11, but the phenotypes are very different (68). More recently, a single case of adult-onset GM2-gangliosidosis type II (Sandhoff disease) presenting PT and cerebellar ataxia has been reported, although inferior olive signal was not described (69).
Toxic HOD
There are few reports of reversible inferior olive MRI hypersignal among diffuse MRI changes associated with toxic-induced encephalopathy, such as metronidazole (70). Although none of them was associated with the clinical syndrome of PT or OPT, toxic lesions have a predilection for dentate nuclei and brainstem tegmentum, suggesting reversible lesion of the Guillain–Mollaret triangle (70). One case of reversible PT induced by fluoxetine has been reported, although HOD on MRI is not mentioned (71).
Neuropathology of the Degenerative Hypertrophic Inferior Olivary Nucleus
Histological features of degenerative olivary hypertrophy had been previously reported by numerous authors, mainly in old French publications (1, 4, 11, 31, 72, 73). On postmortem pathological observations, they described macroscopic hypertrophy of the inferior olives associated with neuron swelling with vacuolation (so-called “fenestrated neurons”), bizarre nerve cell shape, severe fibrillary gliosis, and demyelination of the olive white matter (Figure 3). These pathological hallmarks have been thought to result from transynaptic degeneration secondary to a lesion of the ipsilateral central tegmental tract or the contralateral dentate nucleus. More recent immunohistochemical studies identified various changes in the neurons, their neurites, and presynaptic terminals confirming this hypothesis (74, 75). The main finding is a decreased synaptophysin immunoreactivity confirming the presynaptic abnormalities linked to deafferentation (75). In 1981, Goto and Kaneko published a neuropathological study of eight cases of pontine hemorrhage involving unilaterally or bilaterally central tegmental tracts with different survival periods (76). This study demonstrated six neuropathological stages: (1) no olivary changes (<24 h after onset); (2) degeneration of the olivary amiculum (periphery of the olive, at 2–7 days or more); (3) mild olivary enlargement with neuronal hypertrophy and no glial reaction (at about 3 weeks); (4) culminant hypertrophy of both neurons and astrocytes (at about 8.5 months); (5) olivary pseudohypertrophy with neuronal dissolution (at about 9.5 months and later); and (6) olivary atrophy with neuronal disappearance (after a few years).
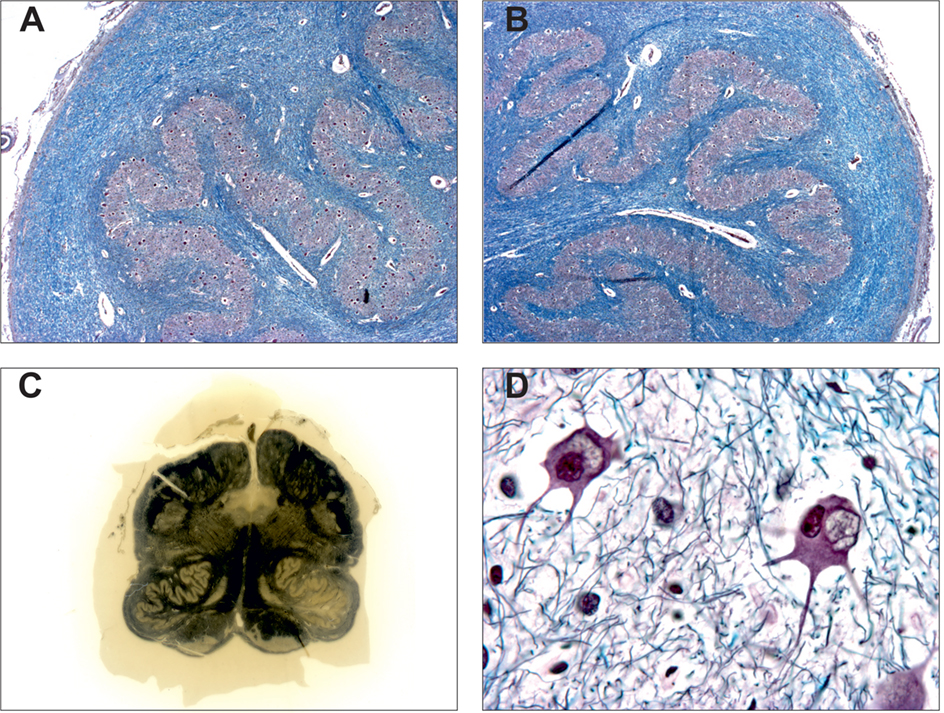
Figure 3. Pathological features of degenerative inferior olive hypertrophy. Hypertrophic inferior olive (A) compared to contralateral side (B) (Bodian Luxol, X200). Note the mild demyelination of the surrounding white matter. (C) Coronal section of the medulla oblongata showing hypertrophy of the left inferior olive (Loyez stain). (D) Swelled and vacuolated nerve cells (“fenestrated neurons”) observed in the hypertrophic inferior olive [from (A), Bodian Luxol, X400]. Courtesy of Charles Duyckaerts and Franck Bielle, Escourolle’s Lab, Pitie-Salpetriere Hospital, Paris, France. Adapted from Ref. (51).
Degenerative olivary hypertrophy is predominantly observed in patients with manifest damage of the dentato–olivary pathway (4, 11, 31, 37, 73, 77). It is predominantly but not always associated with PT/OPT, more specifically following head injury (37). All these neuropathological studies agree with the hypothesis of a unique feature of olivary hypertrophy related to transneuronal degeneration in response to deafferentation following dentato–olivary pathway lesion.
Most interestingly, the only pathological study of PAPT with HOD revealed a unique tau pathology (78). This case showed symmetrical unspecific inferior olivary hypertrophy, without focal brainstem lesion. Strikingly, insoluble tau deposits were exclusively found in some infratentorial neurons, in particular in the inferior olives. Combination of primary tauopathy and secondary degenerative changes in the olives suggested to the authors that “primary degenerative process affecting a portion of olivary neurons could trigger retrograde degeneration of the dentato-olivary fibers, which might cause secondary (deafferentation type) hypertrophic degeneration in other olivary neurons, perhaps through loss of axon collaterals.” Such a hypothesis of a primary focal tauopathy leading to deafferentation-induced hypertrophic degeneration finds an echo with the observations of HOD in patients with pathologically-proven supranuclear palsy tauopathy (56, 57).
Radiological Features
Hypertrophic Olivary Degeneration
The historical observations of neuropathological changes in the inferior olive found their radiological correlates in the observation of increased signal intensity and enlargement of the inferior olive seen on proton density-weighted and T2/FLAIR MRI (6, 7, 79) (Figure 4). The term HOD was then also conventionally used to define these abnormal signals on MRI, even if there is only hypersignal (5, 80). The temporal evolution of these abnormal signals follows pathological changes (81, 82). The hypersignal appears around 1 month after the ictus and persists, while hypertrophy is not usually observed until 6 months after ictus and resolves at approximately 3–4 years after ictus (5) (Figure 5). In some cases, the MRI hypersignal may also return to normal (13, 83). HOD on MRI is unilateral or bilateral in case of symptomatic PT and bilateral in case of PAPT (84, 85). It may also be lacking in familial PAPT (7, 13). In symptomatic PT, HOD usually appears contralateral in case of cerebellar lesion and ipsilateral in case of lateralized central tegmental tract lesion (5, 21, 85). It may precede the clinical manifestations of PT or OPT (80) and even be observed without the development of PT (86). In symptomatic OPT, dissociated pendular nystagmus seems to predict unilateral HOD on MRI with accuracy, while symmetric pendular nystagmus is associated with either unilateral or bilateral HOD (21). Finally, radiological cases of idiopathic HOD without any structural lesion in the Guillain–Mollaret triangle, neither PT, OPT, or PAPT are described (87).
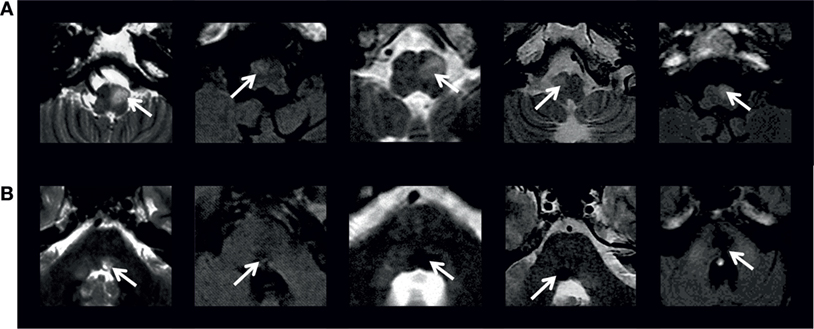
Figure 4. Axial FLAIR or T2 MRI (1.5-T GE scanners) at (A) inferior olive level and (B) midpontine tegmentum level in five patients with symptomatic oculopalatal tremor. White arrows in (A) indicate the abnormal inferior olive hypersignal and in (B) the causative lesion. Adapted from Ref. (24).
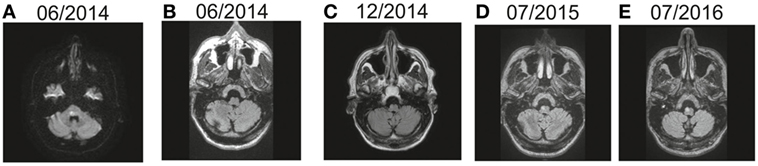
Figure 5. Temporal evolution of right-sided inferior olive hypersignal in a patient with symptomatic oculopalatal tremor. The patient presented a right-sided pontine tegmental lesion in June 2014 seen on the diffusion MRI scan (A), and the medulla showed no abnormal hypersignal on FLAIR MRI (B). Subsequently, right inferior olive hypersignal was observed 6 months later (C), with increasing signal 1 year later (D) and right inferior olive hypertrophy was observed 2 years later (E).
Cerebral Metabolism Imaging
There is discordance in cerebral metabolism imaging; one study found inferior olive hypermetabolism (88), and the other one that used statistical parametric mapping, failed to show metabolic changes in the inferior olive (34).
Cerebellar Changes Associated With HOD
MRI of the cerebellum in patients with symptomatic PT found atrophic changes suggesting a degenerative process involving the dentate nucleus and the cerebellar cortex on the side opposite to the HOD (89). Degeneration of cerebellar cortex secondary to HOD has already been discussed in some neuropathological studies (90).
Physiopathology of PT/OPT Associated with HOD
The main accepted explanation of PT or OPT associated with the development of HOD is that the abnormal inferior olive plays a significant role in PT/OPT (4). First of all, the HOD would develop secondary to dentato–olivary pathway lesion at least for the symptomatic forms, due to a denervation mechanism (77). Normal inferior olivary neurons can generate spontaneous oscillations and are electrically coupled by dendrodendritic gap junctions (91, 92). In case of dentato–olivary pathway lesion, denervated olivary neurons released from inhibitory inputs would enlarge and develop sustained synchronized oscillations (91). Animal models of HOD show the development of spikes on denervated inferior olivary neurons, supporting electrotonic coupling through gap junctions (93). In this hypothesis, inferior olive would be the oscillator of palatal and/or ocular tremor. This is further supported by the observation of disturbed cerebellar function (motor learning) in patients with SPT (94, 95) and the temporal relationship of the development of HOD and the clinical symptoms. This is finally further supported by the observation of inferior olivary nucleus hypermetabolism (86). However, the main criticism against the involvement of inferior olive as part of the mechanism for OPT is the observation of decreased hypertrophy of inferior olive in time while OPT persists, or other observations showing absent inferior olivary nucleus hypermetabolism (34) in patients, and functional imaging showing synchronous decreased cerebellar activity and OPT with clonazepam, but no decrease of inferior olive activity (96). Some authors suggested that inferior olive could be involved in the development of PT/OPT but not in maintaining the symptoms (35).
A fascinating recent model suggested both the implication of inferior olive oscillator generating spike trains at 1–2 Hz and cerebellar modulation/amplification of the motor output (27, 97) (Figure 6). In this model of pendular nystagmus in OPT, the synchronized signal from the inferior olive reaches via climbing fibers Purkinje cells and the deep cerebellar nuclei including vestibular nuclei. In turn, the signal in the vestibular nuclei projects indirectly to the Purkinje cells, via a mossy fiber/granule cells-parallel fiber. The repeated inferior olive pulses would create periodic climbing and parallel fiber inputs to Purkinje cells at approximately the same time and create a learning signal back to the vestibular nuclei, contributing to smoothing and amplifying pulse (27). While this model seems to reproduce many of the aspects of OPT and specifically the 1–2 Hz irregular oscillation, it cannot prove that both inferior olive and cerebellum are necessary to explain it.
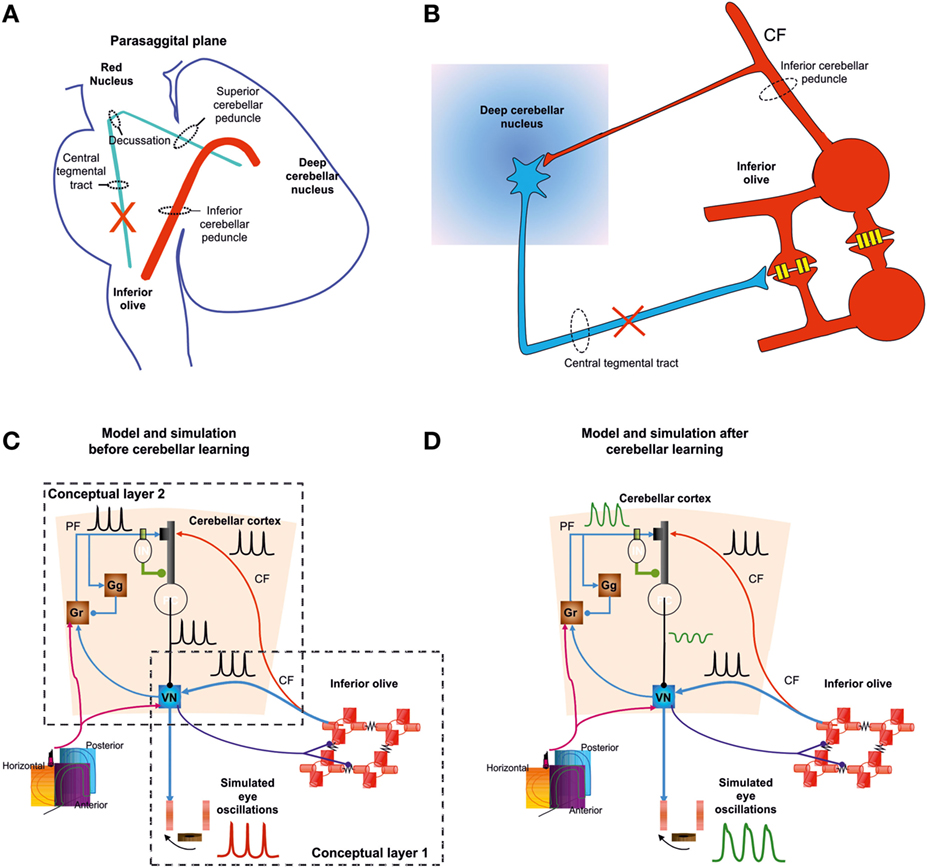
Figure 6. Schematic representation of the Guillain–Mollaret triangle formed by connections between the deep cerebellar nuclei and contralateral inferior olive, which pass near the red nucleus (A). The conduction strength through the dendrodendritic gap junctions (schematized with yellow connexon channels; DD) between adjacent inferior olivary neurons are inhibited by projections from the deep cerebellar nuclei (blue projection) (B). Lesions in the Guillain–Mollaret triangle [red X in (A,B)] also result in hypertrophy of inferior olive neurons causing development of abnormal soma-somatic gap junction. Schematic representation of a model for classical delay conditioning (C,D). Model and traces from simulations after inferior olive hypertrophy but before cerebellar learning (C). Inferior olive and cerebellar modules after hypertrophy and learning (D). Lower left corner shows icon for semicircular canals (C,D). Simulated membrane potentials (black), eye oscillations (magenta). CF, climbing fibers; PF, parallel fibers; DD, dendrodendritic gap junction; SS, soma-somatic gap junction; Gr, granule cell layer; IN, interneurons; PC, Purkinje neurons [(27) with permission for reproduction of material].
The topography of this tremor involving structures corresponding embryologically to the first to fifth branchial arches has received less interest. In 1949, Stern suggested that PT would be the human homolog of a primitive accessory respiratory reflex in gill-breathing vertebrates, leading to the hypothesis of recurrence of an archaic phenomenon (98). The limitation to the branchial arches muscles suggested to authors that the central tegmental tract lesion causes hypersensitivity of the nucleus ambiguous that innervate branchial muscles (99). However, this does not properly explain how 1–2 Hz oscillation develop in the arches, while Shaikh’s model does.
Treatment
Therapeutic trials have been mainly performed on acquired pendular nystagmus, which is the most symptomatic consequence of OPT. The most rigorous treatment trials in acquired pendular nystagmus (due to MS or OPT) led to the proposal of gabapentin or memantine as valuable drugs (26, 100–104). Only one study specifically tested gabapentin, memantine, and baclofen in a group of six patients with acquired pendular nystagmus in OPT with a significant effect of gabapentin and memantine on reduction of nystagmus amplitude and frequency irregularity (102). We have also observed sustained decrease of nystagmus velocity in some patients (Figure 7). Another study found marked improvement of both eye and palate movements as well as complaints by patients (including audible clicks) with trihexyphenidyl; however, patients with PT following a structural lesion, EPT, and MS, but not those with OPT, were included (105). There have been suggestions of testing drugs that reduce electrotonic coupling among hypertrophied inferior olive neurons by blocking connexons like quinine, carbenoxolone, or mefloquine (27), but no study has since been published. Botulinum toxin has been tested on pendular nystagmus in OPT with variable success (106) and in clicking tinnitus in PT (107).
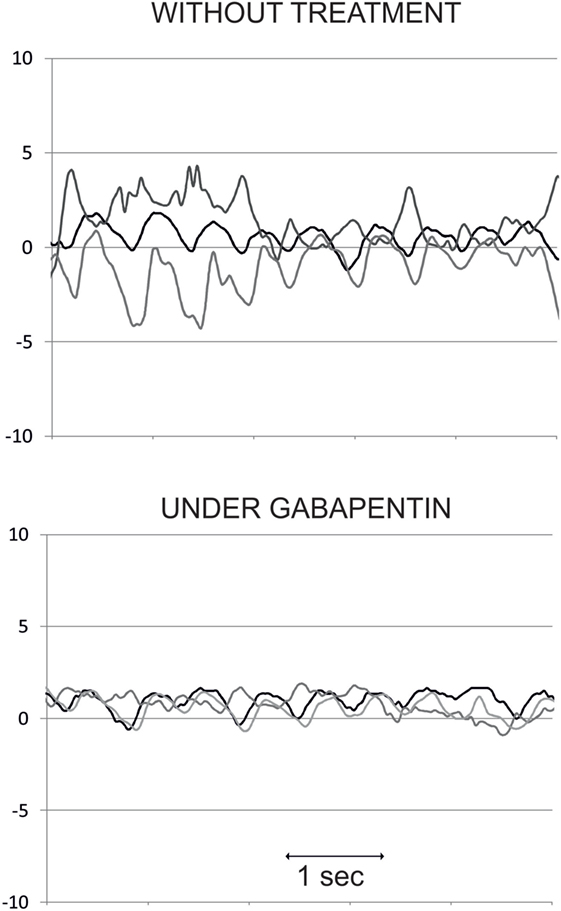
Figure 7. Eye position (in degrees) traces over time (in seconds) in one oculopalatal tremor patient, without treatment (upper panel) and under gabapentin (lower panel). Dark line: horizontal position, gray line: vertical position, and light gray line: torsional position. Note the decrease in nystagmus amplitude, mainly in the torsional plane, under gabapentin.
In a different approach, bilateral deep brain stimulation of the red nucleus in one patient with OPT (and failure of medical treatment) was tested (108). This study failed to show any improvement of eye oscillation. The failure of this intervention may be explained by erroneous interpretation of mechanism of OPT. The hypothesis was to interfere with the rhythmicity of the olivocerebellar circuit, but the target was the afferent dentato–olivary pathway within the red nucleus region.
Author Contributions
Both CT and VD contributed to conception or design of the work; drafting the work and revising; final approval of the version to be published; and the agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The reviewer MW and handling Editor declared their shared affiliation and the handling Editor states that the process nevertheless met the standards of a fair and objective review.
Acknowledgments
We gratefully acknowledge Dr. Franck Bielle and Prof. Charles Duyckaerts for their pathological expertise and for providing microphotographs of postmortem cases (Raymond Escourolle Laboratory, La Salpetriere Hospital, Paris, France). We gratefully acknowledge Philip Robinson for English language editing (Direction de la Recherche Clinique, Hospices Civils de Lyon).
Supplementary Material
The Supplementary Material for this article can be found online at http://journal.frontiersin.org/article/10.3389/fneur.2017.00302/full#supplementary-material.
Video S1. Palatal and chin tremor. This 30-year-old patient presented an acute pontine hemorrhagic stroke resulting in left hemiplegia and right facial palsy. Three months later, he complained of oscillopsia. T2 MRI showed pontine hemorrhagic scar. In addition to pendular nystagmus (not shown), synchronous left-sided palatal and chin tremor around 1 Hz frequency can be observed in this video. T2 MRI showed left-sided hypertrophic olivary degeneration at medullar level.
Video S2. Ocular and upper lip tremor. This 29-year-old patient presented an acute hemorrhagic pontine stroke. He presented with right-sided hemiplegia, left-sided facial palsy, dysarthria, and left-sided gaze palsy. A few months later, he complained of oscillopsia. The video shows binocular incongruent 1.6 Hz pendular nystagmus, and left upper lip synchronous tremor. There was also palatal tremor (not shown). MRI found left-sided hypertrophic olivary degeneration.
Video S3. Decrease of pendular nystagmus with time in a patient with symptomatic oculopalatal tremor (OPT). OPT was diagnosed in 2012 but occurred a few weeks after surgical treatment of bleeding brainstem cavernoma in 2010. During follow-up, the patient described a decrease in oscillopsia and observation disclosed great decrease in nystagmus in 2016, without any pharmacological treatment.
Abbreviations
HOD, hypertrophic olivary degeneration; PT, palatal tremor; OPT, oculopalatal tremor; PAPT, progressive ataxia and palatal tremor; EPT, essential palatal tremor.
References
1. Oppenheim H. Über ein bei Krankheitsprocessen in der hinteren Schadelgrube beobachtetes symptom. Neurol Zbl (1889) 8:132–4.
2. Spencer HR. Pharyngeal and laryngeal “nystagmus”. Lancet (1886) 2:702. doi:10.1016/S0140-6736(01)78357-2
3. Deuschl G, Toro C, Valls-Sole J, Zeffiro T, Zee DS, Hallett M. Symptomatic and essential palatal tremor. 1. Clinical, physiological and MRI analysis. Brain (1994) 117(Pt 4):775–88. doi:10.1093/brain/117.4.775
4. Van Bogaert L, Bertrand I. Sur les myoclonies associées synchrones et rythmiques par lésion en foyer du tronc cérébral. Rev Neurol (1928) 1(2):203.
5. Goyal M, Versnick E, Tuite P, Cyr JS, Kucharczyk W, Montanera W, et al. Hypertrophic olivary degeneration: metaanalysis of the temporal evolution of MR findings. AJNR Am J Neuroradiol (2000) 21(6):1073–7.
6. Sperling MR, Herrmann C Jr. Syndrome of palatal myoclonus and progressive ataxia: two cases with magnetic resonance imaging. Neurology (1985) 35(8):1212–4. doi:10.1212/WNL.35.8.1212
7. Yokota T, Hirashima F, Furukawa T, Tsukagoshi H, Yoshikawa H. MRI findings of inferior olives in palatal myoclonus. J Neurol (1989) 236(2):115–6. doi:10.1007/BF00314408
8. Dubinsky RM, Hallett M. Palatal myoclonus and facial involvement in other types of myoclonus. Adv Neurol (1988) 49:263–78.
9. Matsuo F, Ajax ET. Palatal myoclonus and denervation supersensitivity in the central nervous system. Ann Neurol (1979) 5(1):72–8. doi:10.1002/ana.410050111
10. Lapresle J, Hamida MB. The dentato-olivary pathway. Somatotopic relationship between the dentate nucleus and the contralateral inferior olive. Arch Neurol (1970) 22(2):135–43. doi:10.1001/archneur.1970.00480200041004
11. Guillain G, Mollaret P. Deux cas de myoclonies synchrones et rythmées vélo-pharyngo-laryngo-oculo-diaphragmatiques: le problème anatomique et physiologique. Rev Neurol (1931) 2:545–66.
12. Deuschl G, Mischke G, Schenck E, Schulte-Monting J, Lucking CH. Symptomatic and essential rhythmic palatal myoclonus. Brain (1990) 113(Pt 6):1645–72. doi:10.1093/brain/113.6.1645
13. Samuel M, Torun N, Tuite PJ, Sharpe JA, Lang AE. Progressive ataxia and palatal tremor (PAPT): clinical and MRI assessment with review of palatal tremors. Brain (2004) 127(Pt 6):1252–68. doi:10.1093/brain/awh137
14. Zadikoff C, Lang AE, Klein C. The ’essentials’ of essential palatal tremor: a reappraisal of the nosology. Brain (2006) 129(Pt 4):832–40. doi:10.1093/brain/awh684
15. Jacobs L, Newman RP, Bozian D. Disappearing palatal myoclonus. Neurology (1981) 31(6):748–51. doi:10.1212/WNL.31.6.748
16. Leshin N, Stone TT. Continuous rhythmic movements of the palate, pharynx and larynx. Arch Neurol Psychiat (1931) 31:1236.
17. Riley HA, Brock S. Rhythmic myoclonus of the muscles of the palate, pharynx, larynx and other regions. Arch Neurol Psychiat (1933) 29:726. doi:10.1001/archneurpsyc.1933.02240100045004
18. Deuschl G, Toro C, Hallett M. Symptomatic and essential palatal tremor. 2. Differences of palatal movements. Mov Disord (1994) 9(6):676–8. doi:10.1002/mds.870090615
19. Lapresle J. Rhythmic palatal myoclonus and the dentato-olivary pathway. J Neurol (1979) 220(4):223–30. doi:10.1007/BF00314146
20. Gresty MA, Ell JJ, Findley LJ. Acquired pendular nystagmus: its characteristics, localising value and pathophysiology. J Neurol Neurosurg Psychiatry (1982) 45(5):431–9. doi:10.1136/jnnp.45.5.431
21. Kim JS, Moon SY, Choi KD, Kim JH, Sharpe JA. Patterns of ocular oscillation in oculopalatal tremor: imaging correlations. Neurology (2007) 68(14):1128–35. doi:10.1212/01.wnl.0000258665.37827.f6
22. Nakada T, Kwee IL. Oculopalatal myoclonus. Brain (1986) 109(Pt 3):431–41. doi:10.1093/brain/109.3.431
23. Talks SJ, Elston JS. Oculopalatal myoclonus: eye movement studies, MRI findings and the difficulty of treatment. Eye (1997) 11(Pt 1):19–24. doi:10.1038/eye.1997.4
24. Tilikete C, Jasse L, Pelisson D, Vukusic S, Durand-Dubief F, Urquizar C, et al. Acquired pendular nystagmus in multiple sclerosis and oculopalatal tremor. Neurology (2011) 76(19):1650–7. doi:10.1212/WNL.0b013e318219fa9c
25. Galvez-Ruiz A, Roig C, Muñoz S, Arruga J. Convergent-divergent nystagmus as a manifestation of oculopalatal tremor. Neuroophthalmology (2011) 35(5–6):276–9. doi:10.3109/01658107.2011.609606
26. Thurtell MJ, Joshi AC, Leone AC, Tomsak RL, Kosmorsky GS, Stahl JS, et al. Crossover trial of gabapentin and memantine as treatment for acquired nystagmus. Ann Neurol (2010) 67(5):676–80. doi:10.1002/ana.21991
27. Shaikh AG, Hong S, Liao K, Tian J, Solomon D, Zee DS, et al. Oculopalatal tremor explained by a model of inferior olivary hypertrophy and cerebellar plasticity. Brain (2010) 133(Pt 3):923–40. doi:10.1093/brain/awp323
28. Leigh RJ, Zee DS. The Neurology of Eye Movement. 5th ed. New York: Oxford University Press (2015).
29. Borruat FX. Oculopalatal tremor: current concepts and new observations. Curr Opin Neurol (2013) 26(1):67–73. doi:10.1097/WCO.0b013e32835c60e6
30. Alstadhaug KB. Oculopalatal and cerebellar limb tremor due to hypertrophic olivary degeneration. Eur J Neurol (2007) 14(5):e6–7. doi:10.1111/j.1468-1331.2007.01719.x
31. Guillain G, Thurel R. Myoclonies vélo-pharyngo-laryngo-oculo-diaphragmatiques associées a des myoclonies synchrones squelettiques. Rev Neurol (1932) 2:677–84.
32. Nagaoka M, Narabayashi H. Palatal myoclonus – its remote influence. J Neurol Neurosurg Psychiatry (1984) 47(9):921–6. doi:10.1136/jnnp.47.9.921
33. Shaikh AG, Ghasia FF, DeLong MR, Jinnah HA, Freeman A, Factor SA. Ocular palatal tremor plus dystonia – new syndromic association. Mov Disord Clin Pract (2015) 2(3):267–70. doi:10.1002/mdc3.12193
34. Moon SY, Cho SS, Kim YK, Kim SE, Kim JH, Kim JS. Cerebral glucose metabolism in oculopalatal tremor. Eur J Neurol (2008) 15(1):42–9. doi:10.1111/j.1468-1331.2007.01997.x
35. Nishie M, Yoshida Y, Hirata Y, Matsunaga M. Generation of symptomatic palatal tremor is not correlated with inferior olivary hypertrophy. Brain (2002) 125(Pt 6):1348–57. doi:10.1093/brain/awf126
36. Tilikete C, Hannoun S, Nighoghossian N, Sappey-Marinier D. Oculopalatal tremor and severe late-onset cerebellar ataxia. Neurology (2008) 71(4):301. doi:10.1212/01.wnl.0000318287.29513.6d
37. Jellinger K. Hypertrophy of the inferior olives. Report on 29 cases. Z Neurol (1973) 205(2):153–74. doi:10.1007/BF00316018
38. Yun JH, Ahn JS, Park JC, Kwon DH, Kwun BD, Kim CJ. Hypertrophic olivary degeneration following surgical resection or gamma knife radiosurgery of brainstem cavernous malformations: an 11-case series and a review of literature. Acta Neurochir (2013) 155(3):469–76. doi:10.1007/s00701-012-1567-y
39. Chokroverty S, Barron KD. Palatal myoclonus and rhythmic ocular movements: a polygraphic study. Neurology (1969) 19(10):975–82. doi:10.1212/WNL.19.10.975
40. Dereux MJ. Myoclonies vélo-pharyngo-laryngées et oculaires chez un malade atteint de sclérose en plaques. Rev Neurol (1938) 69:135–8.
41. Itoh Y, Sakata E. Acquired pendular oscillation after brain-stem hemorrhage. Acta Otolaryngol Suppl (1989) 468:205–8. doi:10.3109/00016488909139046
42. Lopez LI, Bronstein AM, Gresty MA, Du Boulay EP, Rudge P. Clinical and MRI correlates in 27 patients with acquired pendular nystagmus. Brain (1996) 119(Pt 2):465–72. doi:10.1093/brain/119.2.465
43. Tranchant C, Bhatia KP, Marsden CD. Movement disorders in multiple sclerosis. Mov Disord (1995) 10(4):418–23. doi:10.1002/mds.870100403
44. Revol A, Vighetto A, Confavreux C, Trillet M, Aimard G. [Oculo-palatal myoclonus and multiple sclerosis]. Rev Neurol (1990) 146(8–9):518–21.
45. Sudhakar P, Parmar H, Cornblath WT. Oculopalatal tremor in multiple sclerosis with spontaneous resolution. Neurol Clin Pract (2012) 2(3):255–7. doi:10.1212/CPJ.0b013e31826af275
46. Tarnutzer AA, Palla A, Marti S, Schuknecht B, Straumann D. Hypertrophy of the inferior olivary nucleus impacts perception of gravity. Front Neurol (2012) 3:79. doi:10.3389/fneur.2012.00079
47. Eggenberger E, Cornblath W, Stewart DH. Oculopalatal tremor with tardive ataxia. J Neuroophthalmol (2001) 21(2):83–6. doi:10.1097/00041327-200106000-00003
48. Kumar N, Eggers SD, Milone M, Keegan BM. Acquired progressive ataxia and palatal tremor: importance of MRI evidence of hemosiderin deposition and vascular malformations. Parkinsonism Relat Disord (2011) 17(7):565–8. doi:10.1016/j.parkreldis.2011.04.018
49. Louis ED, Lynch T, Ford B, Greene P, Bressman SB, Fahn S. Delayed-onset cerebellar syndrome. Arch Neurol (1996) 53(5):450–4. doi:10.1001/archneur.1996.00550120016004
50. Scott BL, Jankovic J. Delayed-onset progressive movement disorders after static brain lesions. Neurology (1996) 46(1):68–74. doi:10.1212/WNL.46.1.68
51. Leger JM, Duyckaerts C, Brunet P. Syndrome of palatal myoclonus and progressive ataxia: report of a case. Neurology (1986) 36(10):1409–10. doi:10.1212/WNL.36.10.1409-a
52. de Jong FJ, Boon AJ. Progressive ataxia and palatal tremor – two cases with an unusual clinical presentation and course. Parkinsonism Relat Disord (2012) 18(7):904–5. doi:10.1016/j.parkreldis.2012.04.002
53. Mongin M, Delorme C, Lenglet T, Jardel C, Vignal C, Roze E. Progressive ataxia and palatal tremor: think about POLG mutations. Tremor Other Hyperkinet Mov (N Y) (2016) 6:382. doi:10.7916/D86M36RK
54. Nicastro N, Ranza E, Antonarakis SE, Horvath J. Pure progressive ataxia and palatal tremor (PAPT) associated with a new polymerase gamma (POLG) mutation. Cerebellum (2016) 15(6):829–31. doi:10.1007/s12311-015-0749-6
55. Hanihara T, Amano N, Takahashi T, Itoh Y, Yagishita S. Hypertrophy of the inferior olivary nucleus in patients with progressive supranuclear palsy. Eur Neurol (1998) 39(2):97–102. doi:10.1159/000007915
56. Suyama N, Kobayashi S, Isino H, Iijima M, Imaoka K. Progressive supranuclear palsy with palatal myoclonus. Acta Neuropathol (1997) 94(3):290–3. doi:10.1007/s004010050706
57. Kulkarni PK, Muthane UB, Taly AB, Jayakumar PN, Shetty R, Swamy HS. Palatal tremor, progressive multiple cranial nerve palsies, and cerebellar ataxia: a case report and review of literature of palatal tremors in neurodegenerative disease. Mov Disord (1999) 14(4):689–93. doi:10.1002/1531-8257(199907)14:4<689::AID-MDS1022>3.0.CO;2-8
58. de Yebenes JG, Vazquez A, Rabano J, de Seijas EV, Urra DG, Obregon MC, et al. Hereditary branchial myoclonus with spastic paraparesis and cerebellar ataxia: a new autosomal dominant disorder. Neurology (1988) 38(4):569–72. doi:10.1212/WNL.38.4.569
59. Howard KL, Hall DA, Moon M, Agarwal P, Newman E, Brenner M. Adult-onset Alexander disease with progressive ataxia and palatal tremor. Mov Disord (2008) 23(1):118–22. doi:10.1002/mds.21774
60. Schwankhaus JD, Parisi JE, Gulledge WR, Chin L, Currier RD. Hereditary adult-onset Alexander’s disease with palatal myoclonus, spastic paraparesis, and cerebellar ataxia. Neurology (1995) 45(12):2266–71. doi:10.1212/WNL.45.12.2266
61. Thyagarajan D, Chataway T, Li R, Gai WP, Brenner M. Dominantly-inherited adult-onset leukodystrophy with palatal tremor caused by a mutation in the glial fibrillary acidic protein gene. Mov Disord (2004) 19(10):1244–8. doi:10.1002/mds.20161
62. Li R, Johnson AB, Salomons G, Goldman JE, Naidu S, Quinlan R, et al. Glial fibrillary acidic protein mutations in infantile, juvenile, and adult forms of Alexander disease. Ann Neurol (2005) 57(3):310–26. doi:10.1002/ana.20406
63. Graff-Radford J, Schwartz K, Gavrilova RH, Lachance DH, Kumar N. Neuroimaging and clinical features in type II (late-onset) Alexander disease. Neurology (2014) 82(1):49–56. doi:10.1212/01.wnl.0000438230.33223.bc
64. Sebesto JR, van Gerpen JA. Teaching video neuroimages: palatal tremor in adult-onset Alexander disease. Neurology (2016) 86(24):e252. doi:10.1212/WNL.0000000000002763
65. Kinghorn KJ, Kaliakatsos M, Blakely EL, Taylor RW, Rich P, Clarke A, et al. Hypertrophic olivary degeneration on magnetic resonance imaging in mitochondrial syndromes associated with POLG and SURF1 mutations. J Neurol (2013) 260(1):3–9. doi:10.1007/s00415-012-6564-9
66. Tchikviladze M, Gilleron M, Maisonobe T, Galanaud D, Laforet P, Durr A, et al. A diagnostic flow chart for POLG-related diseases based on signs sensitivity and specificity. J Neurol Neurosurg Psychiatry (2015) 86(6):646–54. doi:10.1136/jnnp-2013-306799
67. Storey E. Spinocerebellar Ataxia Type 20. Pagon RA, Adam MP, Ardinger HH, Wallace SE, Amemiya A, Bean LJH, et al., editors. Seattle, WA: University of Washington (2007).
68. Storey E, Knight MA, Forrest SM, Gardner RJ. Spinocerebellar ataxia type 20. Cerebellum (2005) 4(1):55–7. doi:10.1080/14734220410019048
69. Pretegiani E, Rosini F, Federighi P, Cerase A, Dotti MT, Rufa A. Pendular nystagmus, palatal tremor and progressive ataxia in GM2-gangliosidosis. Eur J Neurol (2015) 22(6):e67–9. doi:10.1111/ene.12661
70. Seok JI, Yi H, Song YM, Lee WY. Metronidazole-induced encephalopathy and inferior olivary hypertrophy: lesion analysis with diffusion-weighted imaging and apparent diffusion coefficient maps. Arch Neurol (2003) 60(12):1796–800. doi:10.1001/archneur.60.12.1796
71. Bharucha KJ, Sethi KD. Complex movement disorders induced by fluoxetine. Mov Disord (1996) 11(3):324–6. doi:10.1002/mds.870110318
72. Lapresle J, Ben Hamida M. Correspondance somatotopique, secteur par secteur, des dégénérescences de l’olive bulbaire consécutives à des lésions limitées du noyau dentelé contro-latéral. Rev Neurol (1965) 113(4):439–48.
73. Marie P, Foix C. Sur la dégénération pseudo-hypertrophique de l’olive bulbaire. Rev Neurol (1913) 26:48–52.
74. Ogawa K, Kamei S, Ichihara K, Uehara K, Suzuki Y, Uchihara T, et al. Immunohistochemical study of pseudohypertrophy of the inferior olivary nucleus. Clin Neuropathol (2014) 33(1):68–75. doi:10.5414/NP300594
75. Ogawa K, Mizutani T, Uehara K, Minami M, Suzuki Y, Uchihara T. Pathological study of pseudohypertrophy of the inferior olivary nucleus. Neuropathology (2010) 30(1):15–23. doi:10.1111/j.1440-1789.2009.01033.x
76. Goto N, Kaneko M. Olivary enlargement: chronological and morphometric analyses. Acta Neuropathol (1981) 54(4):275–82. doi:10.1007/BF00697000
77. Gauthier JC, Blackwood W. Enlargement of the inferior olivary nucleus in association with lesions of the central tegmental tract or dentate nucleus. Brain (1961) 84(3):341–61. doi:10.1093/brain/84.3.341
78. Mari Z, Halls AJM, Vortmeyer A, Zhukareva V, Kunihiro U, Lee VM, et al. Clinico-pathological correlation in progressive ataxia and palatal tremor: a novel tauopathy. Mov Disord Clin Pract (2014) 50–6. doi:10.1002/mdc3.12014
79. Pierot L, Cervera-Pierot P, Delattre JY, Duyckaerts C, Chiras J, Brunet P. Palatal myoclonus and inferior olivary lesions: MRI-pathologic correlation. J Comput Assist Tomogr (1992) 16(1):160–3.
80. Yokota T, Tsukagoshi H. Olivary hypertrophy precedes the appearance of palatal myoclonus. J Neurol (1991) 238(7):408. doi:10.1007/BF00319861
81. Birbamer G, Buchberger W, Felber S, Aichner F. MR appearance of hypertrophic olivary degeneration: temporal relationships. AJNR Am J Neuroradiol (1992) 13(5):1501–3.
82. Revel MP, Mann M, Brugieres P, Poirier J, Gaston A. MR appearance of hypertrophic olivary degeneration after contralateral cerebellar hemorrhage. AJNR Am J Neuroradiol (1991) 12(1):71–2.
83. Yanagisawa T, Sugihara H, Shibahara K, Kamo T, Fujisawa K, Murayama M. Natural course of combined limb and palatal tremor caused by cerebellar-brain stem infarction. Mov Disord (1999) 14(5):851–4. doi:10.1002/1531-8257(199909)14:5<851::AID-MDS1022>3.0.CO;2-G
84. Hirono N, Kameyama M, Kobayashi Y, Udaka F, Mezaki T, Abe K, et al. MR demonstration of a unilateral olivary hypertrophy caused by pontine tegmental hematoma. Neuroradiology (1990) 32(4):340–2. doi:10.1007/BF00593060
85. Uchino A, Hasuo K, Uchida K, Matsumoto S, Tsukamoto Y, Ohno M, et al. Olivary degeneration after cerebellar or brain stem haemorrhage: MRI. Neuroradiology (1993) 35(5):335–8. doi:10.1007/BF00588362
86. Robin JJ, Alcala H. Olivary hypertrophy without palatal myoclonus associated with a metastatic lesion to the pontine tegmentum. Neurology (1975) 25(8):771–5. doi:10.1212/WNL.25.8.771
87. Gu CN, Carr CM, Kaufmann TJ, Kotsenas AL, Hunt CH, Wood CP. MRI findings in nonlesional hypertrophic olivary degeneration. J Neuroimaging (2015) 25(5):813–7. doi:10.1111/jon.12267
88. Dubinsky RM, Hallett M, Di Chiro G, Fulham M, Schwankhaus J. Increased glucose metabolism in the medulla of patients with palatal myoclonus. Neurology (1991) 41(4):557–62. doi:10.1212/WNL.41.4.557
89. Kim SJ, Lee JH, Suh DC. Cerebellar MR changes in patients with olivary hypertrophic degeneration. AJNR Am J Neuroradiol (1994) 15(9):1715–9.
90. Anderson JR, Treip CS. Hypertrophic olivary degeneration and Purkinje cell degeneration in a case of long-standing head injury. J Neurol Neurosurg Psychiatry (1973) 36(5):826–32. doi:10.1136/jnnp.36.5.826
91. Manor Y, Rinzel J, Segev I, Yarom Y. Low-amplitude oscillations in the inferior olive: a model based on electrical coupling of neurons with heterogeneous channel densities. J Neurophysiol (1997) 77(5):2736–52.
92. Sotelo C, Llinas R, Baker R. Structural study of inferior olivary nucleus of the cat: morphological correlates of electrotonic coupling. J Neurophysiol (1974) 37(3):541–59.
93. Ruigrok TJ, de Zeeuw CI, Voogd J. Hypertrophy of inferior olivary neurons: a degenerative, regenerative or plasticity phenomenon. Eur J Morphol (1990) 28(2–4):224–39.
94. Deuschl G, Toro C, Valls-Sole J, Hallett M. Symptomatic and essential palatal tremor. 3. Abnormal motor learning. J Neurol Neurosurg Psychiatry (1996) 60(5):520–5. doi:10.1136/jnnp.60.5.520
95. Shaikh AG, Wong AL, Optican LM, Zee DS. Impaired motor learning in a disorder of the inferior olive: is the cerebellum confused? Cerebellum (2017) 16(1):158–67. doi:10.1007/s12311-016-0785-x
96. Yakushiji Y, Otsubo R, Hayashi T, Fukuchi K, Yamada N, Hasegawa Y, et al. Glucose utilization in the inferior cerebellar vermis and ocular myoclonus. Neurology (2006) 67(1):131–3. doi:10.1212/01.wnl.0000223837.52895.2e
97. Hong S, Leigh RJ, Zee DS, Optican LM. Inferior olive hypertrophy and cerebellar learning are both needed to explain ocular oscillations in oculopalatal tremor. Prog Brain Res (2008) 171:219–26. doi:10.1016/S0079-6123(08)00631-6
98. Stern MM. Rhythmic palatopharyngeal myoclonus. J Nerv Ment Dis (1949) 109:48–53. doi:10.1097/00005053-194901000-00006
99. Kane SA, Thach WT. Palatal Myoclonus and Function of the Inferior Olive: Are They Related? Strat P, editor. Berlin: Springer (1989).
100. Averbuch-Heller L, Tusa RJ, Fuhry L, Rottach KG, Ganser GL, Heide W, et al. A double-blind controlled study of gabapentin and baclofen as treatment for acquired nystagmus. Ann Neurol (1997) 41(6):818–25. doi:10.1002/ana.410410620
101. Bandini F, Castello E, Mazzella L, Mancardi GL, Solaro C. Gabapentin but not vigabatrin is effective in the treatment of acquired nystagmus in multiple sclerosis: how valid is the GABAergic hypothesis? J Neurol Neurosurg Psychiatry (2001) 71(1):107–10. doi:10.1136/jnnp.71.1.107
102. Shaikh AG, Thurtell MJ, Optican LM, Leigh RJ. Pharmacological tests of hypotheses for acquired pendular nystagmus. Ann N Y Acad Sci (2011) 1233:320–6. doi:10.1111/j.1749-6632.2011.06118.x
103. Starck M, Albrecht H, Pollmann W, Dieterich M, Straube A. Acquired pendular nystagmus in multiple sclerosis: an examiner-blind cross-over treatment study of memantine and gabapentin. J Neurol (2010) 257(3):322–7. doi:10.1007/s00415-009-5309-x
104. Starck M, Albrecht H, Pollmann W, Straube A, Dieterich M. Drug therapy for acquired pendular nystagmus in multiple sclerosis. J Neurol (1997) 244(1):9–16. doi:10.1007/PL00007728
105. Jabbari B, Rosenberg M, Scherokman B, Gunderson CH, McBurney JW, McClintock W. Effectiveness of trihexyphenidyl against pendular nystagmus and palatal myoclonus: evidence of cholinergic dysfunction. Mov Disord (1987) 2(2):93–8. doi:10.1002/mds.870020202
106. Repka MX, Savino PJ, Reinecke RD. Treatment of acquired nystagmus with botulinum neurotoxin A. Arch Ophthalmol (1994) 112(10):1320–4. doi:10.1001/archopht.1994.01090220070025
107. Penney SE, Bruce IA, Saeed SR. Botulinum toxin is effective and safe for palatal tremor: a report of five cases and a review of the literature. J Neurol (2006) 253(7):857–60. doi:10.1007/s00415-006-0039-9
Keywords: symptomatic palatal tremor, progressive ataxia and palatal tremor, pendular nystagmus, hypertrophic degeneration of inferior olive, dentato–olivary pathway, Guillain–Mollaret triangle
Citation: Tilikete C and Desestret V (2017) Hypertrophic Olivary Degeneration and Palatal or Oculopalatal Tremor. Front. Neurol. 8:302. doi: 10.3389/fneur.2017.00302
Received: 05 April 2017; Accepted: 12 June 2017;
Published: 29 June 2017
Edited by:
Aasef G. Shaikh, Case Western Reserve University, United StatesReviewed by:
Mark F. Walker, Case Western Reserve University, United StatesKonrad P. Weber, University of Zurich, Switzerland
Copyright: © 2017 Tilikete and Desestret. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Caroline Tilikete, Y2Fyb2xpbmUudGlsaWtldGVAaW5zZXJtLmZy
 Caroline Tilikete
Caroline Tilikete Virginie Desestret1,2,4
Virginie Desestret1,2,4