- 1Department of Neurological Surgery, University of Louisville School of Medicine, Louisville, KY, United States
- 2Department of Radiology, University of Louisville School of Medicine, Louisville, KY, United States
- 3Department of Bioinformatics and Biostatistics, University of Louisville School of Public Health and Information Sciences, Louisville, KY, United States
Background and purpose: Acute central retinal artery occlusion (CRAO) is a serious ophthalmologic emergency that may result in monocular blindness. To date, studies evaluating intra-arterial thrombolysis (IAT) have not shown a definitive clinical benefit. We have conducted a systematic review with a meta-analysis to effectively evaluate this treatment option.
Methods: A systematic literature search was focused on studies containing five or more patients undergoing IAT that included a control group treated with standard therapy. Pooled meta-analysis was performed.
Results: Five retrospective controlled studies and one randomized clinical trial were identified satisfying all inclusion criteria resulting in the analysis of 236 patients treated with IAT and 255 patients treated with ST. A pooled fixed effects analysis resulted in an estimated odds ratio of 2.52, 95% CI (1.69, 3.77) (P < 0.0001) favoring IAT.
Conclusion: IAT is a promising therapeutic option for CRAO with great potential. Further randomized trials are needed to establish a significant benefit and ensure the safety of the intervention.
Introduction
Acute non-arteritic central retinal artery occlusion (CRAO) is a rare ophthalmologic emergency. Less than 30% of CRAO patients will demonstrate a spontaneous improvement in visual acuity (VA) (1–3). Despite the severity of this condition, few effective therapeutic options exist. Current standard therapy (ST) includes a combination of non-invasive or minimally invasive therapies such as aspirin, topical beta-blockers, carbogen, ocular massage, and anterior chamber paracentesis. These therapies have all largely demonstrated limited efficacy, even though they remain the only therapeutic options commonly available (4, 5).
Intravenous and endovascular methods have been examined in CRAO. Intra-arterial thrombolysis (IAT) may reduce systemic side effects by potentially lowering the total dose needed compared to intravenous administration (6–8). Similarly, direct administration of thrombolytic agents to the target vessel may improve overall efficacy. This approach is an attractive therapeutic option given that systems of care that are currently being upgraded for the endovascular treatment of acute stroke via thrombectomy can be leveraged and utilized for rapid IAT treatment, despite the difference in the treatment paradigm (9).
The published results of IAT for CRAO have been inconsistent resulting in a controversial view of its efficacy. The most recent meta-analysis evaluating IAT for CRAO was conducted in 2000, reported on only 100 patients treated with IAT, and did not utilize any controlled studies (10). In this manuscript, we provide a systematic review of the most relevant literature and perform a meta-analysis of controlled studies evaluating the efficacy of IAT for improving VA in CRAO.
Methods
This meta-analysis was performed in accordance with the ‘Preferred Reporting Items for Systematic Review and Meta-Analyses’ (PRISMA) (11). We conducted a literature search in MEDLINE and EMBASE for studies utilizing IAT in the treatment of CRAO from January 1, 1946 to January 1, 2015. Key words utilized included “retinal artery,” “intra-arterial fibrinolysis,” “intra-arterial thrombolysis,” and “intra-arterial thrombolytic.” Relevant articles were systematically reviewed for content and overall significance. Inclusion criteria for qualitative systematic reviews consisted of studies in English containing five or more patients with acute onset non-arteritic CRAO undergoing IAT with either urokinase or rt-PA. In order to be included for the quantitative meta-analysis, reports had to include a control group treated with ST.
The number of patients with visual improvement was extracted and odds ratios (OR) with 95% confidence intervals were calculated for each study. Visual improvement was defined as any improvement in VA in either the IAT or ST group. When more than one measure of visual improvement was reported, the reported primary outcome was chosen. When a primary outcome for visual improvement was not designated or apparent, the most restrictive definition of improvement was chosen.
The meta-analysis was then conducted using the inverse variance method for weighting studies and the DerSimonian-Laird estimator for quantifying heterogeneity. Both fixed- and random-effects model meta-analysis were performed and a forest plot was produced using Review Manager version 5.3.5. The initial analysis was exploratory and included only the retrospective non-randomized studies due to their presumed similar level of evidence. Our primary analysis was performed in identical fashion, but additionally included the results of the randomized trial.
SAS 9.3 was used to construct a mixed effects model to evaluate the effect of study quality on treatment outcomes and assess for suitability of including studies of different quality in the pooled meta-analysis. The mixed effect model utilized was conducted in accordance with the methods described in Sheu and Suzuki (12). This approach allowed modeling of the log odds ratio (log OR) as the response variable. The variance of the log OR for each study was derived from Argresti et al. (13).
The R package meta (R version 3.1.1, The R Foundation for Statistical Computing) was used to construct a funnel plot to test for publication bias of study outcomes with respect to study size, which included a formal test of funnel plot asymmetry. All studies included in the qualitative analysis were assessed for additional biases including selection bias. A summary of the qualitative analysis is provided in Table 1.
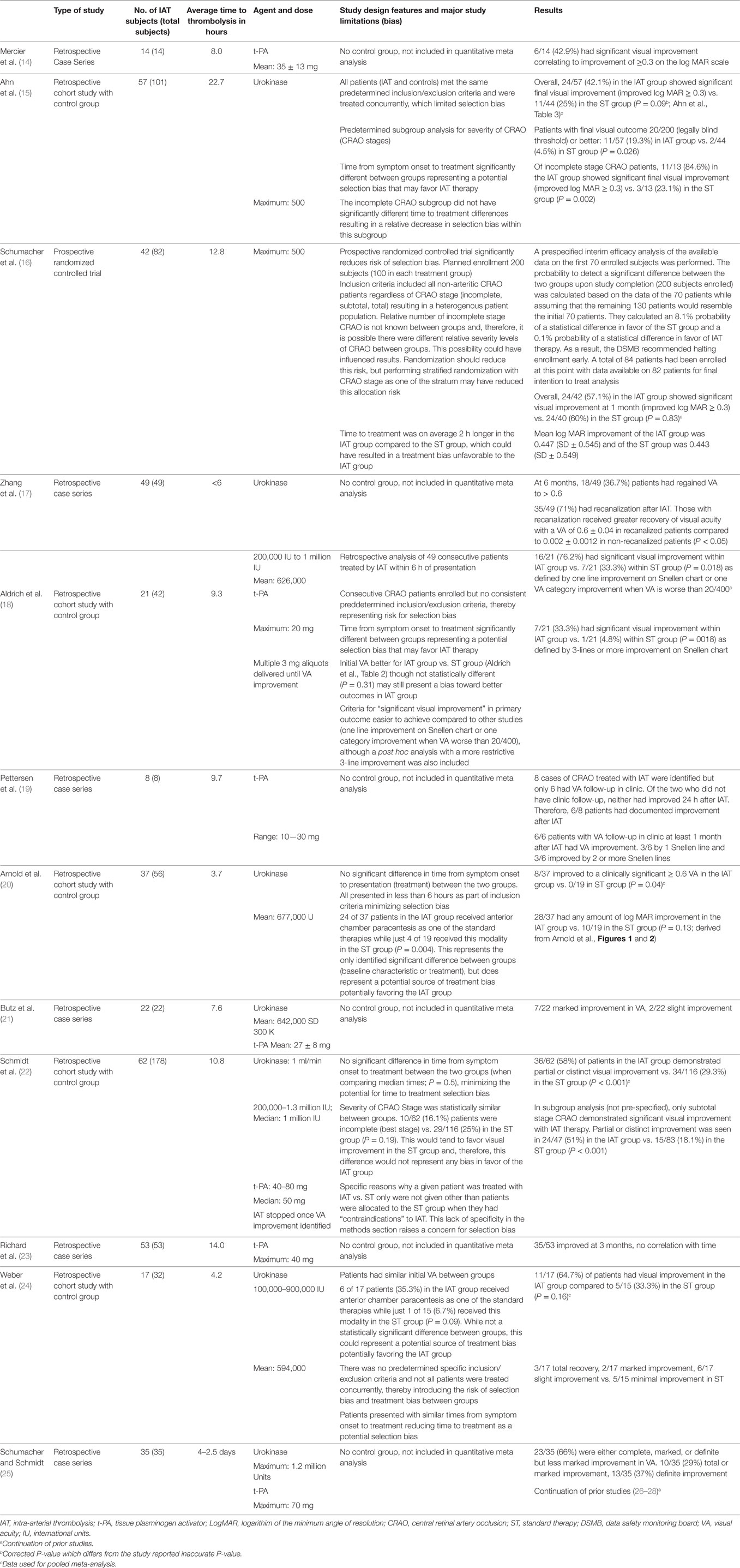
Table 1. Qualitative analysis of systematic review for intra-arterial thrombolysis treatment for central retinal artery occlusion.
Results
Systematic Review
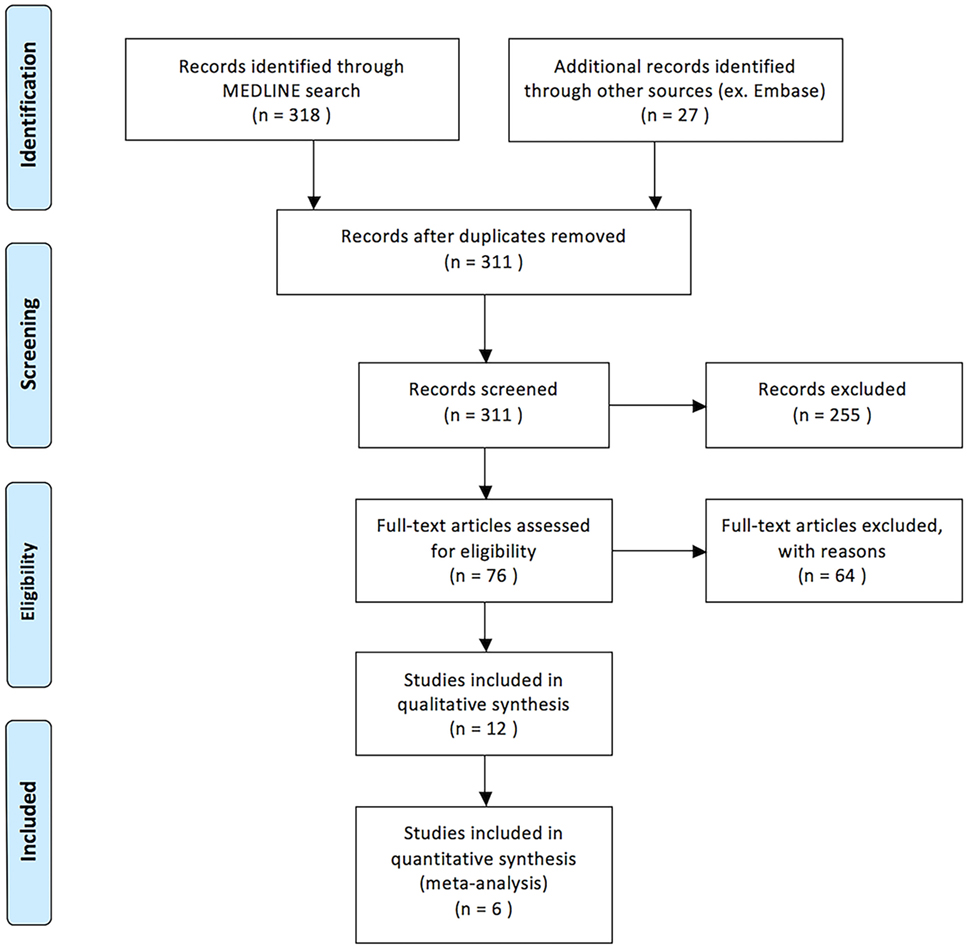
Our search yielded 318 titles from MEDLINE and 27 titles from EMBASE (Figure 1). Seventy-six articles were identified as being relevant to our topic. Twenty-one relevant studies were included as they were containing five or more patients treated with IAT for acute non-arteritic CRAO (14–25, 29–34). Of these, nine were excluded either for not being written in English, representing duplicate data, or both. In total, 12 English language studies were identified as being relevant to the topic of discussion and satisfying all criteria for review. Eleven of these studies were retrospective in nature and one was a randomized controlled trial (14–25). A total of 417 patients treated with IAT are presented.
Upon review of the included studies, 236 of 417 patients (56.5%) treated with IAT demonstrated improvement in VA. Mean time from symptom onset to thrombolysis varied widely between studies ranging from 4.2 to 22.7 h. Mean age of patients treated with IAT was 61.9 years. Either urokinase or rt-PA was administered to patients in the IAT group. Urokinase dosing ranged between 200,000 and 1,300,000 IU. The maximum dose of rt-PA administered across all studies was 80 mg. Table 1 is a detailed summary of all 12 studies including their results and an assessment of bias.
Meta-Analysis
Six studies fulfilled all inclusion criteria for evaluation in the quantitative meta-analysis. Overall, 236 patients were treated with IAT and 255 were managed conservatively with ST. Of those treated with IAT, 119/236 (50.4%) demonstrated an improvement in VA compared with 81/255 (31.8%) of those treated with ST (P < 0.005). Mean time from last known normal to treatment with IAT was 9.51 (range 1–172) h compared with 10.58 h for those treated with conservative treatment (range 2.0–22.5). No studies included demonstrated any statistically significant difference in age, gender, or presenting VA.
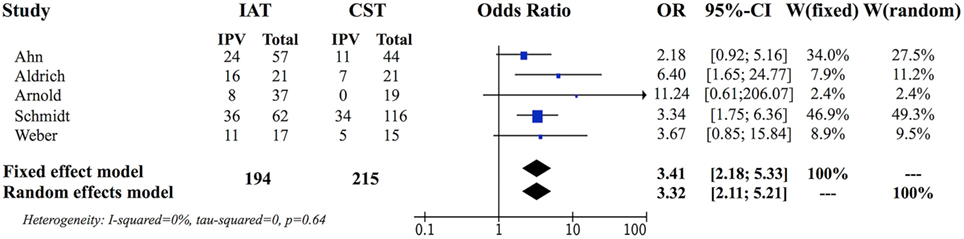
The preliminary weighted pooled analysis included only the five retrospective non-randomized studies of a similar level of evidence (15, 18, 20, 22, 24). The estimated pooled ORs for fixed effects analysis was 3.41, 95% CI (2.18, 5.33), which was statistically significant (P < 0.0001) favoring IAT. The individual study ORs ranged from 2.18 to 11.24 (Figure 2) (15, 18).
We then evaluated the randomized controlled trial by the European Assessment Group for Lysis in the Eye (EAGLE) equally with the retrospective studies (16). The individual OR for the EAGLE study was 0.89, 95% CI (0.37, 2.14) demonstrating no significant difference between IAT and ST in that study. Formal meta-analysis of all six studies, including the EAGLE study, resulted in fixed-effects pooled OR of 2.52, 95% CI (1.69, 3.77), which remained in favor of IAT (P < 0.0001) (Figure 3).
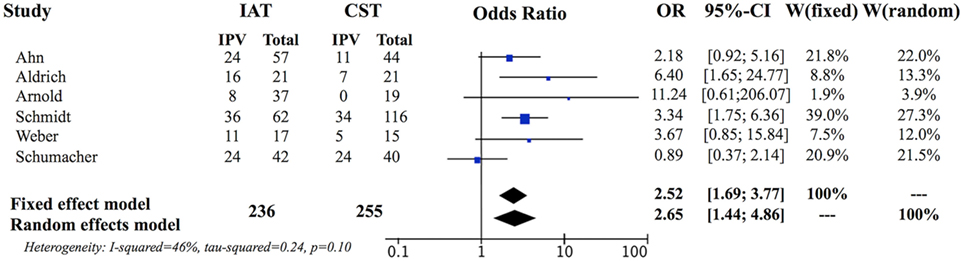
Figure 3. Forest plot evaluating five retrospective controlled studies and a randomized controlled trial. The addition of the randomized trial, which individually does not support IAT therapy, decreased the pooled odds ratio but it was still significantly in favor of intra-arterial therapy over standard therapy controls. There was an increase in heterogeneity, but this remained non-significant (P = 0.10) supporting the use of this method.
A mixed effects model comparing the treatment results of the EAGLE study compared to the retrospective studies showed a fixed effect difference for the estimated log OR that was −1.25 below the estimated log OR of the other five studies. This result did not represent a significant difference between the retrospective studies and the EAGLE study 95% CI (−2.84, 0.34), P = 0.087. Estimated variance of the random effect of study was <0.01 when the fixed effect was included. When the fixed effect was not included, the estimated variance of the random effect was 0.014.
Formal testing of funnel plot asymmetry was non-significant (P = 0.47) supporting a lack of publication bias of study outcomes with respect to study size (Figure 4).
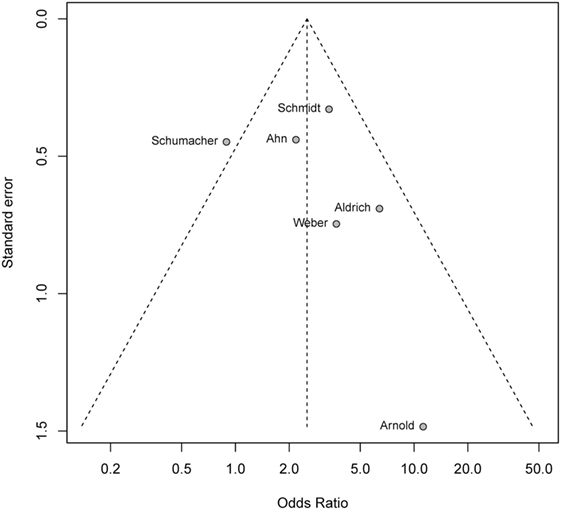
Figure 4. Funnel plot testing for publication bias. Formal test of funnel plot asymmetry with P = 0.47 showign a lack of publication bias.
Of the 236 patients examined, major complications included 4 groin hematomas, 2 intracranial hemorrhages, 5 transient ischemic events, 9 ischemic strokes (as documented on MRI imaging), and 1 hemianopia. Both intracranial hemorrhages were not associated with long-term disabilities with an mRS of 0 at the 1-month follow-up (16). Only two reported ischemic strokes were clinically significant and both resolved without permanent disability with an mRS of 0 at 2 weeks (15, 20). No studies reported long-term neurological deficit. Minor complications were more commonly reported including headache, tinnitus, and hyperesthesia.
Discussion
While the use of IAT for CRAO is highly debated, studies directly comparing outcomes between ST and IAT generally favored the use of IAT. All five of the retrospective studies reported better visual outcomes for the IAT group compared to ST, yet, only 2 of those individual studies reached statistical significance in the chosen outcome measure where the lower end of the 95% confidence interval of the OR was greater than one (18, 22). Nevertheless, the OR for the retrospective studies significantly favored IAT therapy when the data was pooled for meta-analysis (Figure 2). Furthermore, the OR remained significantly favorable for IAT therapy, despite the addition of the EAGLE study, which does not support IAT therapy independently (16).
It is important to consider the varying primary outcomes and follow-up duration for studies included in the analysis. All studies reported the primary outcome for all studies [except for one which was not explicitly defined (24)] to be a change in best-corrected visual acuity (BCVA). In two studies, the primary outcome was determined at 1 month and at final follow-up, a clinically significant improvement being defined as logMAR ≥ 0.3 (18, 19). A single study defined clinically significant improvement as logMAR ≥ 0.6 (22) within 24 h after treatment, while two studies considered a positive primary outcome if there was one VA category improvement after intervention (21, 25).
Our meta-analysis supports the hypothesis that IAT significantly increases the likelihood of experiencing an improvement in VA compared with ST alone. Included studies showed dramatic variations in the efficacy of both IAT (23.5–80% in VA improvement) and conservative therapies alone (1–3, 29, 30).
Assessment and Treatment of Bias within Included Studies
Qualitative analysis serves to address potential individual study design flaws and the potential for biases, specifically selection bias, within the included studies. The single most important driver of the positive results was the study by Schmidt et al. with the largest sample size and a significant treatment effect favoring IAT (22). It also had a favorable assessment of bias with excellent matching of time from symptom onset to time of treatment in both groups and likewise excellent matching of the stage of CRAO occlusion between groups.
Control patients were not always treated concurrently in the various studies. Determination of treatment with ST in some patients depended on the fact that the medical center was not yet performing IAT for CRAO at that time point. A priori application of inclusion and exclusion criteria was not consistently applied to any of the retrospective studies except for Ahn et al. introducing additional risk for selection bias (15). There were no differences in baseline characteristics except in the time from symptom onset to initiation of treatment parameter, which tended to be longer in control groups of retrospective studies potentially favoring IAT.
The variation in therapeutic regimens including type of treatment, dose, duration, and timing varied between patients within each study and between the studies in both IAT and ST groups introducing the potential for treatment bias. All the retrospective studies included in the analysis had considerable variability in both the ST and IAT treatment regimens.
The choice of outcome measure to assess the effectiveness of CRAO therapy varied considerably between studies. Some studies used descriptive terminology for assessments of BCVA improvement, while others used a quantitative analysis involving LogMAR scores derived from BCVA improvement. Likewise, there was significant variation in the definition of a “good outcome” for treatment, ranging from any visual improvement to significant quantified visual score improvement. The timing of follow-up was also inconsistent as some studies evaluated immediate outcomes, while others focused on delayed outcome measures ranging from 1 month to 1 year or more.
Inclusion of Randomized and Non-Randomized Studies in the Pooled Analysis
Different approaches were considered for sensitivity analysis to account for differences in study quality. One strategy involves assigning higher weight to studies with a level of evidence. Most weighting methods are considered arbitrary and study quality should not modify the precision of the pooled estimates (35). An alternative approach focuses the sensitivity analysis on the components of study quality considered most important to the meta-analysis. We performed a mixed effects approach utilizing a fixed covariate to identify the overall “component of study quality,” which we considered important (35, 36). Our study utilized an indicator variable and separated the individual studies into two categories: those that are prospective randomized trials and those that are not. A significant result of this fixed effect covariate would indicate that the EAGLE study has a significant difference in treatment effect compared to the other studies. We used a mixed effects model analysis, which resulted in −1.25 on the log OR scale for the EAGLE study lower than the non-randomized studies. The difference was not statistically significant and, therefore, our analysis of the impact of study quality shows no significant difference in overall treatment effect between studies of different quality. This supports the decision to pool all controlled studies meeting inclusion criteria regardless of level of evidence.
Even though the EAGLE study was a randomized controlled trial, significant concerns have been raised from various experts regarding its study design and its inclusion and exclusion criteria (37–39). We used the inverse variance method was used to calculate OR and P-value and obtained similar results using the Mantel–Haenszel method and the random effects model. The test of heterogeneity was not statistically significant (P = 0.1) and supported the models used.
Clinical Relevance
Despite the positive outcomes of this meta-analysis, we do not recommend universal acceptance of IAT for CRAO. The inconsistent results that we observed based on the lack of efficacy for IAT seen in the only randomized trial and the divergence of these results from prior retrospective studies raises important questions.
Study design plays an important role in evaluating an intervention and significantly affects the likelihood of observing the desired effect. Given the possible flaws seen in the EAGLE trial, efforts should be directed at identifying study design choices of the EAGLE study that may have prevented seeing a positive treatment effect. The inclusion criteria of the EAGLE trial were broad and resulted in a heterogeneous study sample with regard to initial severity of CRAO. CRAO is classified into stages, which include incomplete, subtotal, and total subtypes in the order of increasing severity. Evaluation of the degree of arterial occlusion is based on fundoscopic findings or angiography and has been shown to correlate with visual outcomes after treatment with thrombolytics. Clinically significant improvement in VA was most likely to be seen in cases of incomplete CRAO (50–76.9%) as opposed to subtotal (10.6–14.3%) and total occlusion (0%) (15). A potentially effective strategy for future randomized trials may be to enroll CRAO patients who are in the incomplete stage, the subtotal stage or a combination of both stages. This strategy would exclude the total stage where an IAT treatment effect is not likely to be seen. This would serve to increase average treatment effect, decrease variance, and thereby decrease the sample size needed for future studies.
Time from symptom onset to fibrinolysis is another significant factor in the treatment of CRAO. Current results have inconsistently demonstrated an association between time to treatment and functional outcomes (39–41). While improvements can be observed in patients treated with IAT well after 6 h, we speculate the effectiveness of the intervention to lessen as time to treatment lengthens, in a similar fashion to endovascular stroke therapy (37, 38, 42). Based on prior evidence, multiple time points for intervention should be considered (41, 43).
Fear of serious complications associated with IAT is frequently cited, yet, our review did not demonstrate a heightened risk for long-term neurological complications.
Limitations
While our meta-analysis was able to include a large number of patients, significant limitations are present. One limitation is the inherent treatment selection bias found in non-randomized studies, which can lead to confounders being over-represented in a given cohort. The decision on whether or not to perform IAT was sometimes related to time from symptom onset. Increased time from visual change to therapy in only one group would be expected to produce worse outcomes thus skewing results. An additional limitation is that our study did not evaluate the degree of visual improvement. In future trials, stratification of visual improvement would help quantify the effectiveness of the intervention and would help better confirm the optimal timing of the intervention (44). Another limitation was the inclusion of studies of varying levels of quality of evidence. While it is reasonable to include studies of different quality in a meta-analysis, it is not ideal. An additional important limitation is our own bias approaching this topic. Additional non-biased studies are needed to objectively assess the merit of this intervention.
Conclusion
The EAGLE study is the only randomized trial and, considered in isolation, does not support IAT therapy. However, lessons from the acute stroke trials reinforce that it would be inappropriate to dismiss a promising treatment for CRAO based on a single randomized trial. This meta-analysis evaluating all controlled studies reporting IAT therapy for CRAO demonstrated a pooled OR significantly favoring IAT treatment. Our results support that further investigation with additional well-designed randomized controlled trials is necessary prior to establishing universal recommendations about IAT.
Author Contributions
Conception, approved the final version on behalf of all authors, and study supervision: RJ. Design: RJ, PP, AC, and SR. Acquisition of data: PP, NK, AW, and AC. Analysis and interpretation of data: all authors. Drafting the article: RJ, PP, NK, AW, AC, and GB. Reviewed submitted version: all authors.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
1. Hayreh SS, Zimmerman MB. Central retinal artery occlusion: visual outcome. Am J Ophthalmol (2005) 140(3):376–91. doi:10.1016/j.ajo.2005.03.038
2. Atebara NH, Brown GC, Cater J. Efficacy of anterior chamber paracentesis and Carbogen in treating acute nonarteritic central retinal artery occlusion. Ophthalmology (1995) 102(12):2029–34; discussion 34–5. doi:10.1016/S0161-6420(95)30758-0
3. Mueller AJ, Neubauer AS, Schaller U, Kampik A, European Assessment Group for Lysis in the Eye. Evaluation of minimally invasive therapies and rationale for a prospective randomized trial to evaluate selective intra-arterial lysis for clinically complete central retinal artery occlusion. Arch Ophthalmol (2003) 121(10):1377–81. doi:10.1001/archopht.121.10.1377
4. Rudkin AK, Lee AW, Aldrich E, Miller NR, Chen CS. Clinical characteristics and outcome of current standard management of central retinal artery occlusion. Clin Exp Ophthalmol (2010) 38(5):496–501. doi:10.1111/j.1442-9071.2010.02280.x
5. Fiess A, Cal O, Kehrein S, Halstenberg S, Frisch I, Steinhorst UH. Anterior chamber paracentesis after central retinal artery occlusion: a tenable therapy? BMC Ophthalmol (2014) 14:28. doi:10.1186/1471-2415-14-28
6. Waters RE II, Mahaffey KW, Granger CB, Roe MT. Current perspectives on reperfusion therapy for acute ST-segment elevation myocardial infarction: integrating pharmacologic and mechanical reperfusion strategies. Am Heart J (2003) 146(6):958–68. doi:10.1016/S0002-8703(03)00439-3
7. The National Institute of Neurological Disorders and Stroke rt-PA Stroke Study Group. Tissue plasminogen activator for acute ischemic stroke. N Engl J Med (1995) 333(24):1581–7. doi:10.1056/NEJM199512143332401
8. Avgerinos ED, Chaer RA. Catheter-directed interventions for acute pulmonary embolism. J Vasc Surg (2015) 61(2):559–65. doi:10.1016/j.jvs.2014.10.036
9. Saver JL, Goyal M, van der Lugt A, Menon BK, Majoie CB, Dippel DW, et al. Time to treatment with endovascular thrombectomy and outcomes from ischemic stroke: a meta-analysis. JAMA (2016) 316(12):1279–89. doi:10.1001/jama.2016.13647
10. Beatty S, Au Eong KG. Local intra-arterial fibrinolysis for acute occlusion of the central retinal artery: a meta-analysis of the published data. Br J Ophthalmol (2000) 84(8):914–6. doi:10.1136/bjo.84.8.914
11. Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gotzsche PC, Ioannidis JP, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. Ann Intern Med (2009) 151(4):W65–94. doi:10.7326/0003-4819-151-4-200908180-00136
12. Sheu C, Suzuki S. Meta-analysis using linear mixed models. Behav Res Methods Instrum Comput (2001) 33(2):102–7. doi:10.3758/BF03195354
13. Argresti A. An Introduction to Categorical Data Analysis. New York: John Wiley & Sons, Inc (1996).
14. Mercier J, Kastler A, Jean B, Souteyrand G, Chabert E, Claise B, et al. Interest of local intra-arterial fibrinolysis in acute central retinal artery occlusion: clinical experience in 16 patients. J Neuroradiol (2015) 42(4):229–35. doi:10.1016/j.neurad.2014.02.007
15. Ahn SJ, Kim JM, Hong JH, Woo SJ, Ahn J, Park KH, et al. Efficacy and safety of intra-arterial thrombolysis in central retinal artery occlusion. Invest Ophthalmol Vis Sci (2013) 54(12):7746–55. doi:10.1167/iovs.13-12952
16. Schumacher M, Schmidt D, Jurklies B, Gall C, Wanke I, Schmoor C, et al. Central retinal artery occlusion: local intra-arterial fibrinolysis versus conservative treatment, a multicenter randomized trial. Ophthalmology (2010) 117(7):1367–75.e1. doi:10.1016/j.ophtha.2010.03.061
17. Zhang X, Ji X, Luo Y, Liu D, Guo L, Wu H, et al. Intra-arterial thrombolysis for acute central retinal artery occlusion. Neurol Res (2009) 31(4):385–9. doi:10.1179/174313209X444008
18. Aldrich EM, Lee AW, Chen CS, Gottesman RF, Bahouth MN, Gailloud P, et al. Local intraarterial fibrinolysis administered in aliquots for the treatment of central retinal artery occlusion: the Johns Hopkins Hospital experience. Stroke (2008) 39(6):1746–50. doi:10.1161/STROKEAHA.107.505404
19. Pettersen JA, Hill MD, Demchuk AM, Morrish W, Hudon ME, Hu W, et al. Intra-arterial thrombolysis for retinal artery occlusion: the Calgary experience. Can J Neurol Sci (2005) 32(4):507–11. doi:10.1017/S0317167100004522
20. Arnold M, Koerner U, Remonda L, Nedeltchev K, Mattle HP, Schroth G, et al. Comparison of intra-arterial thrombolysis with conventional treatment in patients with acute central retinal artery occlusion. J Neurol Neurosurg Psychiatry (2005) 76(2):196–9. doi:10.1136/jnnp.2004.037135
21. Butz B, Strotzer M, Manke C, Roider J, Link J, Lenhart M. Selective intraarterial fibrinolysis of acute central retinal artery occlusion. Acta Radiol (2003) 44(6):680–4. doi:10.1080/02841850312331287829
22. Schmidt DP, Schulte-Monting J, Schumacher M. Prognosis of central retinal artery occlusion: local intraarterial fibrinolysis versus conservative treatment. AJNR Am J Neuroradiol (2002) 23(8):1301–7.
23. Richard G, Lerche RC, Knospe V, Zeumer H. Treatment of retinal arterial occlusion with local fibrinolysis using recombinant tissue plasminogen activator. Ophthalmology (1999) 106(4):768–73. doi:10.1016/S0161-6420(99)90165-3
24. Weber J, Remonda L, Mattle HP, Koerner U, Baumgartner RW, Sturzenegger M, et al. Selective intra-arterial fibrinolysis of acute central retinal artery occlusion. Stroke (1998) 29(10):2076–9. doi:10.1161/01.STR.29.10.2076
25. Schumacher M, Schmidt D. Local fibrinolysis in central retinal artery occlusion: follow-up in 36 cases. Proceedings of the XVth Symposium of Neuroradiology. Kumamoto (1995). p. 458–60.
26. Schmidt D, Schumacher M, Wakhloo AK. Microcatheter urokinase infusion in central retinal artery occlusion. Am J Ophthalmol (1992) 113(4):429–34. doi:10.1016/S0002-9394(14)76167-7
27. Schumacher M, Schmidt D, Wakhloo AK. Intra-arterial fibrinolytic therapy in central retinal artery occlusion. Neuroradiology (1993) 35(8):600–5. doi:10.1007/BF00588405
28. Schumacher M, Schmidt D, Wakhloo AK. [Intra-arterial fibrinolysis in central artery occlusion]. Radiologe (1991) 31(5):240–3.
29. Fernandez FJ, Guelbenzu S, Barrena C, Larrosa JM, Gonzalvo FJ, Melcon B, et al. [Selective ophthalmic artery fibrinolysis in acute central retinal artery occlusion]. Arch Soc Esp Oftalmol (2002) 77(2):81–6.
30. Framme C, Spiegel D, Roider J, Sachs HG, Lohmann CP, Butz B, et al. [Central retinal artery occlusion. Importance of selective intra-arterial fibrinolysis]. Ophthalmologe (2001) 98(8):725–30. doi:10.1007/s003470170079
31. Weill A, Cognard C, Piotin M, Laloum L, Castaings L, Moret J. [Persistent value of intra-arterial fibrinolysis 8 hours or more following central retinal artery occlusion or of its branches]. J Fr Ophtalmol (1998) 21(7):466–70.
32. Vulpius K, Hoh H, Lange H, Maercker W, Ruhle H. [Selective percutaneous transluminal thrombolytic therapy with rt-PA in central retinal artery occlusion]. Ophthalmologe (1996) 93(2):149–53.
33. Annonier P, Sahel J, Wenger JJ, Rigolot JC, Foessel M, Bronner A. [Local fibrinolytic treatment in occlusions of the central retinal artery]. J Fr Ophtalmol (1984) 7(11):711–6.
34. Annonier P, Benichou C, Flament J, Bronner A. [Role of fibrinolysis in the treatment of retinal arterial occlusion. Discussion of 5 cases]. Bull Soc Ophtalmol Fr (1988) 88(10):1167–71.
35. Juni P, Altman DG, Egger M. Systematic reviews in health care: assessing the quality of controlled clinical trials. BMJ (2001) 323(7303):42–6. doi:10.1136/bmj.323.7303.42
36. Stram DO. Meta-analysis of published data using a linear mixed-effects model. Biometrics (1996) 52(2):536–44. doi:10.2307/2532893
37. Hayreh SS. Comment re: multicenter study of the European Assessment Group for Lysis in the Eye (EAGLE) for the treatment of central retinal artery occlusion: design issues and implications. Graefes Arch Clin Exp Ophthalmol (2007) 245(3):464–6; author reply 7–70. doi:10.1007/s00417-006-0473-5
38. Agarwal N, Gala NB, Karimi RJ, Turbin RE, Gandhi CD, Prestigiacomo CJ. Current endovascular treatment options for central retinal arterial occlusion: a review. Neurosurg Focus (2014) 36(1):E7. doi:10.3171/2013.11.FOCUS13331
39. Arthur A, Aaron S. Thrombolysis for artery occlusion. Ophthalmology (2011) 118(3):604; author reply 604–5. doi:10.1016/j.ophtha.2010.10.042
40. Biousse V. Thrombolysis for acute central retinal artery occlusion: is it time? Am J Ophthalmol (2008) 146(5):631–4. doi:10.1016/j.ajo.2008.07.025
41. Page PS, Cambon AC, James RF. Visual improvement after intra-arterial thrombolysis for central retinal artery occlusion does not correlate with time to treatment. Interv Neurol (2016) 5(3–4):131–9. doi:10.1159/000446853
42. Fransen PS, Beumer D, Berkhemer OA, van den Berg LA, Lingsma H, van der Lugt A, et al. MR CLEAN, a multicenter randomized clinical trial of endovascular treatment for acute ischemic stroke in the Netherlands: study protocol for a randomized controlled trial. Trials (2014) 15:343. doi:10.1186/1745-6215-15-343
43. Nogueira RG, Jadhav AP, Haussen DC, Bonafe A, Budzik RF, Bhuva P, et al. Thrombectomy 6 to 24 hours after stroke with a mismatch between deficit and infarct. N Engl J Med (2018) 378(1):11–21. doi:10.1056/NEJMoa1706442
Keywords: retinal artery occlusion, thrombolytic therapy, intra-arterial infusions, revascularization, meta analysis
Citation: Page PS, Khattar NK, White AC, Cambon AC, Brock GN, Rai SN and James RF (2018) Intra-Arterial Thrombolysis for Acute Central Retinal Artery Occlusion: A Systematic Review and Meta-Analysis. Front. Neurol. 9:76. doi: 10.3389/fneur.2018.00076
Received: 09 November 2017; Accepted: 01 February 2018;
Published: 21 February 2018
Edited by:
Owen B. White, Monash University, AustraliaReviewed by:
Bruce Campbell, University of Melbourne, AustraliaBernard Yan, University of Melbourne, Australia
Celia S. Chen, Flinders Medical Centre, Australia
Copyright: © 2018 Page, Khattar, White, Cambon, Brock, Rai and James. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Robert F. James, cm9iZXJ0LmphbWVzQGxvdWlzdmlsbGUuZWR1
 Paul S. Page
Paul S. Page Nicolas K. Khattar
Nicolas K. Khattar Andrew C. White2
Andrew C. White2 Robert F. James
Robert F. James