- 1Department of Neurology, Epilepsy Center Frankfurt Rhine-Main, Goethe University Frankfurt, Frankfurt, Germany
- 2Department of Neurology, Epilepsy Center Hessen, Philipps University of Marburg, Marburg, Germany
Introduction: This study was designed to evaluate risk factors and incidence of epilepsy-related injuries and accidents (ERIA) at an outpatient clinic of a German epilepsy center providing healthcare to a mixed urban and rural population of over one million inhabitants.
Methods: Data acquisition was performed between 10/2013 and 09/2014 using a validated patient questionnaire on socioeconomic status, course of epilepsy, quality of life (QoL), depression, injuries and accidents associated with seizures or inadequate periictal patterns of behavior concerning a period of 3 months. Univariate analysis, multiple testing and regression analysis were performed to identify possible variables associated with ERIA.
Results: A total of 292 patients (mean age 40.8 years, range 18–86; 55% female) were enrolled and analyzed. Focal epilepsy was diagnosed in 75% of the patients. The majority was on an antiepileptic drug (AEDs) polytherapy (mean number of AEDs: 1.65). Overall, 41 patients (14.0%) suffered from epilepsy-related injuries and accidents in a 3-month period. Besides lacerations (n = 18, 6.2%), abrasions and bruises (n = 9, 3.1%), fractures (n = 6, 2.2%) and burns (n = 3, 1.0%), 17 mild injuries (5.8%) were reported. In 20 (6.8% of the total cohort) cases, urgent medical treatment with hospitalization was necessary. Epilepsy-related injuries and accidents were related to active epilepsy, occurrence of generalized tonic-clonic seizures (GTCS) and drug-refractory course as well as reported ictal falls, ictal loss of consciousness and abnormal peri-ictal behavior in the medical history. In addition, patients with ERIA had significantly higher depression rates and lower QoL.
Conclusion: ERIA and their consequences should be given more attention and standardized assessment for ERIA should be performed in every outpatient visit.
Introduction
Epilepsy is a common and chronic neurological disorder that affects about 39 million people worldwide (1, 2). People with epilepsy are subject to social and vocational stigma, have only restricted access to the labor market and significantly reduced employment opportunities (3). Moreover, quality of life (QoL) is significantly reduced for themselves and their caregivers (4–9). Active epilepsy with persisting seizures is associated with loss of consciousness, uncontrolled movements, falls or periictal abnormal behavior may predispose to accidents and injuries such as burns, contusions, lacerations or fractures (10–12).
A prospective longitudinal analysis from Finland on 245 children with epilepsy since 1964 showed a significantly increased age- and sex-adjusted mortality. During the 40-year follow up, 60 (24%) subjects died and 33 (55%) of these events were attributed to the underlying epilepsy. Besides sudden unexpected death in epilepsy (SUDEP) and status epilepticus, epilepsy-related injuries and accidents (ERIA), such as peri-ictal drowning, were common causes of death. Within this cohort, pneumonia, cardiovascular diseases and suicide have been reported as most frequent causes of death not related to seizures or epilepsy (13–15). ERIA were shown to be a major cost factor for hospitalizations among patients with epilepsy (16). One major problem with ERIA is unreported cases in daily practice. If not investigated in detail, it can be assumed that approximately 50% of ERIA are falsely not documented as seizure-related in medical documentation (17).
The main aim of our study was to assess the frequency and types of ERIA in a cohort of consecutive patients with epilepsy using a validated questionnaire (18) and to search for variables associated with ERIA. Improved screening parameters for ERIA may help in reducing the frequency and severity of ERIA. A second aim was to assess QoL and depression as possible consequences of ERIA.
Patients and Methods
Study Settings, Patients and Design
This study was performed as a survey at the epilepsy outpatient clinic of the University Hospital Marburg. The University Hospital Marburg is a large multispecialty tertiary care hospital in the center of Germany that provides healthcare to a population of over 1,000,000 inhabitants. Marburg is located within the postal code area 35, which was used previously for a population-based estimation of the incidence of status epilepticus and different cost-of-illness studies (3, 19–21). After receiving written informed consent, all adult epilepsy patients aged 18 years or older were eligible. Socioeconomic data and information on injuries or accidents as well as on QoL were accessed between 10/2013 and 09/2014 using a validated questionnaire (18). Patients were asked to complete the questionnaire concerning their individual epilepsy related experiences during the last 3 months. The diagnosis was based on the definitions proposed by the International League Against Epilepsy and the International Bureau for Epilepsy (22) and was provided by the treating physician. Any patient with at least one seizure during the last 12 months at enrolment was classified as having an “active epilepsy.” Patients were excluded when the diagnosis of epilepsy could not be determined without doubt. The study had the approval of the local ethics committee.
Variables Associated With Epilepsy-Related Injuries and Accidents
Specification of epilepsy syndromes as well as information regarding intake of AEDs was assessed by the attending physician. The following factors were identified as possible parameters influencing injuries or accidents in patients with epilepsy according to published literature (18): patient age, duration of epilepsy, sex, epilepsy syndrome, postictal patterns of behavior as well as seizure frequency and semiology (loss of consciousness, nocturnal seizures). Abnormal postictal behavior patterns were classified in line with the guidelines of the German employers' liability insurance association (Unfallversicherung, DGUV 250-001), which classifies seizures with abnormal postictal behavior as “high risk” for related injuries or accidents at work (23). Refractory epilepsy was classified according to ILAE definitions (24). Moreover, different sociodemographic and clinical characteristics were documented.
Changes in Mood and Quality of Life Due to ERIA
To evaluate the psychological aspects of ERIA, various established neuropsychological inventories were assessed using already established questionnaires: To measure health related QoL, the QOLIE31 score (Quality of Life in Epilepsy Inventory) (25) and EQ5D score (Euro Quality of Life) were used (26). For EQ5D, both relevant parameters, i.e., overall TTO (time trade-off) and VAS (visual analog scale), were determined (26). To evaluate health-related depression, NDDI-E (Neurological Disorders Depression Inventory for Epilepsy) (27) and BDI-II (Becks Depression Inventory) were used (28). As an independent parameter, ABNAS (A-B Neuropsychological Assessment Schedule) was assessed (29). Liverpool Adverse Events Profile (LAEP) was measured to detect AED-specific aspects in QoL (30, 31).
Data Entry and Statistical Analysis
Data entry and analysis were performed using the File Maker Pro 8.5 database (Filemaker Inc., Santa Clara, CA, USA). A double-entry procedure was employed to assure a high level of data accuracy. Statistical analyses were performed using IBM SPSS Statistics 22 (IBM Corporation, Armonk, NY, USA). For the calculation of variables that potentially influence injuries and accidents in patients with active epilepsy, a chi-square test following Pearson and Benjamini-Hochberg-Adjustment was used to exclude multiple testing bias (32, 33). In addition, regression analysis was performed. P-values < 0.05 were considered as significant.
Results
Sociodemographic and Clinical Characteristics
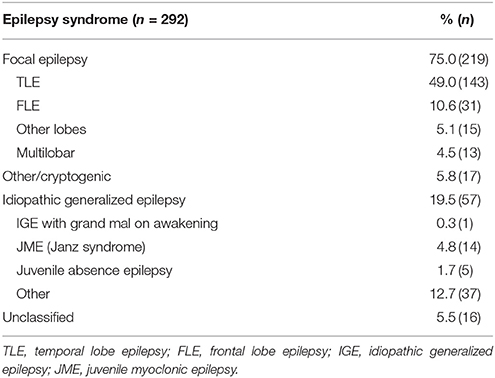
We enrolled 292 patients with epilepsy and a mean age of 40.8 years (SD ± 15.6; range 18–86 years). Gender ratio was nearly balanced with 55% female patients. Two-thirds of patients (67.8%) had an active epilepsy with at least one seizure within the last 12 months. The majority of 75% (n = 219) suffered from focal epilepsy (FE), 19.5% (n = 57) from an idiopathic generalized epilepsy (IGE) and, in 5.5%, the epilepsy syndrome was reported as unclassified; details are presented in Table 1. Between the two cohorts with IGE or FE, there were no significant differences in gender distribution, number of AED or seizure freedom. The mean age in the IGE cohort was significantly younger. Detailed information of sociodemographic and clinical characteristics for all patients and the FE and IGE cohorts are displayed in Table 2.
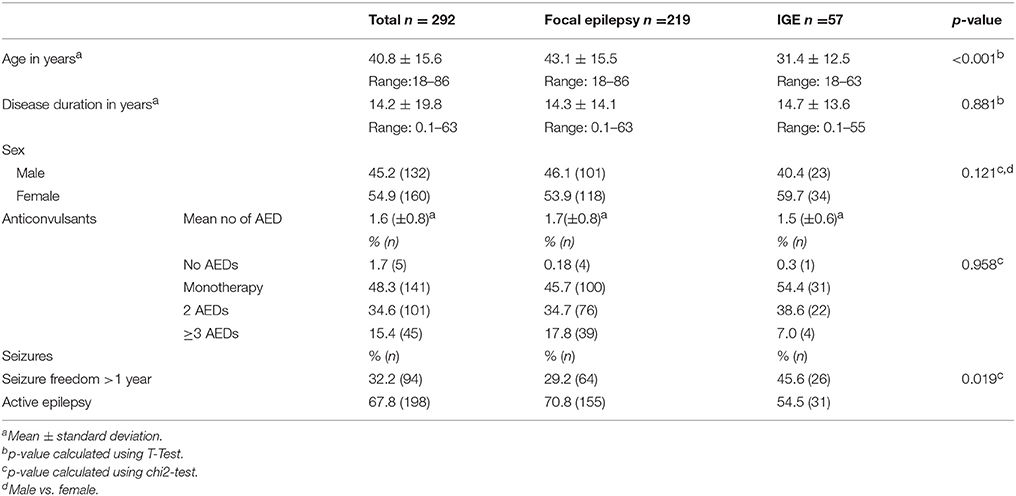
Table 2. Sociodemographic and clinical characteristics of the entire cohort and for patients with focal and idiopathic generalized epilepsies.
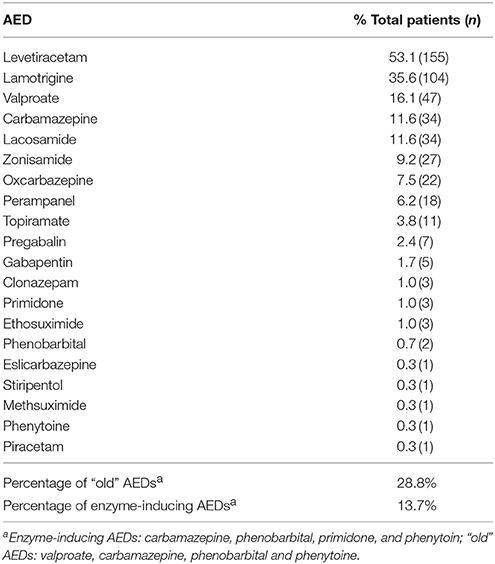
The five most frequently prescribed AEDs were levetiracetam (LEV, 53.1%), lamotrigine (LTG, 35.6%) and valproate (VPA, 16.1%) followed by carbamazepine (CBZ) and lacosamide (LCM) (each 11.6%). A wide variability of newer AEDs (second and third generation) were used, while 28.8% of prescribed AEDs were so called “old” substances like VPA, CBZ, phenobarbital (PB) and phenytoine (PHT). Only 13.7% of the AEDs had enzyme-inducing properties (i.e., CBZ, PB, primidone, PHT). For a more detailed listing, please refer to Table 3.
Clinical Characteristics of Patients With Epilepsy-Related Injuries and Accidents
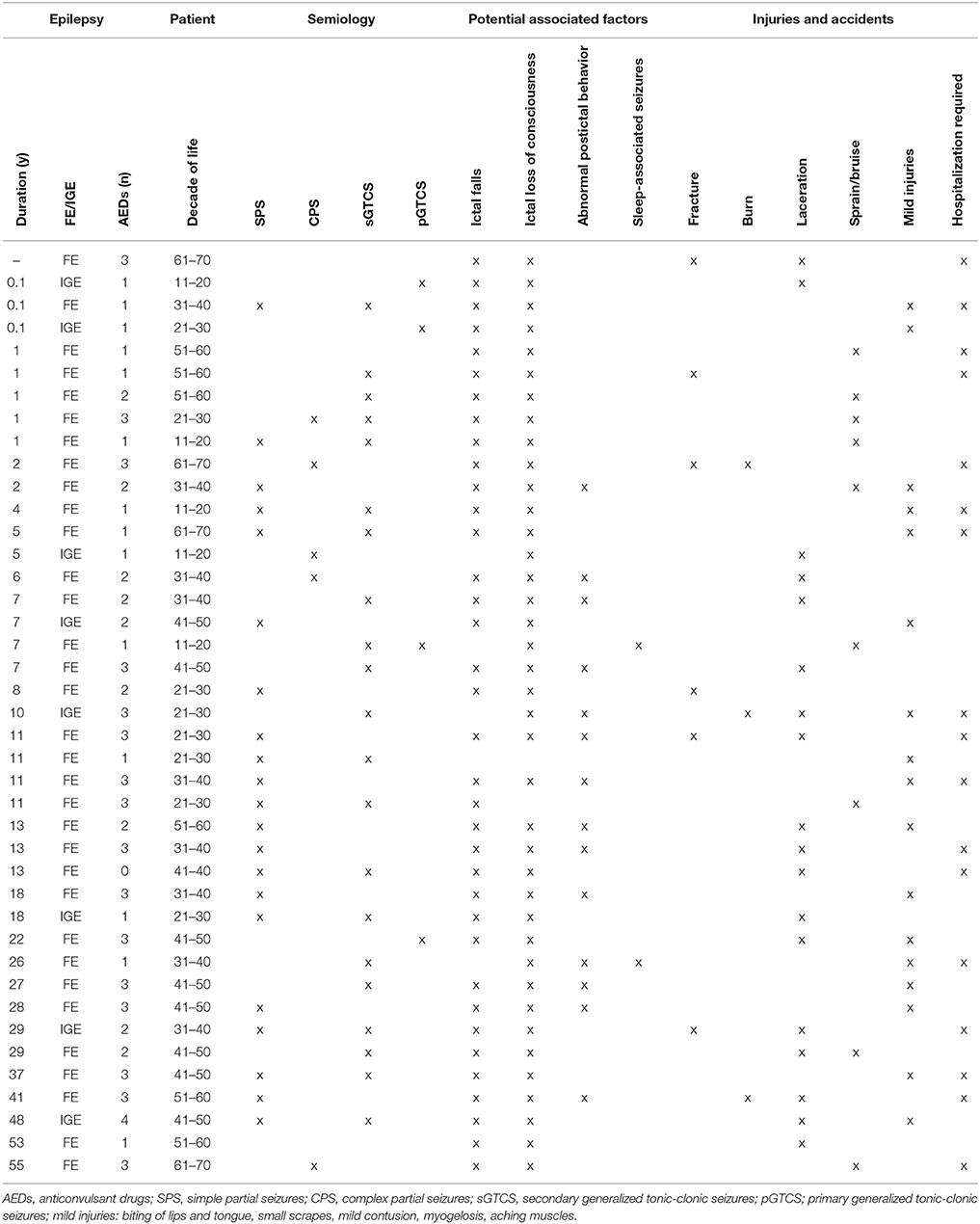
In total, 41 patients (14.0%) suffered from epilepsy-related injuries and accidents (ERIA) within the 3-month observation period. Of these, 20 patients (6.8% of total cohort) required hospitalization. Out of a total of 292 patients, two independent injuries were reported in two cases and one patient suffered from three independent injuries. Besides lacerations (n = 18, 6.2%), abrasions and bruises (n = 9, 3.2%), fractures (n = 6, 2.1%) and burns (n = 3, 1.0%), 17 other mild injuries (5.8%) were reported. Among the patients with injuries, 48.8% were male, the mean age was 39.2 years (SD ± 14.1, range 18–67 years). Mean disease duration in patients with ERIA was 14.7 years (SD ± 15.2, range 0.1–40 years). Only one patient in this group reported no AED therapy, 14 participants (14/41; 34.2%) were under anticonvulsant monotherapy, 26 reported taking two or more AEDs (63.4%), and the mean number of AEDs was 2.0 (SD 1.0, range 0–4). The majority of seizure-related injuries was observed in patients suffering from generalized tonic-clonic seizures (n = 24, 58.5%, i.e., bilateral tonic-clonic seizures). Another 14 patients (34.2%) reported predominantly simple partial seizures (SPS, i.e., focal seizures with preserved awareness) or complex partial seizures (CPS, i.e., focal onset seizures with impaired awareness); in three cases (7.3%), the habitual seizure type was not stated. In nearly all cases (40 of 41 patients, 97.6%) suffering from ERIA, certain semiological features were reported. Most of the injured patients reported ictal loss of consciousness (n = 39, 95.1%) or ictal falls (n = 36, 87.8%) or both (n = 35, 85.4%). Isolated peri-ictal abnormal behavior (n = 14, 34.2%) was less often seen in patients with ERIA, but several patients reported periictal abnormal behavior in association with ictal falls and ictal loss of consciousness (n = 12, 29.3%). Only two patients with ERIA reported to suffer from sleep-bound seizures only (4.9%). For a detailed list of all patients that reported ERIA see Table 4.
Variables Associated With Epilepsy-Related Injuries and Accidents
Reported sociodemographic and clinical characteristics were analyzed as potential variables associated with ERIA. Comparing patients with (n = 41, 14.0%) to patients without (n = 251, 86.0%) ERIA, we identified an active epilepsy (p < 0.001), regular GTCS (p < 0.001) and a refractory course of disease (p = 0.005) as variables associated with ERIA using a univariate analysis. Moreover, ictal falls (p = 0.012), ictal loss of consciousness (p = 0.022) and inadequate postictal behavior (p = 0.017) were significantly increased in patients suffering ERIA. After post hoc multivariate analysis all reported parameters remained significant. No difference was determined for patient age, duration of epilepsy, gender, epilepsy syndrome or sleep-related seizures in our cohort; for details please refer to Table 5. Multiple regression analysis revealed a strong positive correlation of ERIA with active epilepsy (p = 0.006, B = 0.140), all other tested characteristics (i.e., age, sex, disease duration, seizure semiology, epilepsy syndrome, refractive epilepsy) did not reach levels of significance.
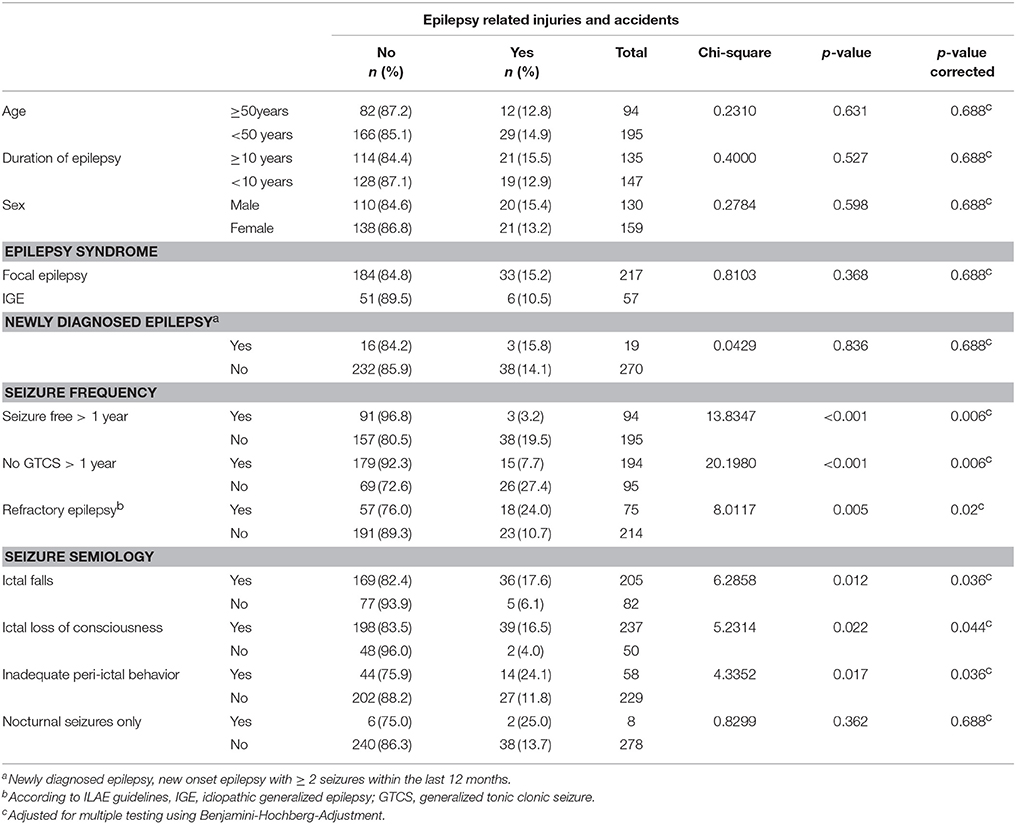
Table 5. Frequency and clinical characteristics of patients with epilepsy-related injuries and accidents (ntotal = 292, nERIA = 41).
Epilepsy-Related Injuries and Accidents are Accompanied by a Decreased Quality of Life (QoL)
People suffering from ERIA reported significantly more symptoms indicative of a manifest depression (BDI ≥ 14 [p = 0.021], NDDIE ≥ 13 [p = 0.002]). Patients with ERIA (overall T 38.9 ± 9.0) had a significant lower (QoL) compared to patients without ERIA (overall T 49.2 ± 10.4 [p < 0.001]) in QOLIE-31. Regarding each single item of the QOLIE-31 inventory, patients with ERIA showed a significantly increased seizure worry (p ≤ 0.001) and decreased overall QoL (p < 0.001), emotional well-being (p = 0.001) and energy level (p = 0.019) as well as cognitive (p < 0.001) and social functions (p < 0.001). Medication effects did not reach any level of significance in QOLIE-31 (p = 0.68), even if the results for Liverpool Adverse Events Profile (LEAP) were significantly different between patients with and without ERIA (p = 0.006). Similar results for QoL were obtained in EQ5D Score [VAS (visual analog scale) 56.0 ±19.1 vs. 68.4 ±18.7, p < 0.001]. In addition, ABNAS showed a significant distinction between patients with (22.2 ± 15.7) and without ERIA (35.8 ± 17.9, p < 0.001). After post hoc multivariate analysis all reported parameters remained significant. For a detailed statistical analysis of QoL and depression scores please refer to Table 6.
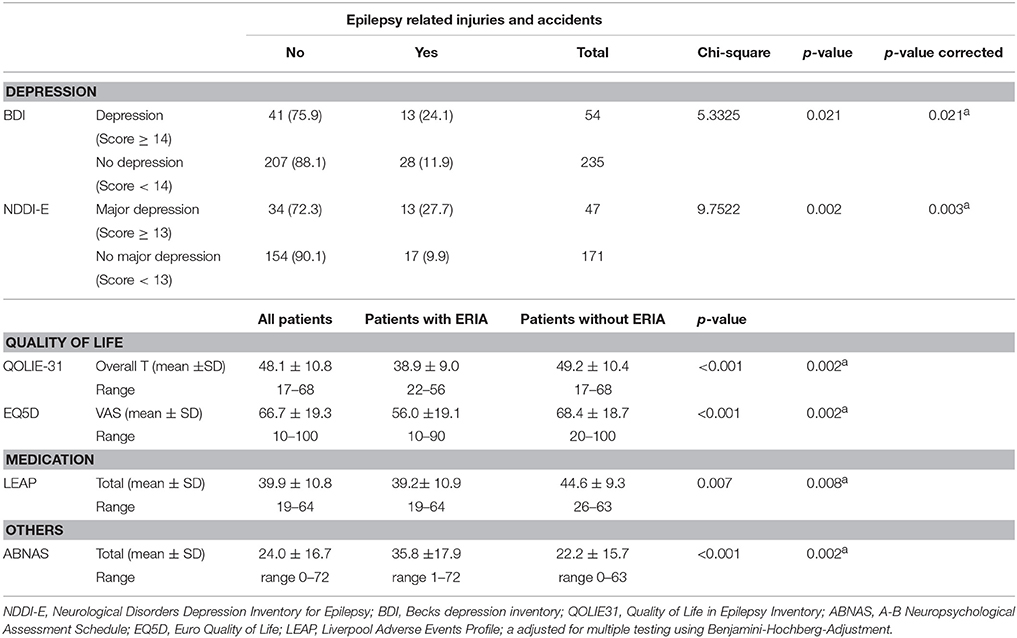
Table 6. Changes in mood and quality of life due to epilepsy-related injuries and accidents (ntotal = 292, nERIA = 41).
Discussion
This study analyzed in detail a cohort of epilepsy patients in Germany showing the high burden imposed on patients due to ERIA. In addition, patients with ERIA showed a decreased QoL and increased depression rates. Our results are in line with other studies on ERIA, showing that number and frequency of injuries and accidents are significantly increased in epilepsy patients compared to matched controls. However, there are certain limitations of the study design (e.g., a missing epidemiologic approach) that may display limitations of the acquired data and findings. The finding that most enrolled patients had focal and active epilepsy may also affect the comparability with other studies.
In a large database study published in 2010, 8,890 subjects with epilepsy were analyzed, resulting in a 1-year incidence of one or more injuries in 20.6% of patients with and 16.1% of patients without epilepsy (p < 0.001) (34). Active epilepsy and refractory course of disease, as demonstrated in our cohort, are variables associated with ERIA. Within the 3-month observation period, 41 of 292 patients (14.0%) reported ERIA. The most common symptoms were lacerations (n = 18, 6.2%) followed by abrasions and bruises (n = 9, 3.1%), fractures (n = 6, 2.1%) and burns (n = 3, 1.0%). In 20 cases, urgent medical treatment and hospitalization was necessary. These results are well in line with previously published data on ERIA from a pilot study from 2008 among our outpatients (18, 35) and other international publications reporting ERIA in children, adolescents and adults with epilepsy in Nigeria, the United States, Australia and the UK (34, 36–41). In addition, a study reporting on 52 patients with refractory focal epilepsy led to comparable results with epilepsy-related injuries in 21% of cases with TLE and 8% with other focal epilepsies within a mean disease duration of 22 and 17 years. Lifetime prevalence of ERIA was estimated to 57%. Moreover, this study reported that only 45% of ERIA in general, and especially severe ones, had been documented (17). This is in contrast to another study with 247 enrolled patients showing a rather low frequency for ERIA of 1 per 44 patient years and which claimed a minor severity for the most reported cases (41).
Another prospective study analyzed ERIA using a control group of healthy relatives, showing that epilepsy patients have a 21% higher likelihood to harm themselves over a 1–2 year observation period, however, mainly mild and trivial injuries were reported (42). In children aged 2–16 years, ERIA were reported in 4.7% (43) and children with epilepsy were shown to have a 18–23% increased risk of fracture, a 49% increased risk of thermal injury and more than twice the risk of poisoning from medicinal products (39). Differences between the mentioned studies are probably due to diverging study designs (prospective vs. retrospective), different cohort characteristics and may be influenced by local factors as well as awareness and ascertainment issues. ERIA are commonly underreported and there is a high estimated number of underreported cases (17). The high frequency of ERIA in our study can be attributed to the targeted questions and precise documentation reducing underascertainment.
The vast majority of patients reporting ERIA (96.0%) presented specific ictal features and we were able to identify a significant correlation between ERIA with epilepsy characteristics and semiological aspects. Variables associated with ERIA were active epilepsy, drug-refractory course as well as reported ictal falls and ictal loss of consciousness or abnormal peri-ictal behavior in the medical history. These findings are similar to the results of past studies (18). Moreover, the presence of GTCS was significantly associated with ERIA, which is in line with previous studies, in which over 80% of ERIA occurred during GTCS (41). Additionally, we were able to show a significant coherence between ERIA and abnormal peri-ictal behavior, which was previously controversially discussed (18). Patients with frequent and prolonged ictal auras seem to be at lower risk, which is probably based on the possibility of informing bystanders, calling for help and creating a secure surrounding. As another high-risk subgroup, individuals with active epilepsy could be identified. There was no significant difference in the occurrence of ERIA between patients with FE or IGE, which is surprising because of the common presence of GTCS in patients with IGE. Overall, 75% of IGE patients presented with GTCS compared to 51% with FE in an ERIA cohort (40).
A second focus of our study was to assess QoL and depression as possible consequence of ERIA using a battery of neuropsychological inventories (BDI-II, NDDIE, EQ5D, QOLIE-31, ABNAS, and LEAP) that have been already established and validated in patients with epilepsy (25–29). Subjects reporting ERIA showed significantly increased depression rates and had a lower QoL, which has not been reported so far. The association of lower QoL and a trend to depression with drug-refractory epilepsy has been already shown (12, 27, 28, 31, 44, 45) and ERIA might be associated or rather aggravate these aspects. However, this study was not designed to further analyse and correlate these aspects. Also, adverse effects due to AEDs were significantly higher in patients with ERIA.
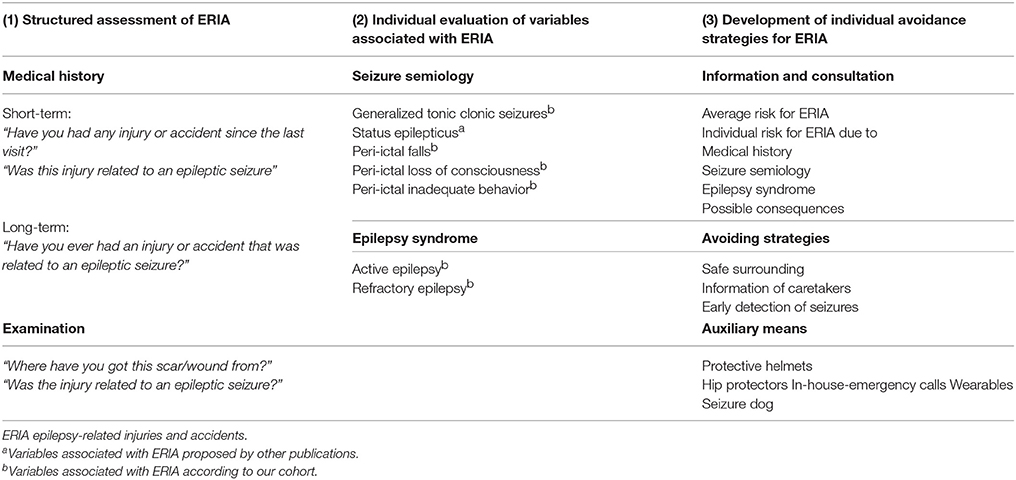
The most practical solution to injury prevention is a better seizure control (40, 41). However, this may not be achieved, especially in patients with drug-refractory epilepsies who have been shown to be a high-risk group for ERIA. Therefore, another focus should aim at reducing the risk for ERIA. We propose a prophylactic pathway focusing on three major aspects: (1) identifying high risk patients by a standardized assessment of ERIA, (2) optimising antiepileptic therapy, and (3) developing individual safety assessments to reduce the future risk of ERIA (see Table 7). Individual safety assessments may contain information and education on the risks and consequences of ERIA, possibilities to avoid injuries (behavior, early detection) and suggestions for auxiliary means (helmets, hip protectors, in-house emergency calls, wearables). Even if the majority of epilepsy-related injuries and accidents occur at home (88%) and not in a public space (10%), a professional and epilepsy-specific assessment of the actual or aspired profession and the concomitant work environment seems to be unavoidable, hence, the fact that only 2% of epilepsy-related injuries and accidents occur at work (46, 47).
Conclusion
ERIA, as well as their clinical consequences, are an underreported but highly relevant aspect in the treatment of patients with active epilepsy. Moreover, ERIA might be associated or rather aggravate neuropsychological symptoms such as depression or decreased QOL in this severely affected subgroup of patients with epilepsy. However, ERIA are only sporadically considered in the development of tailored anti-epileptic therapies. More effort should be spent on the identification and assessment of ERIA or patients at risk as well as on prevention and general education on this topic to increase patient safety, satisfaction and QoL.
Author Contributions
LW and AS generated the research idea, study design, and concept. NW, SR, and AS acquired the data. LW and AS analyzed the data and drafted the work. LW, NW, SR, LK, AH, SK, FR, and AS made critical revisions for important intellectual content and interpreted the data. LW and AS wrote the manuscript. LW, NW, SR, LK, AH, SK, FR, and AS approved the final manuscript.
Conflict of Interest Statement
SK reports personal fees from Desitin, Eisai, UCB, as well as support for scientific meetings from AD-tech, Desitin, Eisai, GW pharma, LivaNova, Nihon Kohden and Novartis outside the submitted work. FR reports personal fees from Eisai, grants and personal fees from UCB, grants and personal fees from Desitin Arzneimittel, personal fees and other from Novartis, personal fees from Medronic, personal fees from Cerbomed, personal fees from ViroPharma and Shire, grants from European Union, grants from Deutsche Forschungsgemeinschaft, outside the submitted work. AS reports personal fees and grants from Desitin Arzneimittel, Eisai, LivaNova, Sage Therapeutics, UCB Pharma and Zogenix, outside the submitted work.
The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
We are grateful to the patients participating in this study and to our colleagues for their assistance in conducting this study.
References
1. GBD 2015 Disease and Injury Incidence and Prevalence Collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990-2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet (2016) 388:1545–602. doi: 10.1016/S0140-6736(16)31678-6
2. Strzelczyk A, Reese JP, Dodel R, Hamer HM. Cost of epilepsy: a systematic review. Pharmacoeconomics (2008) 26:463–76. doi: 10.2165/00019053-200826060-00002
3. Willems LM, Richter S, Watermann N, Bauer S, Klein KM, Reese JP, et al. Trends in resource utilization and prescription of anticonvulsants for patients with active epilepsy in Germany from 2003 to 2013 - a ten-year overview. Epilepsy Behav. (2018) 83:28–35. doi: 10.1016/j.yebeh.2018.03.025
4. Forsgren I, Beghi E, Ekman M. Cost of epilepsy in Europe. Eur J Neurol. (2005) 12(Suppl. 1):54–8. doi: 10.1111/j.1468-1331.2005.01191.x
5. Smeets VMJ, van Lierop BAG, Vanhoutvin JPG, Aldenkamp AP, Nijhuis FJN. Epilepsy and employment: literature review. Epilepsy Behav. (2007) 10:354–62. doi: 10.1016/j.yebeh.2007.02.006
6. Haag A, Strzelczyk A, Bauer S, Kuhne S, Hamer HM, Rosenow F. Quality of life and employment status are correlated with antiepileptic monotherapy versus polytherapy and not with use of “newer” versus “classic” drugs: Results of the “Compliant 2006” survey in 907 patients. Epilepsy Behav. (2010) 19:618–22. doi: 10.1016/j.yebeh.2010.09.037
7. Jacoby A. Stigma, epilepsy, and quality of life. Epilepsy Behav. (2002) 3:S10–20. doi: 10.1016/S1525-5050(02)00545-0
8. Helmers SL, Kobau R, Sajatovic M, Jobst BC, Privitera M, Devinsky O, et al. Self-management in epilepsy: Why and how you should incorporate self-management in your practice. Epilepsy Behav. (2017) 68:220–4. doi: 10.1016/j.yebeh.2016.11.015
9. Jehi L, Tesar G, Obuchowski N, Novak E, Najm I. Quality of life in 1931 adult patients with epilepsy: seizures do not tell the whole story. Epilepsy Behav. (2011) 22:723–7. doi: 10.1016/j.yebeh.2011.08.039
10. Buck D, Baker GA, Jacoby A, Smith DF, Chadwick DW. Patients' experiences of injury as a result of epilepsy. Epilepsia (1997) 38:439–44.
11. Wirrell EC. Epilepsy-related injuries. Epilepsia (2006) 47(Suppl. 1):79–86. doi: 10.1111/j.1528-1167.2006.00666.x
12. Strzelczyk A, Griebel C, Lux W, Rosenow F, Reese JP. The burden of severely drug-refractory epilepsy: a comparative longitudinal evaluation of mortality, morbidity, resource use and cost using German Health insurance data. Front Neurol. (2017) 8:712. doi: 10.3389/fneur.2017.00712
13. Nashef L, So EL, Ryvlin P, Tomson T. Unifying the definitions of sudden unexpected death in epilepsy. Epilepsia (2012) 53:227–33. doi: 10.1111/j.1528-1167.2011.03358.x
14. Annegers JF. United States perspective on definitions and classifications. Epilepsia (1997) 38:S9–12. doi: 10.1111/j.1528-1157.1997.tb06137.x
15. Sillanpaa M, Shinnar S. Long-term mortality in childhood-onset epilepsy. N Engl J Med. (2010) 363:2522–9. doi: 10.1056/NEJMoa0911610
16. Strzelczyk A, Nickolay T, Bauer S, Haag A, Knake S, Oertel WH, et al. Evaluation of health-care utilization among adult patients with epilepsy in Germany. Epilepsy Behav. (2012) 23:451–7. doi: 10.1016/j.yebeh.2012.01.021
17. Friedman DE, Gilliam FG. Seizure-related injuries are underreported in pharmacoresistant localization-related epilepsy. Epilepsia (2010) 51:43–7. doi: 10.1111/j.1528-1167.2009.02170.x
18. Strzelczyk A, Hermsen A, Oertel WH, Knake S, Rosenow F, Hamer HM. Risk factors and incidence of epilepsy-related injuries an accidents. Nervenheilkunde (2014) 2014:331–4. doi: 10.4081/ni.2009.e20
19. Kortland LM, Alfter A, Bahr O, Carl B, Dodel R, Freiman TM, et al. Costs and cost-driving factors for acute treatment of adults with status epilepticus: a multicenter cohort study from Germany. Epilepsia (2016) 57:2056–66.
20. Riechmann J, Strzelczyk A, Reese JP, Boor R, Stephani U, Langner C, et al. Costs of epilepsy and cost-driving factors in children, adolescents, and their caregivers in Germany. Epilepsia (2015) 56:1388–97. doi: 10.1111/epi.13584
21. Strzelczyk A, Haag A, Reese JP, Nickolay T, Oertel WH, Dodel R, et al. Trends in resource utilization and prescription of anticonvulsants for patients with active epilepsy in Germany. Epilepsy Behav. (2013) 27:433–8. doi: 10.1016/j.yebeh.2013.03.007
22. Fisher RS, Cross JH, French JA, Higurashi N, Hirsch E, Jansen FE, et al. Operational classification of seizure types by the International League Against Epilepsy: Position Paper of the ILAE Commission for Classification and Terminology. Epilepsia (2017) 58:522–30. doi: 10.1111/epi.13670
23. Deutsche Gesetzliche Unfallversicherung e.V. (DGUV) DGUV Information 250-001: Berufliche Beurteilung bei Epilepsie und nach erstem epileptischen Anfall. Berlin: Ausschuss Arbeitsmedizin der Gesetzlichen Unfallversicherung (2015).
24. Kwan P, Arzimanoglou A, Berg AT, Brodie MJ, Allen Hauser W, Mathern G, et al. Definition of drug resistant epilepsy: consensus proposal by the ad hoc task force of the ILAE Commission on therapeutic strategies. Epilepsia (2010) 51:1069–77. doi: 10.1111/j.1528-1167.2009.02397.x
25. May TW, Pfafflin M, Cramer JA. Psychometric properties of the german translation of the QOLIE-31. Epilepsy Behav. (2001) 2:106–14. doi: 10.1006/ebeh.2001.0170
26. Rabin R, de Charro F. EQ-5D: a measure of health status from the EuroQol Group. Ann Med. (2001) 33:337–43. doi: 10.3109/07853890109002087
27. Metternich B, Wagner K, Buschmann F, Anger R, Schulze-Bonhage A. Validation of a German version of the neurological disorders depression inventory for Epilepsy (NDDI-E). Epilepsy Behav. (2012) 25:485–8. doi: 10.1016/j.yebeh.2012.10.004
28. de Oliveira GN, Lessa JM, Goncalves AP, Portela EJ, Sander JW, Teixeira AL. Screening for depression in people with epilepsy: comparative study among neurological disorders depression inventory for epilepsy (NDDI-E), hospital anxiety and depression scale depression subscale (HADS-D), and Beck depression inventory (BDI). Epilepsy Behav. (2014) 34:50–4. doi: 10.1016/j.yebeh.2014.03.003
29. Samarasekera SR, Helmstaedter C, Reuber M. Cognitive impairment in adults with epilepsy: the relationship between subjective and objective assessments of cognition. Epilepsy Behav. (2015) 52:9–13. doi: 10.1016/j.yebeh.2015.08.013
30. Panelli RJ, Kilpatrick C, Moore SM, Matkovic Z, D'Souza WJ, O'Brien TJ. The liverpool adverse events profile: relation to AED use and mood. Epilepsia (2007) 48:456–63. doi: 10.1111/j.1528-1167.2006.00956.x
31. Kortland LM, Knake S, von Podewils F, Rosenow F, Strzelczyk A. Socioeconomic outcome and quality of life in adults after status epilepticus: a multicenter, longitudinal, matched case-control analysis from Germany. Front Neurol. (2017) 8:507. doi: 10.3389/fneur.2017.00507
32. Benjamini Y, Drai D, Elmer G, Kafkafi N, Golani I. Controlling the false discovery rate in behavior genetics research. Behav Brain Res. (2001) 125:279–84. doi: 10.1016/S0166-4328(01)00297-2
33. Benjamini Y, Hochberg Y. Controlling the false discovery rate - a practical and powerful approach to multiple testing. J R Stat Soc Ser B-Methodol. (1995) 57:289–300. doi: 10.2307/2346101
34. Kwon CS, Liu M, Quan H, Wiebe S, McChesney J, Wirrell E, Hamiwka L, Jette N. The incidence of injuries in persons with and without epilepsy–a population-based study. Epilepsia (2010) 51:2247–53. doi: 10.1111/j.1528-1167.2010.02697.x
35. Strzelczyk A, Reese JP, Oertel WH, Dodel R, Rosenow F, Hamer HM. Costs of epilepsy and their predictors: Cross-sectional study in Germany and review of literature. Nervenheilkund. Epileptology (2013) 1:55–60. doi: 10.1016/j.epilep.2013.01.004
36. Lagunju IA, Oyinlade AO, Babatunde OD. Seizure-related injuries in children and adolescents with epilepsy. Epilepsy Behav. (2016) 54:131–4. doi: 10.1016/j.yebeh.2015.11.019
37. Baca CB, Vickrey BG, Vassar SD, Cook A, Berg AT. Injuries in adolescents with childhood-onset epilepsy compared with sibling controls. J Pediatr. (2013) 163:1684–91 e4. doi: 10.1016/j.jpeds.2013.07.046
38. Bellon M, Walker C, Peterson C. Seizure-related injuries and hospitalizations: self-report data from the 2010 Australian epilepsy longitudinal survey. Epilepsy Behav. (2013) 26:7–10. doi: 10.1016/j.yebeh.2012.10.019
39. Prasad V, Kendrick D, Sayal K, Thomas SL, West J. Injury among children and young adults with epilepsy. Pediatrics (2014) 133:827–35. doi: 10.1542/peds.2013-2554
40. Camfield C, Camfield P. Injuries from seizures are a serious, persistent problem in childhood onset epilepsy: a population-based study. Seizure (2015) 27:80–3. doi: 10.1016/j.seizure.2015.02.031
41. Lawn ND, Bamlet WR, Radhakrishnan K, O'Brien PC, So EL. Injuries due to seizures in persons with epilepsy: a population-based study. Neurology (2004) 63:1565–70. doi: 10.1212/01.WNL.0000142991.14507.B5
42. Beghi E, Cornaggia C, Group RE. Morbidity and accidents in patients with epilepsy: results of a European cohort study. Epilepsia (2002) 43:1076–83. doi: 10.1046/j.1528-1157.2002.18701.x
43. Sajjan S, Jain P, Sharma S, Seth A, Aneja S. Injuries in children with epilepsy: a hospital based study. Indian Pediatr. (2016) 53:883–885. doi: 10.1007/s13312-016-0952-7
44. Cianchetti C, Bianchi E, Guerrini R, Baglietto MG, Briguglio M, Cappelletti S, et al. Symptoms of anxiety and depression and family's quality of life in children and adolescents with epilepsy. Epilepsy Behav. (2017) 79:146–53. doi: 10.1016/j.yebeh.2017.11.030
45. Xu Y, Hackett ML, Glozier N, Nikpour A, Bleasel A, Somerville E, et al. Frequency and predictors of psychological distress after a diagnosis of epilepsy: a community-based study. Epilepsy Behav. (2017) 75:190–5. doi: 10.1016/j.yebeh.2017.07.044
46. Martin R, Vogtle L, Gilliam F, Faught E. What are the concerns of older adults living with epilepsy? Epilepsy Behav. (2005) 7:297–300. doi: 10.1016/j.yebeh.2005.05.003
Keywords: epilepsy, seizure, falls, laceration, accident
Citation: Willems LM, Watermann N, Richter S, Kay L, Hermsen AM, Knake S, Rosenow F and Strzelczyk A (2018) Incidence, Risk Factors and Consequences of Epilepsy-Related Injuries and Accidents: A Retrospective, Single Center Study. Front. Neurol. 9:414. doi: 10.3389/fneur.2018.00414
Received: 26 February 2018; Accepted: 18 May 2018;
Published: 15 June 2018.
Edited by:
Fernando Cendes, Universidade Estadual de Campinas, BrazilReviewed by:
Dieter Schmidt, Epilepsy Research Group, GermanyMartin Holtkamp, Charité Universitätsmedizin Berlin, Germany
Copyright © 2018 Willems, Watermann, Richter, Kay, Hermsen, Knake, Rosenow and Strzelczyk. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Laurent M. Willems, bGF1cmVudC53aWxsZW1zQGtndS5kZQ==
 Laurent M. Willems
Laurent M. Willems Nina Watermann2
Nina Watermann2 Susanne Knake
Susanne Knake Adam Strzelczyk
Adam Strzelczyk