- 1Nuffield Department of Clinical Neurosciences, University of Oxford, Oxford, United Kingdom
- 2Computational Neuroimaging Group, Academic Unit of Neurology, Trinity College Dublin, Dublin, Ireland
- 3Wellcome Centre for Integrative Neuroimaging, University of Oxford, Oxford, United Kingdom
Advances in neuroimaging, complementing histopathological insights, have established a multi-system involvement of cerebral networks beyond the traditional neuromuscular pathological view of amyotrophic lateral sclerosis (ALS). The development of effective disease-modifying therapy remains a priority and this will be facilitated by improved biomarkers of motor system integrity against which to assess the efficacy of candidate drugs. Functional MRI (FMRI) is an established measure of both cerebral activity and connectivity, but there is an increasing recognition of neuronal oscillations in facilitating long-distance communication across the cortical surface. Such dynamic synchronization vastly expands the connectivity foundations defined by traditional neuronal architecture. This review considers the unique pathogenic insights afforded by the capture of cerebral disease activity in ALS using FMRI and encephalography.
Introduction
Neurodegenerative disorders are increasingly understood as a disintegration of complex cerebral functional networks (1). Amyotrophic lateral sclerosis (ALS) is characterized by loss of upper and lower motor neurones of the corticospinal tract, brainstem, and spinal anterior horns, resulting in progressive weakness of downstream muscles. In addition to protean upstream causes (2, 3), there is firmly established clinical, pathological, and genetic overlap of ALS with frontotemporal dementia (FTD). The diagnosis remains a clinical one, with a lack of biomarkers being a significant barrier to the development of highly-effective disease-modifying therapy. Advanced neuroimaging, in combination with histopathological insights, has brought the brain to the forefront of biomarker development (4).
The earliest studies of cerebral blood flow in ALS employed positron emission tomography, and demonstrated a widened region of cortical activation in response to a simple motor task (5). Among the hypotheses for this “boundary shift effect” was loss of local inhibitory GABA-ergic interneuronal circuits [reviewed in (6)]. A consistent pathological feature of ALS has been the observation of increased cortical excitability, possibly reflecting reduced local inhibitory influences, measured using short-interval paired transcranial magnetic stimulation (TMS) (7, 8). Through the characterization of monogenetic associations, ALS research has expanded to include the study of what is now thought to be a long presymptomatic phase (9), in which cortical functional abnormalities may be the among the earliest detectable manifestations (10).
Blood oxygenation level-dependent (BOLD)-based functional (FMRI), with the major advantages of avoiding ionizing radiation and the greater availability of MRI technology, confirmed a profound alteration in cortical activity inherent to the pathogenesis of ALS. Tools to study cortical neurophysiology in real-time have also undergone significant development in both sensitivity and analysis. This review will consider the unique insights that FMRI and encephalography bring to the understanding of the pathogenesis of ALS at the systems level, which is increasingly of greatest relevance to the long-term goal of neuroprotective therapy.
Functional MRI
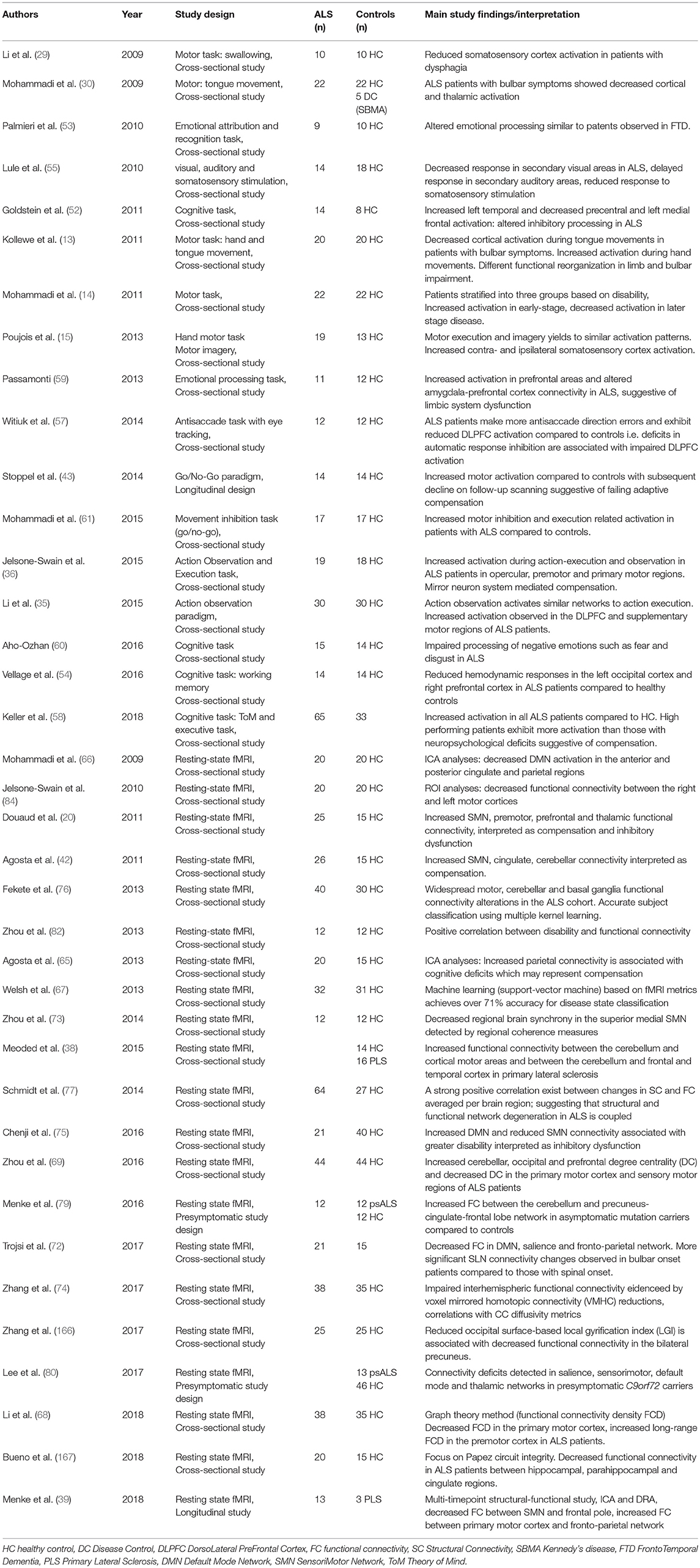
FMRI has been extensively used to characterize network dysfunction in ALS in cross-sectional, longitudinal, and presymptomatic study designs. The practical advantages of FMRI in ALS include the widespread availability of MRI platforms, the large number of freely available analysis suites and the ability to provide crucial biological insights in relatively simple, cost-effective, and non-invasive study designs (11). Despite some inconsistencies in the literature (12), two main themes have consistently emerged from the application of FMRI to ALS: (i) the characterization of compensatory changes, such as the recruitment of additional cortical (13–16), subcortical (14, 17, 18), and cerebellar (17, 19) regions to execute motor and cognitive tasks, and ii) the concept of inhibitory dysfunction as a key facet of ALS-associated pathophysiology (6, 20).
Methodological Considerations
Functional MRI has been extensively used to characterize network dysfunction in ALS in cross-sectional, longitudinal, and presymptomatic study designs (21) (Table 1), but method-associated limitations are rarely articulated. The protracted hemodynamic response to neural activity hampers the temporal resolution of fMRI. Blood oxygen-level dependent (BOLD) signal typically peaks only 5–6s after focal activation therefore careful study designs are indispensable for meaningful temporal inferences (22, 23). Spatial distortions and signal dropout due to susceptibility gradients near air-tissue interfaces lead to decreased BOLD signal in lateral parietal, orbitofrontal and dorsolateral prefrontal regions necessitating meticulous sequence optimization before data acquisition (24–26). Scanner noise may also impact on the interpretation of BOLD signal, particularly in the default-mode network (27), and careful experimental designs are required to minimize the influence of background noise (28). While fMRI findings are often presented by overlaying activation maps upon high-resolution structural images, the inherent spatial resolution of fMRI is limited by the signal-to-noise ratio profile of consecutive, rapid whole-brain imaging. In the majority ALS studies, the voxel size of fMRI protocols is two to four times larger than what is used for structural acquisitions (21).
Motor Paradigms
Pioneering FMRI studies in ALS relied initially on hand movement paradigms (15, 16), which were gradually complemented by innovative bulbar studies (13, 29, 30). In motor-task FMRI studies, different strategies have been utilized to control for limb weakness, motor effort and lower motor neuron involvement for the interpretation of cerebral activation. Motor imagery (31) has attracted considerable attention, not only for emerging brain-machine interface applications (32) but also as an FMRI paradigm for a condition like ALS in which patients typically develop severe motor disability (33). The execution and imagination of specific movements manifest in similar activation patterns in ALS and controls (15) suggesting that this approach may be particularly pertinent to patient cohorts with mixed disability profiles. Some ALS studies however report divergent activation maps in motor imagery and execution (34). Similarly to motor imagery, action observation is also thought to result in comparable cortical activity to action execution which has been used to study the mirror-neuron system in ALS (35, 36). Another approach to control for motor disability and establish ALS-specific activation patterns is the inclusion of disease-controls, i.e., non-ALS patients with motor disability (16, 37). Very few FMRI studies to date have specifically evaluated functional changes in other rarer motor neurone disorders such as the upper motor neurone-only primary lateral sclerosis (PLS) (38, 39) and lower motor neurone-dominated Kennedy's disease (30) using motor paradigms. Patient stratification into separate study groups based on motor disability is another strategy to interpret functional alterations in the context of disability (14). In light of the fundamentally divergent study designs, the inclusion of patients in different stages of their disease and small sample sizes, the inconsistent findings of motor activation studies are not surprising. Whilst, hypo- (29, 30, 40) and hyper-activation (13, 14, 16, 35, 36) of the somatosensory cortex have both been reported in response to motor tasks, the recruitment of premotor areas is a relatively consistent finding. An integrative explanation of the seemingly divergent findings is that the initial hyper-activation represents an early-stage adaptive process to execute movement (14), which gradually gives place to hypo-activation as progressive structural changes ensue (41, 42). Robust multi-timepoint longitudinal studies are required to clarify the timeline of functional changes in ALS as very few task-based longitudinal FMRI studies have been published to date (33, 43). One longitudinal study identified reduced motor activation on 3-month follow-up which was interpreted as compensatory failure due to progressive neural loss (43), while another study reported increased precentral gyrus activity 6-month after initial scanning as evidence of ongoing adaptation (33). In addition to compensatory processes in motor, premotor and supplementary motor areas (44), evidence also exist that the basal ganglia (17, 18, 45, 46), the ipsilateral motor cortex (14, 47), and the cerebellum (17, 19, 47, 48) also contribute to adaptive network reorganization.
Extra-Motor Studies
With the increasing recognition of cognitive impairment in ALS (49, 50), a series of elegant language (51), executive (52), theory-of-mind (36), and memory (43, 53, 54) task-based activation studies have also been published. In addition to the cognitive activation paradigms, visual, auditory and somatosensory stimulation studies have further characterized the spectrum of extra-motor involvement in ALS (55, 56). Other innovative non-motor activation studies in ALS include an anti-saccade study with concurrent eye tracking to investigate dorsolateral prefrontal cortex (DLPFC) function (57). Similar to the divergent findings of motor-task studies, increased activation (36, 52, 58, 59) and impaired activation (51, 60) have both been noted on cognitive tasks, which is likely to represent stages of successful and failing adaptation. More often however a pattern of coexisting hypo- and hyper- activation is reported (37, 54, 61).
Resting-State Studies
The analysis of task-free BOLD signal in the so-called resting-state (rsFMRI) benefits from fast acquisition times with a data-driven, more consistent experimental design, making them an attractive add-on to high-resolution structural protocols. With the establishment of the internationally collaborative Neuroimaging Society in ALS (NiSALS) (62) and successful multi-site initiatives (63), there is interest in FMRI sequence harmonization and potential for multicentre data pooling (12, 64). rsFMRI studies differ considerably in their analysis approaches and their methods span from independent component analysis (65–67), to graph theory (68, 69) and amplitude of low frequency fluctuation (ALFF) (70, 71). rsFMRI studies in ALS identified decreased frontotemporal (72), sensorimotor (70, 73–75), and cortical-subcortical (76) network integrity and increased default mode network (75), and cerebellar (38, 69) connectivity. Large combined structural-FMRI studies suggest that patterns of structural degeneration overlap with functionally impaired regions and that a strong positive correlation exists between functional and structural connectivity alterations (77). Longitudinal rsFMRI studies indicate declining functional connectivity in sensorimotor, thalamic, and visual networks and increasing connectivity in fronto-parietal and temporal circuits (39). Multimodal, structural-functional, multi-timepoint longitudinal studies (39) are best suited to characterize the natural history of progressive neurodegenerative changes (78). Data from presymptomatic carriers of ALS-causing gene mutations revealed increased cerebello-cerebral functional connectivity (79) and decreased salience, sensorimotor, default-mode, and thalamic networks connectivity (80). Despite the controversy around direct clinico-radiological correlations (81), some studies in ALS have reported significant associations, most often with functional measures (73, 82–84), disease duration (59, 73), and progression rates (20, 40, 85).
Practical Limitations
For a condition in which accumulation of physical disability is accompanied by ventilatory compromise with orthopnoea, supine MRI limits longitudinal assessment to those with slower rates of progression (39). The application of such a biomarker as an outcome measure in a small-scale clinical trial would then entail costly statistical compromises, since no ideal solution exists for the imputation of data points selectively lost from those patients with more aggressive disease (86).
Encephalography
Cortical processes, and the diseases that impact on them, are inadequately described without reference to dynamic neural communication (87, 88), but this necessitates temporal precision, without the dispersive effects of the haemodynamic response function that smears neural signals across several seconds (89). Surface electroencephalography (EEG) as a biomarker in ALS is appealingly practical, well tolerated and non-invasive.
Methodological Limitations
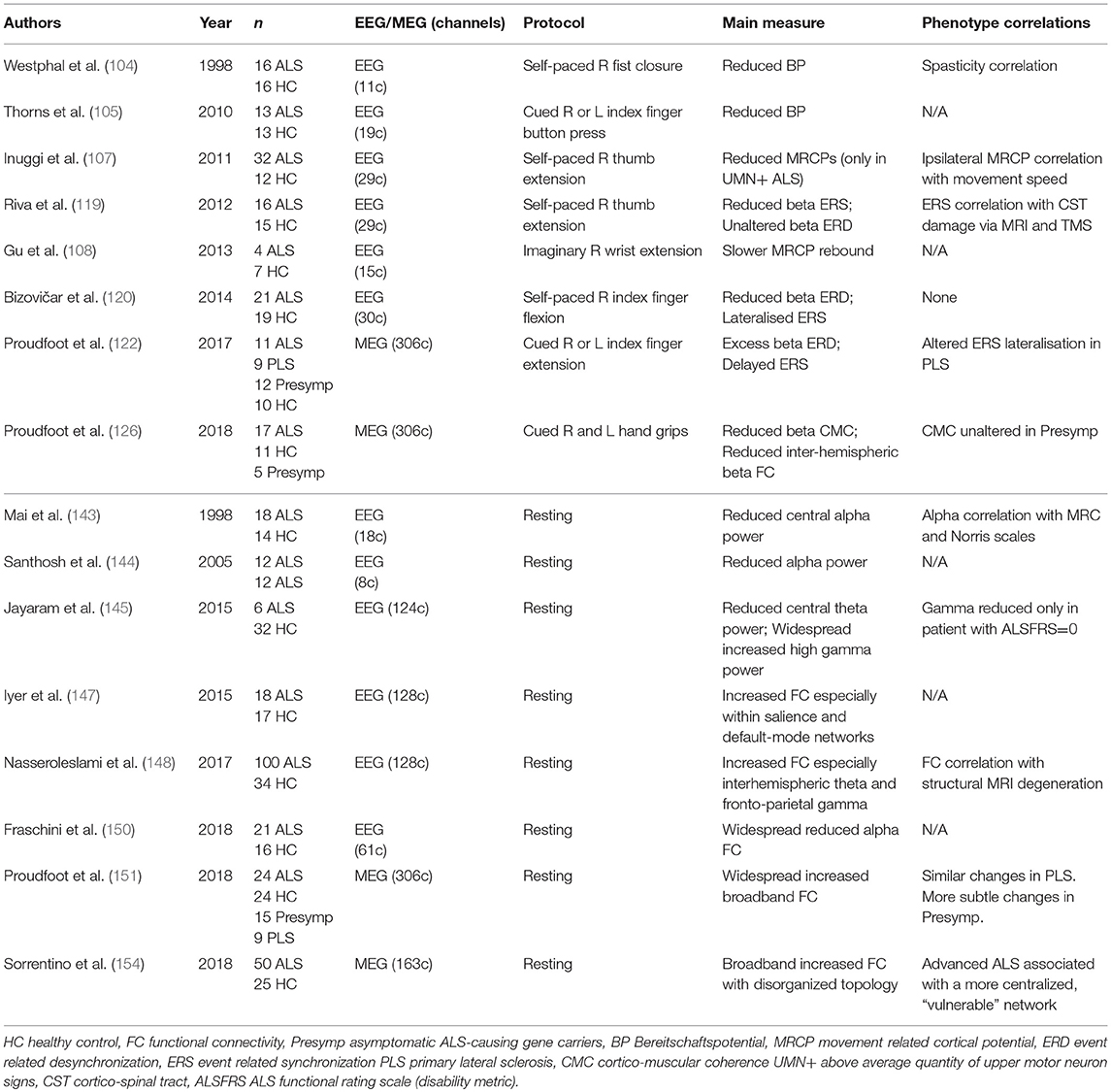
Even a high-density array of surface EEG electrodes still sacrifices spatial resolution owing to the attenuation and mutation of neural signals as they pass through several tissue layers with varying electrical conductivity (89). Magnetoencephalography (MEG) permits recording of tiny (femtoTesla) fluctuations in the magnetic field external to (and undispersed by) the scalp (90). Yet reconstruction of cortical sources remains a mathematically “ill-posed” problem—any given recorded signal could in theory be generated by multiple neural sources and the analytical choice to address this (for example “beamforming”) necessitates certain assumptions (91). MEG's improvement in spatial precision is also offset by expenses and susceptibility to artifact from ferromagnetic interference, albeit mitigated by acquisition and analysis standardization (92, 93). The resulting data is feature-rich, subsequent analysis may necessarily be restricted to a frequency-band of interest or a selected connectivity metric, these choices may in turn influence study conclusions (94) (Table 2).
Evoked Potentials
Small-scale EEG studies have addressed the utility of somatosensory, visual and brainstem evoked potentials in ALS (95–100). Reflecting the inconsistency of reported results, these well-established and standardized assessments have failed to find any routine clinical application in ALS, although they may yet find a role in multimodal assessment (101, 102).
To better reflect the pathological burden in ALS studies have therefore moved toward either motor or cognitive activation paradigms, initially appraising cortical processes via evoked response potentials [ERPs, previously reviewed in (103)]. The “Bereitschaftspotential,” a classical lateralized change in cortical electrical potential, easily recordable during movement preparation, appeared robustly decreased in ALS (104, 105). More recent studies have considered the implications of abnormal movement-related cortical potentials (MRCPs) in ALS in terms of clinical and structural correlates. While a study of 21 ALS patients demonstrated higher MRCPs overall, the effect was shown to be driven by patients with a low burden of clinically detectable UMN morbidity (106). The inference that increased MRCPs reflect cortical compensatory mechanisms was born out by longitudinal study of a sub-set in whom MRCPs declined over 10 months. A comparable study of finger movement in 32 ALS patients revealed reduced MRCPs only in patients with a high UMN burden, alongside evidence of ipsilateral premotor activation to suggest a compensatory “boundary shift” (107). MRCPs are also elicited during imagined movements, but only a limited study in ALS has thus far been performed (108), mandating replication before application of these measures in control and communication devices is to be seriously considered.
Motor Paradigms
Motor events (including self-generated movement) are reflected in frequency-specific changes to continuous “background” neuronal oscillations (109). As might be expected, the neurodegeneration associated with ALS results in distinct alteration to pre-central sensorimotor rhythms. While studies are yet to be widely replicated, they show promise both in terms of relevance to daily motor tasks, and sensitivity to detect early cortical dysfunction in patients still capable of performing the task in question. The results may also contribute to the ongoing efforts to characterize a presymptomatic phase to ALS and have implications for the development of brain-computer interfaces aiming to facilitate environmental control by patients with advanced ALS (110).
Movement is accompanied by reliable and well-characterized fluctuations in neural signal power, particularly within the beta (15–30 Hz) band, with recognizable anatomical localization to motor cortex. Beta-band power is reduced (event related desynchronization, ERD) prior to and during movement execution; movement termination is followed by an equally reliable increase in power well above baseline levels (synchronization, ERS or post-movement beta-rebound) (111). Temporally corresponding to fluctuations in cortical excitability (112), ERD and ERS are adjusted to meet task requirements [including force (113), speed (114), and complexity (115)], are sensitive to pharmacological manipulation [particularly synaptic GABA levels via benzodiazepines (116) or tiagabine (117)] and may be disrupted by other disease states including Parkinson's (118).
Motor Studies (EEG)
Two independent EEG studies have demonstrated attenuation of ERS in ALS. The first involved 16 patients efficiently performing self-paced thumb extensions (119). The degree of ERS attenuation was shown to correlate with corticospinal pathological burden as measured by both mean diffusivity on structural MRI and diminished motor evoked potentials in APB in response to TMS stimulation. The second study included 21 patients performing both sniffing and right index finger flexion (120). Although the patients had detectable weakness in terms of both maximal grip strength and sniff nasal-inspiratory pressure, there were no group differences in the precise pressure produced during the task performance. Neural data from the sniff task were heavily contaminated by facial muscle artifact, but the finger flexion task resulted in reliable ERD/S. The ALS patients were observed to have diminished beta ERD, interpreted as a consequence of pyramidal cell degeneration. Both motor preparation and execution timepoints were affected, while the lateralization of beta ERS was also altered. The study failed to establish clinical correlations with these measures, nor was there any successful correlation with F-wave elicitability (an imperfect measure of corticospinal tract integrity in any case).
Motor Studies (MEG)
The neural signal acquired by MEG is far less susceptible to distortion as it passes through skull and scalp, source modeling is therefore likely to be more accurate than EEG, and an expanding range of MEG studies have specifically appraised sensorimotor rhythms (121). A MEG study involving 11 ALS patients, 9 with PLS, and 12 asymptomatic genetic mutation carriers, investigated sensorimotor rhythms during a laterally-cued motor preparation task requiring speeded index finger extension of either hand (122). Whole-brain source-space data were analyzed pre, during, and post movement, specifically focusing on beta-band frequencies. Although the task was behaviorally performed comparably by ALS patients, the neural data revealed larger beta ERD, 500 ms after cue presentation, during the period of maximal motor preparation, particularly within contra and ipsilateral gyri. Beta ERS, after movement termination, was delayed in both patient groups. The asymptomatic carriers produced excessive beta ERD during motor execution. Conceptually the results are concordant with cross-modality support for cortical hyperexcitability in ALS (123, 124).
The integrity of upper motor neurone pathways can also be non-invasively appraised using MEG. Cortico-muscular coherence (CMC), by which neural oscillations and surface electromyography correlate temporally (particularly during sustained contraction), principally reflects direct corticospinal drive to the peripheral musculature (125). A MEG study of 17 ALS patients was designed to measure CMC during a bilateral forearm grip task (126). As expected, source-space beta CMC was distinctly strongest from the contra-lateral precentral gyrus, but this frequency specific peak was markedly attenuated in the ALS group, despite adequate grip production and without any correlation to force production. The analysis also took advantage of MEG spatial precision to consider motoric cortico-cortical communication during the same task performance. Interhemispheric functional connectivity, in terms of beta band amplitude envelope correlation, was reduced in ALS patients. The inference of reduced CMC, a measure that in health indexes the quality of motor performance (127), is that beta coherence may serve as a novel UMN specific biomarker at the disposal of future therapeutic efforts (128).
Extra-Motor Studies
Taking advantage of the high temporal resolution of encephalographic data, component steps in the complex cognitive dysfunction associated with the ALS-FTD syndrome may be examined. The mismatch negativity (MMN) paradigm considers the attentional modulation of auditory perception. An early EEG study failed to show any abnormalities within ALS patients (129). However, using MEG, plus subtle experimental design adjustments in 12 participants all with bulbar symptoms, MMN response amplitudes were shown to increase relative to healthy controls (130). Given the previously demonstrated sensitivity of MMN responses to ketamine administration, the authors tentatively linked their findings to the glutamatergic excitotoxicity ALS pathogenesis theory. This rare example of “gain of function” was not consistently replicated in two later EEG studies, which interpreted delayed MMN responses as evidence of sub-clinical extra-motor dysfunction (131, 132).
Less well-replicated methodologies have also been applied to ALS patients to consider neural processes underlying working memory (133, 134), selective attention (135, 136), and executive control (137, 138). Broadly, these studies have provided further evidence in favor of sub-clinical disruption to “frontal” cognitive processes in keeping with the extended non-motor ALS phenotype (139). Parietal cortex dysfunction was also implicated in an EEG study involving the Wisconsin Card Sorting Test. While 26 ALS patients did not differ in performance of a “set-shifting” task, even patients without mild cognitive impairment failed to produce the expected enhancement of parietal ERPs during a task-switch (140). Although the attenuation of the “switch potential” failed to correlate with neuropsychological indices, the authors speculated whether such sub-clinical deficits could predict future behavioral disorder.
A study requiring cognitive task performance during data acquisition took a very different analytical approach, using 200 s of data to measure “transfer entropy” between scalp electrodes rather than the millisecond granularity of evoked potentials. The directionality of functional connectivity was appraised via EEG in 18 ALS patients, revealing only feedforward (parietal to frontal) connectivity to increase across a broad frequency band (141). As the patients engaged in a spelling task with a view to brain-computer interfacing, sensory (visual) stimuli were hypothesized to be more readily processed in compensation for the diminished proprioceptive input resulting from physical disability, but an alternative explanation in terms of failing cortical inhibition was also acknowledged (20).
Resting-State Studies (EEG)
The earliest EEG investigations of ALS reflected the emerging concept of cognitive dysfunction within the ALS clinical spectrum, with slowed cortical rhythms noted in non-demented patients (142). A more systematic study of 18 ALS patients conversely revealed sparse differences to healthy controls (143). Only at central electrodes, and only within the alpha band (8–13 Hz), was the power of neural oscillations reduced in ALS. The reduction was interpreted to reflect selective neuronal loss within the sensorimotor cortices. A comparable result was described in a subsequent smaller study (144), and increases in the gamma band (30–90 Hz) power beyond central regions was also reported (145).
Further ALS electrophysiology studies have reflected a growing interest in the so-called “dynome” (146), the extent to which the organization of cortical function is reflected in particular patterns of active connectivity. High-density (128 channel) surface EEG was used to calculate connectivity between both scalp points and projected source nodes in an initial study of 18 patients (147). Fronto-central areas were shown to have increased connectivity, and this was explored across a broad range of measures. A subsequent study expanded this work to 100 patients, including some longitudinal analyses (148), and confirmed EEG-derived connectivity changes in ALS to be more striking than limited group differences in the scalp-recorded power spectrum. This more parsimonious analysis appraised only sensor-space, deriving coherence estimates within 8 consecutive frequency bands. Widespread increases in connectivity were again demonstrated relative to healthy controls, particularly theta band interhemispheric sensorimotor connectivity and gamma band fronto-parietal connectivity. As 59 of the ALS patients had undergone contemporaneous structural MRI, mathematically derived structural “degeneration modes” (accounting for the large-scale gray and white matter changes typical in ALS) were shown to correlate with EEG change, conceptually aligned with the concept of progressive network decline overlying structural disintegration.
Network structures can also be summarized using graph theory metrics, this was explored in sensor-space in 21 patients, demonstrating a more “de-centralised” organization (149). The connectivity metric chosen in this study was phase-based, thus insensitive to any group differences in spectral power, and furthermore was significantly correlated with disability between individuals. This group later re-analyzed the same data reconstructed into source-space (150) and filtered into 3 classical frequency bands to show spatially distributed decreases in connectivity, albeit restricted to the alpha band spectrum.
Resting-State Studies (MEG)
A resting-state MEG study explored functional connectivity in 24 ALS patients using source-space data acquired after co-registration with structural MRI (151). Ten minutes of continuous data was parcellated into 39 regions of interest and the broad-band (3–40 Hz) signal used to calculate “edge” strength between these 39 “nodes,” In keeping with many FMRI studies, functional connectivity was broadly increased in ALS patients relative to age-matched healthy controls, particularly affecting communication links to the posterior cingulate cortex. This finding was aligned with the hypothesis of loss of cortical inhibitory neuronal influences underlying cortical excitability in ALS (5). Comparable posterior non-motor connectivity changes were described using FMRI (152). Nevertheless, the diversity of reported results and interpretations serves to highlight a need for replication and standardization between centers and where possible across modalities (153). A further study of 50 patients, using a different (phase-based) connectivity measure, also described widespread connectivity increases in ALS (154). The increases were not restricted to specific frequency bands and the extracted graph theory metrics suggest global network hyper-centralization to accompany disease progression.
Future Directions
MEG is providing broader insight into cognitive mechanisms underpinning higher cortical function in health (155), and comparable results may eventually prove achievable using surface EEG (156). The next generation of wearable sensors may yet dramatically expand MEG's application (157). The spinal cord is a core but functionally understudied aspect of the motor system disintegration that characterizes ALS. Spinal FMRI is in its infancy (158), but a number of promising studies have already been published in animal models (159, 160), healthy populations (161, 162) and other clinical cohorts (163, 164). The goal of non-invasively studying the integrated activity of upper and lower motor neurone pools looks more feasible with the success in studies involving the dorsal pathways (165). Cerebral FMRI parameters are likely to take an increasing role in emerging machine-learning and classification studies both in diagnostic and prognostic applications (67, 76). Future studies need robust longitudinal design and to capitalize on the growing infrastructure for multicentre studies. This will permit the testing of pathogenic hypotheses within larger cohorts of clinically more homogeneous ALS patients, and define the earliest markers of pathology in presymptomatic individuals essential for the assessment of future neurotherapeutic interventions.
Author Contributions
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
Funding
PB is supported by the Health Research Board (HRB–Ireland; HRB EIA-2017-019), the Irish Institute of Clinical Neuroscience IICN–Novartis Ireland Research Grant, the Iris O'Brien Foundation, and the Research Motor Neuron (RMN-Ireland) Foundation.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
1. Seeley WW, Crawford RK, Zhou J, Miller BL, Greicius MD. Neurodegenerative diseases target large-scale human brain networks. Neuron (2009) 62:42–52. doi: 10.1016/j.neuron.2009.03.024
2. Turner MR, Swash M. The expanding syndrome of amyotrophic lateral sclerosis: a clinical and molecular odyssey. J Neurol Neurosurg Psychiatry (2015) 86:667–73. doi: 10.1136/jnnp-2014-308946
3. Talbot K, Feneberg E, Scaber J, Thompson AG, Turner MR. Amyotrophic lateral sclerosis: the complex path to precision medicine. J Neurol. (2018) 265:2454–62. doi: 10.1007/s00415-018-8983-8
4. Turner MR, Verstraete E. What does imaging reveal about the pathology of amyotrophic lateral sclerosis? Curr Neurol Neurosci Rep. (2015) 15:45. doi: 10.1007/s11910-015-0569-6
5. Kew JJM, Leigh PN, Playford ED, Passingham RE, Goldstein LH, Frackowiak RSJ, et al. Cortical function in amyotrophic lateral sclerosis: a positron emission tomography study. Brain (1993) 116:655–80. doi: 10.1093/brain/116.3.655
6. Turner MR, Kiernan MC. Does interneuronal dysfunction contribute to neurodegeneration in amyotrophic lateral sclerosis? Amyotroph Lateral Scler. (2012) 13:245–50. doi: 10.3109/17482968.2011.636050
7. Yokota T, Yoshino A, Inaba A, Saito Y. Double cortical stimulation in amyotrophic lateral sclerosis. J Neurol Neurosurg Psychiatry (1996) 61:596–600. doi: 10.1136/jnnp.61.6.596
8. Menon P, Geevasinga N, Yiannikas C, Howells J, Kiernan MC, Vucic S. Sensitivity and specificity of threshold tracking transcranial magnetic stimulation for diagnosis of amyotrophic lateral sclerosis: a prospective study. Lancet Neurol. (2015) 14:478–84. doi: 10.1016/S1474-4422(15)00014-9
9. Benatar M, Wuu J, Ravits J. Opportunity and innovation in studying pre-symptomatic amyotrophic lateral sclerosis. Muscle Nerve (2013) 47:629–31. doi: 10.1002/mus.23782
10. Vucic S, Nicholson GA, Kiernan MC. Cortical hyperexcitability may precede the onset of familial amyotrophic lateral sclerosis. Brain (2008) 131:1540–50. doi: 10.1093/brain/awn071
11. Bede P. Deciphering neurodegeneration. Neurology (2017) 89:1758–9. doi: 10.1212/WNL.0000000000004582
12. Bede P, Querin G, Pradat P-F. The changing landscape of motor neuron disease imaging: the transition from descriptive studies to precision clinical tools. Curr Opin Neurol. (2018) 31:431–8. doi: 10.1097/WCO.0000000000000569
13. Kollewe K, Münte TF, Samii A, Dengler R, Petri S, Mohammadi B. Patterns of cortical activity differ in ALS patients with limb and/or bulbar involvement depending on motor tasks. J Neurol. (2011) 258:804–10. doi: 10.1007/s00415-010-5842-7
14. Mohammadi B, Kollewe K, Samii A, Dengler R, Münte TF. Functional neuroimaging at different disease stages reveals distinct phases of neuroplastic changes in amyotrophic lateral sclerosis. Hum Brain Mapp. (2011) 32:750–8. doi: 10.1002/hbm.21064
15. Poujois A, Schneider FC, Faillenot I, Camdessanché J-P, Vandenberghe N, Thomas-Antérion C, et al. Brain plasticity in the motor network is correlated with disease progression in amyotrophic lateral sclerosis. Hum Brain Mapp. (2013) 34:2391–401. doi: 10.1002/hbm.22070
16. Stanton BR, Williams VC, Leigh PN, Williams SCR, Blain CRV, Jarosz JM, et al. Altered cortical activation during a motor task in ALS. Evidence for involvement of central pathways. J Neurol. (2007) 254:1260–7. doi: 10.1007/s00415-006-0513-4
17. Konrad C, Jansen A, Henningsen H, Sommer J, Turski PA, Brooks BR, et al. Subcortical reorganization in amyotrophic lateral sclerosis. Exp Brain Res. (2006) 172:361–9. doi: 10.1007/s00221-006-0352-7
18. Tessitore A, Esposito F, Monsurrò MR, Graziano S, Panza D, Russo A, et al. Subcortical motor plasticity in patients with sporadic ALS: An fMRI study. Brain Res Bull. (2006) 69:489–94. doi: 10.1016/j.brainresbull.2006.01.013
19. Prell T, Grosskreutz J. The involvement of the cerebellum in amyotrophic lateral sclerosis. Amyotroph Lateral Scler Frontotemporal Degener. (2013) 14:507–15. doi: 10.3109/21678421.2013.812661
20. Douaud G, Filippini N, Knight S, Talbot K, Turner MR. Integration of structural and functional magnetic resonance imaging in amyotrophic lateral sclerosis. Brain (2011) 134:3470–9. doi: 10.1093/brain/awr279
21. Bede P, Hardiman O. Lessons of ALS imaging: pitfalls and future directions - A critical review. NeuroImage Clin. (2014) 4:436–43. doi: 10.1016/j.nicl.2014.02.011
22. Buckner RL, Bandettini PA, O'Craven KM, Savoy RL, Petersen SE, Raichle ME, et al. Detection of cortical activation during averaged single trials of a cognitive task using functional magnetic resonance imaging. Proc Natl Acad Sci USA. (1996) 93:14878–83. doi: 10.1073/pnas.93.25.14878
23. Ogawa S, Lee T-M, Stepnoski R, Chen W, Zhu X-H, Ugurbil K. An approach to probe some neural systems interaction by functional MRI at neural time scale down to milliseconds. Proc Natl Acad Sci USA. (2000) 97:11026–31. doi: 10.1073/pnas.97.20.11026
24. Yang QX, Dardzinski BJ, Li S, Eslinger PJ, Smith MB. Multi-gradient echo with susceptibility inhomogeneity compensation (MGESIC): demonstration of fMRI in the olfactory cortex at 3.0 T. Magn Reson Med. (1997) 37:331–5. doi: 10.1002/mrm.1910370304
25. Weiger M, Pruessmann KP, Österbauer R, Börnert P, Boesiger P, Jezzard P. Sensitivity-encoded single-shot spiral imaging for reduced susceptibility artifacts in BOLD fMRI. Magn Reson Med. (2002) 48:860–6. doi: 10.1002/mrm.10286
26. Stenger VA, Boada FE, Noll DC. Three-dimensional tailored RF pulses for the reduction of susceptibility artifacts in T2/(*)-weighted functional MRI. Magn Reson Med. (2000) 44:525–31. doi: 10.1002/1522-2594(200010)44:4<525::AID-MRM5>3.0.CO;2-L
27. Gaab N, Gabrieli JDE, Glover GH. Resting in peace or noise: Scanner background noise suppresses default-mode network. Hum Brain Mapp. (2008) 29:858–67. doi: 10.1002/hbm.20578
28. Gaab N, Gabrieli JDE, Glover GH. Assessing the influence of scanner background noise on auditory processing. I. An fMRI study comparing three experimental designs with varying degrees of scanner noise. Hum Brain Mapp. (2007) 28:703–20. doi: 10.1002/hbm.20298
29. Li S, Chen Q, Yu B, Xue K, Luo C, Xu Y, et al. Structural and functional changes mapped in the brains of amyotrophic lateral sclerosis patients with/without dysphagia: A pilot study. Amyotroph Lateral Scler. (2009) 10:280–7. doi: 10.3109/17482960902893342
30. Mohammadi B, Kollewe K, Samii A, Krampfl K, Dengler R, Münte TF. Decreased brain activation to tongue movements in amyotrophic lateral sclerosis with bulbar involvement but not Kennedy syndrome. J Neurol. (2009) 256:1263–9. doi: 10.1007/s00415-009-5112-8
31. Szameitat AJ, Shen S, Sterr A. Motor imagery of complex everyday movements. An fMRI study. Neuroimage (2007) 34:702–13. doi: 10.1016/j.neuroimage.2006.09.033
32. Marchesotti S, Bassolino M, Serino A, Bleuler H, Blanke O. Quantifying the role of motor imagery in brain-machine interfaces. Sci Rep. (2016) 6:24076. doi: 10.1038/srep24076
33. Lulé D, Diekmann V, Kassubek J, Kurt A, Birbaumer N, Ludolph AC, et al. Cortical plasticity in amyotrophic lateral sclerosis: motor imagery and function. Neurorehabil Neural Repair. (2007) 21:518–26. doi: 10.1177/1545968307300698
34. Stanton BR, Williams VC, Leigh PN, Williams SCR, Blain CRV, Giampietro VP, et al. Cortical activation during motor imagery is reduced in Amyotrophic Lateral Sclerosis. Brain Res. (2007) 1172:145–51. doi: 10.1016/j.brainres.2007.07.044
35. Li H, Chen Y, Li Y, Yin B, Tang W, Yu X, et al. Altered cortical activation during action observation in amyotrophic lateral sclerosis patients: a parametric functional MRI study. Eur Radiol. (2015) 25:2584–92. doi: 10.1007/s00330-015-3671-x
36. Jelsone-Swain L, Persad C, Burkard D, Welsh RC. Action processing and mirror neuron function in patients with amyotrophic lateral sclerosis: an fMRI study. PLoS ONE (2015) 10:e0119862. doi: 10.1371/journal.pone.0119862
37. Lulé D, Diekmann V, Anders S, Kassubek J, Kübler A, Ludolph AC, et al. Brain responses to emotional stimuli in patients with amyotrophic lateral sclerosis (ALS). J Neurol. (2007) 254:519–27. doi: 10.1007/s00415-006-0409-3
38. Meoded A, Morrissette AE, Katipally R, Schanz O, Gotts SJ, Floeter MK. Cerebro-cerebellar connectivity is increased in primary lateral sclerosis. NeuroImage Clin. (2015) 7:288–96. doi: 10.1016/j.nicl.2014.12.009
39. Menke RAL, Proudfoot M, Talbot K, Turner MR. The two-year progression of structural and functional cerebral MRI in amyotrophic lateral sclerosis. NeuroImage Clin. (2018) 17:953–61. doi: 10.1016/j.nicl.2017.12.025
40. Cosottini M, Pesaresi I, Piazza S, Diciotti S, Cecchi P, Fabbri S, et al. Structural and functional evaluation of cortical motor areas in Amyotrophic Lateral Sclerosis. Exp Neurol. (2012) 234:169–80. doi: 10.1016/j.expneurol.2011.12.024
41. Bede P, Hardiman O. Longitudinal structural changes in ALS: a three time-point imaging study of white and gray matter degeneration. Amyotroph Lateral Scler Front Degener. (2018) 19:232–41. doi: 10.1080/21678421.2017.1407795
42. Agosta F, Valsasina P, Absinta M, Riva N, Sala S, Prelle A, et al. Sensorimotor functional connectivity changes in amyotrophic lateral sclerosis. Cereb Cortex (2011) 21:2291–8. doi: 10.1093/cercor/bhr002
43. Stoppel CM, Vielhaber S, Eckart C, Machts J, Kaufmann J, Heinze H-J, et al. Structural and functional hallmarks of amyotrophic lateral sclerosis progression in motor- and memory-related brain regions. NeuroImage Clin. (2014) 5:277–90. doi: 10.1016/j.nicl.2014.07.007
44. Konrad C, Henningsen H, Bremer J, Mock B, Deppe M, Buchinger C, et al. Pattern of cortical reorganization in amyotrophic lateral sclerosis: a functional magnetic resonance imaging study. Exp brain Res. (2002) 143:51–6. doi: 10.1007/s00221-001-0981-9
45. Bede P, Omer T, Finegan E, Chipika RH, Iyer PM, Doherty MA, et al. Connectivity-based characterisation of subcortical grey matter pathology in frontotemporal dementia and ALS: a multimodal neuroimaging study. Brain Imaging Behav. (2018) 12:1696–1707. doi: 10.1007/s11682-018-9837-9
46. Feron M, Couillandre A, Mseddi E, Termoz N, Abidi M, Bardinet E, et al. Extrapyramidal deficits in ALS: a combined biomechanical and neuroimaging study. J Neurol. (2018) 265:1–12. doi: 10.1007/s00415-018-8964-y
47. Schoenfeld MA, Tempelmann C, Gaul C, Kühnel GR, Düzel E, Hopf J-M, et al. Functional motor compensation in amyotrophic lateral sclerosis. J Neurol. (2005) 252:944–52. doi: 10.1007/s00415-005-0787-y
48. Han J, Ma L. Functional magnetic resonance imaging study of the brain in patients with amyotrophic lateral sclerosis. Chinese Med Sci J. (2006) 21:228–33.
49. Elamin M, Bede P, Byrne S, Jordan N, Gallagher L, Wynne B, et al. Cognitive changes predict functional decline in ALS: a population-based longitudinal study. Neurology (2013) 80:1590–7. doi: 10.1212/WNL.0b013e31828f18ac
50. Elamin M, Phukan J, Bede P, Jordan N, Byrne S, Pender N, et al. Executive dysfunction is a negative prognostic indicator in patients with ALS without dementia. Neurology (2011) 76:1263–9. doi: 10.1212/WNL.0b013e318214359f
51. Abrahams S, Goldstein LH, Simmons A, Brammer M, Williams SCR, Giampietro V, et al. Word retrieval in amyotrophic lateral sclerosis: a functional magnetic resonance imaging study. Brain (2004) 127:1507–17. doi: 10.1093/brain/awh170
52. Goldstein LH, Newsom-Davis IC, Bryant V, Brammer M, Leigh PN, Simmons A. Altered patterns of cortical activation in ALS patients during attention and cognitive response inhibition tasks. J Neurol. (2011) 258:2186–98. doi: 10.1007/s00415-011-6088-8
53. Palmieri A, Naccarato M, Abrahams S, Bonato M, D'Ascenzo C, Balestreri S, et al. Right hemisphere dysfunction and emotional processing in ALS: An fMRI study. J Neurol. (2010) 257:1970–8. doi: 10.1007/s00415-010-5640-2
54. Vellage AK, Veit M, Kobeleva X, Petri S, Vielhaber S, Müller NG. Working memory network changes in ALS: An fMRI study. Front Neurosci. (2016) 10:158. doi: 10.3389/fnins.2016.00158
55. Lulé D, Diekmann V, Müller HP, Kassubek J, Ludolph AC, Birbaumer N. Neuroimaging of multimodal sensory stimulation in amyotrophic lateral sclerosis. J Neurol Neurosurg Psychiatry (2010) 81:899–906. doi: 10.1136/jnnp.2009.192260
56. Bede P, Iyer PM, Schuster C, Elamin M, Mclaughlin RL, Kenna K, et al. The selective anatomical vulnerability of ALS: ‘disease-defining’ and ‘disease-defying’ brain regions. Amyotroph Lateral Scler Front Degener. (2016) 8421:1–10. doi: 10.3109/21678421.2016.1173702
57. Witiuk K, Fernandez-Ruiz J, McKee R, Alahyane N, Coe BC, Melanson M, et al. Cognitive deterioration and functional compensation in ALS measured with fMRI using an inhibitory task. J Neurosci. (2014) 34:14260–71. doi: 10.1523/JNEUROSCI.1111-14.2014
58. Keller J, Böhm S, Aho-Özhan HEA, Loose M, Gorges M, Kassubek J, et al. Functional reorganization during cognitive function tasks in patients with amyotrophic lateral sclerosis. Brain Imaging Behav. (2018) 12:771–84. doi: 10.1007/s11682-017-9738-3
59. Passamonti L, Fera F, Tessitore A, Russo A, Cerasa A, Gioia CM, et al. Dysfunctions within limbic-motor networks in amyotrophic lateral sclerosis. Neurobiol Aging (2013) 34:2499–509. doi: 10.1016/j.neurobiolaging.2013.05.016
60. Aho-Özhan HEA, Keller J, Heimrath J, Uttner I, Kassubek J, Birbaumer N, et al. Perception of emotional facial expressions in amyotrophic lateral sclerosis (ALS) at behavioural and brain metabolic level. PLoS ONE (2016) 11:e0164655. doi: 10.1371/journal.pone.0164655
61. Mohammadi B, Kollewe K, Cole DM, Fellbrich A, Heldmann M, Samii A, et al. Amyotrophic lateral sclerosis affects cortical and subcortical activity underlying motor inhibition and action monitoring. Hum Brain Mapp. (2015) 36:2878–89. doi: 10.1002/hbm.22814
62. Turner MR, Grosskreutz J, Kassubek J, Abrahams S, Agosta F, Benatar M, et al. Towards a neuroimaging biomarker for amyotrophic lateral sclerosis. Lancet Neurol. (2011) 10:400–3. doi: 10.1016/S1474-4422(11)70049-7
63. Müller H-P, Turner MR, Grosskreutz J, Abrahams S, Bede P, Govind V, et al. A large-scale multicentre cerebral diffusion tensor imaging study in amyotrophic lateral sclerosis. J Neurol Neurosurg Psychiatry (2016) 87:570–9. doi: 10.1136/jnnp-2015-311952
64. Dansereau C, Benhajali Y, Risterucci C, Pich EM, Orban P, Arnold D, et al. Statistical power and prediction accuracy in multisite resting-state fMRI connectivity. Neuroimage (2017) 149:220–32. doi: 10.1016/J.NEUROIMAGE.2017.01.072
65. Agosta F, Canu E, Valsasina P, Riva N, Prelle A, Comi G, et al. Divergent brain network connectivity in amyotrophic lateral sclerosis. Neurobiol Aging (2013) 34:419–27. doi: 10.1016/j.neurobiolaging.2012.04.015
66. Mohammadi B, Kollewe K, Samii A, Krampfl K, Dengler R, Münte TF. Changes of resting state brain networks in amyotrophic lateral sclerosis. Exp Neurol. (2009) 217:147–53. doi: 10.1016/j.expneurol.2009.01.025
67. Welsh RC, Jelsone-Swain LM, Foerster BR. The utility of independent component analysis and machine learning in the identification of the amyotrophic lateral sclerosis diseased brain. Front Hum Neurosci. (2013) 7:251. doi: 10.3389/fnhum.2013.00251
68. Li W, Zhang J, Zhou C, Hou W, Hu J, Feng H, et al. Abnormal functional connectivity density in amyotrophic lateral sclerosis. Front Aging Neurosci. (2018) 10:215. doi: 10.3389/fnagi.2018.00215
69. Zhou C, Hu X, Hu J, Liang M, Yin X, Chen L, et al. Altered brain network in amyotrophic lateral sclerosis: a resting graph theory-based network study at voxel-wise level. Front Neurosci. (2016) 10:204. doi: 10.3389/fnins.2016.00204
70. Sako W, Abe T, Izumi Y, Yamazaki H, Matsui N, Harada M, et al. Spontaneous brain activity in the sensorimotor cortex in amyotrophic lateral sclerosis can be negatively regulated by corticospinal fiber integrity. Neurol Sci. (2017) 38:755–60. doi: 10.1007/s10072-017-2831-y
71. Luo C, Chen Q, Huang R, Chen X, Chen K, Huang X, et al. Patterns of spontaneous brain activity in amyotrophic lateral sclerosis: a resting-state fMRI study. PLoS ONE (2012) 7:e45470. doi: 10.1371/journal.pone.0045470
72. Trojsi F, Di Nardo F, Santangelo G, Siciliano M, Femiano C, Passaniti C, et al. Resting state fMRI correlates of theory of mind impairment in amyotrophic lateral sclerosis. Cortex (2017) 97:1–16. doi: 10.1016/j.cortex.2017.09.016
73. Zhou F, Xu R, Dowd E, Zang Y, Gong H, Wang Z. Alterations in regional functional coherence within the sensory-motor network in amyotrophic lateral sclerosis. Neurosci Lett. (2014) 558:192–6. doi: 10.1016/j.neulet.2013.11.022
74. Zhang J, Ji B, Hu J, Zhou C, Li L, Li Z, et al. Aberrant interhemispheric homotopic functional and structural connectivity in amyotrophic lateral sclerosis. J Neurol Neurosurg Psychiatry (2017) 88:369.1–70. doi: 10.1136/jnnp-2016-314567
75. Chenji S, Jha S, Lee D, Brown M, Seres P, Mah D, et al. Investigating default mode and sensorimotor network connectivity in amyotrophic lateral sclerosis. PLoS ONE (2016) 11:e0157443. doi: 10.1371/journal.pone.0157443
76. Fekete T, Zach N, Mujica-Parodi LR, Turner MR. Multiple kernel learning captures a systems-level functional connectivity biomarker signature in amyotrophic lateral sclerosis. PLoS ONE (2013) 8:e85190. doi: 10.1371/journal.pone.0085190
77. Schmidt R, Verstraete E, de Reus MA, Veldink JH, van den Berg LH, van den Heuvel MP. Correlation between structural and functional connectivity impairment in amyotrophic lateral sclerosis. Hum Brain Mapp. (2014) 35:4386–95. doi: 10.1002/hbm.22481
78. Schuster C, Elamin M, Hardiman O, Bede P. Presymptomatic and longitudinal neuroimaging in neurodegeneration–from snapshots to motion picture: a systematic review. J Neurol Neurosurg Psychiatry (2015) 86:1089–96. doi: 10.1136/jnnp-2014-309888
79. Menke RAL, Proudfoot M, Wuu J, Andersen PM, Talbot K, Benatar M, et al. Increased functional connectivity common to symptomatic amyotrophic lateral sclerosis and those at genetic risk. J Neurol Neurosurg Psychiatry (2016) 87:580–8. doi: 10.1136/jnnp-2015-311945
80. Lee SE, Sias AC, Mandelli ML, Brown JA, Brown AB, Khazenzon AM, et al. Network degeneration and dysfunction in presymptomatic C9ORF72 expansion carriers. NeuroImage Clin. (2017) 14:286–97. doi: 10.1016/j.nicl.2016.12.006
81. Verstraete E, Turner MR, Grosskreutz J, Filippi M, Benatar M. Mind the gap: The mismatch between clinical and imaging metrics in ALS. Amyotroph Lateral Scler Front Degener. (2015) 16:524–9. doi: 10.3109/21678421.2015.1051989
82. Zhou F, Gong H, Li F, Zhuang Y, Zang Y, Xu R, et al. Altered motor network functional connectivity in amyotrophic lateral sclerosis: a resting-state functional magnetic resonance imaging study. Neuroreport (2013) 24:657–62. doi: 10.1097/WNR.0b013e328363148c
83. Geevasinga N, Korgaonkar MS, Menon P, Van den Bos M, Gomes L, Foster S, et al. Brain functional connectome abnormalities in amyotrophic lateral sclerosis are associated with disability and cortical hyperexcitability. Eur J Neurol. (2017) 24:1507–17. doi: 10.1111/ene.13461
84. Jelsone-Swain LM, Fling BW, Seidler RD, Hovatter R, Gruis K, Welsh RC. Reduced interhemispheric functional connectivity in the motor cortex during rest in limb-onset amyotrophic lateral sclerosis. Front Syst Neurosci. (2010) 4:158. doi: 10.3389/fnsys.2010.00158
85. Verstraete E, van den Heuvel MP, Veldink JH, Blanken N, Mandl RC, Hulshoff Pol HE, et al. Motor network degeneration in amyotrophic lateral sclerosis: a structural and functional connectivity study. PLoS ONE (2010) 5:e13664. doi: 10.1371/journal.pone.0013664
86. Proudfoot M, Jones A, Talbot K, Al-Chalabi A, Turner MR. The ALSFRS as an outcome measure in therapeutic trials and its relationship to symptom onset. Amyotroph Lateral Scler Frontotemporal Degener. (2016) 17:414–25. doi: 10.3109/21678421.2016.1140786
87. Fries P. Rhythms for cognition: communication through coherence. Neuron (2015) 88:220–35. doi: 10.1016/j.neuron.2015.09.034
88. Voytek B, Knight RT. Dynamic network communication as a unifying neural basis for cognition, development, aging, and disease. Biol Psychiatry (2015) 77:1089–97. doi: 10.1016/j.biopsych.2015.04.016
89. Lopes da Silva F. EEG and MEG: relevance to neuroscience. Neuron (2013) 80:1112–28. doi: 10.1016/j.neuron.2013.10.017
90. Proudfoot M, Woolrich MW, Nobre AC, Turner MR. Magnetoencephalography. Pract Neurol. (2014) 14:336–43. doi: 10.1136/practneurol-2013-000768
91. Hall EL, Robson SE, Morris PG, Brookes MJ. The relationship between MEG and fMRI. Neuroimage (2014) 102:80–91. doi: 10.1016/j.neuroimage.2013.11.005
92. Gross J, Baillet S, Barnes GR, Henson RN, Hillebrand A, Jensen O, et al. Good practice for conducting and reporting MEG research. Neuroimage (2013) 65:349–63. doi: 10.1016/j.neuroimage.2012.10.001
93. Hari R, Baillet S, Barnes G, Burgess R, Forss N, Gross J, et al. IFCN-endorsed practical guidelines for clinical magnetoencephalography (MEG). Clin Neurophysiol. (2018) 129:1720–47. doi: 10.1016/j.clinph.2018.03.042
94. van Diessen E, Numan T, van Dellen E, van der Kooi AW, Boersma M, Hofman D, et al. Opportunities and methodological challenges in EEG and MEG resting state functional brain network research. Clin Neurophysiol. (2015) 126:1468–81. doi: 10.1016/j.clinph.2014.11.018
95. Bosch EP, Yamada T, Kimura J. Somatosensory evoked potentials in motor neuron disease. Muscle Nerve (1985) 8:556–62. doi: 10.1002/mus.880080703
96. Ghezzi A, Mazzalovo E, Locatelli C, Zibetti A, Zaffaroni M, Montanini R. Multimodality evoked potentials in amyotrophic lateral sclerosis. Acta Neurol Scand. (1989) 79:353–6. doi: 10.1111/j.1600-0404.1989.tb03799.x
97. Radtke RA, Erwin A, Erwin CW. Abnormal sensory evoked potentials in amyotrophic lateral sclerosis. Neurology (1986) 36:796–801. doi: 10.1212/WNL.36.6.796
98. Cosi V, Poloni M, Mazzini L, Callieco R. Somatosensory evoked potentials in amyotrophic lateral sclerosis. J Neurol Neurosurg Psychiatry (1984) 47:857–61. doi: 10.1136/jnnp.47.8.857
99. Subramaniam JS, Yiannikas C. Multimodality evoked potentials in motor neuron disease. Arch Neurol. (1990) 47:989–94. doi: 10.1001/archneur.1990.00530090063014
100. Soares JJF, Lawoko S, Nolan P. The nature, extent and determinants of violence against psychiatric personnel. Work Stress (2000) 14:105–20. doi: 10.1080/026783700750051630
101. Iglesias C, Sangari S, El Mendili M-M, Benali H, Marchand-Pauvert V, Pradat P-F. Electrophysiological and spinal imaging evidences for sensory dysfunction in amyotrophic lateral sclerosis. BMJ Open (2015) 5:e007659. doi: 10.1136/bmjopen-2015-007659
102. Sangari S, Iglesias C, El Mendili MM, Benali H, Pradat PF, Marchand-Pauvert V. Impairment of sensory-motor integration at spinal level in amyotrophic lateral sclerosis. Clin Neurophysiol. (2016) 127:1968–77. doi: 10.1016/j.clinph.2016.01.014
103. Raggi A, Iannaccone S, Cappa SF. Event-related brain potentials in amyotrophic lateral sclerosis: a review of the international literature. Amyotroph Lateral Scler. (2010) 11:16–26. doi: 10.3109/17482960902912399
104. Westphal KP, Heinemann HA, Grözinger B, Kotchoubey BJ, Diekmann V, Becker W, et al. Bereitschaftspotential in amyotrophic lateral sclerosis (ALS): lower amplitudes in patients with hyperreflexia (spasticity). Acta Neurol Scand. (1998) 98:15–21. doi: 10.1111/j.1600-0404.1998.tb07372.x
105. Thorns J, Wieringa BM, Mohammadi B, Hammer A, Dengler R, Münte TF. Movement initiation and inhibition are impaired in amyotrophic lateral sclerosis. Exp Neurol. (2010) 224:389–94. doi: 10.1016/j.expneurol.2010.04.014
106. Bizovičar N, Koritnik B, Zidar I, Dreo J, Zidar J. Movement-related cortical potentials in ALS increase at lower and decrease at higher upper motor neuron burden scores. Amyotroph Lateral Scler Frontotemporal Degener. (2013) 14:380–9. doi: 10.3109/21678421.2012.760604
107. Inuggi A, Riva N, González-Rosa JJ, Amadio S, Amato N, Fazio R, et al. Compensatory movement-related recruitment in amyotrophic lateral sclerosis patients with dominant upper motor neuron signs: an EEG source analysis study. Brain Res. (2011) 1425:37–46. doi: 10.1016/j.brainres.2011.09.007
108. Gu Y, Farina D, Murguialday AR, Dremstrup K, Birbaumer N. Comparison of movement related cortical potential in healthy people and amyotrophic lateral sclerosis patients. Front Neurosci. (2013) 7:65. doi: 10.3389/fnins.2013.00065
109. Pfurtscheller G, Lopes Da Silva FH. Event-related EEG/MEG synchronization and desynchronization: basic principles. Clin Neurophysiol. (1999) 110:1842–57. doi: 10.1016/S1388-2457(99)00141-8
110. Vansteensel MJ, Pels EGM, Bleichner MG, Branco MP, Denison T, Freudenburg ZV, et al. Fully implanted brain–computer interface in a locked-in patient with ALS. N Engl J Med. (2016) 375:2060–6. doi: 10.1056/NEJMoa1608085
111. Kilavik BE, Zaepffel M, Brovelli A, MacKay WA, Riehle A. The ups and downs of β oscillations in sensorimotor cortex. Exp Neurol. (2013) 245:15–26. doi: 10.1016/j.expneurol.2012.09.014
112. Chen R, Yaseen Z, Cohen LG, Hallett M. Time course of corticospinal excitability in reaction time and self-paced movements. Ann Neurol. (1998) 44:317–25. doi: 10.1002/ana.410440306
113. Stančák A, Riml A, Pfurtscheller G. The effects of external load on movement-related changes of the sensorimotor EEG rhythms. Electroencephalogr Clin Neurophysiol. (1997) 102:495–504. doi: 10.1016/S0013-4694(96)96623-0
114. Toma K, Mima T, Matsuoka T, Gerloff C, Ohnishi T, Koshy B, et al. Movement rate effect on activation and functional coupling of motor cortical areas. J Neurophysiol. (2002) 88:3377–85. doi: 10.1152/jn.00281.2002
115. Hummel F, Kirsammer R, Gerloff C. Ipsilateral cortical activation during finger sequences of increasing complexity: representation of movement difficulty or memory load? Clin Neurophysiol. (2003) 114:605–13. doi: 10.1016/S1388-2457(02)00417-0
116. Hall SD, Stanford IM, Yamawaki N, McAllister CJ, Rönnqvist KC, Woodhall GL, et al. The role of GABAergic modulation in motor function related neuronal network activity. Neuroimage (2011) 56:1506–10. doi: 10.1016/j.neuroimage.2011.02.025
117. Muthukumaraswamy SD, Myers JFM, Wilson SJ, Nutt DJ, Lingford-Hughes A, Singh KD, et al. The effects of elevated endogenous GABA levels on movement-related network oscillations. Neuroimage (2012) 66C:36–41. doi: 10.1016/j.neuroimage.2012.10.054
118. Little S, Pogosyan A, Kuhn AA, Brown P. Beta band stability over time correlates with Parkinsonian rigidity and bradykinesia. Exp Neurol. (2012) 236:383–8. doi: 10.1016/j.expneurol.2012.04.024
119. Riva N, Falini A, Inuggi A, Gonzalez-Rosa JJ, Amadio S, Cerri F, et al. Cortical activation to voluntary movement in amyotrophic lateral sclerosis is related to corticospinal damage: electrophysiological evidence. Clin Neurophysiol. (2012) 123:1586–92. doi: 10.1016/j.clinph.2011.12.013
120. Bizovičar N, Dreo J, Koritnik B, Zidar J. Decreased movement-related beta desynchronization and impaired post-movement beta rebound in amyotrophic lateral sclerosis. Clin Neurophysiol. (2014) 125:1689–99. doi: 10.1016/j.clinph.2013.12.108
121. Cheyne DO. MEG studies of sensorimotor rhythms: a review. Exp Neurol. (2013) 245:27–39. doi: 10.1016/j.expneurol.2012.08.030
122. Proudfoot M, Rohenkohl G, Quinn A, Colclough GL, Wuu J, Talbot K, et al. Altered cortical beta-band oscillations reflect motor system degeneration in amyotrophic lateral sclerosis. Hum Brain Mapp. (2017) 38:237–54. doi: 10.1002/hbm.23357
123. Vucic S, Ziemann U, Eisen A, Hallett M, Kiernan MC. Transcranial magnetic stimulation and amyotrophic lateral sclerosis: pathophysiological insights. J Neurol Neurosurg Psychiatry (2013) 84:1161–70. doi: 10.1136/jnnp-2012-304019
124. Turner MR, Hammers A, Al-Chalabi A, Shaw CE, Andersen PM, Brooks DJ, et al. Distinct cerebral lesions in sporadic and “D90A” SOD1 ALS: studies with [11C]flumazenil PET. Brain (2005) 128:1323–9. doi: 10.1093/brain/awh509
125. Baker SN, Kilner JM, Pinches EM, Lemon RN. The role of synchrony and oscillations in the motor output. Exp. Brain Res. 128, 109–17. doi: 10.1007/s002210050825
126. Proudfoot M, van Ede F, Quinn A, Colclough GL, Wuu J, Talbot K, et al. Impaired corticomuscular and interhemispheric cortical beta oscillation coupling in amyotrophic lateral sclerosis. Clin Neurophysiol. (2018) 129:1479–89. doi: 10.1016/j.clinph.2018.03.019
127. Kristeva R, Patino L, Omlor W. Beta-range cortical motor spectral power and corticomuscular coherence as a mechanism for effective corticospinal interaction during steady-state motor output. Neuroimage (2007) 36:785–92. doi: 10.1016/j.neuroimage.2007.03.025
128. Fisher KM, Zaaimi B, Williams TL, Baker SN, Baker MR. Beta-band intermuscular coherence: a novel biomarker of upper motor neuron dysfunction in motor neuron disease. Brain (2012) 135:2849–64. doi: 10.1093/brain/aws150
129. Hanagasi HA, Gurvit IH, Ermutlu N, Kaptanoglu G, Karamursel S, Idrisoglu HA, et al. Cognitive impairment in amyotrophic lateral sclerosis: evidence from neuropsychological investigation and event-related potentials. Cogn Brain Res. (2002) 14:234–44. doi: 10.1016/S0926-6410(02)00110-6
130. Pekkonen E, Osipova D, Laaksovirta H. Magnetoencephalographic evidence of abnormal auditory processing in amyotrophic lateral sclerosis with bulbar signs. Clin Neurophysiol. (2004) 115:309–15. doi: 10.1016/S1388-2457(03)00360-2
131. Raggi A, Consonni M, Iannaccone S, Perani D, Zamboni M, Sferrazza B, et al. Auditory event-related potentials in non-demented patients with sporadic amyotrophic lateral sclerosis. Clin Neurophysiol. (2008) 119:342–50. doi: 10.1016/j.clinph.2007.10.010
132. Mannarelli D, Pauletti C, Locuratolo N, Vanacore N, Frasca V, Trebbastoni A, et al. Attentional processing in bulbar- and spinal-onset amyotrophic lateral sclerosis: insights from event-related potentials. Amyotroph Lateral Scler Frontotemporal Degener. (2014) 15:30–8. doi: 10.3109/21678421.2013.787628
133. Volpato C, Piccione F, Silvoni S, Cavinato M, Palmieri A, Meneghello F, et al. Working memory in amyotrophic lateral sclerosis: auditory event-related potentials and neuropsychological evidence. J Clin Neurophysiol. (2010) 27:198–206. doi: 10.1097/WNP.0b013e3181e0aa14
134. Zaehle T, Becke A, Naue N, Machts J, Abdulla S, Petri S, et al. Working memory in ALS patients: preserved performance but marked changes in underlying neuronal networks. PLoS ONE (2013) 8:e71973. doi: 10.1371/journal.pone.0071973
135. Pinkhardt EH, Jürgens R, Becker W, Mölle M, Born J, Ludolph AC, et al. Signs of impaired selective attention in patients with amyotrophic lateral sclerosis. J Neurol. (2008) 255:532–8. doi: 10.1007/s00415-008-0734-9
136. Volpato C, Prats Sedano MA, Silvoni S, Segato N, Cavinato M, Merico A, et al. Selective attention impairment in amyotrophic lateral sclerosis. Amyotroph Lateral Scler Front Degener. (2016) 17:236–44. doi: 10.3109/21678421.2016.1143514
137. Seer C, Fürkötter S, Vogts M-B, Lange F, Abdulla S, Dengler R, et al. Executive dysfunctions and event-related brain potentials in patients with amyotrophic lateral sclerosis. Front Aging Neurosci. (2015) 7:225. doi: 10.3389/fnagi.2015.00225
138. Seer C, Joop M, Lange F, Lange C, Dengler R, Petri S, et al. Attenuated error-related potentials in amyotrophic lateral sclerosis with executive dysfunctions. Clin Neurophysiol. (2017) 128:1496–503. doi: 10.1016/j.clinph.2017.05.007
139. Goldstein LH, Abrahams S. Changes in cognition and behaviour in amyotrophic lateral sclerosis: nature of impairment and implications for assessment. Lancet Neurol. (2013) 12:368–80. doi: 10.1016/S1474-4422(13)70026-7
140. Lange F, Lange C, Joop M, Seer C, Dengler R, Kopp B, et al. Neural correlates of cognitive set shifting in amyotrophic lateral sclerosis. Clin Neurophysiol. (2016) 127:3537–45. doi: 10.1016/j.clinph.2016.09.019
141. Blain-Moraes S, Mashour GA, Lee H, Huggins JE, Lee U. Altered cortical communication in amyotrophic lateral sclerosis. Neurosci Lett. (2013) 543:172–6. doi: 10.1016/j.neulet.2013.03.028
142. Gallassi R, Montagna P, Morreale A, Lorusso S, Tinuper P, Daidone R, et al. Neuropsychological, electroencephalogram and brain computed tomography findings in motor neuron disease. Eur Neurol. (1989) 29:115–20. doi: 10.1159/000116391
143. Mai R, Facchetti D, Micheli A, Poloni M. Quantitative electroencephalography in amyotrophic lateral sclerosis. Electroencephalogr Clin Neurophysiol. (1998) 106:383–6. doi: 10.1016/S0013-4694(97)00159-4
144. Santhosh J, Bhatia M, Sahu S, Anand S. Decreased electroencephalogram alpha band [8-13 Hz] power in amyotrophic lateral sclerosis patients: a study of alpha activity in an awake relaxed state. Neurol India (2005) 53:99–101. doi: 10.4103/0028-3886.15071
145. Jayaram V, Widmann N, Förster C, Fomina T, Hohmann M, Müller Vom Hagen J, et al. Brain-computer interfacing in amyotrophic lateral sclerosis: Implications of a resting-state EEG analysis. Conf Proc. (2015) 2015:6979–82. doi: 10.1109/EMBC.2015.7319998
146. Kopell NJ, Gritton HJ, Whittington MA, Kramer MA. Beyond the connectome: the dynome. Neuron (2014) 83:1319–28. doi: 10.1016/j.neuron.2014.08.016
147. Iyer PM, Egan C, Pinto-Grau M, Burke T, Elamin M, Nasseroleslami B, et al. Functional connectivity changes in resting-state EEG as potential biomarker for amyotrophic lateral sclerosis. PLoS ONE (2015) 10:e0128682. doi: 10.1371/journal.pone.0128682
148. Nasseroleslami B, Dukic S, Broderick M, Mohr K, Schuster C, Gavin B, et al. Characteristic increases in EEG connectivity correlate with changes of structural MRI in amyotrophic lateral sclerosis. Cereb Cortex (2017) 29:27–41. doi: 10.1093/cercor/bhx301
149. Fraschini M, Demuru M, Hillebrand A, Cuccu L, Porcu S, Di Stefano F, et al. EEG functional network topology is associated with disability in patients with amyotrophic lateral sclerosis. Sci Rep. (2016) 6:38653. doi: 10.1038/srep38653
150. Fraschini M, Lai M, Demuru M, Puligheddu M, Floris G, Borghero G, et al. Functional brain connectivity analysis in amyotrophic lateral sclerosis: an EEG source-space study. Biomed Phys Eng Express (2018) 4:037004. doi: 10.1088/2057-1976/aa9c64
151. Proudfoot M, Colclough GL, Quinn A, Wuu J, Talbot K, Benatar M, et al. Increased cerebral functional connectivity in ALS: a resting-state magnetoencephalography study. Neurology (2018) 90:e1418–24. doi: 10.1212/WNL.0000000000005333
152. Loewe K, Machts J, Kaufmann J, Petri S, Heinze H-J, Borgelt C, et al. Widespread temporo-occipital lobe dysfunction in amyotrophic lateral sclerosis. Sci Rep. (2017) 7:40252. doi: 10.1038/srep40252
153. Höller Y, Uhl A, Bathke A, Thomschewski A, Butz K, Nardone R, et al. Reliability of EEG measures of interaction: a paradigm shift is needed to fight the reproducibility crisis. Front Hum Neurosci. (2017) 11:1–15. doi: 10.3389/fnhum.2017.00441
154. Sorrentino P, Rucco R, Jacini F, Trojsi F, Lardone A, Baselice F, et al. Brain functional networks become more connected as amyotrophic lateral sclerosis progresses: a source level magnetoencephalographic study. NeuroImage Clin. (2018) 20:564–71. doi: 10.1016/j.nicl.2018.08.001
155. Cope TE, Sohoglu E, Sedley W, Patterson K, Jones PS, Wiggins J, et al. Evidence for causal top-down frontal contributions to predictive processes in speech perception. Nat Commun. (2017) 8:2154. doi: 10.1038/s41467-017-01958-7
156. Baillet S. Magnetoencephalography for brain electrophysiology and imaging. Nat Neurosci. (2017) 20:327–39. doi: 10.1038/nn.4504
157. Boto E, Holmes N, Leggett J, Roberts G, Shah V, Meyer SS, et al. Moving magnetoencephalography towards real-world applications with a wearable system. Nature (2018) 555:657–61. doi: 10.1038/nature26147
158. Bede P, Bokde ALW, Byrne S, Elamin M, Fagan AJ, Hardiman O. Spinal cord markers in ALS: diagnostic and biomarker considerations. Amyotroph Lateral Scler. (2012) 13:407–15. doi: 10.3109/17482968.2011.649760
159. Lawrence J, Stroman PW, Malisza KL. Functional MRI of the cervical spinal cord during noxious and innocuous thermal stimulation in the alpha-chloralose- and halothane-anesthetized rat. Magn Reson Imaging (2008) 26:1–10. doi: 10.1016/j.mri.2007.05.001
160. Malisza KL, Stroman PW. Functional imaging of the rat cervical spinal cord. J Magn Reson Imaging (2002) 16:553–8. doi: 10.1002/jmri.10185
161. Stroman PW, Krause V, Malisza KL, Frankenstein UN, Tomanek B. Functional magnetic resonance imaging of the human cervical spinal cord with stimulation of different sensory dermatomes. Magn Reson Imaging (2002) 20:1–6. doi: 10.1016/S0730-725X(02)00468-X
162. Kornelsen J, Stroman PW. fMRI of the lumbar spinal cord during a lower limb motor task. Magn Reson Med. (2004) 52:411–4. doi: 10.1002/mrm.20157
163. Agosta F, Valsasina P, Caputo D, Stroman PW, Filippi M. Tactile-associated recruitment of the cervical cord is altered in patients with multiple sclerosis. Neuroimage (2008) 39:1542–8. doi: 10.1016/J.NEUROIMAGE.2007.10.048
164. Stroman PW, Kornelsen J, Bergman A, Krause V, Ethans K, Malisza KL, et al. Noninvasive assessment of the injured human spinal cord by means of functional magnetic resonance imaging. Spinal Cord. (2004) 42:59–66. doi: 10.1038/sj.sc.3101559
165. Eippert F, Kong Y, Winkler AM, Andersson JL, Finsterbusch J, Büchel C, et al. Investigating resting-state functional connectivity in the cervical spinal cord at 3T. Neuroimage (2017) 147:589–601. doi: 10.1016/j.neuroimage.2016.12.072
166. Zhang Y, Fang T, Wang Y, Guo X, Alarefi A, Wang J, et al. Occipital cortical gyrification reductions associate with decreased functional connectivity in amyotrophic lateral sclerosis. Brain Imaging Behav. (2017) 11:1–7. doi: 10.1007/s11682-015-9499-9
Keywords: amyotrophic lateral sclerosis, motor neurone disease, biomarker, neuroimaging, neurophysiology, cortex
Citation: Proudfoot M, Bede P and Turner MR (2019) Imaging Cerebral Activity in Amyotrophic Lateral Sclerosis. Front. Neurol. 9:1148. doi: 10.3389/fneur.2018.01148
Received: 15 October 2018; Accepted: 11 December 2018;
Published: 08 January 2019.
Edited by:
Francesca Trojsi, Università Degli Studi Della Campania “Luigi Vanvitelli” Naples, ItalyReviewed by:
Edoardo Gioele Spinelli, Vita-Salute San Raffaele University, ItalyRodolfo Gabriel Gatto, University of Illinois at Chicago, United States
Copyright © 2019 Proudfoot, Bede and Turner. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Martin R. Turner, bWFydGluLnR1cm5lckBuZGNuLm94LmFjLnVr
 Malcolm Proudfoot
Malcolm Proudfoot Peter Bede
Peter Bede Martin R. Turner1,3*
Martin R. Turner1,3*