- 1Department of Neurology, University Hospital Knappschaftskrankenhaus Bochum, Ruhr University Bochum, Bochum, Germany
- 2Institute of Radiology, Neuroradiology and Nuclear Medicine, University Hospital Knappschaftskrankenhaus Bochum, Ruhr University Bochum, Bochum, Germany
- 3Institute of Diagnostic and Interventional Radiology, St. Marien-Hospital Hamm, Hamm, Germany
Cardiac embolism is presumed to cause a significant portion of cryptogenic strokes. Transesophageal echocardiography may detect intracardiac thrombi, but this remains a rare finding, possibly because remnant clots dissolve spontaneously or following thrombolysis. Cardiac imaging within cerebral CT angiography might offer an alternative method for thrombus detection within hyperacute stroke assessment. In a proof-of-concept study we analyzed records of patients aged ≥ 60 years that presented with suspected stroke and underwent extended cerebral CT angiography as part of their emergency assessment. CT imaging of patients with ischemic stroke or transient ischemic attack (TIA) and atrial fibrillation and of those with embolic strokes of undetermined source (ESUS) was reviewed for intracardiac clots and other cardiac or aortic pathology. Over a period of 3 months 59 patients underwent extended CT angiography for suspected stroke, 44 of whom received a final diagnosis of ischemic stroke or TIA. Of those, 17 had atrial fibrillation, and four fulfilled ESUS criteria. Thrombi were detected within atrial structures on CT angiography in three cases. In two ESUS patients complex atheromatosis of the proximal ascending aorta with irregular and ulcerating plaques was detected. Cardiac imaging within emergency cerebral CT angiography is feasible and can provide valuable diagnostic information in a patient group that might not routinely undergo transesophageal echocardiography. A small change to emergency assessment could potentially uncover cardioembolic pathology in cases that would have remained cryptogenic otherwise.
Introduction
A significant proportion of embolic strokes of undetermined source (ESUS) are probably cardiogenic, but cardioembolic pathologies can elude standard assessments (1). Transesophageal echocardiography (TEE) is the gold standard for detection of emboligenic abnormalities, but it is not routinely performed in all patients for various reasons. Furthermore, the utility of routine TEE in cryptogenic stroke has been questioned, not least because of a low rate of atrial clot detection (2). A reason for this might be that remnant clots can dissolve shortly after stroke onset and prior to TEE; moreover, systemic thrombolysis can dissolve intracardiac thrombi (3).
An alternative method for detecting left atrial thrombi (and other cardiac pathology) is computed tomography (CT) (4), but ECG-triggered contrast-enhanced cardiac CT would unjustifiably delay thrombolysis and/or thrombectomy. However, relevant cardiac abnormalities, including left atrial clots, can be detected on non-cardiac CT (5, 6). Thus, when cerebral CT angiography (CCTA) is performed to detect large artery occlusion or stenosis within emergency stroke assessment, extending the scan range of the angiogram by several inches to include the heart seems feasible.
Scan range of emergency CCTA for acute stroke in elderly patients included the heart for a period of 3 months at our center. The potential for detecting intracardiac clots in patients with large artery occlusion or transient ischemic attack (TIA) and atrial fibrillation as well as ESUS was investigated.
Methods
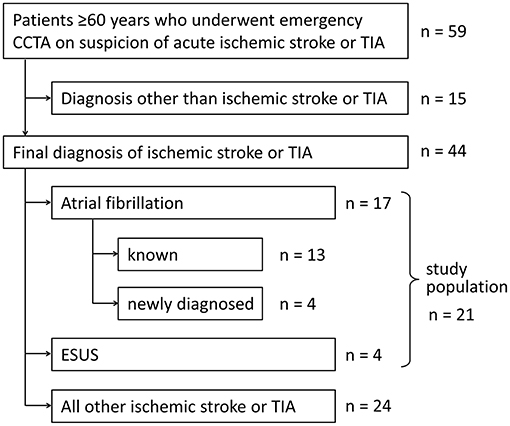
A proof-of-principle study was conducted to assess the feasibility of performing an extended CCTA that includes the heart as well as its potential to detect atrial clots and other emboligenic pathology. At our center emergency CCTA is routinely performed when large artery occlusion or stenosis is suspected as a cause of acute stroke or TIA according to standard protocol as described elsewhere (7). During the period between 30.04.2018 and 03.08.2018, CT technicians had been instructed to set the lower limit of the scan range not to the aortic arch, but to the diaphragm, for any CCTA that was ordered for suspected stroke in patients ≥ 60 years. To increase the pretest probability of atrial thrombi we only analyzed data of acute stroke or TIA patients who had atrial fibrillation or ESUS (Figure 1). Clinical data was collected retrospectively from electronic records of all consecutive patients during the study period who met inclusion criteria. ESUS diagnosis was assigned to patients who fulfilled all diagnostic criteria and had all required assessments (1). For the purpose of this study, findings from CCTA proximal of the aortic arch were not considered for ESUS status. The effective radiation dose was estimated by multiplying the automatically calculated dose length product (DLP) by a standard conversion factor of 0.014 mSv (mGy·cm)−1 (8). This study was approved by the ethics committee of the medical faculty of the Ruhr University Bochum.
Results
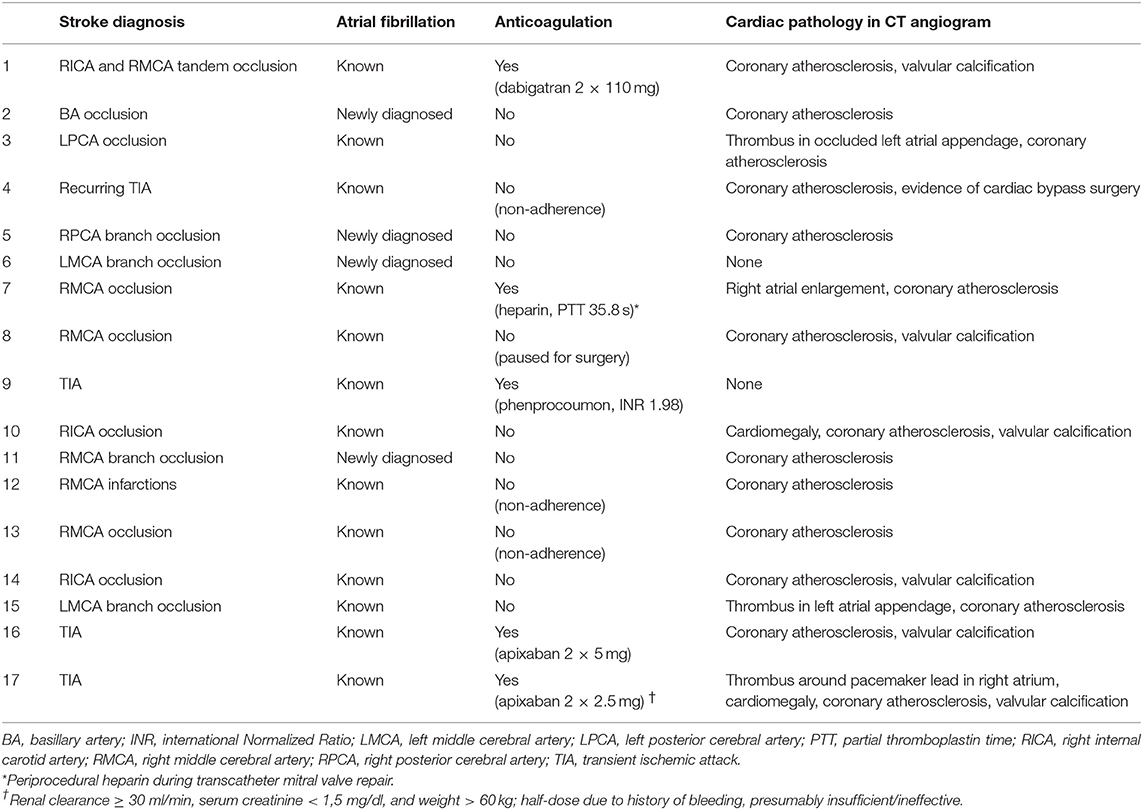
A total of 59 patients underwent emergency CCTA (Figure 2A) for suspected stroke, of which 44 received the final diagnosis ischemic stroke. There were 17 patients who had ischemic stroke or TIA and known or newly diagnosed atrial fibrillation (Table 1; mean age: 77.5 years, standard deviation: 8.4 years; 53% female). As hypothesized, intracardiac thrombi could be visualized: once in an artificially occluded left atrial appendage, once around a pacemaker lead in the right atrium, and once in the left atrial appendage (Figures 2B,C). Four patients were classified as ESUS (Table 2; mean age 80 years, standard deviation 2.7 years; 25% female). Irregular and ulcerating plaques were found in the proximal ascending aorta of two of these patients (Figure 2D).
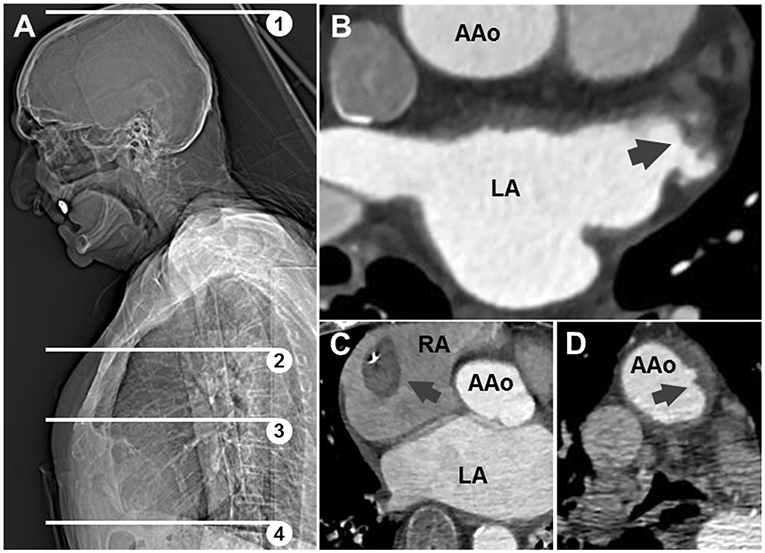
Figure 2. (A) Scout-view showing scan range of conventional (1,2) and extended (1–4) angiography; (3) corresponds to panel (B). Axial CT images: (B) thrombotic material in appendage of left atrium (LA) in patient 15; (C) thrombus around pacemaker lead in right atrium (RA) in patient 17; (D) ulcerating plaque in ascending aorta (AAo) in patient with ESUS.
Median DLP for all 44 ischemic stroke patients was 358 mGy·cm (interquartile range, 305–549 mGy·cm); corresponding radiation doses were thus 5.01 mSv (interquartile range, 4.27–7.69 mSv).
Discussion
Our results demonstrate that co-imaging of cardiac structures within emergency CCTA in elderly patients with ischemic stroke or TIA and atrial fibrillation or ESUS can indeed visualize intracardiac clots before thrombolytic treatment, as well as other emboligenic pathologies such as ulcerating plaques of the ascending aorta.
The main advantage of cardiac imaging within routine CCTA compared to dedicated ECG-gated cardiac CT is the minimal delay of only a few extra seconds, and the sparing of additional contrast agent. A potential disadvantage is lower image quality due to heart movement artifacts and variable contrast filling since bolus timing is optimized for cerebral arterial visualization (9). However, studies suggest that ECG-gating does not markedly improve image quality of the ascending aorta (10) or the left atrium (11). Image quality in this study was adequate in most cases, though larger samples with control imaging would be needed to quantify differences. While TEE remains the gold standard for intracardiac clot detection, it requires specialized expertise and is time-consuming. Performing it in an emergency setting before thrombolysis would introduce an unjustifiable time delay.
The additional radiation exposure during cardiac imaging added to CCTA has to be considered. In our study the median DLP and estimated radiation dose for the entire extended CCTA were less than average for cardiac CT angiography (8). The lifetime risk of radiation-induced fatal cancer for this procedure is <0.05%, added to an average background risk of 21% (12). Although our study design did not allow calculating the added radiation exposure in extended CCTA compared to routine CCTA, the added risk would naturally be even lower. Especially in an elderly population, radiation-associated risks thus can be considered negligible. Moreover, comprehensive risk analysis should also consider periprocedural risks of TEE [0.02% for serious complications (2)] and the risk of cryptogenic stroke recurrence (3–6%/year) (1).
Our study design does not allow estimations of the diagnostic yield of extended CCTA, pre-treatment prevalence of remnant left atrial clots, or direct comparisons with other methods. However, our findings serve as a proof of principle: intracardiac clots can be detected on extended emergency CCTA before they are potentially dissolved by thrombolysis or flushed into the circulation. We looked at patients with known atrial fibrillation to increase pretest probability for cardiac thrombi. However, the biggest clinical benefit of this method would be expected in patients with cryptogenic stroke, where detecting a cardiac thrombus has profound implications for acute treatment (early anticoagulation, follow-up imaging) and secondary prevention (long-term anticoagulation). Prospective multi-center studies should explore this idea in the future. In the long run, a “one-stop shop” multimodal CT program could be developed to optimize emergency stroke diagnostics.
Data Availability
The datasets generated for this study are available on request to the corresponding author.
Ethics Statement
The study protocol was approved by the ethics committee of the Medical Faculty of Ruhr University Bochum. This was a retrospective study based on routine clinical data, so written informed consent was not acquired.
Author Contributions
SP had the idea for the study and served as principal investigator, performed data analysis and wrote the first draft of the manuscript. US and WW contributed to the analysis. IK and JA contributed to clinical and radiological data acquisition and analysis. All authors revised and approved the final manuscript.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
We acknowledge support by the DFG Open Access publication funds of the Ruhr-Universität Bochum.
References
1. Hart RG, Diener HC, Coutts SB, Easton JD, Granger CB, O'Donnell MJ, et al. Embolic strokes of undetermined source: the case for a new clinical construct. Lancet Neurol. (2014) 13:429–38. doi: 10.1016/S1474-4422(13)70310-7
2. McGrath ER, Paikin JS, Motlagh B, Salehian O, Kapral MK, O'Donnell MJ. Transesophageal echocardiography in patients with cryptogenic ischemic stroke: a systematic review. Am Heart J. (2014) 168:706–12. doi: 10.1016/j.ahj.2014.07.025
3. Rose PS, Punjabi NM, Pearse DB. Treatment of right heart thromboemboli. Chest. (2002) 121:806–14. doi: 10.1378/chest.121.3.806
4. Wu X, Wang C, Zhang C, Zhang Y, Ding F, Yan J. Computed tomography for detecting left atrial thrombus: a meta-analysis. Arch Med Sci. (2012) 8:943–51. doi: 10.5114/aoms.2012.32400
5. Kuetting D, Müller A, Feisst A, Luetkens J, Dabir D, Schild HH, et al. Incidental cardiac findings in non-electrocardiogram-gated thoracic computed tomography of intensive care unit patients: assessment of prevalence and underreporting. J Thorac Imaging. (2018) 33:168–75. doi: 10.1097/RTI.0000000000000309
6. Cox M, Balasubramanya R, Hou A, Deshmukh S, Needleman L. Incidental left atrial and ventricular thrombi on routine CT: outcome and influence on subsequent management at an urban tertiary care referral center. Emerg Radiol. (2015) 22:657–60. doi: 10.1007/s10140-015-1342-5
7. Eyding J, Wiebringhaus R, Klein FG, Skodda S, Schlegel U, Alekseyev A, et al. Multimodal CT imaging and recanalizing therapy in acute ischemic stroke: retrospective analysis of a one-year single-center experience. Eur Neurol. (2012) 67:193–9. doi: 10.1159/000334724
8. Hausleiter J, Meyer T, Hermann F, Hadamitzky M, Krebs M, Gerber TC, et al. Estimated radiation dose associated with cardiac CT angiography. JAMA. (2009) 301:500–7. doi: 10.1001/jama.2009.54
9. Husmann L, Valenta I, Gaemperli O, Adda O, Treyer V, Wyss CA, et al. Feasibility of low-dose coronary CT angiography: first experience with prospective ECG-gating. Eur Heart J. (2008) 29:191–7. doi: 10.1093/eurheartj/ehm613
10. Wielandner A, Beitzke D, Schernthaner R, Wolf F, Langenberger C, Stadler A, et al. Is ECG triggering for motion artefact reduction in dual-source CT angiography of the ascending aorta still required with high-pitch scanning? The role of ECG-gating in high-pitch dual-source CT of the ascending aorta. Br J Radiol. (2016) 89:20160174. doi: 10.1259/bjr.20160174
11. Wagner M, Butler C, Rief M, Beling M, Durmus T, Huppertz A, et al. Comparison of non-gated vs. electrocardiogram-gated 64-detector-row computed tomography for integrated electroanatomic mapping in patients undergoing pulmonary vein isolation. Europace. (2010) 12:1090–7. doi: 10.1093/europace/euq135
12. Gerber TC, Carr JJ, Arai AE, Dixon RL, Ferrari VA, Gomes AS, et al. A science advisory from the American Heart Association Committee on Cardiac Imaging of the Council on Clinical Cardiology and Committee on Cardiovascular Imaging and Intervention of the Council on Cardiovascular Radiology and Intervention. Circulation. (2009) 119:1056–65. doi: 10.1161/CIRCULATIONAHA.108.191650
Keywords: angiography, cardioembolic, thrombus, ischemic stroke, atrial fibrillation
Citation: Popkirov S, Schlegel U, Weber W, Kleffner I and Altenbernd J (2019) Cardiac Imaging Within Emergency CT Angiography for Acute Stroke Can Detect Atrial Clots. Front. Neurol. 10:349. doi: 10.3389/fneur.2019.00349
Received: 28 February 2019; Accepted: 21 March 2019;
Published: 10 April 2019.
Edited by:
Guillaume Turc, Centre Hospitalier Sainte-Anne, FranceReviewed by:
Daniel J. Miller, Henry Ford Health System, United StatesGeorge Ntaios, University of Thessaly, Greece
Copyright © 2019 Popkirov, Schlegel, Weber, Kleffner and Altenbernd. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Stoyan Popkirov, c3RveWFuLnBvcGtpcm92QGtrLWJvY2h1bS5kZQ==
†These authors have contributed equally to this work
 Stoyan Popkirov
Stoyan Popkirov Uwe Schlegel
Uwe Schlegel Werner Weber2
Werner Weber2