- 1Balance Disorders Unit, Otolaryngology Department, The Norbert Barlicki Memorial Teaching Hospital, Medical University of Lodz, Lodz, Poland
- 2Department of Otolaryngology, Head and Neck Oncology, The Norbert Barlicki Memorial Teaching Hospital, Medical University of Lodz, Lodz, Poland
This review summarizes current knowledge about the occurrence of hearing and balance disorders after antimalarial drugs treatment. It also examines the clinical applications of antimalarials, their mechanisms behind this ototoxicity and how it can be monitored. It includes studies with larger numbers of patients and those in which auditory function was assessed using audiological tests. Some antimalarials have been repurposed for other conditions like autoimmune disorders, rheumatic diseases, some viral diseases and cancers. While old antimalarial drugs, such as quinoline derivatives, are known to demonstrate ototoxicity, a number of new synthetic antimalarial agents particularly artemisinin derivatives, demonstrate unknown ototoxicity. Adverse audiovestibular effects vary depending on the medication itself, its dose and route of administration, as well as the drug combination, treated disease and individual predispositions of the patient. Dizziness was commonly reported, while vestibular symptoms, hearing loss and tinnitus were observed much less frequently, and most of these symptoms were reversible. As early identification of ototoxic hearing loss is critical to introducing possible alternative treatments with less ototoxic medications, therefore monitoring systems of those drugs ototoxic side effects are much needed.
Introduction
Some medications have ototoxicity effects, that is, they influence the hearing (cochleotoxicity) and/or vestibular (vestibulotoxicity) end organs. The adverse effects (AEs) of ototoxic drugs may cause temporary or permanent hearing loss, tinnitus, dizziness, and vertigo, and may have neurotoxic effects on the auditory and balance systems. The best-known ototoxic drugs are aminoglycoside antibiotics, salicylates, non-steroidal anti-inflammatory drugs, loop diuretics, antimalarials (quinine) and platinum-based cytostatics. Ototoxicity is also reported in patients treated with immunosuppressants (cyclosporin, tacrolimus), antiviral drugs (pegylated and non-pegylated interferons, ribavirin, inhibitors of viral reverse transcriptase—HIV−1 antiretroviral), analgetics (acetaminophen, hydrocodone, methadone), and phosphodiesterase-5 inhibitors (1).
The online databases PubMed (Medline) and Embase (on OVID) were searched for articles published from 1985 to August 2020, with the most recent update on December 2020. The following MeSH terms and keywords were used: ototoxicity, antimalarial drug, antimalarial agent, vestibular symptom, dizziness, vertigo, tinnitus or hearing loss, adverse effect, drugs inducing ototoxicity. The search included publications written in English containing relevant data.
Types of Antimalarial Drugs
Contemporary antimalarial drugs comprise several groups of medications:
- quinoline-type drugs (4-aminoquinolines: chloroquine, hydroxychloroquine, amodiaquine, pyronaridine, piperaquine; 8-aminoquinolines: primaquine, tafenoquine; aryl aminoalcohols: quinine, quinidine, mefloquine, halofantrine, lumefantrine);
- naphthoquinone (Atovaquone);
- antifolates (sulfadoxine-pyrimethamine);
- guanidine derived drugs (proguanil, cycloguanil, chlorproguanil);
- sesquiterpene lactones—artemisinin and its semi-synthetic derivatives (dihydroartemisinin, artemether, artesunate) currently used in artemisinin-based combination therapy (ACT);
- arterolane -a synthetic peroxide-containing compound (2–8).
The WHO recommends five types of ACT for use in children and adults as first and second-line treatment for uncomplicated malaria caused by infection with Plasmodium falciparum:
• artemether plus lumefantrine,
• artesunate plus amodiaquine,
• artesunate plus mefloquine,
• dihydroartemisinin plus piperaquine
• artesunate plus sulfadoxine-pyrimethamine (2).
Artemisinin-based combinations employ a short-acting artemisinin derivative in combination with other complementary compounds (long-acting or with different mechanisms of action) (4). WHO guidelines recommend the use of quinine plus clindamycin for pregnant women in their first trimester, and either ACT or chloroquine for the treatment of uncomplicated malaria caused by other species of Plasmodium (P. vivax, P. ovale, P. malariae, P. knowlesi); however, in case of chloroquine-resistant infections, ACT is used alone (5).
Chloroquine (CQ) and hydroxychloroquine (CHQ) are derivatives of quinine (QN), obtained from the bark of the Peruvian Cinchona tree. QN was first isolated in 1820 by the French scientists Pierre-Joseph Pelletier and Joseph-Bienaimé Caventou (9). CQ was first synthesized in 1934, and hydroxychloroquine sulfate (Plaquenil) was developed in 1946 as a less toxic chloroquine analog (10, 11). Another derivative is Mefloquine (Lariam), which was developed by the United States Army in the 1970s and came into use in the mid-1980s. It is commonly recommended as a prophylaxis for travelers to areas where malaria is endemic (12). Over the years drug-resistant strains of malaria have developed; the first example was found in the 1980s for QN, and since 2006, QN has no longer been recommended as a first-line treatment for malaria.
Artemisinin was discovered in 1972, and dihydroartemisinin in 1973, by Chinese pharmaceutical chemist Tu Youyou, who received the 2015 Nobel Prize in Physiology or Medicine for her discoveries concerning novel therapies against malaria (4). Similarly, Gertrude Elion and George Hitchings (4) also received a Nobel Prize in 1988 for their discovery of pyrimethamine. Some commonly-used antimalarial drugs discussed in the present review are presented in Table 1. Today, often for economic reasons, many antimalarial drugs are manufactured locally in endemic countries under different names (6–8).
Clinical Applications of Antimalarial Drugs
Antimalarials are used not only for the treatment of malaria but also for a range other dermatological, immunological, rheumatological, and severe infectious diseases. In the past, quinine has been used to treat otologic conditions (Menière's disease, herpes zoster, vertigo, purulent otitis media, furuncles in the auditory canal) due to its analgesic as well as ototoxic properties (9). Nowadays, chloroquine and hydroxychloroquine are used to treat autoimmune and connective tissue diseases like systemic lupus erythematosus (SLE), rheumatoid arthritis, Sjögren's syndrome, sarcoidosis, palindromic rheumatism, eosinophilic fasciitis, dermatomyositis, cellulitis, mixed and undifferentiated connective tissue disease (11, 13, 14). They have also demonstrated anticancer properties against different types of cancer (e.g., colon, lung and breast cancer, central nervous system tumors, hematological malignancies), and against cancer cell lines in vitro (11, 15). CQ has also demonstrated antiviral activity, and has been used in clinical trials in infections caused by hepatitis C virus, dengue virus-2, human immunodeficiency virus, and Chikungunya virus (15). Recently, CQ and CHQ were proposed as treatments for severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) infection and were prescribed for almost 12% of COVID-19 patients in Europe (16). The antimalarials have also been used to treat metabolic diseases. Chloroquine shows antihypertensive, antilipidemic, and hypoglycemic effects (11), while artemisinin and its derivatives have been used in the treatment of type 2 diabetes mellitus (17). Currently, CQ and CHQ are being tested for neurological diseases such as neurosarcoidosis, as well as chronic lymphocytic inflammation with pontine perivascular enhancement responsive to corticosteroids, and primary progressive multiple sclerosis (13).
Mechanisms of Ototoxicity
Although they have been studied in animal models, particularly quinine (18), the mechanisms underlying the ototoxicity of antimalarials remain poorly understood. Quinine can negatively affect the auditory system at the level of the central auditory pathway and the auditory periphery. Several hypotheses about changes in cochlea have also been proposed, such as impairment of outer hair cells (OHCs), reduction in the blood flow, and microangiopathy due to quinine-induced thrombocytopenia and disseminated intravascular coagulation (19–21).
Quinine-induced morphological and physiological changes have been observed in the cochlea: in guinea pigs, ultrastructure lesions have been observed, including swellings of the subsurface cisternae and the formation of a central microtubule core in OHCs (22), and high doses of quinine have been found to cause elongation of isolated OHCs followed by contraction (23). Exposure to 5.0 mM QN had the same effect of elongation, but without subsequent relaxation of isolated guinea pig cochlear OHCs, and appeared to reduce active force generation in OHCs without any effect on compliance (24). Recently Davis et al. demonstrated that CQ and CHQ treatment causes damage to hair cells in the zebrafish lateral line while chloroquine causes dose-dependent loss of outer hair cells in cultured neonatal mouse cochlea, with more loss observed in the basal turn than the apical turn, without reduction of supporting cells (25).
In an isolated temporal bone preparation, QN treatment altered the mechanical response of the basilar membrane induced by sound (26). Zheng et al. demonstrated that QN decreased the mean amplitude of the electrically-evoked otoacoustic emissions (OAE), affecting in vivo electromotility of OHCs (27). Dieler et al. report that QN changes the membrane potential of hair cells (hyperpolarization, then depolarization) in a dose-dependent and reversible manner, and causes a diminution of evoked rapid motile responses without any alterations in the turgor, shape or fine structure of OHCs (28). Perilymph perfusion with quinine in guinea pigs was found to result in a reduction of cochlear microphonics and summating potential (at 100 pmol/L), as well as compound action potential input-output function (at all intensities), but no change in endocochlear potential (29). The authors conclude that this lack of change in endocochlear potential excludes the stria vascularis as QN site of action and suggest hearing loss may occur through inhibition of the Ca++-activated K+ channel or an ATP-sensitive K+ channel.
In guinea pigs injected with quinine, the amplitudes of auditory brainstem response (ABR) were reduced at all sound levels but less so at high levels, while distortion product otoacoustic emissions (DPOAE) amplitudes were unchanged at high stimulus levels (30). This could suggest that QN affects both outer and inner hair cells (IHCs) as well as spiral ganglion neurons (SGNs). It has been demonstrated that QN also affects other auditory sites, like the spiral ganglion, and auditory neurons. In adult isolated mice spiral ganglion neurons (SGNs), QN was found to reversibly reduce amplitude and prolong the duration of action potentials; it also blocked the whole-cell potassium and sodium currents, but not calcium currents (31). Quinine modulated the auditory cortex in cats in a different way: it reduced the spontaneous firing rates in the primary auditory cortex and anterior auditory field while increasing them in the secondary auditory cortex (32). Artemisinin derivates (dihydroqinghaosu, artemether, and arteether) may cause necrosis of specific brain stem nuclei in animals, particularly those involved in hearing and balance (33, 34).
There are also few reports about the ototoxicity of other antimalarials. Mefloquine caused dose-dependent damage to hair cells (starting from the cochlear base), supporting cells (prior to hair cells), and SGNs in cultures from postnatal developing rats (35, 36). This mechanism may be due to different apoptotic pathways (the death receptor-mediated signaling pathway or the mitochondrial pathway), as the expression of numerous pro, and anti-apoptotic genes were reported in the cochlear epithelium and SGNs in rat (36, 37). MQ increased oxidative stress in the cochlear hair cells and SGNs, it also induced caspases-3-mediated apoptosis; it is believed that the coenzyme nicotinamide adenine dinucleotide (NAD+) plays a protective role in this process (38). Mefloquine also induced dose-dependent vestibular hair cell loss in the utricle in postnatal rat and the process has been attributed to activation of apoptosis by caspase-8 and caspase-9 (39). Halofantrine affected various cochlear structures, such as IHC, OHCs, SGNs and phalangeal cells in guinea pigs (40). QN also induced tinnitus in the behavioral model of tinnitus in rats (41); however, in another study animals treated with 200 mg/kg/d QN did not exhibit tinnitus-like behavior when assessed using the gap prepulse inhibition of acoustic startle (42).
Interestingly, quinoline drugs have been found to prevent ototoxicity by loop diuretics (furosemide/ethacrynic acid), aminoglycosides, and cisplatin in animal models based on chinchilla and zebrafish (20, 43–45). QN derivatives reduce uptake of gentamicin, neomycin, and cisplatin into hair cells (44, 45). Although the molecular mechanism is unknown, quinine probably acts as a blocker of mechanotransducer (MET) channels for gentamicin (20, 46); however, chloroquine does not appear to reduce MET channel activity (25).
Clinical Manifestations of Ototoxicity
Cochleotoxicity—Hearing Loss and Tinnitus
Antimalarial drugs, such as CQ and HCQ, were considered for a long time as having audio-vestibular side effects (47, 48). QN overdose is generally known as cinchonism. However, audiovestibular toxicity is not a common adverse effect of antimalarials: only 61(2.6%) such complications were reported for all 2339 antimalarial adverse effects listed in the French Pharmacovigilance Network database between January 1986 and December 2010. These complications were nearly equally distributed between CQ and HCQ, 50.8% after therapy for autoimmune diseases and 26.2% in malaria (49). Quinine was not found to have a significant effect on the risk of hearing loss in a study of ototoxic medication use and the 10-year cumulative incidence and progression of hearing loss in 3753 older adults (50). However, this may be due to the low proportion of people using these medications (1.1%) or the fact that they were used for a brief period. In a large group of 19,850 patients who received different regimens containing mefloquine (alone or in combination with artesunate, artemether or sulfadoxine-pyrimethamine) due to uncomplicated malaria, the frequency of dizziness was 47.2% and hearing loss 3.44%; after 28 days of follow-up, these values fell to 6.49 and 0.52% respectively (51).
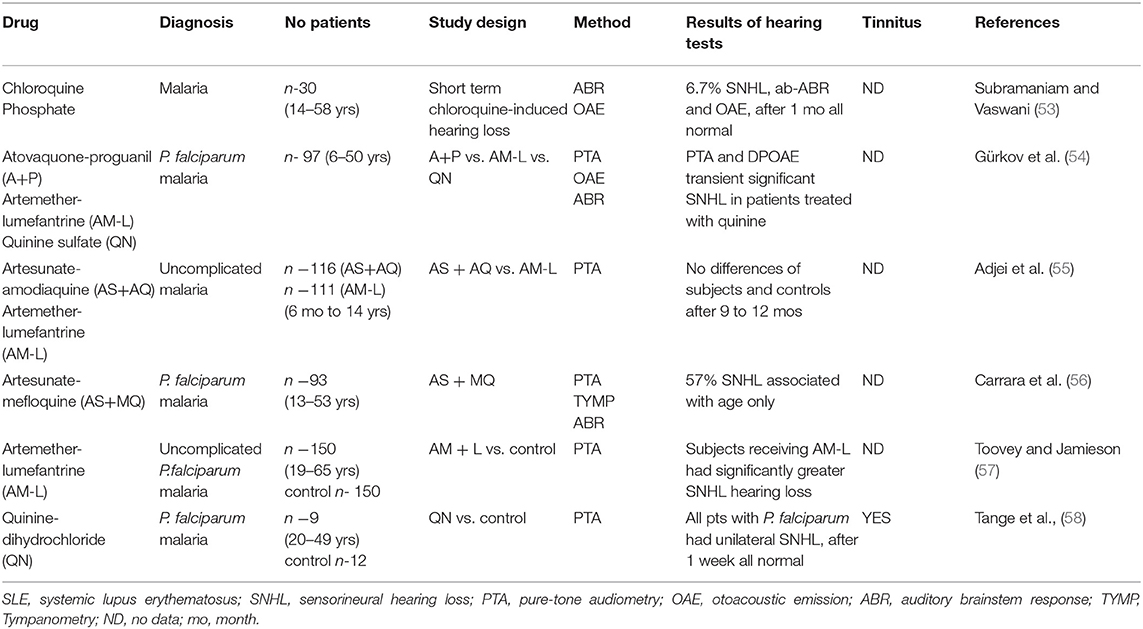
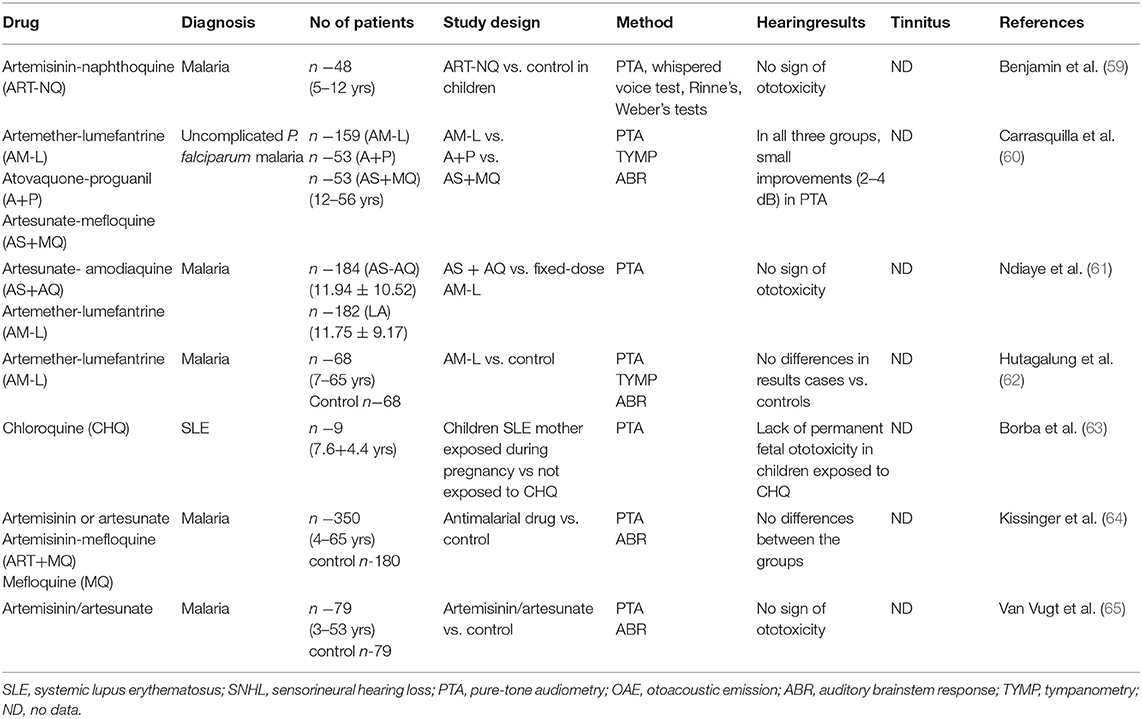
Hearing loss is typically bilateral, mild to moderate sensorineural and mostly reversible (10). Many case reports have described ototoxic effects after treatment with antimalarials, especially CQ and HCQ (52); however, this review focuses on studies based on larger groups of patients. Studies reporting an association between antimalarial use and cochlear ototoxicity are summarized in Table 2, whereas those not finding any such association are listed in Table 3.
The methods used to assess auditory function in the studies varied from subjective to objective audiometry reports. The most common technique was pure tone audiometry, with OAE and ABR being used in children. Most studies used a single hearing test method; however some used three or more. Audiotoxicity manifestations vary depending on the antimalarial drugs. The effects of long-term use of conventional antimalarial drugs like QN have been adequately established. In a study where quinine-dihydrochloride was administered intravenously to patients with malaria caused by P. falciparum, all patients reported severe impaired hearing loss, tinnitus, or a feeling of pressure on the ears. Twenty-four hours later or 1 week later, all audiograms had returned to normal, and the adverse effects disappeared (58). In another study, artemether+lumefantrine (AM-L), atovaquone+proguanil (A+P) and quinine sulfate were compared using a complete audiometric evaluation (PTA, OAE, ABR); it was found that only the patients who were treated with quinine demonstrated a significant but transient SNHL (54). HCQ ototoxicity is less common. Subramaniam and Vaswani found ototoxicity to be infrequent and reversible when chloroquine is administered in regular doses to treat uncomplicated malaria (53).
Artemisinin-based combination therapy (ACT) has contributed remarkably to decrease malaria illnesses and deaths; however, ACTs may affect the auditory system (5, 66). Artesunate+amodiaquine (AS+AQ) and artemether+lumefantrine (AM-L) are two ACT regimens that have been extensively established for the treatment of uncomplicated malaria in Africa (55). Subjects receiving AM-L combination therapy have been found to have a significantly greater risk of SNHL (57). On the other hand, no differences in audiological results were found between a group of 68 patients treated for malaria and control subjects that had never received AM-L from the same endemic malaria region of the Myanmar-Thailand border (62). In addition, hearing loss was found to be common on admission (57%) and only associated with age in patients treated for 3 days with AS+MQ (56). Ndiaye et al. report no signs of ototoxicity in AS+AQ or fixed-dose AM-L (61). In another study, no differences in ABR and PTA were found between patients treated with artemisan or artesunate alone, artesunate + MQ and MQ (64); in addition, no audiometric differences were found between patients treated for acute malaria with an artemisinin derivate and a control group that had never received it (65). A randomized study by Carrasquilla et al. found different combinations of ACT yielded improvements in PTA in acute uncomplicated P. falciparum malaria (60). In addition, a single-dose of artemisinin-naphthoquinone (ART-NQ) was found to be well-tolerated and safe for treating uncomplicated pediatric malaria in patients with P. falciparum and P. vivax (59); in addition, exposure to chloroquine diphosphate during gestation was also found not to result in hearing impairment in children (63).
The Adverse Effects of Antimalarials on the Vestibular System
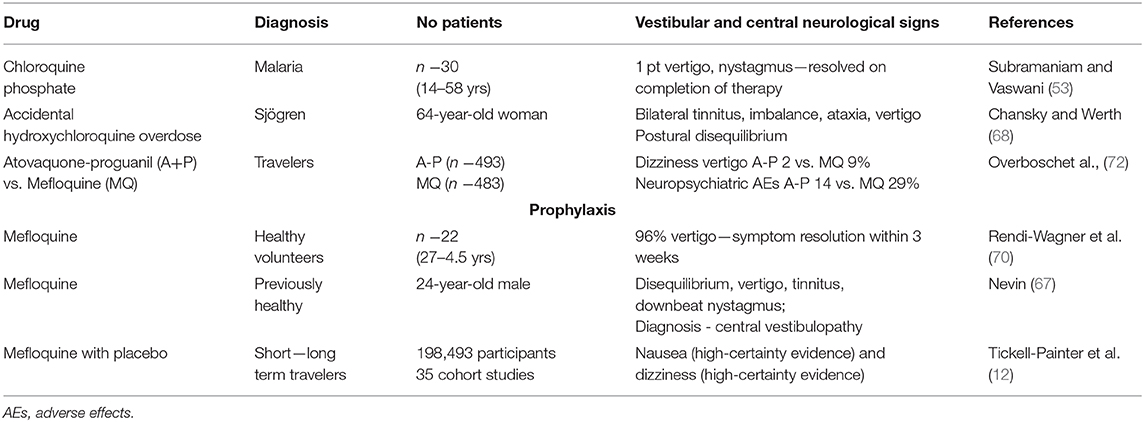
Antimalarial drugs can affect the central and peripheral vestibular systems. However, vestibular ototoxicity related to the peripheral system, such as vertigo, nystagmus and imbalance, is rarely reported either in patients being treated for malaria or autoimmune disease, or in healthy persons taking the drugs for malaria prophylaxis, such as travelers. The peripheral vestibular system can result in either partial or complete impairment of the vestibular end organs. In 2012, lasting vestibular disorder was added to the list of adverse effects of mefloquine by the discovery that vertigo and loss of balance could be permanent in some cases (67).
Only a few authors have reported incidents of acute vertigo in patients treated for malaria. Subramaniam and Vaswani assessed short-term chloroquine-induced ototoxicity in 30 patients with malaria, including only one patient with vestibular adverse effects (53). That patient had vertigo and spontaneous nystagmus, which resolved on completion of therapy. Vestibulopathy in a woman with a history of Sjögren's syndrome was described as a result of an accidental CHQ overdose. The patient had some neurological and otological complaints, such as bilateral tinnitus, imbalance, postural disequilibrium, and vertigo. After 12 weeks, only a mild improvement of symptoms was observed (68). The most critical causes for discontinuation of MQ prophylaxis among travelers are ototoxic and neuropsychiatric adverse effects. The most typical adverse effects in malaria prophylaxis are headaches (15.5%) and dizziness (14.4%), with vertigo and visual difficulties reported in 1–10% of prophylactic users (69). In addition, a meta-analysis of 35 cohort studies (198,493 participants) comparing prophylactic MQ intake with placebo in adults, children and pregnant women indicated that MQ users were more likely to report nausea (high-certainty evidence) and dizziness (high-certainty evidence) (12).
However, a clinical–pharmacological study with 22 healthy volunteers, identified vertigo (96%), nausea (82%), and headache (73%) after administration of MQ (750 and 500 mg at an interval of 6 h), during 21 days of monitoring. In 73% of subjects, severe vertigo was reported, which required bed rest and medication for 1 to 4 days. The majority (77.3%) of the participants showed symptom resolution within 3 weeks after drug administration. The authors emphasize that the severity of adverse effects after the usual therapeutic dosage of MQ in healthy subjects were unexpectedly high and should be taken into account during the treatment of malaria in travelers (70).
Central vestibulopathy was reported in a previously healthy 24-year-old male, taking three doses of MQ for prophylaxis malaria in Africa, despite evolving and worsening symptoms. He developed prolonged psychosis, anxiety, paranoia, short-term memory impairment, personality change and disequilibrium, vertigo, and tinnitus. He had downbeat nystagmus during normal routine vestibular responses in videonystagmography. Vestibular-evoked myogenic potentials on the right-side were enhanced as compared to the left side. Computerized dynamic posturography revealed a dysfunction pattern with falls during sensory organization tests (SOT) 5 and 6, so somatoform disorder was suspected. Radiological imaging tests showed insignificant changes. The author speculates that the symptoms were caused by damage or imbalance between the oculomotor and vestibular nuclei due to multifocal injury to the drug-induced brainstem. In 10 months follow-up, some improvement was observed, but still, he had episodes of vertigo and disequilibrium (71).
To improve efficacy, prevent transmission and reduce resistance, numerous antimalarial drug combinations and newer drugs have been introduced. However, such combinations may increase the level of adverse effects through cumulative toxicity. Similar incidences of adverse effects were observed between groups of travelers receiving atovaquone-proguanil A-P (493 subjects) or MQ (483 subjects) for the prevention of P. falciparum–induced malaria, that is, 71.4% in A-P vs. 67.3% in MQ group. The respective incidences of dizziness and vertigo were found to be 2 and 9%. The authors stated that A-P was better tolerated than MQ, and it was similarly effective for malaria prophylaxis in non-immune travelers (72).
Dizziness—A Sign of Neurotoxicity of the Balance System
The Barany Society Classification of Vestibular Disorders defines dizziness as a non-vertiginous sensation of disturbed or impaired spatial orientation without a false or distorted sense of motion (73). The Barany Society suggests that dizziness and vertigo should be defined separately. Subjects complaining of dizziness describe a range of sensations, such as feeling faint, weak or unsteady. In clinical studies, dizziness is classified as neurological manifestation (69). Headaches (15%) and dizziness (14%) are the most common mental and neurological adverse effects of antimalarial drugs during the treatment of acute malaria or prophylaxis (69).
Among antimalarial drugs, MQ is particularly frequently analyzed because it affects more domains of mental and neurological manifestations. For example, among 5,332 reactions associated with MQ used in malaria prophylaxis, Nevin and Leoutsakos reported vertigo in 6.3% of cases and dizziness in 17.3% (74). Potasman et al. assessed neuropsychiatric problems in 2,500 young travelers to tropical countries who received malaria prophylaxis (75). The most common were sleeping disturbances (52.1%), fatigue (48.7%), and dizziness (39.3%). A study of 1,170 Swedish soldiers who returned from Liberia and used MQ or atovaquone-proguanil (A-P) as malaria chemoprophylaxis found dizziness to be reported by 10.6% of MQ group and by 3.7% of A-P group (76).
Some studies have examined the adverse effects associated with combinations of malaria drugs. Lula et al. analyzed the safety and tolerability of artesunate and amodiaquine combination in the treatment of 387 patients with uncomplicated malaria (77). Dizziness was observed in 16.9% of cases, hypoacusis in 0.3%, tinnitus in 1.6%, and blurred vision in 1.6%. Vugt et al. compared effectiveness of artemether-benflumetol (AB, 177 patients) and artesunate-mefloquine (AM, 232 patients) in the treatment of P. falciparum malaria (78). Significantly higher rates of dizziness were found in the AM group (35%) than the AB group (15%). In addition, Odur et al. (79) reported dizziness in 18.4% of 299 malaria patients treated with dihydroartemisinin-piperaquine; this was the second most frequent adverse effect after weakness (32.6%) (77). Vestibular adverse effects are summarized in Table 4.
Assessment and Monitoring of Ototoxicity
Audiological Monitoring of Ototoxicity
Early identification of ototoxic hearing loss is critical to introducing possible alternative treatments with less ototoxic medications. However, hearing impairment may be unavoidable in some cases, even with ototoxicity monitoring. It is also necessary to bear in mind that some patients are predisposed to ototoxicity (19). One protocol for cochleotoxicity monitoring comprises baseline testing with conventional pure tone audiometry (PTA), high-frequency audiometry (HFA), tympanometry, speech audiometry, OAEs, and ABR. If the PTA could not identify initial ototoxic damage, objective audiological tests are important (80).
Otoacoustic Emissions
OAE tests provide an objective evaluation of subclinical changes in cochlear OHCs (81) and are sensitive tests for detecting and monitoring even early small impairments in the inner ear due to ototoxicity. OAE measurement in children is a particularly attractive approach as an efficient objective test. Ototoxicity monitoring found simultaneous decreases in OAE responses and changes in high-frequency audiometry thresholds (82). However, one consideration in OAE testing is that most applications can produce errors at high frequencies and can be problematic in patients with hearing loss. OAEs do not require a behavioral response and are time efficient. Many significant criteria have been proposed to interpret OAEs, but none has gained universal acceptance so far (80).
High-Frequency Audiometry
HFA comprises air-conduction threshold testing for frequencies above 8000 Hz, ranging up to 16 or 20 kHz. Nowadays, HFA is a well-established and widely-used method in ototoxicity monitoring programs; however, it is not standardized due to the fast decline of high frequencies with age and large inter-subject variability. HFA is more sensitive to early ototoxic changes than conventional audiometry and DPOAEs (19, 80).
Auditory Brainstem Response
The ABR is generated by eight cranial nerve and auditory brainstem structures in response to sound stimuli presented to the ear. It has several clinical applications, including the diagnosis and monitoring of dysfunction in the eight nerves and auditory brainstem, and can be used to estimate the auditory threshold particular in the pediatric population. The ABR can reflect changes in amplitude and/or latency of neural responses as a result of ototoxicity (83). Compared to OAEs, ABR responses can be recorded in ears with more severe pre-existing hearing loss. ABR can be used in cases with moderate hearing loss but output restraint at high frequencies. The limitation of this procedure is its long duration.
Tinnitus Evaluation
No formal tinnitus ototoxicity monitoring procedures have been designed, perhaps because tinnitus is not frequently mentioned as a side effect of antimalarial drugs (58, 68, 71, 77, 78). In the few existing guidelines on ototoxicity, tinnitus is mainly analyzed based on patient self-reporting (18, 19, 80). Konrad-Martin et al. suggest the use of the Tinnitus Ototoxicity Monitoring Interview to detect the onset of tinnitus and its changes in patients (84).
Vestibulotoxicity Monitoring
The vestibular toxicity of some drugs is well-established and can vary from discreet to severe instability, caused by a total bilateral loss of vestibular function. The degree mostly depends on the extent of cellular damage within the vestibular end-organ (85). No widely accepted guidelines for vestibulotoxicity monitoring exist. Dizziness can be assessed using patient self-reporting questionnaire like the Dizziness Handicap Inventory (DHI) (86); patients with a greater DHI score have more extensive functional impairment, as confirmed by clinical tests. When systematically monitoring patients for ototoxicity, DHI questioning is strongly encouraged (80). Electronystagmography (ENG) or videonystagmography (VNG) with rotation chair testing are recommended, and the caloric test is highly sensitive to the presence of peripheral vestibular system impairment. In addition, quantitative techniques like vestibular evoked myogenic potentials (VEMPs) and computerized dynamic posturography may be used (19, 80). Vestibulotoxicity monitoring is essential for patients demonstrating signs of balance disorders in the course of treatment. On the other hand, some clinical vestibular tests like VNG or VEMPs would be impractical, and are not recommended for routine monitoring in the acute phase of the disease as they are poorly tolerated. There is a need to monitor vestibular system function before, during, and after medical therapies that employ drugs with ototoxic properties to allow a possibility to intervene (i.e., by reducing dosages or substituting less damaging medications) to stave-off permanent damage.
Final Remarks
It is challenging to assess ototoxicity in clinical studies because the audiovestibular impairment in diseases treated by antimalarials, such as malaria, SLE or COVID-19, may also be related to the disease itself (87–89). In humans, hearing loss may also be caused by age, adverse effects of other drugs given simultaneously, or the development of concomitant diseases. In malaria, the disease may be affected by the endemicity status of malaria, and individual factors like malaria resistance, which may vary across the eligible studies (69). A 4-year follow-up study found 31 children to show a decrease in transitory evoked otoacoustic emissions (TEOAEs), suggesting that cochlear malfunction perseveres after recuperation from severe malaria (90).
Although for many years, ototoxicity in malaria patients was attributed to antimalarial drugs, studies on animal models suggest that the course of malaria might involve the inner ear. Significant hearing impairment in mice with cerebral malaria was confirmed; however, temporal bone examination revealed no structural alterations in the cochlea or any malaria-typical vascular lesions like leukocyte sequestration or micro-hemorrhages (91, 92). Schmutzhard et al. report an increase of intercellular adhesion molecule-1 (ICAM 1) expression in marginal cells of the stria vascularis in infected mice compared to control; however, there were no differences between malaria mice with and without hearing loss (85). They suggest that the endocochlear potential in the stria vascularis may be altered due to malfunction of the type 1 fibrocytes in the spiral ligament and disruption of the blood labyrinth barrier, which may cause the hearing loss observed in murine cerebral malaria (92, 93).
This review summarizes current knowledge about the occurrence of audiovestibular disorders after antimalarial drug treatment. Antimalarials are widely used not only to treat and prevent malaria but some have been repurposed for treating many autoimmune and rheumatological diseases, viral diseases, particularly SARS-CoV-2 infection, and various types of cancer. The ototoxic manifestations of antimalarial drugs include dizziness and vestibular symptoms; hearing loss and tinnitus were observed much less frequently and most of these symptoms were reversible. Early identification of ototoxic hearing loss is critical for introducing possible alternative treatments with less ototoxic medications. Therefore, more effective monitoring systems are needed.
Author Contributions
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
Funding
This study was founded by the Medical University of Lodz, research project number No 503/1-036-02/503-21-001-19-00.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
The authors would like to express their sincerest gratitude to Bogumila Bruc, MA from Information and Library Center of the Medical University of Lodz for her assistance in the literature search.
Abbreviations
ABR, auditory brainstem response; ACT, artemisinin-based combination therapy; AEs, adverse events; CQ, chloroquine; CHQ, hydroxychloroquine; DPOAE, distortion product otoacoustic emissions; IHCs, inner hair cells; MQ, mefloquine; OAE, otoacoustic emissions; OHCs, outer hair cells; P, plasmodium; PTA, pure tone audiometry; QN, quinine; SGNs, spiral ganglion neurons; SLE, systemic lupus erythematosus; SNHL, sensorineural hearing loss.
References
1. Szczepek AJ. Ototoxicity: old and new foes. In: Hatzopoulos S, editor. Advances in Clinical Audiology. London: IntechOpen. (2017). p. 233–49.
2. Gorobets NY, Sedash YV, Singh BK, Poonam, Rathi B. An overview of currently available antimalarials. Curr Top Med Chem. (2017) 17:2143–57. doi: 10.2174/1568026617666170130123520
3. Hobbs C, Duffy P. Drugs for malaria: something old, something new, something borrowed. F1000 Biol Rep. (2011) 3:24. doi: 10.3410/B3-24
4. Tse EG, Korsik M, Todd MH. The past, present and future of anti-malarial medicines. Malar J. (2019) 18:93. doi: 10.1186/s12936-019-2724-z
5. World Health Organization. Guidelines for the Treatment of Malaria. 3rd ed. Geneva: World Health Organization (2015).
6. The Drugs.com Database. Available online at: https://www.drugs.com (accessed February 17, 2021).
7. The DrugBank Database. Available online at: https://www.drugbank.com (accessed February 17, 2021).
8. U.S. Food Drug Administration, Drugs@FDA: FDA-Approved Drugs. Available online at: https://www.accessdata.fda.gov (accessed February 17, 2021).
9. Semedo MG, Dias-Silva N, Miguéis J, Pita JR. Quinine in otology and neurotology: ototoxicity and historic role in therapy. Otol Neurotol. (2021) 42:145–52. doi: 10.1097/MAO.0000000000002809
10. Della Porta A, Bornstein K, Coye A, Montrief T, Long B, Parris MA. Acute chloroquine and hydroxychloroquine toxicity: a review for emergency clinicians. Am J Emerg Med. (2020) 38:2209–17. doi: 10.1016/j.ajem.2020.07.030
11. Alani BG, Alwash AH, Ibrahim IT. Wide applications of chloroquine other than antimalarial. Pharmacol Pharm. (2020) 11:251–81. doi: 10.4236/pp.2020.1110022
12. Tickell-Painter M, Maayan N, Saunders R, Pace C, Sinclair D. Mefloquine for preventing malaria during travel to endemic areas. Cochrane Database Syst Rev. (2017)10:CD006491. doi: 10.1002/14651858.CD006491.pub4
13. Plantone D, Koudriavtseva T. Current and future use of chloroquine and hydroxychloroquine in infectious, immune, neoplastic, and neurological diseases: a mini-review. Clin Drug Investig. (2018) 38:653–71. doi: 10.1007/s40261-018-0656-y
14. Haładyj E, Sikora M, Felis-Giemza A, Olesińska M. Antimalarials—are they effective and safe in rheumatic diseases? Reumatologia. (2018) 56:164–73. doi: 10.5114/reum.2018.76904
15. Zhou W, Wang H, Yang Y, Chen ZS, Zou C, Zhang J. Chloroquine against malaria, cancers and viral diseases. Drug Discov Today. (2020) 25:2012–22. doi: 10.1016/j.drudis.2020.09.010
16. Lechien JR, Chiesa-Estomba CM, Place S, Van Laethem Y, Cabaraux P, Mat Q, et al. Clinical and epidemiological characteristics of 1420 European patients with mild-to-moderate coronavirus disease 2019. J Intern Med. (2020) 288:335–44. doi: 10.1111/joim.13089
17. Jiang YY, Shui JC, Zhang BX, Chin JW, Yue RS. The potential roles of artemisinin and its derivatives in the treatment of type 2 diabetes mellitus. Front Pharmacol. (2020) 11:585487. doi: 10.3389/fphar.2020.585487
18. Prepageran N, Rutka JA. Salicylates, nonsteroidal anfi-inflammatory drugs, quinine and heavy metals. In: Roland PS, Rutka JA, editors. Ototoxicity. Hamilton, ON: BC Decker Inc. (2004). p. 28–41.
19. Schellack N, Naude A. An overview of pharmacotherapy-induced ototoxicity. S Afr Fam Pract. (2013) 55:357–65. doi: 10.1080/20786204.2013.1087437
20. Alvan G, Berninger E, Gustafsson LL, Karlsson KK, Paintaud G, Wakelkamp M. Concentration-Response relationship of hearing impairment caused by quinine and salicylate: pharmacological similarities but different molecular mechanisms. Basic Clin Pharmacol Toxicol. (2017) 120:5–13. doi: 10.1111/bcpt.12640
21. Smith DI, Lawrence M, Hawkins JE Jr. Effects of noise and quinine on the vessels of stria vascularis. An image analysis study. Am J Otolaryngol. (1985) 6:280–9. doi: 10.1016/s0196-0709(85)80056-9
22. Karlsson KK, Flock B, Flock A. Ultrastructural changes in the outer hair cells of the guinea pig cochlea after exposure to quinine. Acta Otolaryngol. (1991) 111:500–5. doi: 10.3109/00016489109138375
23. Karlsson KK, Flock A. Quinine causes outer hair cells to change length. Neurosci Lett. (1990) 116:101–5. doi: 10.1016/0304-3940(90)90393-n
24. Jarboe SK, Hallworth R. The effect of quinine on outer hair cell shape, compliance and force. Hear Res. (1999) 132:43–50.doi: 10.1016/s0378-5955(99)00031-3
25. Davis SN, Wu P, Camci ED, Simon JA, Rubel EW, Raible DW. Chloroquine kills hair cells in zebrafish lateral line and murine cochlear cultures: Implications for ototoxicity. Hear Res. (2020) 395:108019. doi: 10.1016/j.heares.2020.108019
26. Karlsson KK, Ulfendahl M, Khanna SM, Flock A. The effects of quinine on the cochlear mechanics in the isolated temporal bone preparation. Hear Res. (1991) 53:95–100. doi: 10.1016/0378-5955(91)90216-v
27. Zheng J, Ren T, Parthasarathi A, Nuttall AL. Quinine-induced alterations of electrically evoked otoacoustic emissions and cochlear potentials in guinea pigs. Hear Res. (2001) 154:124–34.doi: 10.1016/s0378-5955(01)00229-5
28. Dieler R, Davies C, Shehata-Dieler WE. Der Einfluss von Chinin auf aktive Motilität und Feinstruktur isolierter äusserer Haarzellen der Meerschweinchenkochlea [The effects of quinine on active motile responses and fine structure of isolated outer hair cells from the Guinea pig cochlea]. Laryngorhinootologie. (2002) 81:196–203. German. doi: 10.1055/s-2002-25041
29. Puel JL, Bobbin RP, Fallon M. Salicylate, mefenamate, meclofenamate, and quinine on cochlear potentials. Otolaryngol. Head Neck Surg. (1990) 102:66–73. doi: 10.1177/019459989010200110
30. Pienkowski M. Ulfendahl. Differential effects of salicylate, quinine, and furosemide on guinea pig inner and outer hair cell function revealed by the input-output relation of the auditory brainstem response. J Am Acad Audiol. (2011) 22:104–12.doi: 10.3766/jaaa.22.2.5
31. Lin X, Chen S, Tee D. Efects of quinine on the excitability and voltage-dependent currents of isolated spiral ganglion neurons in culture. J Neurophysiol. (1998) 79:2503–12. doi: 10.1152/jn.1998.79.5.2503
32. Eggermont JJ, Kenmochi M. Salicylate and quinine selectively increase spontaneous firing rates in secondary auditory cortex. Hear Res. (1998) 117:149–60.doi: 10.1016/s0378-5955(98)00008-2
33. Dayan AD. Neurotoxicity and artemisinin compounds do the observations in animals justify limitation of clinical use? Med Trop. (1998) 58(3 Suppl):32–7.
34. Genovese RF, Newman DB, Li Q, Peggins JO, Brewer TG. Dose-dependent brainstem neuropathology following repeated arteether administration in rats. Brain Res Bull. (1998) 45:199–202. doi: 10.1016/s0361-9230(97)00339-0
35. Ding D, Qi W, Yu D, Jiang H, Salvi R. Ototoxic effects of mefloquine in cochlear organotypic cultures. J Otol. (2009) 4:29–38. doi: 10.1016/S1672-2930(09)50018-9
36. Ding D, Zhang J, Jiang H, Xuan W, Qi W, Salvi R. Some ototoxic drugs destroy cochlear support cells before damaging sensory hair cells. Neurotox Res. (2020) 37:743–52. doi: 10.1007/s12640-020-00170-8
37. Ding D, Someya S, Jiang H, Qi W, Yu D, Tanokura M, et al. Detection of apoptosis by RT-PCR array in mefloquine-induced cochlear damage. J Otol. (2011) 6:1–9. doi: 10.1016/S1672-2930(11)50001-7
38. Ding D, Qi W, Yu D, Jiang H, Han C, Kim MJ, et al. Addition of exogenous NAD+ prevents mefloquine-induced neuroaxonal and hair cell degeneration through reduction of caspase-3-mediated apoptosis in cochlear organotypic cultures. PLoS ONE. (2013) 8:e79817. doi: 10.1371/journal.pone.0079817
39. Yu D, Ding D, Jiang H, Stolzberg D, Salvi R. Mefloquine damage vestibular hair cells in organotypic cultures. Neurotox Res. (2011) 20:51–8. doi: 10.1007/s12640-010-9221-z
40. Iskander NM, Youssef TF, Ahmed MR, Mohamed AA. Is halofantrine ototoxic? Experimental study on guinea pig cochlea model. J Laryngol Otol. (2010) 124:1061–6. doi: 10.1017/s0022215110001301
41. Jastreboff PJ, Brennan JF, Sasaki CT. Quinine-induced tinnitus in rats. Arch Otolaryngol Head Neck Surg. (1991) 117:1162–6. doi: 10.1001/archotol.1991.01870220110020
42. Ralli M, Lobarinas E, Fetoni AR, Stolzberg D, Paludetti G, Salvi R. Comparison of salicylate- and quinine-induced tinnitus in rats: development, time course, and evaluation of audiologic correlates. Otol Neurotol. (2010) 31:823–31. doi: 10.1097/MAO.0b013e3181de4662
43. Rybak LP. Whitworth C. Quinine reduces noxious cochlear effects of furosemide and ethacrynic acid. Am J Otolaryngol. (1988) 9:238–43.doi: 10.1016/s0196-0709(88)80033-4
44. Ou HC, Keating S, Wu P, Simon JA, Raible DW, Rubel EW. Quinoline ring derivatives protect against aminoglycoside-induced hair cell death in the zebrafish lateral line. J Assoc Res Otolaryngol. (2012) 13:759–70. doi: 10.1007/s10162-012-0353-0
45. Rocha-Sanchez SM, Fuson O, Tarang S, Goodman L, Pyakurel U, Liu H, et al. Quinoxaline protects zebrafish lateral line hair cells from cisplatin and aminoglycosides damage. Sci Rep. (2018) 8:15119. doi: 10.1038/s41598-018-33520-w
46. Alharazneh A, Luk L, Huth M, Monfared A, Steyger PS, Cheng AG, et al. Functional hair cell mechanotransducer channels are required for aminoglycoside ototoxicity. PLoS ONE. (2011) 6:e22347. doi: 10.1371/journal.pone.0022347
47. De Luca P, Scarpa A, De Bonis E, Cavaliere M, Viola P, Gioacchini FM, et al. Chloroquine and hydroxychloroquine ototoxicity; potential implications for SARS-CoV-2 treatment. A brief review of the literature. Am J Otolaryngol. (2020) 8:102640. doi: 10.1016/j.amjoto.2020.102640
48. Monteiro EMR, Nascimento MFL, Brito TRC, Lima MC, Sefair LR, Pedrosa MM. Chloroquine and COVID-19: should we care about ototoxicity? Int Arch Otorhinolaryngol. (2020) 24:e359–e363. doi: 10.1055/s-0040-1714142
49. Jourde-Chiche N, Mancini J, Dagher N, Taugourdeau S, Thomas G, Brunet C, et al. Antimalarial ototoxicity: an underdiagnosed complication? A study of spontaneous reports to the French Pharmacovigilance network. Ann Rheum Dis. (2012) 71:1586. doi: 10.1136/annrheumdis-2011-201265
50. Joo Y, Cruickshanks KJ, Klein BEK, Klein R, Hong O, Wallhagen MI. The contribution of ototoxic medications to hearing loss among older adults. J Gerontol A Biol Sci Med Sci. (2020) 75:561–6. doi: 10.1093/gerona/glz166
51. Lee SJ, Ter Kuile FO, Price RN, Luxemburger C, Nosten F. Adverse effects of mefloquine for the treatment of uncomplicated malaria in Thailand: a pooled analysis of 19, 850 individual patients. PLoS ONE. (2017) 12:e0168780. doi: 10.1371/journal.pone.0168780
52. Prayuenyong P, Kasbekar AV, Baguley DM. Clinical implications of chloroquine and hydroxychloroquine ototoxicity for COVID-19 treatment: a mini-review. Front Public Health. (2020) 8:252. doi: 10.3389/fpubh.2020.00252
53. Subramaniam V, Vaswani RV. Assessment of short term chloroquine-induced ototoxicity in malaria patients. Global J Med Res. (2015) 15:14–17.
54. Gürkov R, Eshetu T, Miranda IB, Berens-Riha N, Mamo Y, Girma T, et al. Ototoxicity of artemether/lumefantrine in the treatment of falciparum malaria: a randomized trial. Malar J. (2008) 7:179. doi: 10.1186/1475-2875-7-179
55. Adjei G0, Kurtzhals JAL, Rodrigues OP, Alifrangis M, Hoegberg LCG, Kitcher ED, et al. Amodiaquine-artesunate vs artemether-lumefantrine for uncomplicated malaria in Ghanaian children: a randomized efficacy and safety trial with one year follow-up. Malar J. (2008) 7:127. doi: 10.1186/1475-2875-7-127
56. Carrara VI, Phyo AP, Nwee P, Soe M, Htoo H, Arunkamomkiri J, et al. Auditory assessment of patients with acute uncomplicated Plasmodium falciparum malaria treated with three-day mefloquine-artesunate on the north-western border of Thailand. Malar J. (2008) 7:233. doi: 10.1186/1475-2875-7-233
57. Toovey S, Jamieson A. Audiometric changes associated with the treatment of uncomplicated falciparum malaria with co- artemether. Trans R Soc Trop Med Hyg. (2004) 98:261–7. doi: 10.1016/j.trstmh.2003.11.001
58. Tange RA, Dreschler WA, Claessen FA, Perenboom RM. Ototoxic reactions of quinine in healthy persons and patients with Plasmodium falciparum infection. Auris Nasus Larynx. (1997) 24:131–6.doi: 10.1016/S0385-8146(96)00031-4
59. Benjamin J, Moore B, Lee ST, Senn M, Griffin S, Lautu D, et al. Artemisinin-Naphthoquine combination therapy for uncomplicated pediatric malaria: a tolerability, safety, and preliminary efficacy study. Antimicrob Agents Chemother. (2012) 56:2465–71. doi: 10.1128/AAC.06248-11
60. Carrasquilla G, Barón C, Monsell EM, Cousin M, Walter V, Lefèvre G, et al. Randomized, prospective, three-arm study to confirm the auditory safety and efficacy of artemether-lumefantrine in Colombian patients with uncomplicated Plasmodium falciparum malaria. Am J Trop Med Hyg. (2012) 86:75–83. doi: 10.4269/ajtmh.2012.11-0192
61. Ndiaye JLA, Faye B, Gueye A, Tine R, Ndiaye D, Tchania C, et al. Repeated treatment of recurrent uncomplicated Plasmodium falciparum malaria in Senegal with fixed-dose artesunate plus amodiaquine versus fixed-dose artemether plus lumefantrine: a randomized, open-label trial. Malar J. (2011) 10:237. doi: 10.1186/1475-2875-10-237
62. Hutagalung R, Htoo H, Nwee P, Arunkamomkiri J, Zwang J, Carrara VI, et al. A case-control auditory evaluation of patients treated with artemether-lumefantrine. Am J Trop Med Hyg. (2006) 74:211–4.
63. Borba EF, Turrini-Filho JR, Kurruma KAM, Bertola C, Pedalini ME, Lorenzi MC, et al. Chloroquine gestational use in systemic lupus erythematosus: assessing the risk of child ototoxicity by pure tone audiometry. Lupus. (2004) 13:223–7. doi: 10.1191/0961203304lu528oa
64. Kissinger E, Hien TT, Hung NT, Nam ND, Tuyen NL, Dinh BV, et al. Clinical and neurophysiological study of the effects of multiple doses of artemisinin on brain-stem function in Vietnamese patients. Am J Trop Med Hyg. (2000) 63:48–55. doi: 10.4269/ajtmh.2000.63.48
65. Van Vugt M, Angus BJ, Price RN, Mann C, Simpson JA, Poletto C, et al. Case-control auditory evaluation of patients treated with artemisinin derivatives for multidrug-resistant plasmodium falciparum malaria. Am J Trop Med Hyg. (2000) 62:65–9. doi: 10.4269/ajtmh.2000.62.65
66. Ramos-Martín V, González-Martínez C, Mackenzie I, Schmutzhard J, Pace C, Lalloo DG, et al. Neuroauditory toxicity of artemisinin combination therapies-have safety concerns been addressed? Am J Trop Med Hyg. (2014) 91:62–73. doi: 10.4269/ajtmh.13-0702
67. Nevin RL. Idiosyncratic quinoline central nervous system toxicity: historical insights into the chronic neurological sequelae of mefloquine. Int J Parasitol Drugs Drug Resist. (2014) 4:2. 118–25.doi: 10.1016/j.ijpddr.2014.03.002
68. Chansky PB, Werth VP. Accidental hydroxychloroquine overdose resulting in neurotoxic vestibulopathy. BMJ Case Rep. (2017) 2017:bcr2016218786. doi: 10.1136/bcr-2016-218786
69. Bitta MA, Kariuki SM, Mwita C, Gwer S, Mwai L, Newton CRJC. Antimalarial drugs and the prevalence of mental and neurological manifestations: a systematic review and meta-analysis. Wellcome Open Res. (2017) 2:13. doi: 10.12688/wellcomeopenres.10658.2
70. Rendi-Wagner P, Noedl H, Wernsdorfer WH, Wiedermann G, Mikolasek A, Kollaritsch H. Unexpected frequency, duration and spectrum of adverse events after therapeutic dose of mefloquine in healthy adults. Acta Trop. (2002) 8:2.167–73. doi: 10.1016/s0001-706x(01)00210-8
71. Nevin RL. Limbic encephalopathy and central vestibulopathy caused by mefloquine: a case report. Travel Med Infect Dis. (2012) 10:3. 144–51. doi: 10.1016/j.tmaid.2012.03.006
72. Overbosch D, Schilthuis H, Bienzle U, Behrens RH, Kain KC, Clarke PD, et al. Malarone International Study Team. Atovaquone-proguanil versus mefloquine for malaria prophylaxis in nonimmune travelers: results from a randomized, double-blind study. Clin Infect Dis. (2001) 33:7.1015–21. doi: 10.1086/322694
73. Bisdorff A, Von Brevern M, Lempert T, Newman-Toker DE. Classification of vestibular symptoms: towards an international classification of vestibular disorders. J Vestib Res. (2009) 19:1–2.1–13. doi: 10.3233/VES-2009-0343
74. Nevin RL, Leoutsakos JM. Identification of a syndrome class of neuropsychiatric adverse reactions to mefloquine from latent class modeling of FDA Adverse Event Reporting System data. Drugs RD. (2017) 17:199–210. doi: 10.1007/s40268-016-0167-3
75. Potasman I, Beny A, Seligmann H. Neuropsychiatric problems in 2,500 long-term young travelers to the tropics. J Travel Med. (2000) 7:5–9. doi: 10.2310/7060.2000.00002
76. Andersson H, Askling HH, Falck B, Rombo L. Well-tolerated chemoprophylaxis uniformly prevented Swedish soldiers from Plasmodium falciparum malaria in Liberia, 2004–2006. Mil Med. (2008) 173:1194–8. doi: 10.7205/milmed.173.12.1194
77. Lula Y, Nzolo D, Ntamabyaliro PM, Nanga T, Mesia G, Tona G, et al. Proceedings of the 10th European Congress on Tropical Medicine and International Health; 2017 Oct 16–20 Antwerp, Belgium. Trop Med Int Health. (2017) 22(Suppl 1):1–369. doi: 10.1111/tmi.12981
78. Van Vugt M, Brockman A, Gemperli B, Luxemburger C, Gathmann I, Royce C, et al. Randomized comparison of artemether-benflumetol and artesunate-mefloquine in treatment of multidrug resistant falciparum malaria. Antimicrob Agents Chemother. (1998) 42:1.135–9. doi: 10.1128/AAC.42.1.135
79. Odur A, Ayamba E, Anyorigiya T, Beeri P, Chatio S, Amenga-Etego L, et al. Systems effectiveness and patient adherence to dihydroartemisinin piperaquine in northern Ghana. In: Proceedings of the 10th European Congress on Tropical Medicine and International Health, Vol. 22. Antwerp: Wiley (2017). p. 264–5.
80. American Academy of Audiology. Ototoxicity Monitoring. Position statement and clinical practice guidelines: (2009). Available online at: http://audiology-web.s3.amazonaws.com/migrated/OtoMonGuidelines.pdf_539974c40999c1.58842217.pdf (accessed January 19, 2021).
81. McFadden D, Pasanen EG. Otoacoustic emissions and quinine sulfate. J Acoust Soc Am. (1994) 95:3460–74. doi: 10.1121/1.410022
82. Schmutzhard J, Lackner P, Helbok R, Hurth HV, Aregger FC, Muigg V, et al. Severe malaria in children leads to a significant impairment of transitory otoacoustic emissions–a prospective multicenter cohort study. BMC Med. (2015) 13:125. doi: 10.1186/s12916-015-0366-8
83. Bernard P. Alterations of auditory evoked potentials during the course of chloroquine treatment. Acta Otolaryngol. (1985) 99:387–92. doi: 10.3109/00016488509108928
84. Konrad-Martin D, Reavis KM, McMillan G, Helt WJ, Dille M. Proposed comprehensive ototoxicity monitoring program for VA healthcare (COMP-VA) J Rehabil Res Dev. (2014) 51:81–100. doi: 10.1682/JRRD.2013.04.0092
85. Kisilevsky VE, Tomlinson RD, Ranalli P, Prepageran N. Monitoring vestibular ototoxicity. In: Roland PS, Rutka JA, editors. Ototoxicity. Hamilton, ON: BC Decker Inc. (2004). p. 161–9.
86. Jacobson GP, Newman CW. The development of the dizziness handicap inventory. Arch Otolaryngol Head Neck Surg. (1990) 116:424-7. doi: 10.1001/archotol.1990.01870040046011
87. Polanski JF, Tanaka EA, Barros H, Chuchene AG, Miguel PTG, Skare TL. Chloroquine, hydroxychloroquine and hearing loss: a study in systemic lupus erythematosus patients. Laryngoscope. (2020) 131:E957–E960. doi: 10.1002/lary.28873
88. Mustafa MWM. Audiological profile of asymptomatic Covid-19 PCR-positive cases. Am J Otolaryngol. (2020) 41:102483. doi: 10.1016/j.amjoto.2020.102483
89. Almufarrij I, Uus K, Munro KJ. Does coronavirus affect the audio-vestibular system? A rapid systematic review. Int J Audiol. (2020) 59:487–91. doi: 10.1080/14992027.2020.1776406
90. Reiterer E, Reider S, Lackner P, Fischer N, Dejaco D, Riechelman H, et al. Long-term follow-up study on otoacoustic emissions testing in paediatric patients with severe malaria in Gabon. Malar J. (2019) 18:212. doi: 10.1186/s12936-019-2840-9
91. Schmutzhard J, Kositz CH, Lackner P, Dietmann A, Fischer M, Glueckert R, et al.: Murine malaria is associated with significant hearing impairment. Malar J. (2010) 9:159. doi: 10.1186/1475-2875-9-159
92. Schmutzhard J, Kositz CH, Lackner P, Pritz C, Glueckert R, Fischer M, et al. Murine cerebral malaria: histopathology and ICAM 1 immunohistochemistry of the inner ear. Trop Med Int Health. (2011) 16:914–22. doi: 10.1111/j.1365-3156.2011.02801.x
Keywords: ototoxicity, antimalarials, dizziness, vertigo, tinnitus, hearing loss
Citation: Jozefowicz-Korczynska M, Pajor A and Lucas Grzelczyk W (2021) The Ototoxicity of Antimalarial Drugs—A State of the Art Review. Front. Neurol. 12:661740. doi: 10.3389/fneur.2021.661740
Received: 31 January 2021; Accepted: 04 March 2021;
Published: 20 April 2021.
Edited by:
Agnieszka J. Szczepek, Charité – Universitätsmedizin Berlin, GermanyReviewed by:
Alfonso Scarpa, University of Salerno, ItalyJessica Paken, University of KwaZulu-Natal, South Africa
Pietro De Luca, University of Salerno, Italy
Copyright © 2021 Jozefowicz-Korczynska, Pajor and Lucas Grzelczyk. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Weronika Lucas Grzelczyk, d2Vyb25pa2EubHVjYXNAZ21haWwuY29t; d2Vyb25pa2EubHVjYXMtZ3J6ZWxjenlrQHVtZWQubG9kei5wbA==
 Magdalena Jozefowicz-Korczynska1
Magdalena Jozefowicz-Korczynska1 Weronika Lucas Grzelczyk
Weronika Lucas Grzelczyk