- 1Department of Neurology, St Vincent's Hospital, Sydney, NSW, Australia
- 2School of Medicine, University of New South Wales, Sydney, NSW, Australia
Deep brain stimulation (DBS) of the thalamus is an effective treatment for medically refractory essential, dystonic and Parkinson's tremor. It may also provide benefit in less common tremor syndromes including, post-traumatic, cerebellar, Holmes, neuropathic and orthostatic tremor. The long-term benefit of DBS in essential and dystonic tremor (ET/DT) often wanes over time, a phenomena referred to as stimulation “tolerance” or “habituation”. While habituation is generally accepted to exist, it remains controversial. Attempts to quantify habituation have revealed conflicting reports. Placebo effects, loss of micro-lesional effect, disease related progression, suboptimal stimulation and stimulation related side-effects may all contribute to the loss of sustained long-term therapeutic effect. Habituation often presents as substantial loss of initial DBS benefit occurring as early as a few months after initial stimulation; a complex and feared issue when faced in the setting of optimal electrode placement. Simply increasing stimulation current tends only to propagate tremor severity and induce stimulation related side effects. The report by Paschen and colleagues of worsening tremor scores in the “On” vs. “Off” stimulation state over time, even after accounting for “rebound” tremor, supports the concept of habituation. However, these findings have not been consistent across all studies. Chronic high intensity stimulation has been hypothesized to induce detrimental plastic effects on tremor networks, with some lines of evidence that DT and ET may be more susceptible than Parkinson's tremor to habituation. However, Tsuboi and colleague's recent longitudinal follow-up in dystonic and “pure” essential tremor suggests otherwise. Alternatively, post-mortem findings support a biological adaption to stimulation. The prevalence and etiology of habituation is still not fully understood and management remains difficult. A recent study reported that alternating thalamic stimulation parameters at weekly intervals provided improved stability of tremor control consistent with reduced habituation. In this article the available evidence for habituation after DBS for tremor syndromes is reviewed; including its prevalence, time-course, possible mechanisms; along with expected long-term outcomes for tremor and factors that may assist in predicting, preventing and managing habituation.
Introduction
Tremor is an involuntary, rhythmic, oscillatory movement of a body part (1), with an estimated prevalence of 14.5% in the general adult population (2). The spectrum of tremor extends from the enhanced physiological postural tremor, often only noticeable during states of fatigue and heightened anxiety, to persistent pathological syndromes including essential tremor (ET), where currently available medications are moderately effective at best. For the severe end of the spectrum of tremor syndromes, functional neurosurgical techniques and neuroradiological procedures exists. These have evolved since Cooper (3) made the unintentional observation nearly 70 years ago, that destruction of a portion of the globus pallidus suppressed tremor of a patient with Parkinson's Disease (PD). Stereotactic lesional surgery mainly targeting the ventrolateral thalamus and posterior subthalamic white matter was used since the 1950's. During this period it was discovered that intraoperative high frequency electrical stimulation would suppress tremor and was used to verify the target region prior to thermal abalation (4, 5). These pioneering stereotactic interventions paved the way for the first cases of deep brain stimulation (DBS) performed by Cooper et al. (6). Motivated by a desire to avoid the frequent dysarthria observed following bilateral radiofrequency thalamotomy, DBS of the thalamic ventral intermediate nucleus (VIM) was revisited by Benabid in 1987 for second-side treatment of tremor and popularized following their 1991 publication of a series of 26 PD and six essential tremor (ET) patients treated with VIM DBS who reported to have a sustained tremor response over a 13-month median follow-up period (7). Subsequently large studies confirmed the effectiveness of VIM DBS for ET and PD (8, 9).
DBS remains the most common surgical procedure for medication-refractory tremor. However, the long-term benefits of therapy, particularly in ET, are often observed to wane over time, in a variable, unpredictable pattern. A phenomenon of “tolerance” was first described by Benabid et al. in a series of 80 tremor-dominant PD and 20 ET patients, with either uni- or bi-lateral VIM stimulation (10). Regular increase in stimulation to alleviate tremor was required to a final threshold that could no longer be increased due to the induction of side effects. “Tolerance” was associated with eventual loss of functional benefit and was more commonly observed with those with action tremor, in severe syndromes, with higher stimulation intensity, and where continuous 24-h stimulation had been adopted (10). The phenomenon of “tolerance” has continued to be observed in clinical practice and is now usually referred to as “habituation” (11). Attempts to characterize and quantity habituation have revealed conflicting reports in the medical literature and remain the subject of debate.
In this narrative review article, the available evidence for habituation after DBS for tremor syndromes is reviewed to reappraise perceptions of expected long term outcomes and factors that may assist in predicting this phenomenon. We also provide some information on possible pathophysiological mechanisms underlying ‘habituation' and approaches to its management.
Habituation Definition
Habituation in the context of benefit from DBS was first mentioned in the medical literature by Benabid et al. and described as “tolerance”. It was hypothesized a progressively decreased biological response (habituation) of the neuronal network to be a possible mechanism for the phenomena of “tolerance” (10). Recently the term habituation has been proposed to replace “tolerance”, defined by Fasano and Helmich to be the rapid vanishing of DBS efficacy after programming (11). This definition of habituation can be expanded to include delayed, progressive loss of therapeutic benefit for tremor after DBS, in line with the original concept of “tolerance” due to “decreased biological response (habituation) of the neuronal network” as described by Benabid et al. (10).
Authors have attempted to study habituation in the context of progressive loss of DBS benefit with particular attention given to differentiating progression from the natural history of disease. We agree in theory that comparing the tremor severity in the “off” state at two different time points, after allowing for rebound, represent disease progression; whereas tremor severity in the “on” state is determined by both disease progression and the stimulation effect. The difference (delta) between the on-off state, when compared over time, has been assumed by authors to be a measure of changing stimulation over time and attributed to habituation (12, 13). This is based on the premise that over time, other variables, specifically lead location and optimization of programming remain constant; but further, alternative mechanisms are not contributing or causing the phenomena that has been labeled habituation. Given these provisions, we will proceed on the operational hypothesis, reflected by the change in delta over time, from the definition of habituation known previously as “tolerance”; to be the loss of benefit from electrode reprogramming over time in the setting of optimal electrode placement and programming not explained by disease progression of the tremor syndrome. Habituation should not be explained by loss of micro-lesional implant effect or expected progression due to the natural history of the tremor syndrome. In line with the concept of “rapid vanishing of effect” habituation also refers to temporary improvement in tremor severity following increasing electrical field strength or contact adjustment, followed by subsequent paradoxical worsening.
Although this definition is useful conceptually, determining if an individual patient is experiencing habituation after tremor DBS remains very difficult because of the following; Firstly, there is no absolute agreed definition of what constitutes optimal lead placement; more troubling though, is the fact not all DBS leads placed within the optimal 2 mm radius of the intended target have a concordant clinical response (14). Secondly, optimal DBS programming is highly operator dependent as evidenced by significant clinical improvements achieved after expert reprogramming (15). Lastly, progression of the underlying tremor syndrome as part of the natural history of the disease must be subtracted from any apportionment of habituation, in itself a very difficult distinction, highlighting the inherent complexity and uncertainties surrounding this topic.
Does “Habituation” Really Exist?
Loss of Benefit Over Time
Habituation has most commonly been associated with ET, possibly reflecting the experience of clinicians in practice. Over 20 studies have been published looking at the long-term clinical efficacy of DBS in this condition; most commonly involving uni-or bilateral VIM stimulation (12, 13, 15–32). When looking at studies with a greater than 3-year follow-up, the long-term effect compared to baseline, ranges from 31.2–88.4% improvement (11, 13). The less traditional target, posterior subthalamic area (PSA)/caudal zona incerta (cZi), in comparison has relatively few follow-up studies; but with a similar range of effect size from baseline: 33–76% improvement (30, 33, 34). Some studies have suggested that the PSA/cZI may be less prone to habituation; however, no superiority has ever been clearly established (33–35). Despite this persistent improvement from DBS in the long-term, the majority of studies have shown that the effect diminishes over time.
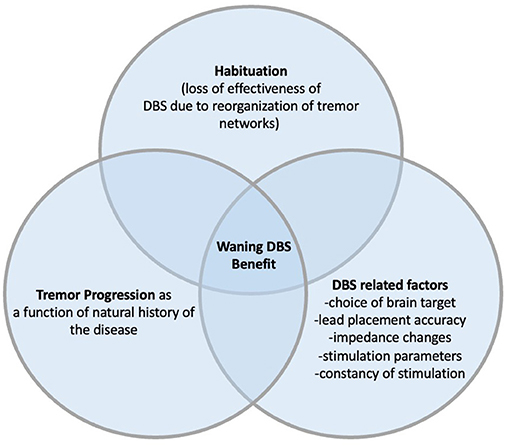
A recent systematic review and meta-analysis by Lu et al., included 26 studies with 439 patients, looked at potential outcome predictors following VIM DBS in ET. The pooled treatment effect was 60.3% improvement in objective Tremor Rating Scale (TRS) scores at 20 months (+/– 17.3). Correlation with outcome was seen only with pre-operative TRS scores and follow-up time; both negatively correlated with the clinical outcome (36). It had previously been reported that pre-operative cerebellar dysfunction was a risk factor for the development of early “tolerance” (37). Natural disease progression and habituation have been proposed as the most plausible factors contributing to VIM-DBS treatment declining overtime (17, 23, 27, 31, 33). Despite the absence of consensus guide lines, electrode placement beyond a 2–3 mm radius of an intended target have been associated with suboptimal tremor control and can be a correctable cause of DBS “failure” (37, 38). Further, in cases of suboptimal clinical benefit, DBS lead adjustment of only a few millimeters can have a meaningful benefit (39). Other possible co-contributing factors include incorrect pre-operative diagnosis (14), loss of microthalamotomy (9) and increased impedance of brain tissue over time (10) (Figure 1). However, effects of varying tissue impedance are minimized by constant current DBS systems now more widely used.
Short-Term Habituation
Continuous increase in DBS stimulation parameters, followed by a temporary improvement in tremor severity but an ultimate paradoxical worsening is the hallmark clinical observation in habituation. This was first demonstrated in the short-term by Barbe et al. in patients treated with VIM DBS for ET. After optimization of stimulation parameters, patients were followed and reassessed at 10-weeks. Statistically significant improvement in TRS hemi-body scores compared to baseline was observed after optimization; but at 10-weeks this stimulation effect was remarkable weaker, abolishing the immediate effect compared to baseline (40). Furthermore, of the 21 patients who agreed to follow-up after initial stimulation changes, only 16 completed the 10-week assessment, with two patients dropping out due to unacceptable worsening of the tremor syndrome. Adaption of the pathological tremor networks to the new DBS interface was proposed, clinically seen as paradoxical worsening of tremor, referred to as “habitation” by the authors.
Long Term Habituation or Disease Progression?
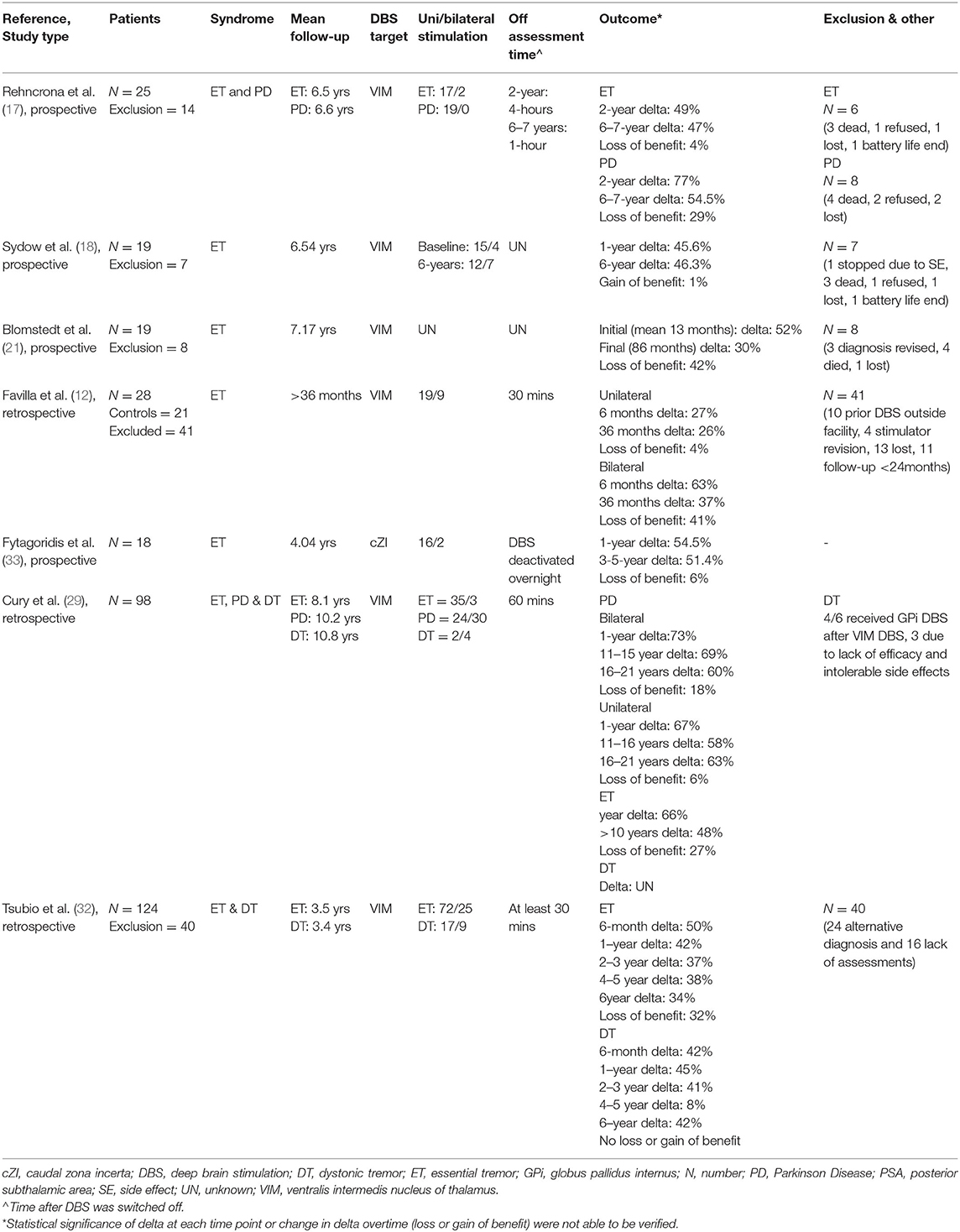
Separating natural disease progression and habituation in the context of gradual loss of DBS benefit overtime is difficult. In theory, comparing the “off” stimulation tremor severity at two different time points should only represent progression secondary to disease. While tremor severity in the “on” stimulation state over similar time points should reflect both disease progression and the stimulation effect (12, 13). Delta, the difference between the “off” and “on” state, when compared overtime, appears our best measure of any changing stimulation effect, and possible habituation. Of all the long-term DBS follow-up studies in ET, only seven (12, 17–19, 29, 32, 33) have “off/on” data at more than one defined time point, that allows the analysis of change in delta overtime, and the possible detection of habituation (Table 1). Further, the most recent study by Paschen et al. have calculated the difference in TRS score in both “off/on” states compared to baseline, allowing for statistical separation of disease progression and habituation (13).
In the seven long-term DBS studies in ET that data is available to determine the percentage of delta change over time, loss of stimulation benefit was seen in all but one study (17). The effect lost over time, as a percentage of the stimulation effect on the first assessment compared to the last assessment, ranged from 4–42% (Table 1). However, owing to the design of the studies, the statistical significance of these changes remains unclear in all but one study, where subgroup analysis revealed the loss of effect to be not significant (p > 0.05). The target of DBS stimulation was the VIM in six (12, 17–19, 29, 33) and the PSA in one (32) of these studies. In five of the studies the exclusion and drop-out rate ranged from 24–59%, with drop-outs often including patients with progressive tremor severity, unacceptable side-effects or even stimulation revision. This could possibly lead to an underestimation of the loss of stimulation benefit over time. During follow-up, stimulation parameters increased across three studies, remained unchanged or statistical insignificant across two studies and were not reported in two.
Limitations beyond the high drop-out rate exists across all these studies. Only one group look at the relationship between lead location and tremor response, despite it being a known cause for chronic loss of DBS benefit over time. Cury et al. reported that stimulation to the caudal part of the right VIM was associated with a worse tremor result at 1-year but how this related to the change in delta overtime and habituation is unknown (29). Favilla et al. who concluded that disease progression is the most likely explanation for worsening tremor after DBS (12), failed to assess the change in response to stimulation over time in patients whose tremor was either stable or improved in the “off” state. Assessment including this cohort reveals a 4 and 41% loss of stimulation benefit between 6 and 36 months in the unilateral and bilateral stimulated groups respectively. Paschen et al. attributed 13% of the worsening “stim-on” to habituation (13). However, overestimation to disease progression is likely to have occurred after the mean monthly worsening of the TRS scores in the “off” and “on” state were calculated from different time points. In which, part of the decline in the “off” state is likely due to the loss of microthalamotomy that is not reflected in the TRS stim-on scores. In the most recent study by Tsuboi et al. delta values need to be interpreted with caution as not all patients were assessed in the “off” state at each separate time point (33).
Emerging Ataxia and Rebound Tremor as Biomarkers for Habituation?
The relationship between stimulation-induced ataxia and habituation is unclear. The two entities are often addressed independently despite the clinical observation that some patients develop a progressive ataxic cerebellar syndrome after bilateral VIM DBS for tremor when the stimulation intensity is increased (41, 42). This syndrome may be characterized by dysarthria, gait unsteadiness, limb incoordination and tremor of a different quality to the original tremor syndrome and often worse (43). DBS induced ataxia is not rare occurring in a third of patients in one large series (44). The cerebellar signs may dissipate if DBS is stopped and allowed to wash-out over several days, usually revealing the original tremor syndrome which may be more manageable than the DBS induced ataxic syndrome (43, 45). Induction and reversibility of the cerebellar dysfunction in these patients implies long-term aberrant plasticity within cerebellar networks. This clinical observation has been supported by functional imaging that has demonstrated stimulation induced hypermetabolism in the cerebellar nodule, exclusive to those with this syndrome, associated with stimulation particularly in the sub-thalamic white matter, the effect linked to antidromic stimulation of cerebello-thalamic fibers (45). Furthermore, a post-mortem study identified preservation of climbing and parallel cerebellum input fibers exclusive to those who had undergone DBS (46). Intriguingly, VIM DBS has also been shown to improve gait and limb ataxia in ET patients, independent of tremor-suppressing effects, provided the stimulation intensity is not excessive, at which point ataxia is worsened (41, 42). These data point to dual contribution of both stimulation proximity to cerebellar outflow tracts and intensity on the development of ataxia after VIM DBS.
Another phenomenon is rebound tremor where tremor severity is much worse than the pre-operative baseline immediately after DBS is switched off (10, 42, 45). This phenomenon has been strongly associated with the stimulation induced ataxic cerebellar syndrome, in which less tremor suppression benefit from stimulation was also observed (45). However, there has not been a clear association between rebound tremor and habituation. Nevertheless, switching the DBS off is a regular occurrence and many DBS groups advise patients to switch their device off at night in an effort to avoid habituation; although controlled studies to confirm this hypothesis are so far lacking. Not infrequently patients with waning tremor benefit and habituation describe more marked rebound tremor with the device off, such that some may become incapable of switching their device off at night owing to unacceptable worsening of tremor (43, 47).
Deep Brain Stimulation and Dystonia–An Insight for Habituation?
DBS of the globus pallidus internus (GPi) is an effective treatment for primary dystonia (48). However, unlike other neurological conditions, maximal clinical benefit can take weeks to months (49). This often occurs with similar stimulation parameters, in the absence of an abrupt but monotonic improvement in dystonia (50). Longitudinal neurophysiological examinations have provided mechanistic insights into excessive muscle activity and overflow characteristics. After GPi DBS a quick absence of enhance per-operative plasticity is seen but the normalization of cortical inhibition takes months to achieve, following a similar time course to the clinical response (51). Further, these physiological changes do not immediately ablate after stopping the stimulation (52). The long-term effects on the cortical circuitry in dystonia are positive, but negative examples, in the form of emergent dystonia, after lesional and DBS surgery of the thalamus have been reported (53). Conceptually, habituation is likely a form of neural reorganization in a negative sense, with many similarities to the changes we observe after GPi DBS for dystonia.
Habituation and Other Tremor Syndromes
Parkinson's Disease Tremor
DBS for the management of PD is a well-established treatment (54–60). It has been shown to be superior to medical therapy in the early (54) through to advanced disease stages (60). The subthalamic nucleus (STN), GPi and VIM have all been shown to be effective DBS targets for tremor suppression (61, 62). Despite the initial reports of habituation involving a cohort predominantly of PD patients (10); little has been published in the literature since. Rechrona et al. demonstrated a 29% loss of effect with VIM stimulation over a 4–5-year follow-up period (17). More recently, Cury et al. showed up to an 18% loss of benefit with the same target over a duration of 16–21 years (29). However, other long-term follow-up studies have not demonstrated a similar waning benefit of DBS commonly associated with ET (61–65). In early-stage PD, STN DBS has been shown to slow the progression of rest tremor and provide long-term symptomatic benefit compared to standard medical care (54). Collectively these data may suggest that habituation is less likely to occur in PD tremor and less likely with DBS targets other than VIM.
Dystonic Tremor
In comparison to ET and PD the long-term effectiveness of DBS for dystonic tremor is not as well established (66). Many studies have reported on the effectiveness of GPi DBS for dystonia without reporting on tremor outcomes (67). The thalamus, commonly the VIM, is the predominant target in DT but alternative or tandem targets (GPi or STN) are often used when dystonic symptoms are more problematic (68). In a systematic review, improvement in TRS motor scores from baseline was approximately 40–50% (66). Recent studies by Tsuboi et al. and Cury et al. reported tremor suppression benefit was not significant at greater than 5–6 years after implantation (29, 32). Moreover, four of six patients in the study by Cury et al. received additional GPi stimulation due to lack of efficacy or intolerable side effects (29). Others have report the similar need to proceed to an alternative DBS target to manage persistent or emergent dystonia and/or tremor after an initial single target DBS implantation (68, 69). The delta change over time could only be assessed in the Tsuboi et al. (32) cohort, where a loss of benefit was seen at the majority of time points (Table 1). The outlying result at greater than 6 years was considered non-significant by the authors (32). It has been hypothesized that these observations represent habituation (32). There is growing evidence dystonia results from widespread multi-level network dysfunction (70), involving the basal ganglia, cerebellum and excessive motor cortical plasticity, with evidence long-term network modification after GPi DBS (71), however the long-term effects of thalamic DBS on these networks in dystonia is unknown.
Uncommon Tremor Syndromes
Apart from one randomized clinical trial in multiple sclerosis (MS)-associated tremor (72), experience with uncommon tremor syndromes; Holmes' tremor (HT) (73), orthostatic tremor (OT) (74), neuropathy-associated tremor and fragile X-associated tremor/ataxia syndrome (75); come from case reports and small case series. Habituation has been reported in neuropathic tremor from demyelinating neuropathy treated with VIM DBS, worse than a comparison ET group (76). Bi-or-unilateral VIM stimulation is the commonest modality of treatment used irrespective of tremor syndrome. Other targets of stimulation used independently or as an adjacent to the VIM included: cZi, GPi, PSA, STN, Ventro-lateral (VL), Ventralis oralis anterior or Ventralis oralis posterior nuclei (VOA/VOP) (75). Data suggests that DBS might be useful for these uncommon syndromes; but both the rarity of these conditions and heterogeneity makes the nature and magnitude of any effect uncertain (75). Furthermore, the same must be said for the development of unwanted events to stimulation including habituation.
Factors That May Predispose to Habituation
Does Habituation Differ Between Tremor Subtypes?
Attempting to identify factors that predisposed to habituation, a phenomenon hard to define and even more difficult to study, should be done with caution. However, the underlying disease seems to be an important factor in predicting long-term outcomes (29, 32). DT appears to be the tremor syndrome least responsive to DBS in the long-term and potentially the most susceptible to habituation (29, 32). Although Tsubio et al. reported comparable long-term tremor suppression results between DT and ET in VIM DBS, loss of stimulation benefit at greater than 6-years was only present in the DT cohort. Furthermore, improvement in activities of daily living tended to be greater in the ET cohort (32). Cury et al. have demonstrated a similar loss of tremor suppression benefit in DT compared with both ET and PD (29). This may reflect disease progression or emergent dystonia (10, 53) rather than habituation (32). Combined VIM and GPi DBS (77) could potentially alleviate some of these issues but long-term follow-up studies are required.
There is evidence to suggest habituation is less common in PD tremor treated with DBS. In the long-term comparison study of thalamic DBS in PD, ET and DT; greatest stimulation benefit was seen with PD tremor (Table 1) (29). In non-comparative studies of VIM, STN and GPi DBS; more stable consistent response to stimulation have been demonstrate (61–65). In some of these studies up to 50% of the cohort experience complete absence of rest tremor (62–65). One interesting observation after both VIM and STN DBS, tremor in the off-medication-off-stimulation condition is often less severe than the off-medication baseline state. This is surprising despite the knowledge that PD tremor doesn't necessarily get worse over time. Structural or neurochemical change leading to the improvement of tremor has been suggested; a persistent micro-thalamotomy effect or residual effect from electrical stimulation have not been excluded (64). This evidence would argue against habituation in PD tremor.
Does Habituation Depend on DBS Target?
The DBS target for tremor suppression is of particular interest in view of the progressive loss of tremor benefit seen with VIM DBS in ET (36). Given the important role the cerebellothalamic tract plays in tremor, the PSA/cZI has been used as an alternative DBS target. A randomized trial comparing the two targets in the treatment of ET, although not statistically significant, favored lower amplitude stimulation of the PSA for tremor control. Owing to the short follow-up duration, any implication on the development of habituation could not be assessed (78). A comparative study comparing the cZI and VIM targets in ET, demonstrated both to be beneficial for tremor suppression; but a potential long-term advantage with applying VIM stimulation due to the gradual worsening of tremor scores in patients stimulated in the cZI region (30). However, other studies have demonstrated persistent long-term benefit with PSA/cZI stimulation and an absence of habituation (33–35). Available evidence suggests PSA/cZI to be equivalent to VIM in effectiveness for tremor suppression but with lower stimulation energy requirements, likely reflecting closer electrode proximity to the dentatorubrothalamic tract. Lower energy requirements for chronic stimulation using the PSA/cZI target could confer some advantage in reducing the risk of habituation but does not appear to eliminate the problem altogether.
PD tremor cohort comparison between VIM-STN and STN-GPi stimulation has occurred with differing strengths of evidence. In a meta-analysis of five randomized control trials, STN and GPi DBS were shown to reduced tremor symptoms without significant differences between the two stimulation targets. STN DBS appeared to reduced tremor severity with a larger effect size in the short-term, while GPi DBS appears to have a steadier and more stable tremor effect in the long-term (62). Further comparison of these two targets, specifically in relationship to action and rest tremor; suggested the initial STN superiority might be due to effective action tremor suppression in the early post-operative period (79). VIM and STN DBS have been compared in a small retrospective analysis, no significant difference in degree of improvement in rest, action or postural tremor was observed (62).
Is Habituation Different With Unilateral vs. Bilateral VIM DBS?
Despite bilateral VIM DBS leading to a greater overall reduction in tremor severity (80) often owing to the bilateral and midline benefits (18), unilateral VIM DBS has been associated with more persistent tremor benefit from stimulation in ET (12). Favilla et al. (12) demonstrated the benefit from stimulation compared with baseline was consistent through 36-months for both unilateral and bilateral stimulation. However, the loss of delta overtime was 4% in the unilateral compared to 41% in the bilateral group (Table 1). A similar finding was seen in the Cury et al. PD cohort who had undergone VIM DBS, tremor suppression was maintain in both unilateral and bilateral groups through 16–21 years of follow-up, but the loss of delta over time was more pronounced in the bilaterally treated group (29) (Table 1). Both observations may reflect a possible propensity for habituation with bilateral stimulation despite the tremor syndrome. Conceptually, bilateral stimulation could exert greater plastic force on cerebellar networks to adopt abnormal configurations, with less potential for compensation from an untreated side. Other factors that should be considered when counseling patients regarding the possible development of habituation include asymmetry of the tremor syndrome, stimulation intensity and continuous vs. interrupted stimulation.
Prevention and Management of Habituation to DBS for Tremor
The accuracy of lead placement is critical to avoid early loss of benefit after VIM or PSA/cZI DBS however this problem, by definition, is distinct from true habituation. For patients with well-placed DBS leads possible preventative strategies for habituation include conservative parameter setting to avoid overstimulation and instructing the patient to switch the device off at night (8). Patients may also be advised to only use stimulation the day when needed rather than continuously, in an on-demand fashion, which may reduce the risk of habituation (81). In clinical practice when patients return in long term follow up and report declining tremor benefit, there is a temptation to reprogram usually with increased stimulation current, which often improves tremor but only temporarily. Such increases when performed repeatedly over time may result in chronic DBS-induced ataxic syndrome. An alternate strategy is to refrain from increasing the DBS, clinical worsening is mild and provided patients remain significantly improved compared with the pre-operative baseline.
There are a few studies evaluating varying stimulation programs in an attempt to reduce the occurrence of habitation. Seier and colleagues found reduce benefit decay in patients varying between two equally effective stimulation programs weekly after 12 weeks compared with those receiving unvarying programs (82). However, another study using daily variation of stimulation programs found no superiority to unvarying stimulation at 10 weeks (83). In clinical practice, with limited options available, installing different effective programming groups for patients to vary between is feasible (84), and may reduce habituation, although weekly changes appear more effective than daily. Cycling of stimulation between ON and OFF in blocks of 1–30 s was reported as helpful in reducing habituation and rebound tremor in three PD tremor patients (85).
For patients with established benefit decay and habituation, particularly those with stimulation induced ataxia, reduction of stimulation current may be helpful, and can be more achievable if performed gradually and predominantly unilaterally. Cessation of stimulation for a few days can be attempted, preferably supervised in hospital, and after initial rebound tremor passes, dissipation of DBS induced cerebellar ataxia is expected with a return to the baseline tremor syndrome, and potential functional improvement if ataxia has become the predominant driver for disability. Cessation of stimulation provides an opportunity to reappraise DBS effectiveness in the On vs. Off stimulation condition. Some patients will experience improvement in DBS effectiveness after a period of complete DBS cessation, so-called “stimulation holiday”, however such improvements are usually unsustained but can be repeated in an attempt to recapture benefits lost to habituation (47).
Related to prevention of habituation should be an attempt to stratify the risk of its occurrence in tremor patients being considered for DBS. The tremor subtype is relevant with PD patients less likely to develop habituation than those with ET or DT. Tremor phenomenology may also be useful in predicting risk of habituation, with patients with more action-predominant tremor (7) and those with signs of cerebellar ataxia at greater risk (37).
Surgical approaches to habitation may include reimplantation of DBS with a repositioned electrode optimized to closer proximity to the dentatorubrothalamic tract (as imaged by diffusion tensor tractography MRI) which may achieve superior tremor control with less habituation (86). Moreover, there is growing evidence that closer targeting of MRI-visualized dentatorubrothalamic tract (DRT) provides more effective and efficient tremor control (87, 88) and it remains to be seen whether this approach of targeting DRT deliberately will confer lower rates of habituation in the longer term. Additional “rescue” leads have been implanted in targets including VIM, PSA/cZI, VOA and STN with moderate additional benefit (89, 90). Thalamotomy has been performed after failed VIM DBS including cases with waning benefit and habituation, with modest additional benefit (91).
The advent of more advanced DBS hardware and programming capabilities including independent, constant current directional leads, implanted pulse generators allowing shorter pulse widths <60 μs and sensing of local field potential (LFP) spectra hold promise to assist in the prevention and long-term management of habituation after tremor DBS. Directional leads allow shaped stimulation fields to maximize benefit with fewer side effects, widening the therapeutic window (92, 93). Similarly shorter pulse widths allow greater stimulation current to be delivered without provoking side effects (94, 95). DBS devices allowing real-time recording of LFPs foreshadow closed loop stimulation; the first study demonstrating successful ambulatory recording of VIM and cZI LFPs corresponding to voluntary movements and tremor with highly effective tremor suppression when DBS was delivered closed-loop triggered by LFP activity (96). Of relevance to closed-loop approaches is the important discovery that delivery of DBS stimuli in specific relation to the phase of tremor (phase-locked stimulation) is more effective and efficient with fewer side effects (97).
Conclusions
Habituation is a real phenomenon after DBS for tremor and is a contributory factor to waning clinical benefit after tremor DBS. The other major contributor to waning benefit is disease progression of the underlying tremor syndrome. Instances of more dramatic loss of clinical benefit over shorter timeframes (short term habituation) may occur and induction of progressive ataxic cerebellar symptoms suggesting aberrant plasticity within cerebellar networks targeted by VIM and PSA DBS. Our current mechanistic understanding of habituation is incomplete and further neurophysiological and imaging studies will be required to elucidate the pathophysiology. In clinical practice, habituation after DBS for tremor remains a feared complication, with available preventative strategies limited to interrupted stimulation regimens (typically switching off at night), minimizing stimulation current or varying programs. It remains to be seen whether technological innovations in DBS such as deliberate MRI-guided targeting of the DRT, directional leads, lower pulse widths or advanced stimulation methods such as phase dependent or on demand DBS will reduce the problem of habituation. In the meantime, it is important that habituation and disease progression of tremor be explained to patients prior to DBS as factors that may result in reduction in clinical benefit over time.
Author Contributions
JP and ST contributed to the first draft and revision of the manuscript. All authors contributed to the article and approved the submitted version.
Funding
ST has received research funding from the Cunningham Family Foundation.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher's Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Bhatia KP, Bain P, Bajaj N, Elble RJ, Hallett M, Louis ED, et al. Consensus Statement on the classification of tremors. from the task force on tremor of the International Parkinson and Movement Disorder Society. Mov Disord. (2018) 33:75–87. doi: 10.1002/mds.27121
2. Wenning GK, Kiechl S, Seppi K, Muller J, Hogl B, Saletu M, et al. Prevalence of movement disorders in men and women aged 50-89 years (Bruneck Study cohort): a population-based study. Lancet Neurol. (2005) 4:815–20. doi: 10.1016/S1474-4422(05)70226-X
3. Cooper IS. Ligation of anterior choroidal artery for involuntary movements parkinsonism. Psychiatric Q. (1953) 27:317–9. doi: 10.1007/BF01562492
4. Hassler R, Riechert T, Mundinger F, Umbach W, Ganglberger JA. Physiological observations in stereotaxic operations in extrapyramidal motor disturbances. Brain. (1960) 83:337–50. doi: 10.1093/brain/83.2.337
5. Walker AE. Stereotactic surgery for tremor. In Schaltenbrand G, Walker AE eds. Stereotaxy of the Human Brain: Anatomical, Physiological and Clinical Applications. Georg Thieme Verlag, Stuttgart (1982). p. 515–21.
6. Cooper S, Upton AR, Amin I. Reversibility of chronic neurologic deficits. Some effects of electrical stimulation of the thalamus and internal capsule in man. Appl Neurophysiol. (1980) 43:244–58. doi: 10.1159/000102263
7. Benabid AL, Pollak P, Gervason C, Hoffmann D, Gao DM, Hommel M, et al. Long-term suppression of tremor by chronic stimulation of the ventral intermediate thalamic nucleus. Lancet. (1991) 337:403–6. doi: 10.1016/0140-6736(91)91175-T
8. Limousin P, Speelman JD, Gielen F, Janssens M. Multicentre European study of thalamic stimulation in parkinsonian and essential tremor. J Neurol Neurosurg Psychiatry. (1999) 66:289–96. doi: 10.1136/jnnp.66.3.289
9. Schuurman PR, Bosch DA, Bossuyt PM, Bonsel GJ, van Someren EJ, de Bie RM, et al. A comparison of continuous thalamic stimulation and thalamotomy for suppression of severe tremor. N Engl J Med. (2000) 342:461–8. doi: 10.1056/NEJM200002173420703
10. Benabid AL, Pollak P, Gao D, Hoffmann D, Limousin P, Gay E, et al. Chronic electrical stimulation of the ventralis intermedius nucleus of the thalamus as a treatment of movement disorders. J Neursurg. (1996) 84:203–14. doi: 10.3171/jns.1996.84.2.0203
11. Fasano A, Helmich RC. Tremor habituation to deep brain stimulation: Underlying mechanisms and solutions. Mov Disord. (2019) 34:1761–73. doi: 10.1002/mds.27821
12. Favilla CG, Ullman D, Shukla AW, Foote KD, Jacobson CE, Okun MS. Worsening essential tremor following deep brain stimulation: disease progression versus tolerance. Brain. (2012) 135:1455–62. doi: 10.1093/brain/aws026
13. Paschen S, Forstenpointner J, Becktepe J, Heinzel S, Hellriegel H, Witt K, et al. Long-term efficacy of deep brain stimulation for essential tremor: an observer-blinded study. Neurology. (2019) 92:e1378–86. doi: 10.1212/WNL.0000000000007134
14. Papavassiliou E, Rau G, Health S, Abosch A, Barbaro NM, Larson PS. Thalamic deep brain stimulation for essential tremor: relation of lead location to outcome. Neurosurgery. (2004) 54:1120–9. doi: 10.1227/01.NEU.0000119329.66931.9E
15. Moro E, Poon YY, Lozano A, Saint-Cyr JA, Lang AE. Subthalamic nucleus stimulation: improvements in outcome with reprogramming. Arch Neurol. (2006) 63:1266–72. doi: 10.1001/archneur.63.9.noc60069
16. Koller WC, Lyons KE, Wilkinson SB, Troster AI, Pahwa R. Long-term safety and efficacy of unilateral deep brain stimulation of the thalamus in essential tremor. Mov Disord. (2001) 16:464–8. doi: 10.1002/mds.1089
17. Rehncrona S, Johnels B, Widner H, Tornqvist AL, Hariz M, Sydow O. Long-term efficacy of thalamic deep brain stimulation for tremor: double-blind assessments. Mov Disord. (2002) 18:163–70. doi: 10.1002/mds.10309
18. Sydow O, Thobois S, Alesch F, Speelman JD. Multicentre European study of thalamic stimulation in essential tremor: a six year follow up. J Neurol Neurosurg Psychiatry. (2003) 74:1387–91. doi: 10.1136/jnnp.74.10.1387
19. Putzke JD, Wharen RE, Obwegeser AA, Wszolek ZK, Lucas JA, Turk MF, et al. Thalamic deep brain stimulation for essential tremor: recommendations for longterm outcome analysis. Can J Neurol Sci. (2004) 31:333–42. doi: 10.1017/S0317167100003413
20. Pahwa R, Lyons KE, Wilkinson SB, Simpson RK, Ondo WG, Tarsy D, et al. Long-term evaluation of deep brain stimulation of the thalamus. J Neurosurg. (2006) 104:506–12. doi: 10.3171/jns.2006.104.4.506
21. Blomstedt P, Hariz GM, Hariz MI, Koskinen LOD. Thalamic deep brain stimulation in the treatment of essential tremor: a long-term follow-up. Br J Neurosurg. (2007) 21:504–9. doi: 10.1080/02688690701552278
22. Schuurman PR, Bosch DA, Merkus MP, Speelman JD. Long-term follow-up of thalamic stimulation versus thalamotomy for tremor suppression. Mov Disord. (2008) 23:1146–53. doi: 10.1002/mds.22059
23. Zhang K, Bhatia S, Oh MY, Cohen D, Angle C, Whiting D. Long-term results of thalamic deep brain stimulation for essential tremor. J Neurosurg. (2010) 112:1271–6. doi: 10.3171/2009.10.JNS09371
24. Nazzaro JM, Pahwa R, Lyons KE. Long-term benefits in quality of life after unilateral thalamic deep brain stimulation for essential tremor. J Neurosurg. (2012) 117:156–61. doi: 10.3171/2012.3.JNS112316
25. Shih LC, LaFaver K, Lim C, Papavassiliou E, Tarsy D. Loss of benefit in VIM thalamic deep brain stimulation (DBS) for essential tremor (ET): how prevalent is it? Parkinsonism Relat Disord. (2013) 19:676–9. doi: 10.1016/j.parkreldis.2013.03.006
26. Baizabal-Carvallo JF, Kagnoff MN, Jimenez-Shahed J, Fekete R, Jankovic J. The safety and efficacy of thalamic deep brain stimulation in essential tremor: 10 years and beyond. J Neurol Neurosurg Psychiatry. (2013) 85:567–72. doi: 10.1136/jnnp-2013-304943
27. Borretzen MN, Bjerknes S, Saehle T, Skjelland M, Skogseid IM, Toft M, et al. Long-term follow-up of thalamic deep brain stimulation for essential tremor - patient satisfaction and mortality. BMC Neurol. (2014) 14:120. doi: 10.1186/1471-2377-14-120
28. Rodriguez Cruz PM, Vargas A, Fernandez-Carballal C, Garbizu J, De La Casa-Fages B, Grandas F. Long-term thalamic deep brain stimulation for essential tremor: clinical outcome and stimulation parameters. Mov Disord Clin Pract. (2016) 3:567–72. doi: 10.1002/mdc3.12337
29. Cury RG, Fraix V, Castrioto A, Perez Fernandez MA, Krack P, Chabardes S, et al. Thalamic deep brain stimulation for tremor in Parkinson disease, essential tremor, and dystonia. Neurology. (2017) 89:1416–23. doi: 10.1212/WNL.0000000000004295
30. Eisinger RS, Wong J, Almeida L, Ramirez-Zamora A, Cagle JN, Giugni JC. Ventral intermediate nucleus versus zona incerta region deep brain stimulation in essential tremor. Mov Disord Clin Pract. (2018) 5:75–82. doi: 10.1002/mdc3.12565
31. Sandoe C, Krishna V, Basha D, Sammartino F, Tatsch J, Picillo M, et al. Predictors of deep brain stimulation outcome in tremor patients. Brain Stimul. (2018) 11:592–9. doi: 10.1016/j.brs.2017.12.014
32. Tsuboi T, Jabarkheel Z, Zeilman PR, Barabas MJ, Foote KD, Okun MS, et al. Longitudinal follow-up with VIM thalamic deep brain stimulation for dystonic or essential tremor. Neurology. (2020) 94:e1073–84. doi: 10.1212/WNL.0000000000008875
33. Fytagoridis A, Sandvik U, Astrom M, Bergenheim T, Blomstedt P. Long term follow-up of deep brain stimulation of the caudal zona incerta for essential tremor. J Neurol Neurosurg Psychiatry. (2012) 83:258–62. doi: 10.1136/jnnp-2011-300765
34. Plaha P, Javed S, Agombar D, O'Farrell G, Khan S, Whone A, et al. Bilateral caudal zona incerta nucleus stimulation for essential tremor: outcome and quality of life. J Neurol Neurosurg Psychiatry. (2011) 82:899–904. doi: 10.1136/jnnp.2010.222992
35. Blomstedt P, Sandvik U, Tisch S. Deep brain stimulation in the posterior subthalamic area in the treatment of essential tremor. Mov Disord. (2010) 25:1350–6. doi: 10.1002/mds.22758
36. Lu G, Luo L, Liu M, Zheng Z, Zhang B, Chen X, et al. Outcomes and adverse effects of deep brain stimulation on the ventral intermediate nucleus in patients with essential tremor. Neural Plast. (2020) 2020:2486065. doi: 10.1155/2020/2486065
37. Okun M, Tagliati M, Pourfar M, Fernandez HH, Rodriguez RL, Alterman RL, Foote KD. Management of referred deep brain stimulation failures: a retrospective analysis from 2 movement disorders centers. Arch Neurol. (2005) 62:1250–5. doi: 10.1001/archneur.62.8.noc40425
38. Merchant SH, Kuo SH, Qiping Y, Winfield L, McKhann G, Sheth S, et al. Objective predictors of ‘early tolerance' to ventral intermediate nucleus of thalamus deep brain stimulation in essential tremor patients. Clin Neurophysiol. (2018) 129:1628–33. doi: 10.1016/j.clinph.2018.05.012
39. Ellis TM, Foote KD, Fernandez HH, Sudhyadom A, Rodriguez RL, Zeilman P, et al. Reoperation for suboptimal outcomes after deep brain stimulation surgery. Neurosurgery. (2008) 63:754–60. doi: 10.1227/01.NEU.0000325492.58799.35
40. Barbe MT, Liebhart L, Runge M, Pauls KA, Wojtecki L, Schnitzler A, et al. Deep brain stimulation in the nucleus ventralis intermedius in patients with essential tremor: habituation of tremor suppression. J Neurol. (2011) 258:434–9. doi: 10.1007/s00415-010-5773-3
41. Fasano A, Herzog J, Raethjen J, Rose FE, Muthuraman M, Volkmann J, et al. Gait ataxia in essential tremor is differentially modulated by thalamic stimulation. Brain. (2010) 133:3635–48. doi: 10.1093/brain/awq267
42. Groppa S, Herzog J, Falk D, Riedel C, Deuschl G, Volkmann J. Physiological and anatomical decomposition of subthalamic neurostimulation effects in essential tremor. Brain. (2014) 137:109–21. doi: 10.1093/brain/awt304
43. Hariz MI, Shamsgovara P, Johansson F, Hariz G, Fodstad H. Tolerance and tremor rebound following long-term chronic thalamic stimulation for Parkinsonian and essential tremor. Stereotact Funct Neurosurg. (1999) 72:208–18. doi: 10.1159/000029728
44. Chiu SY, Nozile-Firth K, Klassen BT, Adams A, Lee K, Van Gompel JJ, et al. Ataxia and tolerance after thalamic deep brain stimulation for essential tremor. Parkinsonism Relat Disord. (2020) 80:47–53. doi: 10.1016/j.parkreldis.2020.09.009
45. Reich MM, Brumberg J, Pozzi NG, Marotta G, Roothans J, Astrom M, et al. Progressive gait ataxia following deep brain stimulation for essential tremor: adverse effect or lack of efficacy? Brain. (2016) 139:2948–56. doi: 10.1093/brain/aww223
46. Kuo SH, Lin CY, Wang J, Liou JY, Pan MK, Louis RJ, et al. Deep brain stimulation and climbing fiber synaptic pathology in essential tremor. Ann Neurol. (2016) 80:461–5. doi: 10.1002/ana.24728
47. Garcia Ruiz P, Muniz de, Igneson J, Lopez Ferro O, Martin C, Magarinos Ascone C. Deep brain stimulation holidays in essential tremor. J Neurol. (2001) 248:725–6. doi: 10.1007/s004150170127
48. Cif L, Vasques X, Gonzalez V, Ravel P, Biolsi B, Collod-Beroud G, et al. Long-term follow-up of DYT1 dystonia patients treated by deep brain stimulation: an open-label study. Mov Disord. (2010) 25:289–99. doi: 10.1002/mds.22802
49. Tisch S, Limousin P, Rothwell JC, Asselman P, Zrinzo L, Jahanshahi M, et al. Changes in forearm reciprocal inhibition following pallidal stimulation for dystonia. Neurology. (2006) 66:1091–3. doi: 10.1212/01.wnl.0000204649.36458.8f
50. Hammond C, Ammari R, Bioulac B, Garcia L. Latest view on the mechanism of action of deep brain stimulation. Mov Disord. (2008) 23:2111–21. doi: 10.1002/mds.22120
51. Ruge D, Tisch S, Hariz MI, Zrinzo L, Bhatia KP, Quinn NP, et al. Deep brain stimulation effects in dystonia: time course of electrophysiological changes in early treatment. Mov Disord. (2011) 26:1913–21. doi: 10.1002/mds.23731
52. Ruge D, Cif L, Limousin P, Gonzalez V, Vasques X, Hariz MI, et al. Shaping reversibility? Long-term deep brain stimulation in dystonia: the relationship between effects on electrophysiology and clinical symptoms. Brain. (2011) 134:2106–15. doi: 10.1093/brain/awr122
53. Picillo M, Paramanandam V, Morgante F, Algarni M, Olszewska DA, Munhoz RP, et al. Dystonia as complication of thalamic neurosurgery. Parkinsonism Relat Disod. (2019) 66:232–6. doi: 10.1016/j.parkreldis.2019.08.008
54. Hacker ML, Turchan M, Heusinkveld LE, Currie AD, Millan SH, Molinari AL, et al. Deep-brain stimulation in early stage Parkinson disease. Neurology. (2020) 95:383–401. doi: 10.1212/WNL.0000000000009946
55. Obeso JA, Olanow CW, Rodriguez-Oroz MC, Krack P, Kumar R, Lang AE. Deep-brain stimulation of the subthalamic nucleus or the pars interna of the globus pallidus in Parkinson's disease, N. Engl J Med. (2001) 345:956–63. doi: 10.1056/NEJMoa000827
56. Anderson VC, Burchiel KJ, Hogarth P, Favre J. Hammerstad JP. Pallidal vs subthalamic nucleus deep brain stimulation in Parkinson disease. Arch Neurol. (2005) 62:554–60. doi: 10.1001/archneur.62.4.554
57. Okun MS, Gallo BV, Mandybur G, Jagid J, Foote KD, Revilla FJ, et al. Subthalamic deep brain stimulation with a constant-current device in Parkinson's disease: an open-label randomised controlled trial. Lancet Neurol. (2012) 11:140–9. doi: 10.1016/S1474-4422(11)70308-8
58. Odekerken VJ, van Laar T, Staal MJ, Mosch A, Hoffmann CF, Nijssen PC, et al. Subthalamic nucleus versus globus pallidus bilateral deep brain stimulation for advanced Parkinson's disease (NSTAPS study): a randomised controlled trial. Lancet Neurol. (2013) 12:37–44. doi: 10.1016/S1474-4422(12)70264-8
59. Weaver F, Follett K, Stern M, Luo P, Harris CL, Hur K, et al. Randomized trial of deep brain stimulation for Parkinson disease: thirty-six-month outcomes. Neurology. (2012) 79:55–65. doi: 10.1212/WNL.0b013e31825dcdc1
60. Weaver F, Follett K, Stern M, Hur K, Harris C, Marks WJ, et al. Bilateral deep brain stimulation vs best medical therapy for patient with advance Parkinson disease: A randomized controlled trial. JAMA. (2009) 301:63. doi: 10.1001/jama.2008.929
61. Wong JK, Cauraugh JH, Ho KWD, Broderick M, Ramirez-Zamora A, Almeida L, et al. STN vs. GPi deep brain stimulation for tremor suppression in Parkinson disease: a systematic review and meta-analysis Parkinsonism Relat Disord. (2019) 58:56–62. doi: 10.1016/j.parkreldis.2018.08.017
62. Parihar R, Alterman R, Papavassiliou E, Tarsy D, Shih LC. Comparison of VIM and STN DBS for Parkinsonian resting and postural/action tremor. Tremor Other Hyperkinet Mov. (2015) 6:321. doi: 10.5334/tohm.247
63. Hariz MI, Krack P, Alesch F, Augustinsson L-E, Bosch A, Ekberg R, et al. Multicetre European study of thalamic stimulation for parkinsonian tremor: a 6 year follow-up. J Neurol Neurosurg Psychiatry. (2008) 79:694–9. doi: 10.1136/jnnp.2007.118653
64. Kim HJ, Jeon BS, Paek SH, Lee JY, Kim HJ, Kim CK, et al. Bilateral subthalamic deep brain stimulation in Parkinson disease patients with severe tremor. Neurosurgery. (2010) 67:626–32. doi: 10.1227/01.NEU.0000374850.98949.D4
65. Savica R, Masumoto JY, Josephs KA, Ahlskog JE, Stead M, Lee KH, et al. Deep brain stimulation in Benign Tremulous parkinsonism. Arch Neurol. (2011) 68:1033–6. doi: 10.1001/archneurol.2011.160
66. Tusboi T, Au KL, Deeb W, Almeida L, Foote KD, Okun MS, et al. Motor outcomes and adverse effects of deep brain stimulation for dystonic tremor: a systematic review. Parkinsonism Relat Disord. (2020) 76:32–41. doi: 10.1016/j.parkreldis.2020.06.008
67. Moro E, LeReun C, Krauss JK, Albanese A, Lin J-P, Walleser Autiero S, et al. Efficacy of pallidal stimulation in isolated dystonia: a systematic review and meta-analysis. Eur J Neurol. (2017) 24:552–60. doi: 10.1111/ene.13255
68. Blomstedt P, Hariz MI, Tisch S, Holmberg M, Bergenheim TA, Forsgren L, et al. family with a hereditary form of torsion dystonia from northern Sweden treated with bilateral pallidal deep brain stimulation. Mov Disord. (2009) 24:2415–9. doi: 10.1002/mds.22842
69. Tsuboi T, Jabarkheel K, Foote KD. Importance of the initial response to GPi deep brain stimulation in dystonia: a nine year quality of life study. Parkinsonism Relat Disord. (2019) 64:249–55. doi: 10.1016/j.parkreldis.2019.04.024
70. Jinnah HA, Neychev V, Hess EJ. The anatomical basis for dystonia: the motor network model. Tremor Other Hyperkinet Mov. (2017) 7:506. doi: 10.5334/tohm.383
71. Tisch S, Limousin P. Neurophysiological insights in dystonia and its response to deep brain stimulation. Exp Brain Res. (2020) 238:1645–57. doi: 10.1007/s00221-020-05833-8
72. Oliveria SF, Rodriguez RL, Bowers D, Kantor D, Hilliard JD, Monari EH, et al. Safety and efficacy of dual-lead thalamic deep brain stimulation for patients with treatment-refractory multiple sclerosis tremor: a single-centre, randomised, single-blind, pilot trial. Lancet Neurol. (2017) 16:691–700. doi: 10.1016/S1474-4422(17)30166-7
73. Espinoza Martinez JA, Arango GJ, Fonoff ET, Reithmeier T, Escobar OA, Furlanetti L, et al. Deep brain stimulation of the globus pallidus internus or ventralis intermedius nucleus of thalamus for Holmes tremor. Neurosurg Rev. (2015) 38:753–63. doi: 10.1007/s10143-015-0636-0
74. Lyons MK, Behbahani M, Boucher OK, Caviness JN, Evidente VG. Orthostatic tremor responds to bilateral thalamic deep brain stimulation. Tremor Other Hyperkinet Mov. (2012) 8:566. doi: 10.5334/tohm.98
75. Artusi CA, Farooqi A, Romagnolo A, Marsili L, Balestrino R, Sokol LL, et al. Deep brain stimulation in uncommon tremor disorders: indications, targets, and programming. J Neurol. (2018) 265:2473–93. doi: 10.1007/s00415-018-8823-x
76. Patel N, Ondo W, Jimenez-Shahed J. Habituation and rebound to thalamic deep brain stimulation in long-term management of tremor associated with demyelinating neuropathy. Int J Neurosci. (2014) 124:919–25. doi: 10.3109/00207454.2014.895345
77. Hedera P, Phibbs FT, Dolhun R, Charles PD, Konrad PE, Neimat JS, et al. Surgical targets for dystonic tremor: considerations between the globus pallidus and ventral intermediate thalamic nucleus. Park Relat Disord. (2013) 19:684–6. doi: 10.1016/j.parkreldis.2013.03.010
78. Barbe MT, Reker P, Hamacher S, Franklin J, Kraus D, Dembek TA, et al. DBS of the PSA and the VIM in essential tremor: a randomized, double-blinded, crossover trial. Neurology. (2018) 9:e543–50. doi: 10.1212/WNL.0000000000005956
79. Wong JK, Viswanathan VT, Nozile-Firth KS, Eisinger RS, Leone EL, Desai AM, et al. STN versus GPi deep brain stimulation for action and rest tremor in Parkinson's disease. Front Hum Neurosci. (2020) 14:578615. doi: 10.3389/fnhum.2020.578615
80. Dallapiazza RF, Lee DJ, De Vloo P, Fomenko A, Hamani C, Hodaie M, et al. Outcomes from stereotactic surgery for essential tremor. J Neurol Neurosurg Psychiatry. (2019) 90:474–82. doi: 10.1136/jnnp-2018-318240
81. Kronenbuerger M, Fromm C, Block F, Coenen VA, Rohde I, Rohde V, et al. On-demand deep brain stimulation for essential tremor: a report on four cases. Mov Disord. (2006) 21:401–5. doi: 10.1002/mds.20714
82. Seier M, Hiller A, Quinn J, Murchison C, Brodsky M, Anderson S. Alternating thalamic deep brain stimulation for essential tremor: a trial to reduce habituation. Mov Disord Clin Pract. (2018) 5:620–6. doi: 10.1002/mdc3.12685
83. Petry-Schmelzer JN, Reker P, Pochmann J, Visser-Vandewalle V, Dembek TA, Barbe MT. Daily alternation of DBS settings does not prevent habituation of tremor suppression in essential tremor patients. Mov Disord Clin Pract. (2019) 6:417–8. doi: 10.1002/mdc3.12777
84. Barbe MT, Pochmann J, Lewis CJ, Allert N, Wirths J, Visser-Vandewalle V, et al. Utilization of predefined stimulation groups by essential tremor patients treated with VIMDBS. Parkinsonism Relat Disord. (2014) 20:1415–8. doi: 10.1016/j.parkreldis.2014.09.021
85. Enatsu R, Kitagawa M, Morishita T, Sasagawa A, Kuribara T, Hirano T, et al. Effect of cycling thalamosubthalamic stimulation on tremor habituation and rebound in parkinson disease. World Neurosurg. (2020) 144:64–7. doi: 10.1016/j.wneu.2020.08.141
86. Anthofer JM, Steib K, Lange M, Ruthenfusser E, Fellner C, Brawanski A, et al. Distance between active electrode contacts and dentatorubrothalamic tract in patients with habituation of stimulation effect of deep brain stimulation in essential tremor. Neurol Surg A Cent Eur Neurosurg. (2017) 78:350–7. doi: 10.1055/s-0036-1597894
87. Dembek TA, Petry-Schmelzer JN, Reker P, Wirths J, Hamacher S, Steffen J, et al. PSA and VIM DBS efficiency in essential tremor depends on distance to the dentatorubrothalamic tract. Neuroimage Clin. (2020) 26:102235. doi: 10.1016/j.nicl.2020.102235
88. Yang AI, Buch VP, Heman-Ackah SM, Ramayya AG, Hitti FL, Beatson N. Thalamic deep brain stimulation for essential tremor: relation of the dentatorubrothalamic tract with stimulation parameters. World Neurosurg. (2020) 137:e89–97. doi: 10.1016/j.wneu.2020.01.039
89. Blomstedt P, Lindvall P, Linder J, Olivecrona M, Forsgren L, Hariz MI. Reoperation after failed deep brain stimulation for essential tremor. World Neurosurg. (2012) 78:554.e1–5. doi: 10.1016/j.wneu.2011.12.013
90. Oyama G, Foote KD, Hwynn N, Jacobson CE, Malaty IA, Rodriguez RL. Rescue leads: a salvage technique for selected patients with a suboptimal response to standard DBS therapy. Parkinsonism Relat Disord. (2011) 17:451–5. doi: 10.1016/j.parkreldis.2011.03.009
91. Bahgat D, Magill ST, Berk C, McCartney S, Burchiel KJ. Thalamotomy as a treatment option for tremor after ineffective deep brain stimulation. Stereotact Funct Neurosurg. (2013) 91:18–23. doi: 10.1159/000342491
92. Steigerwald F, Muller L, Johannes S, Matthies C, Volkmann J. Directional deep brain stimulation of the subthalamic nucleus: A pilot study using a novel neurostimulation device. Mov Disord. (2016) 31:1240–3. doi: 10.1002/mds.26669
93. Rebelo P, Green AL, Aziz TZ, Kent A, Schafer D, Venkatesan L. Thalamic directional deep brain stimulation for tremor: spend less, get more. Brain Stimul. (2018) 11:600–6. doi: 10.1016/j.brs.2017.12.015
94. Bouthour W, Wegrzyk J, Momjian S, Peron J, Fleury V, Tomkova Chaoui E, et al. Short pulse width in subthalamic stimulation in Parkinson's disease: a randomized, double-blind study. Mov Disord. (2018) 33:169–17. doi: 10.1002/mds.27265
95. Kroneberg D, Ewert S, Meyer AC, Kuhn AA. Shorter pulse width reduces gait disturbances following deep brain stimulation for essential tremor. J Neurol Neurosurg Psychiatry. (2019) 90:1046–50. doi: 10.1136/jnnp-2018-319427
96. He S, Debarros J, Khawaldeh S, Pogosyan A, Mostofi A, Baig F, et al. Closed-loop DBS triggered by real-time movement and tremor decoding based on thalamic LFPs for essential tremor. In: 42nd Annual International Conference of the IEEE Engineering in Medicine & Biology Society (EMBC). (2020). p. 3602–5. doi: 10.1109/EMBC44109.2020.9175433
Keywords: tremor, deep brain stimulation, habituation, dystonic tremor, essential tremor, Parkinosn's disease
Citation: Peters J and Tisch S (2021) Habituation After Deep Brain Stimulation in Tremor Syndromes: Prevalence, Risk Factors and Long-Term Outcomes. Front. Neurol. 12:696950. doi: 10.3389/fneur.2021.696950
Received: 18 April 2021; Accepted: 02 July 2021;
Published: 03 August 2021.
Edited by:
Sanjay Pandey, University of Delhi, IndiaReviewed by:
Adolfo Ramirez-Zamora, University of Florida Health, United StatesErwin Montgomery, McMaster University, Canada
Copyright © 2021 Peters and Tisch. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Stephen Tisch, c3RlcGhlbi50aXNjaEBzdmhhLm9yZy5hdQ==
 James Peters
James Peters Stephen Tisch
Stephen Tisch