- 1Department of Orthopedic Surgery, Division of Sports Medicine, Hip Preservation Center, Rush University Medical Center, Chicago, IL, USA
- 2Division of Sports Medicine, Center for Hip Preservation, New England Baptist Hospital, Boston, MA, USA
Femoroacetabular impingement (FAI) is a clinical syndrome resulting from abnormal hip joint morphology and is a common cause of hip pain in young adults. FAI has been posited as a precursor to hip osteoarthritis (OA); however, conflicting evidence exists and the true natural history of the disease is unclear. The purpose of this article is to review the current understanding of how FAI damages the hip joint by highlighting its pathomechanics and etiology. We then review the current evidence relating FAI to OA. Lastly, we will discuss the potential of hip preservation surgery to alter the natural history of FAI, reduce the risk of developing OA and the need for future arthroplasty.
Introduction
The management of femoroacetabular impingement (FAI) is a rapidly developing field in orthopedics. Described by Ganz in 2003, FAI is a pathologic condition resulting from abnormal acetabular and femoral head/neck morphology that has been implicated as a precursor to secondary osteoarthritis (OA) (1–3). However, the relationship between FAI and OA is not straightforward as there exists a large asymptomatic population and without radiographic signs of OA that possesses the morphologic characteristics of FAI (4). While initially managed conservatively, symptomatic FAI is often treated surgically with the goals of relieving pain, increasing range of motion, and preventing or delaying OA and the potential need for total hip arthroplasty (THA). As FAI is increasingly diagnosed in a younger and more active population, the link between high intensity athletic participation during adolescence and the onset of FAI is under investigation (5). The purpose of this article is to review our current understanding of FAI by focusing on the mechanisms of injury, etiology, treatment strategies, and the debate about its predisposition to OA.
How Does FAI Damage the Hip Joint?
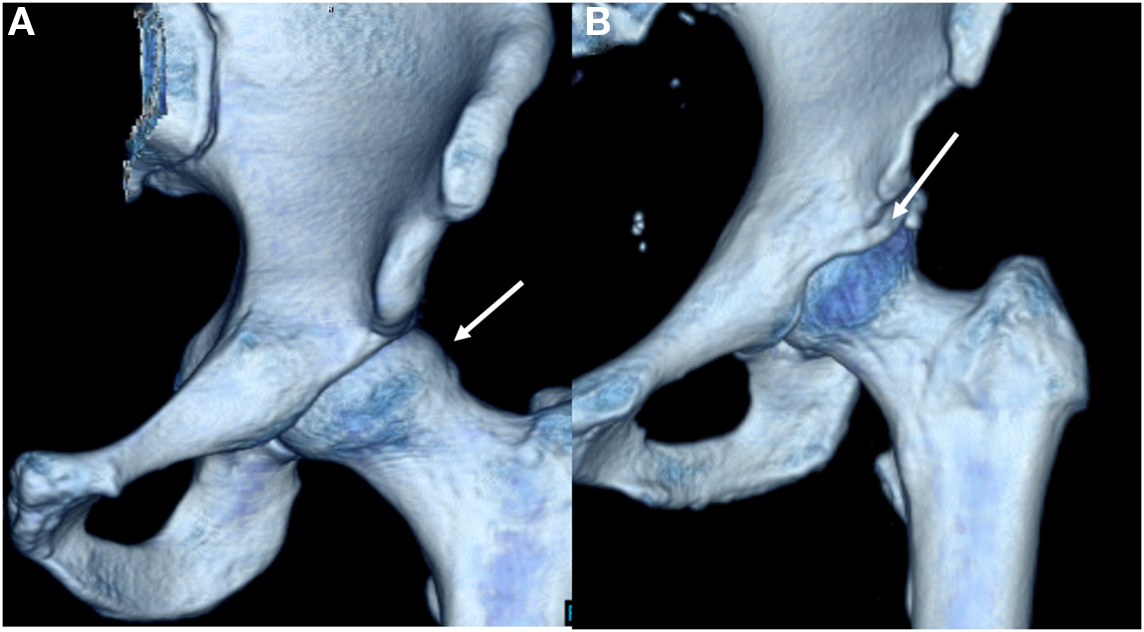
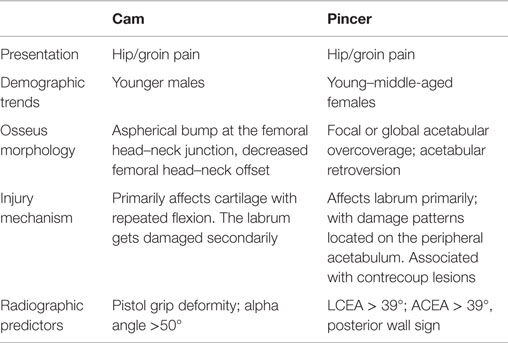
Femoroacetabular impingement results from femoral and acetabular incongruity that induces labral, and chondral damage, causing pain and restricting mobility. Cam lesions at the femoral head/neck junction as well as pincer lesions signifying acetabular overcoverage comprise the osseous deformities of FAI (Figure 1A) (1, 6). Termed “mixed” lesions, commonly FAI is a combination of both with varying degrees, but cam and pincer lesions also occur in isolation. One recent systemic review of 1130 hips found mixed impingement in 45% of cases (7–9). While both lesions are seen in FAI, they result in distinct patterns of articular damage which are markedly different (Table 1). Pincer lesions can vary in severity from focal overgrowth of the anterior acetabular rim with acetabular retroversion to the more global deformities seen with coxa profunda or protrusio acetabuli (Figure 1B) (2). The pincer deformity initially damages the labrum when the hip is in flexion, which brings the acetabular overgrowth into apposition with the femoral neck, thereby compressing the anterior labrum (3, 6). With repeated hip flexion, the labrum sustains repetitive microtrauma gradually separating from the acetabular cartilage and eventually failing (2). As the disease progresses, persistent pressure between the posteroinferior acetabulum and the posteromedial aspect of the femoral head initiates acetabular cartilage damage known as the “contrecoup” lesion (10, 11).
Cam deformities present with femoral head asphericity, seen as a flattening of the anterior contour of the head/neck junction or an osseous bump producing a decreased femoral head–neck offset. The bump is often located in the anterolateral or anterosuperior region of the head–neck junction and can be identified as a “pistol grip” deformity on AP and modified Dunn radiographs (3, 11). Similar to the pincer lesion, cam impingement is most symptomatic with the involved leg in flexion (12). However, unlike pincer lesions, the mechanism of impingement is through shear stress generated as the femoral lesion rotates through the anterosuperior acetabulum (11). From a clinical standpoint, the mixed variant of FAI can present with variable degrees of both injury patterns depending on the predominance of the existing lesions (3).
While osseous deformities underlie FAI, symptoms usually result following labral and chondral injury secondary to the impingement itself. In a recent study, Clohisy et al. found that 93% of patients undergoing surgery for FAI had associated labral injury, and 83% had associated cartilage damage (9). Labral and cartilage injuries occur by different mechanisms for cam and pincer lesions. Patients with pincer impingement present primarily with labral damage, consistent with the pathomechanics of acetabular overcoverage (12). Cartilage lesions in patients with pincer deformities are distinct, with acetabular chondral injury occupying a narrow circumferential band that is less severe than those with cam deformities (7, 12). Additionally, repeated microtrauma to the labrum in pincer abnormalities initiates bone growth at the acetabular rim and promotes eventual labral ossification.
For cam deformities, as the eccentric aspect of the lesion passes through the anterosuperior acetabulum during flexion, the transition zone between the labrum and acetabular cartilage is subjected to compressive and shear stresses (11). This causes the labrum to translate away from the joint while the cartilage is pushed in the opposite direction, preserving the labrum until later in the disease process (7, 12). Consequently, cam lesions initially damage the acetabular cartilage, causing delamination of the cartilage from the labrum, compared to pincer lesions which affect the labrum primarily (3, 11). As the mixed variant of FAI is common, patients frequently present with evidence of both chondral and labral damage resulting from cam and pincer deformities, respectively (3). Notably, poor preoperative cartilage status in symptomatic FAI patients is associated with delayed time till surgery and is a harbinger of potentially worse outcomes (13–15). Overall, the bony lesions of FAI induce variable damage to the hip joint, with cam lesions preferentially affecting the acetabular cartilage and pincer lesions affecting the labrum and peripheral acetabulum in a more circumferential manner (16).
Who Gets FAI?
The collective understanding of the etiology, history, and clinical presentation of FAI has evolved dramatically over the past decade. As FAI represents a syndrome with varying degrees of bony, chondral, and labral pathology at the hip joint, its presentation is similarly diverse. FAI is frequently seen in athletes. One recent systematic review of North American patients undergoing surgery for FAI found that the average age at surgery was 28 years and there was a mild female preponderance FAI at 55% of patients (9). Pincer FAI typically presents in middle-aged women; however, pincer lesions occur commonly in males as well (3, 9, 17). Cam lesions, on the other hand, demonstrate a near 3:1 male predominance and are seen more often in the younger population (3, 17). FAI can be present in the acute or chronic setting, and can be associated with prior trauma, such as malunion of a femoral neck fracture. It has also been associated with pediatric hip diseases, such as developmental dysplasia of the hip (DDH), slipped capital femoral epiphysis (SCFE), and Legg–Calve–Perthes disease (LCPD) (18, 19). Despite this, the most common presentation for FAI is idiopathic, atraumatic pain that has been ongoing between 12 and 16 months (9).
The precise etiology of FAI is still unclear; however, several theories exist linking genetic predisposition, pediatric deformity, and trauma, as well as high intensity adolescent athletic activity to the onset of FAI. Genetic factors involved in FAI pathogenesis were proposed by Pollard et al. who reported that siblings of patients with symptomatic FAI possessed an increased predilection for radiographic and clinical impingement signs (20). These findings, coupled with the increased incidence of cam FAI in males, promote the conclusion that there are intrinsic, although as of yet unidentified, genetic factors influencing hip morphology in the development of FAI (19, 21).
Additionally, pediatric hip disorders can predispose to FAI. SCFE deformities have been shown to predispose to the development of cam impingement in adulthood, which is mechanically consistent with the anterosuperior displacement of the femoral metaphysis in the pediatric disease (5, 19, 22, 23). Similarly, the natural history of LCPD can lead to FAI, in this case resulting from aspherical enlargement of the femoral head (coxa magna) representing the healed osteonecrotic epiphysis (23–25). Unlike SCFE, however, LCPD promotes both intra- and extra-articular impingement, complicating the nature of pain generation (19, 26, 27). Cam lesions have been found in the patients with prior femoral neck fractures, with Mathew et al. finding radiographic FAI in 84% of this cohort (28, 29). Furthermore, FAI can arise as a postsurgical consequence of the Bernese Peri-Acetabular Osteotomy, as the procedure can induce an iatrogenic pincer type acetabular conformation (30, 31). In general, any condition or procedure that alters the native bony anatomy of the hip joint can lead to clinical and radiological signs of impingement and secondary FAI.
While FAI is associated with prior hip pathology, it is most often idiopathic, and particularly common in the athletic population (3). This finding has led to multiple efforts investigating the relationship between sports participation and FAI development (19, 30, 32–37). One recent systematic review of 208 competitive male athletes (300 hips) concluded that athletes participating in high-impact sports (basketball, hockey, and soccer) were significantly more likely to develop cam lesions than non-athletes (odds ratio 1.9–8.0) (35). Furthermore, it has been proposed that the cam lesions develop in response to high intensity activity during development (5, 32, 38). In a study of 77 elite adolescent hockey players, Siebenrock et al. report higher alpha angles in athletes with closed physes as well as higher alpha angles in athletes reporting hip pain (39). In a recent prospective study of pre-professional adolescent soccer players, Agricola et al. measured proximal femur morphology at baseline and 2 years, finding significantly increased radiologic evidence of cam lesions at the 2-year time point (40). While there is evidence that suggests cam lesions can develop in high-intensity adolescent athletes, these studies primarily investigated a western European population. The prevalence of cam deformities in East Asian populations, however, is markedly reduced (41, 42). Thus, the role of genetics likely predisposes certain populations to FAI deformity under given repetitive and supra-physiologic loading conditions (21).
Does FAI Predispose to Arthritis?
Based on multiple in situ observations of the impingement and damage patterns associated with FAI through open surgical dislocation of the femoral head, Ganz et al. proposed FAI as a precursor to the development of OA (1, 2). Their group highlighted the specific labral and chondral injuries affiliated with cam and pincer lesions and argued that prolonged contact between the deformed acetabulum and proximal femur promote further cartilage damage and eventual joint deterioration. Cam lesions, in particular, have demonstrated an increased risk for the development of OA (16, 43–46). One retrospective study analyzed the radiographs of patients with unilateral hip OA and found that the presence of a non-spherical femoral head as seen in cam lesions has a significant association with OA (45). Furthermore, one prospective study of Dutch patients demonstrated that moderate (alpha angle >60°) and severe cam deformities (alpha angle >83°) demonstrated a respective 3.7 and 10 times greater likelihood of developing OA over a 5-year time span when compared to controls (47). This study also identified a positive predictive value of 53% for the future development of OA in patients with cam deformities on X-ray and a positive impingement sign (47). Similarly, in a study investigating the prevalence of FAI deformities in patients undergoing THA for OA, found patients younger than 65 undergoing THA were more likely to have evidence of cam, but not pincer lesions (48). Thus, while cam lesions are linked to the development of OA, the relevance of pincer lesions in OA are less clear (44, 49). However, as isolated pincer lesions are rare, seen in only 7% of FAI cases, the cam lesion present in the other 93% of FAI cases may be the primary driver of OA in FAI (9).
In addition to the epidemiologic and radiographic studies correlating OA development to characteristic FAI lesions, biomarkers seen in OA are being investigated to identify correlations to patients with FAI [Table 2 (72–80)]. While there are over 70 biomarkers that have been studied in OA, validation has proved challenging (50). One recent systematic review identified six biomarkers that were correlated to OA progression: cartilage oligomeric protein (COMP), 25-OH vitamin D, N-terminal telopeptide (NTX), type II collagen C telopeptide (CTX-II), TIMP metalloproteinase inhibitor (TIMP), and vascular cell adhesion molecule 1 (VCAM-1) (50). In support of COMP as a biomarker, Dragomir et al. found that COMP levels were higher in patients with clinical signs of hip dysfunction and Bedi et al. reported COMP levels to be significantly increased in male athletes with FAI compared to a control group (50–52). However, the relevance of COMP has been questioned by several studies that have not found associations between COMP and hip OA (50, 53, 54). One study suggests that deamidated COMP (DCOMP) may be a more useful biomarker as they found a strong association with DCOMP levels and radiographic OA, as well as higher DCOMP concentrations in regions in proximity with OA lesions (54). Additionally, Bedi et al. also found a 276% increase in circulating CRP levels in patients with FAI compared to controls, indicating that there may be an inflammatory component to FAI (52). This observation was supported by a recent histologic study which found significantly increased macrophage and mast cell expression in labrums from patients with FAI compared to labrums from patients with OA (55). While there are currently no validated biomarkers for FAI, studies have shown promising associations that must be confirmed by future research (50).
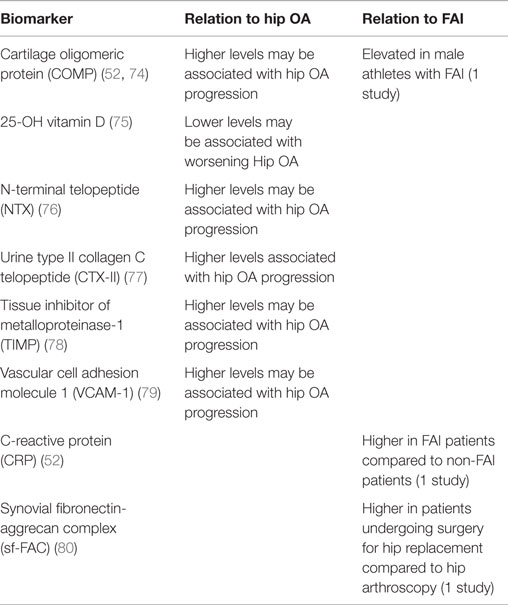
Table 2. Molecular biomarkers associated with the onset and/or progression of hip osteoarthritis and their relation to FAI.
While cam or pincer deformities are a necessary condition for FAI, they are not pathognomonic and are frequently encountered in the asymptomatic population. One recent systematic review of 26 studies with 2114 total asymptomatic hips found an average cam and pincer lesion prevalence of 37 and 63%, respectively (4). Previously reported percentages of cam lesions ranged between 10 and 24%, and the authors attribute their reported increase to the high percentage of athletes in the review population (56–58). Cam lesions, measured on MRI as well as AP and modified Dunn radiographs, are variably defined by alpha angle and standardized cutoff values for normal and abnormal alpha angles are lacking (59–61). Pincer lesion prevalence may also be over-reported as radiographic findings, such as the cross over and posterior wall signs have proven unreliable markers (4, 62). Despite this, it is clear that the radiographic findings of FAI are common in the asymptomatic population, which has brought the correlation between cam and pincer deformities and OA into question (63). One putative explanation for this discrepancy lies in status patient’s articular cartilage. Hogervorst et al. introduced the term “cartilotype” to assess the susceptibility of cartilage to degradation in response to mechanical stress (21). Thus, patients with radiographic FAI may remain asymptomatic if their articular cartilage is able to withstand the impingement produced by the osseous deformities (64). Taken together, the surgeon should relate the patient’s clinical history and findings on physical exam to the radiographic evidence when preparing for the surgical correction of FAI.
Does Surgical Treatment Alter the Natural History?
A number of studies have demonstrated that surgery for FAI is a safe and effective means to improve function and decrease pain levels in the short- and mid-term (65–67). Generally, open and arthroscopic treatment modalities appear to provide comparable outcomes in the mid-term aside from general health-related quality of life, which is significantly higher in the arthroscopic group (68). Intuitively, it makes sense that surgical intervention to remove the osseous mechanical blocks to motion will prevent further damage to the soft tissue structures (cartilage and labrum) of the hip. Studies have corroborated that the severity of cartilaginous and labral degenerative changes are directly associated with the duration of the underlying pathology (69–71). However, the available literature to date cannot assure that surgical intervention either in the asymptomatic or symptomatic patient will prevent the progression to OA and the risk of eventually requiring a THA. Prognostic indicators of early OA following treatment of FAI have yet to be elucidated, thus it is difficult at this time to associate treatment of FAI and the progression of OA.
Research strategies to further investigate the natural history of FAI and the association with OA are currently underway. Such efforts focus on prospective evaluations of younger patients with an early diagnosis of FAI. This study design enables researchers to focus on early interventions that may change the disease course over a long period of time. This study design, ideally in a randomized fashion, will aid in answering long-term questions regarding surgical intervention (both arthroscopic and open) and the ability of these interventions to delay or prevent OA and the need for THA.
Conclusion
The purpose of this article is to review our current understanding of FAI by focusing on the natural history of the disease process. Surgical correction of the underlying osseous pathology in the symptomatic patient will improve function and decrease pain. Although an association between FAI and the development of OA is logical, long-term longitudinal studies have not yet been completed to substantiate cause and effect. Therefore, there currently is insufficient evidence to recommend prophylactic surgery in asymptomatic patients with radiographic evidence of FAI. Future studies targeting the early diagnosis and treatment of FAI will assist in elucidating the etiology of FAI, the natural history of the disease process, and ultimately the association between FAI and the progression to hip OA.
Conflict of Interest Statement
Thomas H. Wuerz MD: Paid consultant, CONMED Linvatec. The remaining co-authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
1. Ganz R, Parvizi J, Beck M, Leunig M, Nötzli H, Siebenrock KA. Femoroacetabular impingement: a cause for osteoarthritis of the hip. Clin Orthop Relat Res (2003) 417:112–20. doi:10.1097/01.blo.0000096804.78689.c2
2. Ganz R, Leunig M, Leunig-Ganz K, Harris WH. The etiology of osteoarthritis of the hip: an integrated mechanical concept. Clin Orthop Relat Res (2008) 466(2):264–72. doi:10.1097/01.blo.0000096804.78689.c2
3. Byrd JW. Femoroacetabular impingement in athletes: current concepts. Am J Sports Med (2014) 42(3):737–51. doi:10.1177/0363546513499136
4. Frank JM, Harris JD, Erickson BJ, Slikker W, Bush-Joseph CA, Salata MJ, et al. Prevalence of femoroacetabular impingement imaging findings in asymptomatic volunteers: a systematic review. Arthroscopy (2015) 31(6):1199–204. doi:10.1016/j.arthro.2014.11.042
5. Siebenrock KA, Ferner F, Noble PC, Santore RF, Werlen S, Mamisch TC. The cam-type deformity of the proximal femur arises in childhood in response to vigorous sporting activity. Clin Orthop Relat Res (2011) 469(11):3229–40. doi:10.1007/s11999-011-1945-4
6. Leunig M, Beaulé PE, Ganz R. The concept of femoroacetabular impingement: current status and future perspectives. Clin Orthop Relat Res (2009) 467(3):616–22. doi:10.1007/s11999-008-0646-0
7. Beck M, Kalhor M, Leunig M, Ganz R. Hip morphology influences the pattern of damage to the acetabular cartilage: femoroacetabular impingement as a cause of early osteoarthritis of the hip. J Bone Joint Surg Br (2005) 87(7):1012–8. doi:10.1302/0301-620X.87B7.15203
8. Bedi A, Kelly BT. Femoroacetabular impingement. J Bone Joint Surg Am (2013) 95(1):82–92. doi:10.2106/JBJS.K.01219
9. Clohisy JC, Baca G, Beaulé PE, Kim Y-J, Larson CM, Millis MB, et al. Descriptive epidemiology of femoroacetabular impingement: a North American cohort of patients undergoing surgery. Am J Sports Med (2013) 41(6):1348–56. doi:10.1177/0363546513488861
10. Pfirrmann CW, Mengiardi B, Dora C, Kalberer F, Zanetti M, Hodler J. Cam and pincer femoroacetabular impingement: characteristic MR arthrographic findings in 50 patients. Radiology (2006) 240(3):778–85. doi:10.1148/radiol.2403050767
11. Beck M, Chegini S, Ferguson S, Hosalkar HS. Mechanism of femoroacetabular impingement. In: Marín-Peña Ó, editor. Femoroacetabular Impingement [Internet]. Berlin: Springer (2012). p. 9–22 [cited 2015 Jul 3]. Available from: http://link.springer.com/chapter/10.1007/978-3-642-22769-1_2
12. Tibor LM, Leunig M. The pathoanatomy and arthroscopic management of femoroacetabular impingement. Bone Joint Res (2012) 1(10):245–57. doi:10.1302/2046-3758.110.2000105
13. Claßen T, Körsmeier K, Kamminga M, Beck S, Rekowski J, Jäger M, et al. Is early treatment of cam-type femoroacetabular impingement the key to avoiding associated full thickness isolated chondral defects? Knee Surg Sports Traumatol Arthrosc (2014). doi:10.1007/s00167-014-3332-7
14. Egerton T, Hinman RS, Takla A, Bennell KL, O’Donnell J. Intraoperative cartilage degeneration predicts outcome 12 months after hip arthroscopy. Clin Orthop Relat Res (2013) 471(2):593–9. doi:10.1007/s11999-012-2594-y
15. Ferro FP, Ho CP, Dornan GJ, Surowiec RK, Philippon MJ. Comparison of T2 values in the lateral and medial portions of the weight-bearing cartilage of the hip for patients with symptomatic femoroacetabular impingement and asymptomatic volunteers. Arthroscopy (2015) 31(8):1497–506. doi:10.1016/j.arthro.2015.02.045
16. Sankar WN, Matheney TH, Zaltz I. Femoroacetabular impingement: current concepts and controversies. Orthop Clin North Am (2013) 44(4):575–89. doi:10.1016/j.ocl.2013.07.003
17. Laborie LB, Lehmann TG, Engesæter I, Eastwood DM, Engesæter LB, Rosendahl K. Prevalence of radiographic findings thought to be associated with femoroacetabular impingement in a population-based cohort of 2081 healthy young adults. Radiology (2011) 260(2):494–502. doi:10.1148/radiol.11102354
18. Nepple JJ, Prather H, Trousdale RT, Clohisy JC, Beaulé PE, Glyn-Jones S, et al. Clinical diagnosis of femoroacetabular impingement. J Am Acad Orthop Surg (2013) 21(Suppl 1):S16–9. doi:10.5435/JAAOS-21-07-S16
19. Chaudhry H, Ayeni OR. The etiology of femoroacetabular impingement: what we know and what we don’t. Sports Health (2014) 6(2):157–61. doi:10.1177/1941738114521576
20. Pollard TC, Villar RN, Norton MR, Fern ED, Williams MR, Murray DW, et al. Genetic influences in the aetiology of femoroacetabular impingement: a sibling study. J Bone Joint Surg Br (2010) 92(2):209–16. doi:10.1302/0301-620X.92B2.22850
21. Hogervorst T, Eilander W, Fikkers JT, Meulenbelt I. Hip ontogenesis: how evolution, genes, and load history shape hip morphotype and cartilotype. Clin Orthop Relat Res (2012) 470(12):3284–96. doi:10.1007/s11999-012-2511-4
22. Klit J, Gosvig K, Magnussen E, Gelineck J, Kallemose T, Søballe K, et al. Cam deformity and hip degeneration are common after fixation of a slipped capital femoral epiphysis. Acta Orthop (2014) 85(6):585–91. doi:10.3109/17453674.2014.957078
23. Millis MB, Lewis CL, Schoenecker PL, Clohisy JC. Legg-Calvé-Perthes disease and slipped capital femoral epiphysis: major developmental causes of femoroacetabular impingement. J Am Acad Orthop Surg (2013) 21(Suppl 1):S59–63. doi:10.5435/JAAOS-21-07-S59
24. Eijer H, Podeszwa DA, Ganz R, Leunig M. Evaluation and treatment of young adults with femoro-acetabular impingement secondary to Perthes’ disease. Hip Int (2006) 16(4):273–80.
25. Accadbled F, Pailhé R, Launay F, Nectoux E, Bonin N, Gicquel P, et al. “Femoroacetabular impingement”. Legg-Calve-Perthes disease: from childhood to adulthood. Orthop Traumatol Surg Res (2014) 100(6):647–9. doi:10.1016/j.otsr.2014.03.029
26. Tannast M, Hanke M, Ecker TM, Murphy SB, Albers CE, Puls M. LCPD: reduced range of motion resulting from extra- and intraarticular impingement. Clin Orthop Relat Res (2012) 470(9):2431–40. doi:10.1007/s11999-012-2344-1
27. Larson AN, Sucato DJ, Herring JA, Adolfsen SE, Kelly DM, Martus JE, et al. A prospective multicenter study of Legg-Calvé-Perthes disease: functional and radiographic outcomes of nonoperative treatment at a mean follow-up of twenty years. J Bone Joint Surg Am (2012) 94(7):584–92. doi:10.2106/JBJS.J.01073
28. Eijer H, Myers SR, Ganz R. Anterior femoroacetabular impingement after femoral neck fractures. J Orthop Trauma (2001) 15(7):475–81. doi:10.1097/00005131-200109000-00003
29. Mathew G, Kowalczuk M, Hetaimish B, Bedi A, Philippon MJ, Bhandari M, et al. Radiographic prevalence of CAM-type femoroacetabular impingement after open reduction and internal fixation of femoral neck fractures. Knee Surg Sports Traumatol Arthrosc (2014) 22(4):793–800. doi:10.1007/s00167-014-2835-6
30. Ziebarth K, Balakumar J, Domayer S, Kim YJ, Millis MB. Bernese periacetabular osteotomy in males: is there an increased risk of femoroacetabular impingement (FAI) after Bernese periacetabular osteotomy? Clin Orthop Relat Res (2011) 469(2):447–53. doi:10.1007/s11999-010-1544-9
31. Albers CE, Steppacher SD, Ganz R, Tannast M, Siebenrock KA. Impingement adversely affects 10-year survivorship after periacetabular osteotomy for DDH. Clin Orthop Relat Res (2013) 471(5):1602–14. doi:10.1007/s11999-013-2799-8
32. Agricola R, Bessems JH, Ginai AZ, Heijboer MP, van der Heijden RA, Verhaar JA, et al. The development of cam-type deformity in adolescent and young male soccer players. Am J Sports Med (2012) 40(5):1099–106. doi:10.1177/0363546512438381
33. Gerhardt MB, Romero AA, Silvers HJ, Harris DJ, Watanabe D, Mandelbaum BR. The prevalence of radiographic hip abnormalities in elite soccer players. Am J Sports Med (2012) 40(3):584–8. doi:10.1177/0363546511432711
34. Lahner M, Walter PA, von Schulze Pellengahr C, Hagen M, von Engelhardt LV, Lukas C. Comparative study of the femoroacetabular impingement (FAI) prevalence in male semiprofessional and amateur soccer players. Arch Orthop Trauma Surg (2014) 134(8):1135–41. doi:10.1007/s00402-014-2008-6
35. Nepple JJ, Vigdorchik JM, Clohisy JC. What is the association between sports participation and the development of proximal femoral cam deformity? A systematic review and meta-analysis. Am J Sports Med (2015) 43(11):2833–40. doi:10.1177/0363546514563909
36. Nepple JJ, Brophy RH, Matava MJ, Wright RW, Clohisy JC. Radiographic findings of femoroacetabular impingement in National Football League combine athletes undergoing radiographs for previous hip or groin pain. Arthroscopy (2012) 28(10):1396–403. doi:10.1016/j.arthro.2012.03.005
37. Ayeni OR, Banga K, Bhandari M, Maizlin Z, de Sa D, Golev D, et al. Femoroacetabular impingement in elite ice hockey players. Knee Surg Sports Traumatol Arthrosc (2014) 22(4):920–5. doi:10.1007/s00167-013-2598-5
38. Zadpoor AA. Etiology of femoroacetabular impingement in athletes: a review of recent findings. Sports Med (2015) 45(8):1097–106. doi:10.1007/s40279-015-0339-2
39. Siebenrock KA, Kaschka I, Frauchiger L, Werlen S, Schwab JM. Prevalence of cam-type deformity and hip pain in elite ice hockey players before and after the end of growth. Am J Sports Med (2013) 41(10):2308–13. doi:10.1177/0363546513497564
40. Agricola R, Heijboer MP, Ginai AZ, Roels P, Zadpoor AA, Verhaar JA, et al. A cam deformity is gradually acquired during skeletal maturation in adolescent and young male soccer players: a prospective study with minimum 2-year follow-up. Am J Sports Med (2014) 42(4):798–806. doi:10.1177/0363546514524364
41. Takeyama A, Naito M, Shiramizu K, Kiyama T. Prevalence of femoroacetabular impingement in Asian patients with osteoarthritis of the hip. Int Orthop (2009) 33(5):1229–32. doi:10.1007/s00264-009-0742-0
42. Fukushima K, Uchiyama K, Takahira N, Moriya M, Yamamoto T, Itoman M, et al. Prevalence of radiographic findings of femoroacetabular impingement in the Japanese population. J Orthop Surg (2014) 9:25. doi:10.1186/1749-799X-9-25
43. Gregory JS, Waarsing JH, Day J, Pols HA, Reijman M, Weinans H, et al. Early identification of radiographic osteoarthritis of the hip using an active shape model to quantify changes in bone morphometric features: can hip shape tell us anything about the progression of osteoarthritis? Arthritis Rheum (2007) 56(11):3634–43. doi:10.1002/art.22982
44. Nicholls AS, Kiran A, Pollard TC, Hart DJ, Arden CP, Spector T, et al. The association between hip morphology parameters and nineteen-year risk of end-stage osteoarthritis of the hip: a nested case-control study. Arthritis Rheum (2011) 63(11):3392–400. doi:10.1002/art.30523
45. Doherty M, Courtney P, Doherty S, Jenkins W, Maciewicz RA, Muir K, et al. Nonspherical femoral head shape (pistol grip deformity), neck shaft angle, and risk of hip osteoarthritis: a case-control study. Arthritis Rheum (2008) 58(10):3172–82. doi:10.1002/art.23939
46. Agricola R, Waarsing JH, Arden NK, Carr AJ, Bierma-Zeinstra SM, Thomas GE, et al. Cam impingement of the hip: a risk factor for hip osteoarthritis. Nat Rev Rheumatol (2013) 9(10):630–4. doi:10.1038/nrrheum.2013.114
47. Agricola R, Heijboer MP, Bierma-Zeinstra SM, Verhaar JA, Weinans H, Waarsing JH. Cam impingement causes osteoarthritis of the hip: a nationwide prospective cohort study (CHECK). Ann Rheum Dis (2013) 72(6):918–23. doi:10.1136/annrheumdis-2012-201643
48. Lafrance R, Williams R, Madsen W, Maloney M, Drinkwater C, Giordano B. The prevalence of radiographic criteria of femoral acetabular impingement in patients undergoing hip arthroplasty surgery. Geriatr Orthop Surg Rehabil (2014) 5(1):21–6. doi:10.1177/2151458514522262
49. Agricola R, Heijboer MP, Roze RH, Reijman M, Bierma-Zeinstra SM, Verhaar JA, et al. Pincer deformity does not lead to osteoarthritis of the hip whereas acetabular dysplasia does: acetabular coverage and development of osteoarthritis in a nationwide prospective cohort study (CHECK). Osteoarthritis Cartilage (2013) 21(10):1514–21. doi:10.1016/j.joca.2013.07.004
50. Nepple JJ, Thomason KM, An TW, Harris-Hayes M, Clohisy JC. What is the utility of biomarkers for assessing the pathophysiology of hip osteoarthritis? A systematic review. Clin Orthop Relat Res (2015) 473(5):1683–701. doi:10.1007/s11999-015-4148-6
51. Dragomir AD, Kraus VB, Renner JB, Luta G, Clark A, Vilim V, et al. Serum cartilage oligomeric matrix protein and clinical signs and symptoms of potential pre-radiographic hip and knee pathology. Osteoarthritis Cartilage (2002) 10(9):687–91. doi:10.1053/joca.2002.0816
52. Bedi A, Lynch EB, Enselman ER, Davis ME, DeWolf PD, Makki TA, et al. Elevation in circulating biomarkers of cartilage damage and inflammation in athletes with femoroacetabular impingement. Am J Sports Med (2013) 41(11):2585–90. doi:10.1177/0363546513499308
53. Garnero P, Mazières B, Guéguen A, Abbal M, Berdah L, Lequesne M, et al. Cross-sectional association of 10 molecular markers of bone, cartilage, and synovium with disease activity and radiological joint damage in patients with hip osteoarthritis: the ECHODIAH cohort. J Rheumatol (2005) 32(4):697–703.
54. Catterall JB, Hsueh MF, Stabler TV, McCudden CR, Bolognesi M, Zura R, et al. Protein modification by deamidation indicates variations in joint extracellular matrix turnover. J Biol Chem (2012) 287(7):4640–51. doi:10.1074/jbc.M111.249649
55. Elias-Jones CJ, Farrow L, Reilly JH, Kerr S, Meek RM, Kelly MP, et al. Inflammation and neovascularization in hip impingement: not just wear and tear. Am J Sports Med (2015) 43(8):1875–81. doi:10.1177/0363546515588176
56. Reichenbach S, Leunig M, Werlen S, Nüesch E, Pfirrmann CW, Bonel H, et al. Association between cam-type deformities and magnetic resonance imaging-detected structural hip damage: a cross-sectional study in young men. Arthritis Rheum (2011) 63(12):4023–30. doi:10.1002/art.30589
57. Leunig M, Jüni P, Werlen S, Limacher A, Nüesch E, Pfirrmann CW, et al. Prevalence of cam and pincer-type deformities on hip MRI in an asymptomatic young Swiss female population: a cross-sectional study. Osteoarthritis Cartilage (2013) 21(4):544–50. doi:10.1016/j.joca.2013.01.003
58. Hack K, Di Primio G, Rakhra K, Beaulé PE. Prevalence of cam-type femoroacetabular impingement morphology in asymptomatic volunteers. J Bone Joint Surg Am (2010) 92(14):2436–44. doi:10.2106/JBJS.J.01280
59. Nötzli HP, Wyss TF, Stoecklin CH, Schmid MR, Treiber K, Hodler J. The contour of the femoral head-neck junction as a predictor for the risk of anterior impingement. J Bone Joint Surg Br (2002) 84(4):556–60. doi:10.1302/0301-620X.84B4.12014
60. Barton C, Salineros MJ, Rakhra KS, Beaulé PE. Validity of the alpha angle measurement on plain radiographs in the evaluation of cam-type femoroacetabular impingement. Clin Orthop Relat Res (2011) 469(2):464–9. doi:10.1007/s11999-010-1624-x
61. Massey PA, Nho SJ, Larson CM, Harris JD. Letter to the editor re: “cam impingement: defining the presence of a cam deformity by the alpha angle data from the CHECK cohort and Chingford cohort”. Osteoarthritis Cartilage (2014) 22(12):2093–4. doi:10.1016/j.joca.2014.09.023
62. Zaltz I, Kelly BT, Hetsroni I, Bedi A. The crossover sign overestimates acetabular retroversion. Clin Orthop Relat Res (2013) 471(8):2463–70. doi:10.1007/s11999-012-2689-5
63. Hartofilakidis G, Bardakos NV, Babis GC, Georgiades G. An examination of the association between different morphotypes of femoroacetabular impingement in asymptomatic subjects and the development of osteoarthritis of the hip. J Bone Joint Surg Br (2011) 93(5):580–6. doi:10.1302/0301-620X.93B5.25236
64. Packer JD, Safran MR. The etiology of primary femoroacetabular impingement: genetics or acquired deformity? J Hip Preserv Surg (2015) 2(3):249–57. doi:10.1093/jhps/hnv046
65. Bedi A, Chen N, Robertson W, Kelly BT. The management of labral tears and femoroacetabular impingement of the hip in the young, active patient. Arthroscopy (2008) 24(10):1135–45. doi:10.1016/j.arthro.2008.06.001
66. Clohisy JC, St John LC, Schutz AL. Surgical treatment of femoroacetabular impingement: a systematic review of the literature. Clin Orthop Relat Res (2010) 468(2):555–64. doi:10.1007/s11999-009-1138-6
67. Ng VY, Arora N, Best TM, Pan X, Ellis TJ. Efficacy of surgery for femoroacetabular impingement: a systematic review. Am J Sports Med (2010) 38(11):2337–45. doi:10.1177/0363546510365530
68. Nwachukwu BU, Rebolledo BJ, McCormick F, Rosas S, Harris JD, Kelly BT. Arthroscopic versus open treatment of femoroacetabular impingement a systematic review of medium- to long-term outcomes. Am J Sports Med (2015).
69. Burnett RS, Della Rocca GJ, Prather H, Curry M, Maloney WJ, Clohisy JC. Clinical presentation of patients with tears of the acetabular labrum. J Bone Joint Surg Am (2006) 88(7):1448–57. doi:10.2106/JBJS.D.02806
70. Ito K, Leunig M, Ganz R. Histopathologic features of the acetabular labrum in femoroacetabular impingement. Clin Orthop Relat Res (2004) 429:262–71. doi:10.1097/01.blo.0000144861.11193.17
71. Nepple JJ, Zebala LP, Clohisy JC. Labral disease associated with femoroacetabular impingement: do we need to correct the structural deformity? J Arthroplasty (2009) 24(6 Suppl):114–9. doi:10.1016/j.arth.2009.06.003
72. Siebenrock KA, Wahab KH, Werlen S, Kalhor M, Leunig M, Ganz R. Abnormal extension of the femoral head epiphysis as a cause of cam impingement. Clin Orthop Relat Res (2004) 418:54–60. doi:10.1097/00003086-200401000-00010
73. Roels P, Agricola R, Oei EH, Weinans H, Campoli G, Zadpoor AA. Mechanical factors explain development of cam-type deformity. Osteoarthritis Cartilage (2014) 22(12):2074–82. doi:10.1016/j.joca.2014.09.011
74. Chaganti RK, Kelman A, Lui L, Yao W, Javaid MK, Bauer D, et al. Change in serum measurements of cartilage oligomeric matrix protein and association with the development and worsening of radiographic hip osteoarthritis. Osteoarthritis Cartilage (2008) 16(5):566–71. doi:10.1016/j.joca.2007.09.008
75. Chaganti RK, Parimi N, Cawthon P, Dam TL, Nevitt MC, Lane NE. Association of 25-hydroxyvitamin D with prevalent osteoarthritis of the hip in elderly men: the osteoporotic fractures in men study. Arthritis Rheum (2010) 62(2):511–4. doi:10.1002/art.27241
76. Kelman A, Lui L, Yao W, Krumme A, Nevitt M, Lane NE. Association of higher levels of serum cartilage oligomeric matrix protein and N-telopeptide crosslinks with the development of radiographic hip osteoarthritis in elderly women. Arthritis Rheum (2006) 54(1):236–43. doi:10.1002/art.21527
77. Reijman M, Hazes JM, Bierma-Zeinstra SM, Koes BW, Christgau S, Christiansen C, et al. A new marker for osteoarthritis: cross-sectional and longitudinal approach. Arthritis Rheum (2004) 50(8):2471–8. doi:10.1002/art.20332
78. Chevalier X, Conrozier T, Gehrmann M, Claudepierre P, Mathieu P, Unger S, et al. Tissue inhibitor of metalloprotease-1 (TIMP-1) serum level may predict progression of hip osteoarthritis. Osteoarthritis Cartilage (2001) 9(4):300–7. doi:10.1053/joca.2000.0389
79. Schett G, Kiechl S, Bonora E, Zwerina J, Mayr A, Axmann R, et al. Vascular cell adhesion molecule 1 as a predictor of severe osteoarthritis of the hip and knee joints. Arthritis Rheum (2009) 60(8):2381–9. doi:10.1002/art.24757
Keywords: femoroacetabular impingement, hip osteoarthritis, hip preservation surgery, FAI etiology, hip arthroscopy
Citation: Kuhns BD, Weber AE, Levy DM and Wuerz TH (2015) The Natural History of Femoroacetabular Impingement. Front. Surg. 2:58. doi: 10.3389/fsurg.2015.00058
Received: 01 September 2015; Accepted: 29 October 2015;
Published: 16 November 2015
Edited by:
Jaimo Ahn, Hospital of the University of Pennsylvania, USAReviewed by:
Manit Arora, University of New South Wales, AustraliaKrisztian Sisak, University of Szeged, Hungary
Copyright: © 2015 Kuhns, Weber, Levy and Wuerz. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Benjamin D. Kuhns, bmhvLnJlc2VhcmNoQHJ1c2hvcnRoby5jb20=
 Benjamin D. Kuhns
Benjamin D. Kuhns Alexander E. Weber1
Alexander E. Weber1 Thomas H. Wuerz
Thomas H. Wuerz