- Department of Neurosurgery, Barrow Neurological Institute, St. Joseph’s Hospital and Medical Center, Phoenix, AZ, USA
Brain metastases are the most common intracranial malignancy, accounting for significant morbidity and mortality in oncology patients. The current treatment paradigm for brain metastasis depends on the patient’s overall health status, the primary tumor pathology, and the number and location of brain lesions. Herein, we review the modern management options for these tumors, including surgical resection, radiotherapy, and chemotherapy. Recent operative advances, such as fluorescence, confocal microscopy, and brachytherapy, are highlighted. With an increased understanding of the pathophysiology of brain metastasis come increased future therapeutic options. Therapy targeted to specific tumor molecular pathways, such as those involved in blood–brain barrier transgression, cell–cell adhesion, and angiogenesis, are also reviewed. A personalized plan for each patient, based on molecular characterizations of the tumor that are used to better target radiotherapy and chemotherapy, is undoubtedly the future of brain metastasis treatment.
Introduction
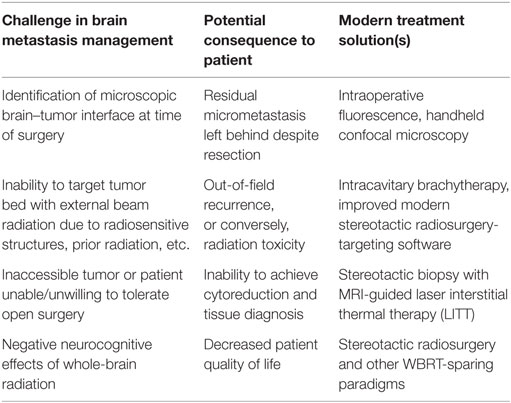
Nearly 200,000 patients are newly diagnosed with brain metastases annually in the United States, and metastases of the lung, skin, kidney, breast, and gastrointestinal tract are the most common intracranial malignancies (1, 2). Historically, overall survival after diagnosis is poor; however, in the last 30 years, improved systemic disease therapies and multimodality brain metastasis treatment have substantially increased survival. This increase in the quantity of life after diagnosis allows clinicians to minimize morbidity and focus on the patient’s quality of life. Choosing an appropriate personalized treatment plan for patients with brain metastasis maximizes survival and minimizes morbidity from unnecessary or futile treatments. The wide variety of tumor types, treatment strategies, and constant innovations within the field requires close collaboration among neurosurgeons, medical oncologists, radiation oncologists, and other specialists. Current treatment paradigms for brain metastases employ several treatment modalities, including open surgical resection, Gamma Knife or CyberKnife stereotactic radiosurgery, focused external beam radiotherapy, whole-brain radiotherapy (WBRT), traditional chemotherapy, and newer targeted biological agents personalized for tumor type. We review the current standards of care for brain metastases and summarize modern advances in their intraoperative diagnosis and treatment (Table 1). Lastly, we provide an overview of recent basic science and translational research leading to better understanding of the personalized biology of brain metastasis through modern genomic, transcriptomic, and proteomic techniques.
Current Treatment Paradigms
Key elements driving decision-making for brain metastasis care are patient factors and tumor factors. Patient factors include the patient’s overall age, condition, and systemic disease burden, summarized as life expectancy independent of central nervous system (CNS) disease. Tumor factors include histological type, number, and location of lesions, and, more recently, the biology of the tumor based on molecular and genetic testing. Patients with poor life expectancy independent of CNS disease may reasonably be offered palliative care or no treatment for the CNS disease, regardless of the nature of the brain involvement. Conversely, patients in good medical condition with a low systemic disease burden, and hence a good survival chance independent of the brain metastases, may warrant aggressive treatment. Similarly, certain histological types of tumors (e.g., small cell lung cancer, breast cancer) are more likely to respond to adjuvant treatment with irradiation or chemotherapy, which can make their use beneficial even for numerous or poorly located lesions. Additionally, the more numerous the brain metastases, the poorer the prognosis is, irrespective of treatment. Lesions in eloquent parts of the brain (i.e., those that subserve a discrete function, such as speech or movement) or in parts of the brain less accessible via open neurosurgery also connote a poorer prognosis.
Neurosurgical resection of individual symptomatic brain metastases remains the standard of care. Lesions causing deficits due to local mass effect and cerebral edema should almost always undergo surgical extirpation once diagnosed, particularly if the lesion is a new diagnosis and tissue is required for pathology. Modern advances in microneurosurgical techniques and intraoperative magnetic resonance imaging-based neuronavigation allow for safe resection of lesions almost anywhere in the cerebrum. For single metastases, Patchell et al.’s landmark randomized clinical trial strongly supports surgical excision (3). Patients with a single brain metastasis underwent surgical excision followed by radiation or biopsy and radiation alone. Local control, overall survival, and quality of life were all significantly improved with surgical resection plus radiation. This study comprised mostly patients with lung cancer metastases who had high function status. Despite its lack of generalizability to all tumor patients, it remains one of the best randomized trials supporting neurosurgical intervention for brain metastases.
Traditionally, WBRT has been used after surgical resection of a single lesion or when there are multiple small asymptomatic lesions. However, WBRT carries a risk of significant cognitive morbidity, and WBRT-sparing strategies are increasingly used (4, 5). Both the American Society for Radiation Oncology and the National Comprehensive Cancer Network Clinical Practice Guidelines in Oncology have published consensus statements supporting stereotactic radiosurgery after surgical resection of a single metastasis, instead of WBRT, in patients with a single lesion and good systemic disease control (6, 7). This WBRT-sparing alternative is not supported by Level 1 randomized trial data, but rather by significant lesser strength evidence (8–12).
Lastly, depending on tumor histology and the organ of origin, standard chemotherapy is implemented at the discretion of the medical oncologist after the surgical site heals.
Intraoperative Advances in Surgical Treatment
Neurosurgical Resection and Tumor Visualization
Nests of tumor cells exist for several millimeters outside the confines of the distinct metastatic brain lesion and its gliotic capsule (13). Aggressive resection of this microscopic margin, when feasible, can reduce the local recurrence of brain metastases (14). Therefore, the intraoperative ability to visualize and resect these microscopic margins using fluorescence-guided surgery is of considerable interest. Fluorescence-guided neurosurgery has become commonplace in glioma surgery; various agents exploit either a degraded blood–brain barrier (e.g., fluorescein) or unique metabolism [e.g., 5-aminolevulinic acid (5-ALA)], with the goal of improving the extent of resection in infiltrative processes (15–19). Within vascular neurosurgery, indocyanine green video angiography is used in cerebral aneurysm, arteriovenous malformation, and dural arteriovenous fistula surgery as an alternative or adjuvant to traditional angiography (20–24). Multiple authors have described intraoperative fluorescence for resection of metastatic tumors, albeit with less robust support than in glioma surgery or neurovascular surgery. Schebesch and colleagues published results from a series of 30 patients with brain metastases who underwent fluorescein-guided resection using a Zeiss microscope filter system (25). Most tumors (90%) avidly expressed fluorescein, and no patients suffered complications attributable to the intravenous dye. No control group was reported, but the gross-total resection rate of 83% and permanent neurological complication rate of 6.7% are similar to reported surgical results (3, 25). Therefore, the use of fluorescein requires additional study before definitive recommendations can be made about its efficacy in improving the safe extent of resection. Some authors have noted that the area of intraoperative fluorescence seems to extend well beyond the gross tumor margins, possibly because of breakdown of the blood–brain barrier induced by the tumor. A prospective trial planned by Schebesch and colleagues will validate the usefulness of fluorescein in this setting. An alternative fluorescent agent is 5-ALA, which has found significant use in glioma surgery. Kamp et al. reported results from a retrospective series of 52 patients undergoing brain metastasis surgery using 5-ALA (26). As with fluorescein, most tumors (62%) expressed 5-ALA positivity. Residual cavity fluorescence was detected in most patients (75%) after gross-total resection of the distinct metastasis. Unfortunately, only one-third of those patients with available histological tissue samples were found to harbor microscopic disease at these 5-ALA-positive margins. Therefore, in non-eloquent areas, the use of 5-ALA seems to drive “supra-maximal” resection of surrounding reactive tissue, which means that caution is required in eloquent areas because 5-ALA positivity was not particularly sensitive for residual micrometastasis. These studies and others demonstrate the pressing need for additional research into novel fluorescent compounds to better define microscopic tumor margins in brain metastases.
Intraoperative Diagnosis
Preoperative diagnosis of a metastatic brain tumor is not always obvious, especially in patients with no known primary malignancy and an isolated lesion. Rapid intraoperative diagnosis via confocal microscopy is now a viable alternative to traditional frozen sectioning with light microscopy. Given the relatively limited literature surrounding fluorescence to date, the in vivo application of intraoperative confocal microscopy is particularly appealing for inspection of the microscopic edges of metastatic lesions within the resection cavity itself. Our group has had success with the use of in vivo, real-time, handheld confocal microscopy for diagnosis of various brain tumor types and visualization of the brain–tumor interface (27–30). Further technological refinement is required for handheld confocal microscopy to be widely adopted, but it remains an appealing method to detect residual tumor.
Brachytherapy
Radioactive brachytherapy seeds used in neurosurgery have had mixed results for a half-century (31–33). Brachytherapy enables delivery of high doses of radiation with quick dose fall-off and custom dosing to areas of residual tumor while sparing nearby radiosensitive structures via selective seed placement within the resection cavity. Isotypes used for intracranial brachytherapy have evolved significantly since the 1960s, with cesium-131 and iodine-125 now replacing older gold- and iridium-based therapies. Modern intracranial brachytherapy has been studied in atypical and anaplastic meningiomas, low- and high-grade gliomas, and metastases (34, 35). Most recently, the use of cesium-131 in brain metastases was reported by Wernicke et al. from a Phase I/II trial (36). Twenty-four patients underwent first-time gross-total resection of a brain metastasis and intraoperative placement of a permanent cesium-131 source with a planned dose of 80 Gy to a surface depth of 5 mm beyond the resection cavity. The patients had no local recurrences, no incidents of symptomatic radiation necrosis, and minimal surgical morbidity. This study was limited by its small size, limited follow-up, and the confounding variable of gross-total resection, which is associated with lower rates of recurrence and progression. Future studies will likely confirm these promising preliminary results with cesium-based brachytherapy for treatment of brain metastases.
Laser Interstitial Thermal Therapy
The use of MR-guided laser interstitial thermal therapy (LITT) for metastasis has been reported in the neurosurgical literature. MR-guided thermal ablation is not a new technology, but recent advances in materials and methods have significantly improved the ability to ablate lesion tissue accurately and safely while sparing nearby brain tissue. Two series have had good results (albeit in small samples with short follow-up) for tumors that failed to respond to traditional radiotherapy and subsequently underwent LITT. The technology allows biopsy and subsequent laser ablation from a small (approximately 4 mm) access port inserted in the operating room. Carpentier et al. described four patients with six tumors treated with LITT without complications; no tumors recurred within the 90-day follow-up (37). Hawasli and colleagues demonstrated similar results using LITT for various lesions, including five metastases (38). Two patients suffered transient neurological morbidity (one aphasia and one hemiparesis), and two had progression of CNS disease 2.2 and 3.5 months after LITT. Nevertheless, LITT has good prospects as a means to extend quantity and quality of life for patients with radiation-resistant brain metastases in eloquent or deep locations who are left with few options. Furthermore, LITT can ablate radiation treatment effect found on biopsy. Further study is warranted.
Personalized Metastasis Treatment
As our technological ability to successfully treat brain metastases has grown in recent decades, so too has our knowledge of the intricate biology of tumorigenesis. The CNS is different from other organs, as blood-borne metastatic cells must first overcome the blood–brain barrier after escaping their primary site of origin. Once these cells pass this barrier, they must establish themselves in a biological niche with a milieu of cytosolic growth factors unlike those of their site of origin. Lastly, once the cells have grown into a macroscopic tumor, different metastatic brain tumors have variable responses to irradiation and chemotherapy due to genetic and epigenetic alterations and poor penetration of the blood–brain barrier by some targeted chemotherapies. Each stage offers the potential for personalized, targeted intervention or, at least, better prognostication based on molecular (vs. histological) disease stratification. Many surgical clinical trials have grouped all metastatic tumors when evaluating new treatment strategies, but these lesions clearly have numerous biological differences despite their commonality as “brain metastases.”
Molecular Initiation of Distal Metastases
The molecular pathophysiology of brain metastasis has been the focus of extensive research. The ability to predict, via primary tumor tissue, which cancer patients will suffer brain metastasis would facilitate prognostication and focus metastasis screening efforts. Numerous lung cancer researchers have attempted to correlate single gene mutations and chromosomal translocations with the development of brain metastases. For example, Lee et al. found that chromosomal amplifications of regions 5q35, 10q23, and 17q23–24 were associated with early development of brain metastases within 3 months of initial tumor diagnosis (39). The exact mechanisms by which these amplifications lead to lung-to-brain metastasis are not yet understood. Genes associated with the development of brain metastases in lung cancer include PLGF, VEGFR1, c-MET, and CXCR4, all of which are targets for further investigation (40–42). HER2-positivity predisposes patients with breast cancer to the development of brain metastases. This predisposition to brain metastases is probably caused by a combination of increased general HER2-positive tumor aggressive behavior as well as HER2-specific neural tropism via downstream pathways, such as TGF-β (43–45). Lastly, the pathophysiology of metastasis is not limited to protein-coding genes. Long non-coding RNA MALAT1 is associated with numerous cancer types and aggressive tumor behavior, despite a relatively poor understanding of the exact function of this highly preserved non-coding RNA. Regardless, high tumor levels of MALAT1 RNA are correlated with poor overall survival in patients with lung-to-brain metastasis, and MALAT1 promotes brain metastasis via the induction of epithelial to mesenchymal transitions (46). Further research is warranted on this long non-coding RNA, which represents an interesting non-protein target for personalized lung metastasis therapy.
Breaching the Blood–Brain Barrier
Once individual tumor cells have hematologically spread to the cerebral microvasculature, they must exit into the perivascular space across the blood–brain barrier to propagate macroscopic tumors. Pharmacological blockade of this transgression is a highly appealing strategy to prevent the formation of brain metastases. Research on multiple tumor types has elucidated the mechanics of this process, although much work remains. An elegant murine model by Kienast and colleagues characterized the individual steps of metastasis formation as tumor cells reach the brain (47). First, individual tumor cells arrest in tiny vessel branches. Next, cells that go on to form macroscopic tumors transmigrate across the vasculature wall within 72 h after being lodged into the capillary. After transmigration, formation of a macroscopic tumor requires that tumor cells proliferate in direct contact with endothelial cells of the brain capillary akin to a pericyte. Cells that do not maintain proximity to the vessel wall regress. Lastly, vessel co-option and angiogenesis must allow for sufficient nutrient delivery to propagate the macrometastasis (47). Each step is driven by complex molecular interactions between the tumor cell and its surroundings, and all these interactions are potential targets for more directed, individualized therapies. The process of cellular transmigration out of the capillary is regulated by complex junctional adhesion molecules, and proteases that degrade these junctional adhesions are implicated in brain metastasis. For example, high levels of cathepsin S are negatively associated with overall brain-metastasis-free survival in patients with breast cancer (48). Depletion of cathepsin S in a murine model reduced in vivo experimental brain metastases, thus identifying another potential personalized target for those patients with high tumor cathepsin S expression (48). The degradation of the blood–brain barrier by tumor cells is regulated not only by protein-protein interactions but also by non-canonical means. Tominaga et al. demonstrated that breast cancer cells release extracellular vesicles, including microRNA such as miR-181C, which promotes the local destruction of the blood–brain barrier via actin fiber delocalization in a PDPK1-mediated fashion (49). Other exosomal microRNAs, such as miR-105, have also been implicated in the loss of cell–cell adhesion at tight junctions (50). The blockade of microRNA signaling pathways is not yet clinically practical but may represent future targeted therapies.
Metastatic Evolution
Once established within the brain parenchyma, metastatic tumor cells continue to evolve (Table 2). Excellent genomic studies have demonstrated that brain metastases harbor gene alterations distinct from the primary tumor. These alterations have widespread ramifications, especially for patients with inoperative brain metastases whose primary lesion is the only tissue available for molecular profiling for additional therapy selection. Brastianos et al. found that in 53% of tumors, clinically relevant alterations occurred in the brain metastasis but not in the primary tumor (51). Many of these mutations arose in the PI3K/AKT/mTOR, CDK, and HER2/EGFR pathways, all of which have inhibitors available for clinical use. In this same cohort, multiple distinct brain lesions were genetically homogeneous compared to the extracranial metastases (51). This genetic homogeneity has significant practical implications because personalized targeted therapies for multiple brain lesions are best chosen on the basis of molecular data from any single brain metastasis rather than from the more divergent primary tumor or extracranial metastatic disease. Similar results demonstrate significant genetic divergence between brain metastasis and primary tumor tissue, specifically for squamous cell lung cancer (52). Further DNA- and RNA-based high-throughput sequencing comparing primary tissue and brain metastases will shed additional light on the metastatic process (and subsequent potential therapies) in coming years.
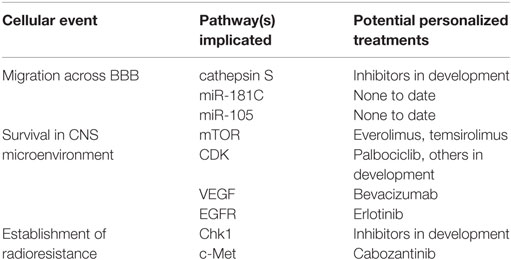
Table 2. Examples of metastatic events, their molecular processes, and potential targeted chemotherapeutics.
Targeted Drug Delivery
Personalization of metastatic cancer treatment aims to improve treatment by using select therapies chosen via molecular profiling to benefit the patient while sparing the patient from biologically irrelevant therapies with potential toxicity. In brain metastases, this strategy is limited because of poor penetration of most novel chemotherapeutics across the blood–brain barrier. Earlier research has used mannitol as a non-specific agent for blood–brain barrier permeation with limited success (53). Researchers have since demonstrated the ability to permeate the blood–brain barrier selectively in murine models at the site of metastases using intravenous tumor necrosis factor (54). Although in its infancy, MR-focused ultrasound combined with microbubbles represents another method of blood–brain barrier disruption but requires significant dedicated infrastructure (55). Many novel targeted delivery strategies are in development in multiple centers, making it likely that more options will become available in the future.
Advances in Radiotherapy
At times, the molecular biology of radiation resistance can be overcome, allowing for more effective delivery of radiotherapy. Xenograft brain metastasis in murine models have demonstrated improved survival and response to external beam radiation after inhibition of Chk1 (DNA damage checkpoint protein) and c-Met (receptor tyrosine kinase with downstream oncogenes) (56–59). Clinical trials are required to demonstrate a benefit in human patients.
Summary and Future Directions
Brain metastases represent a common source of morbidity and mortality for cancer patients. Current treatment paradigms include surgical resection, radiotherapy, and chemotherapy. Recent advances in intraoperative surgical technology (i.e., fluorescence, confocal microscopy, and brachytherapy) hold promise for improved outcomes for brain metastasis resection. The future of brain metastasis management is predicated on personalized therapy targeted to specific tumor molecular pathways, such as those involved in blood–brain barrier transgression, cell–cell adhesion, and angiogenesis. Brain metastases are often biologically distinct lesions compared to the primary tumor. Personalized therapies should therefore be chosen on the basis of brain metastasis tissue whenever available. The multidisciplinary management of patients with brain metastases by neurosurgeons, medical oncologists, and radiation oncologists is essential as therapies become increasingly complex and individualized.
Author Contributions
All authors listed have made substantial, direct, and intellectual contribution to the work and approved it for publication.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Abbreviations
5-ALA, 5-aminolevulinic acid; CNS, central nervous system; LITT, laser interstitial thermal therapy; WBRT, whole-brain radiotherapy.
References
1. Barnholtz-Sloan JS, Sloan AE, Davis FG, Vigneau FD, Lai P, Sawaya RE. Incidence proportions of brain metastases in patients diagnosed (1973 to 2001) in the Metropolitan Detroit Cancer Surveillance System. J Clin Oncol (2004) 22:2865–72. doi:10.1200/JCO.2004.12.149
2. Kohler BA, Ward E, McCarthy BJ, Schymura MJ, Ries LA, Eheman C, et al. Annual report to the nation on the status of cancer, 1975-2007, featuring tumors of the brain and other nervous system. J Natl Cancer Inst (2011) 103:714–36. doi:10.1093/jnci/djr077
3. Patchell RA, Tibbs PA, Walsh JW, Dempsey RJ, Maruyama Y, Kryscio RJ, et al. A randomized trial of surgery in the treatment of single metastases to the brain. N Engl J Med (1990) 322:494–500. doi:10.1056/NEJM199002223220802
4. Greene-Schloesser D, Robbins ME, Peiffer AM, Shaw EG, Wheeler KT, Chan MD. Radiation-induced brain injury: a review. Front Oncol (2012) 2:73. doi:10.3389/fonc.2012.00073
5. Linskey ME, Andrews DW, Asher AL, Burri SH, Kondziolka D, Robinson PD, et al. The role of stereotactic radiosurgery in the management of patients with newly diagnosed brain metastases: a systematic review and evidence-based clinical practice guideline. J Neurooncol (2010) 96:45–68. doi:10.1007/s11060-009-0073-4
6. Tsao MN, Rades D, Wirth A, Lo SS, Danielson BL, Gaspar LE, et al. Radiotherapeutic and surgical management for newly diagnosed brain metastasis(es): an American Society for Radiation Oncology evidence-based guideline. Pract Radiat Oncol (2012) 2:210–25. doi:10.1016/j.prro.2011.12.004
7. Nabors LB, Portnow J, Ammirati M, Brem H, Brown P, Butowski N, et al. Central nervous system cancers, version 2.2014. Featured updates to the NCCN Guidelines. J Natl Compr Canc Netw (2014) 12:1517–23.
8. Soltys SG, Adler JR, Lipani JD, Jackson PS, Choi CY, Puataweepong P, et al. Stereotactic radiosurgery of the postoperative resection cavity for brain metastases. Int J Radiat Oncol Biol Phys (2008) 70:187–93. doi:10.1016/j.ijrobp.2007.06.068
9. Quigley MR, Fuhrer R, Karlovits S, Karlovits B, Johnson M. Single session stereotactic radiosurgery boost to the post-operative site in lieu of whole brain radiation in metastatic brain disease. J Neurooncol (2008) 87:327–32. doi:10.1007/s11060-007-9515-z
10. Mathieu D, Kondziolka D, Flickinger JC, Fortin D, Kenny B, Michaud K, et al. Tumor bed radiosurgery after resection of cerebral metastases. Neurosurgery (2008) 62:817–23; discussion 823–814. doi:10.1227/01.neu.0000316899.55501.8b
11. Jagannathan J, Yen CP, Ray DK, Schlesinger D, Oskouian RJ, Pouratian N, et al. Gamma Knife radiosurgery to the surgical cavity following resection of brain metastases. J Neurosurg (2009) 111:431–8. doi:10.3171/2008.11.JNS08818
12. McGirt MJ, Ambrossi GL, Datoo G, Sciubba DM, Witham TF, Wolinsky JP, et al. Recurrent disc herniation and long-term back pain after primary lumbar discectomy: review of outcomes reported for limited versus aggressive disc removal. Neurosurgery (2009) 64:338–44; discussion 344–335. doi:10.1227/01.NEU.0000337574.58662.E2
13. Berghoff AS, Rajky O, Winkler F, Bartsch R, Furtner J, Hainfellner JA, et al. Invasion patterns in brain metastases of solid cancers. Neuro Oncol (2013) 15:1664–72. doi:10.1093/neuonc/not112
14. Yoo H, Kim YZ, Nam BH, Shin SH, Yang HS, Lee JS, et al. Reduced local recurrence of a single brain metastasis through microscopic total resection. J Neurosurg (2009) 110:730–6. doi:10.3171/2008.8.JNS08448
15. Stummer W, Stocker S, Wagner S, Stepp H, Fritsch C, Goetz C, et al. Intraoperative detection of malignant gliomas by 5-aminolevulinic acid-induced porphyrin fluorescence. Neurosurgery (1998) 42:518–25; discussion 525–516. doi:10.1097/00006123-199803000-00017
16. Shinoda J, Yano H, Yoshimura S, Okumura A, Kaku Y, Iwama T, et al. Fluorescence-guided resection of glioblastoma multiforme by using high-dose fluorescein sodium. Technical note. J Neurosurg (2003) 99:597–603. doi:10.3171/jns.2003.99.3.0597
17. Stummer W, Pichlmeier U, Meinel T, Wiestler OD, Zanella F, Reulen HJ, et al. Fluorescence-guided surgery with 5-aminolevulinic acid for resection of malignant glioma: a randomised controlled multicentre phase III trial. Lancet Oncol (2006) 7:392–401. doi:10.1016/S1470-2045(06)70665-9
18. Koc K, Anik I, Cabuk B, Ceylan S. Fluorescein sodium-guided surgery in glioblastoma multiforme: a prospective evaluation. Br J Neurosurg (2008) 22:99–103. doi:10.1080/02688690701765524
19. Acerbi F, Broggi M, Eoli M, Anghileri E, Cavallo C, Boffano C, et al. Is fluorescein-guided technique able to help in resection of high-grade gliomas? Neurosurg Focus (2014) 36:E5. doi:10.3171/2013.11.FOCUS13487
20. Raabe A, Nakaji P, Beck J, Kim LJ, Hsu FP, Kamerman JD, et al. Prospective evaluation of surgical microscope-integrated intraoperative near-infrared indocyanine green videoangiography during aneurysm surgery. J Neurosurg (2005) 103:982–9. doi:10.3171/jns.2005.103.6.0982
21. Killory BD, Nakaji P, Gonzales LF, Ponce FA, Wait SD, Spetzler RF. Prospective evaluation of surgical microscope-integrated intraoperative near-infrared indocyanine green angiography during cerebral arteriovenous malformation surgery. Neurosurgery (2009) 65:456–62; discussion 462. doi:10.1227/01.NEU.0000346649.48114.3A
22. Hardesty DA, Thind H, Zabramski JM, Spetzler RF, Nakaji P. Safety, efficacy, and cost of intraoperative indocyanine green angiography compared to intraoperative catheter angiography in cerebral aneurysm surgery. J Clin Neurosci (2014) 21:1377–82. doi:10.1016/j.jocn.2014.02.006
23. Zaidi HA, Abla AA, Nakaji P, Chowdhry SA, Albuquerque FC, Spetzler RF. Indocyanine green angiography in the surgical management of cerebral arteriovenous malformations: lessons learned in 130 consecutive cases. Neurosurgery (2014) 10(Suppl 2):246–51; discussion 251. doi:10.1227/NEU.0000000000000318
24. Thind H, Hardesty DA, Zabramski JM, Spetzler RF, Nakaji P. The role of microscope-integrated near-infrared indocyanine green videoangiography in the surgical treatment of intracranial dural arteriovenous fistulas. J Neurosurg (2015) 122:876–82. doi:10.3171/2014.11.JNS14947
25. Schebesch KM, Hoehne J, Hohenberger C, Proescholdt M, Riemenschneider MJ, Wendl C, et al. Fluorescein sodium-guided resection of cerebral metastases – experience with the first 30 patients. Acta Neurochir (Wien) (2015) 157:899–904. doi:10.1007/s00701-015-2395-7
26. Kamp MA, Grosser P, Felsberg J, Slotty PJ, Steiger HJ, Reifenberger G, et al. 5-aminolevulinic acid (5-ALA)-induced fluorescence in intracerebral metastases: a retrospective study. Acta Neurochir (Wien) (2012) 154:223–8; discussion 228. doi:10.1007/s00701-011-1200-5
27. Sanai N, Eschbacher J, Hattendorf G, Coons SW, Preul MC, Smith KA, et al. Intraoperative confocal microscopy for brain tumors: a feasibility analysis in humans. Neurosurgery (2011) 68:282–90; discussion 290. doi:10.1227/NEU.0b013e318212464e
28. Martirosyan NL, Cavalcanti DD, Eschbacher JM, Delaney PM, Scheck AC, Abdelwahab MG, et al. Use of in vivo near-infrared laser confocal endomicroscopy with indocyanine green to detect the boundary of infiltrative tumor. J Neurosurg (2011) 115:1131–8. doi:10.3171/2011.8.JNS11559
29. Eschbacher J, Martirosyan NL, Nakaji P, Sanai N, Preul MC, Smith KA, et al. In vivo intraoperative confocal microscopy for real-time histopathological imaging of brain tumors. J Neurosurg (2012) 116:854–60. doi:10.3171/2011.12.JNS11696
30. Mooney MA, Zehri AH, Georges JF, Nakaji P. Laser scanning confocal endomicroscopy in the neurosurgical operating room: a review and discussion of future applications. Neurosurg Focus (2014) 36:E9. doi:10.3171/2013.11.FOCUS13484
32. Chase NE, Atkins HL, Correll JW. Interstitial irradiation of brain tumors with iridium 192. Radiology (1961) 77:842–3. doi:10.1148/77.5.842
33. Hosobuchi Y, Phillips TL, Stupar TA, Gutin PH. Interstitial brachytherapy of primary brain tumors. Preliminary report. J Neurosurg (1980) 53:613–7. doi:10.3171/jns.1980.53.5.0613
34. Ware ML, Larson DA, Sneed PK, Wara WW, McDermott MW. Surgical resection and permanent brachytherapy for recurrent atypical and malignant meningioma. Neurosurgery (2004) 54:55–63; discussion 63–54. doi:10.1227/01.NEU.0000097199.26412.2A
35. Vitaz TW, Warnke PC, Tabar V, Gutin PH. Brachytherapy for brain tumors. J Neurooncol (2005) 73:71–86. doi:10.1007/s11060-004-2352-4
36. Wernicke AG, Yondorf MZ, Peng L, Trichter S, Nedialkova L, Sabbas A, et al. Phase I/II study of resection and intraoperative cesium-131 radioisotope brachytherapy in patients with newly diagnosed brain metastases. J Neurosurg (2014) 121:338–48. doi:10.3171/2014.3.JNS131140
37. Carpentier A, McNichols RJ, Stafford RJ, Itzcovitz J, Guichard JP, Reizine D, et al. Real-time magnetic resonance-guided laser thermal therapy for focal metastatic brain tumors. Neurosurgery (2008) 63:ONS21–8; discussion ONS28–29. doi:10.1227/01.neu.0000335007.07381.df
38. Hawasli AH, Bagade S, Shimony JS, Miller-Thomas M, Leuthardt EC. Magnetic resonance imaging-guided focused laser interstitial thermal therapy for intracranial lesions: single-institution series. Neurosurgery (2013) 73:1007–17. doi:10.1227/NEU.0000000000000144
39. Lee HW, Seol HJ, Choi YL, Ju HJ, Joo KM, Ko YH, et al. Genomic copy number alterations associated with the early brain metastasis of non-small cell lung cancer. Int J Oncol (2012) 41:2013–20. doi:10.3892/ijo.2012.1663
40. Benedettini E, Sholl LM, Peyton M, Reilly J, Ware C, Davis L, et al. Met activation in non-small cell lung cancer is associated with de novo resistance to EGFR inhibitors and the development of brain metastasis. Am J Pathol (2010) 177:415–23. doi:10.2353/ajpath.2010.090863
41. Chen G, Wang Z, Liu XY, Liu FY. High-level CXCR4 expression correlates with brain-specific metastasis of non-small cell lung cancer. World J Surg (2011) 35:56–61. doi:10.1007/s00268-010-0784-x
42. Li B, Wang C, Zhang Y, Zhao XY, Huang B, Wu PF, et al. Elevated PLGF contributes to small-cell lung cancer brain metastasis. Oncogene (2013) 32:2952–62. doi:10.1038/onc.2012.313
43. Kallioniemi OP, Holli K, Visakorpi T, Koivula T, Helin HH, Isola JJ. Association of c-erbB-2 protein over-expression with high rate of cell proliferation, increased risk of visceral metastasis and poor long-term survival in breast cancer. Int J Cancer (1991) 49:650–5. doi:10.1002/ijc.2910490504
44. Montagna E, Cancello G, D’Agostino D, Lauria R, Forestieri V, Esposito A, et al. Central nervous system metastases in a cohort of metastatic breast cancer patients treated with trastuzumab. Cancer Chemother Pharmacol (2009) 63:275–80. doi:10.1007/s00280-008-0737-3
45. Katsuno Y, Lamouille S, Derynck R. TGF-beta signaling and epithelial-mesenchymal transition in cancer progression. Curr Opin Oncol (2013) 25:76–84. doi:10.1097/CCO.0b013e32835b6371
46. Shen L, Chen L, Wang Y, Jiang X, Xia H, Zhuang Z. Long noncoding RNA MALAT1 promotes brain metastasis by inducing epithelial-mesenchymal transition in lung cancer. J Neurooncol (2015) 121:101–8. doi:10.1007/s11060-014-1613-0
47. Kienast Y, von Baumgarten L, Fuhrmann M, Klinkert WE, Goldbrunner R, Herms J, et al. Real-time imaging reveals the single steps of brain metastasis formation. Nat Med (2010) 16:116–22. doi:10.1038/nm.2072
48. Sevenich L, Bowman RL, Mason SD, Quail DF, Rapaport F, Elie BT, et al. Analysis of tumour- and stroma-supplied proteolytic networks reveals a brain-metastasis-promoting role for cathepsin S. Nat Cell Biol (2014) 16:876–88. doi:10.1038/ncb3011
49. Tominaga N, Kosaka N, Ono M, Katsuda T, Yoshioka Y, Tamura K, et al. Brain metastatic cancer cells release microRNA-181c-containing extracellular vesicles capable of destructing blood-brain barrier. Nat Commun (2015) 6:6716. doi:10.1038/ncomms7716
50. Zhou W, Fong MY, Min Y, Somlo G, Liu L, Palomares MR, et al. Cancer-secreted miR-105 destroys vascular endothelial barriers to promote metastasis. Cancer Cell (2014) 25:501–15. doi:10.1016/j.ccr.2014.03.007
51. Brastianos PK, Carter SL, Santagata S, Cahill DP, Taylor-Weiner A, Jones RT, et al. Genomic characterization of brain metastases reveals branched evolution and potential therapeutic targets. Cancer Discov (2015) 5:1164–77. doi:10.1158/2159-8290.CD-15-0369
52. Paik PK, Shen R, Won H, Rekhtman N, Wang L, Sima CS, et al. Next-generation sequencing of stage IV squamous cell lung cancers reveals an association of PI3K aberrations and evidence of clonal heterogeneity in patients with brain metastases. Cancer Discov (2015) 5:610–21. doi:10.1158/2159-8290.CD-14-1129
53. Siegal T, Rubinstein R, Bokstein F, Schwartz A, Lossos A, Shalom E, et al. In vivo assessment of the window of barrier opening after osmotic blood-brain barrier disruption in humans. J Neurosurg (2000) 92:599–605. doi:10.3171/jns.2000.92.4.0599
54. Connell JJ, Chatain G, Cornelissen B, Vallis KA, Hamilton A, Seymour L, et al. Selective permeabilization of the blood-brain barrier at sites of metastasis. J Natl Cancer Inst (2013) 105:1634–43. doi:10.1093/jnci/djt276
55. Meairs S. Facilitation of drug transport across the blood-brain barrier with ultrasound and microbubbles. Pharmaceutics (2015) 7:275–93. doi:10.3390/pharmaceutics7030275
56. Mitchell JB, Choudhuri R, Fabre K, Sowers AL, Citrin D, Zabludoff SD, et al. In vitro and in vivo radiation sensitization of human tumor cells by a novel checkpoint kinase inhibitor, AZD7762. Clin Cancer Res (2010) 16:2076–84. doi:10.1158/1078-0432.CCR-09-3277
57. Yang H, Yoon SJ, Jin J, Choi SH, Seol HJ, Lee JI, et al. Inhibition of checkpoint kinase 1 sensitizes lung cancer brain metastases to radiotherapy. Biochem Biophys Res Commun (2011) 406:53–8. doi:10.1016/j.bbrc.2011.01.106
58. De Bacco F, Luraghi P, Medico E, Reato G, Girolami F, Perera T, et al. Induction of MET by ionizing radiation and its role in radioresistance and invasive growth of cancer. J Natl Cancer Inst (2011) 103:645–61. doi:10.1093/jnci/djr093
Keywords: brain metastasis, chemotherapy, personalized medicine, radiotherapy
Citation: Hardesty DA and Nakaji P (2016) The Current and Future Treatment of Brain Metastases. Front. Surg. 3:30. doi: 10.3389/fsurg.2016.00030
Received: 01 April 2016; Accepted: 11 May 2016;
Published: 25 May 2016
Edited by:
Eberval Figueiredo, University of São Paulo, BrazilReviewed by:
Hiroki Toda, Kitano Hospital, JapanKareem Zaghloul, National Institute of Neurological Disorders and Stroke, USA
Copyright: © 2016 Hardesty and Nakaji. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Peter Nakaji, bmV1cm9wdWJAZGlnbml0eWhlYWx0aC5vcmc=
 Douglas A. Hardesty
Douglas A. Hardesty Peter Nakaji
Peter Nakaji