- 1Department of Cardiovascular and Thoracic Surgery, Viet-Tiep Friendship Hospital, Hai Phong, Vietnam
- 2Department of Cardiovascular and Thoracic Surgery, Viet Duc University Hospital, Hanoi, Vietnam
- 3Department of Surgery, Hanoi Medical University, Hanoi, Vietnam
- 4Department of Cardiovascular and Thoracic Surgery, Hanoi Medical University Hospital, Hanoi, Vietnam
- 5Institute for Preventive Medicine and Public Health, Hanoi Medical University, Hanoi, Vietnam
Background: Little is known about video-assisted thoracoscopic surgery in the Nuss procedure (VATS-NUSS) and its postoperative outcomes in the resource-scarce conditions in clinical practice such as Vietnam. Available evidence in the literature was mostly reported from large institutions in developed countries. Hence, this study was conducted to review our initial large single-center experience in the use of the VATS-NUSS for patients with pectus excavatum (PE) within 5 years.
Methods: Data from 365 consecutive PE patients between January 2015 and December 2019 who were surgically treated with VATS-NUSS were retrospectively analyzed.
Results: Of 365 patients, median age at operation was 15.61 ± 3.73 years (range = 5–27 years), most being child and adolescent. Three hundred nine patients (84.65%) were male. PE was commonly detected at puberty (n = 328, 89.9%). Postoperatively, early complications consisted of pneumothorax (n = 5, 1.37%), pleural bleeding/pleural fluid (n = 2, 0.55%), pleural hematoma (n = 1, 0.27%), pneumonia (n = 1, 0.27%), surgical wound infection (n = 1, 0.27%), incision fluid accumulation (n = 3, 0.82%), metal bar infection (n = 1, 0.27%), atelectasis (n = 3, 0.82%), and fever (n = 8, 2.19%). Late complications included surgical wound infection (n = 2, 0.55%), metal bar deviation (n = 5, 1.37%), metal bar allergy (n = 10, 2.74%), recurrent PE (n = 2, 0.55%), and persistent PE (n = 5, 1.37%). No deaths occurred. In 175 patients (47.95%) experiencing bar removal, mean operative time for bar removal was 34.09 ± 10.61 min, and the length of hospitalization following bar removal was 2.4 ± 1.34 days; the most frequent complication was pneumothorax (n = 19, 10.85%). One wound infection and one incision fluid accumulation happened following bar removal. Favorable midterm to long-term postoperative outcomes were achieved.
Conclusions: From the beginning of the Vietnamese surgeons' experience, VATS-NUSS application obtained favorable outcomes with minimizing the occurrence of serious intraoperative and postoperative complications. Current rare evidence enables to give a real picture in the application, modification, and development of VATS-NUSS in the countries having similar resource-scarce conditions.
Introduction
With universal acceptance, the additional use of the video-assisted thoracoscopic surgery (VATS) technique in the Nuss procedure (NUSS) demonstrated an increased safety of the operation with decreased frequency of serious intraoperative and postoperative complications (1–6). Bufo et al. firstly, reported VATS use for the prevention of life-threatening intraoperative lesions of the mediastinum (7). The discussion is currently focused on the superiority of left, right, or bilateral thoracoscopic access; the necessity of CO2 insufflation; or the sternal elevation to improve the safety during surgical procedure (8–11). Although serious life-threatening complications after the NUSS are rare (12–14), VATS use during the operation may further reduce the perioperative risk. The available evidence in the literature was mostly reported from large institutions in developed countries, and no publications on the surgical outcomes and safety in the application of the VATS for NUSS were known in the resource-scarce conditions in clinical practice such as Vietnam.
To fill this knowledge gap, this study sought to review our large single-center experience in the use of the VATS technique in NUSS for the Vietnamese patients with pectus excavatum (PE) within 5 years. Our results might have some implications for establishing appropriate treatment strategies in the application, modification, and development of VATS-NUSS in the countries having similar resource-scarce conditions.
Methods
Patients
In this retrospective one-center study, we consecutively selected patients with the diagnosis of PE who underwent VATS for the NUSS procedure from January 2015 to December 2019. A total of 365 patients surgically treated for PE at our institution (Viet Duc University Hospital, Hanoi, Vietnam) were included in final analysis.
A comprehensive evaluation by complete history, physical examination, chest radiographs, electrocardiogram (ECG), pulmonary function test, echocardiogram, and computed tomography (CT) of the chest was performed for all patients. The demographic data, surgical data, early and late complications, length of hospital stay, and early, midterm, and long-term outcomes were also collected. All possible variables were finally included on the basis of clinical judgment, literature review, and availability in our hospital. The indications for surgical repair were two or more of the following criteria demonstrated by Dr. Nuss: (1) progression of the deformity, (2) exercise intolerance, (3) progressive chest pain or dyspnea, (4) restrictive ventilatory impairment, (5) Haller index >3.25, (6) previous failed Ravitch procedure, (7) cardiac compression, and (8) mitral valve prolapse. All procedures were performed with the NUSS, which included left, right, or bilateral VATS.
Surgical Techniques
All patients had a thoracic epidural for intraoperative anesthesia and postoperative pain control. The patient was placed in the supine position after general anesthesia. The arms of patients were kept abducted approximately 70–80° in relation to the body.
In the determination of anatomic landmarks in surgery, we determined the concave area, the center of the concave area, the edges of the concave area, and the highest point of the concave edge on either side of the concave circumference. The metal bar was placed along a straight line that was formed from the center of the concave area and the two highest points of the concave edge. The measurements of the thorax and the bending of the metal bar (sternum lift) were then performed.
A 5-mm trocar was placed in the marked position on the left chest wall. This point was located in the sixth intercostal space on the midaxillary line. After pumping low pressure CO2 (5 mm Hg) into the pleural cavity to collapse the lungs, the camera with a 30-degree optic was taken to the left chest to observe the entire chest and mediastinum during surgery. Skin incision was made above or below the position where the metal rod was intended to be placed in an intercostal space. One 2-cm vertical skin incision was made in the midaxillary line each side. In required cases with two-bar insertion, also only a skin incision was made between the two intended positions to place the two metal bars. Subcutaneous or submuscular dissections were performed from the cutaneous incision to the highest edge of the PE.
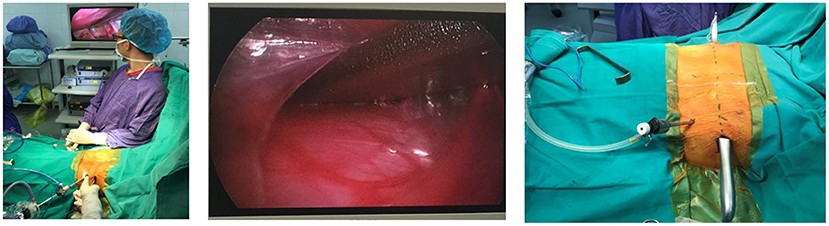
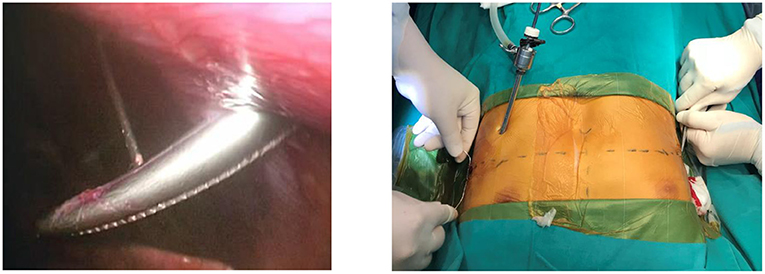
Then, we created a tunnel through the mediastinum under the control of thoracoscopy (Figure 1). The heart-shaped clamp was used to pierce the left pleural space at the highest edge of the pit, slowly went close to the anterior chest wall toward the mediastinum at the deepest point of the pit, and gradually separated the pericardium from the posterior sternum. The introducer reaches the posterior surface of the sternum in the left pleural cavity and further is moved through the mediastinal pleura in the retrosternal space, and then it went across the right pleural space and finally exits the chest through the right intercostal space. The ECG was continuously monitored during the process of created tunnel through the mediastinum.
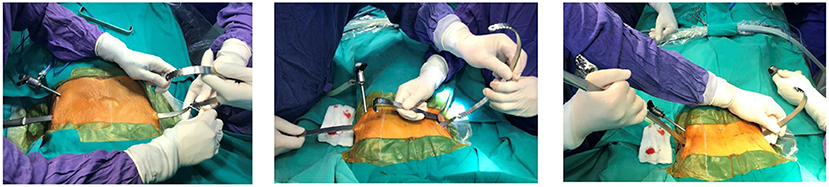
During the stage of reeving sternum lift bar, a Perlon thread was tied to the metal rod and then was tied to the introducer. Following the withdrawn introducer, the metal bar was threaded through the anterior mediastinum in a direction going from right to left under the control of the thoracoscopy (Figure 2). Then, the pectus bar was rotated to suit the patient's chest (Figure 3).
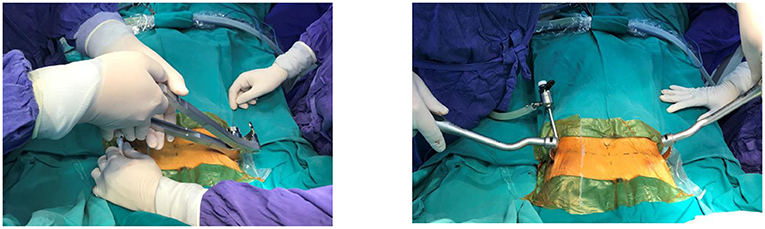
The steel thread was sewn around the ribs and was then tied to the top of the metal bar. Steel sutures on the one side combined with Vicryl sutures on the other were commonly utilized to fix the metal corrective bar. Bilateral steel sutures were used in severe PE patients with high risk of postoperative displacement of the steel support bar (Figure 4). The process of sewing steel threads was very safe under the observation of endoscopy. We did not use screw braces to fix the metal bar like the original Nuss surgery. Finally, the anesthesiologist squeezed the balloon through the endotracheal tube to allow the expanded lungs while gradually withdrawing the trocar and removing air from bilateral pleural space. Pleural drainage catheter was not placed.
Statistical Analysis
A visual inspection was first performed on all data for coding errors, outliers, or funky distributions. No imputation was made for missing data. Continuous variables were expressed as mean, standard deviation (SD) with interquartile ranges. Categorical variables were presented as counts with percentage. t Test, Mann–Whitney U test, χ2 test, and Fisher exact test were applied to examine the differences between patients with bar removal and patients without bar removal. p < 0.05 was considered statistically significant. Data were processed using Stata 15 (StataCorp LLC, USA) for Windows.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. Informed consents were obtained from all individual participants included in the study. If the participants are younger than 18 years, or otherwise legally or medically unable to provide written informed consent, then consent is obtained from their parents or guardian. This study was approved by the Ethics Board of the Hanoi Medical University (no. 01NCS17/HMU IRB).
Results
Preoperative Clinical Characteristics of the Patients
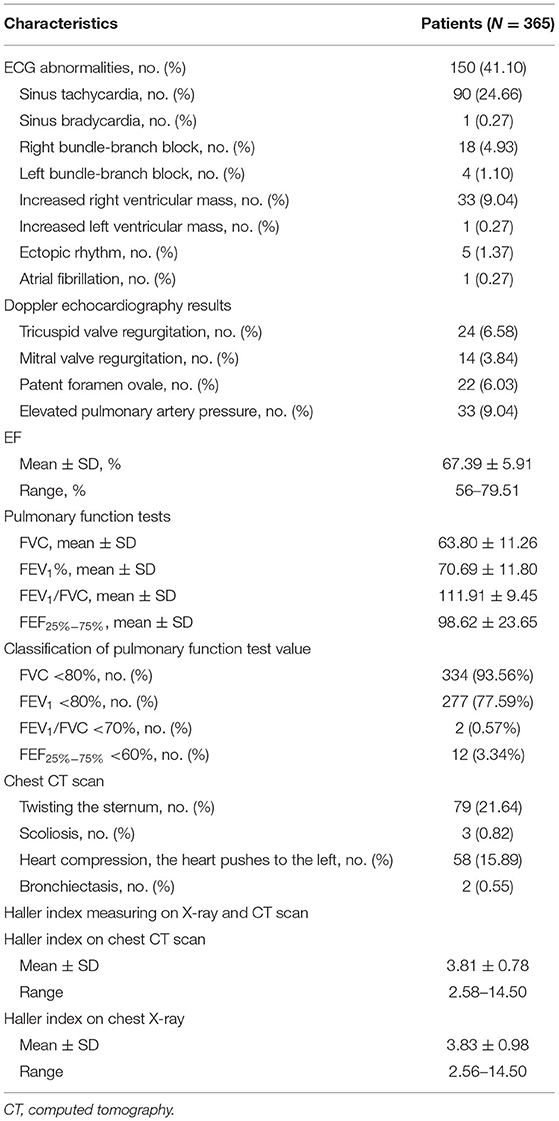
Of 365 PE patients, the median age at operation was 15.61 ± 3.73 years (range = 5–27 years), most being child and adolescent. Three hundred nine patients (84.65%) were male. PE was commonly detected at puberty (n = 328, 89.9%); 84.4% of the patients were underweight [body mass index (BMI) <18.5 kg/m2], and no patient was recorded as overweight and obese. There were 1.37% of the patients who experienced previous PE repair surgery (n = 5). Among 365 patients, 288 patients had symmetric PE (78.90%), and 77 patients had asymmetric PE (21.10%) (Table 1). The numeric distribution of each morphologic type and subtype of pectus is described in Table 1.
Palpitations were present in 81.64% of the patients (n = 298). Other common symptoms were inferiority complex (n = 262, 71.78%), dyspnea on exertion (n = 218, 59.73%), shortness of breath while exercising (n = 203, 55.62%), development of poor physical health (n = 192, 52.60%), and chest pain while exercising (n = 149, 40.82%) (Table 1).
Preoperative Subclinical Characteristics of the Patients
ECG abnormalities were documented in 41.10% of the patients (n = 150); the most common abnormality on ECG was sinus tachycardia (n = 90, 26.66%); increased right ventricular mass was present in 9.04% of the patients (n = 33), right bundle-branch block in 4.93% (n = 18), left bundle-branch block in 1.10% (n = 4), ectopic rhythm in 1.37% (n = 5), sinus bradycardia in 0.27% (n = 1), increased left ventricular mass in 0.27% (n = 1), and atrial fibrillation in 0.27% (n = 1). On Doppler echocardiography, 6.58% of patients had tricuspid valve regurgitation (n = 24), whereas the figure for mitral valve regurgitation was 3.84% (n = 14); patent foramen ovale was detected among 22 patients (6.03%); elevated pulmonary artery pressure was recorded in 33 patients (3.94%). Mean ejection fraction (EF) measured at 67.39 ± 5.91%. In 365 patients with PE presenting for surgical treatment, the mean forced vital capacity (FVC) was 63.80 ± 11.26, the mean forced expiratory volume in 1 second (FEV1) was 70.69 ± 11.8%, the mean FEV1/FVC was 111.91 ± 9.45, and the mean forced expiratory flow at 25–75% of the vital capacity (FEF25%−75%) was 98.62 ± 23.65. The mean Haller index was 3.81 ± 0.78 as measuring on chest CT scan and 3.83 ± 0.98 as measured on chest X-ray (Table 2).
Operative Characteristics of the Patients
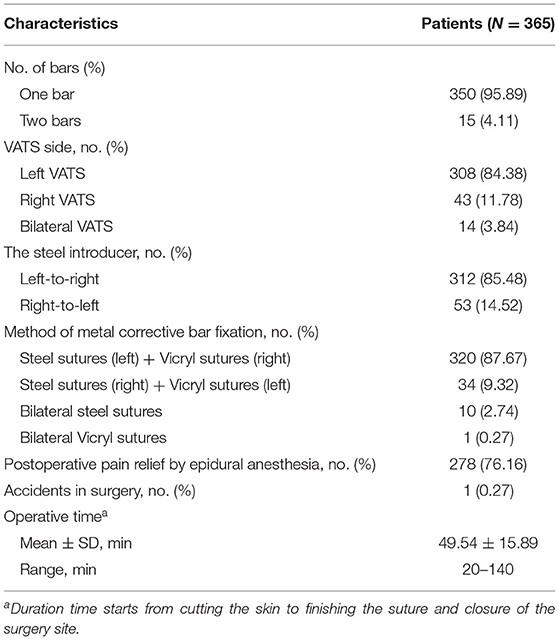
One bar was indicated for most patients (n = 350, 95.89%), and two bars were required in 15 patients (4.11%). The mean operative time (from cutting the skin to finishing the skin suture) was 49.54 min; 85.48% of the patients were carried through the left operative wound from the left to the right pleural cavity (n = 312), whereas 14.52% were carried from the right to the left pleural cavity (n = 53). Steel sutures on the left side combined with Vicryl sutures on the right were commonly utilized to fix metal corrective bar (n = 320, 87.67%). The epidural anesthesia was used to postoperative pain relief in 278 patients (76.16%). Intraoperative bleeding occurred in one case (Table 3).
Postoperative Characteristics of the Patients
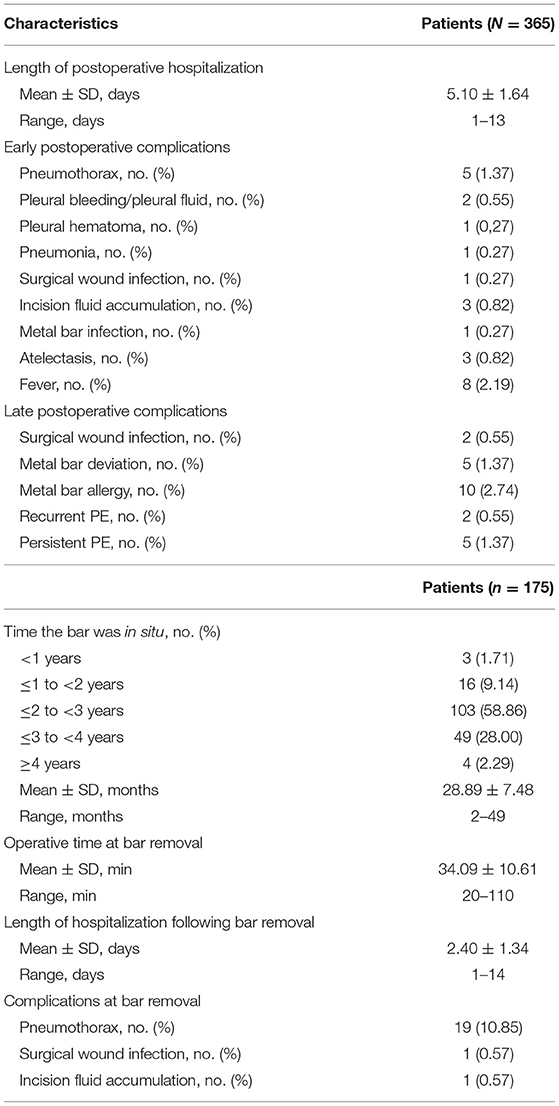
The length of postoperative hospitalization ranged from 1 to 13 days (mean = 5.1 days). Early postoperative complications were considered to be up to 30 days during hospital stay. The most common was pneumothorax (n = 5, 1.37%). Other events largely consisted of pleural bleeding/pleural fluid (n = 2, 0.55%), pleural hematoma (n = 1, 0.27%), pneumonia (n = 1, 0.27%), surgical wound infection (n = 1, 0.27%), incision fluid accumulation (n = 3, 0.82%), metal bar infection (n = 1, 0.27%), atelectasis (n = 3, 0.82%), and fever (n = 8, 2.19%). In addition, surgical wound infection (n = 2, 0.55%), metal bar deviation (n = 5, 1.37%), metal bar allergy (n = 10, 2.74%), recurrent PE (n = 2, 0.55%), and persistent PE (n = 5, 1.37%) were late postoperative complications. No deaths occurred in the present cohort (Table 4).
Bar Removal After the VATS-NUSS
Table 4 also indicated the outcomes related to the bar removal that was carried out in 175 patients (47.95%). The time the bar was in situ ranged from 2 to 49 months (mean = 28.89 ± 7.48 months). In three patients (1.71%), bar removal was performed early, <1 year, because of metal bar allergy. We recorded the mean operative time at bar removal of 34.09 ± 10.61 min and the length of hospitalization following bar removal of 2.4 ± 1.34 days. Pneumothorax, the most frequent complication, was diagnosed in 19 patients (10.85%). Besides, surgical wound infection in one patient and incision fluid accumulation in one patient were documented following bar removal.
Midterm to Long-Term Outcome of Patients Undergoing VATS-NUSS
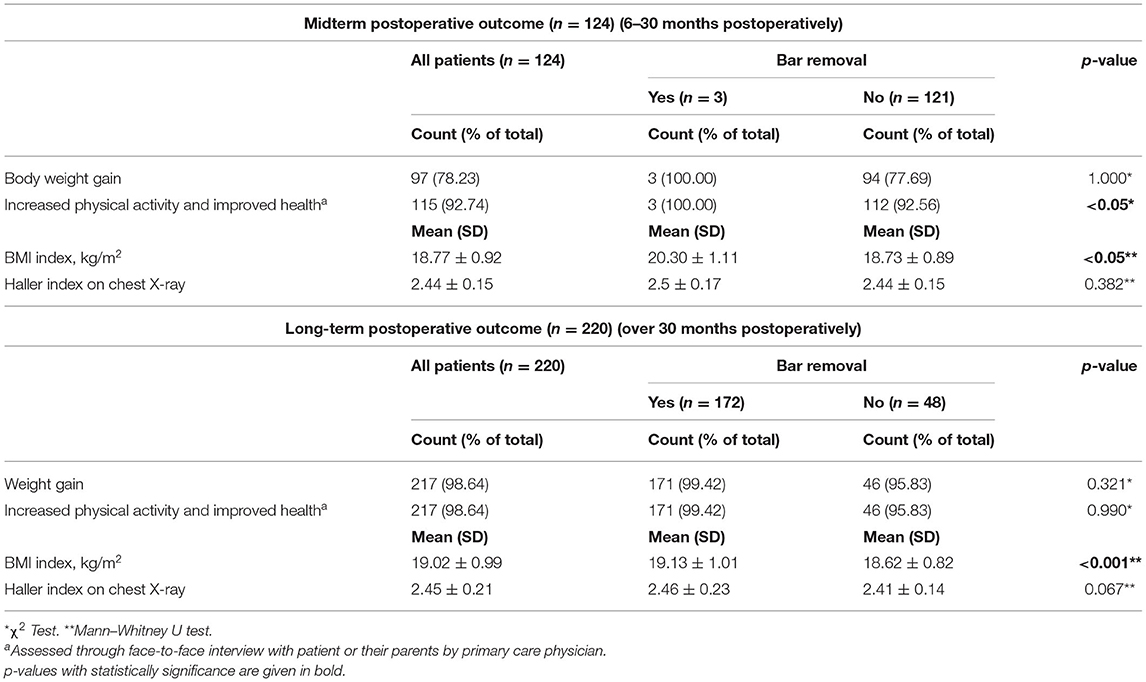
One hundred twenty-four of 365 patients (33.97%) were documented with midterm results at 6–30 months postoperatively. Of 124 patients, most patients had unremoved bar (n = 121, 97.58%), whereas only three patients had the bar removed (2.42%). Most patients had body weight gain (n = 97, 78.23%) and increased physical activity and improved health (n = 115, 92.74%) in the postoperative midterm period. The mean BMI index was 18.77 ± 0.92 kg/m2, and the Haller index on chest X-ray was 2.44 ± 0.15. Postoperative long-term results over 30 months were also assessed with 220 (60.27%) of 365 patients (Table 5). Of these, 172 received bar removal (78.18%), and 48 had not yet received bar removal (21.82%). Two hundred seventeen patients (98.64%) had both body weight gain and increased physical activity and improved health. The mean BMI index was 19.02 ± 0.99 kg/m2, and the Haller index on chest X-ray was 2.45 ± 0.21.
Discussion
In the emerging clinical practice in a developing country such as Vietnam, it difficult for us to acquire all important data for a large cohort of consecutive PE patients who underwent VATS for the NUSS procedure. Hence, from the Vietnamese surgeons' initial experience, we believe that the results from this cohort enable to give a real condition in clinical practice valuably not only for Vietnam but also for other countries having similar resource-scarce settings. In Vietnam, the cost and insurance coverage for the PE treatment have not been the main barriers for the patients and their family. The main issue in our condition is that there is no primary PE screening system at postpartum, evident in the late detection of PE. Although the number of children with PE that was detected at an early stage increased within the recent 5 years in Vietnam's large surgical institutions, many barriers to timely referral exist in remote rural and mountainous areas. With the ongoing development in other developed countries, many charity organizations across the world have come to Vietnam to lend a helping hand in the screening and treatment of PE; however, that concern is not enough to have a general picture in the detection and treatment of PE in Vietnam. To date, several ethnic minority communities lack adequate knowledge about PE in children and its lesions, with our primary healthcare system lacking good communication for birth defects between hospitals and the community. The mean age in our study is 15.61 ± 3.73 years (range = 5–27 years). When dividing the age according to the school-age groups of Vietnamese children, we found that the highest percentage of children undergoing VATS for the NUSS procedure were in 12- to 15-year age group (41.64%), which was known as the child's puberty period. In our institution, usually, the family takes the child to the hospital for examination and treatment when the child has evidently presented PE symptoms. Noticeably, 48.23% was in the age of 16 years or older, partially showing that the families' understanding and awareness of PE are still limited in Vietnam, and also gaps exist in the national surveillance and public health surveillance systems of congenital anomalies. As known from the literature, proper metal bar shape is crucial in NUSS for PE patients. Kelly et al. favored a gentle semicircular curve laterally with a 2- to 4-cm flat segment in the center to support the sternum (16). Slight overcorrection of the depression is to be preferred to undercorrection because the authors believed it minimizes the risk of recurrence and decreases the risk of buckling of the cartilages (16). Park et al. favored an asymmetrically bent bar for patients with an asymmetric depression (17). In this cohort, we used a ruler bent to the desired thoracic shape and then bent the metal bar to the shape and size of the ruler. We began to bend the middle of the bar gradually to its ends; bars are usually taken to be 1 inch smaller compared to the distance between the two middle armpit lines. We have found that symmetrically bent bars tend to be stable on the patients' chest.
In our patient series, most patients received only one bar (95.89%), whereas 4.11% of the patients received two bars. We indicated the two-bar insertion for most elderly patients with severe PE and wide concave area. No case in our institution had three metal bars placed yet. Our finding was consistent with previous reports when both revealed that the number of implanted steel bars was greater with age (18–20). Compared to previous studies (19, 20), the number of patients receiving two metal bars was lower in our study. The patient's chest wall becomes more solid after puberty and harder to bend; hence, introducing more than one bar is done to prevent postoperative pain and pressure on the chest wall by preventing displacement of the bar. Previous authors suggest that the PE patients with Marfan syndrome or grand canyon PE and asymmetrical PE require two metal bars. In addition, the use of two metal bars during the first treatment also becomes the standard of minimally invasive surgery for PE in several reports. To date, not only does the rate of using two metal bars increase gradually in recent years, but also the insertion of three metal bars (21–23).
Previous reports have shown the safety of thoracoscopic NUSS, with decreased frequency of serious intraoperative and postoperative complications, especially in patients with complex PE, a previous history of intrathoracic surgery and recurrent PE (7, 9, 15). VATS technique was acquired for all PE patients in the current cohort, and CO2 insufflation was used to create an artificial pneumothorax with the aim of increasing the empty pleural space. In the early years of implementing VATS in NUSS in our institution, the surgeons stand to the right of the patient using the supportive thoracoscopic approach, consistent with the classic NUSS that chooses the entrance from the right chest wall. Then, the left thoracoscopy is mostly used during recent years (19, 24) because we find this surgical access in line with the doctors' experience in our institution. In particular, bilateral thoracoscopic approach was applied in 14 patients because of severe PE compressing the lungs and heart and failed Ravitch procedure. In the early years of implementing VATS in NUSS, we applied both thoracoscopic approaches including the right chest wall and the left chest wall. In our opinion, the surgical field of view in the left thoracoscopic approach is wide enough to clearly observe the entire pleural cavity, left lung, blood vessel, heart, pericardium, mediastinum, and diaphragm; hence, serious complications can be avoided. Several authors favored placing the trocar in the middle axillary line (11, 25). However, we found that the trocar placed in the middle axillary line can result in that the vision can be hindered by the heart, lungs, and diaphragm, especially in cases of asymmetrical severe PE. We agree with Hendrickson et al. and Palmer et al. that left thoracoscopy has the advantage over right thoracoscopy and does not increase the risk of heart injury (8, 10). Hemostasis is well-controlled, and lung damage is avoided under endoscopic observation. During the process of ballooning through the endotracheal tube to enlarge the bilateral lungs, we clearly observed that the two lungs were enlarged, and until the lungs were maximized, when the pleural space was empty, we withdrew the endoscopic trocar. Therefore, pleural drainage catheter was not placed in surgery.
During the surgery, no serious complications and no fatalities occurred in our series. Operative bleeding complication was observed in one case. This is the case of intercostal arterial injury in the position of attaching the steel bar to the rib on left side. Effective hemostasis is then controlled by thoracoscopy. The most frequent complication in the early postoperative period was pneumothorax. The occurrence of this complication varies widely between 1.3 and 64% in the literature (9, 26–29). In our patients, the pneumothorax resolved spontaneously and did not require additional surgical intervention. Besides, no patient with postoperative bleeding required a blood transfusion, and no other serious complications or death was recorded in our study. Late postoperative complications are events that occur when the patient is discharged from the hospital, such as metal bar deviation, metal bar allergy, or surgical wound infection. Bar displacement is one of the most common complications (30). We recorded a bar displacement rate of 1.37%, and no major bar displacement in all patients. According to early literature, the incidence of bar displacement was 3–20%, and this incidence is lower in our study (1.37%) (15, 17, 31). Our approach with the mechanism-based bar fixation techniques by case-by-case analysis seems to be an effective and safe method of bar fixation in minimally invasive PE repair. Metal bar allergy also occurred in 2.74% of our patients (n = 10) with their manifestations such as mild fever, inflammation, wound fluid buildup, blisters, and erythema. Our study had similar proportion of metal bar allergy compared to others such as that of Nuss and Kelly (32) with 2.9% and Kelly et al. (16) of 2.8%. In particular, we found that 6 of 10 patients with metal bar allergy had a history of allergic diseases such as allergic rhinitis, drug allergy, weather allergy, food allergy, and asthma. Here we queried only for history of allergic diseases prior to surgery. The test for metal allergy before the operation was not available in clinical condition at our institution; therefore, we were incapable of testing routinely those for metal allergy. This helps us gain more experience in the prognosis, diagnosis, and treatment of allergy drugs, as well as closely monitor allergy status after surgery for appropriate management measures.
NUSS is known as a prepared clean surgery. Although very few wound infections were reported, these are extremely serious because a foreign material has been placed inside the body. In our study, there were two patients with late wound infection (0.55%). They received prompt antibiotic treatment and were discharged after 7- to 10-day treatment. There were no cases of deep infection to metal bar. Some studies in the literature showed the incidence of late wound infection was 1.5% (33, 34). In addition, other complications should be also noted in our report. Five patients had chest wall pain lasting 1 and 2 months postoperatively, for which oral pain relievers were taken and breathing exercises were performed; their pain then subsided and disappeared. There were two cases of recurrent PE and five cases of persistent PE after bar removal operation.
Midterm to long-term postoperative outcomes in PE patients were also assessed in the present cohort. Most patients had body weight gain after surgery. Through information self-reported by patients or their family, we also found increased physical activity and improved health. The Haller index, which is the ratio of the widest transverse (T) diameter of the chest to the shortest distance between the anterior spine and posterior sternum, has been used to quantify the severity of PE. Also, the size of the PE was postoperatively evaluated according to the Haller index based on two projection chest X-rays—posteroanterior and right lateral. We measured the mean Haller index of 2.44 ± 0.15 at 6 to 30 months postoperatively, while the Haller index on chest X-ray was 2.45 ± 0.21 over 30 months. In our patient cohort, bar removal was indicated based on the appropriate duration of bar maintenance, removal techniques, and strategies to avoid complications. For children up to 12 years old, the bar was maintained for 2 years, whereas in 12- to 20-year-old individuals, it was maintained for 2.5 years, and that in adults older than 20 years was maintained for 3 years.
For the first time in Vietnam, we conducted a large retrospective cohort of consecutive patients with the diagnosis of PE undergoing VATS for the NUSS procedure at a major academic Medical Center (Viet Duc University Hospital). Therefore, our study should be viewed in light of its main limitations. The study's retrospective nature and being a single-center study need be acknowledged. Because this was a retrospective review of medical records, the follow-up period data from the patients' medical records in our institution were available only for 124 of 365 patients at 6 to 30 months postoperatively and 220 of 365 patients at 30 months postoperatively. Our study with relative sample size and from one institution needs to be highlighted, potentially showing a limited validity and not enabling to represent those of other centers. In addition, because of the unavailability of retrospective medical records, we were unable to provide data about PE recurrence after the removal of metal bars as well as midterm and long-term postoperative pain. Because of the scope of study objectives, it is interesting for the authors to, in detail, evaluate the different postoperative time points and surgical outcomes, as well as to establish prognostic models for postoperative results in a further study. In particular, the metal support is still in place in a number of PE patients, not enabling us to have complete evaluation of postoperative results in the present study.
Conclusions
The VATS technique in NUSS for PE was a safe and effective operation, with minimizing the occurrence of serious intraoperative and postoperative complications. From our initial one-institution experience in a lower-middle income country, favorable midterm to long-term postoperative outcomes in PE patients were obtained with VATS treatment using the minimally invasive Nuss method. Current rare evidence drawn from this cohort enables to give a real picture on the application, modification, and development of VATS in NUSS not only for Vietnam but also for other countries having similar resource-scarce conditions.
Data Availability Statement
The original contributions generated for this study are included in the article/supplementary materials, further inquiries can be directed to the corresponding author/s.
Ethics Statement
The studies involving human participants were reviewed and approved by the Ethics Board of the Hanoi Medical University (No. 01NCS17/HMU IRB). Informed consents were obtained from all individual participants included in the study. If the participants are under the age of 18, or otherwise legally or medically unable to provide written informed consent, then consent is obtained from their parents or guardian.
Author Contributions
T-MN and Q-HD: conceptualization and writing—original draft preparation. T-MN, V-TL, H-UN, and Q-HD: methodology, validation, and resources. N-KD, H-LV, and K-DV: software. H-LV and K-DV: formal analysis. T-MN, H-UN, H-SP, N-TV, V-AN, and Q-HD: investigation and visualization. Q-HD: data curation and supervision. T-MN, H-UN, H-SP, N-TV, V-AN, H-LV, and Q-HD: writing—review and editing. N-KD and K-DV: project administration. T-MN: funding acquisition. All authors have read and agreed to the submitted version of the manuscript.
Disclaimer
The views expressed in this article were solely those of the authors and do not represent the official positions of the organizations that the authors are affiliated with.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
We thank all patients and their families for their support of the research. We would also like to thank the Board of Directors of Viet Duc Hospital for supporting the best conditions for the authors to complete this research.
References
1. Antonoff MB, Saltzman DA, Hess DJ, Acton RD. Retrospective review of reoperative pectus excavatum repairs. J Pediatr Surg. (2010) 45:200–5. doi: 10.1016/j.jpedsurg.2009.10.036
2. Croitoru DP, Kelly RE Jr., Goretsky MJ, Gustin T, Keever R, Nuss D. The minimally invasive Nuss technique for recurrent or failed pectus excavatum repair in 50 patients. J Pediatr Surg. (2005) 40:181–7. doi: 10.1016/j.jpedsurg.2004.09.038
3. Miller K, Ostlie D, Wade K, Chaignaud B, Gittes G, Andrews W, et al. Minimally invasive bar repair for ‘redo’correction of pectus excavatum. J Pediatr Surg. (2002) 37:1090–2. doi: 10.1053/jpsu.2002.33883
4. Redlinger RE Jr., Kelly RE Jr., Nuss D, Kuhn MA, Obermeyer RJ, Goretsky MJ. One hundred patients with recurrent pectus excavatum repaired via the minimally invasive Nuss technique—effective in most regardless of initial operative approach. J Pediatr Surg. (2011) 46:1177–81. doi: 10.1016/j.jpedsurg.2011.03.048
5. Schaarschmidt K, Kolberg-Schwerdt A, Lempe M, Schlesinger F, Bunke K, Strauss J. Extrapleural, submuscular bars placed by bilateral thoracoscopy—a new improvement in modified Nuss funnel chest repair. J Pediatr Surg. (2005) 40:1407–10. doi: 10.1016/j.jpedsurg.2005.05.039
6. Takagi S, Oyama T, Tomokazu N, Kinoshita K, Makino T, Ohjimi H. A new sternum elevator reduces severe complications during minimally invasive repair of the pectus excavatum. Pediatr Surg Int. (2012) 28:623–6. doi: 10.1007/s00383-012-3087-5
7. Bufo AJ, Stone MM. Addition of thoracoscopy to Nuss pectus excavatum repair. Pediatr Endosurg Innov Tech.. (2001) 5:159–62. doi: 10.1089/10926410152403093
8. Hendrickson RJ, Bensard DD, Janik JS, Partrick DA. Efficacy of left thoracoscopy and blunt mediastinal dissection during the Nuss procedure for pectus excavatum. J Pediatr Surg. (2005) 40:1312–4. doi: 10.1016/j.jpedsurg.2005.05.017
9. Cheng Y-L, Lee S-C, Huang T-W, Wu C-T. Efficacy and safety of modified bilateral thoracoscopy-assisted Nuss procedure in adult patients with pectus excavatum. Eur J Cardiothorac Surg.. (2008) 34:1057–61. doi: 10.1016/j.ejcts.2008.07.068
10. Palmer B, Yedlin S, Kim S. Decreased risk of complications with bilateral thoracoscopy and left-to-right mediastinal dissection during minimally invasive repair of pectus excavatum. Eur J Pediatr Surg.. (2007) 17:81–3. doi: 10.1055/s-2007-965012
11. Saxena AK, Castellani C, Höllwarth ME. Surgical aspects of thoracoscopy and efficacy of right thoracoscopy in minimally invasive repair of pectus excavatum. J Thorac Cardiovasc Surg. (2007) 133:1201–5. doi: 10.1016/j.jtcvs.2007.01.040
12. Gips H, Zaitsev K, Hiss J. Cardiac perforation by a pectus bar after surgical correction of pectus excavatum: case report and review of the literature. Pediatr Surg Int. (2008) 24:617–20. doi: 10.1007/s00383-007-2097-1
13. Zhang D-K, Tang J-M, Ben X-S, Xie L, Zhou H-Y, Ye X, et al. Surgical correction of 639 pectus excavatum cases via the Nuss procedure. J Thorac Dis.. (2015) 7:1595. doi: 10.3978/j.issn.2072-1439.2015.09.30
14. Schaarschmidt K, Lempe M, Schlesinger F, Jaeschke U, Park W, Polleichtner S. Lessons learned from lethal cardiac injury by Nuss repair of pectus excavatum in a 16-year-old. Ann Thorac Surg. (2013) 95:1793–5. doi: 10.1016/j.athoracsur.2012.09.038
15. Park HJ, Lee SY, Lee CS, Youm W, Lee KR. The Nuss procedure for pectus excavatum: evolution of techniques and early results on 322 patients. Ann Thorac Surg. (2004) 77:289–95. doi: 10.1016/S0003-4975(03)01330-4
16. Kelly RE, Goretsky MJ, Obermeyer R, Kuhn MA, Redlinger R, Haney TS, et al. Twenty-one years of experience with minimally invasive repair of pectus excavatum by the Nuss procedure in 1215 patients. Ann Surg. (2010) 252:1072–81. doi: 10.1097/SLA.0b013e3181effdce
17. Park HJ, Lee SY, Lee CS. Complications associated with the Nuss procedure: analysis of risk factors and suggested measures for prevention of complications. J Pediatr Surg. (2004) 39:391–5. doi: 10.1016/j.jpedsurg.2003.11.012
18. Pawlak K, Gasiorowski Ł, Gabryel P, Gałecki B, Zieliński P, Dyszkiewicz W. Early and late results of the Nuss procedure in surgical treatment of pectus excavatum in different age groups. Ann Thorac Surg. (2016) 102:1711–6. doi: 10.1016/j.athoracsur.2016.04.098
19. Pawlak K, Gasiorowski Ł, Gabryel P, Dyszkiewicz W. Video-assisted-thoracoscopic surgery in left-to-right Nuss procedure for pectus excavatum for prevention of serious complications–technical aspects based on 1006 patients. Videosurg Other Miniinvas Tech.. (2018) 13:95. doi: 10.5114/wiitm.2018.72683
20. Pilegaard HK, Licht PB. Routine use of minimally invasive surgery for pectus excavatum in adults. Ann Thorac Surg. (2008) 86:952–6. doi: 10.1016/j.athoracsur.2008.04.078
21. Nuss D, Kelly RE Jr., Croitoru DP, Katz ME. A 10-year review of a minimally invasive technique for the correction of pectus excavatum. J Pediatr Surg. (1998) 33:545–52. doi: 10.1016/S0022-3468(98)90314-1
22. Pilegaard HK. Extending the use of Nuss procedure in patients older than 30 years. Eur J Cardiothorac Surg. (2011) 40:334–7. doi: 10.1016/j.ejcts.2010.11.040
23. Stanfill AB, DiSomma N, Henriques SM, Wallace LJ, Vegunta RK, Pearl RH. Nuss procedure: decrease in bar movement requiring reoperation with primary placement of two bars. J Laparoendosc Adv Surg Tech.. (2012) 22:412–5. doi: 10.1089/lap.2011.0080
24. Cafarotti S, Memoli E, Patella M, Rugel G, Minerva E, Mendoza C, et al. Uniportal VATS for pectus excavatum: the Southern Switzerland experience. Eur Rev Med Pharmacol Sci. (2020) 24:9008–11. doi: 10.26355/eurrev_202009_22843
25. Nuss D, Obermeyer RJ, Kelly RE. Nuss bar procedure: past, present and future. Ann Cardiothorac Surg.. (2016) 5:422. doi: 10.21037/acs.2016.08.05
26. Shu Q, Shi Z, Xu W-Z, Li J-H, Zhang Z-W, Lin R, et al. Experience in minimally invasive Nuss operation for 406 children with pectus excavatum. World J Pediatr.. (2011) 7:257. doi: 10.1007/s12519-011-0319-z
27. Park HJ, Sung S-W, Park J-K, Kim JJ, Jeon HW, Wang YP. How early can we repair pectus excavatum: the earlier the better? Eur J Cardiothorac Surg. (2012) 42:667–72. doi: 10.1093/ejcts/ezs130
28. Peter SDS, Sharp SW, Ostlie DJ, Snyder CL, Holcomb GW III, Sharp RJ. Use of a subxiphoid incision for pectus bar placement in the repair of pectus excavatum. J Pediatr Surg. (2010) 45:1361–4. doi: 10.1016/j.jpedsurg.2010.02.115
29. Castellani C, Schalamon J, Saxena AK, Höellwarth ME. Early complications of the Nuss procedure for pectus excavatum: a prospective study. Pediatr Surg Int. (2008) 24:659–66. doi: 10.1007/s00383-008-2106-z
30. Hebra A, Swoveland B, Egbert M, Tagge EP, Georgeson K, Othersen HB, et al. Outcome analysis of minimally invasive repair of pectus excavatum: review of 251 cases. J Pediatr Surg. (2000) 35:252–8. doi: 10.1016/S0022-3468(00)90019-8
31. Croitoru DP, Kelly RE Jr., Goretsky MJ, Lawson ML, Swoveland B, Nuss D. Experience and modification update for the minimally invasive Nuss technique for pectus excavatum repair in 303 patients. J Pediatr Surg. (2002) 37:437–45. doi: 10.1053/jpsu.2002.30851
32. Nuss D, Kelly RE. Minimally invasive surgical correction of chest wall deformities in children (Nuss procedure). Adv Pediatr. (2008) 55:395–410. doi: 10.1016/j.yapd.2008.07.012
33. Shin S, Goretsky MJ, Kelly RE Jr, Gustin T, Nuss D. Infectious complications after the Nuss repair in a series of 863 patients. J Pediatr Surg. (2007) 42:87–92. doi: 10.1016/j.jpedsurg.2006.09.057
Keywords: VATS, Nuss, congenital pectus excavatum, complications, thoracoscopic surgery
Citation: Nguyen T-M, Le V-T, Nguyen H-U, Pham H-L, Phung H-SD, Vu N-T, Nguyen V-A, Do N-K, Vu K-D, Vo H-L and Doan Q-H (2021) An Initial 5-Year Single-Center Experience of 365 Patients Undergoing the Video-Assisted Thoracoscopic Surgery for Nuss Procedure for Pectus Excavatum in Resource-Scare Setting. Front. Surg. 8:693562. doi: 10.3389/fsurg.2021.693562
Received: 11 April 2021; Accepted: 13 May 2021;
Published: 14 June 2021.
Edited by:
Federico Raveglia, ASST-Monza, ItalyReviewed by:
Massimo Torre, Niguarda Ca' Granda Hospital, ItalyJin Yong Jeong, Catholic University of Korea, South Korea
Copyright © 2021 Nguyen, Le, Nguyen, Pham, Phung, Vu, Nguyen, Do, Vu, Vo and Doan. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Quoc-Hung Doan, aHVuZy5kb2FucXVvY0BobXUuZWR1LnZu; The-May Nguyen, bmd1eWVudGhlbWF5QGdtYWlsLmNvbQ==
 The-May Nguyen
The-May Nguyen Van-Thieu Le1
Van-Thieu Le1 Kim-Duy Vu
Kim-Duy Vu Hoang-Long Vo
Hoang-Long Vo Quoc-Hung Doan
Quoc-Hung Doan