- 1Department of Anesthesiology, First Affiliated Hospital of Soochow University, Suzhou, Jiangsu, China
- 2Institute of Anesthesiology, Soochow University, Suzhou, Jiangsu, China
- 3Department of Anesthesiology and Pain Medicine, University of California Davis Health System, Sacramento, CA, United States
Background: Video-assisted thoracoscopic surgery (VATS) has been widely performed for patients with lung cancer. Splenic rupture after VATS lung procedures is a very rare and serious event.
Case Presentation: We reported a case with hemodynamic instability after left lower VATS lobectomy. There was no evidence of diaphragmatic injury during the surgery. Computed tomography (CT) showed spleen injury and large amount of fluid in the abdominal cavity. Emergent laparotomy was performed, and splenic rupture was diagnosed. The patient underwent splenectomy, with two lacerations at the diaphragmatic surface of the spleen. The patient did well postoperatively and was discharged from the hospital on postoperative day 5.
Conclusion: There are few similar cases reported in the literature. Persistent hemodynamic instability due to the rupture of spleen is life-threatening. In the situation of unexplained hypotension during VATS procedures (especially left-sided approaches), the possibility of splenic injury and rupture should be considered. Abdominal ultrasonography and/or CT examinations should be carried out for prompt diagnosis and treatment of such rare complication.
Introduction
Video-assisted thoracoscopic surgical procedures (VATS) are widely performed for treatment of patients with lung cancer. Growing evidence have shown the advantages of VATS lobectomy over thoracotomy (1). Splenic rupture after VATS lung procedures is a very rare but catastrophic event. Here, we reported a case with persistent hemodynamic instability after VATS lobectomy. After the splenic rupture was eventually diagnosed, the patient received splenectomy. This report adheres to the CARE guidelines, with completed CARE checklist in Supplementary File S1.
Case Report
A 67-year-old female (165 cm height and 68 kg weight) was admitted to our hospital for left lower lung adenocarcinoma and underwent left lower VATS lobectomy. The patient was at American Society of Anesthesiologists physical status II with no major comorbidities. Hemoglobin (Hb) was 11.7 g/dL, and platelet count was 119 × 109/L. Preoperative laboratory tests revealed no abnormalities in coagulation function or other parameters.
Pre-induction non-invasive cuff blood pressure was 142/78 mmHg, heart rate was 72 beats/min, and peripheral oxygen saturation was 97%. The patient received an opioid-free anesthesia regimen. Dexmedetomidine 0.6 µg/kg was administered over 10 min. An arterial line was inserted into the right radial artery for measuring arterial blood pressure. Esketamine 0.3 mg/kg, propofol 2 mg/kg, and rocuronium 0.6 mg/kg were used for induction. A left-sided double-lumen endobronchial tube was used for intubation and confirmed by auscultation, capnography, and fiberoptic bronchoscopy. One-lung ventilation (tidal volume 350–400 mL, respiratory rate 12–18 breaths/min, I/E = 1:2, 60%–100% oxygen) was started. Anesthesia was maintained with sevoflurane 1%–3% inhalation, dexmedetomidine infusion 0.3–1.0 µg/kg/h, and intermittent esketamine boluses 0.1–0.2 mg/kg. Dexamethasone 5 mg and ondansetron 8 mg were given for prophylaxis of postoperative nausea and vomiting.
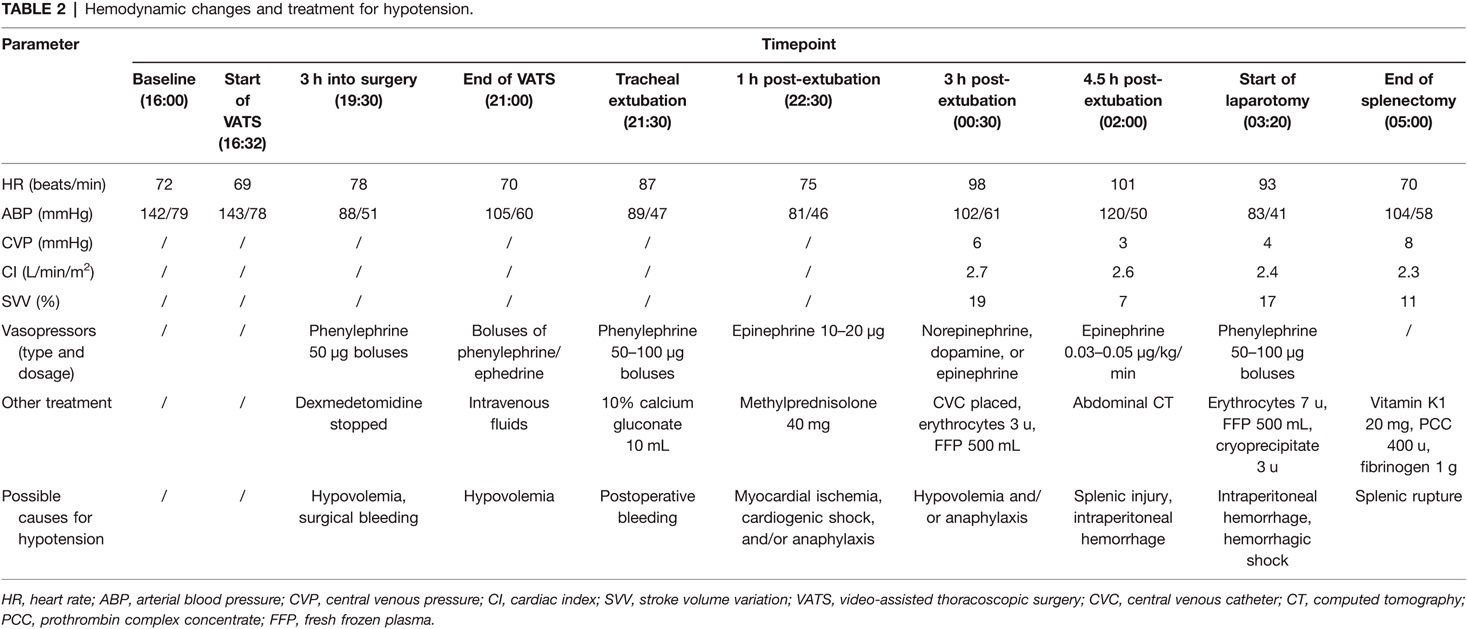
The surgical procedure started at 16:32. The patient’s hemodynamics remained stable for about 3 h into surgery. At 19:30, patient’s blood pressure dropped from 130/75 mmHg to 88/51 mmHg. Phenylephrine 50 µg was administered intravenously to treat the hypotension, and dexmedetomidine infusion was also stopped. The anesthesiologist discussed the situation with the thoracic surgeons. They checked the surgical field and found no obvious bleeding. Using intermittent boluses of phenylephrine and/or ephedrine, blood pressure was maintained within 100–130/60–80 mmHg. The surgery was completed at 21:00. Intraoperative fluid infusion consisted of lactate Ringer’s solution 1,000 mL and gelatin solution 500 mL. Intraoperative blood loss was approximately 300 mL. Intraoperative urine output was 400 mL.
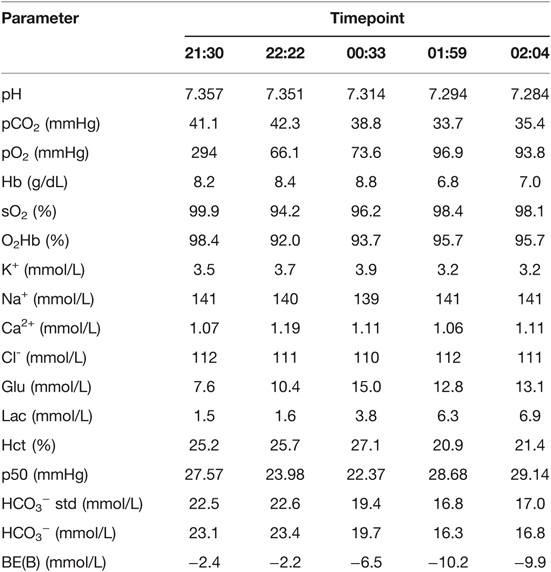
At the end of the surgery, the arterial blood gas and electrolyte analyses showed that pH = 7.357, pCO2 = 41.1 mmHg, pO2 = 294 mmHg, Hb = 8.2 g/dL, Ca2+ = 1.07 mmol/L, and lactate = 1.5 mmol/L (Table 1). At 21:30, the double-lumen endobronchial tube was removed after the patient fully awaked. Supplemental oxygen at 2 L/min was delivered via a nasal catheter. Shortly thereafter, her blood pressure dropped from 120/70 mmHg to 85/46 mmHg, and heart rate was 80 beats/min. 10% calcium gluconate 10 mL was administered intravenously. At 22:22, the Hb was 8.4 g/dL (Table 1). Blood pressure still fluctuated within 70–100/50–65 mmHg. The surgeons rechecked the drainage and confirmed no bleeding in the chest. At this moment, the patient complained of chest tightness. Myocardial ischemia and cardiogenic shock were suspected. The electrocardiogram showed no abnormality in the ST segment, and high-sensitivity troponin T results did not support myocardial injury. Gelatin anaphylaxis was also considered in the differential diagnosis, and methylprednisolone 40 mg and intermittent low-dose epinephrine 10–20 µg were given intravenously. Bedside chest X-ray ruled out pneumothorax, intrathoracic bleeding, or other intrathoracic abnormalities.
Because of sustained hemodynamic instability, a central venous catheter was placed in the internal jugular vein under ultrasonography, for central venous pressure (CVP) monitoring, fluid resuscitation, and infusion of vasopressors (norepinephrine and dopamine). In addition, cardiac index (CI) and stroke volume variation (SVV) were monitored using the FloTrac/Vigileo system. At 23:55, the monitor showed that CVP = 4 mmHg, CI = 2.6 L/min/m2, and SVV = 19%. Based on these, hypovolemia was considered. Lactate Ringer’s solution 500 mL, hydroxyethyl starch 500 mL, erythrocytes 3 units, and fresh frozen plasma 500 mL were given. At 00:33, Hb was 8.8 g/dL, and lactate was 3.8 mmol/L (Table 1). Blood pressure was maintained within 100–120/60–80 mmHg, and heart rate was 80–100 beats/min, with a small-dose epinephrine infusion (0.03–0.05 µg/kg/min).
At 01:59, blood analysis showed that pH decreased to 7.294, Hb dropped to 6.8 g/dL, and lactate increased to 6.3 mmol/L (Table 1). These results were confirmed by an additional test at 02:04. The patient reported upper abdominal pain, and her abdomen was more distended than before. The bedside abdominal ultrasonography showed free intraperitoneal fluids (images not captured). Abdominal computed tomography (CT) confirmed splenic injury and large amount of fluid in the abdominal cavity (Figure 1A). Emergent laparotomy was performed, and splenic rupture was diagnosed. The patient underwent splenectomy, with two lacerations on the diaphragmatic surface of the spleen (Figure 1B). The hemodynamic changes and treatment for hypotension of this patient were summarized in Table 2. After surgery, the patient was admitted to the thoracic intensive care unit. The patient did well postoperatively. She was transferred to the general ward at postoperative day 1 and discharged from the hospital on postoperative day 5.
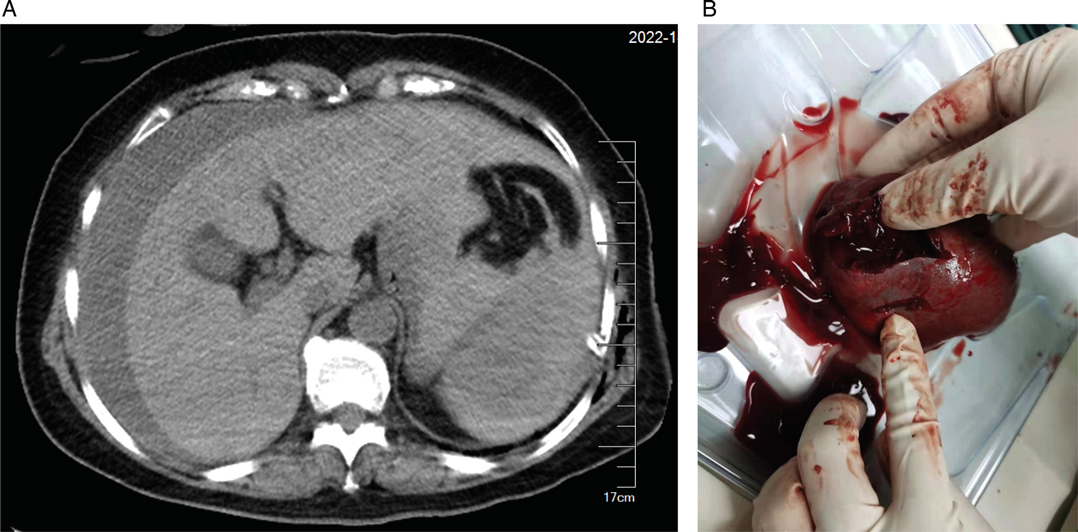
Figure 1. Images of abdominal computed tomography and ruptured spleen. (A) Abdominal computed tomography showing splenic hematoma and large amount of free intraperitoneal fluid. (B) Two lacerations of the ruptured spleen.
Discussion
Splenic rupture is an uncommon and serious complication of VATS procedures. Only four similar cases were previously reported in the literature (2–5). Among them, one patient was managed with angiographic arterial embolization of the spleen, and the other three patients received splenectomy. Our patient also underwent splenectomy. In all reported cases including our patient, there was no evidence of diaphragmatic laceration during the surgical procedures, and radiographic examinations also showed that the diaphragm was intact. The injury to the spleen may be caused by transdiaphragmatic blunt trauma, which was possibly associated with the positioning of thoracoscopic ports and surgical manipulations. In the absence of diaphragmatic injury during surgery, early diagnosis of splenic rupture is usually difficult. In our case, after excluding hypovolemic shock and surgical bleeding, the cause of hemodynamic instability was considered as cardiogenic shock or anaphylactic shock, and eventually back to hypovolemic shock. We continuously monitored the patient’s hemodynamic fluctuations and maintained her blood pressure with vasopressors and volume therapy; however, to some extent, the diagnosis of splenic rupture of this case was delayed.
From this case, we can learn that, in the situation of unexplained hemodynamic instability during the VATS procedures, the possibility of splenic injury and rupture should be considered. For patients with clinical features of hemodynamic instability and abdominal pain after VATS, abdominal ultrasound and/or CT examinations should be carried out as soon as possible for prompt diagnosis and treatment.
Data Availability Statement
The original contributions presented in the study are included in the article/Supplementary Material, further inquiries can be directed to the corresponding authors.
Ethics Statement
Ethical review and approval was not required for the study on human participants in accordance with the local legislation and institutional requirements. The patients/participants provided their written informed consent to participate in this study. Written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article.
Author Contributions
All authors made substantial contributions to the design of this study, data acquisition and interpretation, drafting the manuscript, or revising the manuscript critically. All authors agreed to be accountable for all aspects of the work and gave their final approval of this version to be published. All authors contributed to the article and approved the submitted version.
Funding
Funds from our institution for open access publication fees.
Acknowledgments
We would like to thank the thoracic surgeons and our department nurses for their support in this case report.
Supplementary Material
The Supplementary Material for this article can be found online at: http://journal.frontiersin.org/article/10.3389/fsurg.2022.900396/full#supplementary-material.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher's Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Boffa DJ, Dhamija A, Kosinski AS, Kim AW, Detterbeck FC, Mitchell JD, et al. Fewer complications result from a video-assisted approach to anatomic resection of clinical stage I lung cancer. J Thorac Cardiovasc Surg. (2014) 148(2):637–43. doi: 10.1016/j.jtcvs.2013.12.045
2. Flores RM, Ihekweazu U, Dycoco J, Rizk NP, Rusch VW, Bains MS, et al. Video-assisted thoracoscopic surgery (VATS) lobectomy: catastrophic intraoperative complications. J Thorac Cardiovasc Surg. (2011) 142(6):1412–7. doi: 10.1016/j.jtcvs.2011.09.028
3. Forti Parri SN, Guiducci GM, Domanico A, Tugnoli G. Splenic rupture after videothoracoscopic procedure: an unusual complication conservatively managed. J Thorac Cardiovasc Surg. (2014) 148(5):e236–7. doi: 10.1016/j.jtcvs.2014.08.004
4. Zhang Z. A care report of splenic rupture with subcapsular hematoma after thoracoscopic surgery. Chin J Endosc. (2001) 5(7):27. [Article in Chinese]. doi: 10.3969/j.issn.1007-1989.2001.05.064
Keywords: video-assisted thoracoscopic surgical, splenic rupture, hemodynamic instability, anesthesia management, urgent splenectomy
Citation: Liu C, Wu T, Ding Y, Lin J, Zhou S, Liu H, Ji F and Peng K (2022) Case Report: Hemodynamic Instability Caused by Splenic Rupture During Video-Assisted Thoracoscopic Lobectomy. Front. Surg. 9:900396. doi: 10.3389/fsurg.2022.900396
Received: 20 March 2022; Accepted: 7 April 2022;
Published: 22 April 2022.
Edited by:
Nuria Maria Novoa, University of Salamanca Health Care Complex, SpainReviewed by:
Yener Aydin, Faculty of Medicine, Atatürk University, TurkeyAli Bilal Ulas, Faculty of Medicine, Atatürk University, Turkey
Copyright © 2022 Liu, Wu, Ding, Lin, Zhou, Liu, Ji and Peng. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Ke Peng pengke0422@163.com
†ORCID: Ke Peng orcid.org/0000-0003-2879-1759
Speciality section: This article was submitted to Thoracic Surgery, a section of the journal Frontiers in Surgery
 Chun-tong Liu1,2
Chun-tong Liu1,2 Hong Liu
Hong Liu Fu-hai Ji
Fu-hai Ji Ke Peng
Ke Peng