- 1Food Security and Safety Focus Area, Faculty of Natural and Agricultural Sciences, North-West University, Mmabatho, South Africa
- 2Department of Biological Sciences, Kings University, Ode-Omu, Nigeria
- 3Environmental Pollution Science and Technology, (ENPOST), Ido-Ijesha, Ilesha, Nigeria
The growing rate of urbanization and industrialization has led to an increase in several types of pollution caused by the release of toxic chemicals to the environment. This is usually perpetuated by the manufacturing industry (e.g. detergent and dye), agricultural sectors (e.g. fertilizers and pesticides), mining industry (e.g. cyanide and sulphuric acid) and construction companies (e.g. cement and metals). These pollutants have adverse effects on the health of plants, animals, and humans. They also lead to the destruction of the microbial population in both aquatic and the terrestrial regions, and hence, have necessitated the need for remediation. Although different remediation methods, such as the physical and chemical methods, have been adopted for years, however, the drawbacks and challenges associated with them have promoted the use of an alternative which is bioremediation. Bioremediation involves using biological agents such as plants and microbes to remove or lessen the effects of environmental pollutants. Of the two, microbes are more utilized primarily because of their rapid growth and ability to be easily manipulated, thus enhancing their function as agents of bioremediation. Different groups of bacteria, fungi and algae have been employed to clean up various environmental pollutants. This review discusses the types, mechanisms, and factors affecting microbial bioremediation. It also recommends possible steps that could be taken to promote the use of microbes as bioremediation agents.
1 Introduction
The rise of urbanization and industrialization, has left the environment exposed to numerous pollutants which are toxic to living things. Pollutants arising from different industrial processes are major sources of pollution to the soil and aquatic environment. Different types and quantities of heavy metals are released during the industrial production process and as effluents after further industrial production. For instance, the wastewater from dye-producing companies are associated with antimony, chromium and mercury (Methneni et al., 2021). The application of fertilizers, pesticides and herbicides in the agricultural sector generates pollutants that include aluminium, copper, zinc, nickel, lead and arsenic to the environment (Ayilara et al., 2020; Prabagar et al., 2021). Similarly, untreated pollutants from wastewaters of the agri-food industries disposed into river canals and other waterbodies have harmful effects on the environment (Siric et al., 2022a; AL-Huqail et al., 2022). Crude oil also serves as a major environmental pollutant particularly through pipeline vandalization, transportation leakage, and/or accidental spillage (Ogunlaja et al., 2019). During mining, some chemicals such as lead, arsenic, cadmium, and copper which are toxic to the immediate environment are released (Liu et al., 2020). Some other environmentally toxic chemicals including but not limited to cyanide and sulphuric acid are used during the mining process. (Ayangbenro et al., 2018; Orlovic-Leko et al., 2022). Equally, other industrial wastes such as those produced in cement-making industries release zinc, copper and cadmium and can be found in the top soils (Jafari et al., 2019). Chromium and lead from pharmaceutical effluents (Kumari and Tripathi, 2020), plastics containing lead, manganese, iron, copper, chromium, silver, cadmium, antimony and mercury all pollute water (Zhou et al., 2019). In addition, copper, arsenic, mercury, chromium, lead, nickel, cadmium and zinc from the coal industry serve as environmental pollutant (Sun et al., 2019). These heavy metals are very toxic to aquatic and terrestrial habitats and their inhabitants. In humans, mercury, cadmium and lead alters the central nervous system, especially in infants, while lead results in liver and kidney dysfunction, cardiovascular diseases, malfunctioning of the reproductive and immune system (Zwolak et al., 2019; Fashola et al., 2020a; Fashola et al., 2020b; Ayangbenro and Babalola, 2020). Cadmium causes cancers, skeletal disorders, neurotoxic and nefrotoxic complexities, and dysfunction of the reproductive system (Zwolak et al., 2019; Fashola et al., 2020a; Fashola et al., 2020b; Ayangbenro and Babalola, 2020). Wastes containing heavy metals are often improperly disposed into soil and water environments. When disposed into water bodies, they can lead to the death of fishes, and other aquatic inhabitants, otherwise, they are biomagnified and cause chronic diseases in humans and animals. Therefore, there is need for the remediation of these pollutants using physical, chemical, or biological methods. The physical and chemical methods have been used for years but they come with their drawbacks which include the need for an expert and special equipment for the chemical bioremediation procedure while the physical bioremediation procedure is expensive (Mahmood et al., 2021). This has called for the need for a better alternative which is the biological remediation (Bioremediation). Bioremediation is a most efficient, eco-friendly and cost effective technology for the transformation of contaminants (Sonune, 2021). Biological remediation can be carried out using both plants and microorganism, nonetheless, plants take a longer time to grow and cannot be easily manipulated like the microbes which makes the microbes more preferable (Hussain et al., 2022). In addition, microbes mitigates heavy metals and improve soil fertility and plant development (Chaudhary et al., 2023b). Hence, this review discusses the types, mechanism, challenges as well as the factors affecting microbial bioremediation, with recommendation on how to enhance the use of microbes in aquatic and terrestrial bioremediation.
2 Different pollutants and their toxicity on living things
Exposure of humans to air pollutants can cause developmental disorders, respiratory problems, cancers, cardiovascular diseases, and other health issues (Table 1). For instance, it has been reported that exposure to particulate matter in the air was associated with an increased risk of premature death in humans (Pope et al., 2019). Nitrogen oxides produced by combustion processes, are significant air pollutants. They irritate the respiratory system, cause cough, shortness of breath, and exacerbate asthma (Zhao et al., 2020). Equally, Sulfur dioxide, produced by burning fossil fuels, can cause respiratory and cardiovascular diseases, including bronchoconstriction, shortness of breath, and coughing. A recent study found that exposure to sulfur dioxide was associated with increased mortality from respiratory diseases in China (Luo et al., 2015). Volatile organic compounds (VOCs), emitted by various sources, including paints, cleaning products, and vehicle emissions, can cause eye, nose, and throat irritation, headaches, nausea, and dizziness. Some VOCs (such as benzene) are also carcinogenic, and are associated with an increased risk of leukemia (Bala et al., 2021). Water pollutants which include pesticides, heavy metals, and organic compounds are sometimes ingested by humans either directly or indirectly (through the consumption of aquatic animals). These pollutants can cause various health problems, including cancer, neurological disorders, and reproductive issues. It has been reported that exposure to heavy metals result in a higher risk of hypertension and kidney damage in humans (Wu et al., 2018; Rai et al., 2019).
Similarly, different animal diseases are caused by pollutants. Exposure to particulate matter (PM) can cause inflammation and damage to the respiratory system of animals, leading to respiratory diseases such as chronic obstructive pulmonary disease (COPD) and asthma (Manisalidis et al., 2020). When animals consume water contaminated with heavy metals, pesticides, and pharmaceuticals, it leads to reproductive disorders, liver damage, and cancer (Hitt et al., 2023). Nitrogen dioxide when present in the environment, reduces the growth of plants and the yield of crops while sulfur dioxide causes acid rain and acidification (Manisalidis et al., 2020). An impairment in the photosynthetic rhythm and metabolism is observed in plants exposed to ozone (Zuhara and Isaifan, 2018). In the aquatic environment, eutrophication occurs when there is a high concentration of nitrogen availability. This leads to algal bloom and cause death and disequilibration in the diversity of fish (Zuhara and Isaifan, 2018).
2.1 Types of remediation
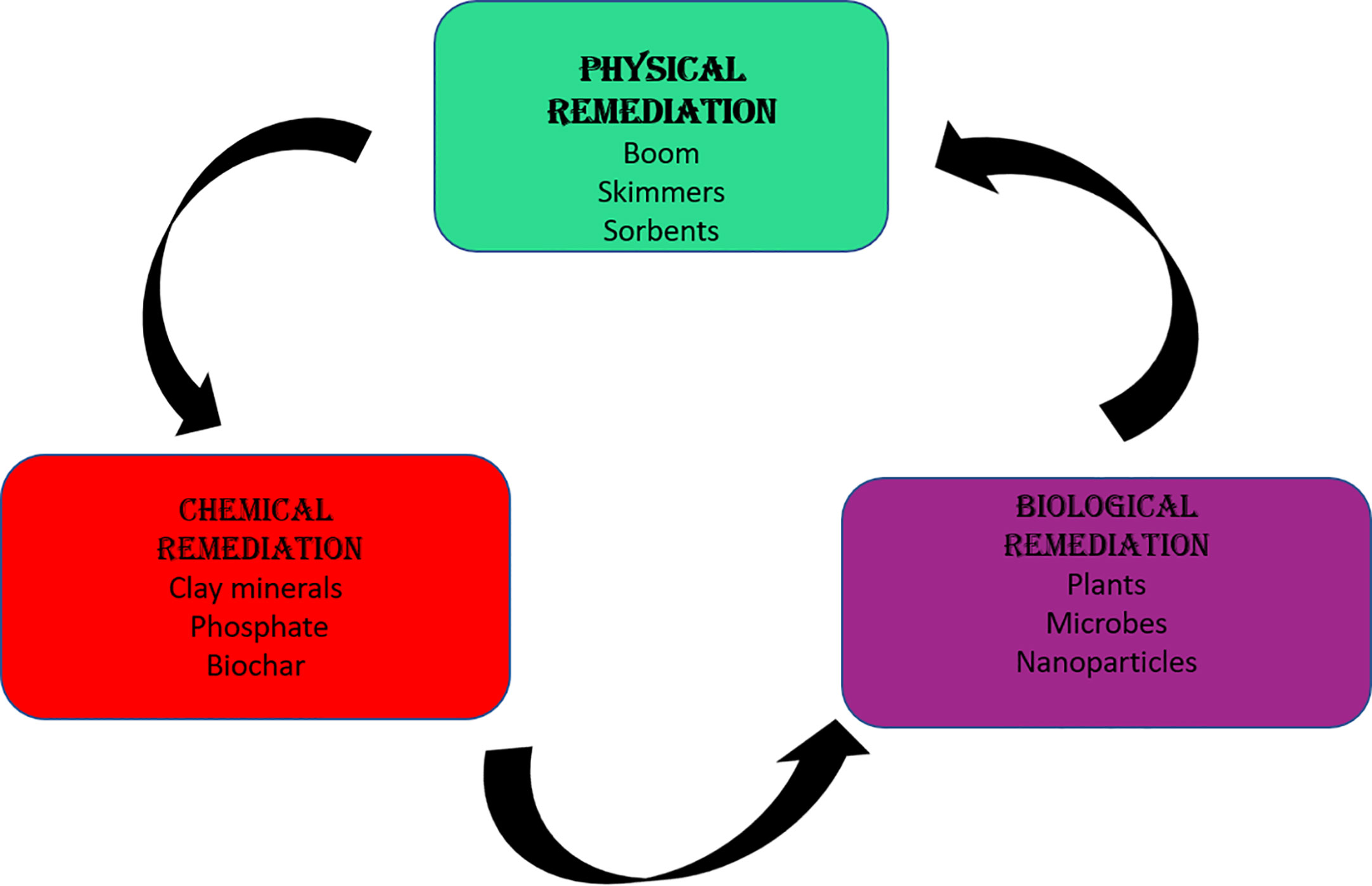
There are different types of remediation, namely the physical, chemical and biological techniques. The physical remediation involves the use of skimmers, sorbent materials and booms. Boom is a physical barrier made of materials that absorbs oil pollutants and prevents it from spreading before a further remediation procedure is carried out (Vocciante et al., 2019) (Figure 1). Skimmers and sorbents are methods that are further used to absorb and adsorb pollutants after booms (Kumari et al., 2019). The major challenge associated with the use of bloom remediation technique is that it is dependent on the buoyancy and roll response. When the boom is buoyant, it floats and remains longer on the water surface. The roll response refers to the torque required to rotate the bloom from its vertical position. That is, an increased roll response results in a higher remediation process (Dhaka and Chattopadhyay, 2021).
Chemical remediation is the process of adding chemicals such as clay minerals, phosphate, biochar, aluminum salts, silicocalcium materials, and sulfide to stabilize and remove heavy metals from the environment. The mechanism behind the use of these chemicals include adsorption, reduction, oxidation, complexation, precipitation and ion exchange (Xu et al., 2022). Chemical remediation technique is an easy, simple, and rapid technique; however, the chemical used can also serve as a source of environmental pollution (Xu et al., 2022) (Figure 1).
Bioremediation is another method of pollution treatment, it is a sustainable, affordable and safe remediation technique (Kumar A. et al., 2021; Kumar G. et al., 2021; Patel A. K. et al., 2022). The technology involves the use of organics such as plants and microbes. The viability of this method depends on the nature, location and level of pollution (Patel A. K. et al., 2022). Microbes on the other hand have proved to be efficient in the remediation of environmental pollutants. They are preferred to plants in remediation, this is due to their ease of growth, rapid growth period and easy manipulation. It is therefore necessary to improve the use of microbes as agent of bioremediation to promote a sustainable environment.
3 Different microbes used as bioremediation agents
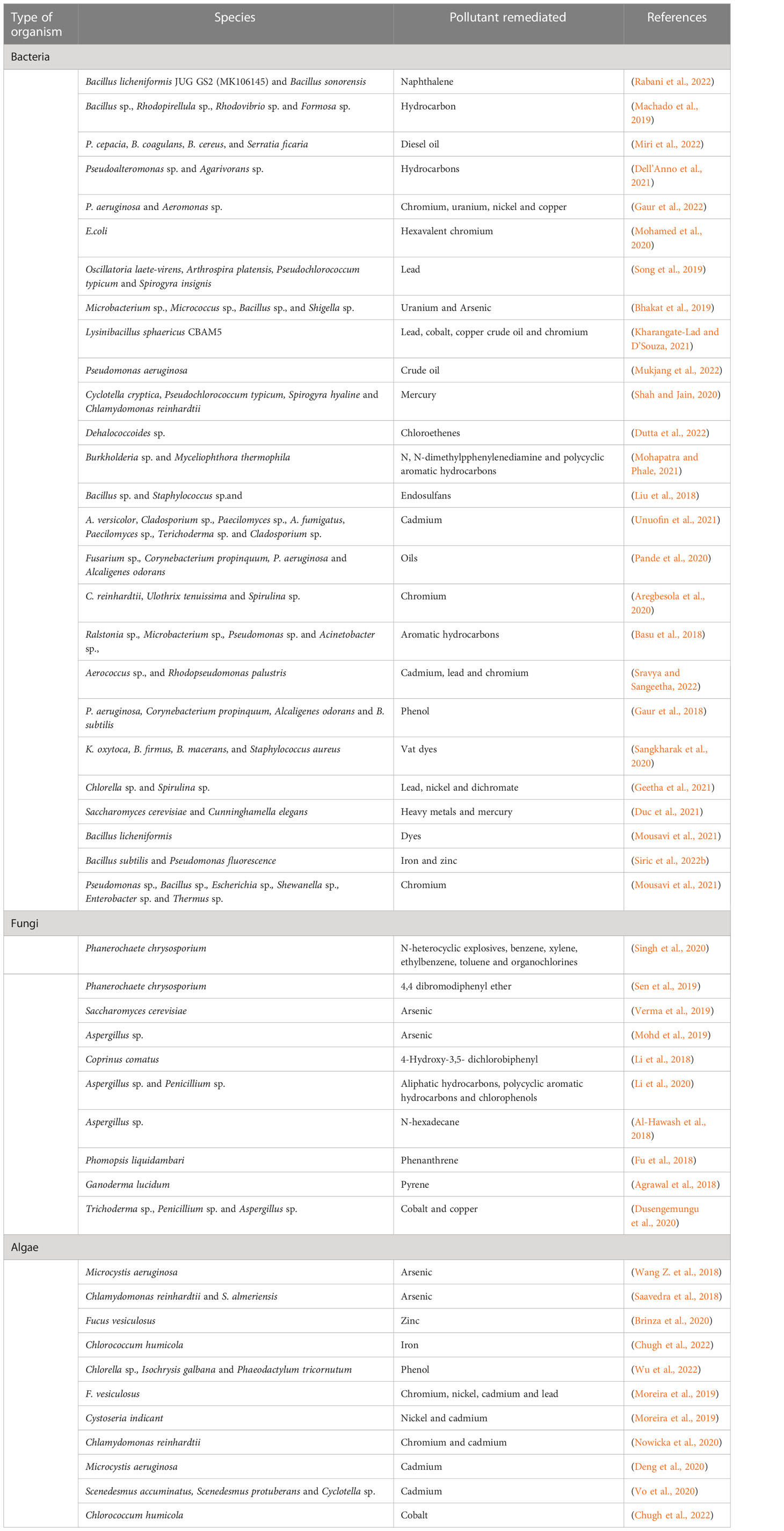
Microorganisms can convert toxic elements into water, carbon dioxide, and other less toxic compounds, which are further degraded by other microbes in a process referred to as mineralization (Mahmoud, 2021; Kumar G. et al., 2022). Bioremediation can be carried out using bacteria, fungi, algae, etc. (Table 2). Microbes are ubiquitous in nature, and they utilize a wide range of substrates as carbon source; hence, they are found in unusual environments where they can absorb a wide range of pollutants (Kour et al., 2022). Also, their ability to survive in odd environments promote their efficiency. For example the acidophiles survive in acidic environments, the psychrophiles thrive in cold climates and the halophiles survive in saline region (Perera and Hemamali, 2022).
4 Mechanisms of microbial bioremediation
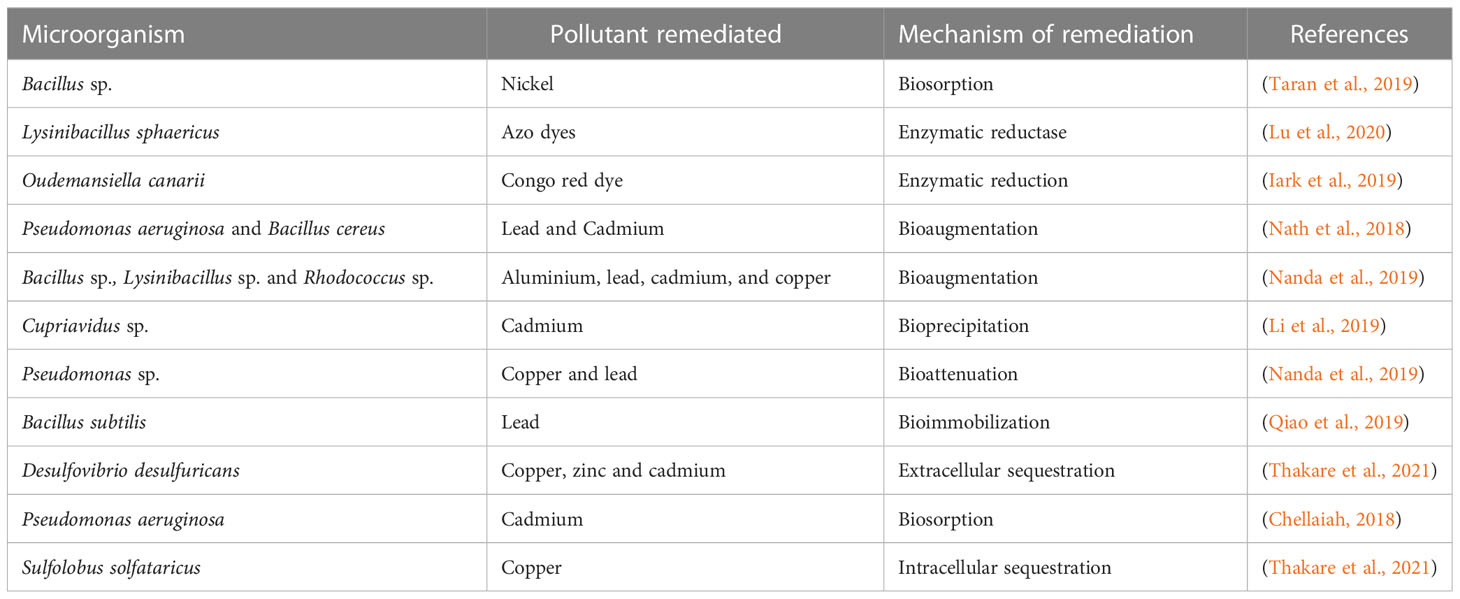
Microbes can remove pollutants from the environment using different mechanisms. These mechanisms can be placed into two broad categories namely immobilization and mobilization (Ndeddy Aka and Babalola, 2016; Verma and Kuila, 2019). Mobilization process involves, enzymatic oxidation, bioleaching, biostimulation, bioaugmentation and enzymatic reduction procedure. On the other hand, immobilization includes bioaccumulation, complexation, biosorption, and precipitation (solidification) (Tak et al., 2012; Ayangbenro et al., 2019). During mineralization, microbes help transform pollutants into end products such as carbon dioxide and water or other intermediate metabolic substances. Similarly, immobilization is the conversion of compounds into a form where it will be unavailable in the environment. For instance, the conversion of nitrate nitrogen into organic nitrogen (Pratush et al., 2018). The method is usually utilized for the bioremediation of heavy metals, especially in highly contaminated environments.
Immobilization can be carried out using the in-situ and the ex-situ methods (Pratush et al., 2018). The ex-situ process involves the removal of polluted soils from the site of pollution to another location where it would undergo a microbial process to immobilize the metal ions responsible for the contamination (Ayangbenro and Babalola, 2017). On the other hand, in the in-situ procedure, the pollution is treated on site (Cao et al., 2020). Microbes such as E. asburiae and B. cereus have been reported to be involved in immobilization of heavy metals which pollute the environment (Fashola et al., 2020a). During microbial bioremediation, microbes protect themselves from toxic compounds by forming hydrophobic or solvent efflux pump that protects the outer membrane of the cell (Verma and Kuila, 2019).
4.1 Enzymatic oxidation
Enzymatic oxidation is the process of oxidizing pollutant compounds from a higher oxidation state to a lower one, during which heavy metals lose an electron and become less toxic. This process utilizes an enzyme (oxidoreductase) released by the microbes involved. This method is highly effective in the remediation of dyes, phenols, and other pollutants which are not easily degraded by bacteria (Unuofin et al., 2019). The oxidative enzymes form radicals which can be broken down into different fractions, eventually forming compounds with high molecular weight (Unuofin et al., 2019). An example of an oxidoreductase enzyme is laccase, which catalyzes the oxidation of aromatic amines (Gangola et al., 2018). Other examples are phenols and polyphenols, which cause the reduction of molecular oxygen to water (Kushwaha et al., 2018; Sahay, 2021). Laccase production has been reported in Pycnoporus sp. and Leptosphaerulina sp. where it was outlined to degrade heavy metals (Copete-Pertuz et al., 2018; Tian et al., 2020).
4.2 Enzymatic reduction
This process is the opposite of enzymatic oxidation, here, the pollutants are converted to a reduced oxidized state where they become insoluble. Obligate and facultative anaerobes carry out the process; this method is effective in the bioremediation of compounds such as polychlorinated dibenzo-p-dioxins and dibenzofurans (Zacharia, 2019). Equally, chrome reductase catalyzes the reduction of hexavalent chromium to trivalent chromium, and azoreductase reduces the azo compounds by cleaving to azo bonds (Saxena et al., 2020). Much more research is needed to unravel other organisms which are capable of bioremediating pollutants in the environment.
4.3 Bioaugmentation
Microorganisms are specially added to polluted sites to feed on toxic pollutants in a process referred to as bioaugmentation. It is a very effective, rapid and cost-effective method of bioremediation (Mahmoud, 2021). External microbes are added to polluted sites to augment the resident microbes. In other cases, it could also involve the isolation and genetic modification of microbes from the site of pollution before returning them to the same site for remediation. Genetic manipulation of resident microbes of polluted sites is carried out because the organisms may naturally not be capable of degrading the pollutant present at a site, and hence are modified to enhance their ability. In some other cases, non-resident microbes are added to polluted areas to promote the degradation of pollutants. The effectiveness of these new strains depends on some factors, which include the ability to compete with the resident microbes and the ability to adapt to the new environment (Fashola et al., 2016; Ayangbenro and Babalola, 2017; Goswami et al., 2018; Babalola et al., 2019). Burkholderia sp. FDS-1 which was added to a polluted site, has been reported to degrade nitrophenolic compound present in pesticides polluted soil to a less toxic form at a slightly acidic pH and a temperature of about 30° C (Goswami et al., 2018; Ojuederie et al., 2021) (Table 3).
4.4 Biostimulation
Biostimulation is the addition of nutrients (such as nitrogen, potassium, phosphorus), metabolites, electron donors, enzymes, electron acceptors, biosurfactants, etc., which are limiting to the soil to enhance the activity of the resident microbes and increase the remediation process (Ojuederie and Babalola, 2017; Ayangbenro and Babalola, 2018). It is an affordable, environmentally friendly and efficient process (Goswami et al., 2018). Compared to the bioaugmentation method, the biostimulation method is preferable because indigenous microbes are more competitive than the introduced ones (Sayed et al., 2021), and this method helps to maintain the natural microbial diversity balance of the environment. Nivetha et al. (2022) reported the effectiveness of Bacillus sp., Rhodococcus sp., Staphylococcus sp., Klebsiella sp., Pseudomonas sp., and Citrobacter sp. in bioremediation of heavy metals through the biostimulation technique. Unfortunately, as effective as this method of bioremediation may be, it could lead to some other environmental complications, including eutrophication due to the excess nutrient present in the environment. Also, if the sources of the nutrients are chemicals (synthetic), they can serve as a source of pollution to the environment defeating the initial purpose of bioremediation (Table 3).
4.5 Bioleaching
Bioleaching is the process of utilizing acidophilic microbes to promote the solubilization of heavy metals which are in a solid state from the sediment matrix. The process is particularly useful for iron or sulfur pollutants (Sun et al., 2021; Bhandari et al., 2023). Therefore, iron- or sulfur-oxidizing bacteria are majorly recruited for this process; examples of such organisms are A. thiooxidans, Aspergillus sp., Mucor sp., Penicillium sp., Cladosporium sp. and Rhizopus sp. (Medfu Tarekegn et al., 2020). These microbes create an acid environment and solubilize heavy metals in an immobilized state, into an aqueous solution (Medfu Tarekegn et al., 2020).
4.6 Biosorption
This is the adsorption of heavy metals from pollutants through proton and ion displacement, complexation, chelation and physical interaction with electrostatic forces (Mahmoud, 2021). It involves the removal of contaminants from solutions as a result of the outer cell shield of bacteria, fungi and algae which are bioremediation agents. Generally, metals are linked through the active groups of the compounds which exist at the cells surface layer. This results in the transfer of ion between metal cations and the negatively charged active group potentials present at the outer part of the microorganism structure. Rhodococcus erythropolis, Streptomyces sp. K11, and Bacillus anthracis have been reported to be capable of bioremediation through the biosorption process (Mathew and Krishnamurthy, 2018; Baltazar et al., 2019; Sedlakova-Kadukova et al., 2019). Oftentimes, heavy metal pollutants (e.g., gold, zinc and copper) have some economic importance and are very useful in industrial processes. Hence, the ability of the compounds to be recovered through a process called desorption (using the solution of weak mineral solution or chelating compounds), which is a reversible step in biosorption makes it a good process (Medfu Tarekegn et al., 2020).
Complexation involves using ligand to form a complex with inorganic metals, which are pollutants in the environment, especially solid wastes (Ayangbenro and Babalola, 2017). Complexation is carried out mainly through different agents, namely the high molecular weight ligands, siderophores and toxic metal-binding compounds as well as the low-molecular weight organic acids (alcohols, tricarboxylic acids and citric acids) (Pratush et al., 2018). Complexation occurs when extracellular polymeric substances, found on the surfaces of microbes interact with heavy metals which pollute the environment (Xie et al., 2020). Xiao et al. (2019) reported the removal of copper (II) oxide and hexavalent chromium from wastewater using biochar in a mechanism which includes complexation. The organisms that have been reported to be involved in complexation include Rhodobacter blasticus (Bai et al., 2019) and B.lichenformisis (Wang et al., 2019).
When microbes are exposed to a polluted environment where there is iron-deficiency, they produce siderophores which are iron chelators. The siderophores have binding groups such as hydroxamate, catecholate and phenolates that form complexes with heavy metals and increase their solubility (Khan et al., 2018). Siderophores are capable of producing reactive oxygen species, which also enhance their function as bioremediation agents for organic contaminants (Albelda-Berenguer et al., 2019). Cyanobacteria have been reported to be effective as bioremediation agents due to the production of siderophores; for example, they are capable of bioremediating complex compounds like polythene and are capable of producing different types of siderophores, which include the anachelin, synechobactin and schizokinen (Arstol and Hohmann-Marriott, 2019; Sarmah and Rout, 2020) (Table 3).
4.7 Bioaccumulation
Bioaccumulation refers to the process where the rate of absorption of a compound is more than the rate at which the compound is lost. This process leads to the (toxic) build-up of compounds in the intracellular portion of the microbes. (Sharma et al., 2022a). Heavy metals move across the membrane of microbes using different mechanisms such as carrier-mediated transport, protein channel and ion pumps (Mir-Tutusaus et al., 2018). Many organisms have been reported to be very active in bioaccumulation of heavy metals. For example, Rhizopus arrhizus, bioremediates mercury, Pseudomonas putida, bioremediates cadmium and Aspergillus niger bioremediates thorium (Sharma et al., 2022a).
4.8 Precipitation
This is the conversion of heavy metals or pollutants into precipitates or crystals, resulting in a reduced toxicity level; this process can occur during the biogeochemical cycling to form deposing of metals (iron and manganese), mineralized manganese and silver as well as microfossils, due to the activity of enzymes and galactosis of secondary metabolites (Sharma et al., 2022a). For instance, sulfate-reducing bacteria are capable of converting organo-phosphate to ortho-phosphate when the pH is alkaline (i.e. above 7) (Pratush et al., 2018). Similarly, Bacillus subtilis and Oceanobacillus indicireducens have also been reported to be associated with the precipitation of heavy metals in the environment (Maity et al., 2019).
5 Factors affecting microbial bioremediation
The ability of microbes to bioremediate heavy metals is determined by different factors, which include the total metal ion concentration, redox potential, chemical forms of the metals, competition among microbes, pH, temperature, soil structure, presence of oxygen, moisture content, nature of the soil and the solubility of the heavy metal in water (Medfu Tarekegn et al., 2020). At acidic pH, free ionic species are formed by heavy metals, leading to the availability of more protons which would saturate the binding site of the metals. The pH of an environment affects the structure of the pollutant and also determines the ability of the microbe to survive in such an environment; the optimum pH that enhances bioremediation falls between 6.5 and 8.5 (Kharangate-Lad and D’Souza, 2021).
Microbes compete for carbon which is a limited resource and serve as an energy source for microbes. Therefore, the inherent ability of the microbes, which compete better to degrade heavy metal pollutant, would affect the biodegradation rate. In addition to carbon, microbes responsible for biodegradation also require nitrogen (N) and phosphorus (P), thus it is important to balance the C:N:P ratio to enhance the rate of biodegradation, in environment when these essential nutrients are limited. They can be added to increase microbial activities (Bala et al., 2022). The type and population of microbes determine the rate and success of a bioremediation process, for instance in the laboratory, a strain of organism might successfully bioremediate a particular heavy metal, which becomes problematic in a field situation where a consortium of microbes would be needed (Patel A. B. et al., 2022). The molecular nature, gene and enzyme induction, metabolite production, growth efficiency and survival rate affect the ability of individual microbes as bioremediation agents (Kebede et al., 2021). In addition, the ionization of the cell wall’s chemical moieties, the configuration of the microbial cell wall and sorption site also affect the rate of microbial biodegradation (Mahmoud, 2021).
The amount of moisture present in an environment affects the solubility of the heavy metals in water, as well as their availability, pH and osmotic pressure (Medfu Tarekegn et al., 2020). At a high moisture content, the microbial biodegradation rate is very low. This might be a result of an anaerobic condition that is created, which prevents the survival of aerobic microbes. Also, at a low moisture content, microbes might not be able to survive; hence an optimum moisture content is required for a successful microbial biodegradation process. In the cold regions where only psychrophiles can survive, the rate of microbial degradation of heavy metals is slow. This is because metabolic activities are reduced as the microbial transport channels is freezed by the sub-zero water; the degradation of each compound also occurs at different temperature even though most bioremediation processes are favored by high temperature (Ren et al., 2018; Bala et al., 2022; Sharma et al., 2022c). At an increased temperature, the rate of heavy metal solubility is increased, which consequently increases their rate of availability as well as the rate of microbial biodegradation (Mahmoud, 2021).
Similarly, the chemical structure, bioavailability, concentration, toxicity and stability of the metal or pollutant determines the rate at which microbial biodegradation takes place (Kebede et al., 2021). For instance, heavy metals with a simple chemical structure and low concentration would be easier to be remediated by microbes compared to those with a complex chemical structure and high temperature. Cycloalkane compounds that are highly condensed as well as high molecular weight polymatic hydrocarbons (those containing four rings and above) are more difficult to degrade compared to the lighter polyhydrocarbons (anthracene, naphthalene and phenanthrene) and unbranched alkanes (alkanes with intermediate length of about C10–C25) (Kebede et al., 2021). Hence, in order of ascending degradation, the n-alkanes are more easily degraded compared to the branched alkanes, low molecular weight aromatics, high molecular weight hydrocarbons and the asphaltenes (Imam et al., 2019). Biodegradation is carried out aerobically and anaerobically. The ability of an organism which degrades a particular nutrient to survive in such an environment depends on the nature of the organism (Jacob et al., 2018). For example, oxygenase associated with organisms that are active in aerobic regions is only produced in the presence of oxygen.
Different soil parameters, including the soil region, moisture-holding capacity texture and particle size, affect the rate of microbial biodegradation (Alvarez et al., 2020). There is a higher population and diversity of microbes at the top layer of the soil (0-10cm). This is due to the increased availability of oxygen and organic matter, which is the opposite of what happens in sediment soils (Ndeddy Aka and Babalola, 2017). In soils with fine particles, such as clayey soils, hydrocarbon retention takes place more at the surface, which renders the nutrient of the soil and oxygen unavailable. Therefore the best soil texture that promotes increased microbial biodegradation is the well-drained soil which supports oxygen availability and inhabits more soil microbes (Huang et al., 2019). The presence of salinity has an effect on the hydrocarbonoclastic activity of the halotolerant and halophilic microbes, and it also exposes the soil microbes to stress conditions. The osmotic pressure of microorganisms increases as the saline concentration of an environment increases. This has a direct negative impact on the metabolic activities, of the microbes as well as the transportation system and solubility of the heavy metals (Imron et al., 2020; Kebede et al., 2021).
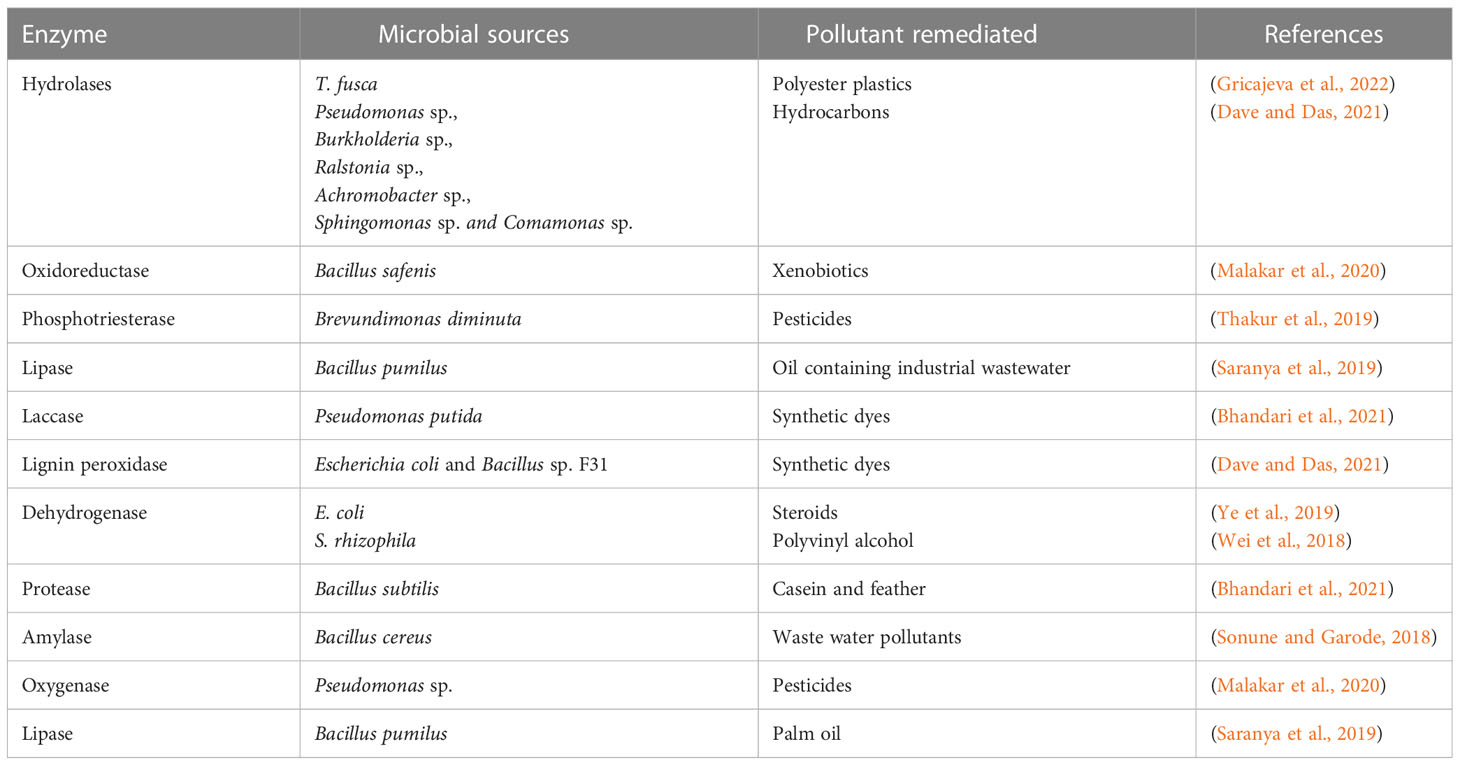
6 Microbial enzymes used in bioremediation
Different microbial enzymes have been reported to be helpful in the removal of pollutants (especially heavy metals) in the environment (Verma and Kuila, 2019; Bhatt et al., 2021a; Chaudhary et al., 2023a) (Table 4). Mechanisms such as elimination, oxidation, ring-opening and reduction are used by enzymes in bioremediation (Bhandari et al., 2021). Different factors which include temperature, contact time, concentration and pH affect the potency of microbial enzymes (Bhandari et al., 2021). Enzyme bioremediation is expensive and time-consuming and therefore cannot be used when there is an urgent need for bioremediation (Narayanan et al., 2023). Equally, the stability and activity of the pollutants, affects the potency of the bioremediation process. It is difficult to determine and discover multiple sources of a particular type of enzyme which might make the procedure unsustainable (Narayanan et al., 2023).
7 Molecular approaches for validating microbial remediation
Molecular mechanisms help to unravel the microbial metabolism, genes, nature, diversity and dynamics of microbes involved in microbial remediation. Diverse molecular mechanisms are utilized in the study of microbes used in bioremediation. Metabolic and protein profiling, sequencing as well as the use of advanced bioinformatics resources are particularly used to unravel the different groups of microbes and the factors affecting them in bioremediation process (Sharma et al., 2022b). On the other hand, conventional and culture-dependent molecular methods are also used in the monitoring of microbial communities during bioremediation. These methods include the use of terminal-restriction fragment (T-RF) length polymorphism, amplified ribosomal DNA restriction analysis, temperature gradient gel electrophoresis, randomly amplified polymorphic DNA analysis, length heterogeneity polymerase chain reaction, amplified fragment length polymorphisms, denaturing gradient gel electrophoresis, length heterogeneity polymerase chain reaction, automated ribosomal intergenic spacer analysis and single strand conformation polymorphism (Bharagava et al., 2019).
Moreover, omics approaches such as transcriptomics, proteomics and metagenomics have greatly contributed in this field. Metagenomics involve the extraction of genomic DNA from all forms of life residing in a sample. Thereafter, the DNA will be fragmented, cloned, transformed and screened in the metagenome library (Bharagava et al., 2019). The approaches to metagenomics include metabolomics, metatranscriptomics, fluxomics and metabolomics. Metatranscriptomics involve the use of metagenomic mRNA which unravel the function and expression of microbes present in a sample (Mukherjee and Reddy, 2020). Metaproteomics involved the assessment of all the protein samples that comes from environmental samples (Bargiela et al., 2015). Metabolomics is the identification and quantification of all the metabolites released into an environment (Liu et al., 2022). Fluxomics refers to the different approaches used to study the rate of metabolic activities in a biological sample (Kumar V. et al., 2022). More recently, the use of Next-Generation sequencing which is viewed as the most powerful technology for gene sequencing has become more popular (Eisenhofer et al., 2019).
8 Other bioremediation metabolites produced by microbes
Microbes produce metabolites such as organic acids, biosurfactants and polymeric substances which are also used in bioremediation. Organic acids improve the bioavailability, mobility and solubility of metals; examples of organic acids include citric acids, malate and acetic acids (Saha et al., 2021). Polymeric substances are beneficial in bioremediation by enhancing the phytostabilization of metals (through mobility), examples of polymeric substances include polyesters, polysaccharides and polyphosphates. Equally, biosurfactants which include viscosin, polymixin, glycoprotein and gramicidin help to solubilise, mobilise and increase the bioavailability of hydrophobic substrates (Ojuederie and Babalola, 2017; Saha et al., 2021).
9 Recent advancements in microbial bioremediate
Lately, many improvements have been observed with the use of microbes as agents of bioremediation. Microbial glycoconjugates help to reduce the surface tension, increase the bioavailability, and create a solvent interface of organic pollutants. This helps to enhance the removal of the pollutants in the environment (Bhatt et al., 2021b). Atakpa et al. (2022) reported the use of microbial glycoconjugates from Scedosporium sp. and Acinetobacter sp. in the biodegradation of petroleum hydocarbons.
Microbial biofilms which consist of polysaccharides, extracellular DNAs and proteins are also lately used in the bioremediation of organic pollutants (Sonawane et al., 2022). They are particularly used in the remediation of recalcitrant pollutants. The technology is presently being made better by improving on the quorum sensing, environmental factors and surface of adhesion (Sonawane et al., 2022). In a research carried out by Andreasen et al. (2018), it was revealed that Exiguobacterium profundum was able to significantly reduce the concentration of arsenic in synthetic wastewater after 48 hours of incubation.
Bioelectrochemical system is another emerging technology which combines the use of biological and electrochemical methods in the control of pollutants (Ambaye et al., 2023). This technology helps to majorly remediate petroleum hydrocarbon pollutants and its efficiency depends mainly on the syntrophic and cooperative interactions between the members of the microbial groups involved (Ambaye et al., 2023). Sharma et al. (2020) stated that Pseudomonas sp., Ralstonia sp., Rhodococcus sp., and Thauera sp. are capable of remediating phenanthrene from petroleum hydrocarbon polluted soils.
Nanotechnology is a thriving method of pollution control globally. Nanomaterials can be sourced from different sources which include the physical and chemical sources (Shanmuganathan et al., 2019). The efficiency of nanoparticles as bioremediation agents is dependent on different factors such as the size, chemical nature, surface coating and shape of the nanoparticles (Tan et al., 2018). Other factors such as the nature of the pollutants, type of media, temperature and the environmental pH affect the potency of nanoparticles in the bioremediation process (Tan et al., 2018). For instance, carbon dots nanoparticles have recently gained attention in the remediation of environmental pollutants owing to their abundance, low toxicity and unique optical properties (Long et al., 2021). It is therefore necessary to carry out further research to unravel technologies and mechanisms to improve the efficiency of the bioremediation process.
10 Future perspectives and conclusions
A number of research endeavours have been carried out on the use of microbial enzymes for bioremediation of waste materials; however, it is very important to improve the process to ensure a safer and more sustainable environment. It is imperative to intensify research to unravel novel microbes that can effectively and rapidly bioremediate different pollutants, especially from industrial sources. Perhaps the novel microbes and their enzymes may have the inherent ability to bioremediate pollutants better than the presently used ones. It is also very important to carry out more studies to innovate rapid detection methods to reveal the progress or help to confirm total biodegradation of pollutants in the environment. Similarly, microbes presently used in bioremediation can be genetically modified to produce more enzymes which will enhance their biodegrading ability. A combination of different microbial consortium other than a single microbial consortium would be a better approach to bioremediation, as this would bring about the presence of different organisms which utilizes different substrate, consequently increasing the rate of microbial biodegradation.
Often, microbes are majorly used to degrade organic substrates, leaving out the persistent inorganic pollutants. Hence, research should be intensified to discover microbes that are capable of degrading inorganic (synthetic) pollutants. In recent years, nuclear wastes generated from the research sectors, hospitals, fuel processing plants and nuclear reactors have remained a global source of pollution. Therefore, the use of microbes and microbial enzymes in the bioremediation of nuclear wastes should be seriously taken into consideration. Occasionally, microbes themselves serve as a source of pollution instead of remediating pollutants. An example of such can be found when microbial biostimulation which results in algal bloom is carried out Consequently, methods to prevent this should be devised to ensure a sustainable environment.
Furthermore, in nature (outside the laboratory), the degradation of different compounds occurs at a different temperature, while the survival of microbes in nature are also environment-specific (temperature). It is therefore essential to carry out more field research to determine the optimum temperature for the degradation of different compounds in nature. In addition, it is also essential to find a balance between the environmental temperature and the temperature for the survival of different microbes in the environment. This would help to prevent bioremediation failure when external microbes are to be recruited or introduced to an environment. As positive and effective microbes might be recruited in the bioremediation of pollutants, it is important to carry out follow-up research to understand their effects on the environment after bioremediation, as some organisms which are introduced to an environment might later constitute pollution to the environment through mutation and other means. Hence, there should be regulatory bodies which would monitor the potential risk associated with microbes in specific environments.
Finally, if enzymes or microbes are directly applied to the soil, they might die or lose their potency before the remediation process begins; therefore, their combination with other agents, such as the nanoparticle could enhance their activity. More awareness is needed on the adoption of microbial degradation, and this will help policymakers as well as the populace to utilize this method. Many people unaware of this procedure might use the available conventional method, which might not be as safe and effective as the microbial biodegradation.
Author contributions
MA and OB conceptualized, wrote, reviewed, and edited the manuscript. All authors contributed to the article and approved the submitted version.
Funding
This research was funded by the National Research Foundation, South Africa, grant number UID: 123634; 132595.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
Agrawal N., Verma P., Shahi S. K. (2018). Degradation of polycyclic aromatic hydrocarbons (phenanthrene and pyrene) by the ligninolytic fungi ganoderma lucidum isolated from the hardwood stump. Bioresour. Bioprocess. 5 (1), 1–9. doi: 10.1186/s40643-018-0197-5
Albelda-Berenguer M., Monachon M., Joseph E. (2019). Siderophores: from natural roles to potential applications. Adv. Appl. Microbiol. 106, 193–225. doi: 10.1016/bs.aambs.2018.12.001
Al-Hawash A. B., Zhang J., Li S., Liu J., Ghalib H. B., Zhang X., et al. (2018). Biodegradation of n-hexadecane by aspergillus sp. RFC-1 and its mechanism. Ecotoxicol. Environ. Saf. 164, 398–408. doi: 10.1016/j.ecoenv.2018.08.049
AL-Huqail A. A., Kumar P., Eid E. M., Adelodun B., Abou Fayssal S., Singh J., et al. (2022). Risk assessment of heavy metals contamination in soil and two rice (Oryza sativa l.) varieties irrigated with paper mill effluent. Agriculture 12 (11), 1864.
Alvarez L. M., Ruberto L. A. M., Gurevich J., Mac Cormack W. P. (2020). Environmental factors affecting reproducibility of bioremediation field assays in Antarctica. Cold Regions Sci. Technol. 169, 102915. doi: 10.1016/j.coldregions.2019.102915
Ambaye T. G., Vaccari M., Franzetti A., Prasad S., Formicola F., Rosatelli A., et al. (2023). Microbial electrochemical bioremediation of petroleum hydrocarbons (PHCs) pollution: recent advances and outlook. Chem. Eng. J. 452, 139372. doi: 10.1016/j.cej.2022.139372
Andreasen R., Li Y., Rehman Y., Ahmed M., Meyer R., Sabri A. (2018). Prospective role of indigenous Exiguobacterium profundum PT2 in arsenic biotransformation and biosorption by planktonic cultures and biofilms. J. Appl. Microbiol. 124 (2), 431–443.
Aragao W. A. B., Teixeira F. B., Fagundes N. C. F., Fernandes R. M., Fernandes L. M. P., da Silva M. C. F., et al. (2018). Hippocampal dysfunction provoked by mercury chloride exposure: evaluation of cognitive impairment, oxidative stress, tissue injury and nature of cell death. Oxid. Med. Cell. Longevity 2018 (1):1–11. doi: 10.1155/2018/7878050
Aregbesola O. A., Kumar A., Mokoena M. P., Olaniran A. O. (2020). Cloning, overexpression, purification, characterization and structural modelling of a metabolically active Fe2+ dependent 2, 6-dichloro-p-hydroquinone 1, 2-dioxygenase (CpsA) from bacillus cereus strain AOA-CPS_1. Int. J. Biol. Macromol. 161, 247–257. doi: 10.1016/j.ijbiomac.2020.05.268
Arstol E., Hohmann-Marriott M. F. (2019). Cyanobacterial siderophores–physiology, structure, biosynthesis, and applications. Mar. Drugs 17 (5), 281.
Atakpa E. O., Zhou H., Jiang L., Ma Y., Liang Y., Li Y., et al. (2022). Improved degradation of petroleum hydrocarbons by co-culture of fungi and biosurfactant-producing bacteria. Chemosphere 290, 133337. doi: 10.1016/j.chemosphere.2021.133337
Ayangbenro A. S., Babalola O. O. (2017). A new strategy for heavy metal polluted environments: a review of microbial biosorbents. Int. J. Environ. Res. Public Health 14 (1), 94. doi: 10.3390/ijerph14010094
Ayangbenro A. S., Babalola O. O. (2018). Metal(loid) bioremediation: strategies employed by microbial polymers. Sustainability 10 (9), 3028. doi: 10.3390/su10093028
Ayangbenro A. S., Babalola O. O. (2020). Genomic analysis of Bacillus cereus NWUAB01 and its heavy metal removal from polluted soil. Sci. Rep. 10 (1), 1–12. doi: 10.1038/s41598-020-75170-x
Ayangbenro A. S., Babalola O. O., Aremu O. S. (2019). Bioflocculant production and heavy metal sorption by metal resistant bacterial isolates from gold mining soil. Chemosphere 231, 113–120. doi: 10.1016/j.chemosphere.2019.05.092
Ayangbenro A. S., Olanrewaju O. S., Babalola O. O. (2018). Sulfate-reducing bacteria as an effective tool for sustainable acid mine bioremediation. Front. Microbiol. 1, 1986. doi: 10.3389/fmicb.2018.01986
Ayilara M. S., Olanrewaju O. S., Babalola O. O., Odeyemi O. (2020). Waste management through composting: challenges and potentials. Sustainability 12 (11), 4456. doi: 10.3390/su12114456
Babalola O. O., Aremu B. R., Ayangbenro A. S. (2019). Draft genome sequence of heavy metal-resistant bacillus cereus NWUAB01. Microbiol. Resour. Announc. 8 (7), e01706–e01718. doi: 10.1128/MRA.01706-18
Bai L., Zhang Q., Wang C., Yao X., Zhang H., Jiang H. (2019). Effects of natural dissolved organic matter on the complexation and biodegradation of 17α-ethinylestradiol in freshwater lakes. Environ. pollut. 246, 782–789. doi: 10.1016/j.envpol.2018.12.098
Bala S., Garg D., Thirumalesh B. V., Sharma M., Sridhar K., Inbaraj B. S., et al. (2022). Recent strategies for bioremediation of emerging pollutants: a review for a green and sustainable environment. Toxics 10 (8), 484. doi: 10.3390/toxics10080484
Bala G.-P., Rajnoveanu R.-M., Tudorache E., Motisan R., Oancea C. (2021). Air pollution exposure–the (in) visible risk factor for respiratory diseases. Environ. Sci. pollut. Res. 28, 19615–19628. doi: 10.1007/s11356-021-13208-x
Balali-Mood M., Naseri K., Tahergorabi Z., Khazdair M. R., Sadeghi M. (2021). Toxic mechanisms of five heavy metals: mercury, lead, chromium, cadmium, and arsenic. Front. Pharmacol. 12, 227. doi: 10.3389/fphar.2021.643972
Baltazar M., Gracioso L. H., Avanzi I. R., Karolski B., Tenório J. A. S., do Nascimento C. A. O., et al. (2019). Copper biosorption by rhodococcus erythropolis isolated from the sossego mine–PA–Brazil. J. Mater. Res. Technol. 8 (1), 475–483. doi: 10.1016/j.jmrt.2018.04.006
Bargiela R., Herbst F. A., Martínez-Martínez M., Seifert J., Rojo D., Cappello S., et al. (2015). Metaproteomics and metabolomics analyses of chronically petroleum-polluted sites reveal the importance of general anaerobic processes uncoupled with degradation. Proteomics 15 (20), 3508–3520. doi: 10.1002/pmic.201400614
Basu S., Rabara R. C., Negi S., Shukla P. (2018). Engineering PGPMOs through gene editing and systems biology: a solution for phytoremediation? Trends Biotechnol. 36 (5), 499–510.
Bhakat K., Chakraborty A., Islam E. (2019). Characterization of arsenic oxidation and uranium bioremediation potential of arsenic resistant bacteria isolated from uranium ore. Environ. Sci. pollut. Res. 26 (13), 12907–12919. doi: 10.1007/s11356-019-04827-6
Bhandari G., Gupta S., Chaudhary P., Chaudhary S., Gangola S. (2023). “Bioleaching: a sustainable resource recovery strategy for urban mining of e-waste,” in Microbial technology for sustainable e-waste management. Eds. Debbarma P., Kumar S., Suyal D. C., Soni R. (Cham: Springer), 157–175.
Bhandari S., Poudel D. K., Marahatha R., Dawadi S., Khadayat K., Phuyal S., et al. (2021). Microbial enzymes used in bioremediation. J. Chem. 2021, 1–17. doi: 10.1155/2021/8849512
Bharagava R. N., Purchase D., Saxena G., Mulla S. I. (2019). “Applications of metagenomics in microbial bioremediation of pollutants: from genomics to environmental cleanup,” in Microbial diversity in the genomic era (Amsterdam: Elsevier), 459–477.
Bhatt P., Gangola S., Joshi C., Chaudhary P., Kumar G., Bhandari G., et al. (2021a). Recent advancements and mechanism of microbial enzymes in sustainable agriculture. Microb. Technol. Sustain. Environ. 1, 247–259. doi: 10.1007/978-981-16-3840-4_15
Bhatt P., Verma A., Gangola S., Bhandari G., Chen S. (2021b). Microbial glycoconjugates in organic pollutant bioremediation: recent advances and applications. Microb. Cell Factories 20 (1), 1–18. doi: 10.1186/s12934-021-01556-9
Brinza L., Geraki K., Cojocaru C., Holdt S. L., Neamtu M. (2020). Baltic Fucus vesiculosus as potential bio-sorbent for zn removal: mechanism insight. Chemosphere 238, 124652. doi: 10.1016/j.chemosphere.2019.124652
Cao H.-L., Liu C., Cai F.-Y., Qiao X.-X., Dichiara A. B., Tian C., et al. (2020). In situ immobilization of ultra-fine Ag NPs onto magnetic Ag@RF@Fe3O4 core-satellite nanocomposites for the rapid catalytic reduction of nitrophenols. Water Res. 179, 115882. doi: 10.1016/j.watres.2020.115882
Chaudhary P., Ahamad L., Chaudhary A., Kumar G., Chen W.-J., Chen S. (2023a). Nanoparticle-mediated bioremediation as a powerful weapon in the removal of environmental pollutants. J. Environ. Chem. Eng. 11, 109591. doi: 10.1016/j.jece.2023.109591
Chaudhary P., Xu M., Ahamad L., Chaudhary A., Kumar G., Adeleke B. S., et al. (2023b). Application of synthetic consortia for improvement of soil fertility, pollution remediation, and agricultural productivity: a review. Agronomy 13 (3), 643. doi: 10.3390/agronomy13030643
Chellaiah E. (2018). Cadmium (heavy metals) bioremediation by Pseudomonas aeruginosa: a minireview. Appl. Water Sci. 8, 1–10. doi: 10.1007/s13201-018-0796-5
Chugh M., Kumar L., Shah M. P., Bharadvaja N. (2022). Algal bioremediation of heavy metals: an insight into removal mechanisms, recovery of by-products, challenges, and future opportunities. Energy Nexus 7, 100129. doi: 10.1016/j.nexus.2022.100129
Copete-Pertuz L. S., Plácido J., Serna-Galvis E. A., Torres-Palma R. A., Mora A. (2018). Elimination of isoxazolyl-penicillins antibiotics in waters by the ligninolytic native Colombian strain leptosphaerulina sp. considerations on biodegradation process and antimicrobial activity removal. Sci. Total Environ. 630, 1195–1204. doi: 10.1016/j.scitotenv.2018.02.244
Dave S., Das J. (2021). Role of microbial enzymes for biodegradation and bioremediation of environmental pollutants: challenges and future prospects. Bioremediat. Environ. Sustainability, 325–346. doi: 10.1016/B978-0-12-820524-2.00013-4
Dell’Anno F., Rastelli E., Tangherlini M., Corinaldesi C., Sansone C., Brunet C., et al. (2021). Highly contaminated marine sediments can host rare bacterial taxa potentially useful for bioremediation. Front. Microbiol. 12, 584850. doi: 10.3389/fmicb.2021.584850
Deng J., Fu D., Hu W., Lu X., Wu Y., Bryan H. (2020). Physiological responses and accumulation ability of microcystis aeruginosa to zinc and cadmium: implications for bioremediation of heavy metal pollution. Bioresour. Technol. 303, 122963. doi: 10.1016/j.biortech.2020.122963
Deng Y., Wang M., Tian T., Lin S., Xu P., Zhou L., et al. (2019). The effect of hexavalent chromium on the incidence and mortality of human cancers: a meta-analysis based on published epidemiological cohort studies. Front. Oncol. 9, 24. doi: 10.3389/fonc.2019.00024
de Souza M. B., de Souza Santos L. R., Borges R. E., Nunes H. F., Vieira T. B., Pacheco S. M., et al. (2020). Current status of ecotoxicological studies of bats in Brazil. Bull. Environ. Contam. Toxicol. 104, 393–399. doi: 10.1007/s00128-020-02794-0
Dhaka A., Chattopadhyay P. (2021). A review on physical remediation techniques for treatment of marine oil spills. J. Environ. Manage. 288, 112428. doi: 10.1016/j.jenvman.2021.112428
Duc H., Hung N., Oanh N. (2021). Anaerobic degradation of endosulfans by a mixed culture of pseudomonas sp. and staphylococcus sp. Appl. Biochem. Microbiol. 57 (3), 327–334. doi: 10.1134/S0003683821030030
Dusengemungu L., Kasali G., Gwanama C., Ouma K. O. (2020). Recent advances in biosorption of copper and cobalt by filamentous fungi. Front. Microbiol. 11, 3285. doi: 10.3389/fmicb.2020.582016
Dutta N., Usman M., Ashraf M. A., Luo G., Zhang S. (2022). Efficacy of emerging technologies in addressing reductive dechlorination for environmental bioremediation: a review. J. Hazard. Mater. Lett. 3, 100065. doi: 10.1016/j.hazl.2022.100065
Eisenhofer R., Minich J. J., Marotz C., Cooper A., Knight R., Weyrich L. S. (2019). Contamination in low microbial biomass microbiome studies: issues and recommendations. Trends Microbiol. 27 (2), 105–117. doi: 10.1016/j.tim.2018.11.003
Fashola M. O., Ngole-Jeme V. M., Babalola O. O. (2016). Heavy metal pollution from gold mines: environmental effects and bacterial strategies for resistance. Int. J. Environ. Res. Public Health 13 (11), 1047. doi: 10.3390/ijerph13111047
Fashola M. O., Ngole-Jeme V. M., Babalola O. O. (2020a). Heavy metal immobilization potential of indigenous bacteria isolated from gold mine tailings. Int. J. Environ. Res. 14 (1), 71–86. doi: 10.1007/s41742-019-00240-6
Fashola M. O., Ngole-Jeme V. M., Babalola O. O. (2020b). Physicochemical properties, heavy metals, and metal-tolerant bacteria profiles of abandoned gold mine tailings in krugersdorp, south Africa. Can. J. Soil Sci. 100 (3), 217–233. doi: 10.1139/cjss-2018-0161
Fay M. J., Alt L. A., Ryba D., Salamah R., Peach R., Papaeliou A., et al. (2018). Cadmium nephrotoxicity is associated with altered microRNA expression in the rat renal cortex. Toxics 6 (1), 16. doi: 10.3390/toxics6010016
Fu W., Xu M., Sun K., Hu L., Cao W., Dai C., et al. (2018). Biodegradation of phenanthrene by endophytic fungus phomopsis liquidambari in vitro and in vivo. Chemosphere 203, 160–169. doi: 10.1016/j.chemosphere.2018.03.164
Gangola S., Sharma A., Bhatt P., Khati P., Chaudhary P. (2018). Presence of esterase and laccase in bacillus subtilis facilitates biodegradation and detoxification of cypermethrin. Sci. Rep. 8 (1), 12755.
Gaur N., Narasimhulu K., PydiSetty Y. (2018). Recent advances in the bio-remediation of persistent organic pollutants and its effect on environment. J. Clean. Prod. 198, 1602–1631. doi: 10.1016/j.jclepro.2018.07.076
Gaur V. K., Tripathi V., Manickam N. (2022). “Bacterial-and fungal-mediated biodegradation of petroleum hydrocarbons in soil,” in Development in wastewater treatment research and processes (Amsterdam: Elsevier), 407–427.
Geetha N., Bhavya G., Abhijith P., Shekhar R., Dayananda K., Jogaiah S. (2021). Insights into nanomycoremediation: secretomics and mycogenic biopolymer nanocomposites for heavy metal detoxification. J. Hazard. Mater. 409, 124541. doi: 10.1016/j.jhazmat.2020.124541
Goswami M., Chakraborty P., Mukherjee K., Mitra G., Bhattacharyya P., Dey S., et al. (2018). Bioaugmentation and biostimulation: a potential strategy for environmental remediation. J. Microbiol. Exp. 6 (5), 223–231.
Gricajeva A., Nadda A. K., Gudiukaite R. (2022). Insights into polyester plastic biodegradation by carboxyl ester hydrolases. J. Chem. Technol. Biotechnol. 97 (2), 359–380. doi: 10.1002/jctb.6745
Hitt L. G., Khalil S., Blanchette A., Finkelstein M. E., Iverson E. N., McClelland S. C., et al. (2023). Lead exposure is correlated with reduced nesting success of an urban songbird. Environ. Res. 227 (1), 115711. doi: 10.1016/j.envres.2023.115711
Huang Y., Pan H., Wang Q., Ge Y., Liu W., Christie P. (2019). Enrichment of the soil microbial community in the bioremediation of a petroleum-contaminated soil amended with rice straw or sawdust. Chemosphere 224, 265–271. doi: 10.1016/j.chemosphere.2019.02.148
Hussain A., Rehman F., Rafeeq H., Waqas M., Asghar A., Afsheen N., et al. (2022). In-situ, ex-situ, and nano-remediation strategies to treat polluted soil, water, and air–a review. Chemosphere 289, 133252. doi: 10.1016/j.chemosphere.2021.133252
Iark D., dos Reis Buzzo A. J., Garcia J. A. A., Côrrea V. G., Helm C. V., Corrêa R. C. G., et al. (2019). Enzymatic degradation and detoxification of azo dye Congo red by a new laccase from Oudemansiella canarii. Bioresour. Technol. 289, 121655. doi: 10.1016/j.biortech.2019.121655
Imam A., Suman S. K., Ghosh D., Kanaujia P. K. (2019). Analytical approaches used in monitoring the bioremediation of hydrocarbons in petroleum-contaminated soil and sludge. TrAC Trends Anal. Chem. 118, 50–64. doi: 10.1016/j.trac.2019.05.023
Imron M. F., Kurniawan S. B., Ismail N. I., Abdullah S. R. S. (2020). Future challenges in diesel biodegradation by bacteria isolates: a review. J. Clean. Prod. 251, 119716. doi: 10.1016/j.jclepro.2019.119716
Jacob J. M., Karthik C., Saratale R. G., Kumar S. S., Prabakar D., Kadirvelu K., et al. (2018). Biological approaches to tackle heavy metal pollution: a survey of literature. J. Environ. Manage. 217, 56–70. doi: 10.1016/j.jenvman.2018.03.077
Jafari A., Ghaderpoori M., Kamarehi B., Abdipour H. (2019). Soil pollution evaluation and health risk assessment of heavy metals around douroud cement factory, Iran. Environ. Earth Sci. 78 (8), 1–9. doi: 10.1007/s12665-019-8220-5
Kebede G., Tafese T., Abda E. M., Kamaraj M., Assefa F. (2021). Factors influencing the bacterial bioremediation of hydrocarbon contaminants in the soil: mechanisms and impacts. J. Chem. 2021, 1–17. doi: 10.1155/2021/9823362
Khan A., Singh P., Srivastava A. (2018). Synthesis, nature and utility of universal iron chelator–siderophore: a review. Microbiol. Res. 212, 103–111. doi: 10.1016/j.micres.2017.10.012
Kharangate-Lad A., D’Souza N. C. (2021). Current approaches in bioremediation of toxic contaminants by application of microbial cells; biosurfactants and bioemulsifiers of microbial origin. Rhizobiont Bioremediat. Hazard. Waste, 217–263. doi: 10.1007/978-981-16-0602-1_11
Kour D., Khan S. S., Kour H., Kaur T., Devi R., Rai P. K., et al. (2022). Microbe-mediated bioremediation: current research and future challenges. J. Appl. Biol. Biotechnol. 10 (2), 6–24. doi: 10.7324/JABB.2022.10s202
Kumar V., Garg V. K., Kumar S., Biswas J. K. (2022). Omics for environmental engineering and microbiology systems (Florida: CRC Press).
Kumar G., Lal S., Maurya S. K., Bhattacherjee A., Chaudhary P., Gangola S., et al. (2021). Exploration of klebsiella pneumoniae M6 for paclobutrazol degradation, plant growth attributes, and biocontrol action under subtropical ecosystem. PloS One 16 (12), e0261338. doi: 10.1371/journal.pone.0261338
Kumar G., Lal S., Soni S. K., Maurya S. K., Shukla P. K., Chaudhary P., et al. (2022). Mechanism and kinetics of chlorpyrifos co-metabolism by using environment restoring microbes isolated from rhizosphere of horticultural crops under subtropics. Front. Microbiol. 13, 2796. doi: 10.3389/fmicb.2022.891870
Kumar A., Sharma A., Chaudhary P., Gangola S. (2021). Chlorpyrifos degradation using binary fungal strains isolated from industrial waste soil. Biologia 76 (10), 3071–3080. doi: 10.1007/s11756-021-00816-8
Kumari A., Kaur R., Kaur R. (2019). A review on fate and remediation techniques of oil spills. Int. J. Res. Pharm. Sci. 10, 111.
Kumari V., Tripathi A. (2020). Remediation of heavy metals in pharmaceutical effluent with the help of bacillus cereus-based green-synthesized silver nanoparticles supported on alumina. Appl. Nanosci. 10 (6), 1709–1719. doi: 10.1007/s13204-020-01351-9
Kushwaha A., Maurya S., Pathak R. K., Agarwal S., Chaurasia P. K., Singh M. (2018). “Laccase from white rot fungi having significant role in food, pharma, and other industries,” in Research advancements in pharmaceutical, nutritional, and industrial enzymology (Pennsylvania: IGI Global), 253–277.
Li Q., Liu J., Gadd G. M. (2020). Fungal bioremediation of soil co-contaminated with petroleum hydrocarbons and toxic metals. Appl. Microbiol. Biotechnol. 104 (21), 8999–9008. doi: 10.1007/s00253-020-10854-y
Li N., Xia Q., Niu M., Ping Q., Xiao H. (2018). Immobilizing laccase on different species wood biochar to remove the chlorinated biphenyl in wastewater. Sci. Rep. 8 (1), 1–9. doi: 10.1038/s41598-018-32013-0
Li F., Zheng Y., Tian J., Ge F., Liu X., Tang Y., et al. (2019). Cupriavidus sp. strain Cd02-mediated pH increase favoring bioprecipitation of Cd2+ in medium and reduction of cadmium bioavailability in paddy soil. Ecotoxicol. Environ. Saf. 184, 109655.
Liu Z., Shao B., Zeng G., Chen M., Li Z., Liu Y., et al. (2018). Effects of rhamnolipids on the removal of 2, 4, 2, 4-tetrabrominated biphenyl ether (BDE-47) by phanerochaete chrysosporium analyzed with a combined approach of experiments and molecular docking. Chemosphere 210, 922–930. doi: 10.1016/j.chemosphere.2018.07.114
Liu X., Shi H., Bai Z., Zhou W., Liu K., Wang M., et al. (2020). Heavy metal concentrations of soils near the large opencast coal mine pits in China. Chemosphere 244, 125360. doi: 10.1016/j.chemosphere.2019.125360
Liu L., Wu Q., Miao X., Fan T., Meng Z., Chen X., et al. (2022). Study on toxicity effects of environmental pollutants based on metabolomics: a review. Chemosphere 286, 131815. doi: 10.1016/j.chemosphere.2021.131815
Long C., Jiang Z., Shangguan J., Qing T., Zhang P., Feng B. (2021). Applications of carbon dots in environmental pollution control: a review. Chem. Eng. J. 406, 126848. doi: 10.1016/j.cej.2020.126848
Lu J., Zhang C., Leong H. Y., Show P. L., Lu F., Lu Z. (2020). Overproduction of lipoxygenase from pseudomonas aeruginosa in Escherichia coli by auto-induction expression and its application in triphenylmethane dyes degradation. J. Biosci. Bioeng. 129 (3), 327–332. doi: 10.1016/j.jbiosc.2019.09.006
Luo H., Yuan D. B., Zhang W. (2015). Association between cadmium exposure and renal cancer risk: a meta-analysis of observational studies. Sci. Rep. 5 (1), 1–8.
Machado L. F., de Assis Leite D. C., da Costa Rachid C. T. C., Paes J. E., Martins E. F., Peixoto R. S., et al. (2019). Tracking mangrove oil bioremediation approaches and bacterial diversity at different depths in an in situ mesocosms system. Front. Microbiol. 10, 2107. doi: 10.3389/fmicb.2019.02107
Mahmood A., Bilal B., Naeem Z., Iram S. (2021). “Physical, chemical, and biological remediation techniques for textile effluents in context with developed and developing countries,” in Rhizobiont in bioremediation of hazardous waste (Singapore: Springer), 409–441.
Mahmoud G. A.-E. (2021). “Microbial scavenging of heavy metals using bioremediation strategies,” in Rhizobiont in bioremediation of hazardous waste. Eds. Vivek K., Ram P., Manoj K. (Singapore: Springer), 265–289.
Maity J. P., Chen G.-S., Huang Y.-H., Sun A.-C., Chen C.-Y. (2019). Ecofriendly heavy metal stabilization: microbial induced mineral precipitation (MIMP) and biomineralization for heavy metals within the contaminated soil by indigenous bacteria. Geomicrobiol. J. 36 (7), 612–623. doi: 10.1080/01490451.2019.1597216
Malakar N., Mitra S., Toppo P., Mathur P. (2020). Understanding the functional attributes of different microbial enzymes in bioremediation. NBU J. Plant Sci. 12 (1), 58–69. doi: 10.55734/NBUJPS.2020.v12i01.005
Manisalidis I., Stavropoulou E., Stavropoulos A., Bezirtzoglou E. (2020). Environmental and health impacts of air pollution: a review. Front. Public Health 1), 14. doi: 10.3389/fpubh.2020.00014
Mathew B. B., Krishnamurthy N. B. (2018). Screening and identification of bacteria isolated from industrial area groundwater to study lead sorption: kinetics and statistical optimization of biosorption parameters. Groundwater Sustain. Dev. 7, 313–327. doi: 10.1016/j.gsd.2018.07.007
Medfu Tarekegn M., Zewdu Salilih F., Ishetu A. I. (2020). Microbes used as a tool for bioremediation of heavy metal from the environment. Cogent Food Agric. 6 (1), 1783174. doi: 10.1080/23311932.2020.1783174
Methneni N., Morales-Gonzalez J. A., Jaziri A., Mansour H. B., Fernandez-Serrano M. (2021). Persistent organic and inorganic pollutants in the effluents from the textile dyeing industries: ecotoxicology appraisal via a battery of biotests. Environ. Res. 196, 110956. doi: 10.1016/j.envres.2021.110956
Miri S., Rasooli A., Brar S. K., Rouissi T., Martel R. (2022). Biodegradation of p-xylene–a comparison of three psychrophilic pseudomonas strains through the lens of gene expression. Environ. Sci. pollut. Res. 29 (15), 21465–21479. doi: 10.1007/s11356-021-17387-5
Mir-Tutusaus J. A., Baccar R., Caminal G., Sarrà M. (2018). Can white-rot fungi be a real wastewater treatment alternative for organic micropollutants removal? a review. Water Res. 138, 137–151. doi: 10.1016/j.watres.2018.02.056
Mohamed M. S., El-Arabi N. I., El-Hussein A., El-Maaty S. A., Abdelhadi A. A. (2020). Reduction of chromium-VI by chromium-resistant escherichia coli FACU: a prospective bacterium for bioremediation. Folia Microbiol. 65 (4), 687–696. doi: 10.1007/s12223-020-00771-y
Mohapatra B., Phale P. S. (2021). Microbial degradation of naphthalene and substituted naphthalenes: metabolic diversity and genomic insight for bioremediation. Front. Bioeng. Biotechnol. 9, 602445. doi: 10.3389/fbioe.2021.602445
Mohd S., Kushwaha A. S., Shukla J., Mandrah K., Shankar J., Arjaria N., et al. (2019). Fungal mediated biotransformation reduces toxicity of arsenic to soil dwelling microorganism and plant. Ecotoxicol. Environ. Saf. 176, 108–118. doi: 10.1016/j.ecoenv.2019.03.053
Moreira V., Lebron Y., Lange L., Santos L. (2019). Simultaneous biosorption of cd (II), Ni (II) and Pb (II) onto a brown macroalgae fucus vesiculosus: mono-and multi-component isotherms, kinetics and thermodynamics. J. Environ. Manage. 251, 109587. doi: 10.1016/j.jenvman.2019.109587
Mousavi S. M., Hashemi S. A., Iman Moezzi S. M., Ravan N., Gholami A., Lai C. W., et al. (2021). Recent advances in enzymes for the bioremediation of pollutants. Biochem. Res. Int. 2021, 1–12.
Mukherjee A., Reddy M. S. (2020). Metatranscriptomics: an approach for retrieving novel eukaryotic genes from polluted and related environments. 3 Biotech. 10 (2), 71. doi: 10.1007/s13205-020-2057-1
Mukjang N., Chitov T., Mhuantong W., Champreda V., Pathom-Aree W., Sattayawat P., et al. (2022). Bacterial communities associated with crude oil bioremediation through composting approaches with indigenous bacterial isolate. Life 12 (11), 1712. doi: 10.3390/life12111712
Nanda M., Kumar V., Sharma D. (2019). Multimetal tolerance mechanisms in bacteria: the resistance strategies acquired by bacteria that can be exploited to ‘clean-up’heavy metal contaminants from water. Aquat. Toxicol. 212, 1–10. doi: 10.1016/j.aquatox.2019.04.011
Narayanan M., Ali S. S., El-Sheekh M. (2023). A comprehensive review on the potential of microbial enzymes in multipollutant bioremediation: MechanAisms, challenges, and future prospects. J. Environ. Manage. 334, 117532. doi: 10.1016/j.jenvman.2023.117532
Nath S., Deb B., Sharma I. (2018). Isolation of toxic metal-tolerant bacteria from soil and examination of their bioaugmentation potentiality by pot studies in cadmium-and lead-contaminated soil. Int. Microbiol. 21, 35–45. doi: 10.1007/s10123-018-0003-4
Ndeddy Aka R. J., Babalola O. O. (2016). Effect of bacterial inoculation of strains of pseudomonas aeruginosa, alcaligenes feacalis and bacillus subtilis on germination, growth and heavy metal (Cd, cr, and Ni) uptake of brassica juncea. Int. J. Phytoremediation 18 (2), 200–209.
Ndeddy Aka R. J., Babalola O. O. (2017). Identification and characterization of cr-, cd-, and Ni-tolerant bacteria isolated from mine tailings. Bioremediat. J. 21 (1), 1–19.
Nivetha N., Srivarshine B., Sowmya B., Rajendiran M., Saravanan P., Rajeshkannan R., et al. (2022). A comprehensive review on bio-stimulation and bio-enhancement towards remediation of heavy metals degeneration. Chemosphere 3, 137099.
Nowicka B., Fesenko T., Walczak J., Kruk J. (2020). The inhibitor-evoked shortage of tocopherol and plastoquinol is compensated by other antioxidant mechanisms in chlamydomonas reinhardtii exposed to toxic concentrations of cadmium and chromium ions. Ecotoxicol. Environ. Saf. 191, 110241. doi: 10.1016/j.ecoenv.2020.110241
Ogunlaja A., Ogunlaja O. O., Okewole D. M., Morenikeji O. A. (2019). Risk assessment and source identification of heavy metal contamination by multivariate and hazard index analyses of a pipeline vandalised area in Lagos state, Nigeria. Sci. Total Environ. 651, 2943–2952. doi: 10.1016/j.scitotenv.2018.09.386
Ojuederie O. B., Babalola O. O. (2017). Microbial and plant-assisted bioremediation of heavy metal polluted environments: a review. Int. J. Environ. Res. Public Health 14 (12), 1504. doi: 10.3390/ijerph14121504
Ojuederie O. B., Chukwuneme F., Olanrewaju O., Ayilara M., Adegboyega T. T., Babalola O. O. (2021). Contribution of microbial inoculants in sustainable maintenance of human health, including test methods and evaluation of safety of microbial pesticide microorganisms. Biopesticides: Botanicals Microorganisms Improving Agric. Hum. Health 2021, 207–240.
Orlovic-Leko P., Farkas B., Galic I. (2022). A short review of environmental and health impacts of gold mining. Reliability: Theory Appl. 17(SI 4 (SI 4 (70), 242–248.
Pande V., Pandey S. C., Sati D., Pande V., Samant M. (2020). Bioremediation: an emerging effective approach towards environment restoration. Environ. Sustainability 3 (1), 91–103. doi: 10.1007/s42398-020-00099-w
Patel A. B., Jain K. R., Manvar T., Desai C., Madamwar D. (2022). Enriched bacterial community efficiently degrade polycyclic aromatic hydrocarbons in soil ecosystem: insights from a mesocosms study. Biochem. Eng. J. 185, 108516. doi: 10.1016/j.bej.2022.108516
Patel A. K., Singhania R. R., Albarico F. P. J. B., Pandey A., Chen C.-W., Dong C.-D. (2022). Organic wastes bioremediation and its changing prospects. Sci. Total Environ. 824, 153889. doi: 10.1016/j.scitotenv.2022.153889
Pavesi T., Moreira J. C. (2020). Mechanisms and individuality in chromium toxicity in humans. J. Appl. Toxicol. 40 (9), 1183–1197. doi: 10.1002/jat.3965
Perera I. C., Hemamali E. H. (2022). Genetically modified organisms for bioremediation: current research and advancements. Bioremediat. Environ. Pollut.: Emerg. Trends Strategies 1, 163–186.
Pope C. A., Lefler J. S., Ezzati M., Higbee J. D., Marshall J. D., Kim S.-Y., et al. (2019). Mortality risk and fine particulate air pollution in a large, representative cohort of US adults. Environ. Health Perspect. 127 (7), 077007.
Prabagar S., Dharmadasa R. M., Lintha A., Thuraisingam S., Prabagar J. (2021). Accumulation of heavy metals in grape fruit, leaves, soil and water: a study of influential factors and evaluating ecological risks in jaffna, Sri Lanka. Environ. Sustainability Indic. 12, 100147. doi: 10.1016/j.indic.2021.100147
Pratush A., Kumar A., Hu Z. (2018). Adverse effect of heavy metals (As, Pb, Hg, and cr) on health and their bioremediation strategies: a review. Int. Microbiol. 21 (3), 97–106. doi: 10.1007/s10123-018-0012-3
Qiao W., Zhang Y., Xia H., Luo Y., Liu S., Wang S., et al. (2019). Bioimmobilization of lead by bacillus subtilis X3 biomass isolated from lead mine soil under promotion of multiple adsorption mechanisms. R. Soc. Open Sci. 6 (2), 181701. doi: 10.1098/rsos.181701
Rabani M. S., Sharma R., Singh R., Gupta M. (2022). Characterization and identification of naphthalene degrading bacteria isolated from petroleum contaminated sites and their possible use in bioremediation. Polycyclic Aromatic Compounds 42 (3), 978–989. doi: 10.1080/10406638.2020.1759663
Rai P. K., Lee S. S., Zhang M., Tsang Y. F., Kim K.-H. (2019). Heavy metals in food crops: health risks, fate, mechanisms, and management. Environ. Int. 125 (1), 365–385. doi: 10.1016/j.envint.2019.01.067
Rehman A. U., Nazir S., Irshad R., Tahir K., ur Rehman K., Islam R. U., et al. (2021). Toxicity of heavy metals in plants and animals and their uptake by magnetic iron oxide nanoparticles. J. Mol. Liquids 321, 114455. doi: 10.1016/j.molliq.2020.114455
Ren X., Zeng G., Tang L., Wang J., Wan J., Wang J., et al. (2018). The potential impact on the biodegradation of organic pollutants from composting technology for soil remediation. Waste Manage. 72, 138–149. doi: 10.1016/j.wasman.2017.11.032
Saavedra R., Muñoz R., Taboada M. E., Vega M., Bolado S. (2018). Comparative uptake study of arsenic, boron, copper, manganese and zinc from water by different green microalgae. Bioresour. Technol. 263, 49–57. doi: 10.1016/j.biortech.2018.04.101
Saha L., Tiwari J., Bauddh K., Ma Y. (2021). Recent developments in microbe–plant-based bioremediation for tackling heavy metal-polluted soils. Front. Microbiol. 12, 731723. doi: 10.3389/fmicb.2021.731723
Sahay R. (2021). Synthetic applications of laccase and its catalytic potentials. Inter J. Adv. Eng. Res. Sci. 8 (6), 112–120. doi: 10.22161/ijaers.86.12
Sangkharak K., Choonut A., Rakkan T., Prasertsan P. (2020). The degradation of phenanthrene, pyrene, and fluoranthene and its conversion into medium-chain-length polyhydroxyalkanoate by novel polycyclic aromatic hydrocarbon-degrading bacteria. Curr. Microbiol. 77 (6), 897–909. doi: 10.1007/s00284-020-01883-x
Saranya P., Selvi P., Sekaran G. (2019). Integrated thermophilic enzyme-immobilized reactor and high-rate biological reactors for treatment of palm oil-containing wastewater without sludge production. Bioprocess Biosyst. Eng. 42, 1053–1064. doi: 10.1007/s00449-019-02104-x
Sarmah P., Rout J. (2020). “Role of algae and cyanobacteria in bioremediation: prospects in polyethylene biodegradation,” in Advances in cyanobacterial biology (Amsterdam: Elsevier), 333–349.
Saxena G., Kishor R., Bharagava R. N. (2020). “Application of microbial enzymes in degradation and detoxification of organic and inorganic pollutants,” in Bioremediation of industrial waste for environmental safety (New York: Springer), 41–51.
Sayed K., Baloo L., Sharma N. K. (2021). Bioremediation of total petroleum hydrocarbons (TPH) by bioaugmentation and biostimulation in water with floating oil spill containment booms as bioreactor basin. Int. J. Environ. Res. Public Health 18 (5), 2226. doi: 10.3390/ijerph18052226
Sedlakova-Kadukova J., Kopcakova A., Gresakova L., Godany A., Pristas P. (2019). Bioaccumulation and biosorption of zinc by a novel streptomyces K11 strain isolated from highly alkaline aluminium brown mud disposal site. Ecotoxicol. Environ. Saf. 167, 204–211. doi: 10.1016/j.ecoenv.2018.09.123
Sen S. K., Patra P., Das C. R., Raut S., Raut S. (2019). Pilot-scale evaluation of bio-decolorization and biodegradation of reactive textile wastewater: an impact on its use in irrigation of wheat crop. Water Resour. Industry 21, 100106. doi: 10.1016/j.wri.2019.100106
Shah H., Jain S. (2020). “Bioremediation: an approach for environmental pollutants detoxification,” in Waste to energy: prospects and applications (New York: Springer), 121–142.
Shanmuganathan R., Karuppusamy I., Saravanan M., Muthukumar H., Ponnuchamy K., Ramkumar V. S., et al. (2019). Synthesis of silver nanoparticles and their biomedical applications-a comprehensive review. Curr. Pharm. Design 25 (24), 2650–2660. doi: 10.2174/1381612825666190708185506
Sharma P., Dutta D., Udayan A., Nadda A. K., Lam S. S., Kumar S. (2022a). Role of microbes in bioaccumulation of heavy metals in municipal solid waste: impacts on plant and human being. Environ. pollut. 305, 119248. doi: 10.1016/j.envpol.2022.119248
Sharma M., Nandy A., Taylor N., Venkatesan S. V., Kollath V. O., Karan K., et al. (2020). Bioelectrochemical remediation of phenanthrene in a microbial fuel cell using an anaerobic consortium enriched from a hydrocarbon-contaminated site. J. Hazard. Mater. 389, 121845. doi: 10.1016/j.jhazmat.2019.121845
Sharma P., Singh S. P., Iqbal H. M., Tong Y. W. (2022b). Omics approaches in bioremediation of environmental contaminants: an integrated approach for environmental safety and sustainability. Environ. Res. 211, 113102. doi: 10.1016/j.envres.2022.113102
Sharma P., Singh S. P., Parakh S. K., Tong Y. W. (2022c). Health hazards of hexavalent chromium (Cr (VI)) and its microbial reduction. Bioengineered 13 (3), 4923–4938. doi: 10.1080/21655979.2022.2037273
Singh R. K., Tripathi R., Ranjan A., Srivastava A. K. (2020). “Fungi as potential candidates for bioremediation,” in Abatement of environmental pollutants (Amsterdam: Elsevier), 177–191.
Siric I., Eid E. M., El-Morsy M. H., Osman H. E., Adelodun B., Abou Fayssal S., et al. (2022a). Health risk assessment of hazardous heavy metals in two varieties of mango fruit (Mangifera indica l. var. dasheri and langra). Horticulturae 8 (9), 832. doi: 10.3390/horticulturae8090832
Siric I., Eid E. M., Taher M. A., El-Morsy M. H., Osman H. E., Kumar P., et al. (2022b). Combined use of spent mushroom substrate biochar and PGPR improves growth, yield, and biochemical response of cauliflower (Brassica oleracea var. botrytis): a preliminary study on greenhouse cultivation. Horticulturae 8 (9), 830. doi: 10.3390/horticulturae8090830
Sonawane J. M., Rai A. K., Sharma M., Tripathi M., Prasad R. (2022). Microbial biofilms: recent advances and progress in environmental bioremediation. Sci. Total Environ. 824, 153843. doi: 10.1016/j.scitotenv.2022.153843
Song J., Zhang S., Xie Y., Li Q. (2019). Purification and characteristics of an aflatoxin B1 degradation enzyme isolated from pseudomonas aeruginosa. FEMS Microbiol. Lett. 366 (5), fnz034. doi: 10.1093/femsle/fnz034
Sonune N. (2021). “Microbes: a potential tool for bioremediation,” in Rhizobiont in bioremediation of hazardous waste. Eds. Vivek K., Ram P., Manoj K. (Singapore: Springer), 391–407.
Sonune N., Garode A. (2018). Isolation, characterization and identification of extracellular enzyme producer bacillus licheniformis from municipal wastewater and evaluation of their biodegradability. Biotechnol. Res. Innovation 2 (1), 37–44. doi: 10.1016/j.biori.2018.03.001
Sravya K., Sangeetha S. (2022). Feasibility study on bioremediation techniques to contaminated soils. Mater. Today: Proc. 51, 2556–2560. doi: 10.1016/j.matpr.2021.12.364
Sun W., Cheng K., Sun K. Y., Ma X. (2021). Microbially mediated remediation of contaminated sediments by heavy metals: a critical review. Curr. pollut. Rep. 7 (2), 201–212. doi: 10.1007/s40726-021-00175-7
Sun L., Guo D., Liu K., Meng H., Zheng Y., Yuan F., et al. (2019). Levels, sources, and spatial distribution of heavy metals in soils from a typical coal industrial city of tangshan, China. Catena 175, 101–109. doi: 10.1016/j.catena.2018.12.014
Tak H. I., Ahmad F., Babalola O. O. (2012). Advances in the application of plant growth-promoting rhizobacteria in phytoremediation of heavy metals. Rev. Environ. Contam. Toxicol. 223 (1), 33–52.
Tan W., Peralta-Videa J. R., Gardea-Torresdey J. L. (2018). Interaction of titanium dioxide nanoparticles with soil components and plants: current knowledge and future research needs–a critical review. Environ. Sci.: Nano 5 (2), 257–278. doi: 10.1039/C7EN00985B
Taran M., Fateh R., Rezaei S., Gholi M. K. (2019). Isolation of arsenic accumulating bacteria from garbage leachates for possible application in bioremediation. Iranian J. Microbiol. 11 (1), 60. doi: 10.18502/ijm.v11i1.707
Thakare M., Sarma H., Datar S., Roy A., Pawar P., Gupta K., et al. (2021). Understanding the holistic approach to plant-microbe remediation technologies for removing heavy metals and radionuclides from soil. Curr. Res. Biotechnol. 3, 84–98. doi: 10.1016/j.crbiot.2021.02.004
Thakur M., Medintz I. L., Walper S. A. (2019). Enzymatic bioremediation of organophosphate compounds–progress and remaining challenges. Front. Bioeng. Biotechnol. 7, 289. doi: 10.3389/fbioe.2019.00289
Tian Q., Dou X., Huang L., Wang L., Meng D., Zhai L., et al. (2020). Characterization of a robust cold-adapted and thermostable laccase from pycnoporus sp. SYBC-L10 with a strong ability for the degradation of tetracycline and oxytetracycline by laccase-mediated oxidation. J. Hazard. Mater. 382, 121084.
Unuofin J. O., Falade A. O., Aladekoyi O. J. (2021). Applications of microbial laccases in bioremediation of environmental pollutants: potential issues, challenges, and prospects. Bioremediat. Environ. Sustainability 1, 519–540. doi: 10.1016/B978-0-12-820524-2.00021-3
Unuofin J. O., Okoh A. I., Nwodo U. U. (2019). Aptitude of oxidative enzymes for treatment of wastewater pollutants: a laccase perspective. Molecules 24 (11), 2064. doi: 10.3390/molecules24112064
Verma S., Kuila A. (2019). Bioremediation of heavy metals by microbial process. Environ. Technol. Innovation 14, 100369. doi: 10.1016/j.eti.2019.100369
Verma S., Verma P. K., Chakrabarty D. (2019). Arsenic bio-volatilization by engineered yeast promotes rice growth and reduces arsenic accumulation in grains. Int. J. Environ. Res. 13 (3), 475–485. doi: 10.1007/s41742-019-00188-7
Vo M.-T., Nguyen V.-T., Dao T.-S. (2020). Responses of green algae and diatom upon exposure to chromium and cadmium. Vietnam J. Sci. Technol. Eng. 62 (1), 69–73. doi: 10.31276/VJSTE.62(1).69-73
Vocciante M., Finocchi A., De Folly D′ Auris A., Conte A., Tonziello J., Pola A., et al. (2019). Enhanced oil spill remediation by adsorption with interlinked multilayered graphene. Materials 12 (14), 2231. doi: 10.3390/ma12142231
Wang L., Liu Y., Shu X., Lu S., Xie X., Shi Q. (2019). Complexation and conformation of lead ion with poly-γ-glutamic acid in soluble state. PloS One 14 (9), e0218742. doi: 10.1371/journal.pone.0218742
Wang Z., Luo Z., Yan C., Rosenfeldt R. R., Seitz F., Gui H. (2018). Biokinetics of arsenate accumulation and release in microcystis aeruginosa regulated by common environmental factors: practical implications for enhanced bioremediation. J. Clean. Prod. 199, 112–120. doi: 10.1016/j.jclepro.2018.07.131
Wang Y., Mandal A. K., Son Y.-O., Pratheeshkumar P., Wise J. T., Wang L., et al. (2018). Roles of ROS, Nrf2, and autophagy in cadmium-carcinogenesis and its prevention by sulforaphane. Toxicol. Appl. Pharmacol. 353, 23–30. doi: 10.1016/j.taap.2018.06.003
Wei Y., Fu J., Wu J., Jia X., Zhou Y., Li C., et al. (2018). Bioinformatics analysis and characterization of highly efficient polyvinyl alcohol (PVA)-degrading enzymes from the novel PVA degrader stenotrophomonas rhizophila QL-P4. Appl. Environ. Microbiol. 84 (1), e01898–e01817. doi: 10.1128/AEM.01898-17
Wu W., Jiang S., Zhao Q., Zhang K., Wei X., Zhou T., et al. (2018). Environmental exposure to metals and the risk of hypertension: a cross-sectional study in China. Environ. pollut. 233, 670–678. doi: 10.1016/j.envpol.2017.10.111
Wu P., Zhang Z., Luo Y., Bai Y., Fan J. (2022). Bioremediation of phenolic pollutants by algae-current status and challenges. Bioresour. Technol. 350, 126930. doi: 10.1016/j.biortech.2022.126930
Xiao F., Cheng J., Cao W., Yang C., Chen J., Luo Z. (2019). Removal of heavy metals from aqueous solution using chitosan-combined magnetic biochars. J. Colloid Interface Sci. 540, 579–584. doi: 10.1016/j.jcis.2019.01.068
Xie Q., Liu N., Lin D., Qu R., Zhou Q., Ge F. (2020). The complexation with proteins in extracellular polymeric substances alleviates the toxicity of cd (II) to chlorella vulgaris. Environ. pollut. 263, 114102. doi: 10.1016/j.envpol.2020.114102
Xu Q., Wu B., Chai X. (2022). In situ remediation technology for heavy metal contaminated sediment: a review. Int. J. Environ. Res. Public Health 19 (24), 16767. doi: 10.3390/ijerph192416767
Ye X., Peng T., Feng J., Yang Q., Pratush A., Xiong G., et al. (2019). A novel dehydrogenase 17β-HSDx from rhodococcus sp. P14 with potential application in bioremediation of steroids contaminated environment. J. Hazard. Mater. 362, 170–177. doi: 10.1016/j.jhazmat.2018.09.023
Zacharia J. T. (2019). Degradation pathways of persistent organic pollutants (POPs) in the environment. Persistent Organic Pollutants, 17–30. doi: 10.5772/intechopen.79645
Zeng F., Qiu B., Wu X., Niu S., Wu F., Zhang G. (2012). Glutathione-mediated alleviation of chromium toxicity in rice plants. Biol. Trace Elem. Res. 148, 255–263. doi: 10.1007/s12011-012-9362-4
Zhang C., Gan C., Ding L., Xiong M., Zhang A., Li P. (2020). Maternal inorganic mercury exposure and renal effects in the wanshan mercury mining area, southwest China. Ecotoxicol. Environ. Saf. 189, 109987. doi: 10.1016/j.ecoenv.2019.109987
Zhao B., Johnston F. H., Salimi F., Kurabayashi M., Negishi K. (2020). Short-term exposure to ambient fine particulate matter and out-of-hospital cardiac arrest: a nationwide case-crossover study in Japan. Lancet Planet. Health 4 (1), e15–e23. doi: 10.1016/S2542-5196(19)30262-1
Zhou Y., Liu X., Wang J. (2019). Characterization of microplastics and the association of heavy metals with microplastics in suburban soil of central China. Sci. Total Environ. 694, 133798. doi: 10.1016/j.scitotenv.2019.133798
Zuhara S., Isaifan R. (2018). The impact of criteria air pollutants on soil and water: a review. J. Environ. Sci. pollut. Res. 4 (2), 278–284. doi: 10.30799/jespr.133.18040205
Keywords: microbial bioremediation, bioaugmentation, biostimulation, siderophores, biosorption
Citation: Ayilara MS and Babalola OO (2023) Bioremediation of environmental wastes: the role of microorganisms. Front. Agron. 5:1183691. doi: 10.3389/fagro.2023.1183691
Received: 10 March 2023; Accepted: 18 May 2023;
Published: 30 May 2023.
Edited by:
Parul Chaudhary, Graphic Era Hill University, IndiaReviewed by:
Viabhav Kumar Upadhayay, Dr. Rajendra Prasad Central Agricultural University, IndiaSami Abou Fayssal, University of Forestry, Sofia, Bulgaria
Copyright © 2023 Ayilara and Babalola. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Olubukola O. Babalola, b2x1YnVrb2xhLmJhYmFsb2xhQG53dS5hYy56YQ==
 Modupe S. Ayilara
Modupe S. Ayilara Olubukola O. Babalola
Olubukola O. Babalola