- 1Department of Medicine, Hualien Armed Forces General Hospital, Hualien City, Taiwan
- 2Department of Stomatology of Periodontology, Mackay Memorial Hospital, Taipei, Taiwan
- 3Departments of Dentistry, Tri-Service General Hospital, National Defense Medical Center, Taipei, Taiwan
- 4Department of Psychiatry, Hualien Tzu Chi General Hospital, Hualien City, Taiwan
- 5Department of Nursing, Hualien Armed Forces General Hospital, Hualien, Taiwan
- 6Nursing Department, China Medical University Hospital, Taichung, Taiwan
- 7School of Nursing and Graduate Institute of Nursing, China Medical University, Taichung, Taiwan
- 8Arnold School of Public Health, University of South Carolina, Columbia, SC, United States
- 9John Ochsner Heart and Vascular Institute, Ochsner Clinical School, The University of Queensland School of Medicine, New Orleans, LA, United States
- 10Departments of Medicine, Tri-Service General Hospital, National Defense Medical Center, Taipei, Taiwan
Background: The association observed between mental stress and metabolic syndrome (MetS) has varied across studies and may be confounded by physical activity (PA) and fitness status.
Method: This study included a military cohort of 2,854 participants in Taiwan who were not taking any medications and were free of baseline MetS. The Brief Symptoms Rating Scale (BSRS-5) includes five domains—depression, anxiety, hostility, insomnia, and interpersonal sensitivity—measured on a five-point Likert-type scale ranging from 0 to 4, with a maximum score of 20. PA (hrs/wk) was categorized into three levels: <150, 150–299 and ≥300. Aerobic fitness was evaluated by the amount of time taken to complete a 3,000 m run. MetS was defined according to the International Diabetes Federation (IDF) criteria. Multivariable Cox proportional hazards regression analysis with adjustments for potential covariates including PA and aerobic fitness was utilized to determine the associations of BSRS-5 scores (each 1-unit score increase) with the incidence of MetS and related features.
Results: During a median follow-up period of 5.8 years, 662 new-onset cases of MetS (23.2%) developed. BSRS-5 scores were not associated with the risk of new-onset MetS [hazard ratio (HR): 1.006 [95% confidence interval (CI): 0.975, 1.039]]. Among the five MetS features, the only one associated with BSRS-5 scores was the risk of new-onset hypertension [HR: 1.038 (95% CI: 1.002, 1.075)], which was defined as blood pressure ≥130/85 mmHg or the use of antihypertensive medications, among 2,405 participants free of baseline hypertension.
Conclusions: Our findings suggest that in young adult military personnel, mental stress was not associated with the incidence of MetS but was associated with its hypertension component, which was independent of PA and aerobic fitness.
Introduction
Metabolic syndrome (MetS), a cluster of conditions that occur together rather than a single ailment, is characterized by abdominal adiposity, hypertension, low high-density lipoprotein cholesterol (HDL-C), increased triglycerides, and insulin resistance (1, 2). People with metabolic abnormalities, whether alone or in synergy with other risk factors, face an increased risk of cardiovascular disease, diabetes, certain cancers, and an increased probability of premature death (3, 4). MetS and related conditions present substantial global public health challenges. Over a quarter of the world's population have MetS (approximately one billion people), and one-third of the Chinese population is affected (4). Recent reports also revealed that more than 50% of military personnel in the armed forces of the US and Taiwan had overweight/obesity (5, 6). Early detection of related risk factors and timely intervention are essential to prevent MetS from occurring or progressing into chronic diseases (7).
Psychological distress is also a significant public health issue in contemporary society (8). Approximately one out of eight individuals worldwide has a mental health issue, and nearly half of all people experience such issues at some point in their lives (9). Mental disorders can trigger physiological responses involving the neurological, endocrine, and immune systems, potentially contributing to MetS (4, 8, 10). For instance, eating in response to stress may result in overweight and obesity. The effect of emotional eating may be stronger among populations in high stress professions (4). Military personnel are obliged to perform regular training to maintain good fitness, which may reduce metabolic abnormalities (11). However, factors such as exposure to combat or rescue operations; witnessing traumatic events; and the demanding nature of military culture, e.g., extended deployments and a strong sense of duty, have tremendous effects on mental health which may cause emotional eating to promote overweight or obesity (12). There were only a few studies for military personnel in this context (4, 11, 13). In a recent statistics report in the U.K. army, the prevalence of overweight/obesity is over 64% in 2022–2023 (13). In the Millennium Cohort Study for U.S. military personnel, Boyko et al. have observed that mental stress, e.g., depression and anxiety was related to trouble sleep and sleep apnea that were associated with incident diabetes (14). We found that physical activity (PA) and aerobic fitness, which are crucial confounders in this association (15), were not controlled at baseline in these studies. In addition, our prior study showed an association of incident MetS with substance use which was highly related to mental stress in military personnel (16). Therefore, this study aimed to examine the association of mental stress with new-onset MetS in military personnel, with adjustments for baseline PA and aerobic fitness.
Materials and methods
Study participants
The cardiorespiratory fitness and health in the Eastern armed forces (CHIEF) (2) study was registered in 2014 and followed for the MetS events throughout 2020. At baseline in 2014, no participants took any antihypertensive, antidiabetic or lipid-lowering agents, and all of them received annual health examinations at the Hualien Armed Forces General Hospital, which is the only military referral hospital in Eastern Taiwan. The baseline (2014) and subsequent annual health examinations (2015–2020) consisted of subjective demographic reports and objective hemodynamic and blood metabolic biomarker measurements. In addition, participants were required to complete a questionnaire concerning their alcohol, betel nut, and tobacco use status (active vs. former/never) and PA levels at baseline (2014). Aerobic fitness was assessed by performance in a 3,000 m run test at baseline. The CHIEF study aimed to clarify the correlations and associations of PA and aerobic fitness with cardiometabolic risk in military personnel. The details of the CHIEF study have been described previously (15–22). The study protocol was approved by the Research Ethics Committee of the Mennonite Christian Hospital in Hualien City, Taiwan (certificate No. 16-05-008), and all participants provided written consent in accordance with ethical standards. Our study adhered strictly to the ethical guidelines outlined in the Declaration of Helsinki, ensuring compliance with all applicable regulations throughout the research process.
Annual health examinations
The anthropometric waist circumference, body height and body weight of each participant were measured while the participants were standing. Body mass index (BMI) was calculated as the body weight in kilograms divided by the square of the body height in meters. The blood pressure (BP) of each participant was measured once in a seated position utilizing an automatic device (FT201 Parama-Tech Co., Ltd., Fukuoka, Japan) (23–25) after the participant had rested for 15 min or more. Venous blood samples were drawn from each participant following a 12-hour overnight fast to assess the serum levels of total cholesterol, low-density lipoprotein cholesterol (LDL-C), HDL-C, triglycerides, and fasting glucose utilizing an automated analyzer (Olympus AU640, Kobe, Japan) (26, 27).
PA and aerobic fitness assessments
PA levels were assessed by self-reported engagement in leisure-time running over the past six months, categorized into <150, 150–299, and ≥300 min per week according to the American guidelines for exercise (12). Aerobic fitness was evaluated by time taken to complete a 3,000 m run test at the Military Physical Training and Testing Center in Hualien City, Taiwan. The 3,000 m run test was performed outdoors and conducted uniformly at 16:00 under suitable weather conditions, for example, with no heavy rain or high outdoor temperatures, e.g., >40 Celsius degrees, in accordance with military testing regulations (28).
Mental stress assessment
Mental stress was assessed using scores on the 5-item Brief Symptom Rating Scale (BSRS-5) (6). This scale assesses mood symptoms, including trouble falling asleep (insomnia), feeling tense (anxiety), feeling easily annoyed or irritated (hostility), being in a low mood (depression), and feeling inferior to others (interpersonal sensitivity). Each domain was scored on a five-point Likert-type scale ranging from 0 (not at all) to 4 (extremely severe). The maximum BSRS-5 score is 20. Participants had to self-report BSRS-5 scores and related domain scores to assess mental stress over the past 3 months at the baseline examination. In a sample of 1,222 Taiwanese military personnel in a prior study (29), the BSRS-5 score and an additional question for suicidal ideation [on a scale from 0 (none) to 3 (severe)] were validated by the Adult Self-Report Scale-4, a self-reported rating scale including 136 items about a wide range of psychiatric symptoms of DSM-IV diagnoses (30); where the results showed moderate to high correlations regarding suicidality [r = 0.534–0.902].
Definition of incident MetS events
In accordance with the International Diabetes Federation criteria specifically for Chinese individuals (1), MetS was defined as the presence of three or more of the following clinical features: (1) waist circumference ≥80 cm in women and ≥90 cm in men; (2) SBP ≥130 mmHg, and/or DBP ≥85 mmHg, or a use of anti-hypertensive medications; (3) HDL-C <50 mg/dl in women and <40 mg/dl in men; (4) triglycerides ≥150 mg/dl, or a used of lipid-lowering medications; and (5) fasting glucose ≥100 mg/dl, or a use of antidiabetic medications.
Statistical analysis
The baseline characteristics of the study cohort are presented as the mean ± standard deviations (SD) for continuous variables and as number (percentage) for categorical variables. Follow-up for each participant began at baseline (2014) and continued until the first occurrence of MetS, loss to follow-up, or the end of the follow-up period (December 31, 2020). The severity of mental stress was classified into three categories based on BSRS-5 scores ≤1, 2, and ≥3 or treated as a continuous variable (each 1-unit increase). We utilized multivariable Cox proportional hazards regression models to investigate the association between BSRS-5 score and incident MetS. Model 1 is crude model. In Model 2, age, sex, waist circumference, SBP, DBP, HDL-C, triglycerides, and fasting glucose are adjusted at baseline. Model 3 includes adjustments for substance use status and the covariates in Model 2. Model 4 includes adjustments for baseline PA levels, aerobic fitness, and the covariates in Model 3. All confounders were chosen for an association with mental stress or MetS in priorly published reports (18, 23, 25). As is known, Cox regression model is the most commonly used method for the analysis of survival data, particularly in some well-known observational cohort studies (11, 31) since it does not require the assumption of survival distributions for the data (32). Statistical significance was defined as two-sided P values <0.05. All statistical analyses were performed using SPSS v26.0 software for Windows (IBM Corp., Armonk, NY, USA).
Results
The CHIEF cohort study recruited 4,080 military personnel at baseline (2014). Participants meeting specific criteria were excluded at baseline, i.e., those with MetS (N = 457), those aged 40 years or older (N = 58), those with missing mental health information (N = 36), and those who left the military bases in Eastern Taiwan area and were lost to follow-up (N = 675). Ultimately, a total of 2,854 participants were included in the final analysis (Figure 1). Over a mean 6-year period of follow-up from January 1, 2014, to December 31, 2020, we identified 662 (23.2%) incident cases with new-onset MetS. Table 1 reveals the baseline demographic and clinical characteristics of the study cohort. The participants who developed MetS were older and had a larger waist circumference; higher BMI, SBP, and DBP; elevated serum total cholesterol, LDL-C, triglycerides and fasting glucose levels; and lower HDL-C levels at baseline (Table 1) than those who did not develop MetS. Additionally, the participants with new-onset MetS were observed to have a higher prevalence of active substance use, i.e., alcohol intake, betel nut chewing and cigarette smoking. Moreover, their aerobic fitness was marginally lower in PA levels (p = 0.09).
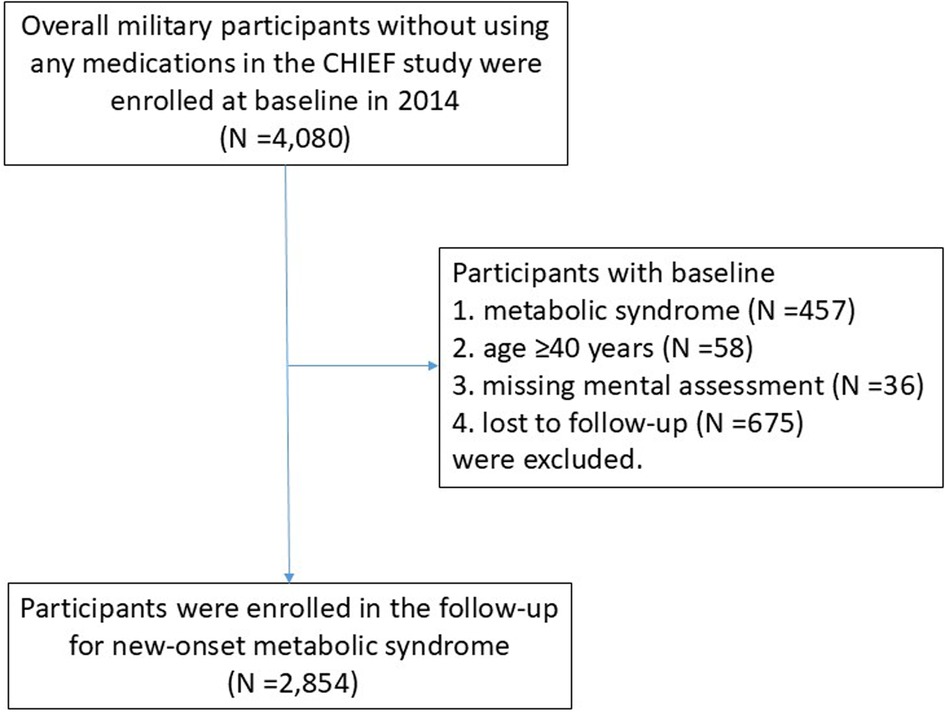
Figure 1. The flowchart to select eligible participants at baseline (2014) for the follow-up of new-onset metabolic syndrome in the CHIEF study, 2014–2020.
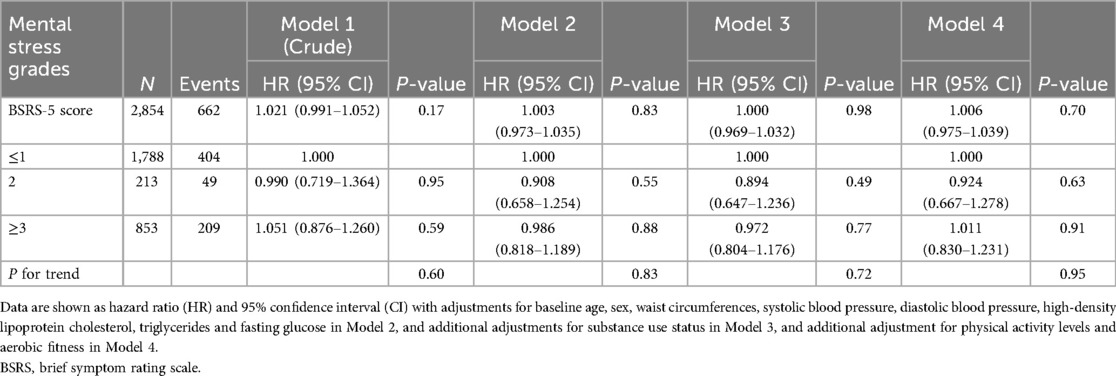
Table 2 demonstrates the results of the multivariable Cox regression analysis examining incident new-onset MetS in relation to baseline BSRS-5 scores. There was no association found in either the crude model or the multivariable models, regardless of whether the BSRS-5 score was treated as a continuous variable or a categorical variable.
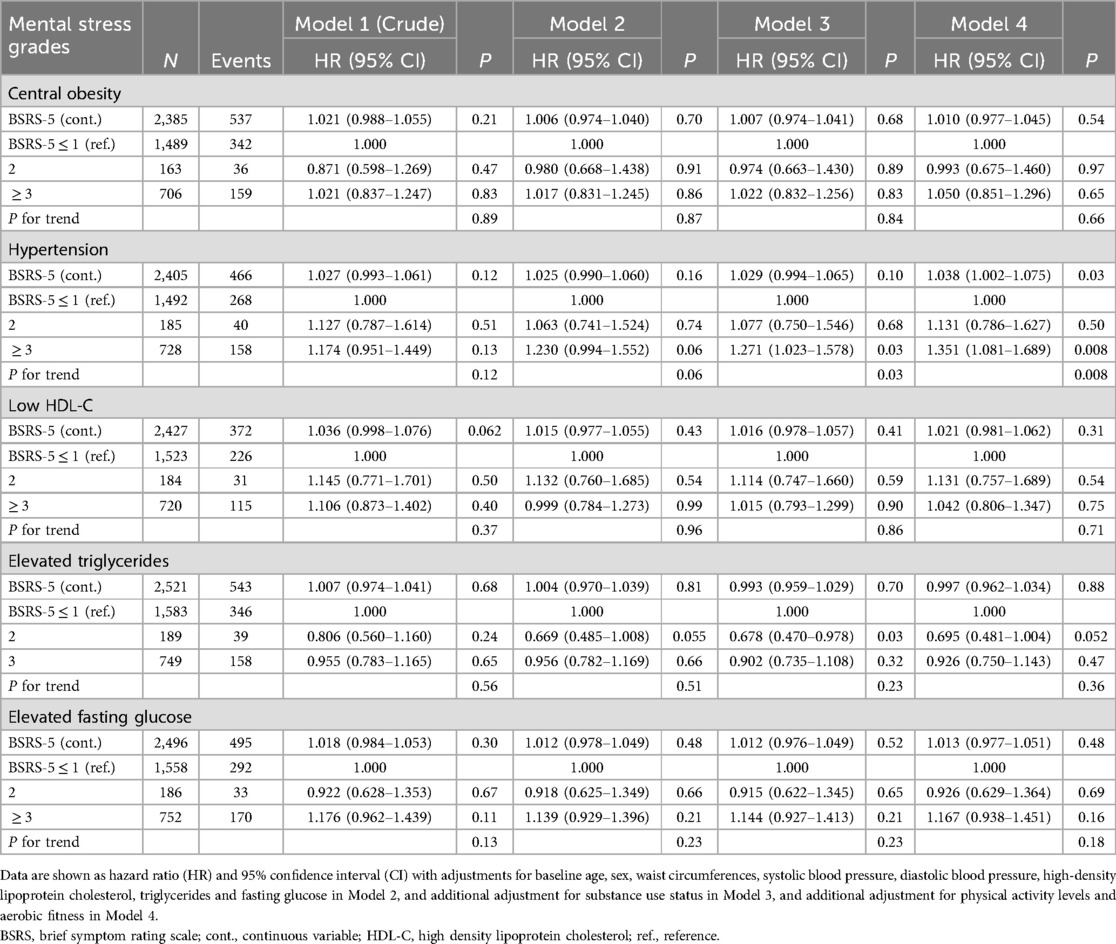
Table 3 shows the results for the incidences of various metabolic abnormalities in relation to BSRS-5 scores. When BSRS-5 scores were treated as a continuous variable, higher scores were associated with a greater incidence of new-onset hypertension [hazard ratio (HR): 1.038 (95% confidence interval (CI): 1.002, 1.075)]. Similarly, when BSRS-5 scores were treated as a categorical variable, a tendency toward a higher BSRS-5 score (2 and ≥3) was associated with a greater risk of new-onset hypertension than the BSRS-5 scores ≤1 [HRs: 1.131 (95% CI: 0.786, 1.627) and 1.351 (95% CI: 1.081, 1.689), respectively; p for trend = 0.008]. However, the other MetS clinical features (abdominal obesity, prediabetes, low HDL-C, and elevated triglycerides) showed no associations with BSRS-5 scores.
Discussion
To the best of our knowledge, this study represents the first investigation into the associations between mental stress and the incidences of new-onset MetS and related metabolic components in a young military population. We did not identify an association between mental stress assessed by the BSRS-5 score and incident new-onset MetS, despite controlling for various potential confounders, including baseline PA levels, aerobic fitness, and other covariates. Nevertheless, our findings suggest a dose‒response association between mental stress grades and new-onset hypertension.
Based on the complex pathomechanisms of stress, a correlation between mental stress and MetS appears to be justified. Mental stress activates the sympathetic nervous system and adrenal medulla, resulting in catecholamine release, and triggers the hypothalamic‒pituitary‒adrenal (HPA) axis which increases blood cortisol levels (31). Both catecholamines and cortisol contribute to elevated blood glucose levels and insulin resistance, which are key factors in MetS. Mental stress also induces the chemokines and inflammatory cytokines release which promote the development of insulin resistance, diabetes and MetS (8, 33, 34). The stress-induced inflammatory process may involve sympathetic nervous system activation and NF-κB transcription factor activation (35). Psychiatric disorders such as bipolar disorder and posttraumatic stress disorder (3, 36–38), and mental stress (8, 39), have been found as risk factors for MetS. However, findings from a cohort study suggested that mental stress might be a consequence rather than a cause of MetS (40). Another study reported a specific association of depression with MetS in a hospital sample in Taiwan (41). Our results may differ from those of prior studies due to variations in the definition of mental stress. Furthermore, previous meta-analyses have revealed varying impacts of stress on MetS, with occupational stress having the strongest impact and general perceived stress having a weaker impact (42). Additionally, our assessment of mental stress relied on self-reports rather than objective clinical diagnoses, which might be another possible reason accounting for the null association between mental stress and incident MetS in this study. Consistent with our findings, several studies also reported no relationship between MetS and mental stress (4, 8, 11, 13, 43).
A growing body of evidence suggests an association between mental stress and hypertension (4, 40, 44–47), particularly affecting DBP instead of SBP (42, 48). In a meta-analysis by Liu et al., those with mental stress had a twofold increase in their probability of hypertension (odds ratio: 2.40, 95% CI: 1.65–3.49) (46). In contrast, a few studies showed no association between mental stress and hypertension (48, 49). A study for Ghanaians in Amsterdam demonstrated that mental stress, e.g., permitted discrimination, depression and financial stress, did not increase the prevalence of hypertension (49). Chronic exposure to mental stress could sustain this heightened sympathetic activity, which may lead to cardiovascular issues such as cardiac arrhythmias, elevated BP, and, in severe cases, sudden death (47). Chronic mental stress may affect neuroglial circuits or compromise antioxidant systems, possibly altering the response of neurons to stress (47). Other metabolic abnormalities, specifically, central obesity, insulin resistance, and dyslipidemia, have also been reported to be linked to mental stress in some prior studies (8, 39). Nevertheless, PA and aerobic fitness were rarely controlled in the prior studies to address this issue, which may lead to a bias (50). Military personnel belong to a distinct group and are commonly required to engage in rigorous PA to have superior fitness. This may counteract the negative impact of mental stress on metabolic health.
Several limitations should be considered when interpreting the data in this study. First, the self-report questionnaire used in this study to assess mental health provided only suboptimal evidence compared with clinical diagnosis. Second, military personnel represent a specific population, and the small sample size of female participants in this study may restrict the generalizability of our results to the broader public. Third, our results do not appear to be adjusted for relevant social and psychological factors. This is a significant limitation, as such factors are known to influence both mental stress and metabolic outcomes. In contrast, this study was strengthened by the detailed data on the baseline characteristics of the participants, which facilitated adjustments for potential confounding factors. Lifestyle factors such as PA and diet can affect both mental stress and the development of MetS in the general population. The uniformity among military personnel regarding their living environment, diets, and health care provisions helps minimize the impact of unmeasured variables. In addition, several crucial confounders that have been relatively understudied in prior research, such as PA and aerobic fitness levels, were controlled for at baseline.
Conclusions
Our findings suggest that mental stress may not be effective in predicting the development of MetS, but it does increase the risk of new-onset hypertension among military populations. Based on our study findings, prevention of new-onset hypertension should be emphasized and initiated in military personnel who experience high mental stress. Future studies specifically focusing on the military population will be required. It is crucial to consider the realities of this population across different countries in order to deepen understanding of the relationships between mental stress, MetS, obesity, and other chronic diseases.
Data availability statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Ethics statement
The studies involving humans were approved by This study conforms to the purpose of the Helsinki Declaration and was approved by the Mennonite Christian Hospital (registration number: 16-05-008). The requirement for clinical trial registration was waived because of the study's retrospective design. The studies were conducted in accordance with the local legislation and institutional requirements. The participants provided their written informed consent to participate in this study.
Author contributions
K-ZT: Formal analysis, Investigation, Writing – original draft. KHL: Investigation, Supervision, Validation, Visualization, Writing – review & editing. A-HH: Writing – review & editing, Data curation, Methodology, Supervision, Project administration, Resources, Visualization. Y-CC: Investigation, Supervision, Validation, Visualization, Writing – review & editing. XS: Investigation, Supervision, Validation, Writing – review & editing. CL: Investigation, Supervision, Validation, Visualization, Writing – review & editing. G-ML: Conceptualization, Data curation, Funding acquisition, Investigation, Methodology, Project administration, Resources, Supervision, Validation, Visualization, Writing – review & editing.
Funding
The author(s) declare that financial support was received for the research and/or publication of this article. The study was supported by the Medical Affairs Bureau Ministry of National Defense (MND-MAB-D-114222) and Hualien Armed Forces General Hospital (HAFGH-D-114008), where was the main place involved in the study design, data collection, analyses and writing of this research.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declared that they were an editorial board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision.
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Alberti KG, Zimmet P, Shaw J. Metabolic syndrome–a new world-wide definition. A consensus statement from the international diabetes federation. Diabet Med. (2006) 23:469–80. doi: 10.1111/j.1464-5491.2006.01858.x
2. Gonzalez-Chavez A, Chavez-Fernandez JA, Elizondo-Argueta S, Gonzalez-Tapia A, Leon-Pedroza JI, Ochoa C. Metabolic syndrome and cardiovascular disease: a health challenge. Arch Med Res. (2018) 49:516–21. doi: 10.1016/j.arcmed.2018.10.003
3. Restrepo Moreno S, Garcia Valencia J, Vargas C, Lopez-Jaramillo C. Cognitive development in patients with bipolar disorder and metabolic syndrome. Rev Colomb Psiquiatr (Engl Ed). (2019) 48:149–55. doi: 10.1016/j.rcp.2017.10.003
4. Li C, Tao T, Tang Y, Lu H, Zhang H, Li H, et al. The association of psychological stress with metabolic syndrome and its components: cross-sectional and bidirectional two-sample Mendelian randomization analyses. Front Endocrinol (Lausanne). (2023) 14:1212647. doi: 10.3389/fendo.2023.1212647
5. Webber BJ, Bornstein DB, Deuster PA, O'Connor FG, Park S, Rose KM, et al. BMI and physical activity, military-aged U.S. population 2015–2020. Am J Prev Med. (2023) 64:66–75. doi: 10.1016/j.amepre.2022.08.008
6. Lin KH, Su FY, Yang SN, Liu MW, Kao CC, Nagamine M, et al. Body mass index and association of psychological stress with exercise performance in military members: the cardiorespiratory fitness and hospitalization events in armed forces (CHIEF) study. Endocr Metab Immune Disord Drug Targets. (2021) 21:2213–9. doi: 10.2174/1871530321666210427090550
7. Tenk J, Matrai P, Hegyi P, Rostas I, Garami A, Szabo I, et al. Perceived stress correlates with visceral obesity and lipid parameters of the metabolic syndrome: a systematic review and meta-analysis. Psychoneuroendocrinology. (2018) 95:63–73. doi: 10.1016/j.psyneuen.2018.05.014
8. Singh B, Olds T, Curtis R, Dumuid D, Virgara R, Watson A, et al. Effectiveness of physical activity interventions for improving depression, anxiety and distress: an overview of systematic reviews. Br J Sports Med. (2023) 57:1203–9. doi: 10.1136/bjsports-2022-106195
9. Haykin H, Rolls A. The neuroimmune response during stress: a physiological perspective. Immunity. (2021) 54:1933–47. doi: 10.1016/j.immuni.2021.08.023
10. Garcez A, Weiderpass E, Canuto R, Lecke SB, Spritzer PM, Pattussi MP, et al. Salivary cortisol, perceived stress, and metabolic syndrome: a matched case-control study in female shift workers. Horm Metab Res. (2017) 49:510–9. doi: 10.1055/s-0043-101822
11. Tsai KZ, Chu CC, Huang WC, Sui X, Lavie CJ, Lin GM. The combined effect of cardiorespiratory and muscular fitness on the incidence of metabolic syndrome before midlife. J Cachexia Sarcopenia Muscle. (2024) 15:1483–90. doi: 10.1002/jcsm.13503
12. Tsai KZ, Liu PY, Lin YP, Chu CC, Huang WC, Sui X, et al. Do the American guideline-based leisure time physical activity levels for civilians benefit the mental health of military personnel? Front Psychiatry. (2023) 14:1255516. doi: 10.3389/fpsyt.2023.1255516
13. Official Statistics. Obesity profile: short statistical commentary May 2024—GOV.UK (2024). Available online at: https://www.gov.uk/government/statistics/update-to-the-obesity-profile-on-fingertips/obesity-profile-short-statistical-commentary-may-2024 (Accessed July 29, 2025).
14. Boyko EJ, Seelig AD, Jacobson IG, Hooper TI, Smith B, Smith TC, et al. Sleep characteristics, mental health, and diabetes risk: a prospective study of U.S. Military service members in the millennium cohort study. Diabetes Care. (2013) 36:3154–61. doi: 10.2337/DC13-0042
15. Lin KH, Chen YJ, Yang SN, Liu MW, Kao CC, Nagamine M, et al. Association of psychological stress with physical fitness in a military cohort: the CHIEF study. Mil Med. (2020) 185:e1240–6. doi: 10.1093/milmed/usz469
16. Liu WN, Hsu YC, Lin YP, Tsai KZ, Chang YC, Liu PY, et al. Substance use and incidence of metabolic syndrome before midlife among military adults: the CHIEF cohort study. Front Public Health. (2024) 12:1406524. doi: 10.3389/fpubh.2024.1406524
17. Lin GM, Li YH, Lee CJ, Shiang JC, Lin KH, Chen KW, et al. Rationale and design of the cardiorespiratory fitness and hospitalization events in armed forces study in Eastern Taiwan. World J Cardiol. (2016) 8:464–71. doi: 10.4330/wjc.v8.i8.464
18. Tsai KZ, Chu CC, Huang WC, Sui X, Lavie CJ, Lin GM. Prediction of various insulin resistance indices for the risk of hypertension among military young adults: the CHIEF cohort study, 2014–2020. Cardiovasc Diabetol. (2024) 23:141. doi: 10.1186/s12933-024-02229-8
19. Liu WN, Feng AC, Hsu CY, Liu PY, Tsai KZ, Zeng HC, et al. Mitral valve prolapse and physical performance in Asian military males: the CHIEF Heart study. J Sports Sci. (2023) 41:1179–86. doi: 10.1080/02640414.2023.2260626
20. Wang SH, Chung PS, Lin YP, Tsai KZ, Lin SC, Fan CH, et al. Metabolically healthy obesity and physical fitness in military males in the CHIEF study. Sci Rep. (2021) 11:9088. doi: 10.1038/s41598-021-88728-0
21. Fan CH, Lin SC, Tsai KZ, Wu TJ, Lin YP, Lin YK, et al. Association of single measurement of dipstick proteinuria with physical performance of military males: the CHIEF study. BMC Nephrol. (2020) 21:287. doi: 10.1186/s12882-020-01948-w
22. Chung PS, Tsai KZ, Lin YP, Lin YK, Lin GM. Association between leukocyte counts and physical fitness in male military members: the CHIEF Study. Sci Rep. (2020) 10:6082. doi: 10.1038/s41598-020-63147-9
23. Lin YP, Fan CH, Tsai KZ, Lin KH, Han CL, Lin GM. Psychological stress and long-term blood pressure variability of military young males: the cardiorespiratory fitness and hospitalization events in armed forces study. World J Cardiol. (2020) 12:626–33. doi: 10.4330/wjc.v12.i12.626
24. Lin YK, Liu PY, Fan CH, Tsai KZ, Lin YP, Lee JM, et al. Metabolic biomarkers and long-term blood pressure variability in military young male adults. World J Clin Cases. (2020) 8:2246–54. doi: 10.12998/wjcc.v8.i11.2246
25. Lin GM, Tsai KZ, Lin CS, Han CL. Physical fitness and long-term blood pressure variability in young male military personnel. Curr Hypertens Rev. (2020) 16(2):156–60. doi: 10.2174/1573402115666191023111351
26. Huang WC, Tsai KZ, Yang KT, Chen HH, Kwon Y, Lin GM. A comparison of various insulin resistance indices and the possibility of hypertension in military adults: CHIEF study. Diabetol Metab Syndr. (2024) 16:78. doi: 10.1186/s13098-024-01323-x
27. Lin YP, Hsu YC, Tsai KZ, Kwon Y, Lin GM. Insulin resistance indices and carotid intima-media thickness in physically fit adults: cHIEF atherosclerosis study. Endocr Metab Immune Disord Drug Targets. (2023) 23:1442–8. doi: 10.2174/1871530323666230324104737
28. Tsai KZ, Lin JW, Lin F, Su FY, Li YH, Lin YP, et al. Association of betel nut chewing with exercise performance in a military male cohort: the CHIEF study. J R Army Med Corps. (2018) 164:399–404. doi: 10.1136/jramc-2017-000899
29. Ma CC, Dai YM. Cut-off values of five-item brief symptom rating scale in evaluating suicidality among military recruits. Taiwanese J Psychiatry. (2014) 28:109–20.
30. Gadow K, Sprafkin J, Weiss M. Adult Self-Report Inventory 4 Manual. Stony Brook, NY: Checkmate Plus (2004).
31. Lin GM, Lloyd-Jones DM, Colangelo LA, Lima JAC, Szklo M, Liu K. Association between secondhand smoke exposure and incident heart failure: the multi-ethnic study of atherosclerosis (MESA). Eur J Heart Fail. (2024) 26:199–207. doi: 10.1002/ejhf.3155
32. George B, Seals S, Aban I. Survival analysis and regression models. J Nucl Cardiol. (2014) 21:686–94. doi: 10.1007/s12350-014-9908-2
33. Seematter G, Binnert C, Martin JL, Tappy L. Relationship between stress, inflammation and metabolism. Curr Opin Clin Nutr Metab Care. (2004) 7:169–73. doi: 10.1097/00075197-200403000-00011
34. Deak T, Quinn M, Cidlowski JA, Victoria NC, Murphy AZ, Sheridan JF. Neuroimmune mechanisms of stress: sex differences, developmental plasticity, and implications for pharmacotherapy of stress-related disease. Stress. (2015) 18:367–80. doi: 10.3109/10253890.2015.1053451
35. Bierhaus A, Wolf J, Andrassy M, Rohleder N, Humpert PM, Petrov D, et al. A mechanism converting psychosocial stress into mononuclear cell activation. Proc Natl Acad Sci U S A. (2003) 100:1920–5. doi: 10.1073/pnas.0438019100
36. Leonard BE. Inflammation as the cause of the metabolic syndrome in depression. Mod Trends Pharmacopsychiatry. (2013) 28:117–26. doi: 10.1159/000343974
37. Bartoli F, Carra G, Crocamo C, Carretta D, Clerici M. Metabolic syndrome in people suffering from posttraumatic stress disorder: a systematic review and meta-analysis. Metab Syndr Relat Disord. (2013) 11:301–8. doi: 10.1089/met.2013.0010
38. Theodosis-Nobelos P, Asimakopoulou E, Madianos M. Pathophysiological mechanisms of major mental disorders related to cardiovascular disease. Psychiatriki. (2022) 33:113–23. doi: 10.22365/jpsych.2021.038
39. Mehdi S, Wani SUD, Krishna KL, Kinattingal N, Roohi TF. A review on linking stress, depression, and insulin resistance via low-grade chronic inflammation. Biochem Biophys Rep. (2023) 36:101571. doi: 10.1016/j.bbrep.2023.101571
40. Raikkonen K, Matthews KA, Kuller LH. The relationship between psychological risk attributes and the metabolic syndrome in healthy women: antecedent or consequence? Metab Clin Exp. (2002) 51:1573–7. doi: 10.1053/meta.2002.36301
41. Chen YJ, Lin CL, Li CR, Huang SM, Chan JY, Fang WH, et al. Associations among integrated psychoneuroimmunological factors and metabolic syndrome. Psychoneuroendocrinology. (2016) 74:342–9. doi: 10.1016/j.psyneuen.2016.09.027
42. Kuo WC, Bratzke LC, Oakley LD, Kuo F, Wang H, Brown RL. The association between psychological stress and metabolic syndrome: a systematic review and meta-analysis. Obes Rev. (2019) 20:1651–64. doi: 10.1111/obr.12915
43. Ortega-Montiel J, Posadas-Romero C, Ocampo-Arcos W, Medina-Urrutia A, Cardoso-Saldana G, Jorge-Galarza E, et al. Self-perceived stress is associated with adiposity and atherosclerosis. The GEA Study. BMC Public Health. (2015) 15:780. doi: 10.1186/s12889-015-2112-8
44. Von Visger TT, Kuntz KK, Phillips GS, Yildiz VO, Sood N. Quality of life and psychological symptoms in patients with pulmonary hypertension. Heart Lung. (2018) 47:115–21. doi: 10.1016/j.hrtlng.2017.12.004
45. Balint EM, Boseva P, Schury K, Guendel H, Rottbauer W, Waller C. High prevalence of posttraumatic stress in patients with primary hypertension. Gen Hosp Psychiatry. (2016) 38:53–8. doi: 10.1016/j.genhosppsych.2015.10.002
46. Liu MY, Li N, Li WA, Khan H. Association between psychosocial stress and hypertension: a systematic review and meta-analysis. Neurol Res. (2017) 39:573–80. doi: 10.1080/01616412.2017.1317904
47. Fontes MAP, Marins FR, Patel TA, de Paula CA, Dos Santos Machado LR, de Sousa Lima EB, et al. Neurogenic background for emotional stress-associated hypertension. Curr Hypertens Rep. (2023) 25:107–16. doi: 10.1007/s11906-023-01235-7
48. Fauvel JP, M'Pio I, Quelin P, Rigaud JP, Laville M, Ducher M. Neither perceived job stress nor individual cardiovascular reactivity predict high blood pressure. Hypertension. (2003) 42:1112–6. doi: 10.1161/01.HYP.0000102862.93418.EE
49. Agyei B, Nicolaou M, Boateng L, Dijkshoorn H, van den Born BJ, Agyemang C. Relationship between psychosocial stress and hypertension among Ghanaians in Amsterdam, The Netherlands–the GHAIA study. BMC Public Health. (2014) 14:692. doi: 10.1186/1471-2458-14-692
Keywords: cohort study, mental stress, metabolic syndrome, military personnel, hypertension
Citation: Tsai K-Z, Lin K-H, Hung A-H, Chang Y-C, Sui X, Lavie CJ and Lin G-M (2025) Mental stress and the risks of metabolic syndrome and related components in military personnel: CHIEF cohort study. Front. Cardiovasc. Med. 12:1432464. doi: 10.3389/fcvm.2025.1432464
Received: 19 May 2024; Accepted: 5 August 2025;
Published: 21 August 2025.
Edited by:
Matilde Otero-Losada, National Scientific and Technical Research Council (CONICET), ArgentinaReviewed by:
Masoudeh Babakhanian, Semnan University of Medical Sciences, IranLuiz Claudio Barreto Silva Neto, Federal University of Espirito Santo, Brazil
Copyright: © 2025 Tsai, Lin, Hung, Chang, Sui, Lavie and Lin. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Gen-Min Lin, ZmFybWVyNTA3QHlhaG9vLmNvbS50dw==
 Kun-Zhe Tsai
Kun-Zhe Tsai Ko-Huan Lin
Ko-Huan Lin Ai-Hsiu Hung5
Ai-Hsiu Hung5 Xuemei Sui
Xuemei Sui Carl J. Lavie
Carl J. Lavie Gen-Min Lin
Gen-Min Lin