Abstract
Coastal wetlands of the Yellow Sea and Bohai Sea, China, along the East Asian–Australasian Flyway (EAAF) migratory route provide important stopover sites for waterbirds. Natural wetland loss caused by external stress has posed serious threats to the population of waterbirds. Elucidating the extent to which species depend on natural wetland and providing conservation and management recommendations for species are important steps toward relieving such population declines. We created a natural landscape (NL) index along the coastal wetlands of the Yellow and Bohai Seas, China, using the inverse distance-weighted nearest-neighbor approach. Then, we used a bootstrapping procedure to combine the NL index with 11,485 occurrence records for 80 waterbird species attributed to four functional groups (shorebirds, ducks, herons, and gulls) to quantify species’ dependence on coastal natural wetlands. Twenty-seven out of the 80 species selected (16 shorebird, 3 duck, 4 heron, and 4 gull species) significantly depended on natural wetlands. The shorebirds [standardized effect size (SES) = 4.37] and herons (SES = 2.56) were more dependent on natural wetlands than the ducks (SES = −0.02) and gulls (SES = −3.22). The threatened species (those classified as critically endangered, endangered, vulnerable, or near threatened) showed significantly higher dependence on natural wetlands than the non-threatened species (t = 2.613, df = 78, p < 0.05). Of the 27 species showing significant dependence on natural wetlands, only nine species were listed as national protected species. Threatened species that highly depend on natural wetlands need more attention as these species could face greater risk due to natural wetland loss.
1 Introduction
Increasing anthropogenic land conversion and climate change threaten species and their habitats (Harte et al., 2004; Newbold et al., 2015). However, how habitat change affects species depends on the species’ habits and their habitat preferences (Wang et al., 2015; Keinath et al., 2016). For example, species that prefer to forage in artificial habitats will be less affected by natural landscape loss than those that forage in natural habitats (Jackson et al., 2020) because they can use artificial habitats converted from natural landscapes to adapt to external disturbance (Lei et al., 2018; Jackson et al., 2019). Therefore, the variation of species’ dependence on natural landscapes could cause different responses to habitat change. Identifying the extent to which species are distributed among natural or human-dominated landscapes can provide information on which species are at greatest risk of habitat loss from land use change (Broms et al., 2014). However, the lack of long-term systematic monitoring data on species abundance limited its understanding (Meretsky et al., 2012).
Citizen science data are collected by atlases, survey reports, and websites (Ma et al., 2012; Hu et al., 2017). These data have long been used to infer the relationship between species distribution and habitat environments to identify priority conservation areas (Long et al., 2019; Huang et al., 2020; Guo et al., 2021). In addition, ecological models combined with citizen science data have also been heavily used to reveal the response of species distribution to climate change and land use change (Hu et al., 2017; Hu et al., 2020). However, how citizen science data are used to reveal the selection preference between natural habitats and artificial wetlands is unclear.
The migratory routes of waterbirds cover thousands of kilometers between breeding and non-breeding sites, and increasing disturbance by human activity at stopover sites interrupts their journeys (Melville et al., 2016; Studds et al., 2017). Decreased habitat quantity and quality at these stopover sites caused by external disturbances pose threats to the populations of waterbirds (Duan et al., 2019; Ma et al., 2019). Migratory waterbirds greatly depend on natural wetlands, but some species are able to use degraded artificial wetlands as alternative habitats (Lei et al., 2018; Jackson et al., 2019). Therefore, species have different dependencies of natural wetlands, and the response of animals to habitat change greatly relies on their dependence on natural wetlands (Xu et al., 2019).
The coastal wetlands of the Yellow Sea and Bohai Sea in China are important stopover sites for migratory waterbirds along the East Asian–Australasian Flyway (EAAF) migratory route (Bai et al., 2015). Since 1960, coastal zones have lost large areas of natural landscapes because of rapid urban development, such as overuse of natural resources and land reclamation (Murray et al., 2018). These activities have led to a dramatic decrease in waterbird populations in the area (Hua et al., 2015). Previous studies mainly explored the natural wetland change caused by disturbance activities using geographic information system (GIS) and remote sensing (RS) (Murray et al., 2014; Li et al., 2021) and how wetland change affected habitat distributions (Duan et al., 2019). A few studies have checked how waterbirds use habitat. Previous results indicated that threatened shorebird species have low occurrence frequency and larger-bodied species have low feeding frequency at artificial sites converted from coastal wetlands (Jackson et al., 2020); therefore, there is differing dependence of species on coastal wetlands. Species that significantly depend on coastal wetlands have experienced a rapid population decline since coastal wetland conversion (Studds et al., 2017). Therefore, it is necessary to clarify species’ dependence on coastal wetlands and provide target species conservation.
We created a natural landscape (NL) index to calculate the degree of naturalization of the specific raster cell. Then, using a bootstrapping procedure, we combined the NL index with citizen science data to quantify the dependence of migratory waterbirds on the coastal natural landscapes of the Yellow and Bohai Seas, China. The higher a species’ dependence on the natural landscape, the greater the risk to its population from habitat change. The aim of our study was to provide information that can facilitate the development of target species conservation to restrict further population decline of migratory waterbirds in the EAAF.
2 Data and Methods
2.1 Study Area
The Yellow and Bohai Seas in China are spread across the provinces and municipalities of Liaoning, Hebei, Tianjin, Shandong, and Jiangsu in Mainland China (Figure 1). The coastal wetlands within this range mainly consist of natural wetland tidal flats, bottomland, estuarine delta, and estuarine water. Artificial wetlands converted from coastal wetlands mainly include salt pan, aquaculture, and paddy fields. These wetlands provide important foraging and resting sites for migratory waterbirds, and the populations of some species along these areas exceed 1% of the global or flyway populations (Bai et al., 2015). The area and function of the coastal wetlands in this region experienced serious loss and degradation between 2000 and 2015 (Duan et al., 2019), which led to rapid population declines.
Figure 1
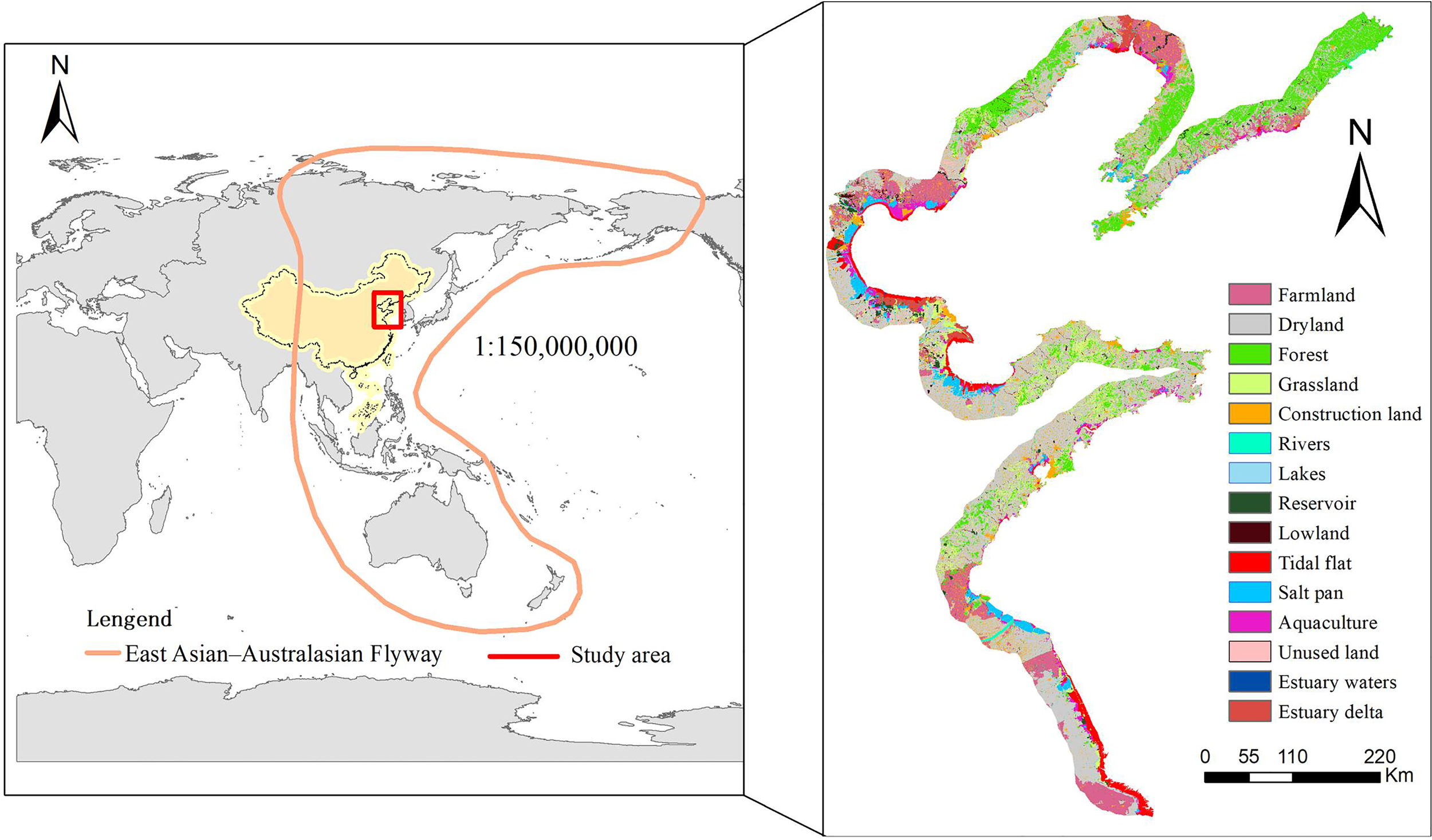
Study area in the Yellow Sea and Bohai Sea, China.
2.2 Citizen Science Data
We collected species occurrence records from multiple sources of citizen science data, including publicly accessible websites: eBird (https://ebird.org/home), Global Biodiversity Information Facility (GBIF; https://www.gbif.org/), BirdReport (http://www.birdreport.cn/), and the China Coastal Waterbird Census Group (https://cms.hkbws.org.hk/cms/resource-tw/publish-tw/regional-publicationtw/category/37-ccwc_report). Each record included the species name, survey site (including the longitude and latitude), place name, survey date, and data source.
Using citizen science data can be problematic because of inconsistencies in the site names, and boundaries can exist (Robinson et al., 2017). For this reason, we used Google Maps 6.5 (https://www.google.com/maps) to verify the coordinates of the survey sites that significantly deviated from a place name (Hu et al., 2017), with the central point of a named area taken to represent the geographical coordinates. To estimate the likelihood of errors, we input data for 30% of the survey sites into Google Earth 6.0, and the accuracy exceeded 90%.
For each species, duplicate records with the same longitudes and latitudes were removed. The model (bootstrapping procedure) also required the exclusion of species with less than 20 occurrence records. These procedures reduced our dataset to 11,485 records for 80 species from four functional groups (44 shorebird, 17 duck, 9 heron, and 10 gull species) (Supplementary Table S1).
2.3 Data Analysis
2.3.1 Natural Landscape Index
To analyze species’ dependence on coastal natural landscapes, an NL index was first created. We used the land cover data along the Yellow and Bohai Seas, China, to develop the NL index. This dataset was obtained from the Yantai Institute of Coastal Zone Research, Chinese Academy of Sciences, with resolution of 100 × 100 m. Land types were divided into 15 classes: paddy, dryland, forest, grassland, construction land, canal, lake, reservoir, bottomland, tidal flat, salt pan, aquaculture, unused, estuarine water, and estuarine delta.
Firstly, for each raster cell, we defined canal, lake, bottomland, tidal flat, estuarine water, and estuarine delta as coastal natural landscapes and assigned a cell value of 1; paddy, dryland, reservoir, salt pan, and aquaculture were classified as coastal artificial landscapes and assigned a cell value of 0.5. Construction land was classified as a completely degraded landscape, with a cell value of 0 (Theobald, 2010).
Secondly, we applied an inverse distance-weighted nearest-neighbor approach to quantify the degree of naturalization in a specific cell location (Equation 1). The value was between 0 and 1; the closer the value to 1, the higher the degree of naturalization of the raster cell.
Ij represents the degree of naturalization at a specific cell location at raster scale j. Pc is the cell value at the central location and Pi is the cell value at the neighboring cells. In our study, j = 1 represents a raster scale of 0.3 km, which indicated that the cell at the central location was encompassed by 81 adjacent cells (n = 81). The remaining raster scales were set to 0.9, 2.7, 8.1, and 20.0 km (Theobald, 2010).
Finally, we calculated the mean Ij of the five distance scales as the NL index (Equation 2). If the central cell and the neighboring cells were all natural landscapes, the central cell was given an NL value of 1. If the central cell and the neighboring cells were all completely degraded landscapes, the central cell was given an NL value of 0.
NL is the natural landscape index of each raster and k is the raster scale.
2.3.2 Dependence of Waterbirds on Natural Wetlands
We combined the waterbird survey data and the NL index and used the bootstrapping procedure in Matlab R2014a to quantify species’ dependence on coastal natural wetlands. This bootstrapping procedure can reduce the sampling error in the citizen science data by comparing the mean NL values of the areas occupied by target species with randomly selected background records in a buffer area to the occurrence records of the target species.
For each target species, we: 1) used spatial analysis in ArcGIS 10.5 to extract the corresponding NL index for each occurrence record; 2) selected the coordinate locations of the above occurrences as the centers of the circles, with each circle having a 10-km radius to create buffers (this buffer range can include most occurrence records of other species); and 3) selected all species in the same functional group within the buffer area and extracted all NL values of the corresponding occurrence records for these species. The sampling frequency was 1,000 times, and the sample size was the same as the number of occurrence records of the target species.
As a last step, for each sample selected, we calculated the average NL index and acquired 1,000 averages. We ranked the 1,000 averages from lowest to highest and compared the real average NL index of the target species with the 1,000 averages. If the real mean NL value was less than the 25th mean value of the 1,000 averages, then the species was considered significantly dependent on coastal artificial wetlands. However, if the real mean NL value was more than the 975th mean, then the species was considered significantly dependent on coastal natural wetlands. Otherwise, there was no significant dependence.
The flowchart of the calculation method for the bootstrapping procedure can be found in Figure 2.
Figure 2
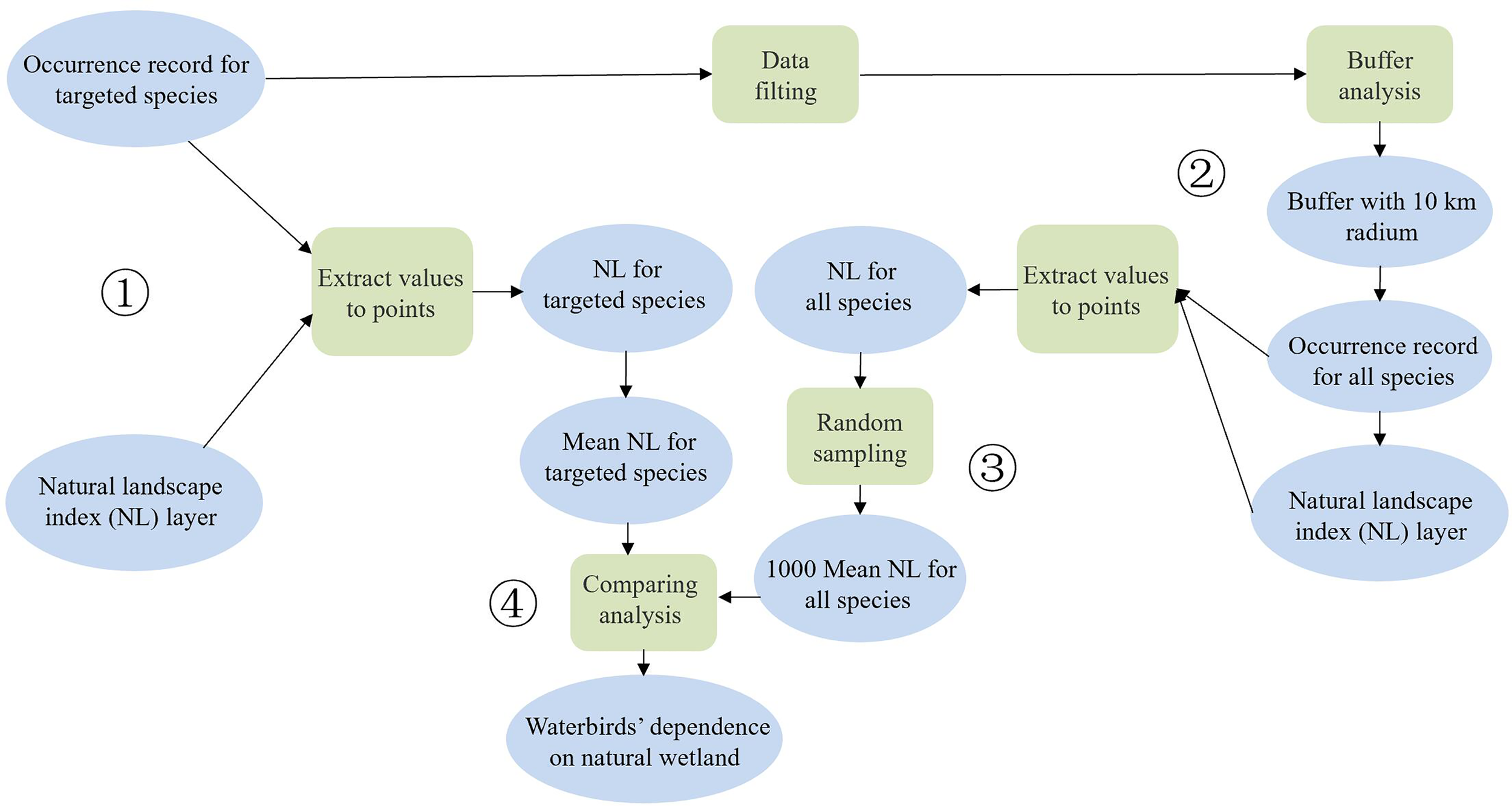
Flowchart demonstrating the calculation method for the bootstrapping procedure.
We calculated the standardized effect size (SES) to quantify the extent how the average NL value of a target species lies above or below the average NL value of the 1,000 samples (Cooke et al., 2019). The higher the SES, the higher the species’ dependence on natural wetlands (Equation 3).
,, and are the average NL index of the target species, the average NL index, and the standard deviation of the 1,000 samples, respectively.
Similar to the calculation process at the species level, we compared the degree of species’ dependence on coastal natural wetlands in different functional groups. For example, for the functional group of shorebirds, we: 1) calculated the average NL index of all the occurrence records of 44 shorebird species to represent the NL index for the shorebird group; 2) selected the coordinate locations of the above occurrence records as the centers of the circles, with each circle having a 10-km radius to create buffers; and 3) selected all species with four functional groups within the buffer area and extracted all NL values of the corresponding occurrence records for these species. Repeating this 1,000 times and the sample size was the same as the occurrence records of 44 shorebird species. The subsequent SES calculation follows Equation 3.
2.3.3 Variation in Species’ Dependence on Coastal Natural Wetlands
To examine the differences in species’ dependence on natural wetlands, we conducted a correlation analysis.
2.3.4 Threatened Status
Threatened species are vulnerable to coastal wetland loss (Clemens et al., 2016; Piersma et al., 2016), which suggests that these species have a low probability of using converted habitats. We hypothesized that threatened species have high dependence on coastal natural wetlands. Threatened status was determined using the IUCN Red List (https://www.iucnredlist.org/; IUCN, 2020). The species identified as critically endangered, endangered, vulnerable, or near threatened, were considered as threatened species, and those species identified as of least concern was considered as non-threatened species (Jackson et al., 2020).
2.3.5 Dispersal Ability
Species with high dispersal ability could better adapt to coastal wetland conversion (Newbold et al., 2015); therefore, these species would have low dependence on natural wetlands. We used their mean wing length (in millimeters) divided by the cube root of their mean mass (in grams) to represent the dispersal ability of the species. The mean wing length of a species can be acquired using the Encyclopedia of Birds in China (Zhao, 2001), whereas the mean mass can be obtained from the Handbook of the Birds of the World (Del Hoyo et al., 1996).
2.3.6 Mean Body Mass
Larger species have low forage frequency in coastal wetlands than smaller species (Jackson et al., 2020). Consequently, we predicted that a relationship could exist between body mass and the species’ dependence on natural wetlands.
Details of the threatened status, dispersal ability, and mean body mass of the target species can be found in Supplementary Table S1. All analyses were conducted using non-parametric tests in SPSS 22.0. Before analysis, for all dispersal ability variables and mean body mass, we used the value of each species divided by the maximum value within the same functional group to exclude differences between groups.
2.3.7 Conservation Status Assessment
Species with significant dependence on coastal natural wetlands in this study were defined as priority conservation targets. Data on the conservation status of these species were retrieved according to the National Protected Species (NPS) List of Key Protected Wildlife of the Wildlife Protection Law of China (Ministry of Forestry in the People’s Republic of China, 2021), including NPS classes I and II. Species in NPS class I or II were assigned as protected and others as not protected (Hu et al., 2017). We recommended which species need to be protected firstly based on the difference in species’ dependence on coastal natural landscapes.
3 Results
3.1 Species’ Dependence on Coastal Natural Landscapes
Of the 80 species, 27 showed significant dependence on coastal natural wetlands, 3 species with significant dependence on coastal artificial wetlands, and 50 species with no significant dependence (Table 1). For shorebirds, 16 species showed significant dependence on coastal natural wetlands [including the spoon-billed sandpiper (Calidris pygmaea), great knot (Calidris tenuirostris), and Nordmann’s greenshank (Tringa guttifer), among others], 1 species showed significant dependence on coastal artificial wetlands, and 27 species had no significant dependence. For ducks, 3 species showed significant dependence on coastal natural wetlands [including Gadwall (Anas strepera), Eurasian teal (Anas crecca), and the common pochard (Aythya ferina)], 1 species with significant dependence on coastal artificial wetlands, and 13 species had no significant dependence. For herons, 4 species showed significant dependence on coastal natural wetlands [cattle egret (Bubulcus ibis), Eurasian spoonbill (Platalea leucorodia), black-faced spoonbill (Platalea minor), and the Chinese egret (Egretta eulophotes)], and 5 species had no significant dependence. For gulls, 4 species showed significant dependence on coastal natural wetlands [little tern (Sterna albifrons), black-tailed gull (Larus crassirostris), Saunders’s gull (Larus saundersi), and relict gull (Larus relictus)], 1 species with significant dependence on coastal artificial wetland, and 5 species had no significant dependence.
Table 1
| Functional groups | Scientific name | Common name | SES | Conservation status |
|---|---|---|---|---|
| Shorebirds | Limosa lapponica a | Bar-tailed godwit | 2.235 | Not listed |
| Calidris tenuirostris a | Great knot | 2.046 | Class II | |
| Tringa guttifer a | Nordmann’s greenshank | 3.739 | Class I | |
| Calidris temminckii a | Temminck’s stint | 2.575 | Not listed | |
| Crocethia alba a | Sanderling | 2.171 | Not listed | |
| Xenus cinereus a | Terek sandpiper | 3.680 | Not listed | |
| Calidris canutus a | Red knot | 2.086 | Not listed | |
| Arenaria interpres a | Ruddy turnstone | 2.027 | Class II | |
| Calidris pygmaea a | Spoon-billed sandpiper | 4.827 | Class I | |
| Calidris alpina a | Dunlin | 2.273 | Not listed | |
| Calidris ferruginea a | Curlew sandpiper | 2.060 | Not listed | |
| Haematopus ostralegus a | Eurasian oystercatcher | 3.604 | Not listed | |
| Charadrius mongolus a | Lesser sand plover | 2.934 | Not listed | |
| Pluvialis squatarola a | Grey plover | 3.968 | Not listed | |
| Numenius phaeopus a | Whimbrel | 3.278 | Not listed | |
| Charadrius leschenaultii a | Greater sand plover | 1.981 | Not listed | |
| Recurvirostra avosetta b | Pied avocet | −2.530 | Not listed | |
| Gallinago gallinago | Common snipe | 1.390 | Not listed | |
| Limosa limosa | Black-tailed godwit | −0.837 | Not listed | |
| Numenius madagascariensis | Far Eastern curlew | 1.828 | Class II | |
| Tringa stagnatilis | Marsh sandpiper | −0.500 | Not listed | |
| Tringa nebularia | Common greenshank | 1.917 | Not listed | |
| Tringa ochropus | Green sandpiper | −0.128 | Not listed | |
| Scolopax rusticola | Eurasian woodcock | −0.263 | Not listed | |
| Numenius minutus | Little curlew | −0.036 | Class II | |
| Actitis hypoleucos | Common sandpiper | 0.604 | Not listed | |
| Limnodromus semipalmatus | Asian dowitcher | −0.812 | Class II | |
| Tringa erythropus | Spotted redshank | −0.192 | Not listed | |
| Tringa totanus | Common redshank | 0.372 | Not listed | |
| Phalaropus lobatus | Red-necked phalarope | 0.317 | Not listed | |
| Calidris acuminata | Sharp-tailed sandpiper | 0.306 | Not listed | |
| Limicola falcinellus | Broad-billed sandpiper | −0.939 | Class II | |
| Tringa glareola | Wood sandpiper | −0.142 | Not listed | |
| Philomachus pugnax | Ruff | −0.975 | Not listed | |
| Glareola maldivarum | Oriental pratincole | −0.767 | Not listed | |
| Himantopus himantopus | Black-winged stilt | −0.448 | Not listed | |
| Charadrius alexandrinus | Kentish plover | 0.964 | Not listed | |
| Calidris ruficollis | Red-necked stint | 0.435 | Not listed | |
| Tringa brevipes | Gray-tailed tattler | 0.286 | Not listed | |
| Pluvialis fulva | Pacific golden plover | −1.057 | Not listed | |
| Charadrius dubius | Little ringed plover | 0.149 | Not listed | |
| Numenius arquata | Eurasian curlew | 0.298 | Class II | |
| Calidris subminuta | Long-toed stint | −0.426 | Not listed | |
| Charadrius placidus | Long-billed plover | −1.860 | Not listed | |
| Ducks | Anas strepera a | Gadwall | 2.498 | Not listed |
| Anas crecca a | Eurasian teal | 2.489 | Not listed | |
| Aythya ferina a | Common pochard | 2.727 | Not listed | |
| Bucephala clangula b | Common goldeneye | −2.021 | Not listed | |
| Tadorna ferruginea | Ruddy shelduck | −0.764 | Not listed | |
| Tadorna tadorna | Common shelduck | −0.528 | Not listed | |
| Anas falcata | Falcated duck | 0.450 | Not listed | |
| Anas penelope | Eurasian wigeon | 1.198 | Not listed | |
| Anas platyrhynchos | Mallard | 0.513 | Not listed | |
| Anas poecilorhyncha | Spot-billed duck | 0.513 | Not listed | |
| Anas clypeata | Northern shoveler | 1.460 | Not listed | |
| Anas acuta | Northern pintail | 0.329 | Not listed | |
| Anas querquedula | Garganey | 0.621 | Not listed | |
| Aythya fuligula | Tufted duck | 1.025 | Not listed | |
| Mergellus albellus | Smew | 0.090 | Class II | |
| Mergus merganser | Common merganser | 0.106 | Not listed | |
| Mergus serrator | Red-breasted merganser | −1.053 | Not listed | |
| Herons | Bubulcus ibis a | Cattle egret | 2.294 | Not listed |
| Platalea leucorodia a | Eurasian spoonbill | 2.820 | Class II | |
| Platalea minor a | Black-faced spoonbill | 2.451 | Class I | |
| Egretta eulophotes a | Chinese egret | 2.471 | Class I | |
| Ardea cinerea | Grey heron | 1.663 | Not listed | |
| Ardea purpurea | Purple heron | 0.723 | Not listed | |
| Casmerodius albus | Great egret | 1.826 | Not listed | |
| Mesophoyx intermedia | Intermediate egret | 1.913 | Not listed | |
| Ardeola bacchus | Chinese pond heron | −1.059 | Not listed | |
| Gulls | Sterna albifrons a | Little tern | 1.988 | Not listed |
| Larus crassirostris a | Black-tailed gull | 5.556 | Not listed | |
| Larus saundersi a | Saunders’s gull | 2.123 | Class I | |
| Larus relictus a | Relict gull | 2.193 | Class I | |
| Larus ridibundus b | Black-headed gull | −3.205 | Not listed | |
| Chlidonias leucopterus | White-winged tern | −1.032 | Not listed | |
| Sterna caspia | Caspian tern | 0.403 | Not listed | |
| Sterna hirundo | Common tern | 0.133 | Not listed | |
| Chlidonias hybrida | Whiskered tern | −0.097 | Not listed | |
| Larus argentatus | Herring gull | 1.295 | Not listed |
Dependence of waterbird species on coastal natural wetlands, standardized effect size (SES), and their conservation status.
Species in National Protected Species class I or II were assigned as protected; others, “Not listed,” as not protected.
Significant dependence on natural wetland.
Significant dependence on artificial wetland.
The functional groups of shorebirds (SES = 4.37) and herons (SES = 2.56) had higher SES values than the functional groups of ducks (SES = −0.02) and gulls (SES = −3.22). These results revealed that these shorebirds and herons are more dependent on coastal natural landscapes than the ducks and gulls (Figure 3).
Figure 3
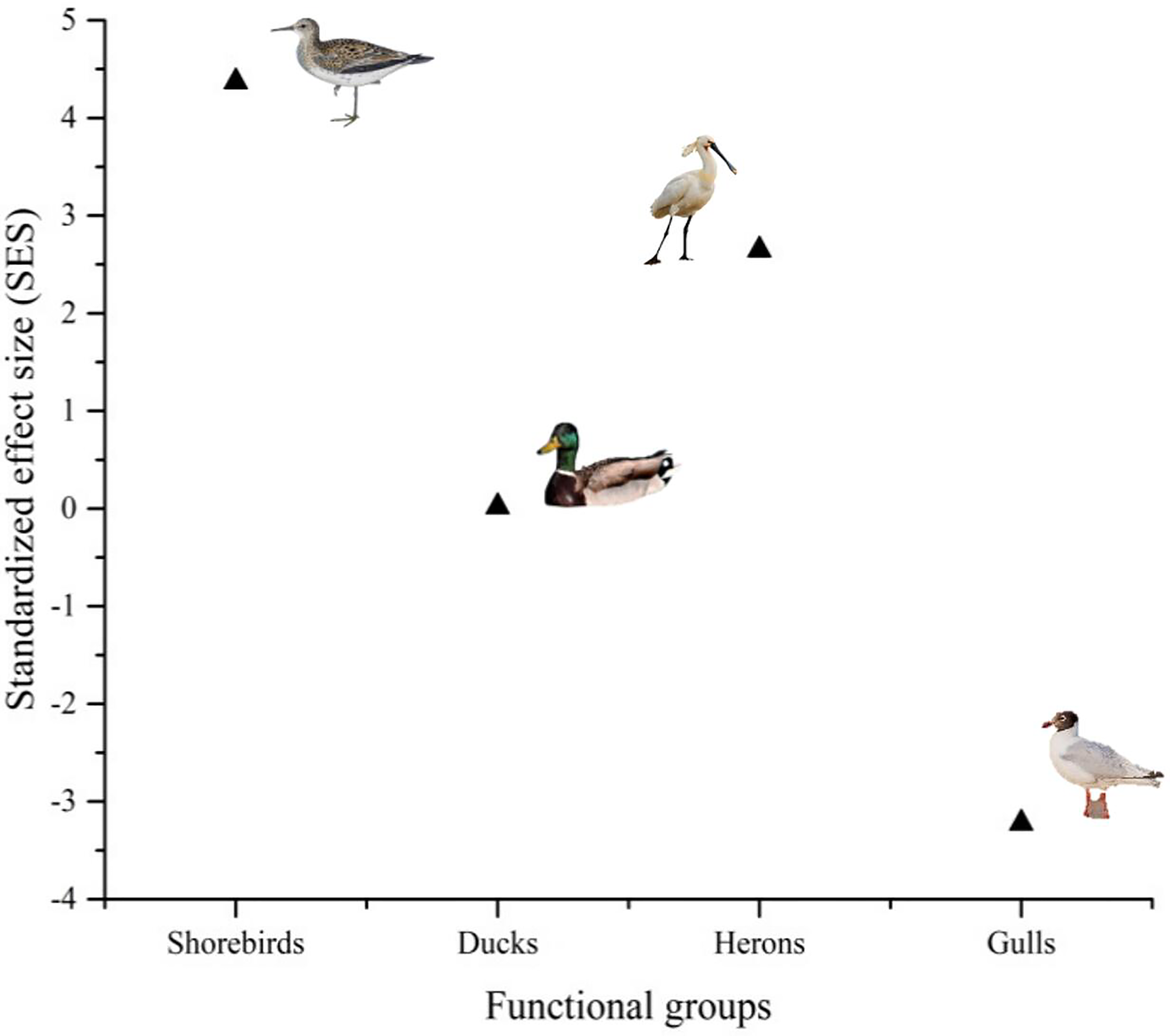
Dependence of waterbird species on coastal natural wetlands and the standardized effect size (SES) of the four functional groups.
3.2 Correlation Between Species’ Dependence on Coastal Natural Wetlands and Variables (Threatened Status, Mean Body Mass, and Dispersal Ability)
The variable that explained species’ dependence on coastal natural wetlands was significant for threatened status (N = 80, r = 0.284*, p < 0.05). Threatened species were significantly dependent on coastal natural wetlands than were non-threatened species (t = 2.613, df = 78, p < 0.05) (Figure 4). The mean body mass and dispersal ability variables were not associated with species’ dependence on coastal natural wetlands (N = 80, r = 0.037, p > 0.05; N = 80, r = 0.004, p > 0.05).
Figure 4
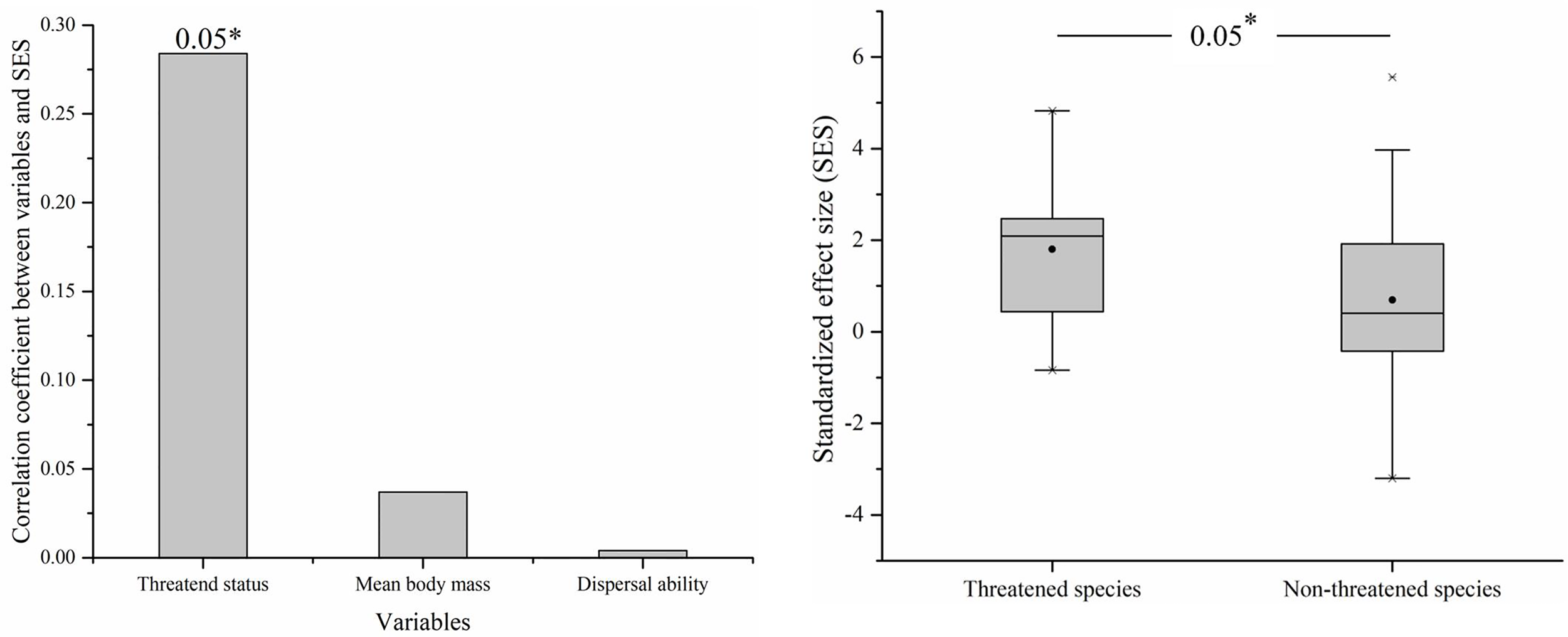
Correlation between standardized effect size (SES) and variables (threatened species, mean body mass, and dispersal ability) (left) and the SES between threatened species and non-threatened species (right). Black circles represent mean values, horizontal bars within boxes represent median values, the upper and lower limits of boxes represent the maxima and minima, respectively, and whiskers represent 1% and 99%. *means significant at the 0.05 level.
3.3 Conservation Status Analysis
Of the 27 species that showed significant dependence on coastal natural wetlands, only 9 were listed as classes I and II according to the NPS List of Key Protected Wildlife of the Wildlife Protection Law of China (Supplementary Table S1 and Table 1). For some unprotected threatened species, such as the common pochard (A. ferina, vulnerable), bar-tailed godwit (Limosa lapponica, near threatened), red knot (Calidris canutus, near threatened), curlew sandpiper (Calidris ferruginea, near threatened), Eurasian oystercatcher (Haematopus ostralegus, near threatened), with significant dependence on natural wetlands, we recommend to firstly list as NPS class I or II.
4 Discussion
4.1 Dependence on Coastal Natural Wetlands
Our results revealed that 33.75% of the species we selected were significantly associated with natural wetlands, which indicated that these species were more susceptible to natural wetland loss. These results are consistent with those of Studds et al. (2017), who found that the populations of seven shorebird species that are highly dependent on natural wetlands in the Yellow Sea ecoregion underwent a rapid decline. For example, the populations of great knot (C. tenuirostris), a global endangered species, and red knot (C. canutus), a global near threatened species with high sensitivity to natural landscapes, declined by up to 5% and 4%, respectively, between 1993 and 2012 because of the rapid loss of tidal flats. It is necessary to conserve these species and to prevent further population decline. We also found that shorebirds and herons had relatively higher dependence on coastal wetlands than ducks and gulls. It could be that most shorebirds and herons along the EAAF prefer to forage on invertebrate communities and rely on tidal flats (Jackson et al., 2020). Conversely, most ducks are generalists and occur inland; their wider ecological niche enables them to exploit more diverse habitats (Ma et al., 2010).
4.2 Correlation Between Species’ Dependence on Coastal Natural Wetlands and Threatened Status, Mean Body Mass, and Dispersal Ability
Threatened status was an important predictor of species’ dependence on natural wetlands. Previous studies have indicated that threatened shorebird species have a lower foraging frequency in artificial wetlands compared with non-threatened species (Jackson et al., 2020). This distinction may be an important reason for the threatened shorebirds having significantly higher dependence on natural wetlands than non-threatened species. In addition, threatened species have greater habitat requirements; therefore, they benefit from less habitat disturbance than the non-threatened species (Peng et al., 2017). The degree of naturalization for each raster cell we created accounted for the surrounding disturbance of non-natural wetlands and construction lands. This may explain why threatened species more strongly prefer natural wetlands than non-threatened species.
Several variables that were shown to be tightly associated with species’ response to habitat loss in other studies (Wang et al., 2015; Todd et al., 2017) were only poorly associated with species’ dependence on natural wetlands in our study. For example, species with lower dispersal ability could be vulnerable to habitat disturbance or have high dependence on natural wetlands. However, our results showed that dispersal ability had a weak positive relationship with species’ dependence on natural wetlands. This difference may have been due to our selection of all migratory species; therefore, dispersal ability does not significantly vary between them. We also found that the mean body mass had low explanatory ability for dependence variation between all species. A previous study indicated that larger shorebird species prefer to forage in coastal wetlands than smaller species (Jackson et al., 2020). However, our study included species from four functional groups, and the variation of the selected forage sites could have led to the lack of a strong correlation between the body size of species and habitat dependence. For example, ducks had lower dependence on coastal natural wetlands than shorebirds.
4.3 Data Limitations
Although we collected several occurrence records for migratory waterbirds in the coastal wetlands of the Yellow and Bohai Seas, China, from a wide range of sources, data limitations still warrant consideration. For example, unequal observation efforts due to the uneven distribution of birdwatchers across the survey region can affect the assessment results (Li et al., 2013; Ma et al., 2012). The continued collection of bird data to augment the survey sites for species along China’s coasts at an even greater scale is necessary and can lead to a better understanding of the species’ dependence on natural landscapes.
4.4 Conservation and Management Implications
Of the 27 species with significant dependence on coastal natural wetlands, more than half were not listed as national protected species according to the NPS list (Ministry of Forestry in the People’s Republic of China, 2021). Some priority conservation actions need to be taken, which are outlined as follows. Common conservation plans are prioritizing species and habitats (Game et al., 2012).
Five unprotected threatened species with significant dependence on coastal natural landscapes need to be listed as national protected species: The NPS list is updated every 5 years. Six of 80 species were updated as national protected species according to the Wildlife Protection Law of China in 2021. This action indicated that the national government pays attention to species conservation. We suggest five unprotected threatened species—the common pochard (A. ferina, vulnerable), bar-tailed godwit (L. lapponica, near threatened), red knot (C. canutus, near threatened), curlew sandpiper (C. ferruginea, near threatened), and Eurasian oystercatcher (H. ostralegus, near threatened)—that can be listed as national protected species when this list is updated in the future.
Take advantage of the opportunities of protected area system reform and nomination of the coasts of the Yellow Sea–Bohai Gulf of China as World Natural Heritage to further expand the coverage of protected areas and fill the gap in terms of the protection of key habitats: The General Office of the CPC Central Committee and the General Office of the State Council jointly issued the Guidelines for Establishing a Natural Protected Area System Focusing on National Parks in June 2019, proposing to build a unified management mechanism on natural protected areas based on different categories and levels and to develop a scheme on the spatial layout of national parks on a scientific basis. In July 2019, Migratory Bird Sanctuaries along the Coast of the Yellow Sea–Bohai Gulf of China (Phase I) was inscribed into the World Heritage List, while phase II of the project is expected to be inscribed into the World Heritage List at the 47th Session of UNESCO World Heritage Convention to be held in 2023. Some important coastal wetlands, including Nandagang, Luannan Coast, Xiehu Qilihai, Dachaoping Beidaihe, Shihenandao, and Changshan Island, should be created as new protected areas. This will make a significant contribution to the conservation of coastal wetlands and migratory waterbirds along EAAF.
Returning marine aquaculture ponds to mudflats and invasive species control to increase the area of estuarine wetlands: The coastal wetlands of the Yellow and Bohai Seas include important estuarine wetlands: Yalujiang Estuary, Liaohe Estuary, and Yellow River Delta. These estuarine wetlands provide important feeding habitats for migratory waterbirds (Jackson et al., 2020). Natural wetland loss is caused by the artificial wetland landscape aquaculture pond occupation in Yalujiang Estuary and Liaohe Estuary (Lei et al., 2017; Ma et al., 2019) and the invasion of the invasive species Spartina alterniflora in the Yellow River Delta (Jackson et al., 2021). Converting aquaculture ponds to mudflats and recovering mudflat wetlands and their ecological functions can be done with hydrology regulation, pollution control, and selection of salt-tolerant plants. Eliminating S. alterniflora and increasing natural wetland areas can be achieved by physical castration and chemical control (Lei et al., 2017). The above actions will increase the distribution range of waterbird species, especially for species with significant dependence on coastal natural landscapes.
Exploring the dependence of waterbirds on natural wetlands at the EAAF scale and providing site-based conservation recommendations in different regions. Waterbirds have diverse habitat uses between breeding areas, stopover sites, and non-breeding areas, and this will cause different preferences on natural landscapes in different regions. Exploring the dependence of waterbirds on natural wetlands at the EAAF scale is important in the future. It is better to provide site-based conservation recommendation in different regions according to the different dependences of waterbirds on natural wetlands.
Creating an EAAF migratory route protected network and building a platform for public participation: The populations of migratory waterbirds are dependent on what happens elsewhere as well, not only in China. Waterbird and habitat conservation needs the participation of more countries along the EAAF migratory route. Creating a protected network around the EAAF and including members from protected areas, wetland management organizations, research institutes, non-governmental organizations (NGOs), and the public to share protection experiences and carry out publicity and education to promote wetland conservation, for example, holding an international birding festival or an international forum of wetland and waterbird conservation to mobilize as many people as possible to participate in waterbird conservation.
Funding
This research was jointly supported by the National Natural Science Foundation of China (42101105) and the Earth Big Data Scientific Engineering Project of the Chinese Academy of Sciences (grant no. XDA19020305).
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Statements
Data availability statement
The datasets presented in this study can be found in online repositories. The names of the repository/repositories and accession number(s) can be found below. The records of species supporting this study can be acquired from the websites of eBird in the USA (https://ebird.org/home), the Global Biodiversity Information Network—Chinese Academy of Sciences (GBIF-CAS) (http://www.gbifchina.org/), and from BirdReport in China (http://www.birdreport.cn/).
Ethics statement
Ethical review and approval was not required for the animal study because this study only collected citizen science data from websites and reports of birds.
Author contributions
XY, SX, and YL: conceptualization, funding acquisition, supervision, and writing—review and editing. HD: data curation, writing—original draft, formal analysis, and visualization. All authors contributed to the article and approved the submitted version.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fmars.2022.888535/full#supplementary-material
References
1
Bai Q. Q. Chen J. Z. Chen Z. H. Dong G. T. Dong J. T. Dong W. X. et al . (2015). Identification of Coastal Wetlands of International Importance for Waterbirds: A Review of China Coastal Waterbird Surveys 2005–2013. Avian Res.6, 1–16. doi: 10.1186/s40657-015-0021-2
2
Broms K. M. Johnson D. S. Altwegg R. Conquest L. L. (2014). Spatial Occupancy Models Applied to Atlas Data Show Southern Ground Hornbills Strongly Depend on Protected Areas. Ecol. Appl.24, 363–374. doi: 10.1890/12-2151.1
3
Clemens R. S. Rogers D. I. Hansen B. D. Gosbell K. Minton C. D. T. Straw P. et al . (2016). Continental-Scale Decreases in Shorebird Populations in Australia. EmuS116, 119–135. doi: 10.1071/MU15056
4
Cooke R. S. C. Bates A. E. Eigenbrod F. (2019). Global Trade-Offs of Functional Redundancy and Functional Dispersion for Birds and Mammals. Global Ecol. Biogeogr.28, 484–495. doi: 10.1111/geb.12869
5
Del Hoyo J. Elliott A. Sargatal J. (1996). “Handbook of the Birds of the World,” in Hoatzinto Auks, Volume 3 (Barcelona: Lynx Edicions).
6
Duan H. L. Xia S. L. Hou X. Y. Liu Y. Yu X. B. (2019). Conservation Planning Following Reclamation of Intertidal Areas Throughout the Yellow and Bohai Seas, China. Biodivers. Conserv.28, 3787–3801. doi: 10.1007/s10531-019-01851-3
7
Game E. T. Kareiva P. Possingham H. P. (2012). Six Common Mistakes in Conservation Priority Setting. Conserv. Biol.27, 480–485. doi: 10.1111/cobi.12051
8
Guo L. Xiao N. B. Luo Z. L. Liu D. M. Zhao Z. P. Guan X. et al . (2021). Identifying Conservation Priority Areas for Gymnosperm Species Under Climate Changes in China. Biol. Conserv.253, 108914. doi: 10.1016/j.biocon.2020.108914
9
Harte J. Ostling A. Green J. L. Kinzig A. (2004). Biodiversity Conservation: Climate Change and Extinction Risk. Nature430, 145–148. doi: 10.1038/nature02718
10
Huang Z. Bai Y. Alatalo J. M. Yang Z. P. (2020). Mapping Biodiversity Conservation Priorities for Protected Areas: A Case Study in Xishuangbanna Tropical Area, China. Biol. Conserv.249, 108741. doi: 10.1016/j.biocon.2020.108741
11
Hua N. Tan K. Chen Y. Ma Z. J. (2015). Key Research Issues Concerning the Conservation of Migratory Shorebirds in the Yellow Sea Region. Bird. Conserv. Int.25, 38–52. doi: 10.1017/S0959270914000380
12
Hu R. C. Gu Y. Y. Lu M. Lu Z. Wei M. Zhong J. (2020). Shifts in Bird Ranges and Conservation Priorities in China Under Climate Change. PloS One15, e0240225. doi: 10.1371/journal.pone.0240225
13
Hu R. Wen C. Gu Y. Wang H. Gu L. Shi X. Y. et al . (2017). A Bird’ s View of New Conservation Hotspots in China. Biol. Conserv.211, 47–55. doi: 10.1016/j.biocon.2017.03.033
14
IUCN . (2020). The IUCN Red List of Threatened Species. Version 2020-2. Available at: https://www.iucnredlist.org (Accessed 20 July 2021).
15
Jackson M. V. Carrasco L. R. Choi C. Y. Li J. Ma Z. J. Melville D. S. et al . (2019). Multiple Habitat Use by Declining Migratory Birds Necessitates Joined-Up Conservation. Ecol. Evol.9, 2505–2515. doi: 10.1002/ece3.4895
16
Jackson M. V. Choi C. Y. Amano T. Estrella S. M. Lei W. P. Moores N. et al . (2020). Navigating Coasts of Concrete: Pervasive Use of Artifcial Habitats by Shorebirds in the Asia-Pacifc. Biol. Conserv.247, 108591. doi: 10.1016/j.biocon.2020.108591
17
Jackson M. V. Fuller R. A. Gan X. Li J. Mao D. H. Melville D. S. (2021). Dual Threat of Tidal Flat Loss and Invasive Spartina Alterniflora Endanger Important Shorebird Habitat in Coastal Mainland China. J. Environ. Manage.278, 111549. doi: 10.1016/j.jenvman.2020.111549
18
Keinath D. A. Doak D. F. Hodges K. E. Prugh L. R. Fagan W. Sekercioglu C. H. et al . (2016). A Global Analysis of Traits Predicting Species Sensitivity to Habitat Fragmentation. Global Ecol. Biogeogr.26, 115–127. doi: 10.1111/geb.12509
19
Lei G. C. Zhang Z. W. Yu X. B. Zhang M. X. (2017). Blueprint of Coastal Wetland Conservation and Management in China (Beijing: Science Press).
20
Lei W. P. Masero J. A. Piersma T. Zhu B. R. Yang H. Y. Zhang Z. W. (2018). Alternative Habitat: The Importance of the Nanpu Saltpans for Migratory Waterbirds in the Chinese Yellow Sea. Bird. Conserv. Int.28, 549–566. doi: 10.1017/S0959270917000508
21
Li X. Y. Liang L. Gong P. Liu Y. Liang F. F. (2013). Bird Watching in China Reveals Bird Distribution Changes. Sci. Bull.58, 649656. doi: 10.1007/s11434-012-5458-7
22
Li Y. F. Mao D. H. Wang Z. M. Wang X. Tan X. Y. Jia M. M. et al . (2021). Identifying Variable Changes in Wetlands and Their Anthropogenic Threats Bordering the Yellow Sea for Water Bird Conservation. Glob. Ecol. Conserv.27, e01613. doi: 10.1016/j.gecco.2021.e01613
23
Long A. M. Pierce B. L. Anderson A. D. Skow K. L. Lopea R. R. (2019). Integrating Citizen Science and Remotely Sensed Data to Help Inform Time-Sensitive Policy Decisions for Species of Conservation Concern. Biol. Conserv.237, 463–469. doi: 10.1016/j.biocon.2019.07.025
24
Ma Z. J. Cai Y. T. Li B. Chen J. K. (2010). Managing Wetland Habitats for Waterbirds: An International Perspective. Wetlands30, 15–27. doi: 10.1007/s13157-009-0001-6
25
Ma Z. J. Cheng Y. Wang J. Fu X. (2012). The Rapid Development of Birdwatching in Mainland China: A New Force for Bird Study and Conservation. Bird. Conserv. Int.23, 259–269. doi: 10.1017/S0959270912000378
26
Ma T. T. Li X. W. Bai J. H. Cui B. S. (2019). Habitat Modification in Relation to Coastal Reclamation and Its Impacts on Waterbirds Along China’s Coast. Glob. Ecol. Conserv.17, e00585. doi: 10.1016/j.gecco.2019.e00585
27
Melville D. S. Chen Y. Ma Z. J. (2016). Shorebirds Along the Yellow Sea Coast of China Face an Uncertain Future-A Review of Threats. Emu116, 100–110. doi: 10.1071/MU15045
28
Meretsky V. L. Maguire L. A. Davis F. W. Stoms D. M. Scott J. M. Figg D. et al . (2012). A State-Based National Network for Effective Wildlife Conservation. BioScience62, 970–976. doi: 10.1525/bio.2012.62.11.6
29
Ministry of Forestry in the People’ s Republic of China and Ministry of Agriculture of the People’s Republic of China . (2021). Available at: http://www.forestry.gov.cn/main/3957/20210205/153020834857061.html (Accessed Dec 1, 2021).
30
Murray N. J. Clemens R. S. Phinn S. R. Possingham H. P. Fuller R. A. (2014). Tracking the Rapid Loss of Tidal Wetlands in the Yellow Sea. Front. Ecol. Environ.12, 267e272. doi: 10.1890/130260
31
Murray N. J. Phinn S. R. DeWitt M. Ferrari R. Johnston R. Lyons M. B. et al . (2018). The Global Distribution and Trajectory of Tidal Flats. Nature565, 222–225. doi: 10.1038/s41586-018-0805-8
32
Newbold T. Hudson L. N. Hill S. L. L. Contu S. Lysenko I. Senior R. A. et al . (2015). Global Effects of Land Use on Local Terrestrial Biodiversity. Nature520, 45–50. doi: 10.1038/nature14324
33
Peng H.-B. Anderson G. Q. A. Chang Q. Choi C.-Y. Chowdhury S. U. Clark N. A. et al . (2017). The Intertidal Wetlands of Southern Jiangsu Province, China-Globally Important for Spoon-Billed Sandpipers and Other Threatened Waterbirds, But Facing Multiple Serious Threats. Bird. Conserv. Int.27, 1–18. doi: 10.1017/S0959270917000223
34
Piersma T. Lok T. Chen Y. Hassell C. J. Yang H. Y. Boyle A. et al . (2016). Simultaneous Declines in Summer Survival of Three Shorebird Species Signals a flyway at Risk. J. Appl. Ecol.53, 479–490. doi: 10.1111/1365-2664.12582
35
Robinson O. J. Ruiz-Gutierrez V. Fink D. (2017). Correcting for Bias in Distribution Modelling for Rare Species Using Citizen Science Data. Divers. Distrib.24, 1–13. doi: 10.1111/ddi.12698
36
Studds C. E. Kendall B. E. Murray N. J. Wilson H. B. Rogers D. I. Clemens R. S. et al . (2017). Rapid Population Decline in Migratory Shorebirds Relying on Yellow Sea Tidal Mudflats as Stopover Sites. Nat. Commun.8, 14895. doi: 10.1038/ncomms14895
37
Theobald D. M. (2010). Estimating Natural Landscape Changes From 1992 to 2030 in the Conterminous US. Landscape Ecol.25, 999–1011. doi: 10.1007/s10980-010-9484-z
38
Todd B. D. Nowakowski A. J. Rose J. P. Price S. J. (2017). Species Traits Explaining Sensitivity of Snakes to Human Land Use Estimated From Community Science Data. Biol. Conserv.206, 31–36. doi: 10.1016/j.biocon.2016.12.013
39
Wang Y. P. Thornton D. H. Ge S. Wang Y. Ding P. (2015). Ecological Correlates of Vulnerability to Fragmentation in Forest Birds on Inundated Subtropical Land-Bridge Islands. Biol. Conserv.191, 251–257. doi: 10.1016/j.biocon.2015.06.041
40
Xu Y. J. Si Y. L. Zhang W. Grishchenko Y. M. Prins H. H. T. Gong P. et al . (2019). Species-Dependent Effects of Habitat Degradation in Relation to Seasonal Distribution of Migratory Waterfowl in the East Asian–Australasian Flyway. Landscape Ecol.34, 243–257. doi: 10.1007/s10980-018-00767-7
41
Zhao Z. J. (2001). Ornithology of China (Changchun: Jilin Science and Technology Press).
Summary
Keywords
citizen science data, bootstrapping procedure, conservation and management, dependence on natural wetland, natural landscape index
Citation
Duan H, Yu X, Xia S and Liu Y (2022) Combining Bootstrapping Procedure and Citizen Science Data to Elucidate Waterbirds’ Dependence on Coastal Wetland. Front. Mar. Sci. 9:888535. doi: 10.3389/fmars.2022.888535
Received
03 March 2022
Accepted
21 March 2022
Published
14 April 2022
Volume
9 - 2022
Edited by
Junhong Bai, Beijing Normal University, China
Reviewed by
Yifei Jia, Beijing Forestry University, China; Dehua Mao, Northeast Institute of Geography and Agroecology (CAS), China
Updates
Copyright
© 2022 Duan, Yu, Xia and Liu.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Xiubo Yu, yuxb@igsnrr.ac.cn
This article was submitted to Marine Ecosystem Ecology, a section of the journal Frontiers in Marine Science
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.