Abstract
Objective:
This systematic review aimed to synthesize current clinical evidence from randomized controlled trial (RCT) and meta-analyses on the efficacy and safety of TCM in the treatment of Alzheimer’s Disease (AD).
Methods:
Systematic searches across eight biomedical databases (PubMed, Embase, Web of Science, Cochrane Library, CNKI, Wanfang, VIP, SinoMed) through October 26, 2024 yielded an evidence matrix, which was analyzed through integrated narrative-graphic synthesis.
Results:
Our analysis encompassed 187 studies (141 RCTs and 46 systematic reviews/meta-analyses), demonstrating cyclical publication growth with recent contraction. Study characteristics included sample sizes of 50–100 participants and intervention durations of 4–24 weeks. Interventions included acupuncture, herbal decoctions, and proprietary medicines. Outcomes focused on clinical efficacy, scale scores, TCM syndrome scores, and safety. While TCM demonstrated therapeutic potential, prescription heterogeneity and diagnostic ambiguity constrained specificity. Methodological quality was generally low, with few high-quality systematic reviews or meta-analyses.
Conclusion:
While TCM shows therapeutic potential in Alzheimer’s disease, methodological limitations persist. Subsequent research requires enhanced trial designs with standardized outcome metrics and rigorous bias control protocols.
Highlights
-
This review elucidates the current evidence base for the treatment of Alzheimer’s disease (AD) with Traditional Chinese Medicine (TCM).
-
The clinical studies included generally feature relatively small sample sizes.
-
A majority of these studies inadequately reflect the principles of syndrome differentiation and treatment, which are fundamental to TCM.
-
While TCM appears to hold potential for the treatment of AD, the overall quality of research in this area necessitates substantial improvement.
1 Introduction
Alzheimer’s disease, a progressive neurodegenerative pathology, manifests as multidomain cognitive deterioration with core manifestations in mnestic deficits, linguistic processing dysfunction, and executive capacity decline (1, 2). Among neurodegenerative dementias, AD represents a substantial proportion (3). Worldwide, AD prevalence is estimated at 55 million cases, projected to reach 139 million by 2050 (4). An estimated 69 million and 315 million individuals have preclinical and prodromal AD, respectively (5, 6). This large patient population severely burdens socioeconomic systems, family structures, and mental health, establishing AD as a critical global public health challenge (7). Despite limited diagnostic and therapeutic options for AD, RCTs employing TCM interventions demonstrate promising clinical utility with enhanced safety. However, comprehensive evidence validating these methods remains inadequate (8, 9). The evidence map, an emerging methodology, provides a systematic approach to evaluate TCM’s current evidence base, therapeutic potential, and limitations in AD diagnosis and treatment (10). Evidence mapping systematically collates and synthesizes domain-specific literature, enabling comprehensive analysis of research progress and knowledge gaps. This approach provides an empirical foundation for guiding future high-quality studies, significantly advancing TCM applications in AD diagnosis and treatment.
2 Materials and methods
2.1 Search strategy
A systematic search was conducted across eight databases, including CNKI, WanFang Data, VIP, SinoMed, PubMed, Embase, Web of Science, and Cochrane Library, for literature related to the treatment of dementia with TCM. The search period ranged from the inception of the databases to October 26, 2024. Chinese Search Terms: (“dementia” OR “Alzheimer’s Disease” OR “Alzheimer” OR “AD”) AND (“Traditional Chinese Medicine” OR “Chinese herbal medicine” OR “acupuncture” OR “needle puncture” OR “integrated Chinese and Western medicine” OR “granules” OR “powders”OR “pills” OR “tablets” OR “decoctions” OR “injections” OR “capsules” OR “preparations” OR “oral liquids” OR “injections” OR “tuina” OR “prepared Chinese medicine” OR “external therapy” OR “syndrome typing”) AND (“randomized controlled trials” OR “RCT” OR “systematic analysis” OR “meta”). English Search Terms: (“dementia” OR “Alzheimer” OR “Alzheimer’s Disease” OR “AD”) AND (“Chinese medicine” OR “acupuncture” OR “Integrative Medicine” OR “granules” OR “powder” OR “pill” OR “tablet” OR “decoction” OR “injection” OR “capsule” OR “massage” OR “syndrome”) AND (“randomized controlled trials” OR “RCT” OR “meta-analysis” OR “systematic reviews”).
2.2 Inclusion criteria
Eligible studies required a confirmed Alzheimer’s disease diagnosis and were limited to RCTs or meta-analyses/systematic reviews incorporating RCTs. Interventions encompassed Traditional Chinese Medicine (TCM) modalities, including oral/topical herbal compounds (all formulations), proprietary medicines, external therapies (e.g., acupuncture, cupping, moxibustion), or integrative Chinese-Western approaches. Comparator groups involved Western pharmacotherapy, placebo, or other relevant controls, with no restrictions imposed on participant demographics (age, gender), disease duration, comorbidities, or adjunct therapies.
2.3 Exclusion criteria
Exclusion criteria comprised preclinical models (animal/cell studies), non-empirical research outputs (case reports, reviews, conference abstracts, expert opinions, patents, technological achievements, media articles), studies investigating vascular dementia or irrelevant pathologies, inaccessible/incomplete datasets, non-anglophone/sinophone publications, and duplicate reports.
2.4 Literature screening and data extraction
Five investigators independently executed literature screening and data extraction against predefined inclusion criteria, with cross-validated screening outcomes ensuring methodological fidelity. EndNote X9 facilitated automated deduplication. Title/abstract screening excluded non-conforming studies, followed by full-text appraisal refining the corpus through exclusion of non-compliant or duplicate publications. A data extraction form was established, including but not limited to title, authors, publication date, journal of publication, therapeutic parameters (syndrome stratification strategies, sample size, pharmacotherapeutic protocols, duration), and outcome metrics (TCM syndrome scoring, validated psychometric scales, safety surveillance).
2.5 Quality assessment
The methodological quality of the RCTs was assessed using the Cochrane risk of bias tool (11). This tool includes seven domains: (1) random sequence generation; (2) allocation concealment; (3) blinding of participants and personnel; (4) blinding of outcome assessment; (5) incomplete outcome data (attrition bias); (6) selective reporting; (7) other potential sources of bias. Each domain was evaluated as “low risk,” “unclear risk,” or “high risk” based on the information provided in the included studies.
The methodological quality of meta-analyses/systematic reviews was assessed using the AMSTAR 2 tool (12). This tool contains 16 items, with items 2, 4, 7, 9, 11, 13, and 15 being considered key. Each study was rated as “Yes,” “Partially Yes,” or “No” based on the content provided.
The quality assessments were performed by five researchers, and any discrepancies in evaluations were resolved through group discussion, reaching a consensus before finalizing the assessment.
2.6 Statistical analysis
The evidence was synthesized through multimodal visualization: Flowcharts delineated literature screening; temporal publication patterns were mapped via chronographic line graphs; bubble matrices quantified intervention-outcome evidence distribution; and network topographies elucidated TCM-acupuncture therapeutic synergies. Methodological rigor was stratified using proportional bar stacking. Quantitative visualizations (line/stacked graphs, bubble matrices) were constructed in Origin 2024, while therapeutic networks were modeled in Cytoscape. Statistical analyses for herbal and acupuncture modalities were computed using SPSS.
3 Result
3.1 Literature screening process and results
The systematic search yielded 6,682 potentially relevant records. EndNote-assisted deduplication retained 6,035 entries for preliminary evaluation. Title/abstract screening preserved 588 potentially eligible entries. Full-text appraisal confirmed eligibility for 187 studies (141 RCTs; 46 systematic reviews/meta-analyses), with the complete selection protocol detailed in Figure 1.
Figure 1

Flow diagram of the literature screening process.
3.2 Trends in publication
As of October 26, 2024, 141 RCTs and 46 systematic reviews/meta-analyses investigating TCM for AD treatment have been cumulatively published. Publication frequency peaked in 2022 (26 articles), with the lowest output in 2002 (2 articles). The annual publication count has demonstrated a fluctuating but overall upward trajectory since 2000, marked by a notable decline in 2023, as depicted in Figure 2.
Figure 2
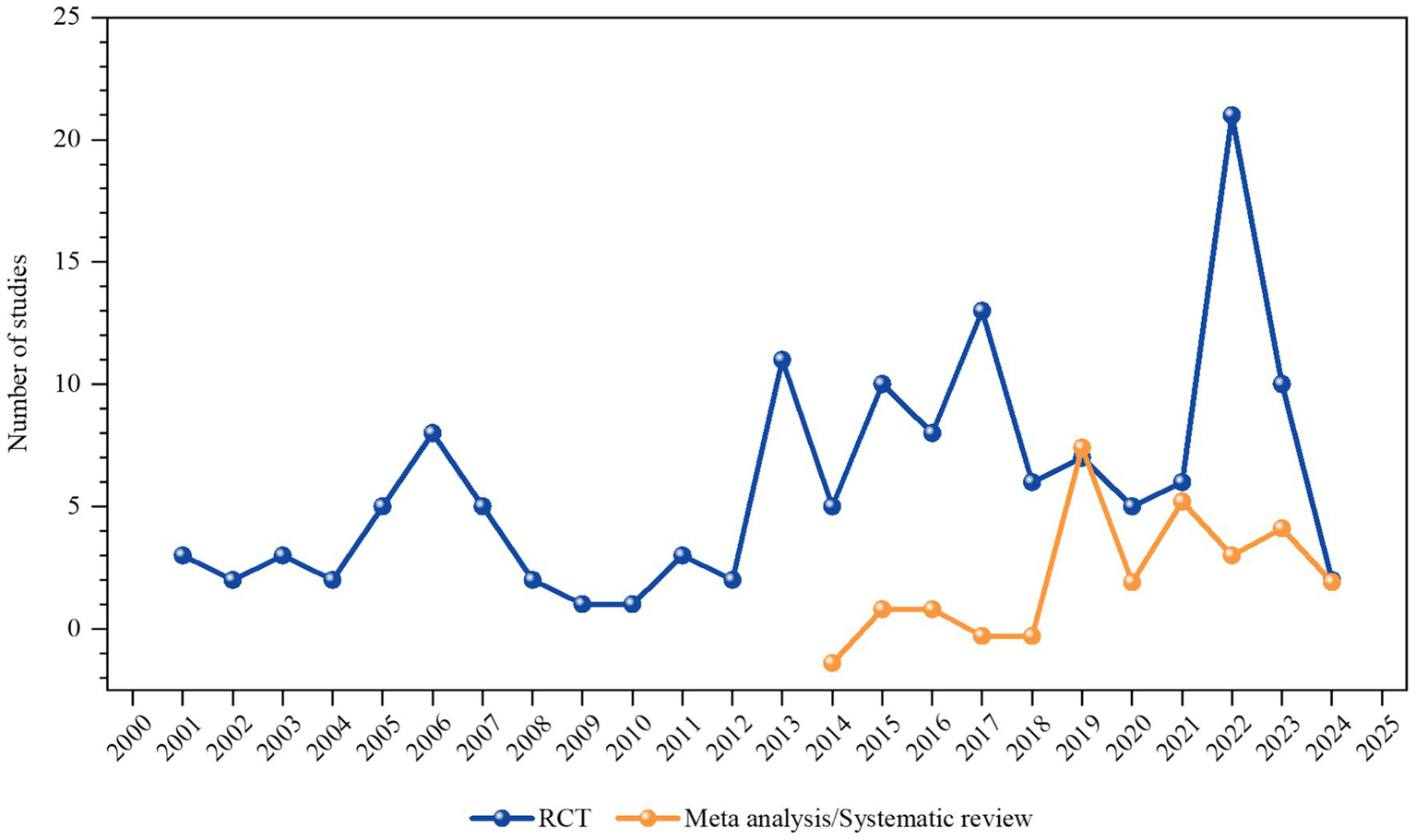
Publication trend graph. The line graph illustrates the number of publications and the trend of publication output from the year 2000 (inclusive) to 2024.
3.3 Study scale
3.3.1 Sample size
The analysis incorporated 141 RCTs comprising 12,411 participants, with enrollment ranging from 22 to 300 per study. Distribution analysis demonstrated predominant representation (60%) in the 51–100 participant stratum, versus minimal frequency (1%) for cohorts of 200–250 participants. as detailed in Table 1.
Table 1
| Sample size (n)a/per | Pub. Volb./per | Fractionc/% |
|---|---|---|
| n<50 | 21 | 15% |
| 50<n ≤ 100 | 84 | 60% |
| 100<n ≤ 150 | 24 | 17% |
| 150<n ≤ 200 | 6 | 4% |
| 200<n ≤ 250 | 2 | 1% |
| n>250 | 4 | 3% |
Sample size.
Sample size per RCT.
A total of 141 RCT publications were categorized by predefined sample size intervals. Collectively, these studies comprised an aggregated participant cohort of 12,411 individuals.
Proportional distribution of RCT publications stratified by sample size intervals, expressed as a percentage of the total publication cohort.
3.3.2 Source of literature
Among the RCTs included in this study, 28 were master’s theses, 5 were doctoral theses, 5 were published in Trials, 4 in New Chinese Medicine, 4 in Chinese Acupuncture, and 4 in Chinese Medicine Modern Distance Education. Other journals included Liaoning Journal of Traditional Chinese Medicine (3 articles), Sichuan Journal of Traditional Chinese Medicine (3 articles), Chinese and Western Medicine Combined in Cardiovascular Diseases (3 articles), Chinese Journal of Gerontology (3 articles), Chinese Journal of Integrative Medicine on Cardio-Cerebrovascular Disease (3 articles), Journal of Chengdu Medical College (2 articles), Henan Traditional Chinese Medicine (2 articles), Shanxi Journal of Traditional Chinese Medicine (2 articles), World Journal of Traditional Chinese Medicine (2 articles), Chinese Journal of Basic Medicine in Traditional Chinese Medicine (2 articles), Chinese Archives of Traditional Chinese Medicine (2 articles), Journal of Integrative Medicine (2 articles), Guiding Journal of Traditional Chinese Medicine and Pharmacy (2 articles), The institutions such as “Integrative Medicine Research,” “Journal of Acupuncture and Tuina Science,” and “Alzheimer’s & Dementia” only accept one submission each. These sources are presented in Table 2.
Table 2
| Source of literature | Pub. Vol./per | Fraction |
|---|---|---|
| Master’s theses | 28 | 19.9% |
| Doctoral theses | 5 | 3.5% |
| Trials | 5 | 3.5% |
| New Chinese Medicine, | 4 | 2.8% |
| Chinese Acupuncture | 4 | 2.8% |
| Chinese Medicine Modern Distance Education. | 4 | 2.8% |
| Liaoning Journal of Traditional Chinese Medicine | 3 | 2.1% |
| Sichuan Journal of Traditional Chinese Medicine | 3 | 2.1% |
| Chinese and Western Medicine Combined in Cardiovascular Diseases | 3 | 2.1% |
| Chinese Journal of Gerontology | 3 | 2.1% |
| Chinese Journal of Integrative Medicine on Cardio-Cerebrovascular Disease | 3 | 2.1% |
| Journal of Chengdu Medical College | 2 | 1.4% |
| Henan Traditional Chinese Medicine | 2 | 1.4% |
| Shanxi Journal of Traditional Chinese Medicine | 2 | 1.4% |
| World Journal of Traditional Chinese Medicine | 2 | 1.4% |
| Chinese Journal of Basic Medicine in Traditional Chinese Medicine | 2 | 1.4% |
| Chinese Archives of Traditional Chinese Medicine | 2 | 1.4% |
| Journal of Integrative Medicine | 2 | 1.4% |
| Guiding Journal of Traditional Chinese Medicine and Pharmacy | 2 | 1.4% |
| Others (1 article) | 60 | 42.5% |
Source of literature.
3.4 Intervention measures
A total of 71 different intervention measures were identified. The most common intervention was the use of Chinese herbal decoctions (61 studies, 43.3%), primarily focusing on kidney-tonifying formulas (17 studies), such as Bu Shen Huo Xue Fang (Kidney-tonifying and Blood-activating formula), Bu Shen Yi Jing Fang (Kidney-tonifying and Essence-nourishing formula), and Bu Shen Yi Sui Fang (Kidney-tonifying and Marrow-nourishing formula), as well as Tong Qiao Huo Xue Tang (Blood-activating and Opening the Orifices Decoction) (5 studies) and Di Huang Yin Zi (Rehmannia Decoction) (4 studies), among more than 60 other herbal decoctions. Acupuncture combined with other TCM therapies (41 studies, 29%) was also common, along with proprietary Chinese medicines (32 studies, 22.7%), including Gui Ling Ji Capsule (3 studies), Fang Huo Hai She Capsule (3 studies), and 28 other types. Other interventions (7 studies, 5%), such as poultices, acupressure, auricular acupuncture, and massage, were often used as part of combined therapy, with some not specifically listed. These interventions are summarized in Figure 3.
Figure 3

Depicts a bubble chart analyzing the clinical efficacy of traditional Chinese medicine (TCM) in Alzheimer’s disease management. Each bubble’s diameter is proportional to the number of published studies, with color gradients employed to optimize data differentiation and visual interpretability. The x-axis categorizes outcome measures (e.g., cognitive function scores), while the y-axis classifies intervention modalities (e.g., herbal formulae or acupuncture). Notably, TCM interventions predominantly involve complex herbal prescriptions. In clinical practice, physicians adapt these base formulae through herb addition or subtraction (jiajian), a therapeutic customization aligned with TCM’s personalized pharmacodynamic principles.
3.5 Influencing factors
A statistical analysis of the sample characteristics provided the following details: Sample age (129 studies)ranged from 45 to 91 years; Disease duration (86 studies) varied from 4 months to 1 year; The stages of Alzheimer’s Disease were clearly defined (28 studies), classified as Stage 1 (mild), Stage 2 (moderate), and Stage 3 (severe); Education (139 studies); Intervention duration (140 studies) ranged from 4 weeks to 1 year; History of smoking and alcohol consumption (10 studies); Syndrome differentiation and treatment (61 studies); Follow-up for efficacy assessment (23 studies).
3.6 Outcome measures
Outcome measures across 141 RCTs were stratified by reporting frequency: (1) Total effective rate; (2) Mini-Mental State Examination (MMSE) score; (3) Adverse reactions: dizziness, nausea, vomiting, loss of appetite, sleep disturbances, gastrointestinal symptoms, chest tightness, and bradykinesia; (4) Activities of Daily Living (ADL) score; (5) Safety indicators: complete blood count, urinalysis, stool routine, occult blood in stool, coagulation profile, liver function, kidney function, electrocardiogram, etc.; (6) Other disease-related assessments: laboratory tests, TCM symptoms and signs, etc.; (7) TCM syndrome score; (8) Alzheimer’s Disease Assessment Scale-Cognitive Subscale (ADAS-cog) score; (9) Inflammatory markers: interleukin-1β (IL-1β), interleukin-6 (IL-6), plasma Aβ1-42, tumor necrosis factor-α (TNF-α), C-reactive protein (CRP), transforming growth factor-β1 (TGF-β1), etc.; (10) Clinical Dementia Rating (CDR) score; (11) Hasegawa Dementia Scale-Revised (HDS-R) score; (12) Montreal Cognitive Assessment (MoCA) score; (13) Neuroimaging; (14) Amyloid protein levels; (15) Oxidative stress markers; (16) Blood lipids; and (17) Behavioral and Psychological Symptoms of Dementia (BEHAVE-AD) score.
The most commonly reported outcome measures in RCTs for TCM treatment of AD were total effective rate (104 studies) and MMSE score (86 studies), followed by adverse reactions (51 studies) and ADL score (43 studies). Safety indicators (43 studies) and other disease-related measurements (31 studies) were less frequently reported, as summarized in Figure 3.
3.7 Frequency analysis of medicinal herb usage
In the included studies, the frequency of individual herbal medicine usage was recorded a total of 2,346 times. The most frequently used herbs in the compound prescriptions were analyzed. The top 15 most frequently used single herbs were: Shichangpu (Acorus tatarinowii Schott) (48 times), Shudi (Rehmanniae Radix Praeparata) (40 times), Yuanzhi (Polygala tenuifolia Willd) (36 times), chuanxiong (Ligusticum chuanxiong Hort) (34 times), Fuling (Poria cocos Wolf) (32 times), Danggui (Angelica sinensis) (29 times), Huangqi (Astragalus membranaceus Bge) (28 times), Shanzhuyu (Cornus officinalis Sieb. et Zucc) (27 times), Gancao (Glycyrrhiza uralensis Fisch) (25 times), Yinyanghuo (Epimedium brevicornu Maxim) (25 times), Danshen (Salvia miltiorrhiza Bge) (22 times), Heshouwu (Polygonum multiflorum Thunb) (22 times), Roucongrong (Cistanche deserticola Y.C.Ma) (21 times), Baizhu (Atractylodes macrocephala Koidz) (17 times), and Gouqi (Lycium chinense Mill) (17 times).
The top 15 most commonly used herb pairings, based on percentage support, were as follows: Rehmanniae Radix Praeparata—Cistanche deserticola Y.C.Ma (20.59%); Acorus tatarinowii Schott—Poria cocos Wolf, Polygala tenuifolia Willd (14.71%); Rehmanniae Radix Praeparata—Bajitian (Morinda officinalis How) (13.73%); Rehmanniae Radix Praeparata—Tusizi (Cuscuta chinensis Lam) (13.73%); Chishao (Paeonia veitchii Lynch)—Taoren (Prunus persica(L.)Batsch) (12.75%); Rehmanniae Radix Praeparata—Cistanche deserticola Y.C.Ma, Polygala tenuifolia Willd (12.75%); Acorus tatarinowii Schott—Cistanche deserticola Y.C.Ma, Polygala tenuifolia Willd (12.75%); Rehmanniae Radix Praeparata—Cornus officinalis Sieb. et Zucc, Polygala tenuifolia Willd (12.75%); Acorus tatarinowii Schott—Cornus officinalis Sieb. et Zucc, Polygala tenuifolia Willd (12.75%); Prunus persica(L.)Batsch—Paeonia veitchii Lynch (11.76%); Glycyrrhiza uralensis Fisch—Chaihu (Bupleurum chinense DC) (11.76%); Acorus tatarinowii Schott—Cistanche deserticola Y.C.Ma, Cornus officinalis Sieb. et Zucc (11.76%); Polygala tenuifolia Willd—Maidong (Ophiopogon ja ponicus Ker-Gawl) (11.76%); Polygala tenuifolia Willd—Morinda officinalis How, Rehmanniae Radix Praeparata (11.76%); Polygala tenuifolia Willd—Wuweizi (Schisandra chinensis Baill), Acorus tatarinowii Schott (11.76%). These results are summarized in Table 3 and Figure 4.
Table 3
| Consequent | Antecedent | Support | Confidence percentage | Gain |
|---|---|---|---|---|
| Rehmanniae Radix Praeparata | Cistanche deserticola Y.C.Ma | 20.59 | 80.95 | 2.06 |
| Acorus tatarinowii Schott | Poria cocos Wolf, Polygala tenuifolia Willd | 14.71 | 80 | 1.7 |
| Rehmanniae Radix Praeparata | Morinda officinalis How | 13.73 | 85.71 | 2.19 |
| Rehmanniae Radix Praeparata | Cuscuta chinensis Lam | 13.73 | 85.71 | 2.19 |
| Paeonia veitchii Lynch | Prunus persica(L.)Batsch | 12.75 | 92.31 | 7.85 |
| Rehmanniae Radix Praeparata | Cistanche deserticola Y.C.Ma, Polygala tenuifolia Willd | 12.75 | 84.62 | 2.16 |
| Acorus tatarinowii Schott | Cistanche deserticola Y.C.Ma, Polygala tenuifolia Willd | 12.75 | 84.62 | 1.8 |
| Rehmanniae Radix Praeparata | Cornus officinalis Sieb. et Zucc, Polygala tenuifolia Willd | 12.75 | 84.62 | 2.16 |
| Acorus tatarinowii Schott | Cornus officinalis Sieb. et Zucc, Polygala tenuifolia Willd | 12.75 | 84.62 | 1.8 |
| Prunus persica(L.)Batsch | Paeonia veitchii Lynch | 11.76 | 100 | 7.85 |
| Glycyrrhiza uralensis Fisch | Bupleurum chinense DC | 11.76 | 19.67 | 3.74 |
| Acorus tatarinowii Schott | Cistanche deserticola Y.C.Ma, Cornus officinalis Sieb. et Zucc | 11.76 | 19.67 | 1.95 |
| Polygala tenuifolia Willd | Ophiopogon ja ponicus Ker-Gawl | 11.76 | 83.33 | 2.36 |
| Polygala tenuifolia Willd | Morinda officinalis How, Rehmanniae Radix Praeparata | 11.76 | 83.33 | 2.36 |
| Polygala tenuifolia Willd | Schisandra chinensis Baill, Acorus tatarinowii Schott | 11.76 | 83.33 | 2.36 |
Statistical correlations of traditional Chinese medicine herb co-occurrences.
Figure 4

Chinese medicine association rules diagram. Node chromatic intensity denotes TCM utilization frequency, with edge weight reflecting herbal pair co-occurrence frequency.
3.8 Acupuncture point selection analysis
Analysis of included studies identified 50 acupuncture points employed in AD treatment, with 15 most utilized: Baihui (GV20) (23 times), Sishencong (EX-HN1) (18 times), Shenting (GV24) (15 times), Neiguan (PC6) (11 times), Shenmen (HT7) (11 times), Zusanli (ST36) (11 times), Taixi (KD3) (10 times), Xuan Zhong (GB39) (9 times), Fengchi (GB20) (8 times), Sanyinjiao (SP6) (8 times), Shenshu (BL23) (7 times), Yintang (EX-HN3) (6 times), Benshen (GB13) (5 times), and Dazhui (GV14), Hegu (LI4), Taichong (LR3), Xuehai (SP10), Zhaohai (KI6), and Zhongwan (RN12) (4 times each).
The top 15 acupuncture point combinations, based on the percentage of support, were: GV20 – GV24 (45.45%); EX-HN1 – GV24 (45.45%); EX-HN1 – GV24, GV20 (39.39%); GV20 – GV24, EX-HN1 (36.36%); EX-HN1 – HT7 (33.33%); GV20 – PC6 (33.33%); GV20 – HT7 (33.33%); EX-HN1 – KD3 (30.30%); GV20 – KD3 (30.30%); GV20 – HT7, EX-HN1 (30.30%); EX-HN1 – HT7, GV20 (27.27%); HT7 – SP6 (24.24%); GV20 – SP6 (24.24%); GV20 – GB20 (24.24%); GV20 – KD3, EX-HN (24.24%); EX-HN1 – KD3, GV20 (24.24%); GV24 – HT7, EX-HN1, GV20 (24.24%). These results are summarized in Table 4 and Figure 5.
Table 4
| Consequent | Antecedent | Support | Confidence percentage | Gain |
|---|---|---|---|---|
| GV20 | GV24 | 45.45 | 86.67 | 1.24 |
| EX-HN1 | GV24 | 45.45 | 80.00 | 1.47 |
| EX-HN1 | GV24, GV20 | 39.39 | 84.62 | 1.55 |
| GV20 | GV24, EX-HN1 | 36.36 | 91.67 | 1.32 |
| EX-HN1 | HT7 | 33.33 | 90.91 | 1.67 |
| GV20 | PC6 | 33.33 | 90.91 | 1.30 |
| GV20 | HT7 | 33.33 | 81.82 | 1.17 |
| EX-HN1 | KD3 | 30.30 | 80.00 | 1.47 |
| GV20 | KD3 | 30.30 | 80.00 | 1.15 |
| GV20 | HT7, EX-HN1 | 30.30 | 80.00 | 1.15 |
| EX-HN1 | HT7, GV20 | 27.27 | 88.89 | 1.63 |
| HT7 | SP6 | 24.24 | 87.50 | 2.63 |
| GV20 | SP6 | 24.24 | 87.50 | 1.26 |
| GV20 | GB20 | 24.24 | 87.50 | 1.26 |
| GV20 | KD3, EX-HN1 | 24.24 | 87.50 | 1.26 |
| EX-HN1 | KD3, GV20 | 24.24 | 87.50 | 1.60 |
| GV24 | HT7, EX-HN1, GV20 | 24.24 | 87.50 | 1.93 |
The graphical representation encapsulates the statistical patterns underlying the co-occurrence of traditional Chinese medicine herbs.
Figure 5
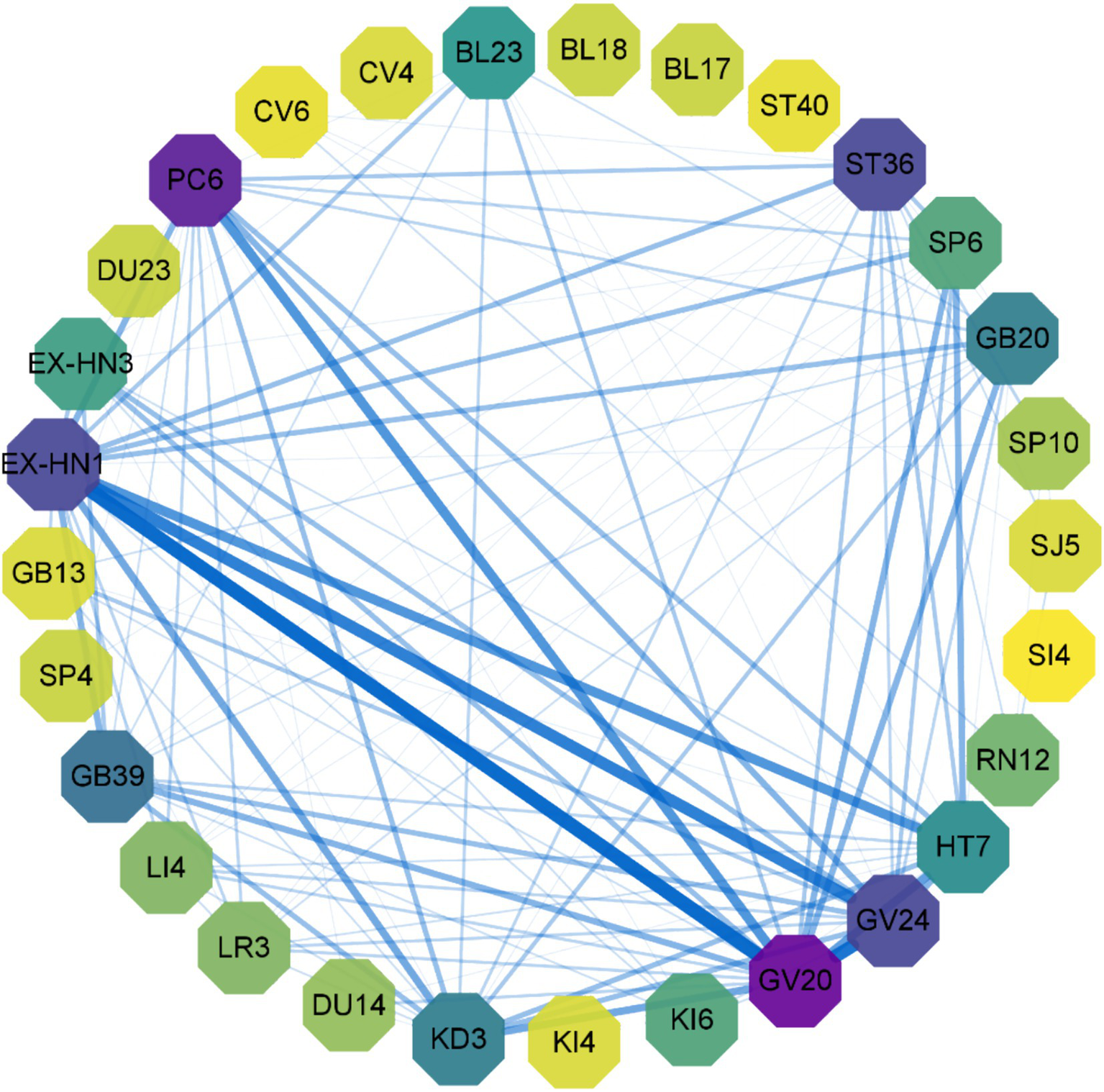
Acupoint association rules graph. Node chromatic intensity denotes acupoint utilization frequency, with edge weight reflecting acupoint co-occurrence frequency.
3.9 Methodological quality assessment
3.9.1 Methodological quality assessment of RCTs
Eighteen studies were excluded for incomplete data, yielding 123 studies for final analysis. Risk assessment demonstrated “low bias” in domains including randomization, allocation concealment, participant/personnel blinding, outcome assessment blinding, data completeness, reporting transparency, and other confounders. “Unclear risk” applied to studies with unreported implementation details, while “high risk” indicated absent methodological safeguards, as detailed in Figure 6.
Figure 6
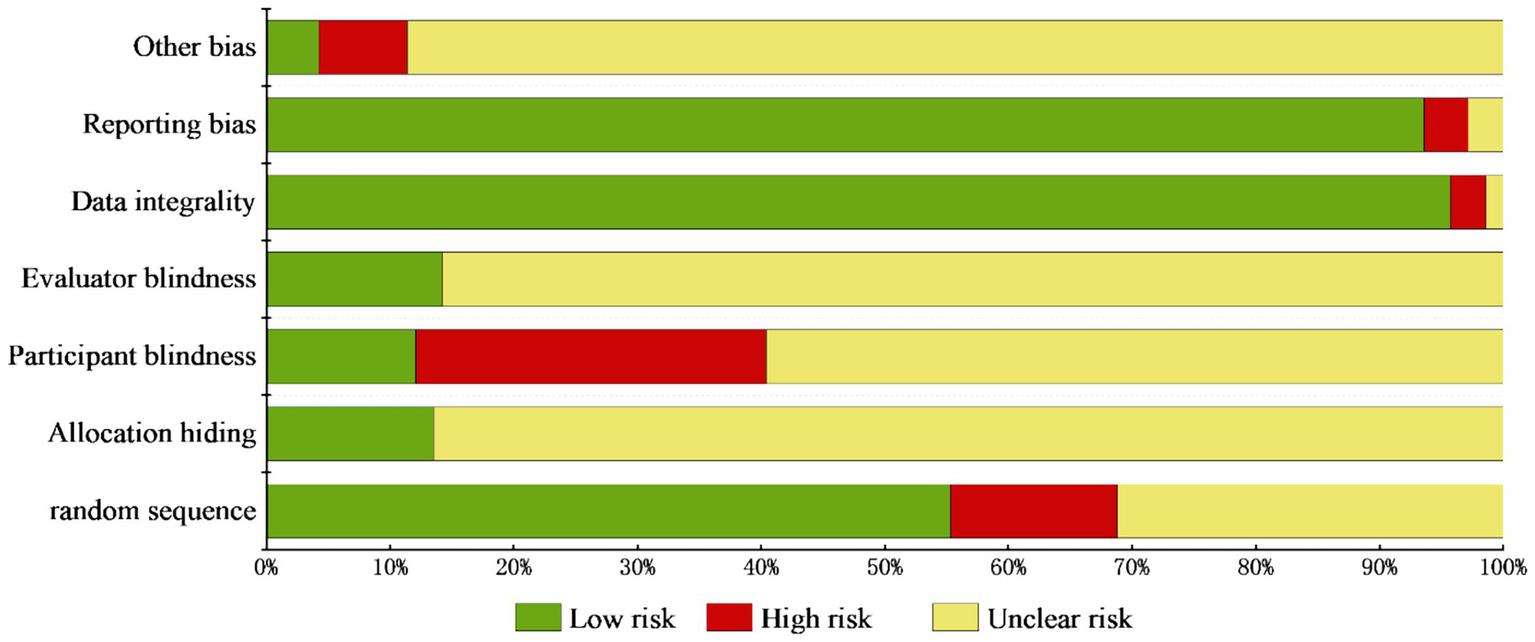
Bar chart representing the percentage of bias risk assessment in included RCTs. The chart utilizes bars of different hues to denote various bias risk domains. The x-axis represents the proportion of studies with different levels of risk, and the y-axis enumerating the seven Cochrane ROB assessment criteria.
3.9.2 Methodological quality assessment of meta-analyses/systematic reviews
The AMSTAR2 tool was used to assess the methodological quality of 46 included meta-analyses/systematic reviews. The results indicated that all included studies employed appropriate methods to assess the risk of bias in each included study, and they reasonably interpreted and discussed the heterogeneity of the research outcomes. However, none of the studies reported the list of excluded studies, funding sources, or conflicts of interest. These findings are illustrated in Figure 7.
Figure 7

Bar graph illustrating the percentage-based bias risk assessment of included meta-analyses/systematic reviews. The visualization employs chromatic bars encoding distinct bias risk domains, with the x-axis represents the proportion of studies with different levels of risk, the y-axis enumerating the 16 AMSTAR 2 methodological criteria.
4 Discussion
This evidence mapping study synthesizes RCTs and systematic evidence syntheses on TCM interventions for AD, delineating the current evidence landscape.
4.1 General characteristics of included studies
Quantitative assessment of RCT sample sizes demonstrated 84 trials (60%) enrolled 50–100 participants, whereas 6 trials (4%) encompassed 200–300 participants. Implementation of sample size estimation formulas remains imperative to establish methodologically robust, cost-effective minimum thresholds ensuring trial validity (13, 14). Intervention durations exhibited significant heterogeneity (4 weeks–1 year), with 95% of trials employing ≤24-week protocols, predominantly 8, 12, and 24-week regimens. Furthermore, the evidence base comprised degree thesis (78%), contrasting with limited institutionally indexed articles (22%). This disparity likely stems from the cost-efficiency and smaller-scale feasibility of degree thesis theses versus institutionally mandated studies requiring multicenter collaborations and methodological rigor thresholds. Delayed institutional review cycles and unmet journal impact criteria may further constrain formal dissemination of lower-evidence RCTs (15).
4.2 Intervention strategies
The RCTs analyzed in this study indicate therapeutic potential of TCM interventions in AD management. The interventions primarily encompass acupuncture, Chinese herbal decoctions, Chinese patent medicines, moxibustion, and external TCM therapies (e.g., Ear Acupoint Bean Pressing, Herbal Plasters). Acupuncture and herbal decoctions constituted the predominant therapeutic modalities. Among 141 RCTs, 40 (28.37%) adopted acupuncture as the primary intervention, 81(57.44%) adopted traditional Chinese medicine decoctions as the primary intervention, including 11 studies (0.08%) that combined acupuncture with traditional Chinese medicine. Primary acupoints GV20 (Baihui) and EX-HN1 (Sishencong) were selected per Zhenjiu Dacheng (The Great Compendium of Acupuncture) for their bidirectional regulatory capacities in cerebral homeostasis—balancing sedative and nootropic effects. Mechanistic analyses indicate these acupoints modulate cortical neurotransmitter dynamics and neuropeptide homeostasis, enhancing neural circuit plasticity through glutamatergic-GABAergic equilibrium restoration (16, 17).
Herbal decoctions for AD predominantly featured Tongqiao Huoxue Decoction, Dihuang Yinzi, and customized formulations. Given the compositional heterogeneity of bespoke prescriptions, analytical focus centered on high-frequency herbal dyads and monocomponent agents. The most prevalent phytotherapeutic pairs included Rehmanniae Radix Praeparata—Cistanche deserticola Y.C.Ma; Acorus tatarinowii Schott—Poria cocos Wolf, Polygala tenuifolia Willd; Rehmanniae Radix Praeparata—Morinda officinalis How. Radix Rehmanniae Preparata is known for nourishing kidney yin (18), Cistanche deserticola Y.C.Ma supplements kidney yang and enhances essence and blood (19), Acorus tatarinowii Schott opens the orifices and clears the mind (20), Poria cocos Wolf strengthens the spleen and calms the heart (21), and Polygala tenuifolia Willd improves memory and calms the mind (22). Morinda officinalis How demonstrates therapeutic efficacy through dual mechanisms of renal yang tonification and osteoarticular reinforcement (23). The Rehmanniae Radix Praeparata—Cistanche deserticola Y.C.Ma dyad, chronicled in Yizong Bidu (Essential Readings of Medical Tradition), achieves cerebrotonic effects through yin-yang equilibrium. Modern mechanistic studies validate its capacity to mitigate senescence-associated cognitive decline via pleiotropic modulation of neuroinflammatory and neurotrophic pathways (24), while Acorus tatarinowii Schott has anti-inflammatory and antioxidant effects (3, 25). Polygala tenuifolia Willd exhibits nootropic efficacy and neuroprotective properties through multi-target pharmacological modulation (26).
4.3 Outcome indicators
Outcome measures constitute fundamental determinants of clinical research validity (27). This investigation revealed outcome measurement limitations: Firstly, evidence standardization deficits, with merely 28 studies operationally defining AD staging criteria, and as the disease progresses, patients’ cognitive functions and abilities in daily living gradually decline, which may significantly impact outcome measure (28). Only 10 studies documented lifestyle (e.g., smoking/alcohol use) risk factors implicated in oxidative-inflammatory pathophysiology that accelerates neurodegeneration/apoptosis, thereby influence AD therapeutic efficacy (29, 30). Only 23 articles recorded long-term follow-up for efficacy, leading to a lack of data on long-term efficacy and potential issues; And Evidence deficits persist in family/past medical history. Secondly, Within TCM therapeutic frameworks, only 14 studies operationally defined bianzheng criteria—the diagnostic paradigm governing TCM therapeutic strategies. This methodology enables etiopathogenic precision through syndrome-specific intervention tailoring, ensuring TCM-consistent clinical evidence generation (31). Lastly, There are some limitations in the evaluation indicators of therapeutic effects: Sparse implementation of TCM-specific symptomatology assessments; Non-standardized adverse event documentation via TESS protocols; commonly used scales, such as the MMSE, ADAS-cog, HDS-R, and ADL, Heterogeneity in assessment criteria may introduce variability in therapeutic outcomes across studies (32). Future clinical research should prioritize rigorous trial design validation, metric standardization, and systematic integration of TCM-specific biomarkers including symptomatology quantification and syndromic stratification to enhance methodological robustness and evidentiary credibility.
4.4 Methodological quality assessment
Literature quality assessment constitutes a methodological cornerstone in systematic reviews, ensuring methodological rigor for evidence-based clinical trial design and guideline formulation (33). Cochrane bias assessment of included RCTs demonstrated predominant unclear risk ratings, suggesting suboptimal methodological quality requiring standardization enhancement. Methodological deficiencies, including deficient randomization and allocation concealment protocols, compromised participant/investigator blinding integrity, potentially introducing placebo effects. Concurrent investigator preferentiality and assessor expectancy bias may collectively introduce measurement inaccuracies that undermine therapeutic effect validity. In terms of study design and implementation, most RCTs have incomplete information, lacking key data such as sample size calculation, long-term efficacy follow-up, and the number of dropouts. The 46 meta-analyses/systematic reviews demonstrate significant outcomes of TCM in treating AD, but the overall credibility is not high. Critical deficiencies encompass absent prospectively registered protocols, omission of excluded study inventories, insufficient declaration of funding provenance, and opacity in conflict-of-interest disclosures. Strengthening research validity requires implementation of rigorous randomization procedures, adherence to evidence-based frameworks, incorporation of longitudinal outcome tracking, enhancement of evidentiary integrity, and systematic mitigation of bias contamination (34).
4.5 Recommendations for future research
Building upon the identified evidence gaps, we recommend four key methodological improvements to advance future studies: (1) stringent implementation of bias-control strategies, including randomization and blinding procedures; (2) utilization of standardized reporting tools, such as TCM syndrome scoring systems; (3) integration of trial designs with the fundamental tenets of TCM syndrome differentiation and treatment; and (4) extension of follow-up durations to evaluate long-term therapeutic outcomes. Collectively, these refinements are designed to strengthen both the methodological rigor and translational relevance of future TCM-based interventions for AD.
5 Conclusion
This evidence mapping study evaluated randomized controlled trials and systematic reviews of Traditional Chinese Medicine interventions for Alzheimer’s disease. While demonstrating therapeutic potential, critical methodological limitations were identified. Underpowered sample cohorts and inherent challenges in blinding manual interventions such as acupuncture contributed to clinical heterogeneity. Additionally, the studies failed to report confidence intervals for clinical efficacy and inadequately addressed methodological heterogeneity. The evidence framework remains incomplete, with insufficient representation of TCM’s syndrome differentiation and treatment principles and a lack of distinct TCM-specific outcome measures.
Methodological appraisal revealed prevalent design flaws including ambiguous randomization protocols and suboptimal blinding implementation, undermining validity. Future research priorities should emphasize large-scale multicenter trials adhering to standardized TCM guidelines, development of syndrome-specific therapeutic protocols, and actively exploring and incorporating TCM-specific evaluation criteria and efficacy indicators is essential for a more comprehensive assessment of TCM’s therapeutic efficacy and safety in treating AD. These initiatives seek to establish a rigorous evidence base validating TCM’s therapeutic efficacy in Alzheimer’s disease management.
Statements
Data availability statement
The original contributions presented in the study are included in the article/supplementary material, further inquiries can be directed to the corresponding author.
Author contributions
SC: Data curation, Writing – original draft, Writing – review & editing, Methodology, Visualization. YZ: Data curation, Methodology, Visualization, Writing – original draft, Writing – review & editing. XW: Writing – review & editing. YH: Writing – review & editing. JY: Writing – review & editing. ZD: Writing – review & editing. YJL: Writing – review & editing. HQ: Writing – review & editing. LW: Writing – review & editing. YL: Writing – review & editing. KW: Writing – review & editing. GZ: Writing – review & editing. QQ: Funding acquisition, Project administration, Supervision, Writing – review & editing.
Funding
The author(s) declare that financial support was received for the research and/or publication of this article. This study was supported by the National Natural Science Foundation of China (Grant No. 82460989), the Guangxi Natural Science Foundation (Grant No. 2023GXNSFAA026084), the Guangxi Higher Education Institutions Middle-aged and Young Teachers’ Basic Scientific Research Capacity Promotion Project (Grant No. 2023KY0325), the Guangxi University of Chinese Medicine Doctoral Research Startup Fund Project (Grant No. 2018BS064), and the Guangxi International Zhuang Medicine Hospital Talent Introduction Research Startup Fund Project (Grant No. GZ2024RC001).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The authors declare that Gen AI was used in the creation of this manuscript. During the research process, no AI technology was employed. However, during manuscript revision, AI-assisted tools were utilized solely for linguistic refinement of certain expressions.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Abbreviations
AD, Alzheimer’s Disease; ADAS-cog, Alzheimer’s Disease Assessment Scale-Cognitive Subscale; ADL, Activities of Daily Living; BEHAVE-AD, Behavioral and Psychological Symptoms of Dementia; CDR, Clinical Dementia Rating; CNKI, China National Knowledge Infrastructure; CRP, C-reactive protein; HDS-R, Hasegawa Dementia Scale-Revised; IL-1β, interleukin-1β; IL-6, interleukin-6; MMSE, Mini-Mental State Examination; MoCA, Montreal Cognitive Assessment; RCTs, randomized controlled trials; TCM, Traditional Chinese Medicine; TGF-β1, transforming growth factor-β1; TNF-α, tumor necrosis factor-α.
References
1.
Alzheimer’s Association . Alzheimer's disease facts and figures. Alzheimers Dement. (2024) 20:3708–821. doi: 10.1002/alz.13809
2.
Roelofs A . Accounting for word production, comprehension, and repetition in semantic dementia, Alzheimer's dementia, and mild cognitive impairment. Brain Lang. (2023) 238:105243. doi: 10.1016/j.bandl.2023.105243
3.
Rosselli M Uribe IV Ahne E Shihadeh L . Culture, ethnicity, and level of education in Alzheimer's disease. Neurotherapeutics. (2022) 19:26–54. doi: 10.1007/s13311-022-01193-z
4.
Grande G Vetrano DL Kalpouzos G Welmer AK Laukka EJ Marseglia A et al . Brain changes and Fast cognitive and motor decline in older adults. J Gerontol A Biol Sci Med Sci. (2023) 78:326–32. doi: 10.1093/gerona/glac177
5.
Sara Evans-Lacko EA Read S Comas-Herrera A Farina N . World Alzheimer report 2024. London, UK: Alzheimer's Disease International (2024). Available at: https://www.alzint.org/resource/world-alzheimer-report-2024/
6.
Gustavsson A Norton N Fast T Frölich L Georges J Holzapfel D et al . Global estimates on the number of persons across the Alzheimer's disease continuum. Alzheimers Dement. (2023) 19:658–70. doi: 10.1002/alz.12694
7.
Bagetta G Bano D Scuteri D . Basic, translational, and clinical research on dementia. Int J Mol Sci. (2024) 25:6861. doi: 10.3390/ijms25136861
8.
Parsons K Smith-Young J Pike A . Understanding how community-dwelling persons with early dementia perceive health and community services: informing the dementia strategy of Newfoundland and Labrador, Canada. Dementia (London). (2025) 24:150–70. doi: 10.1177/14713012241284693
9.
Zhang L Li H Hu L Ou X Tan H Zhang X et al . Reporting characteristics and quality of randomized controlled trial protocols in traditional Chinese medicine: a cross-sectional study. Front Pharmacol. (2024) 15:1389808. doi: 10.3389/fphar.2024.1389808
10.
Alahdab F Murad MH . Evidence maps: a tool to guide research agenda setting. BMJ Evid Based Med. (2019) 24:209–11. doi: 10.1136/bmjebm-2018-111137
11.
Higgins JPT Savović Jelena Page MJ Elbers RG Sterne JAC (2024). Cochrane handbook for systematic reviews of interventions version 6.5. Cochrane. Available online at: https://training.cochrane.org/handbook/current/chapter-08 (Accessed Month 10, 2024).
12.
Shea BJ Reeves BC Wells G Thuku M Hamel C Moran J et al . AMSTAR 2: a critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ. (2017) 358:j4008. doi: 10.1136/bmj.j4008
13.
Andrade C . Poorly recognized and uncommonly acknowledged limitations of randomized controlled trials. Indian J Psychol Med. (2024) 47:02537176241297953. doi: 10.1177/02537176241297953
14.
Verma V Mishra AK Narang R . Determination of sample size for different clinical study designs. J Assoc Physicians India. (2024) 72:34–40. doi: 10.59556/japi.72.0492
15.
Purohit U Sacks CA Raissi A Hughes E Boctor M Manzoor S et al . Press releases of drug-related randomized trial results prior to publication in high-Impact Journals: an observational study. J Gen Intern Med. (2023) 38:3107–14. doi: 10.1007/s11606-023-08313-1
16.
Li F Wang Y Jiang T-X Zhu M-J Ji J-J Wu W-W et al . Acupuncture and moxibustion for vascular dementia and its effect on serum VEGF and AChE. Zhongguo Zhen Jiu. (2021) 41:851–4. doi: 10.13703/j.0255-2930.20200816-0001
17.
Wei Y-T Zhu T-T Jia J Yan X-K . Research progress on the mechanism of acupuncture intervention on Alzheimer's disease. Zhen Ci Yan Jiu. (2022) 47:362–8. doi: 10.13702/j.1000-0607.20210480
18.
Yuan H Yang M Han X Ni X . The therapeutic effect of the Chinese herbal medicine, Rehmanniae Radix Preparata, in attention deficit hyperactivity disorder via reversal of structural abnormalities in the cortex. Evid Based Complement Alternat Med. (2018) 2018:3052058. doi: 10.1155/2018/3052058
19.
Cheng N Wang H Hao H Rahman FU Zhang Y . Research progress on polysaccharide components of Cistanche deserticola as potential pharmaceutical agents. Eur J Med Chem. (2023) 245:114892. doi: 10.1016/j.ejmech.2022.114892
20.
Hao W Chen J Zhang Y Mou T Wang J Zhang C et al . Integration of metabolomics and network pharmacology to validate the mechanism of Schisandra chinensis(Turcz.)Baill- Acorus tatarinowii Schott ameliorating the Alzheimer's disease by regulating the aromatase activity to affect local estrogen in brain of AD model rats. Arab J Chem. (2023) 16:104457. doi: 10.1016/j.arabjc.2022.104457
21.
Zhao MH Guan ZY Tang N Cheng YQ . The differences between the water- and alkaline-soluble Poria cocos a review. Int J Biol Macromol. (2023) 235:123925. doi: 10.1016/j.ijbiomac.2023.123925
22.
Li YY Zheng HM Yao QH Ma YB Wang L Liu Q et al . Preparation, structural characteristics and pharmacological activity of polysaccharides from Polygala tenuifolia: a review. Carbohydr Res. (2024) 539:109117. doi: 10.1016/j.carres.2024.109117
23.
Zhang Y Zhang M . Neuroprotective effects of Morinda officinalis how.: anti-inflammatory and antioxidant roles in Alzheimer's disease. Front Aging Neurosci. (2022) 14:963041. doi: 10.3389/fnagi.2022.963041
24.
Sun R Yuan H Wang J Zhu K Xiong Y Zheng Y et al . Rehmanniae Radix Preparata ameliorates behavioral deficits and hippocampal neurodevelopmental abnormalities in ADHD rat model. Front Neurosci. (2024) 18:1402056. doi: 10.3389/fnins.2024.1402056
25.
Huang J Xu Z Yu C Liu L Ji L Qiu P et al . The volatile oil of Acorus tatarinowii Schott ameliorates Alzheimer's disease through improving insulin resistance via activating the PI3K/AKT pathway. Phytomedicine. (2024) 135:156168. doi: 10.1016/j.phymed.2024.156168
26.
Li S Hou Z Ye T Song X Hu X Chen J . Saponin components in Polygala tenuifolia as potential candidate drugs for treating dementia. Front Pharmacol. (2024) 15:1431894. doi: 10.3389/fphar.2024.1431894
27.
Outcome measures and measurement science are the foundation of clinical research. J Hand Ther. (2023) 36:1–2. doi: 10.1016/j.jht.2023.03.007
28.
Jia J Xu J Liu J Wang Y Wang Y Cao Y et al . Comprehensive Management of Daily Living Activities, behavioral and psychological symptoms, and cognitive function in patients with Alzheimer's disease: A Chinese consensus on the Comprehensive Management of Alzheimer's disease. Neurosci Bull. (2021) 37:1025–38. doi: 10.1007/s12264-021-00701-z
29.
Durazzo TC Mattsson N Weiner MW . Smoking and increased Alzheimer's disease risk: a review of potential mechanisms. Alzheimers Dement. (2014) 10:S122–45. doi: 10.1016/j.jalz.2014.04.009
30.
Miller SE Mogle JA Linden-Carmichael AN Almeida DM . Within-person associations between alcohol use and memory lapses among middle-aged and older adults. Drug Alcohol Rev. (2023) 42:1754–63. doi: 10.1111/dar.13721
31.
Jiang M Lu C Zhang C Yang J Tan Y Lu AP et al . Syndrome differentiation in modern research of traditional Chinese medicine. J Ethnopharmacol. (2012) 140:634–42. doi: 10.1016/j.jep.2012.01.033
32.
Turana Y Farina N Theresia I Fitri FI Suswanti I Jacobs R et al . The dementia severity rating scale: A potential community screening tool for dementia in low- and middle-income countries. Dementia (London). (2024) 23:476–92. doi: 10.1177/14713012231186837
33.
Butcher NJ Monsour A Mew EJ Chan AW Moher D Mayo-Wilson E et al . Guidelines for reporting outcomes in trial reports: the CONSORT-outcomes 2022 extension. JAMA. (2022) 328:2252–64. doi: 10.1001/jama.2022.21022
34.
Howard B Diug B Ilic D . Methods of teaching evidence-based practice: a systematic review. BMC Med Educ. (2022) 22:742. doi: 10.1186/s12909-022-03812-x
Summary
Keywords
traditional Chinese medicine, Alzheimer’s disease, randomized controlled trials, meta-analysis, evidence map
Citation
Cui S, Zhao Y, Wang X, Huang Y, Ye J, Deng Z, Li Y, Qin H, Wang L, Li Y, Wang K, Zheng G and Qin Q (2025) Evaluating the clinical evidence of TCM in Alzheimer’s disease: an evidence map perspective. Front. Neurol. 16:1571361. doi: 10.3389/fneur.2025.1571361
Received
05 February 2025
Accepted
06 August 2025
Published
29 August 2025
Volume
16 - 2025
Edited by
Luca Rastrelli, University of Salerno, Italy
Reviewed by
Gaurav Kandoi, Invaio Sciences, United States
Priyanka Choudhury, Medical College of Wisconsin, United States
Updates
Copyright
© 2025 Cui, Zhao, Wang, Huang, Ye, Deng, Li, Qin, Wang, Li, Wang, Zheng and Qin.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Kaihua Wang, 67232624@qq.comQijing Qin, qinqijing2004@163.com
†These authors have contributed equally to this work
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.