Abstract
Background:
Diabetes mellitus is a major public health issue, and its complication, diabetic peripheral neuropathy (DPN), can significantly impair foot and ankle joint function, affecting daily activities and quality of life. This systematic review and meta-analysis aimed to evaluate the impact of exercise on musculoskeletal function and clinical outcomes in DPN patients.
Methods:
Evaluation of randomized controlled trials (RCTs) of exercise in patients with DPN published from Cochrane Library, PubMed, Embase, Web of Science, Medline, ClinicalKey, CNKI, Wanfang database, VIP Chinese Journal Database, and Chinese Biomedical Literature Database was performed. Revman 5.4 software was used for statistical analysis.
Results:
A total of 625 samples were included in 10 studies. It showed that exercise significantly increased ankle dorsiflexion range of motion (SMD = 0.61, 95%CI (0.13, 1.08), P < 0.05), ankle flexion range of motion (SMD = 0.59, 95% CI (0.12, 1.06), P < 0.05), hallux strength (MD = 1.89, 95% CI (1.00, 2.78), P < 0.001), toes strength (MD = 2.51, 95% CI (1.69, 3.33), P < 0.001) and lower extremity functional strength (MD = −2.82, 95% CI (−3.88, −1.76), P < 0.001), whereas reduced glycosylated hemoglobin (SMD = −1.44, 95% CI (−2.30, −0.57), P < 0.01) and body mass index (MD = −0.86, 95% CI (−1.15, −0.57), P < 0.001). However, discrepancies were observed between pooled results based on different neuropathy assessment tools.
Conclusion:
The available evidence suggests that exercise training is an effective method for improving peripheral neuropathy.
Systematic review registration:
https://inplasy.com/, identifier INPLASY202340112.
1 Introduction
Diabetes mellitus (DM), as a metabolic chronic disease, has become a growing public health challenge worldwide. According to 2021 data, ~537 million adults aged 20 to 79 years worldwide suffer from diabetes, accounting for 9.3% of the population in this age group, and is expected to increase to 783 million by 2045 (1). Diabetic peripheral neuropathy (DPN) is one of the most common microvascular complications of DM and its prevalence is even higher than that of diabetic retinopathy and diabetic nephropathy (2). Epidemiological studies have shown that about 50% of diabetic patients experience different degrees of DPN during the course of the disease (3), and its prevalence is greatly affected by the study population and diagnostic criteria, ranging from 2.4% to 75.1%, and increases significantly with the age and duration of the disease (4, 5).
The development of DPN is closely associated with long-term hyperglycemia. Persistent hyperglycemia can cause damage such as nerve ischemia and demyelination through various metabolic pathways (such as oxidative stress, polyol pathway, and AGEs accumulation), which can lead to peripheral neurological dysfunction (6). DPN usually has an insidious onset, and the clinical manifestations are mainly symmetrical distal paresthesia, accompanied by decreased muscle strength, hyporeflexia and foot circulation disorders (7). If not intervened in time, it can progress to serious complications such as diabetic foot, which is one of the main causes of foot ulcers and non-traumatic amputations (8). Studies show that DPN increases the risk of foot ulcers by 2–3 times, and its associated complications result in up to $176 billion in direct medical expenses annually, significantly increasing the economic burden on patients and the healthcare system (9, 10). In addition, DPN disrupts neuromuscular coordination as well as proprioceptive function, resulting in dysfunction such as abnormal gait and decreased balance (11, 12). Data have shown that patients with DPN have a significantly higher risk of falls than those without DPN, with a fall incidence of 73%, and are closely associated with fractures, disability, and even death (13, 14).
Given the broad clinical impact of DPN and the heavy health burden it imposes, the development of effective interventions has become a focus of current research attention. However, existing treatment strategies primarily focus on strict glycemic control and relief of neuropathic pain (15), with limited effectiveness in improving functional impairments. With the advancement of the concept of non-pharmacological intervention, exercise training as a safe and feasible treatment has been paid more and more attention. The American Diabetes Association (ADA) (16) also explicitly recommends in its guidelines that diabetic patients engage in various forms of exercise to manage DPN. Studies have shown that exercise can effectively enhance skeletal muscle insulin sensitivity, promote blood glucose regulation (17), and improve muscle strength and proprioceptive function, which can improve the overall functional status of DPN patients to some extent (18, 19). Systematic reviews have now assessed the combined effects of exercise interventions in patients with DPN, but most studies (20–23) have focused on their effects on balance function or gait stability, and integrated evidence for improvement in musculoskeletal function and clinical outcomes remains relatively limited. To address the current evidence gap, this meta-analysis aims to systematically assess the effects of different exercise interventions on musculoskeletal function and clinical outcomes in DPN patients, clarify their potential benefits, and provide evidence-based support for optimizing clinical intervention strategies.
2 Methods
This systematic review was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines (24). The review protocol was registered with the International Platform of Registered Systematic Review and Meta-Analysis Protocols (INPLASY) under the registration number INPLASY202340112.
2.1 Search strategy
A comprehensive literature search was conducted in the Cochrane Library, PubMed, Embase, Web of Science, MEDLINE, ClinicalKey, CNKI, Wanfang Data, VIP Database, and the Chinese Biomedical Literature Database, covering publications from database inception to March 2023 in both English and Chinese. Search terms included combinations of subject headings and free-text terms related to the population (“diabetic neuropathies”, “diabetic autonomic neuropathies”, “painful diabetic neuropathies”, “diabetic polyneuropathy”), intervention (“exercise”, “aerobic exercise”, “exercise training”, “physical activity”, “physical exercise”), and study type (“randomized controlled trial”, “RCT”). The detailed search strategy for PubMed is provided in Table 1.
Table 1
| Number | Search terms |
|---|---|
| #1 | (“Diabetic Neuropathies” [MeSH Terms]) OR (“Diabetic Neuropath*” [Text Word]) OR (“Painful Diabetic Neuropath*” [Text Word]) OR (“Diabetic Polyneuropathy” [Text Word]) OR (DPN [Text Word]). |
| #2 | (Exercise [MeSH Terms]) OR (“Exercise Therapy” [Mesh Terms]) OR (“Resistance Training” [Mesh Terms]) OR (“Aerobic Exercise” [Mesh Terms]) OR (“Exercise Training*” [Text Word]) OR (“Physical Activit*” [Text Word]) OR (“Physical Exercise*” [Text Word]). |
| #3 | (“Randomized Controlled Trial” [MeSH Terms]) OR (RCT [Text Word]). |
| #4 | #1 AND #2 AND #3. |
PubMed searchable.
2.2 Eligibility criteria
The inclusion criteria were as follows: ① study subjects: clearly diagnosed DPN patients, aged ≥18 years. ② interventions: control group received usual healthcare, intervention group received exercise training on the basis of usual healthcare. ③ at least one of the following outcome indicators was included: ankle dorsiflexion range of motion left (ROML), ankle plantar flexion range of motion left (ROML), hallux strength, toes strength, glycated hemoglobin (HbA1c), Michigan diabetic neuropathy score (MDNS), Michigan neuropathy screening instrument (MNSI), five-time sit-to-stand test (FTSST test), body mass index (BMI). ④ randomized controlled trials (RCTs) used.
Exclusion criteria were as follows: ① studies of patients with gestational diabetes. ② literatures that cannot get full text or original data. ③ repeated publications. ④ gray literatures such as conference papers.
2.3 Study selection
Two investigators independently screened the literature. Articles with different opinions were arbitrated by a third investigator. Two independent investigators collected and cross-checked the basic information and extracted data. A standardized data extraction form was designed, including author(s)/year, Country, Mean age, Sample size, Duration of diabetes, HbA1c, Exercise program for intervention group, Control group interventions, Follow-up, Outcomes.
2.4 Assessment of bias risk
In this study, we used the risk of bias assessment tool recommended by the Cochrane Collaboration Network (25) to assess the risk of bias of the included RCTs. The tool assesses the risk of systematic bias in studies from seven dimensions: randomization method, allocation concealment, blinded implementation, data integrity, selective reporting, and other potential biases. Each dimension was judged as “low risk”, “unclear risk”, or “high risk” based on literature reports. If all dimensions are low risk, the study is rated as low risk overall; if there is at least one high risk dimension, the study is judged as high risk; if there is one or more unclear risks and the remainder are low risk, the study is rated as unclear risk.
2.5 Data analysis
In this study, meta-analysis was performed using Review Manager software (RevMan 5.4) as recommended by Cochrane Collaboration. Heterogeneity was assessed by the I2 statistic, where I2 values of 25%, 50%, and 75% represented low, moderate, and high heterogeneity (26). If the heterogeneity test results showed P ≥ 0.1 and I2 < 50%, the fixed-effect model was used; if P < 0.1 and I2 ≥ 50%, and the heterogeneity could not be explained by clinical or methodological differences, the random-effects model was used. For the analysis results with high heterogeneity, the possible sources of heterogeneity were further explored through subgroup analysis (with the type of exercise intervention as the grouping variable). When the number of included studies was ≥ 10, Egger's test was used to assess the possibility of publication bias (27). Sensitivity analyses assessed the robustness of the combined results by excluding individual studies one by one. Effect sizes for continuous variables were presented as standardized mean difference (SMD) or weighted mean difference (MD), and 95% confidence intervals (95% CI) were calculated to estimate the precision of the overall effect. The level of heterogeneity was set at α = 0.1, and the remaining tests were set at α = 0.05.
3 Results
3.1 Literature screening
A total of 3,667 relevant studies were retrieved from the databases. After removing duplicates (n = 524) and excluding irrelevant titles and abstracts (n = 3,101), 42 studies were selected for full-text review. Among these, 32 studies were excluded due to not meeting the outcome criteria, not meeting the intervention criteria, or the full text being unavailable. Finally, 10 studies were included in the analysis. The study selection process is illustrated in Figure 1.
Figure 1

Flowchart of the study selection process.
3.2 Characteristics of included studies
The included studies were published between 2013 and 2022, covering six countries. A total of 10 studies were included, comprising seven articles in English and three in Chinese, with a combined sample size of 625 participants (307 in the intervention group and 318 in the control group). The interventions primarily consisted of aerobic and resistance exercises, while other types included tap dance, short-term strength training, and balance training. The frequency of the interventions ranged from two to five times per week, and the duration ranged from 8 to 16 weeks. A brief description of each intervention is provided in Table 2, and detailed information on all included studies is presented in Table 3.
Table 2
| Type of intervention | Definition/description |
|---|---|
| Foot–ankle therapeutic exercise program (28) | An online foot and ankle exercise program targeting both intrinsic and extrinsic muscles with functional, stretching, and strengthening exercises. |
| Progressive resistance training (29) | The external resistance is progressively increased over time to promote gradual and sustained improvements in strength. |
| Supervised resistance training program (30) | All exercise sessions were supervised by an exercise physiologist, with resistance activities adjusted based on real-time blood glucose monitoring. |
| BioDensity™ Resistance Motion Instrument (Resistance exercise) (31) | Resistance training quantified with the bioDensity™ system. |
| Medium-intensity elastic resistance exercise (32) | Moderate-intensity elastic resistance training was performed using a yellow resistance band as an exercise aid. |
| Based on peripheral neuropathy as dominant movement pattern (EPN) (33) | Personalized exercise based on movement impairments caused by nerve damage, such as muscle weakness, sensory loss, and poor balance. |
| Tapping dance (34) | Striking the floor with tap shoes produces rhythm, strengthens ankle and foot muscles, and stimulates the soles of the feet. |
| Short-term strength and balance training (19) | An 8-week program of passive joint and muscle strengthening exercises combined with balance training was implemented. |
| Aerobic exercise (35) | Moderate intensity treadmill exercises. |
Short description of interventions included in this review.
Table 3
| Study | Country | Mean age | Sample size | duration of diabetes | HbA1c (%) | Exercise program for intervention group | Control group interventions | Follow-up (weeks) | Outcomes | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Intervention group | Control group | Intervention group | Control group | Intervention group | Control group | Intervention group | Control group | Type of exercise | Duration (weeks) | Frequency (times/ week) | |||||
| Cruvinel-Júnior et al. (28) | Brazil | 56.5 ± 9.9 | 51.1 ± 10.2 | 15 | 15 | 10.8 ± 7.4 | 18.8 ± 11.8 | — | — | A | 12 | 3 | Usual care | 6, 12, 24 | |
| Monteiro et al. (36) | Brazil | 61.5 ± 11.7 | 60.1 ± 8.9 | 15 | 19 | — | — | — | — | A | 12 | 2 | Usual care | 12, 24 | |
| Khan et al. (29) | Denmark | 63 ± 8 | 63 ± 8 | 15 | 15 | 10 ± 8 | 10 ± 8 | 7.40 ± 1.20 | 7.40 ± 1.20 | B1 | 12 | 2 or 3 | Usual care | 12 | |
| Gholami et al. (30) | Ireland | 63 ± 3 | 64 ± 3 | 15 | 14 | — | — | 9.09 ± 1.82 | 9.97 ± 1.82 | B2 | 12 | 3 | Usual care | 12 | |
| Luo et al. (33) | China | 56.25 ± 2.89 | 55.53 ± 3.80 | 40 | 40 | 11.16 ± 2.26 | 11.33 ± 2.85 | 8.37 ± 0.67 | 8.63 ± 1.16 | C | 12 | — | Educational workshops | 12 | |
| Zhao et al. (34) | China | 63.4 ± 3.55 | 66.6 ± 5.81 | 20 | 20 | 7.81 ± 4.86 | 7.92 ± 3.16 | 6.31 ± 1.21 | 6.86 ± 0.616 | D | 16 | 3 | Usual care | 16 | |
| Yang et al. (31) | China | 59.28 ± 10.18 | 59.52 ± 8.92 | 50 | 50 | — | — | 7.13 ± 1.04 | 6.62 ± 1.21 | B3 | 24 | 4 | Usual care | 12, 24 | |
| Hu and Zhang (32) | China | 75.2 ± 0.5 | 75.8 ± 0.6 | 45 | 45 | — | — | 8.36 ± 0.25 | 8.35 ± 0.21 | B4 | 12 | 3 | Usual care | 12 | |
| Venkataraman et al. (19) | Singapore | 62 | 62 | 67 | 67 | 15.3 ± 10.7 | 15.3 ± 10.7 | 8.5 | 8.5 | E | 8 | 3 | Foot care education | 8, 24 | |
| Dixit et al. (35) | India | 54.40 ± 1.24 | 59.45 ± 1.16 | 29 | 37 | 5.46 ± 0.16 | 6.84 ± 0.14 | — | — | F | 8 | 5 or 6 | Usual care | 8 | |
Characteristics of the studies included in the meta-analysis.
Note: “—”, not reported; A, Foot–ankle therapeutic exercise program; B1, Progressive resistance training; B2, Supervised resistance training program; B3, BioDensity ™ Resistance Motion Instrument (Resistance exercise); B4, Medium-intensity elastic resistance exercise; C, Based on peripheral neuropathy as dominant movement pattern (EPN); D, Tapping dance; E, Short-term strength and balance training; F, Aerobic exercise; , Ankle dorsiflexion range of motion left; , Ankle plantar flexion range of motion left; , Hallux strength; , Toes strength; , glycosylated hemoglobin (HbA1c); , Michigan Diabetic Neuropathy Score (MDNS); , Michigan Neuropathy Screening Instrument (MNSI); , five-time sit-to-stand test (FTSST); , body mass index (BMI).
3.3 Quality of the evidence
Of the 10 included studies, eight studies reported using random number table or lottery to generate random sequences, and the remaining two studies only mentioned randomization and did not describe specific random implementation methods. Two studies were stored in opaque, sequentially numbered, sealed envelopes using an allocation order. Five studies mentioned blinding of outcome assessors, two mentioned that study reports could not blind study subjects and interventions, and the remaining studies did not mention blinding of subjects, researchers, or outcome assessors. All studies described losses to follow-up and withdrawals in detail and reported pre-specified outcome measures. None of the investigators mentioned other risk of bias situations, such as conflicts of interest. The Cochrane risk of bias summary and graph are presented in Figures 2A, B, respectively.
Figure 2

(A) Bias risk assessment of the included studies. (B) Summary of bias risks of the included studies.
3.4 Heterogeneity analysis of HbA1c
A total of five studies (29–33) included HbA1c as the outcome measure. The heterogeneity test showed that there was great heterogeneity among the studies (P < 0.001, I2 = 91%). The random-effects model was used to combine the effect size. The results (Figure 3A) showed that the intervention group was superior to the control group in reducing HbA1c [SMD = −1.44, 95% CI (−2.30, −0.57), P < 0.01]. Sensitivity analysis of included studies revealed little change, suggesting good stability of the results. However, the final results remained statistically significant (P < 0.01), and the intervention group was superior to the control group in reducing HbA1c.
Figure 3

Forest plot and subgroup analysis of HbA1c. (A) Overall analysis. (B) Subgroup analysis.
In the subgroup analysis (Figure 3B), the heterogeneity of resistance exercise was statistically significant among four studies (P < 0.001, I2 = 88%), and significant difference detected between the resistance exercise and control groups [SMD = −1.14, 95% CI (−2.00, −0.27), P < 0.05]. One study reported Based on peripheral neuropathy as dominant movement pattern (EPN), the heterogeneity was not applicable. Overall, there was a difference between the subgroups (P < 0.01, I2 = 87.3%), indicating that the type of exercise was one of the sources of heterogeneity.
3.5 Heterogeneity analysis of BMI and ankle ROML
BMI was included as an outcome measure in three studies (31, 32, 35), and there was no statistically significant difference in heterogeneity (P = 0.49, I2 = 0%, Figure 4A). The fixed effects model combined effect size showed that the intervention group was superior to the control group in reducing BMI [MD = −0.86, 95% CI (−1.15, −0.57), P < 0.001].
Figure 4
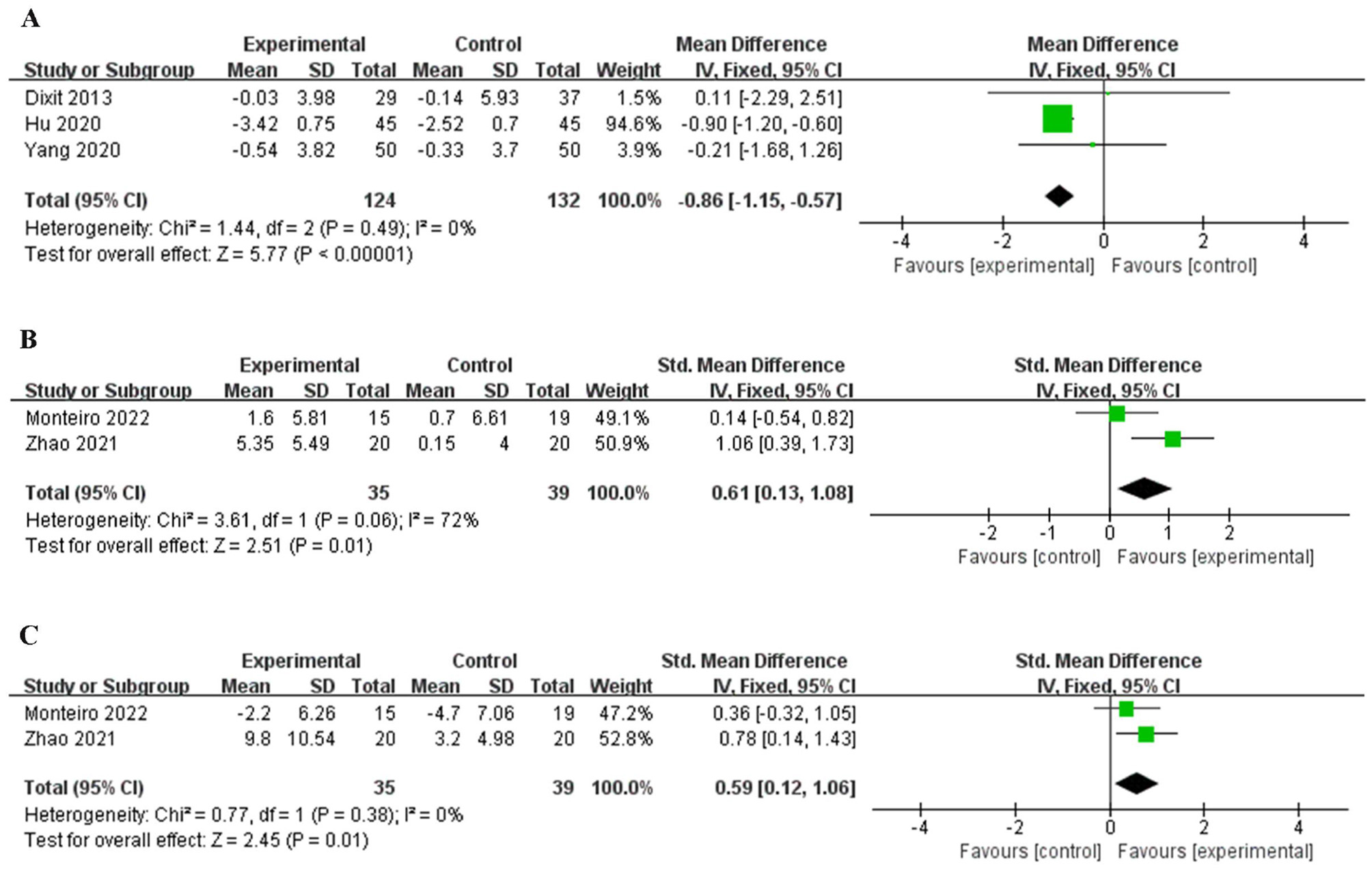
Forest plot of BMI and ankle ROML. BMI (A), ankle dorsiflexion ROML (B), ankle plantar flexion ROML (C).
Two studies (34, 36) included ankle dorsiflexion ROML as the outcome measure. The heterogeneity test showed that there was great heterogeneity among the studies (P = 0.06, I2 = 72%). The results (Figure 4B) showed that the intervention group was superior to the control group in increasing ankle flexion range of motion [SMD = 0.61, 95% CI (0.13, 1.08), P < 0.05].
Two studies (34, 36) included a ankle plantar flexion ROML, but there was no statistically significant difference in heterogeneity (P = 0.38, I2 = 0%, Figure 4C). The random-effects model combined effect size showed that the intervention group was superior to the control group in increasing ankle extension range of motion [SMD = 0.59, 95% CI (0.12, 1.06), P < 0.05].
3.6 Heterogeneity analysis of hallux and toes strength and FTSST test
Two studies (28, 36) included hallux strength and toes strength, and there was no statistically significant difference in heterogeneity between studies (Figures 5A, B). The fixed effects model combined the effect size showed that the intervention group was superior to the control group in increasing hallux strength [MD = 1.89, 95% CI (1.00, 2.78), P < 0.001] and toes strength [MD = 2.51, 95% CI (1.69, 3.33), P < 0.001].
Figure 5

Forest plot of hallux and toes strength and FTSST test. Hallux strength (A), Toes strength (B), FTSST test (C).
Three studies (19, 29, 34) included FTSST test, and there was no statistically significant difference in heterogeneity between studies (P = 0.27, I2 = 24%, Figure 5C). The fixed effects model combined the effect size showed that the intervention group was superior to the control group in enhancing lower extremity functional strength [MD = −2.82, 95% CI (−3.88, −1.76), P < 0.001].
3.7 Heterogeneity analysis of MNSI and MDNS
Three studies (28, 30, 36) used MNSI as an outcome measure, and there was no statistically significant difference in heterogeneity (P = 0.14, I2 = 48%, Figure 6A). The fixed effects model combined the effect size showed that the intervention group was superior to the control group in improving peripheral neuropathy [MD = −1.20, 95% CI (−2.02, −0.38), P < 0.01].
Figure 6

Forest plot of MNSI and MDNS. MNSI (A), MDNS (B).
Other three studies (30, 32, 35) used MDNS as an outcome measure, and there was a statistically significant difference in heterogeneity (P < 0.001, I2 = 98%, Figure 6B). However, there was no statistically significant difference between groups in improving peripheral neuropathy [MD = −3.57, 95% CI (– 7.46, 0.33), P = 0.07].
4 Discussion
This meta-analysis represents a comprehensive synthesis of data currently available for DPN intervention studies. We evaluated 10 randomized controlled trials involving nine different exercise interventions. This systematic review and meta-analysis quantitatively assessed whether exercise training can improve musculoskeletal function and clinical outcomes in DPN patients. The findings indicate that exercise training combined with standard care has a positive effect on improving ankle joint range of motion, hallux and toe strength, and lower limb functional strength, while also reducing HbA1c and BMI levels. Exercise training may have a positive effect on reducing MNSI scores in improving neurological symptom scores, but the existing evidence cannot yet support that exercise training has a clear advantage on MDNS scores.
4.1 Musculoskeletal functional (Ankle ROM, hallux and toes strength, FTSST)
This study showed that exercise intervention, especially ankle joint and distal lower limb muscle training, significantly improved ankle range of motion, toe and lower limb muscle strength in patients with DPN. Meta-analysis of two studies showed that both foot and ankle training and tap dance interventions significantly increased ankle dorsiflexion and plantar flexion range of motion, consistent with previous systematic reviews supporting the effectiveness of physical therapy targeting the foot and ankle in improving joint flexibility (37). The possible mechanism is that this type of training extends joint range of motion by applying mechanical stimulation to the soft tissues around the ankle joint and enhancing its flexibility and neuromuscular control (38). Meta-analysis of two other studies showed that foot and ankle training significantly enhanced hallux and toe muscle strength in patients with DPN, with low heterogeneity among studies, suggesting that it has a consistent effect in enhancing distal muscle strength. This is consistent with the findings of Prókai et al. (39). The latter points out that DPN patients are often accompanied by decreased isokinetic torque of the ankle joint and foot muscle atrophy, especially significantly weakened flexor hallux and minor muscle groups (40). Muscle strength of the hallux and phalanges is key to maintaining gait stability and preventing falls, and its enhancement helps to improve weight-bearing control and dynamic balance, thereby improving overall functional mobility (41).
In addition, a meta-analysis of three studies showed that different types of exercise interventions significantly improve functional strength level of the lower limbs in DPN patients. Among them, progressive resistance training promotes muscle protein synthesis and reverses muscle fiber atrophy by activating the mTOR-p70S6K pathway, delaying muscle loss and enhancing muscle strength more effectively than low-intensity training (42, 43); strength and balance training helps to improve knee extensor strength, motor ability and walking performance (44, 45); while tap dance training improves coordination and balance control ability in addition to enhancing lower limb muscle strength (46). Cruvinel-Júnior et al. (47) further pointed out that foot and ankle exercise training can not only directly improve foot function, but also promote functional recovery by indirect mechanisms such as relieving pain and increasing joint mobility. In summary, exercise intervention, especially foot and ankle training, is effective in improving lower limb function in patients with DPN and can be used as a clinically important rehabilitation strategy.
4.2 Clinical outcomes (BMI, HbA1c)
The clinical manifestations of DPN can be reflected by BMI and HbA1c. This study shows that exercise intervention significantly improved BMI as well as HbA1c in DPN patients. Numerous studies have demonstrated that exercise interventions effectively promote body fat reduction with weight management in different populations (48, 49). This meta-analysis further complements the evidence-based support of exercise for BMI improvement in DPN patients. Exercise lowers BMI by increasing energy expenditure and optimizing metabolic function, particularly by enhancing skeletal muscle insulin sensitivity, promoting lipid metabolism, and reducing fat accumulation (50). The incidence and severity of diabetic neuropathy are positively correlated with the duration of hyperglycemia and blood glucose levels (51), and persistent or fluctuating hyperglycemia can induce irreversible peripheral nerve damage (52). Therefore, active glycemic control is essential for diabetes and its complications, particularly DPN. In this paper, the heterogeneity of different studies of HbA1c is large, but there are statistical differences between the intervention group and the control group, and the intervention group is superior to the control group, which is consistent with the results of previous studies (53, 54). Both EPN and resistance exercise had good effects on improving HbA1c by DPN. Structured resistance exercises >150 min per week in patients with T2DM reduced HbA1c by 0.89% (55). An animal study (56) showed that exercise regulates the metabolic system of the body, and lowers blood glucose levels, thereby reducing the degree of microvascular injury, strengthening nerve conduction function, and ultimately improving peripheral neuropathy symptoms. Therefore, the effect of intensive glycemic control on improving DPN is positive, and the more stringent the glycemic control, the more patients benefit.
4.3 Neuropathy symptom scores (MNSI, MDNS)
MNSI and MDNS are currently among the most commonly used screening tools for assessing DPN. Although studies have shown a significant reduction in the development of abnormal neurological examinations in patients undergoing exercise intervention (57), and improvements in neurological function may be attributed to the strengthening of existing sensory-motor pathways (58), the combined results of this meta-analysis are not fully consistent with previous studies. In the three studies using the MNSI score, the combined results showed a significant advantage of exercise training over usual care in improving neuropathy symptoms. However, in the three studies using the MDNS score, the results did not reach statistical significance and did not clearly indicate a significant effect of exercise training on this scale. The inconsistency in the above results may reflect multiple differences in study populations, intervention protocols, outcome setting, and bias control. Among them, MNSI mainly assesses subjective symptoms and some physical parameters, which are suitable for early screening and are greatly subjectively affected; MDNS focuses on objective neurological function such as muscle strength, reflexes and pain sensation, which is suitable for moderate to severe DPN with more stable results, but requires high study design (59). Although the results of this study suggest that exercise training may help improve neurological function in some indicators, the relevant evidence is still insufficient. In the future, more rigorous, high-quality randomized controlled trials should be conducted in combination with DPN grading and objective indicators (e.g., nerve conduction velocity) to determine the optimal exercise intervention program.
4.4 Strengths and limitations
In this study, a detailed search strategy was employed by searching different types of databases and trial registries. In addition, heterogeneity among studies of most outcome measures was low in comparison to different outcome measures, which indicated that the reliability of this meta-analysis was high. We try to explain the source of heterogeneity through sensitive analysis and subgroup analysis, so that the study results are more persuasive. In addition, we evaluated the effect of exercise training on improving DPN through a meta-analysis with a higher evidence-based level, suggesting that it can improve musculoskeletal function and clinical outcome measures in patients. It can provide clinicians with evidence to help them make better clinical decisions.
However, this study still has several limitations, and caution should be exercised when interpreting the results. First, all included studies lacked long-term follow-up data, and the sustained efficacy of exercise training for DPN remains unclear. Additionally, some studies failed to complete routine follow-up due to the impact of the COVID-19 epidemic, limiting a comprehensive assessment of long-term effects. Second, this study covers a variety of exercise intervention modalities, but most of them are single-center studies with small sample sizes, and there are some limitations in the representativeness and external generalizability of the results. Third, the included studies differed in terms of type of exercise, duration and frequency of intervention, which may lead to some degree of clinical heterogeneity. Finally, some studies lacked sufficient reporting on methodological details such as concealment of random assignment, blinded implementation, and outcome measurement, which may have biased the meta-analysis results. In the future, it is urgent to design standardized large-sample, multicenter randomized controlled trials to verify and expand the findings of this study.
5 Conclusion
Our meta-analysis suggests that exercise interventions, particularly foot and ankle training, contribute to improving DPN related musculoskeletal dysfunction and clinical outcomes compared to usual care measures. These results will provide a basis for adjuvant treatment strategies and clinical decision-making in DPN.
6 Relevance for clinical practice
The American College of Sports Medicine (17) recommends that patients with diabetes, particularly those with DPN, engage in muscle-strengthening exercises 2–4 times per week. However, nurses may encounter challenges in selecting appropriate exercise interventions for patients with DPN, which may affect patients' achievement of optimal treatment outcomes. A better understanding of the latest evidence-based information and potential risks and benefits of exercise interventions for DPN patients is essential for caregivers to optimize clinical decision-making and improve individualized services. In this study, we summarized the effects of structured exercise intervention on musculoskeletal function and clinical outcomes in patients with DPN in recent years through systematic review and meta-analysis, and the results showed that this intervention had a significant effect in improving ankle range of motion, hallux and toe strength, and lower limb muscle strength, and could effectively reduce HbA1c and BMI levels. These findings emphasize the necessity of integrating evidence-based exercise prescriptions into routine care, providing practical intervention directions for nursing practice. Due to heterogeneity in intervention effects across different studies, it is particularly important to develop individualized exercise programs and implement them through a multidisciplinary team comprising rehabilitation therapists, diabetes management specialists, and dietitians. In clinical practice, the effectiveness of exercise interventions should be continuously assessed and dynamically adjusted to ensure long-term patient adherence and achieve optimal treatment outcomes. In addition, future studies should further evaluate the cost-effectiveness and accessibility of exercise interventions and consider the impact of cultural, economic, and psychological factors on patient acceptance. Incorporating patient preferences into intervention design can enhance adherence and promote feasibility.
Statements
Data availability statement
The original contributions presented in the study are included in the article/supplementary material, further inquiries can be directed to the corresponding authors.
Author contributions
JY: Validation, Formal analysis, Software, Visualization, Funding acquisition, Writing – original draft. LL: Formal analysis, Software, Visualization, Writing – original draft. TY: Formal analysis, Software, Visualization, Writing – original draft. YP: Software, Formal analysis, Writing – original draft. QY: Data curation, Software, Writing – original draft. JL: Data curation, Software, Writing – original draft. YH: Data curation, Software, Writing – original draft. XZ: Conceptualization, Supervision, Validation, Writing – review & editing. ZY: Conceptualization, Supervision, Validation, Writing – review & editing.
Funding
The author(s) declare that financial support was received for the research and/or publication of this article. This study was supported by the Science & Technology Programme of the Sichuan Provincial Health Commission (24WSXT033).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that no Gen AI was used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1.
Sun H Saeedi P Karuranga S Pinkepank M Ogurtsova K Duncan BB et al . IDF diabetes atlas: global, regional and country-level diabetes prevalence estimates for 2021 and projections for 2045. Diabetes Res Clin Pract. (2022) 183:109119. 10.1016/j.diabres.2021.109119
2.
Pop-Busui R Ang L Boulton AJM Feldman EL Marcus RL Mizokami-Stout K et al . Diagnosis and treatment of painful diabetic peripheral neuropathy. Compendia. (2022) 2022:1–32. 10.2337/db2022-01
3.
Castelli G Desai KM Cantone RE . Peripheral neuropathy: evaluation and differential diagnosis. Am Fam Physician. (2020) 102:732–9.
4.
Braffett BH Gubitosi-Klug RA Albers JW Feldman EL Martin CL White NH et al . Risk factors for diabetic peripheral neuropathy and cardiovascular autonomic neuropathy in the diabetes control and complications trial/epidemiology of diabetes interventions and complications (DCCT/EDIC) study. Diabetes. (2020) 69:1000–10. 10.2337/db19-1046
5.
Jaiswal M Divers J Dabelea D Isom S Bell RA Martin CL et al . Prevalence of and risk factors for diabetic peripheral neuropathy in youth with type 1 and type 2 diabetes: SEARCH for diabetes in youth study. Diabetes Care. (2017) 40:1226–32. 10.2337/dc17-0179
6.
Herder C Bongaerts BW Rathmann W Heier M Kowall B Koenig W et al . Differential association between biomarkers of subclinical inflammation and painful polyneuropathy: results from the KORA F4 study. Diabetes Care. (2015) 38:91–6. 10.2337/dc14-1403
7.
Chang MC Yang S . Diabetic peripheral neuropathy essentials: a narrative review. Ann Palliat Med. (2023) 12:390–8. 10.21037/apm-22-693
8.
Aso Y . Updates in diabetic neuropathy: a call for new diagnostic and treatment approaches. J Diabetes Investig. (2022) 13:432–4. 10.1111/jdi.13711
9.
Carter MJ . Why is calculating the “true” cost-to-heal wounds so challenging?Adv Wound Care. (2018) 7:371–9. 10.1089/wound.2018.0829
10.
Costa WJT Penha-Silva N Bezerra IMP Paulo dos Santos I Ramos JLS de Castro JM et al . Analysis of diabetes mellitus-related amputations in the state of Espírito Santo, Brazil. Medicina. (2020) 56:287. 10.3390/medicina56060287
11.
Timar B Timar R Gaiţǎ L Oancea C Levai C Lungeanu D . The impact of diabetic neuropathy on balance and on the risk of falls in patients with type 2 diabetes mellitus: a cross-sectional study. PLoS ONE. (2016) 11:e0154654. 10.1371/journal.pone.0154654
12.
Orlando G Balducci S Boulton AJM Degens H Reeves ND . Neuromuscular dysfunction and exercise training in people with diabetic peripheral neuropathy: a narrative review. Diabetes Res Clin Pract. (2022) 183:109183. 10.1016/j.diabres.2021.109183
13.
Khan KS Christensen DH Nicolaisen SK Gylfadottir SS Jensen TS Nielsen JS et al . Falls and fractures associated with type 2 diabetic polyneuropathy: a cross-sectional nationwide questionnaire study. J Diabetes Investig. (2021) 12:1827–34. 10.1111/jdi.13542
14.
Wu B Niu Z Hu F . Study on risk factors of peripheral neuropathy in type 2 diabetes mellitus and establishment of prediction model. Diabetes Metab J. (2021) 45:526–38. 10.4093/dmj.2020.0100
15.
American Diabetes Association . 11. Microvascular complications and foot care: standards of medical care in diabetes-−2021. Diabetes Care. (2021) 44:S151–67. 10.2337/dc21-S011
16.
Colberg SR Sigal RJ Yardley JE Riddell MC Dunstan DW Dempsey PC et al . Physical activity/exercise and diabetes: a position statement of the American diabetes association. Diabetes Care. (2016) 39:2065–79. 10.2337/dc16-1728
17.
Kanaley JA Colberg SR Corcoran MH Malin SK Rodriguez NR Crespo CJ et al . Exercise/physical activity in individuals with type 2 diabetes: a consensus statement from the American College of Sports Medicine. Med Sci Sports Exerc. (2022) 54:353–68. 10.1249/MSS.0000000000002800
18.
Chittrakul J Siviroj P Sungkarat S Sapbamrer R . Multi-system physical exercise intervention for fall prevention and quality of life in pre-frail older adults: a randomized controlled trial. Int J Environ Res Public Health. (2020) 17:3102. 10.3390/ijerph17093102
19.
Venkataraman K Tai BC Khoo EYH Tavintharan S Chandran K Hwang SW et al . Short-term strength and balance training does not improve quality of life but improves functional status in individuals with diabetic peripheral neuropathy: a randomised controlled trial. Diabetologia. (2019) 62:2200–10. 10.1007/s00125-019-04979-7
20.
Thukral N Kaur J Malik M . A systematic review and meta-analysis on efficacy of exercise on posture and balance in patients suffering from diabetic neuropathy. Curr Diabetes Rev. (2021) 17:332–44. 10.2174/1573399816666200703190437
21.
Dixit S Gular K Asiri F . Effect of diverse physical rehabilitative interventions on static postural control in diabetic peripheral neuropathy: a systematic review. Physiother Theory Pract. (2020) 36:679–90. 10.1080/09593985.2018.1491078
22.
de Oliveira Lima RA Piemonte GA Nogueira CR Dos Santos Nunes-Nogueira V . Efficacy of exercise on balance, fear of falling, and risk of falls in patients with diabetic peripheral neuropathy: a systematic review and meta-analysis. Arch Endocrinol Metab. (2021) 65:198–211. 10.20945/2359-3997000000337
23.
Akbari NJ Naimi SS . The effect of exercise therapy on balance in patients with diabetic peripheral neuropathy: a systematic review. J Diabetes Metab Disord. (2022) 21:1861–71. 10.1007/s40200-022-01077-1
24.
Page MJ McKenzie JE Bossuyt PM Boutron I Hoffmann TC Mulrow CD et al . The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. PLoS Med. (2021) 18:e1003583. 10.1371/journal.pmed.1003583
25.
Tarsilla M . Cochrane handbook for systematic reviews of interventions. J Multidiscip Eval. (2010) 6:142–8. 10.56645/jmde.v6i14.284
26.
Huedo-Medina TB Sánchez-Meca J Marín-Martínez F Botella J . Assessing heterogeneity in meta-analysis: Q statistic or I2 index?Psychol Methods. (2006) 11:193–206. 10.1037/1082-989X.11.2.193
27.
Egger M Smith GD Schneider M Minder C . Bias in meta-analysis detected by a simple, graphical test. BMJ. (1997) 315:629–34. 10.1136/bmj.315.7109.629
28.
Cruvinel-Júnior RH Ferreira JSSP Veríssimo JL Monteiro RL Suda EY Silva ÉQ et al . Could an internet-based foot-ankle therapeutic exercise program modify clinical outcomes and gait biomechanics in people with diabetic neuropathy? A clinical proof-of-concept study. Sensors. (2022) 22:9582. 10.3390/s22249582
29.
Khan KS Overgaard K Tankisi H Karlsson P Devantier L Gregersen S et al . Effects of progressive resistance training in individuals with type 2 diabetic polyneuropathy: a randomised assessor-blinded controlled trial. Diabetologia. (2022) 65:620–31. 10.1007/s00125-021-05646-6
30.
Gholami F Khaki R Mirzaei B Howatson G . Resistance training improves nerve conduction and arterial stiffness in older adults with diabetic distal symmetrical polyneuropathy: a randomized controlled trial. Exp Gerontol. (2021) 153:111481. 10.1016/j.exger.2021.111481
31.
Yang XR Liu LY Yang L Li WP Zhang JH . Effect of resistance exercise on peripheral neuropathy in Type 2 diabetes mellitus. J Cent South Univ (Med Sci). (2020) 45:1185–1192. Chinese. 10.11817/j.issn.1672-7347.2020.190505
32.
Hu HY Zhang ZY . Effect of exercise intervention in elderly patients with diabetic peripheral neuropathy. Chin J Gen Pract. (2020) 18:769–72. Chinese. 10.16766/j.cnki.issn.1674-4152.001352
33.
Luo Q Li Y Li J Xiong J . Effects of different exercise interventions on female patients with diabetic peripheral neuropathy. Chin J Woman Child Health Res. (2021) 32:608–13. Chinese.
34.
Zhao Y Cai K Wang Q Hu Y Wei L Gao H . Effect of tap dance on plantar pressure, postural stability and lower body function in older patients at risk of diabetic foot: a randomized controlled trial. BMJ Open Diabetes Res Care. (2021) 9:e001909. 10.1136/bmjdrc-2020-001909
35.
Dixit S Maiya A Shastry B . Effect of aerobic exercise on quality of life in population with diabetic peripheral neuropathy in type 2 diabetes: a single blind, randomized controlled trial. Qual Life Res Int J Qual Life Asp Treat Care Rehabil. (2014) 23:1629–40. 10.1007/s11136-013-0602-7
36.
Monteiro RL Ferreira JSSP Silva ÉQ Cruvinel-Júnior RH Veríssimo JL Bus SA et al . Foot-ankle therapeutic exercise program can improve gait speed in people with diabetic neuropathy: a randomized controlled trial. Sci Rep. (2022) 12:7561. 10.1038/s41598-022-11745-0
37.
Lepesis V Marsden J Rickard A Latour JM Paton J . Systematic review and meta-analysis of the effects of foot and ankle physical therapy, including mobilisations and exercises, in people with diabetic peripheral neuropathy on range of motion, peak plantar pressures and balance. Diabetes Metab Res Rev. (2023) 39:e3692. 10.1002/dmrr.3692
38.
Zvetkova E Koytchev E Ivanov I Ranchev S Antonov A . Biomechanical, healing and therapeutic effects of stretching: a comprehensive review. Appl Sci. (2023) 13:8596. 10.3390/app13158596
39.
Prókai J Murlasits Z Bánhidi M Csóka L Gréci V Atlasz T et al . The effects of a 12-week-long sand exercise training program on neuromechanical and functional parameters in type II diabetic patients with neuropathy. Int J Environ Res Public Health. (2023) 20:5413. 10.3390/ijerph20075413
40.
Le Corre A Caron N Turpin N Dalleau G . Mechanisms underlying altered neuromuscular function in people with DPN. Eur J Appl Physiol. (2023) 123:1433–46. 10.1007/s00421-023-05150-2
41.
Quinlan S Fong Yan A Sinclair P Hunt A . The evidence for improving balance by strengthening the toe flexor muscles: a systematic review. Gait Posture. (2020) 81:56–66. 10.1016/j.gaitpost.2020.07.006
42.
Severinsen K Jakobsen JK Pedersen AR Overgaard K Andersen H . Effects of resistance training and aerobic training on ambulation in chronic stroke. Am J Phys Med Rehabil. (2014) 93:29–42. 10.1097/PHM.0b013e3182a518e1
43.
Schoenfeld BJ Contreras B Vigotsky AD Peterson M . Differential effects of heavy versus moderate loads on measures of strength and hypertrophy in resistance-trained men. J Sports Sci Med. (2016) 15:715–22.
44.
Sadeghi H Jehu DA Daneshjoo A Shakoor E Razeghi M Amani A et al . Effects of 8 weeks of balance training, virtual reality training, and combined exercise on lower limb muscle strength, balance, and functional mobility among older men: a randomized controlled trial. Sports Health. (2021) 13:606–12. 10.1177/1941738120986803
45.
Holviala J Kraemer WJ Sillanpää E Karppinen H Avela J Kauhanen A et al . Effects of strength, endurance and combined training on muscle strength, walking speed and dynamic balance in aging men. Eur J Appl Physiol. (2012) 112:1335–47. 10.1007/s00421-011-2089-7
46.
Rocha P McClelland J Sparrow T Morris ME . The biomechanics and motor control of tap dancing. J Dance Med Sci Off Publ Int Assoc Dance Med Sci. (2017) 21:123–9. 10.12678/1089-313X.21.3.123
47.
Cruvinel-Júnior RH Ferreira JSSP Sacco ICN . How do foot-ankle exercises improve clinical and plantar pressure outcomes in people with diabetic peripheral neuropathy? A mediation analysis of a randomized controlled trial. Braz J Phys Ther. (2025) 29:101213. 10.1016/j.bjpt.2025.101213
48.
Hsu KJ Chien KY Tsai SC Tsai YS Liao YH Chen JJ et al . Effects of exercise alone or in combination with high-protein diet on muscle function, aerobic capacity, and physical function in middle-aged obese adults: a randomized controlled trial. J Nutr Health Aging. (2021) 25:727–34. 10.1007/s12603-021-1599-1
49.
Siu PM Yu AP Chin EC Yu DS Hui SS Woo J et al . Effects of tai chi or conventional exercise on central obesity in middle-aged and older adults : a three-group randomized controlled trial. Ann Intern Med. (2021) 174:1050–7. 10.7326/M20-7014
50.
Hawley JA Lessard SJ . Exercise training-induced improvements in insulin action. Acta Physiol. (2008) 192:127–35. 10.1111/j.1748-1716.2007.01783.x
51.
Sone H Mizuno S Yamada N . Vascular risk factors and diabetic neuropathy. N Engl J Med. (2005) 352:1925–7. 10.1056/NEJM200505053521817
52.
Cameron NE Eaton SE Cotter MA Tesfaye S . Vascular factors and metabolic interactions in the pathogenesis of diabetic neuropathy. Diabetologia. (2001) 44:1973–88. 10.1007/s001250100001
53.
Callaghan BC Little AA Feldman EL Hughes RAC . Enhanced glucose control for preventing and treating diabetic neuropathy. Cochrane Database Syst Rev. (2012) 2012:CD007543. 10.1002/14651858.CD007543.pub2
54.
Kluding PM Bareiss SK Hastings M Marcus RL Sinacore DR Mueller MJ . Physical training and activity in people with diabetic peripheral neuropathy: paradigm shift. Phys Ther. (2017) 97:31–43. 10.2522/ptj.20160124
55.
Umpierre D Ribeiro PAB Kramer CK Leitão CB Zucatti ATN Azevedo MJ et al . Physical activity advice only or structured exercise training and association with HbA1c levels in type 2 diabetes: a systematic review and meta-analysis. JAMA. (2011) 305:1790–9. 10.1001/jama.2011.576
56.
Kosacka J Woidt K Toyka KV Paeschke S Klöting N Bechmann I et al . The role of dietary non-heme iron load and peripheral nerve inflammation in the development of peripheral neuropathy (PN) in obese non-diabetic leptin-deficient ob/ob mice. Neurol Res. (2019) 41:341–53. 10.1080/01616412.2018.1564191
57.
Singleton JR Marcus RL Jackson JE K Lessard M Graham TE Smith AG . Exercise increases cutaneous nerve density in diabetic patients without neuropathy. Ann Clin Transl Neurol. (2014) 1:844–9. 10.1002/acn3.125
58.
de Leon RD Hodgson JA Roy RR Edgerton VR . Locomotor capacity attributable to step training versus spontaneous recovery after spinalization in adult cats. J Neurophysiol. (1998) 79:1329–40. 10.1152/jn.1998.79.3.1329
59.
Wang Q Cao L Luan X . Review of clinical application of peripheral neuropathy scales. J Shanghai Jiaotong Univ (med Sci). (2021) 41:1518. 10.3969/j.issn.1674-8115.2021.11.018
Summary
Keywords
diabetic peripheral neuropathy, clinical outcomes, musculoskeletal function, exercise, meta-analysis
Citation
Yang J, Li L, Ye T, Pu Y, Yao Q, Luo J, Huang Y, Zhang X and Yang Z (2025) Effectiveness of exercise on musculoskeletal function and clinical outcomes in patients with diabetic peripheral neuropathy: a systematic review and meta-analysis. Front. Neurol. 16:1610955. doi: 10.3389/fneur.2025.1610955
Received
14 April 2025
Accepted
11 August 2025
Published
29 August 2025
Volume
16 - 2025
Edited by
Triantafyllos Didangelos, University General Hospital of Thessaloniki AHEPA, Greece
Reviewed by
Lorenzo Brognara, University of Bologna, Italy
Anwar H. Siddiqui, Aligarh Muslim University, India
Updates
Copyright
© 2025 Yang, Li, Ye, Pu, Yao, Luo, Huang, Zhang and Yang.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Xianqin Zhang sclzzxq@hotmail.comZheng Yang yzixjj@163.com
†These authors have contributed equally to this work
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.