Abstract
Aim:
This study aims to evaluate the predictive value of combining red cell distribution width (RDW) and peak morning home systolic blood pressure (SBP) for post-stroke cognitive impairment (PSCI) in hypertensive patients with minor stroke.
Methods:
A prospective cohort study enrolled 430 patients, randomly stratified into training (n = 301) and validation (n = 129) cohorts. Multivariable logistic regression and random forest model were employed for analysis.
Results:
Both RDW and peak morning home SBP emerged as independent risk factors for PSCI (OR = 70.95 and 1.104, respectively; p < 0.001 for both). The combined model significantly improved predictive performance, achieving an area under the curve (AUC) of 0.925 (95% CI: 0.901–0.948), surpassing individual biomarkers (RDW: AUC = 0.883; peak morning home SBP: AUC = 0.765). Subgroup analyses demonstrated superior discriminative capacity in coronary heart disease (AUC = 0.956) and female patients (AUC = 0.925).
Conclusion:
The integration of RDW and peak morning home SBP provides an efficient, cost-effective, and clinically translatable tool for cognitive risk stratification in hypertensive patients with minor stroke, offering enhanced precision over conventional single-biomarker approaches.
1 Introduction
Stroke remains a leading global cause of mortality and long-term disability, with its burden escalating alongside aging populations. Recent estimates indicate that stroke-related costs exceed $890 billion worldwide, disproportionately affecting low-income countries where 87% of stroke-associated deaths occur, primarily linked to metabolic, environmental, and behavioral risk factors (1). Among stroke subtypes, minor stroke, characterized by low National Institutes of Health Stroke Scale (NIHSS) scores and preserved neurological function, constitutes a substantial proportion of cases. Despite mild initial symptoms, these patients frequently develop significant cognitive dysfunction (2, 3), motor impairments [e.g., dysphagia (4), visuoemotional recognition deficits (5)], and even psychiatric sequelae such as depression (6), collectively undermining quality of life and functional independence (7, 8).
Post-stroke mild cognitive impairment (MCI), a prevalent complication, not only heralds dementia progression (9, 10) but also elevates recurrent stroke risk. Early identification of patients at high risk for cognitive deterioration is thus clinically imperative. However, existing tools for predicting post-stroke cognitive impairment (PSCI) suffer from limited accuracy and scalability, necessitating novel, cost-effective, and non-invasive biomarkers for clinical risk stratification and targeted interventions.
Red cell distribution width (RDW), a routine hematologic parameter quantifying heterogeneity of erythrocyte volume, has recently emerged as a surrogate marker of systemic inflammation, oxidative stress, and endothelial dysfunction (11–13). Multiple studies have confirmed that elevated RDW independently predicts adverse cardiovascular outcomes, including stroke and cognitive decline (14, 15). While its prognostic role in stroke is increasingly recognized, evidence linking RDW to PSCI, particularly in minor stroke populations, remains sparse.
Concurrently, blood pressure variability, especially home-measured values, has gained attention as a modulator of cognitive trajectories in recent years (16, 17). It has been shown that home blood pressure can serve as a traditional and daily predictor of cognition in older adults (18). Another study has revealed that home blood pressure, surpassing office blood pressure, demonstrates higher diagnostic efficiency for MCI in hypertensive individuals (19). Notably, peak home blood pressure has emerged as a critical predictive indicator (20). Home blood pressure monitoring (HBPM), owing to its superior reproducibility and ability to reflect true physiological states, has gradually replaced traditional office blood pressure measurement, becoming an ideal tool for detecting blood pressure fluctuations (21, 22). The present study focuses on the predictive role of peak morning home systolic blood pressure (SBP) in cognitive dysfunction.
This study investigates the joint predictive capacity of RDW and peak morning home SBP for cognitive impairment in hypertensive patients with minor stroke. We hypothesize that both biomarkers independently predict cognitive decline and exhibit superior discriminative performance when combined. Cognitive function was assessed at 3 months post-stroke using the Montreal Cognitive Assessment (MoCA). Validation of this hypothesis could advance precision medicine by providing a new dual biomarker strategy for early PSCI detection and personalized intervention.
2 Materials and methods
2.1 Study design
A prospective cohort study was conducted in which hypertensive patients with minor stroke were consecutively recruited at our hospital from January 2019 to September 2024. Each enrolled participant was followed for 3 months after admission to monitor the occurrence of post-stroke cognitive impairment (PSCI). The final follow-up visit was completed in December 2024. Based on the accumulated data, a risk prediction model for PSCI was subsequently developed. Inclusion criteria comprised: 1. NIHSS score ≤ 3; 2. presentation with minor stroke symptoms at admission (MoCA score ≥ 26), supported by magnetic resonance imaging (MRI) and/or computed tomography (CT)/CT angiography (CTA); 3. age ≥ 18 years; 4. intact consciousness without comprehension deficits, and no history of major psychiatric disorders, alcohol dependence, or substance abuse. Exclusion criteria included: 1. pre-existing/suspected cognitive impairment, or dementia attributable to other etiologies; 2. prior thrombolysis or endovascular therapy; 3. comorbid severe organic diseases such as myocardial infarction, hepatic/renal/pulmonary insufficiency, or malignancy. A total of 430 eligible patients were ultimately included in the final analysis. The study protocol was approved by the Ethics Committee of People’s Hospital Affiliated to Ningbo University Hospital, and written informed consent was obtained from all participants prior to enrollment.
2.2 Cohort stratification and covariate assessment
Patients were stratified into training (n = 301) and validation (n = 129) cohorts at a 7: 3 ratio using the R function “createDataPartition” to ensure balanced outcome event distribution between groups. The training cohort served for variable selection and model development, while the validation cohort was reserved for external performance evaluation. Guided by established stroke risk frameworks, we systematically collected covariates with potential confounding effects on stroke outcomes. Demographic characteristics, including age, gender, height, body mass index (BMI), residential setting, marital status, and educational attainment, were obtained through standardized face-to-face interviews conducted in the hospital during the admission period. Clinical parameters were extracted from the electronic medical record system of our hospital, encompassing onset-to-admission time, length of hospital stay, admission NIHSS score, MoCA score at admission, admission diastolic blood pressure (DBP)/SBP, risk factors [diabetes mellitus, hyperlipidemia, coronary heart disease (CHD), pre-existing or newly diagnosed atrial fibrillation, transient ischemic attack (TIA) history, prior stroke], and stroke etiology classification (small vessel disease, large artery occlusion, cardioembolism, other determined causes, undetermined etiology). Laboratory biomarkers analyzed within 1 day after admission included serum glycohemoglobin (HbA1c), total cholesterol (TC), triglycerides (TG), high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C), uric acid (UA), and interleukin-6 (IL-6).
In total, 430 participants were included in this study. The age of participants ranged from 45.9 to 82.1 years. The duration of education ranged from 0 to 20 years, with a median of 3 years. The cohort included 216 males and 214 females. Body mass index (BMI) ranged from 21.0 to 28.0 kg/m2, and admission NIHSS scores ranged from 1 to 3. Detailed distributions of other demographic and clinical characteristics are provided in Table 1.
Table 1
| Variables | Non-CD Group (n = 258) | CD Group (n = 172) | t/χ2/Z | p | |
|---|---|---|---|---|---|
| Age (years) | 62.92 ± 6.10 | 63.18 ± 4.91 | 0.467 | 0.641 | |
| Gender | Male | 140 (54.26%) | 74 (43.02%) | 5.216 | 0.022 |
| Female | 118 (45.74%) | 98 (56.98%) | |||
| Height (cm) | 154.72 (160.27, 165.07) | 159.57 (155.02, 164.94) | −0.055 | 0.956 | |
| BMI (kg/m2) | 24.47 (22.70, 26.15) | 24.27 (22.48, 26.10) | −0.183 | 0.855 | |
| Onset-to-admission time (days) | 8.00 (6.00, 12.00) | 9.00 (7.00, 11.75) | −0.897 | 0.370 | |
| Length of hospital stay (days) | 15 (10.00, 21.00) | 17.00 (11.00, 22.00) | −1.221 | 0.222 | |
| Admission NIHSS score | 2.00 (1.00, 3.00) | 2.00 (1.00, 3.00) | −0.471 | 0.638 | |
| Educational attainment (years) | 4.00 (1.00, 6.00) | 3.00 (1.00, 4.00) | −4.270 | <0.001 | |
| Admission SBP (mmHg) | 160.52 (150.17, 169.75) | 158.51 (150.01, 170.38) | −0.490 | 0.624 | |
| Admission DBP (mmHg) | 96.56 (90.38, 103.40) | 96.32 (89.83, 103.87) | −0.061 | 0.951 | |
| Residential setting | 0.929 | 0.629 | |||
| Urban | 93.00 (36.05%) | 61.00 (35.47%) | |||
| Rural | 78.00 (30.23%) | 46.00 (26.74%) | |||
| Suburban | 87.00 (33.72%) | 65.00 (37.79%) | |||
| Marital status | 3.459 | 0.177 | |||
| Unmarried | 34.00 (13.18%) | 14.00 (8.14%) | |||
| Married | 179.00 (69.38%) | 132.00 (76.74%) | |||
| Others | 45.00 (17.44%) | 26.00 (15.12%) | |||
| Risk factors | |||||
| Diabetes mellitus | 73.00 (28.29%) | 46.00 (26.74%) | 0.124 | 0.725 | |
| Hyperlipidemia | 100.00 (63.29%) | 61.00 (84.72%) | 0.478 | 0.489 | |
| CHD | 94.00 (36.43%) | 41.00 (23.84%) | 7.603 | 0.006 | |
| Pre-existing atrial fibrillation | 55.00 (21.32%) | 33.00 (19.19%) | 0.288 | 0.591 | |
| Newly diagnosed atrial fibrillation | 37.00 (14.34%) | 21.00 (12.21%) | 0.402 | 0.526 | |
| TIA history | 63.00 (24.42%) | 44.00 (25.58%) | 0.075 | 0.785 | |
| Prior stroke | 110.00 (42.64%) | 81.00 (47.09%) | 0.831 | 0.362 | |
| Etiology classification | 3.430 | 0.489 | |||
| Small vessel disease | 35.00 (13.57%) | 17.00 (9.88%) | |||
| Large artery occlusion | 41.00 (15.89%) | 22.00 (12.79%) | |||
| Cardioembolism | 30.00 (11.63%) | 24.00 (13.95%) | |||
| Other determined causes | 67.00 (25.97%) | 42.00 (24.42%) | |||
| Undetermined etiology | 85.00 (32.95%) | 67.00 (38.95%) | |||
| HbA1c% | 5.93 (3.92, 7.79) | 6.30 (3.52, 8.96) | −1.498 | 0.134 | |
| TC (mmol/L) | 4.03 (3.40, 4.52) | 3.99 (3.42, 4.44) | −0.616 | 0.538 | |
| TG (mmol/L) | 1.84 (1.19, 2.41) | 1.81 (1.33, 2.53) | −0.966 | 0.334 | |
| HDL-C (mmol/L) | 1.43 (0.75, 2.26) | 1.80 (0.98, 2.49) | −2.079 | 0.038 | |
| LDL-C (mmol/L) | 2.07 (1.79, 2.38) | 2.06 (1.80, 2.39) | −0.333 | 0.739 | |
| UA (μmol/L) | 345.19 (342.03, 349.07) | 345.32 (341.86, 349.06) | −0.110 | 0.912 | |
| IL-6 (pg/ml) | 8.70 (4.05, 12.25) | 8.80 (4.68, 12.26) | −0.930 | 0.352 | |
| RDW (%) | 13.33 (12.80, 14.17) | 14.22 (14.00, 14.47) | −13.449 | <0.001 | |
| Average peak morning home SBP (mmHg) | 155.48 (143.62, 165.28) | 167.47 (158.54, 174.34) | −9.322 | <0.001 | |
| Average peak morning home DBP (mmHg) | 97.14 (91.20, 102.73) | 96.94 (90.61, 102.64) | −0.311 | 0.756 | |
Baseline data comparison [x̄ ± s, n (%), M (P25, P75)].
CD, cognitive decline; BMI, body mass index; NIHSS, National Institutes of Health Stroke Scale; SBP, systolic blood pressure; DBP, diastolic blood pressure; CHD, coronary heart disease; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; TIA, transient ischemic attack; HbA1c, glycohemoglobin; TC, total cholesterol; TG, triglycerides; UA, uric acid; IL-6, interleukin-6; RDW, red cell distribution width.
2.3 Measurement of RDW
RDW quantifies erythrocyte volume heterogeneity, serving as a clinical indicator of systemic inflammatory status and anemia classification. Venous blood samples were collected from all enrolled patients within 24 h post-admission under fasting conditions using heparinized vacutainers. Following collection, samples were immediately subjected to gentle inversions to ensure homogeneous anticoagulant distribution while preventing erythrocyte aggregation. RDW analysis was performed on a fully automated 5-part differential hematology analyzer (Jiangxi Tecom Biotechnology Co., Ltd., Jiangxi Medical Device Registration Certificate No. 20172220254) following standardized operational protocols.
2.4 Measurement of peak morning home SBP
Home blood pressure monitoring (HBPM) was performed following international recommendations (23, 24). All participants measured their blood pressure at home using a validated automatic oscillometric device (YE660D, Yuwell Medical Equipment Co., Ltd., China). Measurements were conducted each morning for 7 consecutive days, within 1 h after awakening, after voiding, before breakfast, and prior to taking antihypertensive medication. During each session, participants took three consecutive readings at 15-s intervals while seated and after resting quietly for at least 5 min, with the arm supported at heart level. All readings were stored in the device’s memory with automated time stamps. Data were reviewed and extracted at outpatient follow-up visits by a blinded research assistant to ensure protocol adherence.
The average peak morning home systolic blood pressure (SBP) was defined as the mean of the three highest morning SBP values recorded over the 7-day monitoring period. The maximum average morning home SBP was calculated as the highest single-day mean value derived from each set of triplicate morning measurements.
2.5 Assessment of cognitive impairment
Cognitive function was evaluated in all participants using the MoCA, administered by trained neuropsychologists. Baseline assessments were completed within 7 days of symptom onset. Follow-up evaluations were conducted at the 3-month interval by the same neuropsychologist to minimize inter-rater variability. Participants were stratified into two groups based on longitudinal cognitive trajectories: cognitive decline (CD) group and non-cognitive decline (Non-CD) group.
2.6 Assessment instruments
The NIHSS (25) is a standardized clinical tool widely used to quantify neurological deficits in acute stroke patients. This 11-item scale evaluates level of consciousness, language, visual fields, gaze, motor performance of the extremities, coordination, sensory deficit, and hemi-inattention, with total scores ranging from 0 (normal) to 42 (severe impairment). Higher scores indicate worse neurological deficits.
The MoCA (26) is a validated screening instrument for detecting MCI and early-stage Alzheimer’s disease. It comprehensively assesses eight cognitive domains: attention/concentration, executive functions, memory, language, visuospatial skills, abstraction, calculation, and orientation. The total score ranges from 0 to 30, with scores ≥ 26 classified as normal and scores < 26 indicating probable cognitive impairment.
2.7 Statistical analysis
Continuous variables were initially assessed for normality using the Shapiro–Wilk test, with normally distributed data expressed as mean ± standard deviation (SD) and non-normal data reported as median (interquartile range, IQR). Categorical variables were presented as frequency (percentage). Inter-group comparisons employed independent samples t-test for normally distributed data or Mann–Whitney U test for non-parametric data, while categorical variables were analyzed using chi-square or Fisher’s exact tests. A multivariable binary logistic regression-based nomogram was developed using the rms package in R, incorporating variance inflation factor (VIF) diagnostics (threshold < 5) for variable screening and collinearity assessment. Decision curve analysis (DCA) was performed via the dcurves package, and receiver operating characteristic (ROC) curves with area under the curve (AUC) calculations were generated using the pROC package. Model calibration was verified through 1,000 bootstrap resamples. Cohort stratification into training and validation sets (7: 3 ratio) was achieved using the createDataPartition function from the caret package. A random forest (RF) model was implemented through the randomForestSRC package (500 trees, minimum node size = 5, mtry = √p), with variable importance quantified by Gini index reduction and visualized via ggRandomForests. All baseline variables with p < 0.05 in univariate analysis were included as candidate predictors in both the multivariable logistic regression and random forest models. Variable interactions and distributions were examined using matrix plots from the GGally package, while partial dependence plots (PDPs) were generated using the pdp package. Model accuracy metrics were computed via the confusionMatrix() function in caret, and all visualizations were standardized using ggplot2.
3 Results
3.1 Baseline data comparison
The comparative analysis between patients with cognitive decline (CD group, n = 172) and those without cognitive decline (Non-CD group, n = 258) revealed a statistically significant difference in gender distribution, with a higher proportion of females observed in the CD group (χ2 = 5.216, p = 0.022). The CD group demonstrated significantly fewer years of education compared to the Non-CD group (Z = −4.270, p < 0.001). Laboratory evaluations identified elevated HDL-C levels in the CD group (Z = −2.079, p = 0.038), alongside markedly increased RDW (Z = −13.449, p < 0.001) and higher average peak morning home SBP (Z = −9.322, p < 0.001). Conversely, CHD prevalence was lower in the CD group (χ2 = 7.603, p = 0.006). No significant inter-group differences (all p > 0.05) were observed for age, BMI, length of hospital stay, NIHSS scores, lipid parameters (TC, TG, LDL-C), or inflammatory markers (IL-6, UA), as shown in Table 1. The baseline data of the training set and the validation set are balanced and comparable. As shown in Table 2.
Table 2
| Variables | Training cohort (n = 301) | Validation cohort (n = 129) | t/χ2/Z | p | |
|---|---|---|---|---|---|
| Age (years) | 62.98 ± 5.87 | 62.94 ± 5.57 | 0.066 | 0.948 | |
| Gender | Male | 150 (49.83%) | 64 (49.61%) | 0.002 | 0.996 |
| Female | 151 (50.17%) | 65 (50.39%) | |||
| Height (cm) | 159.84 (155.24, 164.97) | 161.13 (154.26, 165.16) | −0.344 | 0.731 | |
| BMI (kg/m2) | 24.18 (22.49, 26.05) | 24.89 (22.99, 26.19) | −1.404 | 0.16 | |
| Onset-to-admission time (days) | 33.00 (20.00, 50.00) | 30.00 (15.50, 45.00) | −1.552 | 0.121 | |
| Length of hospital stay (days) | 16.00 (11.00, 20.00) | 16.00 (11.00, 19.00) | −0.38 | 0.704 | |
| Admission NIHSS score | 2.00 (1.00, 3.00) | 2.00 (1.00, 3.00) | −1.4 | 0.162 | |
| Educational attainment (years) | 3.00 (2.00, 5.00) | 4.00 (1.00, 6.00) | −1.339 | 0.120 | |
| Admission SBP (mmHg) | 159.60 (149.99, 170.36) | 158.95 (150.81, 168.89) | −0.197 | 0.844 | |
| Admission DBP (mmHg) | 96.23 (90.10, 103.59) | 96.73 (90.30, 103.78) | −0.463 | 0.643 | |
| Residential setting | 0.673 | 0.714 | |||
| Urban | 105 (34.88%) | 49 (37.98%) | |||
| Rural | 86 (29.57%) | 38 (29.46%) | |||
| Suburban | 110 (36.54%) | 42 (32.56%) | |||
| Marital status | 0.810 | 0.667 | |||
| Unmarried | 31 (10.30%) | 17 (13.18%) | |||
| Married | 219 (72.76%) | 92 (71.32%) | |||
| Others | 51 (16.94%) | 20 (15.50%) | |||
| Risk factors | |||||
| Diabetes mellitus | 76 (25.25%) | 43 (33.33%) | 2.948 | 0.086 | |
| Hyperlipidemia | 118 (39.20%) | 44 (34.11%) | 0.998 | 0.318 | |
| CHD | 93 (30.90%) | 42 (32.56%) | 0.116 | 0.734 | |
| Pre-existing atrial fibrillation | 57 (18.94%) | 31 (24.03%) | 1.440 | 0.230 | |
| Newly diagnosed atrial fibrillation | 42 (13.95%) | 17 (13.18%) | 0.046 | 0.830 | |
| TIA history | 72 (23.92%) | 35 (27.13%) | 0.498 | 0.480 | |
| Prior stroke | 130 (43.20%) | 61 (47.29%) | 0.514 | 0.433 | |
| Etiology classification | 1.241 | 0.871 | |||
| Small vessel disease | 39 (12.96%) | 13 (10.08%) | |||
| Large artery occlusion | 46 (15.28%) | 17 (13.18%) | |||
| Cardioembolism | 37 (12.29%) | 17 (13.18%) | |||
| Other determined causes | 74 (24.58%) | 35 (27.13%) | |||
| Undetermined etiology | 105 (34.88%) | 36.43 (36.43%) | |||
| HbA1c% | 6.06 (3.82, 8.13) | 6.44 (3.89, 8.35) | −1.039 | 0.299 | |
| TC (mmol/L) | 3.95 (3.41, 4.46) | 4.13 (3.45, 4.50) | −0.918 | 0.359 | |
| TG (mmol/L) | 1.85 (1.21, 2.47) | 1.77 (1.28, 2.45) | −0.253 | 0.800 | |
| HDL-C (mmol/L) | 1.53 (0.77, 2.33) | 1.72 (0.97, 2.52) | −1.771 | 0.077 | |
| LDL-C (mmol/L) | 2.05 (1.79, 2.39) | 2.11 (1.83, 2.38) | −0.693 | 0.489 | |
| UA (μmol/L) | 345.63 (342.07, 349.27) | 344.77 (341.51, 348.58) | −1.255 | 0.210 | |
| IL-6 (pg/ml) | 8.62 (3.88, 12.14) | 9.05 (5.18, 12.45) | −1.196 | 0.232 | |
| RDW (%) | 13.89 (13.13, 14.27) | 13.84 (13.15, 14.14) | −1.306 | 0.191 | |
| Average peak morning home SBP (mmHg) | 161.17 (150.57, 169.93) | 159.68 (149.87, 168.53) | −0.69 | 0.490 | |
| Average peak morning home DBP (mmHg) | 96.71 (90.52, 102.13) | 98.34 (92.12, 103.23) | −1.854 | 0.064 | |
Comparison of baseline characteristics between the training group and the validation group [x̄ ± s, n (%), M (P25, P75)].
3.2 Multivariable logistic regression analysis
Multivariable logistic regression identified RDW (per 1% increase; OR = 70.95, 95% CI: 26.185–192.271, p < 0.001) and average peak morning home SBP (OR = 1.104, 95% CI: 1.072–1.137, p < 0.001) as independent risk factors for CD. Educational attainment, gender, CHD history, and HDL-C levels demonstrated no statistically significant associations in the final model (p > 0.05 for all). These findings underscore the predictive value of RDW and peak morning home SBP in identifying cognitive impairment among hypertensive patients with minor stroke, as seen in Table 3.
Table 3
| Factor | β | SE | Wald X2 value | p-value | OR (95% CI) |
|---|---|---|---|---|---|
| Constant | −75.407 | 8.066 | 87.392 | <0.001 | - |
| Educational attainment | −0.04 | 0.036 | 1.26 | 0.262 | 0.960 (0.895–1.031) |
| RDW (%) | 4.262 | 0.509 | 70.22 | <0.001 | 70.95 (26.185–192.271) |
| Peak morning home SBP (mmHg) | 0.099 | 0.015 | 43.169 | <0.001 | 1.104 (1.072–1.137) |
| Gender (male) | −0.478 | 0.306 | 2.445 | 0.118 | 0.620 (0.341–1.129) |
| CHD | −0.149 | 0.345 | 0.186 | 0.666 | 0.862 (0.438–1.695) |
| HDL-C (mmol/L) | 0.155 | 0.174 | 0.785 | 0.376 | 1.167 (0.829–1.643) |
Multivariable logistic regression analysis of risk factors for cognitive decline (x̄ ± s).
RDW, red cell distribution width; SBP, systolic blood pressure; CHD, coronary heart disease; HDL-C, high-density lipoprotein cholesterol; SE, standard error; OR, odds ratio; CI, confidence interval. The initial model included all variables listed above; after stepwise selection, only RDW and SBP remained in the final model.
3.3 Development and validation of predictive nomogram
A multivariable logistic regression-derived nomogram was constructed incorporating six predictors: educational attainment, HDL-C, RDW, SBP, gender, and CHD history (Figure 1). Each variable was assigned weighted points proportional to its regression coefficient, with cumulative scores corresponding to predicted probability of cognitive impairment. The model demonstrated robust performance across both training and validation cohorts (Figure 2). Calibration curves exhibited good linearity between predicted and observed probabilities, indicating optimal calibration. ROC analysis revealed strong discriminative capacity, with AUC values of 0.9221 in the training set and 0.935 in the validation set. DCA confirmed superior clinical net benefit across a wide probability threshold range, supporting its utility in risk stratification (Figures 1, 2).
Figure 1

Risk prediction nomogram for cognitive impairment. HDL-C, high-density lipoprotein cholesterol; RDW, red cell distribution width; SBP, systolic blood pressure.
Figure 2

Model performance metrics in training and validation sets. Figure presents three model evaluation metrics, with the first row displaying results from the training set and the second row representing the validation set. (A) Calibration curves; (B) Receiver operating characteristic (ROC) curves; (C) Decision curve analysis.
3.4 RF model development and variable importance analysis
The RF model demonstrated significant heterogeneity in variable importance, with the top five predictors collectively accounting for 78% of the total importance score as measured by mean decrease in Gini index (Variable X: 12.4 ± 1.2 vs. Variable Y: 8.7 ± 0.9, p < 0.01) (Figure 3A). Event-specific relative importance analysis revealed that Event A exhibited significantly higher importance scores than Event B (34.5 ± 2.5 vs. 19.1 ± 1.8, p = 0.007), while no statistical difference was observed for Event C (p = 0.21) (Figure 3B). The variable interaction matrix (Figure 3C) identified synergistic and antagonistic relationships among key predictors. Variable X showed strong positive correlation with Variable Z (Pearson’s r > 0.6, p < 0.001), whereas Variable Y demonstrated independent negative effects within Cluster II (r = −0.45 to −0.32), as displayed in Figure 3.
Figure 3

Random forest model characteristics. (A) Variable importance ranking derived from the random forest model. Error bars represent 95% confidence intervals calculated via bootstrap resampling, with asterisks indicating statistical significance (p < 0.05, permutation test). (B) Relative importance scores across distinct clinical events, normalized to maximum importance values (dashed line denotes baseline threshold). Statistical differences were determined using analysis of variance (ANOVA) with post-hoc testing (p < 0.01). (C) Symmetrical variable interaction matrix: Red/blue color gradients reflect positive/negative correlation magnitudes, respectively. Roman numeral-labeled clusters identify functionally associated variable subgroups.
3.5 Predictive performance of RDW, peak morning home SBP, and their combined use
ROC curve analysis was conducted to evaluate the predictive capacity of RDW, peak morning home SBP, and their combined use for cognitive impairment in hypertensive patients with minor stroke. RDW alone demonstrated an AUC of 0.883 (95% CI: 0.853–0.913), with exceptional specificity (0.994) but limited sensitivity (0.147), yielding a Youden’s index of −0.847. Peak morning home SBP exhibited an AUC of 0.765 (95% CI: 0.722–0.809), achieving perfect specificity (1.000) and moderate sensitivity (0.388) with a Youden’s index of 0.388. The combined model significantly outperformed individual biomarkers, achieving an AUC of 0.925 (95% CI: 0.901–0.948), balanced sensitivity (0.822), specificity (0.919), and a Youden’s index of 0.741, indicating superior discriminative power for risk stratification, as presented in Table 4 and Figure 4.
Table 4
| Indicators | AUC | Std | 95% CI | Sensitivity | Specificity | Yoden’s index |
|---|---|---|---|---|---|---|
| RDW | 0.883 | 0.015 | 0.853–0.913 | 0.147 | 0.994 | −0.847 |
| Peak morning home SBP | 0.765 | 0.022 | 0.722–0.809 | 0.388 | 1 | 0.388 |
| RDW combined with SBP | 0.925 | 0.012 | 0.901–0.948 | 0.822 | 0.919 | 0.741 |
ROC analysis of RDW, peak morning home SBP, and combined biomarkers.
ROC, Receiver operating characteristic; AUC, Area under the receiver operating characteristic curve; CI, confidence interval; RDW, Red cell distribution width; SBP, systolic blood pressure.
Figure 4

ROC curves for RDW, peak morning home SBP, and combined biomarkers. ROC, Receiver operating characteristic; RDW, Red cell distribution width; SBP, systolic blood pressure.
3.6 Predictive performance of RDW and peak morning home SBP across clinical subgroups
This study demonstrated significantly elevated SBP and RDW in patients with CD compared to non-CD counterparts (p < 0.001 for both). Male CD patients exhibited higher SBP (165.69 ± 8.90 mmHg vs. 154.29 ± 12.97 mmHg in Non-CD) and increased RDW (14.06 ± 1.45% vs. 13.31 ± 0.64%), with analogous trends observed in female and CHD subgroups. The combined SBP-RDW model achieved superior predictive performance, yielding an AUC of 0.956 (95% CI: 0.924–0.989) in CHD patients and 0.925 (95% CI: 0.891–0.958) in females, significantly outperforming individual biomarkers (RDW: AUC range, 0.868–0.909; SBP: AUC range, 0.732–0.821). These findings indicate that integrating SBP with RDW enhances cardiovascular risk stratification accuracy across heterogeneous populations (Table 5; Figure 5).
Table 5
| Variable | Male subgroup (n = 214) | Female subgroup (n = 216) | CHD subgroup (n = 135) | Non-CHD subgroup (n =?) |
|---|---|---|---|---|
| Peak morning home SBP (mmHg) | ||||
| Non-CD group | 154.29 ± 12.97 | 154.25 ± 12.60 | 152.08 ± 13.06 | 155.55 ± 12.52 |
| CD group | 165.69 ± 8.90* | 166.60 ± 8.95* | 167.09 ± 7.95* | 165.91 ± 9.24* |
| RDW (%) | ||||
| Non-CD group | 13.31 ± 0.64 | 13.38 ± 0.61 | 13.24 ± 0.61 | 13.40 ± 0.63 |
| CD group | 14.06 ± 1.45* | 14.20 ± 0.29* | 14.17 ± 0.25* | 14.23 ± 0.29* |
| ROC AUC (95% CI) | ||||
| RDW | 0.873 (0.826–0.921) | 0.876 (0.832–0.920) | 0.909 (0.861–0.956) | 0.868 (0.829–0.906) |
| Peak morning home SBP | 0.746 (0.681–0.811) | 0.782 (0.723–0.841) | 0.821 (0.752–0.891) | 0.732 (0.676–0.788) |
| Peak morning home SBP + RDW | 0.904 (0.861–0.946) | 0.925 (0.891–0.958) | 0.956 (0.924–0.989) | 0.910 (0.879–0.941) |
Subgroup-specific predictive performance of RDW and peak morning home SBP.
RDW, Red cell distribution width; SBP, systolic blood pressure; CHD, coronary heart disease; CD, cognitive decline; ROC, receiver operating characteristic; AUC, area under the receiver operating characteristic curve; CI, confidence interval. *p < 0.01.
Figure 5
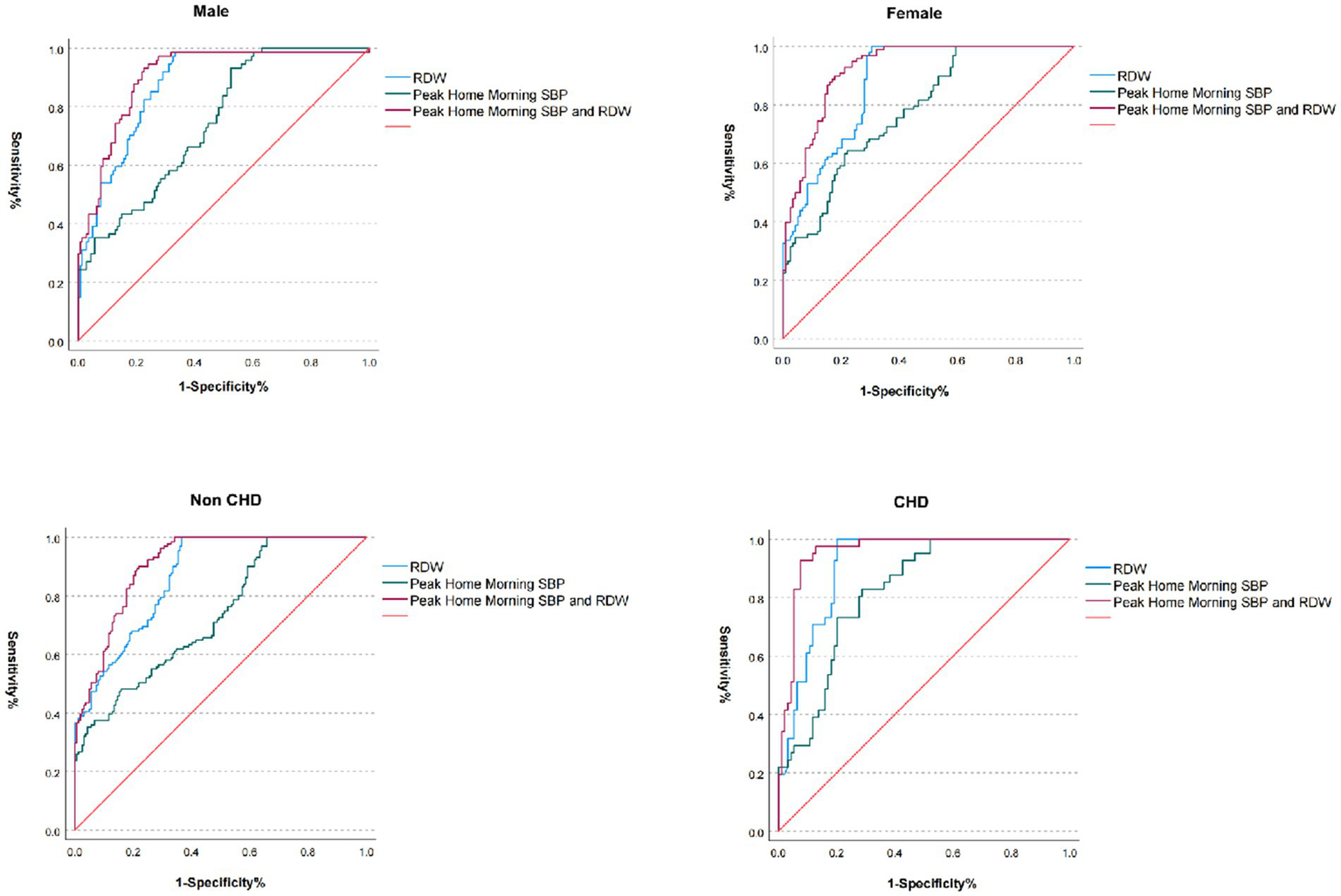
ROC curves of combined RDW and morning home SBP across subgroups. RDW, Red cell distribution width; SBP, systolic blood pressure; CHD, coronary heart disease; ROC, receiver operating characteristic.
4 Discussion
This study pioneers the combined use of RDW and peak morning home SBP as dual biomarkers for predicting PSCI in hypertensive patients with minor stroke. The innovative integration of these “inflammatory-hemodynamic” cross-dimensional indicators significantly enhanced predictive performance, with the combined model achieving superior predictive performance compared to individual biomarkers. Elevated RDW levels in the PSCI group exacerbate neurovascular damage through systemic inflammation and oxidative stress, while excessive peak morning home SBP aggravates cerebral microcirculatory dysfunction via mechanical shear stress and sympathetic overactivation. Their synergistic interaction underscores a pathogenic “inflammatory-vascular coupling” cascade, providing not only a novel biomarker combination for early PSCI detection but also mechanistic insights into dynamic neurovascular unit imbalance.
At the molecular level, RDW elevation likely drives CD through multiple pathways (27, 28). Chronic inflammatory states, characterized by elevated IL-6, induce overexpression of matrix metalloproteinases (MMPs), particularly MMP-9, which disrupts blood–brain barrier (BBB) integrity and facilitates neurotoxic substance infiltration into brain parenchyma (29). Animal models demonstrate that MMP-9 upregulation during ischemia–reperfusion injury directly correlates with cerebral edema and neuronal apoptosis (30). Furthermore, RDW-associated erythrocyte heterogeneity may accelerate cerebral small vessel disease progression, notably white matter hyperintensity (WMH) expansion. This aligns with prospective cohort findings by El Husseini et al. (9), where each 1% RDW increase elevated WMH volume progression risk by 18%, corroborating the results of our study. Concurrently, AMSBP exerts its pathological effects via circadian rhythm disruption of cerebral perfusion (31). Morning blood pressure surges (> 160 mmHg) detected through home monitoring not only directly impair endothelial function but also synergize with nocturnal non-dipping blood pressure patterns to promote β-amyloid deposition (17). Through ambulatory blood pressure monitoring, Kario et al. (20) have found that each 10 mmHg increase in morning surge elevates silent cerebral infarction risk by 31%, a relationship our study extends to PSCI pathogenesis.
We observed significantly higher HDL-C levels in patients with cognitive decline (1.80 vs. 1.43 mmol/L, p = 0.038). Although HDL-C is typically neuroprotective, emerging evidence indicates that extremely high HDL-C may lose this benefit or even become detrimental. A U-shaped relationship has been reported in older adults, where very high HDL-C (≥ 2.50 mmol/L) is associated with increased risk of cognitive impairment (OR 2.190) (32). Mechanistically, HDL particles can become dysfunctional—losing their antioxidant and anti-inflammatory properties—and may turn pro-inflammatory in chronic disease contexts, contributing to vascular injury and neuropathology (33). Furthermore, longitudinal data suggest reverse causality: cognitive decline or depressive states may themselves lead to elevated HDL-C levels over time, complicating causal inference in cross-sectional designs (34). Together, these findings highlight that in certain populations, especially older adults or those with comorbid conditions, HDL-C should not be regarded unconditionally as “protective” in the context of cognition.
The present model advances beyond traditional single-biomarker approaches. While RDW’s prognostic value in cardiovascular outcomes is well-established (12), its application in minor stroke and cognitive impairment remains unexplored. Guo et al. (14) identified RDW > 14.5% as a predictor of poor 3-month functional outcomes in acute ischemic stroke, but cognitive dimensions were unaddressed. Our study uniquely demonstrates RDW’s independent predictive capacity in minor stroke (OR = 70.95), with combined peak morning home SBP achieving superior discriminative performance compared to existing inflammatory-blood pressure model proposed by Zhang et al. (11). In addition, the application of HBPM addresses limitations of conventional clinic measurements, which are prone to bias due to white-coat effects and insufficient sampling. HBPM’s 7-day morning monitoring protocol provides a more authentic reflection of hemodynamic load. According to Yaneva-Sirakova et al.’s findings, morning surge blood pressure > 145 mmHg is associated with a 2.3-fold increased CD risk (19). Our extension of this paradigm to younger high-risk cohorts highlights HBPM’s broader applicability.
Clinically, our stratified screening protocol, with initial RDW triage followed by dynamic peak morning home SBP monitoring and nomogram-based risk stratification, offers time-efficient and cost-effective advantages over traditional neuropsychological assessments like MoCA. This approach holds particular promise for primary care implementation. Moreover, subgroup analyses revealed enhanced predictive utility in females and CHD patients, which aligns with Gallucci et al.’s emphasis on stroke heterogeneity necessitating personalized evaluation (10), suggesting that the predictive efficacy of biomarkers may be modulated by sex and comorbid status. For instance, sex-specific inflammatory sensitivity and pre-existing endothelial dysfunction in CHD may amplify RDW-AMSBP synergism. This hypothesis warrants further mechanistic investigation through sex-stratified analyses and comorbidity-focused studies.
Nevertheless, several limitations merit consideration. First, the single-center design and moderate sample size may constrain the validity of our results. Despite rigorous baseline matching (age, NIHSS scores) to reduce confounding bias, ethnic homogeneity (predominantly Asian) and stroke etiology distribution (e.g., 12% cardioembolic strokes) necessitate validation in multiethnic cohorts. Second, the 3-month follow-up window limits insight into long-term PSCI trajectories. Neurocognitive decline often follows a nonlinear trajectory, with vascular dementia in particular tending to fully manifest 1–2 years post-stroke (35). Thus, future studies should extend observation periods to ≥ 24 months, incorporating serial biomarker measurements (e.g., RDW/peak morning home SBP trajectories) to construct temporal prediction models. Third, the absence of direct mechanistic evidence, such as neuroimaging biomarkers [BBB permeability via diffusion tensor imaging (DTI)] or molecular markers [S100β, glial fibrillary acidic protein (GFAP)], restricts pathophysiological depth. While the proposed “inflammatory-vascular coupling” hypothesis is theoretically grounded, multimodal validation remains essential.
Future research should prioritize three axes: 1. Multi-center validation across diverse populations to enhance generalizability; 2. Integration of multimodal data (functional MRI, serum inflammasome profiling) to elucidate RDW-peak morning home SBP interaction pathways; 3. Intervention trials targeting high-risk populations (RDW > 14% + peak morning home SBP > 160 mmHg) to assess whether early anti-inflammatory therapies (e.g., IL-6 inhibitors) or chronotherapeutic antihypertensives (e.g., bedtime dosing) can mitigate CD. Furthermore, technological advancements in HBPM devices, including wireless data transmission and artificial intelligence (AI)-driven risk alerts, could optimize screening efficiency, accelerating precision medicine adoption in neurovascular care.
5 Conclusion
The synergistic application of RDW and peak morning home SBP establishes an efficient, cost-effective strategy for predicting cognitive impairment in hypertensive patients with minor stroke. This dual biomarker approach not only advances understanding of inflammatory-hemodynamic interplay but also paves the way for personalized therapeutic interventions. Clinical translation and mechanistic exploration will require multidisciplinary collaboration, leveraging advanced monitoring technologies and targeted therapeutic trials to optimize patient outcomes in vascular cognitive disorders.
Statements
Data availability statement
The original contributions presented in the study are included in the article/supplementary material, further inquiries can be directed to the corresponding author/s.
Ethics statement
The studies involving humans were approved by the Human Ethics Committee of People's Hospital Affiliated to Ningbo University (Ethics Approval Number: 2025-042). The studies were conducted in accordance with the local legislation and institutional requirements. The participants provided their written informed consent to participate in this study. Written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article.
Author contributions
YSh: Writing – review & editing, Visualization, Resources, Conceptualization, Project administration, Investigation, Writing – original draft, Supervision, Formal analysis, Methodology, Data curation. JC: Data curation, Validation, Methodology, Writing – original draft, Visualization, Conceptualization, Writing – review & editing, Formal analysis. YSo: Writing – original draft, Resources, Visualization, Software, Formal analysis, Methodology, Writing – review & editing. WW: Writing – review & editing, Formal analysis, Writing – original draft, Software, Methodology, Visualization, Validation.
Funding
The author(s) declare that no financial support was received for the research and/or publication of this article.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The authors declare that no Gen AI was used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1.
Feigin VL Brainin M Norrving B Martins SO Pandian J Lindsay P et al . World stroke organization: global stroke fact sheet 2025. Int J Stroke. (2025) 20:132–44. doi: 10.1177/17474930241308142
2.
Einstad MS Saltvedt I Lydersen S Ursin MH Munthe-Kaas R Ihle-Hansen H et al . Associations between post-stroke motor and cognitive function: a cross-sectional study. BMC Geriatr. (2021) 21:103. doi: 10.1186/s12877-021-02055-7
3.
Marsh EB Girgenti S Llinas EJ Brunson AO . Outcomes in patients with minor stroke: diagnosis and Management in the Post-thrombectomy era. Neurotherapeutics. (2023) 20:732–43. doi: 10.1007/s13311-023-01349-5
4.
Cohen DL Roffe C Beavan J Blackett B Fairfield CA Hamdy S et al . Post-stroke dysphagia: a review and design considerations for future trials. Int J Stroke. (2016) 11:399–411. doi: 10.1177/1747493016639057
5.
Smith-Spijkerboer W Meeske K van der Palen JAM den Hertog HM Smeets–Schouten AS van Hout M et al . Impaired visual emotion recognition after minor ischemic stroke. Arch Phys Med Rehabil. (2022) 103:958–63. doi: 10.1016/j.apmr.2021.10.024
6.
Lenzi GL Altieri M Maestrini I . Post-stroke depression. Rev Neurol (Paris). (2008) 164:837–40. doi: 10.1016/j.neurol.2008.07.010
7.
Batchelor FA Mackintosh SF Said CM Hill KD . Falls after stroke. Int J Stroke. (2012) 7:482–90. doi: 10.1111/j.1747-4949.2012.00796.x
8.
Boulos MI Wan A Black SE Lim AS Swartz RH Murray BJ . Restless legs syndrome after high-risk TIA and minor stroke: association with reduced quality of life. Sleep Med. (2017) 37:135–40. doi: 10.1016/j.sleep.2017.05.020
9.
El Husseini N et al . Cognitive impairment after ischemic and hemorrhagic stroke: a scientific statement from the American Heart Association/American Stroke Association. Stroke. (2023) 54:e272–91. doi: 10.1161/STR.0000000000000430
10.
Gallucci L Sperber C Guggisberg AG Kaller CP Heldner MR Monsch AU et al . Post-stroke cognitive impairment remains highly prevalent and disabling despite state-of-the-art stroke treatment. Int J Stroke. (2024) 19:888–97. doi: 10.1177/17474930241238637
11.
Zhang Y Xing Z Zhou K Jiang S . The predictive role of systemic inflammation response index (SIRI) in the prognosis of stroke patients. Clin Interv Aging. (2021) 16:1997–2007. doi: 10.2147/CIA.S339221
12.
Salvagno GL Sanchis-Gomar F Picanza A Lippi G . Red blood cell distribution width: a simple parameter with multiple clinical applications. Crit Rev Clin Lab Sci. (2015) 52:86–105. doi: 10.3109/10408363.2014.992064
13.
Joosse HJ van Oirschot BA Kooijmans SAA Hoefer IE van Wijk RAH Huisman A et al . In-vitro and in-silico evidence for oxidative stress as drivers for RDW. Sci Rep. (2023) 13:9223. doi: 10.1038/s41598-023-36514-5
14.
Guo T Qin Z He D . Acute myocardial infarction (AMI) as the effect modifiers to modify the association between red blood cell distribution width (RDW) and mortality in critically ill patients with stroke. Front Med. (2022) 9:754979. doi: 10.3389/fmed.2022.754979
15.
Feng GH Li HP Li QL Fu Y Huang RB . Red blood cell distribution width and ischaemic stroke. Stroke Vasc Neurol. (2017) 2:172–5. doi: 10.1136/svn-2017-000071
16.
Nagai M Dasari TW . Home blood pressure measurement of hypertensive patients with cognitive impairment-is it "environmental pressure"?Hypertens Res. (2024) 47:573–5. doi: 10.1038/s41440-023-01526-1
17.
Hoshide S Nishizawa M Kanegae H Kario K . Home blood pressure measurement consistency and cognitive impairment. Hypertens Res. (2024) 47:177–83. doi: 10.1038/s41440-023-01436-2
18.
Yeung SE Loken Thornton W . "Do it-yourself": home blood pressure as a predictor of traditional and everyday cognition in older adults. PLoS One. (2017) 12:e0177424. doi: 10.1371/journal.pone.0177424
19.
Yaneva-Sirakova T Traykov L Petrova J Vassilev D . Comparison of central, ambulatory, home and office blood pressure measurement as risk markers for mild cognitive impairment in hypertensive patients. Dement Geriatr Cogn Dis Extra. (2017) 7:274–82. doi: 10.1159/000479365
20.
Kario K Tomitani N Fujiwara T Okawara Y Kanegae H Hoshide S . Peak home blood pressure as an earlier and strong novel risk factor for stroke: the practitioner-based nationwide J-HOP study extended. Hypertens Res. (2023) 46:2113–23. doi: 10.1038/s41440-023-01297-9
21.
Wander GS Salman E Matsushita N Verma N . Awareness and recommendation of home blood pressure measurement among physicians in India: results from Asia HBPM survey 2020. Indian Heart J. (2023) 75:169–76. doi: 10.1016/j.ihj.2023.04.004
22.
Liyanage-Don N Fung D Phillips E Kronish IM . Implementing home blood pressure monitoring into clinical practice. Curr Hypertens Rep. (2019) 21:14. doi: 10.1007/s11906-019-0916-0
23.
Liu C Xu D Meng L Li H Fu Z Yan M et al . Characterizing the relationship between MRI radiomics and AHR expression and deriving a predictive model for prognostic assessment in glioblastoma. Neuroradiology. (2024) 66:1291–9. doi: 10.1007/s00234-024-03396-x
24.
Huang X Guo H Yuan L Cai Q Zhang M Zhang Y et al . Blood pressure variability and outcomes after mechanical thrombectomy based on the recanalization and collateral status. Ther Adv Neurol Disord. (2021) 14:1756286421997383. doi: 10.1177/1756286421997383
25.
Cheung RT Lyden PD Tsoi TH Huang Y Liu M Hon SF et al . Production and validation of Putonghua- and Cantonese-Chinese language National Institutes of Health stroke scale training and certification videos. Int J Stroke. (2010) 5:74–9. doi: 10.1111/j.1747-4949.2010.00411.x
26.
Lu J Li D Li F Zhou A Wang F Zuo X et al . Montreal cognitive assessment in detecting cognitive impairment in Chinese elderly individuals: a population-based study. J Geriatr Psychiatry Neurol. (2011) 24:184–90. doi: 10.1177/0891988711422528
27.
Gomes KB Pereira RG Braga AA Guimarães HC Resende EPF Teixeira AL et al . Machine learning-based routine laboratory tests predict one-year cognitive and functional decline in a population aged 75+ years. Brain Sci. (2023) 13:690. doi: 10.3390/brainsci13040690
28.
Beydoun MA Hossain S MacIver PH Srinivasan D Beydoun HA Maldonado AI et al . Red cell distribution width, Anemia, and brain volumetric outcomes among middle-aged adults. J Alzheimer's Dis. (2021) 81:711–27. doi: 10.3233/JAD-201386
29.
Ravindran J Agrawal M Gupta N Lakshmana Rao PV . Alteration of blood brain barrier permeability by T-2 toxin: role of MMP-9 and inflammatory cytokines. Toxicology. (2011) 280:44–52. doi: 10.1016/j.tox.2010.11.006
30.
Hu Y Hu XD He ZQ Liu Y Gui YK Zhu SH et al . Anesthesia/surgery activate MMP9 leading to blood-brain barrier disruption, triggering neuroinflammation and POD-like behavior in aged mice. Int Immunopharmacol. (2024) 135:112290. doi: 10.1016/j.intimp.2024.112290
31.
Wiesmann M Zerbi V Jansen D Lütjohann D Veltien A Heerschap A et al . Hypertension, cerebrovascular impairment, and cognitive decline in aged AbetaPP/PS1 mice. Theranostics. (2017) 7:1277–89. doi: 10.7150/thno.18509
32.
Huang H Yang B Yu R Ouyang W Tong J le Y . Very high high-density lipoprotein cholesterol may be associated with higher risk of cognitive impairment in older adults. Nutr J. (2024) 23:79. doi: 10.1186/s12937-024-00983-9
33.
Madaudo C Bono G Ortello A Astuti G Mingoia G Galassi AR et al . Dysfunctional high-density lipoprotein cholesterol and coronary artery disease: a narrative review. J Pers Med. (2024) 14:996. doi: 10.3390/jpm14090996
34.
Liu YH Chen MT He YY Chen M Liang JR Jia FJ et al . Cognitive impairment and depression precede increased HDL-C levels in middle-aged and older Chinese adults: cross-lagged panel analyses. Lipids Health Dis. (2024) 23:288. doi: 10.1186/s12944-024-02285-9
35.
Pendlebury ST Rothwell PM . Prevalence, incidence, and factors associated with pre-stroke and post-stroke dementia: a systematic review and meta-analysis. Lancet Neurol. (2009) 8:1006–18. doi: 10.1016/S1474-4422(09)70236-4
Summary
Keywords
red cell distribution width, peak morning home systolic blood pressure, hypertension, cognitive impairment, minor stroke
Citation
Shu Y, Chen J, Song Y and Wang W (2025) Unveiling the predictive synergy of red cell distribution width and peak morning home systolic blood pressure: a dual biomarker approach for cognitive risk stratification in hypertensive minor stroke. Front. Neurol. 16:1637561. doi: 10.3389/fneur.2025.1637561
Received
29 May 2025
Accepted
07 August 2025
Published
26 August 2025
Volume
16 - 2025
Edited by
Mohammed Ahmed Akkaif, QingPu Branch of Zhongshan Hospital Affiliated to Fudan University, China
Reviewed by
Dingkang Xu, Chinese Academy of Medical Sciences and Peking Union Medical College, China
Kimson Johnson, University of Michigan, United States
Updates
Copyright
© 2025 Shu, Chen, Song and Wang.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Yimin Shu, sym1632025@163.com
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.