- 1Spinal Surgery Department, Mianyang Orthopaedic Hospital, Mianyang, Sichuan, China
- 2Department of Spinal & Bone Tumors (1), The Affiliated Traditional Chinese Medicine Hospital of Southwest Medical University, Luzhou, Sichuan, China
Postoperative infections following spinal fusion procedures are commonly caused by pathogens such as Staphylococcus aureus, coagulase-negative Staphylococcus, Streptococcus species, and Escherichia coli. However, cases involving Prevotella oris as a causative agent are rarely documented in the literature. We report a case of postoperative infection after anterior cervical spinal fusion surgery. After empirical antimicrobial therapy failed, high-throughput nucleic acid sequencing detected Prevotella oris in the patient's cerebrospinal fluid—a rare and previously unreported finding. Upon switching to a more sensitive antibiotic regimen, the patient's symptoms improved significantly. This case highlights an unusual etiology and provides a comprehensive documentation of the treatment process, aiming to assist clinicians in the diagnosis and management of similar infections.
Introduction
Postoperative infections are a common complication following spinal fusion surgery. Although the incidence remains relatively low, these infections often require prolonged antimicrobial therapy, leading to extended hospitalization and significantly increased medical, social, and economic burdens (1, 2). Consistent studies have demonstrated that Staphylococcus aureus is the predominant pathogen responsible for postoperative infections following spinal fusion procedures (3, 4). However, other Gram-positive and Gram-negative bacteria can also contribute to such infections (5, 6). Here, we present a rare case of postoperative infection after anterior cervical spinal fusion surgery caused by Prevotella oris. By sharing the diagnostic and therapeutic approach, we aim to provide clinicians with valuable insights into the management of similar cases.
Case report
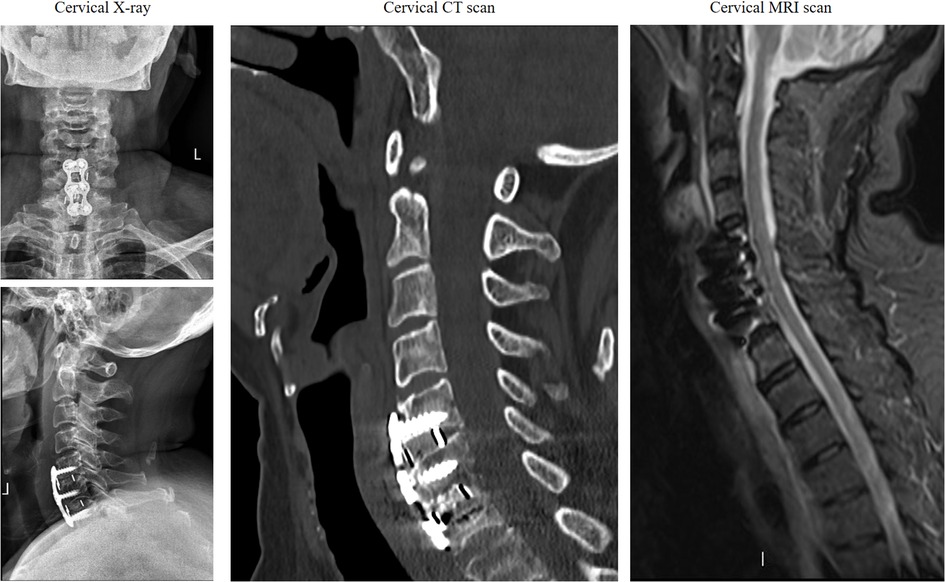
We present a case of postoperative infection in a 70-year-old female patient following anterior cervical spinal fusion. The patient was admitted for the management of neck pain accompanied by left upper extremity pain and numbness. Her medical history included hypertension and diabetes, both of which were managed with a regular medication regimen to control blood pressure and blood glucose levels. Physical examination revealed pronounced tenderness and muscle tension in the left cervical and shoulder regions, hypoesthesia in the lateral aspects of the left upper arm and forearm, and muscle strength graded 4 in the left triceps, wrist extensors, and finger extensors. Additionally, the bilateral foraminal compression tests and left brachial plexus traction test were positive. Preoperative imaging demonstrated a C5–7 intervertebral disc herniation (Figure 1), compressing the nerve roots and dural sac at the corresponding levels. Although the patient's symptoms were attributed to the left-sided herniation at C6/7, we prophylactically addressed the asymptomatic right-sided disc herniation at C5/6, considering both the increased risk of adjacent segment disease following fusion surgery and the potential limitations for future reoperations due to the patient's advancing age and progressively deteriorating physical condition. To alleviate her symptoms, a C5–7 anterior cervical discectomy and fusion (ACDF) was performed, which resulted in significant postoperative symptom relief. Postoperative imaging findings are shown in (Figures 2). Perioperative assessment confirmed the absence of surgically significant complications, including incidental durotomy, cerebrospinal fluid leakage, esophageal perforation, or laryngotracheal injury. This was corroborated by postoperative clinical indicators: absence of dysphagia, cervical swelling, or subcutaneous emphysema, coupled with minimal incisional drainage (<30 ml/day of serosanguineous fluid) and stable vital signs.
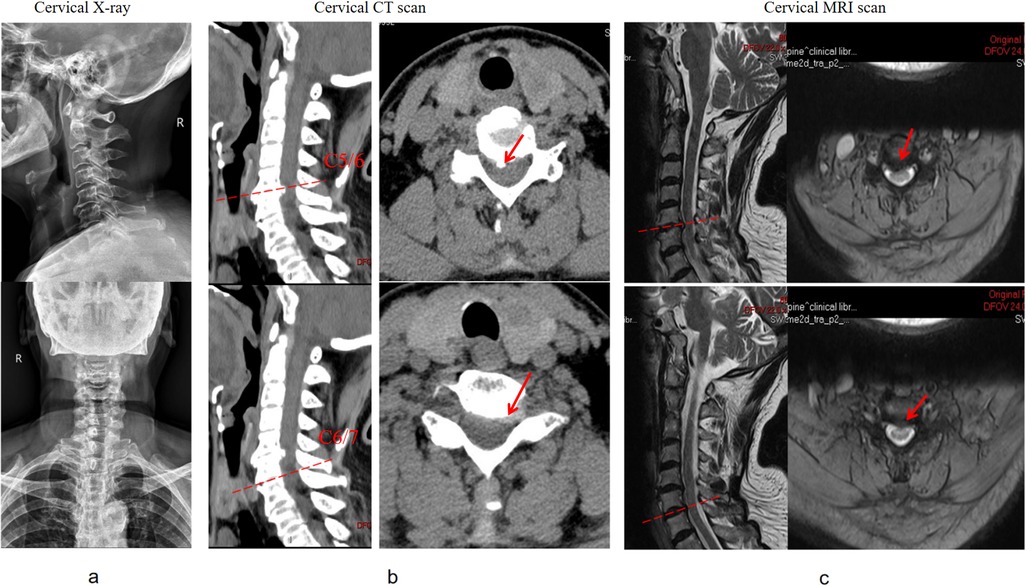
Figure 1. Preoperative imaging examination [including (a) X-ray, (b) CT scan, and (c) MRI] suggested herniation of the C5-7 intervertebral disc with compression of the dural sac and nerve root.
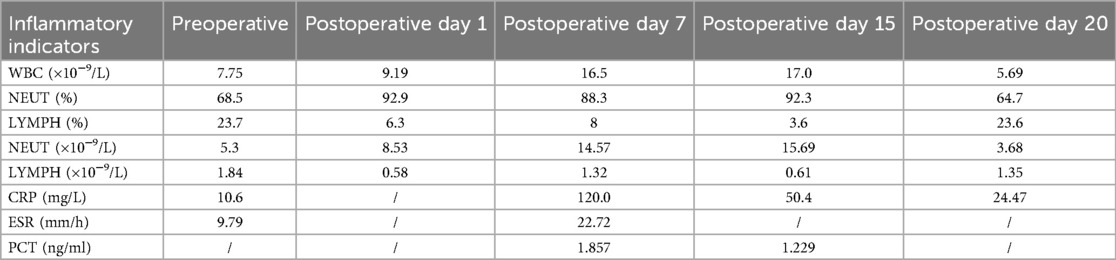
Unfortunately, one week after surgery, the patient developed axial symptoms, including left neck and shoulder pain, stiffness, and muscle tension. Hematological results revealed a white blood cell count of 16.50 × 10−9/L, neutrophil count of 14.57 × 10−9/L (88.3%), procalcitonin level of 1.857 ng/ml, and C-reactive protein level of 120.0 mg/L, all suggesting infection. Since no pathogen was initially identified, empirical treatment with moxalactam sodium (0.5 g every 12 h) was initiated for one week. Despite this, the patient's condition worsened, with left upper limb weakness (about grade 3) and bilateral lower extremity weakness (about grade 3) developing. Follow-up hematological tests showed a white blood cell count of 17.00 × 10−9/L, neutrophil count of 15.69 × 10−9/L (92.3%), procalcitonin level of 1.229 ng/ml, and C-reactive protein level of 50.4 mg/L. MRI revealed spinal cord edema in the surgical area (Figure 3a). To determine the etiology, a lumbar puncture was performed for cerebrospinal fluid analysis, which showed a white blood cell count of 19.00 × 10−6/L. Pathogen nucleic acid high-throughput sequencing of the CSF identified Prevotella oris (relative abundance: 45.76%, confidence: 99%), a microorganism typically found in the oral and gastrointestinal microbiota.
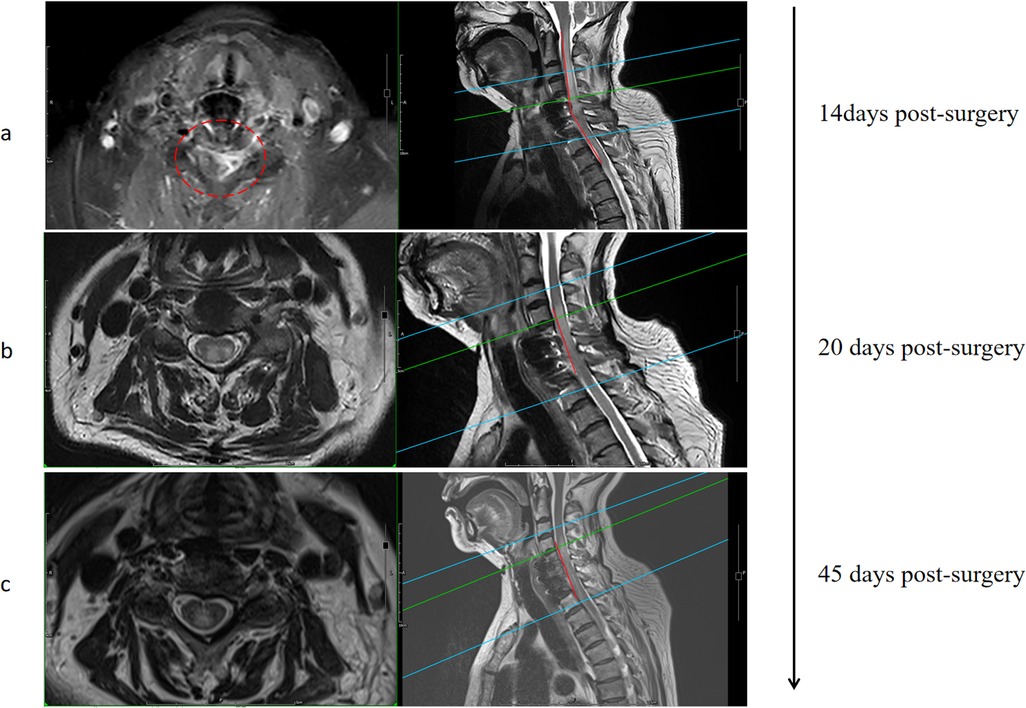
Figure 3. MRI imaging findings at different periods after the occurrence of axial pain after surgery (a) Imaging follow-up after the first empirical use of antibiotics (b) Imaging follow-up after antibiotic administration for one week after metagenomics revealed the causative organism (c) Imaging follow-up about 3 weeks after discharge.
After a multidisciplinary consultation involving neurology and pharmacy, the patient was treated with vancomycin hydrochloride (1.0 g every 12 h), piperacillin sodium/tazobactam (4.5 g every 8 h), and ornidazole (0.5 g every 12 h) for infection control. After 3 weeks of systemic antimicrobial therapy, hematological parameters normalized within institutional reference ranges (Table 1), indicating effective infection control and prompting discontinuation of further serological surveillance. At the last documented follow-up 6 weeks postoperatively, the patient exhibited resolution of left cervicobrachial pain, with improvement in muscle strength to grade 4 in the left upper limb and grade 4/5 in bilateral lower limbs. Concurrent MRI revealed regression of spinal cord edema (Figures 3b,c).
Discussion
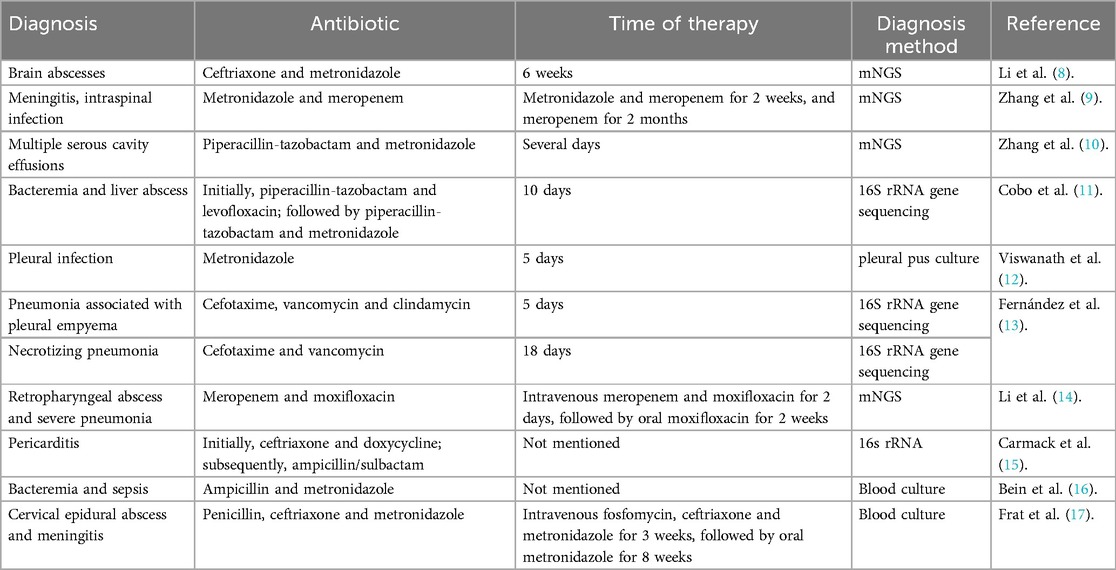
Postoperative infections following spinal fusion are most commonly caused by Staphylococcus aureus. According to a report by the International Spine Research Society, the incidence of surgical site infections after spinal surgery is 1.2%, with Staphylococcus aureus (41.9%) and methicillin-resistant Staphylococcus aureus (MRSA) (17.0%) being the most prevalent pathogens (7). To our knowledge, no cases of Prevotella oris infection following spinal fusion surgery have been reported, based on a comprehensive search of the PubMed database. Only a few studies have identified Prevotella species as causative agents of brain abscesses, meningitis, or spinal canal infections (Table 2) (8–17). All findings were consistently identified through metagenomic and high-throughput sequencing, providing critical guidance for subsequent treatment decisions. Prevotella oris, a commensal bacterium primarily colonizing the oral cavity and gastrointestinal tract, was the detected pathogen. Upon hospital admission, the patient reported no complaints of oral lesions. The absence of dysphagia, cervical swelling, subcutaneous emphysema, coupled with minimal (<30 ml/day) serosanguineous (as opposed to clear) drainage from the surgical incision, effectively excluded esophageal perforation and cerebrospinal fluid leakage as potential postoperative complications in this case. Notably, prior studies have demonstrated a significant association between preoperative nasal bacterial colonization and postoperative surgical site infections in spinal procedures, suggesting potential dissemination of nasal flora to the wound (18). Given the close anatomical proximity between the cervical spine and the oral cavity, it is biologically plausible that Prevotella oris could disseminate from the oral cavity to the surgical site. Anterior cervical spine surgery (e.g., ACDF) requires dissection through the retropharyngeal space, where the posterior pharyngeal wall lies in close proximity to the prevertebral fascia. Intraoperative maneuvers (such as retraction or electrocautery) may cause microtrauma to the pharyngeal mucosa even without full-thickness perforation, potentially allowing oral flora to migrate through microscopic breaches into the surgical field. Furthermore, oropharyngeal procedures (including endotracheal intubation and suctioning) can induce transient bacteremia, enabling hematogenous bacterial seeding at the surgical site-particularly on implant surfaces.
We aimed to summarize and share the insights from this case to guide clinical diagnosis and treatment. First, when infection is suspected after spinal fusion surgery, clinical manifestations such as deterioration or fever (though absent in this case) may occur. Routine diagnostic tests should include a complete blood count, erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), procalcitonin (PCT), cervical x-rays, CT scans, and cervical MRI (including contrast-enhanced MRI). Second, high-throughput genomic sequencing has increasingly identified rare pathogens in recent years, aiding clinicians in accurate pathogen identification and guiding standardized treatment strategies. Although high-throughput genomic sequencing holds promise for rapid diagnosis, its findings must be interpreted in concordance with clinical presentation, laboratory results, and imaging studies. Finally, early initiation of appropriately targeted antibiotics significantly improves patient prognosis.
Data availability statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Ethics statement
The patient provided written consent for the utilization of this case in academic discourse and publication.
Author contributions
CW: Conceptualization, Investigation, Methodology, Visualization, Writing – original draft, Supervision, Validation, Writing – review & editing. YL: Conceptualization, Investigation, Methodology, Visualization, Writing – original draft, Resources.
Funding
The author(s) declare that financial support was received for the research and/or publication of this article. This study was supported by the Scientific Research Project of Sichuan Medical Association (S2024060).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Tan T, Lee H, Huang MS, Rutges J, Marion TE, Mathew J, et al. Prophylactic postoperative measures to minimize surgical site infections in spine surgery: systematic review and evidence summary. Spine J. (2020) 20(3):435–47. doi: 10.1016/j.spinee.2019.09.013
2. Di Martino A, Papalia R, Albo E, Diaz L, Denaro L, Denaro V. Infection after spinal surgery and procedures. Eur Rev Med Pharmacol Sci. (2019) 23(2):173–8. doi: 10.26355/eurrev_201904_17487
3. Rosenthal NA, Heinrich KH, Chung J, Yu H. Cost and hospital resource utilization of staphylococcus aureus infection post elective posterior instrumented spinal fusion surgeries in U.S. hospitals: a retrospective cohort study. Spine. (2019) 44(9):637–46. doi: 10.1097/BRS.0000000000002898
4. Begier E, Rosenthal NA, Richardson W, Chung J, Gurtman A. Invasive Staphylococcus aureus infection among patients undergoing elective, posterior, instrumented spinal fusion surgeries: a retrospective cohort study. Surg Infect. (2022) 23(1):12–21. doi: 10.1089/sur.2021.019
5. Anderson PA, Savage JW, Vaccaro AR, Radcliff K, Arnold PM, Lawrence BD, et al. Prevention of surgical site infection in spine surgery. Neurosurgery. (2017) 80(3S):S114–23. doi: 10.1093/neuros/nyw066
6. Yoon JW, Wanderman NR, Kerezoudis P, Alvi MA, De Biase G, Akinduro OO, et al. Enterobacter infection after spine surgery: an institutional experience. World Neurosurg. (2019) 123:e330–7. doi: 10.1016/j.wneu.2018.11.169
7. Shillingford JN, Laratta JL, Reddy H, Ha A, Lehman RA, Lenke LG, et al. Postoperative surgical site infection after spine surgery: an update from the scoliosis research society (SRS) morbidity and mortality database. Spine Deform. (2018) 6(6):634–43. doi: 10.1016/j.jspd.2018.04.004
8. Li C, Lv C, Yu M, Zhang Y, Ling L, Yue W. A case report of a brain abscess due to Prevotella oris and a review of the literature. BMC Infect Dis. (2023) 23(1):633. doi: 10.1186/s12879-023-08306-9. Published 2023 September 27.37759232
9. Zhang WW, Ai C, Mao CT, Liu DK, Guo Y. Prevotella oris-caused meningitis and spinal canal infection: a case report. World J Clin Cases. (2023) 11(16):3830–6. doi: 10.12998/wjcc.v11.i16.3830
10. Zhang F, Wang J-L, Zhu J, Si S, Guo H, Yue X, et al. When a sore throat turns into deadly multiple serous cavity effusions: the role of Prevotella oris in rapidly progressing infection-a case report. Front Med. (2025) 12:1517389. doi: 10.3389/fmed.2025.1517389. Published 2025 January 29.
11. Cobo F, Pérez-Carrasco V, Pérez-Rosillo MÁ, García-Salcedo JA, Navarro-Marí JM. Bacteremia due to Prevotella oris of probable hepatic origin. Anaerobe. (2022) 76:102586. doi: 10.1016/j.anaerobe.2022.102586
12. Viswanath LS, Gunalan A, Jamir I, Balamurugan S, Anbarasan S, Aswin K, et al. Prevotella oris: a lesser known etiological agent of pleural effusion. Anaerobe. (2022) 78:102644. doi: 10.1016/j.anaerobe.2022.102644
13. Fernández Vecilla D, Roche Matheus MP, Iglesias Hidalgo G, Ugalde Zárraga E, Unzaga Barañano MJ, Díaz de Tuesta Del Arco JL. Two cases of Prevotella oris causing serious pleuropulmonary infections. Rev Esp Quimioter. (2023) 36(4):439–41. doi: 10.37201/req/001.2023
14. Li MJ, Zhou SF, Zhang Y, Zhang Y, Fu WB. Unveiling the dark side of Prevotella: a case of fatal pneumonia from a common probiotic. Front Med. (2024) 11:1382134. doi: 10.3389/fmed.2024.1382134. Published 2024 December 24.
15. Carmack AE, LaRocco AM, Mathew M, Goldberg HV, Patel DM, Saleeb PG. Subacute polymicrobial bacterial pericarditis mimicking tuberculous pericarditis: a case report. Am J Case Rep. (2021) 22:e933684. doi: 10.12659/AJCR.933684
16. Bein T, Brem J, Schüsselbauer T. Bacteremia and sepsis due to Prevotella oris from dentoalveolar abscesses. Intensive Care Med. (2003) 29:856. doi: 10.1007/s00134-003-1697-z
17. Frat JP, Godet C, Grollier G, Blanc JL, Robert R. Cervical spinal epidural abscess and meningitis due to Prevotella oris and peptostreptococcus micros after retropharyngeal surgery. Intensive Care Med. (2004) 30:1695. doi: 10.1007/s00134-004-2265-x
Keywords: anterior cervical spinal fusion, postoperative infection, prevotella oris, high-throughput nucleic acid sequencing, case report
Citation: Wang C and Lei Y (2025) Postoperative infection following anterior cervical fusion surgery caused by Prevotella oris: a case report. Front. Surg. 12:1555039. doi: 10.3389/fsurg.2025.1555039
Received: 3 January 2025; Accepted: 8 August 2025;
Published: 29 August 2025.
Edited by:
Julius Höhne, Paracelsus Medical Private University, GermanyReviewed by:
Kushani Shah, Dana–Farber Cancer Institute, United StatesMichal Ziga, Cantonal Hospital Lucerne, Switzerland
Copyright: © 2025 Wang and Lei. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Yang Lei, NDQ2MDA1NDM3QHFxLmNvbQ==
 Chenglong Wang
Chenglong Wang Yang Lei2*
Yang Lei2*