- 1Department of Integrative Food, Bioscience and Biotechnology, Chonnam National University, Gwangju, Republic of Korea
- 2Department of Plant Protection, College of Agriculture, Yangtze University, Jingzhou, China
- 3Department of Rural and Bio-Systems Engineering, Chonnam National University, Gwangju, Republic of Korea
- 4Kumho Life Science Laboratory, Chonnam National University, Gwangju, Republic of Korea
Sheath blight, caused by the fungal pathogen Rhizoctonia solani, is one of the major diseases affecting rice, leading to significant yield losses. Unmanned aerial vehicle (UAV)-based fungicide spraying has emerged as an efficient method for disease control. In this study, the sensitivity of R. solani to six fungicides (florylpicoxamid, fludioxonil, flutolanil, ipflufenoquin, orysastrobin, and pencycuron) with different modes of action was tested in vitro. These fungicides were also tested in planta, when after exhibiting the highest efficacy, orysastrobin and flutolanil were then selected to evaluate efficacy at reduced concentrations using mobile spraying equipment (MSE). The results revealed that even at 1/4 of the original concentration, both fungicides were sufficient to control sheath blight. This study provides an optimized fungicide application method for controlling sheath blight using UAVs.
1 Introduction
Rice is one of the most widely consumed crops globally and serves as a staple food for more than half of the world’s population, particularly in Asia, Africa, and Latin America (Sackey et al., 2025). Rice diseases caused by various pathogens can significantly reduce grain yield and quality. Among these diseases, sheath blight, caused by Rhizoctonia solani AG1-IA, is particularly destructive. The pathogen initially causes water-soaked lesions on leaf sheaths above the waterline. As the disease progresses, lesions enlarge and become grayish-white with dark margins (Mohapatra et al., 2025). Additionally, R. solani produces sclerotia on leaf sheaths, enhancing pathogen survival under adverse environmental conditions and complicating disease management.
Currently, chemical control is the primary method for managing sheath blight. Fungicides with different modes of action, including methyl benzimidazole carbamates (MBCs), succinate dehydrogenase inhibitors (SDHIs), and quinone outside inhibitors (QoIs), have been widely applied in rice fields (Chen et al., 2012; Ishii and Kurosaki, 2017; Neha et al., 2017). However, the success of fungicide-based control largely depends on proper timing, accurate targeting, and sufficient coverage, as sheath blight tends to become more severe under high humidity during the rainy season and typically develops on the lower parts of the plant, such as the leaf sheaths near the soil surface (Persaud et al., 2019).
Conventional fungicide applications usually rely on manual air-pressure knapsack sprayers (Li et al., 2018; Wang et al., 2019), which are equipped with low-precision nozzles and exhibit low spraying efficiency, leading to significant pesticide losses (Rincón et al., 2017; Wang et al., 2019). Moreover, workers are exposed to chemicals during spraying (Cao et al., 2017). Considering the extensive rice cultivation areas, these limitations pose significant challenges to disease management. The application of unmanned aerial vehicles (UAVs) for pesticide application is an alternative to reduce operators’ exposure to pesticides and also lower labor costs, which can contribute to the development of sustainable agriculture (Umeda et al., 2022; Kuster et al., 2023). Nowadays, UAVs have gained attention for their high spraying efficiency, flexibility under various field conditions, and improved operator safety. UAV-based approaches, which were initially developed for plant disease diagnosis, are now increasingly being utilized for field-level disease control through precise and efficient application of agrochemicals (Wongsuk et al., 2024). However, little research has been conducted on the efficacy of different fungicides and their concentrations for chemical control of specific crops using UAV-based spraying. Mobile spraying equipment (MSE) serves as an indoor system that simulates UAV spraying conditions, allowing controlled evaluations of spraying parameters and fungicide efficacy (Park et al., 2024). Functions as a laboratory simulator, MSE replicates key UAV spray features (e.g., droplet spectrum, velocity, and impaction) under controlled conditions, which helps to establish UAV-relevant spray parameters under reproducible laboratory settings before moving to UAV field applications.
Therefore, this study aims to develop an effective method for controlling rice sheath blight with UAVs by identifying suitable fungicides and evaluating fungicide efficacy at diluted concentrations, as required for UAV-based applications, using MSE.
2 Materials and methods
2.1 Sheath blight pathogen and fungicides
Isolates of the rice sheath blight pathogen Rhizoctonia solani (KACC40101, KACC40106, and KACC45811) were obtained from the Korean Agricultural Culture Collection (KACC) in the Rural Development Administration located in Wanju-gun, Korea. These isolates were confirmed as AG1-IA by KACC records. A total of six fungicides with different modes of action (Supplementary Table S1) were used in this study: florylpicoxamid, (Bupifix 10% SC, Corteva, IN, USA), fludioxonil (Sapphire 20% SC, Syngenta, Basel, Switzerland), flutolanil (Moncut 15% EC, Nihon Nohyaku, Tokyo, Japan), ipflufenoquin (Migiwa 20% SC, Nippon Soda, Tokyo, Japan), orysastrobin (Allclean 12% SC, BASF, Ludwigshafen, Germany), and pencycuron (Monceren 20% SC, Bayer, Leverkusen, Germany).
2.2 In vitro fungicide sensitivity assay
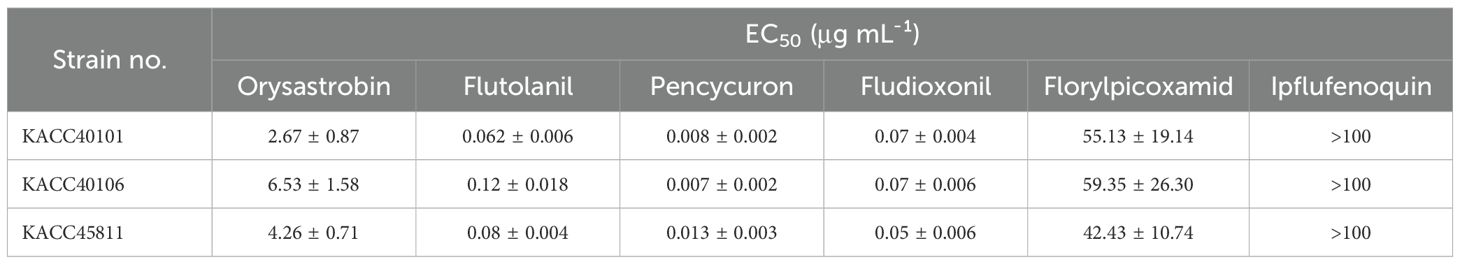
The sensitivity of R. solani isolates to six fungicides was determined by evaluating the 50% effective concentration (EC50) for inhibiting mycelial growth. Isolates were first cultured on potato dextrose agar (PDA) at 25 °C in darkness for 2 days. Mycelial plugs (5 mm in diameter) were then taken from the edges of the colonies and transferred to PDA plates amended with various concentrations of fungicides (0, 0.01, 0.1, 1, 10, and 100 μg mL-¹). For media containing orysastrobin, salicylhydroxamic acid (Sigma-Aldrich, St. Louis, MO, USA) was added at a final concentration of 100 μg mL-¹. The plates were incubated at 25 °C in darkness. Colony diameters were measured after 2 days, and EC50 values were calculated using the R package drc (Ritz et al., 2015). The experiment was performed with 2 technical replicates and 2 biological replicates.
2.3 In planta fungicide control
Mycelial plugs (5 mm in diameter; n = 20) of isolate KACC40106, taken from fresh colonies grown on PDA, were individually transferred into 200 mL of potato dextrose broth (PDB) and incubated at 25 °C on a shaker at 150 rpm. After 5 days, the mycelia were ground using a blender and sprayed evenly onto 6-week-old rice plants grown in pots (0.15 m2 per pot). A total of 500 mL of fungicide solution was applied at the recommended concentration according to their product instructions using mobile spraying equipment (MSE) while moving across the plots prior to pathogen inoculation: florylpicoxamid (100 μg mL-¹), fludioxonil (100 μg mL-¹), flutolanil (75 μg mL-¹), ipflufenoquin (100 μg mL-¹), orysastrobin (60 μg mL-¹), and pencycuron (100 μg mL-¹). After incubation at 25 °C for 7 days, all rice pots were photographed by a Nikon EOS 600D camera, and proportion of blighted area was determined by calculating the ratio of diseased area to the total area of plants (pixel-based analysis) using Photoshop software ™ (Kwack et al., 2005). The proportion of blighted area was calculated using the formula: Proportion of blighted area = (Pixels of blighted area)/(Pixels of total plant area). Control efficacy (%) was then calculated as follows: Control efficacy (%) = (1 - Proportion of blighted area of fungicide treatment/Average Proportion of blighted area of pathogen treatment) × 100 (Lee et al., 2023). Statistical analysis was conducted using Tukey’s honestly significant difference (HSD) test.
2.4 In planta fungicide application using mobile spraying equipment
Two fungicides exhibiting the highest control efficacy were selected for controlling R. solani at reduced concentrations using mobile spraying equipment (MSE). Fungicide concentrations were designated as “1X”, “1/2X”, and “1/4X”, corresponding to 100%, 50%, and 25% of the recommended concentration, respectively. Water was used as the control treatment. Spraying was conducted at a speed of 1 m/s and a pressure of 250 kPa (2.5 bar) using a TeeJet® XR11002 nozzle connected to a water pump and positioned 1 m above the ground. After the rice plants had been air-dried for several hours, the pathogen was inoculated onto them. During fungicide spraying, water-sensitive papers (Syngenta, Basel, Switzerland) were placed at the same height as the rice plants to visualize the dispersion patterns of each fungicide at different concentrations. Spray application efficiency was quantitatively assessed by analyzing the stained areas on water-sensitive papers using a WSP scanner program (Park et al., 2020). MSE spraying was performed twice, separated by an interval of 7 days. Proportion of blighted area was assessed using the method described above. Disease severity was also evaluated by manually rating plants on a conventional 0–5 scale (0: no disease; 1: ≤20%; 2: 21–40%; 3: 41–60%; 4: 61–80%; 5: >80%) (Figure 1), and the trends between disease severity and incidence (percentage of blighted tillers per total tillers) were analyzed.
3 Results
3.1 In vitro fungicide sensitivity
The sensitivity of Rhizoctonia solani isolates (KACC40101, KACC40106, and KACC45811) to six fungicides was evaluated in vitro. Overall, these isolates showed similar fungicide sensitivity patterns. The EC50 values of the three isolates for fludioxonil, flutolanil, orysastrobin, and pencycuron were below 10 μg mL-¹ (Table 1). The EC50 values for florylpicoxamid ranged from 42.43 to 59.35 μg mL-¹. However, these isolates exhibited resistance to ipflufenoquin, with EC50 values exceeding 100 μg mL-¹ (Table 1).
3.2 In planta fungicide control efficacy
Pathogenicity tests using isolates KACC40101, KACC40106, and KACC45811 were conducted on rice plants, among which isolate KACC40106 showed the highest pathogenicity (Supplementary Figure S1). Thus, isolate KACC40106 was selected as the representative isolate for further experiments. In the fungicide efficacy test against sheath blight, the lowest Proportion of blighted area was observed with flutolanil treatment (2.07% ± 0.60), followed by orysastrobin treatment (9.92% ± 1.23), indicating relatively high efficacy of disease control of these two fungicides (Figure 2).
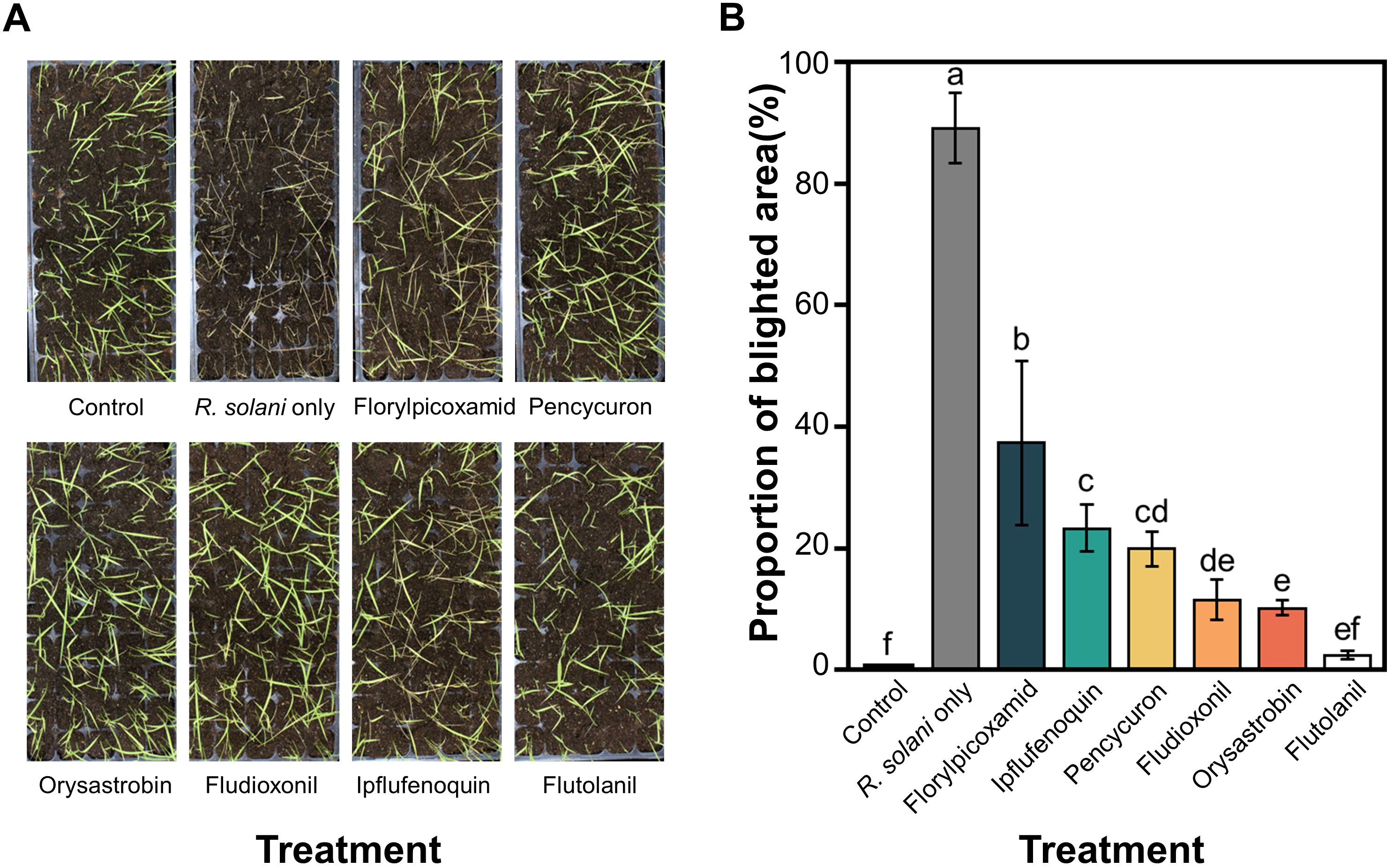
Figure 2. In planta sheath blight control efficacy of 6 different fungicides. (A) Photographs of symptoms on rice plants in each treatment. (B) Bar graph of Proportion of blighted area in each treatment. Statistical differences are indicated by significance letters as determined by Tukey’s honestly significant difference (HSD) test. Treatments sharing the same letter were not significantly different at p < 0.05.
3.3 Control efficacy of two fungicide using mobile spraying equipment
Fungicides flutolanil and orysastrobin, showing higher in planta control potentials against sheath blight, were further used to evaluate their efficacy at reduced concentrations using mobile spraying equipment (MSE). These fungicides were uniformly sprayed at different concentrations, as confirmed by coverage on water-sensitive papers (WSPs) (Figure 3A). The average WSP coverage across all concentrations was approximately 20% (Figure 3B). Fourteen days after MSE spraying, different phenotypes were observed among treatments (Figure 4A). Proportion of blighted area trends closely corresponded to disease severity results. The pathogen-only groups exhibited the highest Proportion of blighted area and disease severity. Control efficacy was maintained even at lower concentrations (1/2X and 1/4X) of both flutolanil and orysastrobin. The concentration with the lowest control efficacy was 1X for both flutolanil and orysastrobin (Figure 4B).
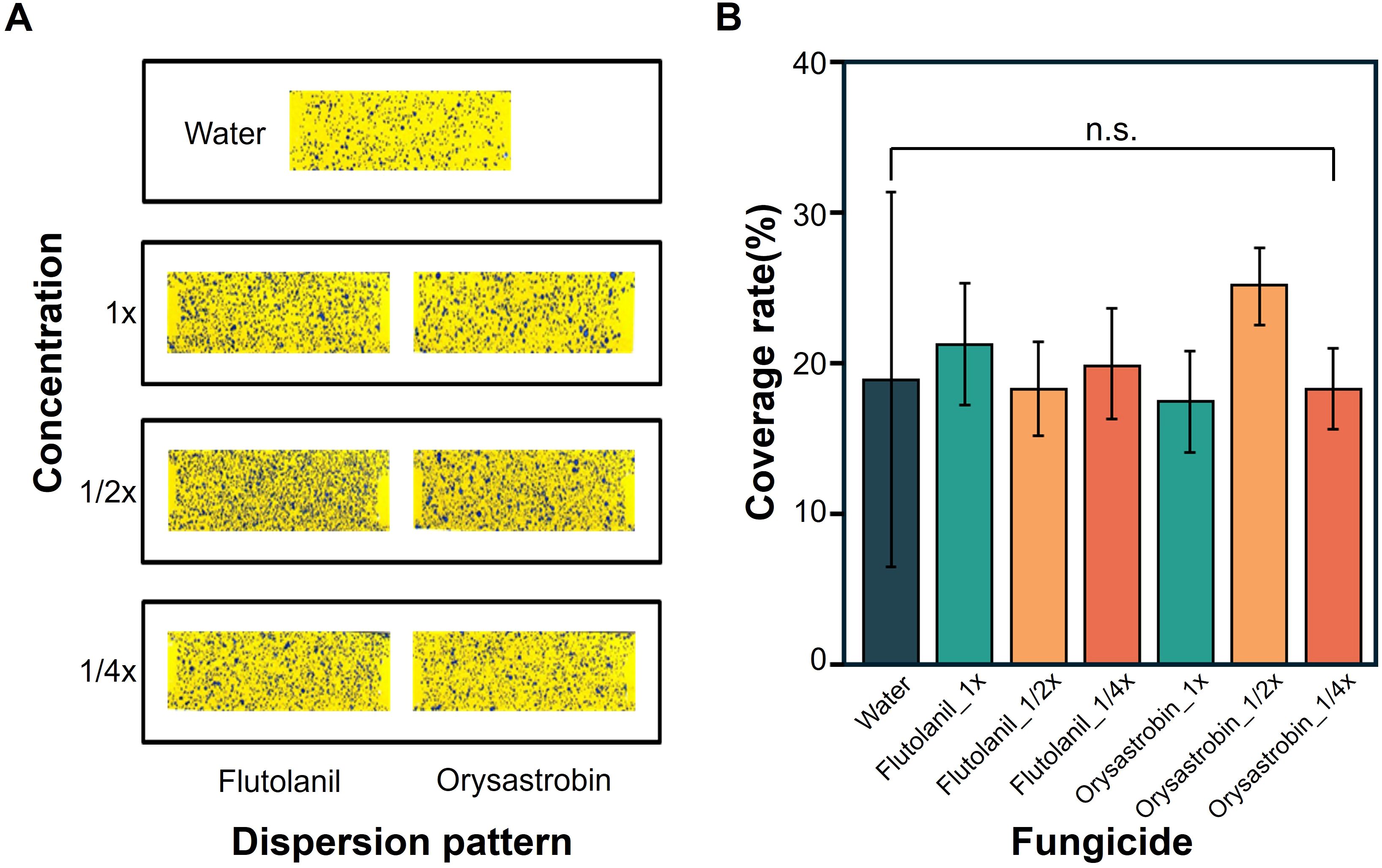
Figure 3. Distribution and coverage patterns of fungicide spray droplets across different treatments as visualized using water-sensitive papers (WSP). (A) Photographs of water-sensitive papers (WSP) with different surface coverages for each treatment. (B) Bar graph of calculated coverage rates in each treatment. Statistical differences based on coverage rate are indicated by significance letters (Tukey’s HSD, p < 0.05).
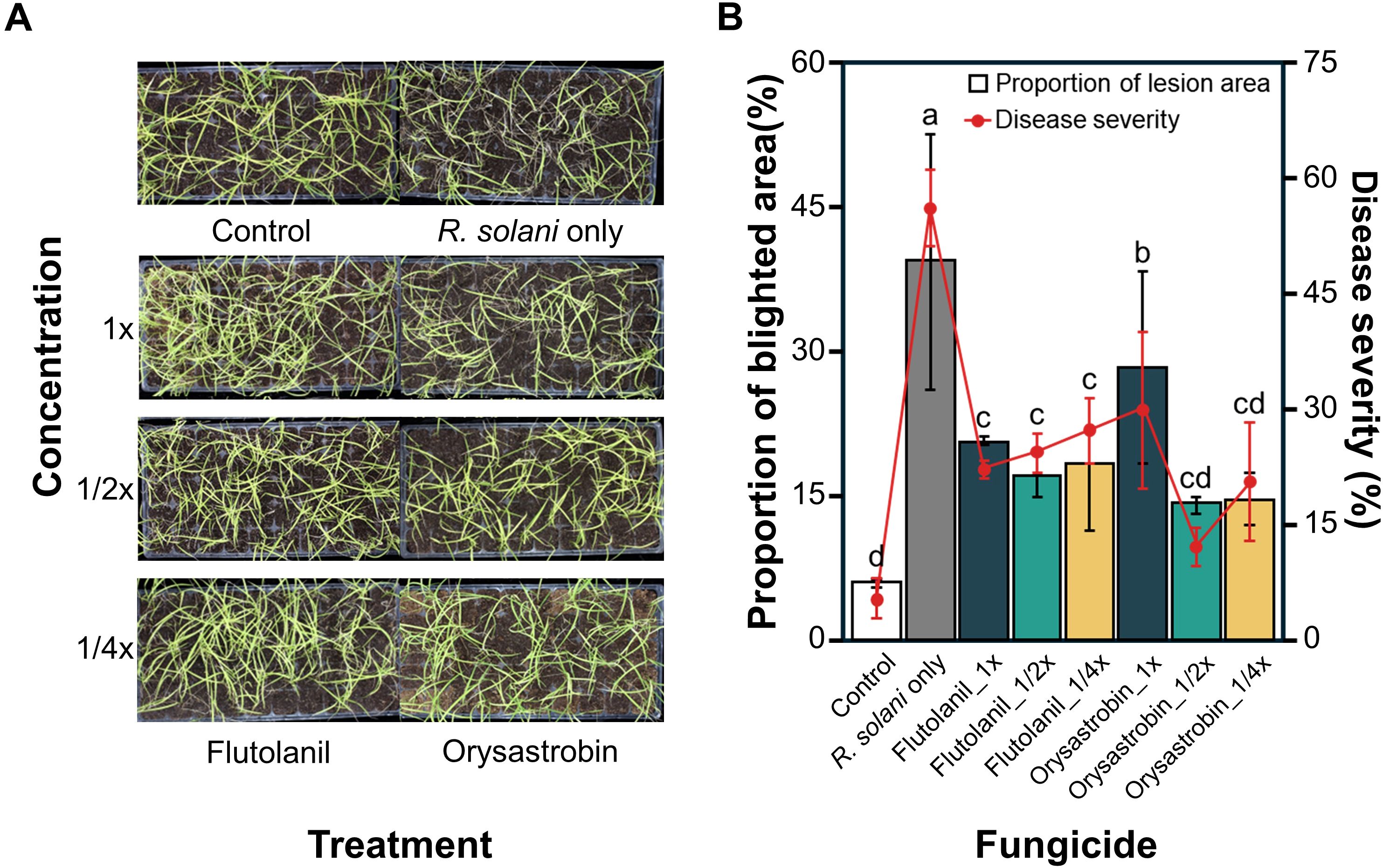
Figure 4. In planta efficacy of flutolanil and orysastrobin at different concentrations against rice sheath blight. (A) Photographs of symptoms on rice plants in each treatment. (B) Proportion of blighted area (bar graph) and disease severity (line graph) across treatments. Statistical differences based on proportion of blighted area are indicated by significance letters (Tukey’s HSD, p < 0.05); different letters indicate significant differences among treatments.
4 Discussion
This study tested the control efficacy of fungicides flutolanil and orysastrobin at diluted concentrations using mobile spraying equipment (MSE) for controlling the sheath blight pathogen Rhizoctonia solani, after an initial evaluation identified them as the most effective fungicides against this pathogen. Mobile spraying equipment reproduced UAV spray behavior in the greenhouse, enabling reproducible spray settings and efficacy benchmarks for subsequent UAV applications, thus helping to determine minimum effective fungicide concentrations. Notably, concentrations as low as 1/2 or even 1/4 of the recommended dose were sufficient to control sheath blight effectively when applied using MSE.
According to the in vitro sensitivity assay, isolates of R. solani were sensitive to the fungicides orysastrobin, flutolanil, pencycuron, and fludioxonil (EC50 values ranged from 0.007 to 6.53 μg mL-¹). Reduced sensitivity was observed for florylpicoxamid and ipflufenoquin, which showed much higher EC50 values (42.43–59.35 μg mL-¹ and >100 μg mL-¹, respectively). Despite reduced in vitro sensitivity, both fungicides showed moderate efficacy in planta. Similar trends have been reported in Sclerotium rolfsii on peanut, where in vitro sensitivity was not significantly correlated with field efficacy, and isolates with lower sensitivity were controlled as effectively as those with higher sensitivity (Franke et al., 1998). The finding indicates that in vitro sensitivity does not always correspond to field performance, likely due to factors such as fungicide uptake, translocation within plant tissues, environmental conditions, and pathogen–host interactions.
Flutolanil and orysastrobin exhibited the highest efficacy in controlling R. solani under in planta conditions, suggesting their potential as effective alternatives for rice sheath blight management. Consistently, flutolanil has been reported to achieve long-term control of sheath blight by exhibiting excellent translocation throughout the plant (Li et al., 2014). Orysastrobin has also demonstrated good fungicidal efficacy against this pathogen in a previous study (Stammler et al., 2007). In this study, these two fungicides showed greater disease control efficacy at concentrations of 1/2X and 1/4X than at 1X, which may be attributed to some complex factors such as plant growth stage or potential negative effects that a higher dosage might have on the plants. It is worth noting, however, that the long-term application of low concentrations in practical settings requires consideration, as it could increase the risk of fungicide resistance, particularly target site-based resistance, such as SDHIs and QoIs (Amaradasa and Everhart, 2016; Ayer et al., 2020).
As an indoor simulation system, MSE provides an effective platform for optimizing UAV operational parameters and application conditions. In this study, MSE was used to refine the fungicide application method for rice sheath blight control by reducing fungicide concentrations. A similar approach was reported by Park et al. (2024), who developed an efficient method using regulated concentrations of fungicides to manage garlic leaf blight.
Unmanned aerial vehicles (UAVs), or drones, offer a promising tool in precision agriculture due to their numerous advantages. Recent research has increasingly focused on developing UAV-based spraying methods for disease prevention and control (Baek et al., 2024; Kayode et al., 2024; Köycü et al., 2024; Li et al., 2024; Murata et al., 2024; Park et al., 2024; Ragiman et al., 2024). In rice production, UAVs have demonstrated effectiveness in managing multiple insect pests and diseases (Murata et al., 2024). In conclusion, this study optimized a fungicide application method suitable for UAV-based management of rice sheath blight. Further research is needed to explore broader practical applications of these optimized methods under field conditions.
Data availability statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Author contributions
HL: Writing – review & editing, Data curation, Investigation, Writing – original draft, Methodology. H-RJ: Methodology, Writing – original draft, Writing – review & editing, Data curation, Investigation. SC: Investigation, Writing – review & editing, Methodology, Writing – original draft. SP: Writing – original draft, Writing – review & editing, Investigation. J-hC: Writing – review & editing, Investigation, Writing – original draft. HC: Writing – review & editing, Writing – original draft, Investigation. S-WP: Writing – review & editing, Investigation, Writing – original draft. WA: Investigation, Writing – review & editing, Writing – original draft. S-WH: Writing – original draft, Writing – review & editing, Methodology, Project administration. HS: Supervision, Writing – original draft, Writing – review & editing, Conceptualization, Project administration.
Funding
The author(s) declare financial support was received for the research and/or publication of this article. This research was funded by Rural Development Administration, project number RS-2022-RD010344 and the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT), grant number RS-2025-00555346. This work was also carried out with the support of Research Program for Agriculture Science and Technology Development (Project No. RS-2025-02305681), Rural Development Administration, Republic of Korea.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fagro.2025.1687036/full#supplementary-material
References
Amaradasa B. S. and Everhart S. E. (2016). Effects of sublethal fungicides on mutation rates and genomic variation in fungal plant pathogen, Sclerotinia sclerotiorum. PloS One 11, e0168079. doi: 10.1371/journal.pone.0168079
Ayer K. M., Choi M.-W., Smart S. T., Moffett A. E., and Cox K. D. (2020). The effects of succinate dehydrogenase inhibitor fungicide dose and mixture on development of resistance in Venturia inaequalis. Appl. Environ. Microbiol. 86, e01196-20. doi: 10.1128/AEM.01196-20
Baek J.-W., Eun H.-R., Kim S.-M., Lee Y.-J., Lee Y.-H., Lee H.-S., et al. (2024). Analysis of azoxystrobin and difenoconazole in brown rice and dried straw using LC-MS/MS, and comparison of efficacy by UAV spraying with different dosages and adjuvants. Korean. J. Environ. Agric. 43, 365–375. doi: 10.5338/KJEA.2024.43.35
Cao L., Cao C., Wang Y., Li X., Zhou Z., Li F., et al. (2017). Visual determination of potential dermal and inhalation exposure using allura red as an environmentally friendly pesticide surrogate. ACS Sustain. Chem. Eng. 5, 3882–3889. doi: 10.1021/acssuschemeng.6b03050
Chen Y., Zhang A., Wang W., Zhang Y., and T. -C. (2012). Baseline sensitivity and efficacy of thifluzamide in Rhizoctonia solani. Ann. Appl. Biol. 161, 247–254. doi: 10.1111/j.1744-7348.2012.00569.x
Franke M. D., Brenneman T. B., and Stevenson K. L. (1998). Stem rot of peanut: Relationship between in vitro fungicide sensitivity and field efficacy of fungicides. Peanut. Sci. 25, 76–80. doi: 10.3146/i0095-3679-25-2-4
Ishii H. and Kurosaki A. (2017). “Sensitivity of fungal strains isolated from rice sheath blight symptom to the SDHI fungicides furametpyl and benzovindiflupyr,” in Modern Fungicides and Antifungal Compounds. Eds. Deising H. B., Fraaije B., Mehl A., Oerke E. C., Sierotzki H., and Stammler G. (Braunschweig, Germany: Braunschweig: Deutsche Phytomedizinische Gesellschaft), 193–196.
Kayode J. F., Amudipe S. O., Nwodo C. W., Afolalu S. A., Akinola A. O., Ikumapayi O. M., et al. (2024). Development of remote-controlled solar-powered pesticide sprayer vehicle. Discov. Appl. Sci. 6, 101. doi: 10.1007/s42452-024-05748-x
Köycü N. D., Özyurt H. B., and Çelen I. H. (2024). Comparison of aerial and ground spraying applications in controlling fusarium crown rot in wheat. Int. J. Agric. Biol. Eng. 17, 73–80. doi: 10.25165/j.ijabe.20241705.8553
Kuster C. J., Kohler M., Hovinga S., Timmermann C., Hamacher G., Buerling K., et al. (2023). Pesticide exposure of operators from drone application: A field study with comparative analysis to handheld data from exposure models. ACS Agric. Sci. Technol. 3, 1125–1130. doi: 10.1021/acsagscitech.3c00253
Kwack M. S., Kim E. N., Lee H., Kim J.-W., Chun S.-C., and Kim K. D. (2005). Digital image analysis to measure lesion area of cucumber anthracnose by Colletotrichum orbiculare. J. Gen. Plant Pathol. 71, 418–421. doi: 10.1007/s10327-005-0233-0
Lee G., Choi H., Liu H., Han Y.-H., Paul N. C., Han G. H., et al. (2023). Biocontrol of the causal brown patch pathogen Rhizoctonia solani by Bacillus velezensis GH1–13 and development of a bacterial strain specific detection method. Front. Plant Sci. 13. doi: 10.3389/fpls.2022.1091030
Li S., Hou Y., Peng D., Meng L., Wang J., Zhou M., et al. (2014). Baseline sensitivity and control efficacy of flutolanil in Rhizoctonia solani. Australas. Plant Pathol. 43, 313–320. doi: 10.1007/s13313-014-0272-0
Li Y., Li Y., Pan X., Li Q. X., Chen R., Li X., et al. (2018). Comparison of a new air-assisted sprayer and two conventional sprayers in terms of deposition, loss to the soil and residue of azoxystrobin and tebuconazole applied to sunlit greenhouse tomato and field cucumber. Pest Manage. Sci. 74, 448–455. doi: 10.1002/ps.4728
Li X., Liang Z., Yang G., Lin T., and Liu B. (2024). Assessing the severity of verticillium wilt in cotton fields and constructing pesticide application prescription maps using unmanned aerial vehicle (UAV) multispectral images. Drones 8, 176. doi: 10.3390/drones8050176
Mohapatra B., Sahoo A., and Jani A. (2025). Sheath blight of rice: An overview of the disease. Vigyan. Varta. 6, 282–284.
Murata H., Masui S., and Tsuchida Y. (2024). Efficacy evaluation of low-volume concentrate application of pesticides by unmanned aerial vehicle (UAV) using an indoor spraying device. Appl. Entomol. Zool. 59, 103–110. doi: 10.1007/s13355-023-00858-1
Neha K., Naveenkumar R., Balabaskar P., and Manikandan P. (2017). Evaluation of fungicides against sheath blight of rice caused by Rhizoctonia solani (Kuhn.). ORYZA-. Int. J. Rice 54, 470. doi: 10.5958/2249-5266.2017.00064.9
Park S.-W., An W., Choi S., Choi H., Choi J.-H., Park S., et al. (2024). Evaluation of fungicidal control of garlic leaf blight using mobile spraying equipment. Korean. J. Pestic. Sci. 28, 291–297. doi: 10.7585/kjps.2024.28.3.291
Park J., Hong S.-W., and Lee I. (2020). Evaluation of efficiency of livestock vehicle disinfection systems using water-sensitive paper. J. Korean. Soc Agric. Eng. 62, 87–97. doi: 10.5389/KSAE.2020.62.4.087
Persaud R., Khan A., Isaac W.-A., Ganpat W., and Saravanakumar D. (2019). Plant extracts, bioagents and new generation fungicides in the control of rice sheath blight in Guyana. Crop Prot. 119, 30–37. doi: 10.1016/j.cropro.2019.01.008
Ragiman S., Talluri K. B., Nrg V., and Vidya B. (2024). Unmanned aerial vehicle (UAV)-assisted pesticide application for pest and disease management in rice. Int. J. Agric. Biol. Eng. 17, 88–95. doi: 10.25165/j.ijabe.20241705.8640
Rincón V. J., Sánchez-Hermosilla J., Páez F., Pérez-Alonso J., and Callejón Á.J. (2017). Assessment of the influence of working pressure and application rate on pesticide spray application with a hand-held spray gun on greenhouse pepper crops. Crop Prot. 96, 7–13. doi: 10.1016/j.cropro.2017.01.006
Ritz C., Baty F., Streibig J. C., and Gerhard D. (2015). Dose-response analysis using R. PloS One 10, e0146021. doi: 10.1371/journal.pone.0146021
Sackey O. K., Feng N., Mohammed Y. Z., Dzou C. F., Zheng D., Zhao L., et al. (2025). A comprehensive review on rice responses and tolerance to salt stress. Front. Plant Sci. 16. doi: 10.3389/fpls.2025.1561280
Stammler G., Itoh M., Hino I., Watanabe A., Kojima K., Motoyoshi M., et al. (2007). Efficacy of orysastrobin against blast and sheath blight in transplanted rice. J. Pestic. Sci. 32, 10–15. doi: 10.1584/jpestics.g06-22
Umeda S., Yoshikawa N., and Seo Y. (2022). Cost and workload assessment of agricultural drone sprayer: A case study of rice production in Japan. Sustainability 14, 10850. doi: 10.3390/su141710850
Wang G., Lan Y., Qi H., Chen P., Hewitt A., and Han Y. (2019). Field evaluation of an unmanned aerial vehicle (UAV) sprayer: Effect of spray volume on deposition and the control of pests and disease in wheat. Pest Manage. Sci. 75, 1546–1555. doi: 10.1002/ps.5321
Keywords: rice, sheath blight, fungicide, disease control, mobile spraying equipment
Citation: Liu H, Jang H-R, Choi S, Park S, Choi J-h, Choi H, Park S-W, An W, Hong S-W and Sang H (2025) Assessment of fungicide-based control of sheath blight using mobile spraying equipment. Front. Agron. 7:1687036. doi: 10.3389/fagro.2025.1687036
Received: 16 August 2025; Accepted: 11 November 2025; Revised: 11 November 2025;
Published: 27 November 2025.
Edited by:
Omer Frenkel, Agricultural Research Organization (ARO), IsraelReviewed by:
Mehi Lal, ICAR-Central Potato Research Institute, IndiaFrancisco José Sautua, University of Buenos Aires, Argentina
Copyright © 2025 Liu, Jang, Choi, Park, Choi, Choi, Park, An, Hong and Sang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Hyunkyu Sang, aGtzYW5nQGpudS5hYy5rcg==
†These authors have contributed equally to this work
 Haifeng Liu
Haifeng Liu Hyeong-Rok Jang
Hyeong-Rok Jang Sungyu Choi1
Sungyu Choi1 Hyunkyu Sang
Hyunkyu Sang