- 1State Key Laboratory for Infectious Disease Prevention and Control, National Institute for Communicable Disease Control and Prevention, Chinese Center for Disease Control and Prevention, Beijing, China
- 2Huadong Medical Institute of Biotechniques, Nanjing, China
- 3Department of Veterinary and Biomedical Sciences, College of Veterinary Medicine, University of Minnesota, St. Paul, MN, United States
The poly-γ-D-glutamic acid capsule and anthrax toxins are major virulence factors of Bacillus anthracis. Genes responsible for capsule biosynthesis are located on pXO2, whereas genes encoding the toxins, which are composed of edema factors, lethal factors, and protective antigens (PA), are located on pXO1. In this study, we found that the pag null mutation not only eliminated the production of the protective antigen, it also eliminated the ability of the B. anthracis Pasteur II strain to form capsules. qPCR analysis revealed that the deletion of pag decreased the transcription levels of the capABCD operon and its regulatory genes acpA and acpB. The introduction of the acpA or acpB plasmid complemented the effect of the pag null mutation on capsule formation. Taken together, the above results suggest that PA probably affects capsule biosynthesis by altering the expression of acpA and acpB. In addition, we found that the deletion mutation of pag remarkably attenuated bacterial pathogenicity in a mouse model of infection. Our results indicate that besides encoding the protective antigen, the pag gene of pXO1 is also involved in the modulation of capsule biosynthesis. Our findings provide new insight into the regulation mechanisms of capsule formation in B. anthracis Pasteur II strain.
Introduction
Anthrax is an anthropozoonosis caused by Bacillus anthracis. Clinical manifestations of this disease include cutaneous, gastrointestinal, or respiratory infections. Sporadic prevalence in humans and animals has led to small outbreaks of this disease worldwide (Liang, 1995). As a biological warfare agent, B. anthracis has the potential to cause devastating societal and economic losses. Therefore, understanding the pathogenic mechanism of B. anthracis remains relevant and has been the focus of research in recent years (Moayeri et al., 2015).
The virulence factors of B. anthracis, including anthrax toxins and the antiphagocytic polyglutamic capsule, are encoded by the pXO1 and pXO2 plasmids, respectively. The loss of these two plasmids leads to the loss of toxins and capsule formation, resulting in a loss of pathogenicity (Mikesell et al., 1983; Uchida et al., 1986). The pathogenicity of B. anthracis toxins and the capsule has been intensively explored in various animal models (Smith and Keppie, 1954; Stanley and Smith, 1961). Toxicity was observed only when the protective antigen (PA) was present with either the edema factor (EF) or lethal factor (LF), indicating that PA is a toxin mediator (Fish et al., 1968; Remmele et al., 1968). These toxin factors are encoded by pag cya, and lef, respectively, and are regulated by the atxA gene on pXO1 (Okinaka et al., 1999).
A 63-kDa fragment of PA, PA63, oligomerizes into heptamers or octamers to form the LF/EF binding sites (Leppla, 1988; Singh et al., 1989, 1999). Upon binding to the cell receptor, the PA binds LF and EF. After endocytosis and endosomal acidification, LF and EF are transferred into the cytoplasm to access their targets, including the mitogen-activated protein kinase kinase (MAPKK) family and ATP (Abrami et al., 2003). The lethal toxin inhibits the MEKs signaling pathway, and in turn, prevents the release of chemotactic factors and cytokines, which can induce toxic shock and is the reason behind the high mortality rate of B. anthracis infections (Milne et al., 1994; Gordon et al., 1995; Erwin et al., 2001; Abrami et al., 2003).
The poly-γ-D-glutamic acid capsule is the other key virulence factor and is synthesized by the capBCAD operon on pXO2 (Makino et al., 1989). Besides the anthrax toxin activator, atxA, encoded on pXO1, the expression of toxin genes, including cya, lef, and pag on pXO1, are affected by the expression levels of the capsule genes on pXO2, as well as increased carbon dioxide (CO2) and bicarbonate () levels during the time the pathogen enters the host (Bartkus and Leppla, 1989; Koehler et al., 1994; Sirard et al., 1994). Both atxA and acpA genes modulate the toxins and capsule production, respectively (Uchida et al., 1993; Guignot et al., 1997; Hoffmaster and Koehler, 1997; Uchida et al., 1997). Our previous studies clearly demonstrated that the B. anthracis Pasteur II vaccine strain possesses low copy numbers of the pXO1 plasmid, which dramatically affect the expression levels of toxin genes (Liang et al., 2016).
It is well established that the acpA gene on pXO2 is responsible for controlling capsule synthesis in B. anthracis. However, the reason why some B. anthracis strains harbor pXO2 but lack capsule production is unclear (Ezzell and Welkos, 1999). In this study, we demonstrated that the pag gene on pXO1 plays an important role in capsule biosynthesis of the B. anthracis Pasteur II strain by using gene deletion and complementation studies. Furthermore, we found that PA affects the transcription of acpA gene, which in turn mediates capsule production.
Materials and Methods
Bacterial Vaccine Strain
B. anthracis vaccine Pasteur II strain was kindly provided by the Institute of Lanzhou Biological Products in China.
Construction of Gene Mutants and Overexpression Strains
Construction of the pag Gene Promoter Deletion Strain
We designed primers directed to the upstream region of the pag gene promoter: 141819F_BamHI and 143181R_crossover (Table 1) and introduced a BamHI endonuclease recognition site into the 141819F_BamHI primer. An upstream homology arm approximately 1,360 bp in length was amplified by PCR with wild-type strain DNA used as template. The downstream primers were 143779F_crossover and 144559R_Bgl with a BglII site introduced into the 144559R_Bgl primer (Table 1). A downstream homology arm approximately 800 bp in length was amplified. Since parts of the 143181R_crossover and 143779F_crossover sequences were complementary to each other, we simply mixed the upstream and downstream PCR products and amplified a 2.2-kb fragment composed of an sequences of the upstream and downstream arms of the pag gene using the 141819F Bam and 144559R Bgl primers. This amplicon was double-digested with BamHI and BglII and cloned into the BamHI/BglII site of pMAD, to produce the recombinant plasmid pag-pMAD. The recombinant plasmid was electroporated into the Pasteur II strain. Integrons containing pag-pMAD were obtained at 42°C and subsequently cultured at 30°C to promote gene replacement. The target gene deletion strain, PasteurII-pagpromoterKO, was identified from all erythromycin sensitive strains by using PCR and confirmed by sequencing.
Construction of pag Gene Complementary Strain
Primers directed to the upstream region of the pag gene promoter were designed using Oligo 6.0 software. Four primer sets, PApr-175SacF and PA-RSacII, PApr-385SacF and PA-RSacII, PApr-432SacF and PA-RSacII, 142918SacF and PApr-604SacR (Table 1), and high-fidelity polymerase Pyrobest (Takara, Dalian, China) were used to amplify different sizes of fragments covering pag and its promoter region on pXO1 (Figure 1). After initial denaturation at 95°C for 5 min, reactions were performed for 30 cycles, with each cycle consisting of denaturation (95°C for 30 s), annealing (55°C for 30 s), and extension (72°C for 2 min). A 2.3-kb fragment was amplified, purified, digested with SacII, and cloned into the SacII site of the expression vector pFF40. The recombinant plasmid was transformed into Escherichia coli (DH5α), and the positive clones were screened by PCR and identified by double digestion. The recombinant plasmid was electroporated into the PasteurII-pag promoterKO strains. The positive clone was sequenced to ensure no mutations. The positive strain was designated PasteurII-pagpromoter-KO-CO.
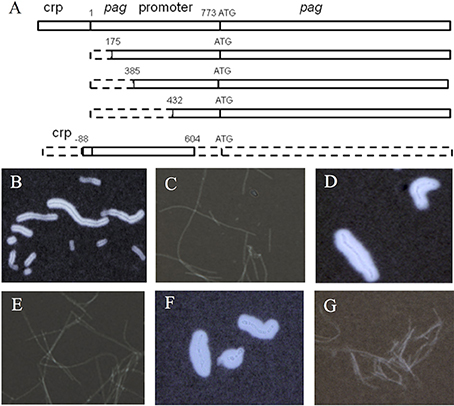
Figure 1. Effect of the pag gene on capsule formation of Pasteur II strain. Diagram of the pag promoter region and pag gene location (A). The dashed boxes indicate truncated regions; the solid boxes indicate untruncated regions. Microscope images of India ink stained Pasteur II strain (B), PasteurII-pag promoter KO strain with the longest fragment deletion of the pag promoter region (C), PasteurII-pag promoterKO-CO strain (D), PasteurII-pagKO strain (E), PasteurII-pagKO-CO strain (F), and negative control, Sterne strain without capsule formation (G). The images are representative of 3 independent experiments.
Construction of pag Gene Deletion Strain
The pag gene deletion strain was obtained using methods similar to those described above. We designed primers to the upstream region of pag: 143120F_BamHI and 143898R_crossover and introduced a BamHI site into the 143120F_Bam primer (Table 1). An upstream homology arm of approximately 800 bp was amplified by PCR using wild-type strain DNA as template. A downstream homology arm approximately 700 bp in length was amplified using the primers: 145987F_crossover and 146648R_Bgl with a BglII site added to the 146648R_Bgl primer (Table 1). The fragment containing both 143898R and 145987F arms was amplified by crossover PCR, double digested with BamHI and BglII, and cloned into the pMAD plasmid, to produce the recombinant plasmid pag-pMAD. The recombinant plasmid was electroporated into the PasteurII strain. The target gene deletion strain PasteurII-pagKO was identified from all erythromycin sensitive strains using PCR (Supplementary Figure 1) and confirmed by sequencing.
Construction of pag Gene Complementary Strain
The pag gene on pXO1 was replaced using methods similar to those described above. Primers directed to pag were designed using Oligo 6.0 software with SacII restriction sites: PA-FSacII and PA-RSacII. The PCR product was digested with SacII and cloned into the SacII site of the expression vector pFF40. The recombinant plasmid was transformed into E. coli DH5α and the positive clones were screened by PCR and identified by double digestion. The resultant plasmid was electroporated into PasteurII-pagKO. The positive clone was selected with kanamycin and designated PasteurII- pagKO-CO after identification by PCR (Supplementary Figure 2) and confirmation by sequencing.
Construction of acpA or acpB Gene Complementation Strain in pag Knockout Mutant
Similar methods were applied to construct the acpA gene complementation strain. The primers used were acpAOE-p40-F and acpAOE-p40-R (Table 1). The PCR product was digested with SacII and cloned into the SacII site of pFF40. The recombinant plasmid was transformed into E. coli DH5α and the positive clone was identified by double digestion. The resultant plasmid was electroporated into PasteurII-pagKO, confirmed by DNA sequencing, and designated PasteurII-pagKO-acpA and PasteurII-pagKO-acpB
RNA Isolation and Quantitative Real-time PCR Analysis (qPCR)
Overnight cultures of strains tested were incubated in LB medium and grown to mid-log phase (OD600 = 0.6) at 37°C with shaking. The cells were harvested by centrifugation, and total RNA was purified using the RNeasy Mini Kit (Qiagen) according to the manufacturer's instructions. The primers used for each target are listed in Table 1. cDNA was synthesized from 1 μg RNA using Superscript III reverse transcriptase (Invitrogen) and random hexamers following the manufacturer's protocol. The cDNA was diluted 5-fold and was amplified by qPCR. The gene expression levels as indicated by CT values were compared among the strains tested. 16S rRNA was used as a reference gene for normalization. The qPCR was repeated three times.
Western-Blotting Analysis
Strains were cultured overnight, and samples of the supernatant were collected and filtered through 0.22 μm-pore-size syringe filters. An aliquot of 5 ml of the filtered supernatant was mixed with an equal volume of 95% ethanol and incubated at 4°C overnight, followed by centrifugation. The precipitate was dried at 22–25°C and resuspended in 50 μl PBS. Following the addition of the loading buffer, the sample was heated to 100°C for 5 min. Twenty microliter of each sample (total protein content 5 μg) was loaded onto a 12% SDS-PAGE gel. The samples were then transferred onto a nitrocellulose membrane (Bio-Rad). The membranes were blocked for 1 h with TBS-T (20 mm Tris base, 137 mm NaCl, 0.1% Tween20) containing 5% dry milk at room temperature. The membranes were then incubated with either an anti-protective antigen (PA) antibody or anti-LF antibody (Thermo Scientific), and then diluted 1:2000 in TBS-T-5% milk overnight at 4°C. Membranes were washed in TBS-T and incubated with HRP-conjugated goat anti-mouse IgG secondary antibody diluted 1:4,000 in TSB-T-5% milk for 1 h at room temperature. After the same washing procedure, proteins were detected using the ECL Western Blot Substrate (Pierce) according to the manufacturer's instructions.
Gel Mobility Shift DNA Binding Assay
To explore whether protective antigen (PA) is able to directly regulate the transcription of acpA gene, we performed a gel shift assay. A DNA fragment of the upstream region of acpA was amplified by PCR using biotin-labeled primers: acpApromo_F and acpApromo_R (Table 1). The amplified DNA fragments were purified using the QIAquick PCR Purification kit (Qiagen) according to the manufacturer's instructions. DNA binding and electrophoresis were performed as described (Yang et al., 2015). Briefly, commercial-grade protective antigen protein (Listbiological Laboratories, Inc.) was added to a 20 μl mixture containing 0.2 pmol biotin-labeled DNA, 1 mg of poly(dI-dC), 25 mM NaH2PO4 (pH 8.0), 50 mM NaCl, 2 mM MgCl2, 1 mM dithiothreitol (DTT), 10% glycerol, and 0.1 mM EDTA. The concentrations of PA protein used were 0.4, 1, 2, 4, and 8 μM (corresponding to 0.2, 0.5, 1, 2, and 4 μg, respectively). Unlabeled DNA fragments of the promoter region were added into the reaction at a 120-fold concentration over the labeled probe to inhibit non-specific binding. Bovine serum albumin (BSA) was added to prevent non-specific protein binding. The binding reaction was initiated by the addition of PA, and the reaction mixture was incubated at room temperature for 25 min. Samples were then loaded onto a 6% native polyacrylamide gel (acrylamide-bisacrylamide [29:1] in 0.5 × Tris-borate-EDTA [TBE] buffer). Electrophoresis was performed for 3 h at 4°C with 7 V cm−1, and the gels were electroblotted onto nylon membranes in 0.5 × TBE at 300 mA for 90 min at 4°C. After cross-linking the DNA-protein hybrids with UV radiation, the membranes were hybridized with stabilized streptavidin-HRP conjugate (Thermo Scientific) followed by incubation with DAB substrate at room temperature for 20–30 min to optimize signal intensity.
Microscopy Analysis of Capsule with India Ink Staining
The B. anthracis isolates were incubated in a LB medium with 0.9% NaHCO3 under 5% CO2 for 8 h. The bacterial cells were stained with India ink and were observed under a microscope. The Sterne strain without capsule formation was used as negative control. The bacterial capsule is visible in India ink because it excludes the ink particles. Each experiment was repeated at least three times.
Mouse Infection
Six to eight week old BALB/C male mice (18–20 g) were used for the assessment of pathogenicity of the Pasteur II pag knockout mutant. Briefly, mice were randomly divided into two groups (n = 6) and injected with 104 CFU ml−1 Pasteur II and PasteurII-pagKO, respectively, via subcutaneous injection to the inner thigh. The mice were observed continuously for 60 min, and afterwards once every 12 h. All experiments involving animals were performed in accordance with the protocols approved by the Animal Care and Use Committee of the National Institute of Allergy and Infectious Diseases, National Institutes of Health, China. Significance in animal survival was determined by Fisher exact test.
Results
The Mutation of pag Promoter or pag Gene Eliminates Capsule Formation
The expression levels of the capsule genes on pXO2 affect the expression of toxin genes, including cya, lef, and pag on pXO1 (Barkus and Leppla, 1989; Koehler et al., 1994; Sirard et al., 1994), indicating the interplay between the two plasmids. During our spontaneous mutagenesis studies, we found that the pag gene on pXO1 might affect the expression of the capsule genes on pXO2. To determine the role of pag in capsule production, we deleted the pag gene promoter region in the pXO1 plasmid (Figure 1A). Compared to the Pasteur II control (Figure 1B), the pag promoter knockout mutant, PasteurII-pagpromoterKO (generated with PApr-175SacF and PA-RSacII), produced dramatically less capsule (Figure 1C), which is similar to the capsule negative control Sterne strain (Figure 1G). To verify if the capsule deficiency was caused by the deletion of the pag promoter, we subcloned fragments of the pag gene and promoter into the pag promoter knockout strain for functional complementation assays (Figure 1A). Our results show that only the strains complemented with fragments encoding the entire pag gene restored capsule synthesis (Figure 1D), suggesting that pag is involved in capsule formation.
To confirm the role of pag gene in capsule formation, we deleted the entire pag gene. Similar to the capsule negative control Sterne strain (Figure 1G), the pag-deletion strain displayed capsule deficiency (Figure 1E). The morphology of PasteurII-pagKO deletion strain showed hair-like, dry, smooth, and non-sticky colonies, which was significantly different from the morphology of the Pasteur II control (data not shown). To further confirm the effect of pag on capsule formation, we conducted the complementation experiment by introducing pFF40 carrying the pag gene into the pag deletion strain. The complementary strain completely recovered the capsule formation, with thicker capsule morphology and increased capsule substance as compared to the Pasteur II strain (Figure 1F).
The Deletion of pag Promoter or pag Entire Gene Abolishes the PA Production
To elucidate whether the capsule defect phenotype of pag null knockout mutants is a result of the lack of PA production, we performed Western blot assays. The results showed that the expression of pag was not detected in either the pag promoter or the entire pag gene null mutants (lane 4 and lane 6 in Figure 2A), whereas the pag null mutation did not affect LF production (lane 4 and lane 6 in Figure 2B). This indicates the specific effect of the pag promoter or pag null mutation on PA production.
Figure 2. Western blot detection of PA and LF expressed in variant strains. PA was detected by hybridization with anti-PA antibody (A). Lane 1, protein molecular size marker; Lane 2, control PA (100 ng); Lane 3, Pasteur II; Lane 4, PasteurII-pag promoter-KO strain; Lane 5, PasteurII-pag promoter-KO-CO strain; Lane 6, PasteurII-pagKO strain; Lane 7, PasteurII-pagKO-CO strain. LF was detected by hybridization with anti-LF antibody (B). Lane 1, protein molecular size marker; Lane 2, control LF (100 ng); Lane 3, Pasteur II; Lane 4, PasteurII-pag promoter-KO strain; Lane 5, PasteurII-pag promoter-KO-CO strain; Lane 6, PasteurII-pagKO strain; Lane 7, PasteurII-pagKO-CO strain.
Taken together, the above results demonstrated that the PA protein is involved in capsule formation.
The Deletion of pag Alters the Transcription of Virulence Genes Located on Both pXO1 and pXO2 Plasmid
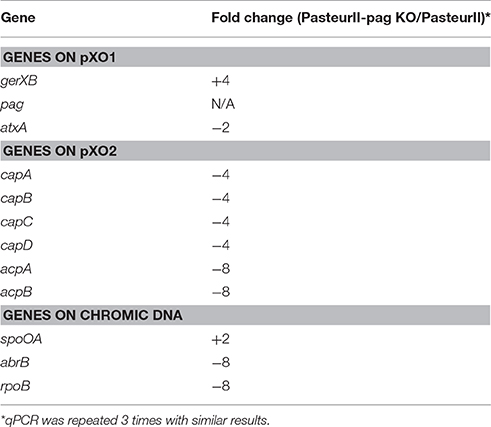
To elucidate the potential mechanism of PA's involvement in capsule formation, we examined the impact of the pag deletion on the transcription of selected virulence genes located on pXO1 and pXO2. The total RNA was extracted from PasteurII-pag KO and the original Pasteur II control, and was analyzed by qPCR. In the pag deletion strain, no pag expression was detected, as had been expected (Table 2). However, the expression of virulence genes gerXB and atxA, which are located in plasmid pXO1, showed a 2-fold increase and 2-fold decrease, respectively, in PasteurII-pag KO compared with that in Pasteur II control. In addition, the expression levels of genes responsible for capsule synthesis, including capA, capB, capC, and capD, decreased 4-fold in the PasteurII-pag KO strain compared with those in Pasteur II control, whereas the expression level of both acpA and acpB decreased 8-fold (Table 2). Surprisingly, the pag deletion also altered the transcription of the spoA, abrB, and rpoB genes located in chromosomal DNA (Table 2).
The Introduction of Constitutive acpA or acpB Expression System Eliminates the Effect of Deleted pag on Capsule Formation
Our qPCR results suggested that PA is probably involved in the capsule formation by mediating the transcription of acpA and acpB. To test it, we constructed a constitutional acpA or acpB expression strain in the pag promoter and pag gene knockout mutant, respectively. Compared to the Pasteur II control (Figure 3) and the pag null mutants (Figures 1C,E), the ability of capsule formation was fully restored in the pag promoter, the entire pag gene knockout mutant carrying on the constitutive acpA expression system (Figures 3B,C), or carrying on the constitutive acpB expression system (Figures 3D,E). To explore the possibility of whether PA directly affects the acpA transcription by binding to the promoter region of acpA, we conducted a gel mobility shift assay using purified recombinant PA, and labeled the acpA gene promoter region. No shift of the DNA/protein complex band was observed (data not shown), indicating that the PA protein did not directly mediate the acpA transcription.
Figure 3. Complementation effect of AcpA and AcpB on capsule formation in the pag null mutants. Microscope images of India ink stained Pasteur II strain (A), acpA complementation strain in the pag promoter deletion mutant (B), and acpA complementation strain in the pag null mutant (C), acpB complementation strain in the pag promoter deletion mutant (D), acpB complementation strain in the pag null mutant (E), and negative control Sterne strain without capsule formation (F). The images are representative of 3 independent experiments.
The Deletion of pag Attenuates the Virulence of the B. anthracis Pasteur II Strain
The role of PA in pathogenicity is controversial in different mouse models of infection (Welkos et al., 1993; Heninger et al., 2006; Moayeri et al., 2015). In this study, we examined the effect of pag on the pathogenicity of B. anthracis Pasteur II strain in the BALB/c mice model of subcutaneous infection. All BALB/c mice infected with PasteurII-pagKO survived for at least 96 h, whereas 50% (3 of 6) of the mice infected with the Pasteur II control strain died by 72 h, and 100% of them died by 96 h after infection. The mortality of mice did not increase when infected with more than 2-fold higher amounts of the pag knockout mutant than wild type control in subsequent experiments. The results indicated that the pag gene is an important virulence factor for B. anthracis Pasteur II strain in subcutaneous infection (Figure 4).
Figure 4. Effect of pag on pathogenicity of B. anthracis in BALB/c mouse model of infection. Six mice each were infected with either Pasteur II (dashed line), or PasteurII-pagKO strain (solid line), Percent of surviving mice shown as a function of time in hours post-infections. The difference of animal survival was statistically analyzed using Fisher exact test. P < 0.01.
Discussion
B. anthracis virulence is plasmid-determined, with the anthrax toxin genes on pXO1 and the capsule genes on pXO2. Both plasmids are required for virulence; strains without pXO1 are avirulent as they do not produce anthrax toxins, whereas strains lacking pXO2, such as the Sterne vaccine strain, are greatly attenuated since they do not form capsules (Koehler et al., 1994; Hoffmaster and Koehler, 1999). Different regulators of B. anthracis control the toxin production and capsule formation. acpA regulator controls the capABCD operon, which is located on pXO2 and is responsible for capsule biosynthesis. In this study, we demonstrated for the first time that the protective antigen protein is also involved in mediating the capsule formation through interaction with acpA and acpB in the B. anthracis Pasteur II strain.
Our results showed that interrupting PA production by either deleting the pag promoter or knocking out the pag gene eliminated the formation of capsules in the B. anthracis Pasteur II strain. Moreover, the expression of pag in trans complemented the capacity of capsule formation. These indicate that the PA plays a role in capsule biosynthesis. However, other investigators have shown that the non-toxigenic (pXO1−, pXO2+) delta Ames strain produces capsule (Welkos et al., 1993; Pomerantsev et al., 2006). This inconsistency is likely due to the different genetic backgrounds between the Pasteur II and Ames strain. Another explanation is that the non-toxigenic Ames strain may still possess the pXO1 plasmid, and as we have demonstrated, there is an existence of the pXO1 plasmid in the Pasteur II strain, and high temperature treatment could not completely eliminate the pXO1 plasmid in B. anthracis (Liang et al., 2016).
The impact of the pag deletion on the transcription of virulence genes on both pXO1 and pXO2 suggests that PA probably indirectly modulates the transcription of the capABCD operon and its regulators acpA and acpB. Although deletion of pag only generated a 4-fold difference of cap transcription levels compared to the control, this suggests a potential accumulative effect of PA protein on capsule formation, as the qPCR analysis provides only a snapshot. Our data showed that the introduction of the episomal acpA or acpB gene restored capsule formation of the pag knockout strain, indicating the critical role of AcpA/AcpB in capsule formation. Drysdale et al. reported that an acpA null mutation slightly affected the cap transcription, whereas an acpA and acpB double mutation totally eliminated cap gene expression (Drysdale et al., 2004). One possible explanation is that the AcpA/AcpB-dependent modulation of capsule formation by PA protein is strain dependent. However, DNA-protein binding analysis revealed that PA could not bind the promoter region of acpA. Taken together, this data demonstrates that PA is involved in regulating capsule formation through interaction with the capsule regulators acpA and acpB gene of Pasteur II strain by an unknown mechanism.
PA can bind and then facilitate EF and LF entry into cells to exert their toxic effects without itself showing any toxic effect, and is an effective protective antigen and strong immunogen. EF is an adenylate cyclase that increases cyclic AMP (cAMP) levels in host cells. The increased cAMP activates PKA and cAMP-activated exchange protein (Epac) signaling, which inhibits the movement and phagocytosis of macrophages (Yeager et al., 2009). LF is the principal virulence factor of anthrax toxin. A high dose of LF can cause macrophage schizolysis, while a small dose of LF can induce macrophage apoptosis by altering cell membrane permeability, depleting the mitochondrial electric potential, or fragmenting DNA. The pXO1 plasmid plays a key role in the regulation of protein function in B. anthracis (Popov et al., 2002; Park et al., 2007). Consistent with previous studies, our results also showed that the pag knockout strain lacked the expression of PA protein and attenuated the virulence of the B. anthracis Pasteur II strain. However, other groups have reported that the pag null mutation had no effect on LD50 values of B. anthracis Ames strain (Welkos et al., 1993; Heninger et al., 2006). These contradictions may result from the different genetic background between the Pasteur II strain and other B. anthracis strains used by other research groups. It is possible that the mouse strain and route of infection also affects the susceptibility to B. anthracis, as we used subcutaneous inoculation of bacteria in the inner thigh of BALB/C mice, whereas Welkos infected CBA/J and A/J mice via subcutaneous or intraperitoneal inoculation (Welkos et al., 1993), and Heninger utilized BALB/C mice through intratracheal infections (Heninger et al., 2006).
Our results indicate that PA indirectly controls the transcription and synthesis of capsule genes by affecting the expression of the capsule-regulating genes, acpA and acpB. The regulated capsule genes are located on pXO2, while PA encoding gene pag is located on pXO1 in B. anthracis. However, we found that the acpA gene on pXO2 had no influence on the expression of the toxin genes (data not shown), which is consistent with previous reports (Bourgogne et al., 2003; Drysdale et al., 2005). AtxA has been shown to regulate capsule through acpA and acpB (Uchida et al., 1997), and pag affected the atxA transcription (Table 2). Thus, it is highly possible that pag affects capsule through atxA. In addition, we found that the deletion of pag also decreased the transcription of and the chromosomal genes, including spoA, abrB, and rpoB. This suggests that other factors may act as intermediate regulators in the interaction with pag and influence capsule synthesis. Nevertheless, our data suggests that the pag gene is an important factor in the expression of B. anthracis virulence factors, including toxins and capsule formation.
In conclusion, to our knowledge, this study is the first to demonstrate that the pag gene is critical to capsule production in the B. anthracis Pasteur II strain. The pXO1 plays a role in the regulation of capsule formation, and is thus a determinant of B. anthracis pathogenicity. The results of our study provide new insights into the mechanisms underlying attenuated virulence in the B. anthracis Pasteur II vaccine strain. These findings will potentially guide the development of more efficient vaccines to prevent anthrax.
Author Contributions
Conceived and designed the experiments: XL. Performed the experiments: XL, ZZ, FZ, HZ. Analyzed the data: XL, JZ, JW, YJ, YDJ. Wrote the paper: XL, YDJ.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
We thank Dr. Shulin Dong and Dr. Chun Wang for suggestions, technical support, and critical reading of the manuscript. This work was partially supported by a grant (2011SKLID210) from the State Key Laboratory for Infectious Disease Prevention and Control.
Supplementary Material
The Supplementary Material for this article can be found online at: http://journal.frontiersin.org/article/10.3389/fcimb.2017.00203/full#supplementary-material
Supplementary Figure 1. The deletion of pag gene promoter as indicated by PCR. Lane 1: DNA molecular marker. Lane 2: PasteurII, and Lane 3: PasteurII- pag promoter-KO.
Supplementary Figure 2. The deletion of pag gene as indicated by PCR. Lane 1: DNA molecular marker. Lane 2: PasteurII, and Lane 3: PasteurII- pag KO.
References
Abrami, L., Liu, S., Cosson, P., Leppla, S. H., and van der Goot, F. G. (2003). Anthrax toxin triggers endocytosis of its receptor via a lipid raft-mediated clathrin-dependent process. J. Cell Biol. 160, 321–328. doi: 10.1083/jcb.200211018
Bartkus, J. M., and Leppla, S. H. (1989). Transcriptional regulation of the protective antigen gene of Bacillus anthracis. Infect. Immun. 57, 2295–2300.
Bourgogne, A., Drysdale, M., Hilsenbeck, S. G., Peterson, S. N., and Koehler, T. M. (2003). Global effects of virulence gene regulators in a Bacillus anthracis strain with both virulence plasmids. Infect. Immun. 71, 2736–2743. doi: 10.1128/IAI.71.5.2736-2743.2003
Drysdale, M., Bourgogne, A., Hilsenbeck, S. G., and Koehler, T. M. (2004). atxA controls Bacillus anthracis capsule synthesis via acpA and a newly discovered regulator, acpB. J. Bacteriol. 186, 307–315. doi: 10.1128/JB.186.2.307-315.2004
Drysdale, M., Heninger, S., Hutt, J., Chen, Y., Lyons, C. R., and Koehler, T. M. (2005). Capsule synthesis by Bacillus anthracis is required for dissemination in murine inhalation anthrax. EMBO J. 24, 221–227. doi: 10.1038/sj.emboj.7600495
Erwin, J. L., DaSilva, L. M., Bavari, S., Little, S. F., Friedlander, A. M., and Chanh, T. C. (2001). Macrophage-derived cell lines do not express proinflammatory cytokines after exposure to Bacillus anthracis lethal toxin. Infect. Immun. 69, 1175–1177. doi: 10.1128/IAI.69.2.1175-1177.2001
Ezzell, J. W., and Welkos, S. L. (1999). The capsule of bacillus anthracis, a review. J. Appl. Microbiol. 87:250. doi: 10.1046/j.1365-2672.1999.00881.x
Fish, D. C., Mahlandt, B. G., Dobbs, J. P., and Lincoln, R. E. (1968). Purification and properties of in vitro-produced anthrax toxin components. J. Bacteriol. 95, 907–918.
Gordon, V. M., Klimpel, K. R., Arora, N., Henderson, M. A., and Leppla, S. H. (1995). Proteolytic activation of bacterial toxins by eukaryotic cells is performed by furin and by additional cellular proteases. Infect. Immun. 63, 82–87.
Guignot, J., Mock, M., and Fouet, A. (1997). AtxA activates the transcription of genes harbored by both Bacillus anthracis virulence plasmids. FEMS Microbiol. Lett. 147, 203–207.
Heninger, S., Drysdale, M., Lovchik, J., Hutt, J., Lipscomb, M. F., Koehler, T. M., et al. (2006). Toxin-deficient mutants of Bacillus anthracis are lethal in a murine model for pulmonary anthrax. Infect. Immun. 74, 6067–6074. doi: 10.1128/IAI.00719-06
Hoffmaster, A. R., and Koehler, T. M. (1997). The anthrax toxin activator gene atxA is associated with CO2-enhanced non-toxin gene expression in Bacillus anthracis. Infect. Immun. 65, 3091–3099.
Hoffmaster, A. R., and Koehler, T. M. (1999). Control of virulence gene expression in Bacillus anthracis. J. Appl. Microbiol. 87, 279–281. doi: 10.1046/j.1365-2672.1999.00887.x
Koehler, T. M., Dai, Z., and Kaufman-Yarbray, M. (1994). Regulation of the Bacillus anthracis protective antigen gene: CO2 and a trans-acting element activate transcription from one of two promoters. J. Bacteriol. 176, 586–595. doi: 10.1128/jb.176.3.586-595.1994
Leppla, S. H. (1988). Production and purification of anthrax toxin. Methods Enzymol. 165, 103–116. doi: 10.1016/S0076-6879(88)65019-1
Liang, X., Zhang, H., Zhang, E., Wei, J., Li, W., Wang, B., et al. (2016). Identification of the pXO1 plasmid in attenuated Bacillus anthracis vaccine strains. Virulence. 7, 578–586. doi: 10.1080/21505594.2016.1164366
Makino, S., Uchida, I., Terakado, N., Sasakawa, C., and Yoshikawa, M. (1989). Molecular characterization and protein analysis of the cap region, which is essential for encapsulation in Bacillus anthracis. J. Bacteriol. 171, 722–730. doi: 10.1128/jb.171.2.722-730.1989
Mikesell, P., Ivins, B. E., Ristroph, J. D., and Dreier, T. M. (1983). Evidence for plasmid-mediated toxin production in Bacillus anthracis. Infect. Immun. 39, 371–376.
Milne, J. C., Furlong, D., Hanna, P. C., Wall, J. S., and Collier, R. J. (1994). Anthrax protective antigen forms oligomers during intoxication of mammalian cells. J. Biol. Chem. 269, 20607–20612.
Moayeri, M., Leppla, S. H., Vrentas, C., Pomerantsev, A. P., and Liu, S. (2015). Anthrax pathogenesis. Annu. Rev. Microbiol. 69, 185–208. doi: 10.1146/annurev-micro-091014-104523
Okinaka, R. T., Cloud, K., Hampton, O., Hoffmaster, A. R., Hill, K. K., Keim, P., et al. (1999). Sequence and organization of pXO1, the large Bacillus anthracis plasmid harboring the anthrax toxin genes. J. Bacteriol. 181, 6509–6515.
Park, S. H., Oh, H. B., Seong, W. K., Kim, C. W., Cho, S. Y., and Yoo, C. K. (2007). Differential analysis of Bacillus anthracis after pX01 plasmid curing and comprehensive data on Bacillus anthracis infection in macrophages and glial cells. Proteomics 7, 3743–3758. doi: 10.1002/pmic.200700338
Pomerantsev, A. P., Sitaraman, R., Galloway, C. R., Kivovich, V., and Leppla, S. H. (2006). Genome engineering in Bacillus anthracis using Cre recombinase. Infect. Immun. 74, 682–693. doi: 10.1128/IAI.74.1.682-693.2006
Popov, S. G., Villasmil, R., Bernardi, J., Grene, E., Cardwell, J., Wu, A., et al. (2002). Lethal toxin of Bacillus anthracis causes apoptosis of macrophages. Biochem. Biophys. Res. Commun. 293, 349–355. doi: 10.1016/S0006-291X(02)00227-9
Remmele, N. S., Klein, F., Vick, J. A., Walker, J. S., Mahlandt, B. G., and Lincoln, R. E. (1968). Anthrax toxin: primary site of action. J. Infect. Dis. 118, 104–113. doi: 10.1093/infdis/118.1.104
Singh, Y., Chaudhary, V. K., and Leppla, S. H. (1989). A deleted variant of Bacillus anthracis protective antigen is non-toxic and blocks anthrax toxin action in vivo. J. Biol. Chem. 264, 19103–19107.
Singh, Y., Klimpel, K. R., Goel, S., Swain, P. K., and Leppla, S. H. (1999). Oligomerization of anthrax toxin protective antigen and binding of lethal factor during endocytic uptake into mammalian cells. Infect. Immun. 67, 1853–1859.
Sirard, J. C., Mock, M., and Fouet, A. (1994). The three Bacillus anthracis toxin genes are coordinately regulated by bicarbonate and temperature. J. Bacteriol. 176, 5188–5192. doi: 10.1128/jb.176.16.5188-5192.1994
Smith, H., and Keppie, J. (1954). Observations on experimental anthrax; demonstration of a specific lethal factor produced in vivo by Bacillus anthracis. Nature 173, 869–870. doi: 10.1038/173869a0
Stanley, J. L., and Smith, H. (1961). Purification of factor I and recognition of a third factor of the anthrax toxin. J. Gen. Microbiol. 26, 49–63. doi: 10.1099/00221287-26-1-49
Uchida, I., Hashimoto, K., and Terakado, N. (1986). Virulence and immunogenicity in experimental animals of Bacillus anthracis strains harbouring or lacking 110 MDa and 60 MDa plasmids. J. Gen. Microbiol. 132, 557–559. doi: 10.1099/00221287-132-2-557
Uchida, I., Hornung, J. M., Thorne, C. B., Klimpel, K. R., and Leppla, S. H. (1993). Cloning and characterization of a gene whose product is a trans-activator of anthrax toxin synthesis. J. Bacteriol. 175, 5329–5338. doi: 10.1128/jb.175.17.5329-5338.1993
Uchida, I., Makino, S., Sekizaki, T., and Terakado, N. (1997). Cross-talk to the genes for Bacillus anthracis capsule synthesis by atxA, the gene encoding the trans-activator of anthrax toxin synthesis. Mol. Microbiol. 23, 1229–1240. doi: 10.1046/j.1365-2958.1997.3041667.x
Welkos, S. L., Vietri, N. J., and Gibbs, P. H. (1993). Non-toxigenic derivatives of the Ames strain of Bacillus anthracis are fully virulent for mice: role of plasmid pX02 and chromosome in strain-dependent virulence. Microb. Pathog. 14, 381–388. doi: 10.1006/mpat.1993.1037
Yang, J., Liang, X., and Ji, Y. (2015). The novel transcriptional regulator SA1804 Is involved in mediating the invasion and cytotoxicity of Staphylococcus aureus. Front. Microbiol. 6:174. doi: 10.3389/fmicb.2015.00174
Keywords: Bacillus anthracis, pXO1 and pXO2, capsule biosynthesis, pag gene, acpA and acpB
Citation: Liang X, Zhu J, Zhao Z, Zheng F, Zhang H, Wei J, Ji Y and Ji Y (2017) The pag Gene of pXO1 Is Involved in Capsule Biosynthesis of Bacillus anthracis Pasteur II Strain. Front. Cell. Infect. Microbiol. 7:203. doi: 10.3389/fcimb.2017.00203
Received: 18 March 2017; Accepted: 08 May 2017;
Published: 26 May 2017.
Edited by:
Dongsheng Zhou, Beijing Institute of Microbiology and Epidemiology, ChinaReviewed by:
Lee-Ann H. Allen, University of Iowa, United StatesChia Y. Lee, University of Arkansas for Medical Sciences, United States
Copyright © 2017 Liang, Zhu, Zhao, Zheng, Zhang, Wei, Ji and Ji. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Xudong Liang, bGlhbmd4dWRvbmdAaWNkYy5jbg==
Yinduo Ji, aml4eHgwMDJAdW1uLmVkdQ==
†These authors have contributed equally to this work.
 Xudong Liang1*†
Xudong Liang1*† Yinduo Ji
Yinduo Ji