- 1Department of Clinical Laboratory, Peking University People's Hospital, Beijing, China
- 2Department of Clinical Laboratory, First Affiliated Hospital of Sun Yat-sen University, Guangzhou, China
Although coexistence of blaNDM-5 and mcr-1 in Escherichia coli has been reported, little is known about the fitness and virulence of such strains. Three carbapenem-resistant Escherichia coli (GZ1, GZ2, and GZ3) successively isolated from one patient in 2015 were investigated for microbiological fitness and virulence. GZ1 and GZ2 were also resistant to colistin. To verify the association between plasmids and fitness, growth kinetics of the transconjugants were performed. We also analyzed genomic sequences of GZ2 and GZ3 using PacBio sequencing. GZ1 and GZ2 (ST10) co-harbored blaNDM-5 and mcr-1, while GZ3 (ST4981) carried only blaNDM-5. GZ3 demonstrated significantly more rapid growth (P < 0.001) and overgrew GZ2 with a competitive index of 1.0157 (4 h) and 2.5207 (24 h). Increased resistance to serum killing and mice mortality was also identified in GZ3. While GZ2 had four plasmids (IncI2, IncX3, IncHI2, IncFII), GZ3 possessed one plasmid (IncFII). The genetic contexts of blaNDM-5 in GZ2 and GZ3 were identical but inserted into different backbones, IncX3 (102,512 bp) and IncFII (91,451 bp), respectively. The growth was not statistically different between the transconjugants with mcr-1 or blaNDM-5 plasmid and recipient (P = 0.6238). Whole genome sequence analysis revealed that 28 virulence genes were specific to GZ3, potentially contributing to increased virulence of GZ3. Decreased fitness and virulence in a mcr-1 and blaNDM-5 co-harboring ST10 E. coli was found alongside a ST4981 strain with only blaNDM-5. Acquisition of mcr-1 or blaNDM-5 plasmid did not lead to considerable fitness costs, indicating the potential for dissemination of mcr-1 and blaNDM-5 in Enterobacteriaceae.
Introduction
Multidrug-resistant Gram-negative bacteria continue to threaten several aspects of public health due to the availability of limited antimicrobials for treatment, especially for the carbapenem-resistant isolates. Colistin has become the last resort to treat serious infections caused by multidrug-resistant organisms. However, colistin resistance has also been reported in carbapenem-resistant bacteria. Since 2015, plasmid-mediated colistin resistance gene, mcr-1, has been reported and identified with increasing incidence in Enterobacteriaceae worldwide, isolated from environment, human and animals (Liu, Y. Y. et al., 2016; Nordmann and Poirel, 2016). The co-existence of mcr-1 and carbapenemase genes, including blaNDM and blaKPC, has already been detected in clinical settings and resulted in a few therapeutic options (Li et al., 2016; Teo et al., 2016). The rapid transmission of mcr-1 and carbapenemase genes located on mobile genetic elements is of great concern. Successful dissemination of resistance plasmids largely depends on the fitness cost imposed on hosts (Andersson and Hughes, 2010). Therefore, understanding the biological cost of resistance is critical for controlling the dissemination of multi-drug resistant strains.
So far, studies on the biological effects of colistin resistance have mainly focused on Acinetobacter baumannii (Hraiech et al., 2013; Beceiro et al., 2014; Wand et al., 2015) with extremely limited data on Enterobacteriaceae. The impact of mcr-1 gene on in vitro and in vivo fitness is yet to be elucidated. Furthermore, there is a lack of studies concerning the effects of coexistence of carbapenemase and mcr-1 on fitness and virulence in Enterobacteriaceae.
Here, we identified three Escherichia coli isolates carrying blaNDM-5 from a single patient with different sequence type and two of them also harbored mcr-1. The purpose of this study was to examine the in vitro and in vivo fitness, virulence and genetic backgrounds of blaNDM-5 and mcr-1 co-producing Enterobacteriaceae and verify the association between resistance plasmids and the fitness.
Materials and Methods
Bacterial Isolation and Clinical Information
Medical records and the patient information were retrospectively reviewed and collected. A 19-year-old female was admitted into a hospital in Guangzhou in April 2015. The patient had a history of pelvic fracture, urethral disruption and pelvic organ injury treated with colostomy and cystostomy. Surgery was performed during her hospitalization. She subsequently presented with fever, fistula of colon and ureter, complex intra-abdominal infections, systemic inflammatory response syndrome and urinary tract infections. From May to July, three carbapenem-resistant E. coli (GZ1, GZ2, and GZ3) were successively isolated from abdominal drainage and urine obtained from cystostomy. While she received various antimicrobials, including cefuroxime, gentamycin, metronidazole, ceftazidime, amikacin, meropenem, imipenem, levofloxacin, cefixime, vancomycin and piperacillin–tazobactam before GZ2 was isolated, due to clinical improvement, the treatment was changed to levofloxacin, cefixime and metronidazole after the isolation. The study was approved by the research ethics board at Peking University People's Hospital. Informed consent was not needed as this study was retrospective and participants were anonymized.
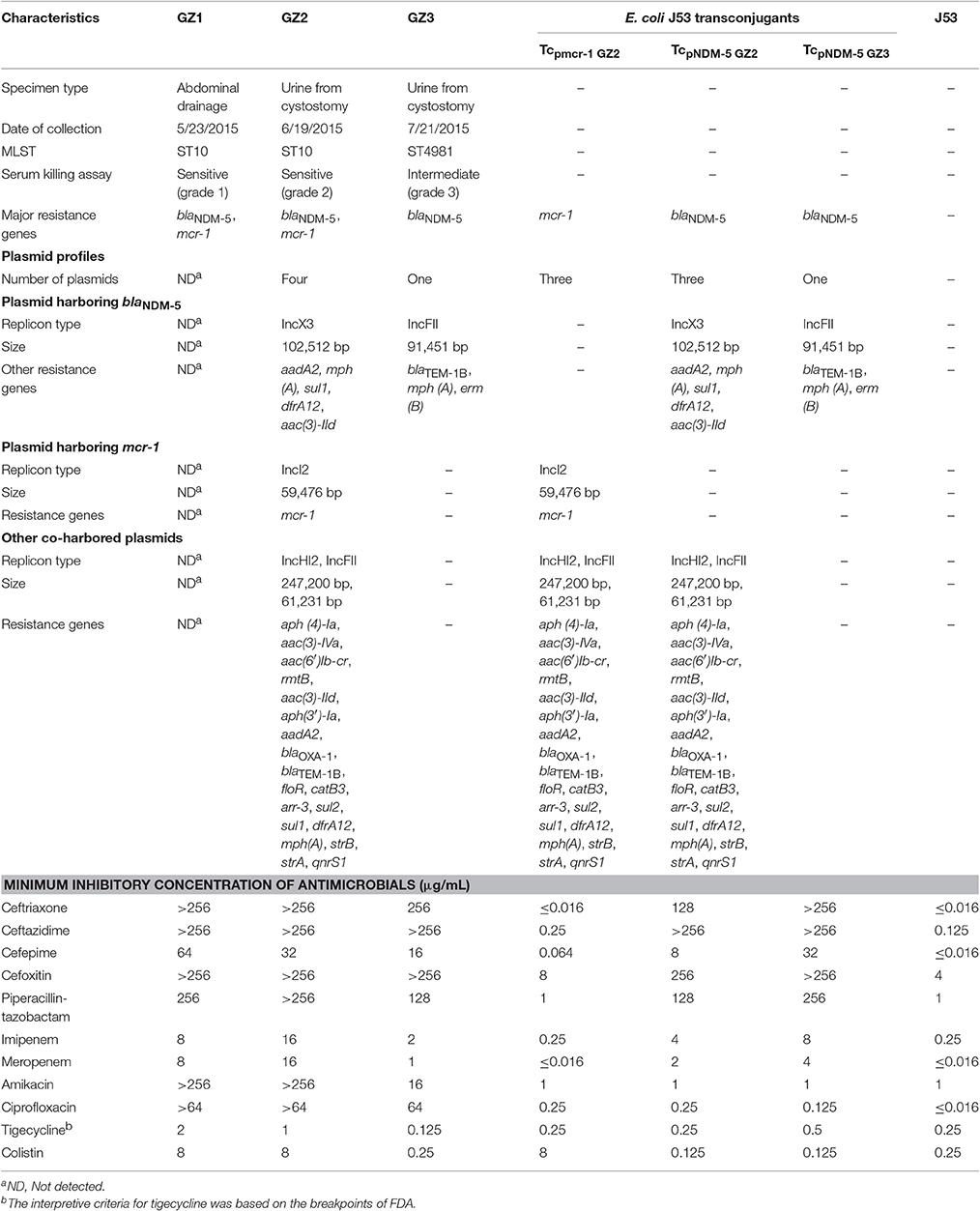
Antimicrobial Susceptibility Testing, Detection of Resistance Genes and Multilocus Sequence Typing
The minimum inhibitory concentrations (MICs) of the following antimicrobials, including ceftriaxone (Roche, Shanghai, China), ceftazidime, cefoxitin, cefepime, amikacin (National Institutes for Food and Drug Control, Beijing, China), piperacillin–tazobactam, tigecycline (Pfizer, NY, USA), imipenem (MSD, Hangzhou, China), meropenem (Sumitomo Pharmaceuticals, Suzhou, China), ciprofloxacin (Bayer, Leverkusen, Germany) and colistin (Amresco, Solon, USA) were determined by agar dilution method, according to Clinical and Laboratory Standards Institute (CLSI) guidelines (Clinical and Laboratory Standards Institute, 2015). The results were interpreted according to CLSI breakpoints (Clinical and Laboratory Standards Institute, 2016). Polymerase chain reaction (PCR) was used to detect the presence of mcr-1, carbapenemase genes and other resistance genes as previously described (Wang et al., 2014; Liu, Y. Y. et al., 2016). Multilocus sequence typing (MLST) was performed as described on the E. coli MLST website (http://mlst.warwick.ac.uk/mlst/dbs/Ecoli).
Growth Assay and In vitro Competition Experiment
Fitness was investigated by using growth curve assay and in vitro competition experiments (Liu, D. et al., 2016). Three isolates (GZ1, GZ2, and GZ3) cultured overnight in LB broth were diluted to an OD600 of 0.01 and grew at 37°C with vigorous aeration (200 rpm). The culture cell density was determined every 0.5 h by measuring the OD600.
In the in vitro competition assays, GZ2 and GZ3 were separately cultured overnight in LB broth at 37°C. The bacteria were diluted and equivalent numbers of GZ2 and GZ3 were pooled and cultured together at 37°C. At 0, 4, and 24 h, aliquots of the mixed bacteria were diluted with 0.9% saline solution and plated on Mueller–Hinton agar (BD, Sparks MD, USA) plates with or without colistin (4 mg/L). Colony-forming unit (CFU) were counted after 0, 4, and 24 h of incubation at 37°C. Each isolate was tested three times. The competitive index (CI) was determined as follows: CI = (GZ3/GZ2)/(Inoculated GZ3/Inoculated GZ2) as previously described (Liu, D. et al., 2016).
Serum Killing Assay
Serum killing assay was conducted to determine the virulence in vitro as previously described (Abate et al., 2012). An inoculum of 25 μL prepared from the mid-log phase was diluted by 0.9% saline solution and was added to 75 μL of pooled human sera contained in a 10 × 75 mm Falcon polypropylene tube (BD Biosciences, Franklin Lakes, NJ, USA). Viable counts were checked at 0, 1, 2, and 3 h of incubation at 37°C. The mean results were expressed as percentage of inoculation and a strain was classified as serum sensitive, intermediately sensitive and resistant.
Mouse Lethality Assay
To determine in vivo virulence, six pathogen-free, 6–8-week-old, male BALB/c mice were used as a sample population for each bacterial concentration. Ten-fold serial dilution of CFU of E. coli was made from a starting concentration of 109 CFU/mL to 105 CFU/mL, and BALB/c mice were infected intraperitoneally with 0.1 mL of each concentration. Symptoms and mortality rates were observed for 14 days. Inoculation dose was confirmed on LB agar and survival curves were assessed by Kaplan–Meier analysis. The study was approved by the research ethics board at Peking University People's Hospital.
Construction of the E. coli J53 Transconjugants Carrying mcr-1 and blaNDM-5 Plasmids
To exclude the impact on fitness caused by different sequence type, we constructed the same strains carrying different plasmids. Conjugation experiment was performed using GZ2 and GZ3 as the donors and E. coli J53 as the recipient according to previous studies (Wang et al., 2014). The donor and the recipient were mixed at a ratio of 1:1 for 24 h. Transconjugants were selected on Chinese Blue lactose agars (OXOID, Basingstoke Hampshire, UK), supplemented with sodium azide (100 mg/L) and imipenem (1 mg/L), sodium azide (100 mg/L) and colistin (4 mg/L) or sodium azide (100 mg/L), colistin (4 mg/L), and imipenem (1 mg/L), respectively. PCR was used to screen for mcr-1 and blaNDM-5 as previously described (Wang et al., 2014; Liu, Y. Y. et al., 2016).
Fitness Measurements of the E. coli J53 Transconjugants
The plasmid profiles of the transconjugants were identified by PCR based on replicons of the major plasmid incompatibility groups as previously described (Carattoli et al., 2005). To verify the association between the resistance plasmids and the fitness, growth kinetics of the transconjugants and the recipient E. coli J53 were investigated (Liu, D. et al., 2016). The strains cultured overnight in LB broth were diluted to an OD600 of 0.01 and grew at 37°C with vigorous aeration (200 rpm). The culture cell density was determined every 0.5 h by measuring the OD600 for 24 h.
Whole Genome Sequencing
Whole genome sequencing (WGS) of GZ2 and GZ3 was conducted using a PacBio RS II system (Pacific Biosciences, Menlo Park, USA) with a 10 kb size library and P6/C4 chemistry. De novo assembly was performed with SMRTanalysis version 2.3 (Pacific Biosciences). The plasmid sequence was annotated using the online Rapid Annotation Subsequencing Technology (RAST) (http://rast.nmpdr.org/) automatically and BLASTn analysis. Antibiotic resistance genes and plasmid incompatibility groups were analyzed through the website of the Center for Genomic Epidemiology (http://www.genomicepidemiology.org/). Virulence genes were identified using the virulence factor database (http://www.mgc.ac.cn/VFs/) and PathogenFinder (https://cge.cbs.dtu.dk/services/PathogenFinder/). The nucleotide sequences of the genome and plasmids of GZ2 and GZ3 have been submitted to GenBank with accession no. MCRE00000000, CP017980, and CP017981.
Statistical Analysis
Statistical analysis was performed with the software GraphPad Prism version 5 using one-way analysis of variance (ANOVA) followed by Tukey–Kramer tests. P < 0.05 was considered to be statistically significant.
Results
Antimicrobial Susceptibility Testing, Molecular Typing and Detection of Resistance Genes
Results pertaining to susceptibility testing showed that all isolates were resistant to most of the tested antimicrobials including carbapenems. While GZ1 and GZ2 exhibited resistance to colistin (Table 1) and co-harbored blaNDM-5 and mcr-1 belonging to ST10, GZ3 (ST4981) carried only blaNDM-5.
In vitro and In vivo Fitness and Virulence of GZ1, GZ2, and GZ3
The growth of GZ3 was significantly higher than the other two isolates (P < 0.001) (Figure 1) and in vitro competition experiment revealed that GZ2 was overgrown by GZ3 with a competitive index (CI) of 1.0157 (4 h) and 2.5207 (24 h). GZ3 demonstrated intermediate to serum killing (grade 3), whereas GZ1 and GZ2 displayed sensitive (grade 1 or 2), thus suggesting that in human sera, GZ3 exhibited higher defense against bactericidal activity than GZ1 and GZ2. A significant difference in mice lethality was identified between GZ3 and the other isolates. All mice injected with GZ3 died within the first day at a concentration of 109 CFU/mL, while mice infected with GZ1, GZ2 and the control survived over 14 days.
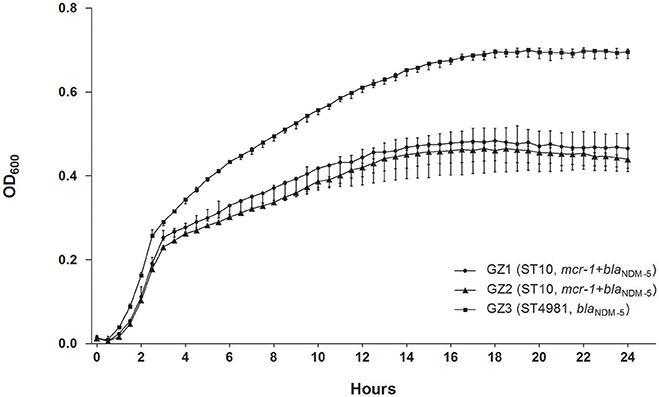
Figure 1. In vitro growth of GZ1, GZ2, and GZ3 cultured at 37°C in LB broth. The growth of GZ3 was significantly higher than the other two strains (P < 0.001). Data points represent the mean (±standard deviations) of three independent experiments.
Conjugation Experiment and Biological Costs of Plasmid Carriage
The characteristics of the transconjugants (designated as Tcpmcr-1GZ2, TcpNDM-5GZ2, TcpNDM-5GZ3) were summarized in Table 1. In the conjugation experiments, blaNDM-5 and mcr-1 could be transferred separately from GZ2 and GZ3 to the recipient E. coli J53. No transconjugants co-harboring mcr-1 and blaNDM-5 were detected. In addition to plasmids containing mcr-1 or blaNDM-5, the plasmid profile of the transconjugants was the same as their corresponding parent strains. According to the plasmid replicon typing results, there were three plasmids in Tcpmcr-1GZ2 (IncI2, IncHI2, IncFII), three plasmids in TcpNDM-5GZ2 (IncX3, IncHI2, IncFII) and one plasmid in TcpNDM-5GZ3 (IncFII).
No significant difference in growth was observed in Tcpmcr-1GZ2, TcpNDM-5GZ2, TcpNDM-5GZ3, and E. coli J53 (P = 0.6238) (Figure 2). The results indicated that no considerable fitness costs were identified in carriage of plasmid containing mcr-1 or blaNDM-5 compared to the recipient strain, suggesting that resistance was not associated with the decreased fitness of GZ2. Chromosomal characteristics, such as different sequence type (ST10 and ST4981) were considered to be the potential explanations.
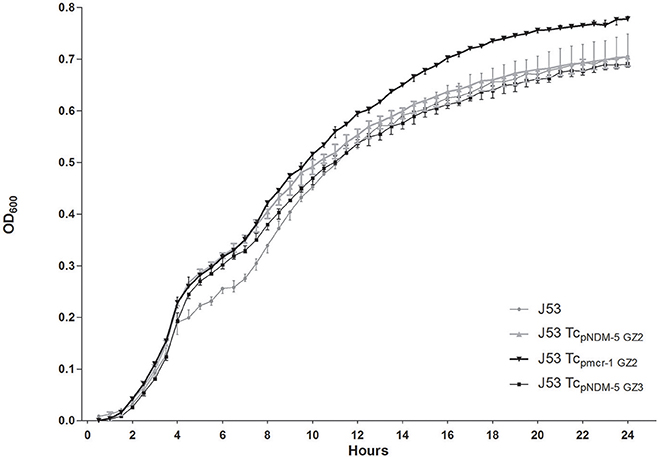
Figure 2. Growth kinetics of E. coli J53 and its transconjugants (TcpNDM-5GZ2, Tcpmcr-1GZ2 and TcpNDM-5GZ3). Values represent the mean ± standard variations obtained from three independent experiments.
Analysis of Virulence Genes in GZ2 and GZ3 by WGS
WGS and virulence factor database analysis revealed that GZ2 and GZ3 had 120 and 124 virulence-associated genes, respectively. Compared with GZ2, GZ3 had a higher number of hemolysin genes and 28 specific virulence genes in the genome. These genes were associated with chorismate binding-like protein, iron acquisition, CFA/I fimbria and type III secretion protein (such as eiv, epr and epa), potentially contributing to the increased virulence in GZ3.
Sequence Analysis of Plasmid Carrying mcr-1
In this study, the plasmids harboring mcr-1 and blaNDM-5 were designated as pGZ2-mcr, pGZ2-NDM, and pGZ3-NDM. The plasmid carrying mcr-1 from GZ2 (named pGZ2-mcr) was 59,476 bp in length, belonging to IncI2 incompatibility group. The results of BLASTn analysis revealed that plasmid pGZ2-mcr with 100% query coverage displayed 99% identity to plasmid pEC13-1 (GenBank accession CP016186.1) and plasmid pBA77-MCR-1 (GenBank accession KX013539.1) which have been isolated from E. coli in Malaysia and United Arab Emirates. No other resistance genes were found in pGZ2-mcr.
Sequence Analysis of Plasmid Containing blaNDM-5
Sequence analysis showed that the two plasmids carrying blaNDM-5 were not identical and belonged to different incompatibility group (Figure 3A). While plasmid pGZ2-NDM was 102,512 bp in length and possessed an IncX3-type backbone, pGZ3-NDM (91,451 bp) belonged to IncFII group. Both the plasmids encoded replication, stability, horizontal transfer, antimicrobial resistance, and maintenance functions (Figure 3A). pGZ2-NDM and pGZ3-NDM were found to harbor additional resistance genes, including aadA2, mph (A), sul1, aac (3)-IId, dfrA12, blaTEM−1B, and erm (B) (Table 1). Sequence alignments with BLAST revealed that pGZ2-NDM shared 53% query coverage and 99% identity with plasmid pNDM-ECN49 (GenBank accession KP765744.1), isolated from an Enterobacter cloacae strain. The genetic environment of blaNDM-5 differed between these two plasmids. pGZ3-NDM exhibited more than 70% query coverage and 99% identity with plasmid pHNFP460-1 (GenBank accession KJ020575.1), whereas no blaNDM-5 gene was found in pHNFP460-1.
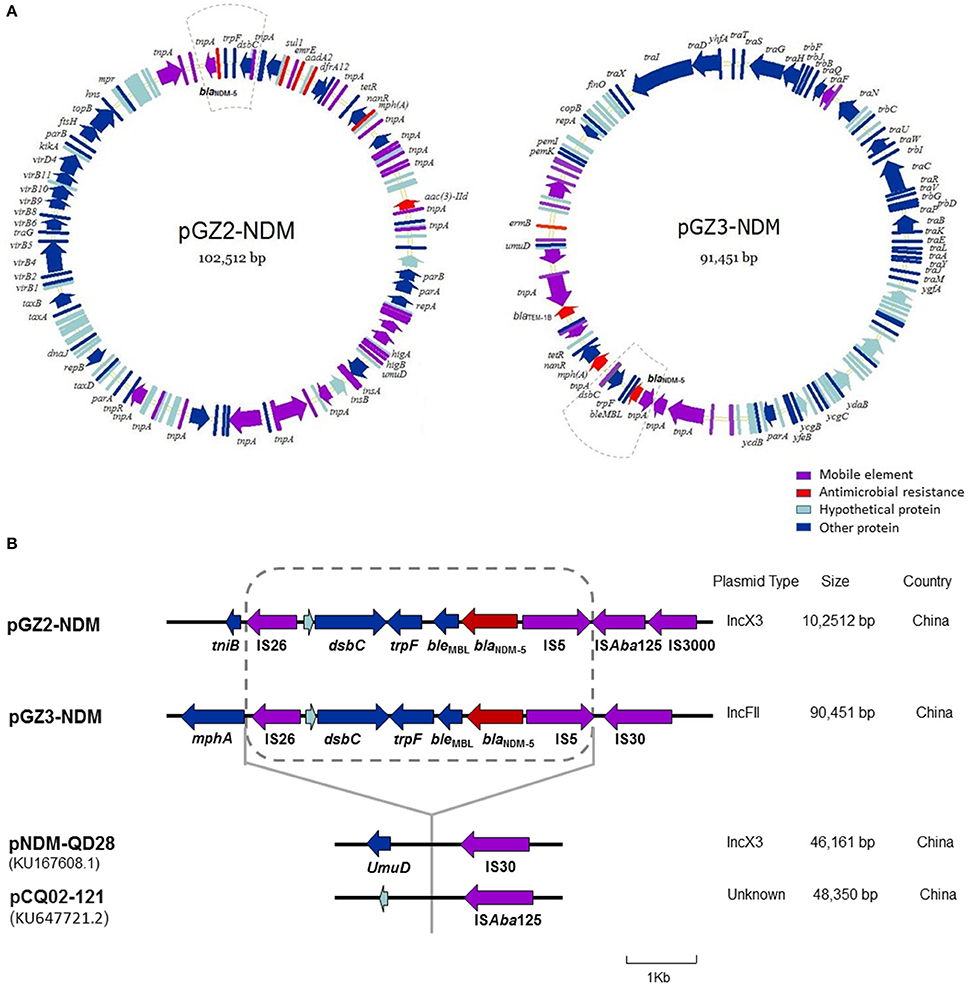
Figure 3. Plasmid analysis of pGZ2-NDM and pGZ3-NDM. Structure map of pGZ2-NDM and pGZ3-NDM (A) and comparative analysis of the genetic contexts of blaNDM-5 in plasmids reported in this study and previously described (B). The gene name is shown next to the corresponding arrow or rod. Genetic regions with dashed lines in pGZ2-NDM and pGZ3-NDM are indicated as blaNDM-5 region. The genetic environment of blaNDM-5 (with dashed line) in pGZ2-NDM and pGZ3-NDM was similar as that previously described in pNDM-QD28 (GenBank accession KU167608.1) and pCQ02-121 (GenBank accession KU647721.2) with 100% query coverage and 99% identity. However, the regions containing blaNDM-5 were inserted into different genes and plasmid backbones.
Comparative analysis of the genetic contexts of blaNDM-5 (~4,900 bp) in pGZ2-NDM and pGZ3-NDM were almost identical and were also similar as that previously reported in pNDM-QD28 (GenBank accession KU167608.1) and pCQ02-121 (GenBank accession KU647721.2) with 100% query coverage and 99% identity (Figure 3B). The regions containing blaNDM-5 were inserted into different genes. Furthermore, these plasmids belonged to different incompatibility groups. Therefore, the results indicated that similar genetic context containing blaNDM-5 have been inserted into different plasmid backbones, thus forming a “new” plasmid.
Discussion
To date, there have been several reports on the coexistence of blaNDM-5 and mcr-1 in Enterobacteriaceae worldwide, but little is known about the fitness and virulence of such strains (Mediavilla et al., 2016; Yang et al., 2016). Several findings are of microbiological and epidemiological interest: (i) blaNDM-5 and mcr-1 co-producing ST10 E. coli showed decreased fitness and virulence, compared to a ST4981 isolate harboring only blaNDM-5 isolated from the same patient (ii) acquisition of the plasmid carrying mcr-1 or blaNDM-5 was not associated with a statistically significant fitness cost to the parent strain.
Diverse sequence types (STs) of mcr-1-positive strains were identified among different regions, with some commonality in clades isolated from clinical samples, such as ST10 (Wang et al., 2017). Furthermore, ST10 has also been found in asymptomatic carriage of E. coli co-harboring blaNDM−1 and mcr-1 from a healthy individual (Zhong et al., 2016). To the best of our knowledge, this is the first report of ST10 in E. coli carrying both blaNDM-5 and mcr-1 from clinical infections. Identification of ST10 in blaNDM- and mcr-1-producing E. coli from both hospi-talized and healthy people warrants our immediate awareness.
Exotoxins, endotoxins, adherence factors, secretion systems, iron acquisition and fimbria are commonly considered to be important virulence features in gram-negative bacteria (Cosentino et al., 2013). Type III secretion systems are associated with transport of virulence proteins across bacterial and host cell membranes into the target cells (Cornelis and Van Gijsegem, 2000). Most bacteria are reported to carry only one type III secretion system, but some isolates have two such systems, which referred as type III secretion 2 (ETT2) in E. coli (Yao et al., 2009). ETT2 comprises the eiv, epr and epa genes and the deletion mutant of ETT2 exhibited defects in invasion and intracellular survival (Yao et al., 2009). In this study, ETT2 was one of the potential explanations for increased virulence in GZ3 respect GZ2 in vitro and in vivo.
Although it is commonly considered that, during the absence of antibiotic pressure, resistance usually imposes a fitness cost on the bacteria (Andersson and Hughes, 2010), several controversies have arisen regarding the association of resistance and fitness. Although a considerable reduction in fitness was observed by newly acquired plasmids and other mobile genetic elements (Starikova et al., 2013; Göttig et al., 2016), some studies showed that carbapenemase-encoding plasmids contributed to low to moderate fitness cost to host (Schaufler et al., 2016; Di Luca et al., 2017). To explore whether blaNDM-5 or mcr-1 plasmid imposed a fitness cost on the host, the growth kinetics were investigated in the transconjugants. No significant fitness cost was observed between the transconjugants and the recipient E. coli J53, indicating the potential dissemination of mcr-1 and blaNDM among Enterobacteriaceae.
For the isolates carrying mcr-1 and blaNDM-5, most studies illustrated that blaNDM-5 was mediated by IncX3 plasmids (Li et al., 2016). pGZ2-NDM belonged to IncX3 in this study, but was much longer in length and carried numerous resistance genes. One explanation was that the patient suffered long course of diseases with complex intra-abdominal infections and received various kinds of antimicrobials. Therefore, the plasmid might have evolved rapidly under great selective pressures.
In previous studies, blaNDM-5 and mcr-1-producing E. coli isolated from a duck was reported in Guangzhou (Yang et al., 2016). Although the patient in this study was not engaged in animal husbandry, she might have had the chance to be in contact with poultry, because she lived in a rural area. The possibility of transmission of strains carrying mcr-1 and blaNDM between animals and humans remains unclear. In addition, there were some limitations in this study. No transconjugants co-harboring mcr-1 and blaNDM-5 were obtained in this study. Thus, we have no access to determine the role of mcr-1 and blaNDM-5 co-producing plasmids in the decreased fitness in GZ2. Further studies on biological effects of acquisition of mcr-1 and blaNDM plasmids are needed.
In conclusion, we reported the decreased fitness and virulence in a mcr-1 and blaNDM-5 co-producing ST10 strain against a ST4981 E. coli harboring blaNDM-5 isolated from the same patient. However, no significant fitness cost was observed by carriage of the mcr-1 or blaNDM-5 plasmid, indicating the potential for dissemination of mcr-1 and blaNDM-5 in Enterobacteriaceae.
Author Contributions
HW conceived and designed the study. KL reviewed the medical records of the case and provided these three isolates. YZ, QW, XW, HL, and RW performed experiments described in this study. HG and YZ did the genomic analysis. YZ wrote the draft and HW revised it. All authors approved the final version.
Funding
This work was partly supported by National Natural Science Foundation of China [no. 81572036, no. 81661138006 and no. 81625014].
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
Abate, G., Koh, T. H., Gardner, M., and Siu, L. K. (2012). Clinical and bacteriological characteristics of Klebsiella pneumoniae causing liver abscess with less frequently observed multi-locus sequences type, ST163, from Singapore and Missouri, US. J. Microbiol. Immunol. Infect. 45, 31–36. doi: 10.1016/j.jmii.2011.09.002
Andersson, D. I., and Hughes, D. (2010). Antibiotic resistance and its cost: is it possible to reverse resistance? Nat. Rev. Microbiol. 8, 260–271. doi: 10.1038/nrmicro2319
Beceiro, A., Moreno, A., Fernández, N., Vallejo, J. A., Aranda, J., Adler, B., et al. (2014). Biological cost of different mechanisms of colistin resistance and their impact on virulence in Acinetobacter baumannii. Antimicrob. Agents Chemother. 58, 518–526. doi: 10.1128/AAC.01597-13
Carattoli, A., Bertini, A., Villa, L., Falbo, V., Hopkins, K. L., and Threlfall, E. J. (2005). Identification of plasmids by PCR-based replicon typing. J. Microbiol. Methods 63, 219–228. doi: 10.1016/j.mimet.2005.03.018
Clinical and Laboratory Standards Institute (2015). Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically, 10th Edn. Approved Standard M07-A10. Wayne, PA: CLSI.
Clinical and Laboratory Standards Institute (2016). Performance Standards For Antimicrobial Susceptibility Testing, 26th Edn. Approved Standard M100S. Wayne, PA: CLSI.
Cornelis, G. R., and Van Gijsegem, F. (2000). Assembly and function of type III secretory systems. Annu. Rev. Microbiol. 54, 735–774. doi: 10.1146/annurev.micro.54.1.735
Cosentino, S., Voldby Larsen, M., Møller Aarestrup, F., and Lund, O. (2013). PathogenFinder–distinguishing friend from foe using bacterial whole genome sequence data. PLoS ONE 8:e77302. doi: 10.1371/journal.pone.0077302
Di Luca, M. C., Sørum, V., Starikova, I., Kloos, J., Hülter, N., Naseer, U., et al. (2017). Low biological cost of carbapenemase-encoding plasmids following transfer from Klebsiella pneumoniae to Escherichia coli. J. Antimicrob. Chemother. 72, 85–89. doi: 10.1093/jac/dkw350
Göttig, S., Riedel-Christ, S., Saleh, A., Kempf, V. A., and Hamprecht, A. (2016). Impact of blaNDM−1 on fitness and pathogenicity of Escherichia coli and Klebsiella pneumoniae. Int. J. Antimicrob. Agents 47, 430–435. doi: 10.1016/j.ijantimicag.2016.02.019
Hraiech, S., Roch, A., Lepidi, H., Atieh, T., Audoly, G., Rolain, J. M., et al. (2013). Impaired virulence and fitness of a colistin-resistant clinical isolate of Acinetobacter baumannii in a rat model of pneumonia. Antimicrob. Agents Chemother. 57, 5120–5121. doi: 10.1128/AAC.00700-13
Li, A., Yang, Y., Miao, M., Chavda, K. D., Mediavilla, J. R., Xie, X., et al. (2016). Complete sequences of mcr-1-harboring plasmids from extended-spectrum-β-lactamase- and carbapenemase-producing Enterobacteriaceae. Antimicrob. Agents Chemother. 60, 4351–4354. doi: 10.1128/AAC.00550-16
Liu, D., Liu, Z. S., Hu, P., Cai, L., Fu, B. Q., Li, Y. S., et al. (2016). Characterization of surface antigen protein 1 (SurA1) from Acinetobacter baumannii and its role in virulence and fitness. Vet. Microbiol. 186, 126–138. doi: 10.1016/j.vetmic.2016.02.018
Liu, Y. Y., Wang, Y., Walsh, T. R., Yi, L. X., Zhang, R., Spencer, J., et al. (2016). Emergence of plasmid-mediated colistin resistance mechanism MCR-1 in animals and human beings in China: a microbiological and molecular biological study. Lancet Infect. Dis. 16, 161–168. doi: 10.1016/S1473-3099(15)00424-7
Mediavilla, J. R., Patrawalla, A., Chen, L., Chavda, K. D., Mathema, B., Vinnard, C., et al. (2016). Colistin- and carbapenem-resistant Escherichia coli harboring mcr-1 and blaNDM-5, causing a complicated urinary tract infection in a patient from the United States. mBio 7:e01191–16. doi: 10.1128/mBio.01191-16
Nordmann, P., and Poirel, L. (2016). Plasmid-mediated colistin resistance: an additional antibiotic resistance menace. Clin. Microbiol. Infect. 22, 398–400. doi: 10.1016/j.cmi.2016.03.009
Schaufler, K., Semmler, T., Pickard, D. J., de Toro, M., de la Cruz, F., Wieler, L. H., et al. (2016). Carriage of extended-spectrum beta-lactamase-plasmids does not reduce fitness but enhances virulence in some strains of pandemic E. coli lineages. Front. Microbiol. 7:336. doi: 10.3389/fmicb.2016.00336
Starikova, I., Al-Haroni, M., Werner, G., Roberts, A. P., Sørum, V., Nielsen, K. M., et al. (2013). Fitness costs of various mobile genetic elements in Enterococcus faecium and Enterococcus faecalis. J. Antimicrob. Chemother. 68, 2755–2765. doi: 10.1093/jac/dkt270
Teo, J. Q., Ong, R. T., Xia, E., Koh, T. H., Khor, C. C., Lee, S. J., et al. (2016). mcr-1 in multidrug-resistant blaKPC−2-producing clinical Enterobacteriaceae isolates in Singapore. Antimicrob. Agents Chemother. 60, 6435–6437. doi: 10.1128/AAC.00804-16
Wand, M. E., Bock, L. J., Bonney, L. C., and Sutton, J. M. (2015). Retention of virulence following adaptation to colistin in Acinetobacter baumannii reflects the mechanism of resistance. J. Antimicrob. Chemother. 70, 2209–2216. doi: 10.1093/jac/dkv097
Wang, X., Xu, X., Li, Z., Chen, H., Wang, Q., Yang, P., et al. (2014). An outbreak of a nosocomial NDM-1-producing Klebsiella pneumoniae ST147 at a teaching hospital in mainland China. Microb. Drug Resist. 20, 144–149. doi: 10.1089/mdr.2013.0100
Wang, Y., Tian, G. B., Zhang, R., Shen, Y., Tyrrell, J. M., Huang, X., et al. (2017). Prevalence, risk factors, outcomes, and molecular epidemiology of mcr-1-positive Enterobacteriaceae in patients and healthy adults from China: an epidemiological and clinical study. Lancet Infect. Dis. 17, 390–399. doi: 10.1016/S1473-3099(16)30527-8
Yang, R. S., Feng, Y., Lv, X. Y., Duan, J. H., Chen, J., Fang, L. X., et al. (2016). Emergence of NDM-5 and MCR-1-producing Escherichia coli clone ST648 and ST156 from a single muscovy duck (Cairina moschata). Antimicrob. Agents Chemother. 60, 6899–6902. doi: 10.1128/AAC.01365-16
Yao, Y., Xie, Y., Perace, D., Zhong, Y., Lu, J., Tao, J., et al. (2009). The type III secretion system is involved in the invasion and intracellular survival of Escherichia coli K1 in human brain microvascular endothelial cells. FEMS Microbiol. Lett. 300, 18–24. doi: 10.1111/j.1574-6968.2009.01763.x
Keywords: colistin-resistant E. coli, blaNDM-5, mcr-1, fitness, virulence
Citation: Zhang Y, Liao K, Gao H, Wang Q, Wang X, Li H, Wang R and Wang H (2017) Decreased Fitness and Virulence in ST10 Escherichia coli Harboring blaNDM-5 and mcr-1 against a ST4981 Strain with blaNDM-5. Front. Cell. Infect. Microbiol. 7:242. doi: 10.3389/fcimb.2017.00242
Received: 27 February 2017; Accepted: 24 May 2017;
Published: 08 June 2017.
Edited by:
Nora Lía Padola, National University of Central Buenos Aires, ArgentinaReviewed by:
Jorge Blanco, Universidade de Santiago de Compostela, SpainMarcela Radice, University of Buenos Aires, Argentina
Copyright © 2017 Zhang, Liao, Gao, Wang, Wang, Li, Wang and Wang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Hui Wang, d2FuZ2h1aUBwa3VwaC5lZHUuY24=; d2h1aWJqQDE2My5jb20=
†These authors have contributed equally to this work.
 Yawei Zhang1†
Yawei Zhang1† Hua Gao
Hua Gao Henan Li
Henan Li Ruobing Wang
Ruobing Wang Hui Wang
Hui Wang