- 1Federal Research Centre ‘Fundamentals of Biotechnology’ of the Russian Academy of Sciences, A. N. Bach Institute of Biochemistry, Moscow, Russia
- 2Department of Immunology, Laboratory for Immunogenetics, Central Institute for Tuberculosis, Moscow, Russia
- 3Department of Immunology, School of Biology, Lomonosov Moscow State University, Moscow, Russia
Earlier we demonstrated that the adenylyl cyclase (AC) encoded by the MSMEG_4279 gene plays a key role in the resuscitation and growth of dormant Mycobacterium smegmatis and that overexpression of this gene leads to an increase in intracellular cAMP concentration and prevents the transition of M. smegmatis from active growth to dormancy in an extended stationary phase accompanied by medium acidification. We surmised that the homologous Rv2212 gene of M. tuberculosis (Mtb), the main cAMP producer, plays similar physiological roles by supporting, under these conditions, the active state and reactivation of dormant bacteria. To test this hypothesis, we established Mtb strain overexpressing Rv2212 and compared its in vitro and in vivo growth characteristics with a control strain. In vitro, the AC-overexpressing pMindRv2212 strain demonstrated faster growth in a liquid medium, prolonged capacity to form CFUs and a significant delay or even prevention of transition toward dormancy. AC-overexpressing cells exhibited easier recovery from dormancy. In vivo, AC-overexpressing bacteria demonstrated significantly higher growth rates (virulence) in the lungs and spleens of infected mice compared to the control strain, and, unlike the latter, killed mice in the TB-resistant strain before month 8 of infection. Even in the absence of selecting hygromycin B, all pMindRv2212 CFUs retained the Rv2212 insert during in vivo growth, strongly suggesting that AC overexpression is beneficial for bacteria. Taken together, our results indicate that cAMP supports the maintenance of Mtb cells vitality under unfavorable conditions in vitro and their virulence in vivo.
Introduction
Asymptomatic forms of tuberculosis (TB), when infected individuals carry the pathogen in dormant state without notable symptoms of the disease for a very long time, are termed latent tuberculosis infection (LTBI). In some of these latently infected individuals, infection eventually transits to the active state, becomes contagious and seriously affects epidemiological situation (Barry et al., 2009). Conditions under which dormant Mycobacterium tuberculosis (Mtb) transits to active multiplication (resuscitates), as well as the mechanisms of transition, are poorly understood. Thus, currently the problem of LTBI development and reactivation is one of the most important in infectious medicine.
In studies aimed at understanding the molecular mechanisms underlying Mtb dormancy and resuscitation, a significant amount of information has been obtained using experimental models in vitro which mimic the development of mycobacterial dormancy and resuscitation (Shleeva et al., 2002, 2011). For instance, the role of the adenylyl cyclase (AC) encoded by the MSMEG_4279 gene has been elucidated through a model of dormant Mycobacterium smegmatis resuscitation. It was shown that the M. smegmatis strain carrying the knock-out mutation in the MSMEG_4279 gene was unable to resuscitate and required the addition of exogenous cAMP for reactivation. Moreover, M. smegmatis and M. tuberculosis transformation with the plasmid containing the MSMEG_4279 gene expressed under the Tet-promoter, which led to its hyper-expression and an increase in intracellular cAMP concentration, prevented the transition of the two species of bacteria to dormancy under stressful conditions (Shleeva et al., 2013). This suggests that in the presence of high amounts of cAMP provided by the AC the bacteria retained their active state. However, direct evidence for the “anti-dormant” role of the M. tuberculosis gene(s) encoding AC for Mtb remained lacking.
The homolog of MSMEG_4279 in Mtb is the Rv2212 gene, which is the major producer of cAMP among 16 biochemically active AC-encoding genes present in the genome (Abdel Motaal et al., 2006; Knapp and McDonough, 2014). We assumed that under stressful conditions the Rv2212 gene, like MSMEG_4279, contributes to the retention of the active metabolic state of Mtb, as well as the reactivation of dormant bacteria due to an increase in cAMP production. To test this hypothesis, we established a novel Mtb strain carrying the plasmid containing Rv2212 expressed under the Tet-promoter and compared the in vitro and in vivo phenotypes of this Rv2212-overexpressing strain with the control strain carrying the “empty” plasmid. Our data clearly demonstrate that the Rv2212-dependent AC activity, which results in elevated cAMP production, supports Mtb vitality under unfavorable conditions in a culture medium and inside the host, as well as its resuscitation from dormant state.
Materials and Methods
Bacterial Strains, Growth Media, and Culture Conditions
The wild type Mtb strain H37Rv and its derivatives carrying plasmids pMindRv2212 (see below) and pMind (empty plasmid control) were used. Hygromycin B was added to the growth media at the 50 μg/ml concentration for the plasmid-containing strains. All strains were routinely maintained on the standard Sauton's medium: 0.5 g KH2PO4, 1.4 g MgSO4·7H2O, 4 g L-asparagine, 60 ml glycerol; 0.05 g ferric ammonium citrate; 2 g sodium citrate, 0.1 ml 1% ZnSO4·7H2O, adjusted to l L with H2O at pH = 7.0 (adjusted with 1 M NaOH) and supplemented with ADC (albumen, glucose and NaCl) with 0.05% Tween-80 (Connell, 1994).
Mycobacterial populations consisting of bacilli that lost capacity to grow on solid media (“non-culturability,” NC) due to gradual acidification of medium during stationary growth phase were developed as described earlier (Shleeva et al., 2011). Briefly, bacteria were kept for 12–15 days in 50 ml of Sauton's medium supplemented with 0.05% Tween-80 and ADC in 150 ml conical flasks on an orbital shaker (200 rpm). These bacterial cultures served for inoculation of modified Sauton's medium aliquots for establishing NC bacteria. The modified Sauton's medium in which Tween-80 is replaced with 0.025% tyloxapol and ADC—with 0.5% bovine albumin, Cohn-Analog (Sigma) compared to the standard Sauton's medium is hereafter termed “glycerol” medium. Another formulation of the modified Sauton's medium containing 4% glucose and 0.2% glycerol is hereafter termed “glucose”medium. Initial pH values in both modified media were 6.2, contrary to pH = 7.0 in the standard Sauton's medium. An 1-ml inoculum of initial culture was added to 200 ml of modified Sauton's medium put into a 500-ml conical flasks (3–5 flasks per experiment), and incubated with shaking at 37°C for 40–60 days. The pH value of medium was measured periodically, and, when reached 6.0–6.2, the cultures were transferred to capped plastic 50-ml tubes, and 2-(N-Morpholino)ethanesulfonic acid (MES) was added at the final 20 mM concentration to prevent further acidification during long-term storage. Incubation was continued under static conditions (i.e., without agitation) at the room temperature for up to 200 days post-inoculation, in the dark. Periodically, samples were collected from different tubes for estimation of CFU counts and the results provided by the most probable numbers (MPN) assay (see below for details) to monitor appearance of NC bacteria. In some experiments, tetracycline hydrochloride was added to medium at the final concentration 20 ng per ml.
Resuscitation of NC bacteria was performed in reactivation medium—the 1:1 mixture of the standard Sauton's medium and the Nutrient Broth E (NBE) medium (HiMedia, India) supplemented with 0.025% tyloxapol (Sigma) in two formats. For the MPN format, 15 ml plastic test tubes (Corning, USA) containing 2 ml reactivation medium were used. Serially diluted NC bacteria were incubated in triplicates at 37°C for 30–50 days without shaking, and the number of tubes with visible bacterial growth was scored. The MPN values were determined using standard statistical tables (de Man, 1975). For the batch format, dormant Mtb bacteria prepared as described above were washed 10 times with PBS, re-suspended in 200 ml of reactivation medium at the initial OD600 = 0.2–0.3, put into a 500-ml flask and incubated at 37°C for 45 days with 100–120 rpm agitation. OD values were periodically in cultural samples.
Estimation of Bacterial Capacity to Form CFU
Bacterial suspensions were serially diluted in fresh Sauton's medium, and triplicate 100-μl samples from each dilution were dropped on Sauton agar. Plates were incubated at 37°C for 21 day, and the numbers of colony forming units (CFU) were counted. The limit of detection was 10 CFU/ml.
DNA Manipulations
The pMindRv2212 overexpression plasmid was constructed as follows:
Amplification of genomic M. tuberculosis DNA was performed using the primers:
Up2212 5′CTGGATCCTCGCTCACGGCGTCCCACCCTA3′,
Low2212 5′CAACTAGTTCGCGACGGCGACGGAGGGGGATAG3′.
The restriction sites are underlined. Amplification products were firstly cloned into pGEM-T vector (Promega), and then sub-cloned into pMind vector using BamHI and SpeI restriction enzymes (Thermo Scientific). Escherichia coli and M. tuberculosis were transformed with the pMindRv2212 vector by electroporation. After growing in selective medium, colonies were checked using PCR, plasmids from colonies were extracted and checked by sequencing.
Ex vivo CFU Analysis
Mycobacterial colonies (N = 50) obtained from lung homogenates were analyzed by PCR in order to confirm the presence of the Rv2212 gene insertion. Amplification was performed using p-Mind specific primers:
Up-pMind 5′CCGGGCCCCGAGCAACACG3′
Low-pMind 5′CCGCAGGCTCGCGTAGGAATCATC3′
All resulting products were about 1,300 bp length, which corresponds to the gene insertion size.
cAMP Determination
Mtb samples from different strains and time points of storage were centrifuged at 13,000 g for 5 min. The pellets were treated with 1 ml of 95°C-heated 0.1N HCl for 10 min and immediately frozen. Samples were disrupted in the bead homogenizer FastPrep-24 (MP Biomedicals, The Netherlands), bacterial debris was removed by centrifugation, and cAMP levels were measured in lysates by ELISA, using rabbit anti-cAMP antibodies (1:5,000) and cAMP-peroxidase conjugate (HRP, 1: 20,000) (GenScript, USA). The results were registered in triplicates at 450 nm using Zenyth 3100 microplate reader (Anthos Labtec Instruments, Austria). At least three independent measurements were performed.
Mice and Infection
Mice of inbred strains I/StSnEgYCit (I/St) and C57BL/6JCit (B6) were bred and maintained under conventional, non-SPF conditions at the Animal Facilities of the Central Institute for Tuberculosis (CIT, Moscow, Russia) in accordance with guidelines from the Russian Ministry of Health # 755, and under the NIH Office of Laboratory Animal Welfare (OLAW) Assurance #A5502-11. Water and food were provided ad libitum. Female mice of 8–12 week of age at the beginning of experiments were used. All experimental procedures were approved by the CIT animal care committee.
Mice were infected intravenously with 5 × 106 bacteria per mouse in 0.5 ml of sterile PBS. Neither tetracycline nor hygromycin B were administrated in these experiments. To assess mycobacterial loads in spleens and lungs, 0.2 ml of serial 10-fold dilutions of individual whole-organ homogenates obtained from 4 mice per group were plated onto Dubos agar, and colonies were counted after 21–23 days of incubation at 37°C. Two independent experiments were performed and their results combined. Mortality was checked weekly.
Statistics
OD, CFU, cAMP concentrations, Resuscitation Index (RI) were expressed as mean ± SEM. MPN were calculated used (95%) confidence limits. Significance of the differences for the in vivo experiments were estimated by ANOVA, P < 0.05 was considered significant.
Results
Growth of Mtb Strains and Resuscitation of Dormant Bacteria In vitro
To study the influence of AC on Mtb growth and resuscitation, we constructed the strain (pMindRv2212) overexpressing AC under the control of the tetracycline (Tet) promoter and demonstrated that this strain displayed a statistically significant increase in the intracellular cAMP levels in exponential phase compared to the empty-vector control strain pMind. The uninduced strain also displayed an elevated level of cAMP expression comparable to that of the induced strain (Figure S1). Apparently, in the absence of the inducer, the background expression level under the Tet promoter in Mtb is sufficient, which is in full agreement with our finding in M. smegmatis for the overexpressed MSMEG_4279 (a homolog of Rv2212) (Shleeva et al., 2013). Thereafter, we compared the growth of these strains in liquid Sauton medium. As shown in Figure 1, the bacteria overexpressing AC grew faster than the control strain. Remarkably, a strong difference in growth rates between the two strains was readily revealed if the starting inoculum contained a low number of bacteria (102 per ml), i.e., provided more unfavorable conditions for growth initiation than larger starting populations did (Mukamolova et al., 2002). Indeed, when a 3-log larger inoculum initiated the growth of the two strains, the speed of multiplication appeared to be almost identical (Figure S2). We also noticed that, in contrast to pMind bacteria, pMindRv2212 quickly recovered from an old inoculum and from dried samples (data not shown).
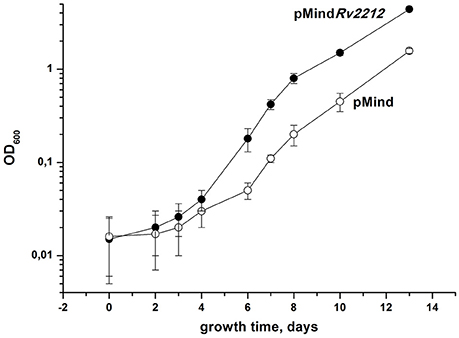
Figure 1. Growth of M. tuberculosis strains in the standard Sauton's medium. Recombinant M. tuberculosis strains containing pMindRv2212 vector (closed circles) or empty pMind vector (open circles) were cultured in the standard Sauton's medium with agitation (200 rpm) at 37°C. Initial size of inoculum was 102 cells per ml. This experiment was repeated three times with similar results. One representative experiment is shown.
Next, we studied whether the overexpression of the Rv2212 gene influences the transition of actively growing bacteria to dormancy. We explored an earlier developed model which makes it possible to accumulate dormant ovoid cells under gradual self-acidification of the growth medium during prolonged stationary phase. These dormant bacteria, which display a distinct morphology and are characterized by “non-culturability” (NC), i.e., a transient loss of capacity to grow on solid media, can be developed in modified Sauton medium, containing either glycerol (Shleeva et al., 2011) or glucose as the major source of carbon. As shown in Figure 2A, in the glycerol-based medium the development of NC pMindRv2212 bacteria was significantly postponed compared to NC pMind bacteria. After their development in the glucose-based medium, pMindRv2212 bacteria provided identical CFU counts on the solid medium throughout the observation period, whereas the control strain gradually acquired the NC phenotype, although much slower than in the glycerol-based medium (Figure 2B). The difference between two media could be due to faster acidification of the glycerol-based medium during pMind strain growth and the lack of acidification of the glucose-based medium during pMindRv2212 strain growth (data not shown).
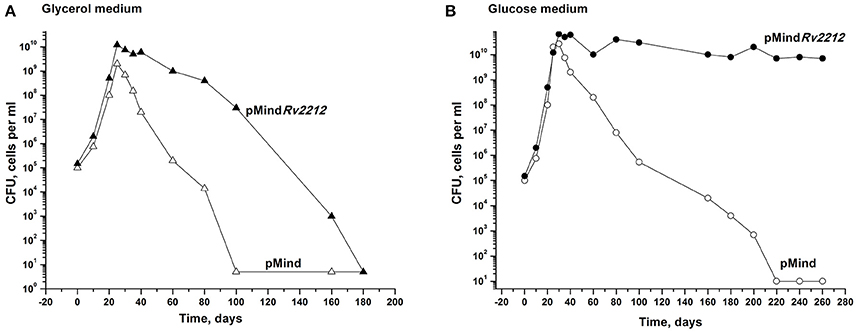
Figure 2. Overexpression of Rv2212 influences transition of M. tuberculosis to dormant “non-culturable” state. Recombinant Mtb strains containing pMindRv2212 vector (closed circles) or empty pMind vector (open circles) were grown in “glycerol” (A) or “glucose” (B) modified Sauton's media with agitation (200 rpm) at 37°C. For details see Materials and Methods. Minimum threshold for CFU determination was 10 cells per ml. SEM for the CFU determination was <20%. Experiments shown in (A) and in (B) were repeated two times and four times, respectively. Typical results are shown.
At least some NC bacteria are able to resuscitate upon cultivation in an appropriate liquid medium. During culture storage, the numbers of bacteria temporarily existing in the NC state but retaining capacity for resuscitation were assessed using the most probable number (MPN) assay based upon serial dilutions of bacteria incubated in a liquid medium (de Man, 1975; Shleeva et al., 2002). By plotting the MPN assay results against the numbers of CFU (the so-called Resuscitation Index, RI) we evaluated the development of the NC state in the two bacterial strains, as well as the contribution of those dormant bacteria, which retained their resuscitation capacity during long storage, to the total size of bacterial population. As shown in Figure 3A, RI varied between 100 (1, corresponds to fully active, multiplying bacteria) and 108 (almost the entire population consists of NC bacteria). Remarkably, pMind bacteria achieved the highest RI value by day 100 of storage, whereas in pMindRv2212 population this condition was achieved not earlier than day 180, indicating significance of the Rv2212 expression for the maintenance of active growth under long storage. In addition, as shown for the wild type bacteria, there was a clear reverse correlation between the RI value and the content of cAMP in bacterial cells, i.e., gradual increase of bacterial population reaching the NC state during prolonged storage (Figure 3B).
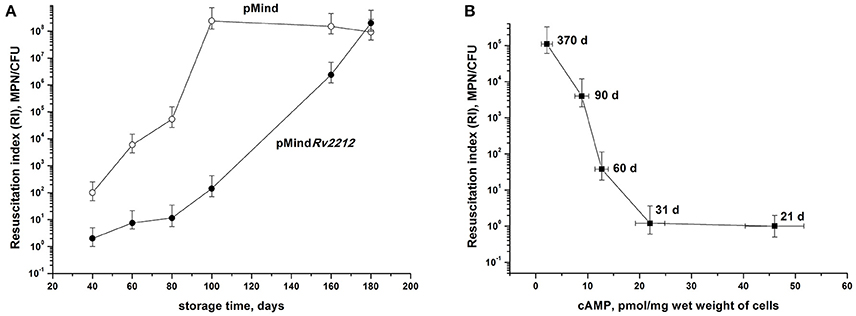
Figure 3. Development of “non-culturability” (A) and cAMP content (B) of M. tuberculosis during dormant bacteria storage. (A) M. tuberculosis pMindRv2212 (closed circles) or pMind (open circles) developed in the “glycerol” modified Sauton's medium were kept statically at the room temperature. Periodically, samples were collected for RI estimation. (B) The wild type mycobacteria were grown and kept under identical conditions for monitoring cAMP intracellular concentrations and RI estimation. Sampling days are indicated. Experiment shown in (A) was repeated twice, one representative experiment is shown. Results shown in (B) display the average from three independent experiments.
The resuscitation of NC bacteria developed in the glycerol-based medium (see Figure 2A, 180 days) was also assessed using the batch format (Shleeva et al., 2013), when identical numbers of bacteria are inoculated in flasks containing resuscitation medium. In this experiment, pMindRv2212 bacteria exhibited significantly shorter lag phase and quicker resuscitation compared to pMind strain (Figure 4).
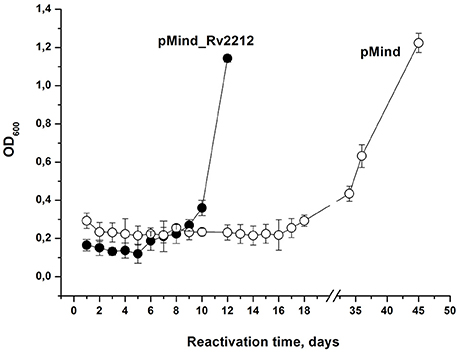
Figure 4. Overexpression of Rv2212 results in faster resuscitation of dormant M. tuberculosis. Non-culturable Mtb pMindRv2212 (closed circles) or pMind (open circles) obtained from the “glycerol” Sauton's modified medium after 180-d incubation (see the legend to Figure 2A) were washed, inoculated in the reactivation medium and incubated with agitation (100 rpm) at 37°C. For the details see Materials and Methods. The experiment was repeated two times with similar results.
Infection Caused by Two Mycobacterial Strains in Mice
In previous experimentation, we and others characterized the phenotypes controlled by the MSMEG_4279 and Rv2212 genes in vitro and in cultured infected macrophages. In order to study the effect of Rv2212 overexpression on the bona fide tuberculosis infection characteristics, mice of two inbred strains, genetically TB-resistant B6 and hyper-susceptible I/St (Nikonenko et al., 2000), were infected with pMindRv2212 and pMind bacteria. First, we obtained an indirect evidence for the AC expression contribution to mycobacterial vitality. Normally, in the absence of specific selection, mycobacteria eject any inserted plasmid if the coding gene does not provide any selection advantage. To assess the stability of the plasmid presence, at week 3 of infection lung homogenates were plated onto Dubo agar and the presence of the plasmid has been evaluated in the resulting bacterial colonies in the PCR format. Even in the absence of selecting hygromycin all colonies analyzed contained the plasmid with an intact Rv2212 insert, strongly suggesting that the AC overexpression is beneficial for the bacteria multiplying in the host. This conclusion was further supported by the observation that the pMindRv2212 colonies displayed markedly increased sizes compared to pMind colonies developed under identical conditions (Figure 5A). At the early stage of infection (3 weeks), the lung CFU counts in genetically susceptible I/St mice were significantly lower for the pMind strain compared to the pMindRv2212, whereas genetically more resistant B6 mice controlled the infection caused by the two mycobacterial strains equally well (Figure 5B). However, at month 6 post-challenge, even B6 mice were unable to control the growth of pMindRv2212 strain in lungs, which was not the case for the pMind strain. Lung CFU counts increased ~1 log compared to the 3-week counts for the pMindRv2212 strain, and significant differences were also evident in spleens (Figure 5C). Importantly, in this experiment the CFU counts were identical for hygromycin-containing and antibiotic-free plates, confirming that the plasmid was retained by bacteria throughout the long, chronic infection course (data not shown). In agreement with the results of mycobacterial CFU evaluation, mortality curves obtained in B6 mice demonstrated that the pMindRv2212 strain was much more virulent in comparison with the control strain (Figure 6).
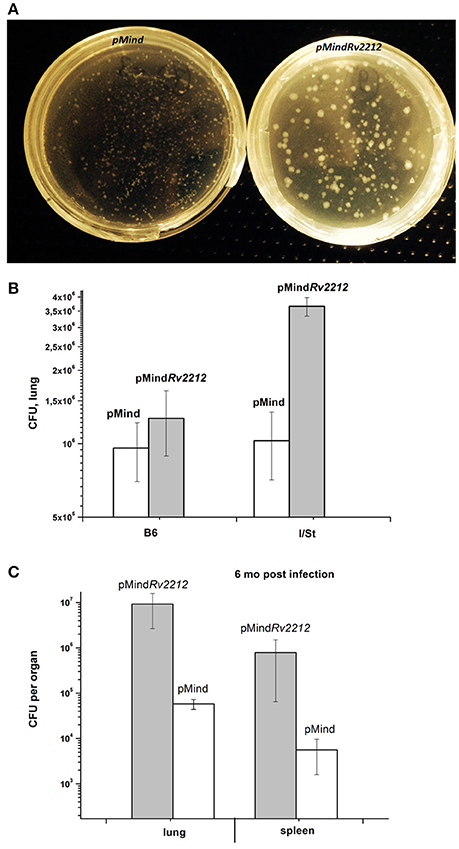
Figure 5. M. tuberculosis pMindRv2212 and pMind phenotypes expressed in vivo. (A) pMindRv2212 strain (left) isolated from lungs provided larger colonies compared to the control strain (right). Lungs of infected B6 mice were homogenized, plated on Dubos agar, kept at 37°C for 3 weeks and photographed. (B) Mice of I/St and B6 inbred strains were infected with 5 × 106 CFUs of M. tuberculosis pMindRv2212 or pMind. At week three post-infection, lungs were homogenized and serial dilutions were plated on Dubo medium. The results of two experiments including 4 mice each (total N = 8) are expressed as mean ± SEM. Significant difference (P < 0.01, ANOVA) was observed between pMindRv2212-infected B6 and I/St mice. (C) At 6 months post-challenge, ~2 log differences (P < 0.001, ANOVA) were observed between multiplication of pMindRv2212 and pMind mycobacteria in lungs and spleens of B6 mice. The results are expressed as mean ± SEM (N = 6).
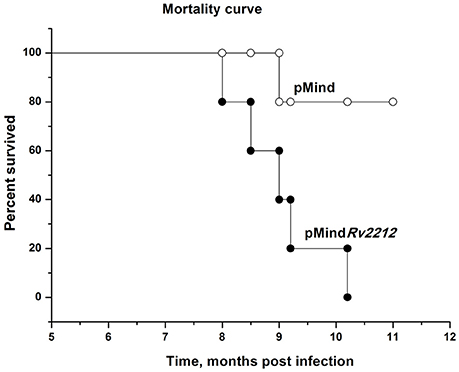
Figure 6. Mortality curve for M. tuberculosis pMindRv2212- and pMind-infected B6 mice. Mice of B6 inbred strain were infected with 5 × 106 CFUs of M. tuberculosis pMindRv2212 (closed circles) or pMind (open circles). Mortality was monitored weekly.
Discussion
In this study, we present a few lines of evidence indicating the importance of AC expression and cAMP production for maintaining Mtb vitality under unfavorable for growth conditions. Firstly, there was a significant difference in in vitro growth dynamics between the AC-overexpressing pMindRv2212 and the control strain if the development of bacterial population started from a small amount of ancestors (Figure 1). Such “low inoculum” cultures are difficult to grow (Mukamolova et al., 2002; Drancourt and Raoult, 2007) and most often require additional external growth factors to initiate their active replication (Mukamolova et al., 2002). On the contrary, if a bulk inoculum is used for the initiation of mycobacterial liquid culture, the overexpression of the Rv2212 gene does not influence growth dynamics (Figure S2). As shown earlier, M. bovis BCG with overexpressed Rv2212 did not show a significant difference in growth in vitro compared to the wild type bacteria; however, only high initial cell concentrations were used in this study. Remarkably, this strain displayed a substantial growth acceleration compared to the control strain within mouse macrophages, i.e., when stressful conditions for intracellular parasite are evident (Pedroza-Roldán et al., 2015). Similarly, the overexpression of Rv2212 in M. smegmatis resulted in an enhanced survival of bacteria within macrophages (Ganaie et al., 2016).
Secondly, the importance of cAMP production was confirmed in experiments on the development of dormant, NC bacteria in vitro under acidic stress. The AC overexpression resulted in the prolongation of bacterial capacity to form CFUs and in a significant delay or even prevention of bacterial transition toward dormancy (Figure 2). Thirdly, elevated Rv2212 expression and cAMP production substantially stimulated the recovery of M. tuberculosis from the NC state and resuscitation of its active growth (Figure 4). Earlier we found that the NC M. smegmatis resuscitation was induced by exogenous fatty acids via MSMEG_4279 (ortolog of Rv2212) activation followed by an increase in intracellular cAMP concentrations. The knocked-out M. smegmatis strain lacking MSMEG_4279 was unable to resuscitate in the presence of fatty acids until exogenous cAMP was added (Shleeva et al., 2013). Finally, the in vivo experiments demonstrated that the increased level of AC expression (and evidently cAMP production) in the mutant stimulated Mtb multiplication in mouse organs.
This latter result is in a sharp contrast with observation by Pedroza-Roldán et al. (2015) concerning the BCG-Rv2212 strain, which exhibited almost identical growth with control strain in mouse organs. We suspect that this difference may be connected with different locations of Mtb and M. bovis BCG within the host cells.
It was established that highly virulent mycobacterial species, e.g., M. tuberculosis and M. leprae, readily migrate from phagosomes into the cytosol of myeloid cells and continue their replication in this new cellular compartment (van der Wel et al., 2007). The fact that this migration is largely dependent upon the ESAT-6 protein, which perturbs the TLR2-MyD88 signaling pathway in the host cells, is significant for the prevention of mycobacterial migration from phagosomes (Rahman et al., 2014). In the genomes of the vast majority of non-virulent or low-virulent mycobacteria, including BCG, the secretory RD1 system, which contains genes for major virulence factors ESAT-6 and cfp10, is lacking (van Ingen et al., 2009), and a very large proportion of the total bacterial population indeed resides within macrophage phagosomes. Differences in intracellular cAMP concentrations might have different impact on the survival of mycobacteria residing within phagosome and cytosol compartments.
A relatively moderate increase in cAMP concentrations (~2.5-fold) appeared to be sufficient to elicit numerous growth-promoting effects of AC overexpression described above. The reason why such a subtle increase results in significant changes in bacterial physiology is unclear, given that during prolonged storage cAMP concentrations dropped 10-fold, whilst the numbers of NC bacteria showed ~5-log changes (Figure 3B). One possibility is that there exists a critical threshold for cAMP concentrations (18–20 pmol per 1 mg of wet weight), below which bacteria rapidly acquire NC phenotype, but even a moderate increase in the cAMP concentrations is sufficient for resuscitation and growth. Our earlier results demonstrating a rapid increase in cAMP content at the initial stage of M. smegmatis resuscitation support this conclusion (Shleeva et al., 2013).
There is ample evidence that cAMP production is essential for the survival of the pathogen within host cells. Thus, high cAMP amounts secreted by Mtb in macrophages modulate phagosome-lysosome fusion, thus increasing mycobacterial survival (Kalamidas et al., 2006). The macrophage “intoxication” by high cAMP concentrations may have a positive effect on mycobacterial survival (Agarwal et al., 2009). cAMP influences the expression of genes involved in mycobacterial response to hypoxic conditions often present in vivo (Gazdik and McDonough, 2005). High cAMP concentrations raise mycobacterial resistance to stressful conditions by elevating the expression of shock proteins GroEL and DnaK (Pedroza-Roldán et al., 2015). In addition, cAMP directly regulates the activity of such enzymes as malate dehydrogenase (Gazdik and McDonough, 2005) and lysine acetylase (Rv0998) (Knapp and McDonough, 2014) involved in the central mycobacterial metabolic pathways (Xu et al., 2011). These diverse effects can plausibly explain the better survival of the Rv2212-overexpressing strain in vivo.
Basing on the in vitro data, we suggest that a high level of cAMP interferes with bacterial transition to dormancy and development of latent-like disease in vivo. Further experiments are needed to elucidate this possibility, especially since the pMindRv2212 strain demonstrated an increased virulence in both genetically TB-susceptible and TB-resistant mice (Figures 5, 6). This is an important observation, suggesting that either mycobacterial AC itself or biochemical cascades upstream AC may be considered as a target for novel anti-TB drugs.
Also of note, that the molecular mechanisms increasing mycobacterial vitality under elevated cAMP concentrations remain obscure. The significance of intracellular cAMP as a second messenger for a wide variety of bacterial biochemical processes is well-established (Baker and Kelly, 2004), but the specific downstream processes linking cAMP levels and bacterial vitality on the transcriptional level are not well-defined. Two transcriptional factors sensing cAMP inside the cell were described: CRPMt (Rv3676) and Cmr (Rv1675c) (Knapp and McDonough, 2014). It was demonstrated that Cmr has more than 300 binding sites along a bacterial chromosome and thus potentially may be involved in regulating a wide variety of metabolic processes. In the context of present work, it is worth mentioning that cAMP modulates Cmr binding with the genes belonging to the DosR regulon, which is important for Mtb survival under hypoxic conditions in a non-replicating state (Ranganathan et al., 2016). CRPMt displays pleotropic effects as well, including the control of amino acid biosynthesis (Bai et al., 2011), expression of the RpfA gene encoding a mycobacterial resuscitation promoting factor (Mukamolova et al., 2003; Rickman et al., 2005; Shleeva et al., 2013), and the expression of succinate dehydrogenase (Rv0247c-Rv0249c) (Knapp et al., 2015).
Our study clearly demonstrates that elevated cAMP production due to the Rv2212-dependent AC activity plays an “anti-dormant” role by supporting, under unfavorable conditions, the active state of Mtb in culture and its multiplication inside the host. The exact metabolic structure of this “vitality supporting” axis remains to be identified.
Author Contributions
AK and MS conceived and designed the experiments. MS, GD, and AG performed the experiments in vitro. TK and ER performed the experiments in vivo. AK, MS, and AA analyzed the data. MS prepared figures and graphs. AK, MS, and AA wrote the manuscript. All the authors read and approved the final manuscript. AK, MS, and AA revised the manuscript.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
This work was financially supported by the Russian Science Foundation grants 16-15-00245 (to AK, in vitro studies) and 15-15-30020 (to TK, in vivo studies). We thank Dr. Brian Robertson for providing the pMind plasmid.
Supplementary Material
The Supplementary Material for this article can be found online at: http://journal.frontiersin.org/article/10.3389/fcimb.2017.00370/full#supplementary-material
Figure S1. cAMP levels in different strains of M. tuberculosis grown on Sauton's medium for 8 days. Recombinant M. tuberculosis strains containing pMindRv2212 vector or empty pMind vector were cultured in the standard Sauton's medium with agitation (200 rpm) at 37°C for 8 days. In some experiments, tetracycline hydrochloride was added to medium at the final concentration 20 ng per ml. Results display the average from three independent experiments.
Figure S2. Growth of different M. tuberculosis strains in the standard Sauton's medium from the 105 inoculum. Recombinant M. tuberculosis strains containing pMindRv2212 vector (closed circles) or empty pMind vector (open circles) were cultured in the standard Sauton's medium with agitation (200 rpm) at 37°C. Initial size of inoculum was 105 bacteria per ml. This experiment was repeated 3 times with similar results, one representative experiment is shown.
References
Abdel Motaal, A., Tews, I., Schultz, J. E., and Linder, J. U. (2006). Fatty acid regulation of adenylyl cyclase Rv2212 from Mycobacterium tuberculosis H37Rv. FEBS J. 273, 4219–4228. doi: 10.1111/j.1742-4658.2006.05420.x
Agarwal, N., Lamichhane, G., Gupta, R., Nolan, S., and Bishai, W. R. (2009). Cyclic AMP intoxication of macrophages by a Mycobacterium tuberculosis adenylate cyclase. Nature 460, 98–102. doi: 10.1038/nature08123
Bai, G., Schaak, D. D., Smith, E. A., and McDonough, K. A. (2011). Dysregulation of serine biosynthesis contributes to the growth defect of a Mycobacterium tuberculosis crp mutant. Mol. Microbiol. 82, 180–198. doi: 10.1111/j.1365-2958.2011.07806.x
Baker, D. A., and Kelly, J. M. (2004). Structure, function and evolution of microbial adenylyl and guanylyl cyclases. Mol. Microbiol. 52, 1229–1242. doi: 10.1111/j.1365-2958.2004.04067.x
Barry, C. E. III., Boshoff, H. I., Dartois, V., Dick, T., Ehrt, S., Flynn, J., et al. (2009). The spectrum of latent tuberculosis: rethinking the biology and intervention strategies. Nat. Rev. Microbiol. 7, 845–855. doi: 10.1038/nrmicro2236
Connell, N. D. (1994). Mycobacterium: isolation, maintenance, transformation, and mutant selection. Methods Cell Biol. 45, 107–125.
Drancourt, M., and Raoult, D. (2007). Cost-Effectiveness of Blood Agar for Isolation of Mycobacteria. PLoS Negl. Trop. Dis. 1:e83. doi: 10.1371/journal.pntd.0000083
Ganaie, A. A., Trivedi, G., Kaur, A., Jha, S. S., Anand, S., Rana, V., et al. (2016). Interaction of Erp protein of Mycobacterium tuberculosis with Rv2212 enhances intracellular survival of Mycobacterium smegmatis. J. Bacteriol. 198, 2841–2852. doi: 10.1128/JB.00120-16
Gazdik, M. A., and McDonough, K. A. (2005). Identification of cyclic AMP-regulated genes in Mycobacterium tuberculosis complex bacteria under low-oxygen conditions. J. Bacteriol. 187, 2681–2692. doi: 10.1128/JB.187.8.2681-2692.2005
Kalamidas, S. A., Kuehnel, M. P., Peyron, P., Rybin, V., Rauch, S., Kotoulas, O. B., et al. (2006). cAMP synthesis and degradation by phagosomes regulate actin assembly and fusion events: consequences for mycobacteria. J. Cell Sci. 119, 3686–3694. doi: 10.1242/jcs.03091
Knapp, G. S., and McDonough, K. A. (2014). Cyclic AMP signaling in Mycobacteria. Microbiol. Spectr. 2, MGM2-0011-2013. doi: 10.1128/microbiolspec.MGM2-0011-2013
Knapp, G. S., Lyubetskaya, A., Peterson, M. W., Gomes, A. L., Ma, Z., Galagan, J. E., et al. (2015). Role of intragenic binding of cAMP responsive protein (CRP) in regulation of the succinate dehydrogenase genes Rv0249c-Rv0247c in TB complex mycobacteria. Nucleic Acids Res. 43, 5377–5393. doi: 10.1093/nar/gkv420
Mukamolova, G. V., Kaprelyants, A. S., Kell, D. B., and Young, M. (2003). Adoption of the transiently non-culturable state–a bacterial survival strategy? Adv. Microb. Physiol. 47, 65–129. doi: 10.1016/S0065-2911(03)47002-1
Mukamolova, G. V., Turapov, O. A., Young, D. I., Kaprelyants, A. S., Kell, D. B., and Young, M. (2002). A family of autocrine growth factors in Mycobacterium tuberculosis. Mol. Microbiol. 46, 623–635. doi: 10.1046/j.1365-2958.2002.03184.x
Nikonenko, B. V., Averbakh, M. M. Jr., Lavebratt, C., Schurr, E., and Apt, A. S. (2000). Comparative analysis of mycobacterial infections in susceptible I/St and resistant A/Sn inbred mice. Tuber. Lung Dis. 80, 15–25. doi: 10.1054/tuld.1999.0225
Pedroza-Roldán, C., Aceves-Sánchez, M. J., Zaveri, A., Charles-Niño, C., Elizondo-Quiroga, D. E., Hernández-Gutiérrez, R., et al. (2015). The adenylyl cyclase Rv2212 modifies the proteome and infectivity of Mycobacterium bovis BCG. Folia Microbiol. 60, 21–31. doi: 10.1007/s12223-014-0335-1
Rahman, V. F., Sobia, P., Gupta, N., Kaer, L. V., and Das, G. (2014). Mycobacterium tuberculosis subverts the TLR-2 – MyD88 pathway to facilitate its translocation into the cytosol. PLoS ONE 9:e86886. doi: 10.1371/journal.pone.0086886
Ranganathan, S., Bai, G., Lyubetskaya, A., Knapp, G. S., Peterson, M. W., Gazdik, M., et al. (2016). Characterization of a cAMP responsive transcription factor, Cmr (Rv1675c), in TB complex mycobacteria reveals overlap with the DosR (DevR) dormancy regulon. Nucleic Acids Res. 44, 134–151. doi: 10.1093/nar/gkv889
Rickman, L., Scott, C., Hunt, D. M., Hutchinson, T., Menéndez, M. C., Walan, R., et al. (2005). A member of the cAMP receptor protein family of transcription regulators in Mycobacterium tuberculosis is required for virulence in mice and controls transcription of the rpfA gene coding for a resuscitation promoting factor. Mol. Microbiol. 56, 1274–1286. doi: 10.1111/j.1365-2958.2005.04609.x
Shleeva, M. O., Bagramyan, K., Telkov, M. V., Mukamolova, G. V., Young, M., Kell, D. B., et al. (2002). Formation and resuscitation of “non-culturable” cells of Rhodococcus rhodochrous and Mycobacterium tuberculosis in prolonged stationary phase. Microbiology 148, 1581–1591. doi: 10.1099/00221287-148-5-1581
Shleeva, M. O., Kudykina, Y. K., Vostroknutova, G. N., Suzina, N. E., Mulyukin, A. L., and Kaprelyants, A. S. (2011). Dormant ovoid cells of Mycobacterium tuberculosis are formed in response to gradual external acidification. Tuberculosis 91, 146–154. doi: 10.1016/j.tube.2010.12.006
Shleeva, M., Goncharenko, A., Kudykina, Y., Young, D., Young, M., and Kaprelyants, A. (2013). Cyclic AMP-dependent resuscitation of dormant Mycobacteria by exogenous free fatty acids. PLoS ONE 8:e82914. doi: 10.1371/journal.pone.0082914
van der Wel, N., Hava, D., Houben, D., Fluitsma, D., van Zon, M., Pierson, J., et al. (2007). M. tuberculosis and M. leprae translocate from the phagosome to the cytosol in myeloid cells. Cell 129, 1287–1298. doi: 10.1016/j.cell.2007.05.059
van Ingen, J., Zwa, R., Dekhuijzen, R., Boeree, M., and van Soolingen, D. (2009). Region of Difference 1 in nontuberculous Mycobacterium species adds a phylogenetic and taxonomical character. J. Bacteriol. 191, 5865–5867. doi: 10.1128/JB.00683-09
Keywords: Mycobacterium tuberculosis, cAMP, adenylyl cyclase, Rv2212, dormant mycobacteria
Citation: Shleeva MO, Kondratieva TK, Demina GR, Rubakova EI, Goncharenko AV, Apt AS and Kaprelyants AS (2017) Overexpression of Adenylyl Cyclase Encoded by the Mycobacterium tuberculosis Rv2212 Gene Confers Improved Fitness, Accelerated Recovery from Dormancy and Enhanced Virulence in Mice. Front. Cell. Infect. Microbiol. 7:370. doi: 10.3389/fcimb.2017.00370
Received: 07 June 2017; Accepted: 02 August 2017;
Published: 17 August 2017.
Edited by:
Anthony Baughn, University of Minnesota, United StatesReviewed by:
Paras Jain, Albert Einstein College of Medicine, United StatesJiaoyu Deng, Wuhan Institute of Virology (CAS), China
Kyle Rohde, University of Central Florida, United States
Copyright © 2017 Shleeva, Kondratieva, Demina, Rubakova, Goncharenko, Apt and Kaprelyants. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Margarita O. Shleeva, bWFyZ29zaGxlZXZhQGdtYWlsLmNvbQ==
 Margarita O. Shleeva
Margarita O. Shleeva Tatyana K. Kondratieva2
Tatyana K. Kondratieva2 Galina R. Demina
Galina R. Demina Alexander S. Apt
Alexander S. Apt