- Department of Animal Sciences, The Robert H. Smith Faculty of Agriculture, Food and Environment, The Hebrew University of Jerusalem, Rehovot, Israel
The gonadotropins follicle-stimulating hormone (FSH) and luteinizing hormone (LH) are key regulators of the reproductive axis in vertebrates. Despite the high popularity of zebrafish as a model organism for studying reproductive functions, to date no transgenic zebrafish with labeled gonadotropes have been introduced. Using gonadotropin regulatory elements from tilapia, we generated two transgenic zebrafish lines with labeled gonadotropes. The tilapia and zebrafish regulatory sequences were highly divergent but several conserved elements allowed the tilapia promoters to correctly drive the transgenes in zebrafish pituitaries. FSH cells reacted to stimulation with gonadotropin releasing hormone by proliferating and showing increased transgene fluorescence, whereas estrogen exposure caused a decrease in cell number and transgene fluorescence. Transgene fluorescence reflected the expression pattern of the endogenous fshb gene. Ontogenetic expression of the transgenes followed typical patterns, with FSH cells appearing early in development, and LH cells appearing later and increasing dramatically in number with the onset of puberty. Our transgenic lines provide a powerful tool for investigating the development, anatomy, and function of the reproductive axis in lower vertebrates.
Introduction
The hypothalamic–pituitary–gonadal (HPG) axis is the master regulator of reproduction in vertebrates. Hypothalamic axons secrete peptides that bind to specific receptors on gonadotrope cells in the anterior pituitary and stimulate the secretion of gonadotropins (GtHs) from vesicles within these cells. The main stimulator of GtH secretion is the decapeptide gonadotropin releasing hormone (GnRH) but recently, several other hypothalamic factors have been shown to act directly on the pituitary cells to enhance GtH expression and release (1–4). In vertebrates, two GtHs have been identified: follicle-stimulating hormone (FSH) and luteinizing hormone (LH). Both GtHs are dimers comprised of a common α subunit and a distinct β subunit that confers their biological specificity. The two GtHs are produced in tetrapods by a single cell type, but play distinct roles in the regulation of gonadal processes. In the testis, FSH regulates Sertoli-cell activity and germ cell maturation whereas in females it induces germ cells and follicular growth. A LH surge is associated with ovulation in females and Leydig cell stimulation in males (5, 6).
Most experimental data in fish suggest that, as in tetrapods, FSH and LH play distinct roles in the regulation of gonadal development and function. In most species of fish, FSH is evident in the pituitary and in the plasma at early developmental stages and controls early stages of gametogenesis in both sexes. FSH is potent at inducing estrogen production by the ovary and its levels in the pituitary and plasma of females coincide with the process of vitellogenesis. LH levels are elevated during more advanced stages of gametogenesis in both males and females and induce secretion of estrogens, androgens, and maturation inducing steroids from the gonad (7, 8). In male fish, expression of the FSH receptor is not restricted to Sertoli cells and is also detected in the steroidogenic Leydig cells. This FSH-induced steroidogenesis probably regulates early stages of spermatogenesis, at a time when a LH stimulus is unavailable (9). In the ovary, both GtHs elicit estrogen production but in the mature follicles LH is more potent in inducing the production of the maturation inducing steroid 17α,20β-dihydroxy-4-pregnen-3-one (10).
A complex feedback of gonadal steroids tightly regulates GnRH receptor expression thereby affecting GtH expression and secretion. Estradiol is probably the most potent regulator of the hypothalamic–pituitary axis and generally considered to decrease lhb and fshb expression. In males, androgens play a similar role in attenuating GtH expression in the pituitary (11).
The HPG axis of fish bears striking resemblance to that of more evolved vertebrates, conserving all the major components and functions found in mammals (1, 8, 12–14). Because studies on the relationship between anatomy and function of the reproductive axis in mammals are often hindered by the inaccessibility of its components in developing and adult animals, fish models, with their unique advantages, are exceptionally valuable as a mean to enhance our understanding of the evolution of the axis and the interplay between its anatomy and function. When compared to other fish models, the zebrafish offers numerous advantages since it presents several distinct traits that make it particularly appropriate for this purpose: A large zebrafish research community has resulted in a solid knowledge base and an ever-growing array of zebrafish-related tools and resources, including methodologies, mutant lines (15), and transgenes (16). These, together with its inherent advantages such as ease of breeding and transgenesis, short generation time, transparency of embryonic stages, and so forth make zebrafish a leading choice for neuroendocrine research (13, 17).
To date several transgenic fish models with labeled GnRH neurons or gonadotrope cells have been introduced. GnRH neurons have been labeled in zebrafish (18) and medaka (19) whereas FSH was targeted in tilapia (20) and GtHs in medaka (21). In zebrafish, a line with labeled common α subunit-expressing cells was recently generated (22), but lines for identifying distinct LH and FSH producing cells have yet to be introduced.
In this study, we used regulatory elements from tilapia to drive fluorescent protein expression in zebrafish gonadotropes. Using these transgenic lines, we describe the ontogeny of GtH expression in this important model species, the anatomy of gonadotropes in the adult pituitary and demonstrate its value for testing the effects of GnRH and estrogens on GtH expression patterns.
Materials and Methods
Fish Husbandry and Breeding
Zebrafish were maintained in a stand-alone unit equipped with central filtration and heating (28 ± 1°C). The fish were fed twice daily with a commercial feed (New Life Spectrum “Grow,” New Life International Inc., Homestead, FL, USA). Breeding was performed by housing fish of both sexes in tanks with a mesh bottom. Eggs were collected in the morning and incubated until the yolk sac was completely absorbed, ca. 5 days postfertilization (dpf). Larvae were then transferred to brackish (6 ppt) water in stand-alone tanks and fed with rotifers (Brachionus plicatilis) until 10 dpf (23). At this age, larvae were transferred to fresh water and fed brine shrimp nauplii and dry, prepared diets. Treatment with 1-phenyl 2-thiourea was omitted in all experiments, because we observed developmental setbacks when using this treatment and because imaging quality was unaffected by the pigments. Under these conditions, fish usually reach sexual maturity at around 60 days of age.
All experimental procedures were in compliance with the Animal Care and Use Guidelines of the Hebrew University and were approved by the local Administrative Panel on Laboratory Animal Care.
Constructs and Transgenesis
The construct used for the labeling of FSH gonadotropes was as described previously (20). For the generation of labeled LH gonadotropes, we cloned a 3.6-kb fragment from tilapia genomic DNA (24) that includes a 3-kb fragment upstream of the first exon, first exon, and first intron (accession number KM575842). The primers (forward: 5′-GGGGACAA CTTTGTATAGAAAAGTTGGGCACTGAAGAAAAACGGTCCTTAA-3′; reverse: 5′-GGGGACTGCTTTTTTGTACAAACTTGGTCTGTAGGCGGCAAGTTGGA TTAGT-3′) included the appropriate attB4 and attB1R adaptors. The fragment was introduced into the pDestTol2CG2 destination clone through a LR Threeway Multisite Gateway reaction (Invitrogen, Carlsbad, CA, USA). The resulting construct drove mCherry expression in LH gonadotropes and EGFP expression in the heart. All Gateway methods and protocols were performed according to the Invitrogen Multisite Gateway Manual. For the purposes of transgenesis, eggs were collected immediately after spawning and injected with a combination of expression plasmid and transposase mRNA (16). After hatching embryos were screened for a signal in the heart and only positive embryos were grown and mated as possible founders. Tilapia transgenesis was performed as described previously (20).
Promoter Analysis
Upstream sequences of tilapia and zebrafish GtH genes were extracted from published databases (UCSC blat, https://genome.ucsc.edu). In silico analysis of the cis elements in the tilapia and zebrafish GtH promoters was performed using the Genomatix Software Suite.
In situ Hybridization, Immunofluorescence, and Imaging
To confirm correct expression of the transgene, fluorescent signals were compared to the in situ hybridization (ISH) staining pattern. ISH was generally performed as described previously (2–4). To detect the GtH mRNA, we cloned a fragment of the zebrafish GtH β subunit using the primers described by Ref. (25). The amplicon was cloned into the TOPO cloning vector (Invitrogen) and used as a template for the preparation of a specific digoxigenin (DIG)-labeled riboprobe (RNA DIG labeling kit, Roche Diagnostics GmbH, Mannheim, Germany). Adult fish heads were fixed overnight in 4% paraformaldehyde (PFA) at 4°C and then decalcified in 0.5 M EDTA at 4°C for 5 days. After cryoprotection [30% sucrose (w/v) in PBS overnight at 4°C] tissues were embedded in tissue freezing medium (Triangle Biomedical Sciences, Inc., Durham, NC, USA), flash frozen in liquid N2 and cryosectioned to 12 μm. Following ISH, the hybridization product was visualized using a fluorescent substrate (Fast Red, Roche). After confirmation of the hybridization signals, immunofluorescence (IF) labeling was performed against EGFP as previously detailed (20). Following staining, sections were mounted in anti-fade solution [2% propyl gallate (w/v), 75% glycerol (v/v) in PBS] and imaged using standard fluorescent microscopy. Since reliable mCherry antibodies were not available, the transgenic signal for this line was imaged before the ISH process. For validation of the correct expression of the LH:mCherry construct in tilapia, we applied IF using specific antibodies raised against tilapia GtH β-subunits (26, 27) and compared the two staining patterns. Sections were imaged using standard or confocal fluorescent microscopy.
For the ontogeny study, transgenic zebrafish from both lines were collected throughout their development from 4 to 65 dpf. Fish were fixed in 4% PFA overnight at 4°C. Young fish, until the age of 21 dpf were transparent enough to allow imaging of the pituitary in intact animals. For this purpose, fixed fish were cleared in 75% glycerol (v/v in PBS), mounted in anti-fade solution, and imaged ventrally on a confocal microscope. Older fish were processed for cryosectioning as described earlier and imaged using standard fluorescent microscopy.
Real-Time PCR
To assess the relative abundance of mRNAs, we used real-time PCR methodology. The genes were normalized to the amount of endogenous reference ef1a by the comparative threshold cycle method. Details of the method can be found elsewhere (28, 29).
In vivo Experiments
Larvae at the age of 10 dpf (n = 20) were placed in 50 ml water in petri dishes, containing the following treatments: control (10 μl ethanol in 50 ml), 100 or 1000 nM salmon GnRH analog (D-Ala6, Pro9-Net)-mammalian GnRH; sGnRHa; (Bachem, Inc., Torrance, CA, USA), 0.5, 5, or 50 ng/ml 17β-estradiol (E2; Sigma Ness Ziona, Israel), or 5 or 50 ng/ml of the aromatase inhibitor fadrozole (Novartis, Basel, Switzerland). In later experiments only a single dose of E2 (5 ng/ml) was used since the two other doses tested gave a similar response. Larvae were maintained in the treated water for 72 h. Every 24 h, all of the water in each dish was changed and treatments were added accordingly. Due to the transparency of zebrafish juveniles, we were able to image whole pituitaries in intact fish. For that purpose, at the end of the experimental period, larvae were fixed in 4% PFA and imaged ventrally using confocal microscopy. For gene-expression assays, total RNA was extracted from larvae heads using Trizol, reverse-transcribed into cDNA, as described previously (2, 29) and subjected to real-time PCR analysis. All experiments were repeated three times and a representative experiment is shown.
Statistical Analysis
Data are presented as mean ± SEM. Data were subjected to one-way ANOVA and Tukey analysis using GraphPad Prism 4 software (San Diego, CA, USA). Means marked with different letters differ significantly (P < 0.05).
Results
Gene Structure and Promoter Comparison
Since we chose to use tilapia promoters to drive fluorescent protein expression in zebrafish gonadotropes, we compared the structure and composition of the GtH genes between the two species. The FSHβ gene in both species is comprised of three exons (Figure 1A). Despite retention of this basic division, the varying sizes of the exons and introns result in a more compact gene in tilapia (~2.6 kb) than in zebrafish (~6 kb). This stems from elongation of the zebrafish introns as well as a long (~600 bp) 3′ untranslated region (UTR) situated within the third exon of the zebrafish gene. In both species, the first exon is relatively short and contains only the 5′ UTR. The structure of the lhb gene of zebrafish varies from that of tilapia in its exon/intron division: whereas the cyprinid genes (zebrafish carp and goldfish) are comprised of three exons, in tilapia the gene is divided into four segments. Four exons are also found in the genes of medaka and stickleback (Figure 1B). The coding region size is relatively conserved at ~140 amino acids. The zebrafish lhb gene contains a large 3′ UTR that is twice the length of the coding region.
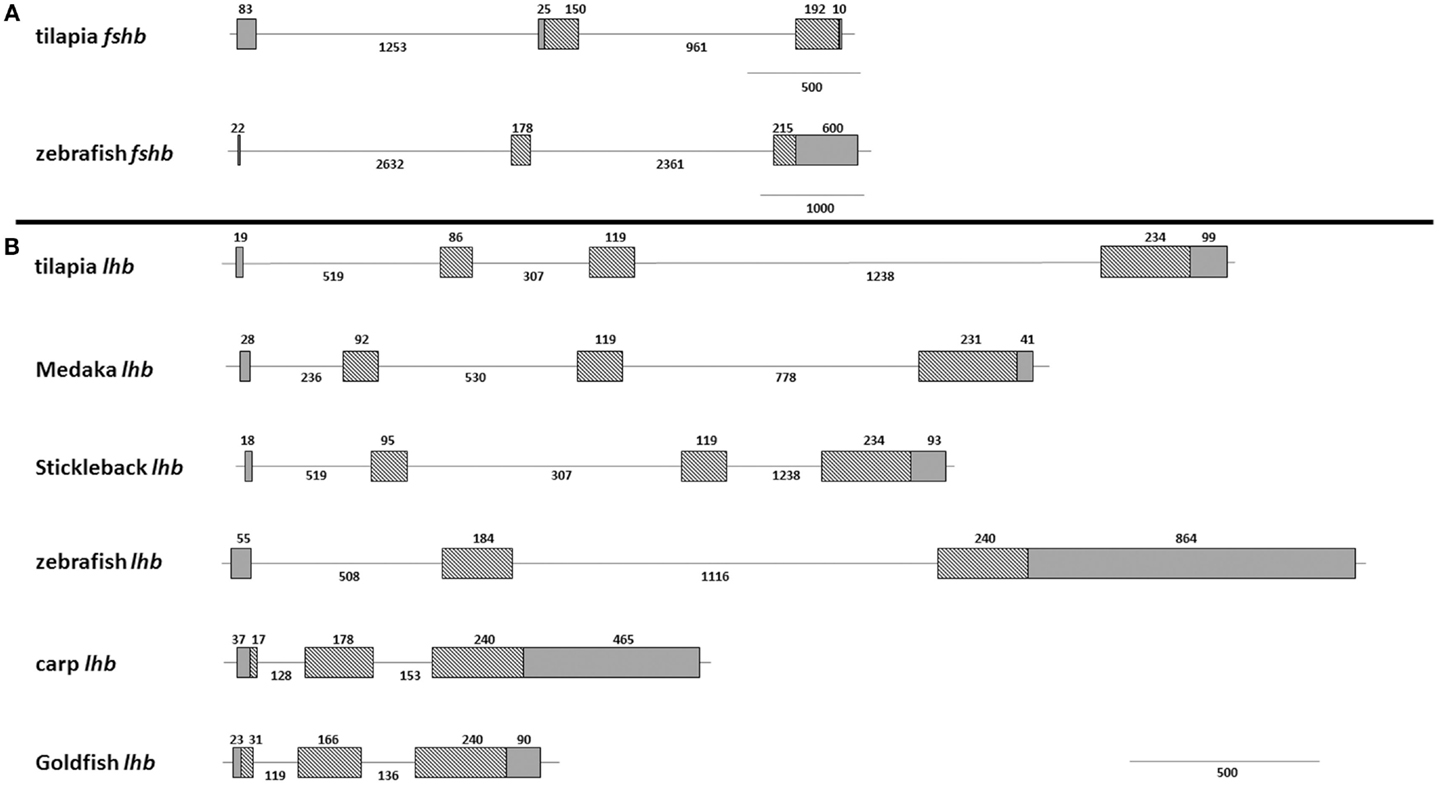
Figure 1. Gene structure of fish gonadotropins. Schematic overview of the exon–intron organization of several fish GtH genes. (A) The fshb genes of tilapia (AY294015.1 and JX887154.1) and zebrafish (NM_205624) are both divided into three exons but vary in intron size and length of untranslated regions. (B) The lhb genes of tilapia (XM_003438349.1) and zebrafish (NM_205622.2) differ in size and in exon–intron numbers. For comparison, medaka (EU_0477621) and stickleback (AJ_534969) genes contain four exons whereas carp (X_59889.1) and goldfish (30) genes are comprised of three exons. Grey boxes, untranslated regions; Hatched boxes, coding regions; lines, introns. Numbers indicate length of specified region in nucleotides.
The promoters of both fshb genes contain a TATA box 25 bp upstream of the transcription start site, although that of tilapia is a non-canonical variant that corresponds to the consensus sequence YYANWY (31). The tilapia promoter contains another consensus TATA sequence, at nucleotide position -95. The conserved smad- and GnRH-responsive steroidogenic factor 1 (SF1) element identified in goldfish (32) is also found in both tilapia and zebrafish proximal promoters and is adjacent to an estrogen-responsive site. Several other smad-binding sites and estrogen-responsive elements (EREs) can be found on both promoters. Pituitary-specific Pitx1 and Pit1 boxes can also be found on both promoters ~700 bp upstream of the transcription start site (Figure 2).
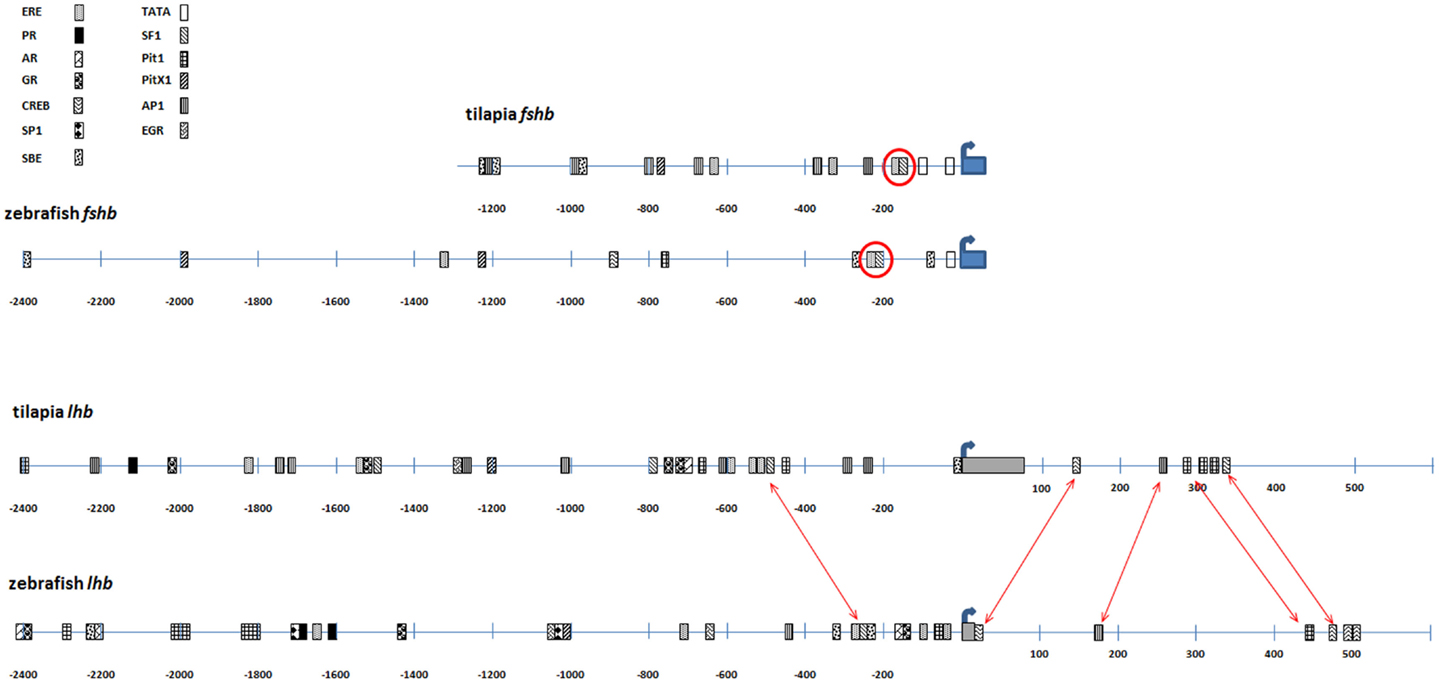
Figure 2. Comparison of regulatory elements of tilapia and zebrafish gonadotropin genes. The 5′ upstream sequence of tilapia and zebrafish GtHs was analyzed for identification of transcription factor-binding (TFB) sites. Upper panel: TFB map of the fshb promoters of tilapia and zebrafish. The common ERE/SF1 site is circled. Lower panel: TFB map of the lhb promoters of tilapia and zebrafish. This analysis also includes the first intron since common TFB sites were identified in the first introns of both species. Common TFB sites are indicated by arrows. Thick blue arrow indicates transcription start site. Gray box, first exon; ERE, estrogen-responsive element; PR, progesterone receptor-binding site; AR, androgen receptor-binding site; GR, glucocorticoid receptor-binding site; SBE, smad-binding element; EGR, early growth response-binding site.
The combination of adjacent SF1 and ERE boxes also appeared in both proximal lhb promoters. Early growth response elements were also found in the zebrafish and tilapia lhb promoters although their position was rather distal (−650 in zf and −1300 in tilapia). Both proximal lhb promoters also contained the pituitary-specific Pit1 element, as well as activator protein 1 (AP1)-binding sites and numerous EREs (Figure 2). Other steroid-responsive sites included androgen and glucocorticoid receptor-binding elements. These steroid-responsive elements were also present in the more distal part of both promoters (−1000 bp and upstream). Trials to label LH gonadotropes with tilapia constructs lacking the region upstream of position-600 failed, although stable genome integration was achieved (data not shown). This implies that some of the elements found in this distal region are mandatory for correct expression of the lhb subunit.
We also analyzed transcription factor-binding (TFB) sites in the first intron since they were found to be important in the regulation of gene expression (33) and were thus included in our constructs. The first introns of the fshb genes did not exhibit significant similarity in composition and order of the TFB sites, but in the lhb genes, both tilapia and zebrafish first introns showed similar composition and order of several TFB sites, including cAMP-responsive elements, AP1 and Pit1 sites (Figure 2).
Transgenes Validation
In tilapia, transgenic LH:mCherry individuals showed a high level of transgene expression in LH cells as evidenced by the colocalization of the mCherry signal in cells immunopositive for LHβ (Figures 3A–C). FSHβ immunoreactive cells represented a different subpopulation and did not express mCherry (Figures 3D–F). Both FSH:EGFP and LH:mCherry transgenic lines exhibited strong and clear expression of the fluorescent reporter in the zebrafish pituitary. Correct expression of the transgene in zebrafish was validated by comparing the expression patterns of the fluorescent reporter and that of GtH as revealed by ISH of the specific GtHβ-subunits (Figure 4). For the FSH:EGFP line, 94% (266 of 283) of the cells showed colocalization of the reporter signal and GtH expression, 3.5% (10 of 283) of the cells showed a GFP signal but no fshb expression (ISH), and 2.5% (7 of 283) of the cells showed fshb expression but no GFP signal detected by IF against GFP (Figures 4A–C). In the LH:mCherry line, the tight clustering of the cells, the unavailability of good antibodies against mCherry and the fact that the reporter tended to form aggregates inside the cells (34) made it difficult to determine the exact degree of overlap between the LHβ subunit and reporter expression. However, comparing the expression patterns of the two showed excellent colocalization of both signals (Figures 4D–F).
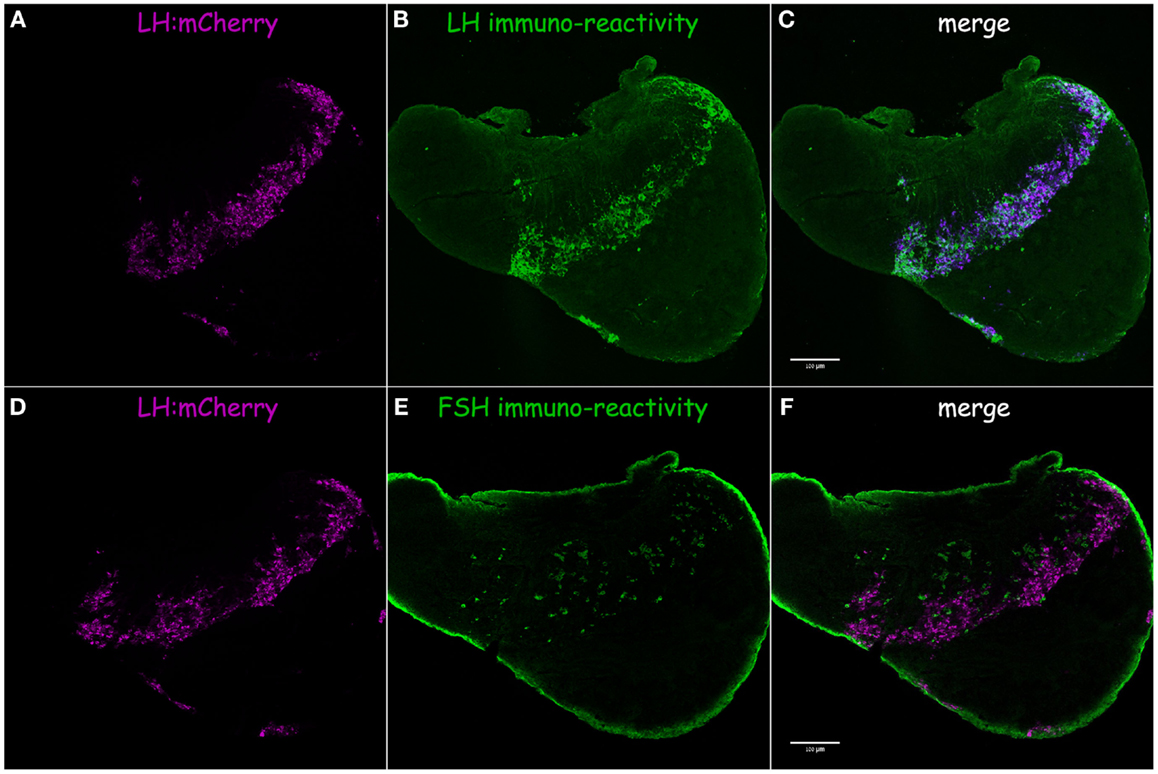
Figure 3. LH:mCherry transgene validation in tilapia. Correct expression of the fluorescent proteins in gonadotrope cells was verified by comparing the fluorescent protein expression pattern (magenta) to immunofluorescence staining using specific antibodies raised against tilapia GtH β-subunit (green). (A) LH:mCherry in the adult pituitary. (B) LHβ immunoreactivity. (C) Merge of A and B shows high level of colocalization of both signals. (D) LH:mCherry in the adult pituitary. (E) FSHβ immunoreactivity. (F) Merge of D and E shows FSH is expressed in a different subset of cells.
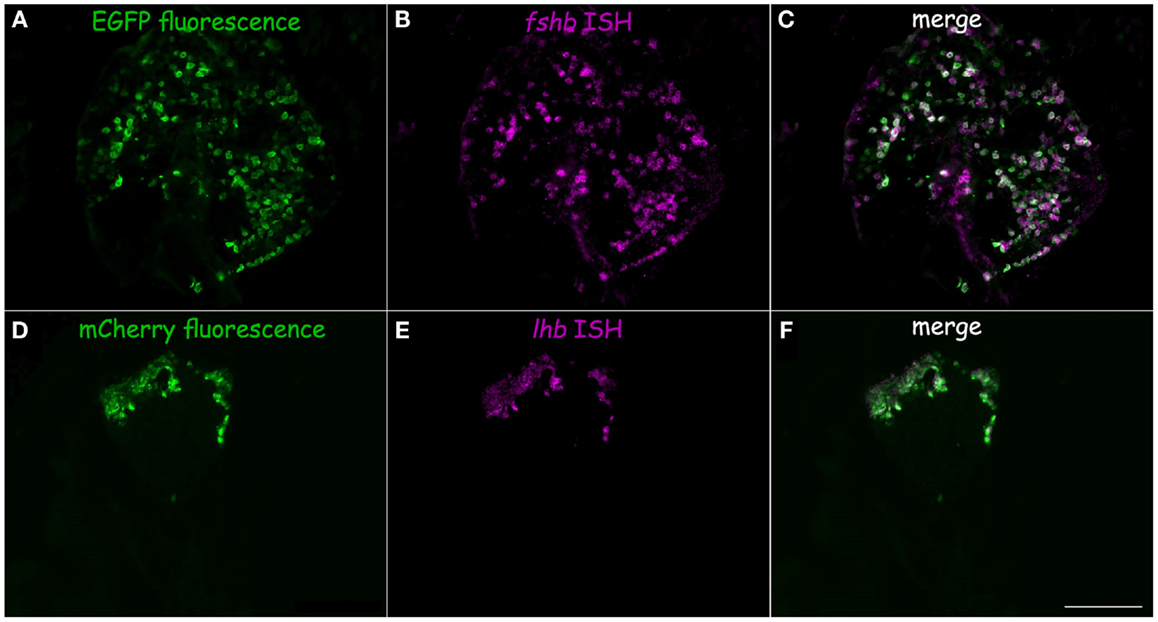
Figure 4. Transgene validation in zebrafish. Correct expression of the fluorescent proteins in gonadotrope cells was verified by comparing the fluorescent protein expression pattern (green) to fluorescent in situ hybridization using probes for GtH β-subunit mRNA (magenta). (A–C) FSH:EGFP line, (D–F): LH:mCherry line. (A) FSH:EGFP in the adult pituitary. (B) fshb mRNA expression. (C) Merge of A and B shows high level of colocalization of both signals. (D) LH:mCherry in the adult pituitary. (E) lhb mRNA expression. (F) Merge of (D) and (E) shows high level of colocalization of both signals. Scale – 100 μm.
In double-labeled zebrafish carrying both transgenes, the two cell types were easily identified and all strongly labeled cells exhibited only one type of reporter (Figure 5). However, in the ventral part of the gland, a population of cells with low-level of expression of both reporters could be identified (Figure 5D). These cells comprised <1.5% of the gonadotropes in the adult pituitary. No differences in expression patterns were found between adult males and females (Figure 5).
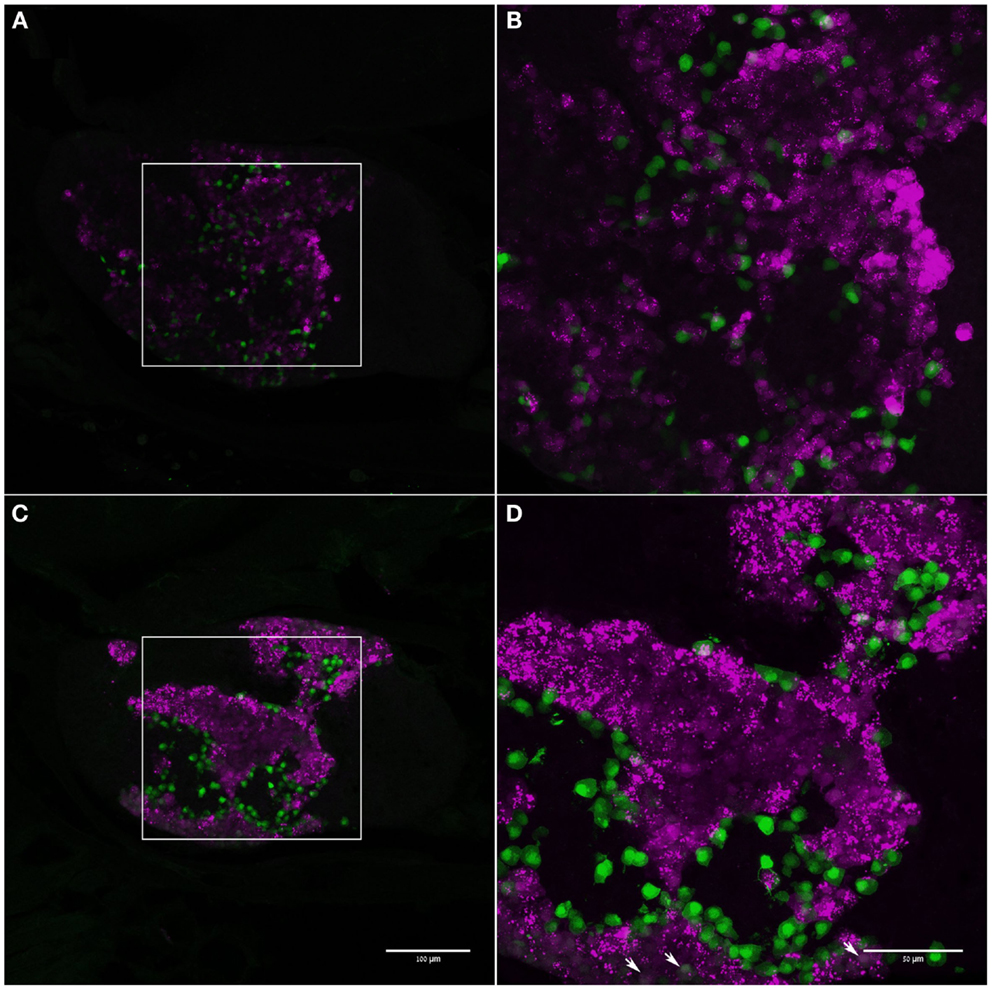
Figure 5. Distribution of LH and FSH gonadotropes in the pituitary of adult zebrafish. Adult male (A,B) and female (C,D) double-transgenic zebrafish expressing EGFP (green) in FSH gonadotropes and mCherry (magenta) in LH cells. Boxed areas in (A) and (C) mark the areas imaged in (B,D), respectively (anterior – left; dorsal – up). In both sexes, FSH cells are situated at the periphery of LH-cell clumps. Strongly labeled cells do not show colocalization of both signals. Some cells in the ventral part of the pituitary cells show low expression of both signals (arrows). Scale bars for (A,B) are the same as for (C,D), respectively.
Contrary to a previous report in medaka (35) identifying significant extra-pituitary expression of the LH transgene, we found the expression of our LH transgene to be highly specific and restricted only to the pituitary.
FSH Transgene Response to GnRH and Estradiol
Application of GnRH in the rearing water caused an increase in FSH cell number when larvae were exposed to concentrations of 100 and 1000 nM sGnRHa from 10 to 13 dpf. In control fish, 8.9 ± 1.12 cells were found in the pituitary at this stage whereas in fish treated with GnRH, the number of cells increased to 13.9 ± 0.5 and 17.6 ± 1.7 cells in the 100 and 1000 nM treatments, respectively. Exposure to estradiol caused a marked decrease in the number of FSH cells, from the basal levels of 8.9 to 3.4 ± 1.2, 2.6 ± 0.7, and 3.7 ± 0.6 cells per pituitary in the 0.5, 5, and 50 ng/ml treatments, respectively (Figure 6B, n = 8). Total fluorescence in the pituitary, composed of number of fluorescent cells as well as GFP intensity, was increased by GnRH (1.7 ± 0.35 and 2.4 ± 0.48 times the control for the 100 and 1000 nM GnRH treatments, respectively; n = 8) and aromatase inhibitor (1.3 ± 0.39 and 2.5 ± 0.15 times basal with 5 and 50 ng/ml fadrozole, respectively; n = 8), and dramatically reduced by exposure to estradiol (0.1 ± 0.04 relative to basal; n = 8) (Figures 6A,D). To determine whether these changes also reflected the actual levels of fshb, we measured the expression of fshb by real-time PCR. The fshb expression levels exhibited a pattern similar to that of the fluorescence intensity in the different treatments (1.1 ± 0.3, 1.7 ± 0.4, 0.14 ± 0.02, 1.5 ± 0.3, 1.6 ± 0.2 relative to control in the control, GnRH 100, GnRH 1000, E2 5, fadrozole 5, and fadrozole 50 treatments, respectively; n = 8; Figure 6C). A high correlation (r2 = 0.81; P = 0.015) was found between fshb expression and gonadotrope fluorescence intensity (Figure 6E). This correlation proves that the tilapia promoter not only correctly labels zebrafish FSH gonadotropes, but also conveys physiologically relevant signals to the transgene, thereby affecting its expression levels in the same way as these signals affect the endogenous gene.
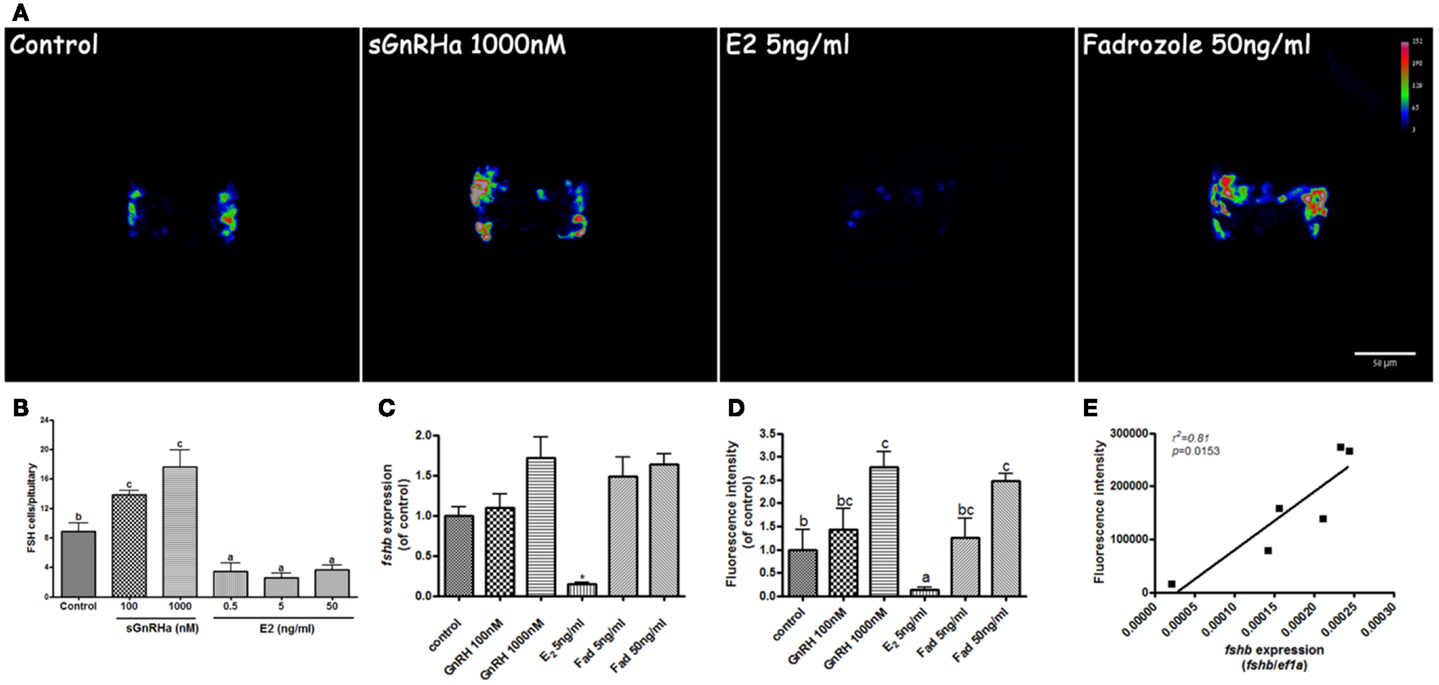
Figure 6. Effect of GnRH and estrogen on fshb and transgene expression. FSH:EGFP larvae at 10 days postfertilization were reared for 72 h in water containing sGnRHa (100 and 1000 nM), 17β-estradiol (E2, 0.5, 5, and 50 ng/ml) or the aromatase inhibitor fadrozole (5 and 50 ng/ml). (A) Representative heat map of the pituitary of treated fish at the end of the treatment (anterior – up). Color coding corresponds to EGFP signal intensity. GnRH and fadrozole treatment caused an increase in cell number and signal intensity. Estradiol decreased both parameters. (B) Quantification of the treatments effects on number of labeled cells per pituitary. GnRH increased and E2 decreased the number of fluorescent cells per pituitary. (C) Real-time quantification of the treatment effect on fshb gene expression also shows a significant decrease in the estradiol treatment. (D) Quantification of the treatment effect on fluorescent signal intensity shows an increase at the high doses of GnRH and fadrozole and a significant decrease in the estradiol treatment. (E) Correlation analysis of fshb expression and EGFP fluorescent signal intensity.
Ontogenic Expression of the Transgene
In the present study, FSH cells appeared as early as 4 dpf, when 5–6 cells per pituitary can be detected (Figure 7A). The number of FSH cells increased with age (Figure 7) to approximately 700 cells per pituitary in the adult. LH cells were completely absent during the early life stages and only appeared (8–10 cells) at around 28 dpf (Figure 7F), their number gradually increasing thereafter (Figure 7H). At sexual maturity (~60 dpf), LH gonadotropes outnumbered FSH cells by a factor of 4.5:1 (Figures 5 and 7I). This shift in gonadotrope population implies that FSH has an important role in the developmental stages of the reproductive system whereas the role of LH becomes dominant as puberty approaches, coinciding with the transition of the ovary from primary growth to vitellogenesis in females and spermatogenesis in males.
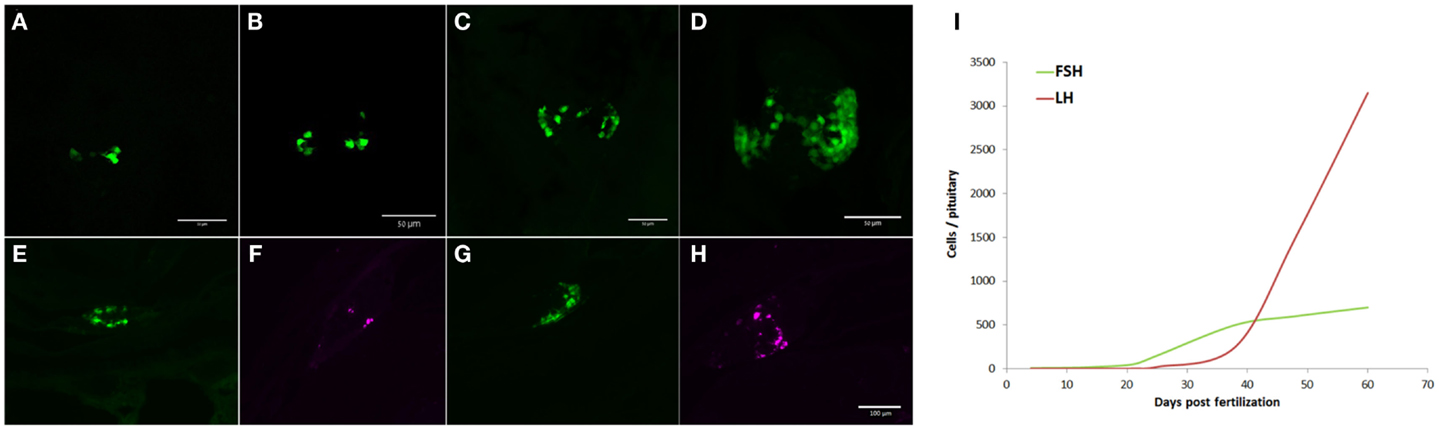
Figure 7. Ontogeny of gonadotrope cells in zebrafish. FSH (green) and LH (magenta) cell patterns were followed throughout development. (A–D) Proliferation of FSH cells during early development. At 4 dpf (A) 5–6 cells can be found in the pituitary. Cell number rises gradually as the fish develops [(B)-7 dpf; (C)-14 dpf; and (D)-21 dpf]. LH cells begin appearing at ~25 dpf (F) and increase in number toward puberty (H). FSH cells continue to proliferate with age [(E)-28 dpf and (G)-38 dpf]. Images (A–D) are ventral whole-mount z-stacks (anterior – up). Images (E–H) are 12-μm thick sagittal sections [anterior – left, common scale bar in (H)]. (I) Summary of gonadotrope population dynamics from 4 dpf to adulthood.
Discussion
In the current study, we introduce three new transgenic fish lines with fluorescently-labeled gonadotropes. The transgenic LH:mCherry tilapia joins our previously reported FSH:EGFP line (20) and completes the task of labeling gonadotropes in this emerging model species. The two zebrafish transgenic lines that were generated further enhance the value of this popular model for the study of reproductive biology.
Although the zebrafish and tilapia lineages are separated by over 250 million years of evolution (36), we could still use the promoter from tilapia to accurately drive reporter proteins in zebrafish gonadotropes. Comparison of the gene structures showed relatively high conservation in the fshb gene, but lhb subunits differed between the two species – the tilapia gene was comprised of four exons whereas the zebrafish had only three. The fourth exon can also be found in other advanced teleosts such as the medaka and stickleback. The zebrafish as well as salmon, carp, and goldfish (37) gene’s division into three exons is more reminiscent of the mammalian organization. Whereas the exon–intron division is a relatively well-conserved feature of closely related genes, addition or deletion of introns occurs at a frequency of ca. 1 intron/gene every 100 million years (38–40), and such events are common in many fish (41, 42). The fact that much of the other observed differences in mRNA size could be attributed to changes in the length of non-coding sequences, and not changes within the open reading frame, may also serve to explain the high tolerance to these significant insertions/deletions since these regions are less subject to functional constraints.
A low-level of conservation was even more salient in the gene promoters. An examination of the regulatory elements of both GtHs in tilapia and zebrafish showed very little resemblance in nucleotide sequence, and in the composition and order of the major TFB sites. Yet, the tilapia promoters drove correct reporter expression in zebrafish, suggesting that they contain enough conserved elements to enable the transgene to recruit the cell machinery in a gonadotrope-specific manner. This plasticity of the regulatory regions allows significant DNA-sequence divergence without loss of functionality (43). Our analysis showed that in the fshb promoters, a functional smad-response element identified in goldfish (32) also exists in both tilapia and zebrafish proximal promoters. In the distal promoters, adjacent AP1 and smad-binding element sites were also apparent in both species and found to be functional in goldfish (32). Apart from these sites, several estrogen-receptor- and SF-binding sites were present in the regulatory regions. The presence of these sites can explain the high responsiveness of the transgene to estrogen exposure. A strong response of fshb expression to estrogen exposure has also been observed in salmon (44) and mammals (45). Since GnRH is the key stimulator of fshb expression and proliferation (28, 46), it is not surprising that exposure of larvae to high doses of GnRH caused an increase in transgene and fshb expression. Because an in vivo model was used in this study, it is difficult to determine whether estrogen and GnRH exerted their effects directly, by binding to their cognate receptors on the gonadotrope, or indirectly by paracrine or autocrine effects – by affecting other regulatory pathways, which control gonadotrope proliferation and expression. Nevertheless, the fact that our transgene can react to external stimuli in a physiologically relevant context strengthens its value and reliability. Moreover, this model can be easily applied to test for the presence and physiological effect of estrogenic substances simply by exposing transgenic fish to potentially contaminated water and then directly quantifying fluorescence intensity of the gonadotropes.
The regulatory region of the lhb gene also showed little similarity in nucleotide sequence and transcription factor organization between tilapia and zebrafish. However, an ERE/SF1 complex that was found in the fshb promoters was also found in both lhb sequences. This combination plays an important role in inducing lhb transcription (47). Early growth response-binding elements are also vital for lhb gene expression (37) and can be found on both promoters in an intermediate position (tilapia at −1332; zebrafish at −707). A PitX1-binding site in the proximal promoter was found to be fundamental for the activation of the lhb gene in chinook salmon (48) and play an important role in mammalian lhb gene regulation (49). The additional Pitx1-binding site is present in the tilapia proximal lhb promoter, close to the ERE/SF1 element, but in the zebrafish, it is located further upstream at −1032. Many other EREs are located in the proximal and distal promoters of tilapia and zebrafish and may account for the reported effects of estrogens on lhb expression. In general, in most fish species examined estrogens had an overall increasing effect on lhb gene expression and secretion whereas the effect on FSH was less consistent (7). In sub-adult salmon, estrogens had a profound up-regulating effect on lhb and a downregulating effect on fshb gene expression (44). In tilapia, estradiol has been shown to decrease fshb and lhb expression in vivo and decrease effect of GnRH on GtH secretion (28). In primary cell cultures from the adult zebrafish pituitary, estrogen increased expression of both GtHs (50). Gonadectomy studies further support the notion of tight feedback mechanisms exerted by gonadal steroids on GtH expression and secretion levels. When gonads are removed pituitaries reacted to the decrease in gonadal steroid levels by increasing GtH production, dependent on the reproductive stage of the fish (51–53).
Introns are incorporated into many transgenic cassettes as a mean of increasing transgene expression in mammals (54–56), fish (57, 58), and invertebrates (59). The effect of introns on transgene performance can be attributed to the fact that the 5′ region (first 500–1000 nt from the splice site) of the first intron in many genes exhibits an exceptional degree of conservation and has been shown to contain functional regulatory elements (33). Since we included the first intron of both genes in our constructs, we also tested for similarities within these sequences. In the lhb introns, a specific sequence of putative binding motifs was found in both zebrafish and tilapia, although the motifs were more densely distributed in the tilapia, corresponding to the trend toward a more compact genome in tilapia than in zebrafish (42).
Our model shows good separation between the expression patterns of the two gonadotropes, evidenced by the fact that all clearly labeled cells expressed only one GtH, in both zebrafish and tilapia. However, very few cells in the ventral-most part of the proximal pars distalis showed low expression levels of both fluorescent proteins. While fish are generally considered to have complete separation of the two GtH types (8), there is some evidence of coexpression of both hormones within the same cells in the Mediterranean yellowtail (60). Another explanation relies on the concept that both LH and FSH are derived from the same lineage (61), and the fact that FSH gonadotropes begin appearing in this area and gradually migrate to their final, more dorsal position (25), thus marking the ventral pituitary as the gonadotrope’s “birthplace.” It is possible that these ventral cells represent a portion of the population that is going through phases of differentiation and may experience a bipotent stage before committing to their final roles.
The developmental pattern of FSH and LH found in our fish corresponds well with that described by Chen and Ge (25) for the same species. FSH was expressed at relatively early stages of development whereas the appearance of LH cells was highly correlated with the onset of puberty, as is the case for tilapia and mummichog (62, 63). This large gap in the temporal expression of FSH and LH differs from the situation in mammals, in which both GtHs are expressed at approximately the same time during development (64). The early expression of FSH may be related to the fact that at these initial stages, FSH seems to play developmental roles in processes other than reproduction, and depletion of its β-subunit mRNA causes severe developmental abnormalities (25). In accordance with the concept of a non-reproductive role for early FSH expression, we could not find any GnRH3 fibers reaching the pituitary until 7 dpf, and even at 14 dpf, some of the fish failed to exhibit GnRH axons in the pituitary (data not shown), although a significant number of FSH cells were present in the pituitary at that time. This observation is strengthened by the fact that in mammals, FSH cells are unresponsive to GnRH at early developmental stages and require LH signaling to begin expressing GnRH receptor (65). This activation by LH is apparently not necessary in fish, as in our model at 14 dpf the FSH cells were receptive to GnRH stimuli, as evidenced by the increase in fshb expression in response to GnRH application (despite a lack of LH at this stage).
Our transgenic zebrafish lines have been validated using accepted methodologies and the transgene response to estrogens and GnRH – the key modulators of GtH secretion – reflects the endogenous gene response patterns. Moreover, the transgene ontogeny profiles closely follow those described for this species. Nevertheless, it is important to note that the tilapia regulatory elements, operating within the zebrafish genomic environment, may respond differently from the endogenous zebrafish genes to some relevant stimuli. Factor-specific validation is therefore required when applying this model to investigate other aspects of GtH regulation.
In summary, we used regulatory elements from an evolutionary distant species to drive correct expression of transgenes in zebrafish gonadotropes. Our results highlight the functional conservation of highly diverged genomic regulatory regions. Our newly introduced transgenic zebrafish lines provide an important and powerful tool for investigating the differential development, anatomy, and function of the reproductive axis in vertebrates.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
The authors would like to thank Mrs. Naama Gruber for her help with the in silico analysis of promoter elements. Fadrozole was a kind gift from Novartis. The research was funded Binational Agricultural Research and Development Fund (BARD IS-4499-12) and by the Israel Science Foundation (ISF) nos. 1350/06 and 237/12.
References
1. Zohar Y, Munoz-Cueto JA, Elizur A, Kah O. Neuroendocrinology of reproduction in teleost fish. Gen Comp Endocrinol (2010) 165:438–55. doi: 10.1016/j.ygcen.2009.04.017
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
2. Biran J, Palevitch O, Ben-Dor S, Levavi-Sivan B. Neurokinin Bs and neurokinin B receptors in zebrafish-potential role in controlling fish reproduction. Proc Natl Acad Sci U S A (2012) 109:10269–74. doi:10.1073/pnas.1119165109
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
3. Biran J, Golan M, Mizrahi N, Ogawa S, Parhar SI, Levavi-Sivan B. Direct regulation of gonadotropin release by neurokinin B in tilapia (Oreochromis niloticus). Endocrinology (2014). doi:10.1210/en.2013-2114
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
4. Biran J, Golan M, Mizrahi N, Ogawa S, Parhar S I, Levavi-Sivan B. LPXRFa, the piscine ortholog of GnIH, and LPXRF-receptor positively regulate gonadotropin secretion in tilapia (Oreochromis niloticus). Endocrinology (2014) 155:4391–401. doi:10.1210/en.2013-2047
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
5. Hedger MP. Chapter 2 hypophyseal-gonadal relationships in the male. In: Bittar EE, Neville Edward B, editors. Principles of Medical Biology. Elsevier (1998). p. 25–55.
6. Mroueh J, Danforth DR. Chapter 3 hypophyseal-ovarian relationships. In: Bittar EE, Neville Edward B, editors. Principles of Medical Biology. Elsevier (1998). p. 57–75.
7. Yaron Z, Gur G, Melamed P, Rosenfeld H, Elizur A, Levavi-Sivan B. Regulation of fish gonadotropins. Int Rev Cytol (2003) 225:131–85. doi:10.1016/S0074-7696(05)25004-0
8. Levavi-Sivan B, Bogerd J, Mananos EL, Gomez A, Lareyre JJ. Perspectives on fish gonadotropins and their receptors. Gen Comp Endocrinol (2010) 165:412–37. doi:10.1016/j.ygcen.2009.07.019
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
9. Schulz RW, De Franca LR, Lareyre JJ, Le Gac F, Chiarini-Garcia H, Nobrega RH, et al. Spermatogenesis in fish. Gen Comp Endocrinol (2010) 165:390–411. doi:10.1016/j.ygcen.2009.02.013
10. Aizen J, Kobayashi M, Selicharova I, Sohn YC, Yoshizaki G, Levavi-Sivan B. Steroidogenic response of carp ovaries to piscine FSH and LH depends on the reproductive phase. Gen Comp Endocrinol (2012) 178:28–36. doi:10.1016/j.ygcen.2012.04.002
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
11. Schulz RW, Vischer HF, Cavaco JE, Santos EM, Tyler CR, Goos HJ, et al. Gonadotropins, their receptors, and the regulation of testicular functions in fish. Comp Biochem Physiol B Biochem Mol Biol (2001) 129:407–17. doi:10.1016/S1096-4959(01)00339-6
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
12. McGonnell IM, Fowkes RC. Fishing for gene function – endocrine modelling in the zebrafish. J Endocrinol (2006) 189:425–39. doi:10.1677/joe.1.06683
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
13. Lieschke GJ, Currie PD. Animal models of human disease: zebrafish swim into view. Nat Rev Genet (2007) 8:353–67. doi:10.1038/nrg2091
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
14. Lohr H, Hammerschmidt M. Zebrafish in endocrine systems: recent advances and implications for human disease. Annu Rev Physiol (2011) 73:183–211. doi:10.1146/annurev-physiol-012110-142320
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
15. Gagnon JA, Valen E, Thyme SB, Huang P, Ahkmetova L, Pauli A, et al. Efficient mutagenesis by Cas9 protein-mediated oligonucleotide insertion and large-scale assessment of single-guide RNAs. PLoS One (2014) 9:e98186. doi:10.1371/journal.pone.0098186
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
16. Kwan KM, Fujimoto E, Grabher C, Mangum BD, Hardy ME, Campbell DS, et al. The Tol2kit: a multisite gateway-based construction kit for Tol2 transposon transgenesis constructs. Dev Dyn (2007) 236:3088–99. doi:10.1002/dvdy.21343
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
17. Friedrich RW, Jacobson GA, Zhu P. Circuit neuroscience in zebrafish. Curr Biol (2010) 20:R371–81. doi:10.1016/j.cub.2010.02.039
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
18. Abraham E, Palevitch O, Ijiri S, Du SJ, Gothilf Y, Zohar Y. Early development of forebrain gonadotrophin-releasing hormone (GnRH) neurones and the role of GnRH as an autocrine migration factor. J Neuroendocrinol (2008) 20:394–405. doi:10.1111/j.1365-2826.2008.01654.x
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
19. Karigo T, Kanda S, Takahashi A, Abe H, Okubo K, Oka Y. Time-of-day-dependent changes in GnRH1 neuronal activities and gonadotropin mRNA expression in a daily spawning fish, medaka. Endocrinology (2012) 153:3394–404. doi:10.1210/en.2011-2022
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
20. Golan M, Levavi-Sivan B. Social dominance in tilapia is associated with gonadotroph hyperplasia. Gen Comp Endocrinol (2013) 192:126–35. doi:10.1016/j.ygcen.2013.04.032
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
21. Karigo T, Aikawa M, Kondo C, Abe H, Kanda S, Oka Y. Whole brain-pituitary in vitro preparation of the transgenic medaka (Oryzias latipes) as a tool for analyzing the differential regulatory mechanisms of LH and FSH release. Endocrinology (2014) 155:536–47. doi:10.1210/en.2013-1642
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
22. Cheng X, Chen X, Jin X, He J, Yin Z. Generation and characterization of gsuα:EGFP transgenic zebrafish for evaluating endocrine-disrupting effects. Toxicol Appl Pharmacol (2014) 278:78–84. doi:10.1016/j.taap.2014.04.009
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
23. Best J, Adatto I, Cockington J, James A, Lawrence C. A novel method for rearing first-feeding larval zebrafish: polyculture with type L saltwater rotifers (Brachionus plicatilis). Zebrafish (2010) 7:289–95. doi:10.1089/zeb.2010.0667
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
24. Brawand D, Wagner CE, Li YI, Malinsky M, Keller I, Fan S, et al. The genomic substrate for adaptive radiation in African cichlid fish. Nature (2014) 513:375–81. doi:10.1038/nature13726
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
25. Chen W, Ge W. Ontogenic expression profiles of gonadotropins (fshb and lhb) and growth hormone (gh) during sexual differentiation and puberty onset in female zebrafish. Biol Reprod (2012) 86:73. doi:10.1095/biolreprod.111.094730
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
26. Kasuto H, Levavi-Sivan B. Production of biologically active tethered tilapia LHβα by the methylotrophic yeast Pichia pastoris. Gen Comp Endocrinol (2005) 140:222–32. doi:10.1016/j.ygcen.2004.10.016
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
27. Aizen J, Kasuto H, Levavi-Sivan B. Development of specific enzyme-linked immunosorbent assay for determining LH and FSH levels in tilapia, using recombinant gonadotropins. Gen Comp Endocrinol (2007) 153:323–32. doi:10.1016/j.ygcen.2007.04.004
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
28. Levavi-Sivan B, Biran J, Fireman E. Sex steroids are involved in the regulation of gonadotropin-releasing hormone and dopamine D2 receptors in female tilapia pituitary. Biol Reprod (2006) 75:642–50. doi:10.1095/biolreprod.106.051540
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
29. Biran J, Ben-Dor S, Levavi-Sivan B. Molecular identification and functional characterization of the kisspeptin/kisspeptin receptor system in lower vertebrates. Biol Reprod (2008) 79:776–86. doi:10.1095/biolreprod.107.066266
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
30. Sohn YC, Yoshiura Y, Suetake H, Kobayashi M, Aida K. Nucleotide sequence of gonadotropin II beta subunit gene in goldfish. Fish Sci. (1999) 65:800–1.
31. Yang C, Bolotin E, Jiang T, Sladek FM, Martinez E. Prevalence of the initiator over the TATA box in human and yeast genes and identification of DNA motifs enriched in human TATA-less core promoters. Gene (2007) 389:52–65. doi:10.1016/j.gene.2006.09.029
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
32. Lau MT, Lin SW, Ge W. Identification of smad response elements in the promoter of goldfish FSHbeta gene and evidence for their mediation of activin and GnRH stimulation of FSHbeta expression. Front Endocrinol (2012) 3:47. doi:10.3389/fendo.2012.00047
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
33. Majewski J, Ott J. Distribution and characterization of regulatory elements in the human genome. Genome Res (2002) 12:1827–36. doi:10.1101/gr.606402
34. Davidson MW, Campbell RE. Engineered fluorescent proteins: innovations and applications. Nat Methods (2009) 6:713–7. doi:10.1038/nmeth1009-713
35. Hildahl J, Sandvik GK, Lifjeld R, Hodne K, Nagahama Y, Haug TM, et al. Developmental tracing of luteinizing hormone beta-subunit gene expression using green fluorescent protein transgenic medaka (Oryzias latipes) reveals a putative novel developmental function. Dev Dyn (2012) 241:1665–77. doi:10.1002/dvdy.23860
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
36. Hedges SB, Dudley J, Kumar S. TimeTree: a public knowledge-base of divergence times among organisms. Bioinformatics (2006) 22:2971–2. doi:10.1093/bioinformatics/btl505
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
37. Chong KL, Koh M, Melamed P. Hormones and their Receptors in Fish Reproduction. Singapore: World Scientific Publishing Co. Pvt. Ltd (2005).
38. Stein LD, Bao Z, Blasiar D, Blumenthal T, Brent MR, Chen N, et al. The genome sequence of Caenorhabditis briggsae: a platform for comparative genomics. PLoS Biol (2003) 1:E45. doi:10.1371/journal.pbio.0000045
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
39. Coghlan A, Wolfe KH. Origins of recently gained introns in Caenorhabditis. Proc Natl Acad Sci U S A (2004) 101:11362–7. doi:10.1073/pnas.0308192101
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
40. Yandell M, Mungall CJ, Smith C, Prochnik S, Kaminker J, Hartzell G, et al. Large-scale trends in the evolution of gene structures within 11 animal genomes. PLoS Comput Biol (2006) 2:e15. doi:10.1371/journal.pcbi.0020015
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
41. Venkatesh B, Ning Y, Brenner S. Late changes in spliceosomal introns define clades in vertebrate evolution. Proc Natl Acad Sci U S A (1999) 96:10267–71. doi:10.1073/pnas.96.18.10267
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
42. Kumar A, Bhandari A, Sinha R, Goyal P, Grapputo A. Spliceosomal intron insertions in genome compacted ray-finned fishes as evident from phylogeny of MC receptors, also supported by a few other GPCRs. PLoS One (2011) 6:e22046. doi:10.1371/journal.pone.0022046
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
43. Taher L, Mcgaughey DM, Maragh S, Aneas I, Bessling SL, Miller W, et al. Genome-wide identification of conserved regulatory function in diverged sequences. Genome Res (2011) 21:1139–49. doi:10.1101/gr.119016.110
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
44. Harding LB, Schultz IR, Goetz GW, Luckenbach JA, Young G, Goetz FW, et al. High-throughput sequencing and pathway analysis reveal alteration of the pituitary transcriptome by 17alpha-ethynylestradiol (EE2) in female coho salmon, Oncorhynchus kisutch. Aquat Toxicol (2013) 14(2–143):146–63. doi:10.1016/j.aquatox.2013.07.020
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
45. Baratta M, West LA, Turzillo AM, Nett TM. Activin modulates differential effects of estradiol on synthesis and secretion of follicle-stimulating hormone in ovine pituitary cells. Biol Reprod (2001) 64:714–9. doi:10.1095/biolreprod64.2.714
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
46. Bernard DJ, Fortin J, Wang Y, Lamba P. Mechanisms of FSH synthesis: what we know, what we don’t, and why you should care. Fertil Steril (2010) 93:2465–85. doi:10.1016/j.fertnstert.2010.03.034
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
47. Le Drean Y, Liu D, Xiong F, Hew CL. Presence of distinct cis-acting elements on gonadotropin gene promoters in diverse species dictates the selective recruitment of different transcription factors by steroidogenic factor-1. Mol Cell Endocrinol (1997) 135:31–40.
48. Melamed P, Koh M, Preklathan P, Bei L, Hew C. Multiple mechanisms for Pitx-1 transactivation of a luteinizing hormone beta subunit gene. J Biol Chem (2002) 277:26200–7. doi:10.1074/jbc.M201605200
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
49. Quirk CC, Lozada KL, Keri RA, Nilson JH. A single Pitx1 binding site is essential for activity of the LHbeta promoter in transgenic mice. Mol Endocrinol (2001) 15:734–46. doi:10.1210/mend.15.5.0628
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
50. Lin S-W, Ge W. Differential regulation of gonadotropins (FSH and LH) and growth hormone (GH) by neuroendocrine, endocrine, and paracrine factors in the zebrafish – an in vitro approach. Gen Comp Endocrinol (2009) 160:183–93. doi:10.1016/j.ygcen.2008.11.020
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
51. Larsen DA, Swanson P. Effects of gonadectomy on plasma gonadotropins I and II in coho salmon, Oncorhynchus kisutch. Gen Comp Endocrinol (1997) 108:152–60. doi:10.1006/gcen.1997.6958
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
52. Borg B, Antonopoulou E, Mayer I, Andersson E, Berglund I, Swanson P. Effects of gonadectomy and androgen treatments on pituitary and plasma levels of gonadotropins in mature male Atlantic salmon, Salmo salar, parr – positive feedback control of both gonadotropins. Biol Reprod (1998) 58:814–20. doi:10.1095/biolreprod58.3.814
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
53. Khan IA, Hawkins MB, Thomas P. Gonadal stage-dependent effects of gonadal steroids on gonadotropin II secretion in the Atlantic croaker (Micropogonias undulatus). Biol Reprod (1999) 61:834–41. doi:10.1095/biolreprod61.3.834
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
54. Brinster RL, Allen JM, Behringer RR, Gelinas RE, Palmiter RD. Introns increase transcriptional efficiency in transgenic mice. Proc Natl Acad Sci U S A (1988) 85:836–40. doi:10.1073/pnas.85.3.836
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
55. Palmiter RD, Sandgren EP, Avarbock MR, Allen DD, Brinster RL. Heterologous introns can enhance expression of transgenes in mice. Proc Natl Acad Sci U S A (1991) 88:478–82. doi:10.1073/pnas.88.2.478
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
56. Furger A, O’sullivan JM, Binnie A, Lee BA, Proudfoot NJ. Promoter proximal splice sites enhance transcription. Genes Dev (2002) 16:2792–9. doi:10.1101/gad.983602
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
57. Moav B, Liu Z, Caldovic LD, Gross ML, Faras AJ, Hackett PB. Regulation of expression of transgenes in developing fish. Transgenic Res (1993) 2:153–61. doi:10.1007/BF01972609
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
58. Rocha A, Ruiz S, Estepa A, Coll JM. Application of inducible and targeted gene strategies to produce transgenic fish: a review. Mar Biotechnol (NY) (2004) 6:118–27. doi:10.1007/s10126-003-0013-9
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
59. Duncker BP, Davies PL, Walker VK. Introns boost transgene expression in Drosophila melanogaster. Mol Gen Genet (1997) 254:291–6. doi:10.1007/s004380050418
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
60. Pilar Garcia Hernandez M, Garcia Ayala A, Zandbergen MA, Agulleiro B. Investigation into the duality of gonadotropic cells of Mediterranean yellowtail (Seriola dumerilii, Risso 1810): immunocytochemical and ultrastructural studies. Gen Comp Endocrinol (2002) 128:25–35. doi:10.1016/S0016-6480(02)00052-7
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
61. Pulichino AM, Vallette-Kasic S, Tsai JP, Couture C, Gauthier Y, Drouin J. Tpit determines alternate fates during pituitary cell differentiation. Genes Dev (2003) 17:738–47. doi:10.1101/gad.1065703
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
62. Shimizu A, Hamaguchi M, Ito H, Ohkubo M, Udagawa M, Fujii K, et al. Appearances and chronological changes of mummichog Fundulus heteroclitus FSH cells and LH cells during ontogeny, sexual differentiation, and gonadal development. Gen Comp Endocrinol (2008) 156:312–22. doi:10.1016/j.ygcen.2008.01.022
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
63. Yan H, Ijiri S, Wu Q, Kobayashi T, Li S, Nakaseko T, et al. Expression patterns of gonadotropin hormones and their receptors during early sexual differentiation in Nile tilapia Oreochromis niloticus. Biol Reprod (2012) 87:116. doi:10.1095/biolreprod.112.101220
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
64. Japon MA, Rubinstein M, Low MJ. In situ hybridization analysis of anterior pituitary hormone gene expression during fetal mouse development. J Histochem Cytochem (1994) 42:1117–25. doi:10.1177/42.8.8027530
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
65. Wen S, Ai W, Alim Z, Boehm U. Embryonic gonadotropin-releasing hormone signaling is necessary for maturation of the male reproductive axis. Proc Natl Acad Sci U S A (2010) 107:16372–7. doi:10.1073/pnas.1000423107
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Keywords: zebrafish, tilapia, gonadotropin, transgene, LH, FSH
Citation: Golan M, Biran J and Levavi-Sivan B (2014) A novel model for development, organization, and function of gonadotropes in fish pituitary. Front. Endocrinol. 5:182. doi: 10.3389/fendo.2014.00182
Received: 16 September 2014; Accepted: 08 October 2014;
Published online: 22 October 2014.
Edited by:
P. Michael Conn, Texas Tech University Health Sciences Center, USAReviewed by:
Gwen V. Childs, University of Arkansas for Medical Sciences, USAWilliam Huckle, Virginia Tech, USA
Copyright: © 2014 Golan, Biran and Levavi-Sivan. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Berta Levavi-Sivan, Department of Animal Sciences, The Robert H. Smith Faculty of Agriculture, Food and Environment, The Hebrew University, P.O. Box 12, Rehovot 76100, Israel e-mail:YmVydGEuc2l2YW5AbWFpbC5odWppLmFjLmls
 Matan Golan
Matan Golan Jakob Biran
Jakob Biran Berta Levavi-Sivan
Berta Levavi-Sivan