- 1First Propedeutic Department of Internal Medicine, Medical School, Aristotle University of Thessaloniki, AHEPA Hospital, Thessaloniki, Greece
- 2Second Department of Obstetrics and Gynecology, Medical School, Aristotle University of Thessaloniki, Hippokration Hospital, Thessaloniki, Greece
Assisted reproduction, including in vitro fertilization and intracytoplasmic sperm injection, is increasingly being used for the management of infertility in patients with polycystic ovary syndrome (PCOS). However, there are limited data regarding the association between obesity and the outcome of assisted reproduction in this specific population as well as on the effects of weight loss. The aim of the present review is to summarize the existing evidence on the association between obesity and the outcome of assisted reproduction in patients with PCOS. Accumulating data suggest that obesity is associated with lower pregnancy and live birth rates in patients with PCOS who are undergoing assisted reproduction therapy. However, it remains unclear whether weight loss improves the outcome of this therapy. Notably, recent guidelines state that the health benefits of postponing pregnancy to achieve weight loss must be balanced against the risk of declining fertility with advancing age. Therefore, if weight loss is not achieved within a reasonable time period, assisted reproduction therapy should be offered in adequately selected patients with PCOS, regardless of the presence of obesity.
Introduction
In recent decades, the prevalence of obesity has increased globally and reached pandemic proportions (1). In high-income countries, approximately one-third of adults are obese and one-third are overweight (1). This rise in the prevalence of obesity has important health-care implications, since obesity is an important risk factor for cardiovascular disease and all-cause mortality (2, 3).
In addition to its cardiometabolic sequelae, obesity is implicated in the pathogenesis of polycystic ovary syndrome (PCOS) (4, 5). Approximately 40–70% of patients with this syndrome are either overweight or obese (4–6). The prevalence of PCOS is also almost four times higher in overweight and obese patients than in lean subjects (7). However, other studies reported that obesity only minimally increases the risk of PCOS (4, 8). Moreover, it has been reported that PCOS might also increase the risk of obesity, potentially by a reduction in basal metabolic rate and an impairment in appetite regulation (9–11).
Polycystic ovary syndrome is the commonest endocrine disorder in women of reproductive age (8, 12, 13) and the leading cause of anovulatory infertility (14). Several studies showed that obese patients with PCOS have more impaired ovulation and lower pregnancy rates than normal-weight patients with this syndrome (15–17).
Assisted reproduction, including in vitro fertilization (IVF) and intracytoplasmic sperm injection (ICSI), is increasingly being used for the management of infertility in patients with PCOS (14). However, there are limited data regarding the association between obesity and the outcome of assisted reproduction in this specific population as well as on the effects of weight loss. The aim of the present review is to summarize the existing evidence on the association between obesity and the outcome of assisted reproduction in patients with PCOS.
Effects of Obesity on the Outcome of Assisted Reproduction in Patients with PCOS
A number of studies evaluated the association between obesity and the outcome of assisted reproduction in patients with PCOS (Table 1). However, most studies were small and retrospective. Moreover, most studies included patients with various causes of fertility and did not perform separate analyses of patients with PCOS.
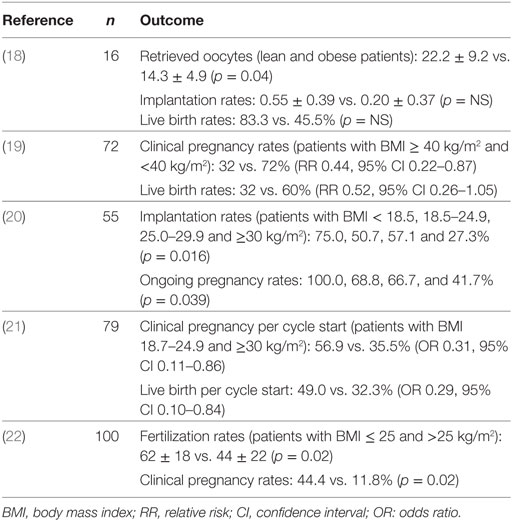
Table 1. Studies evaluating the association between obesity and the outcome of assisted reproduction in patients with polycystic ovary syndrome.
In a small retrospective study (n = 16), lean patients with PCOS had less retrieved oocytes than obese patients with PCOS, even though they required fewer gonadotropin ampoules (18). In contrast, implantation, clinical pregnancy and live birth rates did not differ between the two groups, possibly due to the small number of patients (18). Indeed, in a larger retrospective study (n = 72), patients with PCOS and body mass index (BMI) ≥ 40 kg/m2 had significantly lower clinical pregnancy rates after IVF than patients with PCOS and lower BMI (32 vs. 72%, respectively) and a trend for lower live birth rates (32 vs. 60%, respectively) despite the absence of difference in number of embryos transferred and implantation rates between the two groups (19). Moreover, the former required higher doses of gonadotropin and had fewer oocytes retrieved than the latter (19). In another study that included 55 patients with PCOS, implantation and pregnancy rates declined with increasing BMI (20). More specifically, implantation rates in patients with BMI < 18.5, 18.5–24.9, 25.0–29.9, and ≥30 kg/m2 were 75.0, 50.7, 57.1, and 27.3%, respectively (p = 0.016) whereas ongoing pregnancy rates were 100.0, 68.8, 66.7, and 41.7%, respectively (p = 0.039) (20). Notably, the total gonadotropin dose and the number of oocytes retrieved did not differ between the different BMI categories (20).
In a retrospective study in 79 patients with PCOS undergoing IVF, obese patients with PCOS had 69% lower odds of clinical pregnancy per cycle start than patients with PCOS and normal BMI (p = 0.02) and 77% lower odds of clinical pregnancy per embryo transfer (p = 0.008) (21). In addition, the odds of live birth were 71% lower in obese patients per cycle start (p = 0.02) and 77% lower per embryo transfer (p = 0.01) (21). On the other hand, a trend for lower incidence of ovarian hyperstimulation syndrome (OHSS) was observed (19.6, 10.5, and 3.2% in normal weight, overweight, and obese patients, respectively) (21).
In the largest study performed specifically in patients with PCOS (n = 100), patients with PCOS and BMI > 25 kg/m2 undergoing IVF had lower fertilization rates than patients with PCOS and BMI ≤ 25 kg/m2 undergoing IVF (44 ± 22 vs. 62 ± 18, respectively) and also had lower clinical pregnancy rates (11.8 vs. 44.4%, respectively) (22). Interestingly, these differences were present in both patients undergoing ovarian stimulation with gonadotrophin-releasing hormone (GnRH) agonist and in those undergoing stimulation with GnRH antagonist (22). Of note, GnRH agonists prevent premature luteinizing hormone (LH) surge, thereby increasing the number of retrieved oocytes and pregnancy rates and decreasing the number of cycle cancelations (23). However, they might increase the risk for OHSS (23). On the other hand, GnRH antagonists can competitively block GnRH receptors and cause rapid suppression of gonadotropin release, resulting in fewer complications but appear to be less effective than GnRH agonists (23). Notably, follicle-stimulating hormone (FSH) preparations might offer a more physiologic approach in patients with PCOS, since the LH/FSH ratio is frequently elevated in this population (24).
There are very limited data on the association between obesity and the outcomes of ICSI in patients with PCOS. In a small study in 56 patients with PCOS undergoing IVF or ICSI, obesity was independently related to a lower oocyte count and increased FSH requirement (25). However, pregnancy and live birth rates were not reported and outcomes were not reported separately in patients receiving IVF or ICSI (25).
Effects of Weight Loss on the Outcome of Assisted Reproduction in Patients with PCOS
Several small (n = 18–67) and uncontrolled studies showed that lifestyle-induced weight loss restores ovulation in patients with PCOS (26–29). Weight loss also increased spontaneous pregnancy rates in these studies (27, 28). Case series also suggested that patients with PCOS undergoing bariatric surgery were able to conceive postoperatively (30, 31).
There are very few randomized controlled study (RCTs) that evaluated the effects of weight loss on the outcome of assisted reproduction in patients with PCOS. An early RCT in 38 patients with various causes of infertility showed no benefit of diet and exercise-induced weight loss on pregnancy and live birth rates (32). However, weight loss was modest (3.8 kg) and the change in waist circumference was similar in patients who implemented lifestyle changes and in controls (32). In contrast, a more recent and larger RCT (n = 49) reported higher pregnancy and live birth rates in patients with various causes of infertility who lost approximately 6.6 kg with diet, exercise, and behavioral modification (33). Notably, fewer ART cycles were required to achieve these higher rates in patients who lost weight (33). Nevertheless, neither of these studies reported the effects of weight loss in the subgroup of patients with PCOS (32, 33).
In a secondary analysis of the pregnancy in PPCOS II trial (n = 187) and the treatment of hyperandrogenism versus insulin resistance in infertile PCOS (OWL PCOS) trial (n = 142), lifestyle modification (caloric restriction, antiobesity medication, behavioral modification, and exercise) followed by treatment with clomiphene resulted in higher rates of ovulation and live birth compared with immediate treatment with clomiphene (risk ratio 1.4 and 2.5, respectively) (34). Of note, clomiphene is the treatment of first choice for induction of ovulation in women with PCOS (35, 36). Clomiphene induces ovulation through its anti-estrogen action, which results in a change in GnRH pulse frequency, release of FSH from the anterior pituitary and consequent follicular development (24). Treatment with clomiphene results in ovulation in 75–80% of patients and increases the likelihood of live birth approximately six times more than placebo and three times more than metformin (35–38).
The promising findings of the latter studies were not confirmed in the largest RCT that evaluated the role of weight loss in infertile patients receiving assisted reproduction treatment. In this study, 577 infertile women (201 with PCOS) were randomized to receive a 6-month lifestyle-intervention program followed by 18 months of infertility treatment or prompt infertility treatment for 24 months (39). The primary outcome (vaginal birth of a healthy singleton at term within 24 months after randomization) occurred in a smaller percentage of the women who followed a lifestyle program than in those who received prompt infertility treatment (27.1 vs. 35.2%, respectively; rate ratio 0.77, 95% confidence interval 0.60–0.99, p = 0.04) (39). However, weight loss was rather small (4.4 kg) and only 37.7% of patients randomized to lifestyle changes lost >5% of their body weight (39). Moreover, a considerable proportion of patients in the lifestyle arm discontinued treatment (21.8%) (39). Importantly, rates of pregnancy resulting from natural conception were higher in women assigned lifestyle changes (26.1 vs. 16.2% in patients assigned prompt infertility treatment) (39). Accordingly, the use of ovulation induction or other infertility treatment was less frequent and the number of infertility treatment cycles was lower in the lifestyle arm (39). Subgroup analyses among the 201 PCOS patients that were included in this study were not reported (39). However, among women with anovulatory infertility (n = 269), the rates of the primary outcome, live birth, and ongoing pregnancy did not differ between the intervention and control group (39).
Conclusion
Accumulating data suggest that obesity is associated with lower pregnancy and live birth rates in patients with PCOS who are undergoing assisted reproduction therapy. However, it remains unclear whether weight loss improves the outcome of this therapy. Notably, the American Society for Reproductive Medicine recently concluded that the health benefits of postponing pregnancy to achieve weight loss must be balanced against the risk of declining fertility with advancing age (40). Therefore, if weight loss is not achieved within a reasonable time period, assisted reproduction therapy should be offered in adequately selected patients with PCOS, regardless of the presence of obesity.
Author Contributions
KT drafted the mini review. KD critically revised the draft.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
1. Ng M, Fleming T, Robinson M, Thomson B, Graetz N, Margono C, et al. Global, regional, and national prevalence of overweight and obesity in children and adults during 1980-2013: a systematic analysis for the Global Burden of Disease study 2013. Lancet (2014) 384:766–81. doi:10.1016/S0140-6736(14)60460-8
2. Global Burden of Metabolic Risk Factors for Chronic Diseases Collaboration (BMI Mediated Effects), Lu Y, Hajifathalian K, Ezzati M, Woodward M, Rimm EB, et al. Metabolic mediators of the effects of body-mass index, overweight, and obesity on coronary heart disease and stroke: a pooled analysis of 97 prospective cohorts with 1·8 million participants. Lancet (2014) 383:970–83. doi:10.1016/S0140-6736(13)61836-X
3. Global BMI Mortality Collaboration, Di Angelantonio E, Bhupathiraju ShN, Wormser D, Gao P, Kaptoge S, et al. Body-mass index and all-cause mortality: individual-participant-data meta-analysis of 239 prospective studies in four continents. Lancet (2016) 388:776–86. doi:10.1016/S0140-6736(16)30175-1
4. Yildiz BO, Knochenhauer ES, Azziz R. Impact of obesity on the risk for polycystic ovary syndrome. J Clin Endocrinol Metab (2008) 93:162–8. doi:10.1210/jc.2007-1834
5. Carmina E, Bucchieri S, Esposito A, Del Puente A, Mansueto P, Orio F, et al. Abdominal fat quantity and distribution in women with polycystic ovary syndrome and extent of its relation to insulin resistance. J Clin Endocrinol Metab (2007) 92:2500–5. doi:10.1210/jc.2006-2725
6. Panidis D, Macut D, Tziomalos K, Papadakis E, Mikhailidis K, Kandaraki EA, et al. Prevalence of metabolic syndrome in women with polycystic ovary syndrome. Clin Endocrinol (Oxf) (2013) 78:586–92. doi:10.1111/cen.12008
7. Alvarez-Blasco F, Botella-Carretero JI, San Millán JL, Escobar-Morreale HF. Prevalence and characteristics of the polycystic ovary syndrome in overweight and obese women. Arch Intern Med (2006) 166:2081–6. doi:10.1001/archinte.166.19.2081
8. Diamanti-Kandarakis E, Kouli CR, Bergiele AT, Filandra FA, Tsianateli TC, Spina GG, et al. A survey of the polycystic ovary syndrome in the Greek island of Lesbos: hormonal and metabolic profile. J Clin Endocrinol Metab (1999) 84:4006–11. doi:10.1210/jcem.84.11.6148
9. Georgopoulos NA, Saltamavros AD, Vervita V, Karkoulias K, Adonakis G, Decavalas G, et al. Basal metabolic rate is decreased in women with polycystic ovary syndrome and biochemical hyperandrogenemia and is associated with insulin resistance. Fertil Steril (2009) 92:250–5. doi:10.1016/j.fertnstert.2008.04.067
10. Hirschberg AL, Naessén S, Stridsberg M, Byström B, Holtet J. Impaired cholecystokinin secretion and disturbed appetite regulation in women with polycystic ovary syndrome. Gynecol Endocrinol (2004) 19:79–87. doi:10.1080/09513590400002300
11. Moran LJ, Noakes M, Clifton PM, Wittert GA, Tomlinson L, Galletly C, et al. Ghrelin and measures of satiety are altered in polycystic ovary syndrome but not differentially affected by diet composition. J Clin Endocrinol Metab (2004) 89:3337–44. doi:10.1210/jc.2003-031583
12. Knochenhauer ES, Key TJ, Kahsar-Miller M, Waggoner W, Boots LR, Azziz R. Prevalence of the polycystic ovary syndrome in unselected black and white women of the southeastern United States: a prospective study. J Clin Endocrinol Metab (1998) 83:3078–82. doi:10.1210/jcem.83.9.5090
13. Bozdag G, Mumusoglu S, Zengin D, Karabulut E, Yildiz BO. The prevalence and phenotypic features of polycystic ovary syndrome: a systematic review and meta-analysis. Hum Reprod (2016) 31:2841–55. doi:10.1093/humrep/dew218
14. Balen AH, Morley LC, Misso M, Franks S, Legro RS, Wijeyaratne CN, et al. The management of anovulatory infertility in women with polycystic ovary syndrome: an analysis of the evidence to support the development of global WHO guidance. Hum Reprod Update (2016) 22:687–708. doi:10.1093/humupd/dmw025
15. Kiddy DS, Sharp PS, White DM, Scanlon MF, Mason HD, Bray CS, et al. Differences in clinical and endocrine features between obese and non-obese subjects with polycystic ovary syndrome: an analysis of 263 consecutive cases. Clin Endocrinol (Oxf) (1990) 32:213–20. doi:10.1111/j.1365-2265.1990.tb00857.x
16. Balen AH, Conway GS, Kaltsas G, Techatrasak K, Manning PJ, West C, et al. Polycystic ovary syndrome: the spectrum of the disorder in 1741 patients. Hum Reprod (1995) 10:2107–11. doi:10.1093/oxfordjournals.humrep.a136243
17. Strowitzki T, Capp E, von Eye Corleta H. The degree of cycle irregularity correlates with the grade of endocrine and metabolic disorders in PCOS patients. Eur J Obstet Gynecol Reprod Biol (2010) 149:178–81. doi:10.1016/j.ejogrb.2009.12.024
18. McCormick B, Thomas M, Maxwell R, Williams D, Aubuchon M. Effects of polycystic ovarian syndrome on in vitro fertilization-embryo transfer outcomes are influenced by body mass index. Fertil Steril (2008) 90:2304–9. doi:10.1016/j.fertnstert.2007.10.077
19. Jungheim ES, Lanzendorf SE, Odem RR, Moley KH, Chang AS, Ratts VS. Morbid obesity is associated with lower clinical pregnancy rates after in vitro fertilization in women with polycystic ovary syndrome. Fertil Steril (2009) 92:256–61. doi:10.1016/j.fertnstert.2008.04.063
20. Martinuzzi K, Ryan S, Luna M, Copperman AB. Elevated body mass index (BMI) does not adversely affect in vitro fertilization outcome in young women. J Assist Reprod Genet (2008) 25:169–75. doi:10.1007/s10815-008-9213-6
21. Bailey AP, Hawkins LK, Missmer SA, Correia KF, Yanushpolsky EH. Effect of body mass index on in vitro fertilization outcomes in women with polycystic ovary syndrome. Am J Obstet Gynecol (2014) 211:e1–6. doi:10.1016/j.ajog.2014.03.035
22. Orvieto R, Nahum R, Meltcer S, Homburg R, Rabinson J, Anteby EY, et al. Ovarian stimulation in polycystic ovary syndrome patients: the role of body mass index. Reprod Biomed Online (2009) 18:333–6. doi:10.1016/S1472-6483(10)60090-9
23. Lambalk CB, Banga FR, Huirne JA, Toftager M, Pinborg A, Homburg R, et al. GnRH antagonist versus long agonist protocols in IVF: a systematic review and meta-analysis accounting for patient type. Hum Reprod Update (2017) 23:560–79. doi:10.1093/humupd/dmx017
24. Legro RS. Ovulation induction in polycystic ovary syndrome: current options. Best Pract Res Clin Obstet Gynaecol (2016) 37:152–9. doi:10.1016/j.bpobgyn.2016.08.001
25. Fedorcsák P, Dale PO, Storeng R, Tanbo T, Abyholm T. The impact of obesity and insulin resistance on the outcome of IVF or ICSI in women with polycystic ovarian syndrome. Hum Reprod (2001) 16:1086–91. doi:10.1093/humrep/16.6.1086
26. Clark AM, Ledger W, Galletly C, Tomlinson L, Blaney F, Wang X, et al. Weight loss results in significant improvement in pregnancy and ovulation rates in anovulatory obese women. Hum Reprod (1995) 10:2705–12. doi:10.1093/oxfordjournals.humrep.a135772
27. Clark AM, Thornley B, Tomlinson L, Galletley C, Norman RJ. Weight loss in obese infertile women results in improvement in reproductive outcome for all forms of fertility treatment. Hum Reprod (1998) 13:1502–5. doi:10.1093/humrep/13.6.1502
28. Huber-Buchholz MM, Carey DG, Norman RJ. Restoration of reproductive potential by lifestyle modification in obese polycystic ovary syndrome: role of insulin sensitivity and luteinizing hormone. J Clin Endocrinol Metab (1999) 84:1470–4. doi:10.1210/jcem.84.4.5596
29. Kuchenbecker WK, Groen H, van Asselt SJ, Bolster JH, Zwerver J, Slart RH, et al. In women with polycystic ovary syndrome and obesity, loss of intra-abdominal fat is associated with resumption of ovulation. Hum Reprod (2011) 26:2505–12. doi:10.1093/humrep/der229
30. Jamal M, Gunay Y, Capper A, Eid A, Heitshusen D, Samuel I. Roux-en-Y gastric bypass ameliorates polycystic ovary syndrome and dramatically improves conception rates: a 9-year analysis. Surg Obes Relat Dis (2012) 8:440–4. doi:10.1016/j.soard.2011.09.022
31. Eid GM, Cottam DR, Velcu LM, Mattar SG, Korytkowski MT, Gosman G, et al. Effective treatment of polycystic ovarian syndrome with Roux-en-Y gastric bypass. Surg Obes Relat Dis (2005) 1:77–80. doi:10.1016/j.soard.2005.02.008
32. Moran L, Tsagareli V, Norman R, Noakes M. Diet and IVF pilot study: short-term weight loss improves pregnancy rates in overweight/obese women undertaking IVF. Aust N Z J Obstet Gynaecol (2011) 51:455–9. doi:10.1111/j.1479-828X.2011.01343.x
33. Sim KA, Dezarnaulds GM, Denyer GS, Skilton MR, Caterson ID. Weight loss improves reproductive outcomes in obese women undergoing fertility treatment: a randomized controlled trial. Clin Obes (2014) 4:61–8. doi:10.1111/cob.12048
34. Legro RS, Dodson WC, Kunselman AR, Stetter CM, Kris-Etherton PM, Williams NI, et al. Benefit of delayed fertility therapy with preconception weight loss over immediate therapy in obese women with PCOS. J Clin Endocrinol Metab (2016) 101:2658–66. doi:10.1210/jc.2016-1659
35. Conway G, Dewailly D, Diamanti-Kandarakis E, Escobar-Morreale HF, Franks S, Gambineri A, et al. The polycystic ovary syndrome: a position statement from the European Society of Endocrinology. Eur J Endocrinol (2014) 171:1–29. doi:10.1530/EJE-14-0253
36. Goodman NF, Cobin RH, Futterweit W, Glueck JS, Legro RS, Carmina E. American Association of Clinical Endocrinologists (AACE); American College of Endocrinology (ACE); Androgen Excess and PCOS Society. American Association of Clinical Endocrinologists, American College of Endocrinology, and Androgen Excess and PCOS Society disease state clinical review: guide to the best practices in the evaluation and treatment of polycystic ovary syndrome—part 2. Endocr Pract (2015) 21:1415–26. doi:10.4158/EP15748.DSCPT2
37. Brown J, Farquhar C. Clomiphene and other antioestrogens for ovulation induction in polycystic ovarian syndrome. Cochrane Database Syst Rev (2016) 12:CD002249. doi:10.1002/14651858.CD002249.pub5
38. Legro RS, Barnhart HX, Schlaff WD, Carr BR, Diamond MP, Carson SA, et al. Cooperative multicenter reproductive medicine network. clomiphene, metformin, or both for infertility in the polycystic ovary syndrome. N Engl J Med (2007) 356:551–66. doi:10.1056/NEJMoa063971
39. Mutsaerts MA, van Oers AM, Groen H, Burggraaff JM, Kuchenbecker WK, Perquin DA, et al. Randomized trial of a lifestyle program in obese infertile women. N Engl J Med (2016) 374:1942–53. doi:10.1056/NEJMoa1505297
Keywords: obesity, polycystic ovary syndrome, weight loss, assisted reproduction, in vitro fertilization
Citation: Tziomalos K and Dinas K (2018) Obesity and Outcome of Assisted Reproduction in Patients With Polycystic Ovary Syndrome. Front. Endocrinol. 9:149. doi: 10.3389/fendo.2018.00149
Received: 16 December 2017; Accepted: 20 March 2018;
Published: 04 April 2018
Edited by:
Katherine Samaras, St Vincent’s Hospital Sydney, AustraliaReviewed by:
K. L. Hoehn, University of New South Wales, AustraliaTsegaselassie Workalemahu, Harvard School of Public Health, United States
Copyright: © 2018 Tziomalos and Dinas. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Konstantinos Tziomalos, a3R6aW9tYWxvc0B5YWhvby5jb20=
 Konstantinos Tziomalos
Konstantinos Tziomalos Konstantinos Dinas
Konstantinos Dinas