- 1Department of Epidemiology, School of Public Health, Cheeloo College of Medicine, Shandong University, Jinan, China
- 2Department of Nutrition and Food Hygiene, School of Public Health, Cheeloo College of Medicine, Shandong University, Jinan, China
- 3Center for Primary Care and Public Health (Unisanté), University of Lausanne, Lausanne, Switzerland
Because of the limitation of body mass index (BMI) in distinguishing adipose mass from muscle, the tri-ponderal mass index (TMI) has been proposed as a new indicator for better assessing adiposity in children and adolescents. However, it remains unclear whether TMI performs better than BMI or other adiposity indices in predicting obesity status in childhood and obesity-related cardiovascular risk factors (CVRFs) in childhood or adulthood. We searched PubMed, Cochrane Library, and Web of Science for eligible publications until June 15, 2021. A total of 32 eligible studies were included in this systematic review. We found that TMI had a similar or better ability to predict body fat among children and adolescents than BMI. However, most of the included studies suggested that TMI was similar to BMI in identifying metabolic syndrome although TMI was suggested to be a useful tool when used in combination with other indicators (e.g., BMI and waist circumference). In addition, limited evidence showed that TMI did not perform better than BMI for identifying specific CVRFs, including insulin resistance, high blood pressure, dyslipidemia, and inflammation in children and adolescents, as well as CVRFs in adults.
Systematic Review Registration: https://www.crd.york.ac.uk/prospero, CRD42021260356.
Introduction
The age-standardized prevalence of obesity among children and adolescents aged 5 to 19 years has globally increased from 0.7% in 1975 to 5.6% in 2016 among girls and 0.9% to 7.8% among boys (1). Obesity in children is a cause of several detrimental health outcomes in childhood and later in adulthood, such as left ventricular hypertrophy (2), increased carotid intima-media thickness (3), kidney disease (4), and liver disease (5), cancer, cardiovascular diseases, and death (6–8). Thus, an early and accurate diagnosis of obesity in children and adolescents is urgently needed, in order to reduce the short-term and long-term burden of pediatric obesity-related health outcomes.
Body mass index (BMI, kg/m2) is the most widely used physical indicator of adiposity in both children (with overweight/obesity cutoffs based on age and sex percentiles) and among adults (overweight: BMI 25–29; obesity BMI ≥ 30). Although BMI is strongly correlated with adiposity, the indicator cannot distinguish well between excess weight due to increased fat mass or increased muscle mass (9), especially for changes in body composition during adolescence, leading to weight increase being out of proportion of the change in height squared (10, 11). Although the percentage of body fat is suggested as an accurate method for identifying obesity in children and adolescents, it is less applicable for routine health care, as well as in school-based settings (12).
Tri-ponderal mass index (TMI), calculated as weight (kg)/height (m3), is an emerging indicator, which has been suggested to predict percent body fat (10) and metabolic syndrome (MetS) (13) at least as well as, or better than BMI. However, findings in other previous studies were inconsistent (14–17). For instance, the prevalence of overweight and obesity was higher when identified with BMI (based on standard deviation score, SDS) than with TMI in children and adolescents aged 6–17 years (14). It was also found that BMI (or BMI z-score or BMI-SDS) predicted MetS better than TMI among adolescents aged 10–17 years (15, 16).
It is however unclear whether the emerging TMI can better identify adiposity in childhood or adolescence than the commonly used BMI (10, 14, 18–26) and better predict obesity-related cardiovascular risk factors (CVRFs) such as high blood pressure, dyslipidemia, insulin resistance, and the MetS in childhood (13–16, 18, 21, 26–34) or adulthood (35–37). The misclassification of obesity may lead to either omissions of children who are at high risk of obesity-related diseases or excessive anxiety due to overdiagnosis and then unnecessary waste of medical resources (38). Identifying potential adiposity-related indicators that can accurately predict body fat or related risks has significant implications for prevention, treatment, and management of pediatric obesity.
Therefore, in order to assess whether TMI can be a substitute for BMI in routine pediatric clinical practice to estimate obesity and related CVRFs in children and adolescents or adults, we reviewed articles on the ability of TMI to identify increased body fat, in children and adolescents, and to predict CVRFs in both childhood and adulthood.
Methods
Search Strategy
This review was performed according to the recommendation from the Preferred Reporting Items for Systematic Reviews and Meta-Analyses statement (PRISMA) (http://www.prisma-statement.org/). We searched relevant articles in PubMed, Cochrane Library, and Web of Science until June 15, 2021 using the following search strategy: (“Triponderal mass index” OR “Tri-ponderal mass index” OR “Tri-ponderal index”) AND (“children” OR “childhood” OR “adolescents” OR “adolescence” OR “teenagers” OR “youth” OR “students”) AND (“body mass index” OR “obesity” OR “body fat” OR “cardiovascular disease risk” OR “hypertension” OR “dyslipidemia” OR “insulin resistance” OR “impaired glucose” OR “metabolic syndrome” OR “MetS” OR “inflammation”). We also identified eligible papers from the lists of references in the identified papers. We have registered on PROSPERO (available at: https://www.crd.york.ac.uk/prospero/#aboutpage), and the ID is CRD42021260356.
Inclusion Criteria and Exclusion Criteria
Inclusion criteria were as follows: 1) original article; 2) body fat or adiposity assessed using TMI and BMI in childhood or adolescence; 3) the paper described the association of TMI and BMI measured in childhood and adult with selected CVRFs [i.e., hypertension; dyslipidemia; insulin resistance (IR) or impaired glucose; MetS; and inflammation] measured either in childhood (e.g., at the same time of measurement of the BMI/TMI, e.g., in cross-sectional surveys) or in adulthood (e.g., cohort studies) or both; and 4) cross-sectional, cohort, or retrospective studies. Exclusion criteria were as follows: 1) obviously irrelevant articles; 2) TMI measured in adulthood; 3) other languages rather than English; 4) letter or comment; and 5) studies without data of interest.
Identification of Relevant Studies and Data Extraction
Two independent authors (JS and RY) performed the literature search and extracted the data. In case of disagreement between the two authors, a third expert (BX) was consulted to reach an agreement. The information on the first author, publication year, country of origin, study design, sample size, age and sex distribution of the study population, exposures, outcome definition, adjusted covariates, and results was extracted from each eligible study.
Study Quality Assessment
An 11-item checklist of the cross-sectional study evaluation scale recommended by the Agency for Healthcare Research and Quality (AHRQ) was used to evaluate the quality of cross-sectional studies (39), with answers coded as “Yes” (1) or “No or not clear” (0). The total score of the scale is 11 points. A score of 8 to 11 points is rated as high quality, 4 to 7 points as moderate quality, and less than 4 points as low quality. The Newcastle–Ottawa Scale (NOS) star system (range 0 to 9 stars) was used to evaluate the quality of cohort and case–control studies (40). In brief, four items related to the selection of participants, two items to the comparability of participants, and three items to the exposures or outcomes. A score of “0–3” was regarded as low quality, “4–6” as moderate quality, and “7–9” as high quality.
Results
Study Selection
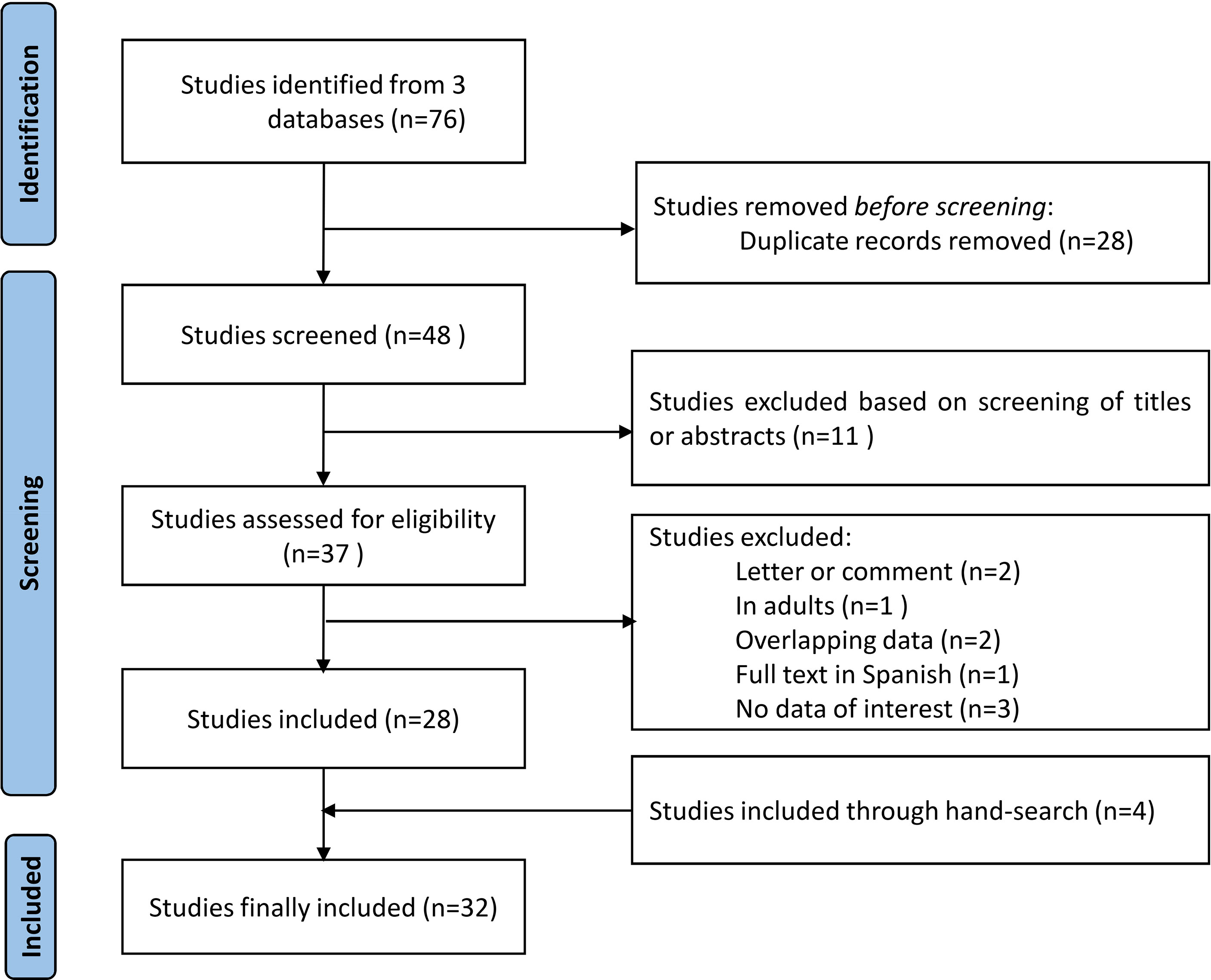
A total of 76 articles were initially identified. After excluding 28 duplicate articles, 48 remained for screening. After excluding 11 irrelevant articles, 2 letters/editorials, 1 in adults, 1 with overlapping data, 1 in Spanish, and 3 with no data of interest, 28 relevant studies were included. In addition, 4 additional articles were identified from the lists of references, resulting in a total of 32 articles eligible for the final systematic review. The detailed PRISMA flowchart of inclusion/exclusion of potential publications is presented in Figure 1.
Study Characteristics
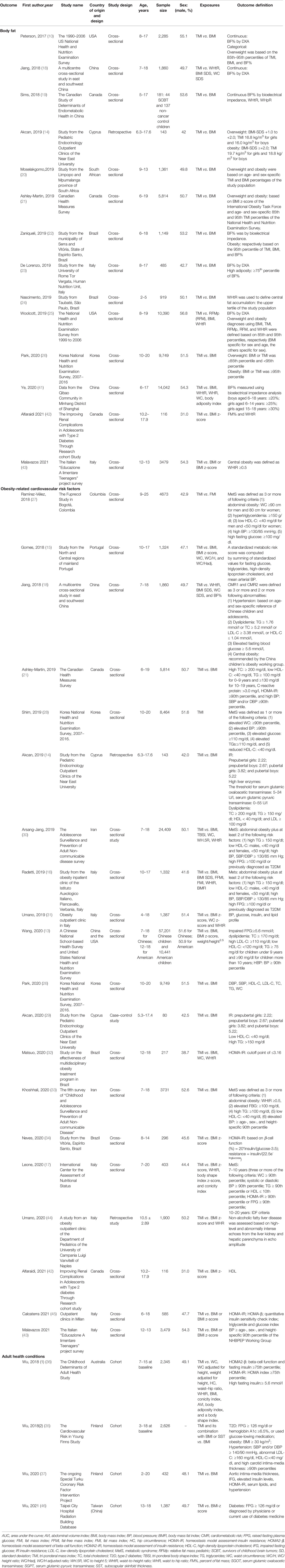
Table 1 describes the characteristics of the included studies including 14 for the association between TMI and body fat in childhood and adolescence (10, 14, 18–26, 41–43), 20 for TMI and CVRFs in childhood and adolescence (13–18, 21, 26–34, 42–45), and 4 for the association in adulthood (35–37, 46). Twenty-five of the included studies used a cross-sectional design (10, 13, 15–28, 30–34, 41–43, 45), 2 a retrospective design (14, 44), 1 a case-control design (29), and 4 a cohort design (35–37, 46).
Results
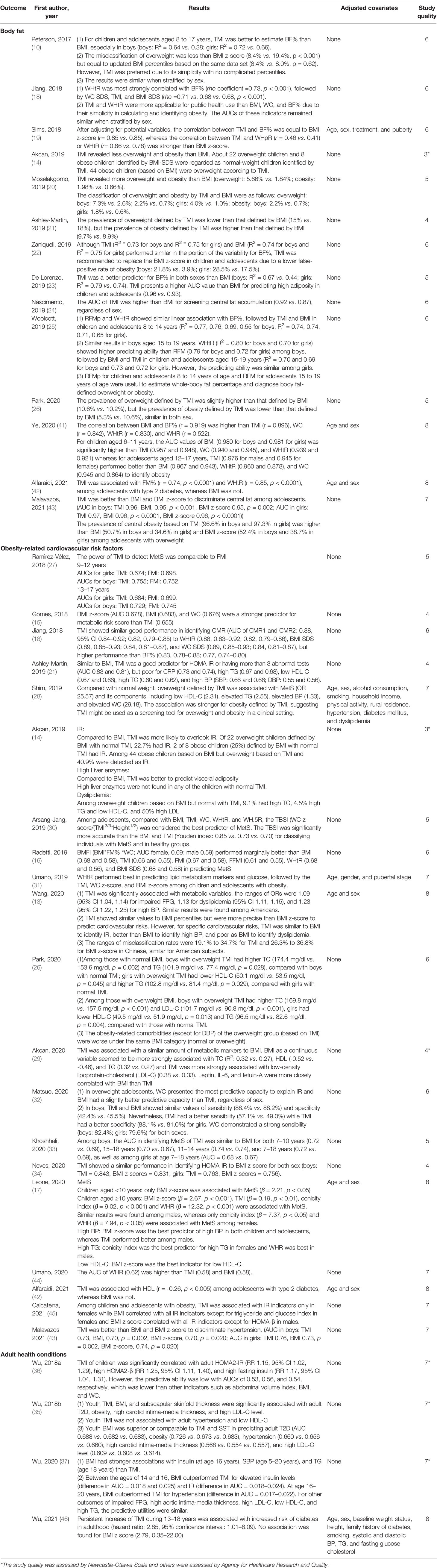
As shown in Table 2, All of the 32 included studies were of moderate to high quality except for one article rated as having a low quality (quality score = 3) (14).
TMI for Screening Body Fat in Children and Adolescents
A total of 14 articles evaluated the ability of TMI to identify body fat mass in children and adolescents compared with BMI (Table 2) (10, 14, 18–26, 41–43).
It has been shown that percent of body fat (BF%) as a gold standard was better predicted by TMI than by BMI (10, 25), although one study reported that both relative fat mass pediatric (RFMp) based on height and waist circumference [WC], and waist-to-height ratio (WHtR) performed better than both TMI and BMI (25). When WHtR was used to define central obesity as the gold standard, three articles showed that TMI was better than BMI correlated with central fat accumulation in both preschool-aged children aged 2–5 years (24) and children and adolescents aged 5–17 years (19, 43).
When BMI and TMI were used as continuous variables, TMI correlated similarly or better than BMI with BF% in children and adolescents (18, 19, 22, 23, 41, 42). Although TMI and BMI among children and adolescents aged 5–18 years explained a similar proportion of the variability for BF%, TMI was recommended to replace BMI z-score in children and adolescents due to its lower false-positive rate of obesity than the BMI z-score (boys: 2.9% vs. 21.8%; girls: 17.5% vs. 28.5%) (18, 19, 22). TMI presented a higher area under the curve (AUC) value than BMI for predicting high BF% (0.96 vs. 0.93, p < 0.001) measured by dual-energy X-ray absorptiometry (DEXA) among children and adolescents aged 8–17 years (23) or more strongly correlated with BF% compared to BMI in adolescents (41, 42).
The remaining four studies could not conclude about a possible advantage of either TMI or BMI to identify overweight or obesity status because of the lack of a gold standard (to objectively assess adiposity) and inconsistent cutoffs (14, 20, 21, 26). Akcan et al. found that TMI identified a lower prevalence of overweight and obesity among children aged 6–17 years compared to BMI-SDS (14), which was contrary to the finding among children aged 9–13 years, independent of sex (20). When considering overweight and obesity separately, Ashley-Martin et al. found that BMI defined more overweight than TMI, whereas TMI defined more obesity than BMI among children and adolescents aged 6–19 years (21), inversely to the findings among children and adolescents aged 10–20 years reported by Park et al. (26).
Overall, studies using a gold standard for comparison and using BMI and TMI as continuous variables suggested that TMI performed equally or better than the widely used BMI to predict BF% and central fat among children and adolescents. TMI was preferred in adolescence due to its stability.
TMI and Obesity-Related Cardiovascular Risk Factors in Children and Adolescents
Twenty articles on the association between TMI and MetS and its components were included in this systematic review (Table 2) (13–18, 21, 26–34, 42–45).
MetS
Ten articles have evaluated the association of TMI and other anthropometric indicators with MetS, metabolic risk score, or cardio-metabolic risk (13, 15–18, 27–30, 33). Three of the 10 articles showed that TMI was not better than other indicators such as BMI (or BMI z-score or BMI-SDS) among children and adolescents aged 10–17 years to predict MetS and a metabolic risk score (15–17). However, the other seven articles suggested that TMI could be a useful screening tool or similar to BMI in predicting MetS or cardio-metabolic risks in children and adolescents aged 5.3 to 25 years (13, 18, 27–30, 33).
TMI was found to be associated with obesity-related CVRFs, including MetS and its components [elevated blood pressure (BP), elevated WC, low high-density lipoprotein cholesterol (HDL-C), and elevated triglycerides (TG)] in late adolescence (28). It was reported that TMI performed similarly to FMI (27) or BMI, or was an auxiliary indicator in addition to BMI, to identify MetS, a metabolic risk score, or CVRFs among children and adolescents aged 5–18 years (13, 18, 29, 33). However, the tri-ponderal body shape index [WC z-score/(TMI2/3*height1/2)] including TMI and WC z-score components performed more accurately in predicting MetS than BMI and TMI (Youden index: 0.85 vs. 0.73 vs. 0.70) among children and adolescents aged 7–18 years, suggesting that the combination of TMI and a WC z-score could be considered as a useful predictor for MetS in children and adolescents (30).
Overall, TMI performed similarly as compared to BMI and other indicators in predicting MetS in most of the included studies, and TMI was also suggested to be a useful tool when used in combination with other adiposity indicators (e.g., BMI and WC) for identifying MetS.
Insulin Resistance
Eight articles compared TMI and BMI for identifying insulin resistance (IR) or impaired glucose in children and adolescents (Table 2) (13, 14, 21, 29, 31, 32, 34, 45). Among these eight articles, seven reported that BMI (used as a continuous variable) performed similarly or marginally better than TMI for identifying IR (13, 21, 29, 31, 32, 34, 45). In addition, compared to BMI, TMI was more likely to underestimate IR (14). The inconsistent cutoffs of TMI and BMI for identifying overweight might lead to different identification of IR. When restricted to children and adolescents aged 4–18 years with overweight or obesity, WHtR or WC, used as continuous variables, seemed to perform best among the four obesity-related indicators (TMI, WC z-score, BMI z-score, and BMI) to predict IR (31, 32).
Overall, TMI did not seem to be superior to BMI for predicting IR in children and adolescents. However, WHtR or WC could be a useful indicator for identifying IR among children and adolescents with overweight and obesity.
Blood Pressure
Only five studies compared the correlation of TMI and BMI with BP, with inconsistent results (Table 2) (13, 17, 21, 33, 43). Although BMI correlated with BP levels stronger than TMI (17, 33), one study based on 5,814 children and adolescents aged 6–19 years showed that, similar to BMI using a continuous variable, TMI (used as a continuous variable) had a low ability to identify high BP, with an AUC of only 0.66 to predict systolic BP and 0.60 to predict diastolic BP (21); similar findings were found among 57,201 Chinese children and adolescents aged 7–18 years, among 10,441 American adolescents aged 12–18 years (13) and among Italian adolescents (43).
Overall, only a few studies examined the question and they tended to suggest that either TMI or BMI performed poorly in identifying high BP in children and adolescents, and the ability varied in different populations.
Dyslipidemia
As shown in Table 2, three articles showed that both TMI and BMI poorly predicted dyslipidemia (13, 14, 21). Although using the same BMI classification, total cholesterol (TC) in boys and HDL-C and TG in girls were worse among children with overweight defined by TMI than among those with normal TMI (26), BMI (as a continuous variable) seemed to be more strongly associated with TC (R2: 0.32 vs. 0.27), HDL (-0.52 vs. -0.46), and TG (0.32 vs. 0.27) compared to TMI, while TMI (as a continuous variable) was more strongly associated with low-density lipoprotein-cholesterol (LDL-C) than BMI (0.38 vs. 0.33) (29), similar to findings on low HDL-C reported by Leone et al. (17), but inversely to findings by Alfaraidi et al. (42).
Overall, there are limited studies on the association of TMI and BMI with dyslipidemia components, and findings suggest that BMI performs better than TMI to identify high TC and TG, whereas TMI is superior to BMI to identify high LDL-C. This will need further evaluation.
Inflammatory and Liver Function Markers
As shown in Table 2, for C-reactive protein (CRP), the prediction accuracy of TMI and BMI z-score was similar (AUC: 0.74 vs. 0.73) (21), whereas other inflammatory markers including leptin, IL-6, and fetuin-A were more closely correlated with BMI than TMI (29). For liver enzymes, overweight and obese status based on TMI could significantly predict elevated serum glutamic oxaloacetic transaminase or elevated serum glutamic pyruvic transaminase, compared with overweight and obesity status based on BMI. However, different cutoffs were defined for BMI vs. TMI, which limits direct comparison (14). For non-alcoholic fatty liver, the discriminating ability of TMI was similarly poor as BMI, with AUC values of only 0.58 (44).
Overall, there is only limited evidence about the performance of TMI and BMI to predict inflammatory markers, which needs further research.
TMI in Childhood or Adolescence for Prediction of Specific CVRFs in Adulthood
Only four articles focused on the association of TMI vs. other obesity-related indicators in childhood or adolescence with CVRFs in adulthood (35–37, 46) (Table 2). BMI at ages 2 to 20 years predicted the presence of CVRFs in young adults aged 20 years as well or better than TMI. For example, the ability to predict adult IR, elevated insulin levels, and hypertension seemed to be stronger for BMI vs. TMI (as assessed in childhood), but similar for the prediction in adults of impaired fasting plasma glucose (FPG), low HDL-C, high LDL-C, high TG, and high aortic intima-media thickness (37). Similarly, another study showed that the AUC values for TMI, or for combination of TMI and BMI, did not outperform BMI alone in predicting adult obesity, diabetes, high carotid intima-media thickness, high LDL-C, and hypertension (35). The AUCs were low for TMI (0.53, 0.56, and 0.54), as well as for other adiposity indicators such as abdominal volume index (0.61, 0.62, and 0.61), BMI (0.59, 0.60, and 0.59), and WC (0.61, 0.61, and 0.61) in childhood to predict adult homeostasis model assessment 2-insulin resistance (HOMA2-IR), HOMA2-β, and high fasting insulin (36). However, when considering growth trajectory instead of a single measurement in childhood, a persistently high TMI during adolescence had predicted diabetes quite well in adults (AUC value as high as 0.81) (46).
Overall, TMI in childhood or adolescence seems to have a lower ability than BMI and other adiposity indicators to predict specific CVRFs in adulthood, whereas TMI trajectory has a higher ability than BMI trajectory in predicting diabetes in adulthood.
Discussion
Main findings
To the best of our knowledge, this is the first review to summarize the evidence regarding TMI as a screening tool for body fat and CVRFs in childhood and adulthood. TMI seemed to perform similarly or better than BMI for predicting body fat and central fat and performed similarly well as BMI in identifying MetS. However, the available evidence on the comparison of TMI and BMI (measured in childhood) for identifying specific CVRFs (in childhood or later in adulthood) including IR, high BP, dyslipidemia, and inflammation was limited and not compelling.
TMI Performed Better Than BMI to Estimate Body Fat in Children and Adolescents
Unlike for adults, no standard BF% cutoff was established to define excess adiposity among children and adolescents until now (47), and objective measurements of fat mass [e.g., DEXA, doubly-labeled water (48), and isotope dilution technique (49)] were much complex and expensive. The components of TMI or BMI (weight and height) can be simply measured using the weight scale and the stadiometer that are widely used for routine pediatric clinical practice. Therefore, in this review, we compared the performance of TMI and BMI and our study suggested that TMI performed better than BMI to estimate body fat in children and adolescents at clinical practice.
The disadvantage of BMI and the advantage of TMI to estimate body fat are as follows. First, although BMI z-score seemed to predict well total fat mass, it predicted BF% weakly with altered body composition among adolescents (50). Second, the definition of overweight and obesity using BMI should be based on sex- and age-specific percentile values in childhood, but this requires using complex tables (10), which may overestimate the actual prevalence of adiposity in children, excessively worrying families and patients (50–53), particularly for adolescents who may be more prone to fat-shaming and weight bias (54). Third, TMI (which is defined independently of age and sex) could be simpler to use compared to age- and sex-stratified BMI cutoffs and a specific cutoff of TMI has been proposed (10). A better relation of TMI with body fat mass across age may be consistent with the fact that BF% may change largely during adolescence (possibly more among girls) due to the height spurt in this age range (55). Fourth, compared to BMI, TMI was more correlated with WHtR, which is a reliable clinical measure of abdominal obesity and is consistently associated with CVRFs (56). TMI could therefore help identify children and adolescents who are overweight or obese based on BMI but also have central obesity and increased risk of CVRFs.
TMI Was More Simple and Accurate Than Other Indicators to Estimate Body Fat in Children and Adolescents
Although the RFMp calculated based on WC and height performed better to estimate BF% than TMI (25), the inter-operator variability between WC measurements is significant, which may cause more misclassification of MetS (57). Furthermore, for tall and thin people, WHtR may be unusually high, causing RFMp and RFM to tend to be 0 or negative (25). Therefore, considering the accuracy and simplicity of the use of TMI in primary health care services and its constancy in predicting adiposity at adolescence, TMI may be useful to evaluate body fat in adolescents. Yet, definite answers about the performance of BMI vs. TMI to predict adiposity in chidden and adolescents needs further studies using objective measurement of body fat mass (e.g., DEXA, isotope dilution) as the gold standard for comparisons, and do so in several populations, and within different ethnic, age, and sex groups.
TMI Performed Similarly as Compared to BMI and Other Indicators in Predicting MetS and Its Components
Although TMI was superior to BMI to screen central fat (19, 24), in this review, it was similar to or not better than BMI to identify MetS and specific CVRFs. One possible reason might be that adiposity defined according to TMI or BMI only accounts for one of the MetS criteria. Another reason might be the inconsistent performances of three indicators (TMI, BMI, and WC) in identifying specific CVRFs including IR, high BP, dyslipidemia, and inflammation (13, 14, 21, 29, 31, 32), which are the main components of MetS.
Age and Trajectory Influence the Association Between TMI vs. BMI in Childhood or Adolescence and Obesity-Related Morbidity in Adulthood
Although BMI in childhood or adolescence seemed to perform marginally better than TMI to predict obesity-related morbidity in adulthood (35, 36), the difference disappeared after adjusting for age (35), suggesting that age might be an important confounding factor that influences the association between BMI in childhood and obesity-related morbidity in adulthood. BMI was better than TMI only in late adolescence to predict adult IR and hypertension, suggesting that the variation of BMI during adolescence influences the strength of the association (10, 37). When considering trajectories, persistently high TMI during 13 and 18 years performed better than the BMI trajectory to predict adult diabetes (46), suggesting that, during adolescence, TMI trajectory (i.e., repeated measurements) may better reflect growth and predict adult CVRD outcomes. Therefore, further prospective studies with large sample sizes, multiethnic populations, and repeated measurements of anthropometric indicators are needed to confirm these findings.
Strengths and Limitations
To the best of our knowledge, this is the first comprehensive review that compared TMI with BMI or other indicators in children and adolescents to predict obesity-related morbidity in both childhood and adulthood. Several limitations should be noted in this review. First, there was high heterogeneity between studies in the considered variables and how the adiposity cutoffs were defined, which limits direct comparisons. Second, most studies on the identification of CVRFs in childhood and adolescence were cross-sectional, which cannot prove causality (55). It must be however mentioned that a marker does not necessarily need to be causally related to an outcome to enable a good prediction. Third, a majority of the included studies came from Western countries, which limits the extrapolation of the results to other populations. Further studies with various ethnic/race groups are needed to confirm the predictive ability of TMI to predict adiposity in children and adolescents. Fourth, although TMI seems better than BMI to predict concomitant fat mass in children and adolescents, neither TMI nor BMI can distinguish fat mass from non-fat mass, and these indicators cannot replace objective measurement of fat mass (e.g., DEXA, isotope dilution). Again, an ultimate fully valid method to compare how BMI or TMI predicts adiposity should rely on objectively measured adiposity as the gold standard (e.g., DEXA, isotope dilution methods) and use a similar dichotomization of categories of elevated BMI or elevated TMI (e.g., using the same percentile cutoffs, e.g., p80 or p90) to enable valid comparisons; this was only rarely performed in the considered studies.
Conclusions
In conclusion, TMI only requires a single threshold according to sex (i.e., no need for sex- and age-specific thresholds) and TMI seems to predict adiposity similarly or better in children and adolescents than BMI. In addition, TMI seemed to perform similarly as BMI for identifying MetS. However, the clinical use of TMI vs. BMI in childhood, in order to predict specific elevated CVRFs in childhood or later in adulthood, is still not definitive and needs further studies, particularly those with a longitudinal design.
Data Availability Statement
The original contributions presented in the study are included in the article/supplementary material. Further inquiries can be directed to the corresponding author.
Author Contributions
BX and PB designed the research. JS and RY conducted the literature search and performed the statistical analysis of the data. JS, BX, and PB wrote the manuscript draft. JS, BX, MZ, and PB contributed to the critical revision of the manuscript for important intellectual content. All authors contributed to the article and approved the submitted version.
Funding
This work was supported by National Natural Science Foundation of China, Grant/Award Number: 81673195; Youth Team of Humanistic and Social Science of Shandong University.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. N. C. D. Risk Factor Collaboration. Worldwide Trends in Body-Mass Index, Underweight, Overweight, and Obesity From 1975 to 2016: A Pooled Analysis of 2416 Population-Based Measurement Studies in 128.9 Million Children, Adolescents, and Adults. Lancet (2017) 390(10113):2627–42. doi: 10.1016/S0140-6736(17)32129-3
2. Jing L, Nevius CD, Friday CM, Suever JD, Pulenthiran A, Mejia-Spiegeler A, et al. Ambulatory Systolic Blood Pressure and Obesity Are Independently Associated With Left Ventricular Hypertrophic Remodeling in Children. J Cardiovasc Magn Reson (2017) 19(1):86. doi: 10.1186/s12968-017-0401-3
3. Park MH, Skow A, De Matteis S, Kessel AS, Saxena S, Viner RM, et al. Adiposity and Carotid-Intima Media Thickness in Children and Adolescents: A Systematic Review. BMC Pediatr (2015) 15:161. doi: 10.1186/s12887-015-0478-5
4. Cho H, Kim JH. Prevalence of Microalbuminuria and its Associated Cardiometabolic Risk Factors in Korean Youth: Data From the Korea National Health and Nutrition Examination Survey. PloS One (2017) 12(6):e0178716. doi: 10.1371/journal.pone.0178716
5. Anderson EL, Howe LD, Jones HE, Higgins JP, Lawlor DA, Fraser A. The Prevalence of Non-Alcoholic Fatty Liver Disease in Children and Adolescents: A Systematic Review and Meta-Analysis. PloS One (2015) 10(10):e0140908. doi: 10.1371/journal.pone.0140908
6. Weihrauch-Bluher S, Schwarz P, Klusmann JH. Childhood Obesity: Increased Risk for Cardiometabolic Disease and Cancer in Adulthood. Metabolism (2019) 92:147–52. doi: 10.1016/j.metabol.2018.12.001
7. Park MH, Falconer C, Viner RM, Kinra S. The Impact of Childhood Obesity on Morbidity and Mortality in Adulthood: A Systematic Review. Obes Rev (2012) 13(11):985–1000. doi: 10.1111/j.1467-789X.2012.01015.x
8. Simmonds M, Burch J, Llewellyn A, Griffiths C, Yang H, Owen C, et al. The Use of Measures of Obesity in Childhood for Predicting Obesity and the Development of Obesity-Related Diseases in Adulthood: A Systematic Review and Meta-Analysis. Health Technol Assess (2015) 19(43):1–336. doi: 10.3310/hta19430
9. Rothman KJ. BMI-Related Errors in the Measurement of Obesity. Int J Obes (Lond) (2008) 32(Suppl 3):S56–9. doi: 10.1038/ijo.2008.87
10. Peterson CM, Su H, Thomas DM, Heo M, Golnabi AH, Pietrobelli A, et al. Tri-Ponderal Mass Index vs Body Mass Index in Estimating Body Fat During Adolescence. JAMA Pediatr (2017) 171(7):629–36. doi: 10.1001/jamapediatrics.2017.0460
11. Schmidt SC, Bosy-Westphal A, Niessner C, Woll A. Representative Body Composition Percentiles From Bioelectrical Impedance Analyses Among Children and Adolescents. The MoMo Study. Clin Nutr (2019) 38(6):2712–20. doi: 10.1016/j.clnu.2018.11.026
12. Cole TJ, Bellizzi MC, Flegal KM, Dietz WH. Establishing a Standard Definition for Child Overweight and Obesity Worldwide: International Survey. BMJ (2000) 320(7244):1240–3. doi: 10.1136/bmj.320.7244.1240
13. Wang X, Dong B, Ma J, Song Y, Zou Z, Arnold L. Role of Tri-Ponderal Mass Index in Cardio-Metabolic Risk Assessment in Children and Adolescents: Compared With Body Mass Index. Int J Obes (Lond) (2020) 44(4):886–94. doi: 10.1038/s41366-019-0416-y
14. Akcan N, Bundak R. Accuracy of Tri-Ponderal Mass Index and Body Mass Index in Estimating Insulin Resistance, Hyperlipidemia, Impaired Liver Enzymes or Thyroid Hormone Function and Vitamin D Levels in Children and Adolescents. J Clin Res Pediatr Endocrinol (2019) 11(4):366–73. doi: 10.4274/jcrpe.galenos.2019.2018.0279
15. Gomes TN, Nevill A, Katzmarzyk PT, Pereira S, Dos Santos MM, Buranarugsa R, et al. Identifying the Best Body-Weight-Status Index Associated With Metabolic Risk in Youth. Scand J Med Sci Sports (2018) 28(11):2375–83. doi: 10.1111/sms.13249
16. Radetti G, Fanolla A, Grugni G, Lupi F, Sartorio A. Indexes of Adiposity and Body Composition in the Prediction of Metabolic Syndrome in Obese Children and Adolescents: Which Is the Best? Nutr Metab Cardiovasc Dis (2019) 29(11):1189–96. doi: 10.1016/j.numecd.2019.06.011
17. Leone A, Vizzuso S, Brambilla P, Mameli C, Ravella S, De Amicis R, et al. Evaluation of Different Adiposity Indices and Association With Metabolic Syndrome Risk in Obese Children: Is There a Winner? Int J Mol Sci (2020) 21(11):4083. doi: 10.3390/ijms21114083
18. Jiang Y, Dou YL, Xiong F, Zhang L, Zhu GH, Wu T, et al. Waist-To-Height Ratio Remains an Accurate and Practical Way of Identifying Cardiometabolic Risks in Children and Adolescents. Acta Paediatr (2018) 107:1629–34. doi: 10.1111/apa.14323
19. Sims ED, Wang KW, Fleming A, Johnston DL, Zelcer SM, Rassekh SR, et al. Tri-Ponderal Mass Index in Survivors of Childhood Brain Tumors: A Cross-Sectional Study. Sci Rep (2018) 8(1):16336. doi: 10.1038/s41598-018-34602-5
20. Moselakgomo VK, Van Staden M. Diagnostic Accuracy of Tri-Ponderal Mass Index and Body Mass Index in Estimating Overweight and Obesity in South African Children. Afr J Prim Health Care Fam Med (2019) 11(1):e1–7. doi: 10.4102/phcfm.v11i1.1949
21. Ashley-Martin J, Ensenauer R, Maguire B, Kuhle S. Predicting Cardiometabolic Markers in Children Using Tri-Ponderal Mass Index: A Cross-Sectional Study. Arch Dis Child (2019) 104(6):577–82. doi: 10.1136/archdischild-2018-316028
22. Zaniqueli D, Oliosa PR, Neves FS, Pani VO, Martins CR, de Souza Pecanha MA, et al. Ponderal Index Classifies Obesity in Children and Adolescents More Accurately Than Body Mass Index Z-Scores. Pediatr Res (2019) 86(1):128–33. doi: 10.1038/s41390-019-0395-7
23. De Lorenzo A, Romano L, Di Renzo L, Gualtieri P, Salimei C, Carrano E, et al. Triponderal Mass Index Rather Than Body Mass Index: An Indicator of High Adiposity in Italian Children and Adolescents. Nutrition (2019) 60:41–7. doi: 10.1016/j.nut.2018.09.007
24. Nascimento VG, Bertoli CJ, Gallo PR, Abreu LC, Leone C. Tri-Ponderal Mass Index: A Screening Tool for Risk of Central Fat Accumulation in Brazilian Preschool Children. Medicina (Kaunas) (2019) 55(9):557. doi: 10.3390/medicina55090577
25. Woolcott OO, Bergman RN. Relative Fat Mass as an Estimator of Whole-Body Fat Percentage Among Children and Adolescents: A Cross-Sectional Study Using NHANES. Sci Rep (2019) 9(1):15279. doi: 10.1038/s41598-019-51701-z
26. Park HK, Shim YS. Distribution of Tri-Ponderal Mass Index and its Relation to Body Mass Index in Children and Adolescents Aged 10 to 20 Years. J Clin Endocrinol Metab (2020) 105(3):e826–e34. doi: 10.1210/clinem/dgaa030
27. Ramírez-Vélez R, Correa-Bautista J, Carrillo H, González-Jiménez E, Schmidt-RioValle J, Correa-Rodríguez M, et al. Tri-Ponderal Mass Index vs. Fat Mass/Height3 as a Screening Tool for Metabolic Syndrome Prediction in Colombian Children and Young People. Nutrients (2018) 10(4):412. doi: 10.3390/nu10040412
28. Shim YS. The Relationship Between Tri-Ponderal Mass Index and Metabolic Syndrome and Its Components in Youth Aged 10-20 Years. Sci Rep (2019) 9(1):14462. doi: 10.1038/s41598-019-50987-3
29. Akcan N, Obaid M, Salem J, Bundak R. Evidence in Obese Children: Contribution of Tri-Ponderal Mass Index or Body Mass Index to Dyslipidemia, Obesity-Inflammation, and Insulin Sensitivity. J Pediatr Endocrinol Metab (2020) 33(2):223–31. doi: 10.1515/jpem-2019-0106
30. Arsang-Jang S, Kelishadi R, Esmail Motlagh M, Heshmat R, Mansourian M. Temporal Trend of Non-Invasive Method Capacity for Early Detection of Metabolic Syndrome in Children and Adolescents: A Bayesian Multilevel Analysis of Pseudo-Panel Data. Ann Nutr Metab (2019) 75(1):55–65. doi: 10.1159/000500274
31. Umano GR, Di Sessa A, Cirillo G, Ursi D, Marzuillo P, Miraglia Del Giudice E. Waist-To-Height Ratio Is More Strongly Associated Than Other Weight-Related Anthropometric Measures With Metabolic Variables. Acta Paediatr (2019) 108(12):2296–97. doi: 10.1111/apa.14992
32. Matsuo AR, Lopes WA, Locatelli JC, Simoes CF, de Oliveira GH, Nardo N Jr. Tri-Ponderal Mass Index as a Tool for Insulin Resistance Prediction in Overweight Adolescents: A Cross-Sectional Study. Nutrition (2020) 74:110744. doi: 10.1016/j.nut.2020.110744
33. Khoshhali M, Heidari-Beni M, Qorbani M, Motlagh ME, Ziaodini H, Heshmat R, et al. Tri-Ponderal Mass Index and Body Mass Index in Prediction of Pediatric Metabolic Syndrome: The CASPIAN-V Study. Arch Endocrinol Metab (2020) 64(2):171–78. doi: 10.20945/2359-3997000000206
34. Neves FS, Alvim RO, Zaniqueli D, Pani VO, Martins CR, Pecanha MAS, et al. Tri-Ponderal Mass Index Is Useful for Screening Children and Adolescents With Insulin Resistance. Rev Paul Pediatr (2020) 38:e2019066. doi: 10.1590/1984-0462/2020/38/2019066
35. Wu F, Buscot MJ, Juonala M, Hutri-Kahonen N, Viikari JSA, Raitakari OT, et al. Association of Youth Triponderal Mass Index vs Body Mass Index With Obesity-Related Outcomes in Adulthood. JAMA Pediatr (2018) 172(12):1192–95. doi: 10.1001/jamapediatrics.2018.3034
36. Wu F, Ho V, Fraser BJ, Schmidt MD, Dwyer T, Venn AJ, et al. Predictive Utility of Childhood Anthropometric Measures on Adult Glucose Homeostasis Measures: A 20-Year Cohort Study. Int J Obes (Lond) (2018) 42(10):1762–70. doi: 10.1038/s41366-018-0177-z
37. Wu F, Buscot MJ, Niinikoski H, Rovio SP, Juonala M, Sabin MA, et al. Age-Specific Estimates and Comparisons of Youth Tri-Ponderal Mass Index and Body Mass Index in Predicting Adult Obesity-Related Outcomes. J Pediatr (2020) 218:198–203 e6. doi: 10.1016/j.jpeds.2019.10.062
38. Karchynskaya V, Kopcakova J, Klein D, Gaba A, Madarasova-Geckova A, van Dijk JP, et al. Is BMI a Valid Indicator of Overweight and Obesity for Adolescents? Int J Environ Res Public Health (2020) 17(13):4815. doi: 10.3390/ijerph17134815
39. Zeng X, Zhang Y, Kwong JS, Zhang C, Li S, Sun F, et al. The Methodological Quality Assessment Tools for Preclinical and Clinical Studies, Systematic Review and Meta-Analysis, and Clinical Practice Guideline: A Systematic Review. J Evid Based Med (2015) 8(1):2–10. doi: 10.1111/jebm.12141
40. Wells GA, Shea B, O’Connell D, Peterson J, Welch V, Losos M, et al. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality If Nonrandomized Studies in Meta-Analyses. Available at: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp (Accessed December 23, 2020).
41. Ye XF, Dong W, Tan LL, Zhang ZR, Qiu YL, Zhang J. Identification of the Most Appropriate Existing Anthropometric Index for Home-Based Obesity Screening in Children and Adolescents. Public Health (2020) 189:20–5. doi: 10.1016/j.puhe.2020.09.007
42. Alfaraidi H, Wicklow B, Dart AB, Sellers E, McGavock J, Thabane L, et al. The Tri-Ponderal Mass Index Is Associated With Adiposity in Adolescent Type 2 Diabetes Mellitus: A Cross-Sectional Analysis. Sci Rep (2021) 11(1):9111. doi: 10.1038/s41598-021-88705-7
43. Malavazos AE, Capitanio G, Milani V, Ambrogi F, Matelloni IA, Basilico S, et al. Tri-Ponderal Mass Index vs Body Mass Index in Discriminating Central Obesity and Hypertension in Adolescents With Overweight. Nutr Metab Cardiovasc Dis (2021) 31(5):1613–21. doi: 10.1016/j.numecd.2021.02.013
44. Umano GR, Grandone A, Di Sessa A, Cozzolino D, Pedulla M, Marzuillo P, et al. Pediatric Obesity-Related Non-Alcoholic Fatty Liver Disease: Waist-to-Height Ratio Best Anthropometrical Predictor. Pediatr Res (2020) 90(1):166-70. doi: 10.1038/s41390-020-01192-w
45. Calcaterra V, Verduci E, Schneider L, Cena H, De Silvestri A, Vizzuso S, et al. Sex-Specific Differences in the Relationship Between Insulin Resistance and Adiposity Indexes in Children and Adolescents With Obesity. Children (Basel) (2021) 8(6):449. doi: 10.3390/children8060449
46. Wu YF, Fan HY, Chen YC, Kuo KL, Chien KL. Adolescent Tri-Ponderal Mass Index Growth Trajectories and Incident Diabetes Mellitus in Early Adulthood. J Clin Endocrinol Metab (2021) 106(8):e2919-27. doi: 10.1210/clinem/dgab235
47. De Lorenzo A, Soldati L, Sarlo F, Calvani M, Di Lorenzo N, Di Renzo L. New Obesity Classification Criteria as a Tool for Bariatric Surgery Indication. World J Gastroenterol (2016) 22(2):681–703. doi: 10.3748/wjg.v22.i2.681
48. LeMura LM, Maziekas MT. Factors That Alter Body Fat, Body Mass, and Fat-Free Mass in Pediatric Obesity. Med Sci Sports Exerc (2002) 34(3):487–96. doi: 10.1097/00005768-200203000-00016
49. Ben Jemaa H, Mankai A, Khlifi S, Minaoui R, Ghozzi D, Zediri M, et al. Development and Validation of Impedance-Based Equations for the Prediction of Total Body Water and Fat-Free Mass in Children Aged 8-11 Years. Clin Nutr (2019) 38(1):227–33. doi: 10.1016/j.clnu.2018.01.028
50. Vanderwall C, Randall Clark R, Eickhoff J, Carrel AL. BMI Is a Poor Predictor of Adiposity in Young Overweight and Obese Children. BMC Pediatr (2017) 17(1):135. doi: 10.1186/s12887-017-0891-z
51. Buss J. Limitations of Body Mass Index to Assess Body Fat. Workplace Health Saf (2014) 62(6):264. doi: 10.1177/216507991406200608
52. Weber DR, Moore RH, Leonard MB, Zemel BS. Fat and Lean BMI Reference Curves in Children and Adolescents and Their Utility in Identifying Excess Adiposity Compared With BMI and Percentage Body Fat. Am J Clin Nutr (2013) 98(1):49–56. doi: 10.3945/ajcn.112.053611
53. Vanderwall C, Eickhoff J, Randall Clark R, Carrel AL. BMI Z-Score in Obese Children Is a Poor Predictor of Adiposity Changes Over Time. BMC Pediatr (2018) 18(1):187. doi: 10.1186/s12887-018-1160-5
54. Ikeda JP, Crawford PB, Woodward-Lopez G. BMI Screening in Schools: Helpful or Harmful. Health Educ Res (2006) 21(6):761–9. doi: 10.1093/her/cyl144
55. Hermanussen M, Largo RH, Molinari L. Canalisation in Human Growth: A Widely Accepted Concept Reconsidered. Eur J Pediatr (2001) 160(3):163–7. doi: 10.1007/s004310000706
56. Abraham TM, Pedley A, Massaro JM, Hoffmann U, Fox CS. Association Between Visceral and Subcutaneous Adipose Depots and Incident Cardiovascular Disease Risk Factors. Circulation (2015) 132(17):1639–47. doi: 10.1161/CIRCULATIONAHA.114.015000
Keywords: children, tri-ponderal mass index, obesity, body fat, cardiovascular risk factors
Citation: Sun J, Yang R, Zhao M, Bovet P and Xi B (2021) Tri-Ponderal Mass Index as a Screening Tool for Identifying Body Fat and Cardiovascular Risk Factors in Children and Adolescents: A Systematic Review. Front. Endocrinol. 12:694681. doi: 10.3389/fendo.2021.694681
Received: 13 April 2021; Accepted: 28 September 2021;
Published: 21 October 2021.
Edited by:
Mostafa Qorbani, Alborz University of Medical Sciences, IranReviewed by:
Motahar Heidari-Beni, Isfahan University of Medical Sciences, IranShirin Djalalinia, Ministry of Health and Medical Education, Iran
Copyright © 2021 Sun, Yang, Zhao, Bovet and Xi. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Bo Xi, eGlibzIwMDdAMTI2LmNvbQ==
 Jiahong Sun1
Jiahong Sun1 Bo Xi
Bo Xi