- 1Laboratory of Immunology and Infectious Diseases, Graduate School of Medical Science and Engineering, Korea Advanced Institute of Science and Technology, Daejeon, South Korea
- 2Department of Internal Medicine and Clinical Oncology, University of Bari Medical School, Bari, Italy
Hepatitis C virus (HCV) infects approximately 170 million people worldwide and is a major cause of life-threatening liver diseases such as liver cirrhosis and hepatocellular carcinoma. Acute HCV infection often progresses to chronic persistent infection, although some patients recover spontaneously. The divergent outcomes of acute HCV infection are known to be determined by differences in virus-specific T-cell responses among patients. Of the two major T-cell subsets, CD8+ T-cells are known to be the key effector cells that control viral infections via cytolytic activity and cytokine secretion. Herein, we review various aspects of HCV-specific CD8+ T-cell responses in acute HCV infection. In particular, we focus on timing of CD8+ T-cell responses, relationship between CD8+ T-cell responses and outcomes of acute HCV infection, receptor expression on CD8+ T-cells, breadth of CD8+ T-cell responses, and viral mutations.
Introduction
Hepatitis C virus (HCV) is a positive-stranded RNA virus of the genus Hepacivirus in the family Flaviviridae that infects approximately 170 million people worldwide (1, 2). Acute HCV infection is spontaneously cleared in 20–30% of patients; however, the majority of patients fail to clear HCV and develop chronic persistent infection, which tends to progress to life-threatening liver diseases such as liver cirrhosis and hepatocellular carcinoma (3). Typically, patients with self-limited acute HCV infection undergo sustained viral clearance within the first 12 weeks of disease onset, whereas viremia beyond 6 months generally indicates chronic evolution (4). The divergent outcomes of acute HCV infection are known to be determined by differences in virus-specific T-cell responses among patients (5–9).
Of the two major T-cell subsets, CD8+ T-cells are known to be the key effector cells that control viral infections via cytolytic activity and cytokine secretion. The importance of cytolytic function in HCV infection is suggested by the fact that CD8+ T-cell responses coincide not only with the decrease of HCV RNA titers in blood, but also with the peak in serum alanine aminotransferase (ALT) (10, 11), which is a marker of hepatocyte injury. Interferon-γ (IFN-γ) secreted by CD8+ T-cells also directly exerts antiviral functions. It was shown that IFN-γ suppresses viral replication in HCV replicon studies (12, 13). In addition, the antiviral functions of CD8+ T-cell-secreted IFN-γ were further demonstrated in cocultures of HCV replicon cells and HCV-specific CD8+ T-cells (14).
In fact, the initial study on immune response in HCV-infected chimpanzees showed that HCV-specific CD8+ T-cell response well correlated with protection against HCV (15). The role of CD8+ T-cells in HCV infection was once clearly demonstrated by a chimpanzee study with antibody-mediated depletion of CD8+ T-cells. In this study, CD8+ T-cells were depleted prior to HCV challenge, and viral load remained at high levels for a prolonged period without CD8+ T-cells (16). Viral clearance was only achieved after HCV-specific CD8+ T-cells recovered in the liver (16).
The importance of CD8+ T-cells in HCV infection is also supported by immunogenetics studies that showed associations between specific human leukocyte antigen (HLA) class I allotypes and clinical outcome of HCV infection. HLA-B27, -B57, and -A3 are known to be associated with a high rate of HCV clearance (17–19).
In this review, we summarize various aspects of CD8+ T-cell responses in acute HCV infection. In particular, we focus on timing of CD8+ T-cell responses, relationship between CD8+ T-cell responses and outcomes of acute HCV infection, receptor expression on CD8+ T-cells, breadth of CD8+ T-cell responses, and viral mutations.
Clinical Course of Acute HCV Infection
In principle, acute HCV infection is defined as the first 6 months following infection with HCV. Although HCV RNA titer is high in the circulation, the majority of patients remain asymptomatic during acute HCV infection with only 15% of infected patients undergoing symptomatic acute hepatitis C (20). In addition, a third of symptomatic infected patients do not produce detectable anti-HCV antibodies at the onset of symptoms (21). Accordingly, it is difficult to identify patients with acute HCV infection and to study immune responses during this period. Therefore, much of the knowledge regarding T-cell responses in acute HCV infection has been gleaned through studies of a chimpanzee model. Since the initial study on CD8+ T-cells in chimpanzees with acute HCV infection (15), monoclonal HCV infection of chimpanzees further enabled precise analyses of CD8+ T-cells during acute HCV infection (11, 22).
After inoculation of 100 chimpanzee infectious dose 50 (CID50) of monoclonal HCV-positive plasma, viral RNA becomes detectable in serum within 1–2 weeks and increases rapidly thereafter (11). However, liver injury only increases minimally despite a high level of viremia during this incubation phase, which lasts about 8–12 weeks. During the incubation period, the expression of IFN-stimulated genes (ISGs) is induced in the infected liver (23). In terms of ISGs induction, HCV infection contrasts with hepatitis B virus (HBV) infection. In fact, ISGs expression is not induced by HBV infection (24), and thus HBV is considered a stealth virus (25).
Serum ALT peaks at 8–12 weeks after HCV infection, and the viral RNA titer begins to decrease at this time (11). Of interest, there are three different patterns of viral RNA decrease as initially proposed by Thimme et al. (10). In the first pattern, HCV RNA titer is decreased below the lower detection limit, and ultimately HCV is cleared (Figure 1A). In the second pattern, HCV RNA titer is decreased below the lower detection limit, but HCV RNA reappears in the serum within several weeks and the infection evolves to a chronic persistent infection (Figure 1B). In the third pattern, HCV RNA titer only undergoes a 2–3 log reduction and the infection evolves to a chronic persistent infection (Figure 1C). Thus, HCV infection of chimpanzees with a single molecular clone results in a relatively uniform course of acute HCV infection with significantly divergent outcomes. Therefore, several interesting immunological questions have arisen and have been studied in this model as described below.
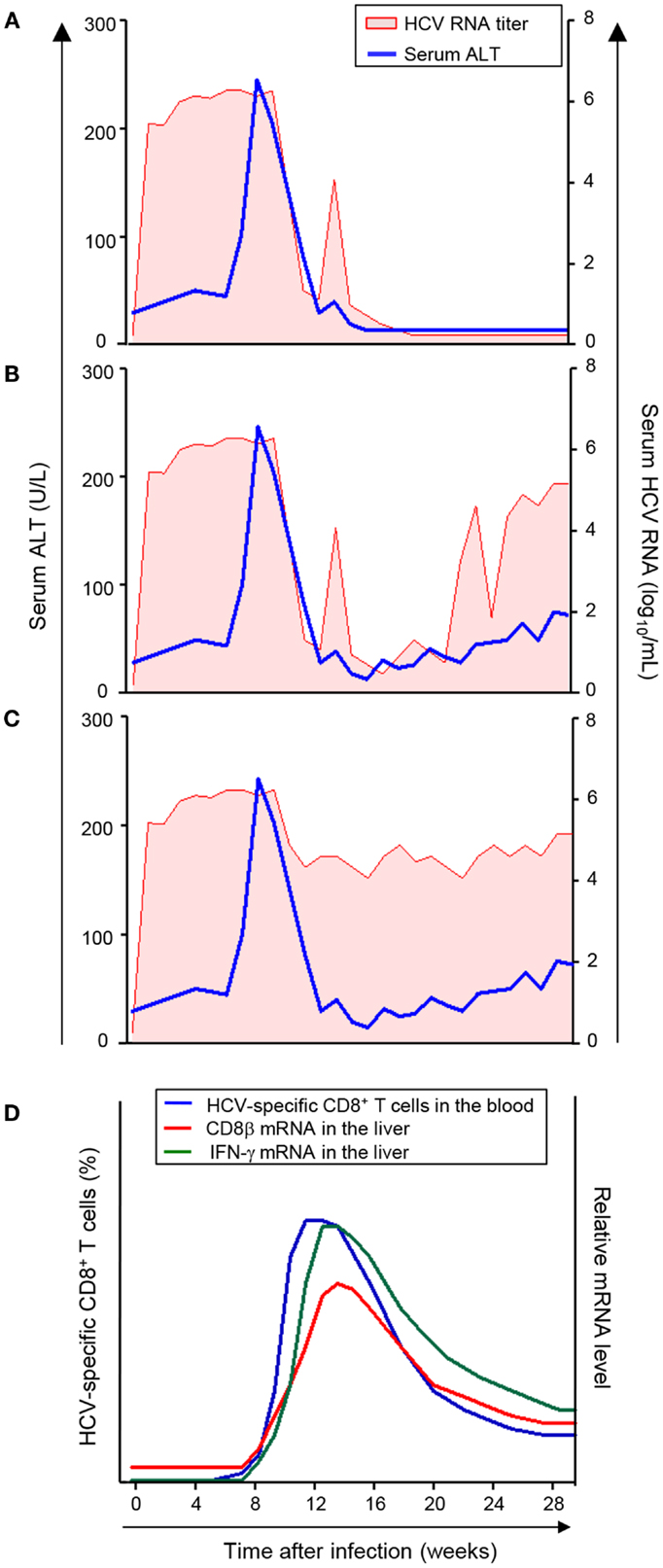
Figure 1. Virologic courses and kinetics of CD8+ T-cell responses in acute HCV infection. (A–C) Three virologic patterns of acute HCV infection (11). (D) Kinetics of CD8+ T-cell response in acute HCV infection (20, 21). The frequency of HCV-specific CD8+ T-cells in the blood and mRNA levels of CD8β and IFN-γ in the liver are presented. The appearance of HCV-specific CD8+ T-cells in the blood coincides with the increase in mRNA levels of CD8β and IFN-γ in the liver and with serum ALT elevation and a decrease in HCV RNA titer.
Timing of CD8+ T-Cell Responses in the Liver and Blood
Initially, a relatively late appearance of virus-specific T-cells in the infected liver was described during acute HCV infection in a chimpanzee study (10). Following infection, HCV replicates rapidly and viral titers increase to high levels in the circulation within days in a chimpanzee model (11, 26). However, HCV-specific T-cell responses do not become detectable in the liver until 8–12 weeks after infection (10). This delay of intrahepatic T-cell responses was shown through functional assays using in vitro expanded, liver-infiltrating T-cells (10). In addition, mRNA analyses showed that the expression of CD8β and IFN-γ increases in the liver at 8–12 weeks after infection (Figure 1D) (10, 11, 26, 27). Further, CD8β and IFN-γ mRNA expression in the liver coincides with a decline in viral titer (Figure 1D), even in the hosts with a chronically evolving course of infection (28).
Recently, the underlying cause of the delayed intrahepatic T-cell responses was revealed through study of HCV-infected chimpanzees (29). In this study, appearance of HCV-specific CD8+ T-cells in the circulation was analyzed using major histocompatibility complex (MHC) class I tetramers. During acute HCV infection, virus-specific CD8+ T-cells appear in the blood at late time points (8–12 weeks after HCV infection) and the appearance of HCV-specific CD8+ T-cells in the blood coincided precisely with the CD8+ T-cell infiltration to the liver, which was quantified by assessing CD8β mRNA levels (Figure 1D) (29). Furthermore, while HCV-specific CD8+ T-cells are induced at late time points, T-cell-recruiting chemokines for CXCR3 and CCR5 are expressed in the HCV-infected liver at early time points (2–8 weeks after HCV infection) (29). Thus, intrahepatic chemokines recruit HCV-specific CD8+ T-cells to the liver as soon as they appear in the blood. Therefore, it can be concluded that the delay in intrahepatic T-cell responses is not due to delayed recruitment, but rather, to delayed priming of HCV-specific CD8+ T-cells (29).
Thus, compared with other viral infections, HCV infection is characterized by delayed induction of virus-specific CD8+ T-cells. This delayed T-cell induction explains the long incubation period prior to development of immune-mediated liver injury (29). Moreover, it may explain why the rate of chronic evolution is so high after HCV infection (10, 11); however, the underlying cause of this delayed induction of HCV-specific CD8+ T-cells is not yet understood. During viral infection, efficient priming of CD8+ T-cells depends on cell death of virus-infected cells and the generation of inflammatory milieu. Virus-specific CD8+ T-cells are primed by cross presentation of viral antigens from virus-infected dying cells (30). HCV is known to be non-cytopathic, therefore it may not kill enough hepatocytes to release viral antigens and may not cause sufficient inflammation to induce cross presentation to CD8+ naïve precursor T-cells. This possibility needs to be investigated in further studies.
Figure 2 summarizes the sequence of major events in acute HCV infection. T-cell-recruiting chemokines for CXCR3 and CCR5 are produced in the liver 2–8 weeks after HCV infection. However, HCV-specific CD8+ T-cells appear in the blood no earlier than 8–12 weeks after infection. These cells express CXCR3 and/or CCR5 (29) and are immediately recruited to the liver. Intrahepatic infiltration of T-cells results in liver injury, manifested by serum ALT elevation and a decrease in HCV RNA titer.
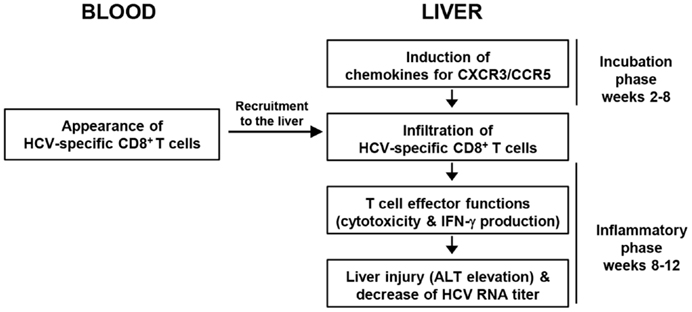
Figure 2. Sequence of events during HCV-specific CD8+ T-cell responses in blood and liver throughout acute HCV infection.
The course of acute HCV infection is modified after re-infection of previously recovered chimpanzees. The duration of the re-infection is substantially shorter than that of the primary infection even after re-challenge with different HCV genotypes (31, 32). In addition, HCV-specific T-cell responses in the blood and liver are detected at much earlier time point after re-infection, compared to the primary infection (33), indicating rapid recall responses of HCV-specific memory T-cells. In a setting of re-infection, the role of CD8+ T-cells was studied by antibody-mediated depletion of CD8+ T-cells. Depletion of CD8+ T-cells and subsequent HCV re-challenge resulted in prolonged viremia, and viral clearance occurred when HCV-specific CD8+ T-cells recovered in the liver (16).
In line with the re-infection studies, successful vaccination shifts the timing of HCV-specific CD8+ T-cell responses earlier. In a study of chimpanzees vaccinated with recombinant adenovirus and DNA against HCV non-structural proteins, HCV RNA was transiently detected and disappeared, and serum ALT was also transiently increased (Figure 3A) (34). In these vaccinated hosts, HCV-specific CD8+ T-cells were detectable in the circulation after vaccination, and they were further expanded after HCV challenge (35). As a result, the frequency of HCV-specific CD8+ T-cells in the vaccinated animals reached peak levels as early as 4 weeks after HCV challenge (Figure 3B), which was 7–15 weeks earlier than in the mock-vaccinated control animals. Accordingly, intrahepatic T-cell responses, evaluated by mRNA levels of CD8β and IFN-γ, were much earlier in the vaccinated animals than in the mock-vaccinated control animals (Figure 3B) (27).
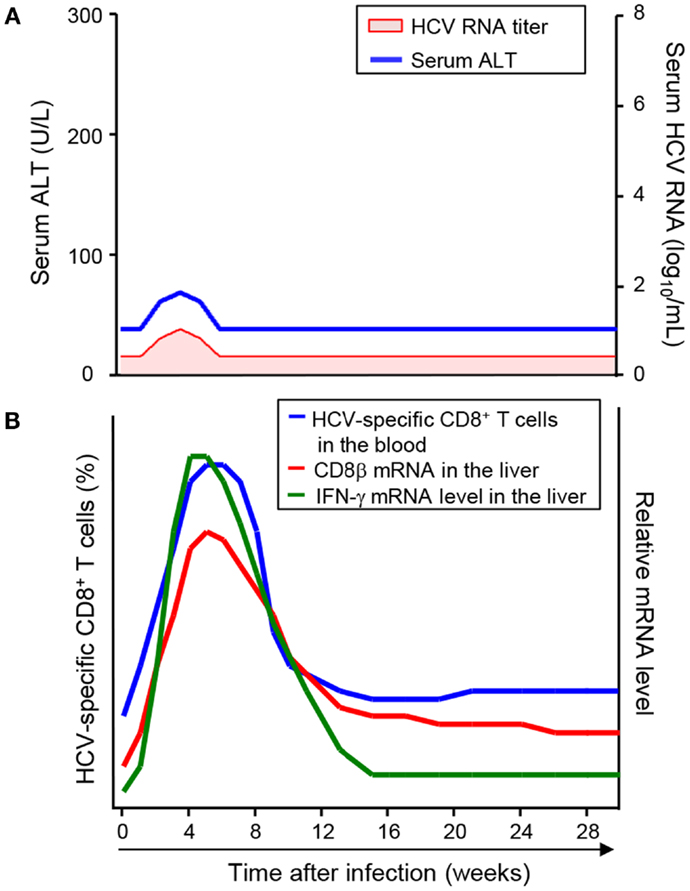
Figure 3. Virologic course and kinetics of CD8+ T-cell responses after HCV challenge in successfully vaccinated hosts. (A) Virologic course after HCV challenge in successfully vaccinated hosts (23). HCV RNA was transiently detected and disappeared, and serum ATL was also transiently elevated. (B) Kinetics of CD8+ T-cell responses after HCV challenge in successfully vaccinated hosts (19, 24). Early CD8+ T-cell responses are shown by the frequency of HCV-specific CD8+ T-cells in the blood and mRNA levels of CD8β and IFN-γ in the liver.
HCV-Specific CD8+ T-Cell Responses and Acute HCV Infection Outcomes
As described above, HCV-specific CD8+ T-cells are induced and appear in the blood at 8–12 weeks after HCV infection and are recruited to the liver at the same time. However, the delayed induction of HCV-specific CD8+ T-cells was observed irrespective of the outcome of acute HCV infection (29). Instead, spontaneous resolution of acute HCV infection correlates with robust and sustained responses of HCV-specific T-cells in patients (6, 8, 36) and chimpanzees (10, 15). In contrast, chronic evolution of acute HCV infection is associated with weak and transient responses of HCV-specific T-cells (8, 10, 15, 36). In addition, the appearance of vigorous HCV-specific CD8+ T-cell responses against multiple epitopes was associated with the onset of viral clearance (6, 8). In a recent study of a chimpanzee model infected with monoclonal HCV, a similar result was reported (37). In this study, HCV-specific T-cell responses in the blood were comprehensively evaluated by IFN-γ enzyme-linked immunosorbent spot (ELISpot) assays with overlapping peptides covering all HCV proteins. While the HCV-specific IFN-γ response was strong in the chimpanzees with a self-limiting course of infection, it was undetectable or delayed in the chimpanzees with a chronically evolving course of infection (37). T-cell response was analyzed also in human immunodeficiency virus-positive patients with acute HCV infection, and spontaneous clearance of acute HCV infection was associated with strong T-cell response against HCV antigens (38).
Early antiviral therapy with pegylated IFN-α and ribavirin is effective in patients with acute HCV infection, and thus it prevents the development of chronic hepatitis. Then what will happen to HCV-specific CD8+ T-cells during successful antiviral therapy in patients with acute HCV infection? HCV-specific CD8+ T-cells observed in the early acute phase of HCV infection are maintained by continuous stimulation with HCV antigens; therefore, initiation of early antiviral therapy during acute HCV infection results in a rapid decline in CD8+ T-cell responses (39, 40). This finding implies that most HCV-specific CD8+ T-cells in the early acute phase of HCV infection are short-lived and furthermore, they are antigen-dependent effector cells rather than self-renewing memory T-cells (40).
As described above, acute HCV infection is often asymptomatic and induces mild inflammation in the liver. In rare cases, however, acute HCV infection results in severe hepatitis, which is known to be mediated by immune responses. In an interesting report regarding two rare severe cases of acute HCV infection, vigorous CD8+ T-cell responses were found to be narrowly focused against HCV NS31073 peptide. Further, HCV NS31073-specific CD8+ T-cells were cross-reactive with a peptide from influenza virus (41), though it was shown later that this cross-reactivity is relatively weak (42). It was inferred that influenza virus peptide-specific CD8+ memory T-cells generated during the previous influenza infection were vigorously activated due to cross-reactivity with HCV and subsequently induced severe liver damage (43). One striking feature of the clinical courses of these two patients was the development chronic HCV infection after severe acute infection in spite of the fact that they exhibited vigorous T-cell responses (41). This finding suggests that the vigor of HCV-specific CD8+ T-cells is not the sole factor determining the outcome of acute HCV infection.
Receptor Expression on HCV-Specific CD8+ T-Cells
In chronic HCV infection, HCV-specific T-cells are functionally impaired, as evidenced by decreased proliferation, cytokine production, and cytolytic activity (44, 45). This impaired function has been attributed to upregulation of inhibitory receptors such as programed cell death-1 (PD-1) (46–53), cytotoxic T-lymphocyte-associated antigen 4 (CTLA-4) (47, 48), T-cell immunoglobulin and mucin domain-containing molecule 3 (Tim-3) (49, 50), 2B4, CD160, and killer cell lectin-like receptor G1 (KLRG1) (51). In chronically HCV-infected patients, HCV-specific CD127loCD8+ T-cells co-expressed the inhibitory receptors PD-1, 2B4, CD160, and KLRG1 (51), and their expression was associated with the absence of viral escape mutations within the corresponding epitopes, indicating that continuous antigenic stimulation is required for the expression of the inhibitory receptors (51). In this report, HCV-specific CD127loCD8+ T-cells could be partly, but not completely, restored functionally by PD-1 blockade alone, suggesting the role of multiple inhibitory receptors in T-cell dysfunction during chronic HCV infection (51).
However, the expression and role of inhibitory receptors has been controversial in acute HCV infection. Initially, high levels of PD-1 on HCV-specific T-cells and PD-1 mRNA in the liver were reported in patients (54) and chimpanzees (55) with acute HCV infection that later progressed to chronic persistent infection. In contrast, other studies showed that PD-1 expression is high on HCV-specific T-cells in acute HCV infection irrespective of the outcome of infection (56, 57). A recent study focused on this issue using chimpanzees acutely infected with monoclonal HCV (37). In this study, the PD-1 level on HCV-specific CD8+ T-cells peaked after appearance of HCV-specific CD8+ T-cells in the blood and decreased thereafter irrespective of the outcome of infection. In addition, intrahepatic mRNA levels of inhibitory receptor genes did not differ among chimpanzees with divergent outcomes of infection. Moreover, upregulation of PD-1 on HCV-specific CD8+ T-cells during acute HCV infection did not interfere with HCV clearance in spontaneously recovered hosts. Thus, following functional analyses of HCV-specific CD8+ T-cells, it was proposed that PD-1 is an activation marker, rather than an exhaustion marker, in acute HCV infection. This is consistent with the fact that PD-1 expression can be transiently induced by T-cell receptor and/or cytokine stimulation (58, 59).
In contrast to naïve chimpanzees, vaccinated chimpanzees have a different expression profile of PD-1 during acute HCV infection. In a study of chimpanzees vaccinated with recombinant adenovirus and DNA against HCV non-structural proteins, HCV-specific CD8+ T-cells in vaccinated animals displayed lower levels of PD-1 than those in the mock-vaccinated control animals (35). It was interpreted that early control of viremia by vaccine-induced T-cells prevented upregulation of PD-1 on HCV-specific CD8+ T-cells. Hence, the low expression of PD-1 might be an indicator of early viral control in vaccinated hosts (35). Taken together, it seems that the functional significance of PD-1 expression on virus-specific CD8+ T-cells differs between acute and chronic HCV infections, and between HCV infections with or without vaccination.
Interestingly, the outcome of acute HCV infection was related to the expression of CD127, also known as IL-7Rα, a marker of memory precursor cells (60). Indeed, a high frequency of CD127+ cells among HCV-specific CD8+ T-cells was observed in hosts that subsequently cleared the acute infection even when virus had been detectable in the blood. However, the frequency of CD127+ cells was low in hosts with chronically evolving infection. This is consistent with the fact that CD127 expression is decreased on HCV-specific T-cells in patients who were studied late in the course of acute HCV infection (9, 51, 61–63). Taken together, the frequency of HCV-specific CD127+CD8+ T-cells predicts the outcome of acute HCV infection when hosts are still viremic (37).
Breadth of CD8+ T-Cell Responses and Viral Mutation
The breadth of CD8+ T-cell responses is an important factor in viral clearance during acute HCV infection (6, 64). Multiple epitopes-specific CD8+ T-cell responses contribute to viral clearance by suppressing the emergence of viral mutations. In fact, HCV-specific CD8+ T-cell responses are narrow over the chronically evolving course of acute HCV infection (6, 65). In contrast, a prospective study with chimpanzees showed that the animals with self-limited acute HCV infection exhibited CD8+ T-cell responses against multiple epitopes (10). During acute HCV infection, the broadest CD8+ T-cell responses occur during the early phase with a subsequent decline in the breadth of response (64, 66). For example, in a longitudinal study of acutely infected injection drug users, patients with progression to chronic infection lost recognition of one or more T-cell epitopes recognized during acute infection (64).
Acute HCV infection progresses to a chronic persistent infection if rapidly generated viral mutations overwhelm and outpace the CD8+ T-cell response. In the initial study with HCV-infected chimpanzees, it was demonstrated that the emergence of viral escape mutants in CD8+ T-cell epitopes correlated with viral persistence (67). A more recent study defined escape mutations in multiple CD8+ T-cell epitopes in patients that developed chronic infection. In this study, patients that cleared HCV had no substitutions within any recognized T-cell epitopes after the initial viremia (68).
During acute HCV infection, virus evolution is driven primarily by positive selection pressure exerted by CD8+ T-cells. This influence of immune pressure on viral evolution appears to subside as chronic infection is established (69). Recently, it was reported that the immunoregulatory changes of pregnancy reduce the selective pressure of HCV-specific CD8+ T-cells on T-cell epitopes, thereby facilitating vertical transmission of viruses with optimized replicative fitness (70). Thus, a strong CD8+ T-cell response eradicates HCV infection and a weak response will cause few mutations with optimized replication fitness. An intermediate immune response, which is usually the case, can stress the virus and select for escape mutants, resulting in viral persistence and a broader viral quasispecies (71).
Conclusion
In this review, we have summarized various aspects of CD8+ T-cell responses in acute HCV infection. Acute HCV infection is characterized by delayed induction of virus-specific CD8+ T-cells, which cause the late onset of inflammation and viral control. In acute HCV infection, the expression of inhibitory receptors such as PD-1 is not related to T-cell exhaustion, while the expression of CD127 predicts the outcome of infection. In addition, the strength and breadth of HCV-specific CD8+ T-cell responses are associated with the outcome of acute HCV infection.
In previous studies, the nature of HCV-specific CD8+ T-cells has been examined using T-cells obtained from patients or chimpanzees with HCV infection. However, HCV-infected cells also need to be studied in the context of interaction between effector CD8+ T-cells and target cells. In particular, the regulation of the antigen processing and presentation machineries in HCV-infected cells is of interest, including immunoproteasomes (28), proteasome activators (72), and MHC molecules. In fact, a recent study demonstrated that HCV attenuates IFN-induced expression of MHC class I molecules, which are required for the recognition of virus-infected cells by CD8+ T-cells. This was suggested as a mechanism of evasion from antiviral CD8+ T-cell responses (73).
Currently, T-cell vaccines against HCV infection are now being developed. In particular, a result of phase I study on recombinant adenoviral vector-based vaccines was recently reported (74). Adenoviral vectors expressing non-structural proteins induced T-cell responses against multiple HCV proteins, and vaccine-induced T-cells recognized heterologous strains. Moreover, they were polyfunctional and sustained for a prolonged period (74). These vaccine candidates might be useful for both prophylactic and therapeutic purposes.
In conclusion, spontaneous recovery from acute HCV infection is associated with effective CD8+ T-cell responses. In contrast, progression to chronic persistent infection is associated with the impaired function of CD8+ T-cells and immune-evading viral mutations. Therefore, the induction of early and robust CD8+ T-cell responses will be a focus for development of successful HCV vaccines in the future.
Author Contributions
Substantial contributions to the conception or design of the work; Pil Soo Sung, Vito Racanelli, Eui-Cheol Shin. Drafting the work or revising it critically for important intellectual content; Pil Soo Sung, Vito Racanelli, Eui-Cheol Shin. Final approval of the version to be published; Vito Racanelli, Eui-Cheol Shin.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
This work was supported by the National Agenda Project grant from Korea Research Council of Fundamental Science and Technology (NTM1311423), the Korea Research Institute for Bioscience and Biotechnology (KRIBB) Initiative program (KGM3121423), and a grant of the Korean Health Technology R&D Project, Ministry of Health and Welfare, South Korea (HI13C1263). This work was also partly supported by the Korea Advanced Institute of Science and Technology (KAIST) Future Systems Healthcare Project from the Ministry of Science, ICT and Future Planning of Korea.
References
1. Shepard CW, Finelli L, Alter MJ. Global epidemiology of hepatitis C virus infection. Lancet Infect Dis (2005) 5(9):558–67. doi:10.1016/S1473-3099(05)70216-4
2. Lavanchy D. Evolving epidemiology of hepatitis C virus. Clin Microbiol Infect (2011) 17(2):107–15. doi:10.1111/j.1469-0691.2010.03432.x
3. Afdhal NH. The natural history of hepatitis C. Semin Liver Dis (2004) 24(Suppl 2):3–8. doi:10.1055/s-2004-832922
4. Bunchorntavakul C, Jones LM, Kikuchi M, Valiga ME, Kaplan DE, Nunes FA, et al. Distinct features in natural history and outcomes of acute hepatitis C. J Clin Gastroenterol (2014). doi:10.1097/MCG.0000000000000076
5. Kaplan DE, Sugimoto K, Newton K, Valiga ME, Ikeda F, Aytaman A, et al. Discordant role of CD4 T-cell response relative to neutralizing antibody and CD8 T-cell responses in acute hepatitis C. Gastroenterology (2007) 132(2):654–66. doi:10.1053/j.gastro.2006.11.044
6. Lechner F, Wong DK, Dunbar PR, Chapman R, Chung RT, Dohrenwend P, et al. Analysis of successful immune responses in persons infected with hepatitis C virus. J Exp Med (2000) 191(9):1499–512. doi:10.1084/jem.191.9.1499
7. Smyk-Pearson S, Tester IA, Klarquist J, Palmer BE, Pawlotsky JM, Golden-Mason L, et al. Spontaneous recovery in acute human hepatitis C virus infection: functional T-cell thresholds and relative importance of CD4 help. J Virol (2008) 82(4):1827–37. doi:10.1128/JVI.01581-07
8. Thimme R, Oldach D, Chang KM, Steiger C, Ray SC, Chisari FV. Determinants of viral clearance and persistence during acute hepatitis C virus infection. J Exp Med (2001) 194(10):1395–406. doi:10.1084/jem.194.10.1395
9. Urbani S, Amadei B, Fisicaro P, Tola D, Orlandini A, Sacchelli L, et al. Outcome of acute hepatitis C is related to virus-specific CD4 function and maturation of antiviral memory CD8 responses. Hepatology (2006) 44(1):126–39. doi:10.1002/hep.21242
10. Thimme R, Bukh J, Spangenberg HC, Wieland S, Pemberton J, Steiger C, et al. Viral and immunological determinants of hepatitis C virus clearance, persistence, and disease. Proc Natl Acad Sci U S A (2002) 99(24):15661–8. doi:10.1073/pnas.202608299
11. Major ME, Dahari H, Mihalik K, Puig M, Rice CM, Neumann AU, et al. Hepatitis C virus kinetics and host responses associated with disease and outcome of infection in chimpanzees. Hepatology (2004) 39(6):1709–20. doi:10.1002/hep.20239
12. Frese M, Schwarzle V, Barth K, Krieger N, Lohmann V, Mihm S, et al. Interferon-gamma inhibits replication of subgenomic and genomic hepatitis C virus RNAs. Hepatology (2002) 35(3):694–703. doi:10.1053/jhep.2002.31770
13. Cheney IW, Lai VC, Zhong W, Brodhag T, Dempsey S, Lim C, et al. Comparative analysis of anti-hepatitis C virus activity and gene expression mediated by alpha, beta, and gamma interferons. J Virol (2002) 76(21):11148–54. doi:10.1128/JVI.76.21.11148-11154.2002
14. Jo J, Aichele U, Kersting N, Klein R, Aichele P, Bisse E, et al. Analysis of CD8+ T-cell-mediated inhibition of hepatitis C virus replication using a novel immunological model. Gastroenterology (2009) 136(4):1391–401. doi:10.1053/j.gastro.2008.12.034
15. Cooper S, Erickson AL, Adams EJ, Kansopon J, Weiner AJ, Chien DY, et al. Analysis of a successful immune response against hepatitis C virus. Immunity (1999) 10(4):439–49. doi:10.1016/S1074-7613(00)80044-8
16. Shoukry NH, Grakoui A, Houghton M, Chien DY, Ghrayeb J, Reimann KA, et al. Memory CD8+ T cells are required for protection from persistent hepatitis C virus infection. J Exp Med (2003) 197(12):1645–55. doi:10.1084/jem.20030239
17. McKiernan SM, Hagan R, Curry M, McDonald GS, Kelly A, Nolan N, et al. Distinct MHC class I and II alleles are associated with hepatitis C viral clearance, originating from a single source. Hepatology (2004) 40(1):108–14. doi:10.1002/hep.20261
18. Fitzmaurice K, Petrovic D, Ramamurthy N, Simmons R, Merani S, Gaudieri S, et al. Molecular footprints reveal the impact of the protective HLA-A*03 allele in hepatitis C virus infection. Gut (2011) 60(11):1563–71. doi:10.1136/gut.2010.228403
19. Kim AY, Kuntzen T, Timm J, Nolan BE, Baca MA, Reyor LL, et al. Spontaneous control of HCV is associated with expression of HLA-B 57 and preservation of targeted epitopes. Gastroenterology (2011) 140(2):686.e–96.e. doi:10.1053/j.gastro.2010.09.042
20. Deterding K, Wiegand J, Gruner N, Hahn A, Jackel E, Jung MC, et al. The German Hep-Net acute hepatitis C cohort: impact of viral and host factors on the initial presentation of acute hepatitis C virus infection. Z Gastroenterol (2009) 47(6):531–40. doi:10.1055/s-0028-1109149
21. Maheshwari A, Ray S, Thuluvath PJ. Acute hepatitis C. Lancet (2008) 372(9635):321–32. doi:10.1016/S0140-6736(08)61116-2
22. Kolykhalov AA, Agapov EV, Blight KJ, Mihalik K, Feinstone SM, Rice CM. Transmission of hepatitis C by intrahepatic inoculation with transcribed RNA. Science (1997) 277(5325):570–4. doi:10.1126/science.277.5325.570
23. Su AI, Pezacki JP, Wodicka L, Brideau AD, Supekova L, Thimme R, et al. Genomic analysis of the host response to hepatitis C virus infection. Proc Natl Acad Sci U S A (2002) 99(24):15669–74. doi:10.1073/pnas.202608199
24. Wieland S, Thimme R, Purcell RH, Chisari FV. Genomic analysis of the host response to hepatitis B virus infection. Proc Natl Acad Sci U S A (2004) 101(17):6669–74. doi:10.1073/pnas.0401771101
25. Wieland SF, Chisari FV. Stealth and cunning: hepatitis B and hepatitis C viruses. J Virol (2005) 79(15):9369–80. doi:10.1128/JVI.79.15.9369-9380.2005
26. Bigger CB, Brasky KM, Lanford RE. DNA microarray analysis of chimpanzee liver during acute resolving hepatitis C virus infection. J Virol (2001) 75(15):7059–66. doi:10.1128/JVI.75.15.7059-7066.2001
27. Shin EC, Capone S, Cortese R, Colloca S, Nicosia A, Folgori A, et al. The kinetics of hepatitis C virus-specific CD8 T-cell responses in the blood mirror those in the liver in acute hepatitis C virus infection. J Virol (2008) 82(19):9782–8. doi:10.1128/JVI.00475-08
28. Shin EC, Seifert U, Kato T, Rice CM, Feinstone SM, Kloetzel PM, et al. Virus-induced type I IFN stimulates generation of immunoproteasomes at the site of infection. J Clin Invest (2006) 116(11):3006–14. doi:10.1172/JCI29832
29. Shin EC, Park SH, Demino M, Nascimbeni M, Mihalik K, Major M, et al. Delayed induction, not impaired recruitment, of specific CD8(+) T cells causes the late onset of acute hepatitis C. Gastroenterology (2011) 141(2):686–95. doi:10.1053/j.gastro.2011.05.006
30. Racanelli V, Behrens SE, Aliberti J, Rehermann B. Dendritic cells transfected with cytopathic self-replicating RNA induce crosspriming of CD8+ T cells and antiviral immunity. Immunity (2004) 20(1):47–58. doi:10.1016/S1074-7613(03)00353-4
31. Bassett SE, Guerra B, Brasky K, Miskovsky E, Houghton M, Klimpel GR, et al. Protective immune response to hepatitis C virus in chimpanzees rechallenged following clearance of primary infection. Hepatology (2001) 33(6):1479–87. doi:10.1053/jhep.2001.24371
32. Major ME, Mihalik K, Puig M, Rehermann B, Nascimbeni M, Rice CM, et al. Previously infected and recovered chimpanzees exhibit rapid responses that control hepatitis C virus replication upon rechallenge. J Virol (2002) 76(13):6586–95. doi:10.1128/JVI.76.13.6586-6595.2002
33. Nascimbeni M, Mizukoshi E, Bosmann M, Major ME, Mihalik K, Rice CM, et al. Kinetics of CD4+ and CD8+ memory T-cell responses during hepatitis C virus rechallenge of previously recovered chimpanzees. J Virol (2003) 77(8):4781–93. doi:10.1128/JVI.77.8.4781-4793.2003
34. Folgori A, Capone S, Ruggeri L, Meola A, Sporeno E, Ercole BB, et al. A T-cell HCV vaccine eliciting effective immunity against heterologous virus challenge in chimpanzees. Nat Med (2006) 12(2):190–7. doi:10.1038/nm1353
35. Park SH, Shin EC, Capone S, Caggiari L, De Re V, Nicosia A, et al. Successful vaccination induces multifunctional memory T-cell precursors associated with early control of hepatitis C virus. Gastroenterology (2012) 143(4):1048.e–60.e. doi:10.1053/j.gastro.2012.06.005
36. Diepolder HM, Zachoval R, Hoffmann RM, Wierenga EA, Santantonio T, Jung MC, et al. Possible mechanism involving T-lymphocyte response to non-structural protein 3 in viral clearance in acute hepatitis C virus infection. Lancet (1995) 346(8981):1006–7. doi:10.1016/S0140-6736(95)91691-1
37. Shin EC, Park SH, Nascimbeni M, Major M, Caggiari L, de Re V, et al. The frequency of CD127(+) hepatitis C virus (HCV)-specific T cells but not the expression of exhaustion markers predicts the outcome of acute HCV infection. J Virol (2013) 87(8):4772–7. doi:10.1128/JVI.03122-12
38. Thomson EC, Fleming VM, Main J, Klenerman P, Weber J, Eliahoo J, et al. Predicting spontaneous clearance of acute hepatitis C virus in a large cohort of HIV-1-infected men. Gut (2011) 60(6):837–45. doi:10.1136/gut.2010.217166
39. Rahman F, Heller T, Sobao Y, Mizukoshi E, Nascimbeni M, Alter H, et al. Effects of antiviral therapy on the cellular immune response in acute hepatitis C. Hepatology (2004) 40(1):87–97. doi:10.1002/hep.20253
40. Park SH, Rehermann B. Immune responses to HCV and other hepatitis viruses. Immunity (2014) 40(1):13–24. doi:10.1016/j.immuni.2013.12.010
41. Urbani S, Amadei B, Fisicaro P, Pilli M, Missale G, Bertoletti A, et al. Heterologous T cell immunity in severe hepatitis C virus infection. J Exp Med (2005) 201(5):675–80. doi:10.1084/jem.20041058
42. Kasprowicz V, Ward SM, Turner A, Grammatikos A, Nolan BE, Lewis-Ximenez L, et al. Defining the directionality and quality of influenza virus-specific CD8+ T cell cross-reactivity in individuals infected with hepatitis C virus. J Clin Invest (2008) 118(3):1143–53. doi:10.1172/JCI33082
43. Rehermann B, Shin EC. Private aspects of heterologous immunity. J Exp Med (2005) 201(5):667–70. doi:10.1084/jem.20050220
44. Spangenberg HC, Viazov S, Kersting N, Neumann-Haefelin C, McKinney D, Roggendorf M, et al. Intrahepatic CD8+ T-cell failure during chronic hepatitis C virus infection. Hepatology (2005) 42(4):828–37. doi:10.1002/hep.20856
45. Wedemeyer H, He XS, Nascimbeni M, Davis AR, Greenberg HB, Hoofnagle JH, et al. Impaired effector function of hepatitis C virus-specific CD8+ T cells in chronic hepatitis C virus infection. J Immunol (2002) 169(6):3447–58. doi:10.4049/jimmunol.169.6.3447
46. Golden-Mason L, Palmer B, Klarquist J, Mengshol JA, Castelblanco N, Rosen HR. Upregulation of PD-1 expression on circulating and intrahepatic hepatitis C virus-specific CD8+ T cells associated with reversible immune dysfunction. J Virol (2007) 81(17):9249–58. doi:10.1128/JVI.00409-07
47. Nakamoto N, Cho H, Shaked A, Olthoff K, Valiga ME, Kaminski M, et al. Synergistic reversal of intrahepatic HCV-specific CD8 T cell exhaustion by combined PD-1/CTLA-4 blockade. PLoS Pathog (2009) 5(2):e1000313. doi:10.1371/journal.ppat.1000313
48. Nakamoto N, Kaplan DE, Coleclough J, Li Y, Valiga ME, Kaminski M, et al. Functional restoration of HCV-specific CD8 T cells by PD-1 blockade is defined by PD-1 expression and compartmentalization. Gastroenterology (2008) 134(7):1927–37. doi:10.1053/j.gastro.2008.02.033
49. Golden-Mason L, Palmer BE, Kassam N, Townshend-Bulson L, Livingston S, McMahon BJ, et al. Negative immune regulator Tim-3 is overexpressed on T cells in hepatitis C virus infection and its blockade rescues dysfunctional CD4+ and CD8+ T cells. J Virol (2009) 83(18):9122–30. doi:10.1128/JVI.00639-09
50. McMahan RH, Golden-Mason L, Nishimura MI, McMahon BJ, Kemper M, Allen TM, et al. Tim-3 expression on PD-1+ HCV-specific human CTLs is associated with viral persistence, and its blockade restores hepatocyte-directed in vitro cytotoxicity. J Clin Invest (2010) 120(12):4546–57. doi:10.1172/JCI43127
51. Bengsch B, Seigel B, Ruhl M, Timm J, Kuntz M, Blum HE, et al. Coexpression of PD-1, 2B4, CD160 and KLRG1 on exhausted HCV-specific CD8+ T cells is linked to antigen recognition and T cell differentiation. PLoS Pathog (2010) 6(6):e1000947. doi:10.1371/journal.ppat.1000947
52. Penna A, Pilli M, Zerbini A, Orlandini A, Mezzadri S, Sacchelli L, et al. Dysfunction and functional restoration of HCV-specific CD8 responses in chronic hepatitis C virus infection. Hepatology (2007) 45(3):588–601. doi:10.1002/hep.21541
53. Radziewicz H, Ibegbu CC, Fernandez ML, Workowski KA, Obideen K, Wehbi M, et al. Liver-infiltrating lymphocytes in chronic human hepatitis C virus infection display an exhausted phenotype with high levels of PD-1 and low levels of CD127 expression. J Virol (2007) 81(6):2545–53. doi:10.1128/JVI.02021-06
54. Rutebemberwa A, Ray SC, Astemborski J, Levine J, Liu L, Dowd KA, et al. High-programmed death-1 levels on hepatitis C virus-specific T cells during acute infection are associated with viral persistence and require preservation of cognate antigen during chronic infection. J Immunol (2008) 181(12):8215–25. doi:10.4049/jimmunol.181.12.8215
55. Rollier CS, Paranhos-Baccala G, Verschoor EJ, Verstrepen BE, Drexhage JA, Fagrouch Z, et al. Vaccine-induced early control of hepatitis C virus infection in chimpanzees fails to impact on hepatic PD-1 and chronicity. Hepatology (2007) 45(3):602–13. doi:10.1002/hep.21573
56. Kasprowicz V, Schulze Zur Wiesch J, Kuntzen T, Nolan BE, Longworth S, Berical A, et al. High level of PD-1 expression on hepatitis C virus (HCV)-specific CD8+ and CD4+ T cells during acute HCV infection, irrespective of clinical outcome. J Virol (2008) 82(6):3154–60. doi:10.1128/JVI.02474-07
57. Urbani S, Amadei B, Tola D, Massari M, Schivazappa S, Missale G, et al. PD-1 expression in acute hepatitis C virus (HCV) infection is associated with HCV-specific CD8 exhaustion. J Virol (2006) 80(22):11398–403. doi:10.1128/JVI.01177-06
58. Agata Y, Kawasaki A, Nishimura H, Ishida Y, Tsubata T, Yagita H, et al. Expression of the PD-1 antigen on the surface of stimulated mouse T and B lymphocytes. Int Immunol (1996) 8(5):765–72. doi:10.1093/intimm/8.5.765
59. Kinter AL, Godbout EJ, McNally JP, Sereti I, Roby GA, O’Shea MA, et al. The common gamma-chain cytokines IL-2, IL-7, IL-15, and IL-21 induce the expression of programmed death-1 and its ligands. J Immunol (2008) 181(10):6738–46. doi:10.4049/jimmunol.181.10.6738
60. Kaech SM, Tan JT, Wherry EJ, Konieczny BT, Surh CD, Ahmed R. Selective expression of the interleukin 7 receptor identifies effector CD8 T cells that give rise to long-lived memory cells. Nat Immunol (2003) 4(12):1191–8. doi:10.1038/ni1009
61. Kasprowicz V, Kang YH, Lucas M, Schulze zur Wiesch J, Kuntzen T, Fleming V, et al. Hepatitis C virus (HCV) sequence variation induces an HCV-specific T-cell phenotype analogous to spontaneous resolution. J Virol (2010) 84(3):1656–63. doi:10.1128/JVI.01499-09
62. Bengsch B, Spangenberg HC, Kersting N, Neumann-Haefelin C, Panther E, von Weizsacker F, et al. Analysis of CD127 and KLRG1 expression on hepatitis C virus-specific CD8+ T cells reveals the existence of different memory T-cell subsets in the peripheral blood and liver. J Virol (2007) 81(2):945–53. doi:10.1128/JVI.01354-06
63. Golden-Mason L, Burton JR Jr, Castelblanco N, Klarquist J, Benlloch S, Wang C, et al. Loss of IL-7 receptor alpha-chain (CD127) expression in acute HCV infection associated with viral persistence. Hepatology (2006) 44(5):1098–109. doi:10.1002/hep.21365
64. Cox AL, Mosbruger T, Lauer GM, Pardoll D, Thomas DL, Ray SC. Comprehensive analyses of CD8+ T cell responses during longitudinal study of acute human hepatitis C. Hepatology (2005) 42(1):104–12. doi:10.1002/hep.20749
65. Gruner NH, Gerlach TJ, Jung MC, Diepolder HM, Schirren CA, Schraut WW, et al. Association of hepatitis C virus-specific CD8+ T cells with viral clearance in acute hepatitis C. J Infect Dis (2000) 181(5):1528–36. doi:10.1086/315450
66. Lechner F, Gruener NH, Urbani S, Uggeri J, Santantonio T, Kammer AR, et al. CD8+ T lymphocyte responses are induced during acute hepatitis C virus infection but are not sustained. Eur J Immunol (2000) 30(9):2479–87. doi:10.1002/1521-4141(200009)30:9<2479::AID-IMMU2479>3.0.CO;2-B
67. Erickson AL, Kimura Y, Igarashi S, Eichelberger J, Houghton M, Sidney J, et al. The outcome of hepatitis C virus infection is predicted by escape mutations in epitopes targeted by cytotoxic T lymphocytes. Immunity (2001) 15(6):883–95. doi:10.1016/S1074-7613(01)00245-X
68. Cox AL, Mosbruger T, Mao Q, Liu Z, Wang XH, Yang HC, et al. Cellular immune selection with hepatitis C virus persistence in humans. J Exp Med (2005) 201(11):1741–52. doi:10.1084/jem.20050121
69. Callendret B, Bukh J, Eccleston HB, Heksch R, Hasselschwert DL, Purcell RH, et al. Transmission of clonal hepatitis C virus genomes reveals the dominant but transitory role of CD8(+) T cells in early viral evolution. J Virol (2011) 85(22):11833–45. doi:10.1128/JVI.02654-10
70. Honegger JR, Kim S, Price AA, Kohout JA, McKnight KL, Prasad MR, et al. Loss of immune escape mutations during persistent HCV infection in pregnancy enhances replication of vertically transmitted viruses. Nat Med (2013) 19(11):1529–33. doi:10.1038/nm.3351
71. Rosen HR. Emerging concepts in immunity to hepatitis C virus infection. J Clin Invest (2013) 123(10):4121–30. doi:10.1172/JCI67714
72. Shin EC, Seifert U, Urban S, Truong KT, Feinstone SM, Rice CM, et al. Proteasome activator and antigen-processing aminopeptidases are regulated by virus-induced type I interferon in the hepatitis C virus-infected liver. J Interferon Cytokine Res (2007) 27(12):985–90. doi:10.1089/jir.2007.0039
73. Kang W, Sung PS, Park SH, Yoon S, Chang DY, Kim S, et al. Hepatitis C virus attenuates interferon-induced major histocompatibility complex class I expression and decreases CD8+ T cell effector functions. Gastroenterology (2014) 146(5):1351–60.e1–4. doi:10.1053/j.gastro.2014.01.054
Keywords: hepatitis C virus, acute infection, outcome, CD8+ T-cell, function
Citation: Sung PS, Racanelli V and Shin E-C (2014) CD8+ T-cell responses in acute hepatitis C virus infection. Front. Immunol. 5:266. doi: 10.3389/fimmu.2014.00266
Received: 31 March 2014; Accepted: 23 May 2014;
Published online: 06 June 2014.
Edited by:
Lynn B. Dustin, University of Oxford, UKReviewed by:
Paul Klenerman, University of Oxford, UKNaglaa H. Shoukry, Centre de Recherche du Centre Hospitalier de l’Université de Montréal, Canada
Copyright: © 2014 Sung, Racanelli and Shin. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Vito Racanelli, Department of Internal Medicine and Clinical Oncology, University of Bari Medical School, Policlinico, 11 Piazza G. Cesare, Bari 70124, Italy e-mail:dml0by5yYWNhbmVsbGkxQHVuaWJhLml0;
Eui-Cheol Shin, Laboratory of Immunology and Infectious Diseases, Graduate School of Medical Science and Engineering, KAIST, 291 Daehak-ro, Yuseong-gu, Daejeon 305-701, South Korea e-mail:ZWNzaGluQGthaXN0LmFjLmty
 Pil Soo Sung
Pil Soo Sung Vito Racanelli
Vito Racanelli Eui-Cheol Shin
Eui-Cheol Shin