- Section of Aquatic Medicine and Nutrition, Department of Basic Sciences and Aquatic Medicine, Faculty of Veterinary Medicine and Biosciences, Norwegian University of Life Sciences, Oslo, Norway
Mucosal organs are principle portals of entry for microbial invasion and as such developing protective vaccines against these pathogens can serve as a first line of defense against infections. In general, all mucosal organs in finfish are covered by a layer of mucus whose main function is not only to prevent pathogen attachment by being continuously secreted and sloughing-off but it serves as a vehicle for antimicrobial compounds, complement, and immunoglobulins that degrade, opsonize, and neutralize invading pathogens on mucosal surfaces. In addition, all mucosal organs in finfish possess antigen-presenting cells (APCs) that activate cells of the adaptive immune system to generate long-lasting protective immune responses. The functional activities of APCs are orchestrated by a vast array of proinflammatory cytokines and chemokines found in all mucosal organs. The adaptive immune system in mucosal organs is made of humoral immune responses that are able to neutralize invading pathogens as well as cellular-mediated immune responses whose kinetics are comparable to those induced by parenteral vaccines. In general, finfish mucosal immune system has the capacity to serve as the first-line defense mechanism against microbial invasion as well as being responsive to vaccination.
Introduction
Mucosal surfaces are important physical barriers whose main function is to protect the systemic environment of the body against microbial invasion. An ideal mucosal vaccine should have the capacity to produce protective immunity that is able to prevent microbial invasion, colonization, and establishment of infection at portals of entry (1). All mucosal organs are endowed with antigen-presenting cells (APCs) that play a vital role in antigen uptake and processing followed by presentation to naïve B- and T-lymphocytes to induce a long-lasting protective immunity (2). Despite so, our understanding of the immunological basis of mucosal vaccine protection has for a long time lagged behind systemic immunity not only in finfish, but also in higher vertebrates, which has led to a corresponding delay in developing highly protective mucosal vaccines across the vertebrate taxa.
The demand for mucosal vaccines in aquaculture has been exacerbated by different stages of fish production cycles in which administering vaccines by injection might not be feasible thereby rendering the use of mucosal vaccines as an alternative. For Atlantic salmon (Salmo salar L), administering vaccines by injection at the freshwater stage is the most commonly applied method while boost vaccination for fish in cages at sea is only applicable by oral vaccination. On the other hand, vaccinating small fish by injection causes stress-related mortalities, which make immersion vaccination a better alternative. While the mode of vaccine delivery is to a large extent dependent on the fish production cycle, developing highly protective vaccines for oral and immersion vaccination has been a serious challenge for a long time because the process of optimizing vaccine delivery methods and measuring immune responses for mucosal vaccines is more complicated than for injectable vaccines (3). For example, the vaccine dose taken up by oral or immersion vaccination is difficult to accurately quantify. Unlike injectable vaccines whose immune response to vaccination is determined by measuring serum antibody levels, there is no optimized quantitative assay established for measuring antibody levels in the mucus of vaccinated fish. Therefore, it is difficult to optimize mucosal vaccine performance for finfish (3). Despite so, gene expression studies show that mucosal vaccines are able to induce immune responses in vaccinated fish. It is anticipated that generating vaccines that have the capacity to induce a combined effect of highly protective mucosal and systemic immune responses could be more effective at attaining sterile immunity. Mucosal immune responses would serve as gatekeepers at the portals of pathogen entry while systemic immunity would serve as a secondary barrier to block the spread of infection to target organs in infected fish.
Considerable progress has been made in optimizating the performance of injectable vaccines in aquaculture (4, 5), but not for mucosal vaccines. However, recent advances in mucosal immunology show that teleosts fish, like all vertebrates, are endowed with a protective immune system although there has been no comprehensive review that puts together a summation of underlying mechanisms of mucosal vaccine protection in finfish, thereby creating the basis for this review. Therefore, this review puts together a collection of different components of the mucosal immune system of finfish with the view to shed insight on how these elements prevent microbial invasion on mucosal surfaces as a basis for designing highly protective mucosal vaccines for finfish.
Immunological Mechanisms of Vaccine Protection in Different Mucosal Organs
Mucosal organs in finfish have been classified into four broad categories, namely the gut, gills, skin, and nasal mucosa by different scientists (6–9). Hence, in this review, we discuss the immunological basis of mucosal vaccine protection based on this classification.
Gut Mucosal Responses to Vaccination
The gut immune system of finfish is made of two components, namely the innate and adaptive immune system whose immune responses to vaccination are discussed below.
Innate Immune Responses to Mucosal Vaccination in the Gut
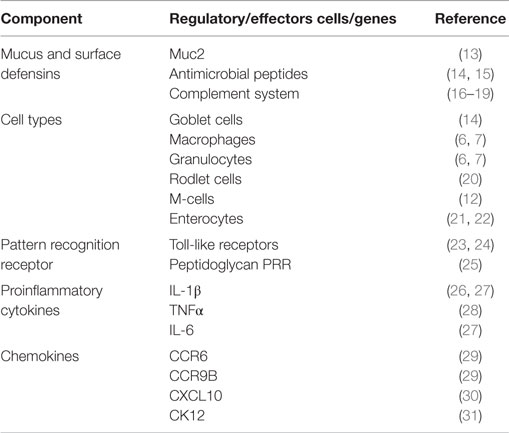
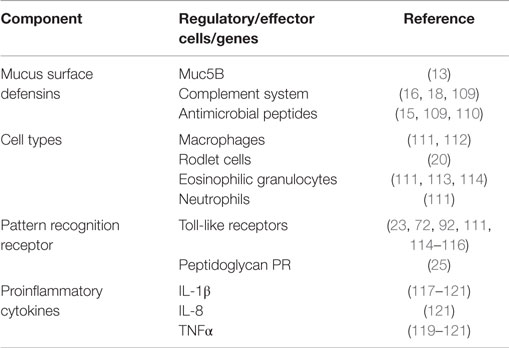
A layer of mucus containing antimicrobial peptides, complement factors, immunoglobulins, and other surface defensins covers the mucosal surface of the gut (Table 1). Rombout et al. (6) have recently reviewed the type of immune cells found in the gut of fish. APCs, such as monocytes and macrophages, are found in the lamina propria (LP) while the intra epithelial lymphocytes (IELs) mainly comprises of B and T-lymphocytes. Zhang et al. (10) and Li et al. (11) have shown that teleosts B-cells possess phagocytic properties that play an important role as APCs. It has been shown that IgT coats gut commensal bacteria in a similar pattern as IgA in mammals (10). In addition, Fuglem et al. (12) have reported of M-cells and dendritic-like cells in the gut of salmonid intestinal epithelium.
Martin et al. (32) compared the antigen uptake ability of intestinal leukocytes with their head-kidney (HK) and peripheral blood (PBL) counterparts and showed that intestinal phagocytes, when activated, ingested as many yeast cells as their HK counterparts, indicating that gut APCs have the same capacity for antigen uptake as their systemic counterparts. Rombout et al. (21, 22) have shown that there are differences in the distribution of APCs between different gut segments with the second segment having more APCs than the first. Similarly, Fuglem et al. (12) also showed that the uptake of gold-BSA was restricted to dendritic-like cells and other epithelial cells located in mucosal folds found in the second gut segment. In another study, Chen et al. (33) recently showed uptake of inactivated infectious pancreatic necrosis virus (IPNV) antigens in the second gut segment following oral and anal intubation, which were also detected in the HK melanomacrophages of Atlantic salmon (S. salar L). Overall, these studies indicate that antigens deposited in the second gut segment were more likely to be taken up by APCs than antigens deposited in other gut segments. Therefore, the challenge is to develop vaccine delivery systems able to deposit antigens in the second gut segment.
Similar to systemic immune responses (5, 34), mucosal antigen uptake is linked to expression of different proinflammatory cytokines, such as IL-1β, TNFα, and IL-8, in the gut (Table 1). In addition, Table 1 shows that chemokines expressed in response to antigen delivery through the gut mucosa are comparable to those induced by parenteral vaccination (35, 36) suggesting that antigen uptake by gut APCs is coordinated by chemokines and cytokines comparable to those induced by systemic antigen delivery systems (35–37). However, there is need for detailed investigations to elucidate the role of these chemokines and cytokines in enhancing the performance of mucosal vaccines in finfish.
Adaptive Immune Responses to Mucosal Vaccination in the Gut
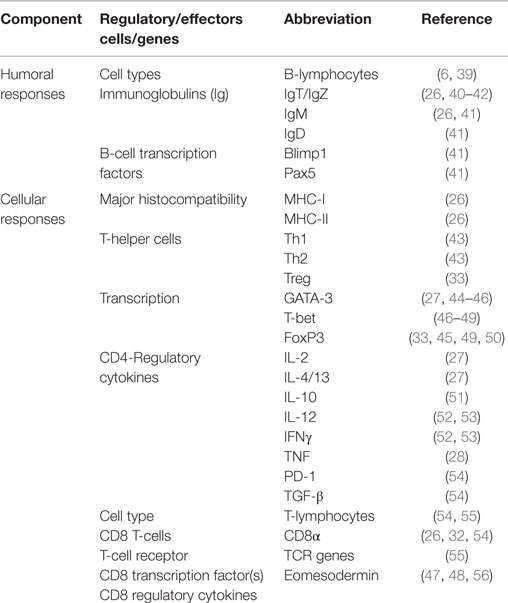
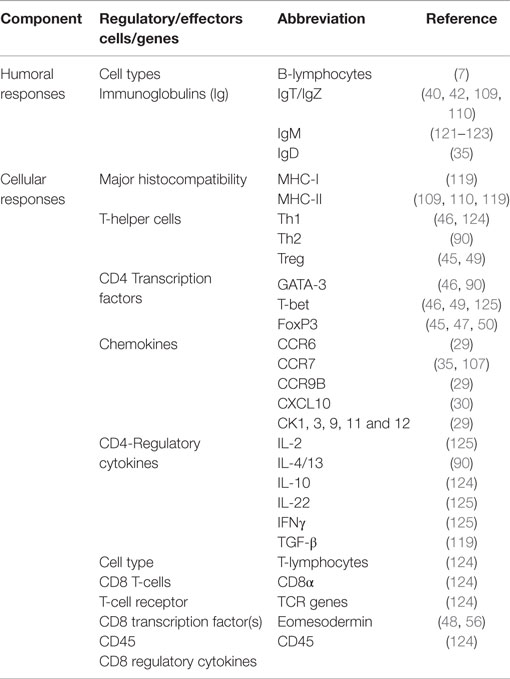
All three immunoglobulin (Ig) isotypes characterized in teleosts fish have been detected in the gut (Table 2). Unlike IgM, IgT is specialized in mucosal surfaces in a similar pattern that IgA functions in mammals and it accounts for the largest proportion of the B-cell population found in the gut of finfish (10). Kai et al. (26) have shown that high IgT levels were only expressed by bath or immersion vaccination unlike IgM that had high levels for vaccines administered by injection (4). Vervarcke et al. (38) compared antibody levels induced by anal intubation of vaccines into the second segment of the gut, oral vaccination through feed and intraperitoneal vaccination against Vibrio anguillarum and observed high antibody levels in the skin mucus and bile for fish vaccinated by intubation and not in the intraperitoneally vaccinated fish. In addition, they detected high antibody levels for fish vaccinated by intubation than the orally vaccinated fish. They further noted that the antigen dose of vaccines administered by anal intubation correlated with post vaccination antibody levels suggesting that deposition of vaccines in the second segment of the gut by anal intubation produced better correlates of vaccine protection than oral vaccination. In summary, these studies show (i) compartmentalization of mucosal responses in which antigens delivered by intubation into the gut mucosa produced high antibody levels in other mucosal organs, which was not the case for vaccines delivered by the intraperitoneal route, (ii) the second segment of the gut is the most antigen absorptive site able to induce high antibody responses, and (iii) antibodies generated by mucosal vaccination were protective against microbial invasion in the gut mucosa.
Several scientists (32, 57, 58) have shown that CD8α, which is a marker for activated cytotoxic T-lymphocytes (CTLs) in fish, accounts for the largest proportion of T-lymphocytes in the gut of teleosts fish. It is interesting to note that upregulation of CD8α cells has been linked to the corresponding upregulation of MHC-I, suggesting that presentation of antigens via the MHC-I pathway could be linked to activation of naïve CD8 cells into effector CTLs in the gut mucosa of finfish. For example, Kai et al. (26) showed upregulation of MHC-I that corresponded with upregulation of CD8α in the gut of grouper larvae (Epinephelus coioides) exposed to nervous necrosis virus (NNV) by oral vaccination, suggesting that activation of CD8α was via the MHC-I pathway. Furthermore, upregulation of T-cell receptor (TCR) and CD3 genes has also been shown to correspond with upregulation of CD8α and MHC-I genes (57, 59, 60). Martin et al. (32) showed a cytotoxic activity that was twice higher in the intestine than in head-kidney leukocytes in rainbow trout while Picchietti et al. (57) showed high cytotoxic activity in lymphocytes purified from the intestinal mucosa, providing further insight into the cellular-mediated activity of activated CTLs in the gut mucosa of finfish. Put together, these studies show that the gut mucosa is endowed with different components of CD8 T-cell responses whose kinetics corresponds with CTL responses shown to eliminate cells infected with intracellular pathogens in higher vertebrates. As for CD4 responses, transcription factors for their specification into Th1 and Th2 subtypes have been characterized in fish and these include T-bet, GATA-3, and FoxP3 (Table 1). Recently, Wang et al. (61) showed that grass carp virus (GCRV) induced upregulation of T-bet when GATA-3 was downregulated in which upregulation of IFNγ correlated with upregulation of T-bet being similar to kinetics induced by parenteral vaccines in finfish (5, 47). Overall, these studies show that the gut mucosa of finfish is endowed with a cellular-mediated immune response orchestrated by CD4 and CD8 genes expressed in response to mucosal vaccination. However, there still remains the challenge of demonstrating the functional role of cellular-mediated immunity in finfish vaccinated by the mucosal route.
Skin Immune Responses to Vaccination
The skin of teleost fish is endowed with different components of the innate and adaptive immune system, which are responsive to vaccination as shown below.
Innate Immune Responses to Mucosal Vaccination in the Skin
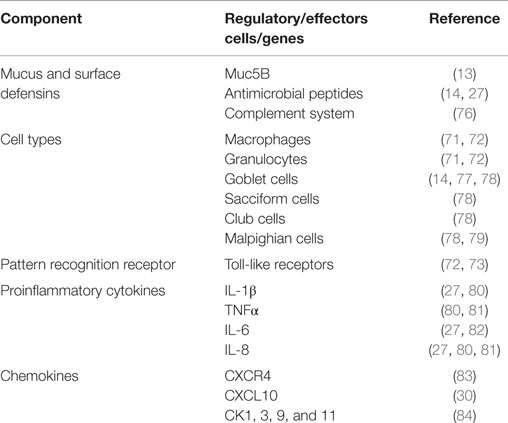
Mucus overlying the epidermis is the first line of defense against microbial invasion on the skin surface. It exerts its protective role by being continuously produced and sloughing off to prevent pathogen adherence. Second, it serves as a vehicle for several immune factors that include lysozomes, proteases, alkaline phosphatases, complement, immunoglobulins, lectins, and C-reactive proteins that prevent pathogen invasion into the skin (14, 15, 62–64). Despite so, bath vaccination studies have shown that the primary site of attachment of V. anguillarum is the skin in which the bacteria cause local inflammatory responses upon attachment (65, 66). This causes the skin epithelial cells to phagocytose pathogens that attach to the skin surface (7, 65). And as such, different phagocytic cells have been characterized that include macrophages, granulocytes, goblet cells, sacciform cells, club cells, and malpighian cells in finfish (Table 3). In addition, dendritic-like cells have also been characterized from the skin mucosa of different fish species (67–69). The presence of monocytes, macrophages, and dendritic-like cells (67–71) indicates that the skin mucosa is endowed with APCs like other mucosal organs found in finfish. Upregulation of different toll-like receptors (TLRs) after pathogen invasions suggests that cells of the skin mucosa have sensors that are able to detect and bind to pathogen-associated molecular patterns (PAMPs) expressed by invading pathogens (72, 73). Li et al. (74) showed upregulation of TLR2 in orange-spotted grouper (E. coioides) exposed to Cryptocaryon irritans infection while Zhao et al. (75) showed increased levels of TLR1, 2, and 19 in response to Ichthyophthirius multifiliis infection in channel catfish (Ictalurus punctatus). It is interesting to note that upregulation of TLRs genes has been shown to correspond with upregulation of proinflammatory genes. For example, Li et al. (74) showed upregulation of TLR2 that coincided with upregulation of IL-1β in orange spotter grouper exposed to C irritans pointing to antigen recognition by APCs orchestrated by proinflammatory genes.
In summary, the skin mucosa of finfish exert its protective mechanisms by (i) having mucus, which prevents pathogen attachment by continuously being produced and sloughing off, (ii) the presence of different host defensins that degrade, opsonize, and neutralize invading pathogens, (iii) carrying out phagocytosis of pathogens that attach onto the skin surface, and (iv) carrying out antigen uptake and processing by APCs that activate the adaptive immune system thereby creating the basis for mucosal vaccination via the skin.
Adaptive Immune Responses to Mucosal Vaccination in the Skin
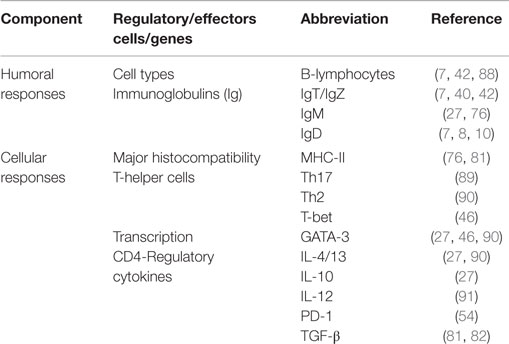
All Ig isotypes characterized in fish have been detected in the skin mucosa (Table 4) and as pointed out by Zhang et al. (10), IgT is the major Ig isotype found in the skin mucosa although IgM is also present but in low quantities. In general, fish antibodies are protective against invading pathogens on mucosal surfaces as shown by Wang et al. (85, 86) and Dickerson and Clark (87) that naive fish exposed to sub-lethal infection of I. multifiliis become resistant to subsequent challenge. In their findings, they observed that resistance correlated with antibody levels in sera and skin mucus of immune fish. They further noted that antibodies from resistant fish easily bound to the immobilization antigens (i-antigens) found on the parasite cell and ciliary membranes. Antibody-mediated cross-linking with i-antigens resulted in expulsion of the parasite from exposed fish indicating that mucosal antibodies have the capacity to eliminate pathogens from mucosal surfaces in finfish. Recovered fish were protected from subsequent exposure to the parasite for a long time indicating that sub-lethal infection of I. multifiliis could serve as a live vaccine. These studies showed that exposure to I. multifiliis infection through the skin mucosa induced both mucosal and systemic antibody responses. In addition, they noted that vaccinating fish using purified i-antigens, provided long-lasting protective immunity against I. multifiliis, which makes the i-antigens to be the most promising candidate for subunit vaccine production.
In another study, Zhao et al. (92) showed that antibody-secreting cells (ASC), which include B-cells, plasmablasts, and non-replicating plasma cells, were found in low numbers in the skin of channel catfish. However, after immunization against I. multifiliis these antibodies increased by 20-fold. Thereafter, the number of ASC in the skin remained high for a long time and they were shown to confer long-term protective immunity against reinfection. However, it is vital to point out that although antibody responses were protective against parasitic infections such as I. multifiliis, there are limited studies that show protective immunity against viral and bacterial infections in the skin (93, 94). Hence, there is need for more studies to determine the protective mechanisms of skin antibodies for other pathogens. However, based on studies carried out this far (85–87, 92, 95), indications are that vaccination via the skin mucosa can induce protective antibody responses thereby creating the basis for vaccination by immersion or bath exposure as shown that fish exposed to I. multifiliis via the skin were protected by mucosal antibodies against subsequent exposure.
As for cellular-mediated immunity, there are no studies that categorically show the ability of vaccine-induced CD8 T-cell responses to prevent microbial invasion into the skin mucosa of vaccinated fish. In general, only a few studies have been carried out on cellular-mediated immunity found in the skin mucosal of finfish (27, 96). However, it is interesting to note that induction of humoral immune responses due to parasitic flagellate Ichthyobodo necator infection correlated with increased CD4+ T-cell expression when CD8α was downregulated in the skin of rainbow trout (27). In their study, Chettri et al. (27) observed upregulation of the transcription factor GATA-3 and IL-4/13A, which correlated with upregulation of IgM+, suggesting that exposure to I. necator induced CD4+ responses in the skin mucosa of infected rainbow trout whose kinetics are similar to those induced by inactivated IPNV vaccines in Atlantic salmon (47). In general, these findings demonstrate that the cellular-mediated immune system of the skin mucosa in finfish is responsive to antigen stimulation and that increased CD4+ and IL-4/13A levels could play an important role in enhancing antibody functions as shown in mammals (97–99).
Gill Mucosal Responses to Vaccination
Similar to other mucosal organs in finfish, immune responses to vaccination in the gills are coordinated by different innate and adaptive immune responses as shown below.
Innate Immune Responses to Mucosal Vaccination in the Gills of Finfish
Similar to other mucosal organs of finfish, the gill surface is covered by a mucus layer that contains complement, antimicrobial compounds, antibodies, and other surface defensins (Table 5). Different phagocytic cells have been characterized in the gills of different fish species and these include monocytes, macrophages, rodlet cells, eosinophilic granular cells, and neutrophils (Table 5). In addition, dendritic-like cells have also been reported from the gills (100). The expression pattern of CD83, a known surface marker of activated dendritic-like cells in fish (101–103), was shown to correlate with increased uptake of poly(I:C) in the gill mucosa of turbot (Scophthalmus maximus) (104), suggesting that dendritic-like cells in finfish express surface markers characteristic of activated APCs after antigen uptake. In addition, upregulation of IL-1β, IL-8, and TNFα has been shown to correspond with upregulation of TLR genes after vaccination in gills of different fish species (23, 25, 74, 105). Furthermore, some studies have also reported of upregulation of chemokines such as CXCL10, CK3, CK9, CK11, and CK12 that correspond with the homing of APCs to antigen deposition sites in the gills (35, 106–108). Put together, these observations show that the gill mucosa in finfish is bestowed with (i) a mucus layer that serves as a vehicle for host defensins, (ii) APCs comprising monocytes, macrophages, and dendritic-like cells, and (iii) regulatory cytokines and chemokines able to coordinate the functional activities of APCs. Therefore, it can be concluded that the gill mucosa is endowed with a functional innate immune system responsive to vaccination able to activate the adaptive immune system.
Adaptive Immune Responses to Mucosal Vaccination in the Gills of Finfish
Adaptive immune components of the gut identified in finfish are shown in Table 6. Olsen et al. (105) have shown that IgT+ B-cells are predominantly located in the epithelial lining of the gill lamellae, suggesting a primary role of this Ig isotype in mucosal defense against pathogen invasion, whereas IgM positive cells are located in gill arterioles and lamellar capillaries. Kai et al. (26) showed that IgM and IgT levels in the gill mucosa were only upregulated by bath or immersion vaccination, suggesting that the route of vaccine delivery had an influence on the induction of humoral immune responses in the gills. Jorgensen et al. (108) showed that IgT and IgM were able to bind to the surface structures of I. multifiliis in the gills of immune rainbow trout shortly after invasion. In their studies, they noted that IgT+ B-cells were predominantly located in the gill epithelia in the secondary lamellae corresponding to observations made by Olsen et al. (105). Parasites in immune fish were stained with IgT and yet no stain was detected from parasites from non-immune fish. They also observed that IgM+ B-cells were only found inside the capillaries of the secondary gill lamellae and yet the parasites located on the gill surface were stained with IgM, suggesting that there is possible diffusion of serum IgM from the systemic compartment to the exterior surface for it to stain the parasites at the mucosal surface within hours after penetration. In summary, these studies show that (i) there is compartmentalization in the distribution of IgT and IgM with the former being on the outer surface of the epithelia lining while the latter being in the capillaries and (ii) both IgM and IgT have the capacity to bind the exterior surfaces of invading pathogens.
Several scientists have shown the activation of naïve CD4 and CD8 T-cells in the gill mucosa of different fish species (58, 126, 127). Takazawa et al. (58) showed that CD8α cells account for a large proportion of the total lymphocytes found in the gills of salmonids. It is interesting to note that induction of MHC-I and CD8+ responses in the gills is mostly by bath or immersion vaccination (26, 52). Kai et al. (26) and Overgard et al. (52) observed that upregulation of CD8α cells coincided with upregulation of MHC-I after vaccination using viral antigens pointing to possible activation of CD8 cells via the MHC-I pathway. Overgard et al. (53) also showed upregulation of TCR genes alongside upregulation of MHC-I and CD8α pointing to possible binding of TCRs to MHC-I molecules leading to activation of CD8α cells. Moreover, Aquilino et al. (128) showed upregulation of CD3 genes and perforins alongside increased levels of CD8α and MHC-I in rainbow trout exposed to viral hemorrhagic septicemia virus (VHSV). Put together, these studies show that the gill mucosa is endowed with different components of CD8 T-cell mediated immune genes ranging from TCRs that bind to antigens presented on MHC-I molecules to CD3 molecules that activate naïve CD8 T-cells to effector CTLs and perforins that carry out cytotoxicity killing of cells infected with intracellular pathogens. However, there is need for detailed investigations to elucidate the role of CTL responses in mucosal vaccine protection in finfish.
As for CD4 T-cell responses, activation of naïve CD4 T-cells into effector subtypes is via the MHC-II pathway (34). Olsen et al. (105) showed that MHC-II cells were distributed across the gill filaments where they accumulated in the hyperplastic tissue in rainbow trout. As shown in Table 6, different transcription factors that specify the differentiation of naïve CD4 cells into different T-helper (Th) subtypes have been identified and characterized in the gills of different fish species. In addition, different cytokines and chemokines that regulate the functional activities of different Th-cells have also been characterized in different fish species. Studies carried out by Takizawa et al. (90) have shown that the constitutive expression of IL-4/13 and GATA-3 skewed toward a Th2 response in gill cells of isogenic rainbow trout exposed to poly(I:C). In their studies (90), they showed that the kinetics of IL-4/13A and IFNγ in the gill cells were consistent with expression patterns observed from fish vaccinated by injection (47). In general, these studies suggest that CD4 T-cells are responsive to antigen stimulation in the gill mucosa of finfish. Therefore, future studies should seek to identify the helper roles of different Th-subtypes in conferring protective immunity in fish vaccinated via the gill mucosa.
Nasal Mucosal Responses to Vaccination
Recent advances have shown that the nasal mucosa is endowed with an innate and adaptive immune system comparable to other mucosal organs in finfish (9). Studies carried out by Tacchi et al. (9) have shown that the nasal mucosa expressed different cytokines, chemokines, antimicrobial peptides, complement factors, and TLR genes in rainbow trout vaccinated against Yersinia ruckeri. In addition, it also expressed MHC-II genes as well as different B-cell receptor and TCR genes in response to vaccination against Y. ruckeri. All three Igs characterized in fish were detected in the nasal mucosa with IgT being the most abundant followed by IgM (9). Tacchi et al. (9) and La Patra et al. (129) showed significant protection against infectious hematopoietic virus infection (IHNV) and Y ruckeri in rainbow trout vaccinated via the nasal mucosa after challenge. In general, indications are that the nasal mucosa uses similar protective mechanisms with those used by other mucosal organs in finfish. However, given that the nasal mucosa is a recently discovered mucosal organ whose protective mechanisms have only been reported in rainbow trout, there is need for follow-up studies in order to consolidate these findings in other fish species.
General Discussion and Conclusion
In general, teleosts fish are endowed with different mucosal organs that include the gills, gut, skin, and nasal mucosa (6, 7, 9, 10). It is interesting to note that all mucosal organs have an innate immune systems, which is made of three important elements, namely (i) mucus whose function is not only to prevent microbial attachment to mucosal surfaces by continuously being excreted and sloughed-off but it serves as a vehicle for antimicrobial peptides, complement, and Igs that degrade, opsonize, and neutralize invading pathogens on mucosal surfaces; (ii) APCs that carry out antigen uptake, processing, and presentation to cells of the adaptive immune systems (130); and finally (iii) proinflammatory cytokines and chemokines that coordinate the functional activities of APCs. Overall, this review shows that the innate immune systems in different mucosal organs is responsive to mucosal vaccination and that it has the capacity to activate cells of the adaptive immune systems in finfish.
It is interesting to note that all three Ig isotypes characterized in finfish have been detected in all mucosal organs (6, 7, 9, 10). Based on studies carried out this far, it is evident that mucosal antibody responses are the only component of the adaptive immune system that has been shown to confer protective immunity in vaccinated fish (85, 86, 131) although the majority of studies that support this observation are based on the neutralizing ability of mucosal antibodies on parasitic infections (85, 86, 131) with only a few studies showing protection against viral and bacterial infections (93, 94). Nevertheless, it is interesting to note that there is compartmentalization in the expression of different Ig isotypes with IgT being the major isotype in mucosal organs while IgM is mostly found in systemic circulation (7, 10, 40). In addition, there is compartmentalization in terms of the physical distribution of IgT and IgM in some mucosal organs as shown that in the gills IgT is mainly found on exterior surfaces of the gill lamellae (105, 131), suggesting that this isotypes functions as a gatekeeper at the portals of entry whereas IgM is mostly found in the arterioles (105, 131), suggesting that its role is to prevent systemic dissemination of pathogens as a secondary defense strategy in situation where IgT on mucosal surfaces fails to prevent the penetration of pathogens into the systemic environment.
Although cellular-mediated immunity is present in all mucosal organs, its existence is mostly demonstrated by gene expression. The ability of these genes to prevent microbial invasion in vaccinated fish has not been clearly elucidated although the kinetics of CD4+ and CD8+ gene expression in different mucosal organs suggests that cellular-mediated immunity is responsive to vaccination in finfish. And as pointed out in our previous studies (5, 34, 35), the route of antigen delivery into APCs is deterministic of the type of cellular-mediated immune response induced by vaccination. Antigens delivered via the intracellular route evoke both CD4+ and CD8+ responses whereas antigens delivered by the extracellular route only induce CD4+ responses. And as pointed out by Howarth and Elliot (132), the most protective vaccines are those that stimulate both CD4+ and CD8+ responses. Therefore, live virus and DNA vaccines that evoke both CD4+ and CD8+ responses are likely to produce better protection than inactivated vaccines. Although live vaccines produce long-lasting protective immunity given that they are replicative and are more immunogenic than in inactivated vaccines (133, 134), they are less used in aquaculture because of the fear of reversion to virulence as shown in the case of IPNV that avirulent strains can revert to virulence under stress conditions (135). On the other hand, DNA vaccines that are also delivered via the intracellular route are not widely used in aquaculture for ethical reasons because of their genetic modified nature (34).This far, only the IHNV-DNA vaccine has been licensed for commercial use in Canada. Hence, the majority of commercial vaccines currently used in aquaculture are inactivated vaccines, which are limited to induction of CD4+ and humoral immune responses. Hence, there still remains the challenge of developing intracellular antigens delivery systems able to evoke of CD4+ and CD8+ T-cell responses that do not pose the danger of reversion to virulence.
This review summarizes the different components of the innate and adaptive immune systems in mucosal tissues of finish, which are activated following mucosal infection and/or vaccination studies. Based on the synopsis put forth in this review, mucosal antibodies appear to play a key role in conferring protective immunity in vaccinated fish (85, 86, 92, 93, 95). On the other hand, there still remains the challenge of elucidating the ability of mucosal vaccine-induced cellular-mediated immune responses to protect fish against microbial invasion in mucosal organs.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
Work leading to preparation of this paper was funded by the TARGETFISH, Targeted Disease Prophylaxis in the European Fish Farming, EU Grant 311993.
References
1. Medina E, Guzman CA. Modulation of immune responses following antigen administration by mucosal route. FEMS Immunol Med Microbiol (2000) 27(4):305–11. doi:10.1111/j.1574-695X.2000.tb01444.x
2. Ogra PL, Faden H, Welliver RC. Vaccination strategies for mucosal immune responses. Clin Microbiol Rev (2001) 14(2):430. doi:10.1128/CMR.14.2.430-445.2001
3. Neutra MR, Kozlowski PA. Mucosal vaccines: the promise and the challenge. Nat Rev Immunol (2006) 6(2):148–58. doi:10.1038/nri1777
4. Munang’andu HM, Fredriksen BN, Mutoloki S, Dalmo RA, Evensen O. Antigen dose and humoral immune response correspond with protection for inactivated infectious pancreatic necrosis virus vaccines in Atlantic salmon (Salmo salar L). Vet Res (2013) 44:7. doi:10.1186/1297-9716-44-7
5. Munang’andu HM, Mutoloki S, Evensen O. Acquired immunity and vaccination against infectious pancreatic necrosis virus of salmon. Dev Comp Immunol (2014) 43(2):184–96. doi:10.1016/j.dci.2013.08.008
6. Rombout JHWM, Abelli L, Picchietti S, Scapigliati G, Kiron V. Teleost intestinal immunology. Fish Shellfish Immunol (2011) 31(5):616–26. doi:10.1016/j.fsi.2010.09.001
7. Salinas I, Zhang YA, Sunyer JO. Mucosal immunoglobulins and B cells of teleost fish. Dev Comp Immunol (2011) 35(12):1346–65. doi:10.1016/j.dci.2011.11.009
8. Xu Z, Parra D, Gómez D, Salinas I, Zhang YA, von Gersdorff Jørgensen L, et al. Teleost skin, an ancient mucosal surface that elicits gut-like immune responses. Proc Natl Acad Sci U S A (2013) 110(32):13097–102. doi:10.1073/pnas.1304319110
9. Tacchi L, Musharrafieh R, Larragoite ET, Crossey K, Erhardt EB, Martin SA, et al. Nasal immunity is an ancient arm of the mucosal immune system of vertebrates. Nat Commun (2014) 5:5205. doi:10.1038/ncomms6205
10. Zhang YA, Salinas I, Li J, Parra D, Bjork S, Xu Z, et al. IgT, a primitive immunoglobulin class specialized in mucosal immunity. Nat Immunol (2010) 11(9):827–U82. doi:10.1038/ni.1913
11. Li J, Barreda DR, Zhang YA, Boshra H, Gelman AE, Lapatra S, et al. B lymphocytes from early vertebrates have potent phagocytic and microbicidal abilities. Nat Immunol (2006) 7(10):1116–24. doi:10.1038/ni1389
12. Fuglem B, Jirillo E, Bjerkås I, Kiyono H, Nochi T, Yuki Y, et al. Antigen-sampling cells in the salmonid intestinal epithelium. Dev Comp Immunol (2010) 34(7):768–74. doi:10.1016/j.dci.2010.02.007
13. Marel MV, Adamek M, Gonzalez SF, Frost P, Rombout JH, Wiegertjes GF, et al. Molecular cloning and expression of two beta-defensin and two mucin genes in common carp (Cyprinus carpio L.) and their up-regulation after beta-glucan feeding. Fish Shellfish Immunol (2012) 32(3):494–501. doi:10.1016/j.fsi.2011.12.008
14. Cole AM, Darouiche RO, Legarda D, Connell N, Diamond G. Characterization of a fish antimicrobial peptide: gene expression, subcellular localization, and spectrum of activity. Antimicrob Agents Chemother (2000) 44(8):2039–45. doi:10.1128/AAC.44.8.2039-2045.2000
15. Gomez D, Sunyer JO, Salinas I. The mucosal immune system of fish: the evolution of tolerating commensals while fighting pathogens. Fish Shellfish Immunol (2013) 35(6):1729–39. doi:10.1016/j.fsi.2013.09.032
16. Løvoll M, Fischer U, Mathisen GS, Bøgwald J, Ototake M, Dalmo RA. The C3 subtypes are differentially regulated after immunostimulation in rainbow trout, but head kidney macrophages do not contribute to C3 transcription. Vet Immunol Immunopathol (2007) 117(3–4):284–95. doi:10.1016/j.vetimm.2007.03.005
17. Løvoll M, Johnsen H, Boshra H, Bøgwald J, Sunyer JO, Dalmo RA. The ontogeny and extrahepatic expression of complement factor C3 in Atlantic salmon (Salmo salar). Fish Shellfish Immunol (2007) 23(3):542–52. doi:10.1016/j.fsi.2007.01.002
18. Lange SR, Bambir S, Dodds AW, Magnadottir B. The ontogeny of complement component C3 in Atlantic cod (Gadus morhua L.) – an immunohistochemical study. Fish Shellfish Immunol (2004) 16(3):359–67. doi:10.1016/j.fsi.2003.06.001
19. Lange S, Bambir S, Dodds AW, Magnadottir B. An immunohistochemical study on complement component C3 in juvenile Atlantic halibut (Hippoglossus hippoglossus L.). Dev Comp Immunol (2004) 28(6):593–601. doi:10.1016/j.dci.2003.10.006
20. Bielek E. Rodlet cells in teleosts: new ultrastructural observations on the distribution of the cores in trout (Oncorhynchus mykiss, Salmo trutta L.). J Submicrosc Cytol Pathol (2002) 34(3):271–8.
21. Rombout JHWM, Lamers CHJ, Helfrich MH, Dekker A, Tavernethiele JJ. Uptake and transport of intact macromolecules in the intestinal epithelium of carp (Cyprinus carpio L) and the possible immunological implications. Cell Tissue Res (1985) 239(3):519–30. doi:10.1007/BF00219230
22. Rombout JHWM, Vandenberg AA. Uptake and transport of ferritin in the epithelium of carp (Cyprinus carpio L) and the possible immunological implications. Cell Biol Int Rep (1985) 9(6):516. doi:10.1016/0309-1651(85)90003-7
23. Su JG, Yang CR, Xiong F, Wang YP, Zhu ZY. Toll-like receptor 4 signaling pathway can be triggered by grass carp reovirus and Aeromonas hydrophila infection in rare minnow Gobiocypris rarus. Fish Shellfish Immunol (2009) 27(1):33–9. doi:10.1016/j.fsi.2009.02.016
24. Takano T, Kondo H, Hirono I, Endo M, Saito-Taki T, Aoki T. Molecular cloning and characterization of toll-like receptor 9 in Japanese flounder, Paralichthys olivaceus. Mol Immunol (2007) 44(8):1845–53. doi:10.1016/j.molimm.2006.10.018
25. Sun L, Liu S, Wang R, Li C, Zhang J, Liu Z. Pathogen recognition receptors in channel catfish: IV. Identification, phylogeny and expression analysis of peptidoglycan recognition proteins. Dev Comp Immunol (2014) 46(2):291–9. doi:10.1016/j.dci.2014.04.018
26. Kai YH, Wu YC, Chi SC. Immune gene expressions in grouper larvae (Epinephelus coioides) induced by bath and oral vaccinations with inactivated betanodavirus. Fish Shellfish Immunol (2014) 40(2):563–9. doi:10.1016/j.fsi.2014.08.005
27. Chettri JK, Kuhn JA, Jaafar RM, Kania PW, Møller OS, Buchmann K. Epidermal response of rainbow trout to Ichthyobodo necator: immunohistochemical and gene expression studies indicate a Th1-/Th2-like switch. J Fish Dis (2014) 37(9):771–83. doi:10.1111/jfd.12169
28. Buchmann K. Immune mechanisms in fish skin against monogeneans – a model. Folia Parasitol (1999) 46(1):1–9.
29. Dixon B, Luque A, Abós B, Castro R, González-Torres L, Tafalla C. Molecular characterization of three novel chemokine receptors in rainbow trout (Oncorhynchus mykiss). Fish Shellfish Immunol (2013) 34(2):641–51. doi:10.1016/j.fsi.2012.12.003
30. Baoprasertkul P, Peatman E, Chen L, He C, Kucuktas H, Li P, et al. Sequence analysis and expression of a CXC chemokine in resistant and susceptible catfish after infection of Edwardsiella ictaluri. Dev Comp Immunol (2004) 28(7–8):769–80. doi:10.1016/j.dci.2003.12.002
31. Montero J, Ordas MC, Alejo A, Gonzalez-Torres L, Sevilla N, Tafalla C. CK12, a rainbow trout chemokine with lymphocyte chemo-attractant capacity associated to mucosal tissues. Mol Immunol (2011) 48(9–10):1102–13. doi:10.1016/j.molimm.2011.02.005
32. Martin E, Trichet VV, Legrand-Frossi C, Frippiat JP. Comparison between intestinal and non-mucosal immune functions of rainbow trout, Oncorhynchus mykiss. Fish Shellfish Immunol (2012) 33(6):1258–68. doi:10.1016/j.fsi.2012.09.019
33. Chen L, Klaric G, Wadsworth S, Jayasinghe S, Kuo TY, Evensen Ø, et al. Augmentation of the antibody response of Atlantic salmon by oral administration of alginate-encapsulated IPNV antigens. PLoS One (2014) 9(10):e109337. doi:10.1371/journal.pone.0109337
34. Munang’andu HM, Evensen O. A review of intra- and extracellular antigen delivery systems for virus vaccines of finfish. J Immunol Res (2015) 2015:960859. doi:10.1155/2015/960859
35. Castro R, Bromage E, Abós B, Pignatelli J, González Granja A, Luque A, et al. CCR7 is mainly expressed in teleost gills, where it defines an IgD(+)IgM(-) B lymphocyte subset. J Immunol (2014) 192(3):1257–66. doi:10.4049/jimmunol.1302471
36. Jimenez N, Coll J, Salguero FJ, Tafalla C. Co-injection of interleukin 8 with the glycoprotein gene from viral haemorrhagic septicemia virus (VHSV) modulates the cytokine response in rainbow trout (Oncorhynchus mykiss). Vaccine (2006) 24(27–28):5615–26. doi:10.1016/j.vaccine.2006.04.061
37. Holvold LB, Fredriksen BN, Bogwald J, Dalmo RA. Transgene and immune gene expression following intramuscular injection of Atlantic salmon (Salmo salar L.) with DNA-releasing PLGA nano- and microparticles. Fish Shellfish Immunol (2013) 35(3):890–9. doi:10.1016/j.fsi.2013.06.030
38. Vervarcke S, Ollevier F, Kinget R, Michoel A. Mucosal response in African catfish after administration of Vibrio anguillarum O2 antigens via different routes. Fish Shellfish Immunol (2005) 18(2):125–33. doi:10.1016/j.fsi.2004.06.004
39. Zhang YA, Salinas I, Sunyer JO. Recent findings on the structure and function of teleost IgT. Fish Shellfish Immunol (2011) 31(5):627–34. doi:10.1016/j.fsi.2011.03.021
40. Hansen JD, Landis ED, Phillips RB. Discovery of a unique Ig heavy-chain isotype (IgT) in rainbow trout: implications for a distinctive B cell developmental pathway in teleost fish. Proc Natl Acad Sci U S A (2005) 102(19):6919–24. doi:10.1073/pnas.0500027102
41. Ballesteros NA, Castro R, Abos B, Rodríguez Saint-Jean SS, Pérez-Prieto SI, Tafalla C. The pyloric caeca area is a major site for IgM(+) and IgT(+) B Cell recruitment in response to oral vaccination in rainbow trout. PLoS One (2013) 8(6):e66118. doi:10.1371/journal.pone.0066118
42. Hu YL, Xiang LX, Shao JZ. Identification and characterization of a novel immunoglobulin Z isotype in zebrafish: implications for a distinct B cell receptor in lower vertebrates. Mol Immunol (2010) 47(4):738–46. doi:10.1016/j.molimm.2009.10.010
43. Kumari J, Bogwald J, Dalmo RA. Vaccination of Atlantic salmon, Salmo salar L., with Aeromonas salmonicida and infectious pancreatic necrosis virus (IPNV) showed a mixed Th1/Th2/Treg response. J Fish Dis (2013) 36(10):881–6. doi:10.1111/jfd.12100
44. Kumari J, Bogwald J, Dalmo RA. Transcription factor GATA-3 in Atlantic salmon (Salmo salar): molecular characterization, promoter activity and expression analysis. Mol Immunol (2009) 46(15):3099–107. doi:10.1016/j.molimm.2009.06.008
45. Zhang ZB, Chi H, Niu CJ, Bogwald J, Dalmo RA. Molecular cloning and characterization of Foxp3 in Atlantic salmon (Salmo salar). Fish Shellfish Immunol (2011) 30(3):902–9. doi:10.1016/j.fsi.2011.01.012
46. Wang TH, Holland JW, Martin SAM, Secombes CJ. Sequence and expression analysis of two T helper master transcription factors, T-bet and GATA3, in rainbow trout Oncorhynchus mykiss and analysis of their expression during bacterial and parasitic infection. Fish Shellfish Immunol (2010) 29(5):705–15. doi:10.1016/j.fsi.2010.06.016
47. Munang’andu HM, Fredriksen BN, Mutoloki S, Dalmo RA, Evensen O. The kinetics of CD4+and CD8+T-cell gene expression correlate with protection in Atlantic salmon (Salmo salar L) vaccinated against infectious pancreatic necrosis. Vaccine (2013) 31(15):1956–63. doi:10.1016/j.vaccine.2013.02.008
48. Takizawa F, Araki K, Ohtani M, Toda H, Saito Y, Lampe VS, et al. Transcription analysis of two Eomesodermin genes in lymphocyte subsets of two teleost species. Fish Shellfish Immunol (2014) 36(1):215–22. doi:10.1016/j.fsi.2013.11.004
49. Mitra S, Alnabulsi A, Secombes CJ, Bird S. Identification and characterization of the transcription factors involved in T-cell development, t-bet, stat6 and foxp3, within the zebrafish, Danio rerio. FEBS J (2010) 277(1):128–47. doi:10.1111/j.1742-4658.2009.07460.x
50. Wang T, Monte MM, Huang W, Boudinot P, Martin SA, Secombes CJ. Identification of two FoxP3 genes in rainbow trout (Oncorhynchus mykiss) with differential induction patterns. Mol Immunol (2010) 47(16):2563–74. doi:10.1016/j.molimm.2010.06.015
51. Skugor S, Glover KA, Nilsen F, Krasnov A. Local and systemic gene expression responses of Atlantic salmon (Salmo salar L.) to infection with the salmon louse (Lepeophtheirus salmonis). BMC Genomics (2008) 9:498. doi:10.1186/1471-2164-9-498
52. Overgard AC, Patel S, Nostbakken OJ, Nerland AH. Atlantic halibut (Hippoglossus hippoglossus L.) T-cell and cytokine response after vaccination and challenge with nodavirus. Vaccine (2013) 31(19):2395–402. doi:10.1016/j.vaccine.2013.01.034
53. Overgard AC, Nerland AH, Fiksdal IU, Patel S. Atlantic halibut experimentally infected with nodavirus shows increased levels of T-cell marker and IFNgamma transcripts. Dev Comp Immunol (2012) 37(1):139–50. doi:10.1016/j.dci.2011.10.003
54. Buonocore F, Castro R, Randelli E, Lefranc MP, Six A, Kuhl H, et al. Diversity, molecular characterization and expression of T cell receptor gamma in a teleost tish, the sea bass (Dicentrarchus labrax, L). PLoS One (2012) 7(10): e47957. doi:10.1371/journal.pone.0047957
55. Marozzi C, Randelli E, Buonocore F, Scapigliati G. T cells from spleen, intestine, and gills of sea bass Dicentrarchus labrax. Fish Shellfish Immunol (2013) 34(6):1664. doi:10.1016/j.fsi.2013.03.094
56. Kumari J, Bogwald J, Dalmo RA. Eomesodermin of Atlantic salmon: an important regulator of cytolytic gene and interferon gamma expression in spleen lymphocytes. PLoS One (2013) 8(2):e55893. doi:10.1371/journal.pone.0055893
57. Picchietti S, Guerra L, Bertoni F, Randelli E, Belardinelli MC, Buonocore F, et al. Intestinal T cells of Dicentrarchus labrax (L.): gene expression and functional studies. Fish Shellfish Immunol (2011) 30(2):609–17. doi:10.1016/j.fsi.2010.12.006
58. Takizawa F, Dijkstra JM, Kotterba P, Korytář T, Kock H, Köllner B, et al. The expression of CD8 alpha discriminates distinct T cell subsets in teleost fish. Dev Comp Immunol (2011) 35(7):752. doi:10.1016/j.dci.2011.02.008
59. Boschi I, Randelli E, Buonocore F, Casani D, Bernini C, Fausto AM, et al. Transcription of T cell-related genes in teleost fish, and the European sea bass (Dicentrarchus labrax) as a model. Fish Shellfish Immunol (2011) 31(5):655–62. doi:10.1016/j.fsi.2010.10.001
60. Randelli E, Scala V, Casani D, Costantini S, Facchiano A, Mazzini M, et al. T cell receptor beta chain from sea bream (Sparus aurata): molecular cloning, expression and modelling of the complexes with MHC class I. Mol Immunol (2008) 45(7):2017–27. doi:10.1016/j.molimm.2007.10.027
61. Wang L, Shang N, Feng H, Guo QL, Dai HP. Molecular cloning of grass carp (Ctenopharyngodon idellus) T-bet and GATA-3, and their expression profiles with IFN-gamma in response to grass carp reovirus (GCRV) infection. Fish Physiol Biochem (2013) 39(4):793–805. doi:10.1007/s10695-012-9741-y
62. Guardiola FA, Cuesta A, Meseguer J, Esteban MA. Constitutive humoral innate defence mechanisms present in epidermal mucus of gilthead seabream (Sparus aurata L.). Fish Shellfish Immunol (2013) 34(6):1709. doi:10.1016/j.fsi.2013.03.222
63. Guardiola FA, Cuesta A, Arizcun M, Meseguer J, Esteban MA. Comparative skin mucus and serum humoral defence mechanisms in the teleost gilthead seabream (Sparus aurata). Fish Shellfish Immunol (2014) 36(2):545–51. doi:10.1016/j.fsi.2014.01.001
64. Woo PT. Protective immunity in fish against protozoan diseases. Parassitologia (2007) 49(3):185–91.
65. Lindell K, Fahlgren A, Hjerde E, Willassen NP, Fällman M, Milton DL. Lipopolysaccharide O-antigen prevents phagocytosis of Vibrio anguillarum by rainbow trout (Oncorhynchus mykiss) skin epithelial cells. PLoS One (2012) 7(5):e37678. doi:10.1371/journal.pone.0037678
66. Rajan B, Lokesh J, Kiron V, Brinchmann MF. Differentially expressed proteins in the skin mucus of Atlantic cod (Gadus morhua) upon natural infection with Vibrio anguillarum. BMC Vet Res (2013) 9:103. doi:10.1186/1746-6148-9-103
67. Bassity E, Clark TG. Functional identification of dendritic cells in the teleost model, rainbow trout (Oncorhynchus mykiss). PLoS One (2012) 7(3):e33196. doi:10.1371/journal.pone.0033196
68. Lugo-Villarino G, Balla KM, Stachura DL, Bañuelos K, Werneck MB, Traver D. Identification of dendritic antigen-presenting cells in the zebrafish. Proc Natl Acad Sci U S A (2010) 107(36):15850–5. doi:10.1073/pnas.1000494107
69. Wittamer V, Bertrand JY, Gutschow PW, Traver D. Characterization of the mononuclear phagocyte system in zebrafish. Blood (2011) 117(26):7126–35. doi:10.1182/blood-2010-11-321448
70. Iger Y, Abraham M, Dotan A, Fattal B, Rahamim E. Cellular-responses in the skin of carp maintained in organically fertilized water. J Fish Biol (1988) 33(5):711–20. doi:10.1111/j.1095-8649.1988.tb05516.x
71. Peleteiro MC, Richards RH. Phagocytic cells in the epidermis of rainbow-trout, Salmo gairdneri Richardson. J Fish Dis (1990) 13(3):225–32. doi:10.1111/j.1365-2761.1990.tb00777.x
72. Franch R, Cardazzo B, Antonello J, Castagnaro M, Patarnello T, Bargelloni L. Full-length sequence and expression analysis of Toll-like receptor 9 in the gilthead seabream (Sparus aurata L.). Gene (2006) 378:42–51. doi:10.1016/j.gene.2006.04.025
73. Pietretti D, Spaink HP, Falco A, Forlenza M, Wiegertjes GF. Accessory molecules for toll-like receptors in teleost fish. Identification of TLR4 interactor with leucine-rich repeats (TRIL). Mol Immunol (2013) 56(4):745–56. doi:10.1016/j.molimm.2013.07.012
74. Li YW, Luo XC, Dan XM, Huang XZ, Qiao W, Zhong ZP, et al. Orange-spotted grouper (Epinephelus coioides) TLR2, MyD88 and IL-1 beta involved in anti-Cryptocaryon irritans response. Fish Shellfish Immunol (2011) 30(6):1230–40. doi:10.1016/j.fsi.2011.04.012
75. Zhao LL, Liu M, Ge JW, Qiao XY, Li YJ, Liu DQ. Expression of infectious pancreatic necrosis virus (IPNV) VP2-VP3 fusion protein in Lactobacillus casei and immunogenicity in rainbow trouts. Vaccine (2012) 30(10):1823–9. doi:10.1016/j.vaccine.2011.12.132
76. Sigh J, Lindenstrom T, Buchmann K. The parasitic ciliate Ichthyophthirius multifiliis induces expression of immune relevant genes in rainbow trout, Oncorhynchus mykiss (Walbaum). J Fish Dis (2004) 27(7):409–17. doi:10.1111/j.1365-2761.2004.00558.x
77. Peleteiro MC, Richards RH. Immunocytochemical studies on immunoglobulin-containing cells in the epidermis of rainbow-trout Salmo-gairdneri Richardson – influence of bath vaccination. J Fish Biol (1988) 32(6):845–58. doi:10.1111/j.1095-8649.1988.tb05428.x
78. Zaccone G, Kapoor BG, Fasulo S, Ainis L. Structural, histochemical and functional aspects of the epidermis of fishes. Adv Mar Biol (2001) 40:253–348. doi:10.1016/S0065-2881(01)40004-6
79. Asbakk K. Elimination of foreign material by epidermal malpighian cells during wound healing in fish skin. J Fish Biol (2001) 58(4):953–66. doi:10.1111/j.1095-8649.2001.tb00547.x
80. Sigh J, Lindenstrom T, Buchmann K. Expression of pro-inflammatory cytokines in rainbow trout (Oncorhynchus mykiss) during an infection with Ichthyophthirius multifiliis. Fish Shellfish Immunol (2004) 17(1):75–86. doi:10.1016/j.fsi.2003.12.005
81. Lindenstrom T, Secombes CJ, Buchmann K. Expression of immune response genes in rainbow trout skin induced by Gyrodactylus derjavini infections. Vet Immunol Immunopathol (2004) 97(3–4):137–48. doi:10.1016/j.vetimm.2003.08.016
82. Musharrafieh R, Tacchi L, Trujeque J, LaPatra S, Salinas I. Staphylococcus warneri, a resident skin commensal of rainbow trout (Oncorhynchus mykiss) with pathobiont characteristics. Vet Microbiol (2014) 169(1–2):80–8. doi:10.1016/j.vetmic.2013.12.012
83. Yeh HY, Klesius PH. Sequence analysis, characterization and mRNA distribution of channel catfish (Ictalurus punctatus Rafinesque, 1818) chemokine (C-X-C motif) receptor 4 (CXCR4) cDNA. Vet Immunol Immunopathol (2010) 134(3–4):289–95. doi:10.1016/j.vetimm.2009.09.022
84. Montero J, Garcia J, Ordas MC, Casanova I, Gonzalez A, Villena A, et al. Specific regulation of the chemokine response to viral hemorrhagic septicemia virus at the entry site. J Virol (2011) 85(9):4046–56. doi:10.1128/JVI.02519-10
85. Wang XT, Clark TG, Noe J, Dickerson HW. Immunisation of channel catfish, Ictalurus punctatus, with Ichthyophthirius multifiliis immobilisation antigens elicits serotype-specific protection. Fish Shellfish Immunol (2002) 13(5):337–50. doi:10.1006/fsim.2001.0410
86. Wang XT, Dickerson HW. Surface immobilization antigen of the parasitic ciliate Ichthyophthirius multifiliis elicits protective immunity in channel catfish (Ictalurus punctatus). Clin Diagn Lab Immunol (2002) 9(1):176–81. doi:10.1128/CDLI.9.1.176-181.2002
87. Dickerson H, Clark T. Ichthyophthirius multifiliis: a model of cutaneous infection and immunity in fishes. Immunol Rev (1998) 166:377–84. doi:10.1111/j.1600-065X.1998.tb01277.x
88. Peleteiro MC, Richards RH. Identification of lymphocytes in the epidermis of the rainbow trout, Salmo gairdneri Richardson. J Fish Dis (1985) 8(2):161–72. doi:10.1111/j.1365-2761.1985.tb01211.x
89. Zhang H, Shen B, Wu H, Gao L, Liu Q, Wang Q, et al. Th17-like immune response in fish mucosal tissues after administration of live attenuated Vibrio anguillarum via different vaccination routes. Fish Shellfish Immunol (2014) 37(2):229–38. doi:10.1016/j.fsi.2014.02.007
90. Takizawa F, Koppang EO, Ohtani M, Nakanishi T, Hashimoto K, Fischer U, et al. Constitutive high expression of interleukin-4/13A and GATA-3 in gill and skin of salmonid fishes suggests that these tissues form Th2-skewed immune environments. Mol Immunol (2011) 48(12–13):1360–8. doi:10.1016/j.molimm.2011.02.014
91. Overgard AC, Nepstad I, Nerland AH, Patel S. Characterisation and expression analysis of the Atlantic halibut (Hippoglossus hippoglossus L.) cytokines: IL-1 beta, IL-6, IL-11, IL-12 beta and IFN gamma. Mol Biol Rep (2012) 39(3):2201–13. doi:10.1007/s11033-011-0969-x
92. Zhao XG, Findly RC, Dickerson HW. Cutaneous antibody-secreting cells and B cells in a teleost fish. Dev Comp Immunol (2008) 32(5):500–8. doi:10.1016/j.dci.2007.08.009
93. Boutier M, Ronsmans M, Ouyang P, Fournier G, Reschner A, Rakus K, et al. Rational development of an attenuated recombinant Cyprinid herpesvirus 3 vaccinevsing vrokaryotic vutagenesis and in vivo bioluminescent imaging. PLoS Pathog (2015) 11(2):e1004690. doi:10.1371/journal.ppat.1004690
94. Liu X, Wu H, Chang X, Tang Y, Liu Q, Zhang Y. Notable mucosal immune responses induced in the intestine of zebrafish (Danio rerio) bath-vaccinated with a live attenuated Vibrio anguillarum vaccine. Fish Shellfish Immunol (2014) 40(1):99–108. doi:10.1016/j.fsi.2014.06.030
95. Dickerson HW, Findly RC. Immunity to Ichthyophthirius infections in fish: a synopsis. Dev Comp Immunol (2014) 43(2):290–9. doi:10.1016/j.dci.2013.06.004
96. Shibasaki Y, Toda F, Kobayashi I, Moritomo T, Nakanishi T. Kinetics of CD4(+) and CD8 alpha(+) T-cell subsets in graft-versus-host reaction (GVHR) in ginbuna crucian carp Carassius auratus langsdorfii. Dev Comp Immunol (2010) 34(10):1075–81. doi:10.1016/j.dci.2010.05.009
97. Carballido JM, Schols D, Namikawa R, Zurawski S, Zurawski G, Roncarolo MG, et al. IL-4 induces human B-cell maturation and IgE synthesis in scid-hu mice – inhibition of ongoing IgE production by in-vivo treatment with an IL-4/IL-13 receptor antagonist. J Immunol (1995) 155(9):4162–70.
98. Defrance T, Carayon P, Billian G, Guillemot JC, Minty A, Caput D, et al. Interleukin-13 is a B-cell stimulating factor. J Exp Med (1994) 179(1):135–43. doi:10.1084/jem.179.1.135
99. Guo BC, Rothstein TL. IL-4 upregulates Ig alpha and Ig beta protein, resulting in augmented IgM maturation and B cell receptor-triggered B cell activation. J Immunol (2013) 191(2):670–7. doi:10.4049/jimmunol.1203211
100. Lovy J, Wright GM, Speare DJ. Morphological presentation of a dendritic-like cell within the gills of chinook salmon infected with Loma salmonae. Dev Comp Immunol (2006) 30(3):259–63. doi:10.1016/j.dci.2005.06.003
101. Doñate C, Roher N, Balasch JC, Ribas L, Goetz FW, Planas JV, et al. CD83 expression in sea bream macrophages is a marker for the LPS-induced inflammatory response. Fish Shellfish Immunol (2007) 23(4):877–85. doi:10.1016/j.fsi.2007.03.016
102. Pettersen EF, Ingerslev HC, Stavang V, Egenberg M, Wergeland HI. A highly phagocytic cell line TO from Atlantic salmon is CD83 positive and M-CSFR negative, indicating a dendritic-like cell type. Fish Shellfish Immunol (2008) 25(6):809–19. doi:10.1016/j.fsi.2008.08.014
103. Xu C, Evensen O, Munang’andu HM. De novo assembly and transcriptome analysis of Atlantic salmon macrophage/dendritic-like TO cells following type I IFN treatment and Salmonid alphavirus subtype-3 infection. BMC Genomics (2015) 16:96. doi:10.1186/s12864-015-1302-1
104. Hu YH, Zhang M, Sun L. Expression of Scophthalmus maximus CD83 correlates with bacterial infection and antigen stimulation. Fish Shellfish Immunol (2010) 29(4):608–14. doi:10.1016/j.fsi.2010.06.014
105. Olsen MM, Kania PW, Heinecke RD, Skjoedt K, Rasmussen KJ, Buchmann K. Cellular and humoral factors involved in the response of rainbow trout gills to Ichthyophthirius multifiliis infections: molecular and immunohistochemical studies. Fish Shellfish Immunol (2011) 30(3):859–69. doi:10.1016/j.fsi.2011.01.010
106. Alvarez-Pellitero P. Fish immunity and parasite infections: from innate immunity to immunoprophylactic prospects. Vet Immunol Immunopathol (2008) 126(3–4):171–98. doi:10.1016/j.vetimm.2008.07.013
107. Ordás MC, Castro R, Dixon B, Sunyer JO, Bjork S, Bartholomew J, et al. Identification of a novel CCR7 gene in rainbow trout with differential expression in the context of mucosal or systemic infection. Dev Comp Immunol (2012) 38(2):302–11. doi:10.1016/j.dci.2012.07.001
108. von Gersdorff JL, Heinecke RD, Skjodt K, Rasmussen KJ, Buchmann K. Experimental evidence for direct in situ binding of IgM and IgT to early trophonts of Ichthyophthirius multifiliis (Fouquet) in the gills of rainbow trout, Oncorhynchus mykiss (Walbaum). J Fish Dis (2011) 34(10):749–55. doi:10.1111/j.1365-2761.2011.01291.x
109. Heinecke RD, Chettri JK, Buchmann K. Adaptive and innate immune molecules in developing rainbow trout, Oncorhynchus mykiss eggs and larvae: expression of genes and occurrence of effector molecules. Fish Shellfish Immunol (2014) 38(1):25–33. doi:10.1016/j.fsi.2014.02.010
110. Heinecke RD, Buchmann K. Inflammatory response of rainbow trout Oncorhynchus mykiss (Walbaum, 1792) larvae against Ichthyophthirius multifiliis. Fish Shellfish Immunol (2013) 34(2):521–8. doi:10.1016/j.fsi.2012.11.036
111. Lin SH, Davidson GA, Secombes CJ, Ellis AE. A morphological study of cells isolated from the perfused gill of dab and Atlantic salmon. J Fish Biol (1998) 53(3):560–8. doi:10.1111/j.1095-8649.1998.tb01001.x
112. Mulero I, Pilar Sepulcre M, Roca FJ, Meseguer J, García-Ayala A, Mulero V. Characterization of macrophages from the bony fish gilthead seabream using an antibody against the macrophage colony-stimulating factor receptor. Dev Comp Immunol (2008) 32(10):1151–9. doi:10.1016/j.dci.2008.03.005
113. Barnett RR, Akindele T, Orte C, Shephard KL. Eosinophilic granulocytes in the epidermis of Oreochromis mossambicus gill filaments studied in situ. J Fish Biol (1996) 49(1):148–56. doi:10.1006/jfbi.1996.0142
114. Mulero I, Sepulcre MP, Meseguer J, Garcia-Ayala A, Mulero V. Histamine is stored in mast cells of most evolutionarily advanced fish and regulates the fish inflammatory response. Proc Natl Acad Sci U S A (2007) 104(49):19434–9. doi:10.1073/pnas.0704535104
115. Buonocore F, Randelli E, Tranfa P, Scapigliati GA. CD83-like molecule in sea bass (Dicentrarchus labrax): molecular characterization and modulation by viral and bacterial infection. Fish Shellfish Immunol (2012) 32(6):1179–84. doi:10.1016/j.fsi.2012.02.027
116. Wang K, Mu Y, Qian T, Ao J, Chen X. Molecular characterization and expression analysis of toll-like receptor 1 from large yellow croaker (Pseudosciaena crocea). Fish Shellfish Immunol (2013) 35(6):2046–50. doi:10.1016/j.fsi.2013.10.022
117. Taechavasonyoo A, Kondo H, Nozaki R, Suzuki Y, Hirono I. Identification of novel interleukin 1 beta family genes in Japanese flounder Paralichthys olivaceus. Fish Shellfish Immunol (2013) 34(1):393–6. doi:10.1016/j.fsi.2012.10.001
118. Taechavasonyoo A, Hirono I, Kondo H. The immune-adjuvant effect of Japanese flounder Paralichthys olivaceus IL-1beta. Dev Comp Immunol (2013) 41(4):564–8. doi:10.1016/j.dci.2013.07.003
119. Dan XM, Zhang TW, Li YW, Li AX. Immune responses and immune-related gene expression profile in orange-spotted grouper after immunization with Cryptocaryon irritans vaccine. Fish Shellfish Immunol (2013) 34(3):885–91. doi:10.1016/j.fsi.2012.12.011
120. Mladineo I, Block BA. Expression of cytokines IL-1 beta and TNF-alpha in tissues and cysts surrounding Didymocystis wedli (Digenea, Didymozoidae) in the Pacific bluefin tuna (Thunnus orientalis). Fish Shellfish Immunol (2010) 29(3):487–93. doi:10.1016/j.fsi.2010.05.008
121. Covello JM, Bird S, Morrison RN, Battaglene SC, Secombes CJ, Nowak BF. Cloning and expression analysis of three striped trumpeter (Latris lineata) pro-inflammatory cytokines, TNF-alpha, IL-1 beta and IL-8, in response to infection by the ectoparasitic, Chondracanthus goldsmidi. Fish Shellfish Immunol (2009) 26(5):773–86. doi:10.1016/j.fsi.2009.03.012
122. Abos B, Castro R, Luque A, Gonzalez L, Tafalla C. Heterogenity of IgM cells in rainbow trout (Oncorhynchus mykiss) tissues. Fish Shellfish Immunol (2013) 34(6):1636. doi:10.1016/j.fsi.2013.03.007
123. Abós B, Castro R, Pignatelli J, Luque A, González L, Tafalla C. Transcriptional heterogeneity of IgM(+) Cells in rainbow trout (Oncorhynchus mykiss) tissues. PLoS One (2013) 8(12):e82737. doi:10.1371/journal.pone.0082737
124. Nuñez Ortiz N, Gerdol M, Stocchi V, Marozzi C, Randelli E, Bernini C, et al. T cell transcripts and T cell activities in the gills of the teleost fish sea bass (Dicentrarchus labrax). Dev Comp Immunol (2014) 47(2):309–18. doi:10.1016/j.dci.2014.07.015
125. Harun NO, Wang T, Secombes CJ. Gene expression profiling in naive and vaccinated rainbow trout after Yersinia ruckeri infection: insights into the mechanisms of protection seen in vaccinated fish. Vaccine (2011) 29(26):4388–99. doi:10.1016/j.vaccine.2011.04.003
126. Chang YT, Kai YH, Chi SC, Song YL. Cytotoxic CD8 alpha(+) leucocytes have heterogeneous features in antigen recognition and class I MHC restriction in grouper. Fish Shellfish Immunol (2011) 30(6):1283–93. doi:10.1016/j.fsi.2011.03.018
127. Somamoto T, Miura Y, Nakanishi T, Nakao M. Local and systemic adaptive immune responses toward viral infection via gills in ginbuna crucian carp. Dev Comp Immunol (2015) 52(1):81–7. doi:10.1016/j.dci.2015.04.016
128. Aquilino C, Castro R, Fischer U, Tafalla C. Transcriptomic responses in rainbow trout gills upon infection, with viral hemorrhagic septicemia virus (VHSV). Dev Comp Immunol (2014) 44(1):12–20. doi:10.1016/j.dci.2013.11.006
129. LaPatra S, Kao S, Erhardt EB, Salinas I. Evaluation of dual nasal delivery of infectious hematopoietic necrosis virus and enteric red mouth vaccines in rainbow trout (Oncorhynchus mykiss). Vaccine (2015) 33(6):771–6. doi:10.1016/j.vaccine.2014.12.055
130. Iliev DB, Thim H, Lagos L, Olsen R, Jorgensen JB. Homing of antigen presenting cells in headkidney and spleen – salmon headkidney hosts diverse APC types. Front Immunol (2013) 4:137. doi:10.3389/fimmu.2013.00137
131. Jorgensen LV, Heinecke RD, Skjodt K, Rasmussen KJ, Buchmann K. Experimental evidence for direct in situ binding of IgM and IgT to early trophonts of Ichthyophthirius multifiliis (Fouquet) in the gills of rainbow trout, Oncorhynchus mykiss (Walbaum). J Fish Dis (2011) 34(10):749–55. doi:10.1111/j.1365-2761.2011.01291.x
132. Howarth M, Elliott T. The processing of antigens delivered as DNA vaccines. Immunol Rev (2004) 199(1):27–39. doi:10.1111/j.0105-2896.2004.00141.x
133. Munang’andu HM, Mutoloki S, Evensen O. Non-replicating vaccines. Fish Vaccin (2014) 3:22–32. doi:10.1002/9781118806913.ch3
134. Munang’andu HM, Sandtrø A, Mutoloki S, Brudeseth BE, Santi N, Evensen Ø. Immunogenicity and cross protective ability of the central VP2 amino acids of infectious pancreatic necrosis virus in Atlantic salmon (Salmo salar L.). PLoS One (2013) 8(1):e54263. doi:10.1371/journal.pone.0054263
Keywords: gill, gut, IgM, IgT, mucosal, oral, skin, vaccine
Citation: Munang’andu HM, Mutoloki S and Evensen Ø (2015) A review of the immunological mechanisms following mucosal vaccination of finfish. Front. Immunol. 6:427. doi: 10.3389/fimmu.2015.00427
Received: 16 May 2015; Accepted: 06 August 2015;
Published: 24 August 2015
Edited by:
Jorge Galindo-Villegas, Murcia University, SpainReviewed by:
Ingunn Sommerset, MSD Animal Health, NorwayEsteban Soto, University of California Davis School of Veterinary Medicine, USA
Copyright: © 2015 Munang’andu, Mutoloki and Evensen. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Hetron Mweemba Munang’andu, Section of Aquatic Medicine and Nutrition, Department of Basic Sciences and Aquatic Medicine, Faculty of Veterinary Medicine and Biosciences, Norwegian University of Life Sciences, Ullevålsveien 72, P.O Box 8146, Oslo NO-0033, Norway,aGV0cm9uZXkubXdlZW1iYS5tdW5hbmdhbmR1QG5tYnUubm8=
 Hetron Mweemba Munang’andu
Hetron Mweemba Munang’andu Stephen Mutoloki
Stephen Mutoloki Øystein Evensen
Øystein Evensen