- 1MediCity Research Laboratory, University of Turku, Turku, Finland
- 2Department of Immunology and Cell Biology, Graduate School of Medicine and Frontier Biosciences, Osaka University, Suita, Japan
- 3WPI Immunology Frontier Research Center, Osaka University, Suita, Japan
- 4Department of Microbiology and Immunology, Graduate School of Medicine, Osaka University, Suita, Japan
- 5Department of Medical Microbiology and Immunology, University of Turku, Turku, Finland
- 6Interdisciplinary Program for Biomedical Sciences, Institute for Academic Initiatives, Osaka University, Suita, Japan
T cell emigration from the thymus is essential for immunological homeostasis. While stromal cell-produced sphingosine-1-phosphate (S1P) has been shown to promote thymocyte egress via the S1P receptor, S1PR1, the significance of S1P/S1PR1 signaling in the thymic stromal cells that surround T cells remains unclear. To address this issue, we developed conditional knockout mice (Lyve1-CRE/S1pr1f/f mice) in which S1pr1 was selectively targeted in cells expressing the lymphatic endothelial cell marker, Lyve1. In these mice, T cells were significantly reduced in secondary lymphoid tissues, and CD62L+ mature CD4 and CD8 single-positive (SP) T cells accumulated in the medulla failed to undergo thymus egress. Using a Lyve1 reporter strain in which Lyve1 lineage cells expressed tdTomato fluorescent protein, we unexpectedly found that a considerable proportion of the thymocytes were fluorescently labeled, indicating that they belonged to the Lyve1 lineage. The CD4 and CD8 SP thymocytes in Lyve1-CRE/S1pr1f/f mice exhibited an egress-competent phenotype (HSAlow, CD62Lhigh, and Qa-2high), but were CD69high and lacked S1PR1 expression. In addition, CD4 SP thymocytes from these mice were unable to migrate to the periphery after their intrathymic injection into wild-type (WT) mice. In contrast, WT T cells could migrate to the periphery in both WT and Lyve1-CRE/S1pr1f/f thymuses. These results demonstrated that thymocyte egress is mediated by T cell-expressed, but not stromal cell-expressed, S1PR1 and caution against using the Lyve1-CRE system for selectively gene deletion in lymphatic endothelial cells.
Introduction
Sphingosine-1-phosphate (S1P) is a polar lipid mediator that is intracellularly generated from sphingomyelin by the successive actions of ceramidase and the sphingosine kinases, such as Sphk1 and Sphk2, and is transported out of the cells by S1P transporters, such as Spns2 (1). Because the S1P concentrations are much higher in blood and lymph than in the parenchyma of lymphoid tissue, S1P gradients are thought to form between lymph and lymphoid tissues and to mediate the lymphocyte egress from lymphoid tissues via the S1P receptor S1PR1, expressed on lymphocytes (2, 3).
The S1PR1 is encoded by the S1pr1 gene in mouse and is a G protein-coupled receptor (GPCR) originally identified by its involvement in endothelial cell (4). S1PR1 couples mainly to Gi/o proteins to induce activation of the Ras–ERK, PI3K–Akt, and small GTPases (Rac and Rho) signaling pathways (5). Both S1pr1-deficient mice (6) and Tie2-CRE/S1pr1f/f mice (7), in which S1pr1 is selectively disrupted in endothelial cells, die during embryogenesis due to vascular network abnormalities. S1PR1 is also highly expressed in lymphocytes, and as described above, lymphocyte-intrinsic S1PR1 is thought to regulate lymphocyte egress from the thymus (8–10) as well as from secondary lymphoid tissues (9).
Paradoxically, however, S1PR1 activation in vivo is found to occur predominantly in the CD31-expressing vascular structures, and not in the majority of lymphocytes in lymphoid tissues, including the thymus, under homeostatic conditions (11). Given that thymocytes leave the thymus via blood vessels (10, 12) and also lymphatics (12–14), the finding that S1PR1 is activated in the thymic vascular endothelial cells suggests that the thymic vasculature (blood vessels and lymphatics) may also play a role in mediating thymocyte egress to the periphery.
The lymphatic vessel endothelial hyaluronan receptor 1 (Lyve1) is a type I integral membrane protein bearing a Link module that binds hyaluronan, one of the most abundant glycosaminoglycans in the extracellular matrix (15). Lyve1 has been shown to bind and internalize hyaluronan (16), and hyaluronan binding activates intracellular signaling that promotes lymphatic endothelial cell proliferation (17). Since Lyve1-null mice exhibit normal lymphatic development and function (18), Lyve1 loss may be compensated by other hyaluronan receptors, or the function of Lyve1 may be more specific than previously thought. Because Lyve1 is expressed by most lymphatic endothelial cells, it has been widely used as a lymphatic endothelial-specific marker, although its expression has been reported in murine ocular (19) and adipose (20) tissue macrophages, murine embryonic blood vessel endothelial cells (21), and in human hepatic blood vessel endothelial cells (22). More recently, Lee demonstrated that Lyve1 is expressed in a fraction of hematopoietic stem cells and in blood vessel endothelial cells in the mouse yolk sac, suggesting that this protein is expressed during hematopoiesis and vascular development in mice (23).
Lyve1-eGFP-CRE mice express Cre recombinase and enhanced green fluorescent protein (eGFP) under control of the Lyve1 promoter (24). Researchers have used these mice for the conditional ablation of genes in the lymphatic endothelium by crossing them with strains carrying loxP-flanked genomic segments of interest (24–26). Here, we developed Lyve1-CRE/S1pr1f/f mice to investigate the role of S1PR1 signaling in lymphatic-dependent thymocyte egress. In these mice, mature CD4 and CD8 single-positive (SP) T cells were unable to leave the thymus and accumulated in the medulla. Unexpectedly, we found that S1PR1 expression was absent in a substantial proportion of the thymocytes, suggesting that Lyve1 may normally be expressed in T cells. Tracking the Lyve1 lineage cells by using a Lyve1-CRE/R26-Tdtomato reporter strain confirmed that Lyve1 was expressed in a substantial proportion of peripheral T cells as well as in thymocytes, particularly those in the thymic medulla, which are thought to emigrate from the thymus (10, 27, 28). Intrathymic injection studies confirmed that Lyve1-CRE/S1pr1f/f T cells were unable to leave the thymus, whereas WT T cells were capable of leaving both the WT and Lyve1-CRE/S1pr1f/f thymuses. Thus, our findings suggest that Lyve1 lineage thymocytes migrate from the thymus in an S1PR1-dependent manner, whereas the S1PR1 signaling in thymic stromal cells appears to be dispensable for thymocyte egress. Finally, our results also indicate that caution should be used when employing the Lyve1-CRE system to selectively target genes in lymphatic endothelial cells.
Materials and Methods
Ethics Statement
All mice were housed at the Central Animal Laboratory at the University of Turku. The animal experiments were approved by the Ethical Committee for Animal Experimentation (under license number 5587/04.10.07/2014) in Finland, and they were performed according to the 3R-principle and in adherence with the Finnish Act on Animal Experimentation (497/2013).
Mice
The B6.129P2-Lyve1tm1.1(EGFP/cre)Cys/J (Lyve1-CRE), B6.129S6(FVB)-S1pr1tm2.1Rlp/J (S1pr1f/f), B6.Cg-Gt(ROSA)26Sortm14(CAG−tdTomato)Hze/J (R26-tdTomato), C57BL6-Tg (CAG-EGFP) 10sb/J (beta-actin-eGFP), and B6.Cg-Tg(CAG-DsRed*MST)1Nagy/J (beta-actin-DsRed) mice were purchased from Jackson Laboratory. The Lyve1-CRE mice were bred with S1pr1f/f or R26-tdTomato mice to generate Lyve1-CRE/S1pr1f/f mice for functional studies and to generate Lyve1-CRE/R26-tdTomato mice for imaging experiments.
Immunohistochemistry
For immunohistochemical analysis of the thymus and lymph nodes (LNs), 6-μm thick frozen sections were fixed with acetone at −20°C and incubated with Alexa647-conjugated ERTR7 (Santa Cruz), Alexa488-conjugated anti-Lyve1 (223322; R&D Systems), and rabbit polyclonal anti-S1PR1 antibodies (H60; Santa Cruz), followed by incubation with Alexa546-conjugated goat anti-rabbit IgG antibody (Life Technologies). The images were captured using a confocal microscope (LSM 780; Zeiss). To detect Qa-2+ cells and tdTomato+ cells, the thymuses were fixed with 4% paraformaldehyde and then subsequently embedded in 4% low-melting agarose (Lonza) in PBS and cut into 200-μm thick sections using a vibratome (VT1200S; Leica). The sections were incubated overnight at 4°C in PBS containing 0.1% BSA and 1% Triton X-100, and Hoechst 33342 (Life Technologies), Alexa647-conjugated anti-Qa-2 antibody (695H1-9-9; BioLegend), DyLight550-conjugated anti-CD31 antibody (MEC13.3; Novus Biologicals), or APC-conjugated anti-CD31 antibody (MEC13.3; BioLegend). The images were captured using a spinning disk confocal microscope (Intelligent Imaging Innovations) and processed using Imaris software (Bitplane). Quantitation of the cortical and medullary area of the thymus was performed using ImageJ software (National Institute of Health).
Flow Cytometry
Thymocytes and lymphocytes were obtained from the thymus, LNs, and spleen by mechanically dissociating the tissues. Peripheral blood lymphocytes were isolated using Ficoll-Paque PLUS (GE Healthcare). Stromal cells in the thymus and LNs were isolated as described previously, with minor modifications (29). Briefly, the tissues were digested with 0.8 mg/ml dispase (Gibco), 0.2 mg/ml Collagenase P (Sigma), and 0.1 mg/ml DNase (Roche). The resulting single-cell suspensions were enriched for non-hematopoietic stromal cells by using CD45 microbeads (Miltenyi Biotech).
The lymphocytes and stromal-enriched cells were resuspended in PBS containing 1 mM EDTA, 2% fetal calf serum, and 0.1% sodium azide and incubated with the following antibodies: APC-Cy7-conjugated anti-CD4 (GK1.5), PerCP-Cy5.5-conjugated anti-CD8 (53-6.7), Pacific Blue-conjugated anti-B220 (RA3-6B2), BV510-conjugated anti-CD62L (MEL-14), FITC-conjugated anti-CD44 (IM7), PE-Cy7-conjugated anti-CD69 (H1.2F3), Alexa647-conjugated anti-Qa-2 (695H1-9-9), Pacific Blue-conjugated anti-HSA (M1/69), anti-6C10 (SM6C10) (30), V450-conjugated anti-CD45 (30-F11), PE-Cy7-conjugated anti-Gp38 (8.1.1), or APC-conjugated anti-CD31 (MEC13.3), all of which were purchased from either BioLegend or BD Biosciences except anti-6C10. To detect 6C10, lymphocytes were further incubated with phycoerythrin-conjugated goat anti-rat IgM antibody (Southern Biotech).
S1PR1 expressed on thymocytes and stromal cells was detected, as described previously (31). Briefly, single-cell suspensions were incubated with rat anti-S1PR1 monoclonal antibody (40 μg/ml, 713412; R&D Systems) followed by incubation with phycoerythrin-conjugated goat anti-rat IgG antibody (Southern Biotech).
All flow cytometric data were acquired on an LSR Fortessa (BD Biosciences) and analyzed using FlowJo software (FlowJo, LLC).
Intrathymic Injection
CD62L+ CD4+ SP cells were isolated from the thymus using a mouse CD4+ T cell isolation kit (StemCell Tech), followed by the positive selection of CD62+ cells (Miltenyi Biotech). To label the cells, they were incubated with 5 μM CFSE or 5 μM CMTMR for 30 min at 37°C in RPMI containing 2% FCS. The cells (1 × 106 cells in 10 μl PBS) were then injected into the thymus of recipient mice, as described previously, with some modifications (32). Young female mice (less than 4 weeks old) were used as the recipient mice to control thymus size, and the single-cell suspension was injected directly through the skin into the thoracic cavity immediately above the sternum using a 10-μl Hamilton syringe equipped with a 33-gauge needle. Two days after the injection, the recipient mice were sacrificed, and the donor-derived cells in the thymus, blood, spleen, and LNs were quantified by flow cytometry.
Adoptive Homing Assays
Lymphocytes were collected from LNs of beta-actin-eGFP mice. The cells (1 × 107 cells in 200 μl PBS) were then intravenously injected into the recipient mice. Twelve hours after the injection, the recipient mice were sacrificed, and the donor-derived cells in the inguinal LNs were quantified by flow cytometry.
Quantitative PCR Analysis
CD62L+CD4+ cells were isolated from the thymus, as described above, prior to RNA purification (RNeasy Plus Micro, QIAGEN). cDNA was generated using Superscript VILO cDNA synthesis kit (Life Technologies) and analyzed by a quantitative TaqMan RT-PCR method with the primers and probes designed by Universal ProbeLibarary Assay Design Center (Roche). S1pr1 primers; forward: CGGTGTAGACCCAGAGTCCT, reverse: AGCTTTTCCTTGGCTGGAG, Actb primers; forward: CTAAGGCCAACCGTGAAAAG, reverse: ACCAGAGGCATACAGGGACA. The expression values were normalized using Actb expression as endogenous controls.
Statistical Analysis
Differences between groups were evaluated with Student’s t-test for single comparisons or one-way ANOVA, followed by post hoc Tukey tests for multiple comparisons. The statistical analyses were performed using Prism software (GraphPad). A P-value <0.05 was considered to be statistically significant. Data are presented as the mean ± SD unless otherwise indicated.
Results
T Cells Are Substantially Reduced in the Secondary Lymphoid Tissues of the Lyve1-CRE/S1pr1f/f Mice
While T cell egress from lymphoid tissues has been shown to require S1PR1 expression on T cells (8, 9), a recent study indicates that S1PR1 activation occurs mainly in the vascular structures rather than on the T cells in lymphoid tissues (11). As shown in Figure 1, S1PR1 was readily detected immunohistologically in the vascular structures including the blood vessels and lymphatics of the thymus and LNs (Figure 1A; Figure S1 in Supplementary Material), and FACS studies confirmed the distinct S1PR1 expression in the blood endothelial cells and lymphatic endothelial cells of these tissues (Figures 1B,C). To address the functional significance of the lymphatic endothelial cell-expressed S1PR1 in lymphocyte egress from lymphoid tissues, we developed Lyve1-CRE/S1pr1f/f mice, in which S1pr1 was deleted selectively in Lyve1 lineage cells due to Cre-mediated excision of the loxP-flanked S1Pr1 allele. The S1pr1f/f mice were used as controls. As expected, Lyve1-CRE/S1pr1f/f mice exhibited a strongly downregulated expression of S1PR1 on the lymphatic endothelial cells of the thymus and LNs (Figure 1C); however, their lymphatics were morphologically indistinguishable from those in S1pr1f/f mice (data not shown). Lyve1-CRE/S1pr1f/f mice also exhibited a markedly reduced S1PR1 expression on blood vessel endothelial cells (Figure 1C), suggesting that the Lyve1 promoter is active in both lymphatic and blood vessel endothelial cells in the thymus.
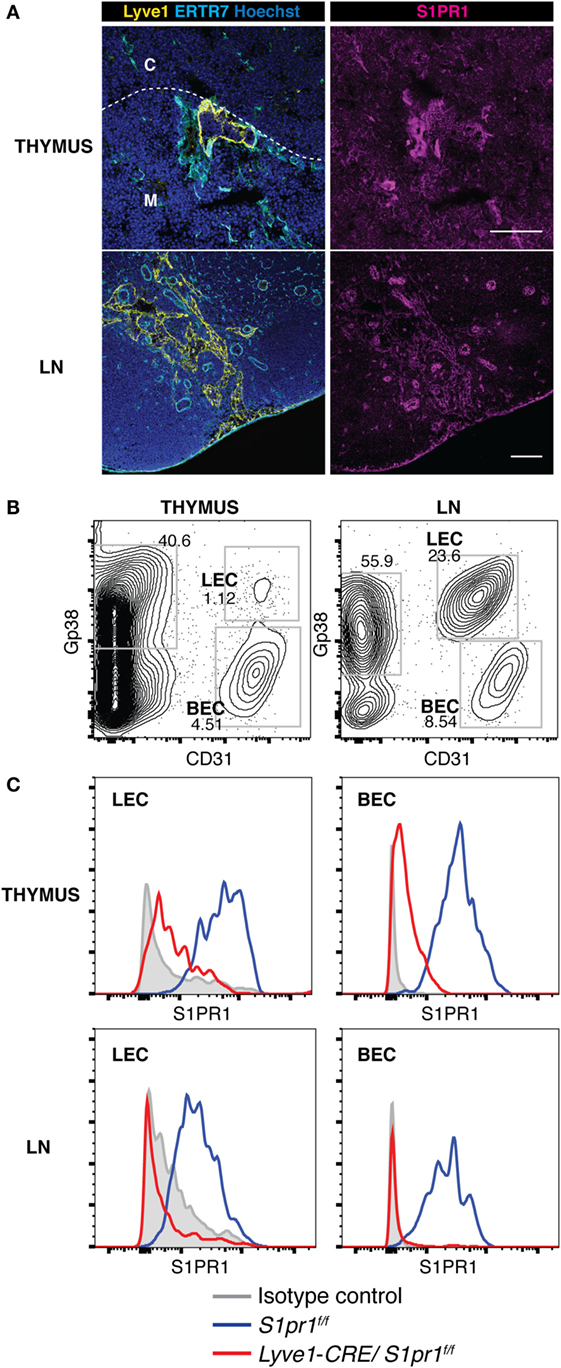
Figure 1. S1PR1 is expressed in lymphatic endothelial cells of the thymus and LNs. (A) S1PR1 expression was examined immunohistologically in the thymus and LNs. Lyve1-positive lymphatics were observed in the vicinity of the cortico-medullary junction (dotted line). C, cortex; M, medulla. Bars, 100 μm. (B,C) Flow cytometric analysis of S1PR1 expression in thymic and LN stromal cells of S1pr1f/f and Lyve1-CRE/S1pr1f/f mice. (B) CD45-negative stromal cell populations from the thymus and LNs. Lymphatic and blood vessel endothelial cells (LECs and BECs) were identified by their expression of Gp38 and CD31. (C) S1PR1 expression in LECs and BECs in the thymus and LNs of S1pr1f/f (blue line) and Lyve1-CRE/S1pr1f/f mice (red line). Data are representative of two (A) or three (B,C) independent experiments.
Analysis of the lymphoid tissues and peripheral blood of Lyve1-CRE/S1pr1f/f mice revealed that the number of both CD4+ T and CD8+ T cells in the LNs, spleen, and peripheral blood was substantially reduced compared with those in S1pr1f/f control mice (Figure 2A). In contrast, there was no significant difference in the number of B cells between the two groups of mice. Further analysis showed that naïve (CD62Lhigh and CD44low) T cells, but not memory-type (CD62Lhigh and CD44high) or effector-type (CD62Llow and CD44high), T cells were markedly reduced in the LN of Lyve1-CRE/S1pr1f/f mice compared to the control mice (Figure 2B). Analysis of the lymphocytes in the primary lymphoid organs showed that the numbers of CD4+ and CD8+ SP cells in the thymus of Lyve1-CRE/S1pr1f/f mice were significantly increased compared to those in S1pr1f/f mice, whereas the number of B cells in the bone marrow and CD4+CD8+ double positive (DP) cells in the thymus was comparable in the two types of mice (Figure 2A).
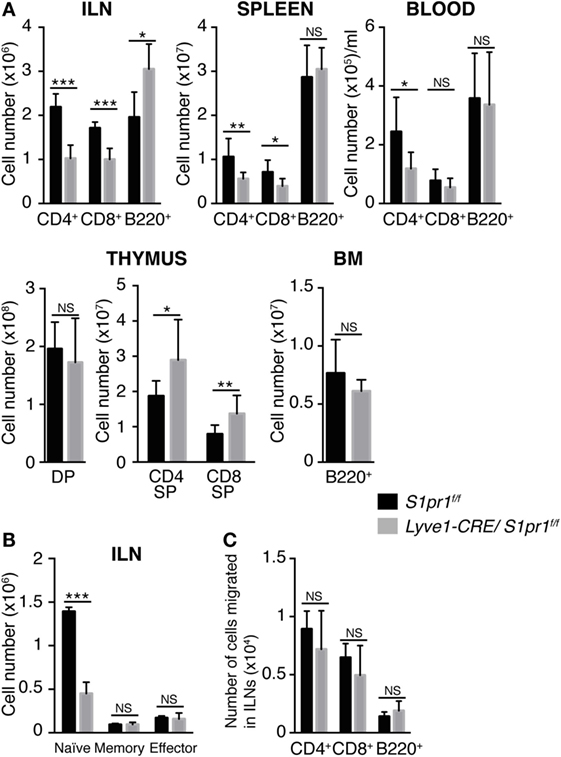
Figure 2. T cells are remarkably decreased in the peripheral lymphoid tissues of Lyve1-CRE/S1pr1f/f mice. (A) T and B cell numbers in the inguinal LNs (ILNs), spleen, blood, thymus, and bone marrow (BM) of S1pr1f/f and Lyve1-CRE/S1pr1f/f mice. DP, CD4+CD8+ double positive cells; CD4 or CD8 SP, CD4+ or CD8+ single-positive cells. (B) Numbers of CD4+ T cell subsets in the inguinal LNs of S1pr1f/f and Lyve1-CRE/S1pr1f/f mice. Naïve, memory, and effector CD4+ T cells were defined as CD62L+CD44−, CD62L+CD44+, and CD62L−CD44+, respectively. (C) Migration of WT lymphocytes into the LNs of S1pr1f/f and Lyve1-CRE/S1pr1f/f mice. Lymphocytes from beta-actin-eGFP mice were injected intravenously into recipient mice. After 12 h, the inguinal LNs were collected, and donor-derived lymphocytes were quantified by flow cytometry. These data are pooled from three independent experiments (n > 4) (A) and are representative of two (B,C) independent experiments with three mice per group. These data were evaluated by Student’s t-test and represent the mean ± SD (*P < 0.05, **P < 0.005, and ***P < 0.0005).
Because S1PR1 expression was disrupted not only in lymphatic endothelial cells but also in blood vessel endothelial cells in Lyve1-CRE/S1pr1f/f mice (Figure 1B), we investigated whether the marked reduction in naïve T and B cells in the peripheral LNs in these mice was due to an impaired ability of the high endothelial venules to mediate lymphocyte recirculation. To this end, we performed a lymphocyte adoptive transfer assay by intravenously injecting LN lymphocytes from beta-actin-eGFP mice into either Lyve1-CRE/S1pr1f/f mice or S1pr1f/f mice. We then examined the migration of the eGFP+ cells into the recipients’ LNs. As shown in Figure 2C, adoptively transferred CD4+ T cells, CD8+ T cells, and B cells were found in comparable numbers in Lyve1-CRE/S1pr1f/f and S1pr1f/f LNs, indicating that S1pr1 deletion in the Lyve1-expressing cells did not compromise the ability of high endothelial venules to mediate lymphocyte trafficking from blood to lymph. These results indicated that S1PR1 deletion in Lyve1-expressing cells reduced the number of circulating T and B cells without affecting high endothelial venule-mediated lymphocyte recirculation.
Both CD4+ and CD8+ SP Subsets Expressing Qa-2 at High Levels Are Markedly Increased in the Thymic Medulla of the Lyve1-CRE/S1pr1f/f Mice
Because naïve T cells were strongly reduced in the LNs of Lyve1-CRE/S1pr1f/f mice, we further examined the T cell subsets in the thymus of these mice. As described above, although the Lyve1-CRE/S1pr1f/f and S1pr1f/f thymuses contained similar numbers of CD4+CD8+ DP cells, there were modest but reproducible increases in the proportion of CD4+ and CD8+ SP subsets in the Lyve1-CRE/S1pr1f/f thymus (Figures 2A and 3A). Among the SP subsets, the CD62LhighCD69high cells, which barely detectable in the WT thymus, were easily detected in the Lyve1-CRE/S1pr1f/f thymus, while the relatively immature (CD62LlowCD69high) and mature (CD62LhighCD69low) SP subsets were proportionately reduced in Lyve1-CRE/S1pr1f/f compared with the S1pr1f/f thymus (Figures 3B,C). The increased CD62LhighCD69high CD4+ (Figure 3D) and CD8+ SP subsets (data not shown) expressed high levels of Qa-2, a cell surface marker that is normally expressed strongly on CD69low recent thymic emigrants (33) (Figure 3D), although CD62LhighCD69high subsets in S1pr1f/f thymus expressed low levels of Qa-2 (Figure 3D), raising the possibility that these cells were defective in thymic egress. Consistent with this possibility, the Lyve1-CRE/S1pr1f/f mice exhibited markedly enlarged thymic medullas (Figures 3E,F). Thus, our results indicated that S1PR1 ablation in Lyve1-expressing cells leads to a thymic accumulation of SP T cells, possibly due to their impaired ability to exit from the thymus.
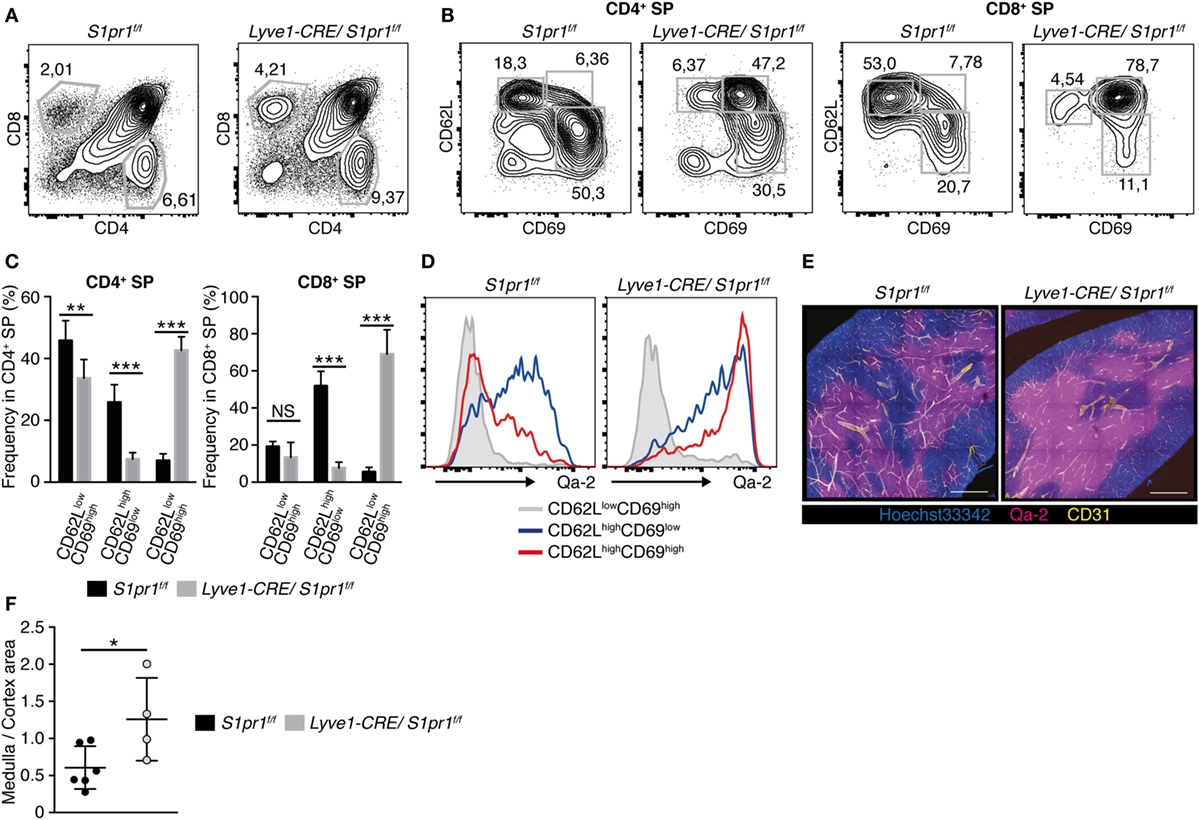
Figure 3. Mature CD4+ and CD8+ SP cells are accumulated in the Lyve1-CRE/S1pr1f/f thymus. (A) Frequencies of CD4+ and CD8+ SP cells in the thymus of Lyve1-CRE/S1pr1f/f and S1pr1f/f mice (n = 5). (B,C) Frequencies of immature (CD62LlowCD69high), mature (CD62LhighCD69low), and CD62highCD69high cells in the CD4+ or CD8+ SP subset of S1pr1f/f and Lyve1-CRE/S1pr1f/f mice. Data were pooled from nine mice (C). The data were evaluated by Student’s t-test and represent the mean ± SD (**P < 0.005 and ***P < 0.0005). (D) Qa-2 expression in the CD62LlowCD69high, CD62LhighCD69low, and CD62LhighCD69high cells in the CD4+ SP populations of the S1pr1f/f and Lyve1-CRE/S1pr1f/f thymuses (n = 3). (E) Immunohistological analysis of the S1pr1f/f and Lyve1-CRE/S1pr1f/f thymuses. Thymus sections (200-μm thick) were stained with antibodies against Qa-2 (magenta) and CD31 (yellow), nuclear staining was performed using Hoechst 33342 (blue). Qa-2 expression was prominent in the thymic medulla. Bars, 500 μm (n = 2). (F) Quantification of the cortical and medullary area in the thymus of Lyve1-CRE/S1pr1f/f and S1pr1f/f mice. Sections were cut approximate middle of the thymus, and the medullary area was determined based on Qa-2 staining: S1pr1f/f (n = 6) and Lyve1-CRE/S1pr1f/f (n = 4). These data were evaluated by Student’s t-test and represent the mean ± SD (*P < 0.05).
The Medullary SP Subsets in Lyve1-CRE/S1pr1f/f Mice Phenotypically Resemble Egress-Competent Thymocytes in Wild-type Mice but Lack S1PR1 Expression
We next sought to determine why S1PR1 ablation in Lyve1-expressing cells resulted in the accumulation of semi-mature CD4+ and CD8+ SP T cell subsets in the thymic medulla. As shown in Figures 3D and 4A, further analysis indicated that these CD62LhighCD69high cells were phenotypically very similar to the egress-competent cells of the thymus in that they were Qa-2high6C10− HSAlow (33, 34); however, the S1PR1 expression was uniformly low or absent in these cells (Figure 4B). Quantitative PCR analysis confirmed that S1pr1 expression was strongly attenuated in mature SP thymocytes in Lyve1-CRE/S1pr1f/f mice (Figure 4C). This was an unexpected finding, because (1) SP thymocytes most closely resembling recent thymic emigrants (HSAlowCD62LhighQa-2high) normally express very high levels of S1PR1 (9, 35) and (2) S1PR1 was deleted selectively in the cells that expressed the lymphatic endothelial cell-specific marker, Lyve1, in these mice.
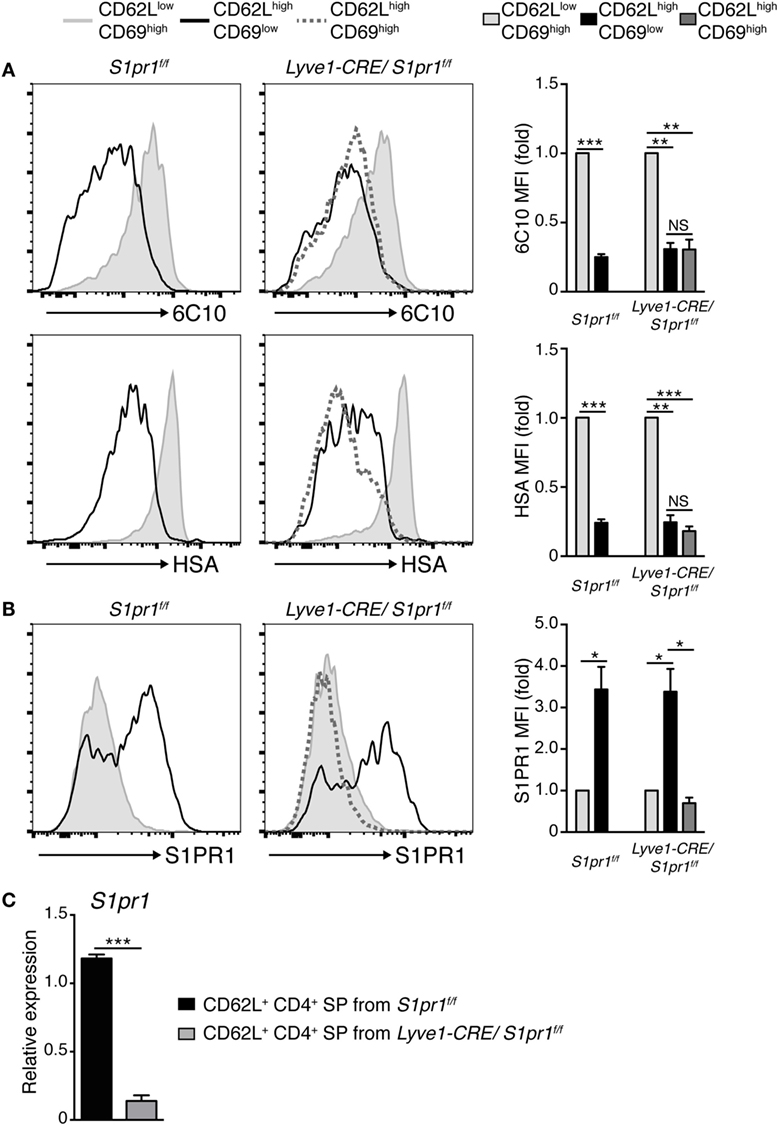
Figure 4. Thymocytes accumulating in the thymus of Lyve1-CRE/S1pr1f/f mice resemble egress-competent cells, but lack S1PR1 expression. (A,B) Expression of 6C10 and heat stable antigen (HSA) (A) or S1PR1 (B) in immature (CD62LlowCD69high), mature (CD62LhighCD69low), and CD62LhighCD69high cells of the CD4+ SP subset in S1pr1f/f and Lyve1-CRE/S1pr1f/f thymuses. The mean fluorescence intensity was normalized against CD62LlowCD69high cells. 6C10 (n = 3), HAS (n = 3), and S1PR1 (n = 3). Data are representative of two independent experiments. These data were evaluated by Student’s t-test and represent the mean ± SD (*P < 0.05, **P < 0.005, and ***P < 0.0005). (C) S1pr1 expression in CD4+ CD62L+ thymocytes of S1pr1f/f (n = 3) and Lyve1-CRE/S1pr1f/f (n = 3) thymuses. The expression intensity was normalized with the expression level of Actb. Data are representative of two independent experiments. These data were evaluated by Student’s t-test and represent the mean ± SD (***P < 0.0005).
A Large Proportion of Thymocytes, Including Those in the Medulla, Belong to the Lyve1 Lineage
The lack of S1PR1 expression in the SP T cell subsets in the Lyve1-CRE/S1pr1f/f mice suggested that these cells may belong to the Lyve1 cell lineage, consistent with a recent lineage tracing study, indicating that a small subset of hematopoietic stem cells arise from the Lyve1 lineage cells (23). To determine if the SP T cells belong to the Lyve1 cell lineage, we crossed Lyve1-CRE mice with the Rosa26-floxed stop-tdTomato (R26-tdTomato) mice to generate Lyve1-CRE reporter mice. In these mice, the Lyve1 promoter-induced expression of Cre recombinase in Lyve1-expressing cells results in excision of a floxed stop cassette preceding the tdTomato sequence at the Rosa26 locus, leading to intracellular tdTomato fluorescence. As shown in Figure 5A, flow cytometric analysis of thymocytes from these mice showed strong tdTomato expression in approximately half of the CD4+CD8+ DP cells and the CD4+ and CD8+ SP T cells, suggesting that a considerable proportion of these cells are derived from the Lyve1 lineage. Consistent with this possibility, histochemical analysis of thymus revealed that tdTomato was expressed in both the thymic cortex and the medulla, with the medulla exhibiting much stronger fluorescence than the cortex (Figure 5B). Both tdTomato-marked and -unmarked mature thymocytes exhibited comparable expression level of CD62L, CD69, and S1PR1 (Figures 5C,D). The peripheral LNs and spleen also contained large numbers of tdTomato-marked and -unmarked T and B cells (Figure 5E), indicating that Lyve1 and non-Lyve1 lineage cells emigrate from the primary lymphoid tissues to repopulate the peripheral lymphoid tissues. Notably, however, we were unable to detect Lyve1 expression on the cell surface of the tdTomato-marked cells either immunohistochemically (Figure 5B) or by flow cytometry (data not shown). These results collectively indicate that Lyve1 is expressed in a large proportion of lymphocytes during ontogeny and that, in the case of the T cell lineage, both Lyve1 and non-Lyve1 lineage lymphocytes seed and emigrate from the thymus to replenish the recirculating T cell pool in the periphery.
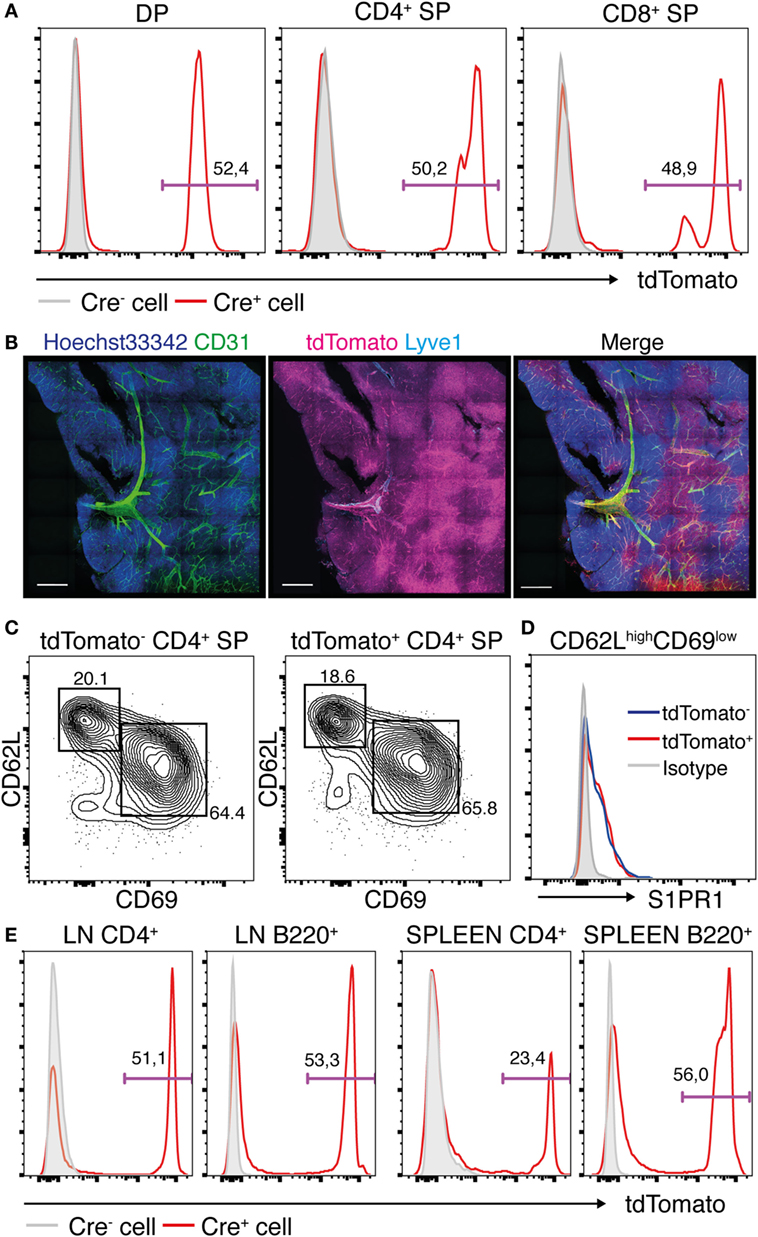
Figure 5. Most of thymic stromal cells and thymocytes belongs to the Lyve1 lineage. (A) tdTomato expression in the thymocytes of R26-tdTomato (Cre−) or Lyve1-CRE/R26-tdTomato (Cre+) mice. (B) Histological sections of the thymus from Lyve1-CRE/R26-tdTomato reporter mice stained with anti-CD31 (green) and anti-Lyve1 (cyan) antibodies, and the Hoechst 33342 nuclear stain (blue). Cre recombinase-mediated tdTomato expression is shown in magenta. Bars: 500 μm. (C,D) Expression of CD62L, CD69, and S1PR1 in tdTomato− or tdTomato+ CD4+ SP cells of Lyve1-CRE/R26-tdTomato mice. S1PR1 expression was examined on mature CD4+ SP cells. (E) tdTomato expression in the T and B cells in the LNs and spleens of R26-tdTomato (Cre−) and Lyve1-CRE/R26-tdTomato (Cre+) mice. n = 3 (A,E) and n = 2 (B–D) per group.
The SP Subsets in the Lyve1-CRE/S1pr1f/f Thymus Are Egress Incompetent
To determine whether the increased SP T cell subsets in the Lyve1-CRE/S1pr1f/f thymus were egress defective, we performed intrathymic adoptive transfer assays (36, 37) using fluorescently labeled CD62L+CD4+ SP thymocytes from Lyve1-CRE/S1pr1f/f and S1pr1f/f mice. As shown in Figure 6, when a 1:1 mixture of S1pr1f/f CD62L+CD4+ SP T cells and Lyve1-CRE/S1pr1f/f CD62L+CD4+ SP T cells labeled with CFSE and CMTMR, respectively, were cotransferred into the thymus of wild-type (WT) recipients, the Lyve1-CRE/S1pr1f/f CD4+ SP T cells were retained in the thymus more abundantly than the S1pr1f/f CD4+ SP T cells, and they migrated much less readily to the recipient’s LNs than did the S1pr1f/f CD4+ SP T cells (Figures 6A,B). Similar results were obtained when the fluorescent labels were swapped (data not shown), indicating that the fluorescent labels had no effect on lymphocyte migration. In contrast, when DsRed-expressing WT CD62L+CD4+ SP T cells were adoptively transferred into the thymus of Lyve1-CRE/S1pr1f/f or S1pr1f/f mice, their migration to the LNs of the two different type of mice was comparable (Figure 6C), indicating that S1PR1 expression of the thymic blood or lymphatic vasculature is dispensable for thymocyte egress. These results collectively demonstrated that Lyve1-CRE/S1pr1f/f CD4+ SP T cells exhibit defective thymus egress and that the T cell intrinsic, but not stromal cell-associated S1PR1 plays a critical role in T cell emigration from the thymus. Our results also suggested that subsets of both T and B cells arise from the Lyve1 linage.
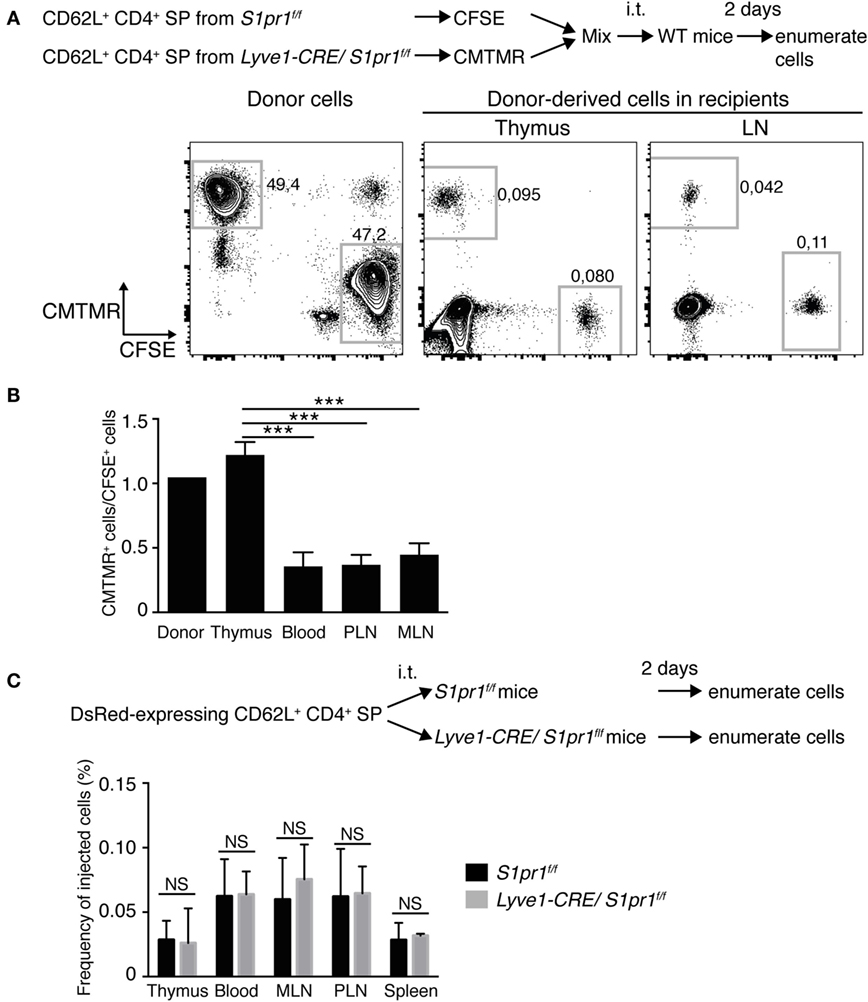
Figure 6. CD4+ SP cells in the Lyve1-CRE/S1pr1f/f thymus are egress-incompetent. (A) Egress of S1pr1f/f or Lyve1-CRE/S1pr1f/f thymocytes from the WT thymus. CD62L+ CD4+ SP cells of the S1pr1f/f or Lyve1-CRE/S1pr1f/f thymus were labeled with CFSE or CMTMR, respectively, and cotransferred into the WT thymus in equal numbers (1 × 106 cells per mouse) by intrathymic (i.t.) injection. Two days after the injection, the numbers of donor-derived cells in the thymus, blood, and LNs of recipient mice were analyzed by flow cytometry. (B) Ratio of Lyve1-CRE/S1pr1f/f CD4+ SP cells (CMTMR) to S1pr1f/f CD4+ SP cells (CFSE) that migrated into the WT recipient’s LNs. PLN, peripheral LNs; MLN, mesenteric LNs. Data are representative of two independent experiments with three mice per group. (C) Egress of WT thymocytes from the S1pr1f/f or Lyve1-CRE/S1pr1f/f thymus. CD62L+CD4+ SP cells from beta-actin-DsRed mice were injected into the S1pr1f/f or Lyve1-CRE/S1pr1f/f thymus. Two days later, the frequencies of DsRed+ cells in the thymus, blood, and LNs were analyzed by flow cytometry. Data are pooled from two independent experiments (n = 3 per group) and represent the mean ± SD. Differences between the groups were evaluated by one-way ANOVA (B) or Student’s t-test (C) (***P < 0.005).
Discussion
The thymus plays an integral role in the production, maturation, and export of T cells to the periphery. Previous studies demonstrated that the thymocyte egress occurs via two different routes, blood vessels (10, 12) and lymphatics (12–14), and that thymocyte egress via blood vessels is critically regulated by lymphocyte-intrinsic S1PR1 and pericyte-derived S1P (10). However, S1PR1 is also abundantly expressed by lymphatic endothelial cells (38) and appears to be activated constitutively in lymphatics (11), although S1PR1’s role in lymphatic endothelial cells remains unresolved. In the present study, we sought to address the S1PR1’s role in lymphatic endothelial cells by deleting the S1pr1 gene in Lyve1-expressing cells by generating Lyve1-CRE/S1pr1f/f mice. Lyve1 is expressed in a majority of lymphatics at the protein level and, hence, has been used as a lymphatic endothelial cell marker in a number of previous studies (16, 39–43), although its expression in other cell types has been reported (19, 21–23).
Our study showed that the Lyve1-CRE/S1pr1f/f mice exhibited marked increases in thymic CD4+ and CD8+ SP T cell subsets and enlarged thymic medullas compared with those of control S1pr1f/f mice or WT mice. The SP T cells from the mutant mice exhibited a mature T cell phenotype (6C10− HSAlow Qa-2+ CD62L+), superficially resembling T cells leaving the thymus (33); however, they lacked S1PR1 expression, which was previously reported to be essential for thymocyte egress (9). The T cells retained in the Lyve1-CRE/S1pr1f/f thymus also exhibited much higher CD69 expression than egress-competent cells (34), which was probably due to the lack of S1PR1 expression, since S1PR1 signaling is critical for suppressing CD69 surface expression (9, 44) (Figure S2 in Supplementary Material). The absence or paucity of S1PR1 expression in these cells was surprising, since we used the Lyve1-CRE system to conditionally disrupt S1pr1 in lymphatic endothelial cells. We therefore sought to track Lyve1 lineage cells and their descendants by generating the Lyve1-CRE/R26-tdTomato mice, in which a red fluorescent protein, tdTomato, is expressed in Lyve1-expressing cells and the cells in which Lyve1 has been expressed. When isolated cells from the peripheral lymphoid tissues of these mice were examined by flow cytometry, reporter gene activation was observed in substantial proportions of T and B lymphocytes, even though they were completely devoid of Lyve1 expression on the cell surface. Furthermore, histochemical analysis confirmed the abundance of tdTomato-marked cells in the thymus of these mice, particularly in the medullary compartments where egress-competent cells are thought to mainly reside (33, 34, 45). A majority of the tdTomato-marked and -unmarked mature thymocytes expressed S1PR1. Collectively, these results strongly suggest that the CD4+ and CD8+ SP T cells accumulating in the Lyve1-CRE/S1pr1f/f mice are Lyve1 lineage descendants that are devoid of S1PR1 and therefore unable to undergo thymus egress.
Upon surveying the literature, we found reports describing the Lyve1 expression in non-lymphatic endothelial cell lineages during ontogeny (21, 23, 46). Of note is a recent study by Lee (23) showing that Lyve1 expression first appears in the mouse yolk sac at E8.5 and that Lyve1 is detected on the cell surface of ~50% of the c-kit+ hematopoietic cell precursors and most of the CD31+ hemogenic endothelial cells in the yolk sac by E10.5 (23). Lyve1-CRE lineage tracing using Lyve1-CRE/R26-YFP mice showed that at least 30% of the hematopoietic precursors in the fetal liver and adult bone marrow express the reporter gene, indicating that they are derived from the Lyve1 lineage, although Lyve1 protein was not detected on the cell surface of bone marrow cells in adult mice. Collectively, these data indicate that Lyve1 expression is developmentally regulated in hematopoietic cells and that hematopoietic precursor cells arise from Lyve1 and non-Lyve1 lineages in mice. Our results are consistent with the Lee’s finding (23) and extend them by showing that a significant proportion of thymocytes and peripheral T cells are derived from the Lyve1 cell lineage and that thymocytes of this lineage in Lyve1-CRE/S1pr1f/f mice fail to leave the thymus due to the absence of S1PR1 expression. Thus, our study verifies that cell exit from the thymus is mediated by thymocytes’ S1PR1 expression by using a completely different model system from that described in previous studies (9, 10). Although our study do not support the involvement of S1PR1 on thymic stromal cells in the regulation of thymocyte egress, future work involving the use of conditional knockout mice in which S1pr1 is postnatally deleted in the thymic stromal cells should clarify this issue.
The biological significance of the Lyve1+/Lyve1− T cell lineages is currently unclear. Flow cytometric analysis of the two populations showed that their phenotypes were comparable, and histochemical analysis indicated that they were randomly distributed in peripheral lymphoid tissues (data not shown). The finding that T and B cells in the LNs and spleen consisted of tdTomato-marked and -unmarked populations suggests that lymphocytes descending from both Lyve1 and non-Lyve1 lineages have the ability to emigrate from the respective primary lymphoid tissues to repopulate the peripheral lymphoid tissues. Further investigation is required to understand the biological significance of Lyve1 expression in the lymphocyte lineage.
Author Contributions
AT conceived and designed the study; acquired, analyzed, and interpreted the data; and drafted the article; MH acquired, analyzed, and interpreted the data; PR, NS, MS, and SJ analyzed and interpreted the data; SS contributed unpublished essential data; MM conceived and designed the study; analyzed and interpreted the data; and drafted the article.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
Microscopic and flow cytometric analyses were performed at the Cell Imaging Core in the Turku Centre for Biotechnology in Finland. We thank Maritta Pohjansolo for technical assistance. We also thank Dr. Kyoko Hayakawa (Fox Chase Cancer Center, USA) for providing the anti-6C10 antibody and Dr. Yousuke Takahama (Tokushima University, Japan) for critically reading the manuscript.
Funding
This study received financial support from the Finland Distinguished Professor Program (FiDiPro) of the Academy of Finland and Grants-in-Aid (JP24111005) from the Ministry of Education, Culture, Sports, Science, and Technology of Japan.
Supplementary Material
The Supplementary Material for this article can be found online at http://journal.frontiersin.org/article/10.3389/fimmu.2016.00489/full#supplementary-material.
References
1. Mendelson K, Evans T, Hla T. Sphingosine 1-phosphate signalling. Development (2014) 141(1):5–9. doi:10.1242/dev.094805
2. Cyster JG, Schwab SR. Sphingosine-1-phosphate and lymphocyte egress from lymphoid organs. Annu Rev Immunol (2012) 30:69–94. doi:10.1146/annurev-immunol-020711-075011
3. Baeyens A, Fang V, Chen C, Schwab SR. Exit strategies: S1P signaling and T cell migration. Trends Immunol (2015) 36(12):778–87. doi:10.1016/j.it.2015.10.005
4. Hla T, Maciag T. An abundant transcript induced in differentiating human endothelial cells encodes a polypeptide with structural similarities to G-protein-coupled receptors. J Biol Chem (1990) 265(16):9308–13.
5. Rivera R, Chun J. Biological effects of lysophospholipids. Rev Physiol Biochem Pharmacol (2008) 160:25–46. doi:10.1007/112_0507
6. Liu Y, Wada R, Yamashita T, Mi Y, Deng CX, Hobson JP, et al. Edg-1, the G protein-coupled receptor for sphingosine-1-phosphate, is essential for vascular maturation. J Clin Invest (2000) 106(8):951–61. doi:10.1172/jci10905
7. Allende ML, Yamashita T, Proia RL. G-protein-coupled receptor S1P1 acts within endothelial cells to regulate vascular maturation. Blood (2003) 102(10):3665–7. doi:10.1182/blood-2003-02-0460
8. Allende ML, Dreier JL, Mandala S, Proia RL. Expression of the sphingosine 1-phosphate receptor, S1P1, on T-cells controls thymic emigration. J Biol Chem (2004) 279(15):15396–401. doi:10.1074/jbc.M314291200
9. Matloubian M, Lo CG, Cinamon G, Lesneski MJ, Xu Y, Brinkmann V, et al. Lymphocyte egress from thymus and peripheral lymphoid organs is dependent on S1P receptor 1. Nature (2004) 427(6972):355–60. doi:10.1038/nature02284
10. Zachariah MA, Cyster JG. Neural crest-derived pericytes promote egress of mature thymocytes at the corticomedullary junction. Science (2010) 328(5982):1129–35. doi:10.1126/science.1188222
11. Kono M, Tucker AE, Tran J, Bergner JB, Turner EM, Proia RL. Sphingosine-1-phosphate receptor 1 reporter mice reveal receptor activation sites in vivo. J Clin Invest (2014) 124(5):2076–86. doi:10.1172/jci71194
12. Miyasaka M, Pabst R, Dudler L, Cooper M, Yamaguchi K. Characterization of lymphatic and venous emigrants from the thymus. Thymus (1990) 16(1):29–43.
13. Smith C. Studies on the thymus of the mammal. VIII. Intrathymic lymphatic vessels. Anat Rec (1955) 122(2):173–9. doi:10.1002/ar.1091220204
14. Kotani M, Seiki K, Yamashita A, Horii I. Lymphatic drainage of thymocytes to the circulation in the guinea pig. Blood (1966) 27(4):511–20.
15. Banerji S, Ni J, Wang SX, Clasper S, Su J, Tammi R, et al. LYVE-1, a new homologue of the CD44 glycoprotein, is a lymph-specific receptor for hyaluronan. J Cell Biol (1999) 144(4):789–801. doi:10.1083/jcb.144.4.789
16. Prevo R, Banerji S, Ferguson DJ, Clasper S, Jackson DG. Mouse LYVE-1 is an endocytic receptor for hyaluronan in lymphatic endothelium. J Biol Chem (2001) 276(22):19420–30. doi:10.1074/jbc.M011004200
17. Wu M, Du Y, Liu Y, He Y, Yang C, Wang W, et al. Low molecular weight hyaluronan induces lymphangiogenesis through LYVE-1-mediated signaling pathways. PLoS One (2014) 9(3):e92857. doi:10.1371/journal.pone.0092857
18. Gale NW, Prevo R, Espinosa J, Ferguson DJ, Dominguez MG, Yancopoulos GD, et al. Normal lymphatic development and function in mice deficient for the lymphatic hyaluronan receptor LYVE-1. Mol Cell Biol (2007) 27(2):595–604. doi:10.1128/mcb.01503-06
19. Xu H, Chen M, Reid DM, Forrester JV. LYVE-1-positive macrophages are present in normal murine eyes. Invest Ophthalmol Vis Sci (2007) 48(5):2162–71. doi:10.1167/iovs.06-0783
20. Cho CH, Koh YJ, Han J, Sung HK, Jong Lee H, Morisada T, et al. Angiogenic role of LYVE-1-positive macrophages in adipose tissue. Circ Res (2007) 100(4):e47–57. doi:10.1161/01.res.0000259564.92792.93
21. Gordon EJ, Gale NW, Harvey NL. Expression of the hyaluronan receptor LYVE-1 is not restricted to the lymphatic vasculature; LYVE-1 is also expressed on embryonic blood vessels. Dev Dyn (2008) 237(7):1901–9. doi:10.1002/dvdy.21605
22. Mouta Carreira C, Nasser SM, di Tomaso E, Padera TP, Boucher Y, Tomarev SI, et al. LYVE-1 is not restricted to the lymph vessels: expression in normal liver blood sinusoids and down-regulation in human liver cancer and cirrhosis. Cancer Res (2001) 61(22):8079–84.
23. Lee L. Markers and Regulators Defining the Development of Hematopoietic Stem Cells and Their Niches [Dissertation]. Los Angeles (CA): University of California, Los Angeles (2014).
24. Pham TH, Baluk P, Xu Y, Grigorova I, Bankovich AJ, Pappu R, et al. Lymphatic endothelial cell sphingosine kinase activity is required for lymphocyte egress and lymphatic patterning. J Exp Med (2010) 207(1):17–27. doi:10.1084/jem.20091619
25. Dellinger MT, Meadows SM, Wynne K, Cleaver O, Brekken RA. Vascular endothelial growth factor receptor-2 promotes the development of the lymphatic vasculature. PLoS One (2013) 8(9):e74686. doi:10.1371/journal.pone.0074686
26. Jang JY, Koh YJ, Lee SH, Lee J, Kim KH, Kim D, et al. Conditional ablation of LYVE-1+ cells unveils defensive roles of lymphatic vessels in intestine and lymph nodes. Blood (2013) 122(13):2151–61. doi:10.1182/blood-2013-01-478941
27. Ushiki T. A scanning electron-microscopic study of the rat thymus with special reference to cell types and migration of lymphocytes into the general circulation. Cell Tissue Res (1986) 244(2):285–98. doi:10.1007/BF00219204
28. Kato S. Thymic microvascular system. Microsc Res Tech (1997) 38(3):287–99. doi:10.1002/(sici)1097-0029(19970801)38:3<287:aid-jemt9>3.0.co;2-j
29. Takeda A, Kobayashi D, Aoi K, Sasaki N, Sugiura Y, Igarashi H, et al. Fibroblastic reticular cell-derived lysophosphatidic acid regulates confined intranodal T-cell motility. Elife (2016) 5:e10561. doi:10.7554/eLife.10561
30. Hayakawa K, Hardy RR. Murine CD4+ T cell subsets defined. J Exp Med (1988) 168(5):1825–38. doi:10.1084/jem.168.5.1825
31. Arnon TI, Xu Y, Lo C, Pham T, An J, Coughlin S, et al. GRK2-dependent S1PR1 desensitization is required for lymphocytes to overcome their attraction to blood. Science (2011) 333(6051):1898–903. doi:10.1126/science.1208248
32. Adjali O, Marodon G, Steinberg M, Mongellaz C, Thomas-Vaslin V, Jacquet C, et al. In vivo correction of ZAP-70 immunodeficiency by intrathymic gene transfer. J Clin Invest (2005) 115(8):2287–95. doi:10.1172/jci23966
33. Jin R, Wang W, Yao JY, Zhou YB, Qian XP, Zhang J, et al. Characterization of the in vivo dynamics of medullary CD4+CD8- thymocyte development. J Immunol (2008) 180(4):2256–63. doi:10.4049/jimmunol.180.4.2256
34. Weinreich MA, Hogquist KA. Thymic emigration: when and how T cells leave home. J Immunol (2008) 181(4):2265–70. doi:10.4049/jimmunol.181.4.2265
35. Drennan MB, Elewaut D, Hogquist KA. Thymic emigration: sphingosine-1-phosphate receptor-1-dependent models and beyond. Eur J Immunol (2009) 39(4):925–30. doi:10.1002/eji.200838912
36. Goldschneider I, Komschlies KL, Greiner DL. Studies of thymocytopoiesis in rats and mice. I. Kinetics of appearance of thymocytes using a direct intrathymic adoptive transfer assay for thymocyte precursors. J Exp Med (1986) 163(1):1–17. doi:10.1084/jem.163.1.1
37. Scollay R, Smith J, Stauffer V. Dynamics of early T cells: prothymocyte migration and proliferation in the adult mouse thymus. Immunol Rev (1986) 91:129–57. doi:10.1111/j.1600-065X.1986.tb01487.x
38. Cahalan SM, Gonzalez-Cabrera PJ, Sarkisyan G, Nguyen N, Schaeffer MT, Huang L, et al. Actions of a picomolar short-acting S1P(1) agonist in S1P(1)-eGFP knock-in mice. Nat Chem Biol (2011) 7(5):254–6. doi:10.1038/nchembio.547
39. Kriehuber E, Breiteneder-Geleff S, Groeger M, Soleiman A, Schoppmann SF, Stingl G, et al. Isolation and characterization of dermal lymphatic and blood endothelial cells reveal stable and functionally specialized cell lineages. J Exp Med (2001) 194(6):797–808. doi:10.1084/jem.194.6.797
40. Odaka C, Morisada T, Oike Y, Suda T. Distribution of lymphatic vessels in mouse thymus: immunofluorescence analysis. Cell Tissue Res (2006) 325(1):13–22. doi:10.1007/s00441-005-0139-3
41. Pfeiffer F, Kumar V, Butz S, Vestweber D, Imhof BA, Stein JV, et al. Distinct molecular composition of blood and lymphatic vascular endothelial cell junctions establishes specific functional barriers within the peripheral lymph node. Eur J Immunol (2008) 38(8):2142–55. doi:10.1002/eji.200838140
42. Lee SJ, Park C, Lee JY, Kim S, Kwon PJ, Kim W, et al. Generation of pure lymphatic endothelial cells from human pluripotent stem cells and their therapeutic effects on wound repair. Sci Rep (2015) 5:11019. doi:10.1038/srep11019
43. Rantakari P, Auvinen K, Jappinen N, Kapraali M, Valtonen J, Karikoski M, et al. The endothelial protein PLVAP in lymphatics controls the entry of lymphocytes and antigens into lymph nodes. Nat Immunol (2015) 16(4):386–96. doi:10.1038/ni.3101
44. Alfonso C, McHeyzer-Williams MG, Rosen H. CD69 down-modulation and inhibition of thymic egress by short- and long-term selective chemical agonism of sphingosine 1-phosphate receptors. Eur J Immunol (2006) 36(1):149–59. doi:10.1002/eji.200535127
45. Gabor MJ, Godfrey DI, Scollay R. Recent thymic emigrants are distinct from most medullary thymocytes. Eur J Immunol (1997) 27(8):2010–5. doi:10.1002/eji.1830270827
Keywords: sphingosine-1-phosphate, S1PR1, thymus, Lyve1, egress
Citation: Takeda A, Hossain MS, Rantakari P, Simmons S, Sasaki N, Salmi M, Jalkanen S and Miyasaka M (2016) Thymocytes in Lyve1-CRE/S1pr1f/f Mice Accumulate in the Thymus due to Cell-Intrinsic Loss of Sphingosine-1-Phosphate Receptor Expression. Front. Immunol. 7:489. doi: 10.3389/fimmu.2016.00489
Received: 17 August 2016; Accepted: 24 October 2016;
Published: 08 November 2016
Edited by:
Takashi Saito, RIKEN, JapanReviewed by:
David L. Wiest, Fox Chase Cancer Center, USABeat A. Imhof, University of Geneva, Switzerland
Copyright: © 2016 Takeda, Hossain, Rantakari, Simmons, Sasaki, Salmi, Jalkanen and Miyasaka. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Masayuki Miyasaka, bW1peWFzYWtAb3JnY3RsLm1lZC5vc2FrYS11LmFjLmpw
 Akira Takeda
Akira Takeda Mohammad Shahadat Hossain
Mohammad Shahadat Hossain Pia Rantakari1
Pia Rantakari1 Szandor Simmons
Szandor Simmons Naoko Sasaki
Naoko Sasaki Sirpa Jalkanen
Sirpa Jalkanen Masayuki Miyasaka
Masayuki Miyasaka