- 1Division of Infectious Diseases, University of Colorado Denver, Aurora, CO, USA
- 2Department of Immunology, University of Colorado Denver, Aurora, CO, USA
- 3Division of Infectious Diseases, Department of Medicine, University of Arizona, Tucson, AZ, USA
T follicular helper cells (TFH) are a specialized subset of CD4 T cells that reside in B cell follicles and promote B cell maturation into plasma cells and long-lived memory B cells. During chronic infection prior to the development of AIDS, HIV-1 (HIV) replication is largely concentrated in TFH. Paradoxically, TFH numbers are increased in early and midstages of disease, thereby promoting HIV replication and disease progression. Despite increased TFH numbers, numerous defects in humoral immunity are detected in HIV-infected individuals, including dysregulation of B cell maturation, impaired somatic hypermutation, and low quality of antibody production despite hypergammaglobulinemia. Clinically, these defects are manifested by increased vulnerability to bacterial infections and impaired vaccine responses, neither of which is fully reversed by antiretroviral therapy (ART). Deficits in TFH function, including reduced HIV-specific IL-21 production and low levels of co-stimulatory receptor expression, have been linked to these immune impairments. Impairments in TFH likely contribute as well to the ability of HIV to persist and evade humoral immunity, particularly the inability to develop broadly neutralizing antibodies. In addition to direct infection of TFH, other mechanisms that have been linked to TFH deficits in HIV infection include upregulation of PD-L1 on germinal center B cells and augmented follicular regulatory T cell responses. Challenges to development of strategies to enhance TFH function in HIV infection include lack of an established phenotype for memory TFH as well as limited understanding of the relationship between peripheral TFH and lymphoid tissue TFH. Interventions to augment TFH function in HIV-infected individuals could enhance immune reconstitution during ART and potentially augment cure strategies.
The Natural History and Function of T Follicular Helper Cells (TFH) and T Follicular Regulatory Cells (TFR)
T follicular helper cells were identified 16 years ago when CD4 T cells with a unique phenotype, notably abundant CXCR5 expression, were identified in the follicles and germinal centers (GCs) of secondary lymphoid tissues (1–3). TFH express a unique transcriptional profile compared to extrafollicular and peripheral CD4 T cell subsets; they are a distinct population of CD4 T cells under the control of the master transcription regulator BCL-6 (4–6). TFH rely on signaling through inducible T cell co-stimulator (ICOS), IL-21, IL-6, and STAT3 to develop and promote the GC response (7–9). Further, interactions with GC B cells support the development of CXCR5hiPD1hi GC TFH via sustained ICOS–ICOSL and CD40–CD40L binding (10). TFH fail to accumulate in lymphoid tissues after immunization in the absence of B cells (11). TFH provide help for maturation of B cells into plasma and memory subsets, as well as drive class switch recombination and expression of enzymes, such as activation-induced deaminase (AID) that promote somatic hypermutation (SHM) to generate highly mutated antibodies (1–3). TFH are one of the main sources of IL-21, a key cytokine that promotes GC formation and maintenance, TFH and B cell proliferation, SHM, and memory B cell/plasma cell differentiation (12–15). IL-21 is primarily produced by CD4 T cells and is particularly critical to generation of antigen-specific IgG antibodies and expansion of class-switched B cells and plasma cells in vivo [reviewed in Ref. (16)]. TFH produce a variety of other cytokines including IL-4 (17), IL-17 (18), and IFNγ (19). In addition, they express increased levels of IL-10, ICOS, and CD40L compared to other T helper subsets, which allows them to positively regulate B cell differentiation and function (3, 20). Due to constraints of studying TFH from lymphoid tissues, recent studies have attempted to establish a marker for TFH in blood (21). While several markers have been used to define peripheral TFH (pTFH), several groups have used CXCR5 and PD1 co-expression (22–24). In rhesus macaques receiving a modified vaccinia virus Ankara SIV vaccine, it was shown that CXCR5+ CD4 T cells accumulated in the blood at peak effector response post-immunization, and proliferating (Ki-67 +) CXCR5+ CD4 T cells in blood were directly correlated to TFH and GC B cell frequency in lymphoid tissues (25). Yet, direct functional studies comparing lymphoid TFH to pTFH have not been done, and their relation to each other, as discussed later, remains uncertain.
More recently, TFR were identified as a unique CD4 T cell subset that controls and regulates GC responses (26–28). Similar to TFH, TFR express high levels of Bcl-6, CXCR5, ICOS, and PD-1 (26–29). TFR are unique in their ability to express Blimp-1 simultaneously with Bcl-6, and express high levels of Foxp3 compared to TFH (27). TFR develop independently of TFH from natural Treg precursors, although they rely on similar signals as TFH, such as CD28 and ICOS, to differentiate (27). TFR are a crucial component of the GC response as they inhibit GC expansion and regulate TFH and GC B cell numbers to prevent development of autoimmunity (26–28). Recent studies have shown that the function of TFR and/or a skew in the balance between TFH and TFR frequency can lead to impaired humoral immunity (30–33). Thus, an imbalance of the TFR-mediated GC regulation and skewing of the GC reaction may counteract this highly regulated response and dampen the immune response to pathogens.
TFH Expand and are the Major Reservoir of HIV Replication in Chronic HIV Infection
In HIV infection prior to the development of AIDS, TFH serve as the major site of virus replication (34–37). A CD4 T cell in the GC is on average 40 times more likely to be productively infected than a CD4 T cell outside of the follicle (36) and a median of 60–75% of HIV-producing cells are found within follicles in lymph nodes of untreated chronically HIV-infected individuals (35, 36). Within B cell follicles, the majority of HIV-producing cells are found in GC (38). Similarly, in chronically SIV-infected rhesus macaques without simian AIDS, virus replication is concentrated in B cell follicles in lymph nodes, spleen, and gut-associated lymphoid tissues, and these differences persist even after controlling for memory CD4 cell populations in the follicular and extrafollicular compartments (39).
Both heightened TFH permissivity and factors in the follicular microenvironment play a role in promoting HIV replication within TFH. Tonsillar TFH and GC TFH are highly permissive to both X4- and R5-tropic HIV compared to other tonsillar T cell subsets ex vivo (38, 40). Heightened permissivity of TFH is not fully explained by differences in memory subsets (as determined by CD95 expression), cellular activation (as measured by HLA-DR and CD38 expression), or chemokine HIV co-receptor expression (38). Within the microenvironment of the B cell follicle, specifically in the GC, follicular dendritic cells (FDC) bind HIV–antibody complexes via FC and complement receptors (41). Although FDC are not productively infected, the virions bound to their surface are adjacent to TFH within GCs (41–43), and these virions are highly infectious to TFH (42), likely contributing to the high viral burden found in TFH. FDC further upregulate HIV replication in CD4 T cells through release of TNFα (40). A relative lack of cytotoxic T lymphocytes (CTL) in the follicle both in HIV (36) and SIV infection (39, 44), likely promotes replication at those sites. Most SIV-specific CTL lack a follicular homing phenotype (CXCR5+CCR7−), which may explain their failure to home to sites of virus replication in B cell follicles (39). Depletion of CD8 cells from SIV-infected macaques leads to increases in virus replication primarily in the extrafollicular zone, further supporting the notion that CTL are primarily active in the extrafollicular compartment and exert little antiviral activity within the follicle (45). Thus, TFH are naturally highly susceptible to HIV, and their location within the immune privileged B cell follicle adjacent to FDC-bound virions further promotes high levels of HIV infection and replication.
Despite being highly permissive to HIV ex vivo and being the major virus-producing T cell subset in chronic HIV infection, the percentages of TFH increase in early- and mid-stage chronic HIV (37, 46) and SIV infection (47). One of the hallmarks of HIV infection prior to AIDS is follicular hyperplasia. The follicles and GCs in HIV-seropositive lymph nodes are substantially larger in size than those in HIV-seronegative lymph nodes (48), suggesting that there are likely numerically more TFH in HIV-seropositive compared to -seronegative lymph nodes in early and midstages of disease as well. Part of this expansion is likely antigen driven. In acute SIV infection, rapid formation of GC and accumulation of TFH, along with high p27 expression, in the follicle has been observed (49). In chronic HIV infection, virus-specific TFH are expanded (46). It has been shown in mice that sustained antigenic stimulation from GC B cells is required to maintain the TFH phenotype (50), further supporting the notion that antigen stimulation is key to TFH expansion. It is likely that other factors besides antigen contribute to TFH expansion. Cytokines known to promote TFH survival, such as IL-6 (47, 51), and interferon-γ (IFN-γ) (52, 53), are increased in HIV and SIV infections, while IL-2, which inhibits TFH differentiation, is decreased (52). Bcl-2, an anti-apoptotic protein, is upregulated on productively infected cells in ex vivo R5 infection (54). The cytokines present in the microenvironment of the TFH, as well as possible resistance to apoptosis, could therefore contribute to the expansion of the TFH population.
TFH Functional Impairments and Their Impact on Humoral Immunity During HIV Infection
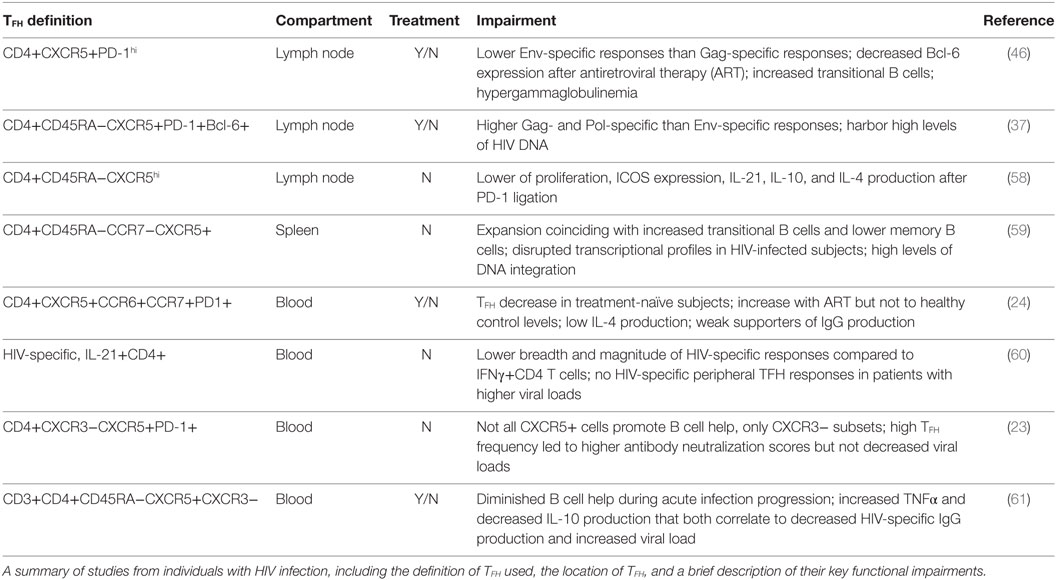
T follicular helper cells provide B cell help via IL-21, IL-4, CD40L, and ICOS to drive antibody production by GC B cells (55). It was shown that TFH and CXCR5−PD1+ CD4 T cell populations from viremic subjects can support IgG1, IgM, and IgA production ex vivo (37), but numerous examples of TFH deficiencies have been demonstrated in HIV infection (Table 1). B cell dysfunction has been well characterized during HIV infection, including the loss of memory B cell function, decreased numbers of GC B cells and plasma cells, hypergammaglobulinemia and spontaneous antibody production, and loss of T-dependent responses (37, 56, 57). Clinically, these deficits are manifested by increased vulnerability to bacterial infections as well as impaired responses to routine vaccinations. Increasing evidence has linked many of these deficits in humoral immunity to impaired TFH function.
In chronic SIV infection, a marked increase of proliferation and turnover of GC B cells was seen as TFH accumulated (49). TFH from lymph nodes of HIV-infected subjects did not produce IL-21 upon HIV antigen stimulation, but were able to after PMA/ionomycin stimulation (37). TFH have high levels of Ki-67 expression but low rates of proliferation in uninfected tonsils (55) and HIV-infected lymph nodes (37). However, IL-21 levels have been reported as deficient in HIV-infected subjects (62), and a longitudinal study demonstrated that HIV-specific IL-21+ CD4 T cells are decreased in viremic subjects (63). In this study, only elite controllers maintained high levels of IL-21 production, and antiretroviral therapy (ART) only partially restored IL-21 levels (63). Interestingly, IL-21+ CD4 T cells from HIV-infected patients have low levels of CD40L expression (64). The loss of CD40–CD40L interactions could lead to impaired stimulation of B cells by CD4 T cells from viremic HIV-infected subjects (65). In another study, splenic TFH from HIV-infected subjects demonstrated impairments in IL-4 production, along with reductions in CD40L and ICOS gene expression (59). Recently, it was demonstrated that chronically SIV-infected rhesus macaques have an expansion of Th1-biased GC TFH, phenotypically distinct from conventional GC TFH, which express CXCR3, produce high levels of IFNγ, and contain higher levels of SIV RNA (66).
Impact of Altered TFH Function on Anti-HIV Antibody Response
Deficits in TFH likely contribute to the failure to develop effective antibody responses to HIV. A recent study of acute HIV seroconverters demonstrated the onset of impairments in the ability of circulating TFH to stimulate HIV-specific antibody production by B cells are associated with peak viremia, suggesting that TFH defects occur very early following infection (61). Most antibodies generated during infection that neutralize across clades of HIV, i.e., broadly neutralizing antibodies (bnAb), are generated after several years and show high levels of SHM resulting from extensive affinity maturation in the GC. These responses are only generated in a small fraction of infected individuals (67), and the critical components of bnAb generation are unknown (68). In both untreated and treated HIV-infected subjects, TFH from lymph nodes were shown to be on average five times more sensitive to Gag than Env, with overall low TFH cytokine production after Env stimulation (46). This could be due to the increased presence of Gag antigen compared to Env antigen in the lymph node of HIV-infected subjects (69) and the persistence of p24 antigen in lymph nodes after long-term ART (70). While it is not surprising there are more Gag-specific TFH than Env-specific TFH, a lack of specificity to the HIV envelope by TFH is likely one of the contributing factors to a lack of bnAb development. A loss of Gag-specific antibody response occurs during disease progression, but there is no simultaneous increase of high affinity Env-specific response (71). Thus, an early and sustained lack of Env-specific TFH response could contribute to the slow development of HIV-neutralizing antibody responses and with the failure of many individuals to generate bnAbs.
While protective neutralizing antibodies and bnAbs have been structurally and genetically well characterized in HIV-infected individuals, it remains unclear how these antibodies are generated and whether or not TFH can promote bnAb development. The development of bnAbs is relatively slow and shown to not strongly correlate with CD4 T cell counts, MHC II alleles, or typical patient demographics (72). However, some evidence suggests that TFH function plays a role in HIV neutralization. Circulating CD4 T cells from HIV controllers and ART-treated individuals produced IL-21 when stimulated with an HIV peptide pool, but not those from HIV progressors (73). In a longitudinal assessment of acute HIV infection (12 months), treated individuals had consistently higher IL-21 production than untreated individuals, and IL-21 contributed to viral control in CD4 and CD8 T cell co-cultures ex vivo (73). In a cohort of chronic aviremic subjects, IL-21 production was reduced in circulating TFH and supplementation of IL-21 or replacement of these subjects’ TFH with TFH from healthy controls led to increased production of HIV-specific antibodies by B cells ex vivo (74). In a cohort of HIV-infected individuals a limited proportion of patients developed bnAbs, but these patients had the highest levels of circulating, functional memory TFH (23). However, their viral loads did not decline after 4 weeks, but began to decline in a few individuals at 40 weeks (23). TFH frequency correlated strongly with bnAb development, thus indicating that TFH are important for generating bnAb. In HIV-infected children receiving ART, circulating memory TFH declined, expressed low levels of ICOS, and had a diminished capacity to produce IL-4 (75). Thus, impairments of TFH function can persist in the absence of viremia. Further, in SHIV-infected rhesus macaques, the quality of TFH response was correlated with the degree of SHM in virus-specific B cells and bnAb production (60). As virus-specific IL-4+ TFH increased (IL-21 was not measured in this study), the amount of IgG+ virus-specific B cells and neutralizing response against HIV increased (60). Specifically, the frequency of IL-4+ and CD40L+ TFH correlated strongly with the frequency of Env-specific IgG + B cells (60). This study also identified a population of IFNγ+ Env-specific TFH, which are less likely to provide B cell help, and these did not correlate to Env-specific IgG+ B cells. In another study, IL-21+ CD4 T cells in the periphery of HIV-infected individuals were shown to be functionally and transcriptionally equivalent to TFH, and Env-specific IL-21+ CD4 T cells provided higher quality B cell help than the Gag-specific subset. Env-specific IL-21+ CD4 T cells also positively correlated to protective responses of subjects who responded to vaccination in the RV144 study (76). Thus, eliciting the right type of TFH help, rather than broad TFH activation, is crucial to bnAb generation. Augmentation and promotion of TFH function to boost this Env-specific IL-21+ CD4 T cell response could benefit future preventive vaccine trials and lead to broader specificity of anti-HIV antibodies and perhaps promote more rapid development of bnAbs in vaccinated individuals.
TFH Impairment and Their Relationship to Vaccine Responses in HIV-Infected Individuals
Individuals with chronic HIV infection typically produce poor antibody responses to immunization (77) and specifically had a high failure rate after a dose of the H1N1/09 influenza vaccine (78). In HIV seronegative individuals, the emergence of blood ICOS+CXCR5+CXCR3+ TFH that are able to produce IL-21 correlated with influenza-specific B cell responses (79) and blood ICOS+IL-21+ influenza-specific TFH expand after immunization and correlate to antibody responses (80). TFH function in HIV-infected individuals could be important to respond to vaccinations, but research in this area is limited. In ART-treated HIV-infected individuals, responders to the H1N1/09 influenza vaccine had upregulated IL-21 production and increased IL-21 receptor expression on B cells (81). Further, B cells from HIV-infected influenza responders secreted high levels of IgG after stimulation with IL-21 and H1N1 antigen, whereas HIV-infected non-responders did not (81). Expression of AID was positively correlated to influenza neutralizing antibody responses in HIV-infected individuals, and those with the highest levels of AID expression carried protective antibodies for the longest amount of time (82).
Recently, the quality of TFH responses to influenza vaccination was characterized in HIV-infected individuals. Of 16 HIV-infected subjects on ART receiving the H1N1/09 influenza vaccine, 8 subjects responded. Antibody responses were linked to the ability of pTFH to proliferate, to the ability of pTFH in responders to proliferate, produce IL-21, and stimulate IgG production (22). In this study, pTFH were not significantly altered in HIV-infected subjects and healthy controls at the time of vaccination, and the HIV-infected group had significantly higher frequencies of central memory pTFH (22). These data indicate that although pTFH were phenotypically similar in HIV-infected subjects compared to healthy controls, recall response and function of pTFH is significantly impaired in HIV-infected subjects even after potent ART regimens. As B cell/pTFH cocultures were performed with sorted cells it remains to be determined if TFR in the periphery play a role in the dichotomy of HIV-infected responders and non-responders.
Mechanisms That Underlie TFH Dysfunction
One of the obvious causes of TFH dysfunction is direct HIV infection of the TFH themselves. Nevertheless, only a minority of TFH are producing virus at any single time point (36), and thus this is unlikely to be the principal cause of their dysfunction. TFH are characterized by high levels of PD-1 expression. Ligation of PD-1 on TFH by lymph node B cells that express PD-L1, which are elevated in HIV-infected individuals, leads to decreases in IL-21 production and ICOS expression (58). Blockade of this interaction, with PD-L1 neutralizing antibodies, restores TFH help to B cells and promotes IgG production (58). One report has shown that HIV infection leads to an expansion of PD-L1 expressing regulatory B cells in peripheral blood that positively correlate with increased viral load and T cell exhaustion (83), however, TFH function was not examined.
Another likely cause of TFH dysfunction in HIV infection is regulation by TFR. In mice, the magnitude of the GC reaction increased and autoimmune responses were generated when TFR were unable to migrate into the follicle (84). Also in mice, it was demonstrated that excessive numbers of TFH are correlated with impaired affinity maturation, and restoring a balanced ratio of TFH to TFR allows for generation of highly mutated, high avidity antibodies (85). Recent studies in rhesus macaques have shown that TFR frequencies in secondary lymphoid tissues are increased in chronically SIV-infected animals (48, 86), while another study found decreases in TFR during chronic infection (87). Reasons for discrepancies among these studies are not clear. In chronic HIV infection, TFR are increased in lymph nodes (48) and spleen (59). They are also increased during acute ex vivo HIV infection of tonsil cells (48). In ex vivo HIV infection of human tonsil cells, our group found that TFR inhibited ICOS expression, IL-21 production, and IL-4 production by TFH (48). In another study of treatment-naïve, chronically HIV-infected subjects, the frequency of memory (CD45RA-CCR7-) TFR and TFH were shown to increase (59). These increases were associated with increased GC B cells; however, these cells were mostly naïve, pre-GC, and transitional B cells as opposed to memory B cells (59). Increases in TFH and TFR from spleen cells of HIV-infected subjects were associated with defects in the memory B cell compartment and reduced B cell help factors such as IL-4 (59). In addition, higher quality of Env-specific (gp120) antibodies in SIV-infected rhesus macaques was correlated with a lower frequency of TFR (87). Neutralizing antibodies to HIV were negatively correlated to Foxp3+ Env-specific TFH (TFR were not excluded from TFH in this work) in SHIV-infected rhesus macaques (60). Collectively, these data suggest that human TFR increase during chronic HIV infection and impair TFH function resulting in disruption of proper B cell differentiation and SHM. It has been shown in mouse models that the loss of TFR function allows for higher levels of antibody production, but the resulting antibody is much lower affinity than if TFR function is not impaired (32). Whether TFR are able to control B cell responses directly, through TFH impairment, or both remains to be shown.
Memory TFH in HIV-Infected Individuals
One clear area requiring more research is the development and fate of memory TFH subsets. It is currently unknown if TFH memory forms and is sustained inside or outside of the GC, or whether effector TFH persist in chronic infections due to prolonged antigen exposure and GC maintenance (88, 89). This is especially difficult to distinguish in HIV-infected subjects, as high levels of antigens persist in the lymph nodes well after ART initiation. Effector TFH are present as long as the GC persists, but if these cells become memory TFH or influence the response to vaccinations in HIV-infected subjects remains to be determined. One challenge in defining memory TFH and effector TFH is the plasticity of phenotype of these cells. Studies in LCMV-infected mice have demonstrated that CXCR5+ memory TFH downregulate PD-1, Bcl-6, IL-21, and ICOS compared to effector populations, but are able to recall effector TFH phenotype upon antigen challenge, suggesting a TFH lineage commitment of these memory cells (90, 91). CXCR5 has been used to distinguish memory TFH, but not all CXCR5+ CD4 T cells possess TFH function after activation (23). Furthermore, in a mouse model, TFH lost expression of Bcl-6, CXCR5, and PD-1 and acquired a memory phenotype when transferred into a mouse that did not express the cognate antigen (92). Thus, lack of a reliable phenotype for effector and memory TFH populations remains a barrier to studying memory TFH development and assessing memory responses (93).
Another important question is the location of the memory TFH pool and whether there is crosstalk between blood and lymphatic tissues. It has been shown that circulating and lymph node-resident memory populations may develop independently and both are antigen specific with potent effector functions (94, 95). In mice, it was demonstrated that effector TFH can circulate to various GCs within the same lymph node, but rarely escape to the periphery (94). Further, pTFH with memory function were shown to develop independent of the GC in mice (96). As sampling pTFH in the blood is more feasible than lymph node TFH and circulating TFH are shown to have memory and migrate to lymph nodes to stimulate B cell effector responses (95), most studies to date have focused on the function of pTFH in vaccine responses of HIV-infected subjects. Highly functional pTFH are reduced in viremic HIV-infected subjects, but rebound after the administration of ART (24). In this study, abundance of IgG+ memory B cells and neutralizing antibody did not strongly correlate with pTFH frequency, however, pTFH from HIV-infected subjects had relatively low IL-21 production in response to either SEB or Gag peptide pool stimulations and had low levels of IL-21 and IL-4 gene expression (24). This suggests that humoral responses and vaccine responses not only need to boost pTFH/TFH numbers, but also elicit highly functional B cell help responses.
The extent to which HIV directly influences TFH function, or whether HIV-driven enhancements in TFR regulatory activity influences TFH dysfunction, leading to poor memory and vaccine response, remains to be fully understood. In mice, circulating TFR were shown to be expanded after viral infection and could potently suppress pTFH function without requiring specific antigens (95). Whether TFR prevent memory TFH function, or prevent memory TFH from reacquiring effector TFH function, is an important area of future research. As TFR frequency has been found to negatively correlate with bnAb generation (60, 87), it will be necessary to determine if they also disrupt the formation or activation of memory TFH. TFR could prevent memory TFH from having high quality effector responses and thus represent a barrier to generating effective vaccine responses. TFR regulatory function could impair the activity of memory TFH and be one of the contributing factors to failure of preventative HIV vaccinations. Further, as TFR act non-specifically on target cells, they could also contribute to the relatively low efficacy of non-HIV vaccine responses in HIV-infected individuals.
Conclusion
T follicular helper cells have a critical role in HIV immunopathogenesis. They proportionately expand compared to total CD4 T cells during chronic disease and are the major virus-producing cells during asymptomatic disease, thereby driving disease progression. They also exhibit multiple functional deficits that impair development of robust humoral immunity to pathogens, including HIV itself. Mechanisms underlying TFH impairment likely include direct infection of TFH, suppressive factors in the GC milieu such as PD-L1 expression on B cells, and TFR. Strategies to augment TFH immunity remain to be developed, but potential interventions include administration of IL-21 as well as inhibition of TFR responses. Such strategies need to be developed cautiously as unintended consequences of these interventions, such as development of autoimmunity due to excessive inhibition of TFR, could be deleterious. A better understanding of the nature of memory TFH populations is also essential in order to develop and test interventions. Knowledge of factors that influence TFH function in HIV infection could lead to improved immune reconstitution in ART-treated individuals and potentially augment strategies to cure HIV infection.
Author Contributions
All the authors listed have made substantial, direct, and intellectual contribution to the work and approved it for publication.
Conflict of Interest Statement
The authors claim no financial conflicts of interest, and no outside parties influenced the writing of this manuscript.
Funding
Funding was provided by NIH/NIAID grants R01 AI096966 to and UM1 AI26617 to EC, T32 AI007447 to BM, and T32 AI007405 to SM.
References
1. Breitfeld D, Ohl L, Kremmer E, Ellwart J, Sallusto F, Lipp M, et al. Follicular B helper T cells express CXC chemokine receptor 5, localize to B cell follicles, and support immunoglobulin production. J Exp Med (2000) 192(11):1545–52. doi: 10.1084/jem.192.11.1545
2. Schaerli P, Willimann K, Lang AB, Lipp M, Loetscher P, Moser B. CXC chemokine receptor 5 expression defines follicular homing T cells with B cell helper function. J Exp Med (2000) 192(11):1553–62. doi:10.1084/jem.192.11.1553
3. Kim CH, Rott LS, Clark-Lewis I, Campbell DJ, Wu L, Butcher EC. Subspecialization of Cxcr5+ T cells B helper activity is focused in a germinal center–localized subset of Cxcr5+ T cells. J Exp Med (2001) 193(12):1373–82. doi:10.1084/jem.193.12.1373
4. Johnston RJ, Poholek AC, DiToro D, Yusuf I, Eto D, Barnett B, et al. Bcl6 and Blimp-1 are reciprocal and antagonistic regulators of T follicular helper cell differentiation. Science (2009) 325(5943):1006–10. doi:10.1126/science.1175870
5. Nurieva RI, Chung Y, Martinez GJ, Yang XO, Tanaka S, Matskevitch TD, et al. Bcl6 mediates the development of T follicular helper cells. Science (2009) 325(5943):1001–5. doi:10.1126/science.1176676
6. Yu D, Rao S, Tsai LM, Lee SK, He Y, Sutcliffe EL, et al. The transcriptional repressor Bcl-6 directs T follicular helper cell lineage commitment. Immunity (2009) 31(3):457–68. doi:10.1016/j.immuni.2009.07.002
7. Rasheed AU, Rahn HP, Sallusto F, Lipp M, Müller G. Follicular B helper T cell activity is confined to CXCR5hiICOShi CD4 T cells and is independent of CD57 expression. Eur J Immunol (2006) 36(7):1892–903. doi:10.1002/eji.200636136
8. Nurieva RI, Chung Y, Hwang D, Yang XO, Kang HS, Ma L, et al. Generation of T follicular helper cells is mediated by interleukin-21 but independent of T helper 1, 2, or 17 cell lineages. Immunity (2008) 29(1):138–49. doi:10.1016/j.immuni.2008.05.009
9. Choi YS, Kageyama R, Eto D, Escobar TC, Johnston RJ, Monticelli L, et al. ICOS receptor instructs T follicular helper cell versus effector cell differentiation via induction of the transcriptional repressor Bcl6. Immunity (2011) 34(6):932–46. doi:10.1016/j.immuni.2011.03.023
10. Akiba H, Takeda K, Kojima Y, Usui Y, Harada N, Yamazaki T, et al. The role of ICOS in the CXCR5+ follicular B helper T cell maintenance in vivo. J Immunol (2005) 175(4):2340–8. doi:10.4049/jimmunol.175.4.2340
11. Haynes NM, Allen CD, Lesley R, Ansel KM, Killeen N, Cyster JG. Role of CXCR5 and CCR7 in follicular Th cell positioning and appearance of a programmed cell death gene-1high germinal center-associated subpopulation. J Immunol (2007) 179(8):5099–108. doi:10.4049/jimmunol.179.8.5099
12. Pène J, Gauchat J-F, Lécart S, Drouet E, Guglielmi P, Boulay V, et al. Cutting edge: IL-21 is a switch factor for the production of IgG1 and IgG3 by human B cells. J Immunol (2004) 172(9):5154–7. doi:10.4049/jimmunol.172.9.5154
13. Good KL, Bryant VL, Tangye SG. Kinetics of human B cell behavior and amplification of proliferative responses following stimulation with IL-21. J Immunol (2006) 177(8):5236–47. doi:10.4049/jimmunol.177.8.5236
14. Bryant VL, Ma CS, Avery DT, Li Y, Good KL, Corcoran LM, et al. Cytokine-mediated regulation of human B cell differentiation into Ig-secreting cells: predominant role of IL-21 produced by CXCR5+ T follicular helper cells. J Immunol (2007) 179(12):8180–90. doi:10.4049/jimmunol.179.12.8180
15. Fazilleau N, Mark L, McHeyzer-Williams LJ, McHeyzer-Williams MG. Follicular helper T cells: lineage and location. Immunity (2009) 30(3):324–35. doi:10.1016/j.immuni.2009.03.003
16. Leonard WJ, Spolski R. Interleukin-21: a modulator of lymphoid proliferation, apoptosis and differentiation. Nat Rev Immunol (2005) 5(9):688–98. doi:10.1038/nri1688
17. Yusuf I, Kageyama R, Monticelli L, Johnston RJ, DiToro D, Hansen K, et al. Germinal center T follicular helper cell IL-4 production is dependent on signaling lymphocytic activation molecule receptor (CD150). J Immunol (2010) 185(1):190–202. doi:10.4049/jimmunol.0903505
18. Hsu H-C, Yang P, Wang J, Wu Q, Myers R, Chen J, et al. Interleukin 17–producing T helper cells and interleukin 17 orchestrate autoreactive germinal center development in autoimmune BXD2 mice. Nat Immunol (2008) 9(2):166–75. doi:10.1038/ni1552
19. Sarra M, Monteleone I, Stolfi C, Fantini MC, Sileri P, Sica G, et al. Interferon-gamma-expressing cells are a major source of interleukin-21 in inflammatory bowel diseases. Inflamm Bowel Dis (2010) 16(8):1332–9. doi:10.1002/ibd.21238
20. Lim HW, Kim CH. Loss of IL-7 receptor α on CD4+ T cells defines terminally differentiated B cell-helping effector T cells in a B cell-rich lymphoid tissue. J Immunol (2007) 179(11):7448–56. doi:10.4049/jimmunol.179.11.7448
21. Morita R, Schmitt N, Bentebibel S-E, Ranganathan R, Bourdery L, Zurawski G, et al. Human blood CXCR5+ CD4+ T cells are counterparts of T follicular cells and contain specific subsets that differentially support antibody secretion. Immunity (2011) 34(1):108–21. doi:10.1016/j.immuni.2011.01.009
22. Pallikkuth S, Parmigiani A, Silva SY, George VK, Fischl M, Pahwa R, et al. Impaired peripheral blood T-follicular helper cell function in HIV-infected nonresponders to the 2009 H1N1/09 vaccine. Blood (2012) 120(5):985–93. doi:10.1182/blood-2011-12-396648
23. Locci M, Havenar-Daughton C, Landais E, Wu J, Kroenke MA, Arlehamn CL, et al. Human circulating PD-1+ CXCR3− CXCR5+ memory Tfh cells are highly functional and correlate with broadly neutralizing HIV antibody responses. Immunity (2013) 39(4):758–69. doi:10.1016/j.immuni.2013.08.031
24. Boswell KL, Paris R, Boritz E, Ambrozak D, Yamamoto T, Darko S, et al. Loss of circulating CD4 T cells with B cell helper function during chronic HIV infection. PLoS Pathog (2014) 10(1):e1003853. doi:10.1371/journal.ppat.1003853
25. Iyer SS, Gangadhara S, Victor B, Gomez R, Basu R, Hong JJ, et al. Codelivery of envelope protein in alum with MVA vaccine induces CXCR3-biased CXCR5+ and CXCR5− CD4 T cell responses in Rhesus macaques. J Immunol (2015) 195(3):994–1005. doi:10.4049/jimmunol.1500083
26. Chung Y, Tanaka S, Chu F, Nurieva RI, Martinez GJ, Rawal S, et al. Follicular regulatory T cells expressing Foxp3 and Bcl-6 suppress germinal center reactions. Nat Med (2011) 17(8):983–8. doi:10.1038/nm.2426
27. Linterman MA, Pierson W, Lee SK, Kallies A, Kawamoto S, Rayner TF, et al. Foxp3+ follicular regulatory T cells control the germinal center response. Nat Med (2011) 17(8):975–82. doi:10.1038/nm.2425
28. Wollenberg I, Agua-Doce A, Hernández A, Almeida C, Oliveira VG, Faro J, et al. Regulation of the germinal center reaction by Foxp3+ follicular regulatory T cells. J Immunol (2011) 187(9):4553–60. doi:10.4049/jimmunol.1101328
29. Sage PT, Francisco LM, Carman CV, Sharpe AH. The receptor PD-1 controls follicular regulatory T cells in the lymph nodes and blood. Nat Immunol (2013) 14(2):152–61. doi:10.1038/ni.2496
30. Kawamoto S, Maruya M, Kato LM, Suda W, Atarashi K, Doi Y, et al. Foxp3+ T cells regulate immunoglobulin a selection and facilitate diversification of bacterial species responsible for immune homeostasis. Immunity (2014) 41(1):152–65. doi:10.1016/j.immuni.2014.05.016
31. León B, Bradley JE, Lund FE, Randall TD, Ballesteros-Tato A. FoxP3+ regulatory T cells promote influenza-specific Tfh responses by controlling IL-2 availability. Nat Commun (2014) 5:3495. doi:10.1038/ncomms4495
32. Sage PT, Paterson AM, Lovitch SB, Sharpe AH. The coinhibitory receptor CTLA-4 controls B cell responses by modulating T follicular helper, T follicular regulatory, and T regulatory cells. Immunity (2014) 41(6):1026–39. doi:10.1016/j.immuni.2014.12.005
33. Wing JB, Ise W, Kurosaki T, Sakaguchi S. Regulatory T cells control antigen-specific expansion of Tfh cell number and humoral immune responses via the coreceptor CTLA-4. Immunity (2014) 41(6):1013–25. doi:10.1016/j.immuni.2014.12.006
34. Hufert FT, van Lunzen J, Janossy G, Bertram S, Schmitz J, Haller O, et al. Germinal centre CD4+ T cells are an important site of HIV replication in vivo. AIDS (1997) 11(7):849–57. doi:10.1097/00002030-199707000-00003
35. Folkvord JM, Armon C, Connick E. Lymphoid follicles are sites of heightened human immunodeficiency virus type 1 (HIV-1) replication and reduced antiretroviral effector mechanisms. AIDS Res Hum Retroviruses (2005) 21(5):363–70. doi:10.1089/aid.2005.21.363
36. Connick E, Mattila T, Folkvord JM, Schlichtemeier R, Meditz AL, Ray MG, et al. CTL fail to accumulate at sites of HIV-1 replication in lymphoid tissue. J Immunol (2007) 178(11):6975–83. doi:10.4049/jimmunol.178.11.6975
37. Perreau M, Savoye A-L, De Crignis E, Corpataux J-M, Cubas R, Haddad EK, et al. Follicular helper T cells serve as the major CD4 T cell compartment for HIV-1 infection, replication, and production. J Exp Med (2013) 210(1):143–56. doi:10.1084/jem.20121932
38. Kohler SL, Pham MN, Folkvord JM, Arends T, Miller SM, Miles B, et al. Germinal center T follicular helper cells are highly permissive to HIV-1 and alter their phenotype during virus replication. J Immunol (2016) 196(6):2711–22. doi:10.4049/jimmunol.1502174
39. Connick E, Folkvord JM, Lind KT, Rakasz EG, Miles B, Wilson NA, et al. Compartmentalization of simian immunodeficiency virus replication within secondary lymphoid tissues of rhesus macaques is linked to disease stage and inversely related to localization of virus-specific CTL. J Immunol (2014) 193(11):5613–25. doi:10.4049/jimmunol.1401161
40. Thacker TC, Zhou X, Estes JD, Jiang Y, Keele BF, Elton TS, et al. Follicular dendritic cells and human immunodeficiency virus type 1 transcription in CD4+ T cells. J Virol (2009) 83(1):150–8. doi:10.1128/JVI.01652-08
41. Spiegel H, Herbst H, Niedobitek G, Foss H-D, Stein H. Follicular dendritic cells are a major reservoir for human immunodeficiency virus type 1 in lymphoid tissues facilitating infection of CD4+ T-helper cells. Am J Pathol (1992) 140(1):15.
42. Heath SL, Tew JG, Tew JG, Szakal AK, Burton GF. Follicular dendritic cells and human immunodeficiency virus infectivity. Nature (1995) 377(6551):740–4. doi:10.1038/377740a0
43. Haase AT, Henry K, Zupancic M, Sedgewick G, Faust RA, Melroe H, et al. Quantitative image analysis of HIV-1 infection in lymphoid tissue. Science (1996) 274(5289):985–9. doi:10.1126/science.274.5289.985
44. Tjernlund A, Zhu J, Laing K, Diem K, McDonald D, Vazquez J, et al. In situ detection of Gag-specific CD8+ cells in the GI tract of SIV infected Rhesus macaques. Retrovirology (2010) 7(1):12. doi:10.1186/1742-4690-7-12
45. Li S, Folkvord JM, Rakasz EG, Abdelaal HM, Wagstaff RK, Kovacs KJ, et al. SIV-producing cells in follicles are partially suppressed by CD8+ cells in vivo. J Virol (2016):1332–1316. doi:10.1128/JVI.01332-16
46. Lindqvist M, van Lunzen J, Soghoian DZ, Kuhl BD, Ranasinghe S, Kranias G, et al. Expansion of HIV-specific T follicular helper cells in chronic HIV infection. J Clin Invest (2012) 122(9):3271–80. doi:10.1172/JCI64314
47. Petrovas C, Yamamoto T, Gerner MY, Boswell KL, Wloka K, Smith EC, et al. CD4 T follicular helper cell dynamics during SIV infection. J Clin Invest (2012) 122(9):3281–94. doi:10.1172/JCI63039
48. Miles B, Miller SM, Folkvord JM, Kimball A, Chamanian M, Meditz AL, et al. Follicular regulatory T cells impair follicular T helper cells in HIV and SIV infection. Nat Commun (2015) 6:8608. doi:10.1038/ncomms9608
49. Hong JJ, Amancha PK, Rogers K, Ansari AA, Villinger F. Spatial alterations between CD4+ T follicular helper, B, and CD8+ T cells during simian immunodeficiency virus infection: T/B cell homeostasis, activation, and potential mechanism for viral escape. J Immunol (2012) 188(7):3247–56. doi:10.4049/jimmunol.1103138
50. Baumjohann D, Preite S, Reboldi A, Ronchi F, Ansel KM, Lanzavecchia A, et al. Persistent antigen and germinal center B cells sustain T follicular helper cell responses and phenotype. Immunity (2013) 38(3):596–605. doi:10.1016/j.immuni.2012.11.020
51. Nakajima K, Martinez-Maza O, Hirano T, Breen E, Nishanian P, Salazar-Gonzalez J, et al. Induction of IL-6 (B cell stimulatory factor-2/IFN-beta 2) production by HIV. J Immunol (1989) 142(2):531–6.
52. Fan J, Bass H, Fahey J. Elevated IFN-gamma and decreased IL-2 gene expression are associated with HIV infection. J Immunol (1993) 151(9):5031–40.
53. Harari A, Rizzardi GP, Ellefsen K, Ciuffreda D, Champagne P, Bart P-A, et al. Analysis of HIV-1–and CMV-specific memory CD4 T-cell responses during primary and chronic infection. Blood (2002) 100(4):1381–7. doi:10.1182/blood-2001-11-0080
54. Haas MK, Levy DN, Folkvord JM, Connick E. Distinct patterns of Bcl-2 expression occur in R5-and X4-tropic HIV-1-producing lymphoid tissue cells infected ex vivo. AIDS Res Hum Retroviruses (2015) 31(3):298–304. doi:10.1089/aid.2014.0155
55. Wang C, Hillsamer P, Kim CH. Phenotype, effector function, and tissue localization of PD-1-expressing human follicular helper T cell subsets. BMC Immunol (2011) 12(1):53. doi:10.1186/1471-2172-12-53
56. Lane HC, Masur H, Edgar LC, Whalen G, Rook AH, Fauci AS. Abnormalities of B-cell activation and immunoregulation in patients with the acquired immunodeficiency syndrome. N Engl J Med (1983) 309(8):453–8. doi:10.1056/NEJM198308253090803
57. De Milito A, Mörch C, Sönnerborg A, Chiodi F. Loss of memory (CD27) B lymphocytes in HIV-1 infection. AIDS (2001) 15(8):957–64. doi:10.1097/00002030-200105250-00003
58. Cubas RA, Mudd JC, Savoye AL, Perreau M, van Grevenynghe J, Metcalf T, et al. Inadequate T follicular cell help impairs B cell immunity during HIV infection. Nat Med (2013) 19(4):494–9. doi:10.1038/nm.3109
59. Colineau L, Rouers A, Yamamoto T, Xu Y, Urrutia A, Pham HP, et al. HIV-infected spleens present altered follicular helper T cell (Tfh) subsets and skewed B cell maturation. PLoS One (2015) 10(10):e0140978. doi:10.1371/journal.pone.0140978
60. Yamamoto T, Lynch RM, Gautam R, Matus-Nicodemos R, Schmidt SD, Boswell KL, et al. Quality and quantity of TFH cells are critical for broad antibody development in SHIVAD8 infection. Sci Transl Med (2015) 7(298):ra120–298. doi:10.1126/scitranslmed.aab3964
61. Muir R, Metcalf T, Tardif V, Takata H, Phanuphak N, Kroon E, et al. Altered memory circulating T follicular helper-B cell interaction in early acute HIV infection. PLoS Pathog (2016) 12(7):e1005777. doi:10.1371/journal.ppat.1005777
62. Iannello A, Tremblay C, Routy JP, Boulassel MR, Toma E, Ahmad A. Decreased levels of circulating IL-21 in HIV-infected AIDS patients: correlation with CD4+ T-cell counts. Viral Immunol (2008) 21(3):385–8. doi:10.1089/vim.2008.0025
63. Iannello A, Boulassel MR, Samarani S, Debbeche O, Tremblay C, Toma E, et al. Dynamics and consequences of IL-21 production in HIV-infected individuals: a longitudinal and cross-sectional study. J Immunol (2010) 184(1):114–26. doi:10.4049/jimmunol.0901967
64. Kaltenmeier C, Gawanbacht A, Beyer T, Lindner S, Trzaska T, van der Merwe JA, et al. CD4+ T cell–derived IL-21 and deprivation of CD40 signaling favor the in vivo development of granzyme B–expressing regulatory B cells in HIV patients. J Immunol (2015) 194(8):3768–77. doi:10.4049/jimmunol.1402568
65. Moir S, Ogwaro KM, Malaspina A, Vasquez J, Donoghue ET, Hallahan CW, et al. Perturbations in B cell responsiveness to CD4+ T cell help in HIV-infected individuals. Proc Natl Acad Sci U S A (2003) 100(10):6057–62. doi:10.1073/pnas.0730819100
66. Velu V, Mylvaganam GH, Gangadhara S, Hong JJ, Iyer SS, Gumber S, et al. Induction of Th1-biased T follicular helper (Tfh) cells in lymphoid tissues during chronic simian immunodeficiency virus infection defines functionally distinct germinal center Tfh cells. J Immunol (2016) 197(5):1832–42. doi:10.4049/jimmunol.1600143
67. Li Y, Migueles SA, Welcher B, Svehla K, Phogat A, Louder MK, et al. Broad HIV-1 neutralization mediated by CD4-binding site antibodies. Nat Med (2007) 13(9):1032–4. doi:10.1038/nm1624
68. Sather DN, Armann J, Ching LK, Mavrantoni A, Sellhorn G, Caldwell Z, et al. Factors associated with the development of cross-reactive neutralizing antibodies during human immunodeficiency virus type 1 infection. J Virol (2009) 83(2):757–69. doi:10.1128/jvi.02036-08
69. Schuurman HJ, Krone WJ, Broekhuizen R, Goudsmit J. Expression of RNA and antigens of human immunodeficiency virus type-1 (HIV-1) in lymph nodes from HIV-1 infected individuals. Am J Pathol (1988) 133(3):516–24.
70. Orenstein JM, Feinberg M, Yoder C, Schrager L, Mican JM, Schwartzentruber DJ, et al. Lymph node architecture preceding and following 6 months of potent antiviral therapy: follicular hyperplasia persists in parallel with p24 antigen restoration after involution and CD4 cell depletion in an AIDS patient. AIDS (1999) 13(16):2219–29. doi:10.1097/00002030-199911120-00004
71. Binley JM, Wrin T, Korber B, Zwick MB, Wang M, Chappey C, et al. Comprehensive cross-clade neutralization analysis of a panel of anti-human immunodeficiency virus type 1 monoclonal antibodies. J Virol (2004) 78(23):13232–52. doi:10.1128/JVI.78.23.13232-13252.2004
72. Doria-Rose NA, Klein RM, Daniels MG, O’Dell S, Nason M, Lapedes A, et al. Breadth of human immunodeficiency virus-specific neutralizing activity in sera: clustering analysis and association with clinical variables. J Virol (2010) 84(3):1631–6. doi:10.1128/JVI.01482-09
73. Chevalier MF, Jülg B, Pyo A, Flanders M, Ranasinghe S, Soghoian DZ, et al. HIV-1-specific interleukin-21+ CD4+ T cell responses contribute to durable viral control through the modulation of HIV-specific CD8+ T cell function. J Virol (2011) 85(2):733–41. doi:10.1128/JVI.02030-10
74. Cubas R, van Grevenynghe J, Wills S, Kardava L, Santich BH, Buckner CM, et al. Reversible reprogramming of circulating memory T follicular helper cell function during chronic HIV infection. J Immunol (2015) 195(12):5625–36. doi:10.4049/jimmunol.1501524
75. Bekele Y, Amu S, Bobosha K, Lantto R, Nilsson A, Endale B, et al. Impaired phenotype and function of T follicular helper cells in HIV-1-infected children receiving ART. Medicine (2015) 94(27):e1125. doi:10.1097/MD.0000000000001125
76. Schultz BT, Teigler JE, Pissani F, Oster AF, Kranias G, Alter G, et al. Circulating HIV-specific interleukin-21(+)CD4(+) T cells represent peripheral Tfh cells with antigen-dependent helper functions. Immunity (2016) 44(1):167–78. doi:10.1016/j.immuni.2015.12.011
77. Kroon FP, van Dissel JT, de Jong JC, van Furth R. Antibody response to influenza, tetanus and pneumococcal vaccines in HIV-seropositive individuals in relation to the number of CD4+ lymphocytes. AIDS (1994) 8(4):469–76. doi:10.1097/00002030-199404000-00008
78. Tebas P, Frank I, Lewis M, Quinn J, Zifchak L, Thomas A, et al. Poor immunogenicity of the H1N1 2009 vaccine in well controlled HIV-infected individuals. AIDS (2010) 24(14):2187–92. doi:10.1097/QAD.0b013e32833c6d5c
79. Bentebibel S-E, Lopez S, Obermoser G, Schmitt N, Mueller C, Harrod C, et al. Induction of ICOS+CXCR3+CXCR5+ TH cells correlates with antibody responses to influenza vaccination. Sci Transl Med (2013) 5(176):ra132–76. doi:10.1126/scitranslmed.3005191
80. Spensieri F, Borgogni E, Zedda L, Bardelli M, Buricchi F, Volpini G, et al. Human circulating influenza-CD4+ ICOS1+ IL-21+ T cells expand after vaccination, exert helper function, and predict antibody responses. Proc Natl Acad Sci U S A (2013) 110(35):14330–5. doi:10.1073/pnas.1311998110
81. Pallikkuth S, Kanthikeel SP, Silva SY, Fischl M, Pahwa R, Pahwa S. Upregulation of IL-21 receptor on B cells and IL-21 secretion distinguishes novel 2009 H1N1 vaccine responders from nonresponders among HIV-infected persons on combination antiretroviral therapy. J Immunol (2011) 186(11):6173–81. doi:10.4049/jimmunol.1100264
82. Cagigi A, Pensieroso S, Ruffin N, Sammicheli S, Thorstensson R, Pan-Hammarström Q, et al. Relation of activation-induced deaminase (AID) expression with antibody response to A (H1N1) pdm09 vaccination in HIV-1 infected patients. Vaccine (2013) 31(18):2231–7. doi:10.1016/j.vaccine.2013.03.002
83. Siewe B, Stapleton JT, Martinson J, Keshavarzian A, Kazmi N, Demarais PM, et al. Regulatory B cell frequency correlates with markers of HIV disease progression and attenuates anti-HIV CD8+ T cell function in vitro. J Leukoc Biol (2013) 93(5):811–8. doi:10.1189/jlb.0912436
84. Vaeth M, Müller G, Stauss D, Dietz L, Klein-Hessling S, Serfling E, et al. Follicular regulatory T cells control humoral autoimmunity via NFAT2-regulated CXCR5 expression. J Exp Med (2014) 211(3):545–61. doi:10.1084/jem.20130604
85. Preite S, Baumjohann D, Foglierini M, Basso C, Ronchi F, Rodriguez BMF, et al. Somatic mutations and affinity maturation are impaired by excessive numbers of T follicular helper cells and restored by Treg cells or memory T cells. Eur J Immunol (2015) 45(11):3010–21. doi:10.1002/eji.201545920
86. Chowdhury A, Del Rio PME, Tharp GK, Trible RP, Amara RR, Chahroudi A, et al. Decreased T follicular regulatory cell/T follicular helper cell (TFH) in simian immunodeficiency virus–infected rhesus macaques may contribute to accumulation of TFH in chronic infection. J Immunol (2015) 195(7):3237–47. doi:10.4049/jimmunol.1502269
87. Blackburn MJ, Zhong-Min M, Caccuri F, McKinnon K, Schifanella L, Guan Y, et al. Regulatory and helper follicular T cells and antibody avidity to simian immunodeficiency virus glycoprotein 120. J Immunol (2015) 195(7):3227–36. doi:10.4049/jimmunol.1402699
88. Bachmann MF, Odermatt B, Hengartner H, Zinkernagel RM. Induction of long-lived germinal centers associated with persisting antigen after viral infection. J Exp Med (1996) 183(5):2259–69. doi:10.1084/jem.183.5.2259
89. Fazilleau N, Eisenbraun MD, Malherbe L, Ebright JN, Pogue-Caley RR, McHeyzer-Williams LJ, et al. Lymphoid reservoirs of antigen-specific memory T helper cells. Nat Immunol (2007) 8(7):753–61. doi:10.1038/ni1472
90. Hale JS, Youngblood B, Latner DR, Mohammed AUR, Ye L, Akondy RS, et al. Distinct memory CD4+ T cells with commitment to T follicular helper-and T helper 1-cell lineages are generated after acute viral infection. Immunity (2013) 38(4):805–17. doi:10.1016/j.immuni.2013.02.020
91. Iyer SS, Latner DR, Zilliox MJ, McCausland M, Akondy RS, Penaloza-MacMaster P, et al. Identification of novel markers for mouse CD4+ T follicular helper cells. Eur J Immunol (2013) 43(12):3219–32. doi:10.1002/eji.201343469
92. Weber JP, Fuhrmann F, Hutloff A. T-follicular helper cells survive as long-term memory cells. Eur J Immunol (2012) 42(8):1981–8. doi:10.1002/eji.201242540
93. Hale JS, Ahmed R. Memory T follicular helper CD4 T cells. Front Immunol (2015) 6:16. doi:10.3389/fimmu.2015.00016
94. Shulman Z, Gitlin AD, Targ S, Jankovic M, Pasqual G, Nussenzweig MC, et al. T follicular helper cell dynamics in germinal centers. Science (2013) 341(6146):673–7. doi:10.1126/science.1241680
95. Sage PT, Alvarez D, Godec J, von Andrian UH, Sharpe AH. Circulating T follicular regulatory and helper cells have memory-like properties. J Clin Invest (2014) 124(12):5191–204. doi:10.1172/JCI76861
Keywords: follicular T helper cells, follicular T regulatory cells, germinal center, broadly neutralizing antibodies, HIV
Citation: Miles B, Miller SM and Connick E (2016) CD4 T Follicular Helper and Regulatory Cell Dynamics and Function in HIV Infection. Front. Immunol. 7:659. doi: 10.3389/fimmu.2016.00659
Received: 05 October 2016; Accepted: 16 December 2016;
Published: 27 December 2016
Edited by:
Smita S. Iyer, Emory University, USAReviewed by:
Anthony Jaworowski, Burnet Institute, AustraliaSilvia Vendetti, Istituto Superiore di Sanità, Italy
Copyright: © 2016 Miles, Miller and Connick. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Elizabeth Connick, Y29ubmlja2VAZW1haWwuYXJpem9uYS5lZHU=
 Brodie Miles
Brodie Miles Shannon M. Miller2
Shannon M. Miller2