- 1Regenerative Medicine Institute (REMEDI), School of Medicine, College of Medicine, Nursing and Health Sciences, National University of Ireland Galway, Galway, Ireland
- 2Discipline of Pharmacology and Therapeutics, School of Medicine, College of Medicine, Nursing and Health Sciences, National University of Ireland Galway, Galway, Ireland
- 3CURAM Centre for Research in Medical Devices, National University of Ireland, Galway, Ireland
Mesenchymal stromal cells (MSC) have been used to treat a broad range of disease indications such as acute and chronic inflammatory disorders, autoimmune diseases, and transplant rejection due to their potent immunosuppressive/anti-inflammatory properties. The breadth of their usage is due in no small part to the vast quantity of published studies showing their ability to modulate multiple immune cell types of both the innate and adaptive immune response. While patient-derived (autologous) MSC may be the safer choice in terms of avoiding unwanted immune responses, factors including donor comorbidities may preclude these cells from use. In these situations, allogeneic MSC derived from genetically unrelated individuals must be used. While allogeneic MSC were initially believed to be immune-privileged, substantial evidence now exists to prove otherwise with multiple studies documenting specific cellular and humoral immune responses against donor antigens following administration of these cells. In this article, we will review recent published studies using non-manipulated, inflammatory molecule-activated (licensed) and differentiated allogeneic MSC, as well as MSC extracellular vesicles focusing on the immune responses to these cells and whether or not such responses have an impact on allogeneic MSC-mediated safety and efficacy.
Introduction
Mesenchymal stem/stromal cells (MSC) are a readily accessible cell source in which interest has expanded hugely in the last two decades (1). The allogeneic use of these cells was touted as being safe owing to their low expression of major histocompatibility complex (MHC) and co-stimulatory molecules and the fact that they can suppress the activity of numerous immune cell populations (2–4). However, in more recent years several groups have shown in different models that MSC do induce anti-donor immune responses from different facets of the immune system, including dendritic cells, T cells, and B-cell mediated allo-antibodies (4–7). Despite the fact that allogeneic MSC can be recognized by the host immune system, administration of these cells to human subjects has been shown, for the most part, to be safe in several different disease settings (5, 8–10). There are a small number of studies describing several treatment emergent adverse events (TEAE) following administration of decidual stromal cells or adipose-derived stromal cells to patients, however, it appears that these are unrelated to the development of anti-donor immune responses (10–13). However, the field should remain vigilant to adverse immune responses which may have detrimental effects in the long term or if the patient receives a transplant later in life which may be rejected as a result of a pre-existing anti-donor memory response, either cellular or humoral (5). To this end, here we have critically assessed recent allogeneic MSC literature as a follow up to our previous review with a focus on studies which have investigated the immune response to allogeneic MSC either in vitro, in vivo, or as part of a clinical trial to update the field on the potential responses to these cells (5).
Immunogenicity and Immunomodulatory Potential of Allogeneic Non-Manipulated MSC
The advantages of using allogeneic over autologous MSC have been well documented with perhaps the most compelling advantage being the ability to obtain cells from healthy donors followed by expansion in vitro to clinically relevant numbers. The cells can then be banked and transported where required with minimal delay. Another commonly touted advantage of allogeneic MSC is their low immunogenicity and while there may be some truth in this when compared to other allogeneic cell types, emerging evidence suggests that allogeneic MSC can indeed induce a strong immune response in vivo which may have severe consequences depending on the disease indication for which the cells are being administered (14). Even in vitro, it has been shown that stimulating MHC class II negative equine MSC with interferon (IFN)-γ for 4 days leads to markedly increased expression of MHC class II (15, 16). One could reasonably assume that, in an allogeneic in vivo setting, administered MSC would encounter significant levels of IFN-γ which may impair their efficacy, depending on the indication. A study by Joswig et al. (17) assessed the clinical response to repeated intra-articular injections of bone marrow-derived equine allogeneic versus autologous MSC. Clinical parameters assessed in the study showed no differences after the first injection of either autologous or allogeneic MSC. However, following the second injection, a significant adverse response of the joint was seen in horses treated with allogeneic compared to autologous MSC, evidenced by elevated synovial total nucleated cell counts. In the same model, another recent study could demonstrate that anti-sera collected from horses injected with MHC-mismatched MSC contained antibodies that caused the death of equine leukocyte antigen-A2 haplotype MSC in cytotoxicity assays (4, 18). By contrast, Ardanaz and colleagues (19) reported no persistent unwanted effects, including the absence of a hypersensitivity response, following single or repeated intra-articular injections of equine allogeneic MSC from pooled donors. In this study, only a transitory inflammatory response was observed which resolved after 10 days.
Owing to the similarities between the immune systems of humans and non-human primates, immunological analysis of allogeneic MSC in these pre-clinical animal models may have important implications from a clinical perspective. Recently, Isakova and co-workers (20) performed such a study. The aim of the study was to evaluate the immune response caused by intra-cranial injection of allogeneic compared to autologous MSC in rhesus macaques. The authors detected clear signs of allo-recognition as evidenced by significantly higher levels of circulating allo-specific antibodies in serum of macaques that received allogeneic compared to autologous MSC. They also demonstrated, in in vitro co-cultures, that peripheral blood mononuclear cells (PBMCs) isolated from allogeneic but not autologous MSC recipients were capable of lysing their respective donor MSC. Furthermore, higher levels of peripheral blood-derived natural killer, B and T cell subsets were recorded in allogeneic MSC recipients with the overall magnitude of the allo-immune response determined by the level of mismatch between the MSC donor and recipient. A detailed understanding of the consequences of these reported changes in immune profiles following allo-MSC therapy is likely to advance the field even further.
Another emerging concept in relation to allo-MSC therapy that should be considered is the idea that allo-MSC cell death following administration may play a role in regulation of ensuing immune responses. Immune responses to dying cells can be affected by multiple factors related to the type of cell death (21). These include, but are not limited to, the cell death pathway, the way dead cells are cleared by innate immune effectors, the nature and phenotype of the effector immune cells, the location of cell clearance, and the immune cell effectors that eventually encounter the antigens presented along with the dead cells (21). Another key consideration is whether to use fresh or cryopreserved cells, as this may impact cell viability and consequent immune responses (22). Indeed, a recent report by Chinnadurai and colleagues demonstrated that cryopreserved MSC were susceptible to T cell-mediated apoptosis (23). In the context of allo-MSC therapy, consideration of all of these issues is likely to shed light on the impact of death of allo-MSC on the immune system, and the implications for the potential of allo-MSC therapeutic efficacy.
Immunogenicity and Immunomodulatory Potential of Licensed Allogeneic MSC
Few studies have investigated the effects of licensing (treatment with pro-inflammatory stimuli in vitro) on allogeneic MSC, particularly in vivo. This is most likely because these cells will be targeted by recipient immune cells upon their first encounter in an in vivo setting and will receive an inflammatory stimulus, thereby, potentially, negating the need to pre-activate these cells in vitro. However, there are some reports using this strategy with the focus primarily being to further enhance the cells’ immunosuppressive ability, as opposed to investigating whether immunogenicity has been increased. In one such study, Mancheno-Corvo and colleagues assessed the ability of adipose-derived MSC (ASC) to inhibit allogeneic T cell proliferation. They correctly pointed out that the majority of similar studies activate T cells at the same time the cells first encounter the MSC in in vitro co-culture assays and that such an approach may not be relevant to typical clinical scenarios in which MSC, administered after the onset of symptoms associated with inflammatory disorder, such as Crohn’s disease or rheumatoid arthritis, will encounter and interact with immune cells (e.g., T cells) that are already activated. Upon analysis, they found that ASC co-cultured with T lymphocytes that had been pre-activated for 48 h beforehand had impaired immunosuppressive capacity. However, this ability to inhibit allogeneic T lymphocyte proliferation was restored when ASC were pre-activated with IFN-γ, provided the co-culture was performed in the medium conditioned by pre-activated ASC and not fresh medium. Intriguingly, pre-treatment with other candidate cytokines tumor necrosis factor (TNF)-α, interleukin (IL)-1β, IL-17, tissue growth factor-β, or stromal cell-derived factor-1α had no reported effect (24).
Little has been reported to date on the use of cytokine-licensed MSC and their immunosuppressive effects on T cell function. Chinnadurai et al., in a recent report, found that only IFN-γ licensed human (h)MSC were able to inhibit the secretion of the key Th1-related cytokines IFN-γ, TNF-α, and IL-2 by allogeneic T cells. In the same experiments, un-licensed MSC had no immunomodulatory effect (23). Moreover, they identified the MSC-expressed, IFN-γ-licensed inhibitory molecules B7H1 and B7DC/programmed death receptor 1 (PD1) pathways as essential effectors in blocking T cell function. The authors also found that, in vivo, licensing-induced efficacy was dependent on whether the MSC were used fresh or from frozen (23). While indoleamine 2,3-dioxygenase (IDO) activity was increased in these licensed MSC, its function was largely dispensable with regard to MSC-driven inhibition of T cell effector function (25).
While the inflammatory cytokines IFN-γ, TNF-α, and IL-1β are by the far the most commonly used molecules to license MSC, Sivanathan et al. employed a novel approach to licensing hMSC by pre-activating the cells with IL-17 (MSC-17). Their rationale for this approach was that, while IFN-γ licensed MSC are more immunosuppressive than their un-licensed counterparts they also upregulate MHC I thereby becoming potentially more immunogeneic in vivo. Unlike IFN-γ licensed MSC, MSC-17 do not upregulate MHC I (or MHC II), nor the T cell co-stimulatory marker CD40. Furthermore, MSC-17 could inhibit the production of Th1-related cytokines IFN-γ, TNF-α, and IL-2 by allogeneic T cells, in addition to inhibiting surface CD25 expression. In addition, and potentially crucial with regard to therapeutic efficacy, MSC-17 but not IFN-γ licensed MSC consistently induced CD4+ CD25high CD127low FoxP3+ regulatory T cells (iTregs) from phytohemagglutinin-activated CD4+ CD25− T cells (26). As mentioned previously, the majority of studies use licensing as a way to further enhance MSC immunosuppressive ability. However, Roemeling van-Rhijn and colleagues (27) focused on the effect(s) of licensing on the immunogenicity of allogeneic MSC by evaluating whether or not repeated exposure to allogeneic MSC [derived from bone marrow (BM-MSC)] and/or adipose tissue (ASC) induced human leukocyte antigen (HLA) class I specific lysis by CD8+ T cells in a human setting. They also tested what effects exposure to IFN-γ had on the MSC and their ability to induce CD8+ cytotoxic T cell reactivity. They found that MSC-educated CD8+ T cells were able to lyse BM-MSC in an HLA-specific manner. Interestingly, in addition to an observed increase in HLA class I expression, percentage lysis of MSC was doubled when the cells were pre-activated with IFN-γ. Furthermore, co-culture of PBMC with IFN-γ-stimulated BM-MSC further increased percentage lysis. A similar trend was observed when using ASC but percentage lysis was significantly lower compared to BM-MSC (16, 27).
Immunogenicity and Immunomodulatory Potential of Allogeneic Differentiated MSC
The cell surface expression of MHC proteins and co-stimulatory molecules can have a major impact on allo-recognition of transplanted cells or organs in an immunocompetent host (5). As mentioned previously, MSC are generally thought to express low or no MHC and co-stimulatory proteins contributing to their perceived “weak” immunogenic profile (5). However, as with exposure to a pro-inflammatory environment, MSC differentiation has been previously shown to result in upregulation of cell surface immunogenic molecules (5, 28, 29). In recent years, several studies have examined upregulation of potentially immunogenic cell surface proteins in an allogeneic and xenogeneic cell therapy context. With regard to the effect of MSC differentiation on the cell surface profile of the cells, there are conflicting results presented in different studies. For example, Li et al. describe an upregulation of HLA-DR on MSC after hepatocyte differentiation (30), while several other studies report no change in cell surface proteins after chondrogenic (31), myogenic (32), and insulin-producing cell (33) differentiation.
More important than cell surface expression levels is functional immunogenicity, which is the response of immune cells to the differentiated cells. Differentiated MSC have been shown to elicit allogeneic lymphocyte proliferation after osteogenic (34), chondrogenic (29, 35), and hepatocytic (30) differentiation, and lymphocyte IFN-γ production in vitro after hepatocytic differentiation (30). Contrary to these studies, it has also been recently shown that differentiated MSC do not induce allogeneic T cell proliferation in vitro after osteogenic (36) or chondrogenic (37) differentiation.
While not directly related to immunogenicity, the secretion of immunoregulatory or immunosuppressive molecules has also been reported as being affected by MSC differentiation. Prostaglandin E2 (PGE2), an important mediator of MSC immunosuppressive ability has been shown to be reduced after myogenic differentiation (38). On the other hand, it has also been shown that MSC, during chondrogenic differentiation can maintain the expression and production of immunomodulatory molecules, such as nitric oxide, IL-6, and IDO (39).
While in vitro data have aided in our understanding of the potential mechanisms occurring during differentiation and the immune response to the transplanted cells, the in vivo immune response to the cells is the most indicative of translational potential. Studies that transplanted allogeneic differentiated MSC into appropriate pre-clinical models have shown varying levels of immune response. For example, hepatocyte differentiated MSC induced significantly more CD3+ and CD45+ cells after transplantation compared to undifferentiated MSC, despite the fact that the differentiated MSC exerted a beneficial effect on the glycemic control of the treated animals (33). Osteogenically differentiated MSC transplantation resulted in significantly more activated immune cells after implantation in a mouse model (34). In a diabetic model, evidence for in situ differentiation was observed, increased numbers of cytotoxic cells in recipients and higher levels of allo-antibody were attributed to this differentiation (40).
Much of the evidence discussed here and elsewhere (5, 28, 29) points to an increase in MSC immunogenicity following induced differentiation. Strategies to reduce this immunogenicity, therefore, would be extremely important for the future development of such therapies. Interestingly, Li et al. showed that when they carried out their hepatocyte differentiation in a 3D scaffold, as opposed to 2D tissue culture plastic, they observed no increase in lymphocyte proliferation, no decrease in production of PGE2 and less IFN-γ production by lymphocytes (30). The maintenance of expression of immunomodulatory molecules which Yang et al. observed was also only seen in 3D culture conditions as opposed to 2D (39). Gene therapy approaches also hold promise as an approach to reduce the detrimental immune effects of differentiation. Dhingra et al. (38), following up on excellent work from Huang et al. (41), showed that restoration of secretion of PGE2 could overcome the rejection and loss of efficacy of cardiomyocyte differentiated MSC (38). It was also shown that interfering with the expression of MHC II on the cell surface of differentiated MSC could increase their survival time in vivo, reduce cytotoxic and allo-antibody responses, and increase therapeutic efficacy (14, 42). Our increased understanding of the immunological changes that occur following differentiation of allo-MSC are already informing alternative strategies to circumvent their increased immunogenicity.
Immunogenicity of Human MSC in Human Subjects
As mentioned earlier, the generation of humoral and/or cellular immune responses against the allogeneic donor cells could potentially lead to adverse immunological effects, impact the efficacy of subsequent allogeneic cell therapy, or compromise the success of future organ transplantation (5). Although it has been shown that allogeneic MSC induce at least a humoral immune response in pre-clinical models, it is not clear whether this occurs on the human setting. To date, approximately 3,000 patients have received allogeneic mesenchymal stem cell treatment for various diseases and no acute adverse events linked to the allogenicity of MSC have been reported (8, 9).
As yet only a limited number of clinical trials have evaluated the generation of potentially harmful anti-HLA antibodies in patients receiving allo-MSCs, and it remains unclear whether this may lead to adverse effects. Four clinical trials have been published that have documented the presence/absence of anti-HLA antibodies in patients after allo-MSC therapy for various disease indications, including osteoarthritis, Crohn’s disease, and Type II diabetes. Results published by Garcia-Sancho et al. on the influence of HLA-matching on the efficacy of allogeneic MSC therapy in osteoarthritis and degenerative disk disease (43), indicated that only a very limited number of patients receiving MSC had developed anti-donor antibodies and, surprisingly, better donor–recipient HLA-matching did not enhance efficacy. In another clinical trial for complex perianal fistulas in Crohn’s disease, Panés et al. reported that allogeneic adipose-derived mesenchymal stem cells (Cx601) treated patients developed donor-specific antibodies (44). At week 12, 34% of Cx601 treated patients generated anti-HLA Class I antibodies compared to none of the placebo-treated group. The authors suggested, however, that the development of donor-specific antibodies had no clinical relevance in terms of affecting the efficacy of Cx601 or in provoking TEAEs. Two other clinical trials have been published by Skyler et al. (45) and Packham et al. (46) using rexlemestrocel-L cells, defined as allogeneic mesenchymal precursor cells (MPC), for the treatment of Type II Diabetes and Diabetic nephropathy. Cross matching between donor cells and recipient was not performed prior to infusion. Both studies indicate that no subjects receiving rexlemestrocel-L treatment developed antibodies specific to the donor HLA or showed clinically relevant increases in either class I or II PRA. The authors suggest that these findings are consistent with the immunotolerant profile of MPCs, which are negative for HLA Class II and CD80 and CD86 co-immunostimulatory molecules, and exert potential immunomodulatory effects, including inhibition of T cell proliferation (47). The observed lack of acute immunological responses to unmatched allogeneic MPC is particularly important in patients who may eventually require organ transplantation. The authors suggest that the lack of any evidence of sustained sensitization and development of antibodies specific to the donor HLA suggests that repeat administration of this therapy may be a feasible option in this patient population.
Taken collectively, these data might indicate that the formation of allo-antibodies may not be as critical as originally thought, though this would likely be dependent on the phenotype of the cells administered as well as disease specificity. However, the implications of the development of donor-specific allo-antibodies will need to be assessed over longer time periods, alongside the tolerability and efficacy of single and repeated administration of allogeneic MSC before definite conclusions can be drawn in this regard.
Immunogenicity of Extracellular Vesicles Secreted from Allogeneic MSC
In recent years exciting results from the use of MSC-derived extracellular vesicles (EVs) or exosomes has come to light (48–50). This research has been primarily focused on transfer of molecular cargoes, such as mRNA and miRNA (48, 51), to enhance regenerative or pro-survival responses. Furthermore, a recent report form Bai and colleagues (52) could demonstrate the efficacy of human MSC-derived exosomes delivered periocularly in a rat model of experimental autoimmune uveitis (52). However, evidence from dendritic cell research indicates that potentially immunogenic proteins such as MHC molecules can also be transferred via EVs (53). This raises the possibility that allogeneic MSC-derived EVs may also possess immunogenic molecules which could lead to potentially detrimental immune responses such as those elicited by allo-MSCs themselves. Although there seems to be evidence that EVs may contain MHC molecules it is not clear at this stage if (i) MSC-derived exosomes contain MHC molecules, (ii) they can be transferred to other cells, and, most notably, (iii) if they may induce allo-immune responses. Another future potential area of interest may be to examine the expression of tissue factor (TF). While TF has been shown to be present in EVs (54), expression has not yet been reported in MSC-derived EVs. Future studies designed to investigate the influence of TF in EVs on anti-donor immune responses or procoagulant activity may yield important insights.
Perspective
While in recent years strong evidence has emerged showing that allogeneic MSC can and do invoke measurable immune responses, there is still ambiguity and uncertainty in the field. The next generation of allo-MSC therapies should be developed through rigorous characterization and fine tuning of MSC immunogenicity, survival, and persistence after transplantation, efficacy, and disease-specific mechanisms of action (Figure 1).
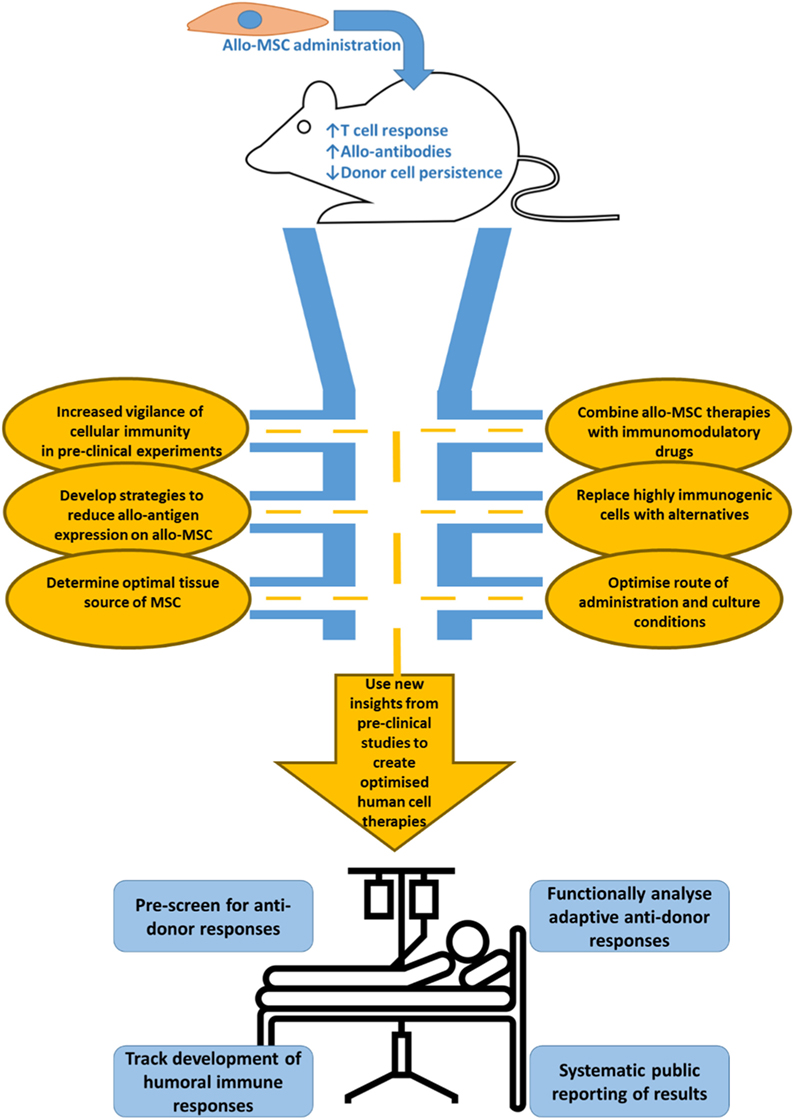
Figure 1. Immunological considerations for the development of next generation allo-MSC therapy. Allogeneic administration of MSC in a variety of pre-clinical models has been shown to induce allo-immune responses. These potentially harmful responses include generation of memory T cells, production of functional allo-antibodies and allo-specific clearance of administered cells. More evidence on the specific effects of allo-MSC administration on a model specific basis is required however and the field would benefit from systematic reporting of relevant allo-immune reactions elicited by MSC in pre-clinical models. The development of the next generation of MSC therapies will require focused and multi-disciplinary pre-clinical experiments to (1) fully characterize and report cell mediated immunity, (2) if necessary, to lower these immune responses in the recipients by working on synergy of allo-MSC with immunomodulatory drugs, (3) by developing strategies to reduce allo-antigen expression on administered cells by using genetic manipulation or small molecules, and (4) if appropriate, replacing highly immunogenic MSC cell therapies with alternatives. In addition to developing bespoke indication specific strategies like those mentioned above, the field as a whole should look to optimize the source, culture conditions and routes of administration of MSC in order to minimize immunogenic reactions to the cells while maintaining efficacy. In the coming years, more detailed pre-clinical data will allow lessons to be learned to create optimized allo-MSC therapies for clinical use. However, we should continue to carefully monitor the allo-immune response once allo-MSC are administered to the patient. This surveillance should include pre-screening patients for pre-existing anti-donor immune responses, monitoring anti-donor cellular and humoral immune responses and or equal importance systematic reporting of results in easily available formats.
Consideration of the points below is likely to lead to considerable advances in our understanding of the anti-donor immune responses induced by allogeneic MSC therapy and potential new strategies to minimize any potential adverse responses while maximizing patient benefit,
1. Establishment and monitoring of safety/efficacy profiles for each specific treatment regimen in different treatment indications and disease stages to evaluate, for example, the risk of allo-immunization relative to the beneficial effects of the treatment.
2. Comprehensive analysis of the full range of characteristics affecting immunogenicity such as cell surface expression of important molecules on the MSC, in vitro responses of dendritic, T, and B cells and in vivo humoral, cytotoxic, and memory responses.
3. The use of immunosuppressive drugs or other molecules in combination with allogeneic MSC to determine if the immune response to the cells can be reduced.
4. Use of gene therapy or small molecule approaches to interfere with the presentation of allo-antigen on the cell surface.
5. Replacement of highly immunogenic cells such as some differentiated MSC (29). Alternative cell types could be investigated to replace damaged tissue (55, 56), or in-depth studies into the mechanism of action of, e.g., licensed MSC may yield candidate molecules which could replace cell therapy altogether.
6. Investigation of alternative routes of administration (e.g., “inert” locations such as the articular cavity or intervertebral disk (43) which may lower the “visibility” of the cells to the host immune system.
7. A detailed understanding of how allo-MSC cell death affects their immunogenic or tolerogenic properties and how (or if) allo-MSC cell death regulates ensuing anti-donor immune responses.
It is clear that MSC therapy, and particularly allogeneic MSC therapy, holds great promise for the treatment of a multitude of diseases. These cells have been shown to be clinically safe despite abundant pre-clinical evidence that they invoke an anti-donor immune response. The systematic collection and dissemination of immunological data should still be undertaken to provide further insight into mechanism of action and so that researchers, clinicians, and regulatory bodies worldwide can remain vigilant to any future adverse effects.
Author Contributions
PL, OT, and AR wrote the manuscript and finalized the draft. MG and TR wrote specific parts of the manuscript and revised the entire draft.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The reviewer DN and handling editor declared their shared affiliation.
Funding
OT and TR are supported by a Science Foundation Ireland grants (12/IA/1624), PL, MG, and TR are supported by EU FP7 [Collaborative Health Project VISICORT (grant number 602470)], Science Foundation Ireland [REMEDI Strategic Research Cluster (grant number 09/SRC-B1794); MG is supported by CÚRAM Research Centre (grant number 13/RC/2073)]; the European Commission [Horizon 2020 Collaborative Health Project NEPHSTROM (grant number 634086), and the European Regional Development Fund. AR is supported by Irish Cancer Society Fellowship (CRF12RYA) and Science Foundation Ireland Starting Investigator Grant (15/SIRG/3456).
References
1. Caplan AI. Mesenchymal stem cells: time to change the name! Stem Cells Transl Med (2017) 6(6):1445–51. doi:10.1002/sctm.17-0051
2. Di Nicola M, Carlo-Stella C, Magni M, Milanesi M, Longoni PD, Matteucci P, et al. Human bone marrow stromal cells suppress T-lymphocyte proliferation induced by cellular or nonspecific mitogenic stimuli. Blood (2002) 99(10):3838–43. doi:10.1182/blood.V99.10.3838
3. Barry FP, Murphy JM, English K, Mahon BP. Immunogenicity of adult mesenchymal stem cells: lessons from the fetal allograft. Stem Cells Dev (2005) 14(3):252–65. doi:10.1089/scd.2005.14.252
4. Eliopoulos N, Stagg J, Lejeune L, Pommey S, Galipeau J. Allogeneic marrow stromal cells are immune rejected by MHC class I- and class II-mismatched recipient mice. Blood (2005) 106(13):4057–65. doi:10.1182/blood-2005-03-1004
5. Griffin MD, Ryan AE, Alagesan S, Lohan P, Treacy O, Ritter T. Anti-donor immune responses elicited by allogeneic mesenchymal stem cells: what have we learned so far? Immunol Cell Biol (2013) 91(1):40–51. doi:10.1038/icb.2012.67
6. Nauta AJ, Westerhuis G, Kruisselbrink AB, Lurvink EG, Willemze R, Fibbe WE. Donor-derived mesenchymal stem cells are immunogenic in an allogeneic host and stimulate donor graft rejection in a nonmyeloablative setting. Blood (2006) 108(6):2114–20. doi:10.1182/blood-2005-11-011650
7. Schu S, Nosov M, O’Flynn L, Shaw G, Treacy O, Barry F, et al. Immunogenicity of allogeneic mesenchymal stem cells. J Cell Mol Med (2012) 16(9):2094–103. doi:10.1111/j.1582-4934.2011.01509.x
8. Le Blanc K, Frassoni F, Ball L, Locatelli F, Roelofs H, Lewis I, et al. Mesenchymal stem cells for treatment of steroid-resistant, severe, acute graft-versus-host disease: a phase II study. Lancet (2008) 371(9624):1579–86. doi:10.1016/S0140-6736(08)60690-X
9. Gregoire C, Lechanteur C, Briquet A, Baudoux E, Baron F, Louis E, et al. Review article: mesenchymal stromal cell therapy for inflammatory bowel diseases. Aliment Pharmacol Ther (2017) 45(2):205–21. doi:10.1111/apt.13864
10. Kaipe H, Carlson LM, Erkers T, Nava S, Mollden P, Gustafsson B, et al. Immunogenicity of decidual stromal cells in an epidermolysis bullosa patient and in allogeneic hematopoietic stem cell transplantation patients. Stem Cells Dev (2015) 24(12):1471–82. doi:10.1089/scd.2014.0568
11. Baygan A, Aronsson-Kurttila W, Moretti G, Tibert B, Dahllof G, Klingspor L, et al. Safety and side effects of using placenta-derived decidual stromal cells for graft-versus-host disease and hemorrhagic cystitis. Front Immunol (2017) 8:795. doi:10.3389/fimmu.2017.00795
12. Melmed GY, Pandak WM, Casey K, Abraham B, Valentine J, Schwartz D, et al. Human placenta-derived cells (PDA-001) for the treatment of moderate-to-severe Crohn’s disease: a phase 1b/2a study. Inflamm Bowel Dis (2015) 21(8):1809–16. doi:10.1097/MIB.0000000000000441
13. Moll G, Ignatowicz L, Catar R, Luecht C, Sadeghi B, Hamad O, et al. Different procoagulant activity of therapeutic mesenchymal stromal cells derived from bone marrow and placental decidua. Stem Cells Dev (2015) 24(19):2269–79. doi:10.1089/scd.2015.0120
14. Ankrum JA, Ong JF, Karp JM. Mesenchymal stem cells: immune evasive, not immune privileged. Nat Biotechnol (2014) 32(3):252–60. doi:10.1038/nbt.2816
15. Schnabel LV, Pezzanite LM, Antczak DF, Felippe MJ, Fortier LA. Equine bone marrow-derived mesenchymal stromal cells are heterogeneous in MHC class II expression and capable of inciting an immune response in vitro. Stem Cell Res Ther (2014) 5(1):13. doi:10.1186/scrt402
16. Gotherstrom C, Ringden O, Tammik C, Zetterberg E, Westgren M, Le Blanc K. Immunologic properties of human fetal mesenchymal stem cells. Am J Obstet Gynecol (2004) 190(1):239–45. doi:10.1016/j.ajog.2003.07.022
17. Joswig AJ, Mitchell A, Cummings KJ, Levine GJ, Gregory CA, Smith R III, et al. Repeated intra-articular injection of allogeneic mesenchymal stem cells causes an adverse response compared to autologous cells in the equine model. Stem Cell Res Ther (2017) 8(1):42. doi:10.1186/s13287-017-0503-8
18. Berglund AK, Schnabel LV. Allogeneic major histocompatibility complex-mismatched equine bone marrow-derived mesenchymal stem cells are targeted for death by cytotoxic anti-major histocompatibility complex antibodies. Equine Vet J (2017) 49(4):539–44. doi:10.1111/evj.12647
19. Ardanaz N, Vazquez FJ, Romero A, Remacha AR, Barrachina L, Sanz A, et al. Inflammatory response to the administration of mesenchymal stem cells in an equine experimental model: effect of autologous, and single and repeat doses of pooled allogeneic cells in healthy joints. BMC Vet Res (2016) 12:65. doi:10.1186/s12917-016-0692-x
20. Isakova IA, Lanclos C, Bruhn J, Kuroda MJ, Baker KC, Krishnappa V, et al. Allo-reactivity of mesenchymal stem cells in rhesus macaques is dose and haplotype dependent and limits durable cell engraftment in vivo. PLoS One (2014) 9(1):e87238. doi:10.1371/journal.pone.0087238
21. Green DR, Ferguson T, Zitvogel L, Kroemer G. Immunogenic and tolerogenic cell death. Nat Rev Immunol (2009) 9(5):353–63. doi:10.1038/nri2545
22. Moll G, Geissler S, Catar R, Ignatowicz L, Hoogduijn MJ, Strunk D, et al. Cryopreserved or fresh mesenchymal stromal cells: only a matter of taste or key to unleash the full clinical potential of MSC therapy? Adv Exp Med Biol (2016) 951:77–98. doi:10.1007/978-3-319-45457-3_7
23. Chinnadurai R, Copland IB, Garcia MA, Petersen CT, Lewis CN, Waller EK, et al. Cryopreserved mesenchymal stromal cells are susceptible to T-cell mediated apoptosis which is partly rescued by IFNgamma licensing. Stem Cells (2016) 34(9):2429–42. doi:10.1002/stem.2415
24. Mancheno-Corvo P, Menta R, del Rio B, Franquesa M, Ramirez C, Hoogduijn MJ, et al. T lymphocyte prestimulation impairs in a time-dependent manner the capacity of adipose mesenchymal stem cells to inhibit proliferation: role of interferon gamma, poly I:C, and tryptophan metabolism in restoring adipose mesenchymal stem cell inhibitory effect. Stem Cells Dev (2015) 24(18):2158–70. doi:10.1089/scd.2014.0508
25. Chinnadurai R, Copland IB, Patel SR, Galipeau J. IDO-independent suppression of T cell effector function by IFN-gamma-licensed human mesenchymal stromal cells. J Immunol (2014) 192(4):1491–501. doi:10.4049/jimmunol.1301828
26. Sivanathan KN, Rojas-Canales DM, Hope CM, Krishnan R, Carroll RP, Gronthos S, et al. Interleukin-17A-induced human mesenchymal stem cells are superior modulators of immunological function. Stem Cells (2015) 33(9):2850–63. doi:10.1002/stem.2075
27. Roemeling-van Rhijn M, Reinders ME, Franquesa M, Engela AU, Korevaar SS, Roelofs H, et al. Human allogeneic bone marrow and adipose tissue derived mesenchymal stromal cells induce CD8+ cytotoxic T cell reactivity. J Stem Cell Res Ther (2013) 3(Suppl 6):004. doi:10.4172/2157-7633.S6-004
28. Lohan P, Coleman CM, Murphy JM, Griffin MD, Ritter T, Ryan AE. Changes in immunological profile of allogeneic mesenchymal stem cells after differentiation: should we be concerned? Stem Cell Res Ther (2014) 5(4):99. doi:10.1186/scrt488
29. Ryan AE, Lohan P, O’Flynn L, Treacy O, Chen X, Coleman C, et al. Chondrogenic differentiation increases antidonor immune response to allogeneic mesenchymal stem cell transplantation. Mol Ther (2014) 22(3):655–67. doi:10.1038/mt.2013.261
30. Li Y, Wu Q, Wang Y, Li L, Chen F, Shi Y, et al. Immunogenicity of hepatic differentiated human umbilical cord mesenchymal stem cells promoted by porcine decellularized liver scaffolds. Xenotransplantation (2017) 24(1). doi:10.1111/xen.12287
31. Lee HJ, Kang KS, Kang SY, Kim HS, Park SJ, Lee SY, et al. Immunologic properties of differentiated and undifferentiated mesenchymal stem cells derived from umbilical cord blood. J Vet Sci (2016) 17(3):289–97. doi:10.4142/jvs.2016.17.3.289
32. Joo S, Lim HJ, Jackson JD, Atala A, Yoo JJ. Myogenic-induced mesenchymal stem cells are capable of modulating the immune response by regulatory T cells. J Tissue Eng (2014) 5:2041731414524758. doi:10.1177/2041731414524758
33. Yang XF, Chen T, Ren LW, Yang L, Qi H, Li FR. Immunogenicity of insulin-producing cells derived from human umbilical cord mesenchymal stem cells. Exp Ther Med (2017) 13(4):1456–64. doi:10.3892/etm.2017.4096
34. Fu X, Yang H, Zhang H, Wang G, Liu K, Gu Q, et al. Improved osteogenesis and upregulated immunogenicity in human placenta-derived mesenchymal stem cells primed with osteogenic induction medium. Stem Cell Res Ther (2016) 7(1):138. doi:10.1186/s13287-016-0400-6
35. Mukonoweshuro B, Brown CJ, Fisher J, Ingham E. Immunogenicity of undifferentiated and differentiated allogeneic mouse mesenchymal stem cells. J Tissue Eng (2014) 5:2041731414534255. doi:10.1177/2041731414534255
36. Niu J, Ding G, Zhang L. Effects of simvastatin on the osteogenic differentiation and immunomodulation of bone marrow mesenchymal stem cells. Mol Med Rep (2015) 12(6):8237–40. doi:10.3892/mmr.2015.4476
37. Kiernan CH, Hoogduijn MJ, Franquesa M, Wolvius EB, Brama PA, Farrell E. Allogeneic chondrogenically differentiated human mesenchymal stromal cells do not induce immunogenic responses from T lymphocytes in vitro. Cytotherapy (2016) 18(8):957–69. doi:10.1016/j.jcyt.2016.05.002
38. Dhingra S, Li P, Huang XP, Guo J, Wu J, Mihic A, et al. Preserving prostaglandin E2 level prevents rejection of implanted allogeneic mesenchymal stem cells and restores postinfarction ventricular function. Circulation (2013) 128(11 Suppl 1):S69–78. doi:10.1161/CIRCULATIONAHA.112.000324
39. Yang J, Chen X, Yuan T, Yang X, Fan Y, Zhang X. Regulation of the secretion of immunoregulatory factors of mesenchymal stem cells (MSCs) by collagen-based scaffolds during chondrogenesis. Mater Sci Eng C Mater Biol Appl (2017) 70(Pt 2):983–91. doi:10.1016/j.msec.2016.04.096
40. Gu LH, Zhang TT, Li Y, Yan HJ, Qi H, Li FR. Immunogenicity of allogeneic mesenchymal stem cells transplanted via different routes in diabetic rats. Cell Mol Immunol (2015) 12(4):444–55. doi:10.1038/cmi.2014.70
41. Huang XP, Sun Z, Miyagi Y, McDonald Kinkaid H, Zhang L, Weisel RD, et al. Differentiation of allogeneic mesenchymal stem cells induces immunogenicity and limits their long-term benefits for myocardial repair. Circulation (2010) 122(23):2419–29. doi:10.1161/CIRCULATIONAHA.110.955971
42. Huang XP, Ludke A, Dhingra S, Guo J, Sun Z, Zhang L, et al. Class II transactivator knockdown limits major histocompatibility complex II expression, diminishes immune rejection, and improves survival of allogeneic bone marrow stem cells in the infarcted heart. FASEB J (2016) 30(9):3069–82. doi:10.1096/fj.201600331R
43. Garcia-Sancho J, Sanchez A, Vega A, Noriega DC, Nocito M. Influence of HLA matching on the efficacy of allogeneic mesenchymal stromal cell therapies for osteoarthritis and degenerative disc disease. Transplant Direct (2017) 3(9):e205. doi:10.1097/TXD.0000000000000724
44. Panes J, Garcia-Olmo D, Van Assche G, Colombel JF, Reinisch W, Baumgart DC, et al. Expanded allogeneic adipose-derived mesenchymal stem cells (Cx601) for complex perianal fistulas in Crohn’s disease: a phase 3 randomised, double-blind controlled trial. Lancet (2016) 388(10051):1281–90. doi:10.1016/S0140-6736(16)31203-X
45. Skyler JS, Fonseca VA, Segal KR, Rosenstock J, Investigators M-D. Allogeneic mesenchymal precursor cells in type 2 diabetes: a randomized, placebo-controlled, dose-escalation safety and tolerability pilot study. Diabetes Care (2015) 38(9):1742–9. doi:10.2337/dc14-2830
46. Packham DK, Fraser IR, Kerr PG, Segal KR. Allogeneic mesenchymal precursor cells (MPC) in diabetic nephropathy: a randomized, placebo-controlled, dose escalation study. EBioMedicine (2016) 12:263–9. doi:10.1016/j.ebiom.2016.09.011
47. Togel F, Westenfelder C. The role of multipotent marrow stromal cells (MSCs) in tissue regeneration. Organogenesis (2011) 7(2):96–100. doi:10.4161/org.7.2.15781
48. Figueroa J, Phillips LM, Shahar T, Hossain A, Gumin J, Kim H, et al. Exosomes from glioma-associated mesenchymal stem cells increase the tumorigenicity of glioma stem-like cells via transfer of miR-1587. Cancer Res (2017) 77(21):5808–19. doi:10.1158/0008-5472.CAN-16-2524
49. Cheng X, Zhang G, Zhang L, Hu Y, Zhang K, Sun X, et al. Mesenchymal stem cells deliver exogenous miR-21 via exosomes to inhibit nucleus pulposus cell apoptosis and reduce intervertebral disc degeneration. J Cell Mol Med (2017). doi:10.1111/jcmm.13316
50. Shimoda A, Tahara Y, Sawada SI, Sasaki Y, Akiyoshi K. Glycan profiling analysis using evanescent-field fluorescence-assisted lectin array: importance of sugar recognition for cellular uptake of exosomes from mesenchymal stem cells. Biochem Biophys Res Commun (2017) 491(3):701–7. doi:10.1016/j.bbrc.2017.07.126
51. Xin H, Li Y, Chopp M. Exosomes/miRNAs as mediating cell-based therapy of stroke. Front Cell Neurosci (2014) 8:377. doi:10.3389/fncel.2014.00377
52. Bai L, Shao H, Wang H, Zhang Z, Su C, Dong L, et al. Effects of mesenchymal stem cell-derived exosomes on experimental autoimmune uveitis. Sci Rep (2017) 7(1):4323. doi:10.1038/s41598-017-04559-y
53. Liu Q, Rojas-Canales DM, Divito SJ, Shufesky WJ, Stolz DB, Erdos G, et al. Donor dendritic cell-derived exosomes promote allograft-targeting immune response. J Clin Invest (2016) 126(8):2805–20. doi:10.1172/JCI84577
54. Park JA, Sharif AS, Tschumperlin DJ, Lau L, Limbrey R, Howarth P, et al. Tissue factor-bearing exosome secretion from human mechanically stimulated bronchial epithelial cells in vitro and in vivo. J Allergy Clin Immunol (2012) 130(6):1375–83. doi:10.1016/j.jaci.2012.05.031
55. Lohan P, Treacy O, Lynch K, Barry F, Murphy M, Griffin MD, et al. Culture expanded primary chondrocytes have potent immunomodulatory properties and do not induce an allogeneic immune response. Osteoarthritis Cartilage (2016) 24(3):521–33. doi:10.1016/j.joca.2015.10.005
Keywords: allogeneic, allo-mesenchymal stromal cell, anti-donor immune response, immunogeniciy, inflammation, immunomodulation
Citation: Lohan P, Treacy O, Griffin MD, Ritter T and Ryan AE (2017) Anti-Donor Immune Responses Elicited by Allogeneic Mesenchymal Stem Cells and Their Extracellular Vesicles: Are We Still Learning? Front. Immunol. 8:1626. doi: 10.3389/fimmu.2017.01626
Received: 25 September 2017; Accepted: 09 November 2017;
Published: 24 November 2017
Edited by:
Reem Al-Daccak, Institut National de la Santé et de la Recherche Médicale, FranceReviewed by:
Guido Moll, Charité Universitätsmedizin Berlin, GermanyDanièle Noël, Institut National de la Santé et de la Recherche Médicale, France
Nuala Mooney, Centre National pour la Recherche Scientifique, France
Copyright: © 2017 Lohan, Treacy, Griffin, Ritter and Ryan. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Aideen E. Ryan, YWlkZWVuLnJ5YW5AbnVpZ2Fsd2F5Lmll
†These authors have contributed equally to this work.
 Paul Lohan
Paul Lohan Oliver Treacy
Oliver Treacy Matthew D. Griffin
Matthew D. Griffin Thomas Ritter
Thomas Ritter Aideen E. Ryan
Aideen E. Ryan