- Department of Hepatic Surgery and Liver Transplantation Center, School of Medicine, Renji Hospital, Shanghai Jiaotong University, Shanghai, China
Currently, liver transplantation is the most effective treatment for end-stage liver disease. Immunosuppressive agents are required to be taken after the operations, which have significantly reduced rejection rates and improved the short-term (<1 year) survival rates. However, post-transplant complications related to the immunosuppressive therapy have led to the development of new protocols aimed at protecting renal function and preventing de novo cancer and dysmetabolic syndrome. Donor specific immune tolerance, which means the mature immune systems of recipients will not attack the grafts under the conditions without any immunosuppression therapies, is considered the optimal state after liver transplantation. There have been studies that have shown that some patients can reach this immune tolerance state after liver transplantation. The intrahepatic immune system is quite different from that in other solid organs, especially the innate immune system. It contains a variety of liver specific cells, such as liver-derived dendritic cells, Kupffer cells, liver sinusoidal endothelial cells, liver-derived natural killer (NK) cells, natural killer T (NKT) cells, and so on. Depending on their specific structures and functions, these intrahepatic innate immune cells play important roles in the development of intrahepatic immune tolerance. In this article, in order to have a deeper understanding of the tolerogenic functions of liver, we summarized the molecular mechanisms of immune tolerance induced by intrahepatic innate immune cells after liver transplantation.
Introduction
The past three decades have seen liver transplantation becoming a major treatment approach for end-stage liver disease. This is because of the dramatic improvement in survival after liver transplantation. Actually, improved use of post-transplant immunosuppression is a major factor. In order to prevent acute and chronic rejection, long-term immunosuppression is required to avoid severe acute and chronic rejection and graft loss. Up to now, the backbone of immunosuppression therapy remains calcineurin inhibitors (CNIs) mostly associated with steroids in the short-term and mycophenolate mofetil or mammalian target of rapamycin (mTOR) inhibitors. However, there are some post-transplant complications related to immunosuppressive therapy, such as renal injury, de novo cancer, dysmetabolic syndrome, and so on. In the future, immunosuppression will be more oriented, aiming to protect the graft from rejection and at reducing the risk of disease recurrence and complications related to immunosuppressive therapy, including promoting stable long-term immunological tolerance of the liver graft. The liver has been shown to be more tolerogenic than other solid organs, and most hepatic allografts can be accepted with low-dose immunosuppressive therapy. It can be exemplified by oral tolerance (which means the mucosal immune system maintains unresponsiveness to antigens that might induce unexpected immune responses) and portal venous tolerance (which means the induction of peripheral tolerance following portal venous delivery of most alloantigens). Other phenomena attributed to hepatic immune tolerance consist of persistent microbial infections and gastrointestinal tumor metastases in the liver (1). Besides this, the liver can induce tolerance for other transplanted organs and decrease the risk of rejection of associated organs, such as heart, kidney, skin, pancreas, and so on (2). There were plenty of signs that showed that the internal microenvironment of the liver could play a role in the development of immune tolerance after transplantation.
Liver is an organ with double blood supply, from the portal vein and the hepatic artery. Arterial and venous blood mix in the liver, resulting in low oxygen tension, low perfusion pressure, and slow and irregular blood flow within the hepatic sinusoids, which assist the intrahepatic cells and molecules to fully contact each other. Generally, the adaptive system in the liver includes humoral immunity and cell-mediated immunity. They are carried by two different lymphocytes (B cells and T cells), which recognize and respond to pathogens in antigen-specific ways. By contrast, the innate immune cells contained in the liver are quite different from those in the peripheral blood, including liver-derived dendritic cells (DCs), Kupffer cells, liver sinusoidal endothelial cells (LSECs), liver-derived natural killer (NK) cells, natural killer T (NKT) cells, and so on. These innate immune cells participate in constituting the immune microenvironment in the liver. Their tolerogenic functions are met by two mechanisms. First, intrahepatic innate immune cells express low or undetectable levels of major histocompatibility complex (MHC) antigens, costimulatory molecules, and other effector molecules, which means it will be difficult for these intrahepatic cells to induce innate or adaptive immune response. Second, innate immune cells in the liver can also exert their immunosuppressive effects by interfering with the functions of other intrahepatic cells, by secreting immunosuppressive cytokines [such as interleukin-10 (IL-10), transforming growth factor- β (TGF-β), indolamine 2,3-dioxygenase (IDO), and so on] or by directly contacting them. In this review, we clarified some potential mechanisms to illustrate human liver allograft tolerance induced by intrahepatic innate immune cells.
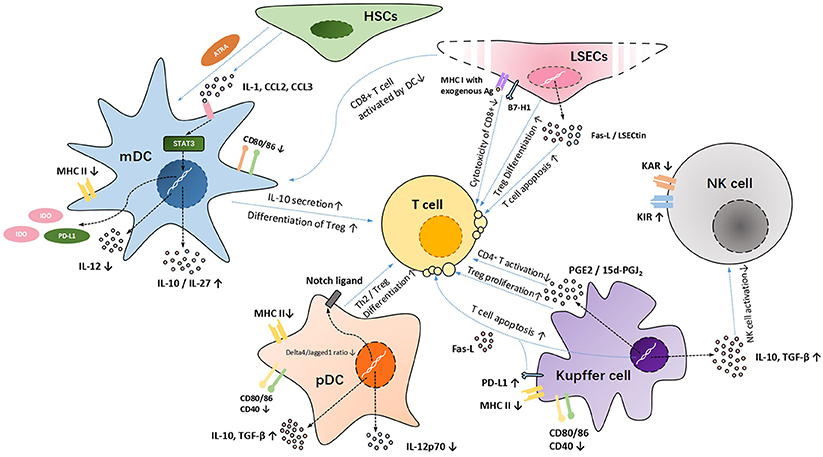
Dendritic Cells (DCs) in Tolerance
Dendritic cells are derived primarily from the bone marrow, as well as from the liver and the spleen. As a heterogeneous population of antigen presenting cells (APCs), DCs play pivotal roles in the initiation of immunity and the induction of immunological tolerance depending on their maturation state and subsets. Recently, regulatory DCs, which mean DCs with immune regulatory functions, have attracted much attention. Up to now, several kinds of regulatory DC had been reported, such as CD11clowI-AlowCD11bhi splenic regulatory DCs (3) and CD11clowCD45RB+DCs (4, 5). They have common characteristics, including immature DC phenotype, high IL-10 and low IL-12p70 secretion, and inhibition of T-cell proliferation. It has been proved that the liver microenvironment could program differentiation of bone marrow derived progenitors into regulatory DCs. In a heterotopic liver transplantation model in rats, He et al. (6) have found that the graft survival rate can be greatly improved by infusing immature DCs (imDCs) into the recipient rats. It has been suggested that the overexpression of the zinc finger protein A20 could effectively inhibit the maturation of DCs that are resident in the liver allograft and consequently suppress acute liver allograft rejection. Dai et al. (7) have demonstrated that A20 treatment could significantly inhibit transplantation induced nuclear factor-κB (NF-κB) mediated activation of DCs resident in the liver allograft, which was consistent with the changes of costimulatory molecule and IL-12 mRNA expression of DCs. Since NF-κB has been shown as a pivotal nuclear transcriptional regulator of DC maturation and immunostimulatory ability (8), it presumably plays an essential role in this process.
In normal conditions, DCs derived from human or mouse livers can secrete certain amounts of IL-10, inducing lower T cell response and promoting regulator T cell (Treg) generation, with a definite effect on immune tolerance (9–11). Besides this, Kushwah et al. (12) have found that viable immature DCs have the ability to uptake apoptotic DC, which suppresses the subsequent maturation of viable DC and mediates the differentiation of naïve T cells into Foxp3+ Tregs (12). However, under inflammatory conditions, quiescent DCs transform from a tolerant state into a responsive state. Dendritic cells located in sinusoids and portal area can fully contact T cells and induce T cell-mediated immune responses (13).
Myeloid Dendritic Cells (mDCs)
In mice, liver myeloid dendritic cells (mDCs) have lower maturity than those in secondary lymphoid tissues, both in terms of phenotype and function (14–16). It is mainly reflected by the lower expression level of MHC-II and costimulatory molecules on the cell surface (16) and lower secretion of IL-12 after toll-like receptor (TLR) activation while there is a higher production of IL-10 and IL-27, which have been found to be closely related to the low response of T cells induced by liver DCs (17). Overall, in in vivo experiments on mice, mDCs derived from the liver show reduced ability to activate allogeneic naïve T cells (14–16).
In addition to the lower maturation state, the liver-derived mDCs can also induce immune tolerance by affecting T cell function. In mice, Khanna et al. (18) injected liver-derived mDCs into allogeneic recipients and found that they can induce T cells to secrete IL-10 (18). Besides this, in vitro experiments have shown that the interactions between NK cells (through their inhibitory receptor NKG2A) and hepatocytes can change the concentrations of some important cytokines in the local microenvironment [for example, an increase in TGF-β and a decrease in tumor necrosise factor- α (TNF-α)], thus, inducing the differentiation of a group of special DCs that have an immune tolerance function. This kind of DCs can induce the differentiation of a special type of Treg, which can inhibit T cell response via the programmed death-1 (PD-1) pathway (19). Based on these results, Liu et al. (20) found that in vitro liver–derived DCs are more likely to induce Treg differentiation than spleen-derived DCs, and this process depended on the high programmed cell death-ligand 1 (PD-L1) expression level of liver-derived DCs (20).
Besides this, the interactions between mDCs and hepatic stellate cells (HSCs) can also induce immune tolerance effects. By secreting all-trans retinoic acid (ATRA), HSCs can induce the syntheses of arginase1 (ARG1) and inducible nitric oxide synthase (iNOS) in mDCs. Among them, ARG1 can degrade the arginine in the local microenvironment, while iNOS can induce the synthesis of NO, thus, inhibiting the function of effector T cells (21). At the same time, HSCs can secrete a variety of cytokines [for example IL-1, chemokine (C-C motif) ligand 2 (CCL2), and CCL3], which can activate the downstream signal transducer and activator of transcription 3 (STAT3) signaling pathway in mDCs and finally prompt mDCs to express IDO and PD-L1. The former (IDO) can catabolize the tryptophan in local microenvironment while the latter (PD-L1) can bind to the PD-1 on the effector T cells (22–24).
Plasmacytoid Dendritic Cells (pDCs)
In general conditions, plasmacytoid dendritic cells (pDCs) can present a small fraction of antigens and then activate T cells, although its function is much weaker than mDCs. It has been found that when compared with spleen-derived pDCs, liver-derived pDCs in mice showed immature phenotype and low secretion level of IL-12p70; thus, they showed a reduced ability to present antigens or activate T cells (25, 26). Besides this, a new subpopulation of CCR9+ pDCs was identified as tolerogenic pDCs in an acute graft-vs.-host disease model (27). It has been demonstrated that this subset of pDCs exist abundantly in the murine liver, produce IL-10 and TGF-β, and induce the differentiation of naïve T cells into a regulatory phenotype through the TLR7/9 pathway (28).
In terms of the interactions with other cells, the liver-derived pDCs have a lower Delta4/Jagged1 ratio of Notch ligands on the cell surface, which means that they can predominantly induce T cells to differentiate into T helper 2 (Th2) cells. As with mDC, pDC can upregulate the expression of PD-L1 through the STAT3 pathway under the impact of IL-27, thereby, inhibiting the function of the effector T cell (29, 30).
Kupffer Cells in Tolerance
Liver has the largest number of residential macrophages, named Kupffer cells, accounting for approximately 20% of the non-parenchymal cells in the liver. In general conditions, Kupffer cells are located in the hepatic sinusoid in order to fully interact with various molecules and cells in the blood. Their main functions are phagocytosing, secreting cytokines [such as IL-1, IL-6, IL-12, IL-18, TNF-α, and IFN-γ (31)], and processing and presenting antigens (32, 33).
Under a resting circumstance, compared with DCs, Kupffer cells express significantly lower levels of MHC-II and costimulatory molecules (such as B7-1, B7-2, CD40). Therefore, as intrahepatic APCs, their functions are significantly weaker than DCs. Moreover, You et al. (34) have found that steady-state Kupffer cells are able to secrete prostaglandin E2 (PGE2) and 15-deoxy-delta 12,14-PGJ2 (15d-PGJ2), which can directly inhibit antigen-specific CD4+ T cells activation (34, 35). In addition, through animal models, Sass et al. (36) confirmed that high doses of TNF can induce hepatocytes apoptosis, whereas low doses of TNF (pretreatment with 10 μ/kg) have the exact opposite role. After the invasion of endotoxin, Kupffer cells secrete anti-inflammatory cytokines such as IL-10 (37) and TGF-β (38) in response to continuous stimulation with low levels of lipopolysaccharide (LPS). This phenomenon is called LPS tolerance. In addition, increased TLR4-mediated expression of adhesion molecules in LSECs and KCs following continuous LPS exposure promoted trapping of T cells within the liver, resulting in lower numbers of circulating primed CD8+ T cells and weak immune responses (39).
Apart from secreting cytokines, Kupffer cells can directly interact with Tregs to stimulate their proliferation and induce them to secrete IL-10, which can inhibit the immune effect of cytotoxic T lymphocyte (CTL) on hepatocyte-expressed antigens (40, 41). Kuniyasu et al. (42) have undertaken the researches into the outcomes of activated CD8+ T cells in the liver and found that some of them can be retained in the liver. In the meantime, Kupffer cells can encourage the proliferation of these activated CD8+ T cells in the early period while they promote apoptosis in the late period. Ultimately, the CTL-mediated cellular immunity can be inhibited by Kupffer cells (42). Through animal experiments, Chen et al. (43) have found that Kupffer cells can promote T cell apoptosis through the Fas/Fas-L pathway. This effect can be inhibited by the Fas-L antibody (43). Similarly, there were researches that showed that Kupffer cells highly expressing PD-L1 can also inhibit the proliferation and functions of T cells by directly contacting them (44). Apart from this, human Kupffer cells activated through TLR2/4 pathways can synthesize IL-10, thus, suppressing IL-18-dependent NK cell activation (45).
Liver Sinusoidal Endothelial Cells (LSECs) in Tolerance
Liver sinusoidal endothelial cells account for about 50% of the non-parenchymal cells in the liver. The structure of LSEC is just like a perforated screen plate. They constitute the wall of the hepatic sinusoid. There is no basement-membrane at the inside/outside of LSECs, with only a few reticular fibers attached on them. In the liver, it has been reported that LSECs can be a unique population of organ-resident APCs; they display scavenger activity with the capacity to (cross) present exogenous antigens on both MHC II and MHC I molecules to CD4 or CD8 T cells, respectively (46, 47). Some animal experiments showed that, in normal conditions, although LSECs express only a few of the MHC class II molecules, they can induce the activation of naive CD4+ T cells and promote their secretion of interferon-γ IFN-γ, IL-4, and IL-10 (48). However, LSECs are not capable of inducing the differentiation of naïve CD4+ T cells into their effector T helper 2 (Th1) cell subpopulation (47).
As APCs, LESCs have specific immune tolerance-inducing effects on both CD4+ and CD8+ T cells. For CD4+ T cells, animal experiments have proved that after injecting allogeneic cells into the portal venous systems, the LSECs in the recipient mice express significantly increased levels of FasL. By this way, LESCs can induce apoptosis of CD4+ T cells via the Fas-FasL pathway, inhibiting the proliferation of CD4+ T cells and their secretion of IL-2 (49). For CD8+ T cells, there is a process called cross-tolerance. As observed with other APCs, LSECs are able to cross-present exogenous antigens to CD8+ T cells via MHC-I molecules. Once LSECs contacted CD8+ T cells, in the early period, CD8+ T cells showed a prominent proliferation effect, and the expression of CD69, CD25, CD44, and PD-1 on the surface of CD8+ T cells were unregulated. But soon afterwards, the interaction between LSECs and CD8+ T cells results in an upregulation of the inhibitory molecules (such as B7-H1) on LSECs, rather than the costimulatory molecules (such as CD80/86). It has been indicated that Bim, a pro-apoptotic Bcl-2 family member, is a pro-apoptotic mediator of this cross-tolerance process (50). Finally, we can obtain a specific subpopulation of LSECs (B7-H1highCD80/86lowLSECs), which can inhibit the cytotoxic effect of CD8+ T cell but fail to induce the clonal deletion of this subpopulation (51).
Besides the mechanism mentioned above, LESCs also have immune tolerance effects that are independent of their antigen presenting function. Through animal experiments, Kruse et al. (52) found that, under the influence of LSECs, CD4+ T cells can differentiate into CD25lowFoxP3−Tregs, which can inhibit the proliferation of naïve CD4+ T cells. The animal model of T cell-mediated autoimmune hepatitis has shown that CD25lowFoxP3− Tregs can downregulate the alanine aminotransferase (ALT; glutamic-pyruvic transaminase) level in peripheral blood and inhibit the infiltration of inflammatory cells in the liver tissue (52). In terms of immune regulation, LESCs can secrete liver and lymph node sinusoidal endothelial cell C-type lectin (LSECtin). The LSECtin can interact with CD44 on the surface of activated T cells, inhibits the secretion of effector cytokines by them (such as IL-2, IFN-γ and so on), and even induces T cell apoptosis (53). After repetitive invasions of LPS, LSECs reduce NK-κB activation and mediate liver tolerance to maintain the homeostasis in the liver (39). In addition, LESCs can interfere with the activation of CD8+ T cells by DCs through direct physical contact with DCs (54).
Natural Killer Cells (NK cells) in Tolerance
Natural killer cells account for the largest proportion (about 30–50%) of all lymphocytes (which consist of NK, NKT, γδT, αβT, and B cells) in a normal adult liver (55, 56). They are important mediators of liver damage in viral and inflammatory liver disease (57–59). Their functions are controlled by balance of activatory and inhibitory signals. In general, NK cells can be divided in two groups based on the molecular markers on their cell surface, CD3−CD56dimCD16+CD27− and CD3−CD56brightCD16−CD27+ NK cells. Among them, the former performs as professional cytotoxic cell, while the latter has a strong ability of cytokine secretion (mainly IFN-γ and TNF-α) (55, 60). Liver-derived NK cells are different from those in the peripheral blood, with higher expression of activation receptors (such as CD69, NKp44, NKp46, NKG2D), inhibition receptors (such as NKG2A+), and TNF related apoptosis inducing ligand (TRAIL), higher secretion of perforin and granzyme, and stronger cytotoxicity (56, 61, 62).
It is generally believed that, in liver transplantation, NK cells play roles in both rejection and tolerance. According to their origins, liver-derived NK cells can be divided in two groups, donor-derived NK cells and recipient-derived NK cells. It has been found that the recipient-derived NK cells play a major role in immune rejection, while the donor-derived NK cells mainly promote immune tolerance (61). Jamil et al. (63) have found that after liver transplantation, recipient NK cells exhibit tolerant phenotypes, with the downregulation of activating receptors and reduced cytotoxicity and cytokine production. Their research also revealed that NK cell tolerance is associated with perturbation of the IL-12/STAT4 signaling pathway, which might be a therapeutic target for clinical practice (63). There are clinical studies that showed that compared with the recipients who developed post-operation immune rejection, those with post-operation immune tolerance had significantly higher percentages and absolute counts of NK cells in their peripheral blood (64). Li et al. (65) have found that there are 13 genes that are significantly overexpressed in the NK cells of immune tolerant recipients after liver transplantation. This phenomenon suggested that NK cells may be involved in the induction of immune tolerance (65).
Natural Killer T (NKt) Cells in Tolerance
Natural killer T cells mostly exist in the liver, spleen, and bone marrow. They can express the molecular markers of NK cells. Beyond this, there are specific T cell receptor (TCR) Vα chains on their cell surface, which can recognize the glycolipid antigens presented by CD1d. Through the mechanism mentioned above, NKT cells can rapidly produce a large amount of cytokine (Th1 or Th2 type) and mediate immune response through direct or indirect ways. It is currently believed that Valpha14 NKT cells play a role in graft immune tolerance. Ikehara et al. (66) used animal experiments to show that mice deficient in Valpha14 NKT cells were not able to induce immune tolerance after pancreatic islets transplantation until the exogenous Valpha14 NKT cells were injected into the mice (66).
Clinical Relevance of Tolerance in Liver Transplantation
As compared with other solid organs, it is clear that human liver allografts show unique immunological features. It can be performed across positive cross matches, with small doses of immunosuppressive regimens and less frequent rejection events. For liver transplantation, some patients can eventually totally withdraw their immunosuppression therapy without undergoing rejection. This is a phenomenon called spontaneous operational tolerance, firstly reported in the early 1990s in Pittsburgh (67–69). Following this, several single centers described their experiences with the discontinuation of immunosuppression because of patient non-compliance (70–75). A US multicenter pediatric trial enrolled 20 recipients of parental living donor liver grafts at least 4 years after transplantation (76). Drug withdrawal was successfully accomplished in 12 patients for more than 6 years. Successful drug withdrawal was defined as 1 year off immunosuppression with normal liver function tests, for example liver biopsies performed at enrollment and 2 and 4 years after complete drug discontinuation that failed to show clinically significant histological changes. From these researches, we can see that the time after transplantation is a key factor associated with spontaneous operational tolerance. At present, a number of clinical trials are put into practice to confirm the high prevalence of tolerance among long-term surviving recipients, to help clarify the mechanisms behind human liver allograft tolerance and to determine whether it is possible to prospectively identify tolerant recipients on immunosuppression by employing diagnostic biomarkers.
Conclusions and Future Challenges
Up to now, it has been more than 40 years since the first time people regarded the liver as an immune-tolerant organ (77). During this period, people researched for the molecular mechanisms inducing intrahepatic immune tolerance and accumulated substantial evidence through animal experiments. However, currently, the relative lack of human studies in this field is one of the major problems that need to be overcome in the future. Although intrahepatic innate immune cells play important roles in inducing immune tolerance after liver transplantation (Figure 1), we seldom hear about clinical cases using their functions to reduce the doses of immunosuppressive agents. In addition, some animal experiments and clinical studies have shown that after the invasion of external pathogens (bacteria or viruses), some receptors (especially TLRs) on the surface of innate immune cells are activated, inducing the release of inflammatory cytokines (such as IL-6, TNF-α, IFN-I, and so on), further interrupting the development of intrahepatic immune tolerance (78). Interestingly, in 2014, the study performed by Bohne et al. (79) showed that for hepatitis C virus (HCV)-infected recipients, tolerance was associated with intrahepatic overexpression of type I IFN and an expansion of exhausted HCV-specific circulating CD8+ T cells. This is in contrast to what we had previously reported, that is, IFN-I can activate STAT4 and contribute to Th 1 response, which play a central role in allograft rejection (80). For this special circumstance of HCV infection, the author hypothesized that high intrahepatic IFN-I signaling induced by HCV infection caused T cell exhaustion, inactivated allo-specific T cell clones, and promoted a tolerogenic liver microenvironment that facilitates operational tolerance. On the basis of these results, we conclude that usually intrahepatic innate immune cells are in balanced states, between pro- and anti-inflammation. Under normal resting conditions, intrahepatic innate immune cells mainly induce intrahepatic immune tolerance, whereas under inflammatory conditions, they promote immune response and release inflammatory cytokines. Further exploration of the essential factors in the transformation of intrahepatic innate immune cells between these two states is of great importance for finding out the mechanisms of intrahepatic immune tolerance.
Author Contributions
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
Funding
This study was supported by the National Natural Science Foundation of China (81700570 and 81601390).
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
1. Thomson AW, Knolle PA. Antigen-presenting cell function in the tolerogenic liver environment. Nat Rev Immunol. (2010) 10:753–66. doi: 10.1038/nri2858
2. Sawada T, Asanuma Y, Furuya T, Shibata S, Yasui O, Sato T, et al. Induction of systemic tolerance in islet allograft by liver transplantation. Transplant Proc. (2001) 33:2995–9. doi: 10.1016/S0041-1345(01)02285-0
3. Zhang M, Tang H, Guo Z, An H, Zhu X, Song W, et al. Splenic stroma drives mature dendritic cells to differentiate into regulatory dendritic cells. Nat Immunol. (2004) 5:1124–33. doi: 10.1038/ni1130
4. Svensson M, Maroof A, Ato M, Kaye PM. Stromal cells direct local differentiation of regulatory dendritic cells. Immunity (2004) 21:805–16. doi: 10.1016/j.immuni.2004.10.012
5. Wakkach A, Fournier N, Brun V, Breittmayer JP, Cottrez F, Groux H. Characterization of dendritic cells that induce tolerance and T regulatory 1 cell differentiation in vivo. Immunity (2003) 18:605–17. doi: 10.1016/S1074-7613(03)00113-4
6. He W, Chen L, Zheng L, Luo L, Gao L. Prolonged survival effects induced by immature dendritic cells and regulatory T cells in a rat liver transplantation model. Mol Immunol. (2016) 79:92–97. doi: 10.1016/j.molimm.2016.10.004
7. Dai FZ, Yang J, Chen XB, Xu MQ. Zinc finger protein A20 inhibits maturation of dendritic cells resident in rat liver allograft. J Surg Res. (2013) 183:885–93. doi: 10.1016/j.jss.2013.01.062
8. Ouaaz F, Arron J, Zheng Y, Choi Y, Beg AA. Dendritic cell development and survival require distinct NF-kappaB subunits. Immunity (2002) 16:257–70. doi: 10.1016/S1074-7613(02)00272-8
9. Rastellini C, Lu L, Ricordi C, Starzl TE, Rao AS, Thomson AW. Granulocyte/macrophage colony-stimulating factor-stimulated hepatic dendritic cell progenitors prolong pancreatic islet allograft survival. Transplantation (1995) 60:1366–70.
10. Thomson AW, Lu L. Are dendritic cells the key to liver transplant tolerance? Immunol Today (1999) 20:27–32.
11. Salerno F, Gerbes A, Ginès P, Wong F, Arroyo V. Diagnosis, prevention and treatment of hepatorenal syndrome in cirrhosis. Postgrad Med J. (2008) 84:662–70. doi: 10.1136/gut.2006.107789
12. Kushwah R, Wu J, Oliver JR, Jiang G, Zhang J, Siminovitch KA, et al. Uptake of apoptotic DC converts immature DC into tolerogenic DC that induce differentiation of Foxp3+ Treg. Eur J Immunol. (2010) 40:1022–35. doi: 10.1002/eji.200939782
13. van den Oord JJ, De Vos R, Facchetti F, Delabie J, De Wolf-Peeters C, Desmet VJ. Distribution of non-lymphoid, inflammatory cells in chronic HBV infection. J Pathol. (1990) 160:223–30. doi: 10.1002/path.1711600308
14. Pillarisetty VG, Shah AB, Miller G, Bleier JI, DeMatteo RP. Liver dendritic cells are less immunogenic than spleen dendritic cells because of differences in subtype composition. J Immunol. (2004) 172:1009–17. doi: 10.4049/jimmunol.172.2.1009
15. Abe M, Tokita D, Raimondi G, Thomson AW. Endotoxin modulates the capacity of CpG-activated liver myeloid DC to direct Th1-type responses. Eur J Immunol. (2006) 36:2483–93. doi: 10.1002/eji.200535767
16. De Creus A, Abe M, Lau AH, Hackstein H, Raimondi G, Thomson AW. Low TLR4 expression by liver dendritic cells correlates with reduced capacity to activate allogeneic T cells in response to endotoxin. J Immunol. (2005) 174:2037–45. doi: 10.4049/jimmunol.174.4.2037
17. Chen Y, Jiang G, Yang HR, Gu X, Wang L, Hsieh CC, et al. Distinct response of liver myeloid dendritic cells to endotoxin is mediated by IL-27. J Hepatol. (2009) 51:510–9. doi: 10.1016/j.jhep.2009.04.026
18. Khanna A, Morelli AE, Zhong C, Takayama T, Lu L, Thomson AW. Effects of liver-derived dendritic cell progenitors on Th1- and Th2-like cytokine responses in vitro and in vivo. J Immunol. (2000) 164:1346–54. doi: 10.4049/jimmunol.164.3.1346
19. Jinushi M, Takehara T, Tatsumi T, Yamaguchi S, Sakamori R, Hiramatsu N, et al. Natural killer cell and hepatic cell interaction via NKG2A leads to dendritic cell-mediated induction of CD4 CD25 T cells with PD-1-dependent regulatory activities. Immunology (2007) 120:73–82. doi: 10.1111/j.1365-2567.2006.02479.x
20. Liu H, Bakthavatsalam R, Meng Z, Li Z, Li W, Perkins JD, et al. PD-L1 signal on liver dendritic cells is critical for Foxp3+CD4+CD25+ Treg and liver tolerance induction in mice. Transplant Proc. (2013) 45:1853–5. doi: 10.1016/j.transproceed.2013.03.015
21. Bhatt S, Qin J, Bennett C, Qian S, Fung JJ, Hamilton TA, et al. All-trans retinoic acid induces arginase-1 and inducible nitric oxide synthase-producing dendritic cells with T cell inhibitory function. J Immunol. (2014) 192:5098–108. doi: 10.4049/jimmunol.1303073
22. Sumpter TL, Dangi A, Matta BM, Huang C, Stolz DB, Vodovotz Y, et al. Hepatic stellate cells undermine the allostimulatory function of liver myeloid dendritic cells via STAT3-dependent induction of IDO. J Immunol. (2012) 189:3848–58. doi: 10.4049/jimmunol.1200819
23. Mellor AL, Keskin DB, Johnson T, Chandler P, Munn DH. Cells expressing indoleamine 2,3-dioxygenase inhibit T cell responses. J Immunol. (2002) 168:3771–6. doi: 10.4049/jimmunol.168.8.3771
24. Wölfle SJ, Strebovsky J, Bartz H, Sähr A, Arnold C, Kaiser C, et al. PD-L1 expression on tolerogenic APCs is controlled by STAT-3. Eur J Immunol. (2011) 41:413–24. doi: 10.1002/eji.201040979
25. Kingham TP, Chaudhry UI, Plitas G, Katz SC, Raab J, DeMatteo RP. Murine liver plasmacytoid dendritic cells become potent immunostimulatory cells after Flt-3 ligand expansion. Hepatology (2007) 45:445–54. doi: 10.1002/hep.21457
26. Tokita D, Sumpter TL, Raimondi G, Zahorchak AF, Wang Z, Nakao A, et al. Poor allostimulatory function of liver plasmacytoid DC is associated with pro-apoptotic activity, dependent on regulatory T cells. J Hepatol. (2008) 49:1008–18. doi: 10.1016/j.jhep.2008.07.028
27. Hadeiba H, Sato T, Habtezion A, Oderup C, Pan J, Butcher EC. CCR9 expression defines tolerogenic plasmacytoid dendritic cells able to suppress acute graft-versus-host disease. Nat Immunol. (2008) 9:1253–60. doi: 10.1038/ni.1658
28. Nakamoto N, Ebinuma H, Kanai T, Chu PS, Ono Y, Mikami Y, et al. CCR9+ macrophages are required for acute liver inflammation in mouse models of hepatitis. Gastroenterology (2012) 142:366–76. doi: 10.1053/j.gastro.2011.10.039
29. Mascanfroni ID, Yeste A, Vieira SM, Burns EJ, Patel B, Sloma I, et al. IL-27 acts on DCs to suppress the T cell response and autoimmunity by inducing expression of the immunoregulatory molecule CD39. Nat Immunol. (2013) 14:1054–63. doi: 10.1038/ni.2695
30. Matta BM, Raimondi G, Rosborough BR, Sumpter TL, Thomson AW. IL-27 production and STAT3-dependent upregulation of B7-H1 mediate immune regulatory functions of liver plasmacytoid dendritic cells. J Immunol. (2012) 188:5227–37. doi: 10.4049/jimmunol.1103382
31. Karimi MH, Geramizadeh B, Malek-Hosseini SA. Tolerance induction in liver. Int J Organ Transplant Med. (2015) 6:45–54.
32. Stienstra R, Saudale F, Duval C, Keshtkar S, Groener JE, van Rooijen N, et al. Kupffer cells promote hepatic steatosis via interleukin-1beta-dependent suppression of peroxisome proliferator-activated receptor alpha activity. Hepatology (2010) 51:511–22. doi: 10.1002/hep.23337
33. Huang HF, Zeng Z, Chen MQ. Roles of Kupffer cells in liver transplantation. Hepatogastroenterology (2012) 59:1251–7. doi: 10.5754/hge12046
34. You Q, Cheng L, Kedl RM, Ju C. Mechanism of T cell tolerance induction by murine hepatic Kupffer cells. Hepatology (2008) 48:978–90. doi: 10.1002/hep.22395
35. Kamei T, Callery MP, Flye MW. Kupffer cell blockade prevents induction of portal venous tolerance in rat cardiac allograft transplantation. J Surg Res. (1990) 48:393–6. doi: 10.1016/0022-4804(90)90001-I
36. Sass G, Shembade ND, Haimerl F, Lamoureux N, Hashemolhosseini S, Tannapfel A, et al. TNF pretreatment interferes with mitochondrial apoptosis in the mouse liver by A20-mediated down-regulation of Bax. J Immunol. (2007) 179:7042–9. doi: 10.4049/jimmunol.179.10.7042
37. Knolle PA, Uhrig A, Protzer U, Trippler M, Duchmann R, Meyer zum Büschenfelde KH, et al. Interleukin-10 expression is autoregulated at the transcriptional level in human and murine Kupffer cells. Hepatology (1998) 27:93–9.
38. Bissell DM, Wang SS, Jarnagin WR, Roll FJ. Cell-specific expression of transforming growth factor-beta in rat liver. Evidence for autocrine regulation of hepatocyte proliferation. J Clin Invest. (1995) 96:447–55.
39. Uhrig A, Banafsche R, Kremer M, Hegenbarth S, Hamann A, Neurath M, et al. Development and functional consequences of LPS tolerance in sinusoidal endothelial cells of the liver. J Leukoc Biol. (2005) 77:626–33. doi: 10.1189/jlb.0604332
40. Breous E, Somanathan S, Vandenberghe LH, Wilson JM. Hepatic regulatory T cells and Kupffer cells are crucial mediators of systemic T cell tolerance to antigens targeting murine liver. Hepatology (2009) 50:612–21. doi: 10.1002/hep.23043
41. Wiegard C, Frenzel C, Herkel J, Kallen KJ, Schmitt E, Lohse AW. Murine liver antigen presenting cells control suppressor activity of CD4+CD25+ regulatory T cells. Hepatology (2005) 42:193–9. doi: 10.1002/hep.20756
42. Kuniyasu Y, Marfani SM, Inayat IB, Sheikh SZ, Mehal WZ. Kupffer cells required for high affinity peptide-induced deletion, not retention, of activated CD8+ T cells by mouse liver. Hepatology (2004) 39:1017–27. doi: 10.1002/hep.20153
43. Chen Y, Liu Z, Liang S, Luan X, Long F, Chen J, et al. Role of Kupffer cells in the induction of tolerance of orthotopic liver transplantation in rats. Liver Transpl. (2008) 14:823–36. doi: 10.1002/lt.21450
44. Gong J, Cao D, Chen Y, Li J, Gong J, Zeng Z. Role of programmed death ligand 1 and Kupffer cell in immune regulation after orthotopic liver transplantation in rats. Int Immunopharmacol. (2017) 48:8–16. doi: 10.1016/j.intimp.2017.04.009
45. Tu Z, Bozorgzadeh A, Pierce RH, Kurtis J, Crispe IN, Orloff MS. TLR-dependent cross talk between human Kupffer cells and NK cells. J Exp Med. (2008) 205:233–44. doi: 10.1084/jem.20072195
46. Limmer A, Ohl J, Kurts C, Ljunggren HG, Reiss Y, Groettrup M, et al. Efficient presentation of exogenous antigen by liver endothelial cells to CD8+ T cells results in antigen-specific T-cell tolerance. Nat Med. (2000) 6:1348–54. doi: 10.1038/82161
47. Knolle PA, Schmitt E, Jin S, Germann T, Duchmann R, Hegenbarth S, et al. Induction of cytokine production in naive CD4+ T cells by antigen-presenting murine liver sinusoidal endothelial cells but failure to induce differentiation toward Th1 cells. Gastroenterology (1999) 116:1428–40. doi: 10.1016/S0016-5085(99)70508-1
48. Neumann K, Rudolph C, Neumann C, Janke M, Amsen D, Scheffold A. Liver sinusoidal endothelial cells induce immunosuppressive IL-10-producing Th1 cells via the Notch pathway. Eur J Immunol. (2015) 45:2008–16. doi: 10.1002/eji.201445346
49. Tokita D, Shishida M, Ohdan H, Onoe T, Hara H, Tanaka Y, et al. Liver sinusoidal endothelial cells that endocytose allogeneic cells suppress T cells with indirect allospecificity. J Immunol. (2006) 177:3615–24. doi: 10.4049/jimmunol.177.6.3615
50. Davey GM, Kurts C, Miller JF, Bouillet P, Strasser A, Brooks AG, et al. Peripheral deletion of autoreactive CD8 T cells by cross presentation of self-antigen occurs by a Bcl-2-inhibitable pathway mediated by Bim. J Exp Med. (2002) 196:947–55. doi: 10.1084/jem.20020827
51. Diehl L, Schurich A, Grochtmann R, Hegenbarth S, Chen L, Knolle PA. Tolerogenic maturation of liver sinusoidal endothelial cells promotes B7-homolog 1-dependent CD8+ T cell tolerance. Hepatology (2008) 47:296–305. doi: 10.1002/hep.21965
52. Kruse N, Neumann K, Schrage A, Derkow K, Schott E, Erben U, et al. Priming of CD4+ T cells by liver sinusoidal endothelial cells induces CD25low forkhead box protein 3- regulatory T cells suppressing autoimmune hepatitis. Hepatology (2009) 50:1904–13. doi: 10.1002/hep.23191
53. Dong H, Zhu G, Tamada K, Flies DB, van Deursen JM, Chen L. B7-H1 determines accumulation and deletion of intrahepatic CD8+ T lymphocytes. Immunity (2004) 20:327–36. doi: 10.1016/S1074-7613(04)00050-0
54. Schildberg FA, Hegenbarth SI, Schumak B, Scholz K, Limmer A, Knolle PA. Liver sinusoidal endothelial cells veto CD8 T cell activation by antigen-presenting dendritic cells. Eur J Immunol. (2008) 38:957–67. doi: 10.1002/eji.200738060
55. Lysakova-Devine T, O'Farrelly C. Tissue-specific NK cell populations and their origin. J Leukoc Biol. (2014) 96:981–90. doi: 10.1189/jlb.1RU0514-241R
56. Moroso V, Metselaar HJ, Mancham S, Tilanus HW, Eissens D, van der Meer A, et al. Liver grafts contain a unique subset of natural killer cells that are transferred into the recipient after liver transplantation. Liver Transpl. (2010) 16:895–908. doi: 10.1002/lt.22080
57. Klugewitz K, Adams DH, Emoto M, Eulenburg K, Hamann A. The composition of intrahepatic lymphocytes: shaped by selective recruitment? Trends Immunol. (2004) 25:590–4. doi: 10.1016/j.it.2004.09.006
58. Dunn C, Brunetto M, Reynolds G, Christophides T, Kennedy PT, Lampertico P, et al. Cytokines induced during chronic hepatitis B virus infection promote a pathway for NK cell-mediated liver damage. J Exp Med. (2007) 204:667–80. doi: 10.1084/jem.20061287
59. Laso FJ, Almeida J, Torres E, Vaquero JM, Marcos M, Orfao A. Chronic alcohol consumption is associated with an increased cytotoxic profile of circulating lymphocytes that may be related with the development of liver injury. Alcohol Clin Exp Res. (2010) 34:876–85. doi: 10.1111/j.1530-0277.2010.01160.x
60. Cooper MA, Fehniger TA, Caligiuri MA. The biology of human natural killer-cell subsets. Trends Immunol. (2001) 22:633–40. doi: 10.1016/S1471-4906(01)02060-9
61. Harmon C, Sanchez-Fueyo A, O'Farrelly C, Houlihan DD. Natural killer cells and liver transplantation: orchestrators of rejection or tolerance? Am J Transplant. (2016) 16:751–7. doi: 10.1111/ajt.13565
62. Harmon C, Robinson MW, Fahey R, Whelan S, Houlihan DD, Geoghegan J, et al. Tissue-resident Eomes(hi) T-bet(lo) CD56(bright) NK cells with reduced proinflammatory potential are enriched in the adult human liver. Eur J Immunol. (2016) 46:2111–20. doi: 10.1002/eji.201646559
63. Jamil KM, Hydes TJ, Cheent KS, Cassidy SA, Traherne JA, Jayaraman J, et al. STAT4-associated natural killer cell tolerance following liver transplantation. Gut (2017) 66:352–361. doi: 10.1136/gutjnl-2015-309395
64. García de la Garza R, Sarobe P, Merino J, Lasarte JJ, D'Avola D, Belsue V, et al. Immune monitoring of immunosuppression withdrawal of liver transplant recipients. Transpl Immunol. (2015) 33:110–6. doi: 10.1016/j.trim.2015.07.006
65. Li L, Wozniak LJ, Rodder S, Heish S, Talisetti A, Wang Q, et al. A common peripheral blood gene set for diagnosis of operational tolerance in pediatric and adult liver transplantation. Am J Transplant. (2012) 12:1218–28. doi: 10.1111/j.1600-6143.2011.03928.x
66. Ikehara Y, Yasunami Y, Kodama S, Maki T, Nakano M, Nakayama T, et al. CD4+ Valpha14 natural killer T cells are essential for acceptance of rat islet xenografts in mice. J Clin Invest. (2000) 105:1761–7. doi: 10.1172/JCI8922
67. Starzl TE, Demetris AJ, Trucco M, Murase N, Ricordi C, Ildstad S, et al. Cell migration and chimerism after whole-organ transplantation: the basis of graft acceptance. Hepatology (1993) 17:1127–52. doi: 10.1002/hep.1840170629
68. Reyes J, Zeevi A, Ramos H, Tzakis A, Todo S, Demetris AJ, et al. Frequent achievement of a drug-free state after orthotopic liver transplantation. Transplant Proc. (1993) 25:3315–9.
69. Takatsuki M, Uemoto S, Inomata Y, Egawa H, Kiuchi T, Fujita S, et al. Weaning of immunosuppression in living donor liver transplant recipients. Transplantation (2001) 72:449–54. doi: 10.1097/00007890-200108150-00016
70. Mañez R, Kusne S, Linden P, Gonzalez-Pinto I, Bonet H, Kramer D, et al. Temporary withdrawal of immunosuppression for life-threatening infections after liver transplantation. Transplantation (1994) 57:149–51.
71. Massarollo PC, Mies S, Abdala E, Leitão RM, Raia S. Immunosuppression withdrawal for treatment of severe infections in liver transplantation. Transplant Proc. (1998) 30:1472–4.
72. Devlin J, Doherty D, Thomson L, Wong T, Donaldson P, Portmann B, et al. Defining the outcome of immunosuppression withdrawal after liver transplantation. Hepatology (1998) 27:926–33. doi: 10.1002/hep.510270406
73. Pons JA, Yélamos J, Ramírez P, Oliver-Bonet M, Sánchez A, Rodríguez-Gago M, et al. Endothelial cell chimerism does not influence allograft tolerance in liver transplant patients after withdrawal of immunosuppression. Transplantation (2003) 75:1045–7. doi: 10.1097/01.TP.0000058472.71775.7d
74. Eason JD, Cohen AJ, Nair S, Alcantera T, Loss GE. Tolerance: is it worth the risk? Transplantation (2005) 79:1157–9. doi: 10.1097/00007890-200407271-00319
75. Behrens-Baumann W, Thiery J. Cataracts by lipid lowering drugs? Three different HMG-CoA reductase inhibitors studied in hypercholesterolemic rabbits. Res Exp Med (Berl) (1992) 192:7–11. doi: 10.1007/BF02576252
76. Feng S, Ekong UD, Lobritto SJ, Demetris AJ, Roberts JP, Rosenthal P, et al. Complete immunosuppression withdrawal and subsequent allograft function among pediatric recipients of parental living donor liver transplants. JAMA (2012) 307:283–93. doi: 10.1001/jama.2011.2014
77. Calne RY, Sells RA, Pena JR, Davis DR, Millard PR, Herbertson BM, et al. Induction of immunological tolerance by porcine liver allografts. Nature (1969). 223:472–6.
78. Goldstein DR. Inflammation and transplantation tolerance. Semin Immunopathol. (2011) 33:111–5. doi: 10.1007/s00281-011-0251-2
79. Bohne F, Londoño MC, Benítez C, Miquel R, Martínez-Llordella M, Russo C, et al. HCV-induced immune responses influence the development of operational tolerance after liver transplantation in humans. Sci Transl Med. (2014) 6:242ra81. doi: 10.1126/scitranslmed.3008793
Keywords: liver transplantation, innate immune cells, immune tolerance, dendritic cells, Kupffer cells, NK cells, LSECs
Citation: Huang H, Lu Y, Zhou T, Gu G and Xia Q (2018) Innate Immune Cells in Immune Tolerance After Liver Transplantation. Front. Immunol. 9:2401. doi: 10.3389/fimmu.2018.02401
Received: 31 May 2018; Accepted: 27 September 2018;
Published: 09 November 2018.
Edited by:
Aurore Saudemont, GlaxoSmithKline, United KingdomReviewed by:
Alain Le Moine, Free University of Brussels, BelgiumAlexandr Bazhin, Klinikum der Universität München, Germany
Copyright © 2018 Huang, Lu, Zhou, Gu and Xia. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Guangxiang Gu, Z3VndWFuZ3hpYW5nQDE2My5jb20=
Qiang Xia, bmpnZ3gyMDA5QDE2My5jb20=
†These authors have contributed equally to this work
 Hongting Huang
Hongting Huang Yefeng Lu†
Yefeng Lu† Guangxiang Gu
Guangxiang Gu