- 1Affiliated Cancer Hospital & Institute of Guangzhou Medical University, State Key Laboratory of Respiratory Diseases, Guangzhou Medical University, Guangzhou, China
- 2Department of Clinical Oncology, Taihe Hospital, Hubei University of Medicine, Shiyan, China
- 3Department of Microbiology and Parasitology, School of Basic Medical Sciences, Shanghai Medical College, Fudan University, Shanghai, China
The pandemic of coronavirus disease 2019 (COVID-19), a disease which causes severe lung injury and multiple organ damage, presents an urgent need for new drugs. The case severity and fatality of COVID-19 are associated with excessive inflammation, namely, a cytokine storm. Metformin, a widely used drug to treat type 2 diabetes (T2D) mellitus and metabolic syndrome, has immunomodulatory activity that reduces the production of proinflammatory cytokines using macrophages and causes the formation of neutrophil extracellular traps (NETs). Metformin also inhibits the cytokine production of pathogenic Th1 and Th17 cells. Importantly, treatment with metformin alleviates various lung injuries in preclinical animal models. In addition, a recent proteomic study revealed that metformin has the potential to directly inhibit SARS-CoV-2 infection. Furthermore, retrospective clinical studies have revealed that metformin treatment reduces the mortality of T2D with COVID-19. Therefore, metformin has the potential to be repurposed to treat patients with COVID-19 at risk of developing severe illness. This review summarizes the immune pathogenesis of SARS-CoV-2 and addresses the effects of metformin on inhibiting cytokine storms and preventing SARS-CoV-2 infection, as well as its side effects.
Introduction
In recent decades, the world has experienced outbreaks of newly emerging viruses, including severe acute respiratory syndrome coronavirus (SARS-CoV-1), Middle East respiratory syndrome coronavirus (MERS-CoV), H5N1 virus, pandemic H1N1 virus, and H7N9 virus (1–4). Currently, coronavirus disease 2019 (COVID-19), caused by SARS-CoV-2 infection, has become a global pandemic. Similar to the H7N9 virus (5, 6), SARS-CoV-2 causes high mortality in elderly patients who have preexisting chronic diseases. Acute lung injury is one of the major causes of the high mortality of patients infected with H5N1, H7N9, MERS-CoV, SARS-CoV-1, and SARS-CoV-2 (1–4). Rising evidence suggests that an elevated inflammatory immune response, characteristic of cytokine storms (7–10), is linked to the acute lung injury and fatality caused by these viruses (11–13).
To date, there is no effective treatment for acute lung injury caused by viral infections. Metformin is a clinically approved anti-diabetes drug. Recent studies have shown that metformin not only has immunomodulatory and antiviral activities but also prevents various acute lung injuries in animal models (14, 15). Thus, this article reviews the immune pathogenesis of SARS-CoV-2 infection and discusses the possibility of using metformin as a drug for mitigating COVID-19 illness severity.
Excessive Inflammation Likely Contributes to the Severe Illness of Patients With COVID-19
SARS-CoV-2 infects human epithelial cells by binding to human angiotensin-converting enzyme 2 (ACE2). Single-cell sequencing analysis shows that the ACE2 gene is expressed in cell clusters in organs including the lung, heart, esophagus, kidney, bladder, testis, and ileum (16–18), indicating that these organs are at risk of SARS-CoV-2 infection. Consistently, multiorgan failure has been observed in some patients with severe COVID-19 (7, 10, 19). The ACE2 expression profile suggests that SARS-CoV-2 infection may initiate or even directly cause organ failure (20).
In addition to direct infection, evidence shows that an elevated inflammatory immune response is also involved in the pathogenesis of SARS-CoV-2 infection. Patients in the intensive care unit (ICU) had higher plasma levels of IL-2, IL-7, IL-10, G-CSF, IP-10, MCP-1, MIP-1α, and TNFα than non-ICU patients (7). Some severe patients also have elevated IL-6 levels in their plasma (21, 22), which correlates with respiratory failure and fatality (19, 22). In addition, the serum levels of C-reactive protein, which is positively regulated by IL-6, are elevated in patients with severe COVID-19 (7, 22). The systemic elevation of cytokines is reminiscent of the cytokine storm or cytokine release syndrome (CRS) that occurs in severe infections of influenza viruses, MERS-CoV, and SARS-CoV-1 (20, 23). Due to the critical role of IL-6 in promoting CRS, inhibition of IL-6 or IL-6 receptor with antibodies has been proposed to treat patients with severe COVID-19 (23, 24).
How SARS-CoV-2 infection triggers cytokine storms remains largely speculative. Concomitant with elevated cytokines, patients with severe COVID-19 exhibit marked lymphopenia. A recent autopsy study of patients with COVID-19 shows that SARS-CoV-2 infection is detected in CD169+ macrophages that express the ACE2 molecule and produce IL-6. The infection of these CD169+ macrophages correlated with lymphocyte apoptosis in the spleen and lymph nodes. This finding may partly explain the excessive inflammation and lymphopenia during severe SARS-CoV-2 infection (25).
Consistent with the involvement of excessive innate immunity in the pathogenesis of severe COVID-19, infiltration of neutrophils in the lung has been found in an autopsy specimen (26). High neutrophil counts are significantly associated with COVID-19 fatality (10), and the neutrophil-to-lymphocyte ratio is an independent risk factor for the illness severity of COVID-19 (27). When a cytokine storm occurs, chemokines recruit neutrophils to lung lesions, and proinflammatory cytokines, such as IL-1β and IL-6, activate neutrophils to produce NETs, which may contribute to lung damage and mortality in patients with COVID-19 (26). It has been reported that aberrant NET formation is linked to pulmonary diseases, particularly acute respiratory distress syndrome (26). Therefore, targeting NETs with existing drugs is also proposed to reduce the clinical severity of COVID-19 (26).
Several pieces of evidence suggest that aberrant activation of immune cells may also contribute to cytokine storms and pathogenic manifestations in the lungs. Single-cell analysis of bronchoalveolar lavage fluid shows that patients with mild COVID-19 have an expansion of clonal CD8 T cells, whereas patients with severe disease have a decline in T cells and NK cells and a concomitant increase in inflammatory FCN1+ macrophages (28). These observations suggest that weak adaptive immunity may result in an inadequate ability to control viral infection. Thus, the persistent presence of SARS-CoV-2 activates alveolar macrophages or epithelial cells to produce various proinflammatory cytokines and chemokines, triggering or recruiting more innate immune cells and thereby amplifying inflammation (20, 29). Furthermore, aberrant Th1 and Th17 cells may also promote the activation of innate immune cells by producing proinflammatory cytokines, including IFNγ, IL-17, and TNFα (13, 24, 30). In addition, damage-associated molecular patterns (DAMPs) released by injured lung epithelial cells could activate innate immune cells as well. The cascade of innate immune responses ultimately results in uncontrolled inflammation (20) (Figure 1).
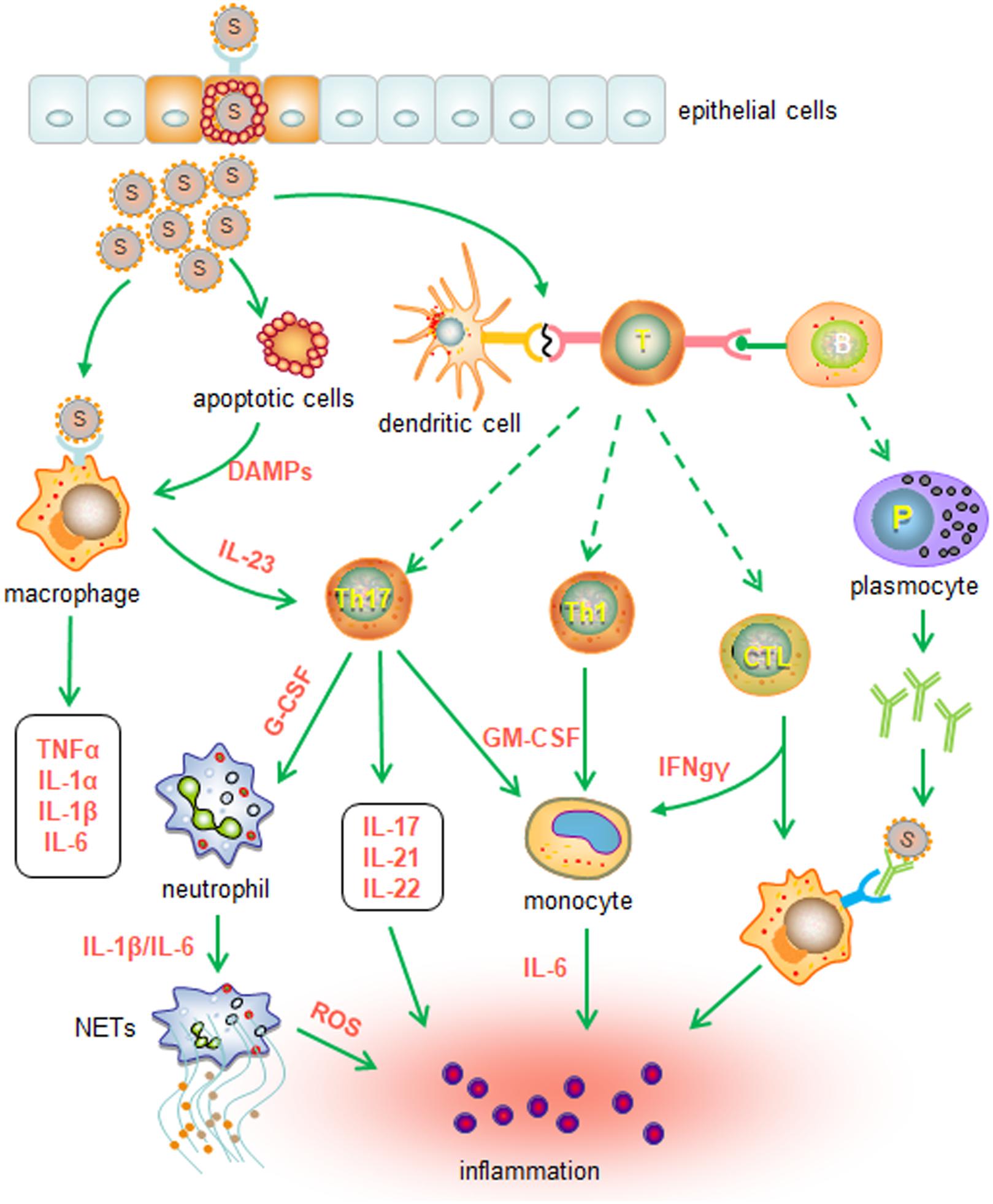
Figure 1. Putative innate immune responses induced by SARS-CoV-2 infection. SARS-CoV-2 and DAMPs released from damaged cells activate macrophages and cause the production of proinflammatory cytokines, such as IL-1α, IL-1β, IL-6, and TNFα, in the early stage of infection. The adaptive immune responses subsequently lead to the secretion of proinflammatory cytokines and chemokines, which may promote further innate cell recruitment and activation. Furthermore, activated Th17 cells and Th1 cells may recruit neutrophils and monocytes through the release of G-CSF and GM-CSF, respectively. Activated CTLs may drive the differentiation of monocytes and macrophages by secreting IFNγ. Inflammatory chemokines and cytokines also recruit and activate neutrophils to produce ROS and NETs. Taken together, the cytokine storm induced by innate immune cells ultimately results in inflammation and injury.
Metformin Inhibits Inflammatory Responses and Alleviates Acute Lung Injuries
Metformin, namely, N,N-dimethylbiguanide, is a first-line drug to treat type 2 diabetes (T2D) and metabolic syndromes. Metformin has a high safety profile and is thus widely used. Its glucose-lowering effect is efficacious when used as monotherapy or in combination with other antidiabetic agents. Patients with T2D who are treated with metformin also had a significant reduction in complications, including myocardial infarction, hypertrophy, and diabetic cardiomyopathy, suggesting that metformin has a cardiovascular protective effect (31). In addition, recent studies have revealed that metformin has numerous other beneficial effects for underlying diseases, including anticancer, anti-aging, neuroprotective, and immunomodulatory effects (31–33).
Similar to its glucose-lowering effect, the immunomodulatory effect of metformin mainly depends on the activation of AMP-activated protein kinase (AMPK) (32–34) (Figure 2). In brief, metformin directly inhibits respiratory-chain complex 1 of the mitochondrial electron transport chain, resulting in a reduction in ATP synthesis and thereby an increase in the AMP/ATP or ADP/ATP ratio, which consequentially activates AMPK through the binding of either AMP or ADP. AMPK activation not only switches off mTOR signaling through direct phosphorylation of TSC2 and RAPTOR but also inhibits the NF-κB pathway (31). It has been demonstrated that metformin has an immunosuppressive activity in both in vitro and in vivo models. For example, metformin inhibits the expression of IL-1β, IL-6, and TNFα by activated macrophages (33, 35–37) but enhances their IL-10 expression (38). Metformin also reduces the release of NETs from neutrophils in patients with diabetes (39, 40). Furthermore, treatment of mice with metformin results in a reduction in the cytokine production of Th1 and Th17 cells and their infiltration in the central nervous system, slowing the disease progression of experimental autoimmune encephalomyelitis (41). Taken together with its suppressive effects on both innate immunity and pathogenic Th cell responses, metformin has the potential to suppress the cytokine storm produced by severe COVID-19.
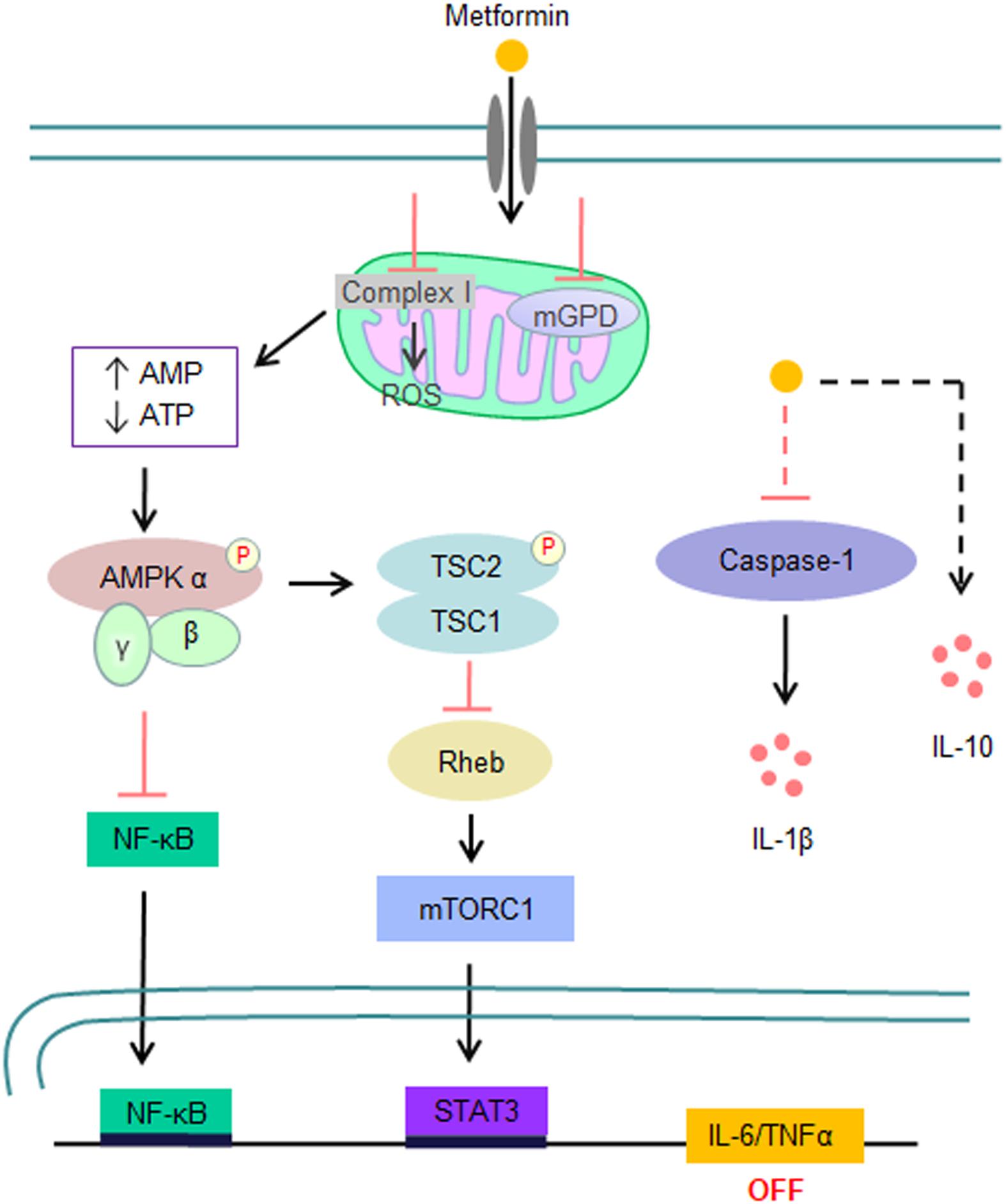
Figure 2. Signaling pathways that are involved in the inhibition of the inflammatory storm by metformin. Metformin directly inhibits complex I activity of the mitochondrial electron transport chain, resulting in an elevated AMP/ATP or ADP/ATP ratio and consequential AMPK activation. Activated AMPK subsequently inhibits the expression of proinflammatory cytokines, such as IL-6 and TNFα, by suppressing the NF-κB and mTOR signaling pathways. In addition, metformin also inhibits IL-1β expression and enhances IL-10 expression by macrophages in response to LPS stimulation in an AMPK-independent manner.
Studies have shown that metformin alleviates inflammation and protects against acute lung injuries in several animal models. First, metformin treatment significantly reduces lipopolysaccharide (LPS)-induced lung destruction and paraquat poisoning-induced acute lung injury (42–45). Second, metformin attenuates lung injury caused by the high pressure of mechanical ventilation (46). Furthermore, metformin reverses established fibrosis of injured lungs in a bleomycin mouse model (47). Together with the anti-inflammatory effects, these findings support that metformin has the potential to mitigate the inflammation and lung injuries of severe COVID-19 infection.
Metformin Is a Potential Antiviral Drug for COVID-19
A recent study has identified 332 protein interactions between SARS-CoV-2 proteins and human proteins with affinity purification-mass spectrometry analysis (48). The analysis has revealed that metformin may target the interactions between viral proteins and host factors, such as viral protein Nsp7 and human NDUFA2, and viral protein Orf9c and human NDUFAF1 or NDUFB9, and thus has antiviral activity.
It has also been reported that metformin has antiviral activities in other viral infections via the activation of AMPK (49–52). Metformin inhibits dengue virus infection by restoring AMPK activity (49), which is attenuated at the early stages of dengue virus infection. Similarly, metformin decreases the viral replication of Coxsackievirus B3 (CVB3) and protects mice from CVB3-induced myocarditis, thereby benefiting the survival rate of infected mice (51). In addition to RNA viral infection, metformin also has antiviral activity in DNA viral infections. Metformin treatment in vitro drastically inhibits viral gene expression and infectious virion production of Kaposi sarcoma herpesvirus (50). Metformin also inhibits the replication of hepatitis B virus (HBV) in primary human hepatocytes by repressing viral transcription-related genes, including LRH1, PPARα, and HNF4α. Meanwhile, a combination of metformin and entecavir inhibits HBV replication more significantly than either alone (52). These findings suggest that metformin might be used as a potential therapeutic agent for SARS-CoV-2 infection and other viral infections, especially in combination with other antiviral agents.
Furthermore, metformin may also inhibit SARS-CoV-2 infection by interfering with its interaction with ACE2 via the activation of AMPK. AMPK phosphorylates ACE2 Ser680 in human endothelial cells and increases ACE2 expression by enhancing its stability. Metformin also enhances the phosphorylation and expression of ACE2 (53). It is believed that phosphorylation will lead to conformational and functional changes in the ACE2 receptor and decrease the binding of SARS-CoV-2 (54). In addition, ACE2 plays an important role in anti-inflammation and antifibrosis (55). The entry of SARS-CoV-2 into cells by binding to ACE2 downregulates its expression and leads to an imbalance in the renin-angiotensin-aldosterone system (RAS), promoting proinflammatory and profibrotic effects. The imbalance in the RAS is likely averted through upregulation of ACE2 expression by metformin (55). Hence, metformin would not only prevent the entry of SARS-CoV-2 but also decrease its deleterious effects.
Two recent independent retrospective studies have revealed that metformin treatment tends to reduce the mortality of patients with COVID-19 with T2D or obesity (56, 57). One large study revealed that a reduction in mortality is only found in female patients and that sex-dependent survival is associated with a reduction in TNFα.
Side Effects of Metformin and Routes of Usage
Although metformin is widely used to treat T2D and has a good safety profile, some treated patients develop cutaneous and gastrointestinal (GI) side effects (58–61). Rare cutaneous side effects include leukocytoclastic vasculitis, bullous pemphigoid, lichen planus, and acute alopecia (58). GI side effects are the most frequent adverse effects (59–61) and occur more frequently in patients of older age and in women than in other patients. Approximately 25% of metformin-treated patients develop GI symptoms, including nausea, vomiting, diarrhea, bloating, and abdominal pain (59, 60). As a result, approximately 5% of cases have to discontinue metformin therapy due to intolerant side effects (59, 60). Intolerant patients are mainly older women (59). The GI side effects may result from complications of treatment with other drugs that reduce the function of organic cation transporter 1 (OCT1). In addition, certain OCT1 genotypes are linked to GI intolerance as well. Furthermore, the OCT1 genotypes and usage of OCT1-interacting drugs have a synergistic effect on GI intolerance (59).
In addition to cutaneous and GI side effects, metformin is rarely associated with other adverse effects, such as chest discomfort, heartburn, flatulence, weakness, myalgia, palpitation, flushing, headache, dyspnea, anemia, increased diaphoresis, and lactic acidosis (58). Both lactic acidosis and anemia are rare side effects. Lactic acidosis occurs in patients who have renal dysfunction. Anemia results from the reduction in vitamin B12 levels due to malabsorption (61).
Lowering the starting dose of oral administration with a gradual increase in dosage is recommended to minimize the GI side effects of patients with diabetes. Actually, oral administration is not necessary to treat other diseases. For example, topical administration has been shown to improve histological, clinical, and radiographic outcomes of chronic periodontitis in patients, and no adverse events occur (62). In addition, topical metformin appears to be a safe and effective treatment for melasma (63). Therefore, topical metformin may be suitable for treating inflammation associated with acute lung injury in COVID-19.
Conclusion and Discussion
Excessive inflammation is involved in the development of severe COVID-19. Inhibition of the inflammatory response might be a promising strategy to mitigate disease severity. Metformin has suppressive activities in the production of proinflammatory cytokines by activated macrophages, the formation of NETs, and the immune responses of pathogenic Th1 and Th17 cells. Studies in animal models have also demonstrated that metformin can alleviate various acute lung injuries. In addition, metformin may directly inhibit SARS-CoV-2 infection by targeting the interaction between human proteins and viral proteins. Furthermore, clinical studies have shown that metformin treatment is associated with a reduction of mortality in diabetic patients with severe COVID-19. Therefore, metformin is promising as a drug candidate to prevent or treat severe COVID-19. Notably, metformin occasionally has adverse side effects, including cutaneous and GI side effects and lactic acidosis. Routes of administration (for example, nebulization) and the synergistic effects with other treatments merit further investigation.
Author Contributions
QL conceived the presented idea. XC wrote the manuscript and prepared the figures. QL, HG, LQ, QD, and CZ revised the manuscript. All authors contributed to the article and approved the submitted version.
Funding
This work was supported by the National Key Project for Infectious Diseases of China (2018ZX10301-208), the Shiyan Science and Technology Bureau Guided Program, the start-up fund of Nanshan Scholarship of Guangzhou Medical University, and the Guangzhou Key Medical Discipline Construction Project Fund.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
1. Al-Tawfiq, JA, Zumla, A, Gautret, P, Gray, GC, Hui, DS, Al-Rabeeah, AA, et al. Surveillance for emerging respiratory viruses. Lancet Infect Dis. (2014) 14:992–1000. doi: 10.1016/S1473-3099(14)70840-0
2. Weber, DJ, Rutala, WA, Fischer, WA, Kanamori, H, Sickbert-Bennett, EE. Emerging infectious diseases: focus on infection control issues for novel coronaviruses (Severe Acute Respiratory Syndrome-CoV and Middle East Respiratory Syndrome-CoV), hemorrhagic fever viruses (Lassa and Ebola), and highly pathogenic avian influenza viruses, A(H5N1) and A(H7N9). Am J Infect Control. (2016) 44(5 Suppl.):e91–100. doi: 10.1016/j.ajic.2015.11.018
3. Chu, H, Chan, JF, Wang, Y, Yuen, TT, Chai, Y, Hou, Y, et al. Comparative replication and immune activation profiles of SARS-CoV-2 and SARS-CoV in human lungs: an ex vivo study with implications for the pathogenesis of COVID-19. Clin Infect Dis. (2020). (in press). doi: 10.1093/cid/ciaa410
4. Payne, B, Bellamy, R. Novel respiratory viruses: what should the clinician be alert for? Clin Med. (2014) 14(Suppl. 6):s12–6. doi: 10.7861/clinmedicine.14-6-s12
5. Han, J, Zhang, N, Zhang, P, Yang, C, Jin, M, Yang, J, et al. Th2-type inflammation under conditions of pre-existing chronic disease is associated with liver damage in patients with avian influenza H7N9 virus. Microbes Infect. (2014) 16:672–7. doi: 10.1016/j.micinf.2014.04.002
6. Yu, H, Cowling, BJ, Feng, L, Lau, EH, Liao, Q, Tsang, TK, et al. Human infection with avian influenza A H7N9 virus: an assessment of clinical severity. Lancet. (2013) 382:138–45. doi: 10.1016/S0140-6736(13)61207-6
7. Huang, C, Wang, Y, Li, X, Ren, L, Zhao, J, Hu, Y, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan. China. Lancet. (2020) 395:497–506. doi: 10.1016/S0140-6736(20)30183-5
8. Chen, N, Zhou, M, Dong, X, Qu, J, Gong, F, Han, Y, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. (2020) 395:507–13. doi: 10.1016/S0140-6736(20)30211-7
9. Jin, YH, Cai, L, Cheng, ZS, Cheng, H, Deng, T, Fan, YP, et al. Promotive association for and C. Health: a rapid advice guideline for the diagnosis and treatment of 2019 novel coronavirus. (2019-nCoV) infected pneumonia (standard version). Mil Med Res. (2020) 7:4. doi: 10.1186/s40779-020-0233-6
10. Wang, D, Hu, B, Hu, C, Zhu, F, Liu, X, Zhang, J, et al. Clinical characteristics of 138 hospitalized patients with 2019 novel Coronavirus-infected pneumonia in Wuhan, China. JAMA. (2020) 323:1061–9. doi: 10.1001/jama.2020.1585
11. Schett, G, Sticherling, M, Neurath, MF. COVID-19: risk for cytokine targeting in chronic inflammatory diseases? Nat Rev Immunol. (2020) 20:271–2. doi: 10.1038/s41577-020-0312-7
12. Yang, X, Yu, Y, Xu, J, Shu, H, Xia, J, Liu, H, et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. Lancet Respir Med. (2020) 8:475–81. doi: 10.1016/S2213-2600(20)30079-5
13. Xu, Z, Shi, L, Wang, Y. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med. (2020) 8:E26–26.
14. Chen, Y, Gut, F, Guan, JL. Metformin might inhibit virus through increasing insulin sensitivity. Chin Med J. (2018) 131:376–7. doi: 10.4103/0366-6999.223856
15. Rangarajan, S, Bone, NB, Zmijewska, AA, Jiang, SN, Park, DW, Bernard, K, et al. Metformin reverses established lung fibrosis in a bleomycin model. Nat Med. (2018) 24:1627–1627. doi: 10.1038/s41591-018-0170-z
16. Qi, F, Qian, S, Zhang, S, Zhang, Z. Single cell RNA sequencing of 13 human tissues identify cell types and receptors of human coronaviruses. Biochem Biophys Res Commun. (2020) 526:135–40. doi: 10.1016/j.bbrc.2020.03.044
17. Zou, X, Chen, K, Zou, J, Han, P, Hao, J, Han, Z. Single-cell RNA-seq data analysis on the receptor ACE2 expression reveals the potential risk of different human organs vulnerable to 2019-nCoV infection. Front Med. (2020) 14:185–92. doi: 10.1007/s11684-020-0754-0
18. Wang, Z, Xu, X. scRNA-seq profiling of human testes reveals the presence of the ACE2 receptor, a target for SARS-CoV-2 infection in spermatogonia, leydig and sertoli cells. Cells. (2020) 9:920. doi: 10.3390/cells9040920
19. Chen, T, Wu, D, Chen, H, Yan, W, Yang, D, Chen, G, et al. Clinical characteristics of 113 deceased patients with coronavirus disease 2019: retrospective study. BMJ. (2020) 368:m1091. doi: 10.1136/bmj.m1091
20. Li, H, Liu, L, Zhang, D, Xu, J, Dai, H, Tang, N, et al. SARS-CoV-2 and viral sepsis: observations and hypotheses. Lancet. (2020) 395:1517–20. doi: 10.1016/S0140-6736(20)30920-X
21. Chen, G, Wu, D, Guo, W, Cao, Y, Huang, D, Wang, H, et al. Clinical and immunological features of severe and moderate coronavirus disease 2019. J Clin Invest. (2020) 130:2620–9. doi: 10.1172/JCI137244
22. Ruan, Q, Yang, K, Wang, W, Jiang, L, Song, J. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Med. (2020) 46:846–8. doi: 10.1007/s00134-020-05991-x
23. Moore, BJB, June, CH. Cytokine release syndrome in severe COVID-19. Science. (2020) 368:473–4. doi: 10.1126/science.abb8925
24. Fu, B, Xu, X, Wei, H. Why tocilizumab could be an effective treatment for severe COVID-19? J Transl Med. (2020) 18:164. doi: 10.1186/s12967-020-02339-3
25. Chen, Y, Feng, Z, Diao, B, Wang, R, Wang, G, Wang, C, et al. The novel severe acute respiratory syndrome Coronavirus 2 (SARS-CoV-2) directly decimates human spleens and lymph nodes. medRxiv [Preprint]. (2020). doi: 10.1101/2020.03.27.20045427
26. Barnes, BJ, Adrover, JM, Baxter-Stoltzfus, A, Borczuk, A, Cools-Lartigue, J, Crawford, JM, et al. Targeting potential drivers of COVID-19: neutrophil extracellular traps. J Exp Med. (2020) 217:e20200652. doi: 10.1084/jem.20200652
27. Liu, J, Liu, Y, Xiang, P, Pu, L, Xiong, H, Li, C, et al. Neutrophil-to-lymphocyte ratio predicts severe illness patients with 2019 novel Coronavirus in the early stage. medRxiv [Preprint]. (2020). doi: 10.1101/2020.02.10.20021584
28. Liao, M, Liu, Y, Yuan, J, Wen, Y, Xu, G, Zhao, J, et al. The landscape of lung bronchoalveolar immune cells in COVID-19 revealed by single-cell RNA sequencing. medRxiv [Preprint]. (2020). doi: 10.1101/2020.02.23.20026690
29. Kindler, E, Thiel, V. SARS-CoV and IFN: too little, too late. Cell Host Microbe. (2016) 19:139–41. doi: 10.1016/j.chom.2016.01.012
30. Zhou, F, Yu, T, Du, R, Fan, G, Liu, Y, Liu, Z, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. (2020) 395:1054–62. doi: 10.1016/S0140-6736(20)30566-3
31. Foretz, M, Guigas, B, Bertrand, L, Pollak, M, Viollet, B. Metformin: from mechanisms of action to therapies. Cell Metab. (2014) 20:953–66. doi: 10.1016/j.cmet.2014.09.018
32. Wang, YW, He, SJ, Feng, X, Cheng, J, Luo, YT, Tian, L, et al. Metformin: a review of its potential indications. Drug Des Devel Ther. (2017) 11:2421–9. doi: 10.2147/DDDT.S141675
33. Ursini, F, Russo, E, Pellino, G, D’Angelo, S, Chiaravalloti, A, De Sarro, G, et al. Metformin and autoimmunity: a “New Deal” of an old drug. Front Immunol. (2018) 9:1236. doi: 10.3389/fimmu.2018.01236
34. Matsushita, M, Kawaguchi, M. Immunomodulatory effects of drugs for effective cancer immunotherapy. J Oncol. (2018) 2018:8653489. doi: 10.1155/2018/8653489
35. Zhou, J, Massey, S, Story, D, Li, L. Metformin: an old drug with new applications. Int J Mol Sci. (2018) 19:2863. doi: 10.3390/ijms19102863
36. Wang, M, Qu, S, Ma, J, Wang, X, Yang, Y. Metformin suppresses LPS-induced inflammatory responses in macrophage and ameliorates allergic contact dermatitis in mice via autophagy. Biol Pharm Bull. (2020) 43:129–37. doi: 10.1248/bpb.b19-00689
37. Kim, J, Kwak, HJ, Cha, JY, Jeong, YS, Rhee, SD, Kim, KR, et al. Metformin suppresses lipopolysaccharide (LPS)-induced inflammatory response in murine macrophages via activating transcription factor-3 (ATF-3) induction. J Biol Chem. (2014) 289:23246–55. doi: 10.1074/jbc.M114.577908
38. Kelly, B, Tannahill, GM, Murphy, MP, O’Neill, LA. Metformin inhibits the production of reactive oxygen species from NADH:ubiquinone oxidoreductase to limit induction of interleukin-1beta (IL-1beta) and boosts interleukin-10 (IL-10) in lipopolysaccharide (LPS)-activated macrophages. J Biol Chem. (2015) 290:20348–59. doi: 10.1074/jbc.M115.662114
39. Menegazzo, L, Scattolini, V, Cappellari, R, Bonora, BM, Albiero, M, Bortolozzi, M, et al. The antidiabetic drug metformin blunts NETosis in vitro and reduces circulating NETosis biomarkers in vivo. Acta Diabetol. (2018) 55:593–601. doi: 10.1007/s00592-018-1129-8
40. Carestia, A, Frechtel, G, Cerrone, G, Linari, MA, Gonzalez, CD, Casais, P, et al. NETosis before and after hyperglycemic control in Type 2 diabetes mellitus patients. PLoS One. (2016) 11:e0168647. doi: 10.1371/journal.pone.0168647
41. Nath, N, Khan, M, Paintlia, MK, Singh, I, Hoda, MN, Giri, S. Metformin attenuated the autoimmune disease of the central nervous system in animal models of multiple sclerosis. J Immunol. (2009) 182:8005–14. doi: 10.4049/jimmunol.0803563
42. Zhang, X, Shang, F, Hui, L, Zang, K, Sun, G. The alleviative effects of metformin for lipopolysaccharide-induced acute lung injury rat model and its underlying mechanism. Saudi Pharm J. (2017) 25:666–70. doi: 10.1016/j.jsps.2017.05.001
43. Wu, K, Tian, R, Huang, J, Yang, Y, Dai, J, Jiang, R, et al. Metformin alleviated endotoxemia-induced acute lung injury via restoring AMPK-dependent suppression of mTOR. Chem Biol Interact. (2018) 291:1–6. doi: 10.1016/j.cbi.2018.05.018
44. Wu, L, Cen, Y, Feng, M, Zhou, Y, Tang, H, Liao, X, et al. Metformin activates the protective effects of the AMPK pathway in acute lung injury caused by paraquat poisoning. Oxid Med Cell Longev. (2019) 2019:1709718. doi: 10.1155/2019/1709718
45. Zmijewski, JW, Lorne, E, Zhao, X, Tsuruta, Y, Sha, Y, Liu, G, et al. Mitochondrial respiratory complex I regulates neutrophil activation and severity of lung injury. Am J Respir Crit Care Med. (2008) 178:168–79. doi: 10.1164/rccm.200710-1602OC
46. Tsaknis, G, Siempos, II, Kopterides, P, Maniatis, NA, Magkou, C, Kardara, M, et al. Metformin attenuates ventilator-induced lung injury. Crit Care. (2012) 16:R134. doi: 10.1186/cc11439
47. Rangarajan, S, Bone, NB, Zmijewska, AA, Jiang, S, Park, DW, Bernard, K, et al. Metformin reverses established lung fibrosis in a bleomycin model. Nat Med. (2018) 24:1121–7. doi: 10.1038/s41591-018-0087-6
48. Gordon, DE, Jang, GM, Bouhaddou, M, Xu, J, Obernier, K, O’Meara, MJ, et al. A SARS-CoV-2-human protein-protein interaction map reveals drug targets and potential drug-repurposing. BioRxiv [Preprint]. (2020). doi: 10.1101/2020.03.22.002386
49. Soto-Acosta, R, Bautista-Carbajal, P, Cervantes-Salazar, M, Angel-Ambrocio, AH, Del Angel, RM. DENV up-regulates the HMG-CoA reductase activity through the impairment of AMPK phosphorylation: a potential antiviral target. PLoS Pathog. (2017) 13:e1006257. doi: 10.1371/journal.ppat.1006257
50. Cheng, F, He, M, Jung, JU, Lu, C, Gao, SJ. Suppression of Kaposi’s sarcoma-associated herpesvirus infection and replication by 5’-AMP-activated protein kinase. J Virol. (2016) 90:6515–25. doi: 10.1128/JVI.00624-16
51. Xie, W, Wang, L, Dai, Q, Yu, H, He, X, Xiong, J, et al. Activation of AMPK restricts coxsackievirus B3 replication by inhibiting lipid accumulation. J Mol Cell Cardiol. (2015) 85:155–67. doi: 10.1016/j.yjmcc.2015.05.021
52. Honda, M, Shirasaki, T, Terashima, T, Kawaguchi, K, Nakamura, M, Oishi, N, et al. Hepatitis B virus (HBV) core-related antigen during nucleos(t)ide analog therapy is related to intra-hepatic HBV replication and development of hepatocellular carcinoma. J Infect Dis. (2016) 213:1096–106. doi: 10.1093/infdis/jiv572
53. Zhang, J, Dong, J, Martin, M, He, M, Gongol, B, Marin, TL, et al. AMP-activated protein kinase phosphorylation of angiotensin-converting enzyme 2 in endothelium mitigates pulmonary hypertension. Am J Respir Crit Care Med. (2018) 198:509–20. doi: 10.1164/rccm.201712-2570OC
54. Sharma, S, Ray, A, Sadasivam, B. Metformin in COVID-19: a possible role beyond diabetes. Diabetes Res Clin Pract. (2020) 164:108183. doi: 10.1016/j.diabres.2020.108183
55. Simoes e Silva, AC, Silveira, KD, Ferreira, AJ, Teixeira, MM. ACE2, angiotensin-(1-7) and Mas receptor axis in inflammation and fibrosis. Br J Pharmacol. (2013) 169:477–92. doi: 10.1111/bph.12159
56. Bramante, C, Ingraham, N, Murray, T, Marmor, S, Hoversten, S, Gronski, J, et al. Observational study of metformin and risk of mortality in patients hospitalized with covid-19. medRxiv [Preprint]. (2020). doi: 10.1101/2020.06.19.20135095
57. Luo, P, Qiu, L, Liu, Y, Liu, XL, Zheng, JL, Xue, HY, et al. Metformin treatment was associated with decreased mortality in COVID-19 patients with diabetes in a retrospective analysis. Am J Trop Med Hyg. (2020) 103:69–72. doi: 10.4269/ajtmh.20-0375
58. Badr, D, Kurban, M, Abbas, O. Metformin in dermatology: an overview. J Eur Acad Dermatol Venereol. (2013) 27:1329–35. doi: 10.1111/jdv.12116
59. Dujic, T, Zhou, K, Donnelly, LA, Tavendale, R, Palmer, CN, Pearson, ER. Association of organic cation transporter 1 with intolerance to metformin in Type 2 diabetes: a GoDARTS study. Diabetes. (2015) 64:1786–93. doi: 10.2337/db14-1388
60. McCreight, LJ, Bailey, CJ, Pearson, ER. Metformin and the gastrointestinal tract. Diabetologia. (2016) 59:426–35. doi: 10.1007/s00125-015-3844-9
61. Pawlyk, AC, Giacomini, KM, McKeon, C, Shuldiner, AR, Florez, JC. Metformin pharmacogenomics: current status and future directions. Diabetes. (2014) 63:2590–9. doi: 10.2337/db13-1367
62. Nicolini, AC, Grisa, TA, Muniz, F, Rosing, CK, Cavagni, J. Effect of adjuvant use of metformin on periodontal treatment: a systematic review and meta-analysis. Clin Oral Investig. (2019) 23:2659–66. doi: 10.1007/s00784-018-2666-9
Keywords: coronavirus disease 2019, SARS-CoV-2, cytokine storm, metformin, antiviral activity
Citation: Chen X, Guo H, Qiu L, Zhang C, Deng Q and Leng Q (2020) Immunomodulatory and Antiviral Activity of Metformin and Its Potential Implications in Treating Coronavirus Disease 2019 and Lung Injury. Front. Immunol. 11:2056. doi: 10.3389/fimmu.2020.02056
Received: 29 May 2020; Accepted: 28 July 2020;
Published: 18 August 2020.
Edited by:
Uwe Ritter, University of Regensburg, GermanyReviewed by:
Jochen Mattner, University of Erlangen-Nuremberg, GermanyBlanca Cárdaba, Health Research Institute Foundation Jimenez Diaz (IIS-FJD), Spain
Copyright © 2020 Chen, Guo, Qiu, Zhang, Deng and Leng. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Qibin Leng, cWJsZW5nQHNpYnMuYWMuY24=
 Xianyang Chen1
Xianyang Chen1 Li Qiu
Li Qiu Chengdong Zhang
Chengdong Zhang Qibin Leng
Qibin Leng