- 1Program in Cell Biology, Peter Gilgan Centre for Research and Learning, Hospital for Sick Children, Toronto, ON, Canada
- 2Department of Biochemistry, University of Toronto, Toronto, ON, Canada
Tissue-resident phagocytes are responsible for the routine binding, engulfment, and resolution of their meals. Such populations of cells express appropriate surface receptors that are tailored to recognize the phagocytic targets of their niche and initiate the actin polymerization that drives internalization. Tissue-resident phagocytes also harbor enzymes and transporters along the endocytic pathway that orchestrate the resolution of ingested macromolecules from the phagolysosome. Solutes fluxed from the endocytic pathway and into the cytosol can then be reutilized by the phagocyte or exported for their use by neighboring cells. Such a fundamental metabolic coupling between resident phagocytes and the tissue in which they reside is well-emphasized in the case of retinal pigment epithelial (RPE) cells; specialized phagocytes that are responsible for the turnover of photoreceptor outer segments (POS). Photoreceptors are prone to photo-oxidative damage and their long-term health depends enormously on the disposal of aged portions of the outer segment. The phagocytosis of the POS by the RPE is the sole means of this turnover and clearance. RPE are themselves mitotically quiescent and therefore must resolve the ingested material to prevent their toxic accumulation in the lysosome that otherwise leads to retinal disorders. Here we describe the sequence of events underlying the healthy turnover of photoreceptors by the RPE with an emphasis on the signaling that ensures the phagocytosis of the distal POS and on the transport of solutes from the phagosome that supersedes its resolution. While other systems may utilize different receptors and transporters, the biophysical and metabolic manifestations of such events are expected to apply to all tissue-resident phagocytes that perform regular phagocytic programs.
Introduction
Phagocytosis, the ingestion of large (>0.5 um) particles, is an evolutionarily conserved, actin-driven process with roles in nutrient acquisition, immunity, and tissue homeostasis (1–3). The ongoing, routine phagocytic programs that maintain tissue homeostasis in the absence of infection or injury are largely performed by tissue-resident phagocytes. These cells are non-migratory and are therefore strategically positioned for phagocytic encounters. In some cases, resident phagocytes are uniformly distributed to optimally survey the whole tissue by probing the space between them (4, 5). In other cases, they are localized to particular regions that favor their collection and engulfment of phagocytic loads [e.g. splenic red pulp macrophages that turnover red blood cells (6) or bone marrow-resident macrophages that remove nuclei extruded during erythropoiesis (7, 8)]. In all cases, the efficient removal of dead cells, parts of cells, and debris by these phagocytes is essential to prevent secondary necrosis, inflammation, and autoimmunity (9, 10).
Within the category of tissue resident phagocytes are “specialized phagocytes”; epithelial derived stromal cells including the retinal pigment epithelium (RPE) of the eye and Sertoli cells of the testes (10, 11). As epithelia, these cells contribute to the formation of blood-tissue barriers while facilitating the directional transport of oxygen, glucose, cholesterol, etc. to the tissue from circulation. As specialized phagocytes, these cells also actively turnover theirs neighbors. Perhaps the best studied example of specialized phagocytes is indeed the RPE that intimately associates with photoreceptors and mediates their turnover (12). A single RPE cell is in contact with ~30 photoreceptor cells (rods and cones) and is responsible for the phagocytosis and removal of the distal portions of the photoreceptor outer segments (POS) that are phagocytosed in a diurnal fashion (13, 14) (Figure 1). The removed mass is not trivial; 7–10% is eliminated daily meaning the entire POS population is turned over every 2 weeks (15). Remarkably, a burst of phagocytosis is timed with the entry of light to the retina and, within hours, the phagosomes formed in each healthy RPE cell are resolved (14, 16).
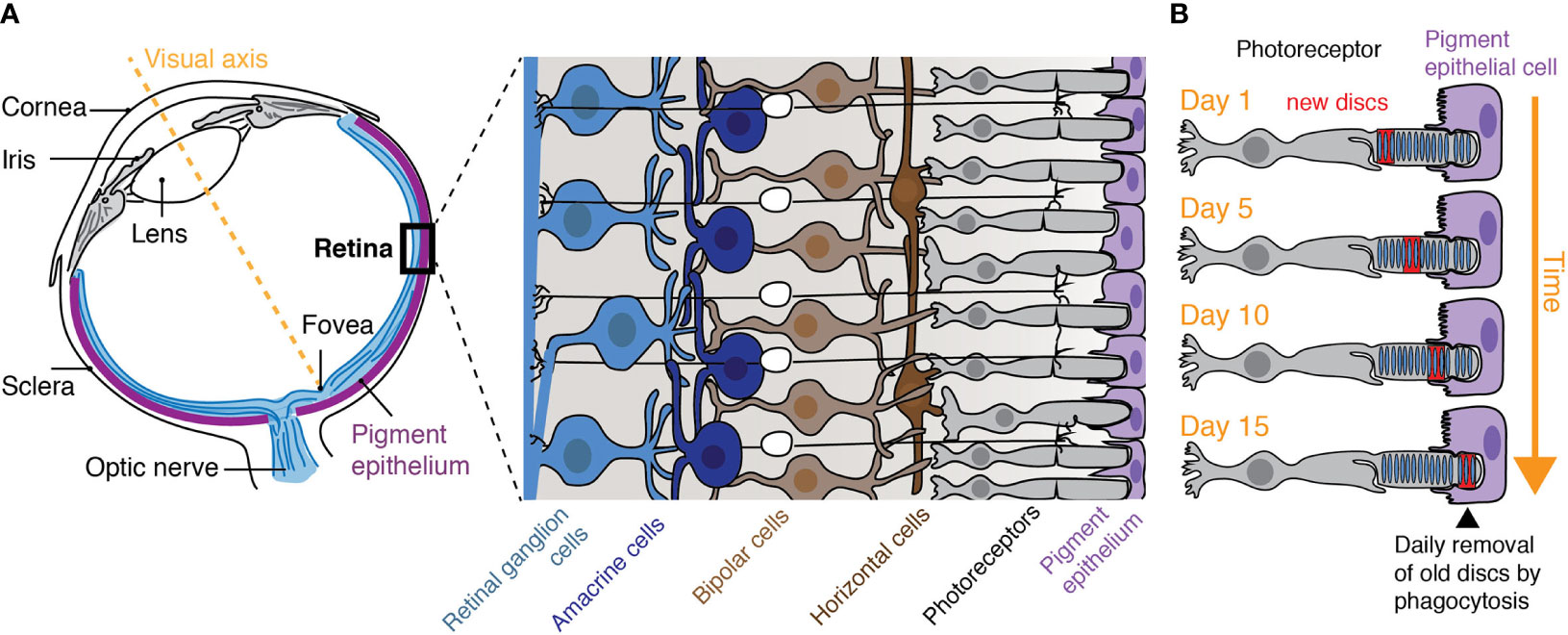
Figure 1 The retinal pigment epithelium. (A) The retina lines the back of the eye and is comprised of five cell types: rod and cone photoreceptors, bipolar, horizontal, amacrine, and retinal ganglion cells. (B) The retinal pigment epithelium that underlies the retina is responsible for the turnover of photoreceptor outer segments (POS) by their phagocytosis and is critical for photoreceptor function. Photoreceptors are choc-a-bloc with membrane discs that harbor opsins which are susceptible to phototoxic damage. The most distal segments of the POS that contain the oldest discs are therefore removed from the live photoreceptors while new discs are made at the beginning of the outer segment. Note: as new discs are formed at the base of the POS, old discs are removed by phagocytosis, a process that takes approximately 2 weeks.
Expectedly, the removal of the aged regions of the POS and of metabolic wastes by the RPE is essential for the homeostasis of the retina (17, 18). Just as critically, the ingested material by the RPE needs to be resolved otherwise it accumulates and forms lipofuscin (comprised of oxidized proteins and lipids), eventually leading to retinal disorders (19). Lipofuscin also accumulates in the eye with healthy aging (20, 21), so an understanding of when and how this causes disease is fundamental. RPE cells are themselves post-mitotic and therefore resolution of the vacuoles formed during phagocytosis is integral to RPE longevity. The resolution process can be envisaged as a sequence of steps that includes the maturation of the nascent phagosome, the digestion of luminal macromolecules, the efflux of solutes from the phagolysosomes, and the resorption of the phagosomal membrane. Interestingly, the building blocks from the ingested photoreceptor outer segments (POS) are thought to be shuttled back to the photoreceptor cells (22), supporting their continuous regeneration and completing a “heterocellular metabolic circuit.” Importantly, in the RPE-photoreceptor relationship, it is appreciated that dysfunction in one cell type leads to degeneration in the other. Such metabolic coupling and the sequence of events that complete these circuits, while recognized in the case of the RPE and photoreceptors, are poorly understood for other tissue-resident phagocytes (23).
Here we describe the phagocytosis, breakdown, and resolution of ingested POS by RPE cells. We first illustrate phagocytosis by the RPE including the binding, ensheathment, and ingestion of the POS driven by the RPE actin cytoskeleton. We subsequently describe the maturation steps of the phagosome that confer on the vacuole its degradative capacity. The eventual efflux of solutes from the phagolysosome is discussed in some detail and we describe how these fluxes are expected to lead to the remodeling of the phagosomal membrane including its vesiculation and tubulation. Finally, we propose that the delivery of building blocks back to the tissue microenvironment supports the health of neighboring cells and we speculate on how such events may occur.
Photoreceptors and the Retinal Pigment Epithelium: Close Encounters
Light that enters the vertebrate eye is focused onto the neural retina, a thin tissue (∼200 µm) comprised of five main classes of cells including photoreceptors, bipolar cells, amacrine cells, horizontal cells, and ganglion cells (Figure 1). These cells work concertedly to process and transmit visual information to the midbrain, thalamus, and visual cortex via the optic nerve (24). Despite their posterior positioning (e.g. at the back layer of the retina and the region farthest from incoming light), it is with the photoreceptor cells where light is captured and the visual pathway is initiated. The light is entirely absorbed in outer segments of the photoreceptor. The POS consists of membranous discs packed with integral membrane proteins called opsins. Opsins are made to be light-sensitive by their covalent association with the chromophore retinal (e.g. rhodopsin in the case of rods) (25) and are perhaps the best studied of the G-protein coupled receptors. Upon light exposure, rhodopsin undergoes immediate bleaching which triggers the visual transduction pathway. Over time, and with repetitive bleaching and regeneration of rhodopsin, phototoxic damage can occur in the neighboring proteins and lipids of the discs (26). To circumvent the accumulation of damaged components, photoreceptors undergo continuous turnover facilitated by the synthesis and assembly of new discs at the base of the outer segment and the simultaneous shedding of the oldest discs from the growing tips of the POS (Figure 1). This mechanism results in photoreceptors that are long-lived and discs of the POS that are short-lived (~2 weeks).
At the tip of the POS facing the RPE, is the marked accumulation of phosphatidylserine (PtdSer) exposed on the outer leaflet of the cell which becomes much more pronounced with light onset (16) (Figure 2). In virtually all other healthy cell types, the asymmetric distribution of PtdSer to the inner leaflet is tightly maintained by the ongoing activity of phospholipid translocases or “flippases” including ATP11A and ATP11C (27–30). Only under apoptotic conditions are these flippases cleaved and inactivated by caspases. The same caspases also cleave and activate scramblases, which can alternatively be activated by Ca2+, that begin to randomly flip membrane phospholipids like PtdSer between the two leaflets (31–34). Given the tight control over the asymmetric distribution of PtdSer in most cells, the polarized and relatively sustained exposure of the phospholipid in the non-apoptotic photoreceptor is a unique phenomenon. It must require 1) the very local disruption of flippases/activation of scramblases and 2) a barrier to the free diffusion of exofacial PtdSer from the distal POS tip.
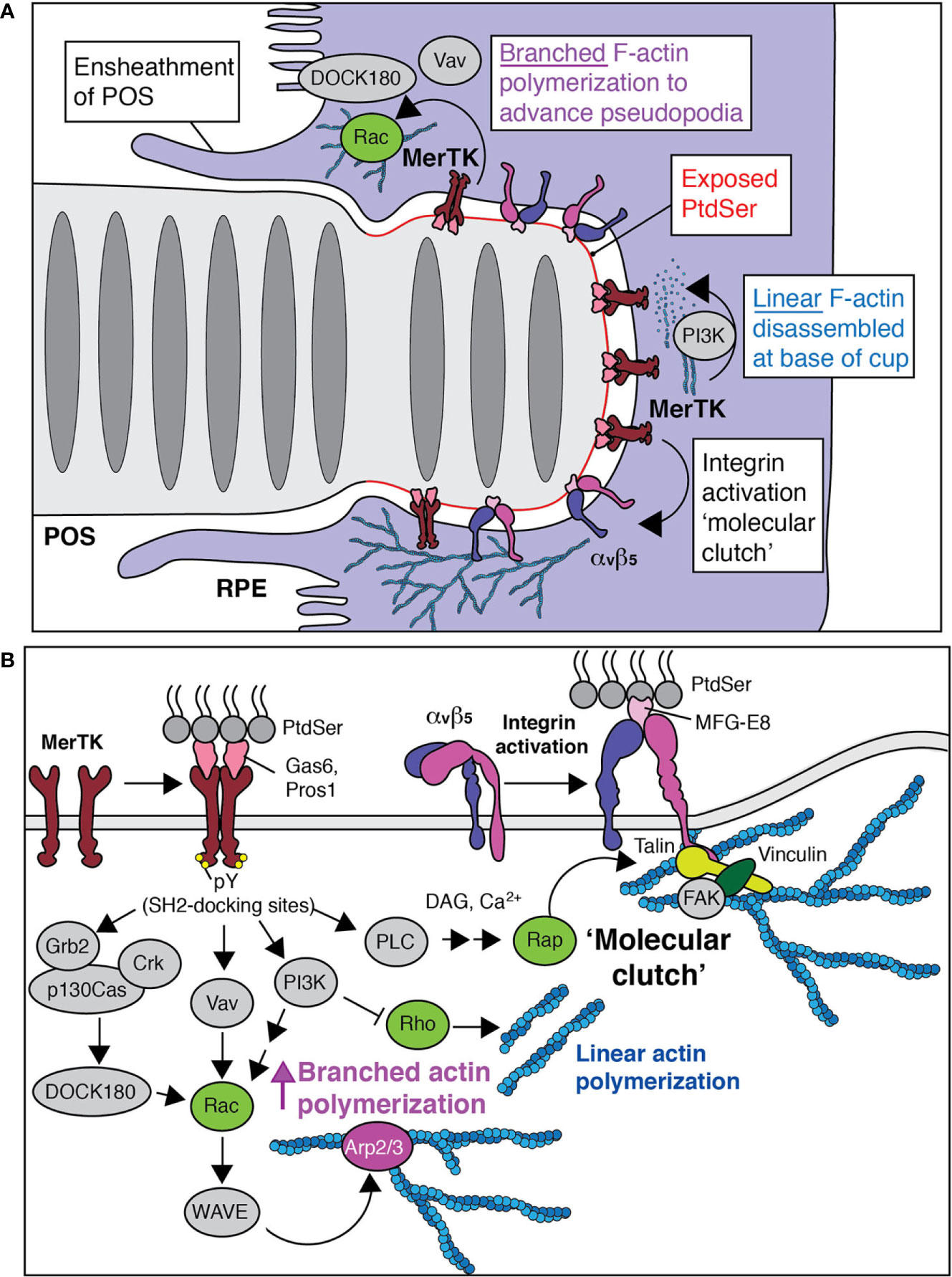
Figure 2 Phagocytosis by the retinal pigment epithelium. (A) Like all types of phagocytosis, the engulfment of POS by the RPE is driven by actin polymerization. PtdSer is first exposed and tethered by bridging molecules that bind the distal portion of the POS and MerTK receptors. To relieve membrane tension, linear networks are removed by the activation of PI3K. Branched networks are stimulated by nucleation promoting factors that bring actin monomers to the Arp2/3 complex. Both activities are mediated by MerTK (see also panel B). (B) MerTK initiates a series of signaling pathways that stimulate branched actin polymerization, networks that attach to the membrane via integrins. The major RPE integrin is αvβ5, which also binds to PtdSer but via MFG-E8. As actin advances the pseudopodia tips of the RPE and facilitates severing of the POS, clearance of actin from the base of the cup allows for the delivery of new membrane, a PI3K-dependent process.
The latter may be achieved by secreted molecules that bridge and tether the PtdSer to RPE receptors. Plenty of bridging molecules may participate in trapping the exposed PtdSer including Gas6, Protein S, Tubby, and Tubby-like protein 1 which connect PtdSer to the RPE-expressed MerTK receptor (35–38) and MFG-E8 which does the same for αvβ5 integrins (39). Given that exposed PtdSer remains polarized to the distal tips of POS in Mfge8-/- and Itgb5-/- (β5 integrin) mice (16), there may be functional redundancy to this effect or the MerTK ligands may be the essential factors. Here, at least Gas6 and Protein S are provided to the interphotoreceptor matrix by their synthesis and release from the RPEs themselves (35, 37, 40). At least some of these bridging molecules were recently shown to be transcribed by the RPE in a circadian fashion (41). Fittingly, experiments challenging RPEs with POS in vitro are often performed by augmenting the culture with exogenous Gas6 and/or Protein S (42) suggesting their requirement and some mechanism(s) of regulation to their expression by light.
How the local inactivation or exclusion of flippases and activation of scramblases at the distal tip of the POS occurs and is regulated in a diurnal fashion, on the other hand, remains entirely unclear. Notably, scramblases of the anoctamin/TMEM16 family show a polarized localization in other cell types (43) and their activation causes membrane expansion and shedding (44). A role for local Ca2+ flux and caspase activation has also been suggested (27).
Photoreceptors and the Retinal Pigment Epithelium: Phagocytosis
The association between the POS of the photoreceptor and the RPEs is constant: The apical microvilli of the RPE probe and become elaborated to “ensheath” the outer segments as they accumulate PtdSer, long before their phagocytosis (Figure 2) (45–47). Such membrane projections from the RPE can reach nearly half-way up the POS (up to ~15 µm), yet little is known about the cytoskeleton and the associated membrane remodeling that supports these structures. It is unlikely to be mediated by the features shared between the RPE microvilli and those found in other epithelial cells like ezrin/radixin/moesin (ERM) proteins (45, 48) and ERM-binding proteins including NHERF1 (49), which tend to oppose broad ruffling events (50, 51). Other features of the RPE apical membrane including the Na+/K+-ATPase pump, while also unique, are unlikely to directly drive ensheathment.
Instead, it is known that the ensheathment of POS requires the engagement of apically targeted MerTK, a member of the Tyro/Axl/Mer family of receptor tyrosine kinases (52, 53). Once MerTK is engaged by Gas6 or Protein S, ligand binding induces its multimerization and trans-autophosphorylation by the kinase domain. This leads to the recruitment of a number of adaptors and effectors that facilitate pseudopod extension (Figure 2). For example, the phosphorylated tyrosines in the cytosolic aspect of the receptor serve as docking sites for Vav, a potent GEF for Rac GTPases (54). The ensheathment structures indeed resemble those of spontaneous ruffles that are observed in macrophages (55) or that can be stimulated by pathogens (56) in a manner dependent on the activation of Rac GTPases. MerTK also stimulates Rac activation by recruiting the p130Cas/CrkII/Dock180 GEF complex (57). Rac-GTP can then bind and activate the WASP-family verprolin-homologous protein (WAVE) regulatory complex (WRC), (58, 59) which functions as a nucleation promoting factor for Arp2/3-branching of the F-actin cytoskeleton. Though less appreciated in recent literature, F-actin branching has long been known to also be mediated by flexible dimers of filamins including filA (60). No matter how they are formed, heavily branched networks of F-actin are indeed associated with broad membrane ruffling events akin to those of ensheathing RPEs (61). That Rac activation is associated with ensheathment is supported by data demonstrating Rac is indeed essential for the phagocytosis of POS (62).
MerTK also recruits and activates PI3K which in turn phosphorylates PtdIns(4,5)P2 to generate PtdIns(3,4,5)P3 a phosphoinositide that can subsequently be converted to PtdIns(3,4)P2 (63). PtdIns(3,4,5)P3 and potentially PtdIns(3,4)P2 support Rac activation by recruiting Rac GEFs and also dismantle linear actin networks by recruiting Rho specific GAPs (64) (Figure 2). The latter is critical for removing the submembrane highly bundled actin cables of the cortex that allows for the focal delivery of membrane from the endocytic pathway. Collectively, these actions relieve membrane tension that opposes membrane ruffling (65–68) and helps to explain why PI3 kinases are critical for the phagocytosis of large particles but not small ones (69, 70). While the phagosomes formed by the RPE are only 1-2 µm in diameter, a single RPE needs to engulf ~30 targets at once—a number that can in fact be much higher in the center of the retina—while maintaining junctions with its neighbors. Given the amount of new membrane delivered and/or the unfurling of the microvillar membrane that would require substantive F-actin disassembly for ensheathment, it makes sense that PI3K activation is indeed a prerequisite for synchronized POS phagocytosis and that its inhibition leads to increased F-actin at aborted phagocytic cups (71). That Rho inactivation is central to the process is supported by experiments demonstrating decreased phagocytosis when Rho kinase is made to be constitutively active (72).
While branched networks of F-actin propel and advance broad regions of the plasma membrane, they must be stabilized and connected to adhered transmembrane proteins to prevent the retrograde flow and collapse of the entire effort (Figure 2). Such points of stability are described as “molecular clutches” that limit the slipping of the branched F-actin networks (73) and yield efficiency to phagocytosis in many contexts (74, 75). A molecular clutch in the RPE membrane could be facilitated by the binding of the RPE cadherins to the neural cell adhesion molecule (N-CAM) expressed in the POS (76). Interestingly, aged whole animal KO Ncam-/- mice have thinned photoreceptor cell layers and premature vision-loss (77). A specific role for N-CAM in the RPE has not been identified but is well-expressed in these cells. Another obvious molecular clutch for the ruffling RPEs is the major RPE integrin, αvβ5 which is targeted apically. A role for the integrin here would explain why ensheathment is not stimulated by peppering the RPEs with MFG-E8 alone (45) but that the efficient, diurnal removal of the portions of the POS indeed requires the integrin and its ligand (16, 78).
Crosstalk Between MerTK and αvβ5
The MerTK and αvβ5 pathways are not mutually exclusive (Figure 2) (57). Like other integrins, αvβ5 is thought to exist primarily in a “bent” or closed conformation with low affinity for ligand (79). Its inside-out activation is triggered by receptor tyrosine kinases like MerTK. Once MerTK is engaged, a number of SH2-containing proteins are then recruited to the phosphorylated tyrosine residues including the adaptors Vav, Crk, and Grb2 as well as the phospholipase C gamma (PLCγ) (80–82) (Figure 2). Importantly, these multi-molecular assemblies favor the recruitment and activation of Rap GTPases which in turn recruit the proteins necessary to disrupt the inactive conformation of the integrin including the Rap1-GTP-interacting adapter molecule (RIAM) and Talin (83) leading to its inside-out activation. Talin, once bound to the β5 subunit of the integrin heterodimer, provides binding sites for F-actin and F-actin-binding proteins like vinculin.
Such an effect is critical in the case of MerTK which itself does not bind F-actin directly or even, to our knowledge, indirectly. Since integrins provide the mechanical linkage between actin polymerization to the ruffling/ensheathing membrane to yield traction, it makes sense that their activation by primary phagocytic receptors is observed in many types of phagocytosis (84). In this regard, it is noteworthy that MerTK-mediated phagocytosis involves focal adhesion kinase (FAK) (85), regulation of FAK activity (62), and the cleavage of PtdIns(4,5)P2 to generate Ins3P/diacylglycerol (DAG), which are normally associated with integrin and Rap activation respectively (86). Still, and as previously described, other ancillary transmembrane clutches are possible. Just as integrins can augment MerTK initiated phagocytosis, other receptors also participate including Tyro3 of the Tyro/Axl/Mer family of receptor tyrosine kinases (87) and the scavenger receptor CD36 (88). The relative contribution of Tyro3 and CD36 is still unclear and MerTK remains the canonical phagocytic receptor of the RPE; only the ablation of MerTK in the RPE leads to severe photoreceptor degeneration.
Sealing of the Nascent RPE Phagosome
The final steps of phagocytosis—sealing of the nascent phagosome by fusion of the pseudopodia tips—is the least understood step in all instances of engulfment and is especially poorly understood in the case of the RPE. This is remarkable since the RPE must apply a sufficient amount of local force on the POS to deform and generate scission through two lipid bilayers of an intact, live cell. The nascent phagosomes formed by RPE cells are indeed regular in their size (1–2 µm in diameter) as observed by electron (18) and light microscopy (89), as would be expected given the regular turnover of the POS, so the steps leading to scission must also be regulated. The site of eventual scission may be demarcated on the POS by the polarized distribution of PtdSer, which expands just before the scission event (16). However, the final, inward constricting force to complete phagocytosis remains enigmatic. In other systems, this has been attributed to the dynamin machinery employed in generic forms of endocytosis (90) and this could also be the case for the RPE. Alternatively, or in addition to dynamin, myosins may provide a “purse-string” constricting force as has been observed during the phagocytosis of targets by macrophages (91). Impressively, such a constricting force is sufficient to deform the skeleton of red cells (91) and to remove parts of live cells during ‘trogocytosis” (92). Indeed, MerTK activation in the RPE leads to the marked recruitment of Myosin II-A and Myosin II-B to the phagocytic cup, and the pharmacological or genetic ablation of Myosin II partially prevents POS engulfment in vitro (93). However, like the majority of the systems used to investigate RPE phagocytosis, here the targets were delivered in a pre-severed state. Clearly, further studies are necessary to visualize these events and to determine the mechanism(s) involved.
Phagosome Maturation
Following its scission from the plasma membrane, the nascent RPE phagosome undergoes graded fusion with endosomes and then lysosomes in a complex process termed “maturation” (Figure 3). Maturation can be envisaged as a series of steps marked by changes in the signaling phosphoinositides found within the limiting membrane of the phagosome and changes in its association with Rab GTPases in particular (12, 94). Importantly, maturation results in the acquisition of the V-ATPase and the delivery of enzymes from lysosomes that grant the vacuole its degradative properties. Maturation also coincides with—and is highly dependent on—the motor-driven movement of the phagosomes toward the basolateral side of the epithelial cell where they collect in a juxtanuclear location (95, 96). While the movement of the POS-containing phagosome through the cytosol differs markedly from the phagosomes formed in myeloid cells, the maturation process otherwise shares many of its salient molecular features. In this section, we therefore describe maturation in general terms and highlight what is known in the RPE system.
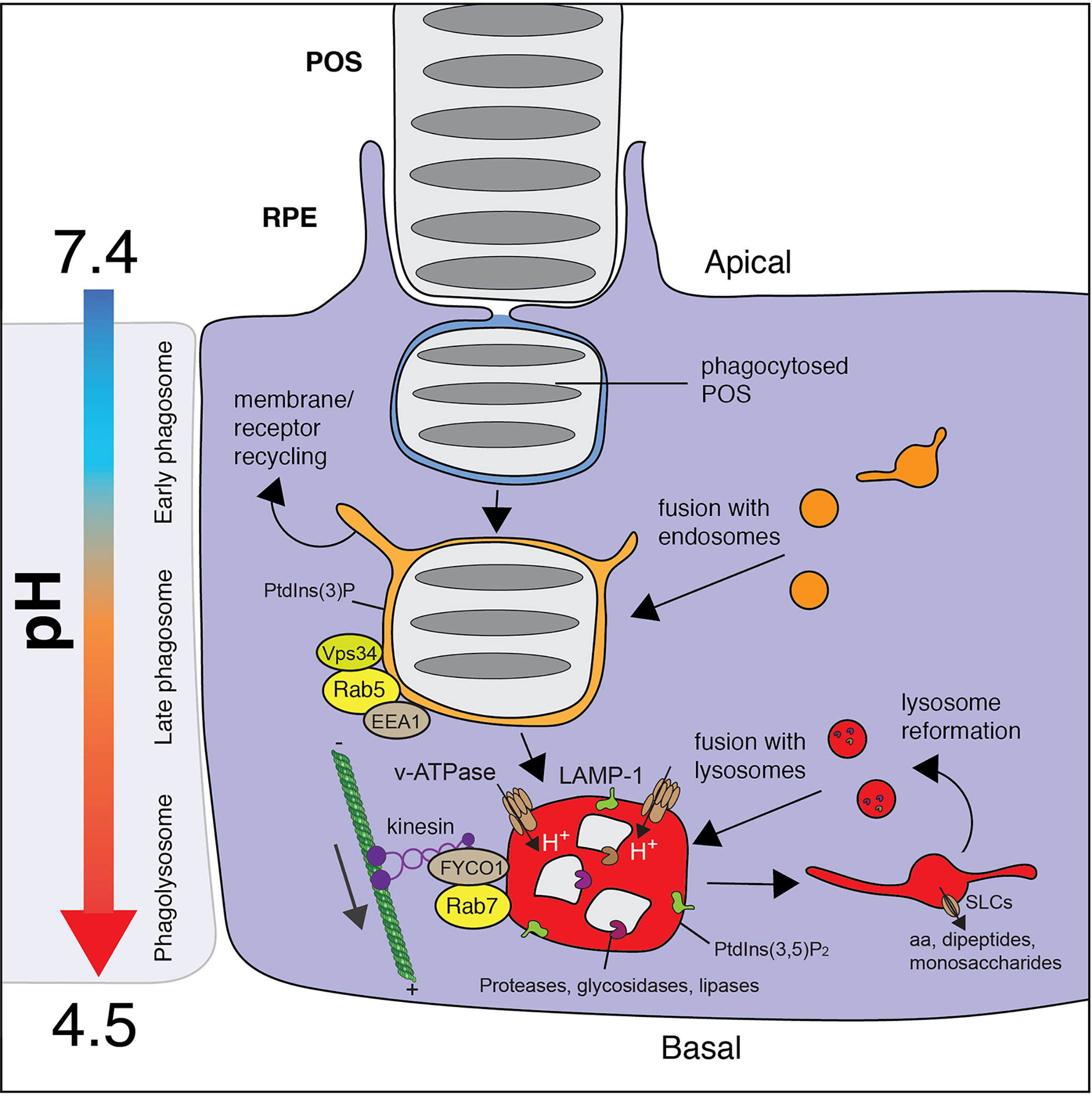
Figure 3 Maturation of the RPE phagosome. Internalized POS particles are sealed in a nascent phagosome that undergoes step-wise maturation which involves the graded fusion of endosomes and then lysosomes. The maturation of the phagosome is delineated by its association with Rab GTPase and its phosphoinositide constituents, which act to coordinate fusion. Maturation also involves microtubule-based movement toward the cell center, a switch in Rabs [Rab5 → Rab7] and phosphoinositide [PI(3)P → PI(3,5)P2] composition, a drop in pH [6.5 → 4.0], and the activation of phagosomal proteases. In the RPE, the movement of the phagosome is dependent on kinesins which traverse the cytosol towards the plus ends of the microtubules on the basal side of the cell. Once solutes are transported from the phagolysosome, the compartment undergoes fragmentation to reform and replenish the pool of lysosomes.
The Rab family of GTPases that orchestrate membrane fusion events and organellar trafficking are indispensable for maturation (94). Following its sealing, Rabex-5, a guanine nucleotide exchange factor (GEF), is targeted to the early phagosome to recruit and activate Rab5, Rab21, and Rab22 (97). While these Rab5 subfamily members all play important roles in phagosome maturation (98), Rab5 remains the best characterized member (99). Once active, Rab5 binds to its effectors including Rabaptin-5 which in turn re-stimulates Rabex-5, triggering a positive-feedback loop (100). Additionally, Rab5 exerts its function through the recruitment and activation of the phosphatidylinositol 3-kinase (PI3K) Vps34, which results in the rapid production of phosphatidylinositol 3-phosphate (PI3P). PI3P is necessary for the recruitment of early endosomal antigen 1 (EEA1), a protein that is essential for phagosome maturation by promoting its fusion with endosomes (99).
Rab5 and EEA1 are indeed reported to localize to the early RPE phagosome along with PI3P (101). Moreover, the conditional knockout of Vps34 leads to disordered phagosome trafficking, impaired lysosome fusion, and retinal degeneration in mice, supporting a similar role for PI3P in the RPE system (102). As the RPE phagosome matures, marked by its incorporation of LAMP proteins, the localization of Rab5 and EEA1 are expectedly depressed (101). Though mechanisms remain to be elucidated, the LAMP family of lysosome-resident glycoproteins is indeed required for the fusion of lysosomes with the phagosome (103). It is therefore noteworthy, that the loss of just one of these family members, LAMP2, in the murine RPE causes delayed POS degradation and the build-up of POS debris (104). In fact, the loss-of-function mutations in LAMP2 that cause Danon disease in humans is also associated with retinal and macular degeneration (105).
In all cases, progression to the late phagosome is marked not just by the acquisition of LAMP proteins and its luminal acidification (described later), but also by the transition from being Rab5- to Rab7-positive, known as a Rab5-Rab7 “switch” (106). The sharp transition between Rab5 and Rab7 is explained by Rab7 GEFs that inhibit Rab5 activation. Specifically, Mon1, a subunit of the Rab7 GEF Mon1-Ccz1 that recruits and activates Rab7, also ousts Rabex-5 and arrests the Rab5 feedback loop (107). The Rab5-Rab7 transition promotes the subunit substitution necessary for the transformation of CORVET, a protein complex associated with early endosomal fusion, to HOPS, associated with later stage endosomal fusion (108).
In addition to facilitating fusion with late endosomes and lysosomes, Rab7 activation also coordinates the inward movement of the phagosome. In myeloid cells, the Rab7 effectors Rab7-interacting lysosomal protein (RILP) (109) and OSBP-related protein 1 (ORP1L) (110) form complexes that cluster dynein motors on the phagosome to cooperatively facilitate retrograde movement (111). The apical to basal movement of phagosomes in the RPE, while indeed dependent on Rab7 and microtubules (112, 113), differs markedly. First, the phagosomes formed by the RPE are initially engaged by Myosin VIIa, an unconventional myosin that moves along F-actin. Myosin VIIa is of particular interest since mutations in the MYO7A gene cause Usher syndrome 1B which involves progressive retinal degeneration (114). Fittingly, Myosin VIIa is largely expressed in the apical region of the RPE where it may facilitate the initial transport of the phagosome through a dense apical F-actin network until it can latch on to microtubule motors (96, 115). Second, the RPE phagosomes are then bound by kinesin motors and their associated kinesin-1 light chain 1 (KLC1), rather than dynein. The RPE phagosomes on the apical side of the cell lose their association with Myosin-VIIa as they begin to associate with KLC1, an event that coincides with their movement toward the basolateral side (95, 116). Mice lacking KLC1 show delayed phagosome progression to the basal region of the cell, the accumulation of debris and drusen, and ultimately the loss of photoreceptors (96, 116). In contrast to myeloid cells, a strong association with dynein motor complexes is in fact associated with delayed phagosome maturation and impaired phagosome motility in RPE (95). The obvious difference in phagosomal transport through the cytosol between cell types is attributed to the polarity of the microtubule tracks that are inversely oriented in the RPE (12): The plus ends of the microtubules are found at the basolateral side of the RPE. Rab7-GTP on the RPE phagosome must therefore mediate connections between the organelle and kinesins. Interestingly, active Rab7 binds FYCO1 and kinesin light chain/KIF5 (117). A role for this complex in orchestrating phagosome motility in the RPE is unknown, though FYCO1 is known to at least control the positioning of lysosomes in RPE cells (118).
The precise regulation of Rabs by their posttranslational modification is also essential for phagosome maturation, especially in the RPE. For example, the ablation of the Rab escort protein (Rep1), a geranylgeranyltransferase that acts on Rab7 and others, causes a delay in phagosome maturation and POS clearance, the accumulation of debris in the RPE, and a pathology resembling age-related macular degeneration (119, 120). Clearly, the efficient mobilization and maturation of the RPE phagosome is critical for retinal health and homeostasis and has some unique features that warrant further study.
Acidification of the Phagosome
As phagosomes form in the RPE every morning, their lumens acidify. The drop in pH is largely driven by the vacuolar ATPase (V-ATPase) that hydrolyzes ATP to pump protons into the lumen. Many of the digestive hydrolases, delivered to the phagosome by its fusion with endolysosomes, are then activated by the decrease in pH. That the acidification of the phagosome is indeed critical for the proper digestion of POS is exemplified by experiments demonstrating that inhibiting the V-ATPase in the RPE causes their accumulation of swollen phagolysosomes that do not degrade opsin proteins (121–123). This ultimately leads to the build-up of undigested debris between the Bruch’s membrane of the choroid and the RPE, a phenotype akin to age-related macular degeneration (AMD) in humans (12).
Despite the imperative role for acidification of the phagosome via the V-ATPase, it is a process that remains remarkably enigmatic in the RPE system. This is partly explained from an operational standpoint. Reliable measurements of phagosomal pH entail the selection of a suitable probe and the ability to target it to the phagosome. In the case of retinal flat mounts, phagocytic targets (i.e. POS) cannot be readily and specifically labeled. Most pH determinations of the RPE phagosome, and even of their lysosomes, have therefore been estimated by qualitative or semiquantitative means using predominantly membrane-permeant weak bases that accumulate in acidic organelles (89, 124). These include fluorescent dyes like LysoTracker (89), that accumulates in the phagosome without altering its fluorescence or LysoSensor that undergoes a pH-dependent spectral shift (124). While such sensors are acceptable indicators of acidity, they come with limitations (125). For example, when used at sufficiently high concentrations, membrane-permeant basic probes can themselves alter the pH of the (phago)lysosome. Nevertheless, LysoSensor has been used to determine that lysosomes in RPE cells in culture are pH 4.5 on average (124).
More recently, a genetically encoded chimera of an intralumenal, pH-sensitive GFP tethered to a cytosolic mCherry via a lysosomal transmembrane domain has been utilized to measure the pH of RPE lysosomes (126). Such a system also has caveats. First, the pKa of GFP is ~6.0 which is not optimal for determinations of the more acidic pH values expected within lysosomes and phagolysosomes. Second, the proteases within the lysosome are expected to degrade the internal GFP thereby conflating measurements of the proteolytic activity and the pH of the compartments.
Generally, the pH of the POS-containing phagosome or the rate at which it acidifies has not been formally assessed but has been inferred by the degradation of its contents (123) or by LysoTracker (89). Other, non-ratiometric dyes like rhodamine-based pHrodo®, that exhibit strong fluorescence in acidic environments have also been used to grossly estimate the pH of RPE phagosomes (127, 128).
The rates and extent of acidification should not be tacitly assumed to occur in all phagocytes similarly. One consideration is that in isolation, the V-ATPase driven proton influx required for acidification is self-limiting as it creates a membrane potential detrimental to the efficient acidification of the phagolysosome. Such an undesirable electrogenic consequence is circumvented by accompanied counterion fluxes that dissipate the building voltage. This can be achieved by cation efflux (e.g. the expulsion of Na+ or K+) or concurrent anion influx (e.g. of Cl−) (129) and these pathways may differ widely between cell types. Little is known about counterion flux mechanisms in any phagocyte, however, it is noteworthy that the whole animal loss of ClC7 or ClC3, chloride exchangers that can provide such counterion fluxes, results in retinal degeneration. While a direct role for ClCs in the RPE is not yet known, this phenotype could suggest that Cl- flux into the RPE phagosome may be critical for its acidification and breakdown of POS (130, 131).
Taken together, given that the rapid acidification supersedes enzymatic breakdown of the POS in the phagosome and is critical in maintaining retinal health, additional experiments should be done in this area. Clearly, a complete picture for the mechanisms that regulate the V-ATPase, pH, and maturation of the RPE phagosome has yet to emerge.
Distilling and Breaking Down Macromolecules
The delivery of the V-ATPase and acidification of the phagosome is concomitant with the enzymatic breakdown of its contents (Figure 3). Though published results vary, as much as 70% of the POS dry mass is heavily glycosylated opsin, and the remaining 30% is polyunsaturated lipids, and therefore the proper digestion in the phagosome requires the delivery and activation of proteases, glycosidases, and lipases (132). The removal of water from the phagosome is also thought to be an important early step that facilitates membrane recycling, decreases the volume required for the pump to acidify, and increases the contact between enzymes and their substrates (133). This early step is initiated by monovalent ion efflux followed by outward movement of osmotically-obliged water (5) that presumably exits through aquaporins or other pores and channels that make mammalian membranes water-permeant.
The proteases involved in breaking down opsins which account for >95% of the protein content of the shed POS (134) into peptides and amino acids are primarily cathepsins. Cathepsin D, a ubiquitously expressed aspartyl protease, has garnered particular attention as it is both highly-expressed in the RPE and its genetic inactivation in mice leads to incompletely digested rhodopsins in the phagosome, the general accumulation of undigested phagocytic debris, and photoreceptor death (135, 136). There are numerous cathepsins that function as cysteine proteases in the RPE, on the other hand, so while these have also been suggested to play a role in proteolytic degradation of the OS target, there is likely functional redundancy between them (12). Making matters more complex, in addition to their autoactivation, aspartyl proteases like cathepsin D can be cleaved and activated by these cysteine proteases in the RPE lysosome (137).
The major phospholipids of the POS are phosphatidylcholine and phosphatidylethanolamine which account for ~80% of the total lipid, followed by phosphatidylserine (13%), and minor contributors like phosphatidylinositol and sphingomyelin (138). As the photoreceptor discs age, their cholesterol content tends to decrease, going from 30% of the molar lipid when first generated by their invagination from the plasma membrane to 10% when shed as part of the distal POS (139). Ingested photoreceptor lipids, including phospholipids and glycolipids, are broken down in the RPE phagolysosome by pH-dependent lipid hydrolases, namely phospholipase A1 and A2 (140). Cholesterol, on the other hand, is liberated from the POS and possibly de-esterified via the lysosomal acid lipase type A (141) and once soluble, transported into the limiting membrane of the phagosome. This requires a system of cholesterol-binding proteins that support movement of the hydrophobic molecule through tunnel-like structures. It can be achieved by the concerted action of Niemann–Pick type C (NPC) 1 and 2 (142) as well as by LIMP-2 (143). Niemann-Pick proteins are indeed necessary for normal retinal function (144). Upon its incorporation into the phagolysosomal membrane, cholesterol transport to extra-lysosomal destinations occurs through vesicular and non-vesicular routes (i.e. membrane contact sites). By these means, cholesterol can be delivered to lysosomes and/or trafficked to the plasma membrane, the Golgi apparatus and the endoplasmic reticulum. Remarkably, as discussed later, the efflux of POS-derived cholesterol back to the photoreceptor by the RPE appears to be a means for its reutilization.
As opsins are replete with glycans, a series of glycosidases are also deployed by the RPE to the phagosome including alpha-mannosidase, beta-glucouronidase and alpha fucosidase (145). Human diseases involving a deficiency of these enzymes commonly have retinal phenotypes, including degeneration, photoreceptor death, vision loss, and undigested phagocytic debris, suggesting that these enzymes play an important role in POS catabolism (146, 147).
Finally, the POS is in fact embedded within an interphotoreceptor matrix (IPM) that has a unique molecular composition that differs from other extracellular matrices. The major insoluble components of the IPM are in fact glycosaminoglycans (i.e. hyaluronic acid and proteoglycans with heparan, chondroitin, and keratan sulfate chains) (148). While the soluble fraction of the IPM contains only small amounts of these, the RPE is expected to take up portions of the IPM along with the POS during phagocytosis and may even facilitate the turnover of the IPM over time. It follows that deficiencies in the enzymes responsible for the breakdown of the glycosaminoglycans (mucopolysaccharidoses) have some sort of retinal phenotype (149).
Solute Efflux and Resolution of the RPE Phagolysosome
Organic and inorganic solutes liberated or accumulated in the phagosome need to be effluxed from the vacuole in order for the compartment to “resolve” (Figure 3). Resolution is a normal part of the phagocytic process but remains the least understood aspect. In the case of the RPE, the return of the phagosomal membrane to replenish the pool of lysosomes is essential. Indeed, there are no observed circadian changes in the expression of prototypical lysosomal proteins in the RPE and so the pool of lysosomes used by the phagosomes must be returned to the cell (89).
In the final stages of resolution, the efflux of solutes is in fact proposed as the mechanism that drives the reformation of lysosomes (Figure 4). Here, tubulation and vesiculation requires extreme deformations to the limiting phagosomal membrane and is opposed by any tension on the vacuole (133). Tension comes in the form of osmotic pressure, generated because the solute concentrations of the phagosome can be higher than those of the cytosol. As solutes flux out of the phagosome, such pressure is relieved and the membrane becomes more pliable to these deformations by clathrin for example (150).
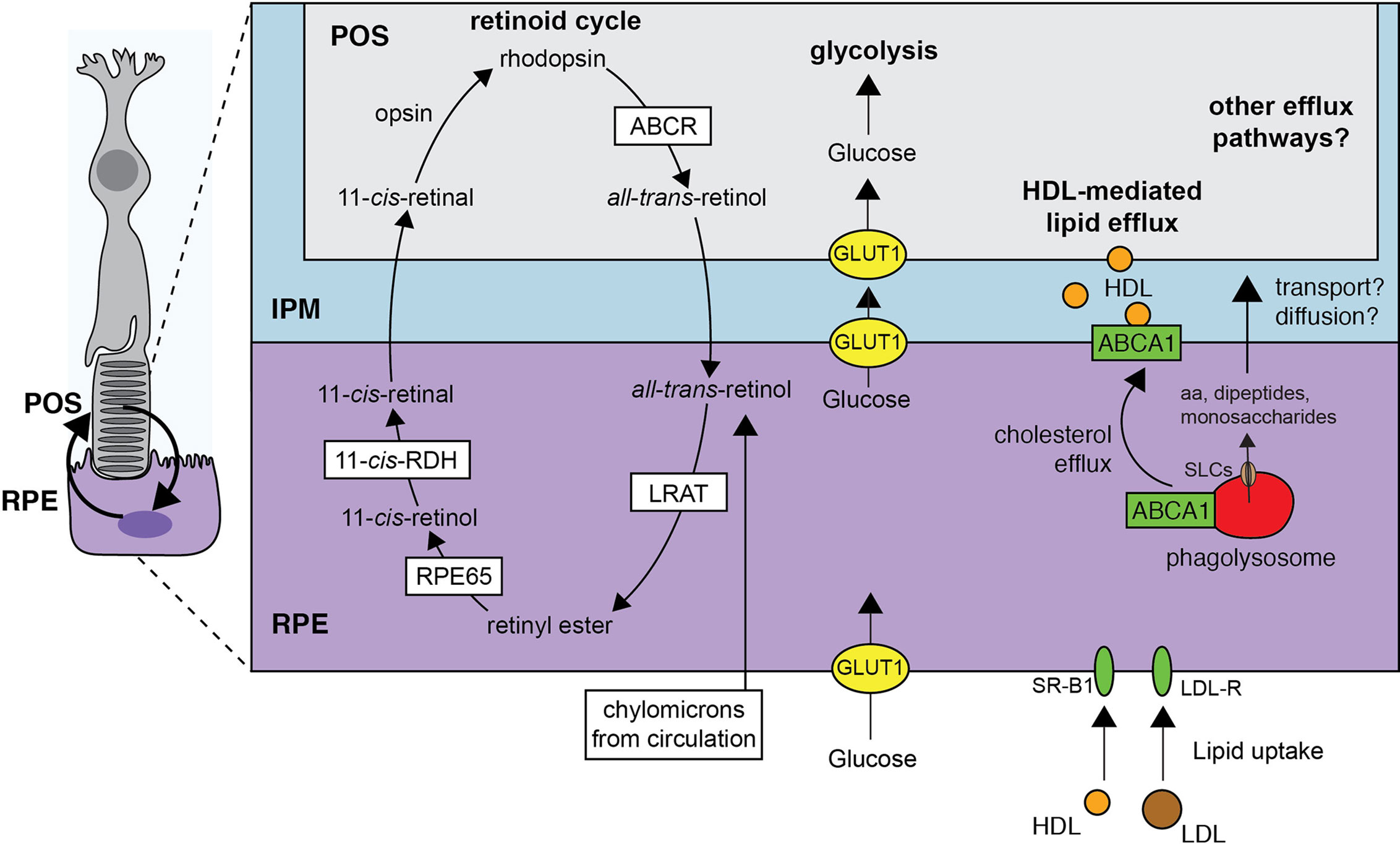
Figure 4 Digestion and solute efflux in the mature RPE phagolysosome. The retinoid cycle necessitates the active participation of the RPE. Upon photoisomerization of 11-cis-retinal to all-trans-retinal, the chromophore activates opsins and phototransduction. The removal of all-trans-retinal can then occurs by active means or passive diffusion but importantly, is delivered to the RPE for its esterification (catalyzed by lecithin:retinol acyltransferase/LRAT). Isomerization to 11-cis-retinol and oxidation to 11-cis-retinal also occurs in the RPE before diffusion back to the POS. The coupling between the POS and the RPE does not stop there. Glucose is transported to the interphotoreceptor matrix from circulation by the RPE and could conceivably also come from the phagolysosome. Similarly, high density lipoprotein mediates lipid efflux from the RPE to be used by the photoreceptors. Lipids, including cholesterol, are either transported from circulation or recycled from the POS-containing phagosome. Finally, other building blocks like amino acids may be supplied to the IPM by means not yet determined.
The exit pathways for solutes come in the form of solute carrier proteins (SLCs), and enormous family of transporters. While SLCs that flux amino acids and dipeptides out of the phagolysosome are the best appreciate in this regard (151–154), others that flux monosaccharides, metals, Na+ (155), Ca2+ (156), etc. will all be critical to the resolution process. The control of the efflux pathways is only recently becoming appreciated. The mammalian target of rapamycin (mTOR) and its associated complexes form major regulatory hubs that can both inhibit and stimulate amino acid efflux by SLCs (151, 152, 157) and even inhibit Na+ channels (155). We anticipate that the “phagosome-lysosome reformation” (PLR) will require exquisite control over its timing. For example, the organic solutes that contribute less osmotically to the compartment overall may be effluxed before more prevalent monovalent ions to ensure that the complete digestion and efflux of proteins and amino acids precedes the reformation of lysosomes. Of course, these ideas remain to be tested.
Recycling Between the RPE and Photoreceptors
The photoreceptor outer segment (POS) grows as it is consumed by the RPE, marking a coupling between a highly anabolic cell type and a catabolic one (Figure 4). Throughout this review, we have emphasized the astonishing amount of material that the RPE must turnover per day. As 90% of the membrane shed in the POS derives from the photosensitive membranes of the internal discs, the total surface membrane of a phagosome is ~30 µm2 and the RPE consumes ~900 µm2 of surface membrane on average daily. It is not surprising that membrane-associated elements can be recycled by the RPE back to the photoreceptor (18). In some cases, the recycling pathways between the RPE and photoreceptors are well-established, especially for the retinoid cycle (158). As described previously, the phototransduction by opsins occur when the bound 11-cis-retinal undergoes isomerization to all-trans-retinal which in turns alters the conformation of the opsin. The return to the photosensitive receptor conformation requires the rejuvenation of the 11-cis-retinal from all-trans-retinal. Aside from the reduction of the all-trans-retinal to retinol, all reactions that support the retinoid cycle in fact take place in the RPE (159). The all-trans-retinol enters the RPE from circulation but a significant portion seems to be shuttled through the IPM (159) or by phagocytosis of the POS. The return of the 11-cis-retinal to the photoreceptor is in turn mediated by binding proteins, passive diffusion through the IPM, or direct transport.
The retinoid cycle represents just one of the recycling pathways between the cells. For example, the phospholipids of the photosensitive membrane discs of the POS are enriched in docosahexanoic acid (DHA), an omega-3 fatty acid. Remarkably, the retina can retain DHA even during long periods of dietary deprivation of omega-3 fatty acids (160). Careful experiments using radiolabeled DHA tracked the fatty acid through the POS and into the RPE and found that phagosomes are indeed packed with DHA to the same extent as the distal POS. Moreover, these types of experiments found that labelling in the RPE cytosol remains low and diffuse throughout extended (days long) periods of tracing, demonstrating that the DHA is quickly recycled back to the photoreceptors (22, 161).
While beyond the scope of this review, it should be mentioned that normal functions of RPE include maintaining an anti-inflammatory microenvironment and this is owed to the ongoing lipid metabolism of the phagosome. In particular, not all of the DHA is necessarily recycled back to the POS as is. The DHA supplied to the RPE can also be enzymatically converted to derive resolvins including neuroprotection D1 (NPD1) (162). NPD1 is a potent anti-inflammatory mediator that inhibits pro-apoptotic proteins and induces anti-apoptotic proteins, yielding cell-protective effects (162). On the other hand, the RPE can also contribute to pro-inflammatory pathways in certain contexts. Under sustained oxidative stress, for example, the RPE makes inflammatory cytokines including IL-6 and IL-1β and this can lead to AMD (163).
In addition to supplying the photoreceptor with retinal and DHA, the RPE sources the photoreceptors with glucose. To do so, the RPE expresses remarkably high levels of GLUT1 at their apical and basolateral surface (164). Glucose in the cytosol would normally be quickly converted for its use in glycolysis, but this is effectively suppressed in the RPE by the lactate that is produced and exported by the photoreceptor and because kinases that act on sugars are inhibited or expressed at sufficient low levels (164, 165). Conversely, such kinases (e.g. pyruvate kinase) are highly enriched in the photoreceptor making the cell highly glycolytic (164). When the RPE is made to consume more glucose, the neighboring photoreceptors become starved and ultimately degenerate. Using the same Glut1-based transport and others, we anticipate monosaccharides could be recycled from the RPE phagosome back to the IPM as well.
Finally, like other phagocytes, the RPE specialize in lipid/cholesterol efflux as emphasized by their robust expression and use of the cholesterol efflux regulatory protein ATP-binding cassette transporter A1 (ABCA1) (166). Here, ABCA1 shuttles cholesterol from the cytoplasm onto HDL. In the RPE, ABCA1 is targeted to both basolateral and apical membranes and can mediate efflux towards the subretinal space (i.e. the IPM) or, to a lesser extent, the choroidal space. Such efflux is more evident when the cells are given liver X receptor agonists or POS targets. Notably, the RPE is well-endowed with other cholesterol efflux proteins like SRB1 and ABCG1 (166). The RPE also metabolizes fatty acids found in the POS like palmitate to produce beta-hydroxybutyrate (167). Interestingly, photoreceptors were also recently found to use palmitate as an energy source through oxidative phosphorylation (168). The shuttling of palmitate or beta-hydroxybutyrate from the RPE to the photoreceptor as potential substates could therefore generally serve to maintain photoreceptor function (169).
It remains to be tested if other building blocks like sugars and amino acids are indeed recycled back to the photoreceptor once fluxed out of the RPE phagosome. Forms of direct transport, where photoreceptors and the RPE could be coupled by GAP junction proteins that support direct connections of the cytosol, are conceivable.
Conclusion
The routine phagocytosis performed by the RPE of their photoreceptor neighbors serves as a reasonable guide for future investigations of heterocellular metabolic circuits as formed by tissue-resident phagocytes and the cells they prune and turnover. Unlike in the retina, other tissues can quite readily expand and contract with abrupt dietary changes or with developmental programs like involution. In these cases, the growth of resident populations of phagocytes are kept in check by the stroma: Well-defined paracrine growth factor circuits between stromal cells and macrophages can stabilize the ratios of these populations and this requires intimate contacts between the cells (170). Supporting mitogenesis in such circuits with nutrient supplies may be an overlooked feature of the coupling. A recent emphasis on cell-cell competition in tissues (171) may benefit from understanding the circuits, cell-cell sharing and recycling, and the metabolic ecosystems that ensure tissue homeostasis. Such studies would immediately lend themselves to an appreciation for how tumor-associated macrophages support the growth of lesions. Additionally, more emphasis on phagosome resolution and the handling of solutes in the endocytic pathway of phagocytes is will certainly lead to strategies that break or support such processes and cycles. The RPE-photoreceptor coupling is a well-defined Yin and Yang relationship but represents one of many more circuits to be uncovered.
Author Contributions
WK and SF constructed figures and wrote/reviewed the manuscript. All authors contributed to the article and approved the submitted version.
Funding
This work was supported by a CIHR Project Grant PJT #169180 (SAF).
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
1. Freeman SA, Grinstein S. Phagocytosis: receptors, signal integration, and the cytoskeleton. Immunol Rev (2014) 262:193–215. doi: 10.1111/imr.12212
2. Underhill DM, Goodridge HS. Information processing during phagocytosis. Nat Rev Immunol (2012) 12:492–502. doi: 10.1038/nri3244
3. Aderem A, Underhill DM. Mechanisms of phagocytosis in macrophages. Annu Rev Immunol (1999) 17:593–623. doi: 10.1146/annurev.immunol.17.1.593
4. Uderhardt S, Martins AJ, Tsang JS, Lammermann T, Germain RN. Resident Macrophages Cloak Tissue Microlesions to Prevent Neutrophil-Driven Inflammatory Damage. Cell (2019) 177:541–555 e17. doi: 10.1016/j.cell.2019.02.028
5. Freeman SA, Uderhardt S, Saric A, Collins RF, Buckley CM, Mylvaganam S, et al. Lipid-gated monovalent ion fluxes regulate endocytic traffic and support immune surveillance. Science (2020) 367:301–5. doi: 10.1126/science.aaw9544
6. Kohyama M, Ise W, Edelson BT, Wilker PR, Hildner K, Mejia C, et al. Role for Spi-C in the development of red pulp macrophages and splenic iron homeostasis. Nature (2009) 457:318–21. doi: 10.1038/nature07472
7. Klei TR, Meinderts SM, van den Berg TK, van Bruggen R. From the Cradle to the Grave: The Role of Macrophages in Erythropoiesis and Erythrophagocytosis. Front Immunol (2017) 8:73. doi: 10.3389/fimmu.2017.00073
8. de Back DZ, Kostova EB, van Kraaij M, van den Berg TK, van Bruggen R. Of macrophages and red blood cells; a complex love story. Front Physiol (2014) 5:9. doi: 10.3389/fphys.2014.00009
9. Mahajan A, Herrmann M, Munoz LE. Clearance Deficiency and Cell Death Pathways: A Model for the Pathogenesis of SLE. Front Immunol (2016) 7:35. doi: 10.3389/fimmu.2016.00035
10. Arandjelovic S, Ravichandran KS. Phagocytosis of apoptotic cells in homeostasis. Nat Immunol (2015) 16:907–17. doi: 10.1038/ni.3253
11. Penberthy KK, Lysiak JJ, Ravichandran KS. Rethinking Phagocytes: Clues from the Retina and Testes. Trends Cell Biol (2018) 28:317–27. doi: 10.1016/j.tcb.2018.01.004
12. Lakkaraju A, Umapathy A, Tan LX, Daniele L, Philp NJ, Boesze-Battaglia K, et al. The cell biology of the retinal pigment epithelium. Prog Retin Eye Res (2020), 100846. doi: 10.1016/j.preteyeres.2020.100846
13. Young RW, Bok D. Participation of the retinal pigment epithelium in the rod outer segment renewal process. J Cell Biol (1969) 42:392–403. doi: 10.1083/jcb.42.2.392
14. LaVail MM. Rod outer segment disk shedding in rat retina: relationship to cyclic lighting. Science (1976) 194:1071–4. doi: 10.1126/science.982063
15. Kevany BM, Palczewski K. Phagocytosis of retinal rod and cone photoreceptors. Physiol (Bethesda) (2010) 25:8–15. doi: 10.1152/physiol.00038.2009
16. Ruggiero L, Connor MP, Chen J, Langen R, Finnemann SC. Diurnal, localized exposure of phosphatidylserine by rod outer segment tips in wild-type but not Itgb5-/- or Mfge8-/- mouse retina. Proc Natl Acad Sci U.S.A. (2012) 109:8145–8. doi: 10.1073/pnas.1121101109
17. Bok D, Hall MO. The role of the pigment epithelium in the etiology of inherited retinal dystrophy in the rat. J Cell Biol (1971) 49:664–82. doi: 10.1083/jcb.49.3.664
18. Strauss O. The retinal pigment epithelium in visual function. Physiol Rev (2005) 85:845–81. doi: 10.1152/physrev.00021.2004
19. Caceres PS, Rodriguez-Boulan E. Retinal pigment epithelium polarity in health and blinding diseases. Curr Opin Cell Biol (2020) 62:37–45. doi: 10.1016/j.ceb.2019.08.001
20. Delori FC, Dorey CK, Staurenghi G, Arend O, Goger DG, Weiter JJ. In vivo fluorescence of the ocular fundus exhibits retinal pigment epithelium lipofuscin characteristics. Invest Ophthalmol Vis Sci (1995) 36:718–29.
21. Feeney-Burns L, Hilderbrand ES, Eldridge S. Aging human RPE: morphometric analysis of macular, equatorial, and peripheral cells. Invest Ophthalmol Vis Sci (1984) 25:195–200.
22. Bazan NG, Gordon WC, Rodriguez de Turco EB. Docosahexaenoic acid uptake and metabolism in photoreceptors: retinal conservation by an efficient retinal pigment epithelial cell-mediated recycling process. Adv Exp Med Biol (1992) 318:295–306. doi: 10.1007/978-1-4615-3426-6_26
23. Han CZ, Ravichandran KS. Metabolic connections during apoptotic cell engulfment. Cell (2011) 147:1442–5. doi: 10.1016/j.cell.2011.12.006
24. Masland RH. The neuronal organization of the retina. Neuron (2012) 76:266–80. doi: 10.1016/j.neuron.2012.10.002
26. Organisciak DT, Vaughan DK. Retinal light damage: mechanisms and protection. Prog Retin Eye Res (2010) 29:113–34. doi: 10.1016/j.preteyeres.2009.11.004
27. Lemke G. How macrophages deal with death. Nat Rev Immunol (2019) 19:539–49. doi: 10.1038/s41577-019-0167-y
28. Nagata S, Suzuki J, Segawa K, Fujii T. Exposure of phosphatidylserine on the cell surface. Cell Death Differ (2016) 23:952–61. doi: 10.1038/cdd.2016.7
29. Segawa K, Kurata S, Nagata S. Human Type IV P-type ATPases That Work as Plasma Membrane Phospholipid Flippases and Their Regulation by Caspase and Calcium. J Biol Chem (2016) 291:762–72. doi: 10.1074/jbc.M115.690727
30. Segawa K, Kurata S, Yanagihashi Y, Brummelkamp TR, Matsuda F, Nagata S. Caspase-mediated cleavage of phospholipid flippase for apoptotic phosphatidylserine exposure. Science (2014) 344:1164–8. doi: 10.1126/science.1252809
31. Suzuki J, Imanishi E, Nagata S. Xkr8 phospholipid scrambling complex in apoptotic phosphatidylserine exposure. Proc Natl Acad Sci U.S.A. (2016) 113:9509–14. doi: 10.1073/pnas.1610403113
32. Suzuki J, Fujii T, Imao T, Ishihara K, Kuba H, Nagata S. Calcium-dependent phospholipid scramblase activity of TMEM16 protein family members. J Biol Chem (2013) 288:13305–16. doi: 10.1074/jbc.M113.457937
33. Suzuki J, Denning DP, Imanishi E, Horvitz HR, Nagata S. Xk-related protein 8 and CED-8 promote phosphatidylserine exposure in apoptotic cells. Science (2013) 341:403–6. doi: 10.1126/science.1236758
34. Suzuki J, Umeda M, Sims PJ, Nagata S. Calcium-dependent phospholipid scrambling by TMEM16F. Nature (2010) 468:834–8. doi: 10.1038/nature09583
35. Burstyn-Cohen T, Lew ED, Traves PG, Burrola PG, Hash JC, Lemke G. Genetic Dissection of TAM Receptor-Ligand Interaction in Retinal Pigment Epithelial Cell Phagocytosis. Neuron (2012) 76:1123–32. doi: 10.1016/j.neuron.2012.10.015
36. Prasad D, Rothlin CV, Burrola P, Burstyn-Cohen T, Lu QX, de Frutos PG, et al. TAM receptor function in the retinal pigment epithelium. Mol Cell Neurosci (2006) 33:96–108. doi: 10.1016/j.mcn.2006.06.011
37. Hall MO, Obin MS, Heeb M, Burgess BL, Abrams TA. Both protein S and Gas6 stimulate outer segment phagocytosis by cultured rat retinal pigment epithelial cells. Exp Eye Res (2005) 81:581–91. doi: 10.1016/j.exer.2005.03.017
38. Caberoy NB, Zhou Y, Li W. Tubby and tubby-like protein 1 are new MerTK ligands for phagocytosis. EMBO J (2010) 29:3898–910. doi: 10.1038/emboj.2010.265
39. Hanayama R, Tanaka M, Miwa K, Shinohara A, Iwamatsu A, Nagata S. Identification of a factor that links apoptotic cells to phagocytes. Nature (2002) 417:182–7. doi: 10.1038/417182a
40. Karl MO, Kroeger W, Wimmers S, Milenkovic VM, Valtink M, Engelmann K, et al. Endogenous Gas6 and Ca2+ -channel activation modulate phagocytosis by retinal pigment epithelium. Cell Signal (2008) 20:1159–68. doi: 10.1016/j.cellsig.2008.02.005
41. Milicevic N, Mazzaro N, de Bruin I, Wils E, Ten Brink J, Asbroek AT, et al. Rev-Erbalpha and Photoreceptor Outer Segments modulate the Circadian Clock in Retinal Pigment Epithelial Cells. Sci Rep (2019) 9:11790. doi: 10.1038/s41598-019-48203-3
42. Law AL, Parinot C, Chatagnon J, Gravez B, Sahel JA, Bhattacharya SS, et al. Cleavage of Mer tyrosine kinase (MerTK) from the cell surface contributes to the regulation of retinal phagocytosis. J Biol Chem (2015) 290:4941–52. doi: 10.1074/jbc.M114.628297
43. Stohr H, Heisig JB, Benz PM, Schoberl S, Milenkovic VM, Strauss O, et al. TMEM16B, a novel protein with calcium-dependent chloride channel activity, associates with a presynaptic protein complex in photoreceptor terminals. J Neurosci (2009) 29:6809–18. doi: 10.1523/JNEUROSCI.5546-08.2009
44. Bricogne C, Fine M, Pereira PM, Sung J, Tijani M, Wang Y, et al. TMEM16F activation by Ca(2+) triggers plasma membrane expansion and directs PD-1 trafficking. Sci Rep (2019) 9:619. doi: 10.1038/s41598-019-43808-0
45. Almedawar S, Vafia K, Schreiter S, Neumann K, Khattak S, Kurth T, et al. MERTK-Dependent Ensheathment of Photoreceptor Outer Segments by Human Pluripotent Stem Cell-Derived Retinal Pigment Epithelium. Stem Cell Rep (2020) 14:374–89. doi: 10.1016/j.stemcr.2020.02.004
46. Matsumoto B, Defoe DM, Besharse JC. Membrane turnover in rod photoreceptors: ensheathment and phagocytosis of outer segment distal tips by pseudopodia of the retinal pigment epithelium. Proc R Soc Lond B Biol Sci (1987) 230:339–54. doi: 10.1098/rspb.1987.0023
47. Steinberg RH, Wood I, Hogan MJ. Pigment epithelial ensheathment and phagocytosis of extrafoveal cones in human retina. Philos Trans R Soc Lond B Biol Sci (1977) 277:459–74. doi: 10.1098/rstb.1977.0028
48. Nandrot EF, Anand M, Sircar M, Finnemann SC. Novel role for alphavbeta5-integrin in retinal adhesion and its diurnal peak. Am J Physiol Cell Physiol (2006) 290:C1256–62. doi: 10.1152/ajpcell.00480.2005
49. Nawrot M, West K, Huang J, Possin DE, Bretscher A, Crabb JW, et al. Cellular retinaldehyde-binding protein interacts with ERM-binding phosphoprotein 50 in retinal pigment epithelium. Invest Ophthalmol Vis Sci (2004) 45:393–401. doi: 10.1167/iovs.03-0989
50. Nijhara R, van Hennik PB, Gignac ML, Kruhlak MJ, Hordijk PL, Delon J, et al. Rac1 mediates collapse of microvilli on chemokine-activated T lymphocytes. J Immunol (2004) 173:4985–93. doi: 10.4049/jimmunol.173.8.4985
51. Takahashi Y, Morales FC, Kreimann EL, Georgescu MM. PTEN tumor suppressor associates with NHERF proteins to attenuate PDGF receptor signaling. EMBO J (2006) 25:910–20. doi: 10.1038/sj.emboj.7600979
52. Rothlin CV, Carrera-Silva EA, Bosurgi L, Ghosh S. TAM receptor signaling in immune homeostasis. Annu Rev Immunol (2015) 33:355–91. doi: 10.1146/annurev-immunol-032414-112103
53. D’Cruz PM, Yasumura D, Weir J, Matthes MT, Abderrahim H, LaVail MM, et al. Mutation of the receptor tyrosine kinase gene Mertk in the retinal dystrophic RCS rat. Hum Mol Genet (2000) 9:645–51. doi: 10.1093/hmg/9.4.645
54. Patel JC, Hall A, Caron E. Vav regulates activation of Rac but not Cdc42 during FcgammaR-mediated phagocytosis. Mol Biol Cell (2002) 13:1215–26. doi: 10.1091/mbc.02-01-0002
55. Swanson JA. Shaping cups into phagosomes and macropinosomes. Nat Rev Mol Cell Biol (2008) 9:639–49. doi: 10.1038/nrm2447
56. Francis CL, Ryan TA, Jones BD, Smith SJ, Falkow S. Ruffles induced by Salmonella and other stimuli direct macropinocytosis of bacteria. Nature (1993) 364:639–42. doi: 10.1038/364639a0
57. Wu Y, Singh S, Georgescu MM, Birge RB. A role for Mer tyrosine kinase in alphavbeta5 integrin-mediated phagocytosis of apoptotic cells. J Cell Sci (2005) 118:539–53. doi: 10.1242/jcs.01632
58. Eden S, Rohatgi R, Podtelejnikov AV, Mann M, Kirschner MW. Mechanism of regulation of WAVE1-induced actin nucleation by Rac1 and Nck. Nature (2002) 418:790–3. doi: 10.1038/nature00859
59. Chen Z, Borek D, Padrick SB, Gomez TS, Metlagel Z, Ismail AM, et al. Structure and control of the actin regulatory WAVE complex. Nature (2010) 468:533–8. doi: 10.1038/nature09623
60. Nakamura F, Osborn TM, Hartemink CA, Hartwig JH, Stossel TP. Structural basis of filamin A functions. J Cell Biol (2007) 179:1011–25. doi: 10.1083/jcb.200707073
61. Svitkina TM, Borisy GG. Arp2/3 complex and actin depolymerizing factor/cofilin in dendritic organization and treadmilling of actin filament array in lamellipodia. J Cell Biol (1999) 145:1009–26. doi: 10.1083/jcb.145.5.1009
62. Mao Y, Finnemann SC. Essential diurnal Rac1 activation during retinal phagocytosis requires alphavbeta5 integrin but not tyrosine kinases focal adhesion kinase or Mer tyrosine kinase. Mol Biol Cell (2012) 23:1104–14. doi: 10.1091/mbc.e11-10-0840
63. Balla T. Phosphoinositides: tiny lipids with giant impact on cell regulation. Physiol Rev (2013) 93:1019–137. doi: 10.1152/physrev.00028.2012
64. Canton J, Schlam D, Breuer C, Gutschow M, Glogauer M, Grinstein S. Calcium-sensing receptors signal constitutive macropinocytosis and facilitate the uptake of NOD2 ligands in macrophages. Nat Commun (2016) 7:11284. doi: 10.1038/ncomms11284
65. Scott CC, Dobson W, Botelho RJ, Coady-Osberg N, Chavrier P, Knecht DA, et al. Phosphatidylinositol-4,5-bisphosphate hydrolysis directs actin remodeling during phagocytosis. J Cell Biol (2005) 169:139–49. doi: 10.1083/jcb.200412162
66. Bajno L, Peng XR, Schreiber AD, Moore HP, Trimble WS, Grinstein S. Focal exocytosis of VAMP3-containing vesicles at sites of phagosome formation. J Cell Biol (2000) 149:697–706. doi: 10.1083/jcb.149.3.697
67. Gauthier NC, Rossier OM, Mathur A, Hone JC, Sheetz MP. Plasma membrane area increases with spread area by exocytosis of a GPI-anchored protein compartment. Mol Biol Cell (2009) 20:3261–72. doi: 10.1091/mbc.e09-01-0071
68. Gauthier NC, Masters TA, Sheetz MP. Mechanical feedback between membrane tension and dynamics. Trends Cell Biol (2012) 22:527–35. doi: 10.1016/j.tcb.2012.07.005
69. Araki N, Johnson MT, Swanson JA. A role for phosphoinositide 3-kinase in the completion of macropinocytosis and phagocytosis by macrophages. J Cell Biol (1996) 135:1249–60. doi: 10.1083/jcb.135.5.1249
70. Cox D, Tseng CC, Bjekic G, Greenberg S. A requirement for phosphatidylinositol 3-kinase in pseudopod extension. J Biol Chem (1999) 274:1240–7. doi: 10.1074/jbc.274.3.1240
71. Bulloj A, Duan W, Finnemann SC. PI 3-kinase independent role for AKT in F-actin regulation during outer segment phagocytosis by RPE cells. Exp Eye Res (2013) 113:9–18. doi: 10.1016/j.exer.2013.05.002
72. Muller C, Charniga C, Temple S, Finnemann SC. Quantified F-Actin Morphology Is Predictive of Phagocytic Capacity of Stem Cell-Derived Retinal Pigment Epithelium. Stem Cell Rep (2018) 10:1075–87. doi: 10.1016/j.stemcr.2018.01.017
73. Case LB, Waterman CM. Integration of actin dynamics and cell adhesion by a three-dimensional, mechanosensitive molecular clutch. Nat Cell Biol (2015) 17:955–63. doi: 10.1038/ncb3191
74. Jaumouille V, Cartagena-Rivera AX, Waterman CM. Coupling of beta2 integrins to actin by a mechanosensitive molecular clutch drives complement receptor-mediated phagocytosis. Nat Cell Biol (2019) 21:1357–69. doi: 10.1038/s41556-019-0414-2
75. Freeman SA, Goyette J, Furuya W, Woods EC, Bertozzi CR, Bergmeier W, et al. Integrins Form an Expanding Diffusional Barrier that Coordinates Phagocytosis. Cell (2016) 164:128–40. doi: 10.1016/j.cell.2015.11.048
76. Marmorstein AD, Gan YC, Bonilha VL, Finnemann SC, Csaky KG, Rodriguez-Boulan E. Apical polarity of N-CAM and EMMPRIN in retinal pigment epithelium resulting from suppression of basolateral signal recognition. J Cell Biol (1998) 142:697–710. doi: 10.1083/jcb.142.3.697
77. Luke MP, LeVatte TL, O’Reilly AM, Smith BJ, Tremblay F, Brown RE, et al. Effect of NCAM on aged-related deterioration in vision. Neurobiol Aging (2016) 41:93–106. doi: 10.1016/j.neurobiolaging.2016.02.003
78. Nandrot EF, Anand M, Almeida D, Atabai K, Sheppard D, Finnemann SC. Essential role for MFG-E8 as ligand for alphavbeta5 integrin in diurnal retinal phagocytosis. Proc Natl Acad Sci U.S.A. (2007) 104:12005–10. doi: 10.1073/pnas.0704756104
79. Springer TA, Dustin ML. Integrin inside-out signaling and the immunological synapse. Curr Opin Cell Biol (2012) 24:107–15. doi: 10.1016/j.ceb.2011.10.004
80. Shelby SJ, Colwill K, Dhe-Paganon S, Pawson T, Thompson DA. MERTK Interactions with SH2-Domain Proteins in the Retinal Pigment Epithelium. PloS One (2013) 8. doi: 10.1371/journal.pone.0053964
81. Mahajan NP, Earp HS. An SH2 domain-dependent, phosphotyrosine-independent interaction between Vav1 and the Mer receptor tyrosine kinase: a mechanism for localizing guanine nucleotide-exchange factor action. J Biol Chem (2003) 278:42596–603. doi: 10.1074/jbc.M305817200
82. Linger RM, Keating AK, Earp HS, Graham DK. TAM receptor tyrosine kinases: biologic functions, signaling, and potential therapeutic targeting in human cancer. Adv Cancer Res (2008) 100:35–83. doi: 10.1016/S0065-230X(08)00002-X
83. Lee HS, Lim CJ, Puzon-McLaughlin W, Shattil SJ, Ginsberg MH. RIAM activates integrins by linking talin to ras GTPase membrane-targeting sequences. J Biol Chem (2009) 284:5119–27. doi: 10.1074/jbc.M807117200
84. Torres-Gomez A, Cabanas C, Lafuente EM. Phagocytic Integrins: Activation and Signaling. Front Immunol (2020) 11:738. doi: 10.3389/fimmu.2020.00738
85. Finnemann SC. Focal adhesion kinase signaling promotes phagocytosis of integrin-bound photoreceptors. EMBO J (2003) 22:4143–54. doi: 10.1093/emboj/cdg416
86. Heth CA, Marescalchi PA, Ye L. IP3 generation increases rod outer segment phagocytosis by cultured Royal College of Surgeons retinal pigment epithelium. Invest Ophthalmol Vis Sci (1995) 36:984–9.
87. Vollrath D, Yasumura D, Benchorin G, Matthes MT, Feng W, Nguyen NM, et al. Tyro3 Modulates Mertk-Associated Retinal Degeneration. PloS Genet (2015) 11:e1005723. doi: 10.1371/journal.pgen.1005723
88. Ryeom SW, Silverstein RL. CD36 participates in the phagocytosis of rod outer segments by retinal pigment epithelium. J Cell Sci (1996) 109( Pt 2):387–95.
89. Mao Y, Finnemann SC. Live Imaging of LysoTracker-Labelled Phagolysosomes Tracks Diurnal Phagocytosis of Photoreceptor Outer Segment Fragments in Rat RPE Tissue Ex Vivo. Adv Exp Med Biol (2016) 854:717–23. doi: 10.1007/978-3-319-17121-0_95
90. Gold ES, Underhill DM, Morrissette NS, Guo J, McNiven MA, Aderem A. Dynamin 2 is required for phagocytosis in macrophages. J Exp Med (1999) 190:1849–56. doi: 10.1084/jem.190.12.1849
91. Swanson JA, Johnson MT, Beningo K, Post P, Mooseker M, Araki N. A contractile activity that closes phagosomes in macrophages. J Cell Sci (1999) 112( Pt 3):307–16.
92. Matlung HL, Babes L, Zhao XW, van Houdt M, Treffers LW, van Rees DJ, et al. Neutrophils Kill Antibody-Opsonized Cancer Cells by Trogoptosis. Cell Rep (2018) 23:3946–3959 e6. doi: 10.1016/j.celrep.2018.05.082
93. Strick DJ, Feng W, Vollrath D. Mertk Drives Myosin II Redistribution during Retinal Pigment Epithelial Phagocytosis. Invest Ophth Vis Sci (2009) 50:2427–35. doi: 10.1167/iovs.08-3058
94. Flannagan RS, Jaumouille V, Grinstein S. The cell biology of phagocytosis. Annu Rev Pathol (2012) 7:61–98. doi: 10.1146/annurev-pathol-011811-132445
95. Esteve-Rudd J, Hazim RA, Diemer T, Paniagua AE, Volland S, Umapathy A, et al. Defective phagosome motility and degradation in cell nonautonomous RPE pathogenesis of a dominant macular degeneration. Proc Natl Acad Sci U.S.A. (2018) 115:5468–73. doi: 10.1073/pnas.1709211115
96. Gibbs D, Kitamoto J, Williams DS. Abnormal phagocytosis by retinal pigmented epithelium that lacks myosin VIIa, the Usher syndrome 1B protein. Proc Natl Acad Sci U.S.A. (2003) 100:6481–6. doi: 10.1073/pnas.1130432100
97. Delprato A, Merithew E, Lambright DG. Structure, exchange determinants, and family-wide rab specificity of the tandem helical bundle and Vps9 domains of Rabex-5. Cell (2004) 118:607–17. doi: 10.1016/j.cell.2004.08.009
98. Khurana T, Brzostowski JA, Kimmel AR. A Rab21/LIM-only/CH-LIM complex regulates phagocytosis via both activating and inhibitory mechanisms. EMBO J (2005) 24:2254–64. doi: 10.1038/sj.emboj.7600716
99. Fratti RA, Backer JM, Gruenberg J, Corvera S, Deretic V. Role of phosphatidylinositol 3-kinase and Rab5 effectors in phagosomal biogenesis and mycobacterial phagosome maturation arrest. J Cell Biol (2001) 154:631–44. doi: 10.1083/jcb.200106049
100. Lippe R, Miaczynska M, Rybin V, Runge A, Zerial M. Functional synergy between Rab5 effector Rabaptin-5 and exchange factor Rabex-5 when physically associated in a complex. Mol Biol Cell (2001) 12:2219–28. doi: 10.1091/mbc.12.7.2219
101. Yu B, Egbejimi A, Dharmat R, Xu P, Zhao Z, Long B, et al. Phagocytosed photoreceptor outer segments activate mTORC1 in the retinal pigment epithelium. Sci Signal (2018) 11. doi: 10.1126/scisignal.aag3315
102. Wensel TG. Phosphoinositides in Retinal Function and Disease. Cells (2020) 9. doi: 10.3390/cells9040866
103. Huynh KK, Eskelinen EL, Scott CC, Malevanets A, Saftig P, Grinstein S. LAMP proteins are required for fusion of lysosomes with phagosomes. EMBO J (2007) 26:313–24. doi: 10.1038/sj.emboj.7601511
104. Notomi S, Ishihara K, Efstathiou NE, Lee JJ, Hisatomi T, Tachibana T, et al. Genetic LAMP2 deficiency accelerates the age-associated formation of basal laminar deposits in the retina. Proc Natl Acad Sci U.S.A. (2019) 116:23724–34. doi: 10.1073/pnas.1906643116
105. Thompson DA, Constable PA, Liasis A, Walters B, Esteban MT. The Physiology of the Retinal Pigment Epithelium in Danon Disease. Retina (2016) 36:629–38. doi: 10.1097/IAE.0000000000000736
106. Fairn GD, Grinstein S. How nascent phagosomes mature to become phagolysosomes. Trends Immunol (2012) 33:397–405. doi: 10.1016/j.it.2012.03.003
107. Poteryaev D, Datta S, Ackema K, Zerial M, Spang A. Identification of the switch in early-to-late endosome transition. Cell (2010) 141:497–508. doi: 10.1016/j.cell.2010.03.011
108. Levin R, Grinstein S, Canton J. The life cycle of phagosomes: formation, maturation, and resolution. Immunol Rev (2016) 273:156–79. doi: 10.1111/imr.12439
109. Harrison RE, Bucci C, Vieira OV, Schroer TA, Grinstein S. Phagosomes fuse with late endosomes and/or lysosomes by extension of membrane protrusions along microtubules: role of Rab7 and RILP. Mol Cell Biol (2003) 23:6494–506. doi: 10.1128/MCB.23.18.6494-6506.2003
110. Johansson M, Lehto M, Tanhuanpaa K, Cover TL, Olkkonen VM. The oxysterol-binding protein homologue ORP1L interacts with Rab7 and alters functional properties of late endocytic compartments. Mol Biol Cell (2005) 16:5480–92. doi: 10.1091/mbc.e05-03-0189
111. Rai A, Pathak D, Thakur S, Singh S, Dubey AK, Mallik R. Dynein Clusters into Lipid Microdomains on Phagosomes to Drive Rapid Transport toward Lysosomes. Cell (2016) 164:722–34. doi: 10.1016/j.cell.2015.12.054
112. Herman KG, Steinberg RH. Phagosome degradation in the tapetal retinal pigment epithelium of the opossum. Invest Ophthalmol Vis Sci (1982) 23:291–304.
113. Keller G, Leuenberger PM. Effects of colchicine on phagosome-lysosome interaction in retinal pigment epithelium. II. In vitro observations on histio-organotypical retinal pigment epithelial cells of the pig (a preliminary report). Albrecht Von Graefes Arch Klin Exp Ophthalmol (1977) 203:253–9. doi: 10.1007/BF00409831
114. Weil D, Blanchard S, Kaplan J, Guilford P, Gibson F, Walsh J, et al. Defective myosin VIIA gene responsible for Usher syndrome type 1B. Nature (1995) 374:60–1. doi: 10.1038/374060a0
115. Williams DS, Lopes VS. The many different cellular functions of MYO7A in the retina. Biochem Soc Trans (2011) 39:1207–10. doi: 10.1042/BST0391207
116. Jiang M, Esteve-Rudd J, Lopes VS, Diemer T, Lillo C, Rump A, et al. Microtubule motors transport phagosomes in the RPE, and lack of KLC1 leads to AMD-like pathogenesis. J Cell Biol (2015) 210:595–611. doi: 10.1083/jcb.201410112
117. Pankiv S, Alemu EA, Brech A, Bruun JA, Lamark T, Overvatn A, et al. FYCO1 is a Rab7 effector that binds to LC3 and PI3P to mediate microtubule plus end-directed vesicle transport. J Cell Biol (2010) 188:253–69. doi: 10.1083/jcb.200907015
118. Hong Z, Pedersen NM, Wang L, Torgersen ML, Raiborg C. PtdIns3P controls mTORC1 signaling through lysosomal positioning. J Cell Biol (2017) 216:4217–33. doi: 10.1083/jcb.201611073
119. Gordiyenko NV, Fariss RN, Zhi C, MacDonald IM. Silencing of the CHM gene alters phagocytic and secretory pathways in the retinal pigment epithelium. Invest Ophthalmol Vis Sci (2010) 51:1143–50. doi: 10.1167/iovs.09-4117
120. Wavre-Shapton ST, Tolmachova T, Lopes da Silva M, Futter CE, Seabra MC. Conditional ablation of the choroideremia gene causes age-related changes in mouse retinal pigment epithelium. PloS One (2013) 8:e57769. doi: 10.1371/journal.pone.0057769
121. Deguchi J, Yamamoto A, Yoshimori T, Sugasawa K, Moriyama Y, Futai M, et al. Acidification of phagosomes and degradation of rod outer segments in rat retinal pigment epithelium. Invest Ophthalmol Vis Sci (1994) 35:568–79.
122. Nuckels RJ, Ng A, Darland T, Gross JM. The vacuolar-ATPase complex regulates retinoblast proliferation and survival, photoreceptor morphogenesis, and pigmentation in the zebrafish eye. Invest Ophthalmol Vis Sci (2009) 50:893–905. doi: 10.1167/iovs.08-2743
123. Wavre-Shapton ST, Meschede IP, Seabra MC, Futter CE. Phagosome maturation during endosome interaction revealed by partial rhodopsin processing in retinal pigment epithelium. J Cell Sci (2014) 127:3852–61. doi: 10.1242/jcs.154757
124. Liu J, Lu W, Reigada D, Nguyen J, Laties AM, Mitchell CH. Restoration of lysosomal pH in RPE cells from cultured human and ABCA4(-/-) mice: pharmacologic approaches and functional recovery. Invest Ophthalmol Vis Sci (2008) 49:772–80. doi: 10.1167/iovs.07-0675
125. Saric A, Grinstein S, Freeman SA. Measurement of Autolysosomal pH by Dual-Wavelength Ratio Imaging. Methods Enzymol (2017) 588:15–29. doi: 10.1016/bs.mie.2016.09.073
126. Webb JCBA, Wittmann T, Barber DL. pHLARE: A Genetically Encoded Ratiometric Lysosome pH Biosensor. bioRxiv (2020). doi: 10.1101/2020.06.03.132720
127. Valapala M, Sergeev Y, Wawrousek E, Hose S, Zigler JS Jr, Sinha D. Modulation of V-ATPase by betaA3/A1-Crystallin in Retinal Pigment Epithelial Cells. Adv Exp Med Biol (2016) 854:779–84. doi: 10.1007/978-3-319-17121-0_104
128. Zigler JS Jr., Zhang C, Grebe R, Sehrawat G, Hackler L Jr., Adhya S, et al. Mutation in the betaA3/A1-crystallin gene impairs phagosome degradation in the retinal pigmented epithelium of the rat. J Cell Sci (2011) 124:523–31. doi: 10.1242/jcs.078790
129. Steinberg BE, Huynh KK, Brodovitch A, Jabs S, Stauber T, Jentsch TJ, et al. A cation counterflux supports lysosomal acidification. J Cell Biol (2010) 189:1171–86. doi: 10.1083/jcb.200911083
130. Kasper D, Planells-Cases R, Fuhrmann JC, Scheel O, Zeitz O, Ruether K, et al. Loss of the chloride channel ClC-7 leads to lysosomal storage disease and neurodegeneration. EMBO J (2005) 24:1079–91. doi: 10.1038/sj.emboj.7600576
131. Stobrawa SM, Breiderhoff T, Takamori S, Engel D, Schweizer M, Zdebik AA, et al. Disruption of ClC-3, a chloride channel expressed on synaptic vesicles, leads to a loss of the hippocampus. Neuron (2001) 29:185–96. doi: 10.1016/S0896-6273(01)00189-1
132. Goldberg AF, Moritz OL, Williams DS. Molecular basis for photoreceptor outer segment architecture. Prog Retin Eye Res (2016) 55:52–81. doi: 10.1016/j.preteyeres.2016.05.003
133. Freeman SA, Grinstein S. Resolution of macropinosomes, phagosomes and autolysosomes: Osmotically driven shrinkage enables tubulation and vesiculation. Traffic (2018) 19:965–74. doi: 10.1111/tra.12614
134. Papermaster DS, Dreyer WJ. Rhodopsin content in the outer segment membranes of bovine and frog retinal rods. Biochemistry (1974) 13:2438–44. doi: 10.1021/bi00708a031
135. Zimmerman WF, Godchaux W,3, Belkin M. The relative proportions of lysosomal enzyme activities in bovine retinal pigment epithelium. Exp Eye Res (1983) 36:151–8. doi: 10.1016/0014-4835(83)90098-2
136. Rakoczy PE, Zhang D, Robertson T, Barnett NL, Papadimitriou J, Constable IJ, et al. Progressive age-related changes similar to age-related macular degeneration in a transgenic mouse model. Am J Pathol (2002) 161:1515–24. doi: 10.1016/S0002-9440(10)64427-6
137. Rakoczy PE, Lai MC, Baines MG, Spilsbury K, Constable IJ. Expression of cathepsin S antisense transcripts by adenovirus in retinal pigment epithelial cells. Invest Ophthalmol Vis Sci (1998) 39:2095–104.
138. Martin RE, Elliott MH, Brush RS, Anderson RE. Detailed characterization of the lipid composition of detergent-resistant membranes from photoreceptor rod outer segment membranes. Invest Ophthalmol Vis Sci (2005) 46:1147–54. doi: 10.1167/iovs.04-1207
139. Albert A, Alexander D, Boesze-Battaglia K. Cholesterol in the rod outer segment: A complex role in a “simple” system. Chem Phys Lipids (2016) 199:94–105. doi: 10.1016/j.chemphyslip.2016.04.008
140. Swartz JG, Mitchell JE. Phospholipase activity of retina and pigment epithelium. Biochemistry (1973) 12:5273–8. doi: 10.1021/bi00750a008
141. Thelen AM, Zoncu R. Emerging Roles for the Lysosome in Lipid Metabolism. Trends Cell Biol (2017) 27:833–50. doi: 10.1016/j.tcb.2017.07.006
142. Infante RE, Wang ML, Radhakrishnan A, Kwon HJ, Brown MS, Goldstein JL. NPC2 facilitates bidirectional transfer of cholesterol between NPC1 and lipid bilayers, a step in cholesterol egress from lysosomes. Proc Natl Acad Sci U.S.A. (2008) 105:15287–92. doi: 10.1073/pnas.0807328105
143. Heybrock S, Kanerva K, Meng Y, Ing C, Liang A, Xiong ZJ, et al. Lysosomal integral membrane protein-2 (LIMP-2/SCARB2) is involved in lysosomal cholesterol export. Nat Commun (2019) 10:3521. doi: 10.1038/s41467-019-11425-0
144. Claudepierre T, Paques M, Simonutti M, Buard I, Sahel J, Maue RA, et al. Lack of Niemann-Pick type C1 induces age-related degeneration in the mouse retina. Mol Cell Neurosci (2010) 43:164–76. doi: 10.1016/j.mcn.2009.10.007
145. Kennedy CJ, Rakoczy PE, Constable IJ. Lipofuscin of the retinal pigment epithelium: a review. Eye (Lond) (1995) 9( Pt 6):763–71. doi: 10.1038/eye.1995.192
146. Usui T, Sawaguchi S, Abe H, Iwata K, Oyanagi K. Late-infantile type galactosialidosis. Histopathology of the retina and optic nerve. Arch Ophthalmol (1991) 109:542–6. doi: 10.1001/archopht.1991.01080040110040
147. Matlach J, Zindel T, Amraoui Y, Arash-Kaps L, Hennermann JB, Pitz S. Retinal and optic nerve degeneration in alpha-mannosidosis. Orphanet J Rare Dis (2018) 13:88. doi: 10.1186/s13023-018-0829-z
148. Ishikawa M, Sawada Y, Yoshitomi T. Structure and function of the interphotoreceptor matrix surrounding retinal photoreceptor cells. Exp Eye Res (2015) 133:3–18. doi: 10.1016/j.exer.2015.02.017
149. Ashworth JL, Biswas S, Wraith E, Lloyd IC. Mucopolysaccharidoses and the eye. Surv Ophthalmol (2006) 51:1–17. doi: 10.1016/j.survophthal.2005.11.007
150. Yu L, McPhee CK, Zheng L, Mardones GA, Rong Y, Peng J, et al. Termination of autophagy and reformation of lysosomes regulated by mTOR. Nature (2010) 465:942–6. doi: 10.1038/nature09076
151. Wyant GA, Abu-Remaileh M, Wolfson RL, Chen WW, Freinkman E, Danai LV, et al. mTORC1 Activator SLC38A9 Is Required to Efflux Essential Amino Acids from Lysosomes and Use Protein as a Nutrient. Cell (2017) 171:642–654 e12. doi: 10.1016/j.cell.2017.09.046
152. Wang S, Tsun ZY, Wolfson RL, Shen K, Wyant GA, Plovanich ME, et al. Metabolism. Lysosomal amino acid transporter SLC38A9 signals arginine sufficiency to mTORC1. Science (2015) 347:188–94. doi: 10.1126/science.1257132
153. Amick J, Tharkeshwar AK, Talaia G, Ferguson SM. PQLC2 recruits the C9orf72 complex to lysosomes in response to cationic amino acid starvation. J Cell Biol (2020) 219. doi: 10.1083/jcb.201906076
154. Smith DE, Clemencon B, Hediger MA. Proton-coupled oligopeptide transporter family SLC15: physiological, pharmacological and pathological implications. Mol Aspects Med (2013) 34:323–36. doi: 10.1016/j.mam.2012.11.003
155. Wang X, Zhang X, Dong XP, Samie M, Li X, Cheng X, et al. TPC proteins are phosphoinositide- activated sodium-selective ion channels in endosomes and lysosomes. Cell (2012) 151:372–83. doi: 10.1016/j.cell.2012.08.036
156. Gomez NM, Lu W, Lim JC, Kiselyov K, Campagno KE, Grishchuk Y, et al. Robust lysosomal calcium signaling through channel TRPML1 is impaired by lysosomal lipid accumulation. FASEB J (2018) 32:782–94. doi: 10.1096/fj.201700220RR
157. Nicklin P, Bergman P, Zhang B, Triantafellow E, Wang H, Nyfeler B, et al. Bidirectional transport of amino acids regulates mTOR and autophagy. Cell (2009) 136:521–34. doi: 10.1016/j.cell.2008.11.044
158. Baehr W, Wu SM, Bird AC, Palczewski K. The retinoid cycle and retina disease. Vision Res (2003) 43:2957–8. doi: 10.1016/j.visres.2003.10.001
159. Travis GH, Golczak M, Moise AR, Palczewski K. Diseases caused by defects in the visual cycle: retinoids as potential therapeutic agents. Annu Rev Pharmacol Toxicol (2007) 47:469–512. doi: 10.1146/annurev.pharmtox.47.120505.105225
160. Tinoco J, Miljanich P, Medwadowski B. Depletion of docosahexaenoic acid in retinal lipids of rats fed a linolenic acid-deficient, linoleic acid-containing diet. Biochim Biophys Acta (1977) 486:575–8. doi: 10.1016/0005-2760(77)90111-4
161. Gordon WC, Rodriguez de Turco EB, Bazan NG. Retinal pigment epithelial cells play a central role in the conservation of docosahexaenoic acid by photoreceptor cells after shedding and phagocytosis. Curr Eye Res (1992) 11:73–83. doi: 10.3109/02713689209069169
162. Bazan NG, Calandria JM, Serhan CN. Rescue and repair during photoreceptor cell renewal mediated by docosahexaenoic acid-derived neuroprotectin D1. J Lipid Res (2010) 51:2018–31. doi: 10.1194/jlr.R001131
163. Kauppinen A, Paterno JJ, Blasiak J, Salminen A, Kaarniranta K. Inflammation and its role in age-related macular degeneration. Cell Mol Life Sci (2016) 73:1765–86. doi: 10.1007/s00018-016-2147-8
164. Kanow MA, Giarmarco MM, Jankowski CS, Tsantilas K, Engel AL, Du J, et al. Biochemical adaptations of the retina and retinal pigment epithelium support a metabolic ecosystem in the vertebrate eye. Elife (2017) 6. doi: 10.7554/eLife.28899
165. Weh E, Lutrzykowska Z, Smith A, Hager H, Pawar M, Wubben TJ, et al. Hexokinase 2 is dispensable for photoreceptor development but is required for survival during aging and outer retinal stress. Cell Death Dis (2020) 11:422. doi: 10.1038/s41419-020-2638-2
166. Storti F, Raphael G, Griesser V, Klee K, Drawnel F, Willburger C, et al. Regulated efflux of photoreceptor outer segment-derived cholesterol by human RPE cells. Exp Eye Res (2017) 165:65–77. doi: 10.1016/j.exer.2017.09.008
167. Adijanto J, Du J, Moffat C, Seifert EL, Hurle JB, Philp NJ. The retinal pigment epithelium utilizes fatty acids for ketogenesis. J Biol Chem (2014) 289:20570–82. doi: 10.1074/jbc.M114.565457
168. Joyal JS, Sun Y, Gantner ML, Shao Z, Evans LP, Saba N, et al. Retinal lipid and glucose metabolism dictates angiogenesis through the lipid sensor Ffar1. Nat Med (2016) 22:439–45. doi: 10.1038/nm.4059
169. Fu Z, Kern TS, Hellstrom A, Smith L. Fatty acid oxidation and photoreceptor metabolic needs. J Lipid Res (2020). doi: 10.1194/jlr.TR120000618
170. Zhou X, Franklin RA, Adler M, Jacox JB, Bailis W, Shyer JA, et al. Circuit Design Features of a Stable Two-Cell System. Cell (2018) 172:744–757 e17. doi: 10.1016/j.cell.2018.01.015
Keywords: cholesterol, integrins, actin cytoskeleton, MerTK, glucose transport, V-ATPase, resolvins, phosphatidylserine (PtdSer)
Citation: Kwon W and Freeman SA (2020) Phagocytosis by the Retinal Pigment Epithelium: Recognition, Resolution, Recycling. Front. Immunol. 11:604205. doi: 10.3389/fimmu.2020.604205
Received: 08 September 2020; Accepted: 20 October 2020;
Published: 13 November 2020.
Edited by:
Gregory D. Fairn, St. Michael’s Hospital, CanadaReviewed by:
Tobias Dallenga, Research Center Borstel (LG), GermanyRaymond B. Birge, The State University of New Jersey, United States
Copyright © 2020 Kwon and Freeman. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Spencer A. Freeman, c3BlbmNlci5mcmVlbWFuQHNpY2traWRzLmNh
 Whijin Kwon
Whijin Kwon Spencer A. Freeman
Spencer A. Freeman