- Division of Dermatology, Department of Medicine, Faculty of Medicine Ramathibodi Hospital, Mahidol University, Bangkok, Thailand
Background: Primary cicatricial alopecia (PCA) is known to be associated with various comorbidities; however, findings regarding the likelihood of specific comorbidities in PCA patients have been inconsistent.
Objective: This study aimed to assess the prevalence and odds of specific comorbidities in patients with PCA compared to controls, and to explore the distribution of comorbidities across various types of PCA.
Methods: Electronic searches were conducted using PubMed, Embase, and Scopus from the dates of their inception until July 2024. A total of 116 studies with 33,494 PCA patients that reported data allowing for the calculation of odds ratios (OR) or prevalences of certain comorbidities in PCA patients were included.
Results: Systemic lupus erythematosus is more prevalent among patients with lichen planopilaris (LPP) [OR 3.10 (95% confidence interval: 2.24–4.29), prevalence 2%], frontal fibrosing alopecia (FFA) [OR 6.92 (2.73–17.56), prevalence 5%], and central centrifugal cicatricial alopecia (CCCA) [OR 3.13 (1.03–9.49), prevalence 5%]. Hypothyroidism is more prevalent among patients with LPP [OR of 1.73 (1.24–2.42), prevalence 17%] and FFA [OR 1.86 (1.36–2.55), prevalence 19%]. LPP patients are prone to having dermatological diseases such as atopic dermatitis [OR 3.96 (1.14–13.81), prevalence 9%], lichen planus [OR 19.21 (1.47–251.02), prevalence 8%], psoriasis [OR 4.75 (2.04–11.06), prevalence 3%], and rosacea [OR 4.62 (2.96–7.19), prevalence 5%], while FFA patients are prone to having allergic contact dermatitis [OR 3.19 (1.44–7.08), prevalence 41%] and rosacea [OR 2.37 (1.72–3.29), prevalence 16%]. Coronary artery disease is found to be more common in LPP than controls [OR 1.63 (1.43–1.86), prevalence 8%], while dyslipidemia is more common among FFA [OR 1.41 (1.06–1.88), prevalence 20%] and CCCA [OR 4.46 (1.01–19.75), prevalence 54%] than controls, and diabetes mellitus is more prevalent among CCCA than controls [OR 1.67 (1.03–2.69), prevalence 26%]. While skin cancer [OR 2.22 (1.33–3.70), prevalence 2%] and melanoma [OR 4.46 (1.70–11.76), prevalence 1%] were found to be more common in LPP than controls, rheumatoid arthritis [OR 1.65 (1.09–2.51), prevalence 4%] was found to be more common in FFA than controls, and allergic rhinitis [OR 11.77 (1.55–89.24), prevalence 24%] and anxiety [OR 4.69 (1.29–16.98), prevalence 17%] were found to be more common in CCCA than controls.
Conclusions: Patients with PCA are at higher risk of developing a wide range of comorbidities. Physicians should remain vigilant and conduct thorough investigations when clinical clues are present.
Systematic Review Registration: https://www.crd.york.ac.uk/prospero/display_record.php?RecordID=564852, identifier CRD42024564852.
1 Introduction
Cicatricial alopecia, also known as scarring alopecia, results from inflammatory damage to the epithelial stem cells in the follicular bulge region, leading to fibrosis and irreversible hair loss (1). Primary cicatricial alopecia (PCA) occurs when the hair follicle is the primary target of inflammation process, while in secondary cicatricial alopecia, the hair follicle is merely a bystander to the disease process (1). The North American Hair Research Society has proposed classifying PCA into lymphocytic, neutrophilic, mixed, and nonspecific types (2). Lymphocytic PCA encompasses chronic cutaneous lupus erythematosus, classic lichen planopilaris (LPP), frontal fibrosing alopecia (FFA), Graham-Little syndrome, pseudopelade of Brocq (PPB), central centrifugal cicatricial alopecia (CCCA), alopecia mucinosa, and keratosis follicularis spinulosa decalvans (2). Neutrophilic PCA includes folliculitis decalvans (FD) and dissecting cellulitis (DC), while mixed PCA consists of acne keloidalis nuchae (AKN), acne necrotica, and erosive pustular dermatosis (EPD) (2).
Understanding the comorbidities associated with PCA is essential, as they can provide insight into disease etiology and are vital for successful interdisciplinary management. Previous studies on PCA have produced inconsistent findings regarding the prevalence of certain comorbidities and odds of PCA patients having them, likely due to the rarity of PCA and small sample sizes. We aimed to systematically investigate the prevalence of comorbidities and their association with each specific type of PCA, helping guide clinical practice and improve patient care.
2 Materials and methods
2.1 Study design
The protocol for this analysis was registered in PROSPERO (International Prospective Register of Systematic Reviews; CRD42024564852, https://www.crd.york.ac.uk/prospero/display_record.php?RecordID=564852). The systematic review followed the Preferred Reporting Items for Systematic Reviews and Meta-analyses guidelines (Supplementary Document) (3). Electronic searches were conducted from the database’s inception to July 2024 using the PubMed, Embase, and Scopus databases. The search strategy was designed to retrieve all studies on PCA using keywords and a controlled vocabulary. There were no restrictions on the language or publication period in the searches. Only full-text articles were considered, and conference abstracts were excluded due to limited detail and lack of peer review. The search included a combination of terms: ‘primary scarring alopecia’, ‘primary cicatricial alopecia’, ‘lichen planopilaris’, ‘frontal fibrosing alopecia’, ‘Graham Little syndrome’, ‘fibrosing alopecia in a pattern distribution’, ‘pseudopelade of Brocq’, ‘central centrifugal cicatricial alopecia’, ‘discoid lupus erythematosus’, ‘alopecia mucinosa’, ‘keratosis follicularis spinulosa decalvans’, ‘folliculitis decalvans’, ‘dissecting cellulitis’, ‘acne keloidalis’, and ‘erosive pustular dermatosis.’ Supplementary Table 1 provides details about the search strategy. Grey literature and unpublished data were not considered. If multiple studies utilized patient data from the same database with an overlapping inclusion period, data from the more recent publication or from a case-control study is selected. The comorbid data were not quantitatively utilized if they were reported in a composite manner, such as LPP/FFA or PCA.
2.2 Study selection
Two reviewers (TY and KT) independently evaluated each article at both the full-text and title/abstract levels. Full texts of potentially eligible studies were assessed for inclusion. Disagreements regarding the studies’ eligibility were resolved via discussion with a third reviewer (PS). Inclusion criteria were: (1) randomized controlled trials, cohort studies, cross-sectional studies, and case-control studies; (2) studies involving patients with confirmed diagnosis of PCA based on clinical and/or histopathological criteria; (3) studies reporting data allowing calculation of odds ratios (OR) or prevalences of specific comorbidities; (4) full-text articles in any language. Exclusion criteria were: (1) conference abstracts; (2) case reports or case series; (3) studies without clear diagnostic criteria for PCA; (4) studies reporting only composite PCA data without subtype specification. Comorbidities were defined based on clinical diagnosis using ICD codes, physician diagnosis, or validated diagnostic criteria as reported in each study.
2.3 Data extraction
Data were extracted from the included studies using a standardized form. The following data were collected: bibliographic data (authors, year of publication), study characteristics (type of study, single or multicenter, study duration, country), alopecia group characteristics (number, age, gender, ethnicity, body mass index (BMI), comorbidity), and treatment information), control group characteristics (number, age, gender, ethnicity, BMI, comorbidity, whether controls were matched for any relevant factors), and comorbidity data.
Corresponding investigators were contacted via email if there was missing data. Two independent reviewers (TY and KT) extracted data, and any discrepancies were discussed and resolved with input from a third reviewer (PS).
2.4 Quality assessment
TY and KT independently assessed the quality of descriptive and case-control studies using the Newcastle-Ottawa Scale (NOS) (4). The NOS is a scoring tool comprised of seven items with nine scores that assess how well the investigators selected their participants (score ranges from 0 to 4), the comparability of their results (score ranges from 0 to 2), and the applicability of the outcomes (score ranges from 0 to 3). The higher the score, the higher the study’s quality and the lower the likelihood of bias. Therefore, we classified studies as having high quality if they received a total score of 7 or more, fair quality if they received a score of 4-6, and low quality if they received a score of less than 4. Sensitivity analyses were performed excluding fair-quality studies to assess the robustness of our findings. Meta-analyses were weighted by study quality, with higher-quality studies given greater influence in pooled estimates. Any discrepancies between reviewers regarding the risk of bias in specific studies were resolved through discussion with a third reviewer (PS).
2.5 Statistical analysis
A meta-analysis was performed, using an inverse variance method, to pool the ORs of a specific PCA and a specific comorbidity, as well as the prevalences of various comorbidities associated with a specific PCA. Each PCA disorder was analyzed separately. Quantitative analyses were conducted on comorbidity, either OR or prevalence, that were reported in at least two studies; otherwise, they were only analyzed qualitatively.
Heterogeneity was assessed and considered present if a Cochrane Q test p-value was < 0.1 or Higgins I2 ≥ 25% (5). The sources of heterogeneity were explored by fitting each covariate (e.g., age, female gender, and BMI) at a time in a meta-regression model. If the τ2 was decreased by ≥ 50% or statistically significant β was revealed, a subgroup analysis was performed based on that covariate if possible (6).
To evaluate publication bias, Deeks funnel plots of the primary outcomes were generated. The Egger linear regression test was applied when a funnel plot suggested possible asymmetry (7). If Egger’s test for a regression intercept gave a p-value < 0.05, a trim and fill method was used to adjust the OR (7). Comprehensive Meta-Analysis software (version 3.3.070, Biostat, Englewood, NJ) was used for all statistical analysis.
3 Results
3.1 Study characteristics
After removing duplicates, 4,797 references were screened by title and abstract. At the full-text stage, 308 full articles met our predefined selection criteria and were sought. We further excluded 192 references for the following reasons: conference abstract or review article (n = 60), not population of interest (i.e., non-alopecia or non-scarring alopecia diagnosis, n = 15), not outcome of interest (i.e., no documented comorbidity prevalence of the patients, n = 115), and duplicate patient data (n = 2) (Figure 1). The review included 116 studies, enrolling a total of 33,494 patients with PCA [8,871 LPP patients (8–31), 6,595 FFA patients (12, 16, 19, 21, 23, 25, 27, 30, 32–83), 3,539 CCCA patients (24, 84–99), 157 PPB patients (20), 24 patients with fibrosing alopecia in a pattern distribution (FAPD) (55), 6,158 FD patients (20, 100–104), 4,752 DC patients (20, 105–108), 3,218 AKN patients (109–120), and 180 EPD patients (121–124)] between 2009 and 2024, were included in the review. Characteristic features of the included studies are provided in Supplementary Tables 2-6.
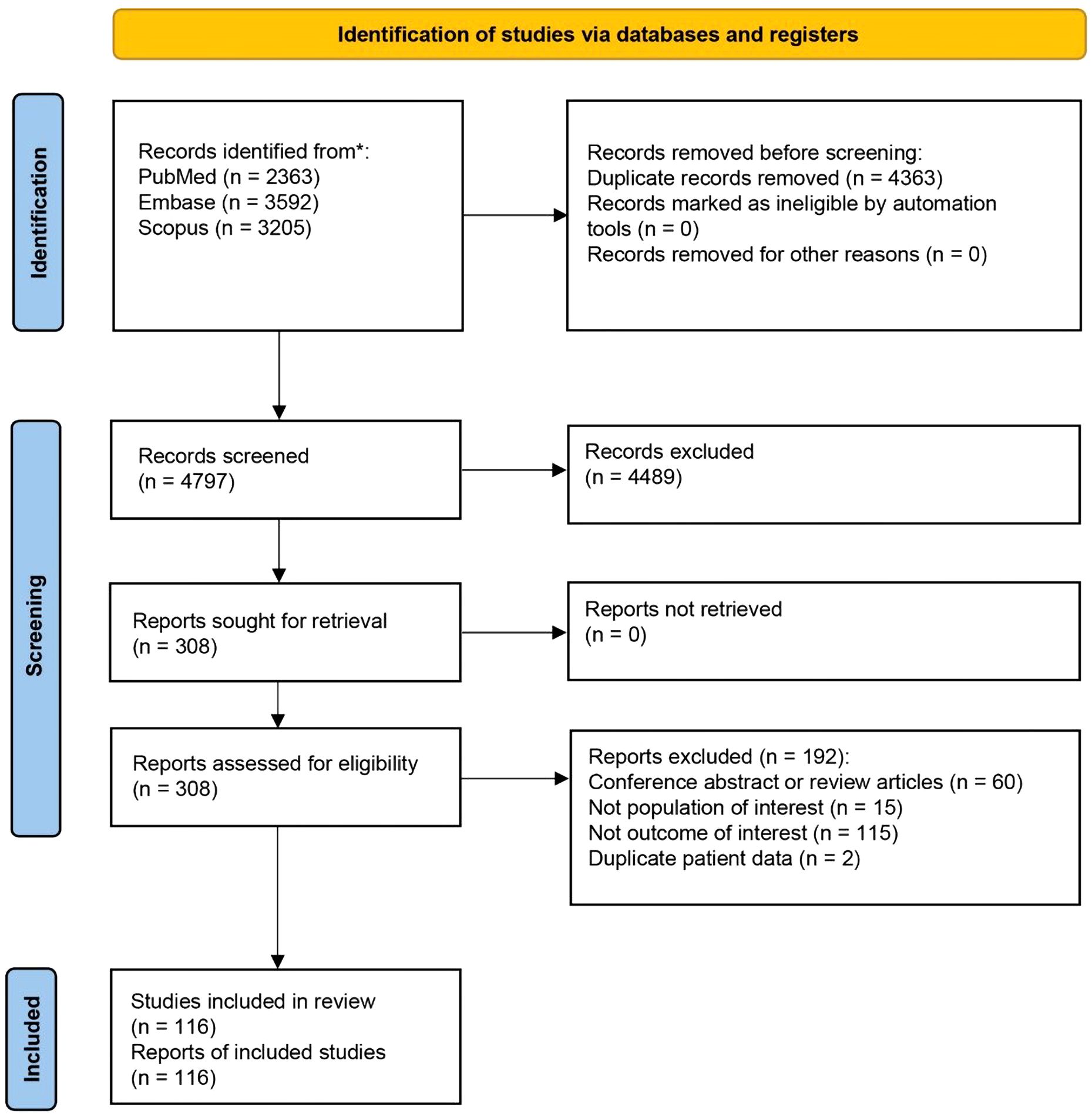
Figure 1. Preferred reporting items for systematic reviews and meta-analyses (PRISMA) flow diagram of search strategy and included studies.
3.2 Lichen planopilaris
3.2.1 Autoimmune diseases
LPP was found to be significantly associated with systemic lupus erythematosus (SLE) [OR 3.10 (95% confidence interval: 2.24–4.29), prevalence 2%]. Inflammatory bowel diseases [OR 2.49 (0.91–6.77), prevalence 5%], including Crohn’s disease [OR 0.75 (0.37–1.49), prevalence 1%] and ulcerative colitis [OR 1.23 (0.32–4.74), prevalence 2%], were not statistically associated with LPP, as were other autoimmune diseases, such as ankylosing spondylitis [OR 1.07 (0.83–1.39), prevalence 1%], celiac disease [OR 1.06 (0.34–3.26), prevalence 2%], multiple sclerosis [OR 1.76 (0.65–4.74), prevalence <1%], rheumatoid arthritis [OR 1.59 (0.90–2.78), prevalence 3%], Sjogren’s syndrome [OR 1.24 (0.81–1.89), prevalence 1%], and type 1 diabetes mellitus [OR 0.77 (0.26–2.27), prevalence 1%].
3.2.2 Cardiovascular and metabolic diseases
Coronary artery disease was found to be more common in LPP than controls [OR 1.63 (1.43–1.86), prevalence 8%]. However, we did not find a statistically significant association between LPP and cardiovascular diseases in general [OR 0.74 (0.48–1.16), prevalence 10%] or other cardiovascular comorbidities like congestive heart failure [OR 0.61 (0.15–2.44), prevalence 4%], myocardial infarction [OR 1.27 (0.87–1.86), prevalence 2%], peripheral artery disease [OR 1.03 (0.68–1.58), prevalence 5%], and stroke [0.57 (0.11–2.84), prevalence 3%]. Interestingly, Kim et al. have reported an adjusted hazard ratio (aHR) of 1.93 (1.07–3.49) for coronary heart disease in their cohort of LPP/FFA patients, while aHR of 1.18 (0.69–2.04) and 0.60 (0.23–1.60) were found for cardiovascular disease and stroke, respectively (125).
None of the metabolic diseases, such as diabetes mellitus [OR 0.87 (0.52–1.47), prevalence 10%], dyslipidemia [OR 1.36 (0.64–2.92), prevalence 27%], hypertension [OR 1.21 (0.63–2.33), prevalence 23%], and obesity [OR 1.82 (0.51–6.45), prevalence 20%], were found to be significantly associated with LPP.
3.2.3 Dermatologic diseases
Lichen planus was found to be strongly associated with LPP [OR 19.21 (1.47–251.02), prevalence 8%]. Other dermatological diseases such as atopic dermatitis [OR 3.96 (1.14–13.81), prevalence 9%], psoriasis [OR 4.75 (2.04–11.06), prevalence 3%], and rosacea [OR 4.62 (2.96–7.19), prevalence 5%] were also found to be significantly associated with LPP. Skin cancer [OR 2.22 (1.33–3.70), prevalence 2%] and melanoma [OR 4.46 (1.70–11.76), prevalence 1%] were also found to be more prevalent in LPP than controls. For alopecia areata (AA), however, the association was not statistically significant [OR 2.56 (0.26–25.76), prevalence 3%].
3.2.4 Thyroid disorders
Thyroid diseases, in general, were found to be associated with LPP [OR 2.64 (1.13–6.21), prevalence 24%]. A statistically significant association was found between LPP and hypothyroidism [OR 1.73 (1.24–2.42), prevalence 17%], hyperthyroidism [OR 1.17 (1.00–1.36), prevalence 1%], and thyroiditis [OR 1.21 (1.00–1.46), prevalence 6%]. Although a statistically significant association was found between Hashimoto thyroiditis [OR 4.24 (1.04–17.24), prevalence 7%] and LPP, a publication bias was identified (p=0.014, Egger’s test). After to a trim and fill analysis to account for the publication bias, the association was no longer statistically significant [adjusted OR 1.72 (0.54–5.41)].
3.2.5 Other diseases
Allergic rhinitis [OR 1.05 (0.50–2.19), prevalence 12%], anxiety [OR 1.19 (0.94–1.51), prevalence 22%], asthma [OR 0.91 (0.53–1.56), prevalence 8%], depression [OR 0.85 (0.45–1.61), prevalence 15%], and vitamin D deficiency [OR 0.91 (0.33–2.54), prevalence 19%] were not statistically associated with LPP.
Table 1 provides a summary of the pooled OR for the described comorbidities of LPP patients, along with the associated pooled prevalences. Figure 2, Supplementary Figure 1 depict the pooled OR and prevalence of the comorbidities in patients with LPP, respectively.
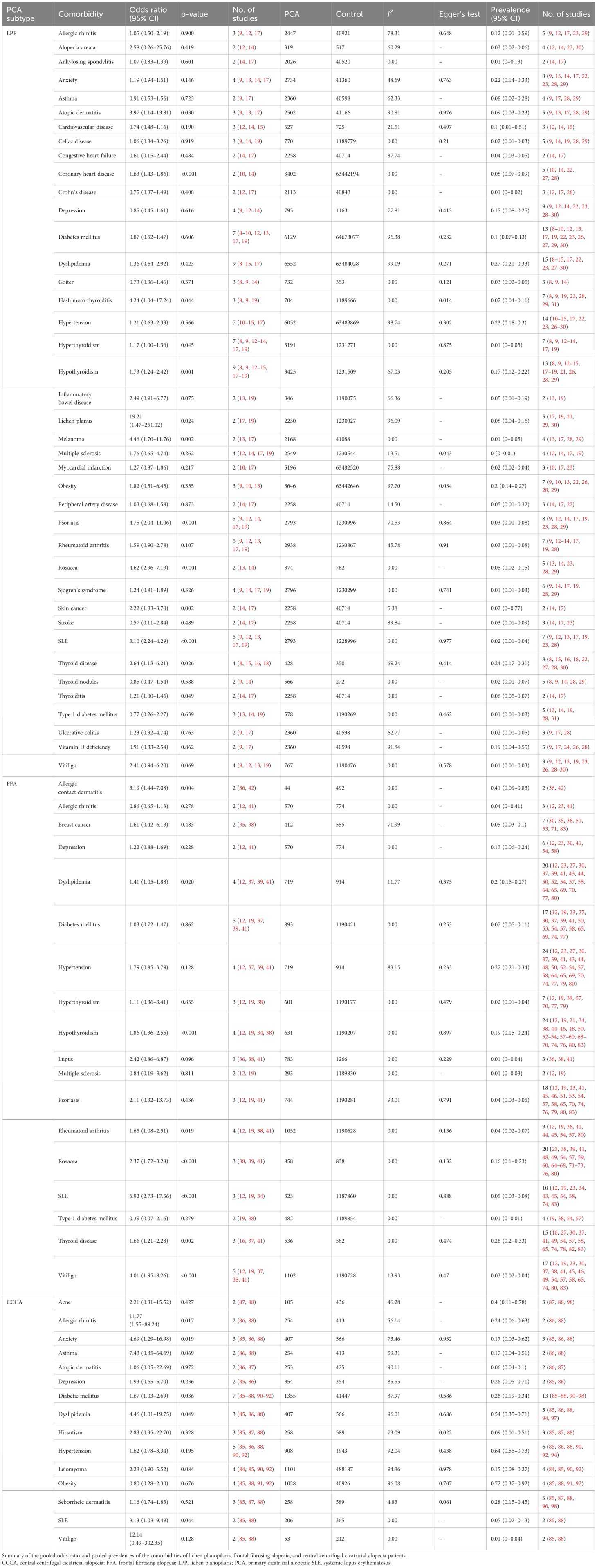
Table 1. Summary of the pooled odds ratio and pooled prevalences of the comorbidities of lichen planopilaris, frontal fibrosing alopecia, and central centrifugal cicatricial alopecia patients.
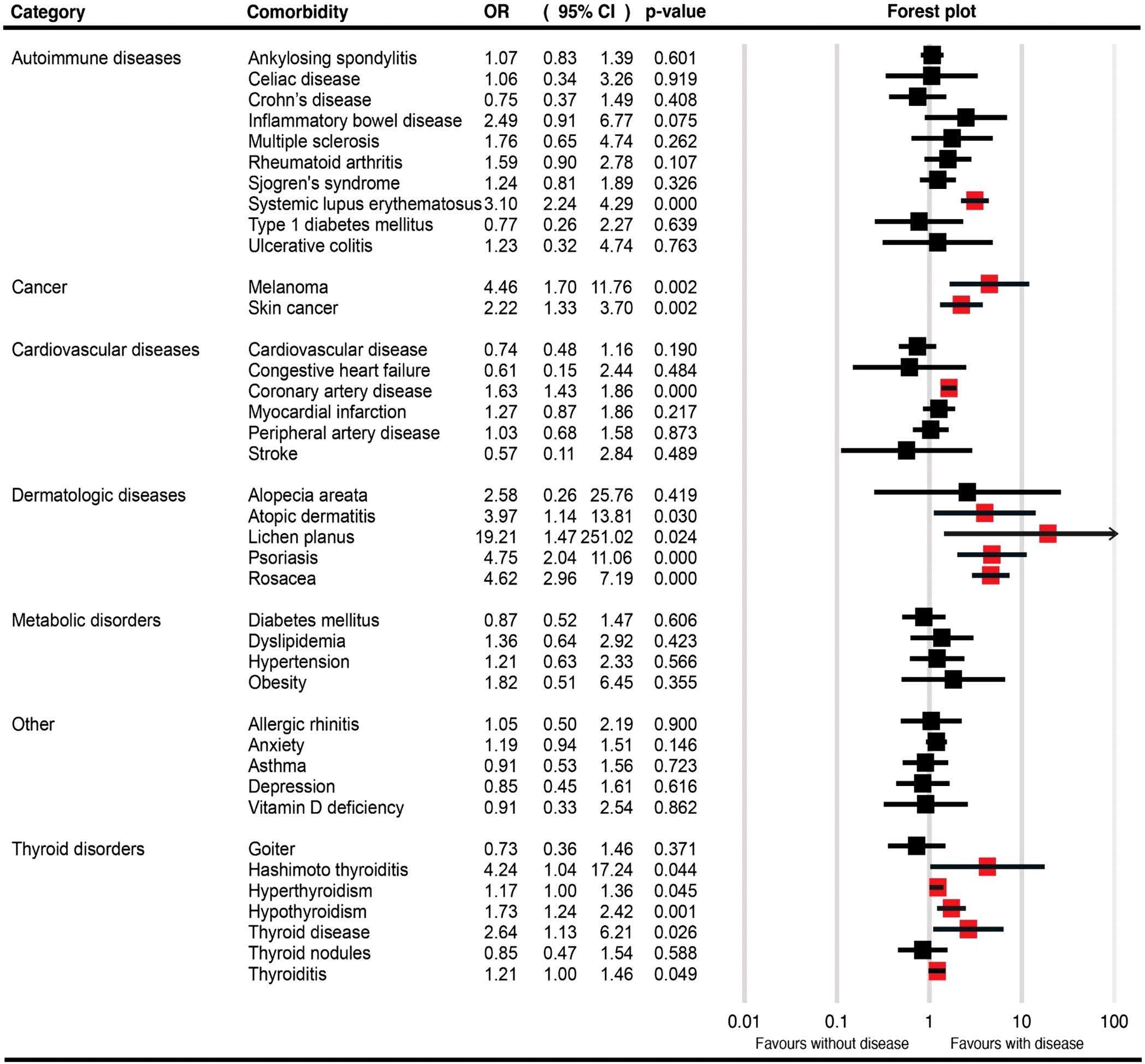
Figure 2. Forest plots for the pooled odds ratio of various comorbidities in patients with lichen planopilaris.
3.3 Frontal fibrosing alopecia
3.3.1 Autoimmune diseases
The results showed a strong association between SLE [OR 6.92 (2.73–17.56), prevalence 5%] and FFA. We also discovered a statistical association between FFA and rheumatoid arthritis [OR 1.65 (1.08–2.51), prevalence 4%], but not with multiple sclerosis [OR 0.84 (0.19–3.62), prevalence 4%] or type 1 diabetes mellitus [OR 0.39 (0.07–2.16), prevalence 1%].
3.3.2 Dermatologic diseases
Allergic contact dermatitis [OR 3.19 (1.44–7.08), prevalence 41%] and rosacea [OR 2.37 (1.72–3.28), prevalence 16%] were found to be associated with FFA; however, for psoriasis [OR 2.11 (0.32–13.73), prevalence 4%], the association was not statistically significant.
3.3.3 Metabolic disorders
Dyslipidemia [OR 1.41 (1.05–1.88), prevalence 20%] was found to be associated with FFA, while diabetes mellitus [OR 1.03 (0.72–1.47), prevalence 7%] and hypertension [OR 1.79 (0.85–3.79), prevalence 27%] were not statistically associated with FFA.
3.3.4 Thyroid disorders
Thyroid disease in general [OR 1.66 (1.21–2.28), prevalence 26%] was found to have statistical association with FFA; however, only hypothyroidism [OR 1.86 (1.36–2.55), prevalence 19%] was statistically associated with FFA, not hyperthyroidism [OR 1.11 (0.36–3.41), prevalence 2%].
3.3.5 Other diseases
Breast cancer [OR 1.61 (0.42–6.13), prevalence 5%], allergic rhinitis [OR 0.86 (0.65–1.13), prevalence 4%], and depression [OR 1.22 (0.88–1.69), prevalence 13%] were not statistically associated with FFA.
Table 1 summarizes the pooled OR for the described comorbidities of FFA patients and the associated pooled prevalences. Figure 3, Supplementary Figure 2 present the pooled OR and prevalence of the comorbidities in patients with FFA, respectively.
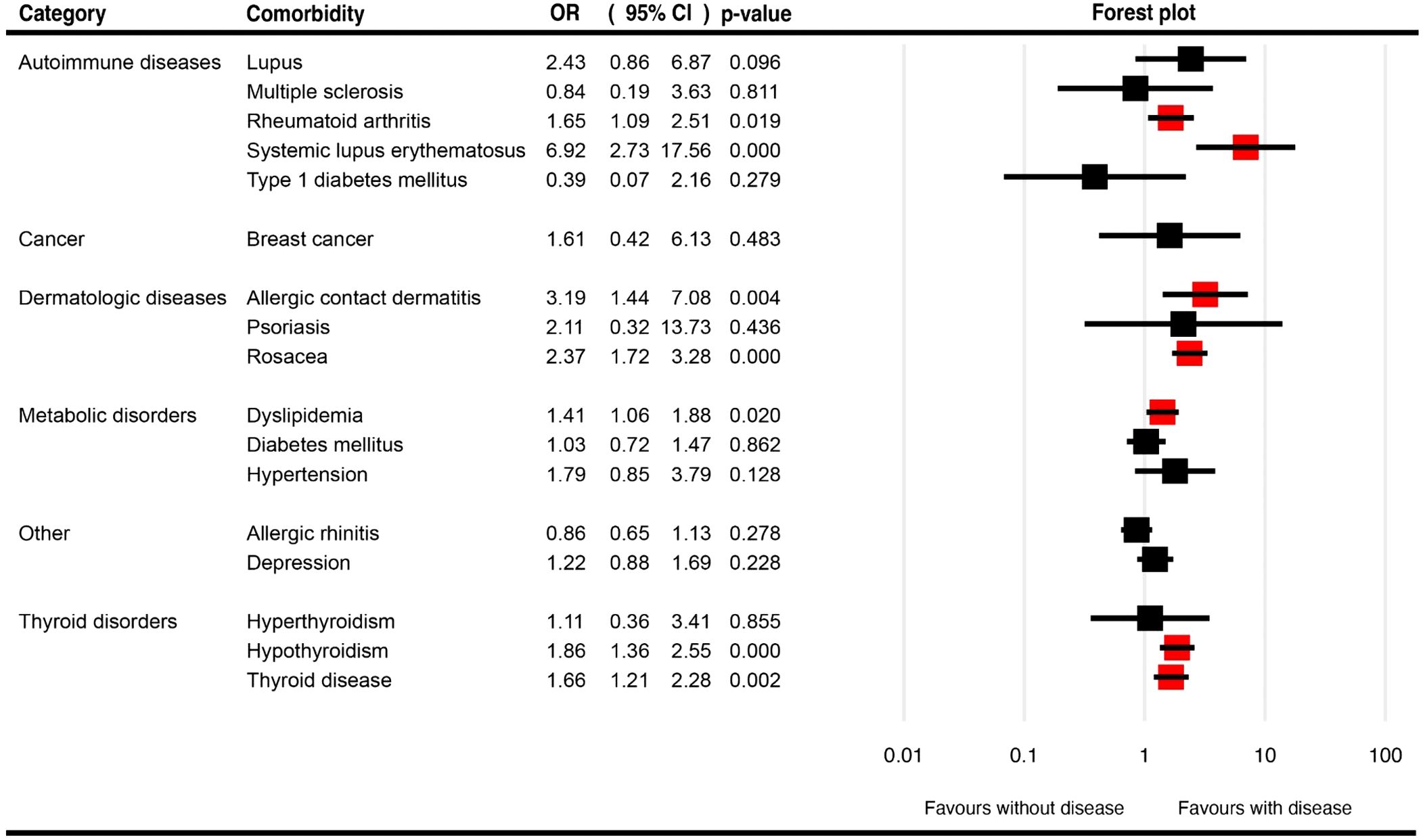
Figure 3. Forest plots for the pooled odds ratio of various comorbidities in patients with frontal fibrosing alopecia.
3.4 Central centrifugal cicatricial alopecia
3.4.1 Autoimmune diseases
SLE [OR 3.13 (1.03–9.49), prevalence 5%] was found to have statistically significant association with CCCA.
3.4.2 Dermatologic diseases
Acne [OR 2.21 (0.31–15.52), prevalence 40%], atopic dermatitis [OR 1.06 (0.05–22.69), prevalence 6%], and seborrheic dermatitis [OR 1.16 (0.74–1.83), prevalence 28%] were not found to be statistically associated with CCCA.
3.4.3 Metabolic disorders
Dyslipidemia [OR 4.46 (1.01–19.75), prevalence 54%] was found to have a strong statistical association with CCCA, while diabetes mellitus [OR 1.67 (1.03–2.69), prevalence 26%] was also statistically associated with CCCA; however, other metabolic diseases such as hypertension [OR 1.62 (0.78–3.34), prevalence 64%], obesity [OR 0.80 (0.28–2.30), prevalence 72%], and hirsutism [OR 2.83 (0.35–22.70), prevalence 9%] were not statistically associated with CCCA.
3.4.4 Other diseases
Allergic rhinitis [OR 11.77 (1.55–89.24), prevalence 24%] and anxiety [OR 4.69 (1.29–16.98), prevalence 17%] were strongly associated with CCCA. Asthma [OR 7.43 (0.85–64.69), prevalence 17%], depression [OR 1.93 (0.65–5.70), prevalence 26%], and leiomyoma [OR 2.23 (0.90–5.52), prevalence 15%] were not associated with CCCA with statistical significance.
Table 1 presents a summary of the pooled OR for the described comorbidities of CCCA patients, as well as the associated pooled prevalences. Figure 4, Supplementary Figure 3 illustrate the pooled OR and prevalence of the comorbidities in patients with CCCA, respectively.
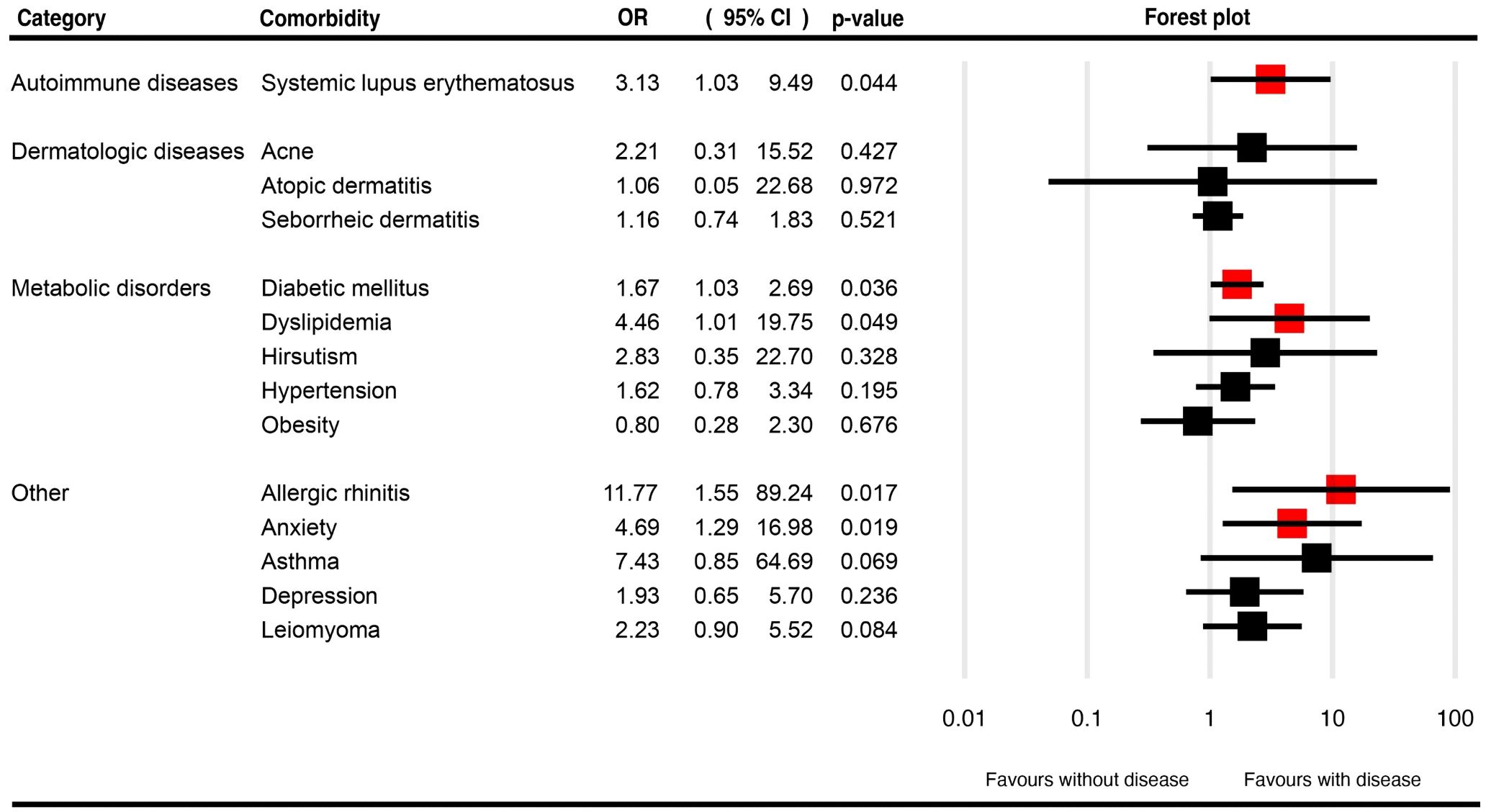
Figure 4. Forest plots for the pooled odds ratio of various comorbidities in patients with central centrifugal cicatricial alopecia.
3.5 Other primary cicatricial alopecia disorders
Among 24 FAPD patients, we identified AA [prevalence 12.5%], celiac disease [prevalence 8.3%], rheumatoid arthritis [prevalence 8.3%], frontal fibrosing alopecia [prevalence 8.3%], pernicious anemia [prevalence 4.2%], scalp psoriasis [prevalence 4.2%], and traction alopecia [prevalence 4.2%]. For AKN, common comorbidities include acne [prevalence 29%], acne conglobata [prevalence 14%], FD [prevalence 21%], and pseudofolliculitis barbae [prevalence 26%]. Acne conglobata [prevalence 24%] is also prevalent in DC, and a higher prevalence of hidradenitis suppurativa was found for DC [prevalence 26%], compared to FD [prevalence 4%] and AKN [prevalence 3%]. And for EPD, patients often report actinic keratosis [prevalence 21%], basal cell carcinoma [prevalence 7%], and squamous cell carcinoma [prevalence 13%] as their comorbidities. Supplementary Figures 4-7 show forest plots for the pooled prevalence of the comorbidities in patients with FD, DC, AKN, and EPD, respectively.
3.6 Quality assessment
Supplementary Table 7 provides a summary of the quality assessment scores for comparative and descriptive studies included in the review. The average quality assessment score was 7.78 (range: 4–9), with 101 high-quality and 15 fair-quality studies. No studies were classified as low-quality. The most common quality concerns in fair-quality studies were related to the comparability of cohorts and adequacy of follow-up. Sensitivity analyses excluding the 15 fair-quality studies did not substantially alter our main findings, with all significant associations remaining statistically significant.
3.7 Meta-regression and subgroup analysis
Meta-regression was performed for all analyses that were found to have significant heterogeneity and did not find any potential source of heterogeneity, except for one analysis in which we found that female proportion seems to play a role in the odds of depression in the LPP cohorts (Supplementary Figure 8). However, we chose not to perform a subgroup analysis due to the limited number of studies that included a narrow range of female proportions, specifically from 72.4 to 91.5%.
4 Discussion
This meta-analysis reveals distinct comorbidity patterns across PCA. The high prevalence and diversity of comorbidities suggest these conditions have systemic implications. Specifically, LPP showed strong associations with autoimmune disorders, particularly thyroid disease and lichen planus. FFA demonstrated links to both autoimmune and metabolic comorbidities, whereas CCCA exhibited significant associations with metabolic and hormonal factors. These distinct patterns underscore the complex, multifactorial nature of PCA and suggest they may be manifestations of broader systemic dysregulation rather than isolated scalp disorders. The elevated ORs for specific comorbidities emphasize the importance of a comprehensive approach to managing patients with these conditions, as they are likely at higher risk for multiple systemic conditions.
4.1 Autoimmune diseases
Statistically significantly higher odds of SLE were found among those with lymphocytic PCA, particularly LPP, FFA, and CCCA, compared with controls. Peroxisome proliferator-activated receptor-γ (PPAR-γ), a nuclear hormone receptor, is involved in the regulation of inflammation and tissue repair (126). It has been proposed to be the link between LPP and SLE (12), as PPAR-γ has been implicated in the pathogenesis of both PCA and SLE (126, 127). Specifically, PPAR-γ expression is altered in both conditions, potentially contributing to dysregulated immune responses and tissue damages (126, 128–130). PPAR-γ is also involved in the pathogenesis of rheumatoid arthritis (131), which we found to have a significant association with FFA. Therefore, further studies exploring the link between PCA and various autoimmune diseases are needed.
The association between PCA and multiple autoimmune conditions aligns with the concept of Multiple Autoimmune Syndrome (MAS), where at least three autoimmune diseases coexist in the same patient. Tatu and Ionescu described a case of MAS type 3, characterized by the coexistence of autoimmune thyroiditis, vitiligo, and AA (132). Although their report focused on non-scarring alopecia, the principle of shared autoimmune pathophysiology may extend to primary cicatricial alopecias, suggesting that PCA could represent a cutaneous manifestation within a broader autoimmune context.
4.2 Thyroid disorders
Similar to a previous meta-analysis, we also found a statistically significant association between LPP and hypothyroidism (133). In addition, we also found FFA to be associated with hypothyroidism, which can be observed in nearly one fifth of patients with either LPP or FFA. The association between thyroid disorders, especially hypothyroidism, and PCA could be explained by the prevalent immune-mediated hypothyroidism, such as Hashimoto thyroiditis. We believe hypothyroidism should be considered when evaluating LPP or FFA patients, given the well-established link between thyroid diseases and autoimmune skin diseases (134). This association underscores the importance of routine thyroid function screening in patients diagnosed with these forms of PCA, especially in cases with clinical suspicion. However, the need for thyroid investigation in asymptomatic patients remains controversial.
4.3 Dermatologic diseases
Compared to previous meta-analysis, we found a similar association and prevalence of rosacea in FFA (135). We also find that rosacea is strongly associated with LPP, although the prevalence is lower than in FFA. Apart from rosacea, LPP is also strongly linked to lichen planus and other immune-mediated skin diseases, including atopic dermatitis and psoriasis.
Although the evidence is limited to two studies, allergic contact dermatitis is also significantly associated with FFA. Given that FFA association with skin care products has been shown previously (136), it may be sensible to inform patients of the risk.
In contrast to other autoimmune skin diseases such as vitiligo and AA (137–140), we found a higher risk of skin cancer and melanoma among LPP patients. Actinic keratosis and non-melanocytic skin cancer are also found to be prevalent among EPD. Given the increased risk of skin cancer and dermatologic diseases, we believe a detailed full-body dermatological examination for patients with PCA is necessary.
4.4 Metabolic disorders
We have confirmed the findings of a previous meta-analysis that dyslipidemia is not statistically associated with LPP (141). Additionally, we also did not identify any association between LPP and other metabolic diseases, including diabetes mellitus, hypertension, and obesity. However, we did find an association between FFA and dyslipidemia, while CCCA is associated with both dyslipidemia and diabetes mellitus. These associations suggest a potential role of metabolic inflammation in their pathogenesis (126). Further studies exploring the association between PCA and metabolic disorders are needed.
4.5 Cardiovascular diseases
Kim et al. have previously found an association between PCA and incident cardiovascular disease, particularly coronary heart disease (125), which is also evident in our LPP findings. Certain interleukins (IL), especially IL-18 (142), are associated with cardiovascular risk, which makes autoimmune patients more prone to coronary diseases. We believe this issue requires further investigation and physicians should be on high alert for coronary events.
4.6 Other diseases
Similar to a previous meta-analysis (143), evidence is limited for vitamin D deficiency and PCA, and evidence so far suggests that vitamin D deficiency is not statistically associated with LPP. In addition, the lack of statistical association between uterine leiomyoma and CCCA is noted in our study, while strong associations between allergic rhinitis, anxiety, and CCCA were found. We suspect that the high prevalence of uterine leiomyoma may not be directly related to the disease. Regular psychosocial assessment and support is crucial for patients with CCCA as well as other PCAs. Further studies exploring the relationship between allergic rhinitis and CCCA are needed.
4.7 Histopathological integration and mechanistic insights
Recent histopathological studies have identified variations in inflammatory patterns across PCA subtypes that may explain their different comorbidity profiles. The predominantly CD8+ T-cell-mediated inflammation in LPP and FFA contrasts with the CD4+ T-cell-predominant mixed inflammatory infiltrate seen in CCCA, potentially explaining their distinct associations with autoimmune and metabolic conditions, respectively (144–146).
The association between PCA and various systemic conditions reflects complex shared pathogenic mechanisms involving inflammatory and immune-mediated pathways. The histopathological hallmark of lymphocytic PCA, destruction of follicular stem cells in the bulge region with perifollicular fibrosis, mirrors inflammatory patterns seen in associated autoimmune conditions. This parallel extends to the molecular level, where PPAR-γ signaling emerges as a crucial link between PCA and systemic disorders.
In LPP and FFA, decreased PPAR-γ expression in hair follicle stem cells triggers proinflammatory cytokine production and subsequent stem cell loss. This mechanism shows striking similarity to PPAR-γ dysfunction in SLE, where impaired PPAR-γ signaling contributes to immune dysregulation and tissue damage (126). The strong association between these conditions likely reflects this shared pathogenic pathway. For CCCA, the significant association with metabolic disorders may similarly reflect common inflammatory mechanisms. PPAR-γ’s role in regulating both immune responses and metabolic homeostasis provides a potential molecular explanation for these associations (147). The presence of metabolic inflammation in conditions such as diabetes and dyslipidemia may exacerbate the follicular inflammation characteristic of CCCA.
FAPD has been recently recognized as a subtype of LPP that presents with a pattern mimicking androgenetic alopecia (148). Histologically, FAPD exhibits a lymphocytic scarring process similar to classic LPP, although detailed characterization of the lymphocyte subtypes in its inflammatory infiltrate remains lacking. While its recognition has expanded our understanding of how scarring alopecia can clinically manifest, data regarding its associated systemic conditions remain limited.
4.8 Clinical significance of risk estimates
The magnitude of associations found in this meta-analysis has important clinical implications. A 3-fold increased risk of SLE in LPP patients translates to an absolute risk of approximately 2%, compared to 0.65% in the general population. While this remains a relatively low absolute risk, it warrants clinical vigilance given SLE’s potential severity. More practically significant is the 17-19% prevalence of hypothyroidism in LPP and FFA, a frequency that justifies routine screening.
The 8% prevalence of coronary artery disease in LPP patients (OR 1.63) represents a clinically meaningful increase that should prompt cardiovascular risk assessment and aggressive management of modifiable risk factors. Similarly, the high prevalence of dyslipidemia in CCCA (54%) and diabetes mellitus (26%) necessitates proactive metabolic screening and intervention in this population.
4.9 Clinical implications and screening recommendations
Based on our findings, we recommend a comprehensive screening approach for PCA patients. Initial assessment should include thyroid function tests and thyroid antibody screening for all PCA patients. Autoimmune screening with antinuclear antibody testing should be considered in patients presenting with systemic symptoms. Metabolic screening including lipid profile and fasting glucose is particularly important in FFA and CCCA patients. Annual full-body skin examinations are warranted in LPP patients due to their increased skin cancer risk.
The complexity of comorbidities in PCA necessitates multidisciplinary care coordination. Establishing collaborative relationships with endocrinology for thyroid management, rheumatology for suspected autoimmune conditions, and maintaining regular dermatologic surveillance are essential components of comprehensive care. Mental health support should be integrated into the management plan, given the significant psychological burden associated with PCA. This coordinated approach ensures that the multiple systemic manifestations of PCA are adequately addressed, ultimately improving patient outcomes and quality of life.
4.10 Limitations
This study has some limitations. Firstly, some comorbidities were infrequently reported and thus were not able to be quantitatively analyzed. Additionally, there is an inadequate number of cohort studies that document incident cases of various comorbidities for quantitative analysis. Furthermore, significant statistical heterogeneity was found in some of our analyses, and despite meta-regression analysis, we were unable to identify the source of heterogeneity, except for one analysis. This is partly due to the limited number of available studies. Potential sources of heterogeneity include variations in diagnostic criteria for PCA subtypes, differences in study populations (such as ethnicity, age distribution, geographic location, disease severity, and comorbidity burden), methodological differences across studies, differences in healthcare systems and access to specialist care, and temporal changes in awareness and screening practices for both PCA and associated conditions. These factors highlight the need for cautious interpretation of pooled estimates for these specific associations. While we employed appropriate statistical methods, the adjusted results may still be influenced by unreported negative findings.
5 Conclusions
This meta-analysis significantly contributes to our understanding of PCA by providing a comprehensive overview of associated comorbidities. PCA patients are at an increased risk of developing a variety of comorbidities, such as SLE, hypothyroidism, metabolic diseases, and various dermatologic conditions. The findings emphasize the need for a multidisciplinary approach to patient care and highlight the importance of considering PCA as potentially systemic disorders with localized scalp manifestations. Therefore, clinicians should maintain a high level of vigilance for comorbid conditions in PCA patients and conduct investigations when clinical signs are observed. Future research should focus on elucidating the mechanisms behind these associations through prospective cohort studies, which would help establish causality and determine whether interventions targeting comorbidities improve outcomes in PCA. While limitations such as study heterogeneity and the challenge of establishing causality exist, this analysis provides valuable insights that can guide clinical practice and future investigations in PCA.
Data availability statement
The original contributions presented in this study are included in the article/Supplementary Material, further inquiries can be directed to the corresponding author.
Author contributions
TY: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Writing – original draft. KT: Conceptualization, Data curation, Investigation, Methodology, Writing – original draft. PS: Conceptualization, Formal analysis, Investigation, Methodology, Software, Supervision, Validation, Writing – original draft, Writing – review & editing.
Funding
The author(s) declare that no financial support was received for the research and/or publication of this article.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declared that they were an editorial board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fimmu.2025.1516407/full#supplementary-material
Supplementary Figure 1 | Forest plots for the pooled prevalence of the comorbidities in patients with lichen planopilaris.
Supplementary Figure 2 | Forest plots for the pooled prevalence of the comorbidities in patients with frontal fibrosing alopecia.
Supplementary Figure 3 | Forest plots for the pooled prevalence of the comorbidities in patients with central centrifugal cicatricial alopecia.
Supplementary Figure 4 | Forest plots for the pooled prevalence of the comorbidities in patients with folliculitis decalvans.
Supplementary Figure 5 | Forest plots for the pooled prevalence of the comorbidities in patients with dissecting cellulitis.
Supplementary Figure 6 | Forest plots for the pooled prevalence of the comorbidities in patients with acne keloidalis nuchae
Supplementary Figure 7 | Forest plots for the pooled prevalence of the comorbidities in patients with erosive pustular dermatosis of the scalp
Supplementary Figure 8 | Bubble plot for the meta-regression analysis of the pooled odds ratio of depression in patients with lichen planopilaris, using the female proportion as a covariate.
References
1. Harries MJ and Paus R. The pathogenesis of primary cicatricial alopecias. Am J Pathol. (2010) 177:2152–62. doi: 10.2353/ajpath.2010.100454
2. Olsen EA, Bergfeld WF, Cotsarelis G, Price VH, Shapiro J, Sinclair R, et al. Summary of North American hair research society (Nahrs)-sponsored workshop on cicatricial alopecia, duke university medical center, february 10 and 11, 2001. J Am Acad Dermatol. (2003) 48:103–10. doi: 10.1067/mjd.2003.68
3. Moher D, Liberati A, Tetzlaff J, and Altman DG. Preferred reporting items for systematic reviews and meta-analyses: the prisma statement. Bmj. (2009) 339:b2535. doi: 10.1136/bmj.b2535
4. Wells G, Shea B, O'Connell D, Peterson J, Welch V, Losos M, et al. The Newcastle–Ottawa scale (Nos) for assessing the quality of non-randomized studies in Meta-analysis. (2000). Available at: http://www.ohri.ca/programs/clinical_epidemiology/oxford.html (Accessed July 10, 2025).
5. Deeks JJ, Altman DG, and Bradburn MJ. Statistical methods for examining heterogeneity and combining results from several studies in meta-analysis. In: Systematic Reviews in Health Care. London: BMJ Publishing Group. (2001). p. 285–312.
6. Thompson SG. Why sources of heterogeneity in meta-analysis should be investigated. Bmj. (1994) 309:1351–5. doi: 10.1136/bmj.309.6965.1351
7. Sterne JA, Egger M, and Smith GD. Systematic reviews in health care: investigating and dealing with publication and other biases in meta-analysis. Bmj. (2001) 323:101–5. doi: 10.1136/bmj.323.7304.101
8. Atanaskova Mesinkovska N, Brankov N, Piliang M, Kyei A, and Bergfeld WF. Association of lichen planopilaris with thyroid disease: A retrospective case-control study. J Am Acad Dermatol. (2014) 70:889–92. doi: 10.1016/j.jaad.2013.12.014
9. Brankov N, Conic RZ, Atanaskova-Mesinkovska N, Piliang M, and Bergfeld WF. Comorbid conditions in lichen planopilaris: A retrospective data analysis of 334 patients. Int J Women’s Dermatol. (2018) 4:180–4. doi: 10.1016/j.ijwd.2018.04.001
10. Conic RRZ, Maghfour J, Damiani G, and Bergfeld W. Exploring the association between lichen planopilaris, cardiovascular and metabolic disorders. J Eur Acad Dermatol Venereol. (2021) 35:e826–e8. doi: 10.1111/jdv.17513
11. Conic RRZ, Piliang M, Bergfeld W, and Atanaskova-Mesinkovska N. Association of lichen planopilaris with dyslipidemia. JAMA Dermatol. (2018) 154:1088–9. doi: 10.1001/jamadermatol.2018.1749
12. Fertig RM, Hu S, Maddy AJ, Balaban A, Aleid N, Aldahan A, et al. Medical comorbidities in patients with lichen planopilaris, a retrospective case-control study. Int J Dermatol. (2018) 57:804–9. doi: 10.1111/ijd.13996
13. Joshi TP, Duruewuru A, Holla S, Naqvi Z, Zhu H, and Ren V. Comorbidities associated with lichen planopilaris: A case-control study using the all of us database. Int J Dermatol. (2023) 62:e396–e8. doi: 10.1111/ijd.16433
14. Manatis-Lornell A, Okhovat JP, Marks DH, Hagigeorges D, and Senna MM. Comorbidities in patients with lichen planopilaris: A retrospective case-control study. J Am Acad Dermatol. (2020) 83:205–8. doi: 10.1016/j.jaad.2019.07.018
15. Nasimi M, Garmaroudi G, Ghiasi M, Lajevardi V, Fooladi Z, Hassan Zadeh Tabatabaei MS, et al. Comorbidities in patients with lichen planopilaris: A case-control study. Skin Appendage Disord. (2022) 8:302–6. doi: 10.1159/000522371
16. Nguyen TQ, Tsai M, Grogan T, and Goh C. Thyroid disease in alopecia areata, lichen planopilaris, and frontal fibrosing alopecia-a case control study of 144 patients. US Endocrinol. (2016) 12:85–6. doi: 10.17925/USE.2016.12.02.85
17. Sung HL, Hyun K, Yeon-Woo H, Won-Soo L, and Solam L. Prevalence and incidence of comorbid diseases and mortality risk associated with lichen planopilaris: A korean nationwide population-based study. Clin Exp Dermatol. (2023) 48:1230–7. doi: 10.1093/ced/llad235
18. Toossi P, Sebti A, and Sarvghadi F. Thyroid diseases and lichen planopilaris: A case control study. Iranian J Dermatol. (2015) 18:104–7.
19. Trager MH, Lavian J, Lee EY, Gary D, Jenkins F, Christiano AM, et al. Medical comorbidities and sex distribution among patients with lichen planopilaris and frontal fibrosing alopecia: A retrospective cohort study. J Am Acad Dermatol. (2021) 84:1686–9. doi: 10.1016/j.jaad.2020.08.015
20. Yu DA, Kim SR, Cho SI, and Kwon O. Endocrine and metabolic comorbidities in primary cicatricial alopecia: A nationwide population-based study. J Dermatol. (2024) 51:429–40. doi: 10.1111/1346-8138.17080
21. Babahosseini H, Tavakolpour S, Mahmoudi H, Balighi K, Teimourpour A, Ghodsi SZ, et al. Lichen planopilaris: retrospective study on the characteristics and treatment of 291 patients. J Dermatol Treat. (2019) 30:598–604. doi: 10.1080/09546634.2018.1542480
22. Cantwell HM, Wieland CN, Proffer SL, Imhof RL, Torgerson RR, and Tolkachjov SN. Lichen planopilaris in men: A retrospective clinicopathologic study of 19 patients. Int J Dermatol. (2021) 60:482–8. doi: 10.1111/ijd.15285
23. Carrascoza GG, Rivitti-MaChado MCM, Macedo T, Romiti R, and Doche I. Lichen planopilaris and frontal fibrosing alopecia-associated conditions: A retrospective study with 241 patients. J Eur Acad Dermatol Venereol. (2024) 38(9):e827–9. doi: 10.1111/jdv.19891
24. Conic RRZ, Piliang M, Bergfeld W, and Atanaskova-Mesinkovska N. Vitamin D status in scarring and nonscarring alopecia. J Am Acad Dermatol. (2021) 85:478–80. doi: 10.1016/j.jaad.2018.04.032
25. Doche I, Romiti R, Rivitti-MaChado MC, Gorbatenko-Roth K, Freese RL, and Hordinsky MK. Quality-of-life impairment is not related to disease activity in lichen planopilaris and frontal fibrosing alopecia. Results of a preliminary cross-sectional study. J Eur Acad Dermatol Venereol. (2022) 36:e288–e90. doi: 10.1111/jdv.17798
26. Gharaei Nejad K, Ghadarjani R, Eftekhari H, and Sheykholeslami S. Most frequent comorbidities in patients with lichen planopilaris: A cross-sectional study. Int J Dermatol Venereology. (2023) 6:229–32. doi: 10.1097/JD9.0000000000000306
27. Kurt BÖ, Şenol Y, and Dicle Ö. Clinical evaluation of primary cicatricial alopecias from Turkey: A retrospective analysis of 97 cases. Turk Dermatoloji Dergisi. (2018) 12:177–82. doi: 10.4274/tdd.3679
28. Larkin SC, Cantwell HM, Imhof RL, Torgerson RR, and Tolkachjov SN. Lichen planopilaris in women: A retrospective review of 232 women seen at mayo clinic from 1992 to 2016. Mayo Clinic Proc. (2020) 95:1684–95. doi: 10.1016/j.mayocp.2020.02.028
29. Lyakhovitsky A, Zilbermintz T, Segal Z, Galili E, Shemer A, Jaworowski B, et al. Exploring remission dynamics and prognostic factors in lichen planopilaris: A retrospective cohort study. Dermatology. (2024) 240(4):531–42. doi: 10.1159/000538355
30. Meinhard J, Stroux A, Lünnemann L, Vogt A, and Blume-Peytavi U. Lichen planopilaris: epidemiology and prevalence of subtypes-a retrospective analysis in 104 patients. JDDG - J German Soc Dermatol. (2014) 12:229–35. doi: 10.1111/ddg.12264
31. Özcan D, Seçkin D, Güleç AT, and Özen O. Lichen planopilaris: demographic, clinical and histopathological characteristics and treatment outcomes of 25 cases. Turkderm Deri Hastaliklari ve Frengi Arsivi. (2015) 49:246–52. doi: 10.4274/turkderm.32767
32. Aldoori N, Dobson K, Holden CR, McDonagh AJ, Harries M, and Messenger AG. Frontal fibrosing alopecia: possible association with leave-on facial skin care products and sunscreens; a questionnaire study. Br J Dermatol. (2016) 175:762–7. doi: 10.1111/bjd.14535
33. Arasu A, Meah N, Eisman S, Wall D, and Sinclair R. Vitamin D status in patients with frontal fibrosing alopecia: A retrospective study. JAAD Int. (2022) 7:129–30. doi: 10.1016/j.jdin.2022.03.008
34. Bazotti LX, Teixeira LM, and Naspolini AP. Risk factors correlated with frontal fibrosing alopecia in criciuma, santa catarina: A case-control study. Surg Cosmetic Dermatol. (2022) 14:e20220042. doi: 10.5935/scd1984-8773.2022140042
35. Buendía-Castaño D, Saceda-Corralo D, Moreno-Arrones OM, Fonda-Pascual P, Alegre-Sánchez A, Pindado-Ortega C, et al. Hormonal and gynecological risk factors in frontal fibrosing alopecia: A case-control study. Skin Appendage Disord. (2018) 4:274–6. doi: 10.1159/000484210
36. Donati A, Lindgren BR, Abreu G, and Hordinsky M. Prevalence of frontal fibrosing alopecia among Brazilian dermatologists: A cross-sectional survey. JAAD Int. (2020) 1:148–50. doi: 10.1016/j.jdin.2020.07.008
37. Leecharoen W, Thanomkitti K, Thuangtong R, Varothai S, Triwongwaranat D, Jiamton S, et al. Use of facial care products and frontal fibrosing alopecia: coincidence or true association? J Dermatol. (2021) 48:1557–63. doi: 10.1111/1346-8138.16063
38. Moreno-Arrones OM, Saceda-Corralo D, Rodrigues-Barata AR, Castellanos-González M, Fernández-Pugnaire MA, Grimalt R, et al. Risk factors associated with frontal fibrosing alopecia: A multicentre case-control study. Clin Exp Dermatol. (2019) 44:404–10. doi: 10.1111/ced.13785
39. Porriño-Bustamante ML, Fernández-Pugnaire MA, and Arias-Santiago S. A cross-sectional study of rosacea and risk factors in women with frontal fibrosing alopecia. Acta Dermato-Venereologica. (2019) 99:1099–104. doi: 10.2340/00015555-3286
40. Porriño-Bustamante ML, Montero-Vílchez T, Pinedo-Moraleda FJ, Fernández-Flores Á, Fernández-Pugnaire MA, and Arias-Santiago S. Frontal fibrosing alopecia and sunscreen use: A cross-sectional study of actinic damage. Acta Dermato-Venereologica. (2022) 102:adv00757. doi: 10.2340/actadv.v102.306
41. Ramos PM, Anzai A, Duque-Estrada B, Farias DC, Melo DF, Mulinari-Brenner F, et al. Risk factors for frontal fibrosing alopecia: A case-control study in a multiracial population. J Am Acad Dermatol. (2021) 84:712–8. doi: 10.1016/j.jaad.2020.08.076
42. Rudnicka L, Rokni GR, Lotti T, Wollina U, Fölster-Holst R, Katsambas A, et al. Allergic contact dermatitis in patients with frontal fibrosing alopecia: an international multi-center study. Dermatologic Ther. (2020) 33(4):e13560. doi: 10.1111/dth.13560
43. Adotama P, Callender V, Kolla A, Young C, Jones P, Svigos K, et al. Comparing the clinical differences in white and black women with frontal fibrosing alopecia. Br J Dermatol. (2021) 185:1074–6. doi: 10.1111/bjd.20605
44. Aslani FS, Saki N, and Sasannia M. Frontal fibrosing alopecia: A clinicopathological study of 22 cases from shiraz, southern Iran. Iranian J Dermatol. (2020) 23:112–9. doi: 10.22034/ijd.2020.111548
45. Banka N, Mubki T, Bunagan MJ, McElwee K, and Shapiro J. Frontal fibrosing alopecia: A retrospective clinical review of 62 patients with treatment outcome and long-term follow-up. Int J Dermatol. (2014) 53:1324–30. doi: 10.1111/ijd.12479
46. Carmona-Rodríguez M, Moro-Bolado F, Romero-Aguilera G, Ruiz-Villaverde R, and Carriel V. Frontal fibrosing alopecia: an observational single-center study of 306 cases. Life (Basel). (2023) 13(6):1344. doi: 10.3390/life13061344
47. Collins MS, Ali S, Wiss IP, and Senna MM. Increased risk of vitamin D deficiency and insufficiency in black patients with central centrifugal cicatricial alopecia. J Am Acad Dermatol. (2022) 87:689–91. doi: 10.1016/j.jaad.2022.02.018
48. Doche I, Nico MMS, Gerlero P, Rebeis M, Melo DF, Tortelly V, et al. Clinical features and sex hormone profile in male patients with frontal fibrosing alopecia: A multicenter retrospective study with 33 patients. J Am Acad Dermatol. (2022) 86:1176–8. doi: 10.1016/j.jaad.2021.04.076
49. Dorgham NA, Hegazy R, Farag A, and Dorgham DA. Frontal fibrosing alopecia: A retrospective clinical review of 58 Egyptian patients with treatment outcome and long-term follow-up. J Eur Acad Dermatol Venereol. (2022) 36:e212–e3. doi: 10.1111/jdv.17747
50. García A, Navarro MR, Ramirez A, Pino A, Navarro A, Moles I, et al. Plasma rich in growth factors as an adjuvant treatment for the management of frontal fibrosing alopecia: A retrospective observational clinical study. J Cutaneous Med Surg. (2023) 27:340–9. doi: 10.1177/12034754231177599
51. Gkini MA, Riaz R, and Jolliffe V. A retrospective analysis of efficacy and safety of intralesional triamcinolone injections in the treatment of frontal fibrosing alopecia either as monotherapy or as a concomitant therapy. Int J Trichology. (2018) 10:162–8. doi: 10.4103/ijt.ijt_46_18
52. Grassi S, Tadiotto Cicogna G, Magri F, Caterina Fortuna M, Caro G, Pernazza A, et al. Frontal fibrosing alopecia and genital lichen sclerosus: single-center experience. J Cosmetic Dermatol. (2021) 20:615–20. doi: 10.1111/jocd.13573
53. Heppt MV, Letulé V, Laniauskaite I, Reinholz M, Tietze JK, Wolff H, et al. Frontal fibrosing alopecia: A retrospective analysis of 72 patients from a german academic center. Facial Plast surgery: FPS. (2018) 34:88–94. doi: 10.1055/s-0037-1615281
54. Imhof RL, Chaudhry HM, Larkin SC, Torgerson RR, and Tolkachjov SN. Frontal fibrosing alopecia in women: the mayo clinic experience with 148 patients, 1992-2016. Mayo Clinic Proc. (2018) 93:1581–8. doi: 10.1016/j.mayocp.2018.05.036
55. Jerjen R, Pinczewski J, Sinclair R, and Bhoyrul B. Clinicopathological characteristics and treatment outcomes of fibrosing alopecia in a pattern distribution: A retrospective cohort study. J Eur Acad Dermatol Venereol. (2021) 35:2440–7. doi: 10.1111/jdv.17604
56. Jiang T and Liu C. Dissecting cellulitis of the scalp with typical clinical features: A retrospective cross-sectional study in a department of dermatology, Beijing, China. J Dermatol. (2022) 49:1173–7. doi: 10.1111/1346-8138.16555
57. Kanti V, Constantinou A, Reygagne P, Vogt A, Kottner J, and Blume-Peytavi U. Frontal fibrosing alopecia: demographic and clinical characteristics of 490 cases. J Eur Acad Dermatol Venereol. (2019) 33:1976–83. doi: 10.1111/jdv.15735
58. Kusano LDC and Brenner FAM. Frontal fibrosing alopecia: follow-up of a Brazilian group. Anais brasileiros dermatologia. (2019) 94:365–6. doi: 10.1590/abd1806-4841.20197941
59. Lobato-Berezo A, Iglesias-Sancho M, Rodríguez-Lomba E, Mir-Bonafé JF, Velasco-Tamariz V, Porriño-Bustamante ML, et al. Frontal fibrosing alopecia in men: A multicenter study of 39 patients. J Am Acad Dermatol. (2022) 86:481–4. doi: 10.1016/j.jaad.2021.09.033
60. Maldonado Cid P, Leis Dosil VM, Garrido Gutiérrez C, Salinas Moreno S, Thuissard Vasallo IJ, Andreu Vázquez C, et al. Frontal fibrosing alopecia: A retrospective study of 75 patients. Actas Dermo-Sifiliograficas. (2020) 111:487–95. doi: 10.1016/j.ad.2020.03.003
61. McSweeney SM, Christou EAA, Dand N, Boalch A, Holmes S, Harries M, et al. Frontal fibrosing alopecia: A descriptive cross-sectional study of 711 cases in female patients from the UK. Br J Dermatol. (2020) 183:1136–8. doi: 10.1111/bjd.19399
62. Melo DF, de Mattos Barreto T, Saceda-Corralo D, MaChado CJ, Xavier de Brito F, Tebet M, et al. Epidemiologic and clinical features of pattern iii frontal fibrosing alopecia (Pseudo fringe type): A multicenter series of 38 patients. J Am Acad Dermatol. (2021) 84:797–8. doi: 10.1016/j.jaad.2020.05.124
63. Moreno-Arrones OM, Saceda-Corralo D, Rodrigues-Barata AR, Castellanos-González M, Fernández-Pugnaire MA, Grimalt R, et al. Factors influencing frontal fibrosing alopecia severity: A multicentre cross-sectional study. J Eur Acad Dermatol Venereol. (2019) 33:e315–e6. doi: 10.1111/jdv.15590
64. Moussa A, Bennett M, Bhoyrul B, Kazmi A, Asfour L, and Sinclair RD. Clinical features and treatment outcomes of frontal fibrosing alopecia in men. Int J Dermatol. (2022) 61:e372–e4. doi: 10.1111/ijd.16313
65. Oulad Ali S, Belcadi J, El Hilali S, Senouci K, and Meziane M. Frontal fibrosing alopecia and comorbidities in a moroccan population. JAAD Int. (2023) 12:37–8. doi: 10.1016/j.jdin.2023.04.003
66. Panchaprateep R, Ruxrungtham P, Chancheewa B, and Asawanonda P. Clinical characteristics, trichoscopy, histopathology and treatment outcomes of frontal fibrosing alopecia in an asian population: A retro-prospective cohort study. J Dermatol. (2020) 47:1301–11. doi: 10.1111/1346-8138.15517
67. Pindado-Ortega C, Saceda-Corralo D, Buendía-Castaño D, Fernández-González P, Moreno-Arrones ÓM, Fonda-Pascual P, et al. Frontal fibrosing alopecia and cutaneous comorbidities: A potential relationship with rosacea. J Am Acad Dermatol. (2018) 78:596–7.e1. doi: 10.1016/j.jaad.2017.09.004
68. Pindado-Ortega C, Saceda-Corralo D, Moreno-Arrones ÓM, Rodrigues-Barata AR, Hermosa-Gelbard Á, Jaén-Olasolo P, et al. Effectiveness of dutasteride in a large series of patients with frontal fibrosing alopecia in real clinical practice. J Am Acad Dermatol. (2021) 84:1285–94. doi: 10.1016/j.jaad.2020.09.093
69. Rocha VB, MaChado CJ, and Contin LA. Uncommon subtypes of frontal fibrosing alopecia: retrospective analysis of clinical characteristics and prognosis. Anais Brasileiros Dermatologia. (2022) 97:260–2. doi: 10.1016/j.abd.2021.02.009
70. Rossi A, Cicogna GT, Caro G, Fortuna MC, Magri F, and Grassi S. Frontal fibrosing alopecia: A new association with lichen sclerosus in men. J Clin Aesthetic Dermatol. (2021) 14:54–8.
71. Saceda-Corralo D, Ortega-Quijano D, Muñoz-Martín G, Moreno-Arrones ÓM, Pindado-Ortega C, Rayinda T, et al. Genotyping of the rs1800440 polymorphism in cyp1b1 gene and the rs9258883 polymorphism in hla-B gene in a spanish cohort of 223 patients with frontal fibrosing alopecia. Acta Derm Venereol. (2023) 103:adv9604. doi: 10.2340/actadv.v103.9604
72. Saceda-Corralo D, Pindado-Ortega C, Moreno-Arrones OM, Ortega-Quijano D, Fernández-Nieto D, Jiménez-Cauhe J, et al. Association of inflammation with progression of hair loss in women with frontal fibrosing alopecia. JAMA Dermatol. (2020) 156:700–2. doi: 10.1001/jamadermatol.2020.0359
73. Salas-Callo CI, Tosti A, Stohmann D, Contarini P, and Pirmez R. Eyelash involvement in frontal fibrosing alopecia: A prospective study. J Am Acad Dermatol. (2022) 87:232–4. doi: 10.1016/j.jaad.2021.07.063
74. Secchin P, Quintella DC, Paula NÁ, Andrade LCDS, and Sodré CT. Clinical-histopathological profile of the frontal fibrosing alopecia: A retrospective study of 16 cases of a university hospital. Anais brasileiros dermatologia. (2019) 94:416–21. doi: 10.1590/abd1806-4841.20197797
75. Starace M, Orlando G, Iorizzo M, Alessandrini A, Bruni F, Mandel VD, et al. Clinical and dermoscopic approaches to diagnosis of frontal fibrosing alopecia: results from a multicenter study of the international dermoscopy society. Dermatol Pract Concept. (2022) 12:e2022080. doi: 10.5826/dpc.1201a80
76. Strazzulla LC, Avila L, Li X, Lo Sicco K, and Shapiro J. Prognosis, treatment, and disease outcomes in frontal fibrosing alopecia: A retrospective review of 92 cases. J Am Acad Dermatol. (2018) 78:203–5. doi: 10.1016/j.jaad.2017.07.035
77. Suchonwanit P, Pakornphadungsit K, Leerunyakul K, Khunkhet S, Sriphojanart T, and Rojhirunsakool S. Frontal fibrosing alopecia in asians: A retrospective clinical study. Int J Dermatol. (2020) 59:184–90. doi: 10.1111/ijd.14672
78. Tan KT and Messenger AG. Frontal fibrosing alopecia: clinical presentations and prognosis. Br J Dermatol. (2009) 160:75–9. doi: 10.1111/j.1365-2133.2008.08861.x
79. Uzunçakmak TK, Özkoca D, Aşkin Ö, and Serdaroğlu S. Evaluation of the demographic characteristics and the factors related to eyebrow involvement in frontal fibrosing alopecia: A retrospective, cross-sectional, single center study. Turkiye Klinikleri Dermatoloji. (2021) 31:93–9. doi: 10.5336/dermato.2021-81322
80. Valesky EM, Maier MD, Kaufmann R, Zöller N, and Meissner M. Single-center analysis of patients with frontal fibrosing alopecia: evidence for hypothyroidism and a good quality of life. J Int Med Res. (2019) 47:653–61. doi: 10.1177/0300060518807335
81. Verma S, Marak A, Paul D, and Dey B. A retrospective study of frontal fibrosing alopecia from North-East India. Indian J Dermatol. (2023) 68:598–602. doi: 10.4103/ijd.IJD_290_23
82. Xavier de Brito FO, Cortez de Almeida RF, MaChado CJ, Lemes LR, Donda ALV, Blanco A, et al. Frontal fibrosing alopecia associated with lichen planus pigmentosus: A multicentre retrospective descriptive analytical study of 104 patients. J Eur Acad Dermatol Venereol. (2023) 37:e1033–7. doi: 10.1111/jdv.19093
83. Zhang M, Zhang L, Rosman IS, and Mann CM. Frontal fibrosing alopecia demographics: A survey of 29 patients. Cutis. (2018) 102:E16–22.
84. Dina Y, Okoye GA, and Aguh C. Association of uterine leiomyomas with central centrifugal cicatricial alopecia. JAMA Dermatol. (2018) 154:213–4. doi: 10.1001/jamadermatol.2017.5163
85. Jafari AJ, Brown C, Echuri H, and Murina AT. Lack of association between comorbidities and central centrifugal cicatricial alopecia: A retrospective cohort study of 153 patients. J Am Acad Dermatol. (2023) 88:e101–e3. doi: 10.1016/j.jaad.2022.09.056
86. Joshi TP, Duruewuru A, Garcia D, Mireles N, Truong P, and Cockerell CJ. Comorbidities in patients with central centrifugal cicatricial alopecia: A case-control study. Int J Dermatol. (2024) 63:e37–e9. doi: 10.1111/ijd.16932
87. Kyei A, Bergfeld WF, Piliang M, and Summers P. Medical and environmental risk factors for the development of central centrifugal cicatricial alopecia: A population study. Arch Dermatol. (2011) 147:909–14. doi: 10.1001/archdermatol.2011.66
88. Leung B, Lindley L, Reisch J, Glass DA, and Ayoade K. Comorbidities in patients with central centrifugal cicatricial alopecia: A retrospective chart review of 53 patients. J Am Acad Dermatol. (2023) 88:461–3. doi: 10.1016/j.jaad.2022.06.013
89. McKenzie SA, Roche FC, Onyekaba G, Williams DM, Ogunleye TA, and Taylor SC. Comorbid anxiety and depression among black women with central centrifugal cicatricial alopecia: A retrospective study. J Dermatol. (2021) 48:e19. doi: 10.1111/1346-8138.15595
90. Narasimman M, De Bedout V, Castillo DE, and Miteva MI. Increased association between previous pregnancies and use of chemical relaxers in 74 women with central centrifugal cicatricial alopecia. Int J Trichology. (2020) 12:176–81. doi: 10.4103/ijt.ijt_37_20
91. Roche FC, Harris J, Ogunleye T, and Taylor SC. Association of type 2 diabetes with central centrifugal cicatricial alopecia: A follow-up study. J Am Acad Dermatol. (2022) 86:661–2. doi: 10.1016/j.jaad.2021.02.036
92. Samrao A, Lyon L, and Mirmirani P. Evaluating the association of central centrifugal cicatricial alopecia (Ccca) and fibroproliferative disorders. Dermatol Online J. (2021) 27(8). doi: 10.5070/D327854688
93. Ali S, Collins M, Taylor SC, Kelley K, Stratton E, and Senna M. Type 2 diabetes mellitus and central centrifugal cicatricial alopecia severity. J Am Acad Dermatol. (2022) 87:1418–9. doi: 10.1016/j.jaad.2022.08.031
94. Balazic E, Chen A, Konisky H, Hawkins K, Choi J, Mhaimeed N, et al. A retrospective chart review of central centrifugal cicatricial alopecia patients at a single urban institution. JAAD Int. (2023) 13:60–2. doi: 10.1016/j.jdin.2023.07.014
95. Jackson TK, Sow Y, Ayoade KO, Seykora JT, Taylor SC, and Ogunleye T. Central centrifugal cicatricial alopecia in males. J Am Acad Dermatol. (2023) 89:1136–40. doi: 10.1016/j.jaad.2023.07.1011
96. Onamusi T, Larrondo J, and McMichael AJ. Clinical factors and hair care practices influencing outcomes in central centrifugal cicatricial alopecia. Arch Dermatol Res. (2023) 315:2375–81. doi: 10.1007/s00403-023-02630-5
97. Shah SK and Alexis AF. Central centrifugal cicatricial alopecia: retrospective chart review. J Cutaneous Med Surg. (2010) 14:212–22. doi: 10.2310/7750.2010.09055
98. Suchonwanit P, Hector CE, Bin Saif GA, and McMichael AJ. Factors affecting the severity of central centrifugal cicatricial alopecia. Int J Dermatol. (2016) 55:e338–e43. doi: 10.1111/ijd.13061
99. Brown-Korsah JB, Roche FC, and Taylor SC. Association of breast and colorectal cancer in patients with central centrifugal cicatricial alopecia: A retrospective, cross-sectional pilot study. J Am Acad Dermatol. (2021) 84:859–60. doi: 10.1016/j.jaad.2020.10.044
100. Bunagan MJ, Banka N, and Shapiro J. Retrospective review of folliculitis decalvans in 23 patients with course and treatment analysis of long-standing cases. J Cutan Med Surg. (2014) 18:1–5. doi: 10.2310/7750.2014.13218
101. Lyakhovitsky A, Segal O, Galili E, Thompson CT, Tzanani I, Scope A, et al. Diagnostic delay, comorbid hidradenitis suppurativa and the prognostic value of bacterial culture in folliculitis decalvans: A cohort study. JDDG - J German Soc Dermatol. (2023) 21:1469–77. doi: 10.1111/ddg.15202
102. Miguel-Gómez L, Rodrigues-Barata AR, Molina-Ruiz A, Martorell-Calatayud A, Fernández-Crehuet P, Grimalt R, et al. Folliculitis decalvans: effectiveness of therapies and prognostic factors in a multicenter series of 60 patients with long-term follow-up. J Am Acad Dermatol. (2018) 79:878–83. doi: 10.1016/j.jaad.2018.05.1240
103. Sarkis A, Cortez de Almeida RF, Lemes LR, Obadia DL, MaChado CJ, Müller-Ramos P, et al. Folliculitis decalvans in women: A retrospective multicentre study of 150 patients. J Eur Acad Dermatol Venereol. (2024) 38:e66–70. doi: 10.1111/jdv.19434
104. Vañõ-Galván S, Molina-Ruiz AM, Fernández-Crehuet P, Rodrigues-Barata AR, Arias-Santiago S, Serrano-Falcõn C, et al. Folliculitis decalvans: A multicentre review of 82 patients. J Eur Acad Dermatol Venereol. (2015) 29:1750–7. doi: 10.1111/jdv.12993
105. Tran AX, Lefante JJ, and Murina A. Risk factors for dissecting cellulitis of the scalp: A case-control study. J Am Acad Dermatol. (2022) 86:941–3. doi: 10.1016/j.jaad.2021.03.076
106. Badaoui A, Reygagne P, Cavelier-Balloy B, Pinquier L, Deschamps L, Crickx B, et al. Dissecting cellulitis of the scalp: A retrospective study of 51 patients and review of literature. Br J Dermatol. (2016) 174:421–3. doi: 10.1111/bjd.13999
107. Feng H, Zhu C, and Jin H. The efficacy and safety of 5-aminolevulinic acid photodynamic therapy (Ala-pdt) as an adjunct therapy for symptoms in patients with dissecting cellulitis of the scalp: A retrospective study. Photodiagnosis Photodyn Ther. (2021) 34:102322. doi: 10.1016/j.pdpdt.2021.102322
108. Melo DF, Ramos PM, MaChado CJ, Anzai A, Blanco A, Mulinari-Brenner F, et al. Dissecting cellulitis in women: A retrospective multicenter study with 17 patients. Int J Dermatol. (2022) 61:e427–e30. doi: 10.1111/ijd.16271
109. Kridin K, Patel PM, Jones VA, Damiani G, Amber KT, and Cohen AD. Hidradenitis suppurativa is associated with acne keloidalis nuchae: A population-based study. Arch Dermatol Res. (2021) 313:333–7. doi: 10.1007/s00403-020-02105-x
110. Kridin K, Solomon A, Tzur-Bitan D, Damiani G, Comaneshter D, and Cohen AD. Acne keloidalis nuchae and the metabolic syndrome: A population-based study. Am J Clin Dermatol. (2020) 21:733–9. doi: 10.1007/s40257-020-00541-z
111. Doche I, Coelho EQ, Quaresma MV, and da Matta Rivitti-MaChado MC. Acne keloidalis nuchae and folliculitis decalvans: same process affecting the follicle or coexisting diseases? A retrospective study. Int J Dermatol. (2019) 58:e200–e3. doi: 10.1111/ijd.14565
112. East-Innis ADC, Stylianou K, Paolino A, and Ho JD. Acne keloidalis nuchae: risk factors and associated disorders - a retrospective study. Int J Dermatol. (2017) 56:828–32. doi: 10.1111/ijd.13678
113. Lobato-Berezo A, Escolà-Rodríguez A, Courtney A, Chim I, Ruiz-Villaverde R, Imbernón-Moya A, et al. Acne keloidalis nuchae: an international multicentric review of 79 patients. J Eur Acad Dermatol Venereol. (2024) 38:e342–e5. doi: 10.1111/jdv.19609
114. Na K, Oh SH, and Kim SK. Acne keloidalis nuchae in asian: A single institutional experience. PLoS One. (2017) 12(12):e0189790. doi: 10.1371/journal.pone.0189790
115. Parker A, Parker MA, Schneider J, Jordaan H, and Visser W. The clinicopathological spectrum of preclinical folliculitis keloidalis with correlation to its dermoscopic features: A cross-sectional analytical study. Int J Dermatol. (2023) 62:1371–7. doi: 10.1111/ijd.16847
116. Umar S, Lee DJ, and Lullo JJ. A retrospective cohort study and clinical classification system of acne keloidalis nuchae. J Clin Aesthetic Dermatol. (2021) 14:E61–E7.
117. Umar S, Lullo JJ, Carter MJ, Shitabata PK, and Lee DJ. Acne keloidalis nuchae is associated with cutis verticis gyrata. Clinical Cosmetic Investigational Dermatol. (2022) 15:1421–7. doi: 10.2147/CCID.S369243
118. Saka B, Teclessou JN, Akakpo SA, Pessinaba S, Gnossike P, Mahamadou G, et al. Acne keloidalis nuchae and hypertension in black subjects: A case-control study. BMC Res Notes. (2020) 13:431. doi: 10.1186/s13104-020-05274-0
119. Shavit E, Cohen A, Zoller L, Onn E, and Kridin K. The burden of gout in acne keloidalis nuchae-insights from a population-based study. J Cosmet Dermatol. (2023) 22:284–8. doi: 10.1111/jocd.15476
120. Valdman-Grinshpoun Y, Kridin K, Schonmann Y, and Cohen AD. Acne keloidalis nuchae and thyroid diseases: A population-based cohort study. Int J Dermatol. (2021) 60:466–70. doi: 10.1111/ijd.15331
121. Michelerio A, Vassallo C, Fiandrino G, and Tomasini CF. Erosive pustular dermatosis of the scalp: A clinicopathologic study of fifty cases. Dermatopathology. (2021) 8:450–62. doi: 10.3390/dermatopathology8040048
122. Shamloul N, Kamrani P, Shamloul G, Kunselman A, Thiboutot D, Billingsley E, et al. Incidence and time to development of Malignancies arising on the scalp of patients with erosive pustular dermatosis based on sex: A retrospective analysis. J Am Acad Dermatol. (2023) 89:1038–9. doi: 10.1016/j.jaad.2023.05.087
123. Starace M, Loi C, Bruni F, Alessandrini A, Misciali C, Patrizi A, et al. Erosive pustular dermatosis of the scalp: clinical, trichoscopic, and histopathologic features of 20 cases. J Am Acad Dermatol. (2017) 76:1109–14.e2. doi: 10.1016/j.jaad.2016.12.016
124. Tomasini C and Michelerio A. Erosive pustular dermatosis of the scalp: A neutrophilic folliculitis within the spectrum of neutrophilic dermatoses: A clinicopathologic study of 30 cases. J Am Acad Dermatol. (2019) 81:527–33. doi: 10.1016/j.jaad.2018.10.029
125. Kim SR, Yu DA, Cho SI, and Kwon O. Association of primary cicatricial alopecia with subsequent cardiovascular disease. J Invest Dermatol. (2024) 144:1166–9. doi: 10.1016/j.jid.2023.10.021
126. Harnchoowong S and Suchonwanit P. Ppar-Γ Agonists and their role in primary cicatricial alopecia. PPAR Res. (2017) 2017:2501248. doi: 10.1155/2017/2501248
127. Oxer DS, Godoy LC, Borba E, Lima-Salgado T, Passos LA, Laurindo I, et al. Pparγ Expression is increased in systemic lupus erythematosus patients and represses cd40/cd40l signaling pathway. Lupus. (2011) 20:575–87. doi: 10.1177/0961203310392419
128. Chanprapaph K, Sutharaphan T, and Suchonwanit P. Scalp biophysical characteristics in males with androgenetic alopecia: A comparative study with healthy controls. Clin Interv Aging. (2021) 16:781–7. doi: 10.2147/cia.S310178
129. Rattanakaemakorn P and Suchonwanit P. Scalp pruritus: review of the pathogenesis, diagnosis, and management. BioMed Res Int. (2019) 2019:1268430. doi: 10.1155/2019/1268430
130. Suchonwanit P, Triyangkulsri K, Ploydaeng M, and Leerunyakul K. Assessing biophysical and physiological profiles of scalp seborrheic dermatitis in the thai population. BioMed Res Int. (2019) 2019:5128376. doi: 10.1155/2019/5128376
131. Li X-F, Sun Y-Y, Bao J, Chen X, Li Y-H, Yang Y, et al. Functional role of ppar-Γ on the proliferation and migration of fibroblast-like synoviocytes in rheumatoid arthritis. Sci Rep. (2017) 7:12671. doi: 10.1038/s41598-017-12570-6
132. Tatu AL and Ionescu MA. Multiple autoimmune syndrome type 3- thyroiditis,Vitiligo and alopecia areata. Acta Endocrinol (Buchar). (2017) 13:124–5. doi: 10.4183/aeb.2017.124
133. Joshi TP, Friske S, and Duvic M. Association of lichen planopilaris with hypothyroidism: A systematic review and meta-analysis. Int J Dermatol. (2023) 62:e606–e8. doi: 10.1111/ijd.16788
134. Baldini E, Odorisio T, Tuccilli C, Persechino S, Sorrenti S, Catania A, et al. Thyroid diseases and skin autoimmunity. Rev Endocr Metab Disord. (2018) 19:311–23. doi: 10.1007/s11154-018-9450-7
135. Liu L, Chen Y, Chen J, Xue Y, Chen T, Li Y, et al. Association between frontal fibrosing alopecia and rosacea: results from clinical observational studies and gene expression profiles. Front Immunol. (2022) 13:985081. doi: 10.3389/fimmu.2022.985081
136. Maghfour J, Ceresnie M, Olson J, and Lim HW. The association between frontal fibrosing alopecia, sunscreen, and moisturizers: A systematic review and meta-analysis. J Am Acad Dermatol. (2022) 87:395–6. doi: 10.1016/j.jaad.2021.12.058
137. Mostaghimi A, Qureshi S, Joyce C, Guo Y, and Huang KP. Reduced incidence of skin cancer in patients with alopecia areata: A retrospective cohort study. Cancer Epidemiol. (2016) 41:129–31. doi: 10.1016/j.canep.2016.02.009
138. Chanprapaph K, Mahasaksiri T, Kositkuljorn C, Leerunyakul K, and Suchonwanit P. Prevalence and risk factors associated with the occurrence of autoimmune diseases in patients with alopecia areata. J Inflammation Res. (2021) 14:4881–91. doi: 10.2147/jir.S331579
139. Mahasaksiri T, Kositkuljorn C, Anuntrangsee T, and Suchonwanit P. Application of topical immunotherapy in the treatment of alopecia areata: A review and update. Drug Des Devel Ther. (2021) 15:1285–98. doi: 10.2147/dddt.S297858
140. Suchonwanit P, Kositkuljorn C, Mahasaksiri T, and Leerunyakul K. A comparison of the efficacy and tolerability of three corticosteroid treatment regimens in patients with alopecia areata. J Dermatolog Treat. (2022) 33:756–61. doi: 10.1080/09546634.2020.1773384
141. Phan K and Smith SD. Lichen planopilaris and dyslipidaemia: systematic review and meta-analysis. Clin Exp Dermatol. (2020) 45:611–3. doi: 10.1111/ced.14190
142. Jefferis BJ, Papacosta O, Owen CG, Wannamethee SG, Humphries SE, Woodward M, et al. Interleukin 18 and coronary heart disease: prospective study and systematic review. Atherosclerosis. (2011) 217:227–33. doi: 10.1016/j.atherosclerosis.2011.03.015
143. Yongpisarn T, Tejapira K, Thadanipon K, and Suchonwanit P. Vitamin D deficiency in non-scarring and scarring alopecias: A systematic review and meta-analysis. Front Nutr. (2024) 11:1479337. doi: 10.3389/fnut.2024.1479337
144. Mobini N, Tam S, and Kamino H. Possible role of the bulge region in the pathogenesis of inflammatory scarring alopecia: lichen planopilaris as the prototype. J Cutan Pathol. (2005) 32:675–9. doi: 10.1111/j.0303-6987.2005.00399.x
145. Del Duca E, Ruano Ruiz J, Pavel AB, Sanyal RD, Song T, Gay-Mimbrera J, et al. Frontal fibrosing alopecia shows robust T helper 1 and janus kinase 3 skewing. Br J Dermatol. (2020) 183:1083–93. doi: 10.1111/bjd.19040
146. Flamm A, Moshiri AS, Roche F, Onyekaba G, Nguyen J, James AJ, et al. Characterization of the inflammatory features of central centrifugal cicatricial alopecia. J Cutan Pathol. (2020) 47:530–4. doi: 10.1111/cup.13666
147. Araoye EF, Thomas JAL, and Aguh CU. Hair regrowth in 2 patients with recalcitrant central centrifugal cicatricial alopecia after use of topical metformin. JAAD Case Rep. (2020) 6:106–8. doi: 10.1016/j.jdcr.2019.12.008
Keywords: scarring alopecia, lichen planopilaris, frontal fibrosing alopecia, central centrifugal cicatricial alopecia, hair loss, immune-mediated alopecia, associated diseases, autoimmune diseases
Citation: Yongpisarn T, Tejapira K and Suchonwanit P (2025) Comorbidities in primary cicatricial alopecia: a systematic review and meta-analysis. Front. Immunol. 16:1516407. doi: 10.3389/fimmu.2025.1516407
Received: 24 October 2024; Accepted: 07 August 2025;
Published: 29 August 2025.
Edited by:
Diana Crisan, University Hospital Ulm, GermanyReviewed by:
Neusa Sakai Valente, University of São Paulo, BrazilIndrashis Podder, Sagore Dutta Hospital, India
Elena Niculet, Dunarea de Jos University, Romania
Copyright © 2025 Yongpisarn, Tejapira and Suchonwanit. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Poonkiat Suchonwanit, cG9vbmtpYXRAaG90bWFpbC5jb20=
†ORCID: Tanat Yongpisarn, orcid.org/0000-0002-1300-9624
Kasama Tejapira, orcid.org/0000-0001-6763-978X
Poonkiat Suchonwanit, orcid.org/0000-0001-9723-0563
 Tanat Yongpisarn
Tanat Yongpisarn Kasama Tejapira
Kasama Tejapira Poonkiat Suchonwanit
Poonkiat Suchonwanit