- 1Department of Rheumatology and Immunology, Affiliated Hospital of Shaoxing University (Shaoxing Municipal Hospital), Zhejiang, China
- 2First Teaching Hospital of Tianjin University of Traditional Chinese Medicine, National Clinical Research Center for Chinese Medicine Acupuncture and Moxibustion, Tianjin, China
- 3Capital Medical University Beijing Hospital of Traditional Chinese Medicine, Beijing, China
- 4Department of Dermatology, Tianjin Institute of Integrative Dermatology, Tianjin Academy of Traditional Chinese Medicine Affiliated Hospital, Tianjin, China
- 5Department of Rheumatology and Immunology, Center for General Practice Medicine, Zhejiang Provincial People's Hospital (Affiliated People's Hospital, Hangzhou Medical College), Zhejiang, China
- 6Institute of Rheumatology and Immunology, Hangzhou Medical College, Zhejiang, China
- 7Zhejiang Provincial Key Laboratory of Traditional Chinese Medicine Cultivation for Arthritis Diagnosis and Treatment, Zhejiang, China
Acrodermatitis continua of Hallopeau (ACH) is a rare aseptic pustular dermatosis for which clinical guidelines are lacking and treatment is largely based on case reports. Biologically targeted therapies offer new therapeutic ideas, with TNF antagonists such as adalimumab showing promising efficacy in both adults and children.The IL-17 and IL-23 axes play a key role in the pathogenesis of ACH, and anti-IL-17A and anti-IL-23 antibodies have shown therapeutic efficacy. In addition, new therapies such as IL-36 inhibitors and JAK inhibitors are being explored. Although biologics provide a new direction for ACH treatment, their safety and efficacy still need to be confirmed by large-scale clinical studies.
1 Introduction
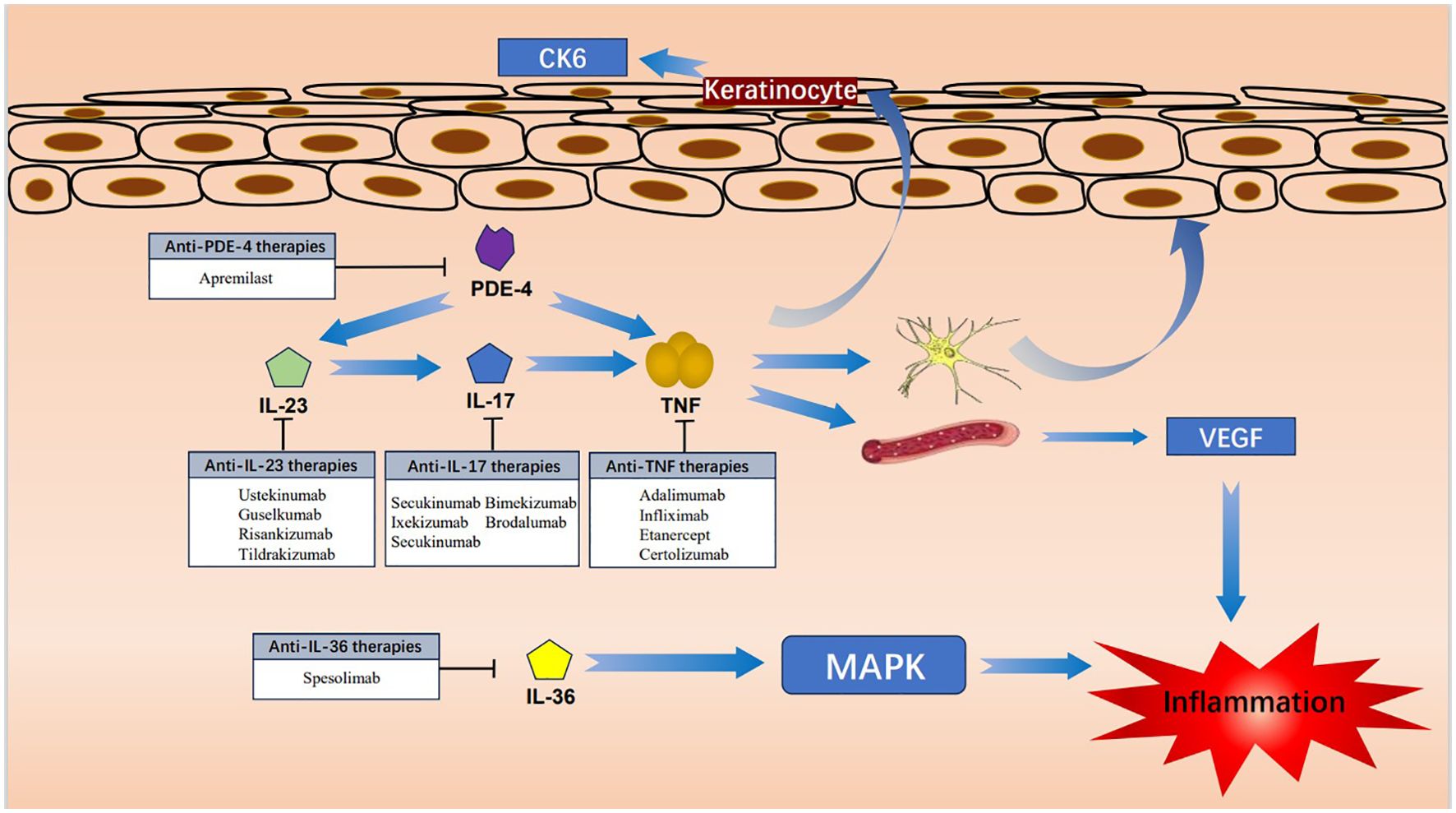
Acrodermatitis continua of Hallopeau (ACH) is considered to be a subtype of pustular psoriasis that presents as a sterile, pustular eruption commonly in the fingertips and toes (1, 2). Given the rarity of ACH and its resistance to conventional treatments, there is a notable absence of established clinical guidelines for ACH. The current approach to treatment is largely informed by case reports. However, some patients who have not responded well to topical therapy and systemic therapy, have found relief with biologic therapy. This development offers a promising new avenue for therapeutic intervention in ACH. In the following we describe the current advances in clinical biologics and immune-targeted agents in the treatment of ACH, as well as the mechanism of action (Figure 1), and summarize the efficacy of currently published biologics and immune-targeted agents for the treatment of ACH (Tables 1, 2).
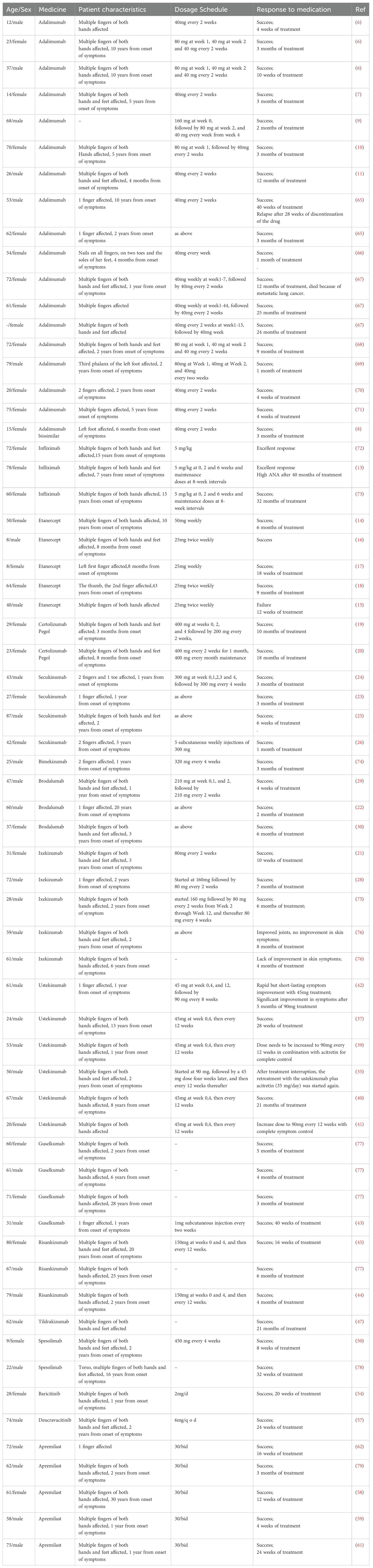
Table 1. Summary of ACH cases treated with biological and small molecule targeted immunomodulatory therapies.
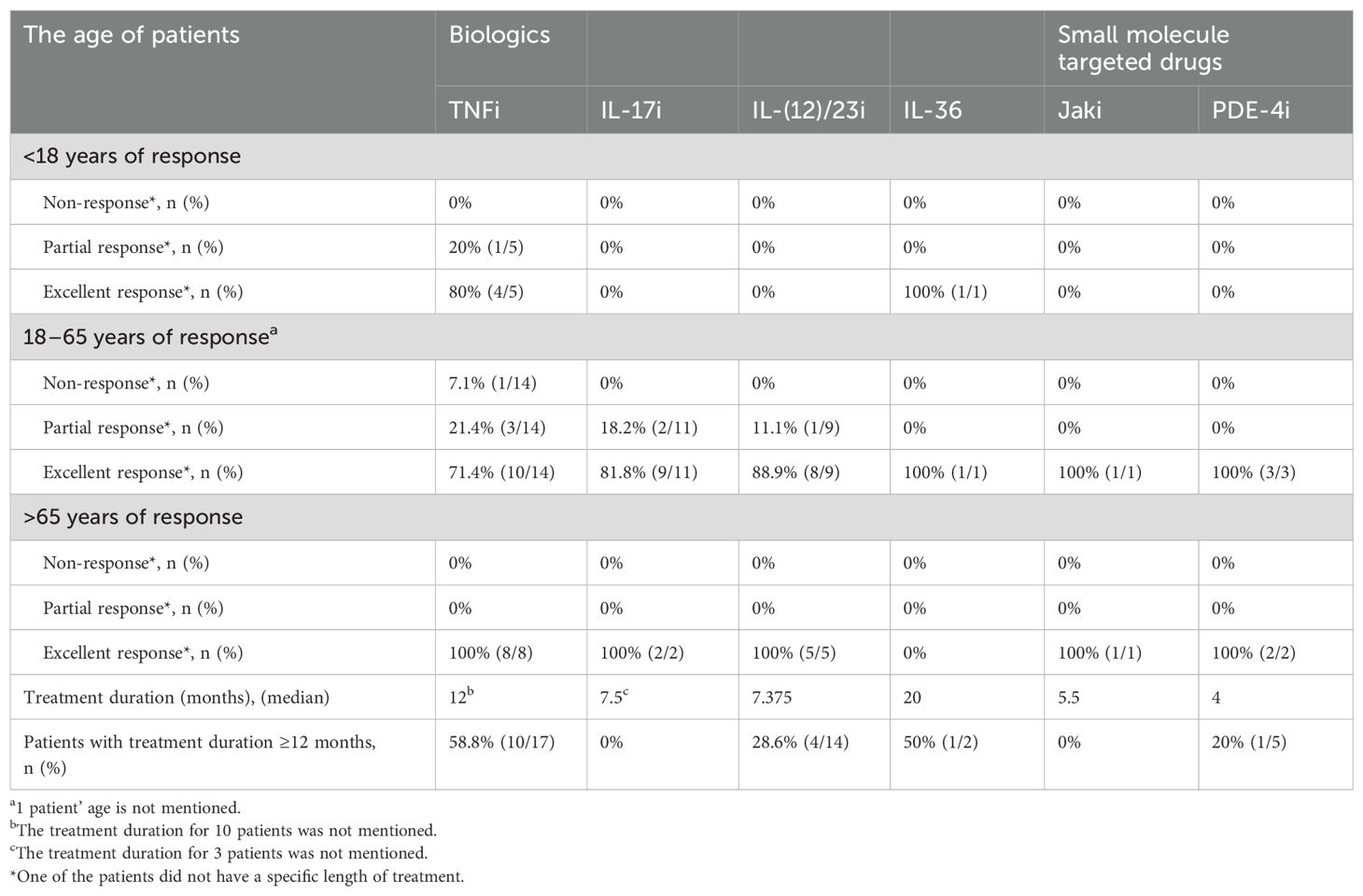
Table 2. Summary of the use of biologics and small molecule targeted drugs in ACH patients of different ages.
2 TNF inhibitor
Recent studies have elucidated the role of tumor necrosis factor (TNF) in the inflammatory response. TNF is able to participate in the inflammatory response by promoting the expression of cytokeratin 6 (CK6) in keratinocytes, recruiting dendritic cells to the dermis (3), stimulating the formation of VEGF (4), and promoting the proliferation of keratinocytes (5), which are involved in the pathogenesis of psoriasis. Nowadays, TNF-α inhibitors have demonstrated promising clinical outcomes in the treatment of patients with ACH.
Adalimumab, a fully human monoclonal antibody, functions by inhibiting the interaction between TNF-α and its p55/p75 cell surface receptors. IL36RN mutations have been identified with increased prevalence in generalized pustular psoriasis (GPP). Furthermore, therapeutic interventions employing IL-17A antagonists have demonstrated efficacy in GPP. Preliminary research suggests that patients with ACH are more likely than patients with other forms of pustular psoriasis to carry two distinct mutations (ie, IL36RN and AP1S3 or IL36RN and CARD14) (5). ACH represents a distinct subtype of GPP, and it is possible for both conditions to coexist. A notable difference in cytokine expression has been observed between those affecting the nail bed and those impacting skin lesions, explaining the suboptimal response to IL-17A inhibitors but improvement in erythema and pustules in ACH patients with severe nail damage, who responded well after switching to adalimumab treatment (6). Several case reports have shown that adalimumab is effective in pediatric patients with ACH, and a case involving a pediatric patient with an IL-36RN mutation exhibited a substantial improvement in their lesions after switching to adalimumab following an unsatisfactory response to secukinumab (7). Additionally, adalimumab biosimilar has been reported for the treatment of ACH in children with sustained improvement (8), demonstrating the efficacy of adalimumab in a rare cohort of pediatric ACH patients. Significant improvements have also been seen in adult cohorts of patients with ACH combined with Crohn’s disease, rheumatoid arthritis, lung cancer, and psoriatic arthritis treated with adalimumab (9–11).
Infliximab is a monoclonal antibody with high specificity for TNF-α, reducing the inflammatory response and inhibiting hyperproliferation of keratinocytes. Although there are many reported cases with favorable results with infliximab, in some cases this treatment is limited by secondary loss of efficacy, positive antinuclear antibody titers, and infusion reactions (12). Infliximab has a greater probability of secondary loss of efficacy than adalimumab. In addition, elevated liver enzymes may occur with infliximab (13).
Etanercept is a fusion protein produced by recombinant DNA technology that targets and binds to soluble TNF-α in the serum, effectively neutralizing its pro-inflammatory effects and reducing its systemic levels. Clinical reports have documented successful treatment outcomes with etanercept in certain cases of ACH (14). However, there are also instances of poor response to etanercept therapy (15), which may correlate with the degree of progression of the patient’s disease. There are reports that children with ACH who developed resistance to infliximab have exhibited a positive response to etanercept. Furthermore, children with ACH and associated bone resorption had a remarkable efficacy of etanercept. These findings provide a compelling basis for the use of etanercept in the pediatric treatment of ACH (16–18). However, etanercept may also have the potential to cause elevated liver enzymes and should be monitored carefully when used.
Certolizumab is a polyethylene glycolated anti-TNF agent, characterized by minimal to negligible placental transfer, which makes it a low-risk therapeutic option for pregnant patients (19). There are case reports of ACH patients who responded well to guselkumab and continued to use guselkumab after successful delivery after switching to certolizumab at 12 weeks of gestation when lesions and pain scores remained significantly improved (20). Although certolizumab is classified by the FDA as acceptable for use in pregnancy, there are still studies reporting traces of the drug detected in neonates. Therefore, clinical benefits and potential risks need to be assessed before using biologics in pregnant patients, and when the clinical benefits surpass the risks, the safer certolizumab may be used.
3 IL-17 inhibitor
There are no clinical guidelines for ACH, but biologics targeting the psoriasis pathway have been shown to be effective against ACH. IL-17 is known to play a major role in the pathogenesis of psoriasis, and both anti-IL17A and anti-IL17RA antibodies have shown promising results in the treatment of psoriasis (21, 22). Therefore, target therapy for ACH patients can be realized by inhibiting IL-17.
Secukinumab, the first IL-17 antibody biologic approved for the treatment of plaque psoriasis, PsA, and ankylosing spondylitis, is a recombinant high-affinity human immunoglobulin G1κ monoclonal antibody. Several cases of successful treatment of ACH with secukinumab have been reported in clinical practice (23–26). And in some patients, it was found that TNFα therapy would lose its efficacy with prolonged treatment time, or need to be increased in dosage or in combination with other drugs, and after switching to secukinumab, significant results were achieved, so secukinumab may be an effective treatment option for patients with ACH who have failed conventional therapies.
Both ixekizumab and secukinumab are monoclonal antibodies that specifically inhibit IL-17 A. However, a recent meta-analysis showed that ixekizumab improved the psoriasis area and severity index (PASI) significantly more than secukinumab over a 12-week course of treatment, which provides new insight into the selection of an appropriate IL-17 inhibitor (27). The main reasons for choosing ixekizumab treatment today are its rapid onset of action, ability to treat concomitant joint symptoms, and safety in patients with underlying HBV infection (28).
Brodalumab, a fully human anti-IL-17 receptor A (IL-17RA) monoclonal antibody, has recently been successfully used in three patients with ACH (22, 29, 30). Brodalumab blocks not only the binding of IL-17A, but also its subtype IL-17F, which may explain why some patients respond to brodalumab but not to IL-17A-blocking antibodies.
Bimekizumab, a monoclonal IgG 1 antibody, has been found to be safe and effective in treating patients with moderate to severe plaque psoriasis (31). A case-control study found a 50%-70% improvement in nail involvement in patients with ACH and complete improvement in skin and joint symptoms in patients with SAPHO syndrome after treatment with bimekizumab. This suggests that bimekizumab may be a safe and effective treatment for patients with PPP, ACH, and SAPHO syndrome (32).
The list of potential therapies for the effective treatment of ACH is expanding with the growing number of biologics targeting IL-17, a cytokine. However, the mechanism of IL-17 treatment for ACH is still unclear, and more clinical data and experimental support are still needed.
4 IL-12/IL-23 inhibitor
Many scholars now believe that ACH is a subtype of pustular psoriasis, and that the immune system and the interleukin (IL)-17/IL-23 axis play key roles in the pathogenic mechanism, so the treatment of ACH is guided by its similarity to pustular psoriasis, and therefore IL-12/23 inhibitors and IL-23 inhibitors appear to be a promising option for the treatment of ACH (33, 34).
Ustekinumab, a monoclonal antibody against the shared p40 subunit of cytokines IL-12 and IL-23, has been used in more than 2,000 patients and has been found to be effective in treating moderate to severe psoriasis (35). In a multicenter retrospective study, ustekinumab showed improvement in 75.0% of patients with ACH (36). Some case reports suggest that ustekinumab is effective in the treatment of ACH, but complete regression of the lesions requires higher doses than in psoriasis vulgaris, often in combination with acitretin. As with other biologics, loss of response to ustekinumab can occur when treatment is interrupted, and reattainment of the therapeutic effect requires an increase in dosage or a combination of other medications (35, 37–39). However, in some case reports patients switched to ustekinumab in combination with acitretin after failing TNF inhibitor therapy and experienced rapid and sustained improvement in their condition (40–42). Indicates that treatment of ACH with ustekinumab may require high-dose maintenance therapy in combination with acitretin and methotrexate and may be an optional alternative therapy after TNF inhibitor therapy.
Guselkumab is also an IL-23 inhibitor, and a case of an ACH patient with single finger involvement whose rash continued to worsen after ixekizumab treatment was reported to have disappeared after switching to guselkumab microinjection therapy. This suggests that local injection of microdose biologics may be a precise and safe treatment for patients with limited ACH. Local injection of microdose biologics is more precise than systemic standard dose biologics, allowing antibodies to reach the target lesion more efficiently, resulting in lower costs and potential side effects (43). However, more clinical cases and clinical studies are still needed.
Risankizumab is a monoclonal antibody that selectively binds IL-23 cytokines and blocks their interaction with the IL-23 receptor. According to multiple case reports, risankizumab has shown good efficacy and safety in the treatment of elderly ACH patients, so we can prioritize risankizumab in elderly ACH patients (44, 45).
Tildrakizumab is a high affinity monoclonal antibody to humanized immunoglobulin IgG1/κ that specifically binds the IL-23p19 subunit. Global Phase III Clinical Trials (reSURFACE 1 and reSURFACE 2) demonstrate efficacy of tildrakizumab in patients with moderate to severe plaque psoriasis (46). And a case of successful treatment of an ACH patient with multiple comorbidities (advanced malignancy, recurrent pyothorax, psoriatic arthritis) using tildrakizumab has been reported (47), so tildrakizumab may be considered for ACH patients with malignancy or high risk of infection.
5 IL-36 inhibitor
A current clinical study in Taiwan suggests that approximately 50% of patients with interleukin-β 36 receptor antagonist (IL 36 RN) deficiency will exhibit symptoms of ACH, which provides pathophysiologic support for the use of IL-36 inhibitors in patients with ACH (48). In 2022, spesolimab, a monoclonal antibody targeting the IL-36 receptor, was approved for the treatment of GPP, and the safety and efficacy of spesolimab was evaluated in a phase I trial in seven patients with moderate GPP, with all seven patients showing a 70%-80% improvement and no significant adverse events in patients or healthy controls (49). Given the similarity between GPP and ACH, spesolimab has also been used in patients with ACH, and in this case, the patient experienced a rapid and significant response to the use of spesolimab (50). And current clinical trials suggest that GPP patients, with or without the IL36RN variant, may respond to spesolimab (51).
6 Jak inhibitor
The main Jak inhibitor currently used for the treatment of patients with ACH is baricitinib, an oral small molecule Janus kinase (JAK1) and (JAK2) inhibitor, which has good use in patients with atopic dermatitis (AD) (52). Baricitinib completely inhibits the JAK-STAT (signal transducer and activator of transcription) signaling pathway, thereby inhibiting targeted cytokines (e.g., TNF-α, IL-17, and IL-23), which could be a potential explanation for baricitinib treatment of ACH (53). A case has been reported in which a patient with ACH who had failed conventional treatment was switched to baricitinib with significant improvement in skin and joint symptoms and no new progression of bone erosion. This suggests that baricitinib is a potential treatment option for ACH and may inhibit the progression of bone disease (54).
TYK2, also a JAK family gene, plays a key role in mediating the signaling of multiple cytokines that cause inflammation. Deucravacitinib is a TYK2 inhibitor that inhibits TYK2-mediated inflammatory and immune responses by selectively inhibiting TYK2 activation and blocking signaling of inflammatory cytokines such as IL-23, IL-12, and type I interferon (IFN) (55). A phase III clinical trial in Japan demonstrated favorable clinical efficacy of deucravacitinib in patients with GPP, TYK2 inhibitor response in GPP may be facilitated by an unregulated IL-36 signaling pathway (56). Only one case has been reported of an elderly patient with ACH after treatment with smoking cessation, tonsillectomy, acitretin and cyclosporine failed, and after 24 weeks of treatment with deucravacitinib, the patient’s symptoms, including nail lesions, completely disappeared (57).
7 PDE-4 inhibitor
Apremilast, an oral phosphodiesterase 4 inhibitor, modulates pro-inflammatory pathways to promote anti-inflammatory activity, increases intracellular concentrations of cAMP, and blocks the production of pro-inflammatory cytokines, such as TNF-α, IFN-γ, IL-17, IL-23, and IL-12 (58, 59), which have been shown to be involved in the inflammatory response in systemic pustulosis and ACH (60). Apremilast is a relatively safe and easy to administer drug compared to other biologics. Based on the case reports reported to date, apremilast can be found to be a potential therapeutic option for ACH patients with comorbidities for biologics (e.g., tuberculosis, cancer, hepatitis B infection) and is also relatively safe and efficacious in elderly ACH patients with multiple comorbidities (58, 61, 62).
With further insights into the pathogenesis and treatment of psoriasis, most of the biologic therapies available for the treatment of plaque psoriasis have also shown significant efficacy in patients with ACH, even though some of the biologics require higher dosages and combination therapies, and still offer new therapeutic ideas for the treatment of ACH. However, with the emergence of new biologics and small molecule targeted drugs, the therapeutic choices for ACH patients will be more promising (63, 64). The number of cases reported in the literature is still small, most studies have small sample sizes, and the safety of biologics in treating ACH patients needs to be confirmed by large-scale clinical studies, and we look forward to further studies to confirm the efficacy of biologically targeted therapies in ACH patients.
Author contributions
QS: Conceptualization, Writing – original draft. LH: Writing – original draft. ZL: Writing – review & editing. YW: Methodology, Conceptualization, Validation, Funding acquisition, Visualization, Writing – review & editing. ZY: Writing – review & editing. CL: Writing – review & editing. ZZ: Writing – review & editing, Methodology, Conceptualization, Validation, Funding acquisition, Visualization.
Funding
The author(s) declare financial support was received for the research and/or publication of this article. This work was supported by the Zhejiang Provincial Key Laboratory of Traditional Chinese Medicine Cultivation for Arthritis Diagnosis and Treatment (grant number:C-2023-W2083); and National Natural Science Foundation of China (grant number: 82473512).
Acknowledgments
The authors are grateful to all participants for their participation in this paper.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The authors declare that no Gen AI was used in the creation of this manuscript.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Bhate C and Schwartz RA. Acrodermatitis continua of Hallopeau: historical perspectives and modern management. Ital J Dermatol Venerol. (2022) 157:132–6. doi: 10.23736/S2784-8671.21.07007-9
2. Chularojanamontri L, Rattanakorn K, Julanon N, Chuamanochan M, and Griffiths CEM. Acrodermatitis continua of Hallopeau and generalised pustular psoriasis: Should they be the same or different entities? Exp Dermatol. (2023) 32:1235–45. doi: 10.1111/exd.14805
3. Sehgal VN, Verma P, Sharma S, Srivastava G, Aggarwal AK, Rasool F, et al. Acrodermatitis continua of Hallopeau: evolution of treatment options. Int J Dermatol. (2011) 50:1195–211. doi: 10.1111/j.1365-4632.2011.04993.x
4. Maliyar K, Crowley EL, Rodriguez-Bolanos F, O'Toole A, and Gooderham MJ. The use of biologic therapy in the treatment of acrodermatitis continua of hallopeau: A review. J Cutan Med Surg. (2019) 23:428–35. doi: 10.1177/1203475419836435
5. Smith MP, Ly K, Thibodeaux Q, Bhutani T, Liao W, and Beck KM. Acrodermatitis continua of Hallopeau: clinical perspectives. Psoriasis (Auckl). (2019) 9:65–72. doi: 10.2147/PTT.S180608
6. Zhang Z, Wu X, Han C, Chen M, and Liu Y. Adalimumab in drei Fällen von Anti-IL-17-refraktärer Acrodermatitis continua Hallopeau. J Dtsch Dermatol Ges. (2023) 21:785–7. doi: 10.1111/ddg.15041_g
7. O'Malley S, Duff E, and Collins S. Paediatric acrodermatitis continua of Hallopeau: perspectives of a patient and their mother. Clin Exp Dermatol. (2023) 49:92–3. doi: 10.1093/ced/llad333
8. Megna M, Fabbrocini G, Costa C, and Ferrillo M. A case of pediatric acrodermatitis continua of Hallopeau successfully treated with adalimumab biosimilar. Dermatol Ther. (2020) 33:e13327. doi: 10.1111/dth.13327
9. Caputo F, Parro S, and Zoli G. Adalimumab for a co-existing clinical condition of Crohn's disease and acrodermatitis continua of Hallopeau. J Crohns Colitis. (2011) 5:649. doi: 10.1016/j.crohns.2011.08.017
10. Crowley EL, Maliyar K, Rodriguez-Bolanos F, O'Toole A, and Gooderham MJ. Acrodermatitis continua of Hallopeau successfully treated with adalimumab: A case report. SAGE Open Med Case Rep. (2019) 7:2050313x19829620. doi: 10.1177/2050313X19829620
11. Lefkir S, Slimani S, Brahimi N, and Ladjouze-Rezig A. Successful treatment of Acrodermatitis continua of Hallopeau associated with psoriatic arthritis with adalimumab. Eur J Rheumatol. (2015) 2:78–9. doi: 10.5152/eurjrheum.2015.0072
12. Hoegler KM, John AM, Handler MZ, and Schwartz RA. Generalized pustular psoriasis: a review and update on treatment. J Eur Acad Dermatol Venereol. (2018) 32:1645–51. doi: 10.1111/jdv.2018.32.issue-10
13. Ahmad K and Rogers S. Three years' experience with infliximab in recalcitrant psoriasis. Clin Exp Dermatol. (2006) 31:630–3. doi: 10.1111/j.1365-2230.2006.02170.x
14. Weisshaar E and Diepgen TL. Successful etanercept therapy in therapy refractory acrodermatitis continua suppurativa Hallopeau. J Dtsch Dermatol Ges. (2007) 5:489–92. doi: 10.1111/j.1610-0387.2007.06332.x
15. Adişen E, Oztaş M, and Gürer MA. Lack of efficacy of etanercept in acrodermatitis continua of Hallopeau. Int J Dermatol. (2007) 46:1205–7. doi: 10.1111/j.1365-4632.2007.03251.x
16. Alorainy M, Alshaya M, Al-Rikabi A, Ayesh M, and Alsaif F. Acrodermatitis continua of hallopeau with bone resorption in an 8-year-old patient: A case report. Case Rep Dermatol. (2017) 9:259–64. doi: 10.1159/000485370
17. Ruggiero A, Patruno C, Napolitano M, Megna M, Gisonni P, Cirillo T, et al. Etanercept for pediatric acrodermatitis continua of Hallopeau: A case report and literature review. Dermatol Ther. (2020) 33:e14046. doi: 10.1111/dth.14046
18. Thielen AM, Barde C, Marazza G, and Saurat JH. Long-term control with etanercept (Enbrel) of a severe acrodermatitis continua of Hallopeau refractory to infliximab (Remicade). Dermatology. (2008) 217:137–9. doi: 10.1159/000134613
19. Rizzetto G, Diotallevi F, Molinelli E, Radi G, and Offidani AM. Certolizumab pegol: A new therapeutic approach for acrodermatitis continua of hallopeau. Skin Appendage Disord. (2022) 8:146–9. doi: 10.1159/000519144
20. Al-Khawaga S, Krishnankutty R, Sher G, Hussain K, Buddenkotte J, and Steinhoff M. Treatment and molecular profiling of acrodermatitis continua of Hallopeau during pregnancy using targeted therapy. JAAD Case Rep. (2021) 16:164–7. doi: 10.1016/j.jdcr.2021.09.002
21. Miller AC, Holland TE, and Cohen DJ. Treatment of acrodermatitis continua of hallopeau with ixekizumab. J Dermatolog Treat. (2021) 32:117–9. doi: 10.1080/09546634.2019.1628170
22. Milani-Nejad N and Kaffenberger J. Treatment of recalcitrant acrodermatitis continua of hallopeau with brodalumab. J Drugs Dermatol. (2019) 18:1047.
23. Galluzzo M, D'Adamio S, Teoli M, Bianchi L, and Talamonti M. Biologic therapy for acrodermatitis continua of Hallopeau: Successful treatment with secukinumab and review of the literature. Dermatol Ther. (2019) 32:e12899. doi: 10.1111/dth.12899
24. Balestri R, Rech G, Tasin L, Rizzoli L, and Girardelli CR. Acrodermatitis continua of Hallopeau successfully treated with secukinumab. J Dermatolog Treat. (2018) 29:3–5. doi: 10.1080/09546634.2018.1527993
25. Muggli D, Maul JT, Anzengruber F, Fopp MW, and Navarini AA. Secukinumab for acrodermatitis continua of hallopeau. JAMA Dermatol. (2017) 153:336–7. doi: 10.1001/jamadermatol.2016.5059
26. Baron JA. Acrodermatitis of Hallopeau and erosive oral mucositis successfully treated with secukinumab. JAAD Case Rep. (2017) 3:215–8. doi: 10.1016/j.jdcr.2017.02.016
27. Papp KA, Leonardi CL, Blauvelt A, Reich K, Korman NJ, Ohtsuki M, et al. Ixekizumab treatment for psoriasis: integrated efficacy analysis of three double-blinded, controlled studies (UNCOVER-1, UNCOVER-2, UNCOVER-3). Br J Dermatol. (2018) 178:674–81. doi: 10.1111/bjd.16050
28. Battista T, Guerrasio G, De Fata Salvatores G, Peduto T, Fabbrocini G, and Di Guida A. Acrodermatitis continua of Hallopeau successfully treated with ixekizumab: A case report. Dermatol Ther. (2022) 35:e15383. doi: 10.1111/dth.15383
29. Bardazzi F, Di Altobrando A, and Lacava R. Brodalumab in the treatment of recalcitrant acrodermatitis continua of Hallopeau. Dermatol Ther. (2020) 33:e14301. doi: 10.1111/dth.14301
30. Passante M, Dastoli S, Nisticò SP, Bennardo L, and Patruno C. Effectiveness of brodalumab in acrodermatitis continua of Hallopeau: A case report. Dermatol Ther. (2020) 33:e13170. doi: 10.1111/dth.13170
31. Reich K, Warren RB, Lebwohl M, Gooderham M, Strober B, Langley RG, et al. Bimekizumab versus secukinumab in plaque psoriasis. N Engl J Med. (2021) 385:142–52. doi: 10.1056/NEJMoa2102383
32. Passeron T, Perrot JL, Jullien D, Goujon C, Ruer M, Boyé T, et al. Treatment of severe palmoplantar pustular psoriasis with bimekizumab. JAMA Dermatol. (2024) 160:199–203. doi: 10.1001/jamadermatol.2023.5051
33. Menter A, Van Voorhees AS, and Hsu S. Pustular psoriasis: A narrative review of recent developments in pathophysiology and therapeutic options. Dermatol Ther (Heidelb). (2021) 11:1917–29. doi: 10.1007/s13555-021-00612-x
34. Genovese G, Moltrasio C, Cassano N, Maronese CA, Vena GA, and Marzano AV. Pustular psoriasis: from pathophysiology to treatment. Biomedicines. (2021) 9. doi: 10.3390/biomedicines9121746
35. Adışen E, Özer İ, Temel B, and Gürer MA. Ustekinumab for the treatment of acrodermatitis continua of Hallopeau refractory to anti-TNF agents. Dermatol Ther. (2017) 30. doi: 10.1111/dth.2017.30.issue-2
36. Gargiulo L, Toso F, Ibba L, Valenti M, Costanzo A, and Narcisi A. Biologics for the treatment of severe acrodermatitis continua of hallopeau: report of two cases successfully treated with ixekizumab and ustekinumab. Dermatol Pract Concept. (2023) 13. doi: 10.5826/dpc.1302a103
37. Shi L, Zeng L, Huang X, Wang B, Zhang Y, and Zhang G. Acrodermatitis continua of hallopeau successfully treated with ustekinumab: A case report and literature review. Clin Cosmet Investig Dermatol. (2023) 16:951–7. doi: 10.2147/CCID.S404860
38. Husson B, Barbe C, Hegazy S, Seneschal J, Aubin F, Mahé E, et al. Efficacy and safety of TNF blockers and of ustekinumab in palmoplantar pustulosis and in acrodermatitis continua of Hallopeau. J Eur Acad Dermatol Venereol. (2020) 34:2330–8. doi: 10.1111/jdv.v34.10
39. Saunier J, Debarbieux S, Jullien D, Garnier L, Dalle S, and Thomas L. Acrodermatitis continua of Hallopeau treated successfully with ustekinumab and acitretin after failure of tumour necrosis factor blockade and anakinra. Dermatology. (2015) 230:97–100. doi: 10.1159/000367690
40. Palacios-Álvarez I, Simal-Gómez G, Mas-Vidal A, and Bernad-Alonso I. Treatment of acrodermatitis continua of Hallopeau with ustekinumab as monotherapy after failure of anti-TNF agents. J Dtsch Dermatol Ges. (2018) 16:611–3.
41. Cymerman RM and Cohen DE. Treatment of acrodermatitis continua of hallopeau with ustekinumab as monotherapy. JAMA Dermatol. (2016) 152:346–8. doi: 10.1001/jamadermatol.2015.3444
42. Adas A, Dadban A, Arnault JP, Chaby G, and Lok C. Acrodermatitis continua of Hallopeau response to optimized biological therapy. Dermatol Online J. (2019) 25. doi: 10.5070/D3252042897
43. Wang Y, Zhou C, Hou Y, Diao Z, Chen D, and Yin Z. Local injection of micro-dose guselkumab for acrodermatitis continua of Hallopeau after failure of systemic ixekizumab treatment. Int J Dermatol. (2024) 63:547–8. doi: 10.1111/ijd.17036
44. Orsini D, Iacovelli P, Frascione P, and Pacifico A. Acrodermatitis continua of Hallopeau in an elderly patient successfully and rapidly treated with risankizumab: a case report. J Dermatolog Treat. (2023) 34:2229468. doi: 10.1080/09546634.2023.2229468
45. Hugo J, Gkalpakioti P, Arenbergerova M, Arenberger P, and Gkalpakiotis S. Acrodermatitis continua Hallopeau successfully treated by risankizumab. Int J Dermatol. (2021) 60:e144–5. doi: 10.1111/ijd.15391
46. Reich K, Warren RB, Iversen L, Puig L, Pau-Charles I, Igarashi A, et al. Long-term efficacy and safety of tildrakizumab for moderate-to-severe psoriasis: pooled analyses of two randomized phase III clinical trials (reSURFACE 1 and reSURFACE 2) through 148 weeks. Br J Dermatol. (2020) 182:605–17. doi: 10.1111/bjd.v182.3
47. Nguyen A, D'Aguanno K, Ridha Z, Tsoukas A, and Netchiporouk E. Successful treatment of acrodermatitis continua of Hallopeau with tildrakizumab: A case report. SAGE Open Med Case Rep. (2023) 11:2050313x231180775. doi: 10.1177/2050313X231180775
48. Hsieh CY, Huang YW, Huang YH, and Tsai TF. Deficiency of interleukin-36 receptor antagonist (DITRA): An analysis of 58 Chinese patients in a tertiary hospital in Taiwan. Exp Dermatol. (2023) 32:1272–8. doi: 10.1111/exd.14783
49. Ratnarajah K, Jfri A, Litvinov IV, and Netchiporouk E. Spesolimab: A novel treatment for pustular psoriasis. J Cutan Med Surg. (2020) 24:199–200. doi: 10.1177/1203475419888862
50. Wang Y, Zhang L, Zheng J, Li X, and Chen L. Spesolimab response in a girl with acrodermatitis continua of hallopeau. JAMA Dermatol. (2024) 160(4):476–7. doi: 10.1001/jamadermatol.2023.6349
51. Burden AD, Okubo Y, Zheng M, Thaçi D, van de Kerkhof P, Hu N, et al. Efficacy of spesolimab for the treatment of generalized pustular psoriasis flares across pre-specified patient subgroups in the Effisayil 1 study. Exp Dermatol. (2023) 32:1279–83. doi: 10.1111/exd.14824
52. Hoy SM. Baricitinib: A review in moderate to severe atopic dermatitis. Am J Clin Dermatol. (2022) 23:409–20. doi: 10.1007/s40257-022-00684-1
53. Mayence A and Vanden Eynde JJ. Baricitinib: A 2018 novel FDA-approved small molecule inhibiting janus kinases. Pharm (Basel). (2019) 12:37. doi: 10.3390/ph12010037
54. Han GM, Yang WS, and Yang B. Inhibition of progression of acrodermatitis continua of hallopeau with baricitinib. JAMA Dermatol. (2021) 157:466–8. doi: 10.1001/jamadermatol.2021.0045
55. Blauvelt A, Rich P, Sofen H, Strober B, Merola JF, Lebwohl M, et al. Deucravacitinib, a selective, allosteric tyrosine kinase 2 inhibitor, in scalp psoriasis: A subset analysis of two phase 3 randomized trials in plaque psoriasis. J Am Acad Dermatol. (2024) 90:775–82. doi: 10.1016/j.jaad.2023.11.060
56. Imafuku S, Okubo Y, Tada Y, Ohtsuki M, Colston E, Napoli A, et al. Deucravacitinib, an oral, selective, allosteric tyrosine kinase 2 inhibitor, in Japanese patients with moderate to severe plaque, erythrodermic, or generalized pustular psoriasis: Efficacy and safety results from an open-label, phase 3 trial. J Dermatol. (2024) 51:365–79. doi: 10.1111/1346-8138.17074
57. Kan Y and Uhara H. Successful treatment of acrodermatitis continua of Hallopeau by TYK2 inhibitor with every other day. J Dermatolog Treat. (2024) 35:2316239. doi: 10.1080/09546634.2024.2316239
58. Fusta-Novell X, Esquius M, and Creus-Vila L. Rapid and sustained response to apremilast in a patient with long-standing acrodermatitis continua of Hallopeau. Dermatol Online J. (2022) 28. doi: 10.5070/D328659731
59. Lanna C, Cesaroni GM, Mazzilli S, Lozzi F, Palumbo V, Diluvio L, et al. Nails as immune-privileged sites: A case of disabling Acrodermatitis continua of Hallopeau successfully treated with Apremilast. Dermatol Ther. (2019) 32:e12946. doi: 10.1111/dth.12946
60. Johnston A, Xing X, Wolterink L, Barnes DH, Yin Z, Reingold L, et al. IL-1 and IL-36 are dominant cytokines in generalized pustular psoriasis. J Allergy Clin Immunol. (2017) 140:109–20. doi: 10.1016/j.jaci.2016.08.056
61. Calleja Algarra A, Aragón Miguel R, Velasco Tamariz V, Prieto Barrios M, Andrés Lencina JJ, Vico Alonso C, et al. Apremilast as a new treatment option for Acrodermatitis continua of Hallopeau. Australas J Dermatol. (2019) 60:e237–8. doi: 10.1111/ajd.12990
62. Megna M, Potestio L, Di Caprio N, Tajani A, Fabbrocini G, and Annunziata A. Acrodermatitis continua of Hallopeau: Is apremilast an efficacious treatment option? Dermatol Ther. (2022) 35:e15423. doi: 10.1111/dth.15423
63. Kiszewski AE, De Villa D, Scheibel I, and Ricachnevsky N. An infant with acrodermatitis continua of Hallopeau: successful treatment with thalidomide and UVB therapy. Pediatr Dermatol. (2009) 26:105–6. doi: 10.1111/j.1525-1470.2008.00838.x
64. Bordignon M, Zattra E, Albertin C, and Belloni-Fortina A. Successful treatment of a 9-year-old boy affected by acrodermatitis continua of Hallopeau with targeted ultraviolet B narrow-band phototherapy. Photodermatol Photoimmunol Photomed. (2010) 26:41–3. doi: 10.1111/j.1600-0781.2009.00468.x
65. Di Costanzo L, Napolitano M, Patruno C, Cantelli M, and Balato N. Acrodermatitis continua of Hallopeau (ACH): two cases successfully treated with adalimumab. J Dermatolog Treat. (2014) 25:489–94. doi: 10.3109/09546634.2013.848259
66. Tobin AM and Kirby B. Successful treatment of recalcitrant acrodermatitis continua of Hallopeau with adalimumab and acitretin. Br J Dermatol. (2005) 153:445–6. doi: 10.1111/j.1365-2133.2005.06759.x
67. Ryan C, Collins P, Kirby B, and Rogers S. Treatment of acrodermatitis continua of Hallopeau with adalimumab. Br J Dermatol. (2009) 160:203–5. doi: 10.1111/j.1365-2133.2008.08893.x
68. Puig L, Barco D, Vilarrasa E, and Alomar A. Treatment of acrodermatitis continua of Hallopeau with TNF-blocking agents: case report and review. Dermatology. (2010) 220:154–8. doi: 10.1159/000277415
69. Sopkovich JA, Anetakis Poulos G, and Wong HK. Acrodermatitis continua of hallopeau successfully treated with adalimumab. J Clin Aesthet Dermatol. (2012) 5:60–2.
70. Müeller RB, Ogilvie A, Schwarz S, Kern P, Schett G, and Sticherling M. Adalimumab treatment of a patient with psoriasis suppurativa Hallopeau associated osteoarthropathy. Clin Exp Rheumatol. (2009) 27:887.
71. Conti A, Ciardo S, Mandel VD, Bigi L, and Pellacani G. Speckled variance optical coherence tomography for the assessment of nail involvement in acrodermatitis continua of Hallopeau: A case study. J Int Med Res. (2016) 44:119–23. doi: 10.1177/0300060515593263
72. Samotij D, Gawron E, Szczęch J, Ostańska E, and Reich A. Acrodermatitis continua of hallopeau evolving into generalized pustular psoriasis following COVID-19: A case report of a successful treatment with infliximab in combination with acitretin. Biologics. (2021) 15:107–13. doi: 10.2147/BTT.S302164
73. Rubio C, Martin MA, Arranz Sánchez DM, Vidaurrazaga C, and Casado M. Excellent and prolonged response to infliximab in a case of recalcitrant acrodermatitis continua of Hallopeau. J Eur Acad Dermatol Venereol. (2009) 23:707–8. doi: 10.1111/j.1468-3083.2008.02998.x
74. Cirone KD and Lovegrove FE. Acrodermatitis continua of Hallopeau successfully treated with bimekizumab: A case report. SAGE Open Med Case Rep. (2023) 11:2050313x231160937. doi: 10.1177/2050313X231160937
75. Xia RY, Liu J, Chen DD, Diao ZY, Gao YM, and Yin ZQ. Ixekizumab for the treatment of acrodermatitis continua of hallopeau and inverse psoriasis. Indian J Dermatol. (2023) 68:234. doi: 10.4103/ijd.ijd_884_22
76. Schmid E, Mohr J, Schön MP, and Mössner R. Two cases of acrodermatitis continua suppurativa (Hallopeau's disease) treated with IL-17A inhibitors. J Dtsch Dermatol Ges. (2019) 17:643–5. doi: 10.1111/ddg.2019.17.issue-6
77. Langer N, Wilsmann-Theis D, Kromer C, Mohr J, and Mössner R. Successful therapy of acrodermatitis continua of Hallopeau with IL-23 blockers -two new cases. J Dtsch Dermatol Ges. (2021) 19:1504–7. doi: 10.1111/ddg.v19.10
78. Wen P, Liu C, Wang T, Jiang X, Wang P, and Wang S. Successful treatment of acrodermatitis continua of Hallopeau coexisting with generalized pustular psoriasis with spesolimab: a case report. Front Immunol. (2024) 15:1338285. doi: 10.3389/fimmu.2024.1338285
Keywords: Acrodermatitis continua of Hallopeau, immune targeted therapy, TNF inhibitor, IL-17 inhibitor, JAK inhibitor
Citation: Sun Q, Han L, Lin Z, Wu Y, Li C, Ying Z and Zhao Z (2025) Acrodermatitis continua of Hallopeau: a review and update on biological and small molecule targeted immunomodulatory therapies. Front. Immunol. 16:1525821. doi: 10.3389/fimmu.2025.1525821
Received: 10 November 2024; Accepted: 23 July 2025;
Published: 15 August 2025.
Edited by:
Nechama Sharon, Laniado Hospital, IsraelReviewed by:
Aikaterini Patsatsi, Aristotle University of Thessaloniki, GreeceMichael A. Firer, Ariel University, Israel
Copyright © 2025 Sun, Han, Lin, Wu, Li, Ying and Zhao. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Zuotao Zhao, emhhb3p1b3Rhb3Rhb0AxNjMuY29t; Chen Li, Y2FzaW8xOTgxQDE2My5jb20=
†These authors have contributed equally to this work and share first authorship
 Qiong Sun1†
Qiong Sun1† Luyao Han
Luyao Han Yuanhao Wu
Yuanhao Wu Chen Li
Chen Li Zhenhua Ying
Zhenhua Ying