- Department of Neurology, The First Affiliated Hospital of Xi’an Jiao Tong University, Xi’an, China
Immune checkpoint inhibitors (ICIs) targeting programmed cell death protein 1 (PD-1), programmed cell death protein 1 ligand, and cytotoxic T-lymphocyte-associated protein 4, are important therapeutic approaches for malignancies. However, these novel treatment measures are associated with immune-related adverse events. We report the first reported case of autoimmune GFAP astrocytopathy-associated myelitis in a patient with hepatocellular carcinoma that was treated with sintilimab (an anti-PD-1 monoclonal antibody) immunotherapy. Additionally, literature review identified 21 previously reported cases of PD-1 inhibitor-associated myelitis, demonstrating similar clinical features. All patients received ICI discontinuation and high-dose glucocorticoid therapy, with the addition of other immune therapies in 15 patients. Clinical improvement was observed in 13 patients. Clinicians should consider autoimmune GFAP astrocytopathy-associated myelitis as a potential differential diagnosis among patients exhibiting neurological symptoms during or following ICI therapy.
1 Introduction
Immune checkpoint inhibitors (ICIs), which specifically target the programmed cell death protein 1/programmed death-ligand 1 (PD-1/PD-L1) axis and cytotoxic T-lymphocyte-associated protein 4, have great clinical efficacy as novel therapeutic agents in contemporary oncology practice. Sintilimab, a selective antibody against PD-1, inhibits the interaction between PD-1 and its ligands and restores anti-tumor immune responses (1). Sintilimab in combination with anti-angiogenesis agents has demonstrated significant clinical benefits in Chinese population with hepatocellular carcinoma (HCC) (2–5). Despite these advances, ICI-mediated immune hyperactivation may trigger diverse immune-related adverse events (irAEs) (6–8), including exacerbation or production of autoimmune neurological disorders (9), such as myasthenia gravis (10), motor polyradiculopathy (11), transvers myelitis (12, 13), and Guillain-Barr´e syndrome (14).
Autoimmune glial fibrillary acidic protein (GFAP) astrocytopathy is a central nervous system (CNS) inflammatory entity characterized by acute meningoencephalomyelitis with pathognomonic linear perivascular gadolinium enhancement on spinal or brain MRI and cerebrospinal fluid (CSF) lymphocytic pleocytosis, elevated protein, and GFAP-IgG positivity (15). Pembrolizumab-associated GFAP meningoencephalomyelitis was reported in a patient with non-small cell lung cancer (NSCLC) (16). Current understanding of ICI-induced GFAP astrocytopathy remains limited.
Herein, we describe the first case of sintilimab-induced GFAP astrocytopathy presenting as isolated myelitis in an HCC patient. And we conducted a literature review of all published cases of PD-1 inhibitor-associated myelitis, analyzing clinical phenotypes, therapeutic strategies, and prognostic outcomes. Our findings highlight the need for heightened clinical suspicion of autoimmune GFAP astrocytopathy in patients developing neurological manifestations during PD-1/PD-L1 blockade.
2 Case presentation
A 40-year-old male presented to our department with a 20-day history of gait disturbance. Neurological examination revealed mild instability on bilateral heel-to-shin testing and instability during the Romberg test with eyes closed. Muscle strength in all limbs, deep tendon reflexes, and superficial sensation were normal.
The patient had a 20-year history of hepatitis B virus (HBV) infection, treated with irregular Chinese herbal medicine for 19 years followed by 1 year of entecavir antiviral therapy. He was diagnosed with hepatocellular carcinoma (HCC, AJCC stage IIIa, T4N0M0) on February 1, 2024. The patient underwent a structured therapeutic regimen beginning with hepatic arterial infusion chemotherapy (HAIC) on February 5, 2024, using idarubicin, oxaliplatin, and fluorouracil. This was followed three days later by combined immunotherapy with sintilimab (200 mg) and bevacizumab (500 mg) on February 8, 2024. The treatment cycle repeated on March 1, 2024 with a second HAIC session using the same triple-agent protocol, subsequently reinforced by another dose of sintilimab and bevacizumab on March 5, 2024. A modified HAIC approach was implemented on April 12, 2024, omitting idarubicin while maintaining oxaliplatin and fluorouracil, followed by the final scheduled dual immunotherapy administration on April 15, 2024. Throughout this 10-week period, the patient received three cycles of HAIC (two with idarubicin-containing regimens) and three doses of combined ICI therapy at standardized intervals.
Laboratory findings revealed chronic hepatitis B infection with HBsAg >250.00 IU/L (normal <0.05), anti-HBe antibodies (0.14 S/CO, positive >1), and anti-HBc antibodies (7.04 S/CO, positive >1). Liver function abnormalities included elevated aspartate aminotransferase (57 U/L, normal 15-40), alkaline phosphatase (212 U/L, normal 45-125), gamma-glutamyl transferase (343 U/L, normal 10-60), direct bilirubin (5.3 μmol/L, normal 0-3.4), hypoalbuminemia (32.1 g/L, normal 40-55), and total bile acids (17.5 μmol/L, normal 3.4-17.1). High-sensitivity HBV DNA was undetectable. Blood electrolyte examination showed hypokalemia (2.86 mmol/L, normal 3.5-5.3), hypernatremia (148.7 mmol/L, normal 137-147), and hyperchloremia (112.4 mmol/L, normal 99-110). Hyperammonemia (68 μmol/L, normal 9-30) and elevated alpha-fetoprotein (15 ng/mL, normal 0-7) were noted. Erythrocyte sedimentation rate was slightly increased (20 mm/h, normal 0-15). His renal function and serum B-vitamin/micronutrient levels were normal.
CSF analysis demonstrated pleocytosis (47 ×106/L, normal 0-8×106/L) and elevated protein (0.92 g/L, normal 0.12-0.6). Anti-GFAP-ϵ antibodies positivity at a titer of 1:3.2 and anti-GFAP-α antibodies reactivity (titer 1:1.1) in CSF were identified by cell-based assay (Figures 1A–C). CSF anti-GFAP antibodies positivity was further confirmed by tissue-based assay (Figures 1D, E). Serum anti-GFAP antibodies were negative. Both CSF and serum samples were negative for anti-aquaporin 4-IgG, anti-myelin oligodendrocyte glycoprotein-IgG, and paraneoplastic antibodies, including anti-Hu, anti-Yo, anti-Ri, anti-Ma2, anti-CV2, and anti-amphiphysin. No oligoclonal bands were detected in CSF or serum.
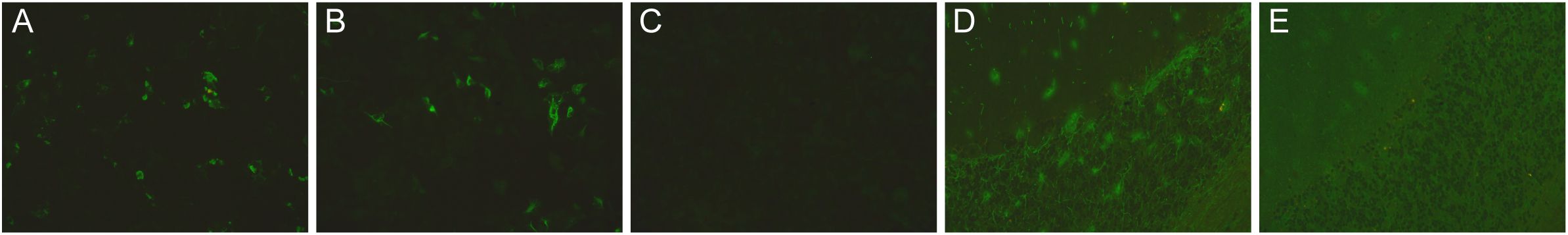
Figure 1. Serum and CSF and anti-GFAP antibodies of the patient detected by cell-based assay and tissue-base assay. Indirect immunofluorescence staining showed positive CSF anti-GFAP-ϵ antibodies at a titer of 1:3.2 (A), anti-GFAP-α antibodies reactivity (B), titer 1:1.1), and negative corresponding isotype controls (C) by cell-based assay. Representative images of CSF anti-GFAP antibodies positivity (D) and corresponding negative isotype controls (E) by tissue-base assay. Original magnification: ×200.
Cervical spine MRI demonstrated T2-weighted hyperintensity at the C4–5 level, notably involving the posterior columns (Figures 2A–D). Gadolinium contrast-enhanced sequences revealed linear enhancement along the dorsal aspect of the lesion and mild leptomeningeal enhancement at the cervicomedullary junction (Figures 2E–H). Brain MRI and thoracolumbar spinal imaging showed no structural abnormalities. Therefore, the diagnosis of autoimmune GFAP astrocytopathy was made.
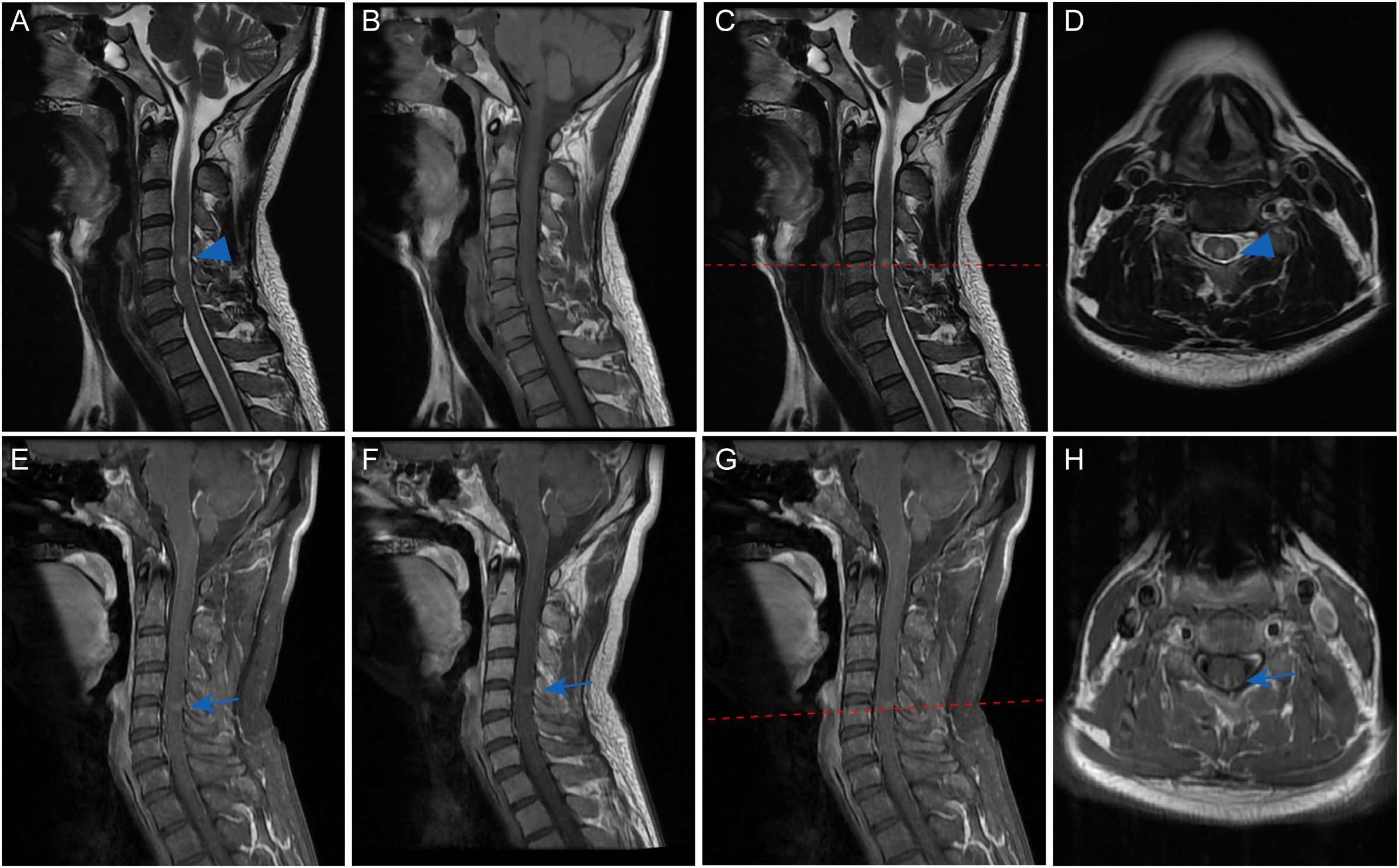
Figure 2. Cervical spinal MRI of the patient. Sagittal T2-weighted imaging demonstrated a discrete intramedullary hyperintensity spanning the C4-C5 levels (blue arrowhead, (A), which mainly located in the posterior part on axial views (blue arrowhead, (C, D). Corresponding T1-weighted sequences revealed iso-intensity without mass effect or cord expansion (B). Contrast-enhanced sequences exhibited subtle linear enhancement along the posterior lesion margin on both sagittal (blue arrow, (E-G) and axial views (blue arrow, H). Mild leptomeningeal enhancement was noted at the cervicomedullary junction (E-G).
Given the patient’s history of chronic hepatitis B virus infection, high-dose intravenous methylprednisolone was deliberately avoided to prevent potential hepatic complications. Then the patient received a 3-day course of intravenous prednisone at 40 mg daily. Significant clinical improvement was observed within 5 days, with near-complete resolution of gait instability. The patient has not undergone any HCC-specific therapeutic interventions to date. Subsequent 9-month follow-up evaluations revealed normal liver function and serum AFP level. The patient sustained neurological remission at the last follow-up assessment.
3 Literature review
Through systematic literature review, we identified 21 documented cases of myelitis developing following PD-1 inhibitor therapy (12, 13, 16–24). Demographic characteristics, clinical features, and therapeutic outcomes treatment were summarized in Table 1. The median onset age of 59 years (range: 16-75) with male predominance (14/21, 66.7%). Malignancies predominantly included non-small cell lung cancer (10/21, 47.6%) and melanoma (7/21, 33.3%). Nivolumab (10/21, 47.6%) and pembrolizumab (12/21, 57.1%) were the most frequently administered PD-1 inhibitors. CSF analysis revealed median pleocytosis of 43.5 cells/μL (range 3-1195) and elevated protein levels (median 0.9 g/L, range 0.32-3.81). Five patients had positive autoantibodies: two serum anti–aquaporin-4 antibodies positive cases (22, 23), one CSF anti-GFAP antibodies positive case (16), and two CSF atypical antibody reactivity cases (16). Spinal MRI demonstrated predominant cervicothoracic involvement, with 95.5% (21/22) exhibiting thoracic segment abnormalities and 59.1% (13/22) showing cervical cord lesions. All patients underwent ICI discontinuation combined with immunosuppressive therapy: high-dose glucocorticoids monotherapy (6/21, 28.6%) or glucocorticoids in combination other immunotherapies (15/21, 71.4%). Most patients were responsive well to glucocorticoids treatment (14/21, 66.7%). Myelitis relapse occurred in 31.8% (7/21) of patients, requiring escalated immunotherapies, including IV immunoglobulin (n=1) (12), plasma exchange (n=1) (12), combination immunoglobulin and plasma exchange (n=1) (16), methylprednisolone with immunoglobulin (n=1) (12), and triple therapy with methylprednisolone, plasma exchange, and cyclophosphamide (n=1) (16). Clinical improvement was achieved in 63.6% (14/22) of patients. The overall mortality rate was 18.2% (4/22) with fatal outcomes attributed to sepsis (n=1) (16), respiratory failure (n=1) (13), or malignancy progression (n=1) (19).
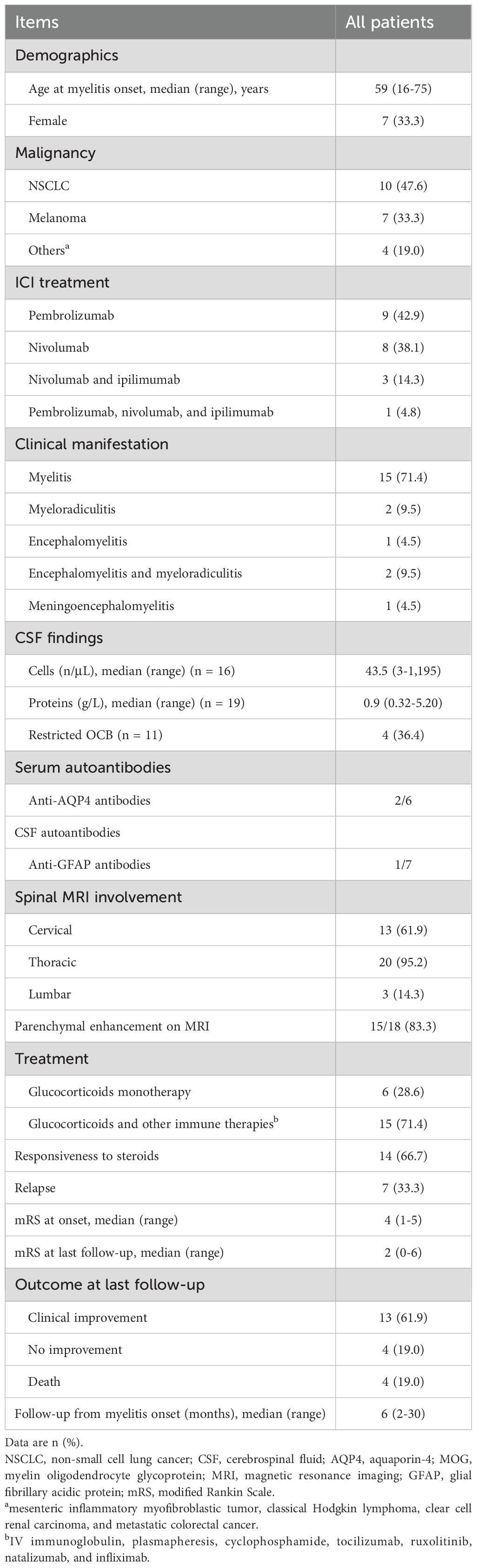
Table 1. Findings of reported cases with myelitis-related adverse events secondary to PD-1 inhibitor therapy.
4 Discussion
To the best of our knowledge, this is the first documented case of autoimmune GFAP astrocytopathy developing as an irAE following sintilimab therapy in an HCC patient. This unique presentation expands the clinical spectrum of ICI-associated neurological complications.
Clinical manifestations of autoimmune GFAP astrocytopathy typically involve meningoencephalitis or meningoencephalomyelitis, with isolated myelitis being a rare presentation (15, 25, 26). Distinct from the longitudinally extensive transverse myelitis commonly described in ICI-associated myelitis (12, 13, 16, 19, 24, 27), our patient exhibited short-segment posterior column involvement with a characteristic linear perivascular enhancement pattern. Notably, while radiation-related myelitis has been reported in some ICI-treated patients (12, 16, 18, 19), our case lacked prior radiotherapy exposure.
Diagnostically, our case fulfilled current criteria through CSF GFAP-IgG positivity confirmed via cell-based assay and tissue-based assay, a finding associated with higher diagnostic specificity compared to serum testing (15). The pleocytosis (47 ×106/L) and elevated protein (0.92 g/L) in CSF of our patient were in accordance with characteristic inflammatory CSF changes in autoimmune GFAP astrocytopathy (16, 25).
A previously reported nivolumab-treated NSCLC patient with GFAP meningoencephalitis demonstrated limited response to high-dose corticosteroids and plasma exchange, ultimately requiring natalizumab rescue therapy (16). This contrasts with our patient’s favorable steroid responsiveness, aligning with the typical corticosteroid-sensitive nature of GFAP astrocytopathy (15). Such therapeutic responses heterogeneity underscores the need for personalized treatment algorithms in ICI-associated autoimmune GFAP astrocytopathy.
Our systematic review reveals universal first-line high-dose glucocorticoids administration across reported cases, consistent with current guideline recommendations (28). However, most patients required additional immunomodulation due to incomplete functional recovery. Seven patients suffered from early relapses within several weeks due to rapid steroids tapering or glucocorticoids monotherapy inadequacy (12, 13, 16, 29). This clinical pattern suggests that patients with extensive myelitis lesions may benefit from aggressive initial immunotherapy regimens. Current evidence supports duration of the first steroid treatment should be continued for at least 3 months to substantiate recovery and prevent early relapses (6). Emerging biological agents including natalizumab, tocilizumab, and ruxolitinib show promise in refractory cases (16, 30), potentially offering targeted immunomodulation without compromising ICI anti-tumor activity.
Autoimmune GFAP astrocytopathy-associated myelitis represents a rare but potentially devastating neurological irAE with generally favorable steroid responsiveness. However, the suboptimal functional outcomes observed in longitudinally extensive myelitis cases necessitate early implementation of intensive, prolonged immunotherapy protocols. Future studies should focus on optimizing risk-stratified treatment algorithms and investigating targeted biological therapies to improve neurological outcomes while preserving anti-tumor immunity.
Data availability statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Ethics statement
The studies involving humans were approved by The Ethics Committee of the First Affiliated Hospital of Xi’an Jiaotong University. The studies were conducted in accordance with the local legislation and institutional requirements. Written informed consent for participation in this study was provided by the participants’ legal guardians/next of kin. Written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article.
Author contributions
XM: Data curation, Funding acquisition, Project administration, Writing – original draft. XQ: Formal Analysis, Supervision, Validation, Writing – review & editing.
Funding
The author(s) declare financial support was received for the research and/or publication of this article. This work was supported by the National Natural Science Foundation of China (grant number 82301531) and the Natural Science Foundation of Shaanxi Province (grant number 2025JC-YBQN-1141).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Gao S, Li N, Gao S, Xue Q, Ying J, Wang S, et al. Neoadjuvant PD-1 inhibitor (Sintilimab) in NSCLC. J Thorac Oncol. (2020) 15:816–26. doi: 10.1016/j.jtho.2020.01.017
2. Fong KY, Zhao JJ, Sultana R, Lee JJX, Lee SY, Chan SL, et al. First-line systemic therapies for advanced hepatocellular carcinoma: A systematic review and patient-level network meta-analysis. Liver Cancer. (2023) 12:7–18. doi: 10.1159/000526639
3. Wang K, Xiang YJ, Yu HM, Cheng YQ, Liu ZH, Qin YY, et al. Adjuvant sintilimab in resected high-risk hepatocellular carcinoma: a randomized, controlled, phase 2 trial. Nat Med. (2024) 30:708–15. doi: 10.1038/s41591-023-02786-7
4. Cappuyns S, Corbett V, Yarchoan M, Finn RS, and Llovet JM. Critical appraisal of guideline recommendations on systemic therapies for advanced hepatocellular carcinoma: A review. JAMA Oncol. (2024) 10:395–404. doi: 10.1001/jamaoncol.2023.2677
5. Ren Z, Xu J, Bai Y, Xu A, Cang S, Du C, et al. Sintilimab plus a bevacizumab biosimilar (IBI305) versus sorafenib in unresectable hepatocellular carcinoma (ORIENT-32): a randomized, open-label, phase 2–3 study. Lancet Oncol. (2021) 22:977–90. doi: 10.1016/S1470-2045(21)00252-7
6. Bruna J, Argyriou AA, Anastopoulou GG, Alemany M, Nadal E, Kalofonou F, et al. Incidence and characteristics of neurotoxicity in immune checkpoint inhibitors with focus on neuromuscular events: Experience beyond the clinical trials. J Peripher Nerv Syst. (2020) 25:171–7. doi: 10.1111/jns.12371
7. Ramos-Casals M and Sisó-Almirall A. Immune-related adverse events of immune checkpoint inhibitors. Ann Intern Med. (2024) 177:p. doi: 10.7326/AITC202402200
8. Khan S and Gerber DE. Autoimmunity, checkpoint inhibitor therapy and immune-related adverse events: A review. Semin Cancer Biol. (2020) 64:93–101. doi: 10.1016/j.semcancer.2019.06.012
9. Marini A, Bernardini A, Gigli GL, Valente M, Muñiz-Castrillo S, Honnorat J, et al. Neurologic adverse events of immune checkpoint inhibitors: A systematic review. Neurology. (2021) 96:754–66. doi: 10.1212/WNL.0000000000011795
10. Algaeed M, Mukharesh L, Heinzelmann M, and Kaminski HJ. Pearls & Oysters: Pembrolizumab-induced myasthenia gravis. Neurology. (2018) 91:e1365–7. doi: 10.1212/WNL.0000000000006278
11. Sepúlveda M, Martinez-Hernandez E, Gaba L, Victoria I, Sola-Valls N, Falgàs N, et al. Motor polyradiculopathy during pembrolizumab treatment of metastatic melanoma. Muscle Nerve. (2017) 56:E162–e167. doi: 10.1002/mus.25672
12. Chatterton S, Xi S, Jia JX, Krause M, Long GV, Atkinson V, et al. Case series: Immune checkpoint inhibitor-induced transverse myelitis. Front Neurol. (2023) 14:1130313. doi: 10.3389/fneur.2023.1130313
13. Charabi S, Engell-Noerregaard L, Nilsson AC, and Stenör C. Case report: longitudinal extensive transverse myelitis with novel autoantibodies following two rounds of pembrolizumab. Front Neurol. (2021) 12:655283. doi: 10.3389/fneur.2021.655283
14. Tanaka R, Maruyama H, Tomidokoro Y, Yanagiha K, Hirabayashi T, Ishii A, et al. Nivolumab-induced chronic inflammatory demyelinating polyradiculoneuropathy mimicking rapid-onset Guillain-Barré syndrome: a case report. Jpn J Clin Oncol. (2016) 46:875–8. doi: 10.1093/jjco/hyw090
15. Kunchok A, Zekeridou A, and McKeon A. Autoimmune glial fibrillary acidic protein astrocytopathy. Curr Opin Neurol. (2019) 32:452–8. doi: 10.1097/WCO.0000000000000676
16. Picca A, Berzero G, Bihan K, Jachiet V, Januel E, Coustans M, et al. Longitudinally extensive myelitis associated with immune checkpoint inhibitors. Neurol Neuroimmunol Neuroinflamm. (2021) 8(3):e967. doi: 10.1212/NXI.0000000000000967
17. Vickers ML, Seidl B, Bigby K, Chern B, Eriksson L, Hartnett G, et al. Inflammatory myeloradiculitis secondary to pembrolizumab: A case report and literature review. Case Rep Oncol Med 2020. (2020) p:8819296. doi: 10.1155/2020/8819296
18. Carausu M, Beddok A, Langer A, Girard N, Bidard FC, Massiani MA, et al. Radiation myelitis after pembrolizumab administration, with favorable clinical evolution and safe rechallenge: a case report and review of the literature. J Immunother Cancer. (2019) 7:317. doi: 10.1186/s40425-019-0803-x
19. Chang VA, Simpson DR, Daniels GA, and Piccioni DE. Infliximab for treatment-refractory transverse myelitis following immune therapy and radiation. J Immunother Cancer. (2018) 6:153. doi: 10.1186/s40425-018-0471-2
20. Chen R, Zinzani PL, Lee HJ, Armand P, Johnson NA, Brice P, et al. Pembrolizumab in relapsed or refractory Hodgkin lymphoma: 2-year follow-up of KEYNOTE-087. Blood. (2019) 134:1144–53. doi: 10.1182/blood.2019000324
21. Kubo K, Wadasaki K, Yamane H, and Doi M. Radiation myelitis after durvalumab administration following chemoradiotherapy for locally advanced non-small cell lung cancer: an illustrative case report and review of the literature. Int Cancer Conf J. (2019) 8:118–21. doi: 10.1007/s13691-019-00367-5
22. Narumi Y, Yoshida R, Minami Y, Yamamoto Y, Takeguchi S, Kano K, et al. Neuromyelitis optica spectrum disorder secondary to treatment with anti-PD-1 antibody nivolumab: the first report. BMC Cancer. (2018) 18:95. doi: 10.1186/s12885-018-3997-2
23. Shimada T, Hoshino Y, Tsunemi T, Hattori A, Nakagawa E, Yokoyama K, et al. Neuromyelitis optica spectrum disorder after treatment with pembrolizumab. Mult Scler Relat Disord. (2020) 37:101447. doi: 10.1016/j.msard.2019.101447
24. Wilson R, Menassa DA, Davies AJ, Michael S, Hester J, Kuker W, et al. Seronegative antibody-mediated neurology after immune checkpoint inhibitors. Ann Clin Transl Neurol. (2018) 5:640–5. doi: 10.1002/acn3.547
25. Hagbohm C, Ouellette R, Flanagan EP, Jonsson DI, Piehl F, Banwell B, et al. Clinical and neuroimaging phenotypes of autoimmune glial fibrillary acidic protein astrocytopathy: A systematic review and meta-analysis. Eur J Neurol. (2024) 31:e16284. doi: 10.1111/ene.16284
26. Shan F, Long Y, and Qiu W. Autoimmune glial fibrillary acidic protein astrocytopathy: A review of the literature. Front Immunol. (2018) 9:2802. doi: 10.3389/fimmu.2018.02802
27. Valentina Poretto MB, Filipponi S, Marangoni S, Piffer S, Chioffi F, and Giometto B. Hunting the real culprit: a complex case of nivolumab-related myelitis. (P4.2-006). Neurology. (2019). doi: 10.1212/WNL.92.15_supplement.P4.2-006
28. Thompson JA, Schneider BJ, Brahmer J, Achufusi A, Armand P, Berkenstock MK, et al. Management of immunotherapy-related toxicities, version 1.2022, NCCN clinical practice guidelines in oncology. J Natl Compr Canc Netw. (2022) 20:387–405. doi: 10.6004/jnccn.2022.0020
29. Dixit NBAK. A case report of recurrent longitudinally extensive holocord transverse myelitis after combination immune checkpoint inhibitor therapy with nivolumab and ipilimumab (815). Neurology. (2020). doi: 10.1212/WNL.94.15_supplement.8
Keywords: autoimmune glial fibrillary acidic protein astrocytopathy, myelitis, immune checkpoint inhibitors, sintilimab, glucocorticoids
Citation: Ma X and Qin X (2025) Autoimmune GFAP astrocytopathy manifesting as sintilimab-induced myelitis: a Case Report and literature review. Front. Immunol. 16:1595653. doi: 10.3389/fimmu.2025.1595653
Received: 14 May 2025; Accepted: 14 August 2025;
Published: 27 August 2025.
Edited by:
Maria Eleftheria Evangelopoulos, National and Kapodistrian University of Athens, GreeceReviewed by:
Anne Haney Cross, Washington University in St. Louis, United StatesShugang Cao, Second Affiliated Hospital of Anhui Medical University, China
Copyright © 2025 Ma and Qin. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Xing Qin, dGhvcm5iaXJkMjAwOEAxNjMuY29t
 Xue Ma
Xue Ma Xing Qin*
Xing Qin*