- 1Department of Agricultural Microbiology, Faculty of Agriculture, Zagazig University, Zagazig, Egypt
- 2Department of Biochemistry, Faculty of Agriculture, Zagazig University, Zagazig, Egypt
- 3Nutrition and Food Sciences Department, National Research Center, Dokki, Giza, Egypt
- 4Molecular Cell Biology Unit, Division of Biochemistry, Department of Chemistry, Faculty of Science, Tanta University, Tanta, Egypt
- 5Natural and Medical Sciences Research Center, University of Nizwa, Nizwa, Oman
- 6Department of Home Economics, Specific Education Faculty, Port Said University, Port Said, Egypt
- 7Department of Poultry Diseases, Faculty of Veterinary Medicine, Cairo University, Giza, Egypt
- 8Plant Production Department (Horticulture-Pomology), Faculty of Agriculture, Saba Basha, Alexandria University, Alexandria, Egypt
- 9Biology Department, Faculty of Science, King Khalid University, Abha, Saudi Arabia
- 10Department of Biology, College of Science, United Arab Emirates University, Al Ain, United Arab Emirates
Curcumin (1,7-bis-(4-hydroxy-3-methoxyphenyl)-hepta-1,6-diene-3,5-dione) is a naturally occurring polyphenol molecule. It is lipophilic and has demonstrated in vitro and in vivo therapeutic effects through multiple pathways. Extensive studies on its pharmacological properties have shown its anti-inflammatory, antioxidant, antinociceptive, antimicrobial, antiparasitic, antimalarial, and wound-healing properties. However, its limited bioavailability in humans due to poor intestinal absorption, rapid metabolism, and rapid systemic elimination remains a significant challenge. Various curcumin formulations have been developed to address this limitation. This article reviews current studies on the biological and pharmacological properties of curcumin. It also examines methods for curcumin isolation, including pressurized fluid extraction, Soxhlet extraction, enzyme-assisted extraction, and microwave extraction. Furthermore, analytical methods for the identification and quantification of curcumin in diverse matrices, as well as procedures for formulating curcumin, will also be addressed. This review consolidates recent studies on curcumin’s chemical, bioactive, and pharmacological properties. It also highlights significant knowledge gaps, indicating the need for future research to elucidate curcumin’s mechanism of action, safety, efficacy, and therapeutic potential for treating various human and animal diseases.
1 Introduction
Traditional medicine practitioners are sources of primary healthcare in many low-income countries. According to the World Health Organization, traditional medicine constitutes the primary healthcare system for over 80% of the world’s population (1). People in industrialized nations are increasingly turning to natural remedies, especially herbal ones, as they are considered safer alternatives to traditional drugs (2). However, the process of discovering new medications from natural sources is a complex and costly endeavor. It involves numerous steps, including gathering plant material, extracting active compounds, isolating and purifying these compounds, and finally, characterizing their properties. The final step of this process is the evaluation of its pharmacological and toxicological properties. Despite these challenges, natural products remain a rich source of compounds with unique chemical structures and mechanisms of action, making them potential candidates for treating various human disorders (3).
Turmeric (Curcuma longa L.) has been extensively researched, and its use is well-documented in the history of Asian traditional medicine. This includes practices from Austronesian peoples with animistic traditions, Siddha, Traditional Chinese Medicine, Unani Medicine, and Ayurveda (4). The safety of turmeric is evidenced by its staple dietary use across various cultures for centuries. Furthermore, it has also been used for managing several disorders, including diabetes, Alzheimer’s disease, cancer, and rheumatic disorders (5). Turmeric supplementation is linked to multiple health benefits, which include its anti-inflammatory and antioxidant properties (6).
Turmeric is a widely used spice available globally, especially in the Indian subcontinent (7, 8). Its rhizomes can be consumed fresh, cooked, dried, and ground into rich orange-yellow powder. The rhizome’s intense yellow color makes it a natural food coloring additive (9). It is also a seasoning agent in Asian cuisine, particularly in curries, and serves as a dye (8, 10, 11). Turmeric powder has an earthy, mustard-like aroma with a hint of black pepper. The highest diversity of Curcuma species is found in Thailand and India. However, many wild species exist in other tropical Asian countries (12, 13).
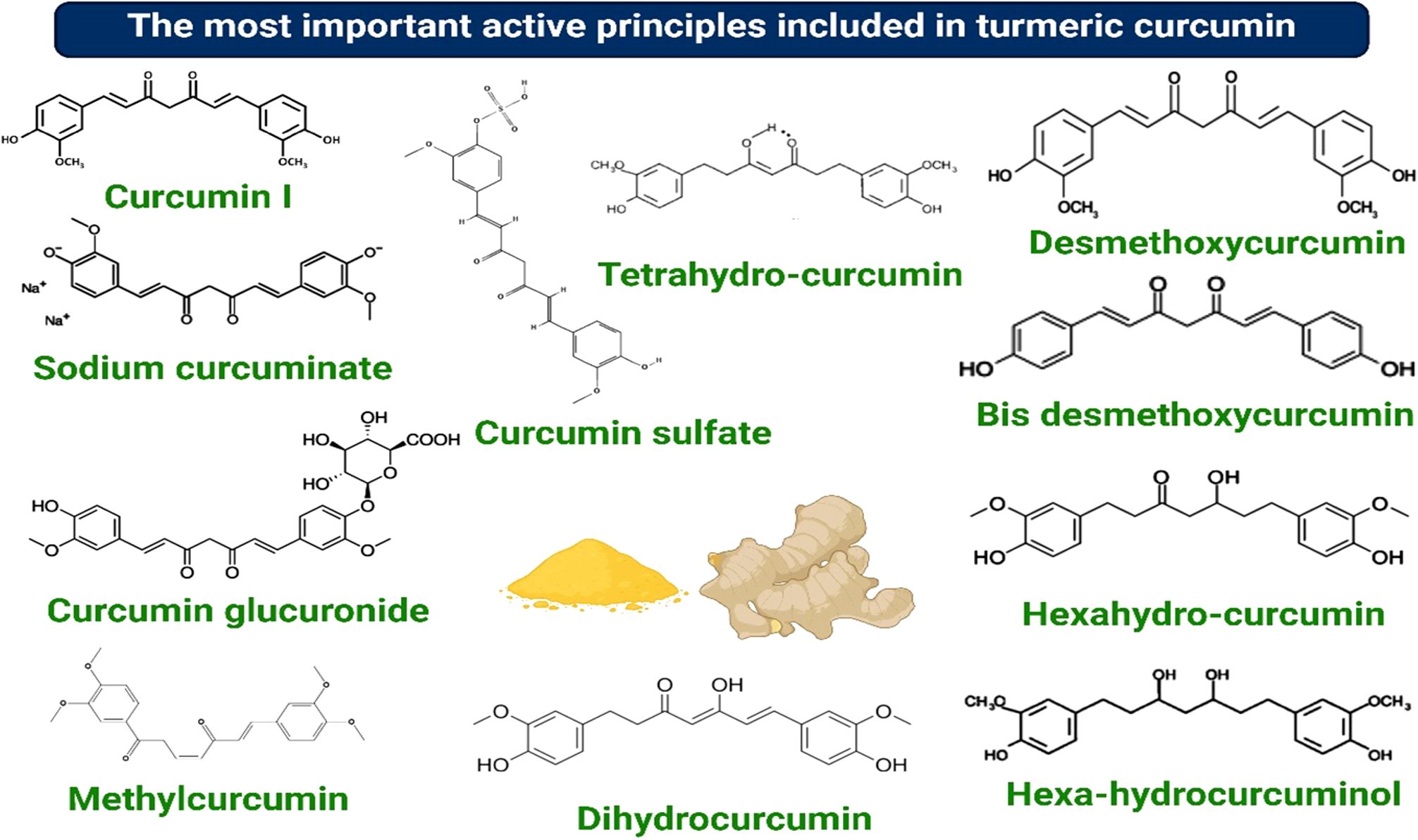
Recent studies have highlighted challenges in classifying Curcuma species, with specimens from South India primarily identified as C. longa. However, the phylogeny, the intra- and interspecific variation, and the relationships among other Curcuma species and cultivars across different regions remain unclear (14). Several species marketed globally as turmeric have been shown to belong to distinct but morphologically similar groups, often sharing comparable local names. C. longa grows similarly to ginger and produces rhizomes that contain curcuminoids, including curcumin, desmethoxycurcumin, and bisdemethoxycurcumin (Figure 1).
Curcumin is a low-molecular-weight, lipophilic polyphenol that can easily cross cell membranes (9, 15). Curcumin interacts with various cellular signaling pathways, enabling it to modulate some chronic illnesses by binding to key molecules (16), such as transcription factors, inflammatory mediators, and enzymes like protein kinase, reductase, and histone acetyltransferase. It effectively regulates epigenetic modifications associated with neurological diseases, inflammation, diabetes, and various cancers (17). Additionally, curcumin selectively inhibits phosphorylase kinase, reducing glycogen metabolism and altering proteasomal pathways (18).
Curcumin has been widely researched for its diverse health benefits, including anti-inflammatory, antidiabetic, neuroprotective, and disease-fighting properties (19). Despite these promising effects, curcumin’s therapeutic potential is significantly limited by its low bioavailability and poor water solubility (20, 21). Its rapid metabolism, limited absorption, and swift systemic elimination further reduce its concentration in blood plasma and tissues, restricting its clinical effectiveness (22). To address these challenges, it is essential to improve curcumin’s physicochemical properties, particularly its solubility and bioavailability.
Employing advanced formulation techniques is crucial for ensuring the safe and effective therapeutic use of curcumin (22, 23). Recent innovations such as phospholipid complexes, nanoparticles, micelles, hydrogels, and liposomes have demonstrated enhanced efficacy and safety profiles, offering new hope for maximizing the clinical benefits of curcumin (21, 23).
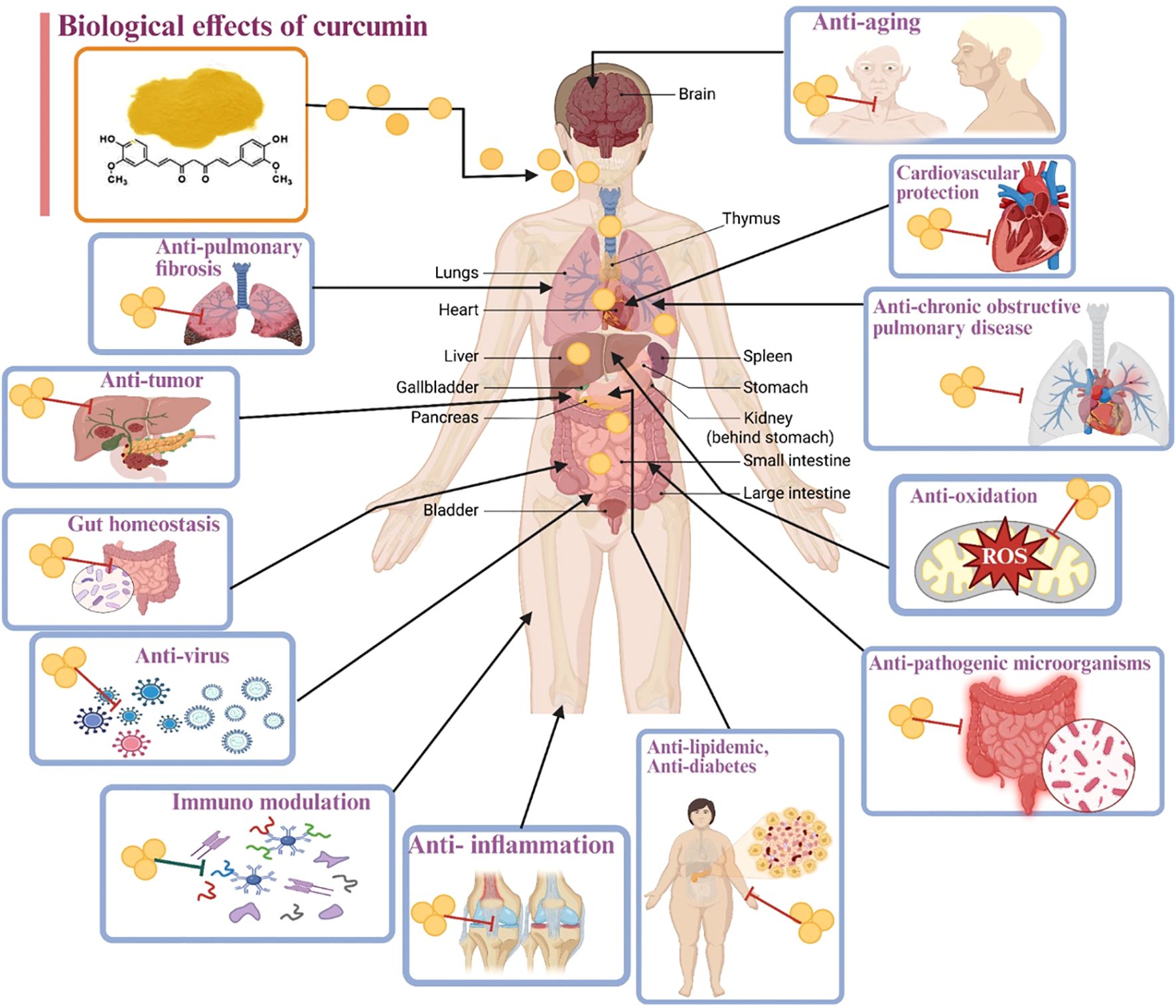
This review distinguishes itself from existing literature by offering a unique and comprehensive synthesis of curcumin research. While prior reviews have covered curcumin’s chemical composition and biological activities, this work goes further by integrating the latest advancements in several key areas. Specifically, it provides an updated understanding of curcumin’s precise immunomodulatory mechanisms, detailing how it influences immune cell function and cytokine pathways, which is crucial for its therapeutic applications in inflammatory and immune-mediated diseases. Furthermore, this review incorporates novel applications, such as its role in managing diseases like COVID-19, including its antiviral properties and ability to enhance vaccine responses, an area of critical recent interest that has not been extensively covered in earlier reviews. Figure 2 displays the different biological effects of curcumin on human health.
Crucially, this review offers a detailed examination of current and emerging strategies to enhance curcumin’s notoriously low bioavailability. It moves beyond traditional methods to discuss novel approaches such as Self-microemulsifying drug delivery systems (SMEDDS), prodrugs, co-crystallization, and amorphous solid dispersions, providing practical insights for researchers and clinicians working to overcome this significant hurdle. By highlighting both the complex synthesis challenges and the latest solutions for enhancing its pharmaceutical properties, this review offers a more comprehensive and forward-looking perspective on curcumin’s evolution from traditional medicine to modern therapeutics.
This comprehensive update on bioavailability strategies, coupled with insights into novel immunological and antiviral roles, offers significant added value compared to existing reviews, addressing the pressing need for effective and safe translation of curcumin into clinical practice.
2 Global distribution of C. longa
C. longa is a rhizome of the family Zingiberaceae primarily cultivated in southwestern and southern Asia (24). It is widely used in traditional Asian cuisine and is colloquially called the “golden spice” because of its intense golden/yellow color and distinct flavor (24). Apart from its use as a condiment, it impacts on the color and flavor of food. Turmeric powder, derived from its rhizome, is a popular spice and the main ingredient in curry. The powder is also utilized globally as a food coloring and, more recently, as a dietary supplement (24).
Curcumin, a yellow-orange pigment extracted from turmeric, is a natural dye recognized as Natural Yellow 3 (E100) and considered eco-friendly (25, 26). Turmeric is cultivated extensively in many countries, particularly in South Asia, Southeast Asia, and the Middle East. India is the largest producer and consumer of turmeric. Other significant producers include Bangladesh, Pakistan, Sri Lanka, and Indonesia, with countries like China, Japan, Korea, and Australia also growing turmeric (27). It has gained significant popularity in Western countries due to its reported health benefits, resulting in a rise in global demand (27).
3 Botanical description of C. longa
Turmeric is a herbaceous perennial plant that grows up to 1 m tall. Its rhizomes are cylindrical and branched, with a pleasant aroma and range in color from bright yellow to orange (12). The plant has two alternating rows of leaves, each comprising three parts: the petiole, blade, and sheath. The leaf sheaths form a pseudostem, while the petioles measure from 50 to 115 cm in length (12, 14). Leaf blades typically range between 76 and 115 cm long, with some reaching up to 230 cm, and are 38–45 cm wide (12, 14, 28, 29). Their shape varies from oblong to elliptical, tapering towards the tip (28).
The stem bracts exhibit colors ranging from white to green, often tinged with reddish-purple hues near the inflorescence apex. The bracts taper towards their apices (29). Turmeric flowers are hermaphroditic, trimerous, and zygomorphic. The three united sepals are white, pubescent, and irregularly toothed (28, 29). The flowers have three triangular petals measuring between 1 and 1.5 cm, with soft, spiny protrusions at their tips (12, 14, 28, 29). The corolla tube, formed by the fusion of three bright yellow petals, measures approximately 3 cm in length (29).
4 Morphology of C. longa
The anther base is equipped with spurs, and staminodes are formed from the residual stamens, with the inner and outer staminodes differing in length (29). The yellow, oval-shaped labellum is between 1.2 and 2.0 cm long and has a yellow ribbon-like marking at its center (14, 29). The bracts are pale green, 3–5 cm long, elliptical to oblong, with a blunt apex (14, 29).
The fruit capsule has three visible sections when opened. In East Asia, the flowering season usually begins in August (29). During this season, an inflorescence stalk, 12–20 cm long and covered with multiple blossoms, develops at the end of the pseudostem (14, 29).
5 Chemical composition of C. longa
C. longa contains various bioactive components, including polysaccharides, essential oils, and curcuminoids (30). It is known for its potential biological and therapeutic effects (31), largely attributed to the quality and concentration of these bioactive components (32). Curcuminoids are polyphenolic compounds responsible for the yellow color of the rhizomes and have many biological roles. The three primary curcuminoids in C. longa are curcumin, bisdemethoxycurcumin, and demethoxycurcumin (33). Curcumin has been extensively studied for its anti-inflammatory, antioxidant, and anticancer properties (34).
The essential oils in C. longa are responsible for its characteristic flavor and aroma (35). The primary components of these oils are turmerone, ar-turmerone, and curlone. Polysaccharides are the third major group of compounds in C. longa (36). The three main polysaccharides found in C. longa are curdlan, glycogen, and turmeric. These polysaccharides have demonstrated antiviral, anticancer, and immunomodulatory properties (37).
Dehydration is the primary method recommended for preserving turmeric quality throughout storage and usage, including freeze-drying, low-temperature drying, and microwave-vacuum drying techniques (38, 39). The hot air-drying method is a widely used and viable option due to its simplicity; however, prolonged exposure to high temperatures may degrade turmeric’s color, flavor, and bioactive compounds (40). While freeze-drying is considered the best preservation method for quality, it is expensive, time-consuming, and energy-intensive (38, 41). In comparison, sun-drying, which is regarded as a conventional technique, requires extended durations and often diminishes the product quality and bioactive components (42). Consequently, more efficient drying techniques are needed to maintain the desired quality and safeguard turmeric’s medicinal properties (42).
C. longa rhizomes comprise carbohydrates (69.4%), protein (6.3%), fats (5.1%), minerals (3.5%), and moisture (13.1%) (30). Essential oils extracted via steam distillation can reach approximately 5.8% of the rhizome content and contain compounds like borneol (0.5%), α-phellandrene (1%), zingiberene (25%), sabinene (0.6%), cineole (1%), and sesquiterpenes (53%) (30).
The primary bioactive compounds in C. longa are a blend of three curcuminoids: diferuloylmethane (94%, C21H20O6, curcumin I), demethoxycurcumin (6%, C20H18O5, curcumin II), and bis-demethoxycurcumin (0.3%, C19H16O4, curcumin III) (32). Curcumin I is considered a principal curcuminoid. The phenolic OH and CH2 groups in the β-diketone moiety of these compounds confer anti-inflammatory and antioxidant properties, among other bioactivities, making curcumin a valuable nutraceutical for chemo-preventive and therapeutic applications (23, 43).
Furthermore, curcumin is soluble in organic solvents such as methanol, ethanol, acetone, and dimethyl sulfoxide (DMSO) but is insoluble in water. Hence, solvent-based extraction is critical for optimal curcumin yield and environmental sustainability (44). Common solvents like methanol, ethanol, and acetonitrile are typically used to obtain antioxidative extracts from C. longa. Deep eutectic solvents have emerged as eco-friendly alternatives due to their non-toxic, biocompatible properties, minimal environmental impact, and enhanced efficiency in extracting bioactive compounds. These solvents, also known as designer solvents, can be synthesized for specific applications, including the extraction of bioactive chemicals (45).
Deep eutectic solvents are synthesized using various hydrogen bond donors and acceptors, such as choline chloride, menthol, and betaine (46). These compounds can be combined with sugars, carboxylic acids, alcohols, amines, or other hydrogen bond-containing compounds to form deep eutectic solvents. Key parameters influencing the synthesis of these solvents include solute-to-solvent ratio, extraction temperature, and duration (47).
Appropriate dosing significantly influences the biological efficacy of curcumin and turmeric extracts (48). Dietary administration of curcumin/turmeric extracts in suitable doses has been shown to inhibit tumor formation in multiple organs of mice and rats. Although high doses of curcumin and turmeric extracts have not increased mortality in mice, adverse effects have been observed in other species (49). For example, turmeric oleoresin administration in pigs has been reported to reduce feed conversion efficiency (or weight gain), increase liver and thyroid weights, and result in histological alterations in the kidney, liver, and urinary bladder. Similarly, high-dose turmeric extract administration in mice significantly changed tissue weights, weight gain, and red and white blood cell levels (49).
Curcumin and turmeric extracts are generally considered nontoxic and highly promising for various biological applications when administered at appropriate doses (50). Beyond the aforementioned biological activities, turmeric has been used in traditional medicine and contemporary and alternative medicine to manage conditions like anemia, indigestion, diabetes, hemorrhoids, edema, hepatitis, atherosclerosis, hysteria, wound healing, urinary diseases, psoriasis, rheumatism, anorexia, dermal diseases, inflammation, hepatic disorders, cough, and sinusitis (51, 52).
6 Techniques for curcumin isolation from turmeric rhizomes
Turmeric rhizomes contain two main pharmacologically active secondary metabolites: curcuminoids and essential oils (53). Demethoxycurcumin, bis-demethoxycurcumin, and curcumin are the primary compounds responsible for the biological activity of the rhizomes (54). Curcuminoids are extracted from turmeric rhizomes using traditional and modern techniques (43).
Traditional techniques include maceration and Soxhlet extraction (55, 56). Contemporary techniques include microwave extraction (57), enzyme-assisted extraction (58), pressurized fluid extraction (59), supercritical fluid extraction (60), and ultrasound extraction (61). Ethanol, dichloromethane, ethyl acetate, isopropanol, methanol, n-butanol, and acetone are the most commonly used solvents for curcuminoid extraction (54, 62, 63). Sahne et al. (55) used acetone for conventional and unconventional extraction because of its strong solubilization capability. Additionally, Muthukumar et al. (63) examined several organic solvents for curcumin extraction and identified acetone as the best solvent.
Curcumin can be extracted using thin-layer chromatography (TLC) on the extraction mixture, a traditional analytical method (54, 62). High-performance liquid chromatography (HPLC) is used to measure the curcumin content in the extract. After extraction, organic solvents are separated from the extract using a vacuum evaporator. The leftover material, or oleoresin, is then dissolved in methanol and analyzed using HPLC, as described by Yadav et al. (64).
The production and stability of curcumin are greatly influenced by the extraction method used. Several cutting-edge techniques for curcumin extraction from turmeric rhizomes have been investigated (55). Their results were compared with Soxhlet extraction, the most commonly used reference technique. The Soxhlet method was observed to achieve a significantly higher curcumin yield (6.9%) compared to enzyme-assisted (4.1%), ultrasound-assisted (3.92%), and microwave-assisted (3.72%) methods (55). The Soxhlet technique offers benefits such as low temperatures, faster extraction times, lower solvent quantities, and higher yields, which are unmatched by the newer extraction techniques (55). Although Soxhlet achieves higher yields (6.9%) than some modern methods like microwave-assisted extraction (3.72%) (55), its disadvantages outweigh this benefit. Advanced techniques offer a 90% reduction in processing time, 50–70% lower solvent consumption, and improved retention of bioactive properties (65).
Soxhlet extraction typically requires 4–6 h per cycle (66), with some processes extending beyond 24 h (67). This prolonged duration makes it impractical for industrial-scale applications where efficiency is critical. The method necessitates large volumes of organic solvents (e.g., ethanol, methanol) (65), raising both economic and environmental concerns due to solvent disposal requirements and potential ecological impacts. Also, continuous heating throughout the extraction cycle results in substantial energy consumption (67), making it cost-prohibitive compared to modern techniques. The prolonged exposure to elevated temperatures may degrade heat-sensitive curcuminoids (67), potentially compromising the bioactive integrity of the extract. On the other hand, the batch-processing nature of Soxhlet extraction hinders continuous production workflows, restricting its viability for commercial manufacturing (67).
Naksuriya et al. (68) investigated the kinetic degradation of curcumin from a naturally occurring curcuminoid mixture under different conditions with varying solvent dielectric constant, pH, and temperature. They also assessed the degradation of pure curcumin under similar settings, using a standard medium composed of a 50:50 (v/v) mixture of an aqueous buffer and methanol. The degradation kinetics of curcumin in the curcuminoid mixture showed a first-order response. The degradation rate increased simultaneously with the medium’s pH, temperature, and dielectric constant (65).
Curcumin underwent rapid degradation by autoxidation in an aqueous buffer (pH: 8) at a steady rate of 0.28−1 h, resulting in a half-life (t1/2) of 2.5 h (65). Mixing curcumin with ω-methoxy poly (ethylene glycol)-b-(N-(2-benzoyloxypropyl) methacrylamide) polymer micelles improved its stability, increasing it approximately 300–500 times compared to pure curcumin in a phosphate buffer and methanol mixture (65). Thus, this stabilization approach offers the potential for developing formulations suitable for further pharmacological and clinical studies (68).
Another study by Liu et al. (69) investigated the use of naturally occurring organic acids and sugars to produce deep eutectic solvents for curcuminoid extraction. Under ideal conditions (temperature: 50°C, solid-to-liquid ratio: 0.1/10 g/mL, and extraction time: 30 min), a solvent containing a 1:1 ratio of citric acid and glucose with 15% water yielded higher extraction efficiency compared to conventional solvents. This process is a promising substitute for extracting natural coloring agents because it is eco-friendly and sustainable (69).
When purifying and separating curcuminoids from the oleoresin, volatile turmeric oil (CP-01) dissolves curcumin, causing issues with recrystallization. To address this, mixtures of several organic solvents were evaluated for the selective recrystallization of curcuminoids (54). A combination of isopropyl alcohol and hexane in a volumetric ratio of 1:1.5 was identified as the optimal solvent for recrystallizing curcuminoids, yielding a recrystallized powder with a purity of up to 99.45% w/w (54). In comparison, the raw curcuminoid powder had a curcumin content of 76.82% w/w (54).
In a study by Ahmed et al. (70), the authors focused on quantifying curcumin from C. longa roots and commercial powder using a green chromatography approach. This method, emphasizing environmental sustainability by minimizing harmful solvents, aligns with renewed interest in green analytical techniques. The proposed method underwent validation according to ICH guidelines, assessing system suitability, linearity, precision, and accuracy. Its simplicity is further enhanced by short retention times, the use of an eco-friendly mobile phase (ethanol: water), and a UV-Vis detector. Overall, this method is more environmentally friendly than previously reported techniques, making it suitable for routine, eco-conscious analysis of curcumin.
7 Physicochemical properties of curcumin
According to Nelson et al. (71), turmeric contains up to ~5% curcumin (1,7-bis-(4-hydroxy-3-methoxyphenyl)-1,6-heptadiene-3,5-dione) (72), also known as diferuloylmethane (73). Curcumin is lipophilic with a strong affinity for fats and oils. It is water-insoluble and also insoluble in acidic or neutral solutions (70). However, it is soluble in organic solvents, like ethanol, dimethylsulfoxide, and acetone, which can be used to extract it from turmeric rhizomes. Its molecular weight is 368.38 g/mol, and its chemical formula is C21H20O6 (70). Structurally, it comprises three primary functional groups: (i) two aromatic ring systems with an alpha, beta-unsaturated beta-diketone moiety, (ii) one o-methoxy phenolic group, and (iii) a seven-carbon linker (74).
Curcumin has a melting point of 183°C and exhibits diketone/keto-enol tautomerism due to its β-diketone moiety (75, 76). The balance between diketone and keto-enol forms is strongly influenced by temperature, pH, and solvent polarity (77), and the enol-to-keto ratio is a significant determinant of curcumin’s pharmacological properties (78). Under acidic and neutral pH conditions, curcumin adopts the keto form, exhibiting chemical stability and acting as a proton donor (79). At pH more than 7, it is unstable and shifts to the enol form, an electron donor that contributes to its antioxidant effects (79, 80).
The lipophilic nature of curcumin is collectively attributed to its nonpolar methyl groups, aromatic rings, and aliphatic bridge (81). However, its three hydroxyl groups undergo protonation and deprotonation depending on pH, which affects its water solubility (81). In neutral and acidic environments, curcumin has poor water solubility due to hydroxyl group protonation. Under alkaline conditions, deprotonation leads to negative charges, increasing water solubility (82). These changes are reflected in shifts in curcumin’s log P value, which decreases (from 3.2) with increased polarity upon hydroxyl group deprotonation, enhancing its water solubility and facilitating its elimination (83).
Manolova et al. (84) utilized advanced ultraviolet-visible spectroscopy (UV-VIS) and quantum chemical calculations to investigate curcumin’s tautomerism in ethanol/water binary solutions (84). Their findings indicate that curcumin in ethanol exists only in the enol-keto tautomer, while water induces a shift to the diketone tautomer (84). Mass spectrometry and liquid chromatography studies confirmed that the enol form predominates in water/acetonitrile solutions (85). In nonpolar solvents such as carbon tetrachloride, curcumin remains in its enol tautomer in solid and liquid states. In solution, curcumin is inherently unstable, with a vibrant yellow color that transitions to deep crimson when exposed to alkaline conditions (86).
Structure-activity relationship studies have highlighted that the two phenyl rings connected by a C-7 linker with keto-enol functionality are crucial for curcumin’s biological activity (87). The unsaturation in the linker, which provides conformational flexibility, is particularly important for its antitumor and anticancer effects, though it is less critical for its redox regulatory or apoptotic activities (80). While synthetic methods for producing curcumin from acetylacetone and vanillin have been established, these approaches typically require lengthy reaction times and yield low amounts of product over multiple steps (88). As a result, there is a need for more efficient and streamlined synthetic methods to produce curcumin quickly and effectively (89).
8 Bioavailability of curcumin
The primary limitation in utilizing curcumin’s therapeutic potential stems from its intrinsic physicochemical characteristics. These characteristics restrict its functional efficacy, rendering less than 2% of curcumin bioavailable to the body and limiting its clinical use (71, 90–92).
8.1 Intestinal stability and permeability of curcumin
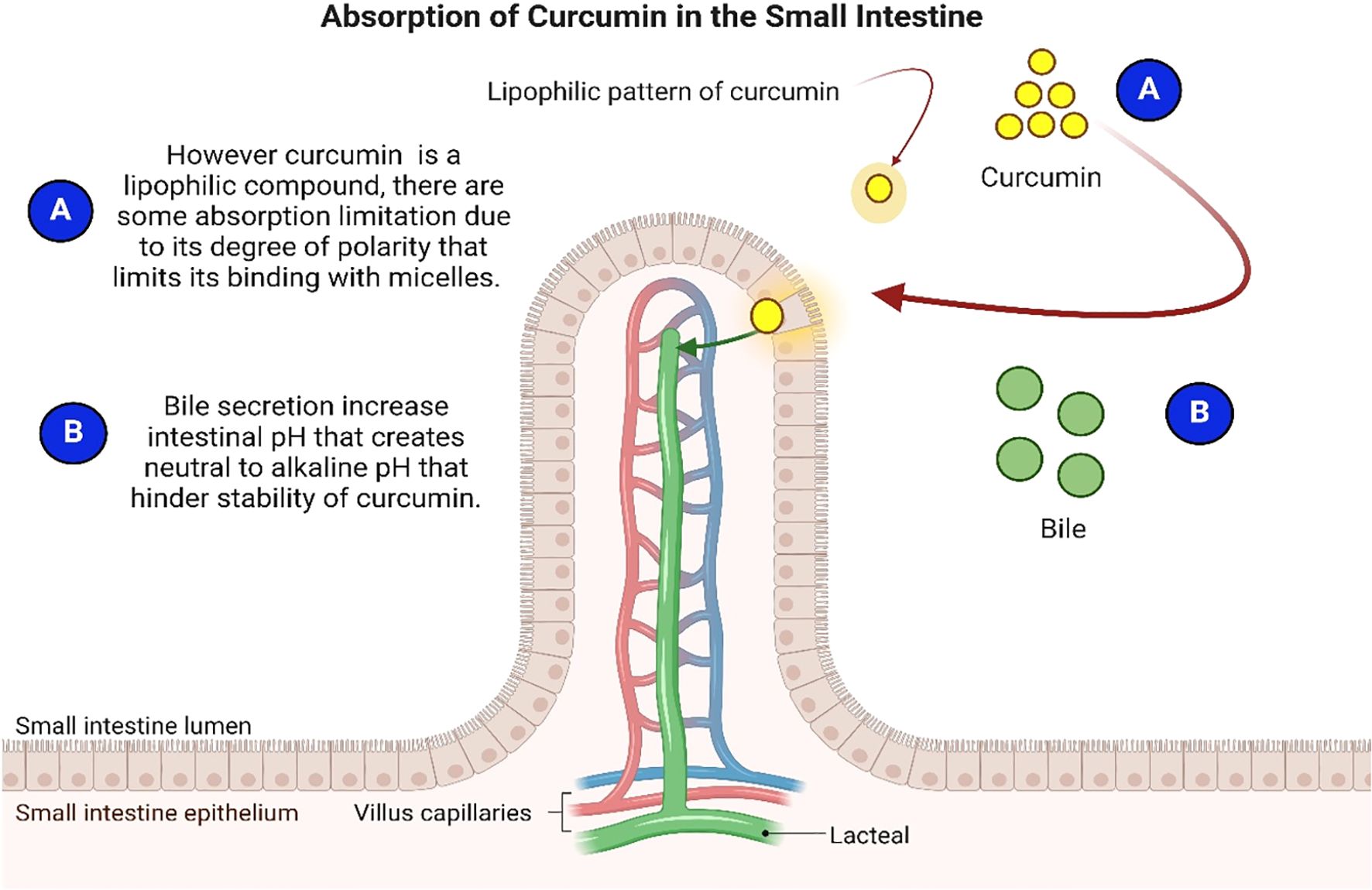
Curcumin’s stability in the intestine is critical to its permeability and absorption. Several physicochemical properties influence its intestinal stability, posing significant challenges for absorption and thus reducing its therapeutic potential across body tissues (86). These challenges include curcumin’s poor solubility in gastrointestinal fluids, which hinders its passage through the mucus layer and subsequent absorption by epithelial cells. Like other lipophilic compounds, only stable and soluble curcumin components post-digestion are absorbed by enterocytes (93). The metabolism of lipophilic nutrients follows a different absorption pathway through the membrane compared to polar nutrients, majorly due to the non-polarity of lipophilic compounds (94). Figure 3 illustrates the process of curcumin absorption in the small intestine.
Lipophilic compounds are transported through membranes by encapsulation with micelles, which the body produces naturally (94). These micelles have polar exteriors and nonpolar interiors, allowing them to bind to nonpolar compounds and facilitate membrane transport (94). As a lipophilic compound, curcumin is expected to follow this pathway; however, its limited polarity can hinder bindings with micelles (94). Upon entering the small intestine, bile secretion increases the pH to a neutral or alkaline state, which reduces curcumin’s stability and solubility, further interfering with its absorption (95).
The intestinal barrier plays a key role in curcumin’s entry into the bloodstream. Although limited information exists on the exact mechanisms, recent studies suggest passive diffusion as the primary mechanism for cellular uptake of natural curcumin, including clathrin-mediated endocytosis (96–98). Notably, curcumin uptake was observed to have a concentration-dependent effect. At relatively lower concentrations, passive transport dominates, while higher concentrations trigger active transport mechanisms, leading to reduced absorption rates into the ileum (99).
At low concentrations, curcumin primarily interacts with the polar heads of the outer membrane surface. Conversely, at higher concentrations, it tends to accumulate within the nonpolar chains of the phospholipid bilayer (100, 101). This accumulation may result in reduced membrane fluidity, as observed in several studies (101, 102). In vivo studies on rats showed poor absorption of orally administered curcumin, with more than 90% excreted in feces within 72 h (92).
Plasma concentrations remained minimal, even at high doses, with most curcumin localized in the small intestine and little reaching systemic circulation, resulting in a bioavailability of approximately 1% (71, 90, 92). Human studies corroborate these findings, with low plasma concentrations of curcumin and its metabolites even at high doses, with bioavailability ranging from 0.16 to 1% (92). Poor curcumin permeability was attributed to intestinal first-pass metabolism and intracellular retention (103). While rodent studies provide valuable findings, their extrapolation to human oral consumption remains unclear, necessitating further research and evidence for validation.
8.2 Metabolism and elimination of curcumin
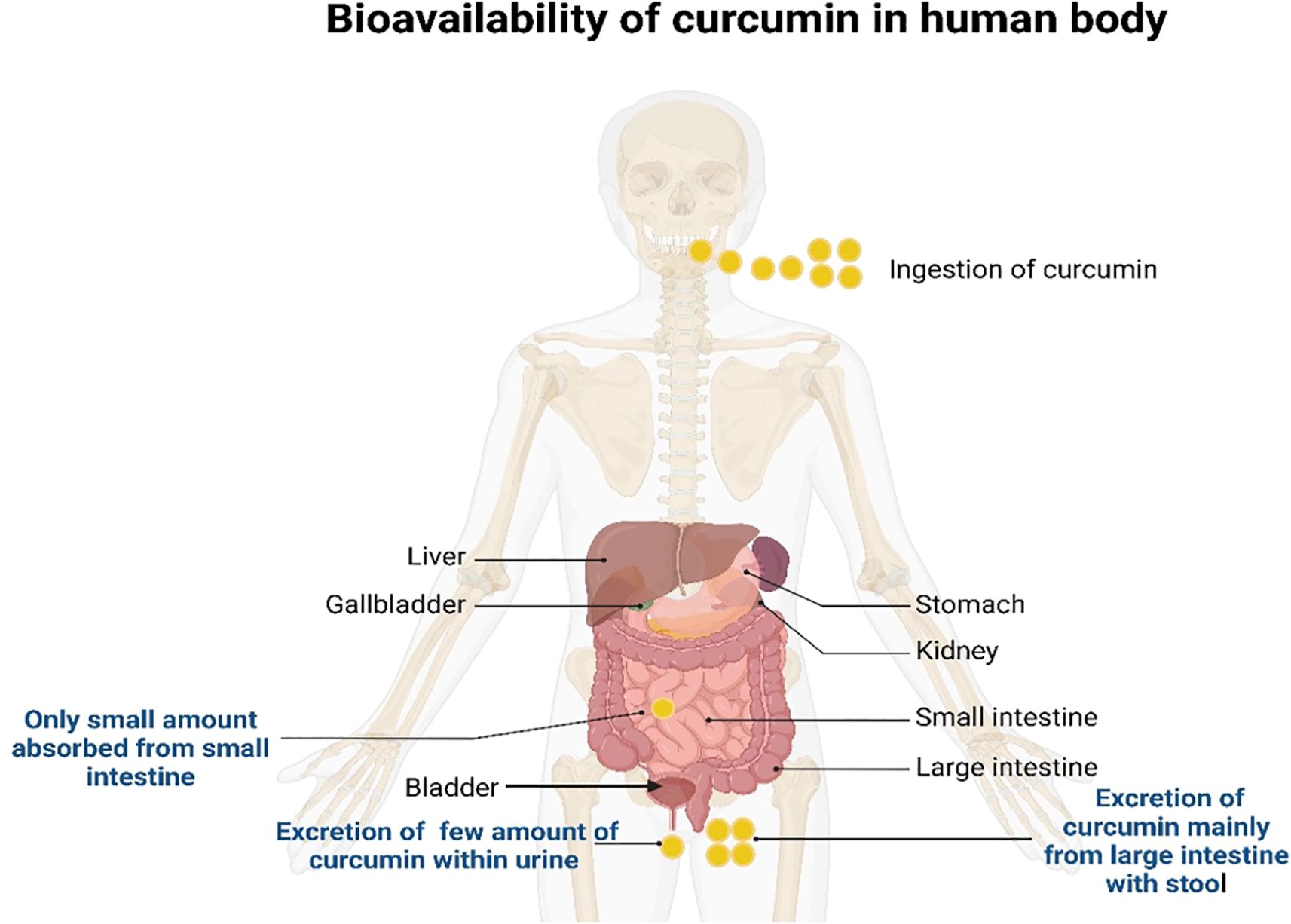
Curcumin’s bioavailability in humans is mostly limited by poor intestinal absorption, rapid hepatic metabolism, and rapid systemic elimination, even at high doses of 12 g/day (104). Most orally ingested curcumin is excreted in feces without undergoing significant metabolic transformation (98). However, the small fraction that is absorbed undergoes a two-stage metabolic pathway. During the initial phase, reductase levels are decreased in enterocytes and hepatocytes (98). Curcumin elimination mostly occurs via feces rather than urine, and approximately 90–98% of orally administered curcumin is eliminated through feces and bile (92, 105, 106). The bioavailability, absorption, and excretion of curcumin inside the human body are indicated in Figure 4.
The remaining curcumin is absorbed by enterocytes, where most of it undergoes xenobiotic metabolism via intestinal and hepatic cell-resident phase I and II enzymes (107, 108). During phase I metabolism, dihydrocurcumin (2HC) is produced when reductases break the double bonds in curcumin, followed by tetrahydrocurcumin (4HC), hexahydrocurcumin (6HC), and octahydrocurcumin (8HC) (108). Phase I metabolism also involves cytochrome P450 (CYP) enzymes, particularly CYP3A4, which is the most prevalent hepatic enzyme and plays a key role in metabolizing dietary phytochemicals like curcumin (109–111).
Dei Cas and Ghidoni (74) reported that 2HC, 4HC, 6HC, and 8HC are produced after reduction. The enzymes involved are nicotinamide adenine dinucleotide phosphate (NADPH)-dependent reductase, alcohol dehydrogenase, and an unknown microsomal enzyme that sped up curcumin degradation (112). Hassaninasab et al. (113) analyzed an enzyme isolated from Escherichia coli that reduces curcumin. They found two reduction phases in the microbial degradation of curcumin by the purified enzyme. Curcumin was initially changed into intermediate 2HC and, ultimately, 4HC, depending on NADPH availability (113).
Curcumin and its reduced metabolites undergo conversion to glutaric acid and sulfate in both in vitro and in vivo conditions. These transformations are facilitated by glucuronyl transferase and sulfotransferase (SULT) enzymes, which mediate glucuronidation and sulfation processes. Sulfated and glucuronidated curcumin has been identified in the intestines and livers of both rats and humans (74), while human plasma contains water-soluble conjugates of sulfate and glucuronide, indicative of curcumin absorption following oral administration (70).
Hager et al. (114) reported that curcumin glucuronidation is catalyzed by uridine diphosphate-glucuronosyltransferase (UDP-UGT). Meanwhile, human phenol SULT1A1 and SULT1A3 are metabolized in rat intestines. Notably, these metabolic modifications result in reduced biological activity (108). Reduced or conjugated curcumin forms, such as 4HC, 6HC, and curcumin sulfate, showed a diminished ability to inhibit cyclooxygenase-2 (COX-2) synthesis (108). Additionally, while other conjugates exhibited decreased inhibition of prostaglandin E2 synthesis, hexahydrocurcuminol is biologically inert (115).
Compared to curcumin itself, the biological effectiveness of curcumin metabolites, excluding 4HC, is considerably reduced (82, 116). Many strategies have been developed to enhance curcumin absorption, including the use of piperine, which inhibits glucuronidation, and the incorporation of curcumin into delivery systems such as liposomes, nanoparticles, phospholipid complexes, or structural analogs of curcumin (110, 111).
Pfeiffer et al. (117) demonstrated the formation of reductive metabolites, such as 4HC, 6HC, and 8HC, during curcumin metabolism in rat liver tissue sections (117). In phase II metabolism, which occurs in the intestinal and hepatic cytosol, UDP-UGT and SULT enzymes catalyze the conjugation of glucuronide and sulfate to curcumin and its reduced metabolites (107, 108). Specifically, curcumin undergoes sulfation mediated by SULT1A1 and SULT1A3 and glucuronidation facilitated by UDP-UGT enzymes (107, 108, 118). This yields curcumin glucuronide (CG), HC glucuronide (2HC-G), 4HC glucuronide (4HC-G), 6HC glucuronide (6HC-G), and 8HC glucuronide (8HC-G), with corresponding sulfate conjugates produced similarly. Notably, glucuronide conjugates are approximately twice as abundant as sulfate conjugates (119).
The principal human curcumin metabolites following oral consumption are predominantly 4HC, 6HC, and glucuronide conjugates such as CG, 4HC-G, and 6HC-G (107, 108, 115). Pan et al. (118) conducted hydrolysis of plasma curcuminoid samples using glucuronidase, revealing that 99% of curcumin metabolites in plasma are glucuronide conjugates (118). These metabolic modifications significantly alter curcumin’s physicochemical properties, shifting its polarity and consequently increasing its water solubility. This shift is evident through curcumin’s log P value, which decreases from 3.2 to 1.6 when conjugated with glucuronide, enhancing its water solubility and facilitating urinary elimination (83). Similar changes are observed with curcumin’s reduced metabolites. For instance, the log P value of 6HC decreases from 2.2 to 1.1 upon glucuronidation (83). Thus, phase I and II metabolism reduces curcumin’s bioavailability and alters the structural and chemical interactions of curcumin and its metabolites (119).
Transporters significantly influence curcumin’s bioavailability by regulating its efflux and influx across cellular barriers (114). Key efflux transporters involved in the transport of curcumin and its metabolites include P-glycoprotein (P-gp), multidrug resistance-associated proteins (MRP), and breast cancer resistance protein (BCRP) (114). These transporters are widely distributed in tissues such as the epithelial cells of the gastrointestinal tract, the blood-brain barrier, and the liver (120). After absorption into enterocytes, soluble curcumin and its metabolites face two potential pathways: efflux back into the intestinal lumen via transporters, such as MRP2, BCRP, and P-gp, or movement into the portal blood through MRP1 and MRP3 transporters (92, 121). Curcumin and its metabolites undergo further metabolism in the liver before being excreted into bile or distributed to tissues or kidneys via systemic circulation (92, 121).
Efflux transporters play a crucial role in limiting intracellular drug accumulation, thereby reducing drug efficacy (120). The dual role of these transporters in facilitating curcumin’s cellular movement underscores their significance when developing strategies to enhance curcumin’s bioavailability (114).
8.3 Tissue distribution and half-life of curcumin
The half-life of curcumin is a key parameter for understanding its pharmacokinetics. In diabetic rats, the half-life of curcumin in systemic circulation following oral intake was 32.70 ± 12.92 min (122). This finding aligns with a prior study on healthy rats, which reported an elimination half-life of 28 min after oral ingestion of curcumin (91). These short half-life values emphasize curcumin’s rapid metabolism and elimination, contributing to its limited systemic bioavailability and therapeutic efficacy after oral intake (117). Maintaining therapeutic levels of curcumin in systemic circulation remains a significant challenge due to this rapid clearance. In humans, calculating the absorption rate and elimination half-life for orally administered curcumin remains challenging, as serum levels often fall below the detection limit (123). However, understanding curcumin’s tissue distribution is essential for optimizing its therapeutic potential, especially in target body regions (117).
A recent review of curcumin’s distribution reported that although curcumin’s tissue distribution has been extensively studied in rats, evaluations in humans are limited (71). Studies using rodent models have shown variable tissue distribution patterns attributed to differences in dose preparations, extraction methods, and detection assays (67). This variability is compounded by curcumin’s rapid degradation and transformation both before and after absorption, complicating the consistency of results and observed distribution patterns (71). Ultimately, these findings suggest that the parent compound does not accumulate significantly in specific organs (71).
While numerous clinical studies in humans have assessed curcumin’s systemic effects following oral intake, none have evaluated its tissue distribution. In rodent studies, orally administered curcumin was primarily detected in the stomach and small intestine, accounting for approximately 90% of the curcumin, with only trace amounts of unchanged curcumin in the liver and kidney (124, 125). At 24 h, only 1% of curcumin remained in the stomach and small intestine (124, 125). Some studies have used radioactive-labeled curcumin in rats to address the difficulties in measuring accurate tissue distribution from orally administered curcumin. These investigations revealed detectable radioactivity in the blood, liver, and kidney after curcumin administration, with consistent absorption percentages regardless of the administered dose (126).
Similarly, in a separate study, mice injected intraperitoneally with radioactive carbon (14C) curcumin showed peak radioactivity levels in various tissues, with the liver and intestinal mucosa having the highest peaks. Subsequently, radioactivity rapidly declined to 20–33% of the peak values within 4 h for most tissues, except the small intestine, which showed a slower decline within 8 h (127). These findings highlight the limitations of using rat models to infer human tissue distribution due to interspecies differences in drug metabolism and pharmacokinetics (128). Consequently, further studies are needed to accurately examine tissue distribution in humans following oral curcumin intake.
8.4 Bypassing curcumin’s poor bioavailability
The physicochemical properties of curcumin significantly limit its clinical utility, as its poor bioavailability restricts its functional capabilities (123). Thus, understanding the challenges associated with curcuminoids’ bioavailability is key to developing effective strategies to overcome these limitations (124). One primary factor contributing to curcumin’s low bioavailability is its rapid conjugation, particularly glucuronidation in the intestine and liver, facilitated by UDP-UGT and accounting for more than 80% of curcumin’s metabolism (129).
Enhancing curcumin’s therapeutic potential requires a focus on prolonging its serum bioavailability by increasing its half-life and reducing its metabolic rate (124, 125). Improving curcumin’s bioavailability would require strategies that inhibit metabolic pathways and slow curcumin’s elimination rate. The coadministration of bio-enhancing substances that modulate these pathways has been widely investigated (125). Piperine stands out for its significant potential to improve the systemic availability of curcumin and, hence, its bioavailability (130). Consequently, formulations integrating curcumin with piperine have gained attention as a viable approach to overcoming curcumin’s poor bioavailability (125).
Figure 5 illustrates the improvement of curcumin bioavailability via integration with drug delivery systems.
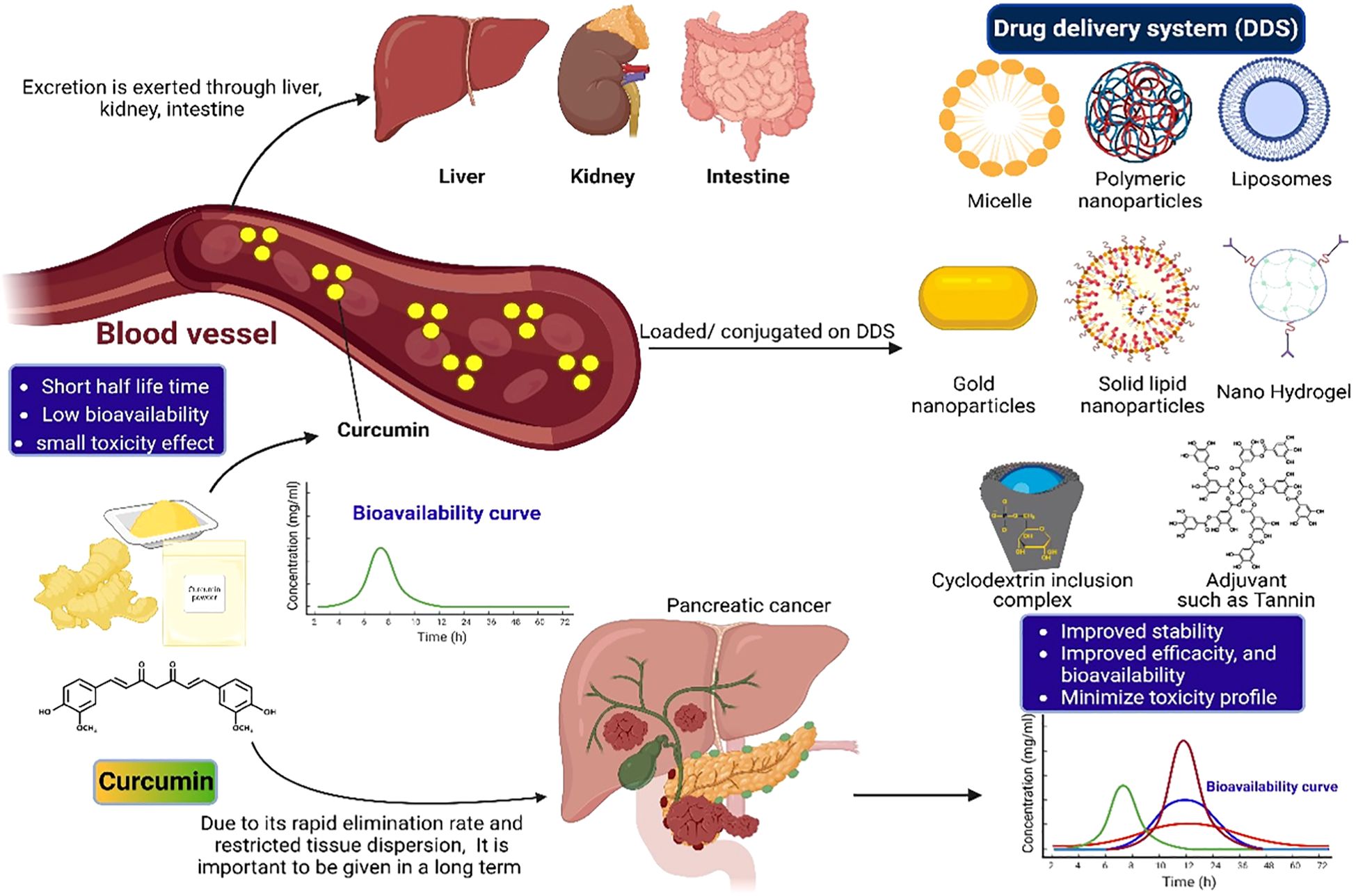
Figure 5. Improvement of curcumin bioavailability through incorporation into medication delivery systems.
8.5 Current strategies to enhance bioavailability
While curcumin demonstrates immense therapeutic promise, its clinical application is significantly hampered by poor bioavailability due to limited absorption, rapid metabolism, and quick systemic elimination. Researchers and clinicians are actively exploring various strategies to overcome these challenges, with innovative formulation techniques and delivery systems offering practical insights. These strategies encompass the procedures outlined below:
8.5.1 Adjuvants and combinations
One of the simplest strategies involves co-administration with adjuvants that inhibit curcumin’s metabolism or enhance its absorption (131). The most well-known example is piperine, a compound found in black pepper. Piperine is known to inhibit enzymes involved in curcumin’s metabolism, thereby increasing its systemic availability. Clinical trials have shown that curcumin combined with piperine can significantly increase curcumin levels in the blood (132).
8.5.2 Nanotechnology-based delivery systems
Nanotechnology offers a revolutionary approach to improve curcumin’s solubility, stability, and bioavailability by encapsulating it within nanocarriers (133). These systems can bypass gastrointestinal barriers and enhance the compound’s reach to target sites (134).
8.5.2.1 Liposomes
These artificial vesicles encapsulate curcumin, making it easier for the body to absorb. Liposomal formulations have demonstrated higher bioavailability compared to free curcumin. They are composed of phospholipid bilayers that can encapsulate curcumin within aqueous compartments or lipid bilayers, enabling targeted delivery and minimizing off-target effects (135).
8.5.2.2 Polymeric nanoparticles
These customizable platforms, often made from biocompatible polymers like poly (lactic-co-glycolic acid) (PLGA) and polyethylene glycol (PEG) derivatives, can encapsulate curcumin to improve its solubility and stability (136). They allow for controlled release kinetics, enhanced cellular uptake, and prolonged circulation times, optimizing therapeutic efficacy.
8.5.2.3 Solid lipid nanoparticles
Solid lipid nanoparticles are biocompatible lipid-based nanocarriers that offer stability and sustained release properties, making them suitable for encapsulating hydrophobic compounds like curcumin (137).
8.5.2.4 Polymeric micelles
These self-assembling colloidal systems spontaneously form when surfactants are dispersed in water. Curcumin can be solubilized within its hydrophobic core, significantly enhancing its bioaccessibility and potentially increasing the permeability of epithelial cells. Micellar curcumin formulations have shown dramatically higher bioavailability in human studies (138, 139).
8.5.2.5 Nanoemulsions
These oil-in-water or water-in-oil formulations provide a stable platform for delivering hydrophobic compounds like curcumin, enhancing its bioavailability and therapeutic efficacy.
8.5.2.6 Cyclodextrin inclusion complexes
Cyclodextrins are cyclic oligosaccharides that can form inclusion complexes with curcumin, improving its solubility and stability (140). This technique has shown improved bioavailability and enhanced antiproliferative effects (141).
8.5.2.7 Nanogels
These crosslinked polymer networks can enhance curcumin’s solubility, improve its controlled release, and prolong its half-life, leading to increased bioavailability and improved therapeutic effects (142).
8.5.3 Specific curcumin formulation
Various specialized formulations have been developed and tested in clinical trials to enhance bioavailability, BCM-95®CG Biocurcumax™, in formulation, combines curcuminoids with essential oils from turmeric rhizome, demonstrating significantly improved bioavailability compared to standard curcumin (143). Furthermore, CuraMed® and Curamin® products often incorporate BCM-95® and other synergistic ingredients to enhance absorption and therapeutic effects (144). Also, Curcuwin® and CurQfen® formulations utilize hydrophilic carriers or fenugreek dietary fibers to improve curcumin’s systemic availability (145).
8.5.4 Novel strategies to enhance bioavailability
Beyond current approaches, cutting-edge research is exploring even more innovative methods.
8.5.4.1 SMEDDS
These systems are isotropic mixtures of oils, surfactants, co-surfactants, and drugs that form fine oil-in-water emulsions or microemulsions upon gentle agitation in aqueous media. This pre-dispersion in the gastrointestinal tract can significantly enhance the dissolution and absorption of poorly soluble drugs like curcumin by presenting them in a finely dispersed, absorbable form (146).
8.5.4.2 Prodrug approaches
Designing curcumin prodrugs involves chemically modifying the curcumin molecule to improve its physicochemical properties, such as water solubility and membrane permeability. These prodrugs are designed to be inert until metabolized in vivo to release the active curcumin, potentially at the target site, thereby overcoming bioavailability issues (147).
8.5.4.3 Co-crystallization
This technique involves forming crystalline solids composed of curcumin and a co-former molecule in a defined stoichiometric ratio. Co-crystals can improve the solubility and dissolution rate of curcumin without altering its chemical structure, leading to enhanced bioavailability (148).
8.5.4.4 Amorphous solid dispersions
By dispersing curcumin in an amorphous state within a polymer matrix, amorphous solid dispersions prevent crystallization and maintain curcumin in a higher energy, more soluble form. This amorphous state can significantly enhance the dissolution rate and saturation solubility, leading to improved absorption (149, 150).
8.5.4.5 Targeted delivery systems with ligands
Incorporating specific ligands (e.g., antibodies, peptides, folate) onto the surface of nanocarriers can enable active targeting of curcumin to specific cells or tissues that overexpress certain receptors. This not only enhances accumulation at the disease site but also potentially improves cellular uptake and efficacy while minimizing off-target effects and systemic exposure to high doses (151).
8.5.4.6 Microencapsulation and spherical crystallization
These techniques aim to encapsulate curcumin within micro-sized particles or create spherical agglomerates to improve flowability, compressibility, and dissolution rate, indirectly contributing to better absorption (152).
8.5.5 General limitations of bioavailability enhancement strategies for curcumin
While significant progress has been made in enhancing curcumin’s bioavailability, several limitations and challenges are associated with the current and novel strategies (153).
8.5.5.1 Complexity and cost of manufacturing
Many advanced formulations, particularly those involving nanotechnology (liposomes, polymeric nanoparticles, SMEDDS), require complex manufacturing processes, specialized equipment, and stringent quality control. This can lead to higher production costs, potentially making the final product less accessible or affordable for widespread use (153).
8.5.5.2 Scalability issues
Translating laboratory-scale production of nanoformulations to industrial-scale manufacturing can be challenging. Ensuring consistent particle size, stability, and drug loading at larger scales requires significant investment and expertise (153).
8.5.5.3 Stability concerns
While some formulations improve stability, others might introduce new stability issues. For example, liquid SMEDDS can suffer from stability and leakage problems, though solid SMEDDS aims to address this. Amorphous solid dispersions, while improving solubility, have a natural tendency to transform into a more stable crystalline form over time, which can lead to a loss of the bioavailability benefit (153).
8.5.5.4 Toxicity of excipients and carriers
Some excipients or carrier materials used in these formulations, especially surfactants in SMEDDS or certain polymers in nanoparticles, might have inherent toxicity concerns, particularly with long-term use or at high doses. Immunotoxicity assessment is crucial for nanoparticles, as their intrinsic properties can influence potential adverse effects on the immune system (153).
8.5.5.5 Regulatory hurdles
Novel drug delivery systems and formulations often face rigorous regulatory scrutiny. Demonstrating long-term safety, efficacy, and batch-to-batch consistency for these complex systems can be a lengthy and expensive process (153).
8.5.5.6 Predictive in vitro models
A lack of robust and predictive in vitro models to assess the performance of these complex formulations remains a significant limitation. This makes it difficult to accurately predict in vivo behavior from laboratory data, necessitating more extensive animal and human trials (153).
Table 1 presents the advanced strategies that may be utilized to improve the bioavailability of curcumin.
9 Chemical degradation of curcumin
Curcumin’s extensive health benefits, minimal side effects, and low supply cost have driven significant research efforts to develop it as a supplement, therapeutic food product, or potentially a pharmaceutical product (186). Its popularity has contributed to a growing global trend of commercial food and non-food products containing turmeric, including beverages, supplements, creams, and soaps (186).
However, curcumin is highly susceptible to chemical degradation, which limits its stability and bioavailability (126). This is particularly problematic for oral ingestion because the gastrointestinal tract is unfavorable for curcumin’s stability, as will be discussed below.
9.1 Alkaline degradation of curcumin
The structural stability of curcumin may diminish under alkaline conditions. In basic environments, it undergoes hydrolytic degradation and alpha, beta-unsaturated beta-diketone moiety cleavage (88). This degradation gives rise to trans-6-(4’-hydroxy-3’-methoxyphenyl)-2, 4-dioxo-5-hexanal, which then undergoes further cleavage to yield compounds like ferulic acid, feruloymethane, and vanillin (95).
Reports have shown that 90% of curcumin degradation under alkaline and neutral conditions occurred within 15–30 min, while in acidic incubations, degradation was substantially slower, with less than 20% of total curcumin degraded within 60 min (71, 92). This suggests the importance of pH when considering the development of curcumin-based functional food products (85).
9.2 Autooxidation of curcumin
Curcumin is also susceptible to autooxidation, which occurs through radical chain reactions and spontaneously occurs in aqueous solutions (92). Autoxidation occurs at physiological pH and is initiated by the surrounding free radicals, which autoxidize the phenolic hydroxyls on the curcumin molecule (74). Autooxidation produces a succession of bicylopentadione compounds, where the seven-carbon chain is oxygenated and doubly cyclized (74), hence yielding bicyclopentadione, vinyl ether, and spiro epoxide (74, 92). Additionally, during curcumin autoxidation, small amounts of two configurational isomers of bicyclopentadione are produced as by-products of lipoxygenase-catalyzed oxygenation (187).
9.3 Photodegradation of curcumin
Curcumin is susceptible to photodegradation when exposed to light in both crystalline and solubilized forms (128). This is visually detected as color fading. Chemical degradation of curcumin occurs at the alpha, beta-unsaturated beta-diketone moiety, yielding compounds such as vanillin, vanillic acid, p-hydroxybenzaldehyde, ferulic aldehyde, and ferulic acid (188).
10 Characterization of curcumin
Curcuminoids are extensively employed in the food processing and pharmaceutical sectors due to their unique properties. Hence, accurate identification and characterization of curcuminoids in various substances are crucial, with the selection of an appropriate analytical technique influenced by factors such as sample type, analytical objectives, and detection limits (189, 190). Chromatography and electrophoresis-based methods are among the preferred approaches for curcuminoid analysis (130).
According to Nurjanah and Saepudin (62) TLC is one method used to fractionate turmeric extracts. Although it is selective, easy to use, and cost-effective, its limitations—such as low resolution and long separation times—have reduced its popularity in turmeric research (62). Recent advancements in TLC have introduced new high-performance TLC (HPTLC), which effectively addresses the constraints associated with traditional TLC (191). HPTLC provides many benefits, including enhanced resolution, reduced detection limit, and increased image scanning capabilities (190).
HPLC remains the most widely used chromatographic technique for qualitative and quantitative analysis of curcumin. Curcuminoids may be analyzed using different HPLC techniques. When combined with a UV–VIS detector, HPLC offers high precision, accuracy, and sensitivity (129). For more complex matrices, liquid chromatography-mass spectrometry techniques have been developed to detect and measure curcumin traces in food, biological fluids, and other samples (189). Several liquid chromatography-mass spectrometry (LC/MS) techniques have been established to identify and measure curcumin in various matrices (192, 193).
Tandem mass spectrometry and ultra-HPLC have demonstrated high throughput, sensitivity, and selectivity for curcuminoid quantification, significantly reducing analytical time and improving sensitivity (194). Additionally, curcuminoids can be quantified using UV-VIS if they absorb within the matrix or sample components. Curcumin exhibits maximum absorption at 425 nm (112, 190, 195).
Curcumin is also characterized using various spectroscopic techniques, including nuclear magnetic resonance, fluorescence, Fourier transform Raman spectroscopy, near-infrared spectroscopy, and Fourier transform infrared spectroscopy (196, 197).
Curcumin exists in three polymorphic forms: two orthorhombic and one monoclinic. Differential scanning calorimetry and X-ray diffraction analyses revealed that these polymorphs are monotropically related, with the monoclinic form being the most stable (198).
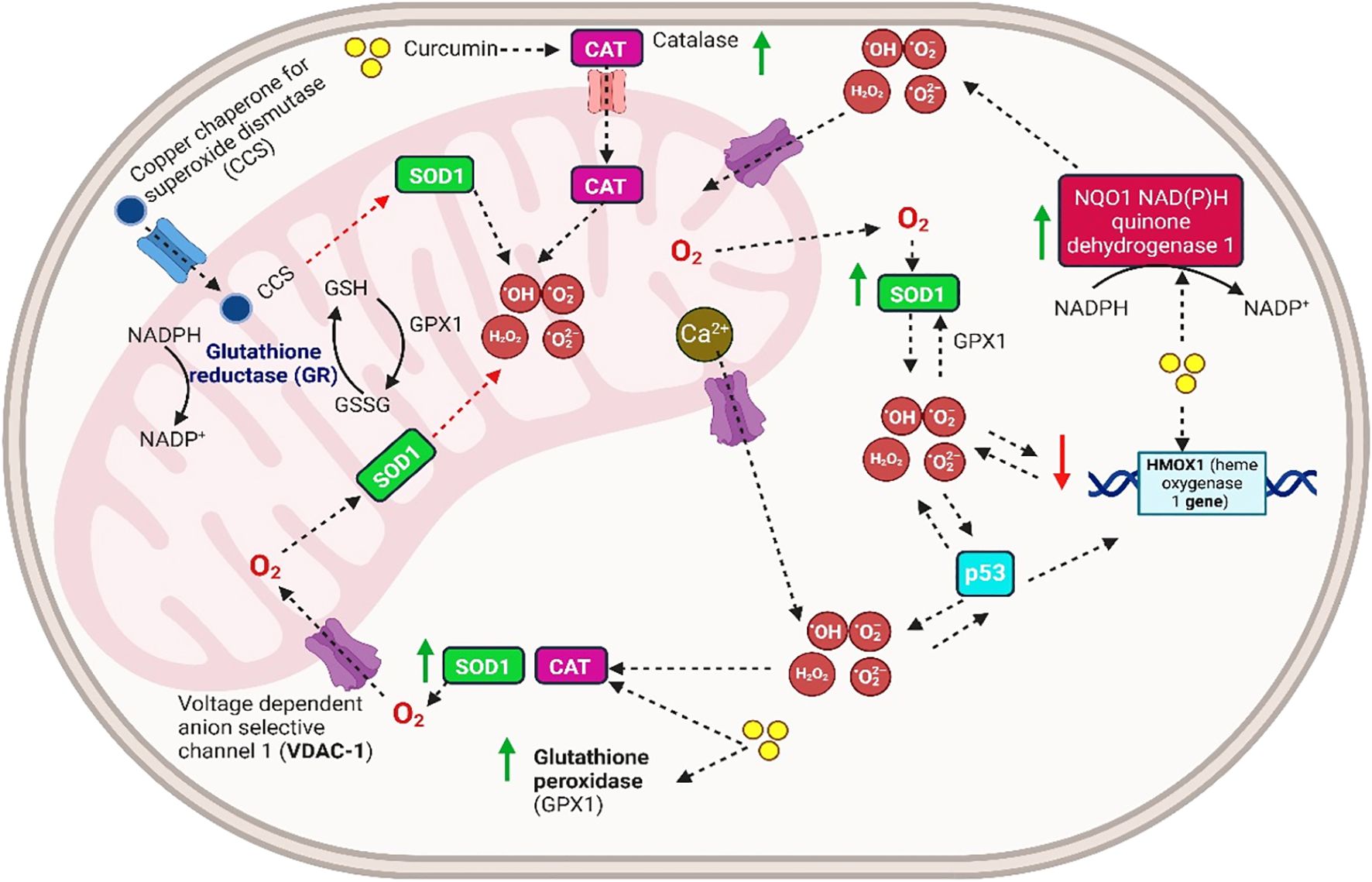
Electron paramagnetic resonance (EPR) spectroscopy is a non-invasive and efficient technique for examining materials containing unpaired electrons. According to Iravani and Soufi (199), EPR spectroscopy identifies different types of radicals and evaluates the antioxidative properties of compounds. Curcumin’s antioxidant properties have been assessed using EPR spectroscopy with free radicals such as 1,1-diphenyl2-picryl hydrazyl (DPPH), nitric oxide (NO·), hydroxyl (HO·), and superoxide (O2) (200, 201).
A study by Nikolic et al. (202) employed EPR spectroscopy to assess the antioxidant properties of low-energy nanoemulsions loaded with curcumin based on the stability of the tempol nitroxide free radical. The results demonstrated that the curcumin-containing nano-emulsion rapidly neutralized free radicals within the first 5 min (202).
11 Formulations of curcumin
Several curcumin formulations incorporating CP-01, piperine, and lecithin have been developed to improve absorption following oral administration compared to pure curcumin (203, 204). Innovative formulations with considerable potential include micelles, liposomes, phospholipid complexes, nanoparticles, cyclodextrins, emulsions, hydrogels, and phytosomes. These formulations enhance curcumin’s efficacy by increasing its circulation over extended periods, improving uptake and resisting metabolic processes, boosting absorption through the small intestine, and extending its plasma half-life (205, 206).
A study on healthy volunteers tested various formulations, including curcumin-containing phytosomes, CP-01, and hydrophilic carrier formulations containing cellulose derivatives and natural antioxidants (CHC) (144). These formulations were compared to a standard curcumin preparation. The results indicated that curcumin prepared using the CHC method achieved significantly higher blood curcuminoid levels than conventional preparations (204).
Cyclodextrins form molecular inclusion complexes with lipophilic substances, enhancing active components’ water solubility, dispersion, and absorption (20). One previous study examined the bioavailability of a curcumin formulation with γ-cyclodextrin. This formulation was compared to turmeric essential oils, curcumin phytosomes derived from rhizomes, and a standardized curcumin extract (20). The results showed that γ-cyclodextrin formulations significantly improved curcuminoid absorption in healthy individuals (20).
The coprecipitation method created a curcumin-β-cyclodextrin inclusion complex, significantly increasing curcumin’s water solubility from 0122 to 0.72100 mg/mL. Under simulated gastrointestinal conditions, the release of this inclusion complex was tested using standard poly (N-isopropyl acrylamide/sodium alginate) hydrogels cross-linked with nano-clay and N, N0-methylene bis(acrylamide) (BIS) (147). These nanocomposite hydrogels exhibited the minimum release-swelling ratio at a pH of 1.2 and the maximum at a pH of 6.8 (147). In nanocomposite hydrogels, increasing the nano-clay concentration resulted in a decrease in both the swelling coefficient and cumulative release. In contrast, with conventional hydrogels, the swelling ratio and cumulative release increased as the BIS ratio increased (207).
Kongkaneramit et al. (208) synthesized curcumin-containing liposomes using the polyol dilution technique. The lipid phase consisted of carbohydrates and hydrogenated phosphatidylcholine combined in a 9:1 molar ratio. Propylene glycol, glycerin, and polyethylene glycol 400 were used as polyol solvents. The curcumin content and liposome size were influenced by the type and concentration of polyol used, as well as the preparation temperature, which is a critical factor in liposome development (208).
Tai et al. (209) investigated curcumin’s stability and release properties in liposomes with varying hydrogenated phospholipid concentrations. They identified chitosan-coated liposomes as a potential drug delivery system. Cuomo et al. (210) further examined the efficacy of anionic liposomes and chitosan-coated liposomes containing curcumin. They tested the formulations in vitro by assessing the bioavailability of ingested curcumin. The positively charged surface of chitosan-coated liposomes enhanced curcumin’s overall bioavailability by facilitating its improved absorption in the small intestine (210).
A low-energy curcumin nano-emulsion was developed and converted into a nano-emulgel by incorporating cross-linked polyacrylic acid (Carbopol® 934) as a gelling agent (151). This formulation was designed to enhance the solubility and absorption of curcumin when applied topically (151). In psoriatic mice, the nano-emulgel showed earlier and faster wound healing compared to both curcumin and betamethasone-17-valerate gel, indicating its potential for long-term psoriasis treatment (211). Additionally, curcumin nanoemulsions were shown to prevent postoperative tumor metastasis and recurrence effectively (212).
A thermosensitive hydrogel incorporating latanoprost and curcumin nanoparticles was recently developed as a formulation with a dual drug delivery system (153). This formulation demonstrated biocompatibility in both in vitro and in vivo studies, along with a delayed release profile (153). It also decreased inflammation and apoptosis while protecting trabecular mesh cells from oxidative damage (213). PLGA-based curcumin nanoparticles have shown enhanced oral and intravenous bioavailability (214). Saber-Moghaddem et al. (215) reported that administering oral nano-curcumin could significantly reduce recovery time in hospitalized patients with COVID-19. A curcumin-phospholipid complex, combining both curcumin and phospholipids, was employed for oral drug delivery to prevent metastases of breast and lung cancers (216).
Wang et al. (217) developed a curcumin-phospholipid complex with enhanced flow properties, solubility, and oral bioavailability, resulting in a high-performance formulation. Furthermore, polymer micelles composed of methoxy-poly(caprolactone)-poly(ethylene glycol) enabled the delayed release of curcumin (218). Liu et al. (219) developed the amylopectin–chitosan composite hydrogel (LRA–CS) for curcumin delivery and observed the dissolution characteristics of curcumin encapsulated in artificial stomachs and intestinal fluids (191). The results revealed that the LRA-CS hydrogel effectively maintained the stability of curcumin in the stomach and facilitated its controlled release in the small intestine (219). Furthermore, a hydrogel made of chitosan, nanocellulose, and a non-ionic surfactant was developed for curcumin delivery (220).
Transdermal administration of resveratrol and curcumin was achieved using cyclodextrin nanospongoid-based hydrogel (CDNS). This system significantly enhanced the in vitro release of curcumin and resveratrol by factors of 10 and 2.5, respectively, compared to their conventional forms (193). The combination of curcumin-CDNS and resveratrol-CDNS exhibited a synergistic cytotoxic effect on breast cancer MCF-7 cells lines. These formulations were integrated into a hydrogel base containing carbomer and propylene glycol (193). Adding CDNS to the hydrogel improved the photostability of resveratrol and curcumin approximately fivefold and sevenfold, respectively, compared to a hydrogel without CDNS. The use of a CDNS-hydrogel base notably increased the consumption of curcumin and resveratrol (221).
Curcumin was also incorporated into a hydrogel system based on oxidized cellulose and polyvinyl alcohol through a freezing process (222). Shefa et al. (222) demonstrated its efficacy in promoting spontaneous wound healing in in vitro rats studies. Sahin et al. (223) developed advanced ultrasol curcumin (AUC), a novel curcumin formulation with enhanced bioavailability and intestinal stability. AUC was demonstrated to improve the pathophysiology of experimentally induced osteoarthritis in rats significantly (222).
In clinical trials with healthy volunteers, the oral bioavailability of a novel curcumin formulation, Curene®, was compared to a formulation containing CP-01 and conventional curcuminoids 95%. Panda et al. (224) demonstrated that Curene® had significantly better oral bioavailability (95%) than CP-01 and regular curcuminoids and was safe for use under trial conditions. Additionally, Longvida® improved curcumin (LC) was studied for its anti-inflammatory properties in two-month-old wild-type and GFAP-IL6 mice. LC reduced inflammation and limited neurodegeneration and motor deficits in GFAP-IL6 mice (225).
Various commercial curcumin formulations exhibit bioavailability exceeding that of standard curcumin by more than 100-fold (21). These include products for oral administration and other formulations like Curcumin Rich, Liposomal Curcumin, Biomor, Dr. Mercola Curcumin Advanced, and Liposomal Curcumin Mango, which are available (205).
12 Health benefits and safety of curcumin
All three curcuminoids have shown potential therapeutic effects (226); however, studies have primarily focused on curcumin due to the growing evidence of its potential as a therapeutic agent in humans. A recent systematic review and meta-analysis of randomized-controlled trials (RCTs) using the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) approach inferred that curcumin could improve inflammation and oxidative stress in adults ≥ 18 years of age across various health conditions (227).
The therapeutic potential of curcumin has become increasingly evident over the past several decades. Numerous studies have demonstrated its effectiveness against a range of cancers, including chemoresistant colon cancer cells, esophageal cancer, thyroid carcinoma, and skin cancer (228). Additionally, curcumin has shown strong anti-inflammatory properties (229). It exerts its anticancer effects by regulating various growth factors, protein kinases, inflammatory cytokines, and transcription factors, thereby inhibiting tumor proliferation and metastasis (228). Beyond its anticancer and antitumor activities, curcumin has also been found beneficial in treating various diseases such as respiratory tract infections, hepatic steatosis, skin photoaging, Parkinson’s disease, obesity, diabetes, HIV-associated diarrhea, and Alzheimer’s disease, primarily by inhibiting amyloid beta oligomer formation (230, 231).
Curcumin interacts with several targets within cancer pathways, notably protein kinases. It inhibits protein kinase C by forming hydrogen bonds with specific residues in the C1B subdomain and fits well within the binding pocket of glycogen synthase kinase-3β through interactions with key amino acids (232). Curcumin is also reported to act as a non-competitive inhibitor of phosphorylase kinase. Cyclin-dependent kinases (CDKs) are recognized as important cancer targets, with CDK1, CDK2, and CDK4/6 playing significant roles in cell cycle regulation. Disruptions in CDK activity are linked to cancer development (232).
Additional claims regarding curcumin’s antidiabetic, anticancer (233, 234), and hepatic benefits have been made. However, they are considered weak based on rigorous systematic reviews and meta-analyses (226, 235, 236). One systematic review linked curcumin’s potential antidiabetic effects to its anti-inflammatory and antioxidant properties (235).
Regarding safety, a recent literature review of double-blinded RCTs examined the safety and toxicity profiles of turmeric and curcumin in medical applications (237). Both turmeric and curcumin were safe for human use, particularly when taken orally. They were also considered safe in animal studies, showing non-mutagenic properties and safety during pregnancy. However, further studies in humans are recommended (237). Oral administration of curcumin at a dose of 6 g/day for 4–7 weeks was reported to be safe. However, minor gastrointestinal disturbances may occur (237).
Although curcumin is considered the primary curcuminoid for its therapeutic potential, recent studies have highlighted similar therapeutic potentials of several curcumin metabolites (238), such as CG, 2HC, 4HC, 6HC, and 8HC. These metabolites exhibit potential therapeutic effects, including antioxidative, anticancer, anti-inflammatory, and antiseptic properties, for various diseases, including liver disorders, neurological conditions, cancer, cardiovascular diseases, and lung diseases, as shown in multiple in vitro and in vivo studies on humans and mice (114, 116, 239).
However, the extent of therapeutic activity of curcumin metabolites compared to curcumin itself remains unclear. While 4HC and 6HC have been more extensively studied, research on 2HC and 8HC is relatively limited, necessitating further investigation into their therapeutic activities (114, 116, 239). Although in vitro and in vivo preclinical studies have demonstrated the therapeutic potential of these metabolites, further validation through animal models and subsequent clinical trials is required to provide robust evidence (206).
Clinical studies have consistently shown curcumin’s safety, tolerability, and effectiveness in managing various chronic human illnesses (19). Soleimani et al. (237) reported no adverse effects in humans after orally administering 6 g of curcumin per day for 4–7 weeks. Additionally, Greil et al. (240) investigated the safety, tolerability, and efficacy of liposomal curcumin (Lipocurc™) in patients with metastatic or locally advanced cancer. Their findings showed that a 300 mg/m2 dose of Lipocurc™ was the maximum dose that could be safely administered to individuals undergoing cancer therapy (240). Saghatelyan et al. (241) evaluated the combination of paclitaxel with intravenous curcumin infusion in patients with metastatic breast cancer. Following a 12-week treatment regimen, intravenous curcumin did not result in any significant adverse effects. Additionally, it did not negatively impact patients’ overall quality of life (241).
A cautionary note regarding potential herb-drug interactions is indeed warranted, particularly concerning curcumin with anticoagulants or chemotherapeutic agents.
12.1 Curcumin and anticoagulants
Curcumin, the bioactive component of turmeric, has demonstrated antiplatelet and anticoagulant properties in numerous studies. This indicates its capacity to impede blood coagulation. The concomitant use of anticoagulant drugs (including warfarin, clopidogrel, aspirin, or dabigatran) elevates the risk of bleeding, bruising, and hematoma development (242). The combined effect can lead to an amplified anticoagulant effect, potentially pushing the patient beyond their therapeutic range and increasing the likelihood of hemorrhagic complications (242).
12.2 Curcumin and chemotherapeutic agents
The interaction between curcumin and chemotherapeutic agents is more complex and can be either beneficial or detrimental, depending on the specific chemotherapy drug and cancer type (243).
12.2.1 Potential synergistic effects
In some preclinical studies, curcumin has shown the ability to enhance the efficacy of certain chemotherapeutic agents (e.g., paclitaxel, gemcitabine, cisplatin) by sensitizing cancer cells to the drugs, inhibiting drug resistance, and promoting apoptosis. This could potentially allow for lower doses of chemotherapy or improve treatment outcomes (243).
12.2.2 Potential antagonistic effects
Conversely, curcumin has been shown to interfere with the action of other chemotherapeutic drugs (e.g., cyclophosphamide, doxorubicin) by activating certain detoxification pathways (like ABC transporters) that pump the drugs out of cancer cells, thereby reducing their effectiveness (244). There are also concerns that curcumin’s antioxidant properties could, in some contexts, protect cancer cells from the oxidative damage induced by certain chemotherapies (244).
12.3 General cautionary points for herb-drug interactions
The combination of herbs and drugs can lead to an exaggeration of either the drug’s or the herb’s effects, or introduce new, unpredictable adverse reactions. Herbs can sometimes accelerate the metabolism or excretion of drugs, leading to sub-therapeutic drug levels and reduced treatment effectiveness (245, 246).
Herbs can influence drug absorption, distribution, metabolism (especially via cytochrome P450 enzymes), and excretion, leading to altered drug concentrations in the body. Herbal products often lack the rigorous standardization and regulation of pharmaceutical drugs, leading to variability in their active compound content and potential contamination (246, 247).
While in vitro and animal studies exist, robust human clinical trials on many herb-drug interactions are often limited, making it difficult to predict precise effects in patients (245–247).
Patients must inform their healthcare providers (doctors, pharmacists, nurses) about all herbal supplements, traditional medicines, and dietary supplements they are taking, even if they seem innocuous. Healthcare professionals should proactively inquire about such use (245, 247). This allows for identifying potential interactions and assessing the patient’s risk factors and modifying drug dosages if necessary. Continuous monitoring for adverse effects or altered drug efficacy is essential (246). Moreover, it is essential to inform patients about the significance of avoiding self-medication with herbal remedies while undergoing conventional pharmacological therapy, as well as the possible risks of interactions (245–247).
In conclusion, while herbs such as curcumin offer potential health benefits, their interaction with prescription medications can have significant clinical implications, ranging from increased toxicity to reduced therapeutic efficacy (245). A cautious and informed approach, with open communication between patients and healthcare providers, is paramount to ensuring patient safety and optimal treatment outcomes (246, 247).
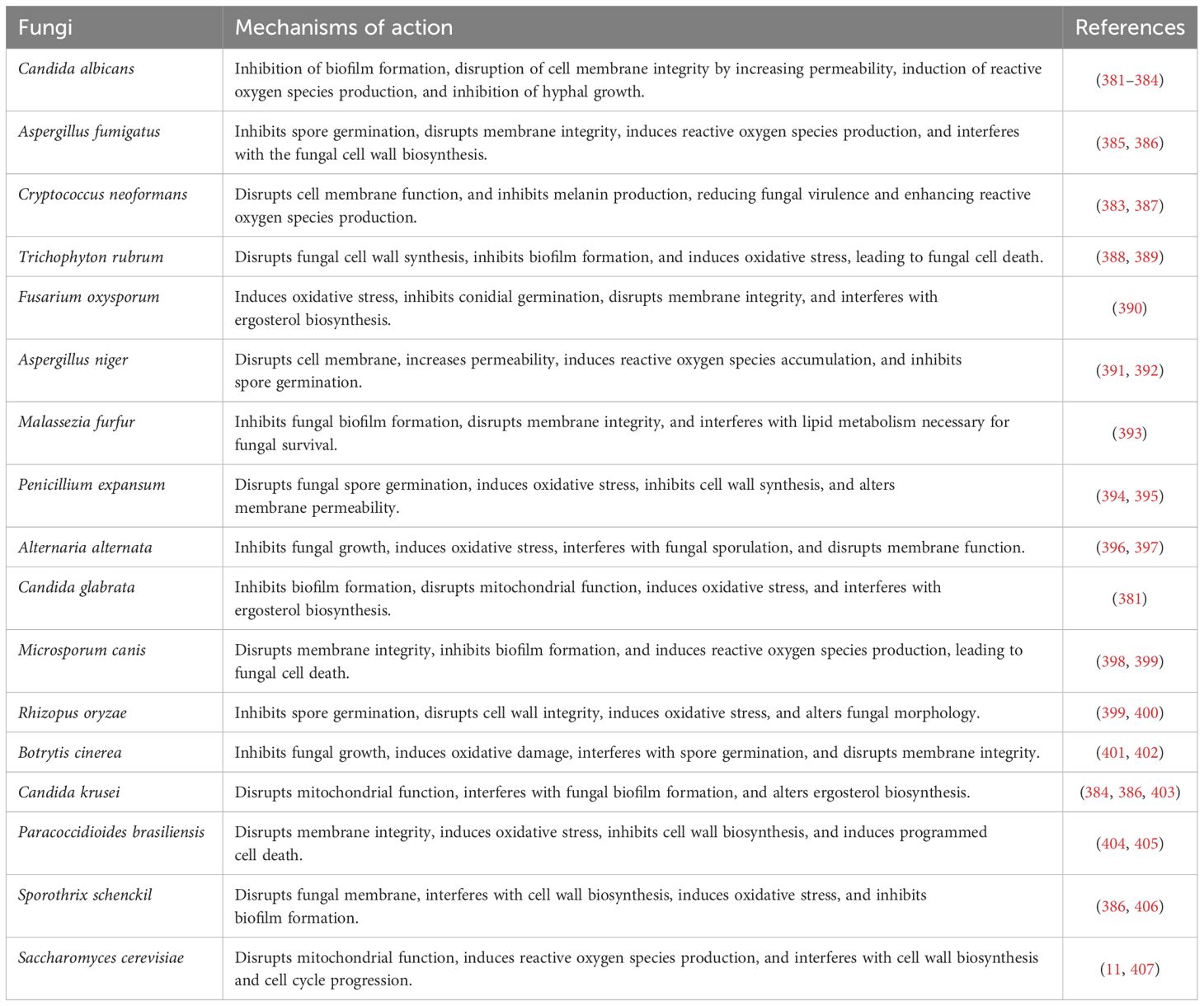
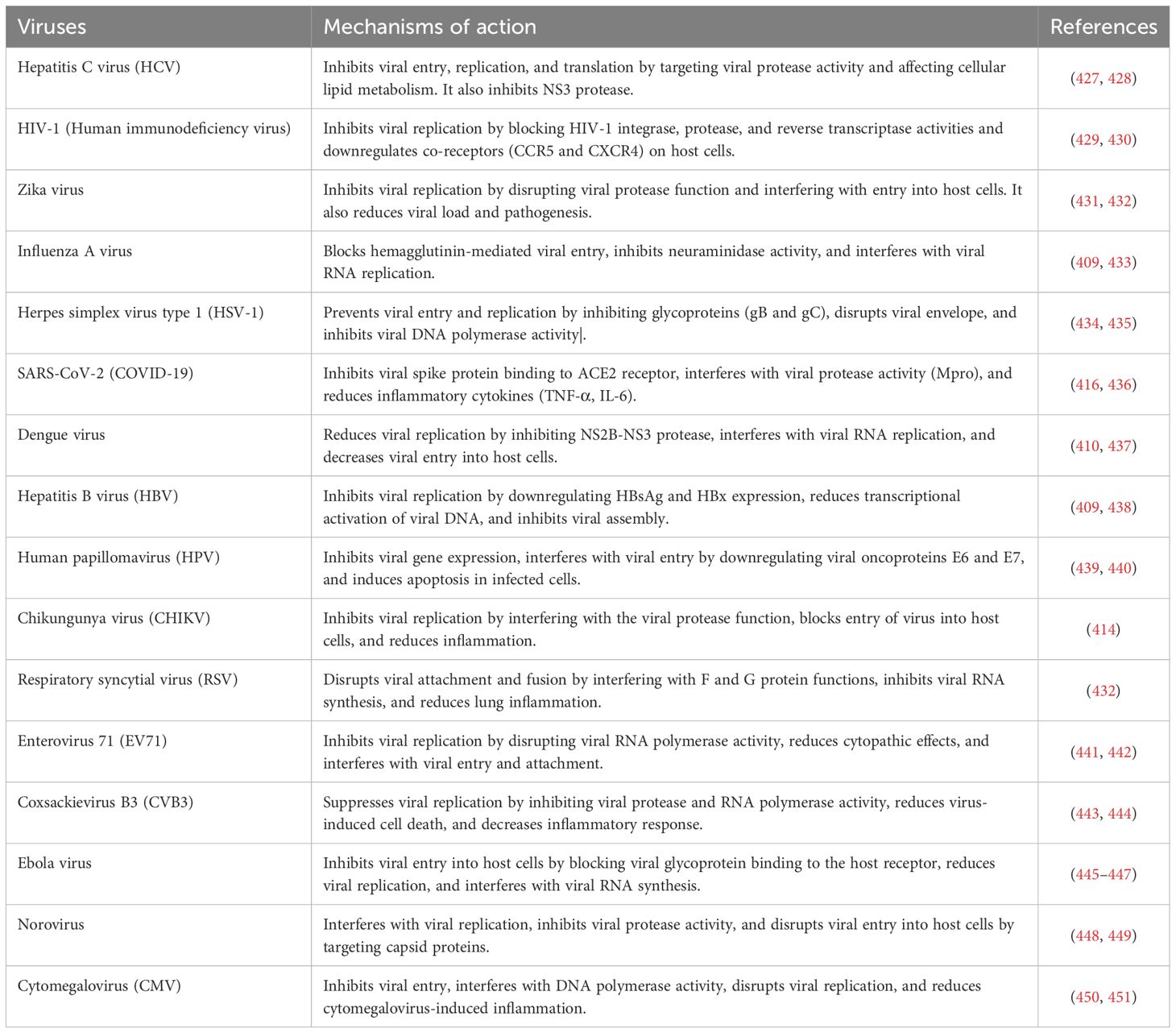
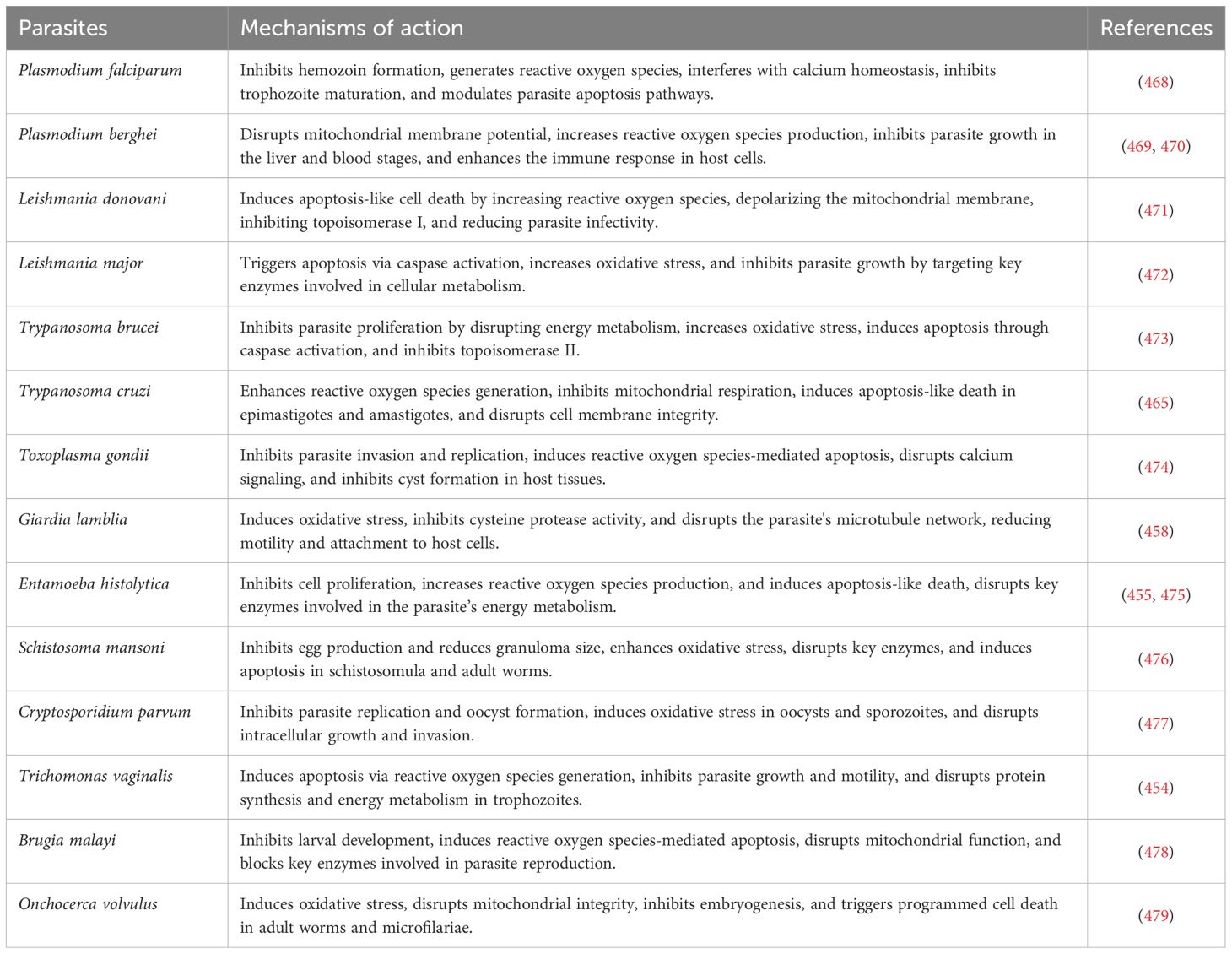
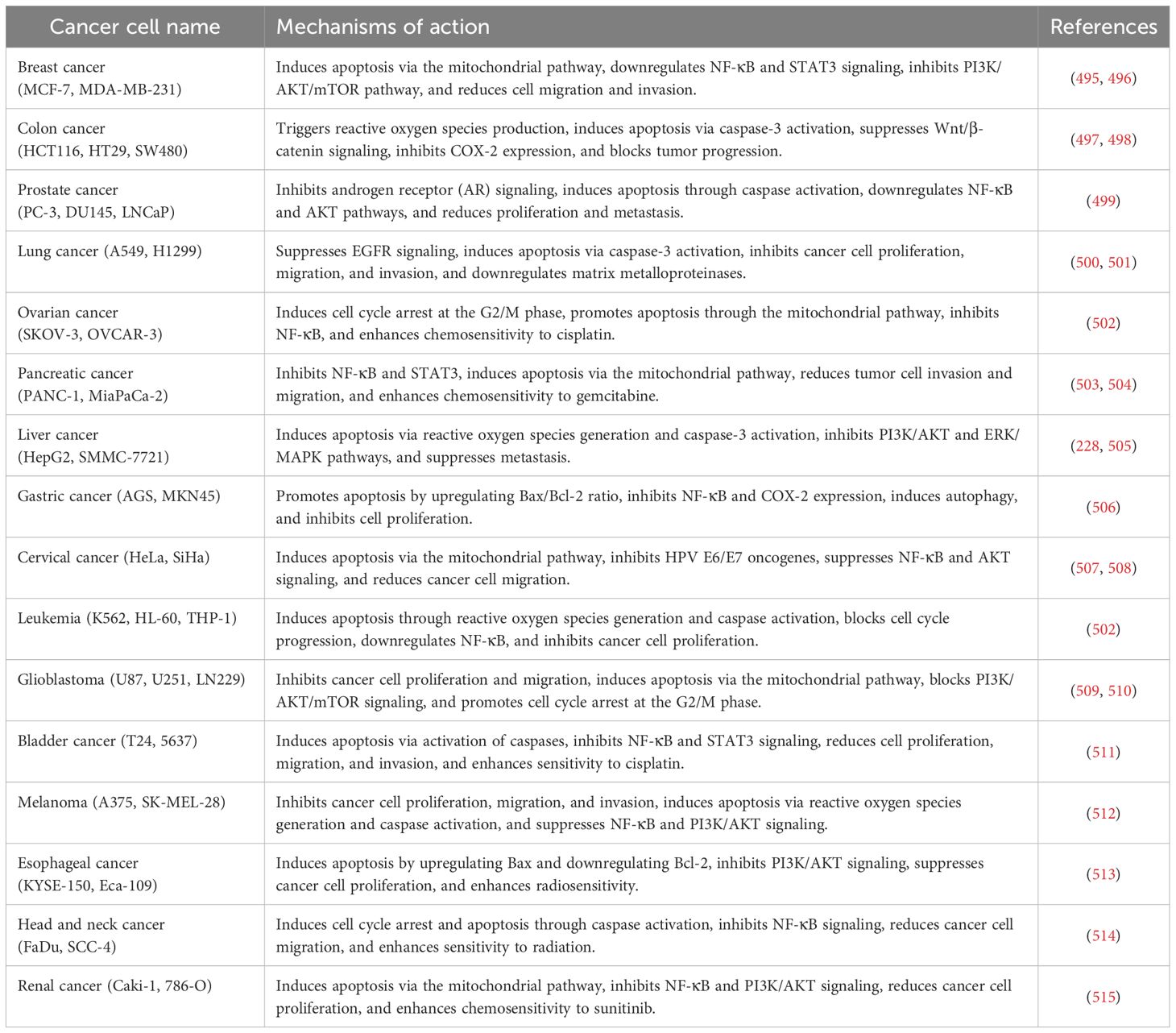
13 Biological activities of curcumin
The therapeutic properties of turmeric are quite diverse, primarily due to the physiological benefits attributed to curcumin (248). In animal studies, oral administration of curcumin has demonstrated significant anti-inflammatory and antiparasitic efficacy (249, 250). Additionally, in vitro studies have shown its anti-inflammatory, antispasmodic, anti-carcinogenic, and gastrointestinal protective properties (248–250).
Table 2 illustrates the biological activity, mechanisms, and therapeutic uses of curcumin and its derivatives.
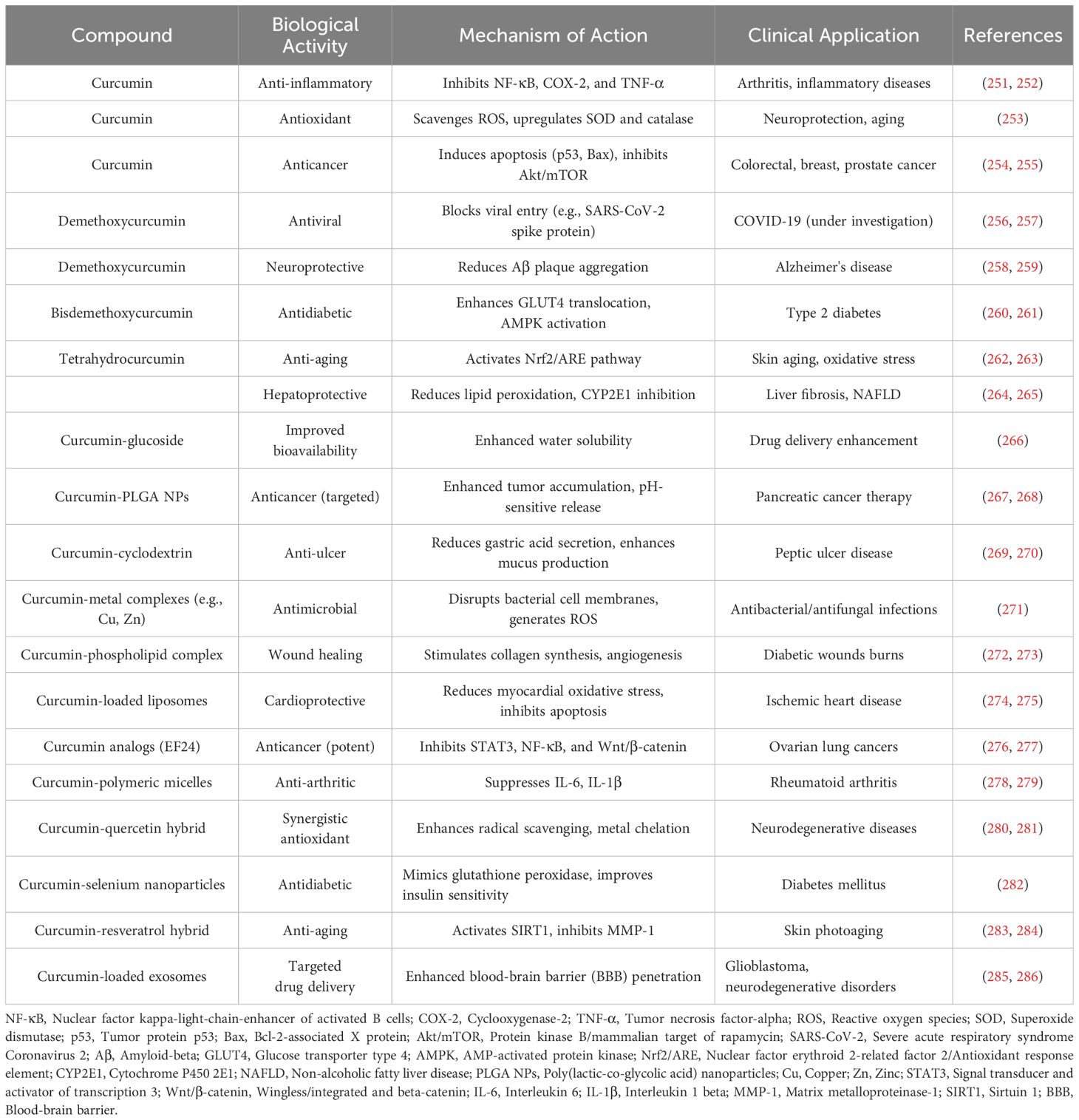
Table 2. Biological activity, mechanisms, and clinical applications of curcumin and its derivatives.
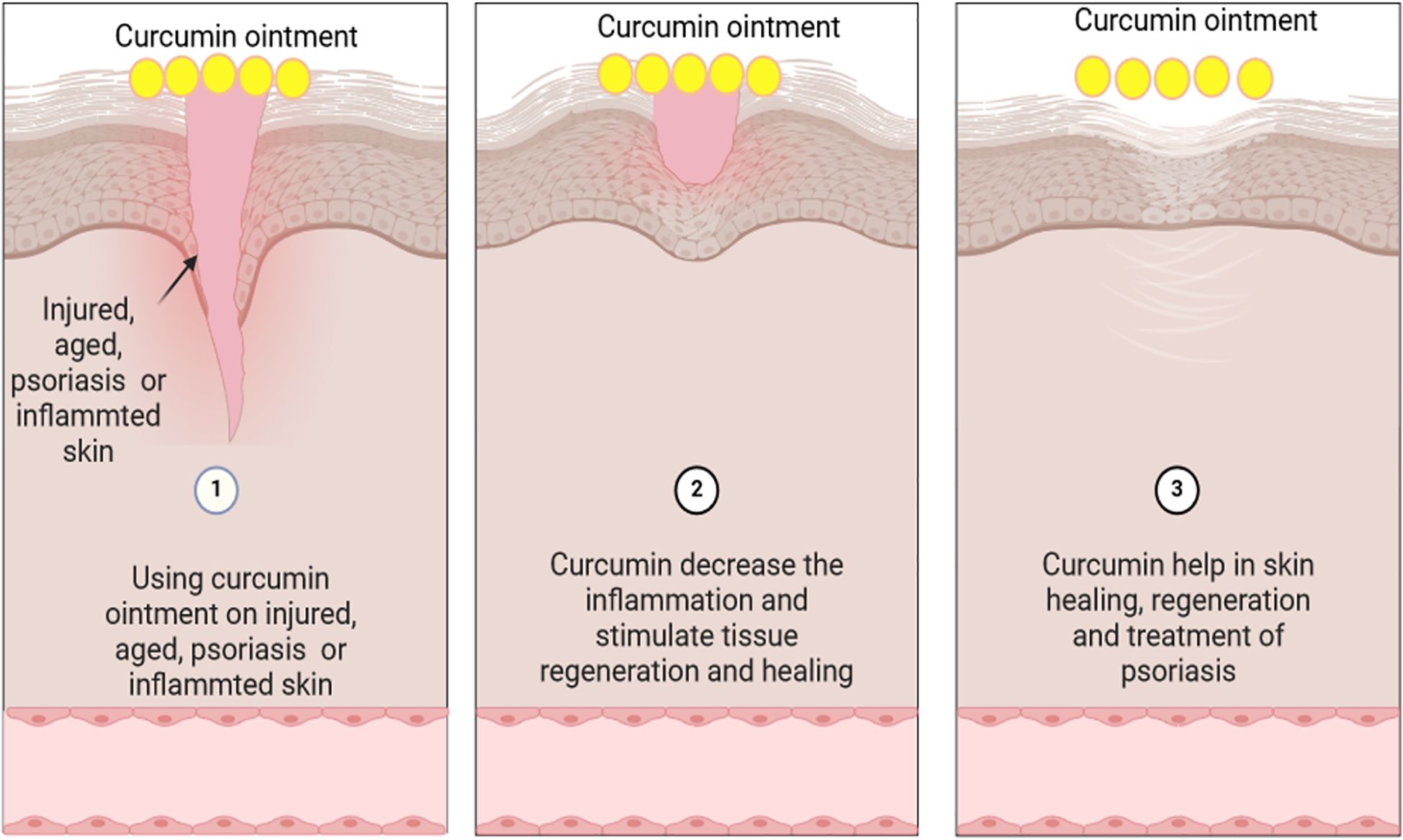
13.1 Wound healing activity of curcumin
Curcumin plays a significant role in enhancing the wound-healing process, which comprises four stages: coagulation, inflammation, proliferation, and remodeling (287). In the initial stage, curcumin promotes programmed cell death of inflammatory cells, hinders the transcription factor NF-κB, decreases cytokine production (tumor necrosis factor-alpha (TNF-α) and interleukin (IL)-1), and lowers reactive oxygen species (ROS) levels (287). These effects enhance antioxidant enzyme synthesis, decrease inflammation, and accelerate the resolution of the inflammatory phase. During the proliferative phase, curcumin promotes collagen deposition, fibroblast migration, granulation tissue development, and re-epithelialization. In the final phase, curcumin boosts TGF-β synthesis, promoting wound contraction and fibroblast development (287).
Various topical formulations, including films, emulsions, fibers, hydrogels, and nano- formulations, have been created to deliver curcumin selectively to wound sites (287–289). Zakerikhoob et al. (288) developed sodium alginate-g-poly (N-isopropyl acrylamide) (Alg-pNIPAM) loaded with curcumin, a thermosensitive hydrogel, for in vivo wound dressing. Studies demonstrated that the Alg-pNIPAM formulation accelerated collagen production, epithelial cell regrowth, and wound contraction. The formulation also exhibited superior anti-inflammatory effects compared to free curcumin solutions. The thermosensitive formulation combines curcumin’s antioxidant and anti-inflammatory properties with the moisture-retaining capability of alginate to expedite the wound healing process (288).
13.2 Anti-inflammatory activity of curcumin
Inflammation plays a pivotal role in the development of many health conditions, including cardiovascular diseases (290), cancer (291), diabetes (292), and neurodegenerative disorders (293). TNF-α is a key mediator in the signal transduction pathways linked to inflammatory diseases, along with other inflammatory mediators. Tak and Firestein (294) identified NF-κB as a potential therapeutic target due to its involvement in these disorders. ROS also contributes significantly to inflammation in various illnesses by modulating transcription factors like NF-κB and activator protein 1 (AP-1) through nuclear histone acetylation and deacetylation (295). Dysregulation of COX-2 and iNOS has been implicated in inflammatory diseases and the pathogenesis of several cancers (296).
Several studies have demonstrated significant anti-inflammatory effects of turmeric, particularly its active component curcumin, through the inhibition of TNF-α (297–299). A systematic review and meta-analysis by Daily et al. (300) reported that curcumin supplementation could effectively decrease inflammatory markers, including C-reactive protein (CRP) and IL-6, in both healthy individuals and those with chronic diseases (300, 301).
Additionally, curcumin has been shown to suppress the production of inflammatory cytokines such as interferon, TNF-α, IL-1, IL-6, and IL-8 (302, 303). A preclinical study by Banik et al. (296) explored curcumin analogs like DM1, which inhibits iNOS and COX2, further highlighting curcumin’s anti-inflammatory potential (304).
Curcumin, often combined with rutin, decreases COX-2 levels and tumor-related inflammation in HPV16-expressing mice (305). In preclinical animal models of invasive pneumonia, curcumin modulates pro- and anti-inflammatory factors (COX-2, IL-6, IL-8, and IL-10), induces apoptosis in polymorphonuclear neutrophilic cells, and mitigates ROS damage (235). Moreover, curcumin has demonstrated efficacy in the treatment of acute lung injury and fatal acute respiratory distress syndrome caused by human coronaviruses in multiple trials (306).
13.3 Immunomodulatory mechanisms and novel applications
Curcumin shifts macrophages from pro-inflammatory (M1) to anti-inflammatory (M2) phenotypes by suppressing TLR4/NF-κB and JAK-STAT pathways, reducing IL-6, TNF-α, and nitric oxide production. This is being explored in rheumatoid arthritis and neuroinflammatory diseases (307, 308). Also, It inhibits Th17 differentiation (reducing IL-17) while promoting regulatory T-cell (Treg) activity via TGF-β1 upregulation. This rebalances immune responses in autoimmune conditions like systemic lupus erythematosus (SLE) and psoriasis (43, 309). Furthermore, curcumin blocks dendritic cell (DC) maturation and antigen presentation, dampening adaptive immune activation. This is leveraged in colitis models to induce intestinal Treg differentiation (310, 311).
Curcumin suppresses NF-κB, MAPK, and JAK/STAT signaling, downregulating pro-inflammatory cytokines (IL-1β, IL-6, TNF-α) while enhancing anti-inflammatory IL-10. It also inhibits ROS-generating enzymes (COX-2, iNOS), mitigating oxidative stress (312–314). Also, It reduces PD-1 expression on exhausted T cells, potentially reversing age-related immune decline and improving vaccine responses (315).
Curcumin also enhances NK cell activity and CD8+ T-cell cytotoxicity against tumors. Derivatives like FLLL32 degrade STAT3, suppressing tumor angiogenesis (VEGF) and metastasis (MMP-2/9) in preclinical osteosarcoma models (316–318).
13.4 Antioxidant activity of curcumin
Oxidative stress arises from an imbalance between endogenous antioxidants and ROS naturally produced in the human body (237). ROS generated during normal cellular processes, such as respiration, include singlet oxygen (¹O2), hydroxyl radicals (HO·), superoxide radicals (O2), and hydrogen peroxide (H2O2). Excess ROS can oxidize biological components, leading to tissue damage (237). The human body uses antioxidant defense systems, including superoxide dismutase (SOD), catalase (CAT), and reduced glutathione to prevent ROS-induced harm (319). Sankar et al. (320) suggested that both free and encapsulated curcumin can indirectly enhance the activity of antioxidant enzymes such as glutathione reductase, SOD, and CAT (320). Curcumin also demonstrates synergistic antioxidant effects when combined with other antioxidants (321).
Kanwal et al. (322) stated that curcumin nanoparticles of different sizes provide increased surface area to expose the functional phenolic groups more effectively, which makes them able to display their free radical scavenging ability effectively. Curcumin exhibits potential biological activities to fight against chronic diseases and can act on several molecular pathways (322). However, the antioxidant feature of curcumin is considered the most important one. Oxidative stress is a result of an imbalance between the elimination and production of ROS and can be related to many chronic diseases and the aging process (323). The properties of curcumin responsible for the removal of reactive nitrogen and oxygen, regulation of different enzymes, and metal chelation are because of the action of curcumin on markers of oxidative stress. This proves that curcumin acts as a potential antioxidant (324).
Curcumin contains various functional groups, and the phenolic functional groups trap electrons to deter the production of H2O2 and scavenge the superoxide radicals, and the β-dike to group produces metal-ligand complexation and carbon-to-carbon double bonds. Thus, the molecule shows some unique antioxidant characteristics (325). Some researchers reported the antioxidant activities of curcumin versus oxidative stress caused by diabetes mellitus. The study was performed on cochlear fibroblasts in rat models of diabetes mellitus, and the authors concluded that an increased expression of superoxide dismutase causes curcumin to confer antioxidant protection (285).
The antiradical power of an antioxidant can be determined with the DPPH method by measuring a decrease in absorbance of DPPH at 515 nm. An antioxidant scavenges the DPPH by donating hydrogen to form a stable DPPH molecule, and the absorbance decreases as a result. The molecules, in the radical form, give an absorbance at 515 nm, which disappears after acceptance of hydrogen or an electron from an antioxidant compound to become a diamagnetic stable molecule (326).
Studies on turmeric leaves further demonstrate the antioxidant properties attributed to its bioactive compounds. Braga et al. (327) highlighted curcumin, total phenolic compounds, and flavonoids as some bioactive substances that contribute to the antioxidant capacity of the leaves. Over the last two decades, many studies have examined the underlying mechanisms of curcumin’s antioxidant properties and its ability to scavenge free radicals, thus reducing cellular oxidative damage (27). Additional tests by Kuncha et al. (328) showed that curcumin can decrease oxidative stress and inflammation in the liver by augmenting antioxidant enzyme activity (328).
Similarly, Jagetia et al. (325) reported that curcumin can prevent stress-induced oxidative damage in the liver, kidneys, and brain of rats. It also shields cells from oxidative damage caused by radiation by reducing ROS production and lipid peroxidation (27). However, despite these studies, the precise antioxidant mechanism of curcumin remains debated. The key point of contention is whether the activity stems from the enolic hydrogen, the phenolic hydrogen, or the central methylenic hydrogen of the heptadiene moiety (329).
Jovanovic et al. (330) demonstrated that curcumin acts as a potent hydrogen atom donor, primarily through the central methylenic group rather than the phenolic group (330). This finding contrasts with the traditional view of curcumin as a phenolic chain-breaking antioxidant donating hydrogen atoms from the phenolic group (331).
Recent studies have explored several curcumin analogs to evaluate their antioxidant efficacy (246, 247). Even with their lack of phenolic hydrogen, these analogs exhibited antioxidant activity comparable to curcumin. Oglah et al. (332) suggested that their ability to donate enolic hydrogen may be a significant contributor to their antioxidant properties (332). Other studies have highlighted the necessity of curcumin’s phenolic hydroxyl group for scavenging free radicals, with the antioxidant effect further enhanced by the addition of a methoxy group to this hydroxyl group (333).
Numerous curcumin derivatives have been synthesized over the last two decades to develop molecules with enhanced antioxidant activity. Shang et al. (334) assessed the antioxidant potential of three series of curcumin derivatives. Compounds containing O-diphenoxyl- and O-dimethoxy-phenoxyl groups exhibited significantly greater antioxidant activity than those without these moieties. Their findings also showed that the antioxidant activity depends on the presence of a seven-carbon spacer; reducing the spacer to five carbons significantly diminished the activity. Additionally, they proposed that lipophilicity, which is enhanced by increasing the amount of carbon atoms, influences antioxidant efficacy (334).
Curcumin has been shown to boost enzymatic antioxidant activity by increasing the levels of enzymes such as SOD, CAT, glutathione peroxidase, and methionine sulfoxide reductase (335). For example, curcumin protects diabetic rats from oxidative stress by upregulating SOD expression in cochlear fibroblasts (336). Comparatively, curcumin demonstrated similar antioxidant activity to ascorbic acid in the DPPH radical scavenging test. At a concentration of 0.1 mM, curcumin achieved a 69% free radical elimination rate, compared to 62% for ascorbic acid, indicating no significant difference between the two antioxidants (337).
Curcumin appears to be especially effective at scavenging smaller oxidative molecules, including H2O2, HO•, and ROO•. Chen et al. (338) demonstrated that curcumin is a potent antioxidant capable of protecting cell cytoplasm from ROS. Stabilized formulations of curcumin with specific carriers have been developed to enhance its stability and antioxidant potential under certain stressful conditions (339, 340).
Curcumin not only neutralizes harmful free radicals but also improves the efficacy of other antioxidants. While these findings are promising, further clinical studies in humans are required to validate the full extent of curcumin’s antioxidant benefits. The antioxidant capabilities of curcumin are demonstrated in Figure 6.
13.5 Antibacterial activity of curcumin
Curcumin’s antibacterial properties were first reported by Schraufstätter and Berntet (341), and over the past 70 years, extensive research has confirmed its broad-spectrum activity against various microorganisms. Curcumin exhibits notable antibacterial effects against Gram-positive and Gram-negative bacteria (342, 343).
The global rise in antibiotic resistance and the associated risk of treatment failures have intensified the search for novel antimicrobial agents (344–346). For instance, Staphylococcus aureus infections, particularly methicillin-resistant S. aureus (MRSA), pose significant challenges in low-resource settings and hospital environments (347). These infections have contributed to increased morbidity and mortality rates over time (348). Recent studies indicate that curcumin shows potent antibacterial activity against both MRSA and methicillin-sensitive S. aureus (MSSA) (349, 350).
Curcumin not only displays intrinsic antibacterial properties but also enhances the efficacy of ampicillin, ciprofloxacin, norfloxacin, gentamicin, and amikacin when used in combination against S. aureus (351). Furthermore, it has been shown to inhibit the growth of various harmful bacteria, including Enterococcus faecalis, Pseudomonas aeruginosa, Escherichia coli, and Klebsiella pneumoniae (352). Its mechanism of action involves disrupting bacterial membranes in both Gram-positive bacteria (S. aureus and E. faecalis) and Gram-negative bacteria (E. coli and P. aeruginosa) (353).
Curcumin exerts its antibacterial effects through several pathways: it inhibits bacterial growth, reduces the production of biofilms and virulence factors, blocks bacterial adhesion to host cells, and suppresses the generation of oxidative compounds (246). Advances in curcumin analogs have further enhanced its antibacterial potential. For example, the curcumin analog CA2, which replaces guaiacol rings with halogenated coumarin rings, demonstrated greater water solubility and stronger antibacterial activity than curcumin against pathogens, such as Haemophilus influenzae, E. coli, P. aeruginosa, and K. pneumonia (332).
Curcumin’s activity extends to light-dependent mechanisms. Under blue light, it acts as a photosensitizer, inducing phototoxicity to inhibit bacterial growth (354). A study by Adamczeck et al. (355) evaluated curcumin’s antibacterial efficacy against over 100 pathogens from 19 species using the broth microdilution method. Results showed that Gram-positive bacteria are generally more susceptible than Gram-negative bacteria. Notably, curcumin displayed potent activity against Streptococcus pyogenes (MIC = 31.25 µg/mL), methicillin-susceptible S. aureus (MIC = 250 µg/mL), Acinetobacter lwoffii (MIC = 250 µg/mL), and certain isolates of P. aeruginosa and E. faecalis ((MIC = 62.5 µg/mL) (355). However, curcumin had limited efficacy against clinical isolates of Candida species (305). Despite its selective activity, curcumin holds promise as a potential antibacterial agent, particularly against resistant bacterial strains. These findings highlight its potential to complement existing treatments and address the growing challenge of antibiotic resistance. The antibacterial properties of curcumin are outlined in Table 3 and Figure 7.
13.6 Antifungal activity of curcumin
In recent decades, fungal infections have significantly increased worldwide, with Candida species being among the most common culprits (376). Among these, Candida albicans stands out as the most virulent and is recognized as a primary fungal pathogen in humans (377, 378). Studies have demonstrated that curcumin not only effectively inhibits the growth of C. albicans isolates but also enhances the efficacy of fluconazole, thereby reducing the pathogen’s resistance to this widely used antifungal agent (379).
In addition, two curcumin derivatives—1,7-bis (3,4,5- trimethoxy phenyl)-1,6-heptadiene-3,5-dione (L1) and 1,7-di(9-anthracenyl)-1,6-heptadiene-3,5-dione (L2) were synthesized and evaluated for their antifungal activity against the genera Aspergillus, Penicillium, and Alternaria. Both derivatives displayed inhibitory effects on the tested cultures, with L1 demonstrating superior antifungal activity compared to L2 (380). These findings suggest curcumin’s potential as a versatile antifungal agent in its natural form or as a chemically modified derivative. The effectiveness of curcumin as an antifungal agent is presented in Table 4.
13.7 Antiviral activity of curcumin
C. longa, particularly its bioactive compound curcumin, has been extensively studied for its potential antiviral properties (311). Research indicates that curcumin can inhibit the replication of various viruses and modulate immune responses. For example, combining curcumin with acyclovir significantly enhances the antiviral effect against the herpes simplex virus compared to acyclovir alone (408). Curcumin’s antiviral mechanisms include suppressing viral replication and targeting critical cellular signaling pathways such as the phosphatidylinositol 3-kinase/protein kinase B (PI3K/Akt and NF-κB (409).
Studies have shown curcumin’s broad-spectrum antiviral efficacy against DNA and RNA viruses (410). For instance, Jeong et al. (411) demonstrated that curcumin pretreatment in fathead minnow cells effectively blocked the early stage of viral hemorrhagic septicemia virus (VHSV) infection. This was attributed to curcumin’s ability to alter the F-actin/G-actin ratio, impeding viral entry into cells (314). In addition, curcumin has shown significant inhibitory effects on herpes simplex virus type 2 (HSV-2) and HIV-1 in vaginal epithelial cells and T lymphocytes, respectively (412).
Curcumin’s impact on various viral diseases is partly due to its inhibition of apurinic/apyrimidinic endonuclease 1 (APE1’s redox function, which influences numerous genes. Li et al. (413) reported that curcumin efficiently blocks the replication of Kaposi’s sarcoma-associated herpesvirus and inhibits angiogenesis and cellular invasion. Moreover, curcumin exhibits antiviral activity against several other viruses, including chikungunya virus, Zika virus, dengue virus, hepatitis C virus, coxsackievirus, human papillomavirus, and SARS-CoV-2 (414, 415).
In the context of COVID-19, curcumin has garnered attention due to its multifaceted therapeutic properties, including its ability to reduce inflammation, modulate the immune system, alleviate pain, and protect lung tissue (416–418). Research suggests that curcumin may interact with Angiotensin 2 (ACE2) or spike proteins involved in the SARS-CoV-2 signaling pathway, thereby disrupting viral processes. Curcumin also inhibits key signaling pathways and transcription factors associated with viral inflammation, such as NF-κB, signal transducer and activator of transcription 3 (STAT-3), Vnt/b-catenin, nuclear factor E2-related factor (Nrf2), and p38/MAPK (419, 420).
A study by Valizadeh et al. (421) demonstrated the potential of nano curcumin in modulating inflammatory responses in COVID-19 patients. Real-time polymerase chain reaction (PCR) and the enzyme-linked immunosorbent assay (ELISA) analyses revealed significant reductions in mRNA levels and cytokine secretion of IL-1β and IL-6, respectively, following nano-curcumin administration (324). These findings highlight curcumin’s potential as a novel therapeutic agent for managing COVID-19 by reducing inflammation and improving immune regulation (421).
Curcumin has shown the potential to enhance the immune response to COVID-19 vaccination (422). Several studies indicated that curcumin supplementation can significantly increase SARS-CoV-2 antibody production in vaccinated individuals (423, 424). It also has dose-dependent immunoediting potential, influencing T-cell production and enhancing the function of effector T-cells while reducing regulatory T-cells (316). Curcumin may prevent critical COVID-19 by blocking ACE2 production (necessary for viral entry) and stimulating anticoagulation and fibrinolysis (425). It has also shown efficacy in preventing severe pneumonia, potentially by acting on the IL-6 trans signal and HM-GB1 system (426).
Further investigation into the specific molecular pathways and cellular targets by which curcumin exerts its immunomodulatory and antiviral actions is necessary. It is also essential to conduct more rigorous and large-scale clinical trials to test curcumin’s safety and efficacy, determine appropriate doses, and assess long-term results for specific immunological disorders and infectious diseases, such as COVID-19, across diverse human populations. The antiviral properties of curcumin are displayed in Table 5.
13.8 Antiparasitic, anti-insects, and antimalarial activity of curcumin
Curcumin demonstrates significant antiparasitic potential, inhibiting the growth of various parasites in the in vitro and in vivo studies. These include Plasmodium falciparum (452), Leishmania major, Leishmania donovani (453), Trichomonas vaginalis (454), Entamoeba histolytica (455), Toxoplasma gondii (456), Neospora caninum (457), and Giardia lamblia (458).
When coupled with artemisinin, curcumin exhibits additive efficacy against P. falciparum and synergistic antiprotozoal activity. In cases involving the highly virulent Plasmodium berghei in mice, curcumin has been shown to improve survival rates (459). The extensive resistance of Plasmodium species to standard antimalarial medicines, such as chloroquine, presents a significant challenge in malaria management (330). However, curcumin remains effective against chloroquine-resistant P. falciparum in vitro and artemisinin-resistant Plasmodium chabaudi in vivo (330).
Martinelli et al. (460) emphasized the potential of curcumin as a foundation for innovative malaria treatments. Furthermore, da Silvaa et al. (461) synthesized curcumin monocarbonyl derivatives, which demonstrated superior efficacy against Trichomonas vaginalis— a causative agent of for trichomoniasis—when compared to metronidazole (461). These derivatives (3a, 3e, and 5e) exhibited enhanced chemical stability and more significant anti-trichomoniasis activity than natural curcumin (332).
Curcumin’s synergistic potential extends to combination with existing drugs. For example, combining metronidazole with curcumin has shown promise against leishmaniasis, while netilmicin paired with curcumin effectively treats amoebiasis (455, 462). In addition, curcumin displays anthelmintic activity against Ascaridia galli and the cestode Raillietina cesticillus (463, 464). In malaria treatment, curcumin not only offers direct therapeutic benefits but also enhances the efficacy of existing antimalarial drugs (465).
Recent advancements highlight the use of curcumin in nanoparticle-based drug delivery. Busari et al. (466) demonstrated that curcumin encapsulated in poly(lactic-co-glycolic) nanoparticles exhibits superior antimalarial activity compared to free curcumin (465). Lower doses of the nanoparticle-based formulation showed enhanced efficacy, with in vivo toxicity studies confirming its safety at the tested levels (466).
Moreover, Novaes et al. (465) explored the role of curcumin as an adjuvant in benznidazole-based chemotherapy for Trypanosoma cruzi infections. Their findings revealed that curcumin enhances the antiparasitic effects of benznidazole while mitigating its side effects, making the combination a promising therapeutic option for Chagas’ disease triggered by T. cruzi.
Moreover, Kausar et al. (467) highlighted the significant clinical implications of curcumin derivatives by demonstrating their potential as novel insecticidal agents. Initial computational studies (molecular docking) investigated how curcumin derivatives bind to the Helicoverpa armigera Sterol Carrier Protein-2 (HaSCP-2). These simulations revealed that the derivatives predominantly form hydrophobic interactions with key residues in the active site: Phe53, Phe110, and Phe89 (467).
To validate these computational findings, laboratory experiments using fluorescence binding and displacement assays were conducted to determine the actual binding affinities of the curcumin derivatives. Among those tested, Cur10 demonstrated the strongest binding, exhibiting the lowest IC50 value of 9.64 μM, while Cur07 and Cur06 showed IC50 values of 19.86 μM and 20.79 μM, respectively (467). Crucially, a strong inverse correlation was observed between the ability of the curcumin derivatives to displace a fluorescent probe from HaSCP-2’s sterol binding site and their capacity to inhibit Sf9 insect cell growth in culture. This finding is clinically important because it supports a novel mechanism of action. These curcumin derivatives likely exert their insecticidal effects by blocking sterol uptake in insects (467).
Since insects, unlike humans and other mammals, cannot synthesize sterols and must obtain them from their diet, inhibiting sterol uptake represents a promising, species-specific insecticidal strategy with potentially low toxicity to non-target organisms. This provides a foundation for developing environmentally friendly and safe pest control alternatives to conventional insecticides (467). This persuasive data underscores curcumin’s significance in antiparasitic and antimalarial therapeutics, facilitating the development of novel treatments aimed at resistant strains and improving the efficacy of current medications. The antiparasitic properties of curcumin are shown in Table 6.
13.9 Anti-diabetic activity of curcumin
Curcumin, known for its inflammatory and blood sugar-regulating properties, shows promise in preventing and managing type 2 diabetes. A study involving 240 individuals with prediabetes over nine months revealed that curcumin supplementation significantly reduced their risk of developing diabetes (480). While research is still ongoing, most findings thus far are based on animal studies rather than human trials (338). Curcumin’s hypoglycemic, hypolipidemic, antioxidative, and anti-inflammatory effects have been extensively documented in animal models, highlighting its therapeutic potential in diabetes management (339).
Curcumin may enhance insulin sensitivity through multiple mechanisms: (1) improved glucose homeostasis: curcumin may increase hepatic glucokinase activity, facilitating better glucose regulation; (2) reduced hypertriglyceridemia: by boosting lipoprotein lipase activity, curcumin can lower very-low-density lipoproteins (VLDL) and triglycerides, addressing lipid imbalances (481); and (3) enhanced glucose uptake: it promotes the expression of glucose transporter-4 (GLUT4), improving peripheral glucose absorption (482). According to Kim et al. (483) the ability of curcumin to reduce glucose levels may be attributed to its ability to decrease the production of hepatic glucose in the liver by inhibiting gluconeogenesis.
Moreover, curcumin has been proposed to mitigate the vascular complications of diabetes, including diabetic retinopathy, cardiomyopathy, and diabetic nephropathy (484, 485). These findings highlight curcumin’s potential as a multifaceted agent in diabetes prevention and treatment.
13.10 Anti-cholesterol activity of curcumin
A study conducted in India demonstrated that curcumin has significant anti-cholesterol properties (346). In the study, ten participants consumed 50 mg of curcumin daily for seven days. This resulted in a 29% increase in high-density lipoprotein (HDL) levels and a 6.11% reduction in blood cholesterol levels. These findings suggested that turmeric may help prevent cardiovascular and vascular disorders in humans (486).
13.11 Anti-cancer activity of curcumin
Carcinogenesis involves a complex series of steps, activating multiple metabolic pathways and mediators (487–490). Key molecules such as proliferative enhancers, cytokines, transcription factors, growth factors, apoptosis inhibitors, and growth factor receptors play crucial roles in cancer development (349, 350). Research indicates that curcumin can target these molecules, influencing cancer progression by regulating cell growth and inducing apoptosis (programmed cell death; PCD) (491). Curcumin has also been found to counteract the carcinogenic effects of tobacco condensates (492).
Curcumin suppresses cancer growth and invasion while promoting apoptosis through various cellular signaling pathways (344). By targeting these pathways, curcumin may inhibit tumor development and angiogenesis (354). Although clinical studies highlight its potential anticancer effects, results vary, with some showing significant benefits while others were inconclusive. This variability may be attributed to curcumin’s limited bioavailability, as it is rapidly metabolized and excreted from the body (493, 494). Current research focuses on developing enhanced formulations and delivery methods to increase therapeutic efficacy. The anti-cancer properties of curcumin are shown in Table 7.
13.11.1 Breast cancer
Breast cancer, the most prevalent malignancy in women, is heavily influenced by estrogen and its receptors (ER-α and ER-β). Approximately two-thirds of breast cancer cases involve overexpression of these receptors, making them critical therapeutic agents (516). Shao et al. (517) revealed that curcumin exhibits potent anti-invasive properties in estrogen-negative MCF-7 breast cancer cell lines. In estrogen-positive lines, curcumin’s antiproliferative effects are estrogen-dependent. These actions involve upregulation of TIMP-1 (tissue inhibitor of metalloproteinase) and MMP-2 (matrix metalloproteinase), both key factors in tumor metastasis (517).
Further research by Calaf et al. (518) demonstrated that curcumin disrupts microtubule formation and cell division checkpoints, inducing apoptosis and inhibiting cell proliferation in MCF-7 cells. When combined with paclitaxell, curcumin enhanced apoptotic activity more effectively than either agent alone (377).
Several analogs have been developed and tested to improve curcumin’s anticancer efficacy. Two promising analogs, PAC and EAC, showed superior blood stability, water solubility, bioavailability, and distribution compared to curcumin. Remarkably, these analogs were five times more effective in inducing apoptosis in breast cancer-causing toxicity (519).
13.11.2 Lung cancer
Lung cancer is among the most lethal types of cancer worldwide, contributing significantly to morbidity and mortality rates (520). Non-small cell lung cancer (NSCLC) represents approximately 85% of all lung cancer cases. Unfortunately, two-thirds of NSCLC cases are diagnosed at advanced stages, making treatment challenging due to drug resistance (521). This highlights the urgent need for innovative adjunctive chemotherapy strategies to enhance current treatments, mitigate side effects, and reduce toxicity without compromising efficacy (380).
Curcumin has emerged as a promising candidate for this purpose. Several studies have demonstrated its ability to inhibit NF-κB activation, a key factor in promoting carcinogen-induced processes such as apoptosis suppression, cellular transformation, invasion, metastasis, chemoresistance, and inflammation (522, 523). A novel curcumin analog, JZ534, has shown enhanced anticancer properties in lung cancer cell lines (524). This compound effectively inhibited cell proliferation, induced PCD, and elevated apoptosis-related proteins such as caspase 3, Bax, and p53. Remarkably, JZ534 exhibited greater anticancer efficacy than curcumin at equivalent doses (410).
13.11.3 Cervical cancer
Curcumin has also shown significant potential against cervical cancer, particularly due to its antimetastatic properties. Research indicates that curcumin inhibits the migration and invasion of cancer cells by suppressing matrix metalloproteinases (MMP-2) and (MMP-9), which are enzymes that facilitate cancer spread by degrading the extracellular matrix (525, 526). In addition, curcumin effectively suppresses telomerase activity, a critical factor in cervical cancer progression, making this mechanism one of its most potent anticancer effects in this context (527).
A novel curcumin analog, EF24, has demonstrated superior anticancer potential. According to Adams et al. (528), EF24 exhibits enhanced bioavailability and robust biological effects compared to curcumin. Furthermore, studies by Tan et al. (529) suggest that EF24 is 10–20 times more effective than curcumin in treating cervical cancer. This improved efficacy indicates the potential of EF24 as a powerful therapeutic option for combating cervical cancer (415).
13.11.4 Prostate cancer
Prostate cancer responds well to anti-androgen treatment when detected early. However, as the disease progresses, cancer cells often develops resistance to hormone deprivation therapy, leading to castration-resistant prostate cancer (CRPC) (530). In a recent clinical trial, more than half of CRPC patients showed a prostate-specific antigen (PSA) response when treated with a combination of curcumin and docetaxel. Remarkably, 88% of responders exhibited a PSA response within the first three treatment cycles (531).
The novel curcumin analogs, RL118 and RL121 on PC3 and DU145 cell lines demonstrated a potent cytotoxic effect on CRPC. These analogs induced apoptosis, suppressed nuclear factor NF-kB activity and arrested cells in the G2/M phase of the cell cycle (532).
13.11.5 Pancreatic cancer
Pancreatic cancer is one of the leading causes of cancer-related deaths globally, responsible for approximately 7% of all such fatalities. Unfortunately, this cancer shows limited responsiveness to radiation and chemotherapy treatments (533). As a result, alternative therapies, including the use of phytochemicals, have been explored. In vitro studies revealed that difluorinated-curcumin (CDF), a curcumin derivative, effectively inhibits the growth and survival of pancreatic cancer cells across various cell lines (534). Another derivative, GO-Y030, exhibited stronger inhibitory effects on pancreatic cell lines than curcumin, likely through the suppression of STAT3 signaling pathways (535).
13.11.6 Colorectal cancer
Colorectal cancer ranks as the fourth most diagnosed malignancy in high-income countries and the fifth in low-income regions. Chemo-preventive strategies have been developed to slow or halt carcinogenesis (536). Since 1995, numerous studies have demonstrated curcumin’s ability to inhibit and reduce the proliferation of colorectal cancer cells (537, 538).
13.12 Anticoagulant activity of curcumin
Turmeric, primarily due to its active compound curcumin, has been extensively studied for its anticoagulant properties. Curcumin inhibits blood clotting by interacting with coagulation factors and platelets. Unlike traditional anticoagulant drugs like warfarin, curcumin demonstrates fewer adverse effects, making it a promising natural alternative (425, 426).
Soni and Salh (539) revealed that curcumin effectively inhibits platelet aggregation and prevents blood clot formation. Similarly, Palathy et al. (540) found that curcumin enhances fibrinolytic factor activity while reducing coagulation factor activity facilitating clot breakdown. These findings suggest that C. longa may possess anticoagulant properties beneficial for individuals at risk of cardiovascular diseases or thrombosis (426).
While curcumin holds potential as a therapeutic anticoagulant, further studies are essential to elucidate its mechanisms and optimize its clinical applications fully.
13.13 Antinociceptive activity of curcumin
Curcumin has shown significant antinociceptive effects in preclinical studies, particularly for neuropathic and inflammatory pain. A study by Zhu et al. (541) demonstrated that curcumin alleviates postoperative pain in rats and accelerates recovery. However, preoperative administration of curcumin did not influence the postoperative pain threshold or recovery rates (542).
Further research evaluated the antinociceptive effects of curcumin encapsulated in PLGA-based curcumin administered intravenously or intrathecally in mice. Intravenous PLGA-based curcumin effectively reduced pain response in formalin and zymosan-induced hyperalgesia models. Intrathecal administration of low doses significantly mitigated allodynia caused by sciatic nerve ligation, while high doses provided prolonged antinociceptive effects (454).
In contrast, pure curcumin administered intrathecally induced only short-term, strong pain relief at high doses. The enhanced efficacy of PLGA-based curcumin is attributed to reduced production of cytokines and brain-derived neurotrophic factor (BDNF) in the spinal cord, as observed in neuropathic pain models (454). This study highlights PLGA-based curcumin potential as an innovative approach to pain management and underscores the therapeutic promise of curcumin nano-formulation (543).
13.14 The anti-Alzheimer’s activity of curcumin
Alzheimer’s disease (AD), a progressive neurodegenerative condition, remains without a definitive cure despite extensive research. Its complex etiology and pathophysiology suggest that the disease arises from multiple factors rather than a single cause (455). A hallmark feature of AD is the aggregation of extracellular amyloid plaques. Additionally, oxidative damage, driven by ROS and biometals like iron, plays a significant role in the disease’s progression (544).
Given the multifaceted nature of AD, there is an urgent need for therapeutic agents capable of targeting multiple pathological pathways (545). Curcumin has emerged as a promising candidate due to its potent anti-inflammatory and antioxidant properties (546). Chen et al. (545) developed a series of curcumin-based compounds and evaluated their potential for treating AD. These compounds demonstrated enhanced inhibitory activity compared to curcumin itself. They also outperformed the reference antioxidant Trolox, exhibiting superior metal-chelating abilities (iron and copper), antioxidant properties, and a capacity to mitigate metal-induced amyloid aggregation (456). Among the tested derivatives, a compound known as A4 showed the most promising results, surpassing other curcumin derivatives in efficacy (456). These findings highlight the potential of A4 as a lead compound for developing multifunctional anti-Alzheimer drugs and highlight the importance of further structural optimization to enhance its therapeutic effects (545).
Moreover, Lan et al. (547) revealed a crucial clinical application of curcumin in mitigating cerebral ischemia-reperfusion (I/R) injury, a significant cause of neuronal damage and neurological dysfunction. The findings indicate that cerebral I/R injury not only causes a specific type of programmed neuronal death called PANoptosis but also triggers microglia to adopt a pro-inflammatory (M1) phenotype, both in living organisms and in vitro. The research demonstrates that pretreatment with curcumin significantly enhanced the proliferative capacity and anti-inflammatory potential of olfactory mucosa-derived mesenchymal stem cells (OM-MSCs). The “curcumin-primed” OM-MSC (CUR-OM-MSC) group exhibited a more pronounced reduction in PANoptotic neuronal death and showed better recovery of neurological function compared to the group treated with OM-MSCs alone (547).
The bioinformatic analysis provided a key mechanistic insight: microRNA-423–5p (miRNA-423–5p) expression was notably upregulated in CUR-OM-MSCs compared to unprimed OM-MSCs (547). This suggests that CUR-OM-MSC treatment induces a beneficial shift of microglia to an anti-inflammatory (M2) phenotype by releasing miRNA-423–5p. This miRNA-423–5p targets nucleotide-binding oligomerization domain 2 (NOD2), which is an upstream regulator of the NF-kappaβ and Mitogen-Activated Protein Kinase (MAPK) signaling pathways. By modulating these pathways, the intervention attenuates PANoptotic neuronal death resulting from cerebral I/R injury (547).
The clinical importance of these findings is substantial. They provide the first demonstration of PANoptotic neuronal death in cerebral I/R conditions and highlight a novel mechanism by which curcumin-primed stem cells can reduce neuroinflammation and improve outcomes (547). This combined approach of curcumin and OM-MSCs offers a promising and potentially efficacious therapeutic strategy for ischemic stroke, addressing multiple facets of injury, including inflammation and specific forms of cell death (547). Curcumin itself has been shown to offer neuroprotection in cerebral ischemia through various mechanisms, including anti-oxidation, anti-inflammation, anti-apoptosis, and protection of the blood-brain barrier, making this combined strategy particularly compelling (547).
14 Other applications of curcumin
14.1 Enhancing skin glow
Numerous physiological and pharmacological processes are carried out by curcumin, known for its physiological and pharmacological properties, including its role in skin health (548). Turmeric extract containing curcuminoids is widely used in topical and oral skincare products. It has been suggested to combat signs of aging skin caused by sun exposure, injuries, increased skin thickness, and reduced elasticity. However, these claims are supported by limited experimental evidence (549).
Sebum, a crucial secretion produced by sebaceous glands, plays a key role in protecting the skin against harmful chemicals and pathogens. Sebum, which constitutes about 95% of skin, includes triglycerides, free fatty acids, waxes, squalene, sterols, and glycophospholipids. By retaining moisture, it enhances the skin’s emollient function and overall resilience (550).
The effect that curcumin has on skin lesions is illustrated in Figure 8.
14.2 Psoriasis treatment
Psoriasis, a chronic inflammatory disease, primarily affects the skin and sometimes the joints, bones, tendons, and nails. The most common form, psoriasis vulgaris, is characterized by oval lesions with white-silver scales, typically found symmetrically on the scalp, lower back, elbows, and knees (551). Recent studies highlight curcumin’s potential in managing psoriasis due to its antioxidant and anti-inflammatory properties (552).
Research suggests curcumin reduces oxidative stress in psoriatic lesions and inhibits elevated phosphorylase kinases, a key factor in psoriasis pathology (553). In vitro studies using doses of 25 and 50 µM showed that curcumin suppressed the growth of psoriatic-like cells (HaCaT cells) by reducing the production of proinflammatory cytokines such as interleukin 17, tumor necrosis factor-α (TNF-α), interferon-γ, and interleukin-6. In addition, curcumin improves skin barrier function by upregulating involucrin (iNV) and filaggrin (FLG) proteins, which are critical for maintaining healthy skin (554).
14.3 Scabies treatment
Traditional Indian medicine systems, including Ayurvedic and Sidha, have long utilized Azadirachta indica (neem) and C. longa (turmeric) for treating chronic ulcers and scabies (465). In a large-scale study, a paste made from neem and turmeric was applied to 814 individuals with scabies (465). Remarkably, 97% of cases were cured within 3–15 days. This method offers cost-effective, accessible, highly effective treatment, particularly for rural communities in resource-limited settings. Furthermore, no adverse reactions or toxicity were observed during the treatment (555).
14.4 Depression management
Curcumin has garnered significant attention in recent years for its potential role in addressing the underlying mechanisms of depression (466). Research suggests that curcumin interacts with various systems implicated in the pathophysiology of major depressive disorder (MDD). It appears to regulate neurotransmitter levels, inflammatory pathways, neuroplasticity, excitotoxicity, and disturbances in the hypothalamus-pituitary-adrenal (HPA) axis (466). In addition, curcumin mitigates oxidative and nitrosative stress, insulin resistance, and imbalances in the endocannabinoid system, making it a promising candidate for managing MDD (556).
14.5 Reducing asthma attacks
As a chronic inflammatory disease of the airways, bronchial asthma remains challenging to treat, with no current therapies altering its progression (467). However, curcumin’s potent anti-inflammatory properties have shown promise in mitigating symptoms. Both in vitro and in vivo studies highlight curcumin’s pharmacological potential to reduce airway inflammation, making it a valuable adjunct in managing bronchial asthma (557).
14.6 Treatment for irritable bowel syndrome
Irritable bowel syndrome (IBS), a prevalent gut-brain axis disorder, is characterized by abdominal pain, discomfort, and altered bowel habits without anatomical abnormalities (480). Despite its global impact, the pathophysiology of IBS remains unclear, and treatment primarily involves supportive therapies (480). Recent studies in animals and humans suggest that curcumin may offer therapeutic benefits for IBS, alleviating symptoms and improving quality of life through its anti-inflammatory and gut-modulating properties (558).
15 Conclusion
This review highlights turmeric’s (C. longa) medicinal potential, primarily due to its active compound, curcumin. While clinical trials confirm curcumin’s safety and efficacy at appropriate dosages for various human and animal diseases, its clinical utility is hindered by poor bioavailability due to limited absorption and rapid metabolism. To overcome these challenges, strategies that combine curcumin with complementary components are being explored, thereby enhancing its therapeutic potential. Emerging research highlights curcumin’s diverse biological activities and its potential as a therapeutic agent for various conditions, provided optimal dosages are achieved. Curcumin demonstrates significant therapeutic promise across a range of conditions, but its poor bioavailability remains a critical challenge. The current review suggests that ongoing efforts to combine curcumin with complementary components are crucial for enhancing its therapeutic potential.
Future research should prioritize the development of novel delivery systems to enhance curcumin’s absorption and stability, as well as further explore synergistic combinations with other compounds. Additionally, more in-depth studies are needed to fully elucidate curcumin’s diverse mechanisms of action, particularly in specific disease contexts. Rigorous and comprehensive clinical trials are essential to establish optimal dosages, assess long-term safety profiles across diverse patient populations, and confirm their efficacy in treating various human diseases. These comprehensive investigations will be crucial for unlocking the full potential of curcumin as an effective and widely applicable therapeutic agent.
Author contributions
ME: Data curation, Formal analysis, Investigation, Writing – original draft, Writing – review & editing. AS: Formal analysis, Software, Visualization, Writing – original draft, Writing – review & editing. DM: Data curation, Investigation, Resources, Writing – original draft, Writing – review & editing. SSA: Data curation, Investigation, Writing – original draft, Writing – review & editing. SG: Data curation, Software, Writing – review & editing. SN: Data curation, Writing – original draft, Writing – review & editing. HS: Investigation, Methodology, Writing – original draft, Writing – review & editing. MF: Formal analysis, Writing – original draft, Writing – review & editing. WM: Data curation, Investigation, Writing – original draft, Writing – review & editing. EI: Conceptualization, Methodology, Writing – review & editing. SFA: Conceptualization, Methodology, Supervision, Writing – original draft, Writing – review & editing. KE: Conceptualization, Formal analysis, Methodology, Resources, Supervision, Writing – original draft, Writing – review & editing.
Funding
The author(s) declare financial support was received for the research and/or publication of this article. This research was funded by the Khalifa Center for Genetic Engineering and Biotechnology, grant number 12R255 to SFA; and UAEU Program for Advanced Research, grant number 12S169 to KE.
Acknowledgments
The authors extend their appreciation to the Deanship of Research and Graduate Studies at King Khalid University for funding this work through Large Research Project under grant number (R. G. P. 1/104/45).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Easmin MS, Sarker MZI, Ferdosh S, Shamsudin SH, Yunus KB, Uddin MS, et al. Bioactive compounds and advanced processing technology: Phaleria macrocarpa (Scheff.) Boerl, a review. J Chem Technol Biotechnol. (2015) 90:981–91. doi: 10.1002/jctb.4603
2. Van Andel T and Carvalheiro LG. Why urban citizens in developing countries use traditional medicines: the case of Suriname. Evid Based Complement Alternat Med. (2013) 2013:687197. doi: 10.1155/2013/687197
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
3. Thomford NE, Senthebane DA, Rowe A, Munro D, Seele P, Maroyi A, et al. Natural products for drug discovery in the 21st century: innovations for novel drug discovery. Int J Mol Sci. (2018) 19:1578. doi: 10.3390/ijms19061578
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
4. Hussain Z, Thu HE, Amjad MW, Hussain F, Ahmed TA, and Khan S. Exploring recent developments to improve antioxidant, anti-inflammatory and antimicrobial efficacy of curcumin: a review of new trends and future perspectives. Mater Sci Eng C. (2017) 77:1316–26. doi: 10.1016/j.msec.2017.03.226
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
5. Alrawaiq NS and Abdullah A. A review of antioxidant polyphenol curcumin and its role in detoxification. Int J Pharm Tech Res. (2014) 6:280–9.
6. Aggarwal BB and Harikumar KB. Potential therapeutic effects of curcumin, the anti-inflammatory agent, against neurodegenerative, cardiovascular, pulmonary, metabolic, autoimmune and neoplastic diseases. Int J Biochem Cell Biol. (2009) 41:40–59. doi: 10.1016/j.biocel.2008.06.010
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
7. Hussain W, Haleem KS, Khan I, Tauseef I, Qayyum S, Ahmed B, et al. Medicinal plants: a repository of antiviral metabolites. Future Virol. (2017) 12:299–308. doi: 10.2217/fvl-2016-0110
8. Krishnaswamy K. Traditional Indian spices and their health significance. Asia Pac J Clin Nutr. (2008) 17:265–8.
9. Hosseini A and Hosseinzadeh H. Antidotal or protective effects of Curcuma longa (turmeric) and its active ingredient, curcumin, against natural and chemical toxicities: a review. BioMed Pharmacother. (2018) 99:411–21. doi: 10.1016/j.biopha.2018.01.072
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
10. Kapoor S and Saraf SL. Topical herbal therapies: an alternative and complementary choice. Res J Med Plant. (2011) 5:650–69. doi: 10.3923/rjmp.2011.650.669
11. Zorofchian Moghadamtousi S, Abdul Kadir H, Hassandarvish P, Tajik H, Abubakar S, and Zandi K. A review on antibacterial, antiviral, and antifungal activity of curcumin. BioMed Res Int. (2014) 2014:186864. doi: 10.1155/2014/186864
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
12. Ody P. Turmeric: Nature’s Miracle Healer: Fact or Fiction. London, UK: Souvenir Press (2018). 224 p.
13. Mathew AG and Pushpanath S. Indian Spices. Kottayam, India: Dee Bee Info Publications (2005). 79 p.
14. Sasikumar B. Genetic resources of Curcuma: diversity, characterization and utilization. Plant Genet Resour. (2005) 3:230–51. doi: 10.1079/PGR200574
15. Den Hartogh DJ, Gabriel A, and Tsiani E. Antidiabetic properties of curcumin II: evidence from in vivo studies. Nutrients. (2019) 12:58. doi: 10.3390/nu12010058
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
16. Hassan F, Rehman MS, Khan MS, Ali MA, Javed A, Nawaz A, et al. Curcumin as an alternative epigenetic modulator: mechanism of action and potential effects. Front Genet. (2019) 10:514. doi: 10.3389/fgene.2019.00514
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
17. Giordano A and Tommonaro G. Curcumin and cancer. Nutrients. (2019) 11:2376. doi: 10.3390/nu11102376
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
18. Heng M. Phosphorylase kinase inhibition therapy in burns and scalds. BioDiscovery. (2017) 20:e11207. doi: 10.3897/biodiscovery.20.e11207
19. Kunnumakkara AB, Bordoloi D, Padmavathi G, Monisha J, Roy NK, Prasad S, et al. Curcumin, the golden nutraceutical: multitargeting for multiple chronic diseases. Br J Pharmacol. (2017) 174:1325–48. doi: 10.1111/bph.13621
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
20. Purpura M, Lowery RP, Wilson JM, Mannan H, Münch G, and Razmovski-Naumovski V. Analysis of different innovative formulations of curcumin for improved relative oral bioavailability in human subjects. Eur J Nutr. (2018) 57:929–38. doi: 10.1007/s00394-016-1376-9
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
21. Jamwal R. Bioavailable curcumin formulations: A review of pharmacokinetic studies in healthy volunteers. J Integr Med. (2018) 16:367–74. doi: 10.1016/j.joim.2018.07.001
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
22. Ma Z, Wang N, He H, and Tang X. Pharmaceutical strategies of improving oral systemic bioavailability of curcumin for clinical application. J Control Release. (2019) 316:359–80. doi: 10.1016/j.jconrel.2019.10.053
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
23. Zheng B and McClements DJ. Formulation of more efficacious curcumin delivery systems using colloid science: enhanced solubility, stability, and bioavailability. Molecules. (2020) 25:2791. doi: 10.3390/molecules25122791
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
24. Prasad S and Aggarwal BB. Turmeric, the golden spice. In: Benzie IFF and Wachtel-Galor S, editors. Herbal Medicine: Biomolecular and Clinical Aspects, 2nd ed. CRC Press, Taylor & Francis, Boca Raton, FL, USA (2011). pp. 263–88.
25. Kocaadam B and Şanlier N. Curcumin, an active component of turmeric (Curcuma longa), and its effects on health. Crit Rev Food Sci Nutr. (2017) 57:2889–95. doi: 10.1080/10408398.2015.1077195
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
26. Almeida HHS, Barros L, Barreira JCM, Calhelha RC, Heleno SA, Sayer C, et al. Bioactive evaluation and application of different formulations of the natural colorant curcumin (E100) in a hydrophilic matrix (yogurt). Food Chem. (2018) 261:224–32. doi: 10.1016/j.foodchem.2018.04.056
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
27. Riaz T, Awais A, Akram M, Rangasamy S, Garcia-Sierra F, Al Hasibuzzaman Md, et al. Phytochemistry and phytochemical potential of Curcuma longa: A narrative review. J Sci Technol Res. (2024) 5:44–56.
28. Kandiannan K, Anandaraj M, Prasath D, Zachariah TJ, Krishnamurthy KS, and Srinivasan V. Evaluation of short and tall true turmeric (Curcuma longa) varieties for growth, yield and stability. Indian J Agric Sci. (2015) 85:718–20. doi: 10.56093/ijas.v85i5.48514
29. Nair RR, Shiva KN, Anchu S, and Zachariah TJ. Characterization of open-pollinated seedling progenies of turmeric (Curcuma longa L.) based on chromosome number, plant morphology, rhizome yield and rhizome quality. Cytologia. (2010) 75:443–9. doi: 10.1508/cytologia.75.443
30. Fuloria S, Mehta J, Chandel A, Sekar M, Rani NNIM, Begum MY, et al. A comprehensive review on the therapeutic potential of Curcuma longa Linn. in relation to its major active constituent curcumin. Front Pharmacol. (2022) 13:820806. doi: 10.3389/fphar.2022.820806
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
31. Razavi BM, Rahbardar MG, and Hosseinzadeh H. A review of therapeutic potentials of turmeric (Curcuma longa) and its active constituent, curcumin, on inflammatory disorders, pain, and their related patents. Phytother Res. (2021) 35:6489–513. doi: 10.1002/ptr.7224
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
32. Vo TS, Vo TTBC, Vo TTTN, and Lai TNH. Turmeric (Curcuma longa L.) chemical components and their effective clinical applications. J Turk Chem Soc A Chem. (2021) 8:883–98. doi: 10.18596/jotcsa.913136
33. Zagórska J, Kukula-Koch W, Czop M, Iłowiecka K, and Koch W. Impact of thermal processing on the composition of Curcuma longa rhizome. Foods. (2023) 12:3086. doi: 10.3390/foods12163086
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
34. Aminnezhad S, Zonobian MA, Moradi Douki M, Mohammadi MR, and Azarakhsh Y. Curcumin and their derivatives with anti-inflammatory, neuroprotective, anticancer, and antimicrobial activities: a review. Micro Nano Bio Asp. (2023) 2:25–34. doi: 10.22034/mnba.2023.417415.1047
35. Sharmeen JB, Mahomoodally FM, Zengin G, and Maggi F. Essential oils as natural sources of fragrance compounds for cosmetics and cosmeceuticals. Molecules. (2021) 26:666. doi: 10.3390/molecules26030666
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
36. AlSaidy HAH, Alhasan DAH, and Ali AA. Reviews on plants gum and mucilage I: The composition and pharmacological significance of turmeric (Curcuma longa) rhizomes polysaccharides and mucilage/their aqueous extracts commercial products. Univ Thi-Qar J. (2021) 8:14–25. doi: 10.32792/utq/utjsci/v8/1/3
37. Guo Q, Huang X, Kang J, Ding H, Liu Y, Wang N, et al. Immunomodulatory and antivirus activities of bioactive polysaccharides and structure-function relationship. Bioact Carbohydr Diet Fiber. (2022) 27:100301. doi: 10.1016/j.bcdf.2021.100301
38. Majumder P, Sinha A, Gupta R, and Sablani SS. Drying of selected major spices: Characteristics and influencing parameters, drying technologies, quality retention and energy saving, and mathematical models. Food Bioproc Tech. (2021) 14:1028–54. doi: 10.1007/s11947-021-02646-7
39. Llano SM, Gómez AM, and Duarte-Correa Y. Effect of drying methods and processing conditions on the quality of Curcuma longa powder. Processes. (2022) 10:702. doi: 10.3390/pr10040702
40. Fathi F, Ebrahimi SN, Matos LC, Oliveira MBPP, and Alves RC. Emerging drying techniques for food safety and quality: A review. Compr Rev Food Sci Food Saf. (2022) 21:1125–60. doi: 10.1111/1541-4337.12898
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
41. Long Y, Zhang M, Mujumdar AS, and Chen J. Valorization of turmeric (Curcuma longa L.) rhizome: Effect of different drying methods on antioxidant capacity and physical properties. Dry Technol. (2022) 40:1609–19. doi: 10.1080/07373937.2022.2032135
42. Belwal T, Cravotto C, Prieto MA, Venskutonis PR, Daglia M, Devkota HP, et al. Effects of different drying techniques on the quality and bioactive compounds of plant-based products: A critical review on current trends. Dry Technol. (2022) 40:1539–61. doi: 10.1080/07373937.2022.2068028
43. Tripathy S, Verma DK, Thakur M, Patel AR, Srivastav PP, Singh S, et al. Curcumin extraction, isolation, quantification and its application in functional foods: a review with a focus on immune enhancement activities and COVID-19. Front Nutr. (2021) 8:747956. doi: 10.3389/fnut.2021.747956
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
44. Hessel V, Tran NN, Asrami MR, Tran QD, Long NVD, Escribà-Gelonch M, et al. Sustainability of green solvents–review and perspective. Green Chem. (2022) 24:410–37. doi: 10.1039/D1GC03662A
45. Socas-Rodríguez B, Torres-Cornejo MV, Álvarez-Rivera G, and Mendiola JA. Deep eutectic solvents for the extraction of bioactive compounds from natural sources and agricultural by-products. Appl Sci. (2021) 11:4897. doi: 10.3390/app11114897
46. Rahman MS, Roy R, Jadhav B, Hossain MN, Halim MA, and Raynie DE. Formulation, structure, and applications of therapeutic and amino acid-based deep eutectic solvents: An overview. J Mol Liq. (2021) 321:114745. doi: 10.1016/j.molliq.2020.114745
47. Bras T, Neves LA, Crespo JG, and Duarte MF. Effect of extraction methodologies and solvent selection upon cynaropicrin extraction from Cynara cardunculus leaves. Sep Purif Technol. (2020) 236:116283. doi: 10.1016/j.seppur.2019.116283
48. Orellana-Paucar AM. Turmeric essential oil constituents as potential drug candidates: A comprehensive overview of their individual bioactivities. Molecules. (2024) 29:4210. doi: 10.3390/molecules29174210
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
49. Vafaeipour Z, Razavi BM, and Hosseinzadeh H. Effects of turmeric (Curcuma longa) and its constituent (curcumin) on the metabolic syndrome: An updated review. J Integr Med. (2022) 20:193–203. doi: 10.1016/j.joim.2022.02.008
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
50. Chopra H, Dey PS, Das D, Bhattacharya T, Shah M, Mubin S, et al. Curcumin nanoparticles as promising therapeutic agents for drug targets. Molecules. (2021) 26:4998. doi: 10.3390/molecules26164998
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
51. Elhawary EA, Moussa AY, and Singab ANB. Genus Curcuma: chemical and ethnopharmacological role in aging process. BMC Complement Med Ther. (2024) 24:31. doi: 10.1186/s12906-023-04317-w
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
52. Bora H, Kamle M, Mahato DK, Tiwari P, and Kumar P. Citrus essential oils (CEOs) and their applications in food: An overview. Plants. (2020) 9:357. doi: 10.3390/plants9030357
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
53. Guimarães AF, Vinhas ACA, Gomes AF, Souza LH, and Krepsky PB. Essential oil of Curcuma longa L. rhizomes: chemical composition, yield variation and stability. Quím Nova. (2020) 43:909–13. doi: 10.21577/0100-4042.20170547
54. Pawar HA, Gavasane AJ, and Choudhary PD. A novel and simple approach for extraction and isolation of curcuminoids from turmeric rhizomes. Nat Prod Chem Res. (2018) 6:1–4. doi: 10.4172/2329-6836.1000300
55. Sahne F, Mohammadi M, Najafpour GD, and Moghadamnia AA. Extraction of bioactive compound curcumin from turmeric (Curcuma longa L.) via different routes: A comparative study. Pak J Biotechnol. (2016) 13:173–80.
56. Monton C, Settharaksa S, Luprasong C, and Songsak T. An optimization approach of dynamic maceration of Centella asiatica to obtain the highest content of four centelloids by response surface methodology. Rev Bras Farmacogn. (2019) 29:254–61. doi: 10.1016/j.bjp.2019.01.001
57. Liang H, Wang W, Xu J, Zhang Q, Shen Z, and Li Q. Optimization of ionic liquid-based microwave-assisted extraction technique for curcuminoids from Curcuma longa L. Food Bioprod Process. (2017) 104:57–65. doi: 10.1016/j.fbp.2017.04.003
58. Sahne F, Mohammadi M, Najafpour GD, and Moghadamnia AA. Enzyme-assisted ionic liquid extraction of bioactive compound from turmeric (Curcuma longa L.): Isolation, purification and analysis of curcumin. Ind Crops Prod. (2017) 95:686–94. doi: 10.1016/j.indcrop.2016.11.037
59. Chao IC, Wang CM, Li SP, Lin LG, Ye WC, and Zhang QW. Simultaneous quantification of three curcuminoids and three volatile components of Curcuma longa using pressurized liquid extraction and high-performance liquid chromatography. Molecules. (2018) 23:1568. doi: 10.3390/molecules23071568
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
60. Nagavekar N and Singhal RS. Supercritical fluid extraction of Curcuma longa and Curcuma amada oleoresin: Optimization of extraction conditions, extract profiling, and comparison of bioactivities. Ind Crops Prod. (2019) 134:134–45. doi: 10.1016/j.indcrop.2019.03.061
61. Patil SS and Rathod VK. Synergistic effect of ultrasound and three phase partitioning for the extraction of curcuminoids from Curcuma longa and its bioactivity profile. Process Biochem. (2020) 93:85–93. doi: 10.1016/j.procbio.2020.02.031
62. Nurjanah N and Saepudin E. Curcumin isolation, synthesis and characterization of curcumin isoxazole derivative compound. AIP Conf Proc. (2019) 2168:020065. doi: 10.1063/1.5132492
63. Muthukumar VP, Vaishnavi M, Theepapriys S, and Saravanaraj A. Process development for the effective extraction of curcumin from Curcuma longa L. (turmeric). Int J Eng Technol. (2018) 7:151–5. doi: 10.14419/ijet.v7i3.34.18926
64. Yadav DK, Sharma K, Dutta A, Kundu A, Awasthi A, Goon A, et al. Purity evaluation of curcuminoids in the turmeric extract obtained by accelerated solvent extraction. J AOAC Int. (2017) 100:586–91. doi: 10.5740/jaoacint.17-0057
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
65. Valizadeh Kiamahalleh M, Najafpour-Darzi G, Rahimnejad M, Moghadamnia AA, and Valizadeh Kiamahalleh M. High performance curcumin subcritical water extraction from turmeric (Curcuma longa L.). J Chromatogr B Analyt Technol BioMed Life Sci. (2016) 1022:191–8. doi: 10.1016/j.jchromb.2016.04.021
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
66. Slaček G, Kotnik P, Osmić A, Postružnik V, Knez Ž, Finšgar M, et al. The extraction process, separation, and identification of curcuminoids from turmeric Curcuma longa. Foods. (2023) 12:4000. doi: 10.3390/foods12214000
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
67. Manasa PSL, Kamble AD, and Chilakamarthi U. Various extraction techniques of curcumin—a comprehensive review. ACS Omega. (2023) 8:34868–78. doi: 10.1021/acsomega.3c04205
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
68. Naksuriya O, van Steenbergen MJ, Torano JS, Okonogi S, and Hennink WE. A kinetic degradation study of curcumin in its free form and loaded in polymeric micelles. AAPS J. (2016) 18:777–87. doi: 10.1208/s12248-015-9863-0
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
69. Liu Y, Li J, Fu R, Zhang L, Wang D, and Wang S. Enhanced extraction of natural pigments from Curcuma longa L. using natural deep eutectic solvents. Ind Crops Prod. (2019) 140:111620. doi: 10.1016/j.indcrop.2019.111620
70. Ahmed M, Ahmad S, and Irfan M. A green ultra-fast liquid chromatographic method for quantification of curcumin in extract of Curcuma longa L. followed by confirmation via spectroscopic techniques. Sep Sci Plus. (2021) 4:118–27. doi: 10.1002/sscp.202170016
71. Nelson KM, Dahlin JL, Bisson J, Graham J, Pauli GF, and Walters MA. The essential medicinal chemistry of curcumin: miniperspective. J Med Chem. (2017) 60:1620–37. doi: 10.1021/acs.jmedchem.6b00975
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
72. Nair DS, Krishnakumar K, and Krishnan B. Pharmacological profile of curcumin: a review. J Bio Innov. (2017) 6:533–41.
73. Rathore S, Mukim M, Sharma P, Devi S, Nagar JC, and Khalid M. Curcumin: a review for health benefits. Int J Res Rev. (2020) 7:273–90.
74. Dei Cas M and Ghidoni R. Dietary curcumin: correlation between bioavailability and health potential. Nutrients. (2019) 11:2147. doi: 10.3390/nu11092147
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
75. Zielińska A, Alves H, Marques V, Durazzo A, Lucarini M, Alves TF, et al. Properties, extraction methods, and delivery systems for curcumin as a natural source of beneficial health effects. Medicina. (2020) 56:336. doi: 10.3390/medicina56070336
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
76. Angelini G, Pasc A, and Gasbarri C. Curcumin in silver nanoparticles aqueous solution: kinetics of keto-enol tautomerism and effects on AgNPs. Colloids Surf A Physicochem Eng Asp. (2020) 603:125235. doi: 10.1016/j.colsurfa.2020.125235
77. Girardon M, Parant S, Monari A, Dehez F, Chipot C, Rogalska E, et al. Triggering tautomerization of curcumin by confinement into liposomes. Chem Photo Chem. (2019) 3:1034–41. doi: 10.1002/cptc.201900159
78. Rege S, Arya M, and Momin S. Mini review on keto-enol ratio of curcuminoid. Ukr Food J. (2019) 7:27–32. doi: 10.24263/2310-1008-2019-7-1-5
79. Shen L and Ji HF. Theoretical study on physicochemical properties of curcumin. Spectrochim Acta A Mol Biomol Spectrosc. (2007) 67:619–23. doi: 10.1016/j.saa.2006.08.018
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
80. Racz LZ, Racz CP, Pop LC, Tomoaia G, Mocanu A, Barbu I, et al. Strategies for improving bioavailability, bioactivity, and physical-chemical behavior of curcumin. Molecules. (2022) 27:6854. doi: 10.3390/molecules27206854
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
81. Grynkiewicz G and Ślifirski P. Curcumin and curcuminoids in quest for medicinal status. Acta Biochim Pol. (2012) 59:201–12. doi: 10.18388/abp.2012_2139
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
82. Schneider C, Gordon ON, Edwards RL, and Luis PB. Degradation of curcumin: from mechanism to biological implications. J Agric Food Chem. (2015) 63:7606–14. doi: 10.1021/acs.jafc.5b00244
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
83. Kim S, Chen J, Cheng T, Gindulyte A, He J, He S, et al. PubChem 2023 update. Nucleic Acids Res Spec Publ. (2023) 51:D1373–80. doi: 10.1093/nar/gkac956
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
84. Manolova Y, Deneva V, Antonov L, Drakalska E, Momekova D, and Lambov N. The effect of the water on the curcumin tautomerism: a quantitative approach. Spectrochim Acta A Mol Biomol Spectrosc. (2014) 132:815–20. doi: 10.1016/j.saa.2014.05.096
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
85. Kawano S, Inohana Y, Hashi Y, and Lin JM. Analysis of keto-enol tautomers of curcumin by liquid chromatography/mass spectrometry. Chin Chem Lett. (2013) 24:685–7. doi: 10.1016/j.cclet.2013.05.006
86. Liu J, Wang H, Wang P, Guo M, Jiang S, Li X, et al. Films based on κ-carrageenan incorporated with curcumin for freshness monitoring. Food Hydrocoll. (2018) 83:134–42. doi: 10.1016/j.foodhyd.2018.05.012
87. Bhandari SV, Kuthe P, Patil SM, Nagras O, and Sarkate AP. A review: exploring synthetic schemes and structure-activity relationship (SAR) studies of mono-carbonyl curcumin analogues for cytotoxicity inhibitory anticancer activity. Curr Org Synth. (2023) 20:821–37. doi: 10.2174/1570179420666230126142238
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
88. Ferrari E, Pignedoli F, Imbriano C, Marverti G, Basile V, Venturi E, et al. Newly synthesized curcumin derivatives: crosstalk between chemico-physical properties and biological activity. J Med Chem. (2011) 54:8066–77. doi: 10.1021/jm200872q
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
89. Vyas A, Dandawate P, Padhye S, Ahmad A, and Sarkar F. Perspectives on new synthetic curcumin analogs and their potential anticancer properties. Curr Pharm Des. (2013) 19:2047–69. doi: 10.2174/138161213805289309
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
90. Liu W, Zhai Y, Heng X, Che FY, Chen W, Sun D, et al. Oral bioavailability of curcumin: problems and advancements. J Drug Targeting. (2016) 24:694–702. doi: 10.3109/1061186X.2016.1157883
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
91. Yang KY, Lin LC, Tseng TY, Wang SC, and Tsai TH. Oral bioavailability of curcumin in rat and the herbal analysis from Curcuma longa by LC–MS/MS. J Chromatogr B. (2007) 853:183–9. doi: 10.1016/j.jchromb.2007.03.010
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
93. Garrett DA, Failla ML, and Sarama RJ. Development of an in vitro digestion method to assess carotenoid bioavailability from meals. J Agric Food Chem. (1999) 47:4301–9. doi: 10.1021/jf9903298
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
94. Guo Z, Liu Y, and Luo Y. Mechanisms of carotenoid intestinal absorption and the regulation of dietary lipids: lipid transporter-mediated transintestinal epithelial pathways. Crit Rev Food Sci Nutr. (2024) 64:1791–816. doi: 10.1080/10408398.2022.2119204
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
95. Jankun J, Wyganowska-Świątkowska M, Dettlaff K, Jelińska A, Surdacka A, Wątróbska-Świetlikowska D, et al. Determining whether curcumin degradation/condensation is actually bioactivation. Int J Mol Med. (2016) 37:1151–8. doi: 10.3892/ijmm.2016.2524
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
96. Huang Y, Deng S, Luo X, Liu Y, Xu W, Pan J, et al. Evaluation of intestinal absorption mechanism and pharmacokinetics of curcumin-loaded galactosylated albumin nanoparticles. Int J Nanomed. (2019) 14:9721–30. doi: 10.2147/IJN.S229992
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
97. Zeng Z, Shen ZL, Zhai S, Xu JL, Liang H, Shen Q, et al. Transport of curcumin derivatives in Caco-2 cell monolayers. Eur J Pharm Biopharm. (2017) 117:123–31. doi: 10.1016/j.ejpb.2017.04.004
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
98. Yu H and Huang Q. Investigation of the absorption mechanism of solubilized curcumin using Caco-2 cell monolayers. J Agric Food Chem. (2011) 59:9120–6. doi: 10.1021/jf201451m
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
99. Xue MCY, Xu L, and Zhang L. Study of the intestinal absorption characteristics of curcumin in vivo and in vitro. J Appl Pharm. (2017) 9:246. doi: 10.21065/1920-4159.1000246
100. Sun Y, Lee CC, Hung WC, Chen FY, Lee MT, and Huang HW. The bound states of amphipathic drugs in lipid bilayers: study of curcumin. Biophys J. (2008) 95:2318–24. doi: 10.1529/biophysj.108.133736
PubMed Abstract | PubMed Abstract | Crossref Full Text | Google Scholar
101. Barry J, Fritz M, Brender JR, Smith PES, Lee DK, and Ramamoorthy A. Determining the effects of lipophilic drugs on membrane structure by solid-state NMR spectroscopy: the case of the antioxidant curcumin. J Am Chem Soc. (2009) 131:4490–8. doi: 10.1021/ja809217u
102. Filippov AV, Kotenkov SA, Munavirov B, and Antzutkin ON. Effect of curcumin on lateral diffusion of phosphatidylcholines in saturated and unsaturated bilayers. Langmuir. (2014) 30:10686–90. doi: 10.1021/la502338c
103. Wahlang B, Pawar YB, and Bansal AK. Identification of permeability-related hurdles in oral delivery of curcumin using the Caco-2 cell model. Eur J Pharm Biopharm. (2011) 77:275–82. doi: 10.1016/j.ejpb.2010.12.006
104. Prasad S, Tyagi AK, and Aggarwal BB. Recent developments in delivery, bioavailability, absorption and metabolism of curcumin: the golden pigment from golden spice. Cancer Res Treat. (2014) 46:2–18. doi: 10.4143/crt.2014.46.1.2
105. Schiborr C, Kocher A, Behnam D, Jandasek J, Toelstede S, and Frank J. The oral bioavailability of curcumin from micronized powder and liquid micelles is significantly increased in healthy humans and differs between sexes. Mol Nutr Food Res. (2014) 58:516–27. doi: 10.1002/mnfr.201300724
106. Holder GM, Plummer JL, and Ryan AJ. The metabolism and excretion of curcumin (1,7-bis-(4-hydroxy-3-methoxyphenyl)-1,6-heptadiene-3,5-dione) in the rat. Xenobiotica. (1978) 8:761–8. doi: 10.3109/00498257809069589
107. Asai A and Miyazawa T. Occurrence of orally administered curcuminoid as glucuronide and glucuronide/sulfate conjugates in rat plasma. Life Sci. (2000) 67:2785–93. doi: 10.1016/S0024-3205(00)00868-7
108. Ireson CR, Jones DJL, Orr S, Coughtrie MWH, Boocock DJ, Williams ML, et al. Metabolism of the cancer chemopreventive agent curcumin in human and rat intestine. Cancer Epidemiol Biomarkers Prev. (2002) 11:105–11.
109. Watkins PB. The barrier function of CYP3A4 and P-glycoprotein in the small bowel. Adv Drug Delivery Rev. (1997) 27:161–70. doi: 10.1016/S0169-409X(97)00041-0
110. Cho YA, Lee W, and Choi JS. Effects of curcumin on the pharmacokinetics of tamoxifen and its active metabolite, 4-hydroxytamoxifen, in rats: possible role of CYP3A4 and P-glycoprotein inhibition by curcumin. Die Pharmazie. (2012) 67:124–30. doi: 10.1691/ph.2012.1099
111. Huang SM, Temple R, Throckmorton DC, and Lesko LJ. Drug interaction studies: study design, data analysis, and implications for dosing and labeling. Clin Pharmacol Ther. (2007) 81:298–304. doi: 10.1038/sj.clpt.6100054
112. Kotha RR and Luthria DL. Curcumin: biological, pharmaceutical, nutraceutical, and analytical aspects. Molecules. (2019) 24:2930. doi: 10.3390/molecules24162930
113. Hassaninasab A, Hashimoto Y, Tomita-Yokotani K, and Kobayashi M. Discovery of the curcumin metabolic pathway involving a unique enzyme in an intestinal microorganism. Proc Natl Acad Sci USA. (2011) 108:6615–20. doi: 10.1073/pnas.1016217108
114. Heger M, van Golen RF, Broekgaarden M, and Michel MC. The molecular basis for the pharmacokinetics and pharmacodynamics of curcumin and its metabolites in relation to cancer. Pharmacol Rev. (2014) 66:222–307. doi: 10.1124/pr.110.004044
115. Ireson C, Orr S, Jones DJL, Verschoyle R, Lim CK, Luo JL, et al. Characterization of metabolites of the chemopreventive agent curcumin in human and rat hepatocytes and in the rat in vivo, and evaluation of their ability to inhibit phorbol ester-induced prostaglandin E2 production. Cancer Res. (2001) 61:1058–64.
116. Aggarwal BB, Deb L, and Prasad S. Curcumin differs from tetrahydrocurcumin for molecular targets, signaling pathways and cellular responses. Molecules. (2014) 20:185–205. doi: 10.3390/molecules20010185
117. Pfeiffer E, Hoehle SI, Walch SG, Riess A, Sólyom AM, and Metzler M. Curcuminoids form reactive glucuronides in vitro. J Agric Food Chem. (2007) 55:538–44. doi: 10.1021/jf0623283
118. Pan MH, Huang TM, and Lin JK. Biotransformation of curcumin through reduction and glucuronidation in mice. Drug Metab Dispos. (1999) 27:486–94. doi: 10.1016/s0090-9556(24)15211-7
119. Vareed SK, Kakarala M, Ruffin MT, Crowell JA, Normolle DP, Djuric Z, et al. Pharmacokinetics of curcumin conjugate metabolites in healthy human subjects. Cancer Epidemiol Biomarkers Prev. (2008) 17:1411–7. doi: 10.1158/1055-9965.EPI-07-2693
120. Sodani K, Patel A, Kathawala RJ, and Chen ZS. Multidrug resistance associated proteins in multidrug resistance. Chin J Cancer. (2012) 31:58–72. doi: 10.5732/cjc.011.10329
121. Berginc K, Trontelj J, Basnet NS, and Kristl A. Physiological barriers to the oral delivery of curcumin. Die Pharmazie. (2012) 67:518–24. doi: 10.1691/ph.2012.1112
122. Gutierres VO, Campos ML, Arcaro CA, Assis RP, Baldan-Cimatti HM, Peccinini RG, et al. Curcumin pharmacokinetic and pharmacodynamic evidences in streptozotocin-diabetic rats support the antidiabetic activity to be via metabolite(s). Evid Based Complement Alternat Med. (2015) 2015:678218. doi: 10.1155/2015/678218
123. Garcea G, Berry DP, Jones DJL, Singh R, Dennison AR, Farmer PB, et al. Consumption of the putative chemopreventive agent curcumin by cancer patients: assessment of curcumin levels in the colorectum and their pharmacodynamic consequences. Cancer Epidemiol Biomarkers Prev. (2005) 14:120–5. doi: 10.1158/1055-9965.120.14.1
124. Ravindranath V and Chandrasekhara N. Absorption and tissue distribution of curcumin in rats. Toxicology. (1980) 16:259–65. doi: 10.1016/0300-483X(80)90122-5
125. Anand P, Kunnumakkara AB, Newman RA, and Aggarwal BB. Bioavailability of curcumin: problems and promises. Mol Pharmaceutics. (2007) 4:807–18. doi: 10.1021/mp700113r
126. Ravindranath V and Chandrasekhara N. Metabolism of curcumin-studies with [3H] curcumin. Toxicology. (1981) 22:337–44. doi: 10.1016/0300-483X(81)90027-5
127. Perkins S, Verschoyle RD, Hill K, Parveen I, Threadgill MD, Sharma RA, et al. Chemopreventive efficacy and pharmacokinetics of curcumin in the min/+ mouse, a model of familial adenomatous polyposis. Cancer Epidemiol Biomarkers Prev. (2002) 11:535–40.
128. Naritomi Y, Sanoh S, and Ohta S. Chimeric mice with humanized liver: application in drug metabolism and pharmacokinetics studies for drug discovery. Drug Metab Pharmacokinet. (2018) 33:31–9. doi: 10.1016/j.dmpk.2017.11.001
129. Manach C, Scalbert A, Morand C, Rémésy C, and Jiménez L. Polyphenols: food sources and bioavailability. Am J Clin Nutr. (2004) 79:727–47. doi: 10.1093/ajcn/79.5.727
130. Volak LP, Ghirmai S, and Cashman JR. Curcuminoids inhibit multiple human cytochromes P450, UDP-glucuronosyltransferase, and sulfotransferase enzymes, whereas piperine is a relatively selective CYP3A4 inhibitor. Drug Metab Dispos. (2008) 36:1594–605. doi: 10.1124/dmd.108.020552
131. Chaachouay N. Synergy, additive effects, and antagonism of drugs with plant bioactive compounds. Drugs Drug Candidates. (2025) 4:4. doi: 10.3390/ddc4010004
132. Alikiaii B, Khatib N, Badpeyma M, Hasanzadeh E, Abbasi S, Amini S, et al. Therapeutic effects of curcumin and piperine combination in critically ill patients with sepsis: a randomized double-blind controlled trial. Trials. (2025) 26:1–13. doi: 10.1186/s13063-025-08916-5
133. Zhuo Y, Zhao YG, and Zhang Y. Enhancing drug solubility, bioavailability, and targeted therapeutic applications through magnetic nanoparticles. Molecules. (2024) 29:4854. doi: 10.3390/molecules29204854
134. Zheng G, Zhang B, Yu H, Song Z, Xu X, Zheng Z, et al. Therapeutic applications and potential biological barriers of nano-delivery systems in common gastrointestinal disorders: a comprehensive review. Adv Compos Hybrid Mater. (2025) 8:1–22. doi: 10.1007/s42114-025-01292-3
135. Karatayli E, Sadiq SC, Schattenberg JM, Grabbe S, Biersack B, and Kaps L. Curcumin and its derivatives in hepatology: therapeutic potential and advances in nanoparticle formulations. Cancers. (2025) 17:484. doi: 10.3390/cancers17030484
136. Tomoaia-Cotisel M, Kun AZ, Rácz CP, Tomoaia G, Mocanu A, Forizs E, et al. Enhanced stability of curcumin and polyethylene glycol composites in the presence of flavonoids and whey protein concentrate: synthesis, structural evaluation and thermal analysis. J Therm Anal Calorim. (2025) 1:1–13. doi: 10.1007/s10973-025-14111-0
137. Dolatabadi S, Karimi M, Nasirizadeh S, Hatamipour M, Golmohammadzadeh S, and Jaafari MR. Preparation, characterization and in vivo pharmacokinetic evaluation of curcuminoids-loaded solid lipid nanoparticles (SLNs) and nanostructured lipid carriers (NLCs). J Drug Delivery Sci Technol. (2021) 62:102352. doi: 10.1016/j.jddst.2021.102352
138. Anjudikkal J, Shukla A, and Pulikkal AK. Effects of micellar media on the bioavailability, solubility, and stability of curcumin. Next Nanotechnol. (2025) 7:100124. doi: 10.1016/j.nxnano.2024.100124
139. Redha AA, Kodikara C, and Cozzolino D. Does encapsulation improve the bioavailability of polyphenols in humans? A concise review based on in vivo human studies. Nutrients. (2024) 16:3625. doi: 10.3390/nu16213625
140. Sedghizadeh N, Taghipoor AD, Ozbıl E, and Pojarani LB. Preparation and evaluation of water-soluble curcumin-cyclodextrin-PVP inclusion complexes. EMU J Pharm Sci. (2024) 7:106–21. doi: 10.54994/emujpharmsci.1597925
141. Low ZX, Teo MYM, Juliana Nordin F, Palanirajan VK, Morak-Młodawska B, Saleem Qazi A, et al. Enhancing the solubility and potency of tetrahydrocurcumin as an anti-cancer agent using a β-cyclodextrin inclusion complex approach. PloS One. (2024) 19:e0305171. doi: 10.1371/journal.pone.0305171
142. Iqubal MK, Md S, Ali J, and Baboota S. Formulation and evaluation of lipid nanogel loaded with quercetin and curcumin for improvement of topical bioavailability. Indian J Pharm Educ Res. (2025) 59:585–601. doi: 10.5530/ijper.20250569
143. Nunes YC, Mendes NM, Pereira de Lima E, Chehadi AC, Lamas CB, Haber JFS, et al. Curcumin: a golden approach to healthy aging: a systematic review of the evidence. Nutrients. (2024) 16:2721. doi: 10.3390/nu16162721
144. Yakubu J and Pandey AV. Innovative delivery systems for curcumin: exploring nanosized and conventional formulations. Pharmaceutics. (2024) 16:637. doi: 10.3390/pharmaceutics16050637
145. Alam MS, Anwar MJ, Maity MK, Azam F, Jaremko M, and Emwas AH. The dynamic role of curcumin in mitigating human illnesses: recent advances in therapeutic applications. Pharmaceuticals. (2024) 17:1674. doi: 10.3390/ph17121674
146. Fu DW, Tian Y, Wang ZH, Li JZ, Zhou DY, Zhu BW, et al. Development of an Antarctic krill oil-based self-microemulsion drug delivery system and its enhancement of bioaccessibility for curcumin. Food Biosci. (2023) 53:102762. doi: 10.1016/j.fbio.2023.102762
147. Abughazala N. Bioavailability-enhancing effects of piperinoids upon curcumin uptake in an experimental functional beverage containing kawakawa and turmeric [Doctoral dissertation]. ResearchSpace@Auckland, New Zealand (2024).
148. Wang H, Zheng C, Tian F, Xiao Z, Sun Z, Lu L, et al. Improving the dissolution rate and bioavailability of curcumin via co-crystallization. Pharmaceuticals. (2024) 17:489. doi: 10.3390/ph17040489
149. Fan S, Zheng W, Ren S, Xiao W, Chen F, and Xue R. Boundary between amorphously and molecularly dispersed curcumin of its amorphous solid dispersions determined by fluorescence spectroscopy. Crystals. (2025) 15:512. doi: 10.3390/cryst15060512
150. Samsoen S, Dudognon E, Le Fer G, Fournier D, Woisel P, and Affouard F. Impact of the polymer dispersity on the properties of curcumin/polyvinylpyrrolidone amorphous solid dispersions. Int J Pharm. (2024) 653:123895. doi: 10.1016/j.ijpharm.2024.123895
151. Zhao J, Jia W, Zhang R, Wang X, and Zhang L. Improving curcumin bioavailability: targeted delivery of curcumin and loading systems in intestinal inflammation. Food Res Int. (2024) 181:115079. doi: 10.1016/j.foodres.2024.115079
152. Zhao B, Yang Z, Li Z, and Niu B. Encapsulation of curcumin in chitosan–phytic acid (CS–PA) capsule: bioavailability, simulated gastrointestinal digestion, and colonic fermentation in vitro. Food Biosci. (2025) 68:106612. doi: 10.1016/j.fbio.2025.106612
153. Ahmed M, Basheer S, Mughram MHA, Iqbal DN, Qamar S, Saeed A, et al. Structural development of curcumin: a natural product arsenal for diverse therapeutic targets—seizing opportunities through serendipity and rational design. J Mol Struct. (2024) 1324:140815. doi: 10.1016/j.molstruc.2024.140815
154. Gonçalves RF, Martins JT, Abrunhosa L, Baixinho J, Matias AA, Vicente AA, et al. Lipid-based nanostructures as a strategy to enhance curcumin bioaccessibility: Behavior under digestion and cytotoxicity assessment. Food Res Int. (2021) 143:110278. doi: 10.1016/j.foodres.2021.110278
155. Pei J, Palanisamy CP, Natarajan PM, Umapathy VR, Roy JR, Srinivasan GP, et al. Curcumin-loaded polymeric nanomaterials as a novel therapeutic strategy for Alzheimer’s disease: A comprehensive review. Ageing Res Rev. (2024) 99:102393. doi: 10.1016/j.arr.2024.102393
156. Alam J, Dilnawaz F, Sahoo SK, Singh DV, Mukhopadhyay AK, Hussain T, et al. Curcumin encapsulated into biocompatible co-polymer PLGA nanoparticle enhanced anti-gastric cancer and anti-Helicobacter pylori effect. Asian Pac J Cancer Prev. (2022) 23:61. doi: 10.31557/APJCP.2022.23.1.61
157. Rezagholizade-Shirvan A, Masrournia M, Fathi Najafi M, and Behmadi H. Synthesis and characterization of nanoparticles based on chitosan-biopolymers systems as nanocarrier agents for curcumin: study on pharmaceutical and environmental applications. Polym Bull. (2023) 80:1495–517. doi: 10.1007/s00289-022-04095-4
158. Asif HM, Zafar F, Ahmad K, Iqbal A, Shaheen G, Ansari KA, et al. Synthesis, characterization and evaluation of anti-arthritic and anti-inflammatory potential of curcumin loaded chitosan nanoparticles. Sci Rep. (2023) 13:10274. doi: 10.1038/s41598-023-37152-7
159. Shahriari M, Kesharwani P, Johnston TP, and Sahebkar A. Anticancer potential of curcumin-cyclodextrin complexes and their pharmacokinetic properties. Int J Pharm. (2023) 631:122474. doi: 10.1016/j.ijpharm.2022.122474
160. Zeng Y, Lv Y, Hu M, Guo F, and Zhang C. Curcumin-loaded hydroxypropyl-β-cyclodextrin inclusion complex with enhanced dissolution and oral bioavailability for epilepsy treatment. Xenobiotica. (2022) 52:718–28. doi: 10.1080/00498254.2022.2136044
161. Chaudhary K and Rajora A. Phytosomes: a critical tool for delivery of herbal drugs for cancer. Phytochem Rev. (2025) 24:165–95. doi: 10.1007/s11101-024-09947-7
162. Singh A, Srivastav S, Singh MP, Singh R, Kumar P, and Kush P. Recent advances in phytosomes for the safe management of cancer. Phytomed Plus. (2024) 5:100540. doi: 10.1016/j.phyplu.2024.100540
163. Cerqueira R, Domingues C, Veiga F, Jarak I, and Figueiras A. Development and characterization of curcumin-loaded TPGS/F127/P123 polymeric micelles as a potential therapy for colorectal cancer. Int J Mol Sci. (2024) 25:7577. doi: 10.3390/ijms25147577
164. Ding Y, Zhao T, Fang J, Song J, Dong H, Liu J, et al. Recent developments in the use of nanocrystals to improve bioavailability of APIs. Wiley Interdiscip Rev Nanomed Nanobiotechnol. (2024) 16:e1958. doi: 10.1002/wnan.1958
165. Jacob S, Kather FS, Boddu SHS, Attimarad M, and Nair AB. Nanosuspension innovations: expanding horizons in drug delivery techniques. Pharmaceutics. (2025) 17:136. doi: 10.3390/pharmaceutics17010136
166. Kaurav M, Gautam L, and Minz S. PEGylated liposomes. In: Saraf S, Sahu RK, and Dave V, editors. Advanced Pharmaceutical and Herbal Nanoscience for Targeted Drug Delivery Systems Part I. Bentham Science Publishers, Sharjah, United Arab Emirates (2022) pp. 133–68. doi: 10.2174/9789815036510122010009
167. Peter S, Khwaza V, Alven S, Naki T, and Aderibigbe BA. PEGylated nanoliposomes encapsulated with anticancer drugs for breast and prostate cancer therapy: an update. Pharmaceutics. (2025) 17:190. doi: 10.3390/pharmaceutics17020190
168. Mahmoudi A, Kesharwani P, Majeed M, Teng Y, and Sahebkar A. Recent advances in nanogold as a promising nanocarrier for curcumin delivery. Colloids Surf B Biointerf. (2022) 215:112481. doi: 10.1016/j.colsurfb.2022.112481
169. Amini SM, Emami T, Rashidi M, and Zarrinnahad H. Curcumin-gold nanoformulation: synthesis, characterizations and biomedical application. Food Biosci. (2024) 57:103446. doi: 10.1016/j.fbio.2023.103446
170. González-Sarrías A, Iglesias-Aguirre CE, Cortés-Martín A, Vallejo F, Cattivelli A, del Pozo-Acebo L, et al. Milk-derived exosomes as nanocarriers to deliver curcumin and resveratrol in breast tissue and enhance their anticancer activity. Int J Mol Sci. (2022) 23:2860. doi: 10.3390/ijms23052860
171. Sable AA, Kunwar A, and Barik A. Alginate and chitosan-based delivery systems for improving the bioavailability and therapeutic efficacy of curcumin. Pharmaceutics. (2024) 16:423. doi: 10.3390/pharmaceutics16030423
172. Raghunath I, Koland M, and Narayanan AV. Piperine: a possible permeation enhancer for oral protein delivery. J Appl Pharm Sci. (2024) 14:035–45. doi: 10.7324/JAPS.2024.157160
173. Cheng W, Xia K, Wu S, and Li Y. Herb-drug interactions and their impact on pharmacokinetics: an update. Curr Drug Metab. (2023) 24:28–69. doi: 10.2174/1389200224666230116113240
174. Nocito MC, De Luca A, Prestia F, Avena P, La Padula D, Zavaglia L, et al. Antitumoral activities of curcumin and recent advances to improve its oral bioavailability. Biomedicines. (2021) 9:1476. doi: 10.3390/biomedicines9101476
175. Sazdova I, Keremidarska-Markova M, Dimitrova D, Mitrokhin V, Kamkin A, Hadzi-Petrushev N, et al. Anticarcinogenic potency of EF24: an overview of its pharmacokinetics, efficacy, mechanism of action, and nanoformulation for drug delivery. Cancers. (2023) 15:5478. doi: 10.3390/cancers15225478
176. Kafali M, Finos MA, and Tsoupras A. Vanillin and its derivatives: a critical review of their anti-inflammatory, anti-infective, wound-healing, neuroprotective, and anti-cancer health-promoting benefits. Nutraceuticals. (2024) 4:522–61. doi: 10.3390/nutraceuticals4040030
177. Ataei M, Roufogalis BD, Majeed M, Shah MA, and Sahebkar A. Curcumin nanofibers: a novel approach to enhance the anticancer potential and bioavailability of curcuminoids. Curr Med Chem. (2023) 30:286–303. doi: 10.2174/0929867329666220322110348
178. Forouzande R, Gharehaghaji AA, and Mohammadi D. Advances in electrospinning techniques for synthesis of nanofibers loaded with herbal extracts and natural ingredients: a comprehensive review. Arch Group. (2025) 5:1–40. doi: 10.53063/synsint.2025.51226
179. Cai J, Guo Y, Liang Y, and He Y. A curcumin sustained-release hydrogel prepared from Pickering emulsions stabilized by modified amorphous calcium phosphate nanoparticles. Colloid Polym Sci. (2024) 302:781–90. doi: 10.1007/s00396-024-05236-z
180. Hamdi NAM, Azmi NA, Sabari NHM, Harun AF, and Haris MS. An insight into the use and advantages of Carbopol in topical mucoadhesive drug delivery system: a systematic review. J Pharm. (2023) 3:53–65. doi: 10.31436/jop.v3i1.156
181. Casula L, Craparo EF, Lai E, Scialabba C, Valenti D, Schlich M, et al. Encapsulation of nanocrystals in mannitol-based inhalable microparticles via spray-drying: a promising strategy for lung delivery of curcumin. Pharmaceuticals. (2024) 17:1708. doi: 10.3390/ph17121708
182. Rawat A, Chourasiya R, Yadav P, Kaushik S, Vishwakarma A, Arya S, et al. Polymeric microspheres for herbal extract encapsulation: advanced formulation techniques, biomedical applications, and future prospects. Curr Appl Polym Sci. (2025) e24522716338115. doi: 10.2174/0124522716338115241226033817
183. Ilyas AH, Deshmuhk R, Majaz Q, Khan GJ, Salim SA, Ahamad S, et al. Formulation and evaluation of clarithromycin and curcumin loaded floating microsphere for sustained release. Biochem Cell Arch. (2024) 24:2313. doi: 10.51470/bca.2024.24.2.2313
184. Abe T, Horisawa Y, Kikuchi O, Ozawa-Umeta H, Kishimoto A, Katsuura Y, et al. Pharmacologic characterization of TBP1901, a prodrug form of aglycone curcumin, and CRISPR-Cas9 screen for therapeutic targets of aglycone curcumin. Eur J Pharmacol. (2022) 935:175321. doi: 10.1016/j.ejphar.2022.175321
185. Kadry MO and Abdel-Megeed RM. CRISPR-Cas9 genome and long non-coding RNAs as a novel diagnostic index for prostate cancer therapy via liposomal-coated compounds. PloS One. (2024) 19:e0302264. doi: 10.1371/journal.pone.0302264
186. Sharifi-Rad J, Rayess YE, Rizk AA, Sadaka C, Zgheib R, Zam W, et al. Turmeric and its major compound curcumin on health: bioactive effects and safety profiles for food, pharmaceutical, biotechnological and medicinal applications. Front Pharmacol. (2020) 11:1021. doi: 10.3389/fphar.2020.01021
187. Tóth G, Roth M, Weckerle M, and Schreier P. Structural elucidation of two novel products from the soybean lipoxygenase-catalyzed dioxygenation of curcumin. Magn Reson Chem. (2000) 38:51–4. doi: 10.1002/(SICI)1097-458X(200001)38:1<51::AID-MRC593>3.0.CO;2-2
188. Priyadarsini KI. Photophysics, photochemistry and photobiology of curcumin: studies from organic solutions, bio-mimetics and living cells. J Photochem Photobiol C. (2009) 10:81–95. doi: 10.1016/j.jphotochemrev.2009.05.001
189. Zaghary W, Hanna E, Zanoun M, Abdallah N, and Sakr T. Curcumin: analysis and stability. J Adv Pharm Res. (2019) 3:47–58. doi: 10.21608/aprh.2019.6191.1069
190. Kotra VSR, Satyabanta L, and Goswami TK. A critical review of analytical methods for determination of curcuminoids in turmeric. J Food Sci Technol. (2019) 56:5153–66. doi: 10.1007/s13197-019-03986-1
191. Kushwaha P, Shukla B, Dwivedi J, and Saxena S. Validated high-performance thin-layer chromatographic analysis of curcumin in the methanolic fraction of Curcuma longa L. rhizomes. Futur J Pharm Sci. (2021) 7:178. doi: 10.1186/s43094-021-00330-3
192. Bhuket PRN, Niwattisaiwong N, Limpikirati P, Khemawoot P, Towiwat P, Ongpipattanakul B, et al. Simultaneous determination of curcumin diethyl disuccinate and its active metabolite curcumin in rat plasma by LC–MS/MS: application of esterase inhibitors in the stabilization of an ester-containing prodrug. J Chromatogr B. (2016) 1033:301–10. doi: 10.1016/j.jchromb.2016.08.039
193. Ramalingam P and Ko YT. A validated LC-MS/MS method for quantitative analysis of curcumin in mouse plasma and brain tissue and its application in pharmacokinetic and brain distribution studies. J Chromatogr B. (2014) 969:101–8. doi: 10.1016/j.jchromb.2014.08.009
194. Ashraf K, Mujeeb M, Ahmad A, Ahmad N, and Amir M. Determination of curcuminoids in Curcuma longa Linn. by UPLC/Q-TOF–MS: an application in turmeric cultivation. J Chromatogr Sci. (2015) 53:1346–52. doi: 10.1093/chromsci/bmv023
195. Van Nong H, Hung LX, Thang PN, Chinh VD, Vu LV, Dung PT, et al. Fabrication and vibration characterization of curcumin extracted from turmeric (Curcuma longa) rhizomes of the northern Vietnam. Springerplus. (2016) 5:1147. doi: 10.1186/s40064-016-2812-2
196. Sathisaran I and Dalvi SV. Crystal engineering of curcumin with salicylic acid and hydroxyquinol as coformers. Cryst Growth Des. (2017) 17:3974–88. doi: 10.1021/acs.cgd.7b00599
197. Ali Z, Saleem M, Atta BM, Khan SS, and Hammad G. Determination of curcuminoid content in turmeric using fluorescence spectroscopy. Spectrochim Acta A Mol Biomol Spectrosc. (2019) 213:192–8. doi: 10.1016/j.saa.2019.01.028
198. Pandey KU and Dalvi SV. Understanding stability relationships among three curcumin polymorphs. Adv Powder Technol. (2019) 30:266–76. doi: 10.1016/j.apt.2018.11.002
199. Iravani S and Soufi GJ. Electron paramagnetic resonance (EPR) spectroscopy: Food, biomedical and pharmaceutical analysis. BioMed Spectrosc Imaging. (2020) 9:165–82. doi: 10.3233/BSI-200206
200. Dudylina AL, Ivanova MV, Shumaev KB, and Ruuge EK. Superoxide formation in cardiac mitochondria and effect of phenolic antioxidants. Cell Biochem Biophys. (2019) 77:99–107. doi: 10.1007/s12013-018-0857-2
201. Morales NP, Sirijaroonwong S, Yamanont P, and Phisalaphong C. Electron paramagnetic resonance study of the free radical scavenging capacity of curcumin and its demethoxy and hydrogenated derivatives. Biol Pharm Bull. (2015) 38:1478–83. doi: 10.1248/bpb.b15-00209
202. Nikolic I, Mitsou E, Damjanovic A, Papadimitriou V, Antic-Stankovic J, Stanojevic B, et al. Curcumin-loaded low-energy nanoemulsions: Linking EPR spectroscopy-analyzed microstructure and antioxidant potential with in vitro evaluated biological activity. J Mol Liq. (2020) 301:112479. doi: 10.1016/j.molliq.2020.112479
203. Gopi S, Jacob J, Varma K, Jude S, Amalraj A, Arundhathy C, et al. Comparative oral absorption of curcumin in a natural turmeric matrix with two other curcumin formulations: An open-label parallel-arm study. Phytother Res. (2017) 31:1883–91. doi: 10.1002/ptr.5931
204. Jäger R, Lowery RP, Calvanese AV, Joy JM, Purpura M, and Wilson JM. Comparative absorption of curcumin formulations. Nutr J. (2014) 13:11. doi: 10.1186/1475-2891-13-11
205. Carvalho Henriques M, Faustino MAF, and Santos Braga S. Curcumin innovative delivery forms: Paving the ‘Yellow Brick Road’ of antitumoral phytotherapy. Appl Sci. (2020) 10:8990. doi: 10.3390/app10248990
206. Stohs SJ, Chen O, Ray SD, Ji J, Bucci LR, and Preuss HG. Highly bioavailable forms of curcumin and promising avenues for curcumin-based research and application: a review. Molecules. (2020) 25:1397. doi: 10.3390/molecules25061397
207. Kasapoglu-Calik M and Ozdemir M. Synthesis and controlled release of curcumin-β-cyclodextrin inclusion complex from nanocomposite poly (N-isopropylacrylamide/sodium alginate) hydrogels. J Appl Polym Sci. (2019) 136:47554. doi: 10.1002/app.47554
208. Kongkaneramit L, Aiemsum-ang P, and Kewsuwan P. Development of curcumin liposome formulations using polyol dilution method. Songklanakarin J Sci Technol. (2016) 38:605–10.
209. Tai K, Rappolt M, Mao L, Gao Y, Li X, and Yuan F. The stabilization and release performances of curcumin-loaded liposomes coated by high and low molecular weight chitosan. Food Hydrocoll. (2020) 99:105355. doi: 10.1016/j.foodhyd.2019.105355
210. Cuomo F, Cofelice M, Venditti F, Ceglie A, Miguel M, Lindman B, et al. In vitro digestion of curcumin loaded chitosan-coated liposomes. Colloids Surf B Biointerf. (2018) 168:29–34. doi: 10.1016/j.colsurfb.2017.11.047
211. Algahtani MS, Ahmad MZ, and Ahmad J. Nanoemulsion loaded polymeric hydrogel for topical delivery of curcumin in psoriasis. J Drug Delivery Sci Technol. (2020) 59:101847. doi: 10.1016/j.jddst.2020.101847
212. Guerrero S, Inostroza-Riquelme M, Contreras-Orellana P, Diaz-Garcia V, Lara P, Vivanco-Palma A, et al. Curcumin-loaded nanoemulsion: A new safe and effective formulation to prevent tumor reincidence and metastasis. Nanoscale. (2018) 10:22612–22. doi: 10.1039/C8NR06173D
213. Cheng YH, Ko YC, Chang YF, Huang SH, and Liu CJL. Thermosensitive chitosan-gelatin-based hydrogel containing curcumin-loaded nanoparticles and latanoprost as a dual-drug delivery system for glaucoma treatment. Exp Eye Res. (2019) 179:179–87. doi: 10.1016/j.exer.2018.11.017
214. Gera M, Sharma N, Ghosh M, Lee SJ, Min T, Kwon T, et al. Nanoformulations of curcumin: an emerging paradigm for improved remedial application. Oncotarget. (2017) 8:66680. doi: 10.18632/oncotarget.19164
215. Saber-Moghaddam N, Salari S, Hejazi S, Amini M, Taherzadeh Z, Eslami S, et al. Oral nano-curcumin formulation efficacy in management of mild to moderate hospitalized coronavirus disease-19 patients: An open label nonrandomized clinical trial. Phytother Res. (2021) 35:2616–23. doi: 10.1002/ptr.7004
216. Liu Y, Huang P, Hou X, Yan F, Jiang Z, Shi J, et al. Hybrid curcumin–phospholipid complex-near-infrared dye oral drug delivery system to inhibit lung metastasis of breast cancer. Int J Nanomed. (2019) 14:3311–30. doi: 10.2147/IJN.S200847
217. Wang J, Wang L, Zhang L, He D, Ju J, and Li W. Studies on the curcumin phospholipid complex solidified with Soluplus®. J Pharm Pharmacol. (2018) 70:242–9. doi: 10.1111/jphp.12857
218. Gupta A, Costa AP, Xu X, Lee SL, Cruz CN, Bao Q, et al. Formulation and characterization of curcumin loaded polymeric micelles produced via continuous processing. Int J Pharm. (2020) 583:119340. doi: 10.1016/j.ijpharm.2020.119340
219. Liu K, Huang RL, Zha XQ, Li QM, Pan LH, and Luo JP. Encapsulation and sustained release of curcumin by a composite hydrogel of lotus root amylopectin and chitosan. Carbohydr Polym. (2020) 232:115810. doi: 10.1016/j.carbpol.2019.115810
220. Gunathilake TMSU, Ching YC, Chuah CH, Illias HA, Ching KY, Singh R, et al. Influence of a nonionic surfactant on curcumin delivery of nanocellulose reinforced chitosan hydrogel. Int J Biol Macromol. (2018) 118:1055–64. doi: 10.1016/j.ijbiomac.2018.06.147
221. Pushpalatha R, Selvamuthukumar S, and Kilimozhi D. Cyclodextrin nanosponge based hydrogel for the transdermal co-delivery of curcumin and resveratrol: Development, optimization, in vitro and ex vivo evaluation. J Drug Delivery Sci Technol. (2019) 52:55–64. doi: 10.1016/j.jddst.2019.04.025
222. Shefa AA, Sultana T, Park MK, Lee SY, Gwon JG, and Lee BT. Curcumin incorporation into an oxidized cellulose nanofiber-polyvinyl alcohol hydrogel system promotes wound healing. Mater Des. (2020) 186:108313. doi: 10.1016/j.matdes.2019.108313
223. Sahin K, Orhan C, Er B, Durmus AS, Ozercan IH, Sahin N, et al. Protective effect of a novel highly bioavailable formulation of curcumin in experimentally induced osteoarthritis rat model. Curr Dev Nutr. (2020) 4:nzaa066_020. doi: 10.1093/cdn/nzaa066_020
224. Panda SK, Parachur VA, Mohanty N, Swain T, and Sahu S. A comparative pharmacokinetic evaluation of a bioavailable curcumin formulation Curene® with curcumin formulation containing turmeric volatile oil and standard curcuminoids 95% in healthy human subjects. Funct Food Health Dis. (2019) 9:134–44. doi: 10.31989/ffhd.v9i2.548
225. Ullah F, Asgarov R, Venigalla M, Liang H, Niedermayer G, Münch G, et al. Effects of a solid lipid curcumin particle formulation on chronic activation of microglia and astroglia in the GFAP-IL6 mouse model. Sci Rep. (2020) 10:2365. doi: 10.1038/s41598-020-58838-2
226. de Melo ISV, Dos Santos AF, and Bueno NB. Curcumin or combined curcuminoids are effective in lowering the fasting blood glucose concentrations of individuals with dysglycemia: systematic review and meta-analysis of randomized controlled trials. Pharmacol Res. (2018) 128:137–44. doi: 10.1016/j.phrs.2017.09.010
227. Dehzad MJ, Ghalandari H, Nouri M, and Askarpour M. Antioxidant and anti-inflammatory effects of curcumin/turmeric supplementation in adults: A GRADE-assessed systematic review and dose–response meta-analysis of randomized controlled trials. Cytokine. (2023) 164:156144. doi: 10.1016/j.cyto.2023.156144
228. Meybodi SM, Rezaei P, Faraji N, Jamehbozorg K, Ashna S, Shokri F, et al. Curcumin and its novel formulations for the treatment of hepatocellular carcinoma: new trends and future perspectives in cancer therapy. J Funct Foods. (2023) 108:105705. doi: 10.1016/j.jff.2023.105705
229. Peng Y, Ao M, Dong B, Jiang Y, Yu L, Chen Z, et al. Anti-inflammatory effects of curcumin in the inflammatory diseases: status, limitations and countermeasures. Drug Des Devel Ther. (2021) 15:4503–25. doi: 10.2147/DDDT.S327378
230. Sun H, Liu T, Wang Z, Shen W, Yuan X, Xie J, et al. Role of curcumin in chronic liver diseases: A comprehensive review. Drug Des Devel Ther. (2025) 19:3395–406. doi: 10.2147/DDDT.S518547
231. Hassanizadeh S, Shojaei M, Bagherniya M, Orekhov AN, and Sahebkar A. Effect of nano-curcumin on various diseases: a comprehensive review of clinical trials. Biofactors. (2023) 49:512–33. doi: 10.1002/biof.1932
232. Ahmed M, Qadir MA, Shafiq MI, Muddassar M, Hameed A, Arshad MN, et al. Curcumin: Synthesis optimization and in silico interaction with cyclin dependent kinase. Acta Pharm. (2017) 67:385–95. doi: 10.1515/acph-2017-0023
233. De Waure C, Bertola C, Baccarini G, Chiavarini M, and Mancuso C. Exploring the contribution of curcumin to cancer therapy: A systematic review of randomized controlled trials. Pharmaceutics. (2023) 15:1275. doi: 10.3390/pharmaceutics15041275
234. Farzaei MH, Zobeiri M, Parvizi F, El-Senduny FF, Marmouzi I, Coy-Barrera E, et al. Curcumin in liver diseases: a systematic review of the cellular mechanisms of oxidative stress and clinical perspective. Nutrients. (2018) 10:855. doi: 10.3390/nu10070855
235. Marton LT, Pescinini-e-Salzedas LM, Camargo MEC, Barbalho SM, Haber JFS, Sinatora RV, et al. Cincotto dos Santos Bueno P. The effects of curcumin on Diabetes mellitus: a systematic review. Front Endocrinol (Lausanne). (2021) 12:669448. doi: 10.3389/fendo.2021.669448
236. White CM and Lee JY. The impact of turmeric or its curcumin extract on nonalcoholic fatty liver disease: a systematic review of clinical trials. Pharm Pract (Granada). (2019) 17:1350. doi: 10.18549/pharmpract.2019.1.1350
237. Soleimani V, Sahebkar A, and Hosseinzadeh H. Turmeric (Curcuma longa) and its major constituent (curcumin) as nontoxic and safe substances. Phytother Res. (2018) 32:985–95. doi: 10.1002/ptr.6054
238. Pandey A, Chaturvedi M, Mishra S, Kumar P, Somvanshi P, and Chaturvedi R. Reductive metabolites of curcumin and their therapeutic effects. Heliyon. (2020) 6:e05469. doi: 10.1016/j.heliyon.2020.e05469
239. Huang Y, Cao S, Zhang Q, Zhang H, Fan Y, Qiu F, et al. Biological and pharmacological effects of hexahydrocurcumin, a metabolite of curcumin. Arch Biochem Biophys. (2018) 646:31–7. doi: 10.1016/j.abb.2018.03.030
240. Greil R, Greil-Ressler S, Weiss L, Schönlieb C, Magnes T, Radl B, et al. A phase 1 dose-escalation study on the safety, tolerability and activity of liposomal curcumin (Lipocurc™) in patients with locally advanced or metastatic cancer. Cancer Chemother Pharmacol. (2018) 82:695–706. doi: 10.1007/s00280-018-3654-0
241. Saghatelyan T, Tananyan A, Janoyan N, Tadevosyan A, Petrosyan H, Hovhannisyan A, et al. Efficacy and safety of curcumin in combination with paclitaxel in patients with advanced, metastatic breast cancer: A comparative, randomized, double-blind, placebo-controlled clinical trial. Phytomedicine. (2020) 70:153218. doi: 10.1016/j.phymed.2020.153218
242. Hussain Y, Abdullah, Khan F, Alsharif KF, Alzahrani KJ, Saso L, et al. Regulatory effects of curcumin on platelets: an update and future directions. Biomedicines. (2022) 10:3180. doi: 10.3390/biomedicines10123180
243. Cacciola NA, Cuciniello R, Petillo GD, Piccioni M, Filosa S, and Crispi S. An overview of the enhanced effects of curcumin and chemotherapeutic agents in combined cancer treatments. Int J Mol Sci. (2023) 24:12587. doi: 10.3390/ijms241612587
244. Koss-Mikołajczyk I, Todorovic V, Sobajic S, Mahajna J, Gerić M, Tur JA, et al. Natural products counteracting cardiotoxicity during cancer chemotherapy: the special case of doxorubicin, a comprehensive review. Int J Mol Sci. (2021) 22:10037. doi: 10.3390/ijms221810037
245. Chen XW, Sneed KB, Pan SY, Cao C, Kanwar JR, Chew H, et al. Herb-drug interactions and mechanistic and clinical considerations. Curr Drug Metab. (2012) 13:640–51. doi: 10.2174/1389200211209050640
246. Chan K. An overview on safety issues of interactions between traditional herbal medicines and pharmaceutical medicines. Bol Latinoam Caribe Plant Med Aromat. (2008) 7:312–31.
247. Shen C, Ren ZY, Lan HD, Kong LY, Yang M, Su YZ, et al. Perspectives, experiences, and practices of healthcare professionals and patients towards herb–drug interaction: A systematic review of qualitative studies. Phytother Res. (2025) 39:505–20. doi: 10.1002/ptr.8384
248. Araujo CAC and Leon LL. Biological activities of Curcuma longa L. Mem Inst Oswaldo Cruz. (2001) 96:723–8. doi: 10.1590/S0074-02762001000500026
249. Davis JM, Murphy EA, Carmichael MD, Zielinski MR, Groschwitz CM, Brown AS, et al. Curcumin effects on inflammation and performance recovery following eccentric exercise-induced muscle damage. Am J Physiol Regul Integr Comp Physiol. (2007) 292:R2168–73. doi: 10.1152/ajpregu.00858.2006
250. Thangapazham RL, Sharma A, and Maheshwari RK. Beneficial role of curcumin in skin diseases. In: Aggarwal BB, Surh YJ, and Shishodia S, editors. The Molecular Targets and Therapeutic Uses of Curcumin in Health and Disease., Springer, Boston, MA (2007). pp. 343–57. doi: 10.1007/978-0-387-46401-5_15
251. Zeng L, Yang T, Yang K, Yu G, Li J, Xiang W, et al. Efficacy and safety of curcumin and Curcuma longa extract in the treatment of arthritis: a systematic review and meta-analysis of randomized controlled trial. Front Immunol. (2022) 13:891822. doi: 10.3389/fimmu.2022.891822
252. Yang YZ, Li JD, Zhang JG, Zhang K, Zhang AR, Li PP, et al. Mechanism of action and new developments in the study of curcumin in the treatment of osteoarthritis: a narrative review. Inflammopharmacology. (2025) 33:929–40. doi: 10.1007/s10787-025-01665-6
253. He Y, Liu Y, and Zhang M. The beneficial effects of curcumin on aging and age-related diseases: from oxidative stress to antioxidant mechanisms, brain health and apoptosis. Front Aging Neurosci. (2025) 17:1533963. doi: 10.3389/fnagi.2025.1533963
254. Zoi V, Kyritsis AP, Galani V, Lazari D, Sioka C, Voulgaris S, et al. The role of curcumin in cancer: a focus on the PI3K/Akt pathway. Cancers. (2024) 16:1554. doi: 10.3390/cancers16081554
255. Yang ZJ, Huang SY, Zhou DD, Xiong RG, Zhao CN, Fang AP, et al. Effects and mechanisms of curcumin for the prevention and management of cancers: an updated review. Antioxidants. (2022) 11:1481. doi: 10.3390/antiox11081481
256. Sharma A, Sharma T, Bhaskar R, Ola M, Sharma A, and Sharma PC. Promising potential of curcumin and related compounds for antiviral drug discovery. Med Chem. (2024) 20:597–612. doi: 10.2174/0115734064277371240325105016
257. Hidayah RN, Nafisa BB, Saiful’Arifin M, Santosaningsih D, and Muti’ah R. Antiviral activity of curcumin, demethoxycurcumin, bisdemethoxycurcumin and cyclocurcumin compounds of Curcuma longa against NSP3 on SARS-CoV-2. Indones J Cancer Chemoprevention. (2022) 13:166–74.
258. Rizvi SA, Kashanian S, and Alavi M. Demothoxycurcumin as a curcumin analogue with anticancer, antimicrobial, anti-inflammatory, and neuroprotective activities: micro and nanosystems. Nano Micro Biosyst. (2023) 2:7–14. doi: 10.22034/nmbj.2023.417924.1029
259. Khajuria P, Kour D, Sharma K, Singh L, Banoo R, Manhas D, et al. Bisdemethoxycurcumin mitigates Alzheimer disease pathology through autophagy-mediated reduction of senescence and amyloid beta. bioRxiv. (2025) 23:2025.05.19.654834. doi: 10.1101/2025.05.19.654834
260. Ajala OS, Innocent-Ugwu DO, Okechukwu PU, and Dada OH. Demethoxylated curcuminoids as antidiabetic complication drug leads–in silico studies. Afr J Pharm Res Dev. (2024) 16:14–25.
261. Ding X, Chen Y, Zhou L, Wu R, Jian T, Lyu H, et al. Bisdemethoxycurcumin attenuated renal injury via activation of Keap1/Nrf2 pathway in high-fat diet-fed mice. Int J Mol Sci. (2022) 23:7395. doi: 10.3390/ijms23137395
262. Astuti IY, Yupitawati A, and Nurulita NA. Anti-aging activity of tetrahydrocurcumin, Centella asiatica extract, and its mixture. Adv Tradit Med. (2021) 21:57–63. doi: 10.1007/s13596-020-00532-9
263. Zhu L, Xue Y, Feng J, Wang Y, Lu Y, and Chen X. Tetrahydrocurcumin as a stable and highly active curcumin derivative: a review of synthesis, bioconversion, detection and application. Food Biosci. (2023) 53:102591. doi: 10.1016/j.fbio.2023.102591
264. Chen KY, Luo S, Liu YT, Wang X, and Wang JB. Progress of research on tetrahydrocurcumin against liver injury. Gastroenterol Hepatol Res. (2023) 5:14. doi: 10.53388/ghr2023-03-078
265. Wu J, Guan F, Huang H, Chen H, Liu Y, Zhang S, et al. Tetrahydrocurcumin ameliorates hepatic steatosis by restoring hepatocytes lipophagy through mTORC1-TFEB pathway in nonalcoholic steatohepatitis. BioMed Pharmacother. (2024) 178:117297. doi: 10.1016/j.biopha.2024.117297
266. Emani LS, Rao JK, Kumar Dasappa J, Pecchio M, Lakey-Beitia J, Rodriguez H, et al. Studies on curcumin-glucoside in the prevention of alpha-synuclein aggregation. J Alzheimers Dis Rep. (2025) 9:25424823251347260. doi: 10.1177/25424823251347260
267. Pandey P, Verma M, Lakhanpal S, Pandey S, Kumar MR, Bhat M, et al. An updated review summarizing the anticancer potential of poly (lactic-co-glycolic acid) (PLGA) based curcumin, epigallocatechin gallate, and resveratrol nanocarriers. Biopolymers. (2025) 116:e23637. doi: 10.1002/bip.23637
268. Keshavarz Shahbaz S, Koushki K, Izadi O, Penson PE, Sukhorukov VN, Kesharwani P, et al. Advancements in curcumin-loaded PLGA nanoparticle delivery systems: progressive strategies in cancer therapy. J Drug Targeting. (2024) 32:1207–32. doi: 10.1080/1061186X.2024.2389892
269. Arya P and Raghav N. In vitro studies of curcumin-β-cyclodextrin inclusion complex as sustained release system. J Mol Struct. (2021) 1228:129774. doi: 10.1016/j.molstruc.2020.129774
270. Wu X and Yang Y. Research progress on drug delivery systems for curcumin in the treatment of gastrointestinal tumors. World J Gastrointest Oncol. (2023) 15:1342. doi: 10.4251/wjgo.v15.i8.1342
271. Al-Thubaiti EH. Antibacterial and antioxidant activities of curcumin/Zn metal complex with its chemical characterization and spectroscopic studies. Heliyon. (2023) 9:e17468. doi: 10.1016/j.heliyon.2023.e17468
272. Zhang B, Guo W, Chen Z, Chen Y, Zhang R, Liu M, et al. Physicochemical characterization and oral bioavailability of curcumin–phospholipid complex nanosuspensions prepared based on microfluidic system. Pharmaceutics. (2025) 17:395. doi: 10.3390/pharmaceutics17030395
273. Mishra N. Comparative efficacy of Curcuma longa, Azadirachta indica and Centella asiatica in canine wound healing: a review. Indian J Vet Public Health. (2024) 10:32. doi: 10.62418/ijvph.10.2.2024.32-40
274. Moutabian H, Ghahramani-Asl R, Mortezazadeh T, Laripour R, Narmani A, Zamani H, et al. The cardioprotective effects of nano-curcumin against doxorubicin-induced cardiotoxicity: a systematic review. Biofactors. (2022) 48:597–610. doi: 10.1002/biof.1823
275. Albaker AB, Alhusaini AM, Albadrani HM, Sarawi WS, Alsabhan JF, Mansy TW, et al. Involvement of MEF-2 and Apo-1/Fas in the modulatory mechanism of liposomal curcumin against cardiac hypoxia. Nat Prod Commun. (2025) 20:1934578X251334719. doi: 10.1177/1934578X251334719
276. Li J, Wang SH, Liu YT, Zhang Q, and Zhou GZ. Inhibition of autophagic flux by the curcumin analog EF-24 and its antiproliferative effect on MCF-7 cancer cells. J Biochem Mol Toxicol. (2023) 37:e23307. doi: 10.1002/jbt.23307
277. Tang C, Liu J, Yang C, Ma J, Chen X, Liu D, et al. Curcumin and its analogs in non-small cell lung cancer treatment: challenges and expectations. Biomolecules. (2022) 12:1636. doi: 10.3390/biom12111636
278. Rakotondrabe TF, Fan MX, Muema FW, and Guo MQ. Modulating inflammation-mediated diseases via natural phenolic compounds loaded in nanocarrier systems. Pharmaceutics. (2023) 15:699. doi: 10.3390/pharmaceutics15020699
279. Unde JS and Shukla R. Polymeric micelles in the delivery of therapeutic phytoconstituents. In: Singh SK, Gulati M, Mutalik S, Dhanasekaran M, and Dua K, editors. Polymeric Micelles: Principles, Perspectives and Practices. Springer, Singapore (2023). pp. 175–201. doi: 10.1007/978-981-99-0361-0_9
280. Matacchione G, Valli D, Silvestrini A, Giuliani A, Sabbatinelli J, Giordani C, et al. Curcumin, polydatin and quercetin synergistic activity protects from high-glucose-induced inflammation and oxidative stress. Antioxidants. (2022) 11:1037. doi: 10.3390/antiox11061037
281. Grabarczyk M, Justyńska W, Czpakowska J, Smolińska E, Bielenin A, Glabinski A, et al. Role of plant phytochemicals: resveratrol, curcumin, luteolin and quercetin in demyelination, neurodegeneration, and epilepsy. Antioxidants. (2024) 13:1364. doi: 10.3390/antiox13111364
282. Mohammed R and Al-Okaily BN. Insights into curcumin-selenium nanoparticles in modulating hormonal levels and promoting PTEN gene expression in doxorubicin treated rats. J Anim Health Prod. (2024) 12:429–36. doi: 10.17582/journal.jahp/2024/12.3.429.436
283. Zhang L, Wang X, and Si H. Synergistic anti-inflammatory effects and mechanisms of the combination of resveratrol and curcumin in human vascular endothelial cells and rodent aorta. J Nutr Biochem. (2022) 108:109083. doi: 10.1016/j.jnutbio.2022.109083
284. Micale N, Molonia MS, Citarella A, Cimino F, Saija A, Cristani M, et al. Natural product-based hybrids as potential candidates for the treatment of cancer: focus on curcumin and resveratrol. Molecules. (2021) 26:4665. doi: 10.3390/molecules26154665
285. Mohi-Ud-Din R, Mir RH, Wani TU, Shah AJ, Mohi-Ud-Din I, Dar MA, et al. Novel drug delivery system for curcumin: Implementation to improve therapeutic efficacy against neurological disorders. Comb Chem High Throughput Screen. (2022) 25:607–15. doi: 10.2174/1386207324666210705114058
286. Chen M, Wang S, Chen Y, Shen H, Chen L, Ding L, et al. Precision cardiac targeting: empowering curcumin therapy through smart exosome-mediated drug delivery in myocardial infarction. Regener Biomater. (2024) 11:rbad108. doi: 10.1093/rb/rbad108
287. Mohanty C and Sahoo SK. Curcumin and its topical formulations for wound healing applications. Drug Discov Today. (2017) 22:1582–92. doi: 10.1016/j.drudis.2017.07.001
288. Zakerikhoob M, Abbasi S, Yousefi G, Mokhtari M, and Noorbakhsh MS. Curcumin-incorporated crosslinked sodium alginate-g-poly (N-isopropyl acrylamide) thermo-responsive hydrogel as an in situ forming injectable dressing for wound healing: In vitro characterization and in vivo evaluation. Carbohydr Polym. (2021) 271:118434. doi: 10.1016/j.carbpol.2021.118434
289. Krausz AE, Adler BL, Cabral V, Navati M, Doerner J, Charafeddine RA, et al. Curcumin-encapsulated nanoparticles as innovative antimicrobial and wound healing agent. Nanomed Nanotechnol Biol Med. (2015) 11:195–206. doi: 10.1016/j.nano.2014.09.004
290. Libby P. Inflammation and cardiovascular disease mechanisms. Am J Clin Nutr. (2006) 83:456S–60S. doi: 10.1093/ajcn/83.2.456S
291. Coussens LM and Werb Z. Inflammation and cancer. Nature. (2002) 420:860–7. doi: 10.1038/nature01322
292. Wellen KE and Hotamisligil GS. Inflammation, stress, and diabetes. J Clin Invest. (2005) 115:1111–9. doi: 10.1172/JCI25102
293. Amor S, Puentes F, Baker D, and van der Valk P. Inflammation in neurodegenerative diseases. Immunology. (2010) 129:154–69. doi: 10.1111/j.1365-2567.2009.03225.x
294. Tak PP and Firestein GS. NF-κB: a key role in inflammatory diseases. J Clin Invest. (2001) 107:7–11. doi: 10.1172/JCI11830
295. Shehzad A, Qureshi M, Anwar MN, and Lee YS. Multifunctional curcumin mediate multitherapeutic effects. J Food Sci. (2017) 82:2006–15. doi: 10.1111/1750-3841.13793
296. Banik U, Parasuraman S, Adhikary AK, and Othman NH. Curcumin: the spicy modulator of breast carcinogenesis. J Exp Clin Cancer Res. (2017) 36:98. doi: 10.1186/s13046-017-0566-5
297. Hatcher H, Planalp R, Cho J, Torti FM, and Torti SV. Curcumin: from ancient medicine to current clinical trials. Cell Mol Life Sci. (2008) 65:1631–52. doi: 10.1007/s00018-008-7452-4
298. Gao X, Kuo J, Jiang H, Deeb D, Liu Y, Divine G, et al. Immunomodulatory activity of curcumin: suppression of lymphocyte proliferation, development of cell-mediated cytotoxicity, and cytokine production in vitro. Biochem Pharmacol. (2004) 68:51–61. doi: 10.1016/j.bcp.2004.03.015
299. Lestari MLAD and Indrayanto G. Curcumin. In: Brittain HG, editor. Profiles of Drug Substances, Excipients and Related Methodology. Academic Press, Cambridge, MA, USA (2014). pp. 113–204. doi: 10.1016/B978-0-12-800173-8.00003-9
300. Daily JW, Yang M, and Park S. Efficacy of turmeric extracts and curcumin for alleviating the symptoms of joint arthritis: a systematic review and meta-analysis of randomized clinical trials. J Med Food. (2016) 19:717–29. doi: 10.1089/jmf.2016.3705
301. Menon VP and Sudheer AR. Antioxidant and anti-inflammatory properties of curcumin. In: Aggarwal BB, Surh YJ, and Shishodia S, editors. The Molecular Targets and Therapeutic Uses of Curcumin in Health and Disease. Springer, New York, USA (2007). pp. 105–25. doi: 10.1007/978-0-387-46401-5_3
302. Surh YJ. Anti-tumor promoting potential of selected spice ingredients with antioxidative and anti-inflammatory activities: a short review. Food Chem Toxicol. (2002) 40:1091–7. doi: 10.1016/S0278-6915(02)00037-6
303. Jiménez-Osorio AS, Monroy A, and Alavez S. Curcumin and insulin resistance—molecular targets and clinical evidences. Biofactors. (2016) 42:561–80. doi: 10.1002/biof.1302
304. Kim T, Davis J, Zhang AJ, He X, and Mathews ST. Curcumin activates AMPK and suppresses gluconeogenic gene expression in hepatoma cells. Biochem Biophys Res Commun. (2009) 388:377–82. doi: 10.1016/j.bbrc.2009.08.018
305. Liu Z and Ying Y. The inhibitory effect of curcumin on virus-induced cytokine storm and its potential use in the associated severe pneumonia. Front Cell Dev Biol. (2020) 8:479. doi: 10.3389/fcell.2020.00479
306. Pizzino G, Irrera N, Cucinotta M, Pallio G, Mannino F, Arcoraci V, et al. Oxidative stress: harms and benefits for human health. Oxid Med Cell Longev. (2017) 2017:8416763. doi: 10.1155/2017/8416763
307. Deng T, Xu J, Wang Q, Wang X, Jiao Y, Cao X, et al. Immunomodulatory effects of curcumin on macrophage polarization in rheumatoid arthritis. Front Pharmacol. (2024) 15:1369337. doi: 10.3389/fphar.2024.1369337
308. Mohseni S, Tavakoli A, Ghazipoor H, Pouralimohamadi N, Zare R, Rampp T, et al. Curcumin for the clinical treatment of inflammatory bowel diseases: a systematic review and meta-analysis of placebo-controlled randomized clinical trials. Front Nutr. (2025) 12:1494351. doi: 10.3389/fnut.2025.1494351
309. Zeng L, Yang T, Yang K, Yu G, Li J, Xiang W, et al. Curcumin and Curcuma longa extract in the treatment of 10 types of autoimmune diseases: a systematic review and meta-analysis of 31 randomized controlled trials. Front Immunol. (2022) 13:896476. doi: 10.3389/fimmu.2022.896476
310. Ghoushi E, Poudineh M, Parsamanesh N, Jamialahmadi T, and Sahebkar A. Curcumin as a regulator of Th17 cells: Unveiling the mechanisms. Food Chem Mol Sci. (2024) 8:100198. doi: 10.1016/j.fochms.2024.100198
311. Mentucci FM, Ferrara MG, Ercole A, Rumie Vittar NB, and Lamberti MJ. Interplay between cancer-associated fibroblasts and dendritic cells: implications for tumor immunity. Front Immunol. (2025) 16:1515390. doi: 10.3389/fimmu.2025.1515390
312. Allegra A, Mirabile G, Ettari R, Pioggia G, and Gangemi S. The impact of curcumin on immune response: an immunomodulatory strategy to treat sepsis. Int J Mol Sci. (2022) 23:14710. doi: 10.3390/ijms232314710
313. Su J, Tan Q, Wu S, Zhou F, Xu C, Zhao H, et al. Administration of turmeric kombucha ameliorates lipopolysaccharide-induced sepsis by attenuating inflammation and modulating gut microbiota. Front Microbiol. (2024) 15:1452190. doi: 10.3389/fmicb.2024.1452190
314. Kali A and Charles MV. Curcumin as a promising therapy for COVID-19: A review. Glob J Med Pharm BioMed Update. (2024) 19:2. doi: 10.25259/GJMPBU_78_2023
315. Verma DK, Hasan A, Rengaraju M, Devi S, Sharma G, Narayanan V, et al. Evaluation of Withania somnifera-based supplement for immunomodulatory and antiviral properties against viral infection. J Ayurveda Integr Med. (2024) 15:100955. doi: 10.1016/j.jaim.2024.100955
316. Wang Y, Zeng Y, Yang W, Wang X, and Jiang J. Targeting CD8+ T cells with natural products for tumor therapy: revealing insights into the mechanisms. Phytomedicine. (2024) 129:155608. doi: 10.1016/j.phymed.2024.155608
317. Limsakul P, Srifa P, Huang Z, Zhu L, Wu Y, and Charupanit K. Immunomodulatory effects of curcumin on CAR T-cell therapy. Antioxidants. (2025) 14:454. doi: 10.3390/antiox14040454
318. Hong Q, Lyu W, Zhang C, Yao W, Han Y, and Chen N. Research trajectory and future trends in curcumin related to immunity: a bibliometric analysis of publications from last two decades. Front Immunol. (2025) 16:1559670. doi: 10.3389/fimmu.2025.1559670
319. Batinić-Haberle I, Rebouças JS, and Spasojević I. Superoxide dismutase mimics: chemistry, pharmacology, and therapeutic potential. Antioxid Redox Signal. (2010) 13:877–918. doi: 10.1089/ars.2009.287
320. Sankar P, Gopal Telang A, Kalaivanan R, Karunakaran V, Manikam K, and Sarkar SN. Effects of nanoparticle-encapsulated curcumin on arsenic-induced liver toxicity in rats. Environ Toxicol. (2015) 30:628–37. doi: 10.1002/tox.21940
321. Aftab N and Vieira A. Antioxidant activities of curcumin and combinations of this curcuminoid with other phytochemicals. Phytother Res. (2010) 24:500–2. doi: 10.1002/ptr.2960
322. Kanwal Q, Ahmed M, Hamza M, Ahmad M, Yousaf N, Javaid A, et al. Curcumin nanoparticles: physicochemical fabrication, characterization, antioxidant, enzyme inhibition, molecular docking and simulation studies. RSC Adv. (2023) 13:22268–80. doi: 10.1039/D3RA01432K
323. Jabczyk M, Nowak J, Hudzik B, and Zubelewicz-Szkodzińska B. Curcumin in metabolic health and disease. Nutrients. (2021) 13:4440. doi: 10.3390/nu13124440
324. Qiu L, Gao C, Wang H, Ren Y, Li J, Li M, et al. Effects of dietary polyphenol curcumin supplementation on metabolic, inflammatory, and oxidative stress indices in patients with metabolic syndrome: a systematic review and meta-analysis of randomized controlled trials. Front Endocrinol. (2023) 14:1216708. doi: 10.3389/fendo.2023.1216708
325. Jagetia GC and Rajanikant GK. Curcumin stimulates the antioxidant mechanisms in mouse skin exposed to fractionated γ-irradiation. Antioxidants. (2015) 4:25–41. doi: 10.3390/antiox4010025
326. Kebede BH, Forsido SF, Tola YB, and Astatkie T. Free radical scavenging capacity, antibacterial activity and essential oil composition of turmeric (Curcuma domestica) varieties grown in Ethiopia. Heliyon. (2021) 7:e06239. doi: 10.1016/j.heliyon.2021.e06239
327. Braga MC, Vieira ECS, and de Oliveira TF. Curcuma longa L. leaves: Characterization (bioactive and antinutritional compounds) for use in human food in Brazil. Food Chem. (2018) 265:308–15. doi: 10.1016/j.foodchem.2018.05.096
328. Kuncha M, Naidu VGM, Sahu BD, Gadepalli SG, and Sistla R. Curcumin potentiates the anti-arthritic effect of prednisolone in Freund’s complete adjuvant-induced arthritic rats. J Pharm Pharmacol. (2014) 66:133–44. doi: 10.1111/jphp.12156
329. Guo YL, Li XZ, and Kuang CT. Antioxidant pathways and chemical mechanism of curcumin. Adv Mater Res. (2011) 236:2311–4. doi: 10.4028/www.scientific.net/AMR.236-238.2311
330. Jovanovic SV, Steenken S, Boone CW, and Simic MG. H-atom transfer is a preferred antioxidant mechanism of curcumin. J Am Chem Soc. (1999) 121:9677–81. doi: 10.1021/ja991446m
331. Barclay LRC, Vinqvist MR, Mukai K, Goto H, Hashimoto Y, Tokunaga A, et al. On the antioxidant mechanism of curcumin: classical methods are needed to determine antioxidant mechanism and activity. Org Lett. (2000) 2:2841–3. doi: 10.1021/ol000173t
332. Oglah MK and Mustafa YF. Curcumin analogs: synthesis and biological activities. Med Chem Res. (2020) 29:479–86. doi: 10.1007/s00044-019-02497-0
333. Priyadarsini KI, Maity DK, Naik GH, Kumar MS, Unnikrishnan MK, Satav JG, et al. Role of phenolic OH and methylene hydrogen on the free radical reactions and antioxidant activity of curcumin. Free Radic Biol Med. (2003) 35:475–84. doi: 10.1016/S0891-5849(03)00325-3
334. Shang YJ, Jin XL, Shang XL, Tang JJ, Liu GY, Dai F, et al. Antioxidant capacity of curcumin-directed analogues: structure–activity relationship and influence of microenvironment. Food Chem. (2010) 119:1435–42. doi: 10.1016/j.foodchem.2009.09.024
335. Munir D, Maria A, and Bashiruddin J. The antioxidant effect of curcumin on cochlear fibroblasts in rat models of Diabetes mellitus. Iran J Otorhinolaryngol. (2017) 29:197–202.
336. Asouri M, Ataee R, Ahmadi AA, Amini A, and Moshaei MR. Antioxidant and free radical scavenging activities of curcumin. Asian J Chem. (2013) 25:7593. doi: 10.14233/ajchem.2013.15308
337. Barzegar A and Moosavi-Movahedi AA. Intracellular ROS protection efficiency and free radical-scavenging activity of curcumin. PloS One. (2011) 6:e26012. doi: 10.1371/journal.pone.0026012
338. Chen S, Wu J, Tang Q, Xu C, Huang Y, Huang D, et al. Nano-micelles based on hydroxyethyl starch-curcumin conjugates for improved stability, antioxidant and anticancer activity of curcumin. Carbohydr Polym. (2020) 228:115398. doi: 10.1016/j.carbpol.2019.115398
339. Ma Q, Ren Y, and Wang L. Investigation of antioxidant activity and release kinetics of curcumin from tara gum/polyvinyl alcohol active film. Food Hydrocoll. (2017) 70:286–92. doi: 10.1016/j.foodhyd.2017.04.018
340. Zhu Q, Sun Y, Yun X, Ou Y, Zhang W, and Li JX. Antinociceptive effects of curcumin in a rat model of postoperative pain. Sci Rep. (2014) 4:4932. doi: 10.1038/srep04932
341. Schraufstätter E and Bernt H. Antibacterial action of curcumin and related compounds. Nature. (1949) 164:456–7. doi: 10.1038/164456a0
342. Liu M, Lu Y, Gao P, Xie X, Li D, Yu D, et al. Effect of curcumin on laying performance, egg quality, endocrine hormones, and immune activity in heat-stressed hens. Poult Sci. (2020) 99:2196–202. doi: 10.1016/j.psj.2019.12.001
343. Hajavi J, Momtazi AA, Johnston TP, Banach M, Majeed M, and Sahebkar A. Curcumin: a naturally occurring modulator of adipokines in diabetes. J Cell Biochem. (2017) 118:4170–82. doi: 10.1002/jcb.26121
344. Akinyemi KO, Al-Khafaji NSK, Al-Alaq FT, Fakorede CO, Al-Dahmoshi HOM, Iwalokun BA, et al. Extended-spectrum beta-lactamases encoding genes among Salmonella enterica serovar Typhi isolates in patients with typhoid fever from four academic medical centers in Lagos, Nigeria. Rev Invest Clin. (2022) 74:165–71. doi: 10.24875/ric.22000078
345. Helmy YA, Taha-Abdelaziz K, Hawwas HAEH, Ghosh S, AlKafaas SS, Moawad MM, et al. Antimicrobial resistance and recent alternatives to antibiotics for the control of bacterial pathogens with an emphasis on foodborne pathogens. Antibiotics. (2023) 12:274. doi: 10.3390/antibiotics12020274
346. Alduhaidhawi AHM, AlHuchaimi SN, Al-Mayah TA, Al-Ouqaili MTS, Alkafaas SS, Muthupandian S, et al. Prevalence of CRISPR-Cas systems and their possible association with antibiotic resistance in Enterococcus faecalis and Enterococcus faecium collected from hospital wastewater. Infect Drug Resist. (2022) 15:1143–54. doi: 10.2147/IDR.S358248
347. Tong SYC, Davis JS, Eichenberger E, Holland TL, and Fowler VG Jr. Staphylococcus aureus infections: epidemiology, pathophysiology, clinical manifestations, and management. Clin Microbiol Rev. (2015) 28:603–61. doi: 10.1128/cmr.00134-14
348. Colomb-Cotinat M, Lacoste J, Brun-Buisson C, Jarlier V, Coignard B, and Vaux S. Estimating the morbidity and mortality associated with infections due to multidrug-resistant bacteria (MDRB), France, 2012. Antimicrob Resist Infect Control. (2016) 5:1–11. doi: 10.1186/s13756-016-0154-z
349. Ribeiro APD, Pavarina AC, Dovigo LN, Brunetti IL, Bagnato VS, Vergani CE, et al. Phototoxic effect of curcumin on methicillin-resistant Staphylococcus aureus and L929 fibroblasts. Lasers Med Sci. (2013) 28:391–8. doi: 10.1007/s10103-012-1064-9
350. Wang J, Zhou X, Li W, Deng X, Deng Y, and Niu X. Curcumin protects mice from Staphylococcus aureus pneumonia by interfering with the self-assembly process of α-hemolysin. Sci Rep. (2016) 6:28254. doi: 10.1038/srep28254
351. Mun SH, Joung DK, Kim YS, Kang OH, Kim SB, Seo YS, et al. Synergistic antibacterial effect of curcumin against methicillin-resistant Staphylococcus aureus. Phytomedicine. (2013) 20:714–8. doi: 10.1016/j.phymed.2013.02.006
352. Gunes H, Gulen D, Mutlu R, Gumus A, Tas T, and Topkaya AE. Antibacterial effects of curcumin: an in vitro minimum inhibitory concentration study. Toxicol Ind Health. (2016) 32:246–50. doi: 10.1177/0748233713498458
353. Tyagi P, Singh M, Kumari H, Kumari A, and Mukhopadhyay K. Bactericidal activity of curcumin I is associated with damaging of bacterial membrane. PloS One. (2015) 10:e0121313. doi: 10.1371/journal.pone.0121313
354. Zheng D, Huang C, Huang H, Zhao Y, Khan MRU, Zhao H, et al. Antibacterial mechanism of curcumin: a review. Chem Biodivers. (2020) 17:e2000171. doi: 10.1002/cbdv.202000171
355. Adamczak A, Ożarowski M, and Karpiński TM. Curcumin, a natural antimicrobial agent with strain-specific activity. Pharmaceuticals. (2020) 13:153. doi: 10.3390/ph13070153
356. Shome S, Talukdar AD, and Upadhyaya H. Antibacterial activity of curcumin and its essential nanoformulations against some clinically important bacterial pathogens: A comprehensive review. Biotechnol Appl Biochem. (2022) 69:2357–86. doi: 10.1002/bab.2289
357. Teow SY, Liew K, Ali SA, Khoo ASB, and Peh SC. Antibacterial action of curcumin against Staphylococcus aureus: a brief review. J Trop Med. (2016) 2016:2853045. doi: 10.1155/2016/2853045
358. Yun DG and Lee DG. Antibacterial activity of curcumin via apoptosis-like response in Escherichia coli. Appl Microbiol Biotechnol. (2016) 100:5505–14. doi: 10.1007/s00253-016-7415-x
359. Gholami M, Zeighami H, Bikas R, Heidari A, Rafiee F, and Haghi F. Inhibitory activity of metal-curcumin complexes on quorum sensing related virulence factors of Pseudomonas aeruginosa PAO1. AMB Express. (2020) 10:111. doi: 10.1186/s13568-020-01045-z
360. Hettiarachchi SS, Perera Y, Dunuweera SP, Dunuweera AN, Rajapakse S, and Rajapakse RMG. Comparison of antibacterial activity of nanocurcumin with bulk curcumin. ACS Omega. (2022) 7:46494–500. doi: 10.1021/acsomega.2c05293
361. Leyva-Diaz AA, Hernandez-Patlan D, Solis-Cruz B, Adhikari B, Kwon YM, Latorre JD, et al. Evaluation of curcumin and copper acetate against Salmonella Typhimurium infection, intestinal permeability, and cecal microbiota composition in broiler chickens. J Anim Sci Biotechnol. (2021) 12:23. doi: 10.1186/s40104-021-00545-7
362. Huang J, Chen B, Li H, Zeng QH, Wang JJ, Liu H, et al. Enhanced antibacterial and antibiofilm functions of the curcumin-mediated photodynamic inactivation against Listeria monocytogenes. Food Control. (2020) 108:106886. doi: 10.1016/j.foodcont.2019.106886
363. Morão LG, Polaquini CR, Kopacz M, Torrezan GS, Ayusso GM, Dilarri G, et al. A simplified curcumin targets the membrane of Bacillus subtilis. Microbiol Open. (2019) 8:e00683. doi: 10.1002/mbo3.683
364. Barua N and Buragohain AK. Therapeutic potential of curcumin as an antimycobacterial agent. Biomolecules. (2021) 11:1278. doi: 10.3390/biom11091278
365. Dai C, Lin J, Li H, Shen Z, Wang Y, Velkov T, et al. The natural product curcumin as an antibacterial agent: Current achievements and problems. Antioxidants. (2022) 11:459. doi: 10.3390/antiox11030459
366. Karami-Zarandi M, Ghale HEG, and Ranjbar R. Characterization of virulence factors and antibacterial activity of curcumin in hypervirulent Klebsiella pneumoniae. Future Microbiol. (2022) 17:529–40. doi: 10.2217/fmb-2021-0222
367. Neelakantan P, Subbarao C, Sharma S, Subbarao CV, Garcia-Godoy F, and Gutmann JL. Effectiveness of curcumin against Enterococcus faecalis biofilm. Acta Odontol Scand. (2013) 71:1453–7. doi: 10.3109/00016357.2013.769627
368. De R, Kundu P, Swarnakar S, Ramamurthy T, Chowdhury A, Nair GB, et al. Antimicrobial activity of curcumin against Helicobacter pylori isolates from India and during infections in mice. Antimicrob Agents Chemother. (2009) 53:1592–7. doi: 10.1128/aac.01242-08
369. Hu P, Huang P, and Chen MW. Curcumin reduces Streptococcus mutans biofilm formation by inhibiting sortase A activity. Arch Oral Biol. (2013) 58:1343–8. doi: 10.1016/j.archoralbio.2013.05.004
370. Nasution HR, Septama AW, and Nugraha SE. Antibiofilm formation activities of ethanol extract of Curcuma domestica Val. rhizome against multidrug-resistant Acinetobacter baumannii. Int J Sci Technol Manage. (2023) 4:809–12. doi: 10.46729/ijstm.v4i4.883
371. Li LM, Li J, and Zhang XY. Antimicrobial and molecular interaction studies on derivatives of curcumin against Streptococcus pneumoniae which caused pneumonia. Electron J Biotechnol. (2016) 19:8–14. doi: 10.1016/j.ejbt.2015.09.011
372. Alikiaii B, Bagherniya M, Askari G, Sathyapalan T, and Sahebkar A. Evaluation of the effect of curcumin on pneumonia: A systematic review of preclinical studies. Phytother Res. (2021) 35:1939–52. doi: 10.1002/ptr.6939
373. Mody D, Athamneh AIM, and Seleem MN. Curcumin: A natural derivative with antibacterial activity against Clostridium difficile. J Glob Antimicrob Resist. (2020) 21:154–61. doi: 10.1016/j.jgar.2019.10.005
374. Sun GQ, Zhang ZCL, Wang WJ, Lan LHL, Lu LLF, Yan YHZ, et al. Antibacterial activity of the curcumin derivative FM0817 against Neisseria gonorrhoeae. Afr J Microbiol Res. (2014) 8:446–51. doi: 10.5897/AJMR2012.2457
375. Sun GQ, Lan HL, Lu LF, Wang J, Yan HZ, Wu XJ, et al. Comparison of the antimicrobial activity of the curcumin derivatives between FM0817 and FM04. Int J Clin Exp Med. (2016) 9:12130–4.
376. Bongomin F, Gago S, Oladele RO, and Denning DW. Global and multi-national prevalence of fungal diseases—estimate precision. J Fungi. (2017) 3:57. doi: 10.3390/jof3040057
377. Karkowska-Kuleta J, Rapala-Kozik M, and Kozik A. Fungi pathogenic to humans: molecular bases of virulence of Candida albicans, Cryptococcus neoformans and Aspergillus fumigatus. Acta Biochim Pol. (2009) 56:211–24. doi: 10.18388/abp.2009_2452
378. Leroy O, Gangneux JP, Montravers P, Mira JP, Gouin F, Sollet JP, et al. Epidemiology, management, and risk factors for death of invasive Candida infections in critical care: a multicenter, prospective, observational study in France (2005–2006). Crit Care Med. (2009) 37:1612–8. doi: 10.1097/CCM.0b013e31819efac0
379. Garcia-Gomes AS, Curvelo JAR, Soares RMA, and Ferreira-Pereira A. Curcumin acts synergistically with fluconazole to sensitize a clinical isolate of Candida albicans showing a MDR phenotype. Med Mycol. (2012) 50:26–32. doi: 10.3109/13693786.2011.578156
380. Thomachan S, Sindhu S, and John VD. Synthesis, characterization, antibacterial, antifungal and cytotoxic activity of curcuminoid analogues with trisubstituted phenyl and anthracenyl ring and their zinc (II), copper (II) and vanadyl (IV) chelates. Int J Pharmac Chem. (2016) 6:78–86. doi: 10.7439/IJPC.V6I3.2960
381. Neelofar K, Shreaz S, Rimple B, Muralidhar S, Nikhat M, and Khan LA. Curcumin as a promising anticandidal of clinical interest. Can J Microbiol. (2011) 57:204–10. doi: 10.1139/W10-117
382. Dovigo LN, Pavarina AC, Ribeiro APD, Brunetti IL, Costa CAS, Jacomassi DP, et al. Investigation of the photodynamic effects of curcumin against Candida albicans. Photochem Photobiol. (2011) 87:895–903. doi: 10.1111/j.1751-1097.2011.00937.x
383. Freitas DD, Silva CR, Andrade Neto JB, Campos RS, Sampaio LS, Nascimento FBSA, et al. In vitro evaluation of the curcumin against fluconazole-resistant strains of Candida spp. and Cryptococcus neoformans. Int J Curr Microbiol Appl Sci. (2018) 4:1191–202.
384. Kumar A, Dhamgaye S, Maurya IK, Singh A, Sharma M, and Prasad R. Curcumin targets cell wall integrity via calcineurin-mediated signaling in Candida albicans. Antimicrob Agents Chemother. (2014) 58:167–75. doi: 10.1128/aac.01385-13
385. Zhang D, Yang Y, Yao B, Hu T, Ma Z, Shi W, et al. Curcumin inhibits Aspergillus flavus infection and aflatoxin production possibly by inducing ROS burst. Food Res Int. (2023) 167:112646. doi: 10.1016/j.foodres.2023.112646
386. Martins CVB, Da Silva DL, Neres ATM, Magalhaes TFF, Watanabe GA, Modolo LV, et al. Curcumin as a promising antifungal of clinical interest. J Antimicrob Chemother. (2009) 63:337–9. doi: 10.1093/jac/dkn488
387. Da Silva DL, Magalhães TFF, Dos Santos JRA, De Paula TP, Modolo LV, De Fatima A, et al. Curcumin enhances the activity of fluconazole against Cryptococcus gattii-induced cryptococcosis infection in mice. J Appl Microbiol. (2016) 120:41–8. doi: 10.1111/jam.12966
388. Brasch J, Beck-Jendroschek V, and Mahn V. Photochemical inhibition of Trichophyton rubrum by different compoundings of curcumin. Mycoses. (2018) 61:393–9. doi: 10.1111/myc.12758
389. Shakib P, Ali ASM, Javanmard E, Mardanshah O, Mohaghegh MA, Zivdari M, et al. Anti-Trichophyton effects of curcumin: A systematic review. Antiinfect Agents. (2021) 19:29–34. doi: 10.2174/2211352519666210202085849
390. Sontsa-Donhoung AM, Bahdjolbe M, Hawaou, and Nwaga D. Selecting e ndophytes for rhizome production, curcumin content, biocontrol potential, and antioxidant activities of turmeric (Curcuma longa). BioMed Res Int. (2022) 2022:8321734. doi: 10.1155/2022/8321734
391. Ferreira FD, Kemmelmeier C, Arrotéia CC, da Costa CL, Mallmann CA, Janeiro V, et al. Inhibitory effect of the essential oil of Curcuma longa L. and curcumin on aflatoxin production by Aspergillus flavus Link. Food Chem. (2013) 136:789–93. doi: 10.1016/j.foodchem.2012.08.003
392. Ferreira FD, Mossini SAG, Ferreira FMD, Arrotéia CC, da Costa CL, Nakamura CV, et al. The inhibitory effects of Curcuma longa L. essential oil and curcumin on Aspergillus flavus Link growth and morphology. Sci World J. (2013) 2013:343804. doi: 10.1155/2013/343804
393. Rukayadi Y and Hwang JK. In vitro anti-Malassezia activity of xanthorrhizol isolated from Curcuma xanthorrhiza Roxb. Lett Appl Microbiol. (2007) 44:126–30. doi: 10.1111/j.1472-765X.2006.02062.x
394. Song L, Zhang F, Yu J, Wei C, Han Q, and Meng X. Antifungal effect and possible mechanism of curcumin mediated photodynamic technology against Penicillium expansum. Postharvest Biol Technol. (2020) 167:111234. doi: 10.1016/j.postharvbio.2020.111234
395. Pang J, Zhang F, Wang Z, Wu Q, Liu B, and Meng X. Inhibitory effect and mechanism of curcumin-based photodynamic inactivation on patulin secretion by Penicillium expansum. Innov Food Sci Emerg Technol. (2022) 80:103078. doi: 10.1016/j.ifset.2022.103078
396. Qi C, Zhang H, Chen W, and Liu W. Curcumin: An innovative approach for postharvest control of Alternaria alternata-induced black rot in cherry tomatoes. Fungal Biol. (2024) 128:1691–7. doi: 10.1016/j.funbio.2024.02.005
397. Kipkogei K, Kiptui K, and Kiprop E. Antifungal potential of Curcuma longa (Turmeric) and Zingiber officinale (Ginger) against Alternaria alternata infecting spinach in Kenya. World J Agric Res. (2019) 7:124–31. doi: 10.12691/wjar-7-4-2
398. Brasch J, Freitag-Wolf S, Beck-Jendroschek V, and Huber M. Inhibition of dermatophytes by photodynamic treatment with curcumin. Med Mycol. (2017) 55:754–62. doi: 10.1093/mmy/myw139
399. Zarrinfar H, Behnam M, Hatamipour M, and Sahebkar A. Antifungal activities of curcuminoids and difluorinated curcumin against clinical dermatophyte isolates. In: Barreto GE and Sahebkar A, editors. Pharmacological Properties of Plant-Derived Natural Products and Implications for Human Health. Advances in Experimental Medicine and Biology. Springer, Cham, Switzerland (2021). pp. 101–7. doi: 10.1007/978-3-030-64872-5_8
400. Kim EJ, Song BN, Jeong DS, Kim SY, Cho YS, and Park SY. Anti-oxidative and anti-inflammatory activities of fermented turmeric (Curcuma longa L.) by Rhizopus oryzae. J Life Sci. (2017) 27:1315–23. doi: 10.5352/JLS.2017.27.11.1315
401. Wei C, Zhang F, Song L, Chen X, and Meng X. Photosensitization effect of curcumin for controlling plant pathogen Botrytis cinerea in postharvest apple. Food Control. (2021) 123:107683. doi: 10.1016/j.foodcont.2020.107683
402. Hua C, Kai K, Bi W, Shi W, Liu Y, and Zhang D. Curcumin induces oxidative stress in Botrytis cinerea, resulting in a reduction in gray mold decay in kiwifruit. J Agric Food Chem. (2019) 67:7968–76. doi: 10.1021/acs.jafc.9b00539
403. Cheraghipour K, Ezatpour B, Masoori L, Marzban A, Sepahvand A, Rouzbahani AK, et al. Anti-Candida activity of curcumin: A systematic review. Curr Drug Discov Technol. (2021) 18:379–90. doi: 10.2174/1570163817666200518074629
404. Rocha OB, e Silva KSF, de Carvalho Junior MAB, Moraes D, Alonso A, Alonso L, et al. Proteomic alterations in Paracoccidioides brasiliensis caused by exposure to curcumin. J Proteom. (2022) 266:104683. doi: 10.1016/j.jprot.2022.104683
405. Rocha OB, do Carmo Silva L, de Carvalho Júnior MAB, de Oliveira AA, de Almeida Soares CM, and Pereira M. In vitro and in silico analysis reveals antifungal activity and potential targets of curcumin on Paracoccidioides spp. Braz J Microbiol. (2021) 52:1897–911. doi: 10.1007/s42770-021-00548-6
406. Huang L, Zhang J, Song T, Yuan L, Zhou J, Yin H, et al. Antifungal curcumin promotes chitin accumulation associated with decreased virulence of Sporothrix schenckii. Int Immunopharmacol. (2016) 34:263–70. doi: 10.1016/j.intimp.2016.03.010
407. Sharma M, Manoharlal R, Puri N, and Prasad R. Antifungal curcumin induces reactive oxygen species and triggers an early apoptosis but prevents hyphae development by targeting the global repressor TUP1 in Candida albicans. Biosci Rep. (2010) 30:391–404. doi: 10.1042/BSR20090151
408. Ide H, Tokiwa S, Sakamaki K, Nishio K, Isotani S, Muto S, et al. Combined inhibitory effects of soy isoflavones and curcumin on the production of prostate-specific antigen. Prostate. (2010) 70:1127–33. doi: 10.1002/pros.21147
409. Mathew D and Hsu WL. Antiviral potential of curcumin. J Funct Foods. (2018) 40:692–9. doi: 10.1016/j.jff.2017.12.017
410. Balasubramanian A, Pilankatta R, Teramoto T, Sajith AM, Nwulia E, Kulkarni A, et al. Inhibition of dengue virus by curcuminoids. Antiviral Res. (2019) 162:71–8. doi: 10.1016/j.antiviral.2018.12.002
411. Jeong EH, Vaidya B, Cho SY, Park MA, Kaewintajuk K, Kim SR, et al. Identification of regulators of the early stage of viral hemorrhagic septicemia virus infection during curcumin treatment. Fish Shellfish Immunol. (2015) 45:184–93. doi: 10.1016/j.fsi.2015.03.042
412. Ferreira VH, Nazli A, Dizzell SE, Mueller K, and Kaushic C. The anti-inflammatory activity of curcumin protects the genital mucosal epithelial barrier from disruption and blocks replication of HIV-1 and HSV-2. PloS One. (2015) 10:e0124903. doi: 10.1371/journal.pone.0124903
413. Li H, Zhong C, Wang Q, Chen W, and Yuan Y. Curcumin is an APE1 redox inhibitor and exhibits an antiviral activity against KSHV replication and pathogenesis. Antiviral Res. (2019) 167:98–103. doi: 10.1016/j.antiviral.2019.04.011
414. Mounce BC, Cesaro T, Carrau L, Vallet T, and Vignuzzi M. Curcumin inhibits Zika and chikungunya virus infection by inhibiting cell binding. Antiviral Res. (2017) 142:148–57. doi: 10.1016/j.antiviral.2017.03.014
415. Babaei F, Nassiri-Asl M, and Hosseinzadeh H. Curcumin (a constituent of turmeric): New treatment option against COVID-19. Food Sci Nutr. (2020) 8:5215–27. doi: 10.1002/fsn3.1858
416. Soni VK, Mehta A, Ratre YK, Tiwari AK, Amit A, Singh RP, et al. Curcumin, a traditional spice component, can hold the promise against COVID-19? Eur J Pharmacol. (2020) 886:173551. doi: 10.1016/j.ejphar.2020.173551
417. Alkafaas SS, Abdallah AM, Ghosh S, Loutfy SA, Elkafas SS, Abdel Fattah NF, et al. Insight into the role of clathrin-mediated endocytosis inhibitors in SARS-CoV-2 infection. Rev Med Virol. (2023) 33:e2403. doi: 10.1002/rmv.2403
418. Alkafaas SS, Abdallah AM, Hussien AM, Bedair H, Abdo M, Ghosh S, et al. A study on the effect of natural products against the transmission of B.1.1.529 Omicron. Virol J. (2023) 20:191. doi: 10.1186/s12985-023-02160-6
419. Subhan F, Khalil AAK, Zeeshan M, Haider A, Tauseef I, Haleem SK, et al. Curcumin: from ancient spice to modern anti-viral drug in COVID-19 pandemic. Life Sci. (2020) 1:69–73. doi: 10.37185/LnS.1.1.137
420. Thimmulappa RK, Mudnakudu-Nagaraju KK, Shivamallu C, Subramaniam KJT, Radhakrishnan A, Bhojraj S, et al. Antiviral and immunomodulatory activity of curcumin: A case for prophylactic therapy for COVID-19. Heliyon. (2021) 7:e06350. doi: 10.1016/j.heliyon.2021.e06350
421. Valizadeh H, Abdolmohammadi-Vahid S, Danshina S, Gencer MZ, Ammari A, Sadeghi A, et al. Nano-curcumin therapy, a promising method in modulating inflammatory cytokines in COVID-19 patients. Int Immunopharmacol. (2020) 89:107088. doi: 10.1016/j.intimp.2020.107088
422. Zaman R, Ravichandran V, and Tan CK. Role of dietary supplements in the continuous battle against COVID-19. Phytother Res. (2024) 38:1071–88. doi: 10.1002/ptr.8096
423. Nittayananta W, Lerdsamran H, Chutiwitoonchai N, Promsong A, Srichana T, Netsomboon K, et al. A novel film spray containing curcumin inhibits SARS-CoV-2 and influenza virus infection and enhances mucosal immunity. Virol J. (2024) 21:26. doi: 10.1186/s12985-023-02282-x
424. Taha AE, Amer I, Sharawy SE, and Ghazy AA. Estimation of anti-SARS-CoV-2 IgM/IgG seroprevalence among non-vaccinated and vaccinated university students: a cross-sectional Egyptian study. Viruses. (2025) 17:378. doi: 10.3390/v17030378
425. Rajak P, Chakraborty A, Dutta S, Banerjee D, Adhikary S, Bhattacharya S, et al. Antiallergic implications of curcumin during COVID-19: current status and perspectives. In: Gantait S, Majumder J, and Sharangi AB, editors. Biotechnology of Medicinal Plants with Antiallergy Properties: Research Trends and Prospects. Springer Nature Singapore, Singapore (2024). pp. 155–73. doi: 10.1007/978-981-97-1467-4_4
426. Najimi N, Amssayef A, Bouadid I, Hakmi M, Festali R, Kadi C, et al. Phytochemical inhibitors of SARS-CoV-2 entry: targeting the ACE2-RBD interaction with L-tartaric acid, L-ascorbic acid, and Curcuma longa extract. ChemistrySelect. (2025) 10:e202406035. doi: 10.1002/slct.202406035
427. Anggakusuma, Colpitts CC, Schang LM, Rachmawati H, Frentzen A, Pfaender S, et al. Turmeric curcumin inhibits entry of all hepatitis C virus genotypes into human liver cells. Gut. (2014) 63:1137–49. doi: 10.1136/gutjnl-2012-304299
428. Kim KJ, Kim KH, Kim HY, Cho HK, Sakamoto N, and Cheong J. Curcumin inhibits hepatitis C virus replication via suppressing the Akt-SREBP-1 pathway. FEBS Lett. (2010) 584:707–12. doi: 10.1016/j.febslet.2009.12.019
429. Prasad S and Tyagi AK. Curcumin and its analogues: a potential natural compound against HIV infection and AIDS. Food Funct. (2015) 6:3412–9. doi: 10.1039/C5FO00485C
430. Kumari N, Kulkarni AA, Lin X, McLean C, Ammosova T, Ivanov A, et al. Inhibition of HIV-1 by curcumin A, a novel curcumin analog. Drug Des Devel Ther. (2015) 9:5051–60. doi: 10.2147/DDDT.S86558
431. Pacho MN, Pugni EN, Díaz Sierra JB, Morell ML, Sepúlveda CS, Damonte EB, et al. Antiviral activity against Zika virus of a new formulation of curcumin in poly lactic-co-glycolic acid nanoparticles. J Pharm Pharmacol. (2021) 73:357–65. doi: 10.1093/jpp/rgaa045
432. Ardebili A, Pouriayevali MH, Aleshikh S, Zahani M, Ajorloo M, Izanloo A, et al. Antiviral therapeutic potential of curcumin: an update. Molecules. (2021) 26:6994. doi: 10.3390/molecules26226994
433. Chen DY, Shien JH, Tiley L, Chiou SS, Wang SY, Chang TJ, et al. Curcumin inhibits influenza virus infection and hemagglutination activity. Food Chem. (2010) 119:1346–51. doi: 10.1016/j.foodchem.2009.09.011
434. Zandi K, Ramedani E, Mohammadi K, Tajbakhsh S, Deilami I, Rastian Z, et al. Evaluation of antiviral activities of curcumin derivatives against HSV-1 in Vero cell line. Nat Prod Commun. (2010) 5:1935–8. doi: 10.1177/1934578X1000501220
435. Flores DJ, Lee LH, and Adams SD. Inhibition of curcumin-treated herpes simplex virus 1 and 2 in Vero cells. Adv Microbiol. (2016) 6:276–87. doi: 10.4236/aim.2016.64027
436. Marín-Palma D, Tabares-Guevara JH, Zapata-Cardona MI, Flórez-Álvarez L, Yepes LM, Rugeles MT, et al. Curcumin inhibits in vitro SARS-CoV-2 infection in Vero E6 cells through multiple antiviral mechanisms. Molecules. (2021) 26:6900. doi: 10.3390/molecules26226900
437. Ichsyani M, Ridhanya A, Risanti M, Desti H, Ceria R, Putri DH, et al. Antiviral effects of Curcuma longa L. against dengue virus in vitro and in vivo. IOP Conf Ser Earth Environ Sci. (2017) 101:12005. doi: 10.1088/1755-1315/101/1/012005
438. Kim HJ, Yoo HS, Kim JC, Park CS, Choi MS, Min JS, et al. Antiviral effect of Curcuma longa Linn extract against hepatitis B virus replication. J Ethnopharmacol. (2009) 124:189–96. doi: 10.1016/j.jep.2009.04.046
439. Mishra A and Das BC. Curcumin as an anti-human papillomavirus and anti-cancer compound. Future Oncol. (2015) 11:2487–90. doi: 10.2217/fon.15.166
440. Divya CS and Pillai MR. Antitumor action of curcumin in human papillomavirus associated cells involves downregulation of viral oncogenes, prevention of NFkB and AP-1 translocation, and modulation of apoptosis. Mol Carcinog. (2006) 45:320–32. doi: 10.1002/mc.20170
441. Huang HI, Chio CC, and Lin JY. Inhibition of EV71 by curcumin in intestinal epithelial cells. PloS One. (2018) 13:e0191617. doi: 10.1371/journal.pone.0191617
442. Qin Y, Lin L, Chen Y, Wu S, Si X, Wu H, et al. Curcumin inhibits the replication of enterovirus 71 in vitro. Acta Pharm Sin B. (2014) 4:284–94. doi: 10.1016/j.apsb.2014.06.006
443. Si X, Wang Y, Wong J, Zhang J, McManus BM, and Luo H. Dysregulation of the ubiquitin-proteasome system by curcumin suppresses coxsackievirus B3 replication. J Virol. (2007) 81:3142–50. doi: 10.1128/jvi.02028-06
444. Lin YJ, Wang LC, Tsai HP, Chi CY, Chang CP, Chen SH, et al. Antiviral and immunoregulatory effects of curcumin on coxsackievirus B3-infected hepatitis. Virus Res. (2023) 336:199203. doi: 10.1016/j.virusres.2023.199203
445. Baikerikar S. Curcumin and natural derivatives inhibit Ebola viral proteins: An in silico approach. Pharmacogn Res. (2017) 9:S15–22. doi: 10.4103/pr.pr_30_17
446. Karkute SG, Mishra R, Rashmi M, and Singh M. A bioinformatics approach to target Ebola virus with curcumin. In: Rao RK and Tripathi JS, editors. Recent Advances on the Role of Basic Sciences in Ayurvedic Medicine. Mahima Publications, Varanasi (2014). pp. 1–14.
447. Adepoju AJ, Latona DF, Olafare OG, Oyebamiji AK, Abdul-Hammed M, and Semire B. Molecular docking and pharmacokinetics studies of (curcumin) potency against Ebola virus. Ovidius Univ Ann Chem. (2022) 33:22–35. doi: 10.2478/auoc-2022-0004
448. Yang M, Lee GJ, Si J, Lee SJ, You HJ, and Ko GP. Curcumin shows antiviral properties against norovirus. Molecules. (2016) 21:1401. doi: 10.3390/molecules21101401
449. Sarowska J, Wojnicz D, Jama-Kmiecik A, Frej-Mądrzak M, and Choroszy-Król I. Antiviral potential of plants against noroviruses. Molecules. (2021) 26:4669. doi: 10.3390/molecules26154669
450. Lv Y, Lei N, Wang D, An Z, Li G, Han F, et al. Protective effect of curcumin against cytomegalovirus infection in Balb/c mice. Environ Toxicol Pharmacol. (2014) 37:1140–7. doi: 10.1016/j.etap.2014.04.017
451. Lv YL, Lan AJ, Lan YY, Chao L, Ning L, Cheng W, et al. Activity of curcumin against human cytomegalovirus in vitro. Afr J Pharm Pharmacol. (2012) 6:30–5. doi: 10.5897/AJPP11.439
452. Cui L, Miao J, and Cui L. Cytotoxic effect of curcumin on malaria parasite Plasmodium falciparum: inhibition of histone acetylation and generation of reactive oxygen species. Antimicrob Agents Chemother. (2007) 51:488–94. doi: 10.1128/aac.01238-06
453. Saleheen D, Ali SA, Ashfaq K, Siddiqui AA, Agha A, and Yasinzai MM. Latent activity of curcumin against leishmaniasis in vitro. Biol Pharm Bull. (2002) 25:386–9. doi: 10.1248/bpb.25.386
454. Mallo N, Lamas J, Sueiro RA, and Leiro JM. Molecular targets implicated in the antiparasitic and anti-inflammatory activity of the phytochemical curcumin in Trichomoniasis. Molecules. (2020) 25:5321. doi: 10.3390/molecules25225321
455. Rangel-Castañeda IA, Hernández-Hernández JM, Pérez-Rangel A, González-Pozos S, Carranza-Rosales P, Charles-Niño CL, et al. Amoebicidal activity of curcumin on Entamoeba histolytica trophozoites. J Pharm Pharmacol. (2018) 70:426–33. doi: 10.1111/jphp.12867
456. El-Shafey AAM, Hegab MHA, Seliem MME, Barakat AMA, Mostafa NE, Abdel-Maksoud HA, et al. Curcumin@metal-organic frameworks nano-composite for treatment of chronic toxoplasmosis. J Mater Sci Mater Med. (2020) 31:1–13. doi: 10.1007/s10856-020-06429-y
457. Qian W, Wang H, Shan D, Li B, Liu J, and Liu Q. Activity of several kinds of drugs against Neospora caninum. Parasitol Int. (2015) 64:597–602. doi: 10.1016/j.parint.2015.08.002
458. Pérez-Arriaga L, Mendoza-Magana ML, Cortés-Zárate R, Corona-Rivera A, Bobadilla-Morales L, Troyo-Sanromán R, et al. Cytotoxic effect of curcumin on Giardia lamblia trophozoites. Acta Trop. (2006) 98:152–61. doi: 10.1016/j.actatropica.2006.03.005
459. Nandakumar DN, Nagaraj VA, Vathsala PG, Rangarajan P, and Padmanaban G. Curcumin-artemisinin combination therapy for malaria. Antimicrob Agents Chemother. (2006) 50:1859–60. doi: 10.1128/aac.50.5.1859-1860.2006
460. Martinelli A, Rodrigues LA, and Cravo P. Plasmodium chabaudi: efficacy of artemisinin+curcumin combination treatment on a clone selected for artemisinin resistance in mice. Exp Parasitol. (2008) 119:304–7. doi: 10.1016/j.exppara.2008.02.011
461. da Silva CC, Pacheco BS, das Neves RN, Alves MSD, Sena-Lopes A, Moura S, et al. Antiparasitic activity of synthetic curcumin monocarbonyl analogs against Trichomonas vaginalis. BioMed Pharmacother. (2019) 111:367–77. doi: 10.1016/j.biopha.2018.12.058
462. Khanra S, Kumar YP, Dash J, and Banerjee R. In vitro screening of known drugs identified by scaffold hopping techniques shows promising leishmanicidal activity for suramin and netilmicin. BMC Res Notes. (2018) 11:319. doi: 10.1186/s13104-018-3446-y
463. Bazh EKA and El-Bahy NM. In vitro and in vivo screening of anthelmintic activity of ginger and curcumin on Ascaridia galli. Parasitol Res. (2013) 112:3679–86. doi: 10.1007/s00436-013-3541-x
464. El-Bahy NM and Bazh EKA. Anthelmintic activity of ginger, curcumin, and praziquantel against Raillietina cesticillus (in vitro and in vivo). Parasitol Res. (2015) 114:2427–34. doi: 10.1007/s00436-015-4416-0
465. Novaes RD, Sartini MVP, Rodrigues JPF, Gonçalves RV, Santos EC, Souza RLM, et al. Curcumin enhances the anti-Trypanosoma cruzi activity of benznidazole-based chemotherapy in acute experimental Chagas disease. Antimicrob Agents Chemother. (2016) 60:3355–64. doi: 10.1128/aac.00343-16
466. Busari ZA, Dauda KA, Morenikeji OA, Afolayan F, Oyeyemi OT, Meena J, et al. Antiplasmodial activity and toxicological assessment of curcumin PLGA-encapsulated nanoparticles. Front Pharmacol. (2017) 8:622. doi: 10.3389/fphar.2017.00622
467. Kausar N, Shier WT, Ahmed M, Albekairi NA, Alshammari A, Saleem M, et al. Investigation of the insecticidal potential of curcumin derivatives that target the Helicoverpa armigera sterol carrier protein-2. Heliyon. (2024) 10:e29695. doi: 10.1016/j.heliyon.2024.e29695
468. Ekawardhani S and Berbudi A. The role of curcumin as an antimalarial agent. Syst Rev Pharm. (2020) 11:18–25.
469. Olanlokun JO, Abiodun WO, Ebenezer O, Koorbanally NA, and Olorunsogo OO. Curcumin modulates multiple cell death, matrix metalloproteinase activation and cardiac protein release in susceptible and resistant Plasmodium berghei-infected mice. BioMed Pharmacother. (2022) 146:112454. doi: 10.1016/j.biopha.2021.112454
470. Shakib P, Kalani H, Ho J, Dolatshah M, Amiri S, and Cheraghipour K. A systematic review on curcumin and anti-Plasmodium berghei effects. Curr Drug Discov Technol. (2022) 19:67–72. doi: 10.2174/1570163819666220315140736
471. Marcolino LMC, Pereira AHC, Pinto JG, Mamone LA, and Strixino JF. Cellular and metabolic changes after photodynamic therapy in Leishmania promastigotes. Photodiagnosis Photodyn Ther. (2021) 35:102403. doi: 10.1016/j.pdpdt.2021.102403
472. Elamin M, Al-Olayan E, Abdel-Gaber R, and Yehia RS. Anti-proliferative and apoptosis induction activities of curcumin on Leishmania major. Rev Argent Microbiol. (2021) 53:240–7. doi: 10.1016/j.ram.2020.08.004
473. Di Chio C, Previti S, Totaro N, De Luca F, Allegra A, Schirmeister T, et al. Dipeptide nitrile CD34 with curcumin: A new improved combination strategy to synergistically inhibit rhodesain of Trypanosoma brucei rhodesiense. Int J Mol Sci. (2023) 24:8477. doi: 10.3390/ijms24108477
474. Adeyemi OS, Ishii K, and Kato K. The in vitro anti-parasitic activities of emodin toward Toxoplasma gondii. Pharmaceuticals. (2023) 16:447. doi: 10.3390/ph16030447
475. Rangel-Castañeda IA, Carranza-Rosales P, Guzmán-Delgado NE, Hernández-Hernández JM, González-Pozos S, Pérez-Rangel A, et al. Curcumin attenuates the pathogenicity of Entamoeba histolytica by regulating the expression of virulence factors in an ex-vivo model infection. Pathogens. (2019) 8:127. doi: 10.3390/pathogens8030127
476. de Paula Aguiar D, Moscardini MBM, Morais ER, de Paula RG, Ferreira PM, Afonso A, et al. Curcumin generates oxidative stress and induces apoptosis in adult Schistosoma mansoni worms. PloS One. (2016) 11:e0167135. doi: 10.1371/journal.pone.0167135
477. Rahman SU, Zhou K, Zhou SS, Sun T, Mi R, Huang Y, et al. Curcumin mitigates Cryptosporidium parvum infection through modulation of gut microbiota and innate immune-related genes in immunosuppressed neonatal mice. Microb Pathog. (2022) 164:105424. doi: 10.1016/j.micpath.2022.105424
478. Kumar R, Harilal S, Gautam A, Nigam M, and Mishra AP. Phytopharmaceuticals as an alternative treatment against parasites. In: Nigam M and Mishra AP, editors. Parasitic Infections. John Wiley & Sons, Inc. Hoboken, New Jersey, NJ (2023). pp. 251–302. doi: 10.1002/9781119878063.ch12
479. Chithra A, Dhivya K, Suresh AR, and Balasubramaniyan M. Phytocompounds as therapeutic agents against neglected tropical diseases. In: Radhakrishnan N, Vasantha S, and Pandurangan AK, editors. Pharmacological Benefits of Natural Agents. IGI Global, Hershey, Pennsylvania, USA (2023). pp. 200–24. doi: 10.4018/978-1-6684-6737-4.ch012
480. Pivari F, Mingione A, Brasacchio C, and Soldati L. Curcumin and type 2 Diabetes mellitus: prevention and treatment. Nutrients. (2019) 11:1837. doi: 10.3390/nu11081837
481. Vaithiyalingam M, Sumathi DL, and Sabarathinam S. Isolation and in silico study of curcumin from Curcuma longa and its anti-diabetic activity. Appl Biochem Biotechnol. (2023) 195:947–57. doi: 10.1007/s12010-022-04173-3
482. Sharma S, Kulkarni SK, and Chopra K. Curcumin, the active principle of turmeric (Curcuma longa), ameliorates diabetic nephropathy in rats. Clin Exp Pharmacol Physiol. (2006) 33:940–5. doi: 10.1111/j.1440-1681.2006.04468.x
483. Kim BH, Lee ES, Choi R, Nawaboot J, Lee MY, Lee EY, et al. Protective effects of curcumin on renal oxidative stress and lipid metabolism in a rat model of type 2 diabetic nephropathy. Yonsei Med J. (2016) 57:664–73. doi: 10.3349/ymj.2016.57.3.664
484. Ran Z, Zhang Y, Wen X, and Ma J. Curcumin inhibits high glucose-induced inflammatory injury in human retinal pigment epithelial cells through the ROS-PI3K/AKT/mTOR signaling pathway. Mol Med Rep. (2019) 19:1024–31. doi: 10.3892/mmr.2018.9749
485. Bajda M, Guzior N, Ignasik M, and Malawska B. Multi-target-directed ligands in Alzheimer’s disease treatment. Curr Med Chem. (2011) 18:4949–75. doi: 10.2174/092986711797535245
486. Zhang HA and Kitts DD. Turmeric and its bioactive constituents trigger cell signaling mechanisms that protect against diabetes and cardiovascular diseases. Mol Cell Biochem. (2021) 476:3785–814. doi: 10.1007/s11010-021-04201-6
487. Alkafaas SS, Elsalahaty MI, Ismail DF, Radwan MA, Elkafas SS, Loutfy SA, et al. The emerging roles of sphingosine 1-phosphate and SphK1 in cancer resistance: a promising therapeutic target. Cancer Cell Int. (2024) 24:89. doi: 10.1186/s12935-024-03221-8
488. Alkafaas SS, Diab T, Shalaby T, and Hessien M. Dexamethasone improves the responsiveness of hepatoma cells for both free and solvent containing paclitaxel in vitro. Egypt J Biochem Mol Biol. (2019) 37:95–110.
489. Alkafaas SS, Loutfy SA, Diab T, and Hessien M. Vasopressin induces apoptosis but does not enhance the antiproliferative effect of dynamin 2 or PI3K/Akt inhibition in luminal A breast cancer cells. Med Oncol. (2022) 40:35. doi: 10.1007/s12032-022-01889-4
490. Alkafaas SS, Obeid OK, Radwan MA, Elsalahaty MI, ElKaffas SS, Hafez W, et al. Novel insight into mitochondrial dynamin-related protein-1 as a new chemo-sensitizing target in resistant cancer cells. Bioorg Chem. (2024) 150:107574. doi: 10.1016/j.bioorg.2024.107574
491. Kunnumakkara AB, Anand P, and Aggarwal BB. Curcumin inhibits proliferation, invasion, angiogenesis and metastasis of different cancers through interaction with multiple cell signaling proteins. Cancer Lett. (2008) 269:199–225. doi: 10.1016/j.canlet.2008.03.009
492. Liang Z, Wu R, Xie W, Zhu M, Xie C, Li X, et al. Curcumin reverses tobacco smoke-induced epithelial-mesenchymal transition by suppressing the MAPK pathway in the lungs of mice. Mol Med Rep. (2018) 17:2019–25. doi: 10.3892/mmr.2017.8028
493. Siviero A, Gallo E, Maggini V, Gori L, Mugelli A, Firenzuoli F, et al. Curcumin, a golden spice with a low bioavailability. J Herb Med. (2015) 5:57–70. doi:10.1016/j.hermed.2015.03.001
494. Kunnumakkara AB, Harsha C, Banik K, Vikkurthi R, Sailo BL, Bordoloi D, et al. Is curcumin bioavailability a problem in humans: Lessons from clinical trials. Expert Opin Drug Metab Toxicol. (2019) 15:705–33. doi: 10.1080/17425255.2019.1650914
495. Farghadani R and Naidu R. Curcumin: modulator of key molecular signaling pathways in hormone-independent breast cancer. Cancers. (2021) 13:3427. doi: 10.3390/cancers13143427
496. Wang H, Zhang K, Liu J, Yang J, Tian Y, Yang C, et al. Curcumin regulates cancer progression: Focus on ncRNAs and molecular signaling pathways. Front Oncol. (2021) 11:660712. doi: 10.3389/fonc.2021.660712
497. Ismail NI, Othman I, Abas F, Lajis NH, and Naidu R. Mechanism of apoptosis induced by curcumin in colorectal cancer. Int J Mol Sci. (2019) 20:2454. doi: 10.3390/ijms20102454
498. Brockmueller A, Ruiz de Porras V, and Shakibaei M. Curcumin and its anti-colorectal cancer potential: From mechanisms of action to autophagy. Phytother Res. (2024) 38:3525–51. doi: 10.1002/ptr.8220
499. Liu J, Li M, Wang Y, and Luo J. Curcumin sensitizes prostate cancer cells to radiation partly via epigenetic activation of miR-143 and miR-143 mediated autophagy inhibition. J Recept Signal Transduct Res. (2017) 25:645–52. doi: 10.1080/1061186X.2017.1315686
500. Dai X, Zhang J, Guo G, Cai Y, Cui R, Yin C, et al. A mono-carbonyl analog of curcumin induces apoptosis in drug-resistant EGFR-mutant lung cancer through the generation of oxidative stress and mitochondrial dysfunction. Cancer Manag Res. (2018) 10:3069–82. doi: 10.2147/CMAR.S159660
501. Ashrafizadeh M, Najafi M, Makvandi P, Zarrabi A, Farkhondeh T, and Samarghandian S. Versatile role of curcumin and its derivatives in lung cancer therapy. J Cell Physiol. (2020) 235:9241–68. doi: 10.1002/jcp.29819
502. Kabir MT, Rahman MH, Akter R, Behl T, Kaushik D, Mittal V, et al. Potential role of curcumin and its nanoformulations to treat various types of cancers. Biomolecules. (2021) 11:392. doi: 10.3390/biom11030392
503. Kunnumakkara AB, Bordoloi D, Harsha C, Banik K, Gupta SC, and Aggarwal BB. Curcumin mediates anticancer effects by modulating multiple cell signaling pathways. Clin Sci Lond. (2017) 131:1781–99. doi: 10.1042/CS20160935
504. Li W, Sun L, Lei J, Wu Z, Ma Q, and Wang Z. Curcumin inhibits pancreatic cancer cell invasion and EMT by interfering with tumor−stromal crosstalk under hypoxic conditions via the IL−6/ERK/NF−κB axis. Oncol Rep. (2020) 44:382–92. doi: 10.3892/or.2020.7600
505. Bai C, Zhao J, Su J, Chen J, Cui X, Sun M, et al. Curcumin induces mitochondrial apoptosis in human hepatoma cells through BCLAF1-mediated modulation of PI3K/AKT/GSK-3β signaling. Life Sci. (2022) 306:120804. doi: 10.1016/j.lfs.2022.120804
506. Li W, Zhou Y, Yang J, Li H, Zhang H, and Zheng P. Curcumin induces apoptotic cell death and protective autophagy in human gastric cancer cells. Oncol Rep. (2017) 37:3459–66. doi: 10.3892/or.2017.5637
507. Zhang X, Zhu L, Wang X, Zhang H, Wang L, and Xia L. Basic research on curcumin in cervical cancer: Progress and perspectives. BioMed Pharmacother. (2023) 162:114590. doi: 10.1016/j.biopha.2023.114590
508. Lewinska A, Adamczyk J, Pajak J, Stoklosa S, Kubis B, Pastuszek P, et al. Curcumin-mediated decrease in the expression of nucleolar organizer regions in cervical cancer (HeLa) cells. Mutat Res Genet Toxicol Environ Mutagen. (2014) 771:43–52. doi: 10.1016/j.mrgentox.2014.07.001
509. Mohamadian M, Ahmadi SS, Bahrami A, and Ferns GA. Review on the therapeutic potential of curcumin and its derivatives on glioma biology. Neurochem Res. (2022) 47:2936–53. doi: 10.1007/s11064-022-03666-1
510. Trotta T, Panaro MA, Prifti E, and Porro C. Modulation of biological activities in glioblastoma mediated by curcumin. Nutr Cancer. (2019) 71:1241–53. doi: 10.1080/01635581.2019.1604978
511. Pourhanifeh MH, Mottaghi R, Razavi ZS, Shafiee A, Hajighadimi S, and Mirzaei H. Therapeutic applications of curcumin and its novel formulations in the treatment of bladder cancer: A review of current evidence. Anticancer Agents Med Chem. (2021) 21:587–96. doi: 10.2174/1871520620666200807223832
512. Chandra Pal H, Marchiony Hunt K, Diamond A, Elmets CA, and Afaq F. Phytochemicals for the management of melanoma. Mini Rev Med Chem. (2016) 16:953–79. doi: 10.2174/1389557516666160211120157
513. Shad AN, Fanoodi A, Maharati A, Akhlaghipour I, and Moghbeli M. Molecular mechanisms of microRNA-301a during tumor progression and metastasis. Pathol Res Pract. (2023) 247:154538. doi: 10.1016/j.prp.2023.154538
514. Kaushik M and Tiku A. Therapeutic potential of phytochemicals as adjuvants in head and neck cancer. Nutr Rev. (2025) 83:1552–70. doi: 10.1093/nutrit/nuaf009
515. Hayakawa S, Ohishi T, Oishi Y, Isemura M, and Miyoshi N. Contribution of non-coding RNAs to anticancer effects of dietary polyphenols: chlorogenic acid, curcumin, epigallocatechin-3-gallate, genistein, quercetin and resveratrol. Antioxidants. (2022) 11:2352. doi: 10.3390/antiox11122352
516. Herynk MH and Fuqua SAW. Estrogen receptors in resistance to hormone therapy. In: Yu D and Hung MC, editors. Breast Cancer Chemosensitivity. Advances in Experimental Medicine and Biology, vol. 608 . Springer, New York, NY (2007). pp. 130–43. doi: 10.1007/978-0-387-74039-3_10
517. Shao ZM, Shen ZZ, Liu CH, Sartippour MR, Go VL, Heber D, et al. Curcumin exerts multiple suppressive effects on human breast carcinoma cells. Int J Cancer. (2002) 98:234–40. doi: 10.1002/ijc.10183
518. Calaf GM, Ponce-Cusi R, and Carrión F. Curcumin and paclitaxel induce cell death in breast cancer cell lines. Oncol Rep. (2018) 40:2381–8. doi: 10.3892/or.2018.6603
519. Al-Hujaily EM, Mohamed AG, Al-Sharif I, Youssef KM, Manogaran PS, Al-Otaibi B, et al. PAC, a novel curcumin analogue, has anti-breast cancer properties with higher efficiency on ER-negative cells. Breast Cancer Res Treat. (2011) 128:97–107. doi: 10.1007/s10549-010-1089-3
520. Siegel R, Ma J, Zou Z, and Jemal A. Cancer statistics. CA Cancer J Clin. (2014) 64:9–29. doi: 10.3322/caac.21208
521. Detillon DD and Veen EJ. Postoperative outcome after pulmonary surgery for non-small cell lung cancer in elderly patients. Ann Thorac Surg. (2018) 105:287–93. doi: 10.1016/j.athoracsur.2017.07.032
522. Shishodia S, Potdar P, Gairola CG, and Aggarwal BB. Curcumin (diferuloylmethane) down-regulates cigarette smoke-induced NF-κB activation through inhibition of IκBα kinase in human lung epithelial cells: correlation with suppression of COX-2, MMP-9 and cyclin D1. Carcinogenesis. (2003) 24:1269–79. doi: 10.1093/carcin/bgg078
523. Tsai JR, Liu PL, Chen YH, Chou SH, Cheng YJ, Hwang JJ, et al. Curcumin inhibits non-small cell lung cancer cells metastasis through the adiponectin/NF-κB/MMPs signaling pathway. PloS One. (2015) 10:e0144462. doi: 10.1371/journal.pone.0144462
524. Wu J, Cai Z, Wei X, Chen M, Ying S, Shi L, et al. Anti-lung cancer activity of the curcumin analog JZ534 in vitro. BioMed Res Int. (2015) 2015:504529. doi: 10.1155/2015/504529
525. Clarke MA, Wentzensen N, Mirabello L, Ghosh A, Wacholder S, Harari A, et al. Human papillomavirus DNA methylation as a potential biomarker for cervical cancer. Cancer Epidemiol Biomarkers Prev. (2012) 21:2125–37. doi: 10.1158/1055-9965.EPI-12-0905
526. Momtazi-Borojeni AA, Mosafer J, Nikfar B, Ekhlasi-Hundrieser M, Chaichian S, Mehdizadehkashi A, et al. Curcumin in advancing treatment for gynecological cancers with developed drug- and radiotherapy-associated resistance. In: de Tombe P, Gudermann T, Jahn R, and Lill R, editors. Reviews of Physiology, Biochemistry and Pharmacology. Springer, Cham, Switzerland (2018). pp. 107–29. doi: 10.1007/112_2018_11
527. Singh M and Singh N. Curcumin counteracts the proliferative effect of estradiol and induces apoptosis in cervical cancer cells. Mol Cell Biochem. (2011) 347:1–11. doi: 10.1007/s11010-010-0606-3
528. Adams BK, Ferstl EM, Davis MC, Herold M, Kurtkaya S, Camalier RF, et al. Synthesis and biological evaluation of novel curcumin analogs as anti-cancer and anti-angiogenesis agents. Bioorg Med Chem. (2004) 12:3871–83. doi: 10.1016/j.bmc.2004.05.006
529. Tan X, Sidell N, Mancini A, Huang RP, Wang S, Horowitz IR, et al. Multiple anticancer activities of EF24, a novel curcumin analog, on human ovarian carcinoma cells. Reprod Sci. (2010) 17:931–40. doi: 10.1177/19337191103742
530. Karantanos T, Corn PG, and Thompson TC. Prostate cancer progression after androgen deprivation therapy: mechanisms of castrate resistance and novel therapeutic approaches. Oncogene. (2013) 32:5501–11. doi: 10.1038/onc.2013.206
531. Mahammedi H, Planchat E, Pouget M, Durando X, Curé H, Guy L, et al. The new combination docetaxel, prednisone and curcumin in patients with castration-resistant prostate cancer: a pilot phase II study. Oncology. (2016) 90:69–78. doi: 10.1159/000441148
532. Chen S, Nimick M, Cridge AG, Hawkins BC, and Rosengren RJ. Anticancer potential of novel curcumin analogs towards castrate-resistant prostate cancer. Int J Oncol. (2017) 52:579–88. doi: 10.3892/ijo.2017.4207
533. Tang SC and Chen YC. Novel therapeutic targets for pancreatic cancer. World J Gastroenterol. (2014) 20:10825–1044. doi: 10.3748/wjg.v20.i31.10825
534. Bao B, Ali S, Banerjee S, Wang Z, Logna F, Azmi AS, et al. Curcumin analogue CDF inhibits pancreatic tumor growth by switching on suppressor microRNAs and attenuating EZH2 expression. Cancer Res. (2012) 72:335–45. doi: 10.1158/0008-5472.CAN-11-2182
535. Hutzen B, Friedman L, Sobo M, Lin L, Cen L, De Angelis S, et al. Curcumin analogue GO-Y030 inhibits STAT3 activity and cell growth in breast and pancreatic carcinomas. Int J Oncol. (2009) 35:867–72. doi: 10.3892/ijo_00000401
536. Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, et al. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. (2015) 136:E359–86. doi: 10.1002/ijc.29210
537. Carroll RE, Benya RV, Turgeon DK, Vareed S, Neuman M, Rodriguez L, et al. Phase IIa clinical trial of curcumin for the prevention of colorectal neoplasia. Cancer Prev Res. (2011) 4:354–64. doi: 10.1158/1940-6207.CAPR-10-0098
538. Rao CV, Rivenson A, Simi B, and Reddy BS. Chemoprevention of colon carcinogenesis by dietary curcumin, a naturally occurring plant phenolic compound. Cancer Res. (1995) 55:259–66.
539. Soni D and Salh B. A neutraceutical by design: the clinical application of curcumin in colonic inflammation and cancer. Scientifica. (2012) 2012:757890. doi: 10.6064/2012/757890
540. Palatty PL, Azmidah A, Rao S, Jayachander D, Thilakchand KR, Rai MP, et al. Topical application of a sandal wood oil and turmeric based cream prevents radiodermatitis in head and neck cancer patients undergoing external beam radiotherapy: a pilot study. Br J Radiol. (2014) 87:20130490. doi: 10.1259/bjr.20130490
541. Zhu J, Sanidad KZ, Sukamtoh E, and Zhang G. Potential roles of chemical degradation in the biological activities of curcumin. Food Funct. (2017) 8:907–14. doi: 10.1039/C6FO01770C
542. Pieretti S, Ranjan AP, Di Giannuario A, Mukerjee A, Marzoli F, Di Giovannandrea R, et al. Curcumin-loaded poly (D,L-lactide-co-glycolide) nanovesicles induce antinociceptive effects and reduce pronociceptive cytokine and BDNF release in spinal cord after acute administration in mice. Colloids Surf B Biointerf. (2017) 158:379–86. doi: 10.1016/j.colsurfb.2017.07.027
543. Barchitta M, Maugeri A, Favara G, Magnano San Lio R, Evola G, Agodi A, et al. Nutrition and wound healing: an overview focusing on the beneficial effects of curcumin. Int J Mol Sci. (2019) 20:1119. doi: 10.3390/ijms20051119
544. Belkacemi A, Doggui S, Dao L, and Ramassamy C. Challenges associated with curcumin therapy in Alzheimer disease. Expert Rev Mol Med. (2011) 13:e34. doi: 10.1017/S1462399411002055
545. Chen SY, Chen Y, Li YP, Chen SH, Tan JH, Ou TM, et al. Design, synthesis, and biological evaluation of curcumin analogues as multifunctional agents for the treatment of Alzheimer’s disease. Bioorg Med Chem. (2011) 19:5596–604. doi: 10.1016/j.bmc.2011.07.033
546. Sanjeeva NCG, Sarkar A, Ahmed I, and Pande A. Large non-restrictive apical ventricular septal defect curtailed by anomalous right ventricular muscle band: a rare variant double-chambered right ventricle. J Cardiovasc Dis Res. (2015) 6:28–30. doi: 10.5530/jcdr.2015.1.5
547. Lan Z, Tan F, He J, Liu J, Lu M, Hu Z, et al. Curcumin−primed olfactory mucosa−derived mesenchymal stem cells mitigate cerebral ischemia/reperfusion injury−induced neuronal PANoptosis by modulating microglial polarization. Phytomedicine. (2024) 129:155635. doi: 10.1016/j.phymed.2024.155635
548. Jyotirmayee B and Mahalik G. A review on selected pharmacological activities of Curcuma longa L. Int J Food Prop. (2022) 25:1377–98. doi: 10.1080/10942912.2022.2082464
549. Vollono L, Falconi M, Gaziano R, Iacovelli F, Dika E, Terracciano C, et al. Potential of curcumin in skin disorders. Nutrients. (2019) 11:2169. doi: 10.3390/nu11092169
550. Lovászi M, Szegedi A, Zouboulis CC, and Törőcsik D. Sebaceous-immunobiology is orchestrated by sebum lipids. Dermatoendocrinol. (2017) 9:e1375636. doi: 10.1080/19381980.2017.1375636
551. Püttgen KB and Cohen BA. Neonatal dermatology. In: Cohen BA, editor. Pediatric Dermatology, 4th ed, vol. . p . Elsevier Saunders Inc, Philadelphia, USA (2013). pp. 29–31.
552. Blagov A, Sukhorukov V, Guo S, Zhang D, Eremin I, and Orekhov A. The role of oxidative stress in the induction and development of psoriasis. Front Biosci. (2023) 28:118. doi: 10.31083/j.fbl2806118
553. Zhang S, Wang J, Liu L, Sun X, Zhou Y, Chen S, et al. Efficacy and safety of curcumin in psoriasis: preclinical and clinical evidence and possible mechanisms. Front Pharmacol. (2022) 13:903160. doi: 10.3389/fphar.2022.903160
554. Idreess HG, Saed ES, Radhi OA, Alqaseer KA, Shnain WD, AL-Roubaey DAA, et al. Turmeric: An important medicinal plant in the treatment of many diseases: A review study. Kufa J Nurs Sci. (2023) 13:229–37. doi: 10.36321/kjns.vi20232.13720
555. Di Lorenzo R, Forgione F, Bernardi A, Sacchi A, Laneri S, and Greco G. Curcumin as a topical agent in clinical studies. Skin Pharmacol Physiol. (2023) 36:235–48. doi: 10.1159/000535100
556. Lara-Espinosa JV, Arce-Aceves MF, López-Torres MO, Lozano-Ordaz V, Mata-Espinosa D, Barrios-Payán J, et al. Effect of curcumin in experimental pulmonary tuberculosis: Antimycobacterial activity in the lungs and anti-inflammatory effect in the brain. Int J Mol Sci. (2022) 23:1964. doi: 10.3390/ijms23041964
557. Khdair SA, Abdulridha MK, and Shafek MA. The effect of curcumin adjuvant therapy on pulmonary function and levels of interleukin-6 (IL-6) and superoxide dismutase-3 (EC-SOD3) in patients with chronic bronchial asthma. Indones J Pharm. (2021) 32:232–40.
Keywords: bioavailability, Curcuma longa, medicinal properties, pharmacological actions, traditional medicine, turmeric
Citation: El-Saadony MT, Saad AM, Mohammed DM, Alkafaas SS, Ghosh S, Negm SH, Salem HM, Fahmy MA, Mosa WFA, Ibrahim EH, AbuQamar SF and El-Tarabily KA (2025) Curcumin, an active component of turmeric: biological activities, nutritional aspects, immunological, bioavailability, and human health benefits - a comprehensive review. Front. Immunol. 16:1603018. doi: 10.3389/fimmu.2025.1603018
Received: 31 March 2025; Accepted: 04 July 2025;
Published: 21 August 2025.
Edited by:
Wenlong Wang, Pingdingshan University, ChinaReviewed by:
Muthukumar Serva Peddha, Central Food Technological Research Institute (CSIR), IndiaMahmood Ahmed, University of Education Lahore, Pakistan
Lu Wang, Zhejiang Chinese Medical University, China
Copyright © 2025 El-Saadony, Saad, Mohammed, Alkafaas, Ghosh, Negm, Salem, Fahmy, Mosa, Ibrahim, AbuQamar and El-Tarabily. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Synan F. AbuQamar, c2FidXFhbWFyQHVhZXUuYWMuYWU=; Khaled A. El-Tarabily, a3RhcmFiaWx5QHVhZXUuYWMuYWU=
 Mohamed T. El-Saadony
Mohamed T. El-Saadony Ahmed M. Saad
Ahmed M. Saad Dina Mostafa Mohammed
Dina Mostafa Mohammed Samar Sami Alkafaas
Samar Sami Alkafaas Soumya Ghosh
Soumya Ghosh Shaimaa H. Negm
Shaimaa H. Negm Heba M. Salem
Heba M. Salem Mohamed A. Fahmy
Mohamed A. Fahmy Walid F. A. Mosa
Walid F. A. Mosa Essam H. Ibrahim
Essam H. Ibrahim Synan F. AbuQamar
Synan F. AbuQamar Khaled A. El-Tarabily
Khaled A. El-Tarabily