- 1Department of Medical Laboratory, Jiangsu Provincial People’s Hospital Chongqing Hospital, Chongqing, China
- 2Department of Medical Laboratory, Qijiang District People’s Hospital, Chongqing, China
- 3Department of Public Health, Jiangsu Provincial People’s Hospital Chongqing Hospital, Chongqing, China
- 4Department of Intensive Care Medicine, Jiangsu Provincial People’s Hospital Chongqing Hospital, Chongqing, China
The tumor microenvironment (TME) is a complex ecosystem and cancer-associated fibroblasts (CAFs) are critical drivers of the immunosuppressive TME. The dynamic interactions between CAFs and immune cells play a crucial role in tumor progression and immune evasion. This review systematically investigates the interactions between CAFs and different immune cells and elaborates on the molecular mechanisms of CAF-mediated immune suppression, with a focus on their multifaceted interactions with various immune cell populations. The present study discusses how CAFs utilize cytokine networks, metabolic reprogramming and immune checkpoint regulation to establish an immunosuppressive TME. Clinical translation should prioritize FAP-directed therapies alongside αPD-1 to concurrently target CAF-immune crosstalk and metabolic competition in the TME.
1 Introduction
Tumor immune escape represents one of the critical biological characteristics enabling the initiation, progression and metastasis of malignant tumors (1). Under normal physiological conditions, the body’s immune system identifies and eliminates abnormal cells through sophisticated multi-layered defense mechanisms, which is called “immune surveillance” (2). However, cancer cells have developed multiple complex mechanisms to evade recognition and attack by the immune system, a capability termed “immune escape” (3). From a molecular perspective, tumor immune escape primarily involves three key aspects: defective antigen presentation, formation of an immunosuppressive microenvironment, and activation of immune checkpoints (4). Cancer remains one of the leading causes of death worldwide, despite significant advancements in treatment modalities such as chemotherapy, radiation therapy and immunotherapy (5, 6). One of the major challenges in effectively treating cancer is immune evasion (7). Immune evasion not only facilitates tumor progression but also limits the efficacy of immunotherapies, which are designed to enhance the body’s natural defenses against cancer (8, 9).
The tumor microenvironment (TME) is a complex ecosystem comprising cancer cells, immune cells, and stromal cells such as cancer-associated fibroblasts (CAFs) (10, 11). CAFs are the most abundant stromal cells in the TME and have significant impacts on tumor growth, invasion and drug resistance (12). Recent studies have also shown that CAFs play a crucial role in maintaining the anti-tumor immune response (13). Through direct cell-cell interactions and paracrine signaling, CAFs modulate the function of various immune cells, including T cells, macrophages, and myeloid-derived suppressor cells (MDSCs), fostering an immunosuppressive milieu (14). This review discusses the key molecular mechanisms by which CAFs facilitate immune escape, which provides insights into potential therapeutic strategies to counteract CAF-mediated immunosuppression in cancer.
2 CAFs and their subtypes
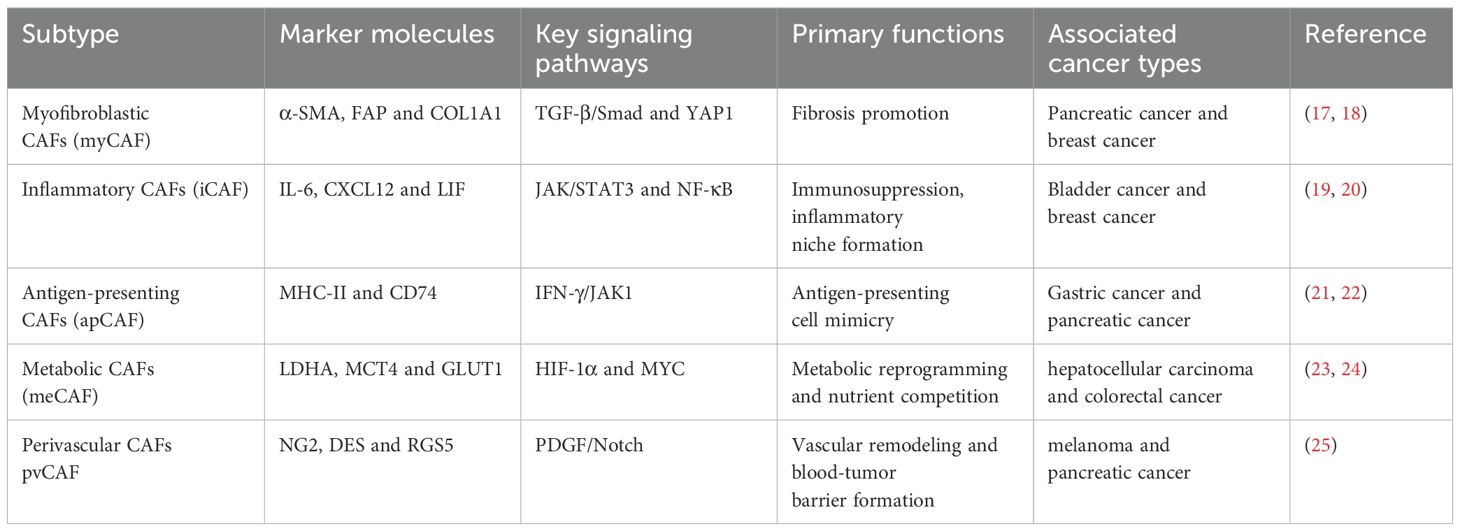
CAFs are a diverse group of cells with distinct phenotypes and functions (15). CAFs lack a universal marker but are commonly identified by the expression of α-smooth muscle actin (α-SMA, encoded by acta2), fibroblast activation protein (FAP), platelet-derived growth factor receptors (PDGFRα/β), vimentin, and fibronectin (16). Based on transcriptomic and functional analyses, CAFs can be broadly categorized into several major subtypes (Table 1). Emerging spatial transcriptomic studies reveal that these subtypes exhibit distinct spatial niches within tumors, with FAP+ myCAFs predominantly localizing to collagen-rich invasive fronts while α-SMA+ iCAFs cluster near angiogenic vasculature, illustrating their specialized roles in stromal remodeling versus immune modulation (26).
3 CAFs-Immune cells interactions and immune evasion
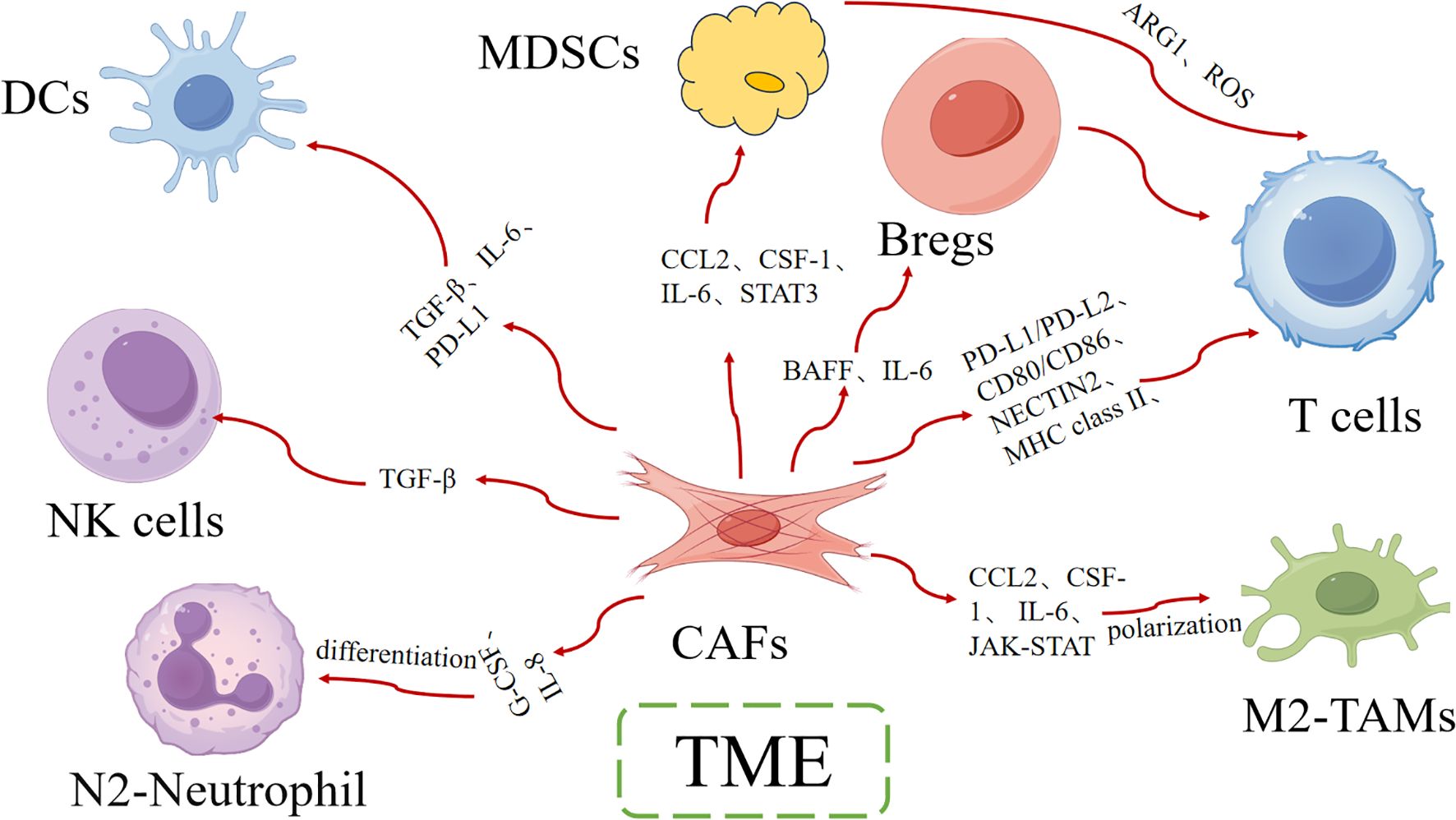
Some studies suggest that CAFs dynamically regulated antitumor immunity through multifaceted interactions with immune cells, creating an immunosuppressive microenvironment that promotes therapy resistance (27). Understanding how CAFs interact with immune cells is essential for developing more effective cancer therapies (Figure 1).
3.1 CAFs and T cells
CAFs, as the predominant stromal cell component in the TME, shape a highly immunosuppressive microenvironment through multidimensional interactions with T cells, thereby promoting tumor immune evasion and therapeutic resistance (28, 29). These interactions involve multiple mechanisms, including direct cell-to-cell contact, secretion of soluble factors, metabolic reprogramming, and the formation of physical barriers (30, 31). Their complexity and dynamics have become a major focus in current cancer immunotherapy research.
CAFs directly regulate T cell functions. CAFs induce T cell exhaustion by upregulating co-inhibitory molecules such as PD-L1/PD-L2 and CD80/CD86, which bind to receptors including PD-1 and CTLA-4 on T cell surfaces (32–34). A single-cell sequencing study reveal that approximately 40% of CAFs in pancreatic cancer highly express PD-L2, which exhibits 30% higher binding affinity to PD-1 compared to PD-L1 (35). Recent mechanistic studies using 3D CAF-T cell coculture systems have demonstrated that CAF-derived NECTIN2 directly engages CD226 on CD8+ T cells, inducing caspase-3-dependent apoptosis. CRISPR-mediated knockout of NECTIN2 in pancreatic CAFs reduced T cell apoptosis by 67% in vitro (13). Flow cytometry analysis revealed that TNBC-derived CAFs exhibit 3.2-fold higher surface NECTIN2 expression compared to normal mammary fibroblasts (p<0.001), suggesting tumor-specific regulation of this pathway. Building upon these established mechanisms, the novel single-cell RNA-seq data identified IGFBP5 as a key upstream regulator of NECTIN2 expression in metastatic CAF subsets (26). Furthermore, certain CAF subsets, such as apCAFs, aberrantly express MHC class II molecules, driving CD4+ T cell differentiation toward regulatory T cells rather than effector T cells (21, 26). In the lung cancer microenvironment, this “pseudo-antigen presentation” can increase regulatory T cell (Treg) proportions by 3- to 5-fold (21).
CAFs further suppress T cell function through soluble factor-mediated paracrine signaling mechanisms. As a master regulator of immune homeostasis, transforming growth factor-beta (TGF-β) plays a multifaceted role in tumor immunology, with CAFs constituting the predominant cellular source of this cytokine in the TME (36–39). TGF-β signaling induces profound dysfunction in CD8+ cytotoxic T lymphocytes through inducing the epigenetic silencing of effector molecules, the transcriptional downregulation of EOMES and TBX21 (the critical transcription factors for cytotoxic differentiation) and the inhibition of mitochondrial oxidative phosphorylation (40–43).
In addition to the aforementioned mechanisms, CAFs can also influence T cell-mediated immune responses by affecting T cell metabolism and remodeling the ECM. For instance, CAFs significantly reduce the glucose concentration in the TME to one-tenth of that in normal tissues by overexpressing glucose transporter 1 and hexokinase 2, which notably decreased the proliferation of T cells and inhibited the mTOR signaling pathway within these cells (44). Moreover, CAFs reshape the ECM through the secretion of matrix-degrading enzymes, leading to a dense fibrotic stroma that physically impedes T cell infiltration and interaction with tumor cells (45). Altogether, these multifaceted interactions highlight the complex role of CAFs in modulating the tumor immune landscape and underscore the need for targeted strategies to disrupt their immunosuppressive functions.
3.2 CAFs and tumor-associated macrophages
Beyond their multifaceted immunosuppressive effects on T cells, CAFs also establish critical crosstalk with tumor-associated macrophages (TAMs), forming another key immunosuppressive axis in the tumor microenvironment. The crosstalk between CAFs and TAMs constitutes a critical axis in tumor immune evasion, creating a profoundly immunosuppressive microenvironment (46). CAFs secrete key chemokines and cytokines, including CCL2, CSF-1 and IL-6, which promotes monocyte egress from peripheral blood, resulting in a 3-5-fold enhancement of TAM infiltration within the tumor microenvironment (47–49). The CAF-derived factors induce profound phenotypic changes in recruited macrophages in hepatocellular carcinoma through JAK-STAT pathway activation (50). This signaling cascade drives macrophage polarization toward an immunosuppressive phenotype (51). Notably, this paracrine signaling axis also upregulates immune checkpoint molecule expression, with CAF-conditioned media inducing a great increase in PD-L1 surface expression on TAMs as measured by flow cytometry. Mechanistic studies reveal that this effect is mediated through both STAT3-dependent transcriptional activation and post-translational stabilization of PD-L1 protein (52).
The biological consequences of this CAF-TAM crosstalk include: enhanced phagocytic resistance of tumor cells, increased production of immunosuppressive IL-10, impaired antigen presentation capacity and promotion of angiogenesis through VEGF secretion (22, 53–55). In the clinical context, the alliance of CAF-TAM correlates with several adverse outcomes. Specifically, in liver cancer, the interaction between TAMs and CAFs is crucial in shaping the immune barrier, and the disruption of this communication potentially enhance immunotherapy effectiveness and improve clinical outcomes of patients (56). In conclusion, this interaction between CAFs and TAMs is a key mechanism of immune evasion in the TME.
3.3 CAFs and myeloid-derived suppressor cells
Building upon their interactions with TAMs, CAFs also establish critical immunosuppressive networks with myeloid-derived suppressor cells (MDSCs), further reinforcing the immune-evasive nature of the tumor microenvironment. MDSCs are a heterogeneous population of myeloid cells at various stages of differentiation, with immunosuppressive activity as the main feature (57). MDSCs can promote immune evasion and tolerance by recruiting Treg via CD40-CD40 ligand signaling (58). Besides, indoleamine 2,3-dioxygenase (IDO) or arginase 1 (ARG1) expressed by MDSCs contribute to the immunosuppressive metabolic microenvironment (59, 60). In the TME, MDSCs and CAFs are two key immune-suppressive cells that promote tumor immune evasion and progression through complex interactions (61). CAFs recruit MDSCs from the bone marrow to the tumor site by secreting cytokines such as CCL2, CSF-1 and IL-6. These cytokines also can activate MDSCs, thereby enhancing their ability to suppress T cell function (62). A recent study found that cytokines by CAFs enhanced the immune-suppressive capabilities of MDSCs and further secreting ARG1 and reactive ROS to inhibit T cell proliferation and activity (63). CAFs and MDSCs jointly deplete arginine and tryptophan in the tumor microenvironment, leading to metabolic dysfunction in T cells (64). This metabolic competition deprives T cells of essential nutrients, inhibiting their proliferation and function. Additionally, the ROS produced by MDSCs further damage the T cell receptor (TCR), reducing T cell reactivity to tumor antigens (65). CAFs and MDSCs also have a synergistic effect in immune suppression (66). CAFs reshape the ECM to form a physical barrier that restricts immune cell infiltration. MDSCs further suppress T cell function by secreting immune-suppressive molecules such as TGF-β and IL-10 (67). This synergistic effect makes immune suppression in the tumor microenvironment more pronounced, thereby promoting tumor progression and resistance to treatment.
3.4 CAFs and dendritic cells
Beyond their interactions with MDSCs, CAFs also engage in complex crosstalk with dendritic cells (DCs), another crucial component of the tumor immune microenvironment. One of the key interactions that contribute to immune evasion involves the relationship between CAFs and DCs. This interaction is complex and multifaceted, influencing both the function and phenotype of DCs, thereby facilitating tumor progression and resistance to immune surveillance (67). CAFs significantly impact the maturation and function of DCs. In a study involving the fusion of DCs and CAFs, it was observed that DC/CAF fusion cells expressed higher levels of co-stimulatory molecules such as CD80, CD86 and MHC II compared to immature DCs, suggesting that CAFs influence the maturation state of DCs, potentially skewing their function towards a more immunosuppressive phenotype (68). CAFs also impair the antigen-presenting function of DCs, which is crucial for initiating T cell-mediated immune responses. For instance, CAFs secrete factors such as PGE2 and TGF-β, which downregulate the expression of molecules important for antigen presentation, including MHC class II in DCs (28). This impairment in DC function reduces the ability of activation of CD4+ and CD8+ T cells, thereby facilitating immune evasion. Additionally, CAFs also influence the recruitment of DCs to the tumor site. By secreting chemokines such as CXCL12, CAFs attract DCs into the TME (69, 70). Emerging evidence suggests that while CAFs actively recruit DCs into the TME, these DCs often exhibit functional impairment due to CAF-mediated immunosuppression. This creates a paradoxical scenario where DCs are physically present but functionally compromised, ultimately failing to initiate proper T cell activation and contributing significantly to tumor immune evasion (71).
3.5 CAFs and natural killer cells
Complementing their suppressive effects on adaptive immune cells, CAFs also exert profound inhibitory effects on innate immunity, particularly by impairing the cytotoxic function of natural killer cells (NKCs). NKCs are innate immune cells that can recognize and kill tumor cells without prior sensitization. However, the presence of CAFs significantly impair the function of NKCs, contributing to immune evasion (72). Cytokines and chemokines produced by CAFs which are shown to suppress the cytotoxic activity of NKCs (72). These cytokines also induce the expression of inhibitory receptors on NKCs, further dampening their ability to recognize and kill tumor cells (72). CAFs deplete essential nutrients in the TME, creating a hostile environment for NKCs. Malchiodi et al. point out that CAFs consume a large of arginine and tryptophan, which are critical for NKCs activation and function (72). CAFs also interact directly with NKCs through cell surface molecules, leading to the downregulation of activating receptors on NKCs (71). This direct contact can inhibit NKCs cytotoxicity and promote immune tolerance within the TME. In summary, inhibiting the secretion of immunosuppressive cytokines by CAFs or blocking the direct cell-to-cell contact between CAFs and NKCs restore NKCs function and improve anti-tumor immunity (73).
3.6 CAFs and B cells
Extending their immunosuppressive influence beyond innate and T cell immunity, CAFs also engage in critical interactions with B cells, further shaping the tumor immune landscape. The interactions between CAFs and B cells are multifaceted and can influence both the function and phenotype of B cells within the TME. Recent studies have shown that CAFs modulate the immune landscape by interacting with various immune cells, including B cells (74). CAFs secrete a variety of cytokines to inhibit B cell activation, proliferation and antibody production, as well as reducing the overall immune response (75). Regulatory B cells (Bregs) is a subpopulation which are known for their immunosuppressive properties (74). CAFs utilizes at least two pathways to interact with Bregs: (1) secreting TGF-β to promote the differentiation of B cells into Bregs (74), and (2) secreting CXCL13 to recruit mature Bregs into TME (76, 77). In melanoma and colorectal cancer models, Bregs recruited by CAFs directly impairing cytotoxic T cell responses and contributing to anti-PD-1 therapy resistance (76, 77). All in all, understanding these interactions and developing strategies to disrupt them could significantly enhance the efficacy of cancer immunotherapies improve patient outcomes.
3.7 CAFs and neutrophils
Completing the spectrum of their immunomodulatory effects, CAFs additionally orchestrate critical interactions with tumor-associated neutrophils (TANs), establishing yet another immunosuppressive axis in the tumor microenvironment. The bidirectional communication between CAFs and tumor-associated neutrophils has emerged as a critical axis in facilitating immune evasion. CAFs serve as the primary architects of neutrophil recruitment within tumors, secreting an array of cytokines and chemokines. The most potent of these is IL-8 (22). Subsequently, these recruited neutrophils polarize into an pro-tumor phenotype, characterized by significantly increased ARG1 expression, elevated PD-L1 surface levels and enhanced MMP-9 secretion, collectively establishing a profoundly immunosuppressive microenvironment (36, 78, 79). The central role of CAF and neutrophils’ interaction in creating and maintaining an immunosuppressive tumor microenvironment is an attractive target for cancer therapy.
4 Molecular mechanisms of CAFs-mediated immune evasion
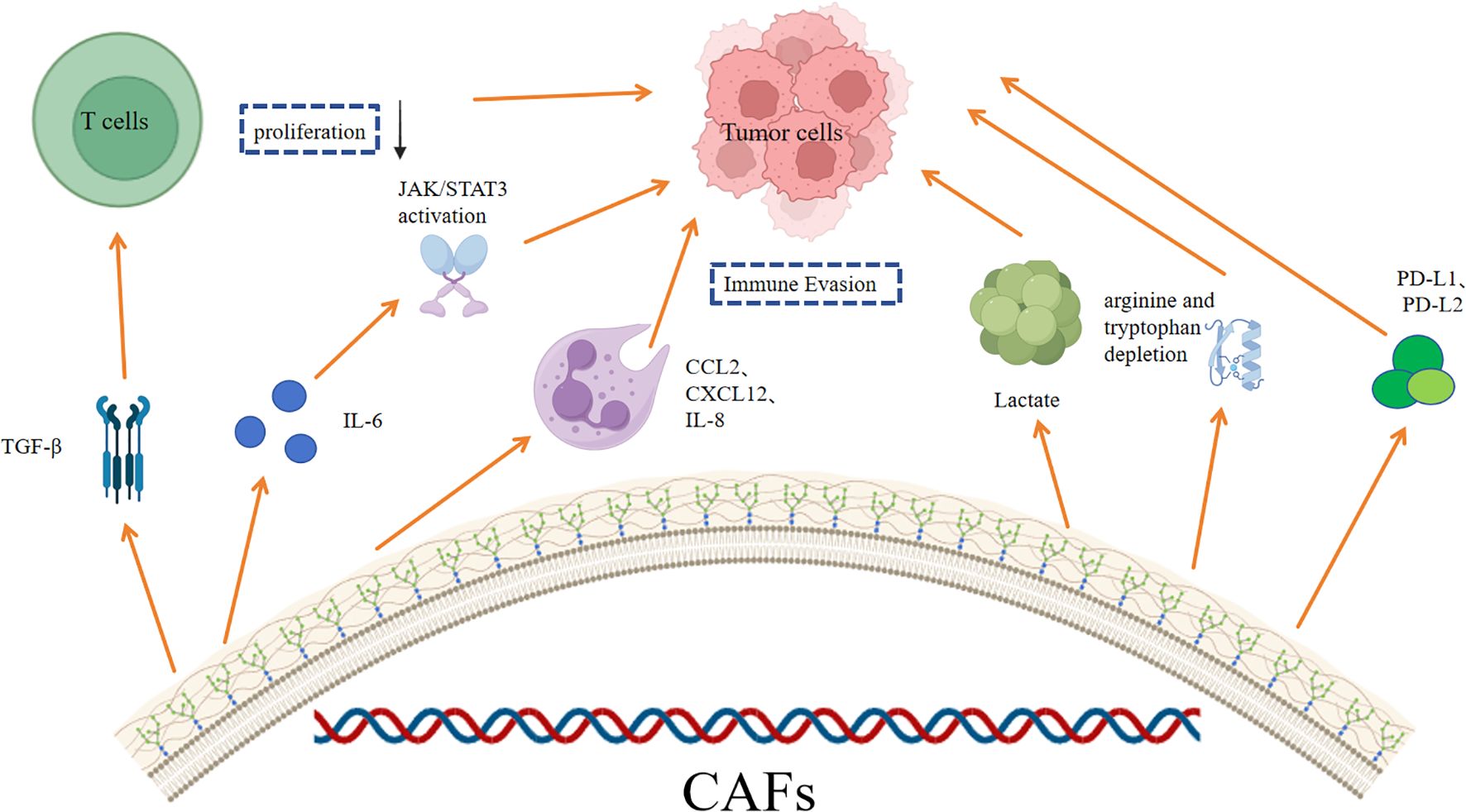
CAFs play a crucial role in the immune evasion through various mechanisms (Figure 2). Understanding the molecular mechanisms of CAFs-mediated immune evasion has important implications for cancer therapy.
4.1 Cytokine-mediated immunosuppression
CAFs modulate the immune microenvironment through the secretion of various cytokines and chemokines, which influence the recruitment, activation, and function of immune cells within the TME (80).
TGF-β is a key cytokine secreted by CAFs that has potent immunosuppressive effects. TGF-β can inhibit the proliferation and function of T cells, promote the differentiation of regulatory T cells, and enhance the immunosuppressive phenotype of other immune cells such as macrophages and dendritic cells (81). Additionally, TGF-β signaling can induce the expression of immune checkpoint molecules like PD-L1 on tumor cells and stromal cells, further contributing to immune evasion (82). The TGF-β signaling axis is thus a critical pathway through which CAFs mediate immune suppression in the TME.
IL-6 is another important cytokine produced by CAFs that drives immune suppression via the JAK/STAT3 signaling pathway. IL-6 activates the JAK/STAT3 pathway in immune cells, leading to the upregulation of anti-inflammatory and immunosuppressive genes (83). This pathway also promotes the differentiation of Tregs, inhibits the function of cytotoxic T cells and enhances the production of immunosuppressive cytokines such as IL-10 (8). Moreover, the IL-6/JAK/STAT3 pathway influence the function of myeloid cells, including macrophages and neutrophils, skewing them towards a more immunosuppressive phenotype (84). Targeting the IL-6/JAK/STAT3 pathway has emerged as a promising strategy to overcome CAF-mediated immune suppression in cancer.
CAFs secrete a variety of chemokines, such as CCL2, CXCL12, and IL-8 playing crucial roles in the recruitment and positioning of immune cells. For example, CCL2 attracts monocytes and macrophages, while CXCL12 is involved in the recruitment of T cells and dendritic cells (85). Additionally, these chemokines influence the phenotype and function of immune cells, further contributing to immune evasion (86).
4.2 Metabolic reprogramming of TME
Lactate is a byproduct of glycolysis and is often produced in high amounts by cancer cells and CAFs (87). The accumulation of lactate in the TME leads to acidification, creating a low pH environment that is immunosuppressive. This acidic environment also suppresses the function of T cells and other immune cells, thereby facilitating immune evasion (88). Additionally, lactate can activate the Wnt/β-catenin signaling pathway in tumor cells, further promoting their energy metabolism and proliferation (89).
CAFs deplete essential amino acids in the TME, such as arginine and tryptophan, through increased metabolic activity. This depletion can impair the function of immune cells, particularly T cells, which require these amino acids for activation and proliferation (90). Recent research showed that CAFs express high levels of ARG1, an enzyme that degrades arginine, leading to a deficiency of this amino acid in the TME (91). Similarly, the expression of tryptophan-degrading enzymes like indoleamine-2,3-dioxygenase by CAFs lead to tryptophan depletion, further suppressing T cell function (92). This metabolic competition between CAFs and immune cells creates an immunosuppressive environment that promotes tumor progression.
CAFs exhibit significant alterations in lipid metabolism, which can impact the TME and immune cell function. High expression of fatty acid synthase, carnitine palmitoyl transferase 1 and CD36 in CAFs promotes fatty acid oxidation and the accumulation of intracellular lipid droplets (90). These metabolic changes can lead to the exhaustion of cytotoxic T lymphocytes and provide fuel for tumor cell fatty acid metabolism, contributing to tumor proliferation and metastasis (93, 94). Additionally, the altered lipid metabolism in CAFs affect the function of other immune cells, further contributing to immune suppression (90).
Hypoxia is a common feature of the TME and induce significant metabolic changes in CAFs. Under hypoxic conditions, CAFs upregulate the expression of hypoxia-inducible factor, which drives the expression of genes involved in glycolysis and other metabolic pathways (95). This metabolic reprogramming allows CAFs to adapt to the low-oxygen environment and further supports the growth and survival of tumor cells. Hypoxia also enhances the secretion of immunosuppressive cytokines and chemokines by CAFs, such as TGF-β and IL-6, which inhibit the function of immune cells and promote immune evasion (96). Moreover, hypoxia-induced metabolic changes in CAFs can lead to the production of reactive oxygen species (ROS), which can damage immune cells and further suppress their function (97).
4.3 Immune checkpoint regulation
CAFs significantly influence the expression of immune checkpoint molecules such as PD-L1 and PD-L2 within the TME (98). Studies have shown that IFN-γ can significantly upregulate the expression of PD-L1 and PD-L2 in various tumor cell lines, thereby enhancing immune evasion (99). The upregulation of PD-L1 and PD-L2 on CAFs and other cells in the TME contributes to the suppression of T cell-mediated immune responses, facilitating tumor progression (100).
In addition to the well-known PD-1/PD-L1 axis, CAFs also modulate the expression of other non-canonical immune checkpoints, such as VISTA and LAG-3. VISTA (V-domain Ig suppressor of T cell activation) is an inhibitory receptor that can suppress T cell activation and proliferation. Similarly, LAG-3 (lymphocyte-activation gene 3) is another immune checkpoint molecule that can inhibit T cell function and contribute to immune exhaustion (101). The expression of these non-canonical checkpoints by CAFs or other cells in the TME can further dampen the immune response, creating a more immunosuppressive environment. Targeting these non-canonical checkpoints, in combination with PD-1/PD-L1 inhibitors, may provide a more comprehensive approach to overcoming immune evasion in cancer.
Fas/FasL is a critical death receptor-ligand system that mediates apoptosis signaling, which is another pathway affected by CAFs in immune evasion (102). Fas ligand is expressed by various cells in the TME, including CAFs, and can induce apoptosis in Fas-expressing T cells. This interaction leads to the elimination of activated T cells, thereby reducing the overall immune response against the tumor (101). The expression of FasL by CAFs can create a local environment that is hostile to T cells, promoting immune evasion and tumor progression. Targeting the Fas/FasL pathway may help to preserve the function of T cells and enhance anti-tumor immunity. At the same time, emerging clinical evidence supports targeted CAF immune crosstalk, and ongoing trials of FAP/PD-L1 dual targeted CAR-T cell (NCT04328026) and TGF-β/PD-1 combination therapy (NCT0366871) have shown a 36-41% improvement in response to treatment resistant tumors (57). CAFs modulate the immune microenvironment through the CD73/adenosine pathway (103). CD73 is an ectoenzyme that converts extracellular AMP into adenosine, a potent immunosuppressive molecule (104). High levels of adenosine in the TME inhibit the function of T cells and other immune cells, promoting immune evasion. CAFs express high levels of CD73, contributing to the accumulation of adenosine in the TME (101, 105). This pathway is targeted by inhibitors of CD73 or adenosine receptors, potentially enhancing the efficacy of cancer immunotherapies.
5 Conclusion
Cancer associated fibroblasts coordinate immune suppression through three core mechanisms: cytokine network (TGF-β/IL-6), metabolic symbiosis (lactate/arginine consumption), and immune checkpoint crosstalk (PD-L1/CD73). These pathways drive T cell dysfunction, macrophage polarization, and bone marrow suppression, forming a therapeutic barrier. Emerging strategies such as JAK/STAT inhibitors, metabolic disruptors, and adenosine blockade have shown promise in epithelial cancers where CAF immune interactions dominate, in stark contrast to the limited role of the matrix in hematopoietic malignancies (94). Space single-cell localization reveals tissue-specific CAF heterogeneity, guiding precise treatment that combines matrix reprogramming with immunotherapy. Future translation requires CAF derived extracellular vesicle biomarkers for patient stratification and targeted delivery systems to avoid drug resistance.
Author contributions
JL: Writing – original draft. XX: Writing – original draft. LJ: Writing – original draft. GG: Writing – review & editing, Writing – original draft.
Funding
The author(s) declare that no financial support was received for the research and/or publication of this article.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Setiadi AF, David MD, Seipp RP, Hartikainen JA, Gopaul R, and Jefferies WA. Epigenetic control of the immune escape mechanisms in Malignant carcinomas. Mol Cell Biol. (2007) 27:7886–94. doi: 10.1128/mcb.01547-07
2. Swann JB and Smyth MJ. Immune surveillance of tumors. J Clin Invest. (2007) 117:1137–46. doi: 10.1172/jci31405
3. Martínez-Jiménez F, Priestley P, Shale C, Baber J, Rozemuller E, and Cuppen E. Genetic immune escape landscape in primary and metastatic cancer. Nat Genet. (2023) 55:820–31. doi: 10.1038/s41588-023-01367-1
4. Tang S, Ning Q, Yang L, Mo Z, and Tang S. Mechanisms of immune escape in the cancer immune cycle. Int Immunopharmacol. (2020) 86:106700. doi: 10.1016/j.intimp.2020.106700
5. Barjasteh AH, Saebi M, Mahmoudi M, Kheder RK, Hashemy SI, Forouzanfar F, et al. Revolutionizing cancer treatment: unveiling the power of car T-cell therapy. Curr Pharm design. (2025) 31:1020–36. doi: 10.2174/0113816128336391241107112957
6. Meng W, Yang B, Huang B, Chen C, Zhu J, Jian D, et al. Impact of preoperative transcatheter rectal arterial chemoembolization with concurrent chemoradiotherapy on surgery and prognosis of patients with locally advanced rectal cancer. J Surg Oncol. (2021) 124:1451–8. doi: 10.1002/jso.26673
7. Zagozdzon R, Winiarska M, and Firczuk M. Immune evasion as the main challenge for immunotherapy of cancer. Cancers. (2022) 14:3622. doi: 10.3390/cancers14153622
8. Zhang S, Gan X, Qiu J, Ju Z, Gao J, Zhou J, et al. Il-10 derived from hepatocarcinoma cells improves human induced regulatory T cells function via jak1/stat5 pathway in tumor microenvironment. Mol Immunol. (2021) 133:163–72. doi: 10.1016/j.molimm.2021.02.014
9. Li YR, Halladay T, and Yang L. Immune evasion in cell-based immunotherapy: unraveling challenges and novel strategies. J Biomed Sci. (2024) 31:5. doi: 10.1186/s12929-024-00998-8
10. Cui K, Wang K, and Huang Z. Ferroptosis and the tumor microenvironment. J Exp Clin Cancer Res. (2024) 43:315. doi: 10.1186/s13046-024-03235-0
11. Meng W, Huang L, Guo J, Xin Q, Liu J, and Hu Y. Innovative nanomedicine delivery: targeting tumor microenvironment to defeat drug resistance. Pharmaceutics. (2024) 16:1549. doi: 10.3390/pharmaceutics16121549
12. Avagliano A and Arcucci A. Insights into melanoma fibroblast populations and therapeutic strategy perspectives: friends or foes? Curr medicinal Chem. (2022) 29:6159–68. doi: 10.2174/0929867329666220620124138
13. Ziani L, Chouaib S, and Thiery J. Alteration of the antitumor immune response by cancer-associated fibroblasts. Front Immunol. (2018) 9:414. doi: 10.3389/fimmu.2018.00414
14. Oya Y, Hayakawa Y, and Koike K. Tumor microenvironment in gastric cancers. Cancer Sci. (2020) 111:2696–707. doi: 10.1111/cas.14521
15. Yasmin H, Ramesh RP, Joseph AM, and Kishore U. Fibroblast heterogeneity and its role in generating protective immunity in the secondary lymphoid organs. Front Immunol. (2025) 16:1519789. doi: 10.3389/fimmu.2025.1519789
16. Zhu L, Zhang X, Zhang S, Zhang Q, Cao L, Zhang Y, et al. Cancer-associated fibroblasts in papillary thyroid carcinoma. Clin Exp Med. (2023) 23:2209–20. doi: 10.1007/s10238-023-00998-2
17. Mucciolo G, Araos Henriquez J, Jihad M, Pinto Teles S, Manansala JS, Li W, et al. Egfr-activated myofibroblasts promote metastasis of pancreatic cancer. Cancer Cell. (2024) 42:101–18 e11. doi: 10.1016/j.ccell.2023.12.002
18. Fan G, Yu B, Tang L, Zhu R, Chen J, Zhu Y, et al. Tspan8(+) myofibroblastic cancer-associated fibroblasts promote chemoresistance in patients with breast cancer. Sci Transl Med. (2024) 16:eadj5705. doi: 10.1126/scitranslmed.adj5705
19. Chen Z, Zhou L, Liu L, Hou Y, Xiong M, Yang Y, et al. Single-cell rna sequencing highlights the role of inflammatory cancer-associated fibroblasts in bladder urothelial carcinoma. Nat Commun. (2020) 11:5077. doi: 10.1038/s41467-020-18916-5
20. Gao H, Wong SQR, Subel E, Huang YH, Lee YC, Hayashi K, et al. Caspase-1-dependent pyroptosis converts Αsma(+) cafs into collagen-iii(High) icafs to fuel chemoresistant cancer stem cells. Sci Adv. (2025) 11:eadt8697. doi: 10.1126/sciadv.adt8697
21. Huang H, Wang Z, Zhang Y, Pradhan RN, Ganguly D, Chandra R, et al. Mesothelial cell-derived antigen-presenting cancer-associated fibroblasts induce expansion of regulatory T cells in pancreatic cancer. Cancer Cell. (2022) 40:656–73.e7. doi: 10.1016/j.ccell.2022.04.011
22. Song J, Wei R, Liu C, Zhao Z, Liu X, Wang Y, et al. Antigen-presenting cancer associated fibroblasts enhance antitumor immunity and predict immunotherapy response. Nat Commun. (2025) 16:2175. doi: 10.1038/s41467-025-57465-7
23. Wang H, Liu F, Wu X, Zhu G, Tang Z, Qu W, et al. Cancer-associated fibroblasts contributed to hepatocellular carcinoma recurrence and metastasis via cd36-mediated fatty-acid metabolic reprogramming. Exp Cell Res. (2024) 435:113947. doi: 10.1016/j.yexcr.2024.113947
24. Wu C, Yu S, Wang Y, Gao Y, Xie X, and Zhang J. Metabolic-suppressed cancer-associated fibroblasts limit the immune environment and survival in colorectal cancer with liver metastasis. Front Pharmacol. (2023) 14:1212420. doi: 10.3389/fphar.2023.1212420
25. Verginadis II, Avgousti H, Monslow J, Skoufos G, Chinga F, Kim K, et al. A stromal integrated stress response activates perivascular cancer-associated fibroblasts to drive angiogenesis and tumour progression. Nat Cell Biol. (2022) 24:940–53. doi: 10.1038/s41556-022-00918-8
26. Tsoumakidou M. The advent of immune stimulating cafs in cancer. Nat Rev Cancer. (2023) 23:258–69. doi: 10.1038/s41568-023-00549-7
27. Guo S, Yuan J, Meng X, Feng X, Ma D, Han Y, et al. Cancer-associated fibroblasts: just on the opposite side of antitumour immunity? Int Immunopharmacol. (2023) 122:110601. doi: 10.1016/j.intimp.2023.110601
28. Maia A, Schöllhorn A, Schuhmacher J, and Gouttefangeas C. Caf-immune cell crosstalk and its impact in immunotherapy. Semin immunopathology. (2023) 45:203–14. doi: 10.1007/s00281-022-00977-x
29. Hilmi M, Nicolle R, Bousquet C, and Neuzillet C. Cancer-associated fibroblasts: accomplices in the tumor immune evasion. Cancers. (2020) 12:2969. doi: 10.3390/cancers12102969
30. Liao T, Chen X, Qiu F, Zhang X, Wu F, Zhao Z, et al. Regulation of cancer-associated fibroblasts for enhanced cancer immunotherapy using advanced functional nanomedicines: an updated review. J nanobiotechnology. (2025) 23:166. doi: 10.1186/s12951-025-03217-0
31. Dzobo K, Senthebane DA, and Dandara C. The tumor microenvironment in tumorigenesis and therapy resistance revisited. Cancers. (2023) 15:376. doi: 10.3390/cancers15020376
32. Tekguc M, Wing JB, Osaki M, Long J, and Sakaguchi S. Treg-expressed ctla-4 depletes cd80/cd86 by trogocytosis, releasing free pd-L1 on antigen-presenting cells. Proc Natl Acad Sci United States America. (2021) 118:e2023739118. doi: 10.1073/pnas.2023739118
33. Afroj T, Mitsuhashi A, Ogino H, Saijo A, Otsuka K, Yoneda H, et al. Blockade of pd-1/pd-L1 pathway enhances the antigen-presenting capacity of fibrocytes. J Immunol (Baltimore Md: 1950). (2021) 206:1204–14. doi: 10.4049/jimmunol.2000909
34. Dezutter-Dambuyant C, Durand I, Alberti L, Bendriss-Vermare N, Valladeau-Guilemond J, Duc A, et al. A novel regulation of pd-1 ligands on mesenchymal stromal cells through mmp-mediated proteolytic cleavage. Oncoimmunology. (2016) 5:e1091146. doi: 10.1080/2162402x.2015.1091146
35. Malech HL and Notarangelo LD. Gene therapy for inborn errors of immunity: base editing comes into play. Cell. (2023) 186:1302–4. doi: 10.1016/j.cell.2023.03.001
36. Wu Y, Ma J, Yang X, Nan F, Zhang T, Ji S, et al. Neutrophil profiling illuminates anti-tumor antigen-presenting potency. Cell. (2024) 187:1422–39.e24. doi: 10.1016/j.cell.2024.02.005
37. Ungefroren H. Tgf-Β Signaling in cancer: control by negative regulators and crosstalk with proinflammatory and fibrogenic pathways. Cancers. (2019) 11:384. doi: 10.3390/cancers11030384
38. Hosseini R, Hosseinzadeh N, Asef-Kabiri L, Akbari A, Ghezelbash B, Sarvnaz H, et al. Small extracellular vesicle tgf-Β in cancer progression and immune evasion. Cancer Gene Ther. (2023) 30:1309–22. doi: 10.1038/s41417-023-00638-7
39. Ghahremanifard P, Chanda A, Bonni S, and Bose P. Tgf-Β Mediated immune evasion in cancer-spotlight on cancer-associated fibroblasts. Cancers. (2020) 12:3650. doi: 10.3390/cancers12123650
40. Yin C, Zhang C, Wang Y, Liu G, Wang N, Liang N, et al. Aldob/kat2a interactions epigenetically modulate tgf-Β Expression and T cell functions in hepatocellular carcinogenesis. Hepatol (Baltimore Md). (2025) 81:77–93. doi: 10.1097/hep.0000000000000704
41. Mishra S, Liao W, Liu Y, Yang M, Ma C, Wu H, et al. Tgf-Β and eomes control the homeostasis of cd8+ Regulatory T cells. J Exp Med. (2021) 218:e20200030. doi: 10.1084/jem.20200030
42. Priyadharshini B, Loschi M, Newton RH, Zhang JW, Finn KK, Gerriets VA, et al. Cutting edge: tgf-Β and phosphatidylinositol 3-kinase signals modulate distinct metabolism of regulatory T cell subsets. J Immunol (Baltimore Md: 1950). (2018) 201:2215–9. doi: 10.4049/jimmunol.1800311
43. Chen W. Tgf-beta regulation of T cells. Annu Rev Immunol. (2023) 41:483–512. doi: 10.1146/annurev-immunol-101921-045939
44. Yadav D, Yadav A, Bhattacharya S, Dagar A, Kumar V, and Rani R. Glut and hk: two primary and essential key players in tumor glycolysis. Semin Cancer Biol. (2024) 100:17–27. doi: 10.1016/j.semcancer.2024.03.001
45. Awaji M and Singh RK. Cancer-associated fibroblasts’ Functional heterogeneity in pancreatic ductal adenocarcinoma. Cancers. (2019) 11:290. doi: 10.3390/cancers11030290
46. Timperi E, Croizer H, Khantakova D, Rana M, Molgora M, Guerriero JL, et al. At the interface of tumor-associated macrophages and fibroblasts: immune-suppressive networks and emerging exploitable targets. Clin Cancer Res. (2024) 30:5242–51. doi: 10.1158/1078-0432.Ccr-24-1690
47. Tomassetti C, Insinga G, Gimigliano F, Morrione A, Giordano A, and Giurisato E. Insights into csf-1r expression in the tumor microenvironment. Biomedicines. (2024) 12:2381. doi: 10.3390/biomedicines12102381
48. Li C, Zhang C, Chen X, Zhang Y, Chen J, Kang L, et al. Relative depletion of soluble interleukin 6 receptors abolished the development of cytokine release syndrome after cart19/22 and lenalidomide treatment for lymphoma. Blood. (2019) 134:5313. doi: 10.1182/blood-2019-126821
49. Flaming J, Chandra R, Girard L, Ganguly D, Toombs J, Minna JD, et al. Abstract po021: lung cancer cells and cancer-associated fibroblasts drive macrophage polarization in a co-culture model. Cancer Immunol Res. (2021) 9:PO021–PO. doi: 10.1158/2326-6074.TUMIMM20-PO021
50. Chen S, Morine Y, Tokuda K, Yamada S, Saito Y, Nishi M, et al. Cancer−Associated fibroblast−Induced M2−Polarized macrophages promote hepatocellular carcinoma progression via the plasminogen activator inhibitor−1 pathway. Int J Oncol. (2021) 59:59. doi: 10.3892/ijo.2021.5239
51. Feng N, Li Y, Guo F, Song J, Wang L, Li M, et al. Fibroblast growth factor 10 alleviates lps-induced acute lung injury by promoting recruited macrophage M2 polarization. Inflammation. (2024) 48:1828–38. doi: 10.1007/s10753-024-02158-4
52. Chuangchot N, Jamjuntra P, Yangngam S, Luangwattananun P, Thongchot S, Junking M, et al. Enhancement of pd-L1-attenuated car-T cell function through breast cancer-associated fibroblasts-derived il-6 signaling via stat3/akt pathways. Breast Cancer research: BCR. (2023) 25:86. doi: 10.1186/s13058-023-01684-7
53. Horii Y, Matsuda S, Watari K, Nagasaka A, Kurose H, and Nakaya M. An assay to determine phagocytosis of apoptotic cells by cardiac macrophages and cardiac myofibroblasts. Bio-protocol. (2017) 7:e2553. doi: 10.21769/BioProtoc.2553
54. Timperi E, Gueguen P, Molgora M, Magagna I, Kieffer Y, Lopez-Lastra S, et al. Lipid-associated macrophages are induced by cancer-associated fibroblasts and mediate immune suppression in breast cancer. Cancer Res. (2022) 82:3291–306. doi: 10.1158/0008-5472.Can-22-1427
55. Tang PC, Chung JY, Xue VW, Xiao J, Meng XM, Huang XR, et al. Smad3 promotes cancer-associated fibroblasts generation via macrophage-myofibroblast transition. Advanced Sci (Weinheim Baden-Wurttemberg Germany). (2022) 9:e2101235. doi: 10.1002/advs.202101235
56. Long F, Zhong W, Zhao F, Xu Y, Hu X, Jia G, et al. Dab2 (+) macrophages support fap (+) fibroblasts in shaping tumor barrier and inducing poor clinical outcomes in liver cancer. Theranostics. (2024) 14:4822–43. doi: 10.7150/thno.99046
57. Lasser SA, Ozbay Kurt FG, Arkhypov I, Utikal J, and Umansky V. Myeloid-derived suppressor cells in cancer and cancer therapy. Nat Rev Clin Oncol. (2024) 21:147–64. doi: 10.1038/s41571-023-00846-y
58. Pan PY, Ma G, Weber KJ, Ozao-Choy J, Wang G, Yin B, et al. Immune stimulatory receptor cd40 is required for T-cell suppression and T regulatory cell activation mediated by myeloid-derived suppressor cells in cancer. Cancer Res. (2010) 70:99–108. doi: 10.1158/0008-5472.Can-09-1882
59. Zoso A, Mazza EM, Bicciato S, Mandruzzato S, Bronte V, Serafini P, et al. Human fibrocytic myeloid-derived suppressor cells express ido and promote tolerance via treg-cell expansion. Eur J Immunol. (2014) 44:3307–19. doi: 10.1002/eji.201444522
60. Pang B, Zhen Y, Hu C, Ma Z, Lin S, and Yi H. Myeloid-derived suppressor cells shift th17/treg ratio and promote systemic lupus erythematosus progression through arginase-1/mir-322-5p/tgf-beta pathway. Clin Sci (Lond). (2020) 134:2209–22. doi: 10.1042/CS20200799
61. Huang J, Chen X, Chang Z, Xiao C, and Najafi M. Boosting anti-tumour immunity using adjuvant apigenin. Anti-cancer Agents medicinal Chem. (2023) 23:266–77. doi: 10.2174/1871520622666220523151409
62. Magaña-Maldonado R, Chávez-Cortez EG, Olascoaga-Arellano NK, López-Mejía M, Maldonado-Leal FM, Sotelo J, et al. Immunological evasion in glioblastoma. BioMed Res Int. (2016) 2016:7487313. doi: 10.1155/2016/7487313
63. Weber R, Riester Z, Hüser L, Sticht C, Siebenmorgen A, Groth C, et al. Il-6 regulates ccr5 expression and immunosuppressive capacity of mdsc in murine melanoma. J immunotherapy Cancer. (2020) 8:e000949. doi: 10.1136/jitc-2020-000949
64. Zhu X, Pribis JP, Rodriguez PC, Morris SM Jr., Vodovotz Y, Billiar TR, et al. The central role of arginine catabolism in T-cell dysfunction and increased susceptibility to infection after physical injury. Ann Surg. (2014) 259:171–8. doi: 10.1097/SLA.0b013e31828611f8
65. Li YR, Wilson M, and Yang L. Target tumor microenvironment by innate T cells. Front Immunol. (2022) 13:999549. doi: 10.3389/fimmu.2022.999549
66. Lin Y, Li B, Yang X, Cai Q, Liu W, Tian M, et al. Fibroblastic fap promotes intrahepatic cholangiocarcinoma growth via mdscs recruitment. Neoplasia (New York NY). (2019) 21:1133–42. doi: 10.1016/j.neo.2019.10.005
67. Mao X, Xu J, Wang W, Liang C, Hua J, Liu J, et al. Crosstalk between cancer-associated fibroblasts and immune cells in the tumor microenvironment: new findings and future perspectives. Mol Cancer. (2021) 20:131. doi: 10.1186/s12943-021-01428-1
68. Mu G, Zhang W, Huang J, Chen Z, and Wang J. Research status of tumor-associated fibroblasts regulating immune cells. Zhongguo fei ai za zhi = Chin J Lung Cancer. (2022) 25:207–13. doi: 10.3779/j.issn.1009-3419.2022.101.04
69. Kryczek I, Lange A, Mottram P, Alvarez X, Cheng P, Hogan M, et al. Cxcl12 and vascular endothelial growth factor synergistically induce neoangiogenesis in human ovarian cancers. Cancer Res. (2005) 65:465–72. doi: 10.1158/0008-5472.465.65.2
70. Nagarsheth N, Wicha MS, and Zou W. Chemokines in the cancer microenvironment and their relevance in cancer immunotherapy. Nat Rev Immunol. (2017) 17:559–72. doi: 10.1038/nri.2017.49
71. Ben-Shmuel A, Gruper Y, Halperin C, Levi-Galibov O, Rosenberg-Fogler H, Barki D, et al. Cancer-associated fibroblasts serve as decoys to suppress nk cell anti-cancer cytotoxicity in breast cancer. Cancer Discov. (2025) 15:1247–69. doi: 10.1158/2159-8290.Cd-24-0131
72. Malchiodi ZX and Weiner LM. Understanding and targeting natural killer cell-cancer-associated fibroblast interactions in pancreatic ductal adenocarcinoma. Cancers. (2021) 13:405. doi: 10.3390/cancers13030405
73. Ielpo S, Barberini F, Dabbagh Moghaddam F, Pesce S, Cencioni C, Spallotta F, et al. Crosstalk and communication of cancer-associated fibroblasts with natural killer and dendritic cells: new frontiers and unveiled opportunities for cancer immunotherapy. Cancer Treat Rev. (2024) 131:102843. doi: 10.1016/j.ctrv.2024.102843
74. Mukherjee P, Ansell SM, and Mondello P. Unraveling the role of cancer-associated fibroblasts in B cell lymphoma. Front Immunol. (2024) 15:1451791. doi: 10.3389/fimmu.2024.1451791
75. Milosevic V and Östman A. Interactions between cancer-associated fibroblasts and T-cells: functional crosstalk with targeting and biomarker potential. Upsala J Med Sci. (2024) 129:e6118. doi: 10.48101/ujms.v126.10710
76. Ren J, Lan T, Liu T, Liu Y, Shao B, Men K, et al. Cxcl13 as a novel immune checkpoint for regulatory B cells and its role in tumor metastasis. J Immunol. (2022) 208:2425–35. doi: 10.4049/jimmunol.2100341
77. Shen L, Li J, Liu Q, Das M, Song W, Zhang X, et al. Nano-trapping cxcl13 reduces regulatory B cells in tumor microenvironment and inhibits tumor growth. J Control Release. (2022) 343:303–13. doi: 10.1016/j.jconrel.2022.01.039
78. Cheng Y, Li H, Deng Y, Tai Y, Zeng K, Zhang Y, et al. Cancer-associated fibroblasts induce pdl1+ Neutrophils through the il6-stat3 pathway that foster immune suppression in hepatocellular carcinoma. Cell Death Dis. (2018) 9:422. doi: 10.1038/s41419-018-0458-4
79. Munir H, Jones JO, Janowitz T, Hoffmann M, Euler M, Martins CP, et al. Stromal-driven and amyloid beta-dependent induction of neutrophil extracellular traps modulates tumor growth. Nat Commun. (2021) 12:683. doi: 10.1038/s41467-021-20982-2
80. Monteran L and Erez N. The dark side of fibroblasts: cancer-associated fibroblasts as mediators of immunosuppression in the tumor microenvironment. Front Immunol. (2019) 10:1835. doi: 10.3389/fimmu.2019.01835
81. Dai Y, Wu J, Wang J, Wang H, Guo B, Jiang T, et al. Magnesium ions promote the induction of immunosuppressive bone microenvironment and bone repair through hif-1α-tgf-Β Axis in dendritic cells. Small (Weinheim an der Bergstrasse Germany). (2024) 20:e2311344. doi: 10.1002/smll.202311344
82. Shrestha R, Bridle KR, Crawford DHG, and Jayachandran A. Immune checkpoint molecules are regulated by transforming growth factor (Tgf)-Β1-induced epithelial-to-mesenchymal transition in hepatocellular carcinoma. Int J Med Sci. (2021) 18:2466–79. doi: 10.7150/ijms.54239
83. Forsthuber A, Aschenbrenner B, Korosec A, Jacob T, Annusver K, Krajic N, et al. Cancer-associated fibroblast subtypes modulate the tumor-immune microenvironment and are associated with skin cancer Malignancy. Nat Commun. (2024) 15:9678. doi: 10.1038/s41467-024-53908-9
84. Jeong H, Koh J, Kim S, Yim J, Song SG, Kim H, et al. Cell-intrinsic pd-L1 signaling drives immunosuppression by myeloid-derived suppressor cells through il-6/jak/stat3 in pd-L1-high lung cancer. J immunotherapy Cancer. (2025) 13:e010612. doi: 10.1136/jitc-2024-010612
85. Dill R and Walker AM. Role of prolactin in promotion of immune cell migration into the mammary gland. J mammary gland Biol neoplasia. (2017) 22:13–26. doi: 10.1007/s10911-016-9369-0
86. Chen R, Chen C, Han N, Guo W, Deng H, Wang Y, et al. Annexin-1 is an oncogene in glioblastoma and causes tumour immune escape through the indirect upregulation of interleukin-8. J Cell Mol Med. (2022) 26:4343–56. doi: 10.1111/jcmm.17458
87. Zang S, Huang K, Li J, Ren K, Li T, He X, et al. Metabolic reprogramming by dual-targeting biomimetic nanoparticles for enhanced tumor chemo-immunotherapy. Acta biomaterialia. (2022) 148:181–93. doi: 10.1016/j.actbio.2022.05.045
88. Rahman MA, Yadab MK, and Ali MM. Emerging role of extracellular ph in tumor microenvironment as a therapeutic target for cancer immunotherapy. Cells. (2024) 13:1924. doi: 10.3390/cells13221924
89. Wang H, Zhang Y, and Du S. Integrated analysis of lactate-related genes identifies polrmt as a novel marker promoting the proliferation, migration and energy metabolism of hepatocellular carcinoma via wnt/Β-catenin signaling. Am J Cancer Res. (2024) 14:1316–37. doi: 10.62347/zttg4319
90. Huang K, Han Y, Chen Y, Shen H, Zeng S, and Cai C. Tumor metabolic regulators: key drivers of metabolic reprogramming and the promising targets in cancer therapy. Mol Cancer. (2025) 24:7. doi: 10.1186/s12943-024-02205-6
91. Akinjiyan FA, Ibitoye Z, Zhao P, Shriver LP, Patti GJ, Longmore GD, et al. Ddr2-regulated arginase activity in ovarian cancer-associated fibroblasts promotes collagen production and tumor progression. Oncogene. (2024) 43:189–201. doi: 10.1038/s41388-023-02884-3
92. Liu K, Yang Y, Chen Y, Li S, Gong Y, and Liang Y. The therapeutic effect of dendritic cells expressing indoleamine 2,3-dioxygenase (Ido) on an iga nephropathy mouse model. Int Urol Nephrol. (2020) 52:399–407. doi: 10.1007/s11255-019-02365-1
93. Broz MT, Ko EY, Ishaya K, Xiao J, De Simone M, Hoi XP, et al. Metabolic targeting of cancer associated fibroblasts overcomes T-cell exclusion and chemoresistance in soft-tissue sarcomas. Nat Commun. (2024) 15:2498. doi: 10.1038/s41467-024-46504-4
94. Kay EJ and Zanivan S. The tumor microenvironment is an ecosystem sustained by metabolic interactions. Cell Rep. (2025) 44:115432. doi: 10.1016/j.celrep.2025.115432
95. Krieg M, Haas R, Brauch H, Acker T, Flamme I, and Plate KH. Up-regulation of hypoxia-inducible factors hif-1alpha and hif-2alpha under normoxic conditions in renal carcinoma cells by von hippel-lindau tumor suppressor gene loss of function. Oncogene. (2000) 19:5435–43. doi: 10.1038/sj.onc.1203938
96. Wu Q, You L, Nepovimova E, Heger Z, Wu W, Kuca K, et al. Hypoxia-inducible factors: master regulators of hypoxic tumor immune escape. J Hematol Oncol. (2022) 15:77. doi: 10.1186/s13045-022-01292-6
97. Alves JL, Quinta-Ferreira RM, Quinta-Ferreira ME, and Matias CM. Exploring different mechanisms of reactive oxygen species formation in hypoxic conditions at the hippocampal ca3 area. Mol Cell Endocrinol. (2025) 601:112517. doi: 10.1016/j.mce.2025.112517
98. Jiang J, Xu Y, Chen D, Li J, Zhu X, Pan J, et al. Pan-cancer analysis of immune checkpoint receptors and ligands in various cells in the tumor immune microenvironment. Aging. (2024) 16:11683–728. doi: 10.18632/aging.206053
99. Larsen TV, Daugaard TF, Gad HH, Hartmann R, and Nielsen AL. Pd-L1 and pd-L2 immune checkpoint protein induction by type iii interferon in non-small cell lung cancer cells. Immunobiology. (2023) 228:152389. doi: 10.1016/j.imbio.2023.152389
100. Mathios D, Ruzevick J, Jackson CM, Xu H, Shah SR, Taube JM, et al. Pd-1, pd-L1, pd-L2 expression in the chordoma microenvironment. J neuro-oncology. (2015) 121:251–9. doi: 10.1007/s11060-014-1637-5
101. Du W, Tang X, Zhou Y, Li M, Jin Z, Dou J, et al. Immune checkpoints mediate tumor immune regulation through metabolic pathways. Zhongguo fei ai za zhi = Chin J Lung Cancer. (2025) 28:213–20. doi: 10.3779/j.issn.1009-3419.2025.106.08
102. Yuan Y, Zhang Y, Zhao S, Chen J, Yang J, Wang T, et al. Cadmium-induced apoptosis in neuronal cells is mediated by fas/fasl-mediated mitochondrial apoptotic signaling pathway. Sci Rep. (2018) 8:8837. doi: 10.1038/s41598-018-27106-9
103. Saigí M, Mesía-Carbonell O, Barbie DA, and Guillamat-Prats R. Unraveling the intricacies of cd73/adenosine signaling: the pulmonary immune and stromal microenvironment in lung cancer. Cancers. (2023) 15:5706. doi: 10.3390/cancers15235706
104. Strickland LN, Liu W, Hussein U, Mardik N, Chen X, Mills T, et al. Preventive treatment with a cd73 small molecule inhibitor enhances immune surveillance in K-ras mutant pancreatic intraepithelial neoplasia. Cancer Prev Res (Philadelphia Pa). (2024) 17:457–70. doi: 10.1158/1940-6207.Capr-24-0200
Keywords: cancer-associated fibroblasts, immune evasion, immune cells, tumor microenvironment, CAFs
Citation: Luo J, Xiang X, Gong G and Jiang L (2025) Cancer-associated fibroblast-mediated immune evasion: molecular mechanisms of stromal-immune crosstalk in the tumor microenvironment. Front. Immunol. 16:1617662. doi: 10.3389/fimmu.2025.1617662
Received: 24 April 2025; Accepted: 19 June 2025;
Published: 26 August 2025.
Edited by:
Qiong Lu, Central South University, ChinaReviewed by:
Roslyn Kemp, University of Otago, New ZealandYudong Ning, Chinese Academy of Medical Sciences and Peking Union Medical College, China
Copyright © 2025 Luo, Xiang, Gong and Jiang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Guangyuan Gong, cmVteWVuQDEyNi5jb20=; Lang Jiang, ajAwMTAxMDUyMUAxNjMuY29t
†These authors have contributed equally to this work
 Junling Luo
Junling Luo Xuehua Xiang2,3†
Xuehua Xiang2,3† Guangyuan Gong
Guangyuan Gong