- Department of Rheumatology, Renji Hospital, School of Medicine, Shanghai Jiao Tong University, Shanghai, China
Objective: Rituximab remains the standard-of-care anti-CD20 therapy for anti-neutrophil cytoplasmic antibody (ANCA)-associated vasculitis (AAV). Obinutuzumab, a next-generation, glycoengineered anti-CD20 monoclonal antibody with enhanced B-cell-depleting capacity, may offer superior efficacy. We evaluated the efficacy and safety of reduced-dose obinutuzumab in 16 patients with active, refractory AAV at a single center in China.
Methods: In this retrospective chart review, we evaluated 16 consecutive patients who received reduced-dose obinutuzumab (most commonly 1,000 mg for induction) after failure to achieve remission with cyclophosphamide (CTX) and/or rituximab (RTX) or who presented with severe, treatment-naïve disease. Primary endpoints were complete remission (CR) rates at 24 and 76 weeks. Secondary endpoints included changes in renal function, inflammatory biomarkers, and immune reconstitution. Adverse events were prospectively recorded.
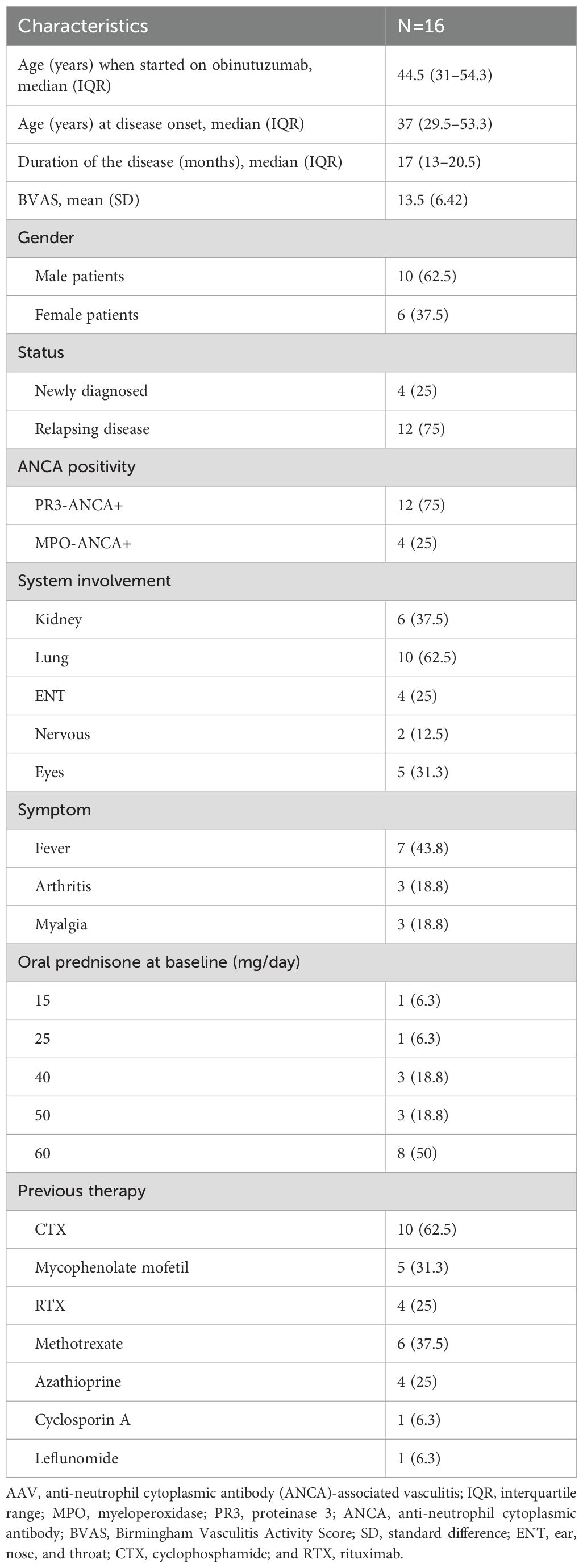
Results: The median age at obinutuzumab initiation was 44.5 years (IQR 31–54.3); 10 (62.5%) were men. The mean Birmingham Vasculitis Activity Score (BVAS) was 13.5 ± 6.4. There were 12 patients (75%) who had relapsing disease refractory to CTX/RTX, whereas four treatment-naïve patients presented with multiorgan failure. CR was achieved in 8/16 patients (50%) at week 24 and 13/16 patients (81.3%) at week 76. Obinutuzumab induced rapid clinical remission, suppressed systemic inflammation, achieved peripheral B-cell depletion, rendered ANCA-negative, and improved renal and pulmonary outcomes. No severe infections occurred. Seven patients (43.8%) developed treatment-emergent infections, predominantly respiratory (75%).
Conclusion: Reduced-dose obinutuzumab demonstrates sustained remission in refractory or relapsing active AAV, achieving high long-term remission rates with an acceptable safety profile. No severe invasive infections were observed.
Highlights
• Reduced-dose obinutuzumab induced sustained remission in patients with relapsed or refractory ANCA-associated vasculitis.
Introduction
Anti-neutrophil cytoplasmic antibody (ANCA)-associated vasculitis (AAV) is a systemic autoimmune condition characterized by necrotizing inflammation of small- to medium-sized blood vessels, frequently resulting in multiorgan involvement. The kidneys are affected in more than 75% of cases, followed by the lungs (50%–70%), the upper respiratory tract, the skin, and, less commonly, the eyes and the peripheral nervous system (1, 2). The clinical course of the disease is typified by rapid progression, cumulative organ damage, and frequent relapses; approximately 40% of patients relapse within 5 years. Renal involvement is particularly ominous, with 20%–40% of patients progressing to end-stage renal disease (ESRD), a complication that substantially worsens the long-term prognosis.
B cells are central to the pathogenesis of AAV. The 2022 EULAR guidelines recommend a combination of glucocorticoids in combination with either rituximab (RTX) or cyclophosphamide (CTX) for life-threatening or organ-threatening AAV (3). However, there is no universally accepted alternative treatment for patients who fail or are intolerant to RTX/CTX, highlighting the need for additional options.
Therapeutic anti-CD20 monoclonal antibodies are classified as Type I or Type II according to their mechanism of action. Obinutuzumab is a humanized, glycoengineered Type II anti-CD20 monoclonal antibody that induces more profound and durable B-cell depletion than Type I agents, such as RTX, via enhanced FcγRIII binding and direct induction of programmed cell death (4–6). Although currently licensed for rituximab-refractory follicular lymphoma, obinutuzumab has demonstrated efficacy in other autoimmune diseases, such as systemic lupus erythematosus and PLA2R-associated membranous nephropathy (7, 8). Preliminary data also indicate renal-protective effects, which are particularly relevant for AAV (9, 10). However, robust evidence for obinutuzumab in AAV is limited. The present study was designed to evaluate obinutuzumab’s ability to induce and maintain remission in patients with AAV refractory to CTX and/or RTX or presenting with severe disease activity.
Methods
Study design and patients
This single-center, retrospective chart review enrolled 16 adults with active AAV who either had severe disease activity or were refractory to CTX and/or RTX. Given that ANCA serotype (proteinase 3 [PR3]-ANCA and myeloperoxidase [MPO]-ANCA) is a stronger predictor of clinical presentation, disease course, treatment response, and comorbidities compared with traditional granulomatosis with polyangiitis (GPA) and microscopic polyangiitis (MPA) classification, patients were stratified by ANCA subtype. The majority of patients received a single 1,000-mg exploratory induction dose of obinutuzumab; some were maintained on either RTX or obinutuzumab. All underwent 76 weeks of follow-up in the Rheumatology Department at Shanghai Renji Hospital. Baseline characteristics and treatment details are listed in Table 1 and Supplementary Table 1. The study was approved by the Renji Hospital Ethics Committee (IRB no. LY2025 - 197-A), and all patients provided written informed consent.
All patients met the MPA or GPA criteria, as defined by the American College of Rheumatology/European Alliance of Associations for Rheumatology (11, 12) and the Chapel Hill Consensus Conference (13).
Outcome definitions
The primary endpoint was the complete remission (CR) rate at 24 weeks and throughout the 76-week follow-up period. CR was defined as a Birmingham Vasculitis Activity Score (BVAS) of zero and an oral prednisone dose of 10 mg/day or less (14). Response was defined as a ≥50% reduction in BVAS with no new manifestations (3). Relapse was defined as the new emergence or recurrence of one or more BVAS items after remission (15).
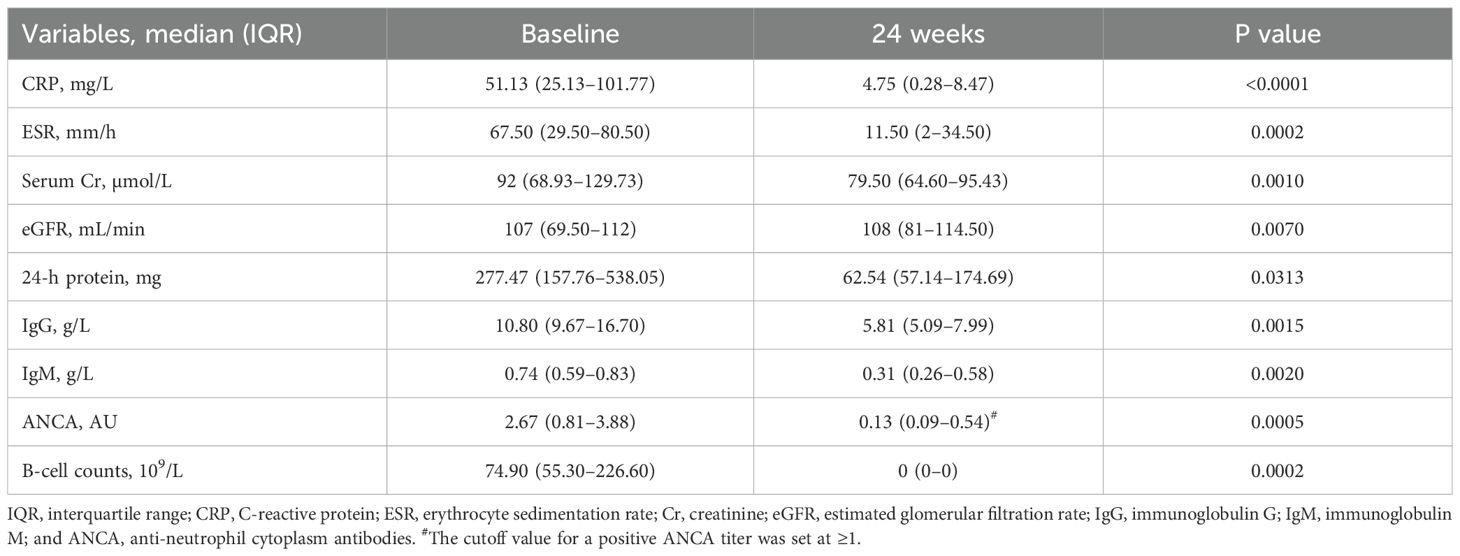
Secondary outcomes included changes in laboratory parameters and radiographic improvements on chest imaging. Laboratory parameters included inflammatory markers [C-reactive protein level (CRP), erythrocyte sedimentation rate (ESR)] and immunological markers [ANCA titers (quantified using a commercial MPO/PR3-ELISA kit, positive cutoff ≥1 AU), B-cell counts, and serum immunoglobulin levels]. Renal function was monitored using serum creatinine, estimated glomerular filtration rate (eGFR), and change in proteinuria as a surrogate for long-term renal survival (16).
Adverse events were carefully recorded to assess the safety profile of obinutuzumab treatment. These included infections and infusion-related reactions occurring within 24 h post-administration.
Statistical analysis
Categorical variables are presented as percentages (%), whereas continuous variables are presented as medians with interquartile range (IQR). Statistical significance was determined using a two-sided P-value ≤0.05. All statistical analyses were performed using the R software, version 4.3.2 (R Foundation for Statistical Computing), and GraphPad Prism 8.
Results
Patient characteristics
The baseline characteristics of the 16 enrolled patients are presented in Table 1 and Supplementary Table 1. The median age at the first obinutuzumab infusion was 44.5 years (IQR 31–54.3); 10 participants (62.5%) were men and 6 (37.5%) were women. PR3-ANCA was detected in 12 patients (75%), and the mean BVAS was 13.5 ± 6.4. Multiorgan involvement was observed in 13 patients: renal (37.5%), pulmonary (62.5%), and ear–nose–throat (25%). Fever was documented in seven patients (43.8%). There were 12 patients (75%) who received obinutuzumab for relapsing disease, of whom 11 had been previously treated with CTX and/or RTX. Four patients (all PR3-ANCA-positive) received obinutuzumab as first-line therapy immediately after diagnosis; each exhibited multiorgan disease and markedly elevated disease activity. In this subgroup, two developed acute bilateral profound sensorineural hearing loss accompanied by neuroimaging evidence of central nervous system vasculitis, and one presented with fulminant renal failure requiring urgent dialysis.
Outcomes
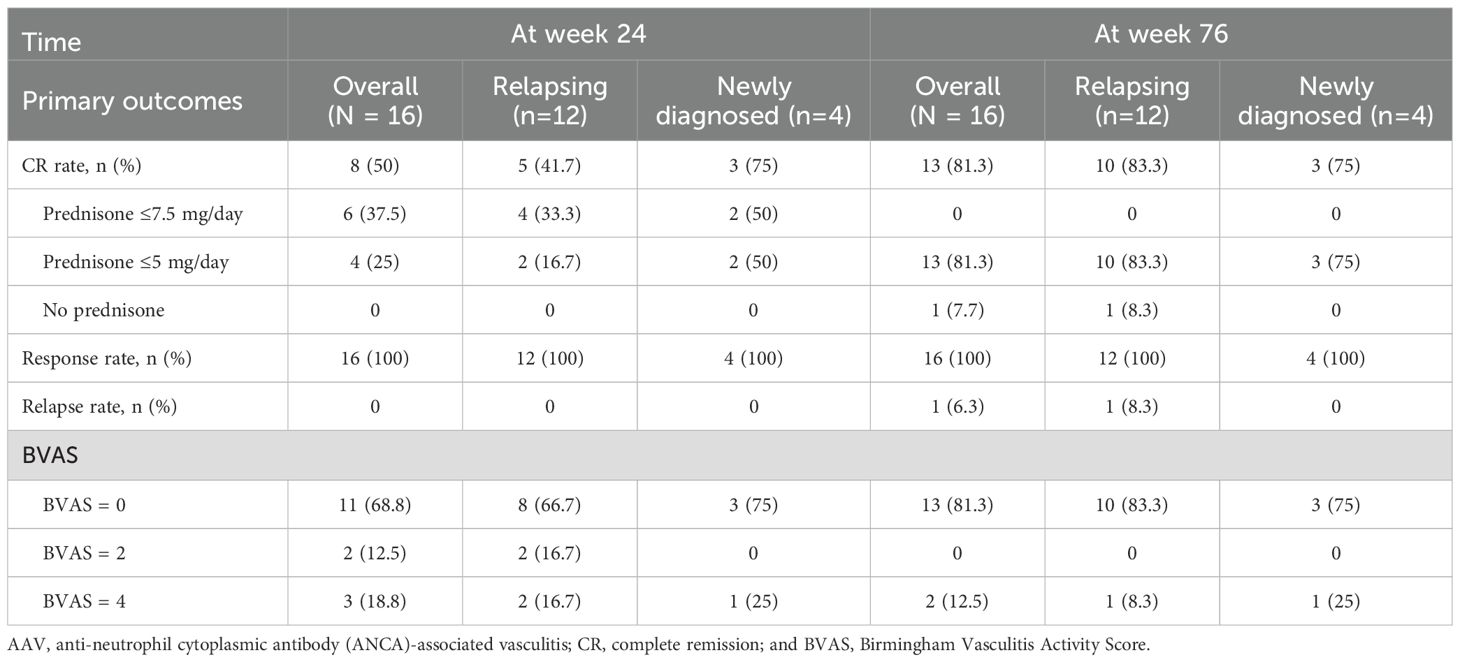
Primary outcomes
We assessed both short-term (24 weeks) and long-term (76 weeks) outcomes following obinutuzumab administration. The primary outcomes are summarized in Table 2. All 16 patients completed the 24-week assessment; one patient was subsequently lost to follow-up. Every patient demonstrated a clinical response at both time points. At 24 weeks, 50% achieved CR, with 41.7% of relapsing patients and 75% of newly diagnosed patients achieving this outcome. Nearly 70% attained a BVAS of 0. By week 76, the CR rate had risen to 81.3%, with all patients receiving ≤5 mg/day of oral prednisone and one patient entirely glucocorticoid-free. One relapse occurred at week 36. Detailed treatment regimens are provided in Supplementary Table 1.
Secondary outcomes
Secondary outcomes confirmed marked improvements in both systemic inflammation and immune parameters (Figure 1A and Table 3). Significant reductions in CRP and ESR levels were observed. Renal function, as measured by serum creatinine and eGFR, also notably improved. Although baseline proteinuria was only modestly elevated in our cohort, 24-h urinary protein excretion improved markedly after treatment. Additionally, improvement in lung condition was also observed (Figure 1B). Immunological analyses revealed profound B-cell depletion (median 74.9 × 109/L pre-infusion versus 0 post-infusion; p = 0.0002) accompanied by a parallel decline in total immunoglobulin levels. The median immunoglobulin G (IgG) level decreased from 10.8 g/L (IQR 9.67–16.7) to 5.81 g/L (IQR 5.09–7.99; p = 0.0015), and all patients became ANCA-negative within 24 weeks of the first infusion.
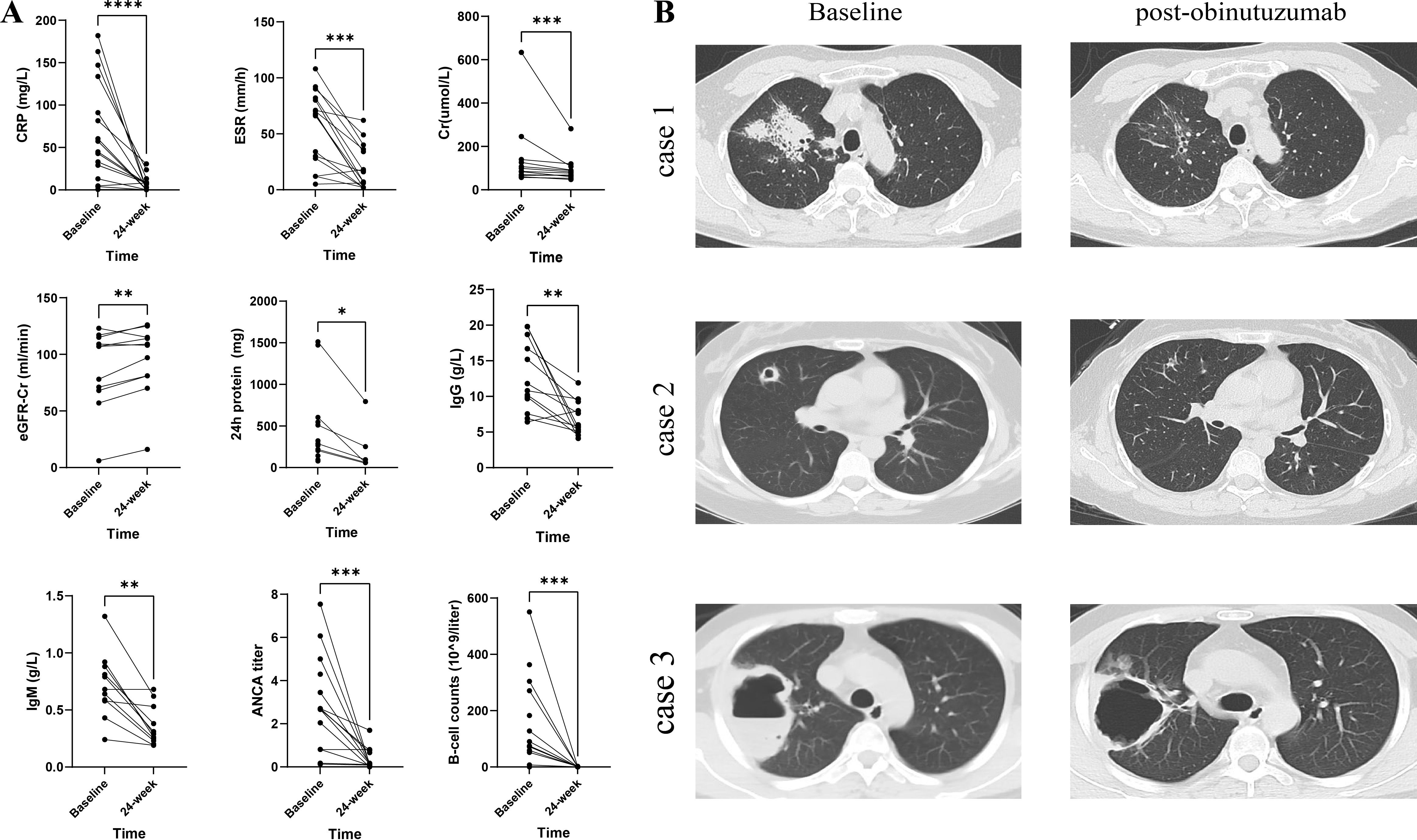
Figure 1. Laboratory parameters and chest CT images at baseline and 24 weeks post-obinutuzumab treatment. (A) Changes in the laboratory parameters of 16 patients at baseline and 24 weeks after obinutuzumab treatment. (B) High resolution CT (HRCT) images at baseline and post-obinutuzumab treatment (4, 8, and 12 weeks) in three cases. CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; Cr, creatinine; ANCA, antineutrophil cytoplasm antibodies; eGFR, estimated glomerular filtration rate; IgG, immunoglobulin G; and IgM, immunoglobulin M. *p<0.05 **p<0.01, ***p<0.001 ****p<0.0001.
Longitudinal analysis of five key biomarkers (ANCA titers, B-cell counts, IgG levels, eGFR, and serum creatinine) demonstrated durable therapeutic effects through 76 weeks (Figure 2). Rapid clinical improvement was evident within the first 4 weeks of obinutuzumab and persisted for approximately 52 weeks, as reflected by sustained ANCA negativity and profound B-cell depletion. Beyond week 52, a gradual rise in ANCA titers and B-cell repopulation was observed, indicating a potential return to baseline immune status.
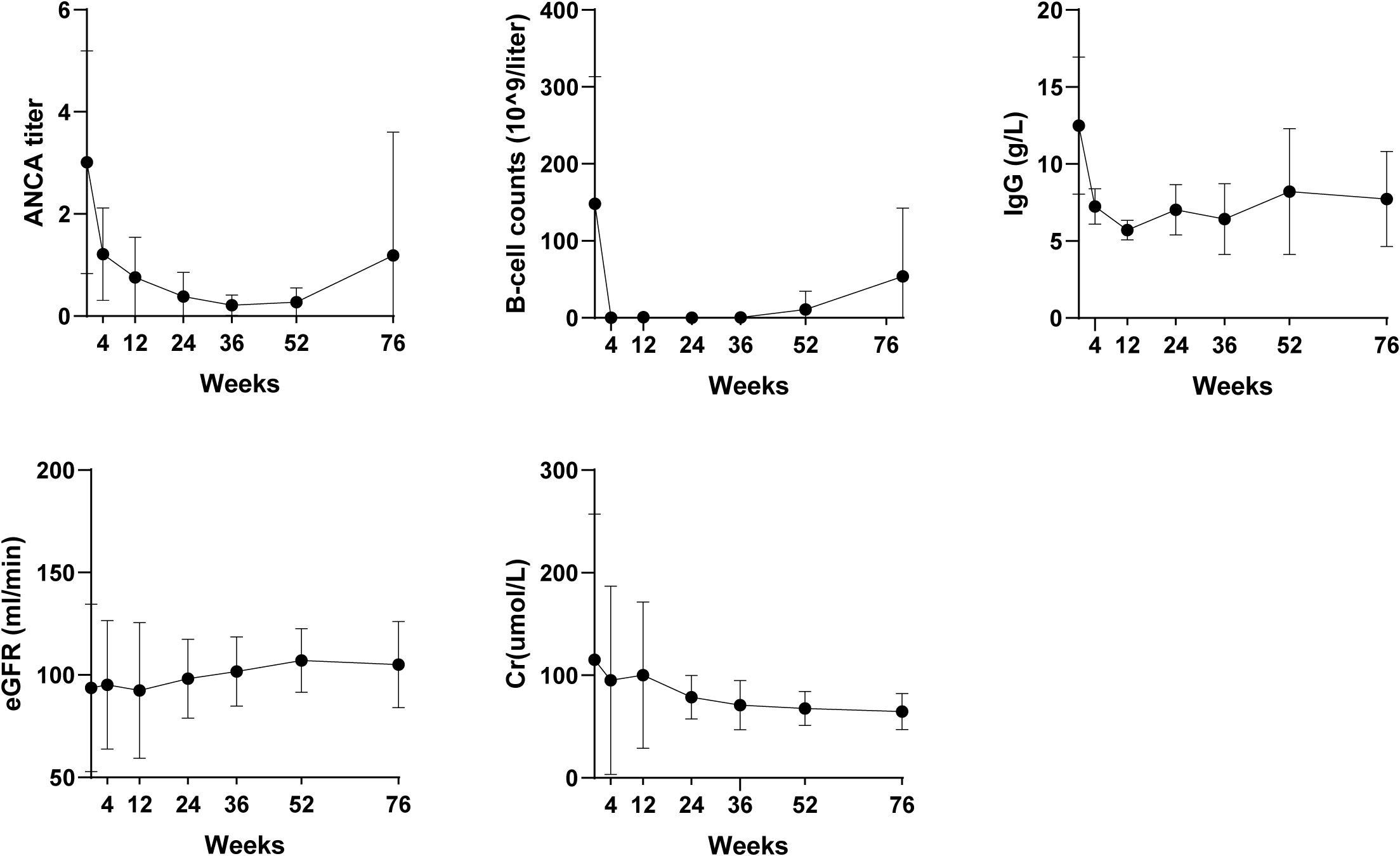
Figure 2. Temporal changes in five key laboratory parameters over 76 weeks. ANCA, antineutrophil cytoplasm antibodies; IgG, immunoglobulin G; eGFR, estimated glomerular filtration rate; Cr, creatinine.
Adverse events
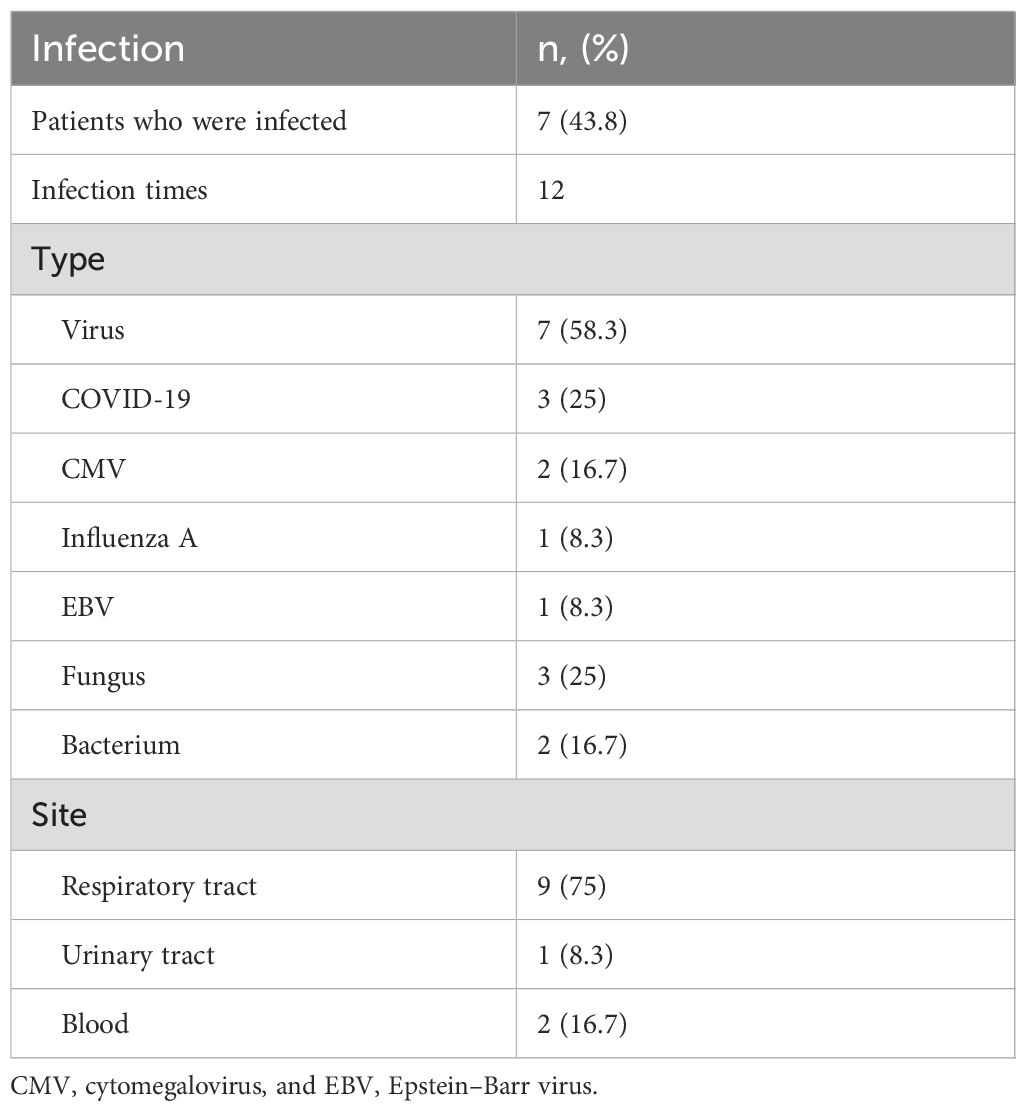
All patients received sulfamethoxazole–trimethoprim prophylaxis against Pneumocystis jirovecii pneumonia (PJP) beginning immediately after the first obinutuzumab infusion. No severe infections occurred. Infections occurred in seven patients (43.8%) with a median onset of 12 weeks (range: 5–25 weeks) after infusion. A total of 12 infectious episodes were recorded; four patients experienced recurrent infections. Viral infections predominated (7, 58.3%), followed by fungal (3, 25%) and bacterial (2, 16.7%) infections. Among the viral infections, three were caused by COVID - 19, two by cytomegalovirus (CMV), one by influenza A virus, and one by Epstein–Barr virus (EBV). The respiratory tract was the most common site of infection (9/12, 75%), with COVID - 19 accounting for half of these cases (Table 4).
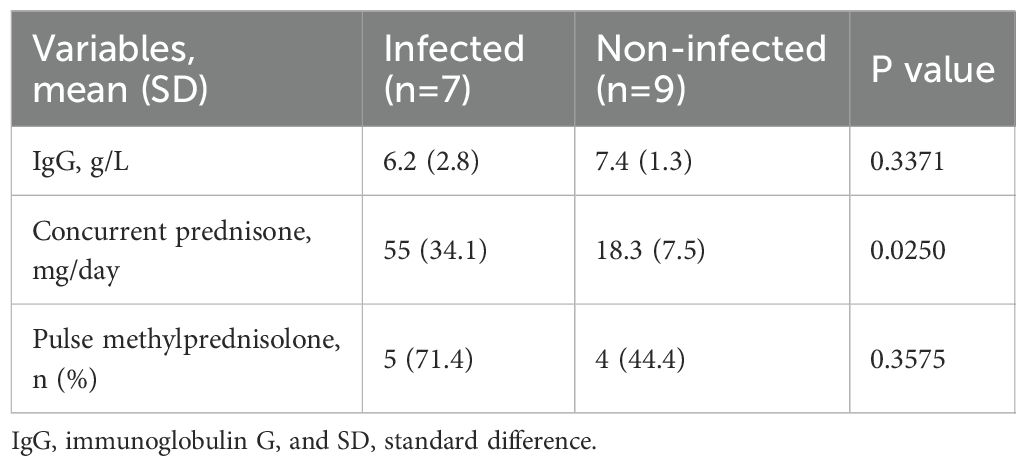
A subgroup analysis examined potential risk factors for infection, including immunoglobulin levels, concurrent prednisone doses, and prior pulse methylprednisolone therapy (Table 5). Infections occurred predominantly during the induction phase and were associated with significantly higher daily prednisone doses (infected group: mean 55 mg/day vs. non-infected group: mean 18.3 mg/day; p = 0.0250), indicating that elevated glucocorticoid exposure increases the risk of infection. However, neither prior pulse methylprednisolone treatment nor immunoglobulin levels at the time of infection differed between the two groups.
Importantly, no patient developed severe hypogammaglobulinemia (IgG <4 g/L) during follow-up; nevertheless, five patients with a marked IgG decline (mean 5.3 g/L) received intravenous immunoglobulin supplementation. Three infusion-related reactions, namely, sinus bradycardia, transient hypertension, and a mild febrile episode, resolved promptly after reducing the infusion rate.
Discussion
Obinutuzumab has already shown benefits for a spectrum of connective-tissue diseases, including systemic lupus erythematosus, anti-Jo1 syndrome, and Calcinosis cutis–Raynaud’s phenomenon–esophageal dysmotility–sclerodactylia–teleangiectasia (CREST) syndrome (17). In the present cohort, we extended these observations to refractory or relapsing AAV. At 76 weeks, CR was attained by 81% of the patients while successfully tapering prednisone to ≤5 mg/day. This was accompanied by marked reductions in ANCA titers and preservation of renal function. Our results are consistent with a case series in which three rituximab-refractory AAV patients achieved sustained remission after treatment with obinutuzumab (18).
Elevated glucocorticoid exposure is a well-established risk factor for infections. Guided by this principle, we adopted an aggressive tapering strategy: oral prednisone was initiated at 1 mg/kg/day (maximum 60 mg) and reduced to 30 mg/day by week 4 and approximately 15 mg/day by month 3, whenever clinically feasible. In this real-world cohort, the precise schedule remained at the treating physician’s discretion. Consequently, attainment of CR (prednisone ≤ 10 mg/day) often took longer than in controlled trials. Consistent with prior reports, infections clustered during the induction period and correlated with significantly higher daily prednisone doses.
Long-term follow-up results revealed that, although ANCA titers and B-cell counts began to rebound after 52 weeks, both Cr and eGFR levels remained stable through 76 weeks. This stability further supports the potential role of obinutuzumab in preserving kidney function. This benefit is especially relevant for the MPA subset, which carries the highest renal risk and is disproportionately prevalent in Asia. In our cohort, all four MPO-ANCA-positive patients, each of whom was previously refractory to CTX or RTX, achieved CR by week 76 while maintained on only 5 mg/day prednisone, highlighting obinutuzumab’s superior efficacy in treating this refractory population.
To minimize the risks of adaptive immune dysfunction and susceptibility to opportunistic infections, we administered prophylactic medications, such as sulfamethoxazole-trimethoprim prophylaxis for PJP prevention, isoniazid for tuberculosis prevention, and entecavir for hepatitis B prevention. Additionally, we would recommend vaccination for every patient, although it is not mandatory. Despite pre-medication with intravenous methylprednisolone and paracetamol to mitigate severe infusion reactions, three infusion-related adverse events were documented. All were mild, and each resolved after a transient reduction in infusion rate. Obinutuzumab-induced thrombocytopenia (19) has been well-documented in a previous study. However, this symptom was not observed in our cohort.
A key strength of this study is that it represents the largest real-world cohort of refractory or relapsing AAV patients treated with obinutuzumab to date. Despite this strength, the small sample size constrained the statistical power and limited the generalizability of our findings. Furthermore, the reduced-dose obinutuzumab regimen employed was deliberately more conservative compared with lymphoma protocols, reflecting its status as an exploratory, non-standard approach for AAV. While this lower-dose strategy may potentially reduce adverse effects, it also raises concerns regarding its ability to achieve optimal therapeutic efficacy. The absence of an evidence-based dosing framework for obinutuzumab in AAV underscores the urgent need for well-designed, randomized, controlled trials to establish standardized, effective, and safe treatment protocols. Nonetheless, this exploratory approach represents an important step toward understanding the potential role of obinutuzumab in managing refractory AAV. In our study, CR was achieved when a patient was maintained on a prednisone dosage of ≤10 mg/day. However, more stringent criteria are now favored in current clinical research and practice. The 2022 EULAR update recommends that patients with AAV achieve a dosage of ≤5 mg/day by 4–5 months (3). Furthermore, the TAPIR trial (20) supports the notion that a dosage of ≤5 mg/day, or ideally complete glucocorticoid discontinuation, may represent a more contemporary and clinically relevant definition of remission. Nevertheless, this study offers critical real-world evidence supporting obinutuzumab as an experimental treatment option for AAV, leveraging its potent B-cell-depleting activity. Future investigations should prioritize dose optimization strategies, rigorous immune function surveillance, and extended follow-up to definitively establish the therapeutic role of obinutuzumab in AAV.
Conclusions
Reduced-dose obinutuzumab demonstrated rapid and durable efficacy with a favorable safety profile in patients with refractory or relapsing active AAV, establishing it as a promising therapeutic alternative, particularly after RTX or CTX failure.
Data availability statement
The data supporting the findings of this study are available within the article, further inquiries can be directed to the corresponding author.
Ethics statement
The studies involving humans were approved by the Renji Hospital Ethics Committee (IRB no. LY2025 - 197-A) and obtained informed consent from all patients. The studies were conducted in accordance with the local legislation and institutional requirements. The participants provided their written informed consent to participate in this study. Written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article.
Author contributions
WW: Formal Analysis, Conceptualization, Investigation, Writing – review & editing, Writing – original draft. JW: Writing – review & editing, Conceptualization, Data curation. SC: Resources, Writing – review & editing, Data curation, Supervision, Conceptualization.
Funding
The author(s) declare that no financial support was received for the research and/or publication of this article.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fimmu.2025.1624234/full#supplementary-material
References
1. Geetha D and Jefferson JA. ANCA-associated vasculitis: core curriculum 2020. Am J Kidney Dis. (2020) 75:124–37. doi: 10.1053/j.ajkd.2019.04.031
2. Kronbichler A, Bajema IM, Bruchfeld A, Mastroianni Kirsztajn G, and Stone JH. Diagnosis and management of ANCA-associated vasculitis. Lancet. (2024) 403:683–98. doi: 10.1016/S0140-6736(23)01736-1
3. Hellmich B, Sanchez-Alamo B, Schirmer JH, Berti A, Blockmans D, Cid MC, et al. EULAR recommendations for the management of ANCA-associated vasculitis: 2022 update. Ann Rheumatic Dis. (2024) 83:30–47. doi: 10.1136/ard-2022-223764
4. Abeles I, Palma C, Meednu N, Payne AS, Looney RJ, and Anolik JH. B cell–directed therapy in autoimmunity. Annu Rev Immunol. (2024) 42:103–26. doi: 10.1146/annurev-immunol-083122-044829
5. Meyer S, Evers M, Jansen JHM, Buijs J, Broek B, Reitsma SE, et al. New insights in Type I and II CD20 antibody mechanisms-of-action with a panel of novel CD20 antibodies. Br J Haematology. (2018) 180:808–20. doi: 10.1111/bjh.15132
6. Marinov AD, Wang H, Bastacky SI, van Puijenbroek E, Schindler T, Speziale D, et al. The type II anti-CD20 antibody obinutuzumab (GA101) is more effective than rituximab at depleting B cells and treating disease in a murine lupus model. Arthritis Rheumatol. (2021) 73:826–36. doi: 10.1002/art.41608
7. Klomjit N, Fervenza FC, and Zand L. Successful treatment of patients with refractory PLA2R-associated membranous nephropathy with obinutuzumab: A report of 3 cases. Am J Kidney Dis. (2020) 76:883–8. doi: 10.1053/j.ajkd.2020.02.444
8. McGovern DP, McClure ME, Coates M, Bond S, Martinez Del Pero M, Mynard K, et al. Study protocol for a randomised, phase II, double-blind, experimental medicine study of obinutuzumab versus rituximab in ANCA-associated vasculitis: ObiVas. BMJ Open. (2024) 14. doi: 10.1136/bmjopen-2023-083277
9. Dall’Era M, Cisternas MG, Smilek DE, Straub L, Houssiau FA, Cervera R, et al. Predictors of long-term renal outcome in lupus nephritis trials: lessons learned from the Euro-Lupus Nephritis cohort. Arthritis Rheumatol. (2015) 67:1305–13. doi: 10.1002/art.39026
10. Tamirou F, Lauwerys BR, Dall’Era M, Mackay M, Rovin B, Cervera R, et al. A proteinuria cut-off level of 0.7 g/day after 12 months of treatment best predicts long-term renal outcome in lupus nephritis: data from the MAINTAIN Nephritis Trial. Lupus Sci Med. (2015) 2:e000123. doi: 10.1136/lupus-2015-000123
11. Suppiah R, Robson JC, Grayson PC, Ponte C, Craven A, Khalid S, et al. 2022 American College of Rheumatology/European Alliance of Associations for Rheumatology classification criteria for microscopic polyangiitis. Ann Rheumatic Dis. (2022) 81:321–6. doi: 10.1136/annrheumdis-2021-221796
12. Robson JC, Grayson PC, Ponte C, Suppiah R, Craven A, Judge A, et al. 2022 american college of rheumatology/european alliance of associations for rheumatology classification criteria for granulomatosis with polyangiitis. Arthritis Rheumatol. (2022) 74:393–9. doi: 10.1002/art.41986
13. Jennette JC, Falk RJ, Bacon PA, Basu N, Cid MC, Ferrario F, et al. 2012 revised international chapel hill consensus conference nomenclature of vasculitides. Arthritis Rheum. (2013) 65:1–11. doi: 10.1002/art.37715
14. Mukhtyar C, Lee R, Brown D, Carruthers D, Dasgupta B, Dubey S, et al. Modification and validation of the birmingham vasculitis activity score (version 3). Ann Rheum Dis. (2009) 68:1827–32. doi: 10.1136/ard.2008.101279
15. Chung SA, Langford CA, Maz M, Abril A, Gorelik M, Guyatt G, et al. 2021 american college of rheumatology/vasculitis foundation guideline for the management of antineutrophil cytoplasmic antibody–associated vasculitis. Arthritis Care Res. (2021) 73:1088–105. doi: 10.1002/acr.24634
16. Furie RA, Rovin BH, Garg JP, Santiago MB, Aroca-Martínez G, Zuta Santillán AE, et al. Efficacy and safety of obinutuzumab in active lupus nephritis. New Engl J Med. (2025). doi: 10.1056/NEJMoa2410965
17. Kvacskay P, Merkt W, Günther J, Blank N, and Lorenz HM. Obinutuzumab in connective tissue diseases after former rituximab-non-response: a case series. Ann Rheum Dis. (2022) 81:744–6. doi: 10.1136/annrheumdis-2021-221756
18. Amudala NA, Boukhlal S, Sheridan B, Langford CA, Geara A, Merkel PA, et al. Obinutuzumab as treatment for ANCA-associated vasculitis. Rheumatology. (2022) 61:3814–7. doi: 10.1093/rheumatology/keab916
19. Mechelfekh Y, Pontrucher A, Paillassa J, Temple M, and Houot R. Obinutuzumab-induced acute thrombocytopenia: Report of two cases and review of literature. Br J Haematology. (2023) 202:168–72. doi: 10.1111/bjh.18826
20. Cronholm PF, Applequist J, Krischer J, Fontenot E, Davis T, Burroughs C, et al. A study of implementation factors for a novel approach to clinical trials: constructs for consideration in the coordination of direct-to-patient online-based medical research. BMC Med Res Methodol. (2024) 24:244. doi: 10.1186/s12874-024-02352-w
Keywords: ANCA-associated vasculitis, obinutuzumab, efficacy, safety, B cells
Citation: Wu W, Wang J and Chen S (2025) Reduced-dose obinutuzumab induces remission in refractory ANCA-associated vasculitis: a report of 16 cases. Front. Immunol. 16:1624234. doi: 10.3389/fimmu.2025.1624234
Received: 08 May 2025; Accepted: 12 August 2025;
Published: 29 August 2025.
Edited by:
Alexandre Wagner Silva De Souza, Federal University of São Paulo, BrazilReviewed by:
Roberto Padoan, University of Padua, ItalyHarikrishnan Gangadharan, Government College Kottayam, India
Victor Pimentel-Quiroz, Hospital Base Guillermo Almenara Irigoyen, Peru
Copyright © 2025 Wu, Wang and Chen. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Juan Wang, anVhbi53YW5nMjAxNkBmb3htYWlsLmNvbQ==; Sheng Chen, MTM5MTc1NTYwNTJAMTM5LmNvbQ==
 Wenyi Wu
Wenyi Wu Juan Wang
Juan Wang Sheng Chen*
Sheng Chen*