- Oncology Department, Cancer Center, The First Hospital of Jilin University, Changchun, China
Antibody-drug conjugates (ADCs) enhance tumour immunogenicity through multidimensional immune modulation beyond targeted cytotoxicity. The immune remodelling of the tumour microenvironment (TME) suggests potential synergistic mechanisms with immune checkpoint inhibitors (ICIs): ICIs amplify antitumour immune responses by blocking inhibitory signals. Preclinical studies and preliminary clinical evidence demonstrate their synergistic efficacy; however, mechanistic synergy requires further experimental validation. Current challenges encompass the impact of heterogeneous TME on therapeutic outcomes and toxicity risks including interstitial lung disease. Advancing the translational potential of combination therapies necessitates optimised linker designs, development of immunostimulatory payloads, and establishment of precise biomarker frameworks. This review investigates the immunomodulatory mechanisms of ADCs, providing a theoretical foundation and novel directions for antitumour combination therapies and next-generation ADC development.
1 Introduction
ADCs, or antibody-drug conjugates, are a class of targeted-cytotoxic anticancer therapies with three major components: an antibody, a linker, and a payload (1). The payload of a classical antibody-drug conjugate is usually borne by a chemotherapeutic drug (cytotoxic drug), the antibody plays a targeting role, and the linker combines the two main components mentioned above to ensure that the drug exerts its efficacy only after it reaches the target tissue (2). The anti-tumour effects of ADCs are achieved through three main mechanisms: the target-specific cytotoxicity, the blockade of cell signal transduction pathways, and the immunologic regulation (3–5). ADCs are targeted, cytotoxic, and relatively long-term anti-neoplastic agents. Moreover, the long-term anti-tumour effect comes from its positive modulation of the anti-tumour immune effect, which enhances the immunogenicity of the tumour microenvironment (6, 7).
The regulation of anti-tumour immune activity by ADCs covers many aspects, the most critical being its regulation of various immune components in the tumour immune microenvironment (7, 8). The immunogenicity of the TME affects the response level of tumour tissues to anti-tumour drugs, and the more active the TME is, the better the prognosis of patients will be (9–11). A large number of studies have shown that ADCs can regulate the phenotypic differentiation of immune cells, the infiltration level, the secretion level of inflammatory factors, and the immunomemory formation in multiple dimensions. As a result, the immunogenicity of TME could be improved and the strength of anti-tumour immune response could be significantly increased (6, 12–15).
Considering the upward regulating effect of ADCs on the tumour immune microenvironment, some researchers have combined it with anti-tumour immunotherapy, intending to obtain better efficacy (14, 16–18). The anti-tumour effect of ICI(immune checkpoint inhibitor), depends on the target antigen expression level and the tumour microenvironment’s immunogenicity (19–22). The combination of ADCs and ICIs has been shown in clinical studies to provide patients with more significant benefits than either therapy alone (23–27).
Contemporary investigations into antibody-drug conjugates remain disproportionately centred on their tumour-killing efficacy, with scant attention accorded to their immunomodulatory functionalities. This review focuses on the modulating effect of ADCs on anti-tumour immunity. It aims to provide a more comprehensive and systematic account of the immunomodulatory effects of ADCs. And it also collate information on the relevant clinical trials of the antibody-drug conjugate and immune checkpoint inhibitor (ADC-ICI) therapy to provide theoretical support for the new therapy’s clinical application.
2 Immunomodulatory mechanisms of antibody-drug conjugates
Antibody-drug conjugates (ADCs) exert antitumour effects through a three-pronged mechanism: (1) Targeted cytotoxicity against antigen-expressing tumour cells; (2) Bystander effect-mediated elimination of adjacent malignant cells; and (3) Immunomodulatory remodelling of the tumour microenvironment (TME), enabling sustained therapeutic efficacy (4, 5). The immunoregulatory functions of ADCs primarily arise from payload-induced immunogenic cell death (ICD) and synergistic contributions of the antibody component (Figure 1).
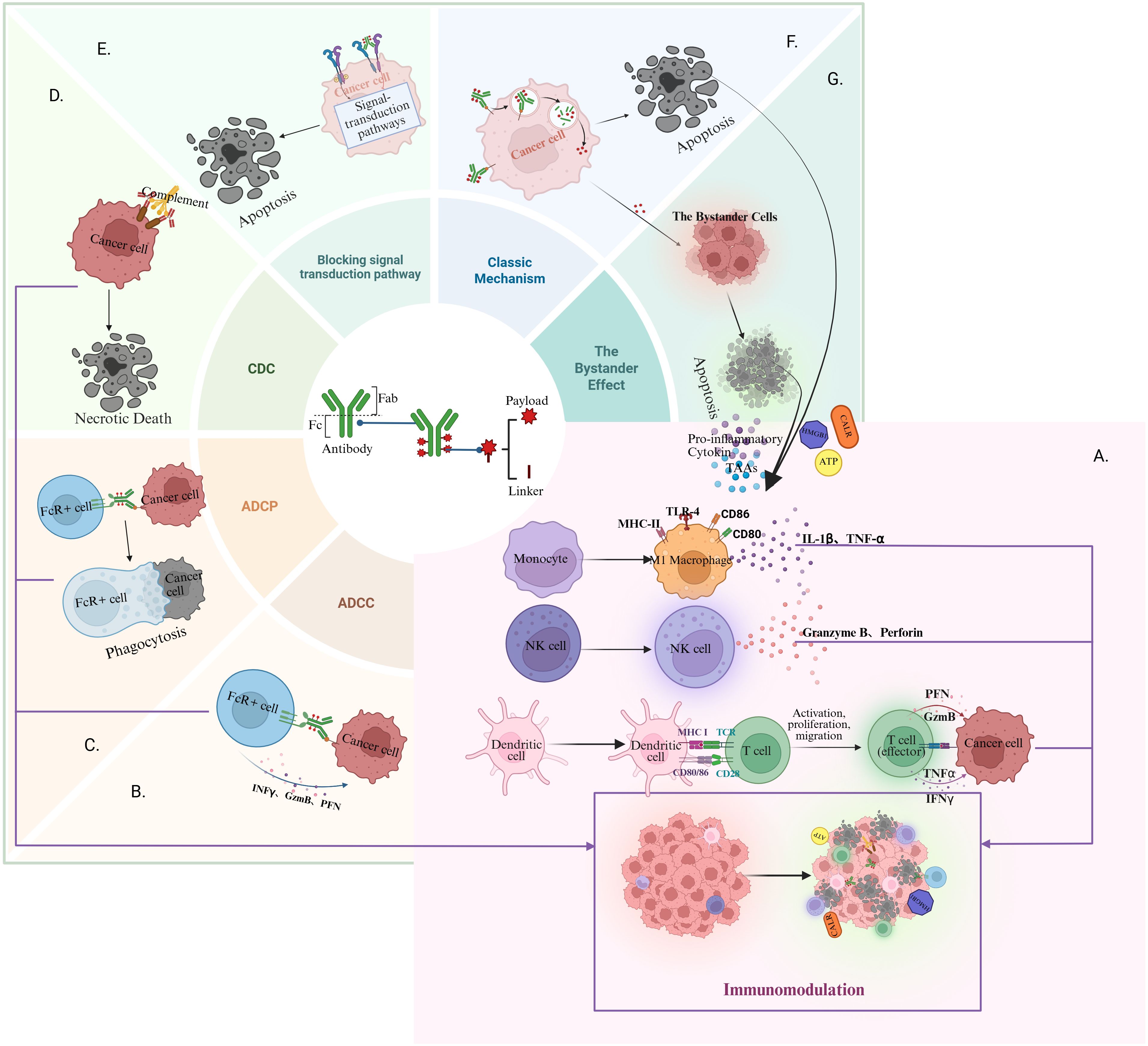
Figure 1. Key components and mechanisms of action of ADCs: (A) ADC enhances immune component activity within the TME; (B) ADC mediates tumour cell killing via the ADCC pathway; (C) ADC induces tumour cell elimination through the ADCP pathway; (D) ADC triggers the necrotic death of tumour cells via the CDC pathway; (E) ADC blocks cell signalling transduction to inhibit tumour growth; (F) ADC is recognised and internalised, and the cytotoxic payloads are released into the TME; (G) The released cytotoxic load can cause apoptosis of surrounding tumour cells via the bystander pathway. Created with Biorender.
2.1 Payload-mediated immunoregulation in ADCs
Cytotoxic payloads, including topoisomerase I inhibitors and monomethyl auristatin E (MMAE), ICD by disrupting DNA replication or microtubule dynamics. This process releases three key immunogenic signals: (1) Increase in damage-associated molecular patterns (DAMPs) such as extracellular adenosine triphosphate (ATP), high mobility group box 1 (HMGB1), and surface-exposed calreticulin (CALR), activating the anti-tumour immune effects; (2) Enhancement of cross-presentation of tumour-associated antigens (TAAs), which could prime T cell responses; and (3) High expression of pro-inflammatory cytokines like IFN-γ, which recruit and activate NK cells and T lymphocytes (6, 28–30). Brentuximab vedotin (BV), an anti-CD30-MMAE conjugate, exemplifies this mechanism by elevating IL-10 and IL-18 levels, thereby reprogramming the TME towards an immunostimulatory state (28). The ICD cascade establishes a self-amplifying loop that enhances immune-mediated tumour clearance and sustains DAMP release (14).
2.2 Antibody-dependent immune activation
The Fc domain of ADCs engages innate immunity through antibody-dependent cellular cytotoxicity (ADCC), phagocytosis (ADCP), and complement activation (CDC). These mechanisms enhance tumour lysis by natural killer cells(NK cells) and macrophages (31–33). Cleavable linkers (e.g., valine-citrulline) further amplify cytotoxicity by enabling payload diffusion to neighbouring cells, a phenomenon termed the “bystander effect.” This not only broadens tumour cell killing but also releases TAAs for cross-presentation, thereby bridging innate and adaptive immunity (34). High drug-to-antibody ratios (DARs), as exemplified by trastuzumab deruxtecan (T-DXd, DAR=8), maximise bystander activity, whereas non-cleavable linkers (e.g., in trastuzumab emtansine [T-DM1], DAR=3.5) restrict payload diffusion (35). T-DXd eradicates antigen-heterogeneous tumours via this mechanism while promoting dendritic cells (DCs) activation and durable immune memory (36).
2.3 ADC-driven remodelling of the tumour immune landscape
ADCs orchestrate multicellular immune activation within the TME: For dendritic cell activation, ADCs upregulate co-stimulatory molecules (CD80/CD86) and MHC-II on intratumoural DCs, enhancing antigen presentation (13, 37). Dolastatin-derived payloads (e.g., monomethyl auristatin E [MMAE]) further augment DC maturation, migration to lymph nodes, and T cell priming (30). Moreover, ADCs polarize tumour-associated macrophages (TAMs) towards pro-inflammatory M1 phenotypes by upregulating toll-like receptor 4 (TLR4) and suppressing scavenger receptor class A member 5 (SCARA5), thereby enhancing phagocytosis and IL-12 secretion (38). STING-targeting ADCs (e.g., αEGFR-172) synergize with DCs and NK cells via IFN-I signalling to amplify antitumour immunity (39). As well as NK cell engagement: ADCs activate NK cells via Fc-mediated ADCC (e.g., CD107a degranulation triggered by gemtuzumab ozogamicin) and ICD-derived CALR binding to NKp30 receptors (40, 41). CD25-targeted ADCs deplete immunosuppressive Tregs while preserving NK cell cytotoxicity (42). What’s more, ADCs elevate tumour-infiltrating CD8+ and Th1 cells while suppressing Treg-derived IL-10/TGF-β, thus improving the Teff/Treg ratio (42, 43). Notably, T-DXd induces epitope spreading, enabling cured mice to reject both HER2+ and HER2− tumours, indicative of antigen-agnostic immune memory (36).
2.4 Bidirectional regulation between ADCs and the TME
ADCs dynamically interact with the TME: (1) ADC-induced immunomodulation enhances sensitivity to subsequent therapies (17, 44); (2) Baseline TME features (e.g., tumour-infiltrating lymphocytes [TILs] density, IFN-γ signalling) predict ADC efficacy (12). For example, advanced triple-negative breast cancer (TNBC) patients with PD-1+ TIL-rich microenvironments exhibit superior responses to antibody-drug conjugate and ADC-ICI combinations (45). Thus it can be seen that the assessment of tumour immunogenicity within the microenvironment may provide critical guidance for ADC-based therapeutic strategies. It is also exemplified in pancreatic ductal adenocarcinoma (PDAC), where spatial heterogeneity analysis reveals that high-immunogenicity tumours (HI-PDAC) require T cell reinvigoration, whereas low-immunogenicity tumours (LI-PDAC) necessitate ICD-mediated antigen release (46). With this in mind, similar analytical methods may also enable stratification of patients according to targetable immune profiles, thereby informing precision treatment selection.
3 Combination therapies based on the immunomodulatory effects of ADCs
3.1 ADC-ICI therapies
Recent advances in combining ADCs with ICIs have demonstrated promising therapeutic potential across multiple malignancies. ADCs exert antitumour activity through targeted cytotoxic payload delivery while concurrently ICD, which promotes tumour antigen release and dendritic cell activation. This immunomodulatory mechanism provides a rational basis for synergy with ICIs, though clinical outcomes exhibit notable heterogeneity depending on tumour types and combination strategies.
3.1.1 Urothelial carcinoma
In urothelial carcinoma, the phase III EV-302 trial (N=886) (47) established enfortumab vedotin (Nectin-4-targeting ADC with MMAE payload) plus pembrolizumab as a new frontline standard, showing superior progression-free survival (median PFS 12.5 vs. 6.3 months; HR=0.45, 95% CI 0.38–0.54) and overall survival (median OS 31.5 vs. 16.1 months; HR=0.47, 95% CI 0.38–0.58) compared to platinum-based chemotherapy. Despite grade ≥3 treatment-related adverse events (TRAEs) occurring in 55.9% of patients, severe toxicities like interstitial lung disease (ILD) remained rare (<1%). In contrast, a Chinese phase II study (N=16) (48) of disitamab vedotin (HER2-targeting ADC with MMAE) combined with tislelizumab reported an objective response rate (ORR) of 62.5% in pretreated patients, with HER2-positive subgroups achieving 70% ORR. While these results highlight HER2 expression as a potential predictive biomarker, small sample sizes and lack of control arms necessitate further validation.
3.1.2 Breast cancer
Breast cancer research reveals divergent outcomes depending on payload characteristics. The phase Ib DS8201-A-U105 trial (N=82) (49) demonstrated trastuzumab deruxtecan (HER2-targeting ADC with topoisomerase I inhibitor deruxtecan [DXd]) plus nivolumab achieved ORRs of 65.6% in HER2-positive and 50% in HER2-low metastatic breast cancer. Preclinical evidence (38) suggests DXd upregulates PD-L1 and MHC-I expression, potentially enhancing T-cell recognition. However, a 20.7% incidence of ILD warrants stringent monitoring. Conversely, the phase II KATE2 trial (N=202) of trastuzumab emtansine (DM1 payload) with atezolizumab (50) showed no significant PFS improvement (median 8.2 vs. 6.8 months; HR=0.82, p=0.33), though PD-L1-positive subgroups trended towards benefit (HR=0.60), underscoring how payload immunomodulatory properties may influence therapeutic synergy. In addition, a trial of sacituzumab Govitecan combined with Pembrolizumab in patients with advanced breast cancer (N=104) (51) is ongoing, with a higher median PFS in the ADC-ICI arm than in the monotherapy arm in the preliminary results analysis.
3.1.3 Hodgkin lymphoma
In classical Hodgkin lymphoma, brentuximab vedotin (CD30-targeting MMAE ADC) combined with nivolumab post-autologous haematopoietic stem cell transplantation yielded exceptional 18-month PFS rates of 94% (N=59) (44). Mechanistically, MMAE-induced CD30+ tumour apoptosis may enhance antigen presentation and PD-1 inhibitor-mediated immune memory. Nevertheless, peripheral neuropathy (53%) and neutropenia (42%) highlight cumulative toxicity concerns with microtubule-disrupting payloads.
3.1.4 Gastric cancer
Gastric cancer studies illustrate the expanding potential of pan-HER2 strategies. A phase I trial (N=56) (17) of disitamab vedotin plus toripalimab demonstrated 50% ORR in HER2-expressing gastric/gastroesophageal junction cancer at the recommended dose, with activity maintained in HER2-low subgroups. Preclinical models further revealed complete tumour eradication and durable immune memory upon rechallenge when combining HER2-targeted ADCs with PD-1 blockade (52). Real-world data (N=38) (53) corroborated clinical efficacy (63.2% ORR, median PFS 8.2 months) without grade ≥3 TRAEs.
Despite these advances, key challenges persist. Efficacy heterogeneity across tumour types—exemplified by enfortumab vedotin’s success in urothelial carcinoma (18, 47, 54) versus trastuzumab emtansine’s limited impact in breast cancer (50)—may reflect differences in payload-mediated immunogenic potential. Furthermore, biomarker development remains inadequate, as most trials lack stratification by PD-L1 status or tumour immune microenvironment profiles. The I-SPY2.2 (55) trial’s observation of 72% pathological complete response rates in HER2-negative/immune-activated breast cancer subtypes hints at microenvironment-driven predictive factors. Information on the above researches can be found in (Table 1).
ADC-ICI combinations leverage mechanistic synergies to remodel immunosuppressive tumour microenvironments (Figure 2). However, clinical translation requires resolving biomarker deficiencies, managing toxicity management, and optimising temporal optimisation. Future directions should prioritise three axes: optimised sequencing strategies to balance efficacy and toxicity, novel ADC designs incorporating immune-priming payloads, and multidimensional biomarker profiling to identify patient subsets most likely to benefit from combinatorial approaches.
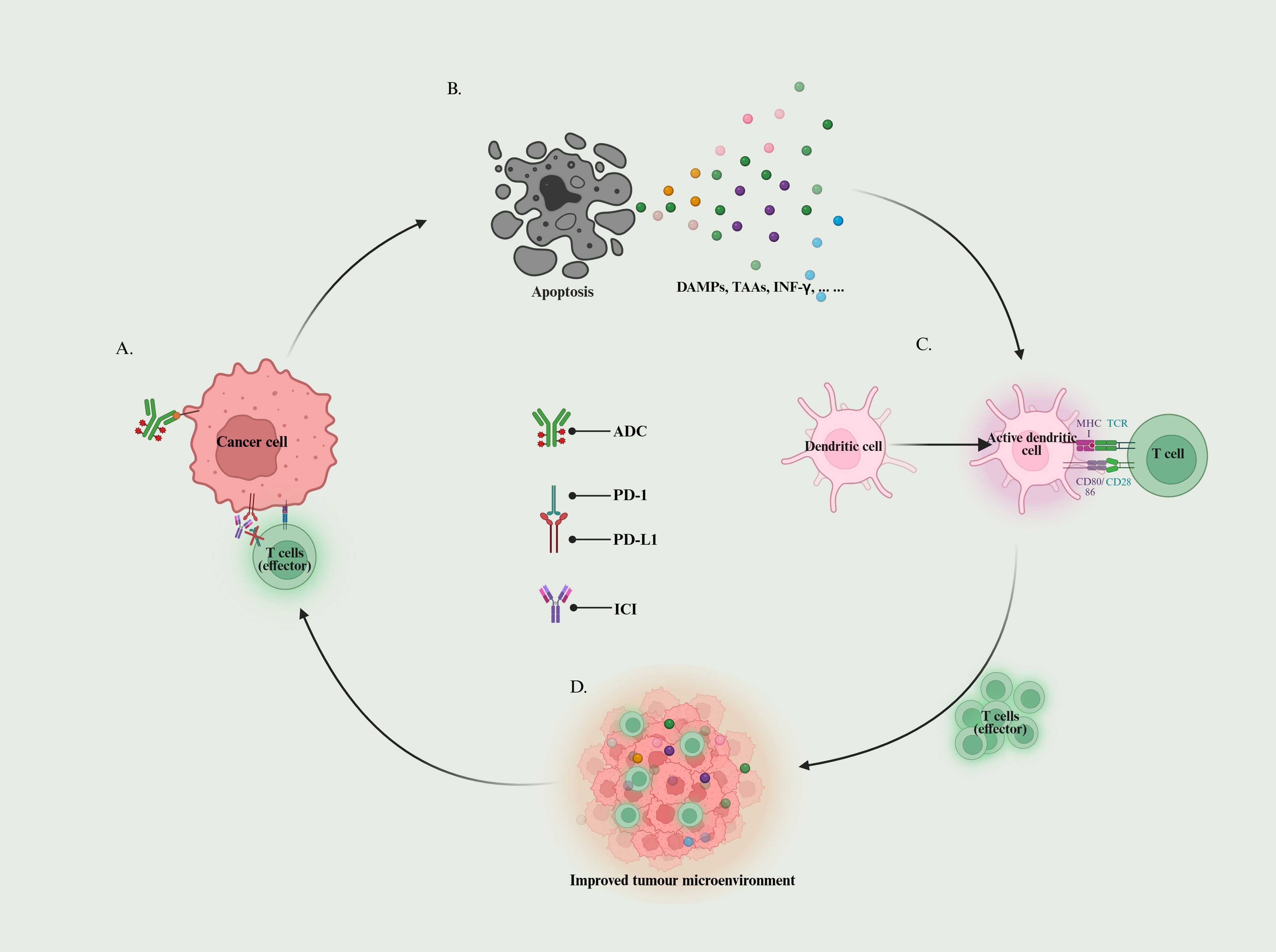
Figure 2. ADC-ICI synergistic cycle: (A) ADC recognises target antigens and ICI antagonises immune checkpoints; (B) Tumour cells release DAMPs, TAAs, IFN-γ, etc. to promote the activation and infiltration of immune cells; (C) Dendritic cells recognise TAAs and activate T cells; (D) Activated T cells migrate into tumour tissues to reduce the immune-suppressive state of the tumour microenvironment, and to enhance the sensitivity to ICI.
3.2 Combination strategies of ADCs with other immune-based therapies
ADCs reshape antitumour immunity by triggering immunogenic tumour cell apoptosis and enhancing cytotoxic T lymphocyte infiltration which primes the microenvironment for synergistic engagement with immunotherapies (12, 56). Beyond checkpoint blockade ADC-mediated tumour antigen modulation facilitates adoptive cell therapies as shown by polatuzumab vedotin enabling chimeric antigen receptor T-cell therapy (CAR-T) bridging in refractory lymphomas via CD79b-directed payload delivery (57). Emerging platforms integrate innate immune activation through TLR7 (Toll-like receptor 7)-agonist ISACs (Immune-Stimulating Antibody Conjugates) that conditionally stimulate myeloid cells upon tumour recognition (58) while surface-engineered NK cells conjugated with hydrophobic ADCs enable spatially controlled dual chemo-immunotherapy (59). Building on existing evidence, antibody-drug conjugates (ADCs) may synergise with cell-engineered immunotherapies (e.g., CAR-T) through target antigen upregulation. ADCs potentially enhance pro-inflammatory cytokine release and tumour-associated antigen presentation (56, 60). Additionally, they may promote infiltration of immune effector cells (such as TILs and NK cells) into the tumour microenvironment. These mechanisms collectively support ADC-immunotherapy combinations, though precise operational dynamics require further mechanistic validation through dedicated studies. Accordingly, ADCs may be combined with multiple immunotherapies to enhance therapeutic outcomes by ameliorating the tumour immune microenvironment. Future advances require temporal optimisation of combination schedules mechanistic biomarker identification and tumour-agnostic evaluation of ADC-immunotherapy partnerships to address heterogeneous resistance mechanisms.
4 Challenges and future perspectives
Multiple ongoing clinical trials are evaluating ADC-ICI combinations across cancer types. Despite encouraging early-phase data, these combinations confront multifaceted translational barriers. First, the predominance of phase I/II trials limits access to long-term survival data (e.g., overall survival [OS] and progression-free survival [PFS]), precluding definitive conclusions on sustained benefits. Second, combination therapy may amplify toxicity risks (e.g., immune-related adverse events and ADCs off-target effects). While some trials focus on target-specific biomarkers (e.g., HER2 or TROP2 expression), the absence of predictive biomarkers hinders patient stratification, particularly in malignancies with tumour heterogeneity. Logistical complexities (e.g., dosing sequence and timing) further complicate clinical implementation. Critically, slow trial progression (with many studies in early recruitment or planning phases) restricts data availability, impeding rapid therapeutic development. (Table 2)
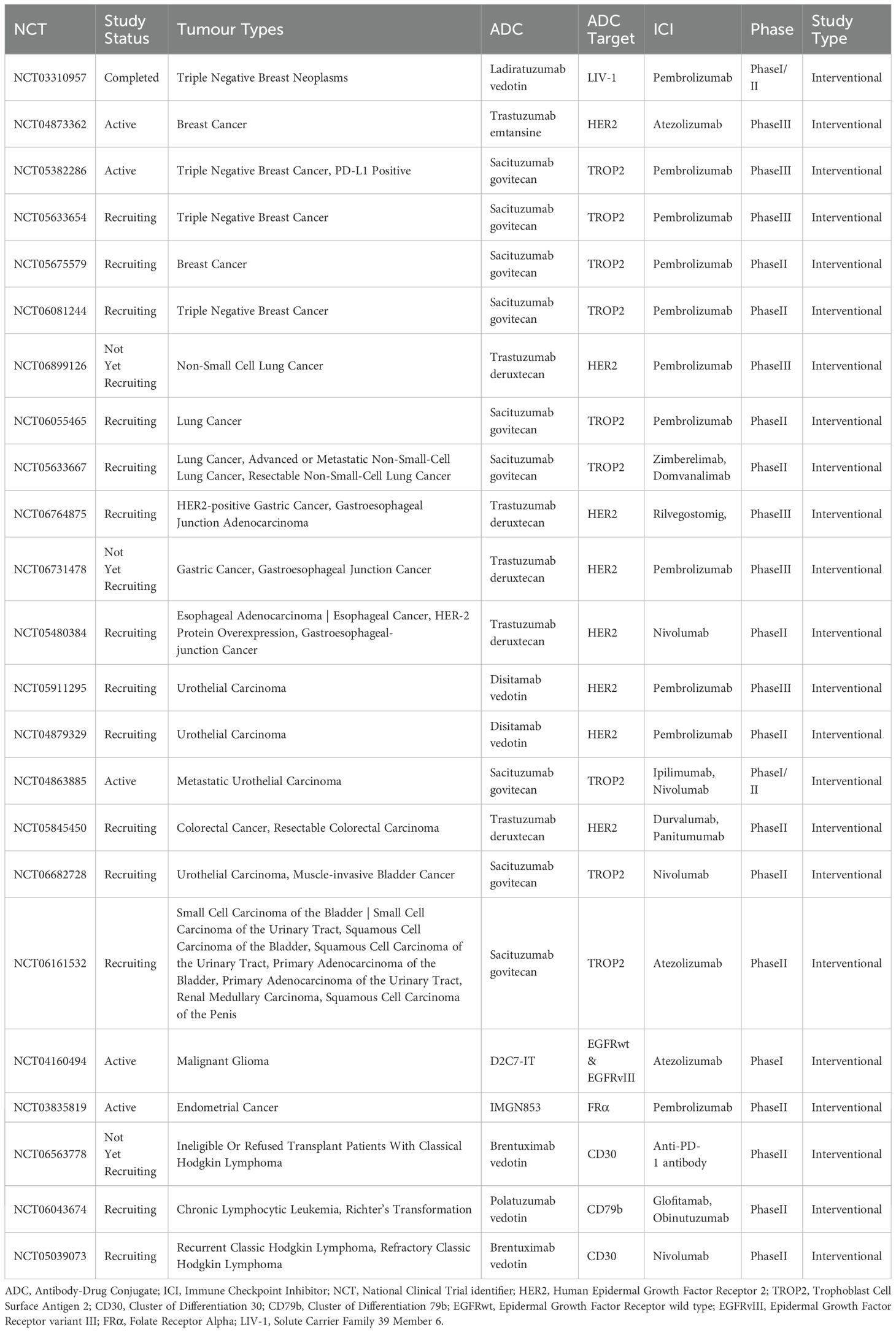
Table 2. ADC-ICI clinical trials registered on ClinicalTrials.gov (ongoing or recruiting).
Despite the remarkable anti-tumour potential of ADCs through ICD and TME remodelling, their clinical translation faces critical challenges. The immunomodulatory potential of contemporary antibody-drug conjugates remains insufficiently harnessed. First, the lack of validated biomarkers limits precision-patient stratification. Current markers like PD-L1 expression and TIL density inadequately reflect TME heterogeneity. For example, spatial immunosuppressive gradients in pancreatic cancer compromise single-biopsy assessments (46), while dynamic TME changes — such as IFN-γ signalling fluctuations—further obscure response prediction. Standardisation of ICD-related biomarkers (e.g., HMGB1, ATP) across payload classes is also urgently needed.
Toxicity management remains a major hurdle for ADC-ICI combinations. Overlapping adverse events, notably ILD and neurotoxicity, require proactive mitigation. Topoisomerase inhibitor-based ADCs (e.g., trastuzumab deruxtecan) combined with PD-1 inhibitors exhibit ILD rates up to 20% (49), while microtubule-targeting payloads (e.g., MMAE) exacerbate peripheral neuropathy, often necessitating dose reductions. Resistance mechanisms further complicate outcomes: antigen heterogeneity limits bystander effects (e.g., T-DM1’s inefficacy in HER2-low tumours due to non-cleavable linkers), while ADC-induced PD-L1 upregulation may accelerate adaptive immune evasion (61).
Novel payload research continues to target the remodelling of the immunosuppressive tumour microenvironment (39, 62). The microbially inspired CD47-listeriolysin O (LLO) conjugate exemplifies this untapped capacity by employing mechanism (63): disruption of phagocytic checkpoints coupled with lysosomal escape to activate the cGAS–STING pathway and strengthen tumour antigen cross-presentation, thereby remodelling TME immunogenicity. However, clinical translation of such novel ADCs necessitates an equilibrium between immunostimulatory intensity and drug safety. Furthermore, explorations of other strategies — including the double antibody-drug conjugate (DAD) (64) and the dual-payload antibody-drug conjugate (65, 66) — could also circumvent the limitations of traditional cytotoxicity-centric directions. Future progress depends on the engineering and mechanistic synergy exploration of ADCs.
5 Conclusion
In conclusion, the immunomodulatory mechanism of ADCs play an important part in the anti-tumour immune response. Partial researches confirm that ADCs may have the potential of synergy with ICIs. However, the effect of combination therapy demands resolution of biomarker, safety, and resistance challenges to complete a multifaceted assessment. In addition, the immune regulatory mechanism of ADCs also needs to be clarified at a more profound level to provide theoretical basis for novel research focuses.
Author contributions
RS: Investigation, Visualization, Writing – original draft, Writing – review & editing. LJ: Writing – review & editing. JC: Writing – review & editing, Supervision. ZL: Writing – review & editing, Supervision.
Funding
The author(s) declare that no financial support was received for the research, and/or publication of this article.
Acknowledgments
We acknowledge the use of BioRender.com to create the schematic diagrams presented in Figures 1, 2 of this manuscript. Moreover, We sincerely thank Prof. Jiuwei Cui and Prof. Zheng Lv for invaluable guidance and critical feedback.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Abbreviations
ADC, Antibody-Drug Conjugate; ADCC, Antibody-Dependent Cellular Cytotoxicity; ADCP, Antibody-Dependent Cellular Phagocytosis; AE, Adverse Event; CALR, Calreticulin; CAR-T, Chimeric Antigen Receptor T-Cell; CDC, Complement-Dependent Cytotoxicity; CD30, Cluster of Differentiation 30; CD79b, Cluster of Differentiation 79b; CR, Complete Response; DAMP, Damage-Associated Molecular Pattern; DAR, Drug-to-Antibody Ratio; DC, Dendritic Cell; DCR, Disease Control Rate; EGFRvIII, Epidermal Growth Factor Receptor Variant III; EGFRwt, Epidermal Growth Factor Receptor Wild Type; FRα, Folate Receptor Alpha; GEJ, Gastroesophageal Junction; HER2, Human Epidermal Growth Factor Receptor 2; HMGB1, High Mobility Group Box 1; HR, Hazard Ratio; ICI, Immune Checkpoint Inhibitor; ICD, Immunogenic Cell Death; IFN-γ, Interferon-Gamma; ILD, Interstitial Lung Disease; ISAC, Immune-Stimulating Antibody Conjugate; LIV-1, Solute Carrier Family 39 Member 6; mBC, Metastatic Breast Cancer; NCT, National Clinical Trial Identifier; ORR, Objective Response Rate; PDAC, Pancreatic Ductal Adenocarcinoma; PFS, Progression-Free Survival; PMID, PubMed Identifier; RC48, Disitamab Vedotin; SCARA5, Scavenger Receptor Class A Member 5; TAA, Tumour-Associated Antigen; T-DM1, Trastuzumab Emtansine; T-DXd, Trastuzumab Deruxtecan; TIL, Tumour-Infiltrating Lymphocyte; TLR4, Toll-Like Receptor 4; TME, Tumour Microenvironment; TNBC, Triple-Negative Breast Cancer; TRAE, Treatment-Related Adverse Event; TROP2, Trophoblast Cell Surface Antigen 2.
References
1. Khera E and Thurber GM. Pharmacokinetic and immunological considerations for expanding the therapeutic window of next-generation antibody-drug conjugates. BioDrugs. (2018) 32:465–80. doi: 10.1007/s40259-018-0302-5
2. Sang H, Liu J, Zhou F, Zhang X, Zhang J, Liu Y, et al. Target-responsive subcellular catabolism analysis for early-stage antibody-drug conjugates screening and assessment. Acta Pharm Sin B. (2021) 11:4020–31. doi: 10.1016/j.apsb.2021.05.024
3. Ruan D-Y, Wu H-X, Meng Q, and Xu R-H. Development of antibody-drug conjugates in cancer: Overview and prospects. Cancer Commun (Lond). (2024) 44:3–22. doi: 10.1002/cac2.12517
4. Chau CH, Steeg PS, and Figg WD. Antibody-drug conjugates for cancer. Lancet. (2019) 394:793–804. doi: 10.1016/S0140-6736(19)31774-X
5. Dumontet C, Reichert JM, Senter PD, Lambert JM, and Beck A. Antibody-drug conjugates come of age in oncology. Nat Rev Drug Discov. (2023) 22:641–61. doi: 10.1038/s41573-023-00709-2
6. Chang HL, Schwettmann B, McArthur HL, and Chan IS. Antibody-drug conjugates in breast cancer: overcoming resistance and boosting immune response. J Clin Invest. (2023) 133:1–10. doi: 10.1172/JCI172156
7. Nicolò E, Giugliano F, Ascione L, Tarantino P, Corti C, Tolaney SM, et al. Combining antibody-drug conjugates with immunotherapy in solid tumors: current landscape and future perspectives. Cancer Treat Rev. (2022) 106:102395. doi: 10.1016/j.ctrv.2022.102395
8. Guo J, Tang B, Fu J, Zhu X, Xie W, Wang N, et al. High-plex spatial transcriptomic profiling reveals distinct immune components and the HLA class I/DNMT3A/CD8 modulatory axis in mismatch repair-deficient endometrial cancer. Cell Oncol (Dordr). (2024) 47:573–85. doi: 10.1007/s13402-023-00885-8
9. Køstner AH, Nielsen PS, Georgsen JB, Parner ET, Nielsen MB, Kersten C, et al. Systemic inflammation associates with a myeloid inflamed tumor microenvironment in primary resected colon cancer-may cold tumors simply be too hot? Front Immunol. (2021) 12:716342. doi: 10.3389/fimmu.2021.716342
10. Wang K, Zerdes I, Johansson HJ, Sarhan D, Sun Y, Kanellis DC, et al. Longitudinal molecular profiling elucidates immunometabolism dynamics in breast cancer. Nat Commun. (2024) 15:3837. doi: 10.1038/s41467-024-47932-y
11. Wang SJ, Dougan SK, and Dougan M. Immune mechanisms of toxicity from checkpoint inhibitors. Trends Cancer. (2023) 9:543–53. doi: 10.1016/j.trecan.2023.04.002
12. Zhang S, Kohli K, Black RG, Yao L, Spadinger SM, He Q, et al. Systemic interferon-γ Increases MHC class I expression and T-cell infiltration in cold tumors: results of a phase 0 clinical trial. Cancer Immunol Res. (2019) 7:1237–43. doi: 10.1158/2326-6066.CIR-18-0940
13. Ogitani Y, Hagihara K, Oitate M, Naito H, and Agatsuma T. Bystander killing effect of DS-8201a, a novel anti-human epidermal growth factor receptor 2 antibody-drug conjugate, in tumors with human epidermal growth factor receptor 2 heterogeneity. Cancer Sci. (2016) 107:1039–46. doi: 10.1111/cas.12966
14. Rios-Doria J, Harper J, Rothstein R, Wetzel L, Chesebrough J, Marrero A, et al. Antibody-drug conjugates bearing pyrrolobenzodiazepine or tubulysin payloads are immunomodulatory and synergize with multiple immunotherapies. Cancer Res. (2017) 77:2686–98. doi: 10.1158/0008-5472.CAN-16-2854
15. Tan X, Fang P, Li K, You M, Cao Y, Xu H, et al. A HER2-targeted antibody-novel DNA topoisomerase I inhibitor conjugate induces durable adaptive antitumor immunity by activating dendritic cells. MAbs. (2023) 15:2220466. doi: 10.1080/19420862.2023.2220466
16. Pascual-Pasto G, McIntyre B, Shraim R, Buongervino SN, Erbe AK, Zhelev DV, et al. GPC2 antibody-drug conjugate reprograms the neuroblastoma immune milieu to enhance macrophage-driven therapies. J Immunother Cancer. (2022) 10:e004704. doi: 10.1136/jitc-2022-004704
17. Wang Y, Gong J, Wang A, Wei J, Peng Z, Wang X, et al. Disitamab vedotin (RC48) plus toripalimab for HER2-expressing advanced gastric or gastroesophageal junction and other solid tumours: a multicentre, open label, dose escalation and expansion phase 1 trial. EClinicalMedicine. (2024) 68:102415. doi: 10.1016/j.eclinm.2023.102415
18. O’Donnell PH, Milowsky MI, Petrylak DP, Hoimes CJ, Flaig TW, Mar N, et al. Enfortumab vedotin with or without pembrolizumab in cisplatin-ineligible patients with previously untreated locally advanced or metastatic urothelial cancer. J Clin Oncol. (2023) 41:4107–17. doi: 10.1200/JCO.22.02887
19. Sun Q, Hong Z, Zhang C, Wang L, Han Z, and Ma D. Immune checkpoint therapy for solid tumours: clinical dilemmas and future trends. Signal Transduct Target Ther. (2023) 8:320. doi: 10.1038/s41392-023-01522-4
20. Banday AH and Abdalla M. Immune checkpoint inhibitors: recent clinical advances and future prospects. Curr Med Chem. (2023) 30:3215–37. doi: 10.2174/0929867329666220819115849
21. Morad G, Helmink BA, Sharma P, and Wargo JA. Hallmarks of response, resistance, and toxicity to immune checkpoint blockade. Cell. (2021) 184:5309–37. doi: 10.1016/j.cell.2021.09.020
22. Wang Z, Wang Y, Gao P, and Ding J. Immune checkpoint inhibitor resistance in hepatocellular carcinoma. Cancer Lett. (2023) 555:216038. doi: 10.1016/j.canlet.2022.216038
23. Choi J, Jang H, Choi J, Choi Y, Yang Y, Shim MK, et al. Immune checkpoint-targeted drug conjugates: A promising tool for remodeling tumor immune microenvironment. J Control Release. (2023) 359:85–96. doi: 10.1016/j.jconrel.2023.05.031
24. Gerber H-P, Sapra P, Loganzo F, and May C. Combining antibody-drug conjugates and immune-mediated cancer therapy: What to expect? Biochem Pharmacol. (2016) 102:1–6. doi: 10.1016/j.bcp.2015.12.008
25. Rha SY and Chung HC. Breakthroughs in the systemic treatment of HER2-positive advanced/metastatic gastric cancer: from singlet chemotherapy to triple combination. J Gastric Cancer. (2023) 23:224–49. doi: 10.5230/jgc.2023.23.e6
26. Saini KS, Punie K, Twelves C, Bortini S, de Azambuja E, Anderson S, et al. Antibody-drug conjugates, immune-checkpoint inhibitors, and their combination in breast cancer therapeutics. Expert Opin Biol Ther. (2021) 21:945–62. doi: 10.1080/14712598.2021.1936494
27. Zhao M, Yan C-Y, Wei Y-N, and Zhao X-H. Breaking the mold: Overcoming resistance to immune checkpoint inhibitors. Antiviral Res. (2023) 219:105720. doi: 10.1016/j.antiviral.2023.105720
28. Advani RH, Moskowitz AJ, Bartlett NL, Vose JM, Ramchandren R, Feldman TA, et al. Brentuximab vedotin in combination with nivolumab in relapsed or refractory Hodgkin lymphoma: 3-year study results. Blood. (2021) 138:427–38. doi: 10.1182/blood.2020009178
29. Izumi Y, Kanayama M, Shen Z, Kai M, Kawamura S, Akiyama M, et al. An antibody-drug conjugate that selectively targets human monocyte progenitors for anti-cancer therapy. Front Immunol. (2021) 12:618081. doi: 10.3389/fimmu.2021.618081
30. Müller P, Martin K, Theurich S, Schreiner J, Savic S, Terszowski G, et al. Microtubule-depolymerizing agents used in antibody-drug conjugates induce antitumor immunity by stimulation of dendritic cells. Cancer Immunol Res. (2014) 2:741–55. doi: 10.1158/2326-6066.CIR-13-0198
31. Cardillo TM, Govindan SV, Sharkey RM, Trisal P, Arrojo R, Liu D, et al. Sacituzumab govitecan (IMMU-132), an anti-trop-2/SN-38 antibody-drug conjugate: characterization and efficacy in pancreatic, gastric, and other cancers. Bioconjug Chem. (2015) 26:919–31. doi: 10.1021/acs.bioconjchem.5b00223
32. Golfier S, Kopitz C, Kahnert A, Heisler I, Schatz CA, Stelte-Ludwig B, et al. Anetumab ravtansine: a novel mesothelin-targeting antibody-drug conjugate cures tumors with heterogeneous target expression favored by bystander effect. Mol Cancer Ther. (2014) 13:1537–48. doi: 10.1158/1535-7163.MCT-13-0926
33. Weisser NE, Sanches M, Escobar-Cabrera E, O’Toole J, Whalen E, Chan PWY, et al. An anti-HER2 biparatopic antibody that induces unique HER2 clustering and complement-dependent cytotoxicity. Nat Commun. (2023) 14:1394. doi: 10.1038/s41467-023-37029-3
34. Giugliano F, Corti C, Tarantino P, Michelini F, and Curigliano G. Bystander effect of antibody-drug conjugates: fact or fiction? Curr Oncol Rep. (2022) 24:809–17. doi: 10.1007/s11912-022-01266-4
35. Duro-Sánchez S, Nadal-Serrano M, Lalinde-Gutiérrez M, Arenas EJ, Bernadó Morales C, Morancho B, et al. Therapy-induced senescence enhances the efficacy of HER2-targeted antibody-drug conjugates in breast cancer. Cancer Res. (2022) 82:4670–9. doi: 10.1158/0008-5472.CAN-22-0787
36. Iwata TN, Ishii C, Ishida S, Ogitani Y, Wada T, and Agatsuma T. A HER2-targeting antibody-drug conjugate, trastuzumab deruxtecan (DS-8201a), enhances antitumor immunity in a mouse model. Mol Cancer Ther. (2018) 17:1494–503. doi: 10.1158/1535-7163.MCT-17-0749
37. Ramamoorthi G, Kodumudi K, Snyder C, Grover P, Zhang H, Greene MI, et al. Intratumoral delivery of dendritic cells plus anti-HER2 therapy triggers both robust systemic antitumor immunity and complete regression in HER2 mammary carcinoma. J Immunother Cancer. (2022) 10:e004704. doi: 10.1136/jitc-2022-004841
38. Haratani K, Yonesaka K, Takamura S, Maenishi O, Kato R, Takegawa N, et al. U3–1402 sensitizes HER3-expressing tumors to PD-1 blockade by immune activation. J Clin Invest. (2020) 130:374–88. doi: 10.1172/JCI126598
39. Wu Y-T, Fang Y, Wei Q, Shi H, Tan H, Deng Y, et al. Tumor-targeted delivery of a STING agonist improvescancer immunotherapy. Proc Natl Acad Sci U.S.A. (2022) 119:e2214278119. doi: 10.1073/pnas.2214278119
40. Hashimoto Y, Koyama K, Kamai Y, Hirotani K, Ogitani Y, Zembutsu A, et al. A novel HER3-targeting antibody-drug conjugate, U3-1402, exhibits potent therapeutic efficacy through the delivery of cytotoxic payload by efficient internalization. Clin Cancer Res. (2019) 25:7151–61. doi: 10.1158/1078-0432.CCR-19-1745
41. Nicoletti R, Lopez S, Bellone S, Cocco E, Schwab CL, Black JD, et al. T-DM1, a novel antibody-drug conjugate, is highly effective against uterine and ovarian carcinosarcomas overexpressing HER2. Clin Exp Metastasis. (2015) 32:29–38. doi: 10.1007/s10585-014-9688-8
42. Zammarchi F, Havenith K, Bertelli F, Vijayakrishnan B, Chivers S, and van Berkel PH. CD25-targeted antibody-drug conjugate depletes regulatory T cells and eliminates established syngeneic tumors via antitumor immunity. J Immunother Cancer. (2020) 8:e000860. doi: 10.1136/jitc-2020-000860
43. Xue D, Liu P, Chen W, Zhang C, and Zhang L. An anti-CD103 antibody-drug conjugate prolongs the survival of pancreatic islet allografts in mice. Cell Death Dis. (2019) 10:735. doi: 10.1038/s41419-019-1980-8
44. Herrera AF, Chen L, Nieto Y, Holmberg L, Johnston P, Mei M, et al. Brentuximab vedotin plus nivolumab after autologous haematopoietic stem-cell transplantation for adult patients with high-risk classic Hodgkin lymphoma: a multicentre, phase 2 trial. Lancet Haematol. (2023) 10:e14–23. doi: 10.1016/S2352-3026(22)00318-0
45. Qin G, Wang X, Ye S, Li Y, Chen M, Wang S, et al. NPM1 upregulates the transcription of PD-L1 and suppresses T cell activity in triple-negative breast cancer. Nat Commun. (2020) 11:1669. doi: 10.1038/s41467-020-15364-z
46. Karamitopoulou E, Wenning AS, Acharjee A, Aeschbacher P, Marinoni I, Zlobec I, et al. Spatial heterogeneity of immune regulators drives dynamic changes in local immune responses, affecting disease outcomes in pancreatic cancer. Clin Cancer Res. (2024) 30:4215–26. doi: 10.1158/1078-0432.CCR-24-0368
47. Powles T, Valderrama BP, Gupta S, Bedke J, Kikuchi E, Hoffman-Censits J, et al. Enfortumab vedotin and pembrolizumab in untreated advanced urothelial cancer. N Engl J Med. (2024) 390:875–88. doi: 10.1056/NEJMoa2312117
48. Zhu K, Chang Y, Zhao D, Guo A, Cao J, Wu C, et al. Expression of HER2 in high-grade urothelial carcinoma based on Chinese expert consensus and the clinical effects of disitamab vedotin-tislelizumab combination therapy in the treatment of advanced patients. Front Pharmacol. (2024) 15:1355081. doi: 10.3389/fphar.2024.1355081
49. Hamilton E, Galsky MD, Ochsenreither S, Del Conte G, Martín M, De Miguel MJ, et al. Trastuzumab deruxtecan with nivolumab in HER2-expressing metastatic breast or urothelial cancer: analysis of the phase ib DS8201-A-U105 study. Clin Cancer Res. (2024) 30:5548–58. doi: 10.1158/1078-0432.CCR-24-1513
50. Emens LA, Esteva FJ, Beresford M, Saura C, De Laurentiis M, Kim S-B, et al. Trastuzumab emtansine plus atezolizumab versus trastuzumab emtansine plus placebo in previously treated, HER2-positive advanced breast cancer (KATE2): a phase 2, multicentre, randomised, double-blind trial. Lancet Oncol. (2020) 21:1283–95. doi: 10.1016/S1470-2045(20)30465-4
51. Garrido-Castro AC, Kim SE, Desrosiers J, Nanda R, Carey LA, Clark AS, et al. SACI-IO HR+: A randomized phase II trial of sacituzumab govitecan with or without pembrolizumab in patients with metastatic hormone receptor-positive/HER2-negative breast cancer. J Clin Oncol. (2024) 42:LBA1004–LBA1004. doi: 10.1200/JCO.2024.42.17_suppl.LBA1004
52. Huang L, Wang R, Xie K, Zhang J, Tao F, Pi C, et al. A HER2 target antibody drug conjugate combined with anti-PD-(L)1 treatment eliminates hHER2+ tumors in hPD-1 transgenic mouse model and contributes immune memory formation. Breast Cancer Res Treat. (2022) 191:51–61. doi: 10.1007/s10549-021-06384-4
53. Xu J, Zhang H, Zhang L, Chu X, Li Y, Li G, et al. Real-world effectiveness and safety of RC48-ADC alone or in combination with PD-1 inhibitors for patients with locally advanced or metastatic urothelial carcinoma: A multicenter, retrospective clinical study. Cancer Med. (2023) 12:21159–71. doi: 10.1002/cam4.6680
54. Hoimes CJ, Flaig TW, Milowsky MI, Friedlander TW, Bilen MA, Gupta S, et al. Enfortumab vedotin plus pembrolizumab in previously untreated advanced urothelial cancer. J Clin Oncol. (2023) 41:22–31. doi: 10.1200/JCO.22.01643
55. Shatsky RA, Trivedi MS, Omene CO, Kalinsky K, Torres ETR, Thomas B, et al. Rates of pathologic complete response (pCR) after datopotamab deruxtecan (Dato) plus durvalumab (Durva) in the neoadjuvant setting: Results from the I-SPY2.2 trial. J Clin Oncol. (2024) 42:LBA501–1. doi: 10.1200/JCO.2024.42.17_suppl.LBA501
56. Fuentes-Antrás J, Genta S, Vijenthira A, and Siu LL. Antibody-drug conjugates: in search of partners of choice. Trends Cancer. (2023) 9:339–54. doi: 10.1016/j.trecan.2023.01.003
57. Liebers N, Duell J, Fitzgerald D, Kerkhoff A, Noerenberg D, Kaebisch E, et al. Polatuzumab vedotin as a salvage and bridging treatment in relapsed or refractory large B-cell lymphomas. Blood Adv. (2021) 5:2707–16. doi: 10.1182/bloodadvances.2020004155
58. Kang M, Moon S-J, Kim JY, Beck A, Allen M, Nelson J, et al. Abstract 5616: Tumor-targeted immune activation via a site-specific TLR7-agonist antibody-drug conjugate. Cancer Res. (2022) 82:5616–6. doi: 10.1158/1538-7445.AM2022-5616
59. Lee DY, Lim KS, Valencia GM, Jung M, Bull DA, and Won Y-W. One-step method for instant generation of advanced allogeneic NK cells. Adv Sci (Weinh). (2018) 5:1800447. doi: 10.1002/advs.201800447
60. Hutchings M, Sureda A, Terol MJ, Bosch Albareda F, Corradini P, Larsen TS, et al. Glofitamab (Glofit) in combination with polatuzumab vedotin (Pola): phase ib/II preliminary data support manageable safety and encouraging efficacy in relapsed/refractory (R/R) diffuse large B-cell lymphoma (DLBCL). Blood. (2021) 138:525. doi: 10.1182/blood-2021-148359
61. Yao J, Ji L, Wang G, and Ding J. Effect of neutrophils on tumor immunity and immunotherapy resistance with underlying mechanisms. Cancer Commun (Lond). (2025) 45:15–42. doi: 10.1002/cac2.12613
62. Malli Cetinbas N, Monnell T, Soomer-James J, Shaw P, Lancaster K, Catcott KC, et al. Tumor cell-directed STING agonist antibody-drug conjugates induce type III interferons and anti-tumor innate immune responses. Nat Commun. (2024) 15:5842. doi: 10.1038/s41467-024-49932-4
63. Schrank BR, Wang Y, Wu A, Tran N, Lee D, Edwards J, et al. An antibody-toxin conjugate targeting CD47 linked to the bacterial toxin listeriolysin O for cancer immunotherapy. Nat Cancer (2025) 6:511–27. doi: 10.1038/s43018-025-00919-0
64. McGregor BA, Sonpavde GP, Kwak L, Regan MM, Gao X, Hvidsten H, et al. The Double Antibody Drug Conjugate (DAD) phase I trial: sacituzumab govitecan plus enfortumab vedotin for metastatic urothelial carcinoma. Ann Oncol. (2024) 35:91–7. doi: 10.1016/j.annonc.2023.09.3114
65. Zhou ZZ, Si Y, Zhang J, Chen K, George A, Kim S, et al. A dual-payload antibody-drug conjugate targeting CD276/B7-H3 elicits cytotoxicity and immune activation in triple-negative breast cancer. Cancer Res. (2024) 84:3848–63. doi: 10.1158/0008-5472.CAN-23-4099
Keywords: antibody-drug conjugates, immune checkpoint inhibitors, tumour micro-environment, immunoregulation, tumour immunology
Citation: Shi R, Jia L, Lv Z and Cui J (2025) Another power of antibody-drug conjugates: immunomodulatory effect and clinical applications. Front. Immunol. 16:1632705. doi: 10.3389/fimmu.2025.1632705
Received: 21 May 2025; Accepted: 05 August 2025;
Published: 20 August 2025.
Edited by:
Jack Shih-Hsun Chen, AbbVie, United StatesReviewed by:
Whi-An Kwon, Hanyang University, Republic of KoreaCopyright © 2025 Shi, Jia, Lv and Cui. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Zheng Lv, bHZ6QGpsdS5lZHUuY24=; Jiuwei Cui, Q3VpandAamx1LmVkdS5jbg==
†ORCID: Zheng Lv, orcid.org/0009-0007-8546-5681
Jiuwei Cui, orcid.org/0000-0001-6496-7550
 Ruotong Shi
Ruotong Shi Lin Jia
Lin Jia Zheng Lv
Zheng Lv Jiuwei Cui
Jiuwei Cui