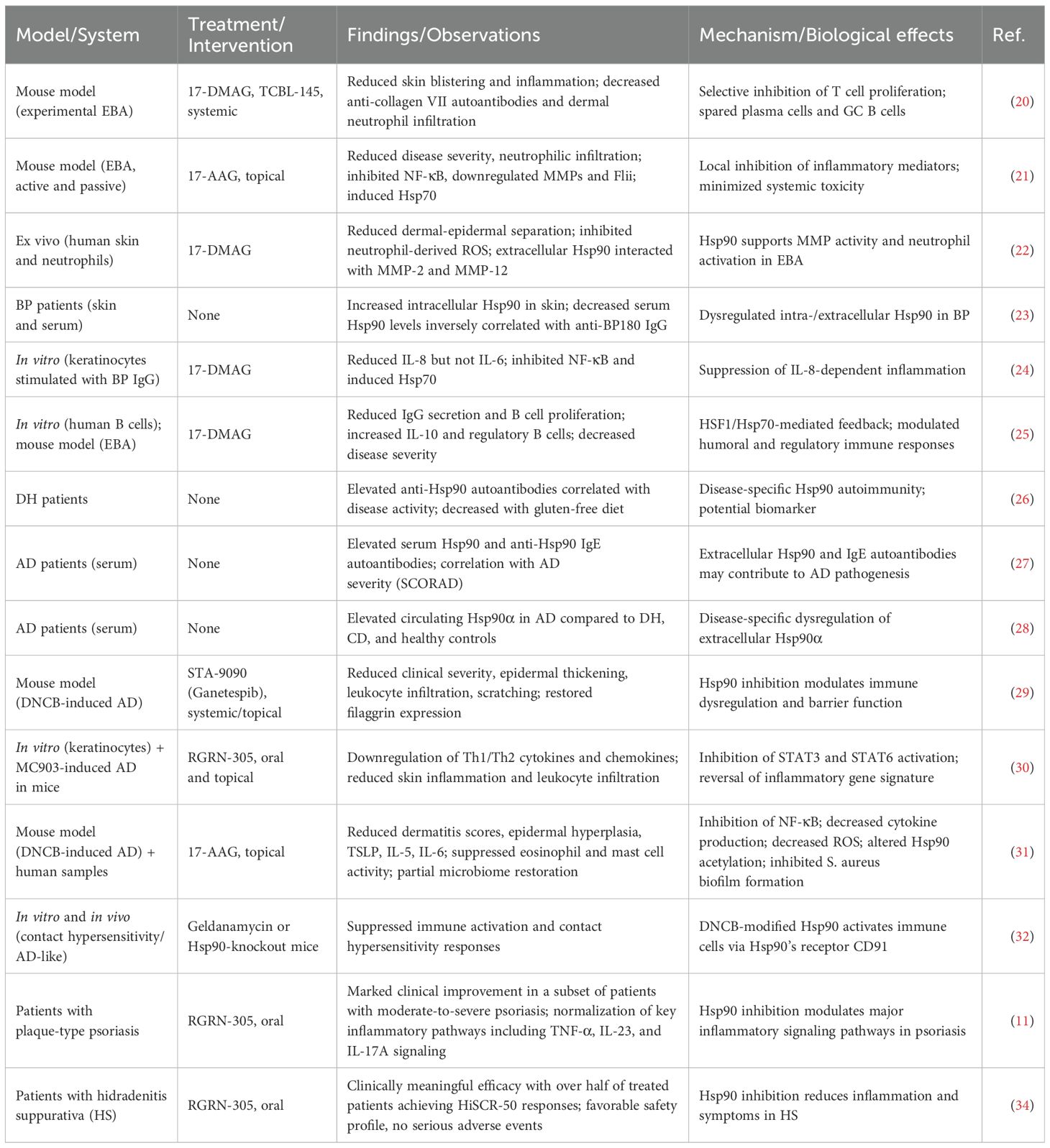
- Laboratory of Cellular and Molecular Immunology, Department of Molecular Biology, Faculty of Biology, University of Gdansk, Gdansk, Poland
Heat shock protein 90 (Hsp90) is a molecular chaperone that plays a critical role in stabilizing and regulating numerous client proteins involved in inflammation, immune activation, and skin barrier homeostasis. Emerging evidence suggests that Hsp90 contributes to the pathophysiology of atopic dermatitis (AD), a chronic inflammatory skin disorder characterized by immune dysregulation, epidermal barrier dysfunction, and microbial imbalance. Notably, elevated intracellular Hsp90 activity has been reported in peripheral blood leukocytes of AD patients, alongside increased extracellular Hsp90 and anti-Hsp90 IgE antibodies. Preclinical studies employing murine models of AD, including dinitrochlorobenzene (DNCB)- and calcipotriol (MC903)-induced dermatitis, have demonstrated that both topical and systemic inhibition of Hsp90 ameliorates disease severity. These improvements correlate with reduced epidermal hyperplasia, decreased expression of Th/Th2 cytokines, attenuation of keratinocyte-derived alarmins, and suppression of inflammation. Additionally, Hsp90 inhibition limits the infiltration or activation of immune cells such as T cells, neutrophils, eosinophiles, and mast cells in the skin. Mechanistic investigations reveal that Hsp90 blockade downregulates key signaling pathways implicated in AD pathogenesis, notably NF-κB and JAK-STAT. In vitro studies further corroborate that Hsp90 inhibition reduces proinflammatory responses in keratinocytes, CD4+ T cells, and eosinophils. Beyond modulating skin inflammation, Hsp90 blockade partially restores gut microbiota dysbiosis and impairs Staphylococcus aureus biofilm formation, both relevant to AD pathogenesis. Although clinical data on Hsp90 inhibitors in AD are still lacking, early-phase trials in psoriasis and hidradenitis suppurativa suggest potential therapeutic benefit. Collectively, these findings underscore a multifaceted role for Hsp90 in AD and support its potential as a promising novel therapeutic target.
Introduction
Atopic dermatitis (AD) is a highly prevalent chronic inflammatory skin disease characterized by relapsing eczematous lesions, pruritus, and profound impact on quality of life. Although recent therapeutic developments have improved disease management, AD remains incurable for most patients, with a high burden of disease persistence and relapse. Its multifactorial pathogenesis—encompassing genetic predisposition, environmental triggers, epidermal barrier dysfunction, and immune dysregulation—poses substantial diagnostic and therapeutic challenges (1–4). Recent advances in immunological research have expanded our understanding AD beyond the traditional Th2-dominated paradigm. AD exhibits a complex immune landscape, including significant involvement of Th22, Th1, and Th17 pathways depending on the subtype of the disease. This heterogeneity is further supported by single-cell transcriptomic studies and biomarker-based endotyping, which reveal distinct immune signatures and anatomical site–specific inflammation, underscoring the need for personalized therapeutic approaches targeting multiple immune axes (5, 6). Simultaneously, the role of the microbiome has gained prominence: Staphylococcus aureus colonization and microbial imbalance correlate with barrier defects and disease severity, while gut–skin axis perturbations may influence systemic immune priming (7). Environmental factors, including urbanization, pollution, and climate variability, further contribute to AD exacerbations and chronicity. The exposome framework is increasingly used to interpret environmental contributions to AD, recognizing the cumulative effect of chemical, physical, and psychosocial exposures throughout life (8). Despite the advent of targeted systemic therapies—such as biologics directed against IL - 4Rα, IL - 13, IL - 31 and small-molecule JAK inhibitors—a large subset of patients do not achieve long-term remission. Primary non-response, secondary loss of efficacy, and treatment discontinuation remain common (9, 10). These limitations underscore an urgent need to identify and therapeutically target upstream pathological mechanisms that drive chronicity, immune imbalance, and barrier dysfunction in AD. Among emerging intracellular regulators, heat shock protein 90 (Hsp90) has gained increasing attention as a therapeutic target, with growing evidence from in vitro studies, preclinical models, and a phase I clinical proof-of-concept trial in patients with plaque-type psoriasis (11, 12). The aim of this mini-review is to provide a focused analysis of the role of Hsp90 in the pathophysiology of atopic dermatitis and to evaluate the therapeutic potential of its inhibitors as a novel class of disease-modifying agents.
HSP90 as a master regulator of cellular signaling and immune responses
Heat shock proteins (HSPs) are a highly conserved family of molecular chaperones that preserve cellular proteostasis by assisting in the folding, assembly, stabilization, and refolding of client proteins. Among them, heat shock protein 90 (HSP90) is one of the most abundant and versatile, comprising up to 2% of total cellular protein under basal conditions and increasing further under stress. HSP90 plays a crucial role in maintaining protein homeostasis and mediating various signal transduction pathways in both physiological and pathological contexts. The HSP90 family consists of multiple isoforms, including cytosolic HSP90α (HSP90AA1, inducible) and HSP90β (HSP90AB1, constitutive), GRP94/gp96 located in the endoplasmic reticulum, and TRAP1 in mitochondria (13). Each HSP90 homolog performs distinct roles tailored to its cellular localization. HSP90AA1 (HSP90α), inducible under stress, predominantly assists in folding and stabilizing client proteins involved in signal transduction and stress responses in the cytosol. In contrast, HSP90AB1 (HSP90β) maintains essential proteostasis during normal conditions, supporting constitutive cellular functions. GRP94, residing in the endoplasmic reticulum, is a pivotal component of the unfolded protein response (UPR), ensuring protein quality control and assisting in the maturation of secretory and membrane proteins. Meanwhile, TRAP1 functions within mitochondria to regulate bioenergetics, modulate reactive oxygen species (ROS) levels, and inhibit apoptosis, thereby contributing to cellular survival. These compartment-specific activities have been predominantly explored within oncology, revealing their contributions to cancer cell metabolism, proliferation, and resistance to stress. Crucially, the intracellular functions of these homologs are functionally and mechanistically distinct from their extracellular roles, underscoring the complexity of HSP90 biology in health and disease (14).
HSP90 operates through a dynamic ATPase cycle governed by its N-terminal, middle, and C-terminal domains. This cycle is tightly regulated by post-translational modifications (PTMs) such as acetylation, phosphorylation, and SUMOylation, which modulate HSP90’s interaction with co-chaperones and its repertoire of client proteins (15, 16). Functionally, HSP90 stabilizes a broad range of client proteins, including kinases (e.g., JAK), nuclear hormone receptors, and transcription factors. Many of these proteins are central to signaling pathways that control proliferation, differentiation, immunity, and survival. Key cascades supported by HSP90 include JAK/STAT, NF-κB, MAPK/ERK, PI3K/AKT, and TLR signaling. By facilitating the proper folding and function of these proteins, HSP90 orchestrates critical cellular responses to environmental stimuli (17). Importantly, HSP90 is also found outside cells. Extracellular HSP90 (eHSP90), particularly the α isoform, can be actively secreted in response to stress or hypoxia, or passively released from necrotic or damaged cells. eHSP90 functions as a damage-associated molecular pattern (DAMP) and engages with pattern recognition receptors such as Toll-like receptor 4 (TLR4), CD91, and LOX - 1 to modulate immune and inflammatory signaling. These interactions activate downstream NF-κB and MAPK cascades, increasing the production of pro-inflammatory cytokines, chemokines, and matrix metalloproteinases (MMPs), and facilitating leukocyte recruitment, tissue remodeling, and cell motility (18, 19). Beyond HSP90α, other HSP90 homologs also contribute to the extracellular signaling landscape. GRP94 and HSP90β have been detected in secretomes of stressed or transformed cells, where they can influence antigen presentation, modulate macrophage and dendritic cell activation, and participate in the regulation of innate immune checkpoints. Extracellular GRP94, in particular, has been implicated in chaperoning immunogenic peptides and facilitating their transfer to antigen-presenting cells, thereby linking cellular stress to adaptive immune responses. TRAP1, although predominantly mitochondrial, may also exert extracellular immunoregulatory effects upon non-canonical release, though the mechanisms remain less well defined. Collectively, these extracellular activities of HSP90 isoforms expand their canonical intracellular functions and position them as key mediators of intercellular communication in immunity and disease contexts (14).
Beyond its roles in immunity and inflammation, HSP90 is a critical enabler of oncogenic transformation. It stabilizes a range of oncogenic client proteins, including mutated p53, BCR-ABL, HER2, and others, thereby supporting tumor growth, metastasis, and resistance to therapy. As a result, HSP90 inhibitors such as geldanamycin derivatives (e.g., 17-AAG) and newer small molecules have been developed to disrupt these interactions. By promoting proteasomal degradation of oncogenic proteins, HSP90 inhibitors exhibit potent anti-cancer activity. Several such compounds are in clinical trials across various malignancies, highlighting the translational promise of targeting HSP90 in oncology (13, 17).
Given the central involvement of HSP90 in cellular signaling, immune regulation, and oncogenesis—both inside and outside the cell—this chaperone remains a critical target for therapeutic intervention. A detailed understanding of its molecular structure, regulation, and diverse biological functions forms the foundation for exploring its roles in human disease and for developing targeted strategies to modulate its activity.
Hsp90 as a potential therapeutic target in autoimmune bullous skin diseases
Although this review focuses primarily on AD, it is important to acknowledge the roles and therapeutic potential of Hsp90 in other inflammatory and autoimmune skin disorders. Certain signaling pathways and pathophysiological mechanisms, while disease-specific, share common elements across conditions such as autoimmune bullous dermatoses, where Hsp90 has also emerged as a promising target. Autoimmune bullous skin diseases (AIBDs), such as bullous pemphigoid (BP), pemphigus vulgaris (PV), and epidermolysis bullosa acquisita (EBA), represent a group of rare, chronic, and potentially life-threatening disorders characterized by autoantibody-mediated blister formation at different levels of the skin. These diseases involve autoantibodies targeting structural proteins of the skin, resulting in loss of cell adhesion and subsequent blistering. Given the pathogenic role of autoantibodies and immune cells, targeting immune mechanisms has been central to therapeutic development. Recently, the molecular chaperone heat shock protein 90 (Hsp90) has emerged as a promising target in AIBDs due to its multifaceted roles in immune regulation and inflammatory signaling (12).
Initial evidence for the involvement of Hsp90 in AIBDs was demonstrated in EBA, an AIBD characterized by autoantibodies targeting type VII collagen. The administration of two Hsp90 inhibitors, 17-DMAG and TCBL - 145, in a mouse model of experimental EBA produced significant therapeutic effects. Both inhibitors markedly improved clinical signs of disease, as evidenced by reduced skin blistering and inflammation. Additionally, treatment suppressed the production of pathogenic autoantibodies against type VII collagen and diminished dermal infiltration of neutrophils, a key driver of tissue damage in EBA. Importantly, these beneficial effects were achieved without reducing the overall numbers of plasma cells, including those specific for type VII collagen, or germinal center B cells. Instead, the inhibitors exerted their immunomodulatory effects primarily through potent inhibition of T cell proliferation, demonstrated by decreased responsiveness of lymph node T cells to restimulation in vitro with anti-CD3/CD28 antibodies or autoantigen. These results indicate that Hsp90 blockade selectively targets pathogenic T cell responses while sparing plasma cells, offering a focused approach to modulating T-cell mediated autoimmunity in EBA (20).
Extending these findings to the skin microenvironment, topical application of the Hsp90 inhibitor 17AAG significantly ameliorated disease severity in both passive transfer and active immunization mouse models of EBA. This treatment reduced neutrophilic infiltration, inhibited NF-κB activation, downregulated MMPs and Flii (an actin-remodeling protein), and induced anti-inflammatory Hsp70 expression in the skin. The topical route offers the advantage of localized inhibition, minimizing systemic toxicity while effectively modulating key inflammatory pathways involved in autoantibody-mediated skin inflammation (21).
In another study, 17-DMAG dose-dependently reduced dermal–epidermal separation ex vivo and inhibited neutrophil-derived ROS production in response to fMLP, LPS, and EBA immune complexes. Additionally, extracellular Hsp90 was found to interact with MMP - 2 and MMP - 12 in EBA patient sera, suggesting their dependence on its chaperone function (22).
In bullous pemphigoid (BP), the most common subepidermal autoimmune blistering disease, Hsp90 expression was found to be dysregulated. Although intracellular Hsp90 levels were increased in perilesional skin biopsies from BP patients, circulating serum levels of Hsp90 were decreased and inversely correlated with IgG autoantibody titers against the immunodominant NC16A domain of BP180, a hemidesmosomal protein targeted by pathogenic autoantibodies. This suggests an abnormal regulation of intracellular and extracellular Hsp90 in BP pathogenesis. Importantly, Hsp90 was induced in keratinocytes by BP patient sera and purified anti-BP180 autoantibodies but was secreted in a restricted manner, further implicating this chaperone in the local skin inflammatory milieu (23).
Mechanistically, Hsp90 modulates keratinocyte responses to autoantibodies. In vitro studies demonstrated that Hsp90 inhibition with 17-DMAG suppressed IL - 8 production by keratinocytes stimulated with BP IgG, while IL - 6 secretion remained unaffected. This effect was mediated by the inhibition of NF-κB transcriptional activity and concomitant induction of Hsp70, a known negative regulator of inflammation. Since IL - 8 plays a critical role in neutrophil recruitment and activation in BP lesions, Hsp90 blockade interferes with a key pathogenic axis, thereby reducing local inflammation and tissue damage (24).
Humoral immune responses are also modulated by Hsp90 inhibition. In vitro treatment of activated human B cells with the Hsp90 blocker 17-DMAG reduced proliferation and IgG secretion, accompanied by induction of heat shock factor 1 (HSF1) and Hsp70, which may contribute to immunoregulatory feedback. In vivo, early treatment of type VII collagen-immunized mice with 17-DMAG altered splenic B cell subsets, increasing regulatory B cell fractions and serum IL - 10 levels while reducing circulating autoantibodies and disease severity. These findings illustrate that Hsp90 inhibition impacts both cellular and humoral arms of autoimmunity, supporting its therapeutic potential in antibody-mediated skin diseases (25).
Lastly, elevated circulating autoantibodies against Hsp90 have been detected in patients with dermatitis herpetiformis (DH), a blistering cutaneous manifestation of celiac disease, but not in BP or PV patients. In DH, levels of anti-Hsp90 autoantibodies correlated positively with disease activity and decreased during remission following gluten-free diet, suggesting a disease-specific autoimmune response against Hsp90 that may contribute to pathogenesis or serve as a biomarker (26).
Collectively, these studies provide converging evidence that Hsp90 plays critical roles in the pathophysiology of autoimmune bullous skin diseases by supporting pathogenic autoantibody production, neutrophil effector functions, and keratinocyte inflammatory responses. Pharmacologic inhibition of Hsp90 modulates these processes at multiple levels, offering a promising therapeutic strategy with potential for both systemic and topical application.
The findings outlined above establish Hsp90 as a critical mediator of autoimmune inflammation in the skin. Although AIBDs and AD differ in their primary immune pathways, they share important downstream mechanisms, including T cell-driven inflammation, keratinocyte activation, neutrophil infiltration, and cytokine-driven tissue damage. These shared effector pathways are regulated in part by Hsp90, as demonstrated in both in vitro and in vivo studies. Therefore, the role of Hsp90 in AIBDs provides valuable precedent and mechanistic rationale for its exploration as a therapeutic target in AD. By demonstrating how Hsp90 modulates immune activation, tissue inflammation, and skin-specific immune responses in AIBDs, this section supports the broader concept of Hsp90 as a central regulator of cutaneous immunopathology - setting the stage for the subsequent focus on its role and inhibition in AD (Table 1).
Hsp90 as a potential therapeutic target in atopic dermatitis
Atopic dermatitis (AD) remains a challenging chronic inflammatory skin disorder characterized by a complex interplay of immune dysregulation, epidermal barrier dysfunction, and altered skin microbiome. Despite advances in understanding its pathophysiology, new therapeutic strategies are continuously sought to improve disease control and patient outcomes. Among emerging targets, heat shock protein 90 (Hsp90), a highly conserved molecular chaperone, has attracted increasing attention due to its involvement in regulating multiple signaling pathways relevant to inflammation and immune responses. Experimental data accumulated over recent years highlight the potential of Hsp90 inhibition as a novel therapeutic approach in AD, supported by in vitro, in vivo, and translational studies.
Initial explorations into the role of Hsp90 in skin inflammation and autoimmunity underscored its importance in maintaining the stability and function of numerous client proteins, including kinases and transcription factors pivotal in immune signaling cascades. AD is a chronic inflammatory skin disorder associated with IgE-mediated immune dysregulation. The first study suggesting a potential role of heat shock protein 90 (Hsp90) in the pathogenesis of AD revealed a significant elevation of serum Hsp90 levels and anti-Hsp90 IgE autoantibodies in AD patients compared to healthy controls. Notably, approximately 50% of AD patients exhibited seropositivity for anti-Hsp90 IgE, in contrast to less than 3% of healthy individuals. A positive correlation between serum Hsp90 levels and disease severity, as measured by SCORAD, was also demonstrated. These findings indicate that extracellular Hsp90 and IgE autoantibodies targeting Hsp90 may contribute to the immunopathology of AD, supporting the hypothesis of an autoimmune component in the disease (27).
Extending this concept, further studies demonstrated that circulating Hsp90α - a major extracellular isoform - was markedly elevated in AD patients when compared not only to healthy controls but also to individuals with dermatitis herpetiformis (DH) and celiac disease (CD). While serum Hsp90α levels remained comparable across DH, CD, and healthy groups, AD patients showed a distinct overexpression, suggesting disease-specific dysregulation of this chaperone. These findings indicate that extracellular Hsp90α may not only contribute to the inflammatory processes in AD but could also aid in distinguishing AD from other pruritic or autoimmune dermatoses such as DH. However, the diagnostic utility of Hsp90α remains preliminary and warrants further investigation (28).
Building on the emerging role of extracellular Hsp90 in AD, recent preclinical findings provided functional evidence that pharmacological inhibition of this chaperone may hold therapeutic promise. For the first time, the effects of the Hsp90 inhibitor STA - 9090 (Ganetespib) were evaluated in an experimental mouse model of dinitrochlorobenzene (DNCB)-induced AD. Both systemic (intraperitoneal) and topical administration of STA - 9090 significantly alleviated disease manifestations. Treated animals exhibited reduced clinical severity, diminished epidermal thickening, and lower dermal leukocyte infiltration, along with a marked decrease in scratching behavior - an indicator of pruritus. Topical treatment with STA - 9090 also resulted in reduced serum IgE concentrations and restored epidermal barrier function, as evidenced by increased filaggrin expression in lesional skin. These findings not only support the involvement of Hsp90 in the immunopathogenesis of AD but also demonstrate, for the first time, the therapeutic potential of Hsp90 blockade in vivo. Together, these data underscore the relevance of targeting Hsp90 as a novel strategy for modulating both immune dysregulation and barrier dysfunction in AD (29).
Subsequent studies have employed both cellular and murine models to further delineate the immunomodulatory effects of Hsp90 inhibition in the context of AD. Using both in vitro and in vivo systems, the immunomodulatory potential of Hsp90 inhibition has been systematically explored. In cultured primary human keratinocytes stimulated with proinflammatory cytokine combinations (TNF/IFNγ or TNF/IL-4), pharmacological blockade of Hsp90 with the small-molecule inhibitor RGRN - 305 led to a substantial downregulation of genes involved in both Th1 and Th2 immune responses, including proinflammatory cytokines (TNF, IL1B, IL6) and chemokines typically upregulated in AD (CCL17, CCL22, TSLP). In parallel, application of RGRN - 305 in a murine model of AD induced by topical MC903 not only alleviated visible skin symptoms such as erythema and edema, but also significantly reduced leukocyte infiltration within the dermis, including T cells, mast cells, and neutrophils. Notably, the therapeutic efficacy of topical RGRN - 305 was comparable to that of topical corticosteroids, such as dexamethasone, yet was associated with fewer systemic effects, including minimal weight loss. Transcriptomic profiling of inflamed skin revealed that treatment with RGRN - 305 reversed the disease-associated gene expression signature, particularly genes linked to immune activation and cytokine signaling (Il1b, Il4, Il6, Il13). Mechanistic investigations further demonstrated that Hsp90 inhibition interfered with intracellular signal transduction by reducing activation of STAT3 and STAT6 - transcription factors critically involved in cytokine-driven inflammation—thus providing a plausible molecular basis for the observed anti-inflammatory effects. Taken together, these results underscore the central involvement of Hsp90 in AD pathogenesis and support the concept that its inhibition - especially through agents like RGRN - 305 - may represent a novel therapeutic strategy capable of modulating key inflammatory and immune pathways in AD (30).
Adding a crucial piece to the puzzle, Sitko et al. (2025) provided a comprehensive analysis of the effects of topical 17-AAG, a well-characterized Hsp90 inhibitor, in the DNCB-induced murine AD model, alongside translational data from human AD patients. The study showed significant clinical improvement in treated mice, evidenced by reduced dermatitis scores. Histologically, 17-AAG treatment correlated with reduced epidermal hyperplasia and decreased expression of TSLP, IL - 5, and IL - 6 in skin biopsies. Importantly, while the number of infiltrating leukocytes - including eosinophils, mast cells, and CD4+ T cells - was not significantly altered by treatment, the activity of eosinophils and mast cells was suppressed, as indicated by significantly reduced eosinophil peroxidase (EPX) activity and lowered serum histamine levels in 17-AAG-treated AD mice. Molecular analyses demonstrated effective downregulation of proinflammatory cytokines and inhibition of NF-κB activation in the skin. In vitro, 17-AAG inhibited the proliferation and inflammatory cytokine production of activated human keratinocytes and CD4+ T lymphocytes, including suppression of IL - 5, IL - 17A, and IL - 22, characteristic of Th2, Th17, and Th22 responses. Furthermore, 17-AAG dose-dependently reduced reactive oxygen species (ROS) production in activated human eosinophils, indicating decreased eosinophil activation status. Microbiome analysis by next-generation sequencing revealed that topical 17-AAG partially restored the gut and skin microbial balance disrupted in AD mice. Although the overall abundance of Staphylococcus aureus on the skin was not significantly changed in vivo, 17-AAG potently inhibited S. aureus biofilm formation in vitro in a dose-dependent manner, suggesting an additional mechanism contributing to the clinical benefit. Importantly, analysis of tape-strip samples from human AD patients revealed no significant differences in the gene expression of four HSP90 homologs (HSP90AA, HSP90AB, Grp94/gp96, and TRAP1) between lesional and non-lesional skin. Similarly, total HSP90 protein levels in polymorphonuclear leukocytes (PMNs) and peripheral blood mononuclear cells (PBMCs) did not differ significantly between AD patients and healthy individuals. However, Sitko et al. observed a significantly lower level of acetylated HSP90 (acetyl-HSP90) in PBMCs from AD patients compared to controls, while treatment of human keratinocytes (HaCaT cells) with 17-AAG led to increased acetylation of HSP90. Additionally, elevated eosinophil peroxidase (EPX) activity was detected in leukocytes from AD patients, consistent with increased eosinophil activation in the disease. These findings highlight that the pathological role of Hsp90 in AD is not due to its expression levels but rather its functional state and post-translational modifications such as acetylation. Together, the data support the potential of topical Hsp90 inhibitors like 17-AAG to modulate immune activation, restore barrier function, and normalize the microbiome in AD, offering a promising therapeutic avenue (31).
These translational findings align with mechanistic insights into the role of Hsp90 in contact hypersensitivity, a model immunologically related to AD. Kim et al. (2022) demonstrated that both pharmacological inhibition and shRNA-mediated knockdown of Hsp90 markedly suppressed DNCB-induced immune activation in vitro. Moreover, genetic deletion of Hsp90 significantly attenuated contact hypersensitivity responses in mice. Additional experiments suggested that DNCB-modified Hsp90 activates immune responses via the CD91 receptor, as CD91 blockade in cell lines and in vivo led to comparable reductions in hypersensitivity (32).
Together, these studies underscore Hsp90 as a nodal regulator of key pathological processes in AD, including keratinocyte activation, T cell differentiation, cytokine production, epidermal barrier integrity, and skin microbiome composition. The consistent therapeutic benefits observed with diverse Hsp90 inhibitors, such as Geldanamycin, Ganetespib, 17-AAG, and RGRN - 305, administered systemically or topically, provide strong rationale for further clinical evaluation in patients with AD (Table 1). The proposed pathophysiological mechanisms and therapeutic targets in preclinical models of AD modulated by Hsp90 inhibition are also presented in Figure 1.
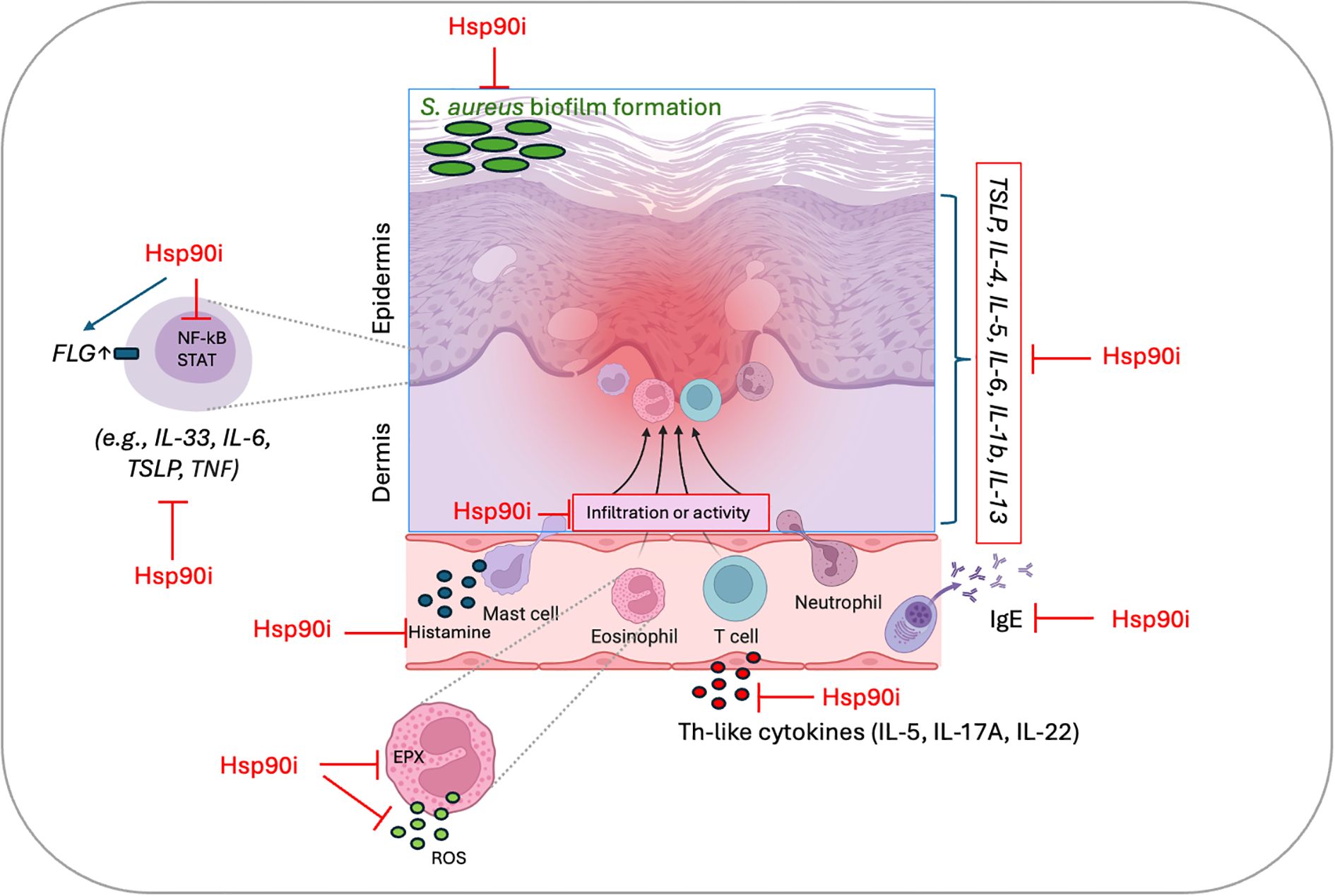
Figure 1. Proposed pathophysiological mechanisms and therapeutic targets in preclinical models of atopic dermatitis (AD) modulated by Hsp90 inhibition. Hsp90 inhibition affects multiple pathways, including suppression of proinflammatory cytokine and IgE production, inhibition of histamine release, attenuation of NF-κB/STAT signaling and EPX activity, reduction of ROS generation, inhibition of dermal leukocyte infiltration, and restoration of epidermal barrier integrity through FLG upregulation. The diagram summarizes current evidence described in references (29–32). Hsp90i, heat shock protein 90 inhibitor; EPX, eosinophil peroxidase; ROS, reactive oxygen species; FLG, filaggrin; NF-κB, nuclear factor kappa-light-chain-enhancer of activated B cells; STATs, signal transducer and activator of transcription proteins; TSLP, thymic stromal lymphopoietin; IL, interleukin; Th, T helper cells; IgE, immunoglobulin E.
Notably, while extracellular Hsp90α is significantly elevated in the serum of patients with AD (27), corresponding increases in total Hsp90 protein within lesional skin are not consistently observed (31). This suggests compartment-specific regulation of Hsp90 in AD. Extracellular Hsp90 may be actively secreted or released into circulation in response to systemic inflammation or skin barrier impairment, where it can function as a danger-associated molecular pattern (DAMP) and modulate immune responses. In contrast, intracellular Hsp90 expression in keratinocytes and other skin-resident cells appears to remain relatively stable, possibly due to homeostatic regulation. Moreover, functional modifications of Hsp90, such as acetylation, rather than changes in overall protein abundance, may critically influence its activity in AD pathogenesis. These post-translational modifications can alter Hsp90’s chaperone function and interactions with client proteins, thereby affecting inflammatory signaling without necessitating altered expression levels in the skin. This compartmentalized and functional regulation of Hsp90 underscores the complexity of its role in AD and supports the rationale for targeted therapeutic modulation (31).
In conclusion, the accumulated evidence supports Hsp90 as a promising therapeutic target in AD. Its inhibition exerts multi-level effects on inflammation, epidermal barrier function, and skin microbiota, all critical components of AD pathogenesis. Notably, recent findings indicate that the four Hsp90 isoforms - Hsp90α, Hsp90β, GRP94, and TRAP1- may exert distinct and even opposing effects on inflammatory pathways in the skin. Selective inhibition of TRAP1 has been shown to consistently suppress proinflammatory cytokine expression in keratinocytes, fibroblasts, and lesional tissue from patients with hidradenitis suppurativa (33). Although such data are not yet available in the context of atopic dermatitis, this raises the possibility that isoform-specific targeting – particularly of TRAP1 – could offer a more refined therapeutic approach. Future studies will be required to determine whether similar mechanisms operate in AD, and whether TRAP1 inhibition might prove beneficial in this specific disease context.
Perspective
Recent clinical investigations of the oral Hsp90 inhibitor RGRN - 305 in plaque-type psoriasis and hidradenitis suppurativa (HS) have provided promising evidence supporting the therapeutic relevance of Hsp90 inhibition in chronic inflammatory skin disorders (11, 34). In an open-label proof-of-concept trial, treatment with RGRN - 305 led to marked clinical improvement in a subset of patients with moderate-to-severe psoriasis, accompanied by molecular and histological normalization of key inflammatory pathways, including TNF-α, IL - 23, and IL - 17A signaling (11). Similarly, in a double-blind randomized trial in HS, RGRN - 305 demonstrated clinically meaningful efficacy and a favorable safety profile, with over half of treated patients achieving HiSCR-50 responses and no serious adverse events reported (34). These findings highlight the potential of Hsp90 as a viable therapeutic target beyond oncological indications (Table 1).
Although Hsp90 inhibitors have not yet been clinically evaluated in patients with AD, existing data suggest a manageable safety profile that may translate to this population. In the psoriasis study, four of seven patients receiving 500 mg/day of RGRN - 305 developed a mild-to-moderate exanthematous drug-induced eruption, leading two to discontinue treatment (11). Importantly, no severe adverse events were reported, and treatment-emergent events were comparable in frequency between RGRN - 305 and placebo groups in the HS trial (34). While systemic administration at higher doses may pose tolerability challenges, these findings underscore the importance of optimizing dosing strategies. For AD, where barrier function is compromised and long-term safety is paramount, lower systemic doses or localized (e.g., topical) delivery of Hsp90 inhibitors could offer a more targeted and better-tolerated approach. Future studies in AD populations will be critical to determine the risk–benefit profile in this context.
Given the mechanistic overlap in cytokine signaling and barrier dysfunction across psoriasis, HS, and AD, Hsp90 inhibition may hold promise in AD, where conventional therapies often fall short in patients with severe, chronic, or treatment-resistant disease. Notably, preclinical data - including those discussed in this mini-review - indicate that Hsp90 contributes to AD pathophysiology at multiple levels: from modulating T cell differentiation and cytokine production to regulating keratinocyte stress responses and skin–microbiota interactions. In parallel, recent studies have shed light on the role of Hsp90 in AIBDs, such as epidermolysis bullosa acquisita and bullous pemphigoid (12). Collectively, these studies provide converging evidence that Hsp90 plays critical roles in AIBD pathogenesis by supporting pathogenic autoantibody production, neutrophil effector functions, and keratinocyte inflammatory responses. Pharmacological inhibition of Hsp90 has been shown to modulate disease-driving mechanisms at multiple levels, offering a promising therapeutic strategy with potential for both systemic and topical application. The molecular chaperone Hsp90 has thus emerged as a key regulator of inflammatory and autoimmune mechanisms in dermatology. Taken together, the emerging clinical efficacy of Hsp90 inhibitors in related dermatoses, coupled with robust preclinical support in AD and AIBD models, strongly supports the initiation of early-phase clinical trials in patients with AD. Such studies may pave the way for a new class of host-directed, immunomodulatory therapies targeting Hsp90 in inflammatory skin disease.
Author contributions
ST: Writing – review & editing, Writing – original draft, Funding acquisition.
Funding
The author(s) declare that financial support was received for the research and/or publication of this article. This study was supported by the Polish National Science Centre (NCN), grant no. 2020/39/B/NZ6/00357.
Conflict of interest
The author declares that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declared that they were an editorial board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Bieber T, Paller AS, Kabashima K, Feely M, Rueda MJ, Ross Terres JA, et al. Atopic dermatitis: pathomechanisms and lessons learned from novel systemic therapeutic options. J Eur Acad Dermatol Venereol. (2022) 36:1432–49. doi: 10.1111/jdv.18225
2. Wang SP, Stefanovic N, Orfali RL, Aoki V, Brown SJ, Dhar S, et al. Impact of climate change on atopic dermatitis: A review by the International Eczema Council. Allergy. (2024) 79:1455–69. doi: 10.1111/all.16007
3. Brunner PM, Guttman-Yassky E, and Leung DYM. The immunology of atopic dermatitis and its reversibility with broad-spectrum and targeted therapies. J Allergy Clin Immunol. (2017) 139:S65–76. doi: 10.1016/j.jaci.2017.01.011
4. Guttman-Yassky E, Renert-Yuval Y, and Brunner PM. Atopic dermatitis. Lancet. (2025) 405:583–96. doi: 10.1016/S0140-6736(24)02519-4
5. Simpson EL, De Benedetto A, Boguniewicz M, Ong PY, Lussier S, Villarreal M, et al. Phenotypic and endotypic determinants of atopic dermatitis severity from the atopic dermatitis research network (ADRN) registry. J Allergy Clin Immunol Pract. (2023) 11:2504–15. doi: 10.1016/j.jaip.2023.04.052
6. He H, Suryawanshi H, Morozov P, Gay-Mimbrera J, Del Duca E, Kim HJ, et al. Single-cell transcriptome analysis of human skin identifies novel fibroblast subpopulation and enrichment of immune subsets in atopic dermatitis. J Allergy Clin Immunol. (2020) 145:1615–28. doi: 10.1016/j.jaci.2020.01.042
7. Okamoto H, Li S, and Nakamura Y. The role of skin dysbiosis and quorum sensing in atopic dermatitis. JID Innov. (2025) 5:100377. doi: 10.1016/j.xjidi.2025.100377
8. Lee E, Yang SI, Suh DI, Kim HB, Lee SY, Kwon SO, et al. Environmental factors shaping atopic dermatitis: Lessons from longitudinal cohort studies. Pediatr Allergy Immunol. (2025) 36:e70130. doi: 10.1111/pai.70130
9. D’Auria E, Indolfi C, Acunzo M, Dinardo G, Comberiati P, Peroni D, et al. Biologics and small molecules: the re-evolution in the treatment of atopic dermatitis in children and adolescents. Current state of the art and future perspectives. Expert Rev Clin Immunol. (2025) 21:493–505. doi: 10.1080/1744666X.2025.2452247
10. Ständer S, Yosipovitch G, Simpson EL, Kim BS, Kabashima K, Thaçi D, et al. Onset and long-term maintenance of optimal itch response in adult patients with moderate-to-severe atopic dermatitis treated with dupilumab: post hoc analysis from two phase 3 trials. Adv Ther. (2025) 42:1800–10. doi: 10.1007/s12325-025-03124-8
11. Bregnhøj A, Thuesen KKH, Emmanuel T, Litman T, Grek CL, Ghatnekar GS, et al. HSP90 inhibitor RGRN - 305 for oral treatment of plaque-type psoriasis: efficacy, safety and biomarker results in an open-label proof-of-concept study. Br J Dermatol. (2022) 186:861–74. doi: 10.1111/bjd.20880
12. Tukaj S and Sitko K. Heat shock protein 90 (Hsp90) and hsp70 as potential therapeutic targets in autoimmune skin diseases. Biomolecules. (2022) 12:1153. doi: 10.3390/biom12081153
13. Schopf FH, Biebl MM, and Buchner J. The HSP90 chaperone machinery. Nat Rev Mol Cell Biol. (2017) 18:345–60. doi: 10.1038/nrm.2017.20
14. Albakova Z. HSP90 multi-functionality in cancer. Front Immunol. (2024) 15:1436973. doi: 10.3389/fimmu.2024.1436973
15. Backe SJ, Sager RA, Woodford MR, Makedon AM, and Mollapour M. Post-translational modifications of Hsp90 and translating the chaperone code. J Biol Chem. (2020) 295:11099–117. doi: 10.1074/jbc.REV120.011833
16. Stetz G, Tse A, and Verkhivker GM. Dissecting structure-encoded determinants of allosteric cross-talk between post-translational modification sites in the hsp90 chaperones. Sci Rep. (2018) 8:6899. doi: 10.1038/s41598-018-25329-4
17. Li ZN and Luo Y. HSP90 inhibitors and cancer: Prospects for use in targeted therapies (Review). Oncol Rep. (2023) 49:6. doi: 10.3892/or.2022.8443
18. Li W, Li Y, Guan S, Fan J, Cheng CF, Bright AM, et al. Extracellular heat shock protein-90alpha: linking hypoxia to skin cell motility and wound healing. EMBO J. (2007) 26:1221–33. doi: 10.1038/sj.emboj.7601579
19. Eustace BK, Sakurai T, Stewart JK, Yimlamai D, Unger C, Zehetmeier C, et al. Functional proteomic screens reveal an essential extracellular role for hsp90 alpha in cancer cell invasiveness. Nat Cell Biol. (2004) 6:507–14. doi: 10.1038/ncb1131
20. Kasperkiewicz M, Müller R, Manz R, Magens M, Hammers CM, Somlai C, et al. Heat-shock protein 90 inhibition in autoimmunity to type VII collagen: evidence that nonmalignant plasma cells are not therapeutic targets. Blood. (2011) 117:6135–42. doi: 10.1182/blood-2010-10-314609
21. Tukaj S, Bieber K, Kleszczyński K, Witte M, Cames R, Kalies K, et al. Topically applied hsp90 blocker 17AAG inhibits autoantibody-mediated blister-inducing cutaneous inflammation. J Invest Dermatol. (2017) 137:341–9. doi: 10.1016/j.jid.2016.08.032
22. Tukaj S, Hellberg L, Ueck C, Hänsel M, Samavedam U, Zillikens D, et al. Heat shock protein 90 is required for ex vivo neutrophil-driven autoantibody-induced tissue damage in experimental epidermolysis bullosa acquisita. Exp Dermatol. (2015) 24:471–3. doi: 10.1111/exd.12680
23. Tukaj S, Kleszczyński K, Vafia K, Groth S, Meyersburg D, Trzonkowski P, et al. Aberrant expression and secretion of heat shock protein 90 in patients with bullous pemphigoid. PloS One. (2013) 8:e70496. doi: 10.1371/journal.pone.0070496
24. Tukaj S, Grüner D, Zillikens D, and Kasperkiewicz M. Hsp90 blockade modulates bullous pemphigoid IgG-induced IL - 8 production by keratinocytes. Cell Stress Chaperones. (2014) 19:887–94. doi: 10.1007/s12192-014-0513-8
25. Tukaj S, Tiburzy B, Manz R, de Castro Marques A, Orosz A, Ludwig RJ, et al. Immunomodulatory effects of heat shock protein 90 inhibition on humoral immune responses. Exp Dermatol. (2014) 23:585–90. doi: 10.1111/exd.12476
26. Kasperkiewicz M, Tukaj S, Gembicki AJ, Silló P, Görög A, Zillikens D, et al. Evidence for a role of autoantibodies to heat shock protein 60, 70, and 90 in patients with dermatitis herpetiformis. Cell Stress Chaperones. (2014) 19:837–43. doi: 10.1007/s12192-014-0505-8
27. Sitko K, Bednarek M, Mantej J, Trzeciak M, and Tukaj S. Circulating heat shock protein 90 (Hsp90) and autoantibodies to Hsp90 are increased in patients with atopic dermatitis. Cell Stress Chaperones. (2021) 26:1001–7. doi: 10.1007/s12192-021-01238-w
28. Sitko K, Kárpáti S, Węgrzyn G, Mincewicz G, Trzeciak M, Kasperkiewicz M, et al. Differences in the detection of circulating Hsp90 alpha between patients with atopic dermatitis and dermatitis herpetiformis. Front Med (Lausanne). (2024) 10:1327144. doi: 10.3389/fmed.2023.1327144
29. Sitko K, Starke M, and Tukaj S. Heat shock protein 90 (Hsp90) inhibitor STA - 9090 (Ganetespib) ameliorates inflammation in a mouse model of atopic dermatitis. Cell Stress Chaperones. (2023) 28:935–42. doi: 10.1007/s12192-023-01387-0
30. Ben Abdallah H, Bregnhøj A, Ghatnekar G, Iversen L, and Johansen C. Heat shock protein 90 inhibition attenuates inflammation in models of atopic dermatitis: a novel mechanism of action. Front Immunol. (2024) 14:1289788. doi: 10.3389/fimmu.2023.1289788
31. Sitko K, Piotrowska E, Podlacha M, Zagórska N, Starke MD, Trzeciak M, et al. Topical application of the HSP90 inhibitor 17-AAG reduces skin inflammation and partially restores microbial balance: implications for atopic dermatitis therapy. Sci Rep. (2025) 15:21245. doi: 10.1038/s41598-025-05307-3
32. Kim SM, Studnitzer B, and Esser-Kahn A. Heat shock protein 90’s mechanistic role in contact hypersensitivity. J Immunol. (2022) 208:2622–31. doi: 10.4049/jimmunol.2101023
33. Ben Abdallah H, Iversen L, and Johansen C. The differential roles of HSP90 isoforms in skin inflammation: anti-inflammatory potential of TRAP1 inhibition. J Invest Dermatol. (2025) 145:2281–92.e3. doi: 10.1016/j.jid.2025.02.006.
Keywords: molecular chaperones, inflammation, NF-κB, STAT, eosinophils, skin barrier, microbiome, Staphylococcus aureus
Citation: Tukaj S (2025) Hsp90 as a pathophysiological factor and emerging therapeutic target in atopic dermatitis. Front. Immunol. 16:1658399. doi: 10.3389/fimmu.2025.1658399
Received: 02 July 2025; Accepted: 18 August 2025;
Published: 28 August 2025.
Edited by:
Abhinav Vasudevan, Eastern Health, AustraliaReviewed by:
William D. Shipman, Skin & Beauty Center- Board Certified Dermatologist, United StatesZarema Albakova, Lomonosov Moscow State University, Russia
Copyright © 2025 Tukaj. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Stefan Tukaj, c3RlZmFuLnR1a2FqQHVnLmVkdS5wbA==
 Stefan Tukaj
Stefan Tukaj