- 1Centre for Neglected Tropical Diseases, Department of Tropical Disease Biology, Liverpool School of Tropical Medicine, Liverpool, United Kingdom
- 2Filariasis Elimination, STH Control and Little Doctor Programme, Ministry of Health and Family Welfare, Dhaka, Bangladesh
- 3Centre for Injury Prevention and Research Bangladesh, Dhaka, Bangladesh
- 4National Podoconiosis Action Network, Addis Ababa, Ethiopia
- 5Diseases Prevention and Control Directorate, Federal Ministry of Health, Addis Ababa, Ethiopia
Lymphedema is a life-long sequelae to several neglected tropical diseases (NTD). In Bangladesh the main cause is lymphatic filariasis (LF) and Ethiopia is endemic for both LF and podoconiosis. The World Health Organization (WHO) recommends daily self-care including meticulous washing and drying of affected skin and attention to entry lesions, limb exercises and elevation. Adherence to this regime reduces secondary infections which cause disabling episodes of acute dermato-lymphangitis (ADL). Self-care practices must be integrated into family life, supported by community and monitored by health staff; however, little is known about the influence of personal and socio-demographic factors on adherence. People affected by lymphedema (n=272), adult caregivers (n=272), and health workers (n=68) in Bangladesh and Ethiopia were trained in lymphedema management according to WHO recommendations. Surveys on the causes and management of lymphedema were collected at baseline and 24-weeks, and patients completed a daily journal of self-care activities and symptoms. At baseline knowledge on causes and management of lymphedema was greater among health workers (>70%) than patients and caregivers (<20%) in both countries, and there were significant between-country differences in patient reported use of limb washing (Bangladesh = 7.7%. Ethiopia = 51.1%, p = 0.001). At 24-weeks knowledge on lymphedema causes and management had increased significantly among patients and caregivers, there was <70% adherence to limb washing and exercises, but lesser use of limb elevation in both countries. A range of patient characteristics were associated with significant variation in self-care, except for limb washing. Performance of fewer leg exercises was significantly associated with increased age or severe lymphedema in Bangladesh, and with being female or in paid work in Ethiopia. Patient journals recorded ADL symptoms and working days lost due to lymphedema more frequently than were reported by recall during the 24-week survey. Core elements of lymphedema self-care education, training and monitoring are the same for multiple etiologies. This creates opportunities for cross-cutting implementation of integrated service delivery across several skin NTDs. Sustainability will depend on community level ownership and research on factors affecting adherence to lymphedema self-care are urgently needed.
1 Introduction
The World Health Organization (WHO) Roadmap to End Neglected Tropical Diseases (NTD) includes the elimination of lymphatic filariasis (LF) as a public health problem in at least 58 of the 72 LF-endemic countries by 2030. Elimination of LF as a public health problem is defined as 1) an infection rate sustained below transmission thresholds, and 2) the availability of an essential package of care for people with existing clinical disease (1). The clinical manifestations of LF are hydrocele and lymphedema, and these conditions are exacerbated by secondary infections causing acute dermato-lymphangitis (ADL). Disability from LF affects 42 million people resulting in an estimated loss of 5.25 million disability adjusted life years (DALY) (2). Hydrocelectomy surgery offers a permanent cure for people affected by hydrocele, but lymphedema cannot be cured and treatment entails lifelong self-management by affected persons with the support of their family and broader community (3). The WHO recommends similar self-management interventions for people affected by podoconiosis (4), which causes lower limb lymphedema after bare-foot exposure to irritant soils (5). Together LF- and podoconiosis-related lymphedema cause pain, disfigurement, disability, and social exclusion for 20 million people globally (6, 7).
The extreme suffering of people affected by hydrocele and lymphedema was the impetus for establishing the Global Program to Eliminate LF (GPELF), which sets out validation targets for the implementation of 1) mass drug administration (MDA) of preventive chemotherapy, and 2) morbidity management and disability prevention (MMDP) services for people with existing disease (8). Since the inception of the GPELF in 2000, targets for MMDP have lagged behind MDA (9) and by 2019, 62 endemic countries reported that MDA had been scaled up to all endemic areas or completed, but only 41 countries reported availability of the essential package of care in any endemic areas (10). The Roadmap targets a 75% reduction in DALYs by 2030 (1) and a significant acceleration of interventions to reduce and prevent morbidity and disability will be needed in order to achieve this.
A distinct feature of the Roadmap is greater ownership by national and local governments, including communities. Achieving this in LF- and podoconiosis-endemic areas will depend on educating all affected persons in evidence-based and effective lymphedema self-care, and on providing support to perform the recommended self-care activities. Currently, WHO recommendations include education on the causes of lymphedema, a daily regime of washing and drying the affected body parts, meticulous skin care to prevent secondary infections (ADL), and elevation and exercises to promote lymphatic circulation (8). There have been multiple studies and two systematic reviews which show the benefits of these core self-care practices for the individual (11, 12), and the economic gains to the whole community (13). Implementation of lymphedema-management at this level is only sustainable if it addresses any challenges and barriers to performing the daily self-care regime, and these may be specific to country, region or individual. Here, we report on the results of patient, caregiver and local health worker questionnaires, and patient journals collected during a study on an enhanced self-care protocol (14) for people affected by moderate and severe lymphedema in Bangladesh (15) and Ethiopia (16). Insights from these diverse populations may be transferable to other LF- and podoconiosis-endemic counties and contribute to achieving the overarching goal to leave no-one behind.
2 Materials and Methods
2.1 Study Design and Research Personnel
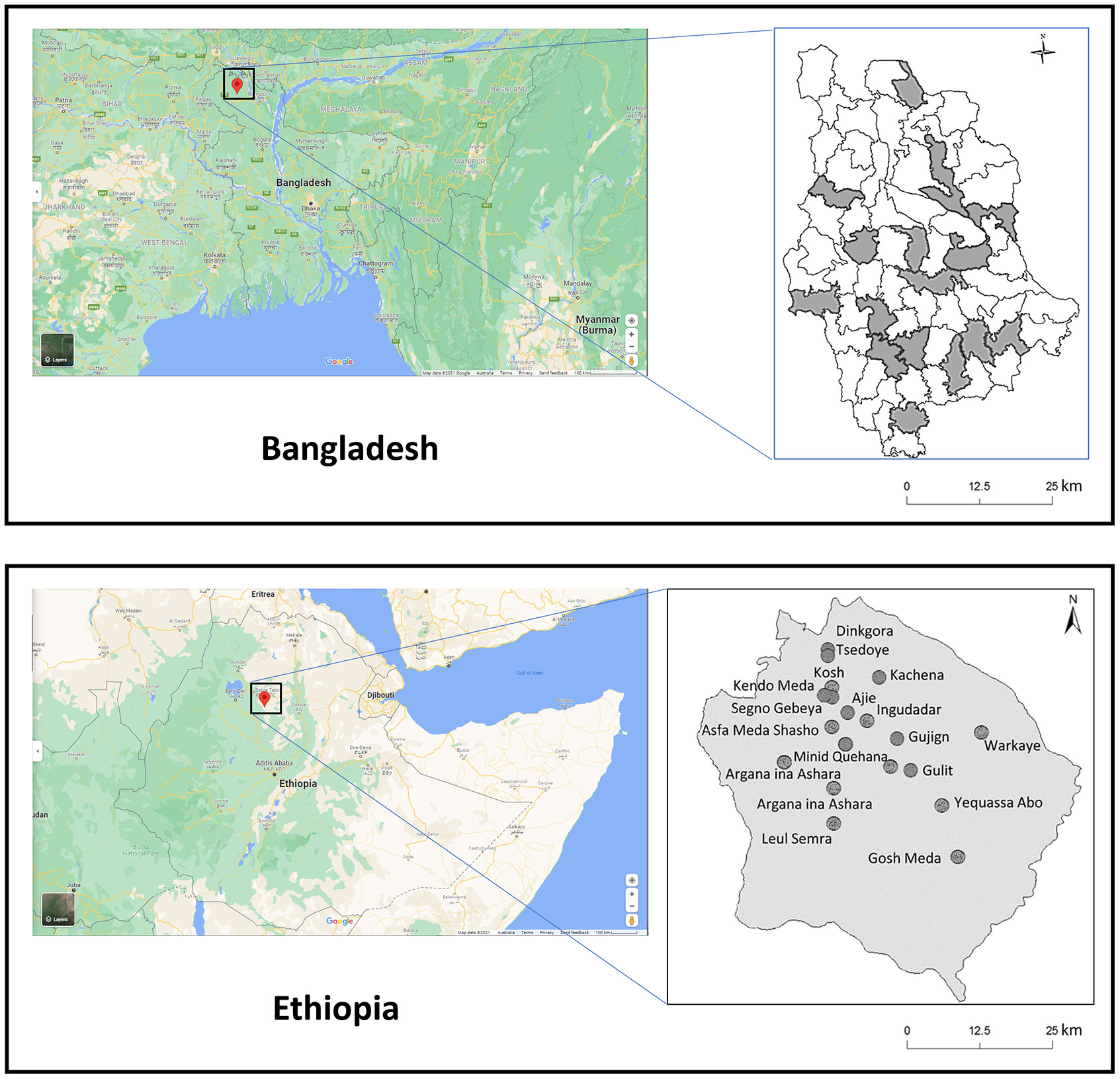
A multicenter cluster randomized, controlled trial (RCT) on an enhanced lymphedema self-care protocol was conducted in Bangladesh and Ethiopia over 24-weeks during late 2018 and early 2019. The enhanced self-care protocol, participant recruitment and selection, and data collected methods have been reported in detail (14–16). Briefly, both countries have a high prevalence of LF, and podoconiosis in Ethiopia (17, 18). Nilphamari District in north-western Bangladesh and Simada District in north-western Ethiopia were chosen based on the absence of any planned MMDP training programs during the study period. The chosen locations also have distinctly different lived environments, Nilphimari is less than 50 m above sea-level and has a tropical climate and Simada has an elevation around 2000m and a cooler and dryer climate. Figure 1 shows the location of study sites in each country.
Patients were included if they had moderate or severe lymphedema in at least one leg, and if they had an adult caregiver who was available to participate in the study and provide support as needed. Caregivers and two community level health workers at each of the included health centers were also enrolled into the study. Data collection and outcome measures on lymphedema status have been published for each country (15, 16). Clinical personnel in the research teams assessed lymphedema status and conducted the participant surveys. All participants were provided with a written information sheet in the local language and the information was also explained to them by native speakers. A maximum of ten patients were enrolled into each study center to avoid over-loading the health workers, but all volunteers who were not enrolled into the study were invited to participate in the lymphedema self-care training. The study was conducted in accordance with the Declaration of Helsinki (19), and approved by the Bangladesh Medical Research Council, and the Amhara Public Health Institute Research Ethics Review Committee, and the Liverpool School of Tropical Medicine Research Ethics Committee (approval no. 012-18), and registered on the ISRCTN, trial number 16,764,792 (20). All participants gave their informed consent for inclusion before they participated in the study.
2.2 Data Collection and Outcome Measures
Questionnaires were used to collect demographic information (age and sex) and knowledge on lymphedema causes and management from all participants (patients, caregivers, health workers). Patients and caregivers were asked about their marital status, number of dependent children living at home, literacy, source of income and how household water was sourced. Patients were also asked about their medical status and lymphedema history, frequency and duration of ADL, days of work lost due to lymphedema, and use of any lymphedema self-care practices. Caregivers were asked how long they had been providing support for someone affected by lymphedema. Since the majority of patients were affected by LF-related lymphedema, the presenting features on each leg were assessed according to the Dreyer 7-stage system (21). Dreyer stages 3 and 4 were considered moderate and stages 5 – 7 as severe lymphedema. The presence or absence and type of any footwear was recorded. Patients were also asked the source of washing and drinking water, the distance travelled to collect household water, and the sex of the person who usually collected the water. Questionnaires were administered at baseline with follow-up questionnaires administered at 4-, 12-, and 24-weeks. Patient, caregiver and health worker responses were entered to the Open Data Kit Collect (ODK Collect) application (22) which had been loaded onto an electronic tablet (Samsung Galaxy Tab A 10.1). Patients recorded their lymphedema self-care activities in a daily journal and also noted the presence of any acute symptoms, days they were unable to work due to lymphedema, and the amount of caregiver time required.
2.3 Lymphedema Self-Care Intervention
A full description of the intervention has been published (14). Briefly, all participants (patients, caregivers, health workers) were trained in lymphedema self-care as recommended by the WHO (3) and in accordance with the LF MMDP Program implementation policy in each country. This included education on the causes of lymphedema in their region, (LF in Bangladesh and LF and podoconiosis in Ethiopia) and on management of ADL episodes. The need for daily hygiene and meticulous skin care were explained, and patients were shown how to thoroughly wash and dry their legs and feet, to look for and apply medicated creams to entry lesions and interdigital lesions. Lower leg exercises performed while seated and standing (5 repetitions of each exercise = 1 event) were demonstrated and practiced, and leg elevation was recommended overnight while sleeping, and during the day when possible. Caregivers and health workers were included in the training and patients were advised to access support and additional medication from the health center if needed.
Patients were provided with a bowl, clean towels, and enough soap and topical creams to complete the study, a paper journal for each 4-week period, and a pencil (14). Patients and caregivers were instructed in how to enter tick marks to a weekly activities chart in the journal and given time to practice during the training session. Self-care activities and the amount of caregiver time needed were entered daily. Patients also recorded how much they walked (15-minutes = 1 event), their water intake (glasses per day), fresh fruit and vegetable consumption (days per week), any acute symptoms present, and if they were unable to work due to lymphedema. At each follow-up, the completed journal(s) were collected and checked, participants were reminded of the daily self-care activities, and enough journals for the next period were provided.
2.4 Analysis
Chi Squared tests were used for between country comparison on participant characteristics (patients, caregivers, health workers). Survey data on knowledge of the causes and management of lymphedema, and self-reported lymphedema practices at 24-weeks were given as a proportion of participants within each country. If during the study any caregiver was replaced with a new caregiver, the new caregiver data were not included in before-and-after comparison on lymphedema knowledge. For the patient journals, a weekly score for the average number of events per activity were calculated from the number of tick marks entered to the chart in first week of each 4-week journal. A random number generator was used to select 30% of patients in each country and data from all available journal pages was extracted on the presence of any acute symptoms, and days of work lost due to lymphedema. Student’s T-tests were used for between-country comparison of journal scores at baseline. Linear regression was used to identify patient characteristics associated with variance in weekly journal scores at week-21 (the first page of the final journal).
3 Results
Patients in both countries experienced significant improvement in objective measures of lymphedema status and these results have been published for each country (15, 16).
3.1 Participants and Water Sources
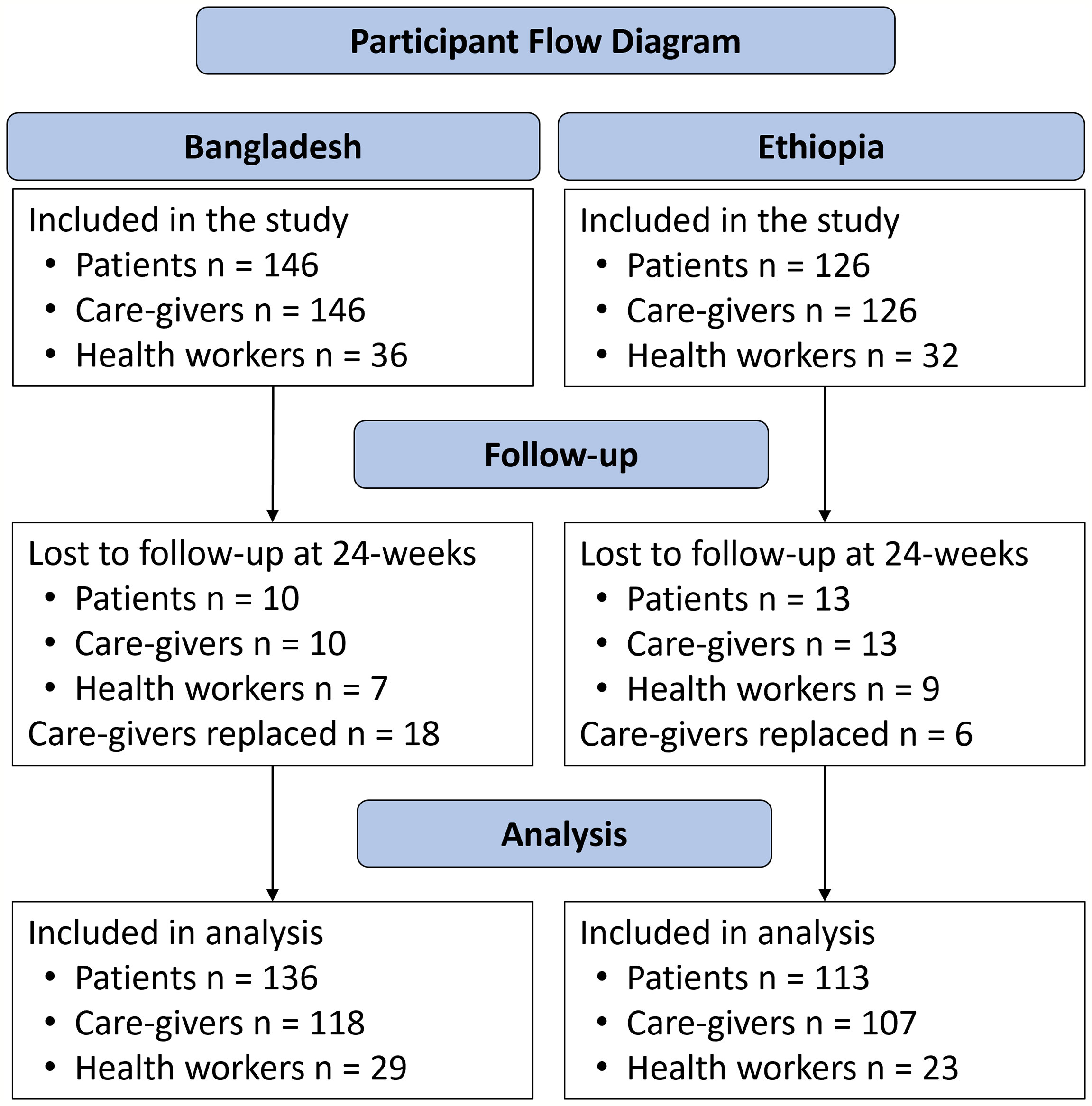
A total of 272 people affected by moderate and severe lower limb lymphedema, 272 designated caregivers, and 68 community health workers were enrolled in the study. In Bangladesh, 142 patients, 142 caregivers, and 36 health workers were enrolled, and during the course of the study 18 caregivers were replaced with a new caregiver. In Ethiopia, 126 patients, 126 caregivers, and 32 health workers were enrolled, and during the course of the study 6 caregivers were replaced with a new caregiver. The flow of all surveyed participants is shown in Figure 2.
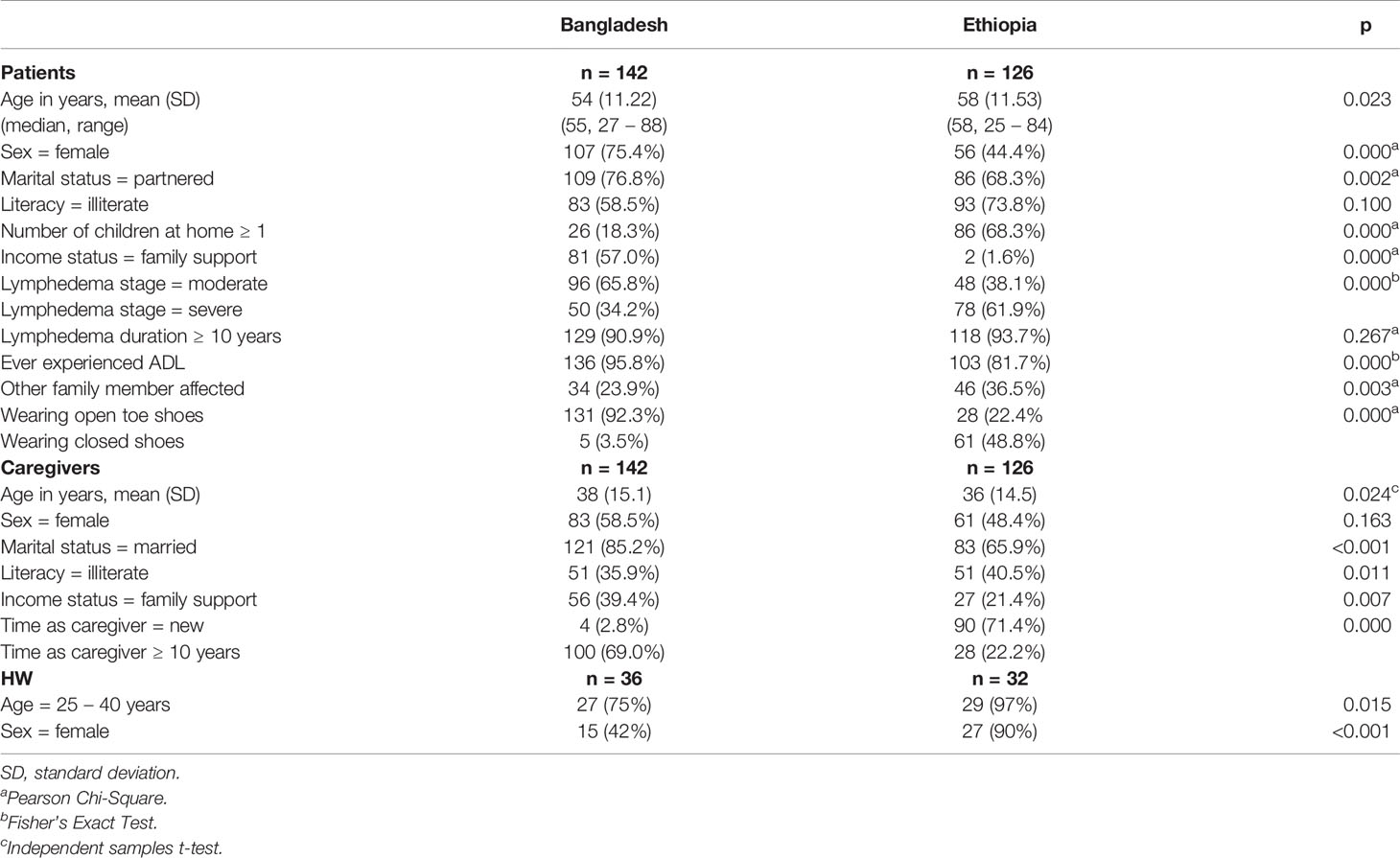
There were significant between-country differences in participant characteristics as shown in Table 1. Mean patient ages were 54 ± 11.22 years in Bangladesh (median = 55, range 27 – 88) and 58 ± 11.53 years in Ethiopia (range 25 – 84, p = 0.023). There were more female patients in Bangladesh (n = 107; 75.4%) than in Ethiopia (n = 56; 44.4%, p = 0.000). More than 90% of patients in both countries had experienced lymphedema for 10 years or more (Bangladesh n = 129; 90.9%, Ethiopia n = 118; 93.7%, p = 0.267) but more patients in Ethiopia were affected by severe lymphedema (n = 78; 61.9%) than in Bangladesh (n = 50; 34.2% p = 0.000). Mean caregiver ages were 38 ± 15.1 years in Bangladesh, and 36 ± 14.5 years in Ethiopia (p = 0.024). Most caregivers in Bangladesh had provided assistance to someone with lymphedema prior to the study (n = 142; 97.2%), and for 10 years or more (n = 100; 69.0%) whereas most caregivers in Ethiopia were new (n = 90; 71.4%, p = 0.000) and fewer had provided assistance for 10 years or more (n = 28; 22.2%). Most health workers were aged 25 – 40 with more female health workers in Ethiopia (n = 27; 90%) than in Bangladesh (n = 15; 42%, <0.001).
In Bangladesh 92.3% of patients sourced water for washing from a tube well, and 83.3% had to walk less than 10 minutes to collect household water. In Ethiopia 64.3% of patients sourced water from a public tap and 15.9% from a protected well. Most had to walk up to 30 minutes to collect water (52.4%) and 19% had to walk 60 minutes or more. Water was collected by both sexes in Bangladesh (female = 75; 51.4%) but few adult men collected water in Ethiopia (n = 4; 3.2%).
3.2 Knowledge on Causes of Lymphedema
Knowledge on the causes of lymphedema increased in every participant group in both countries over the study period (Table 2). At baseline, 16.2% of patients in Bangladesh knew that mosquitos, or worms in mosquitos, were a cause of lymphedema, while 17.5% of patients in Ethiopia knew that not wearing shoes was a cause of podoconiosis. Erroneous causes of lymphedema were given in both countries (Bangladesh; bad water n = 5, bad luck n = 1, Ethiopia; bad water n = 29, witchcraft n = 15, punishment from god n = 13, bad luck n = 11). Caregivers had similar knowledge to patients at baseline. In Bangladesh 25 (17.1%) caregivers knew that mosquitos, or worms in mosquitos, were a cause of lymphedema while 29 (23.0%) caregivers in Ethiopia knew that not wearing shoes was a cause of lymphedema. At 24-weeks no patient or caregiver in either country gave any superstitious or erroneous cause of lymphedema. In both countries, health workers knew at least one correct cause of lymphedema at baseline (Bangladesh n = 33; 91.7%, Ethiopia n = 29; 90.6%) and this was increased to 100% in both countries at 24-weeks.
3.3 Knowledge on Lymphedema Management and Prevention
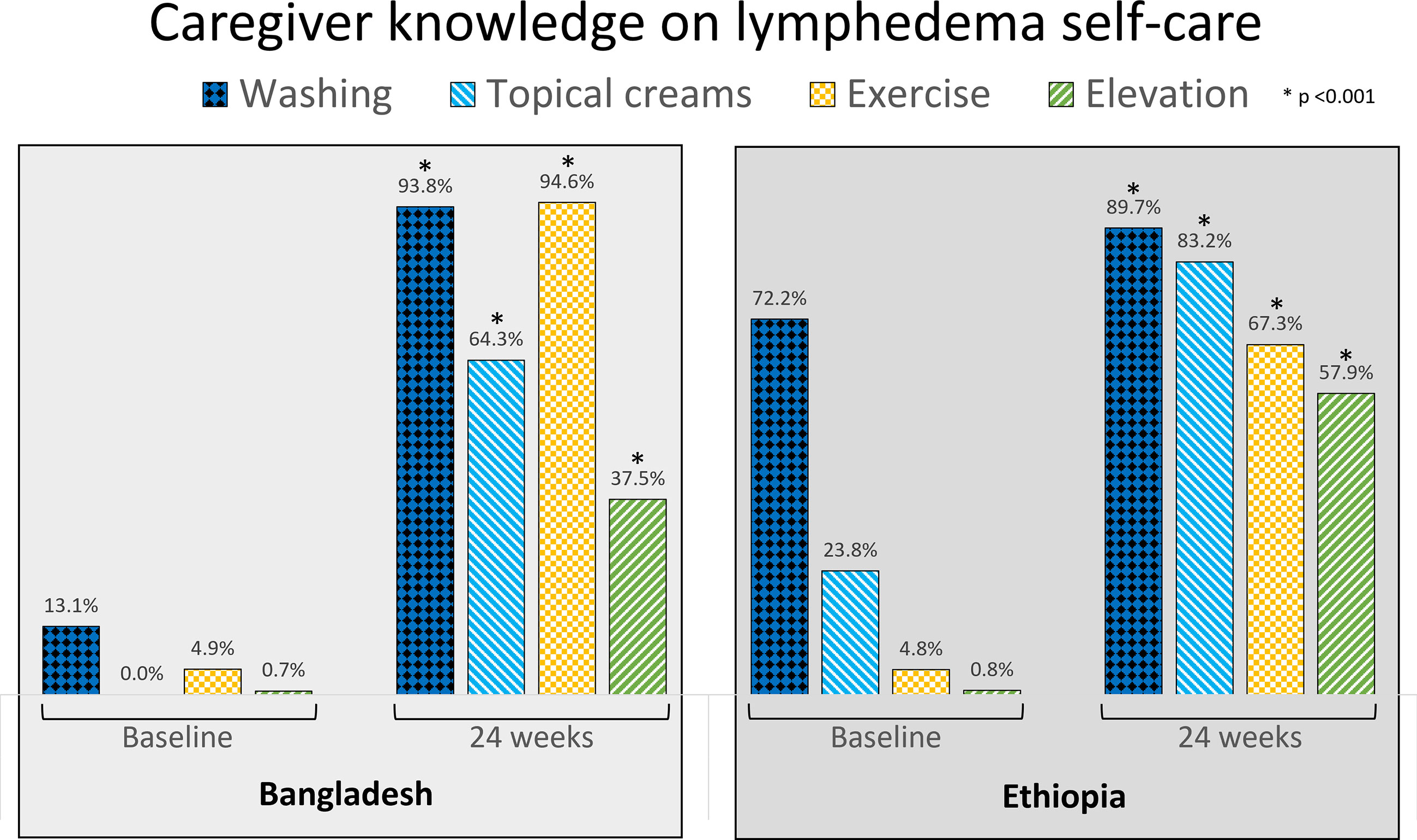
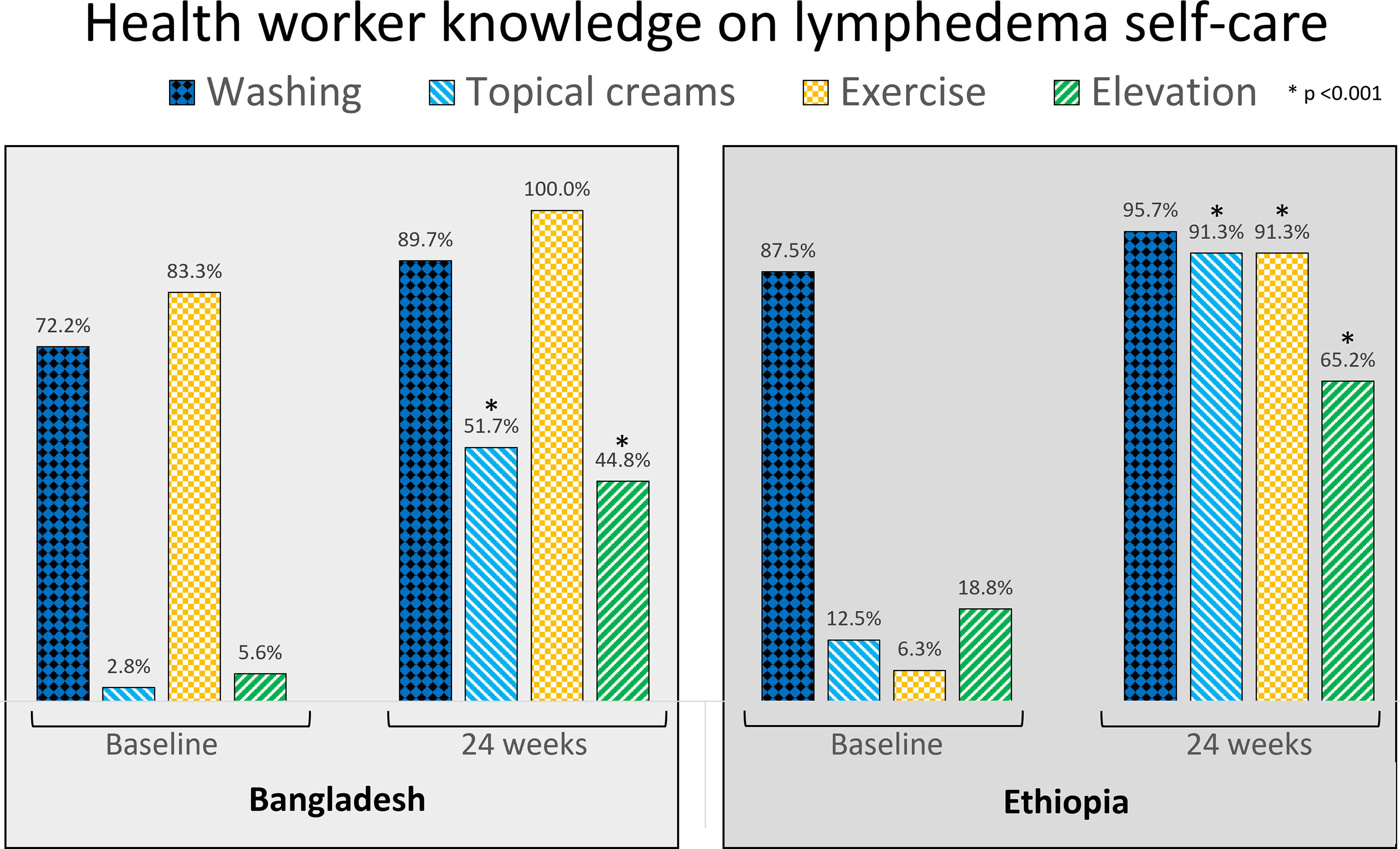
As shown in Figures 3–5 knowledge on lymphedema self-care increased significantly among patients, caregivers and health workers in both countries from baseline to 24-weeks, and 50% of patients in Bangladesh and 64.6% of patients in Ethiopia were able to identify all core activities.
There were notable between country differences at baseline for knowledge on limb washing among patients (Bangladesh n = 11; 7.7%, Ethiopia n = 64; 51.1%, Figure 3) and caregivers (Bangladesh n = 44; 31.0%, Ethiopia n = 91; 72.2%, Figure 4), but not between health workers (Bangladesh n = 26; 72.2%, Ethiopia n = 28; 87.5%). At baseline 83.3% of health workers in Bangladesh knew about exercise (n = 30), and 18.8% of health workers in Ethiopia knew about limb elevation (n = 6, Figure 5).
3.4 Lymphedema Self-Care Practice
3.4.1 Self-Reported Self-Care Practice
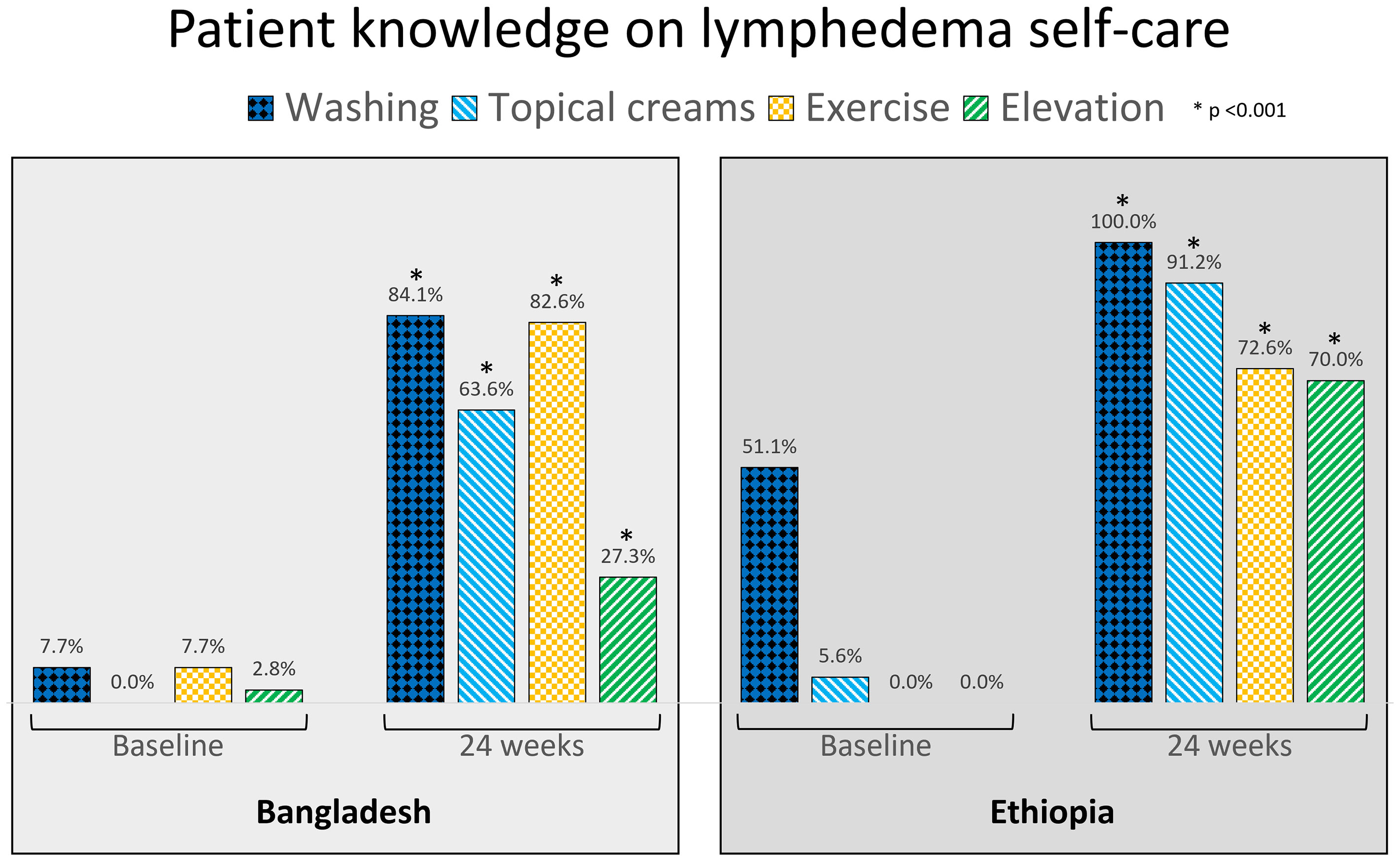
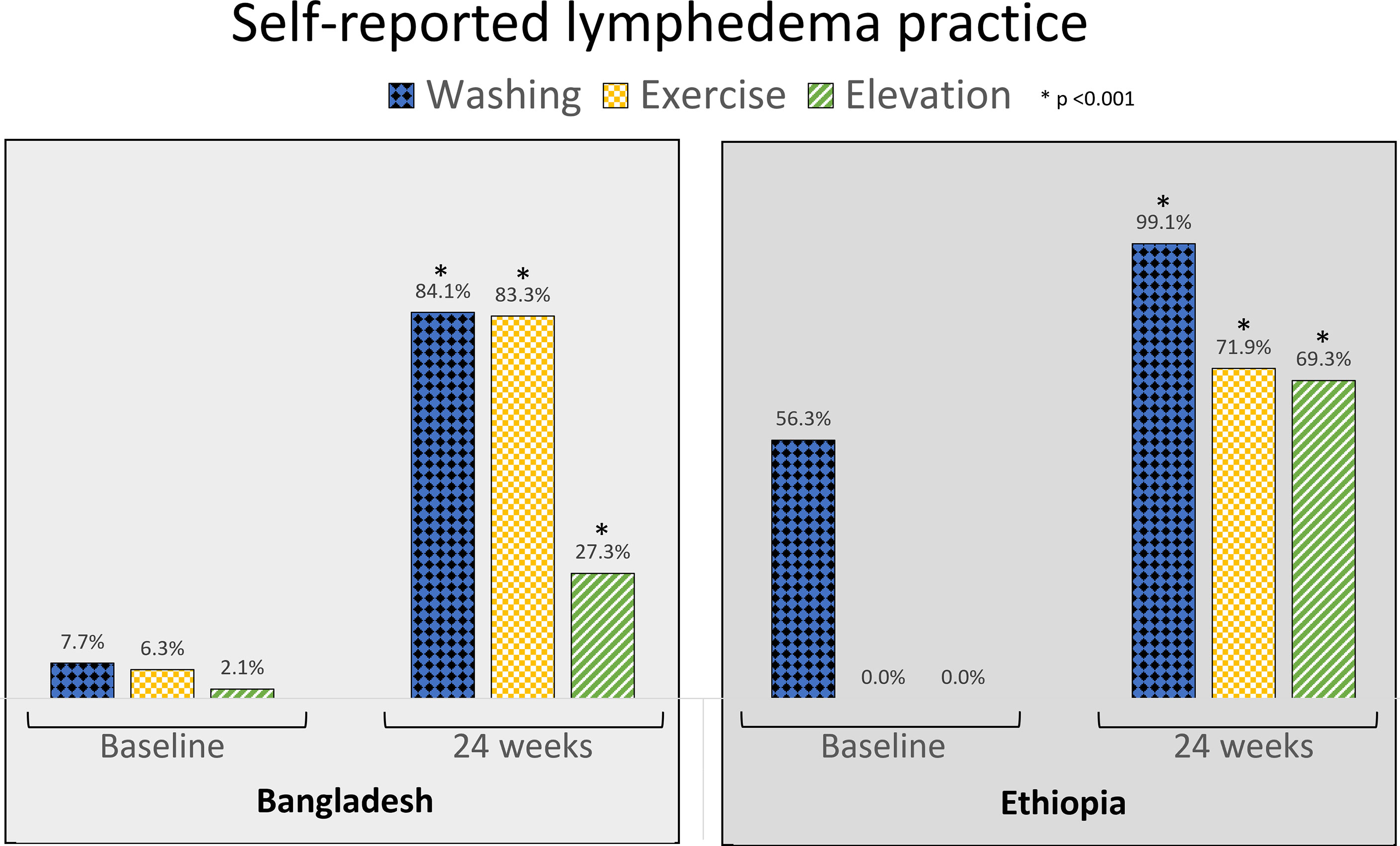
During baseline surveys, few patients in Bangladesh reported performing any lymphedema self-care activities (<10% for all activities) and in Ethiopia 56.3% (n = 71) of patients reported that they practiced limb washing but no-one reported using exercise or elevation. There was a significant improvement for all activities at 24-weeks in both countries (<0.001 for all comparisons, Figure 6).
3.4.2 Adherence to Self-Care Practices
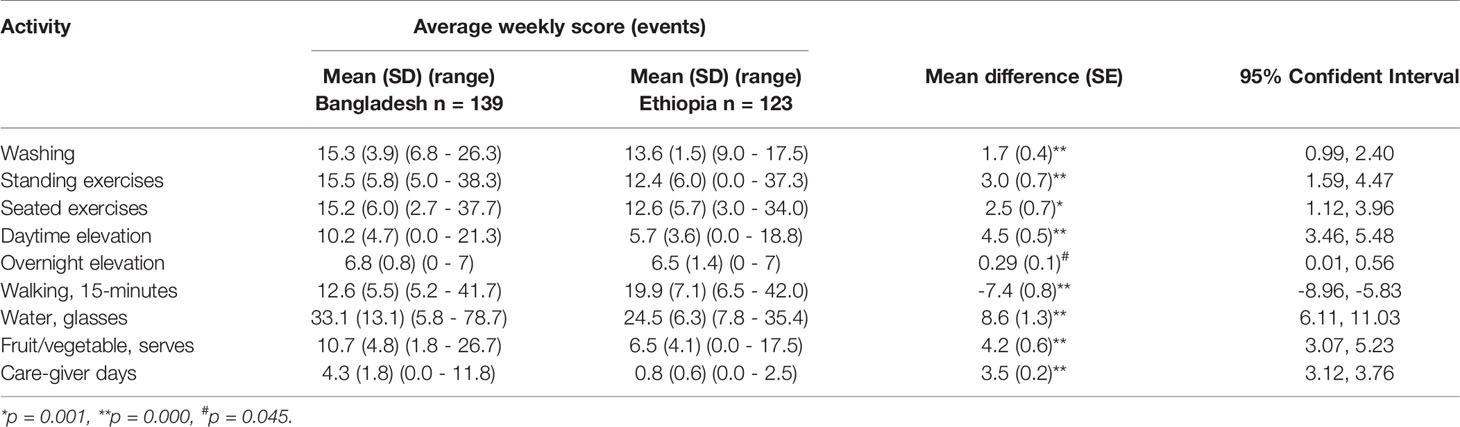
The journals of 123 patients in Bangladesh (97.6%) and 108 patients in Ethiopia (95.6%) were collected at 24-weeks and a weekly average score for each activity was calculated from the number of events recorded in the first week of each journal (Table 3). The highest score in each country was for limb washing (≥ 10 events per week) which was reported by 87.8% of patients in Bangladesh (n = 108) and 92.6% of patients in Ethiopia (n = 100). The next highest scores were for leg exercises (≥ 25 events per week) which were performed by 81.3% of patients in Bangladesh (n = 100) and 64.8% in Ethiopia (n = 70). The lowest scores were for the use of elevation which was used ≥ 5 times per week by 44.4% of patients in Bangladesh (n = 48) and 9.3% in Ethiopia (n = 10). Average weekly scores were significantly higher in Bangladesh than in Ethiopia for washing, exercise and elevation but the mean differences were small (p ≥ 0.001for all comparisons). There were larger and significant differences for; time spent walking which was higher in Ethiopia (mean difference 7.4, 95% CI 5.83, 8.96, p = 0.000), and water intake which was higher in Bangladesh (mean difference 8.6, 95% CI 6.11, 11.03, p = 0.000). The average number of days per week that caregiver assistance was given was also significantly higher in Bangladesh than in Ethiopia (mean difference 3.5 days, 3.12, 3.76, p = 0.000).
3.4.3 Factors Associated With Variation in Self-Care Adherence
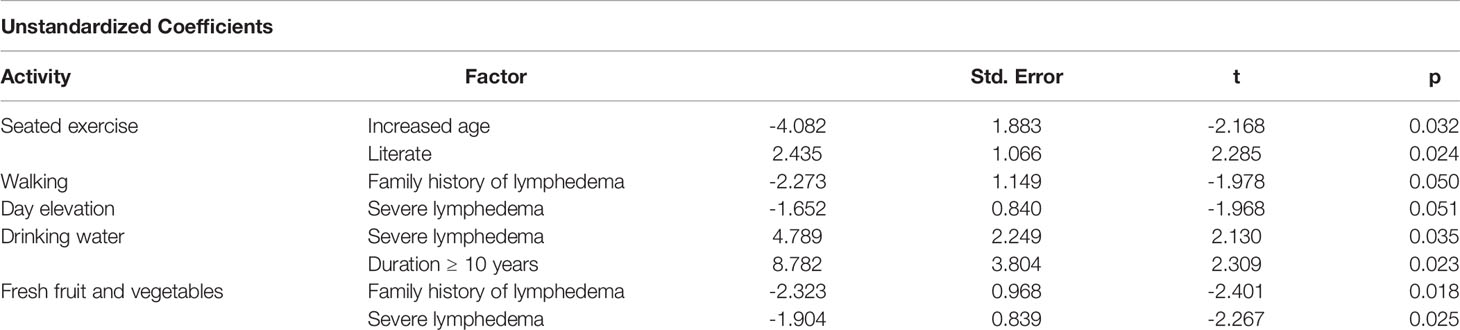
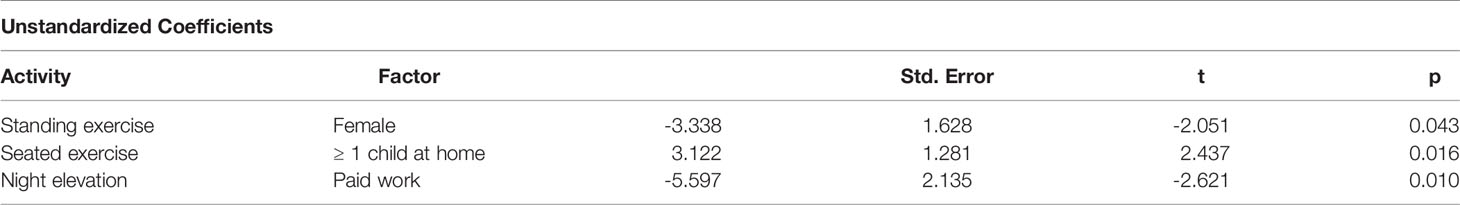
There were significant associations between participant characteristics and variation in journal scores at follow-up (average score for week-21) in both countries, for all activities except for limb washing (Tables 4 and 5). There were no significant associations in either country between performance of self-care activities and either marital status, or the amount of caregiver time required.
Factors associated with significant variation in self-care in Bangladesh are shown in Table 4. An increased age was associated with lower weekly scores for performance of the leg exercises (seated = 4 fewer events, p = 0.032, standing = 3 fewer events, p = 0.068), while literacy was associated with higher scores for performance of the seated exercise (2 more events, p = 0.024). A family history of lymphedema was associated with lower weekly scores for walking (2 fewer events p = 0.050) and consumption of fresh fruit and vegetables (2 fewer days, p = 0.018). A more advanced stage of lymphedema was associated with lower scores for day elevation (2 fewer events, p = 0.0510) and consumption of fresh fruit and vegetables (2 fewer days, p = 0.025), but with higher scores for water intake (4 more glasses, p = 0.035). Lymphedema duration of ≥ 10 years was associated with higher weekly scores for water intake (9 more glasses, p = 0.023), but lower scores for night elevation (2 fewer nights, p = 0.070). There was a large association between an increased age and lower scores for water intake (7 fewer glasses, p = 0.070).
Factors associated with significant variation in self-care in Ethiopia are shown in Table 5. Being female was associated with lower weekly scores for both exercises (seated = 3 fewer events, p = 0.062, standing = 3 fewer events, p = 0.043), while having one or more dependent children at home was associated with higher scores for seated exercises (3 more events, p = 0.016). There was a large and significant association between being in paid work and lower weekly scores for night elevation (6 fewer nights, p = 0.010). There were no significant factors associated with fresh fruit and vegetable consumption or water intake in Ethiopia.
3.4.4 Reporting of Acute Symptoms and Days of Work Lost Due to Lymphedema
Data on acute symptoms and days of work lost due to lymphedema during the study period were extracted from the journals of 46 patients in Bangladesh and 29 patients in Ethiopia (Table 6). In both countries a greater proportion of patients recorded ADL symptoms such as pain and redness in their journals, than patients who recalled any ADL events during the study period at 24-weeks. Similarly, a greater proportion of patients in both countries recorded days of work lost due to lymphedema in their journals, than patients who reported days off work by recall at 24-weeks.
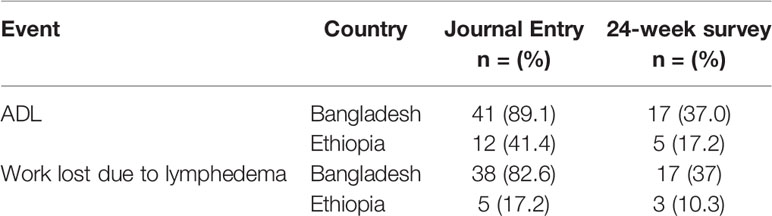
Table 6 Proportion of patients who reported ADL and days of work lost due to lymphedema by journal record and 24-week survey.
4 Discussion
The WHO Roadmap to end NTDs obliges us to measure the public health impact of interventions (1). However, countries seeking validation in the GPELF need only measure actions such as mapping cases, and reporting the number of health centers capable of offering services (8). There is no requirement to report on the number of people who are trained in lymphedema self-management, or the frequency of their self-care practice (8). However, results presented here from both Bangladesh and Ethiopia showed that while most health workers had good knowledge on lymphedema causes (>90%) and the need for limb hygiene (>70%), knowledge among the end-users, the patients and their caregivers was much lower, less than 25% for either population. This knowledge gap must be addressed, and the goal to reduce the suffering of people affected by lymphedema requires that effective tools are placed in the hands of those affected and their families (23). New approaches and tools are being developed to help health workers objectively assess the patient’s condition (24–26), but without measures in the validation dossier on meaningful actions taken by the end user in terms of quality-of-life, improvement in productivity, or reduction in disability, we fail to capture the public health impact of lymphedema interventions.
Chronic skin disease from LF, podoconiosis and other NTDs are managed by similar practices and present considerable potential for cross-cutting service delivery in an integrated health management system (4). To realize this, general and community specific challenges and barriers must be considered, and in our study contrasts in topography and climate provided a backdrop to knowledge and behavior in each community. In Bangladesh where water is plentiful and communities are closely clustered around low-lying rice paddies and vegetable gardens, people washed more frequently, performed more leg exercises but walked less. In Ethiopia where villages are separated by large tracks of dry farmland and water is scarce, people washed less frequently and walked more, doing fewer of the leg exercises. Elevation was used the least in both countries, which is consistent with other reports from Ethiopia (27), Zambia (28), Sri Lanka (29), and in Haiti (30) where compliance with leg washing was 88% while less than 50% of the study cohort were sleeping with the foot of the bed raised. We found several patient specific factors associated with adherence in each country but few previous reports for comparison. Age, sex and literacy have been discussed in regard to lymphedema knowledge and health seeking behaviors (31, 32), or in association with lymphedema stage or ADL frequency (23). Addiss etal. (33) reported that overall adherence to lymphedema self-care practices was significantly associated with being female (OR 1.6, 95% Confidence Interval [CI] 1.0–2.5, P = 0.05) and over 40 years of age (OR 1.5, 95% CI 1.1– 2.0, P = 0.02), but no other studies have reported on factors associated with performance of individual self-care activities. Adapting which practices are recommended or emphasized to accommodate environmental, personal and cultural factors may increase the potential for widespread acceptance and integration of beneficial practices across the whole community.
Data on adherence to self-care practices is typically collected either by survey in which participants are asked to recall events retrospectively, or via a patient journal where activities are recorded on a daily or weekly basis. In our study we collected adherence data using both methods with general concordance in the results on performance of the self-care activities, suggesting either method may be suitable for assessing adherence. However, in both countries, more acute symptoms were reported in the journals than via the surveys, which indicates that people may have experienced ADL symptoms more frequently during the study period than were captured by recall at the end. Likewise, more days where the patient was unable to work due to lymphedema appeared in the journals than were recalled by survey. It is our view that a daily journal or chart kept by the patient and/or their caregiver provides more accurate data on theses markers of disease than retrospective questionnaires.
Unfortunately, we did not ask patients or caregivers if they had received prior MDA or training in lymphedema self-management and data on this may have helped to elucidate the disconnect between knowledge held at the health center and general awareness found among patients and caregivers at baseline. Furthermore, in each country some of the original caregivers were replaced by a new caregiver during the course of the study. While we accounted for this in our before and after comparison on caregiver knowledge, it should be noted that repeated training of new stakeholders will be needed. The original study design and sample size calculations were based on the primary outcomes in the RCT and the results are limited by the lack of longer term follow up. A further shortcoming was in only extracting data from the first week of each 4-week journal to calculate the weekly adherence scores, however it was noted that the first page of each journal generally had the most complete entries with blank spaces and skipped pages more frequently among later pages. The study strengths were in the inclusion of a large number of participants in two contrasting settings, and the collection of data on caregivers and health workers as well as patients. We anticipate these observations on knowledge and adherence in Bangladesh and Ethiopia will be useful to other endemic countries with similar populations.
It is well documented that people affected by lymphedema at all stages have impaired qualify of life and increased financial pressures (34), and that introduction of a hygiene centered self-care program can improve symptoms and reduce ADL episodes (11, 12). In our study, people affected by moderate and severe lymphedema and their caregivers were able to learn and apply lymphedema self-care with a high level of adherence over 24-weeks. Such community based and patient-oriented health services are fundamental to achieving the Sustainable Development Goals (SDG) in universal health coverage, and for achieving global targets for action on NTDs (SDG 3), alleviating poverty (SDG 1) and hunger (SDG 2), enabling people to pursue an education (SDG 4) and lead productive working lives (SDG 8) (1), and to meet the core Roadmap agenda to move away from external drivers and enable in-country autonomy in program delivery (1).
5 Conclusion
In Bangladesh and Ethiopia, lymphedema knowledge and self-care practice increased significantly 24-weeks after lymphedema self-care training. Daily performance of lymphedema self-care can improve clinical outcomes and social inclusion for sufferers, but patient and community level ownership of the program is essential for sustainability. More needs to be done to ensure that knowledge held at the health center is transferred to the patients and caregivers and repeated for new stakeholders. Research on the lived experience of affected persons, on the effectiveness of individual components of self-care, and on factors affecting patient adherence are urgently needed to guide service delivery. People faced with lifelong chronic disease deserve to be guided by appropriate and acceptable recommendations for self-care and provided with the necessary tools and support to achieve it.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Ethics Statement
The studies involving human participants were reviewed and approved by Liverpool School of Tropical Medicine Research Ethics Committee (approval no. 012-18), the Bangladesh Medical Research Council, and the Amhara Public Health Institute Research Ethics Review Committee. The participants provided their written informed consent to participate in this study.
Author Contributions
Conceptualization, study design, methodology, manuscript preparation, JD, SM, HM, HB, LK-H, ASM, and FH. Contribution of resources, STJ, MJK, SMC, MT, MJT. Reviewing and editing MMR, AAK, AFR, AK, FS, TB, MN, KM, and GA. All authors contributed to the article and approved the submitted version.
Funding
This work was supported by funding from the Department for International Development (DFID) and GlaxoSmithKline (GSK) to the Centre for Neglected Tropical Diseases, Liverpool School of Tropical Medicine, for programmatic support for the elimination of lymphatic filariasis as a public health problem. Neither funder was involved in the study design, collection, analysis, interpretation of data, the writing of this article or the decision to submit it for publication.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Acknowledgments
Grateful thanks are given to all the people that volunteered to be in the study and to our research assistants in Ethiopia, Engdaw Getachew Abera, Gizachew Mezgeb Asfaw, Shimels Bahiru Adugna, Wondie Ayele Golnetie, Aklilu Ayehu Tassew, Fentaw Ayalew Nebebe, Kassahun Ewnetu Melese, Ewunetyhun Wale Gebeyehu and Ambachew Birhan Adugna, and in Bangladesh, Mahmud Hasan, Islam Md. Faizur Rashid, Farzana Mohsin, Imran Nazir, Mst Mansia Khatun, S. M. Raihan Nesar Jewel, Dulal Hossain, Kazi Burhan Uddin, and Samid Siddiqi.
References
1. WHO. Ending the Neglect to Attain the Sustainable Development Goals. A Road Map for Neglected Tropical Diseases 2021–2030. (2021) Geneva: World Health Organization, Roadmap v8_modif_feb_19-02.indd.
2. Mathew CG, Bettis AA, Chu BK, English M, Ottesen EA, Bradley MH, et al. The Health and Economic Burdens of Lymphatic Filariasis Prior to Mass Drug Administration Programs. Clin Infect Dis (2019) 70(12):2561–7. doi: 10.1093/cid/ciz671
3. WHO. Lymphatic Filariasis: Managing Morbidity and Preventing Disability: An Aide-Memoire for National Programme Managers. Geneva: World Health Organization (2013).
4. Deribe K, Kebede B, Tamiru M, Mengistu B, Kebede F, Martindale S, et al. Integrated Morbidity Management for Lymphatic Filariasis and Podoconiosis, Ethiopia. Belletin WHO (2017) 95(9):652–6. doi: 10.2471/BLT.16.189399
5. Fuller LC. Podoconiosis: Endemic Nonfilarial Elephantiasis. Curr Opin Infect Dis (2005) 18:119–22. doi: 10.1097/01.qco.0000160899.64190.15
6. Deribe K, Brooker SJ, Pullan RL, Sime H, Gebretsadik A, Assefa A, et al. Epidemiology and Individual, Household and Geographical Risk Factors of Podoconiosis in Ethiopia: Results From the First Nationwide Mapping. Am J Trop Med Hygiene (2015) 92(1):148–58. doi: 10.4269/ajtmh.14-0446
7. Caprioli T, Martindale S, Mengiste A, Assefa D, Hailekiros F, Tamiru M, et al. Quantifying the Socio-Economic Impact of Leg Lymphoedema on Patient Caregivers in a Lymphatic Filariasis and Podoconiosis Co-Endemic District of Ethiopia. PLoS Negl Trop Dis (2020) 14(3):e0008058. doi: 10.1371/journal.pntd.0008058
8. WHO. Validation of Elimination of Lymphatic Filariasis as a Public Health Problem. Geneva: World Health Organization (2017).
9. WHO. “Progress Report 2000-2009 and Strategic Plan 2010-2020 of the Global Programme to Eliminate Lymphatic Filariasis: Halfway Towards Eliminating Lymphatic Filariasis”. In: WHO Library Catalogue. Geneva: World Health Organization (2010).
11. Stocks ME, Freeman MC, Addiss DG. The Effect of Hygiene-Based Lymphedema Management in Lymphatic Filariasis-Endemic Areas: A Systematic Review and Meta-Analysis. PLoS Neglected Trop Dis [electronic resource] (2015) 9(10):e0004171. doi: 10.1371/journal.pntd.0004171
12. Douglass J, Graves P, Gordon S. Self-Care for Management of Secondary Lymphedema: A Systematic Review. PLoS Neglected Trop Dis (2016) 10(6):e0004740. doi: 10.1371/journal.pntd.0004740
13. Sawers L, Stillwaggon E. Economic Costs and Benefits of Community-Based Lymphedema-Management Programs for Lymphatic Filariasis in India. Am J Trop Med Hygiene (2020) 103(1):295–302. doi: 10.4269/ajtmh.19-0898
14. Douglass J, Mableson HE, Martindale S, Kelly-Hope LA. An Enhanced Self-Care Protocol for People Affected by Moderate to Severe Lymphedema. Methods Protoc (2019) 2(3):77. doi: 10.3390/mps2030077
15. Douglass J, Mableson H, Martindale S, Jhara ST, Karim MJ, Rahman MM, et al. Effect of an Enhanced Self-Care Protocol on Lymphedema Status Among People Affected by Moderate to Severe Lower-Limb Lymphedema in Bangladesh, a Cluster Randomized Controlled Trial. J Clin Med (2020) 9(8):2444. doi: 10.3390/jcm9082444
16. Douglass J, Hailekiros F, Martindale S, Mableson H, Seife F, Bishaw T, et al. Addition of Lymphatic Stimulating Self-Care Practices Reduces Acute Attacks Among People Affected by Moderate and Severe Lower-Limb Lymphedema in Ethiopia, a Cluster Randomized Controlled Trial. J Clin Med (2020) 9(12):4077. doi: 10.3390/jcm9124077
17. Shamsuzzaman AKM, Haq R, Karim MJ, Azad MB, Mahmood ASMS, Khair A, et al. The Significant Scale Up and Success of Transmission Assessment Surveys ‘Tas’ for Endgame Surveillance of Lymphatic Filariasis in Bangladesh: One Step Closer to the Elimination Goal of 2020. PLoS Neglected Trop Dis (2017) 11(1):e0005340. doi: 10.1371/journal.pntd.0005340
18. Kebede B, Martindale S, Mengistu B, Kebede B, Mengiste A, H/Kiros F, et al. Integrated Morbidity Mapping of Lymphatic Filariasis and Podoconiosis Cases in 20 Co-Endemic Districts of Ethiopia. PLoS Neglected Trop Dis (2018) 12(7):e0006491. doi: 10.1371/journal.pntd.0006491
19. World Medical Association. World Medical Association Declaration of Helsinki: Ethical Principles for Medical Research Involving Human Subjects. JAMA (2013) 310(20):2191–4. doi: 10.1001/jama.2013.281053
20. Registry, I.S.R.C.T.N. (2021). Available at: https://www.isrctn.com/ISRCTN16764792?q=16764792&filters=&sort=&offset=1&totalResults=1&page=1&pageSize=10&searchType=basic-search (Accessed 16 August 2021).
21. Dreyer G, Addiss D, Dreyer P, Noroes J. Basic Lymphoedema Management, Treatment and Prevention Problems Associated With Lymphatic Filariasis. New Hampshire, USA: Hollis Publishing Company (2002).
22. Hartung C, Lerer A, Anokwa Y, Tseng C, Brunette W, Borriello G. “Open Data Kit: Tools to Build Information Services for Developing Regions”. In: The 4th ACM/IEEE International Conference on Information and Communication Technologies and Development. London, United Kingdom: ACM (2010).
23. Negussie H, Molla M, Ngari M, Berkley JA, Kivaya E, Njuguna P, et al. Lymphoedema Management to Prevent Acute Dermatolymphangioadenitis in Podoconiosis in Northern Ethiopia (GoLBeT): A Pragmatic Randomised Controlled Trial. Lancet Global Health (2018) 6:e795–803. doi: 10.1016/S2214-109X(18)30124-4
24. Douglass J, Graves P, Lindsay D, Becker L, Roineau M, Masson J, et al. Lymphatic Filariasis Increases Tissue Compressibility and Extracellular Fluid in Lower Limbs of Asymptomatic Young People in Central Myanmar. Trop Med Infect Dis (2017) 2(4):50. doi: 10.3390/tropicalmed2040050
25. Yahathugoda C, Weiler MJ, Rao R, De Silva L, Dixon JB, Weerasooriya MV, et al. Use of a Novel Portable Three-Dimensional Scanner to Measure Limb Volume and Circumference in Patients With Filarial Lymphedema. Am J Trop Med Hygiene (2017) 97(6):1836–42. doi: 10.4269/ajtmh.17-0504
26. Kelly-Hope LA, Karim MJ, Sultan Mahmood A, Al Kawsar A, Khair A, Betts H, et al. Infrared Thermal Imaging as a Novel Non-Invasive Point-Of-Care Tool to Assess Filarial Lymphoedema. J Clin Med (2021) 10(11):2301. doi: 10.3390/jcm10112301
27. Churko C, Yohanes T, Kassahun AB, Desalegn N, Endashaw G, Asfaw MA. Foot Care Practice and Associated Factors Among Patients With Lymphoedema in Boreda District, Gamo Zone, Southern EthiopiImplications for Elimination of Podoconiosis and Lymphatic Filariasis. J Foot Ankle Res (2021) 14(1):51. doi: 10.1186/s13047-021-00490-8
28. Mathieu E, Dorkenoo AM, Datagni M, Cantey PT, Morgah K, Harvey K, et al. It Is Possible: Availability of Lymphedema Case Management in Each Health Facility in Togo. Program Description, Evaluation, and Lessons Learned. Am J Trop Med Hygiene (2013) 89(1):16–22. doi: 10.4269/ajtmh.12-0453
29. Wijesinghe RS, Wickremasinghe AR, Ekanayake S, Perera MS. Efficacy of a Limb-Care Regime in Preventing Acute Adenolymphangitis in Patients With Lymphoedema Caused by Bancroftian Filariasis, in Colombo, Sri Lanka. Ann Trop Med Parasitol (2007) 101(6):487–97. doi: 10.1179/136485907X193806
30. Addiss DG, Louis-Charles J, Roberts J, Leconte F, Wendt JM, Milord MD, et al. Feasibility and Effectiveness of Basic Lymphedema Management in Leogane, Haiti, an Area Endemic for Bancroftian Filariasis. PLoS Neglected Trop Dis (2010) 4(4):e668. doi: 10.1371/journal.pntd.0000668
31. Babu BV, Hazra RK, Chhotray GP, Satyanarayana K. Knowledge and Beliefs About Elephantiasis and Hydrocele of Lymphatic Filariasis and Some Socio-Demographic Determinants in an Endemic Community of Eastern India. Public Health (2004) 118(2):121–7. doi: 10.1016/S0033-3506(03)00144-6
32. Maritim P, Silumbwe A, Zulu JM, Sichone G, Michelo C. Health Beliefs and Health Seeking Behavior Towards Lymphatic Filariasis Morbidity Management and Disability Prevention Services in Luangwa District, Zambia: Community and Provider Perspectives. PLoS Neglected Trop Dis (2021) 15(2):e0009075. doi: 10.1371/journal.pntd.0009075
33. Addiss DG, Michel MC, Michelus A, Radday J, Billhimer W, Louis-Charles J, et al. Evaluation of Antibacterial Soap in the Management of Lymphoedema in Leogane, Haiti. Trans R Soc Trop Med Hygiene (2011) 105(1):58–60. doi: 10.1016/j.trstmh.2010.08.011
Keywords: lymphedema, self-care, lymphatic filariasis (LF), podoconiosis, knowledge - attitude - practice, neglected tropical disease (NTD), gaps, insight
Citation: Douglass J, Martindale S, Mableson H, Jhara ST, Karim MJ, Rahman MM, Kawsar AA, Khair A, Mahmood ASMS, Rahman AKMF, Chowdhury SM, Hailekiros F, Tamiru M, Seife F, Bishaw T, Nigussie M, Meribo K, Agidew G, Betts H, Taylor MJ and Kelly-Hope LA (2021) Insights on Lymphedema Self-Care Knowledge and Practice in Filariasis and Podoconiosis-Endemic Communities in Bangladesh and Ethiopia. Front. Trop. Dis 2:767045. doi: 10.3389/fitd.2021.767045
Received: 30 August 2021; Accepted: 22 September 2021;
Published: 08 October 2021.
Edited by:
Alexander Yaw Debrah, Kwame Nkrumah University of Science and Technology, GhanaReviewed by:
Abdallah Ngenya, National Institute of Medical Research, TanzaniaInge Kroidl, Ludwig Maximilian University of Munich, Germany
Copyright © 2021 Douglass, Martindale, Mableson, Jhara, Karim, Rahman, Kawsar, Khair, Mahmood, Rahman, Chowdhury, Hailekiros, Tamiru, Seife, Bishaw, Nigussie, Meribo, Agidew, Betts, Taylor and Kelly-Hope. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Janet Douglass, amFuLmRvdWdsYXNzQGpjdS5lZHUuYXU=
†These authors have contributed equally to this work and share first authorship
 Janet Douglass
Janet Douglass Sarah Martindale1†
Sarah Martindale1† Muhammad Mujibur Rahman
Muhammad Mujibur Rahman Salim Mahmud Chowdhury
Salim Mahmud Chowdhury Mark J. Taylor
Mark J. Taylor Louise A. Kelly-Hope
Louise A. Kelly-Hope