- 1Department of Environmental Sciences, Informatics and Statistics, Ca’ Foscari University of Venice, Venice, Italy
- 2Economic Analysis of Climate Impacts and Policy Division, Euro-Mediterranean Center on Climate Change, Venice, Italy
- 3RFF-CMCC European Institute on Economics and the Environment, Venice, Italy
Controlling malaria is one of the top health sector priorities in Ethiopia. The concrete prevention, control, and treatment interventions undertaken in the past two decades have substantially reduced the morbidity and mortality attributable to malaria. Emboldened by these past achievements, Ethiopia envisages to eliminate malaria by 2030. Realizing this ambition, however, needs to further strengthen the financial, technical, and institutional capacities to address the current as well as emerging challenges. It particularly needs to step up measures pertaining to diagnosis, domestic resource mobilization, vector surveillance, and seasonal weather forecasting.
Highlights
● Ethiopia has made a remarkable progress in terms of controlling malaria, especially, since 2004.
● It is further campaigning towards a “malaria free Ethiopia”.
● This requires building strong cross-sectoral and cross-border coordination capacity.
● It also needs to scale up research and surveillance on emerging malaria vectors.
● The implications of irrigation and hydropower dams on malaria transmission should not be undermined.
Introduction
Malaria is one of the major infectious tropical diseases with substantial socio-economic repercussions in the sub-Saharan Africa region. In 2019, WHO’s African Region accounted for about 94% of malaria cases while only six African countries (Nigeria, Democratic Republic of the Congo, Tanzania, Mozambique, Niger, and Burkina Faso) accounted for about 51% of all malaria deaths globally (1). The number of estimated malaria cases in Africa in 2019 were 215 million (1).
Malaria-related morbidity and mortality entail substantial private costs (e.g., direct costs due to clinical treatments, and indirect costs due to reduced labor productivity) and societal costs (e.g., increasing public health expenditure, and effects on labor, investment, and tourism flows) in many tropical countries (2).
Currently, more than 50% of the population in Ethiopia is exposed to the risk of malaria infection (3–5). Despite the range of prevention measures undertaken in the last two decades, malaria remains to be one of the top ten causes of morbidity and mortality in Ethiopia (6) with substantial repercussions for the macroeconomy (7, 8). The effects of malaria propel into the macroeconomy through two main channels. The first is through agricultural labor productivity changes as the malaria transmission seasons (September to December, and April to May) coincide with the main agriculture harvest seasons (5, 7, 8). Agriculture is the main source of employment (≈ 75%) and merchandise export earnings (≈ 80%) in Ethiopia (9). The second way is through government budget allocation and fiscal balance as government is the main health service provider (4). Seen against the forgoing conditions in Ethiopia, even a marginal increase in the risk of malaria has profound socio-economic implications making malaria disease public health as well as economic problem (8).
It is therefore important to assess the status of malaria risk, and to continuously evaluate the prevention and control measures in the country. It is equally important to identify the existing policy and implementation gaps, and emerging challenges that may undermine (or even reverse) the progress made so far. This paper aims to contribute its part in this regard. It briefly discusses the status of malaria risk, the past achievements, gaps, and emerging challenges in fighting malaria in Ethiopia. The study is a narrative overview that aims to briefly synthesize the existing knowledge, and to draw implications for future research and policy makers (10–12).
The remainder of the paper is structured as follows. Section 2 gives a brief overview of the malaria risk in Ethiopia. Section 3 succinctly presents the malaria prevention and control measures followed by the past achievements, the current gaps, and emerging issues related to malaria control measures in Section 4. This is followed by some recommendations in Section 5, and conclusions in Section 6.
Overview of Malaria Risk
Malaria transmission in Ethiopia is seasonal, unstable, and often characterized by highly focal and large-scale cyclic epidemics (1, 7). Areas lying at altitudes between 1600 and 2000 meters above sea level (masl) are in general epidemic prone hypo-endemic zones of malaria (4) although some studies could also detected malaria in areas higher than 2000 masl (5, 7). Altitude, climate, environmental changes (e.g., due to dams, roads construction, agricultural projects), and housing conditions are important determinants of malaria risk and transmission in Ethiopia (3, 7). Overall, more than 50% of the total population in Ethiopia is exposed to the risk of malaria infection (3–5). Every year, four to five million people are affected by malaria (3, 13) while a major epidemic occurs every five to eight years (7).
The reported malaria cases remain higher than one million cases per annum. On an average, about 1.6 million malaria cases (more than 60% is related to the P. falciparum species) are reported between 2010 and 2019 (1, 5). The rate of the P. falciparum species is especially higher in the lowland regions (7).
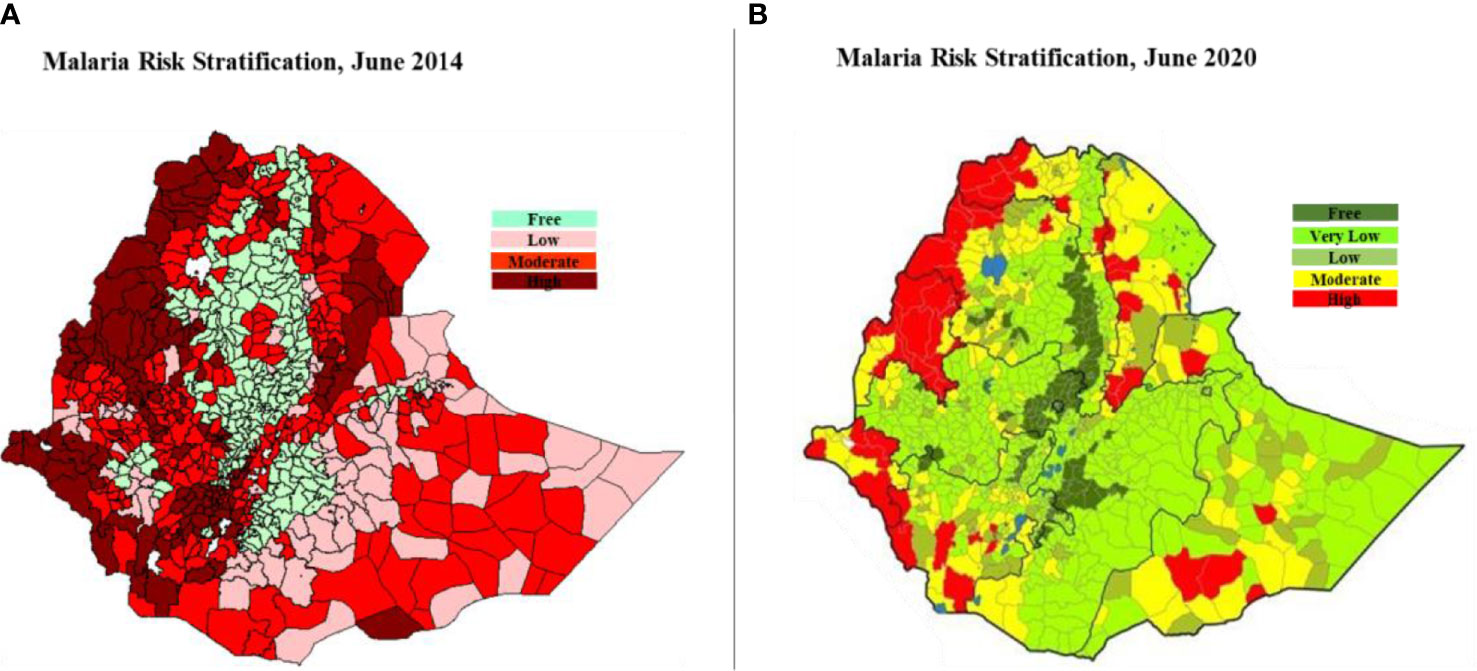
There are five distinct malaria risk strata classified based on annual parasite incidence (API) per 1,000 population, elevation, and expert opinions (7). According to the latest malaria risk classification, about 18% of the population lives in high (API ≥ 50) and moderate (10 ≤ API < 50) risk strata. Such classifications are important as they facilitate the design and implementation of most appropriate interventions per strata (5, 7, 14). It worth noting here that the latest stratification (7) and percentage distribution of population living under each risk stratum is different from the previous stratifications and percentage distributions such as, for example, the one in 2014 (14). Figure 1 collates the two stratification maps together. See also that the latest stratification adds one more stratum which is very low risk.
Policy Responses to Malaria
Ethiopia has been fighting malaria through formal institutions for more than five decades (5, 13). Today, malaria is one of its top national health and economic development priorities (7). In line with this, especially since 2004, the Government of Ethiopia together with its international partners has implemented a series of malaria prevention, control, and elimination programs (7) including preparing malaria guidelines (15, 16) and strategies (7, 17–19), and conducting surveys (14, 20, 21). The guidelines present detailed procedures on implementing and reporting various malaria vector control, diagnosis and treatment, and surveillance and response activities (15, 16). The periodic surveys produce evidence needed to formulate as well as evaluate different policy measures. All in all, policy wise, Ethiopia is stepping up its efforts to move from controlling (17) to eliminating (7) malaria.
Ethiopia started scaling up prevention and control measures in 2004 (5). Notable large-scale interventions include distribution of long-lasting insecticidal nets (LLINs), and indoor residual spraying (IRS), and introduction of additional diagnosis and treatment mechanisms such as artemisinin-based combination therapy (ACT), artemether lumefantrine (AL), and rapid diagnostic test (RDT) (5, 7, 13). Furthermore, in 2010 it started implementing test-treat policy, i.e., administration of antimalarial drugs based on test results (5). Figure 2, which is adapted from (5), depicts the timeline for major interventions between 1995 and 2016.
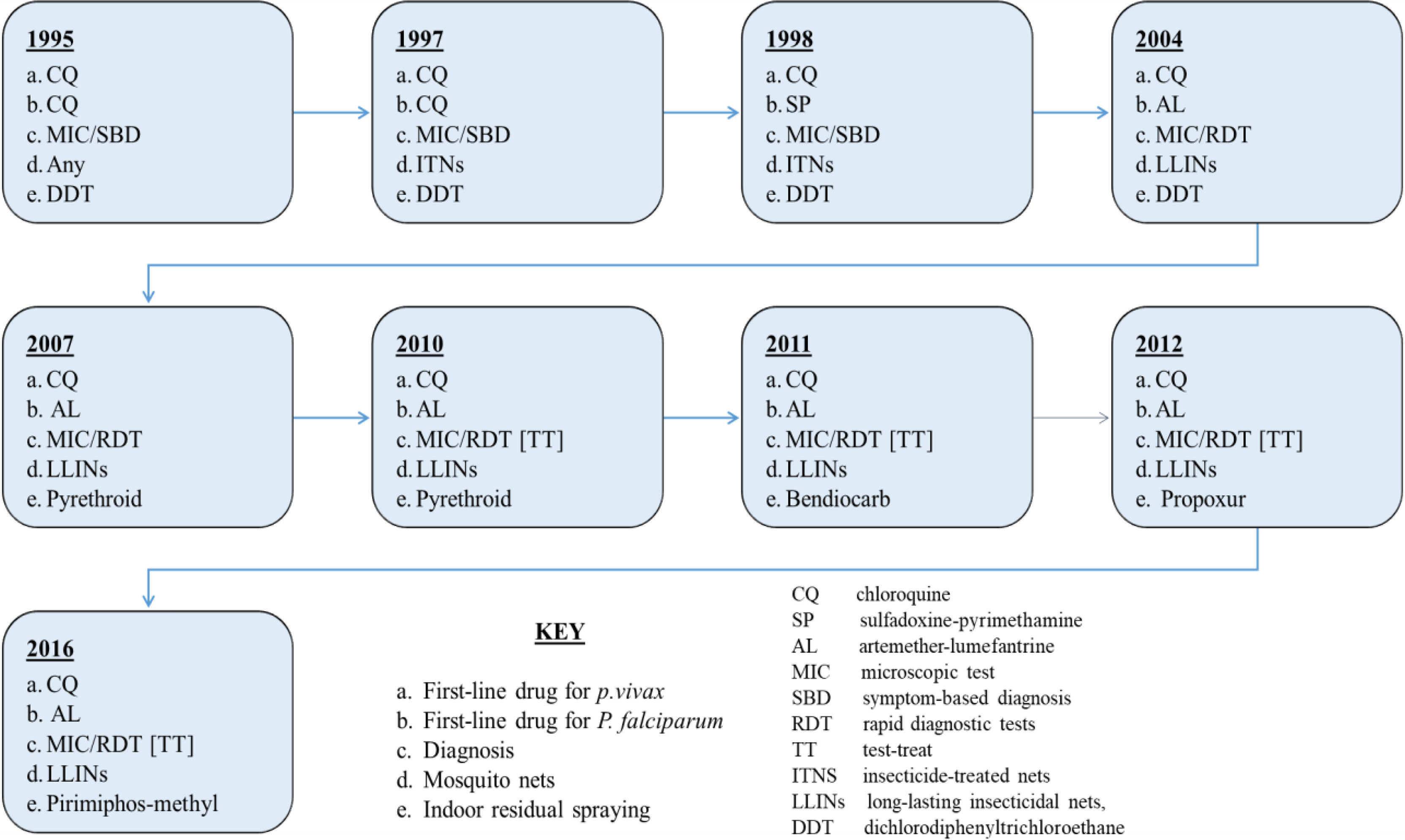
Figure 2 Timeline of major malaria control, diagnosis, and treatment interventions in Ethiopia, 1995-2016.
Remarkable progress has been made afterwards. The share of households in malarious areas (≤ 2000 masl) that possess at least one LLINs and received IRS have, respectively, reached 85% and 93% in 2019 (22). The introduction of RDTs in 2004 was a significant step forward in terms of case detection and management (3) since RDTs are easy to use and to deploy in rural areas (23) where 80% of Ethiopians live. These mass scale deployment of malaria specific inputs were also accompanied by the deployment of health extension workers (HEW) in rural areas which conduct home-to-home outreach activities, and provide basic curative, promotive, and preventive services at health posts (24).
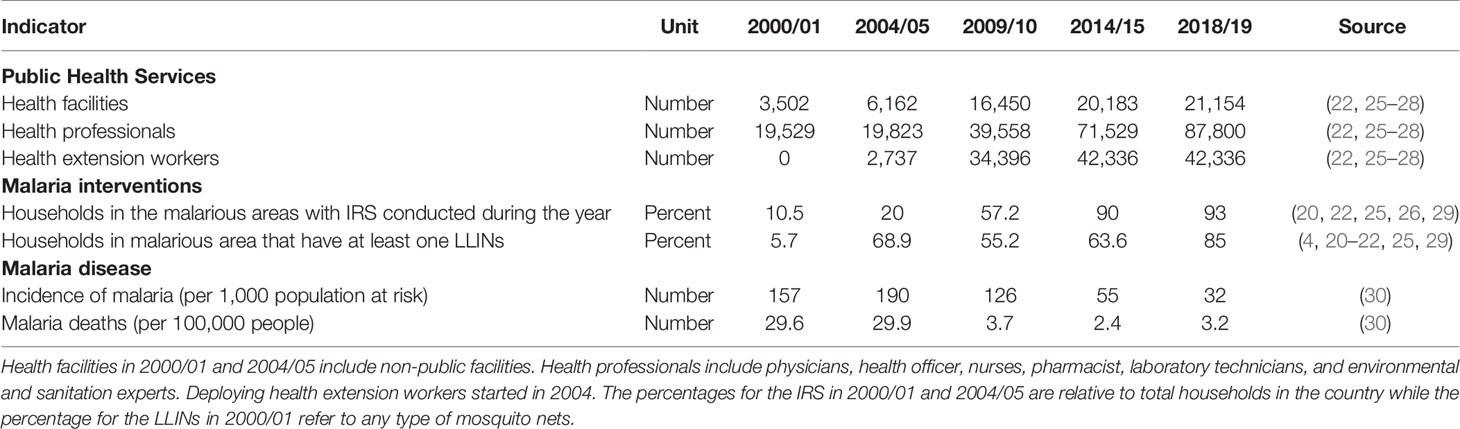
The malaria-specific interventions are also complemented with the overall increase in the number of health infrastructure (e.g., public health facilities and professionals) which profoundly improved malaria case management (13, 24). Notwithstanding the limitations, currently, about 68% of the facilities in the country offers malaria diagnosis or treatment (4). Table 1 presents the key trends in public health services, in malaria control measures, and in malaria disease in the past two decades.
Achievements, Gaps, and Emerging Issues
Achievements
The range of malaria prevention and control measures discussed in the preceding section are paying off. Mortality and morbidity attributable to malaria have significantly declined (7, 30). The number of deaths due to malaria declined by 54% between 2000 and 2016 (31) while the age-standardized mortality rate of malaria has declined by 96.5% between 1990 and 2015 (32). Ethiopia is also on a good track in terms of meeting its Global Technical Strategy for Malaria (2016-2030) (1, 7).
These past achievements encouraged the country to set more ambitious future goals. It envisages to reduce malaria morbidity and mortality, relative to 2020, by 50% in 2025 and to eliminate malaria by 2030 (7). More specifically, it aims to reduce malaria deaths per 100,000 population at risk (from 0.36 to 0.1), and the number of reported cases (from 1.7 million to 0.7 million) between 2019/20 and 2025/26 (7). Seen against these past achievements and the ongoing efforts, Ethiopia’s aim to eliminate malaria by 2030 seems ambitious but attainable. (13). There are however outstanding gaps and emerging challenges that should be addressed to keep this momentum of fighting malaria. Some of them are highlighted below.
Gaps
Diagnosis is one of the key factors in controlling and eliminating malaria. In Ethiopia, 63% of the health facilities provide malaria diagnosis testing (4). Of the alternative methods of malaria diagnosis (33), a microscopic diagnosis allows for the identification of parasitemia percentage, parasitic morphology, and speciation (23). The microscopic diagnosis in Ethiopia is limited. Health facilities that offered malaria diagnosis by microscopy (17%) was much lower than health facilities that offered diagnosis by clinical symptoms (42%) and RDT (54%) (4). Diagnosis by microscopy is available only in 7% of the rural health facilities (4). This represents a major diagnosis capacity gap seen against the fact that about 80% of the population lives in rural areas (7) with poor housing conditions, and thus at higher risk of infection (3). A recent review on the relative advantages and limitations of different malaria diagnostic methods can be found in (23). The readiness for malaria diagnostic capacity is 55% in private health facilities compared to 80% in government health facilities (4).
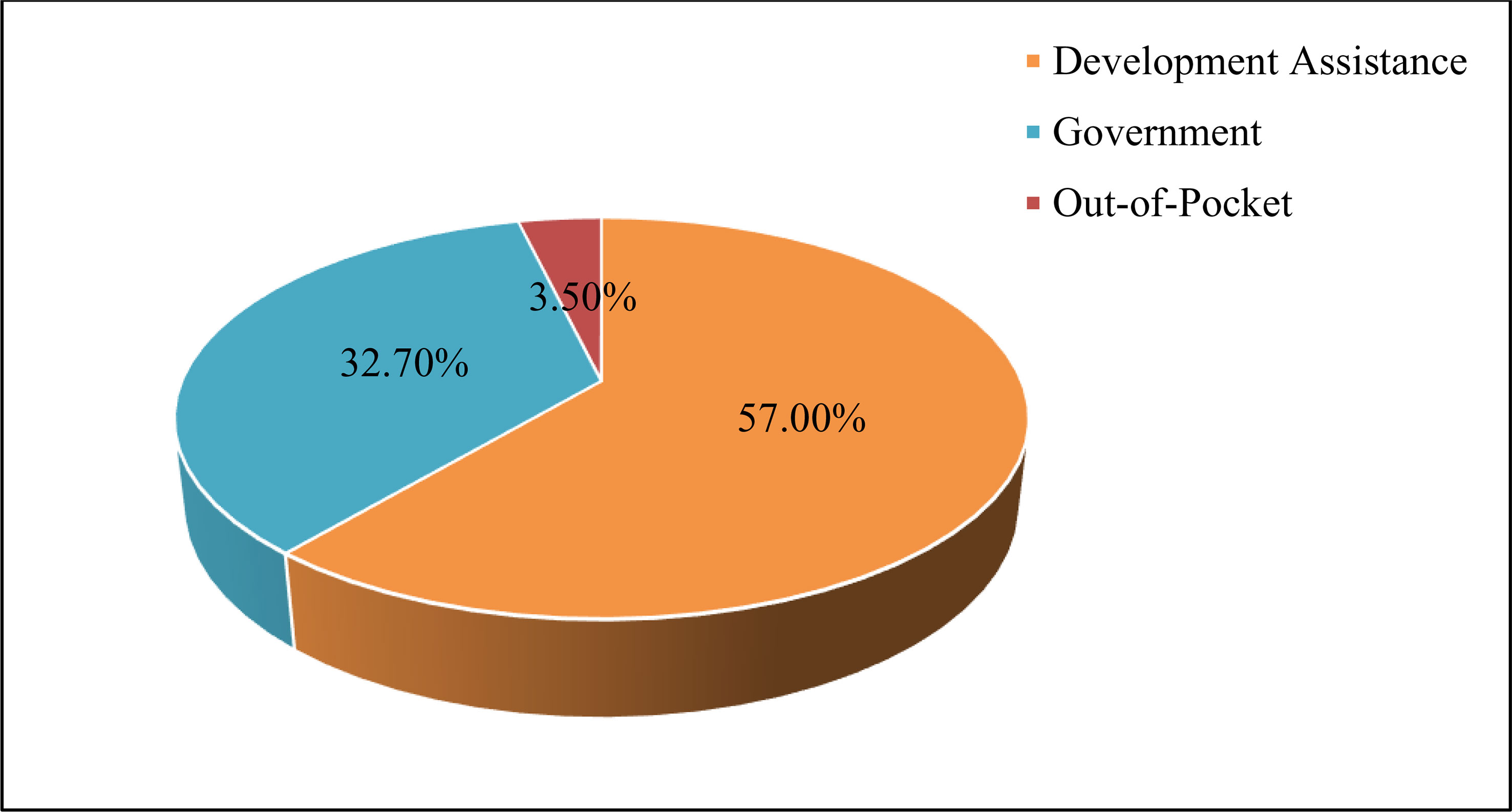
On the other hand, sufficient and sustained amount of funding is required to keep the momentum of fighting malaria (24). The generous funding from external sources has been one of the major reasons behind the past success (5, 13). Development assistance, for instance, contributes about 57% of an estimated US$ 81.2 million total spending on malaria in 2016 (34). See also Figure 3 which is based on (34). The Global Fund and the US Presidents’ Malaria Initiative (PMI) are the two main sources of external funds (7). Ethiopia received close to US$ 0.5 billion between 2008 and 2021 from the PMI funds (35). The biggest share of the external funds is spent on fixed costs and commodities such as LLINs, ACTs, and RDTs (7, 35). As such, it is fair to argue that the prospects of malaria elimination goals partly hinges on the financial commitments by the international donors. The amount of external funds are however expected to decline, and thus domestic sources should fill the gap. For example, the Government of Ethiopia anticipates financing about 56% of the total spending required to implement the malaria elimination plan by 2025/26 (7). In spite of this increased budgetary commitment from the government, however, implementing the current Ethiopia Malaria Elimination Strategic Plan (2021/22-2025/26) will still face about US$ 167.9 million financial gap (7). Diversifying the sources of funds is particularly important in light of unforeseen global and domestic challenges such as the COVID-19 pandemics that may affect the priority areas of the government as well as international partners (7).
Emerging Challenges
The past progress in fighting malaria is threatened by a set of emerging challenges due to such as mosquito resistance to insecticides, the emergence of new vectors, the potential side effects of irrigation and hydropower reservoirs, and climate change and variability (7). Recent evidence shows that local vectors are generally resistant to dichloro-diphenyl-trichloroethane (DDT) and pyrethroids (35), and the LLINs (36). The use of DDT for IRS was of course discontinued in 2007 (5). The emergence of new vectors such as the A.stephensi, which were not previously widely known, poses yet another challenge (1, 7). Anopheles stephensi mosquitoes breed predominately in urban settings preferably in man-made water containers and poses risk for increased transmission of P. falciparum and P.vivax (37). Currently, the A. stephensi vector is widely distributed and established in the eastern parts of Ethiopia (38). On the other hand, Ethiopia is expanding irrigation and hydropower dams that were found intensifying malaria transmission (39). For instance, malaria incidence was about 32% among households in villages with irrigation micro-dams compared to 19% in villages with no micro-dams in northern Ethiopia (40). The side effects are much more pronounced in the lowland and midland ecological settings (41) where the country is recently eyeing to expand its large-scale irrigated agriculture. On top of this, temperature suitability for malaria is climbing into the highlands of Ethiopia (42, 43) because of which the prevalence of malaria is projected to increase (44). A case study in northern Ethiopia shows that climate change may increase area suitable for malaria transmission by 94 to 114% by 2050 (45). Overall, countrywide, up to 130 million people may be at risk of malaria by 2070 (46) that could induce substantial economic costs (8, 42).
Actionable Recommendations
Therefore, in order to maintain the momentum of fighting malaria, it needs to scale up measures related to funding, climate services, and vector control. More specifically, it needs to:
● Raise the domestic resource mobilization capacity (7).
● Enhance climate information processing capacity (47).
● Consider dam reservoir management as one of malaria vector control tools (48).
● Scale up research (38), and surveillance (37) capacity particularly with regard to the emerging vectors.
● Pursue regional cooperation to control cross-border malaria transmission through migration (24), and to surveille the emerging vectors (38).
Conclusions
Ethiopia has been undertaking a wide range of policy measures to control malaria, especially, after 2004. Consequently, mortality and morbidity attributable to malaria have declined significantly. To keep the momentum of fighting malaria, however, Ethiopia needs to strengthen its institutional capacity pertaining to domestic resource mobilization, diagnosis by microscopy, vector surveillance, and climate information processing and seasonal weather forecasting. These actions need, among others, to layout and enhance cross-sectoral coordination (e.g., with irrigation, hydropower, and climate change), and cross-border cooperation (e.g., for better surveillance of vectors) mechanisms. Future public budget allocation to fight malaria should factor in these and other emerging challenges.
Author Contributions
The author confirms being the sole contributor of this work and has approved it for publication.
Conflict of Interest
The author declares that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
2. Gallup JL, Sachs JD. The Economic Burden of Malaria. Am J Trop Med Hyg (2001) 64(1):85–96. doi: 10.4269/ajtmh.2001.64.85
3. Ayele DG, Zewotir TT, Mwambi HG. Prevalence and Risk Factors of Malaria in Ethiopia. Malaria J (2012) 11:195. doi: 10.1186/1475-2875-11-195
4. Ethiopian Public Health Institute. Services Availability and Readiness Assessment. Addis Ababa (2018).
5. Taffese HW, Hemming-Schroeder E, Koepfli C, Tesfaye G, Lee M, Kazura J, et al. Malaria Epidemiology and Interventions in Ethiopia From 2001 to 2016. Infect Dis Poverty (2018) 7:103. doi: 10.1186/s40249-018-0487-3
6. Misganaw A, Melaku YA, Tessema GA, Deribew A, Deribe K, Abera S, et al. National Disability-Adjusted Life Years (DALYs) for 257 Diseases and Injuries in Ethiopia, 1990–2015: Findings From the Global Burden of Disease Study 2015. Popul Health Metrics (2017) 15:28. doi: 10.1186/s12963-017-0146-0
8. Yalew AW. Revisiting Economic Burdens of Malaria in the Face of Climate Change: A Conceptual Analysis for Ethiopia. Int J Climate Change Strategies Manage (2021) 13(1):1–18. doi: 10.1108/IJCCSM-05-2020-0045
10. Pare G, Trudel M, Jaana M, Kitsiou S. Synthesizing Information Systems Knowledge: A Typology of Literature Reviews. Inf Manage (2015) 52:183–99. doi: 10.1016/j.im.2014.08.008
11. Cronin P, Ryan F, Coughlan M. Undertaking a Literature Review: A Step-by-Step Approach. Br J Nurs (2008) 17(1):38–43. doi: 10.12968/bjon.2008.17.1.28059
12. Green BN, Johnson CD, Adams A. Writing Narrative Literature Reviews for Peer-Reviewed Journals: Secrets of the Trade. J Chiropr Med (2006) 5(3):101–17. doi: 10.1016/S0899-3467(07)60142-6
13. Bugssa G, Tedla K. Feasibility of Malaria Elimination in Ethiopia. Ethiopian J Health Sci (2020) 30(4):607–14. doi: 10.4314/ejhs.v30i4.16
14. Ethiopian Public Health Institute. Ethiopian National Malaria Indicator Survey 2015. Addis Ababa (2016).
17. Ministry of Health. National Five-Year Strategic Plan for Malaria Prevention and Control in Ethiopia: 2006-2010. Addis Ababa (2006).
18. Ministry of Health. National Strategic Plan for Malaria Prevention, Control and Elimination in Ethiopia: 2011-2015. Addis Ababa (2010).
21. Ethiopian Health and Nutrition Research Institute. Ethiopia National Malaria Indicator Survey 2011. Addis Ababa (2012).
23. Mbanefo A, Kumar N. Evaluation of Malaria Diagnostic Methods as a Key for Successful Control and Elimination Programs. Trop Med Infect Dis (2020) 5:102. doi: 10.3390/tropicalmed5020102
29. Central Statistical Agency and ORC Macro. Ethiopia Demographic and Health Survey 2005. Addis Ababa (2006).
30. Roser M, Ritchie H. Malaria (2019). Available at: https://ourworldindata.org/malaria (Accessed November 17, 2021).
31. Girum T, Shumbej T, Shewangizaw M. Burden of Malaria in Ethiopia, 2000-2016: Findings From the Global Health Estimates 2016. Trop Dis Travel Med Vaccines (2019) 5:11. doi: 10.1186/s40794-019-0090-z
32. Deribew A, Dejene T, Kebede B, Tessema GA, Melaku YA, Misganaw A, et al. Incidence, Prevalence and Mortality Rates of Malaria in Ethiopia From 1990 to 2015: Analysis of the Global Burden of Diseases 2015. Malaria J (2017) 16:271. doi: 10.1186/s12936-017-1919-4
33. Center for Disease Control and Prevention. Malaria Diagnosis (United States) (2021). Available at: https://www.cdc.gov/malaria/diagnosis_treatment/diagnosis.html (Accessed November 15, 2021).
34. Haakenstad A, Harle AC, Tsakalos G, Micah AE, Dieleman JL. Tracking Spending on Malaria by Source in 106 Countries, 2000-16: An Economic Modelling Study. Lancet Infect Dis (2019) 19(7):703–16. doi: 10.1016/S1473-3099(19)30165-3
35. The President’s Malaria Initiative. Ethiopia Malaria Operational Plans (2021). Available at: https://www.pmi.gov/where-we-work/ethiopia/ (Accessed July 19, 2021).
36. Kendie FA, W/Kiros TH, Semegn EN, Ferede MW. Prevalence of Malaria Among Adults in Ethiopia: A Systematic Review and Meta-Analysis. J Trop Med (2021) 8863002:9. doi: 10.1155/2021/8863002
37. Tadesse FG, Ashine T, Teka H, Esayas E, Messenger LA, Chali W, et al. Anopheles Stephensi Mosquitoes as Vectors of Plasmodium Vivax and P. Falciparum, Horn of Africa, 2019. Emerg Infect Dis (2021) 27(2):603–8. doi: 10.3201/eid2702.200019
38. Balkew M, Mumba P, Dengela D, Yohannes G, Getachew D, Yared S, et al. Geographical Distribution of Anopheles Stephensi in Eastern Ethiopia. Parasit Vectors (2020) 13:135. doi: 10.1186/s13071-020-3904-y
39. Yewhalaw D, Legesse W, Van Bortel W, Gebre-Selassie S, Kloos H, Duchateau L, et al. Malaria and Water Resource Development: The Case of Gilgel-Gibe Hydroelectric Dam in Ethiopia. Malaria J (2009) 8:21. doi: 10.1186/1475-2875-8-21
40. Ersado L. Small-Scale Irrigation Dams, Agricultural Production, and Health: Theory and Evidence From Ethiopia. In: World Bank Policy Research Working Paper, World Bank, Washington, DC. (2005). p. 3494.
41. Kibret S, Wilson GG, Ryder D, Tekie H, Petros B. Malaria Impact of Large Dams at Different Eco-Epidemiological Settings in Ethiopia. Trop Med Health (2017) 45:4. doi: 10.1186/s41182-017-0044-y
42. Ministry of Health. National Health Adaptation Plan to Climate Change: 2018-2020. Addis Ababa (2018).
43. Lyon B, Dinku T, Raman A, Thomson MC. Temperature Suitability for Malaria Climbing the Ethiopian Highlands. Environ Res Lett (2017) 12:064015. doi: 10.1088/1748-9326/aa64e6
44. Taye G, Kaba M, Woyessa A, Deressa W, Simane B, Kumie A, et al. Modeling Effect of Climate Variability on Malaria in Ethiopia. Ethiopian J Health Dev (2015) 29(3):183–96.
45. Abrha H, Hagos H, Brhane E, Hadgu M, Mamo G. Spatio-Temporal Dynamics of Malaria Expansion Under Climate Change in Semi-Arid Areas of Ethiopia. Environ Hazards (2019) 18(5):400–13. doi: 10.1080/17477891.2019.1609405
46. World Health Organization. Climate and Health Country Profile: Ethiopia (2015). Available at: https://apps.who.int/iris/handle/10665/208861 (Accessed July 10, 2021).
47. Ghebreyesus TA, Tadesse Z, Jima D, Bekele E, Mihretie A, Yihdego YY, et al. Using Climate Information in the Health Sector. Field Actions Sci Rep (2009) 2(2):63–7. doi: 10.5194/facts-2-63-2009
Keywords: malaria risk, malaria control, malaria funding, health policy, Ethiopia
Citation: Yalew AW (2022) Achievements, Gaps, and Emerging Challenges in Controlling Malaria in Ethiopia. Front. Trop. Dis 2:771030. doi: 10.3389/fitd.2021.771030
Received: 05 September 2021; Accepted: 09 December 2021;
Published: 25 January 2022.
Edited by:
Son H. Nghiem, Griffith Health, Griffith University, AustraliaReviewed by:
Megha Raj Banjara, Tribhuvan University, NepalJun Feng, Shanghai Municipal Center for Disease Control and Prevention (SCDC), China
Copyright © 2022 Yalew. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Amsalu Woldie Yalew, YW1zYWx1d29sZGllLnlhbGV3QHVuaXZlLml0
 Amsalu Woldie Yalew
Amsalu Woldie Yalew