- 1Faculty of Natural Sciences, University of Stirling, Stirling, United Kingdom
- 2African Aquatic Conservation Fund, Joal, Senegal
- 3Convention on the Conservation of Migratory Species of Wild Animals Aquatic Wild Meat Working Group, Bonn, Germany
- 4Whale and Dolphin Conservation, Chippenham, United Kingdom
- 5Convention on the Conservation of Migratory Species of Wild Animals, Bonn, Germany
- 6Grupo de Ecología Animal, Universidad del Valle, Cali, Colombia
- 7Institute for Terrestrial and Aquatic Wildlife Research, University of Veterinary Medicine Hannover, Foundation, Hannover, Germany
- 8Global Conservation Program, Wildlife Conservation Society, Bronx, NY, United States
- 9Wild Earth Foundation, Puerto Pirámides, Argentina
- 10College of Science and Engineering, James Cook University, Townsville, QLD, Australia
- 11Institute of Environment, Florida International University, North Miami, FL, United States
- 12Pro Delphinus, Lima, Peru
- 13Centro Peruano de Estudios Cetológicos, Lima, Peru
- 14Benin Environment & Education Society, Cotonou, Benin
- 15Department of Environment, Parks and Water Security, Palmerston, NT, Australia
- 16OceanCare, Wädenswil, Switzerland
- 17Elasmo Project, Dubai, United Arab Emirates
- 18Gharial Ecology Project, Madras Crocodile Bank Trust, Mahabalipuram, India
- 19Crocodile Specialist Group, Wildlife Management International Pty. Limited, Karama, NT, Australia
- 20Southeast Asia Marine Mammal Research, Hong Kong, Hong Kong SAR, China
- 21Scientific Committee (Small Cetaceans) of the International Whaling Commission, Cambridge, United Kingdom
Wild animals are captured or taken opportunistically, and the meat, body parts, and/or eggs are consumed for local subsistence or used for traditional purposes to some extent across most of the world, particularly in the tropics and subtropics. The consumption of aquatic animals is widespread, in some places has been sustained for millennia, and can be an important source of nutrition, income, and cultural identity to communities. Yet, economic opportunities to exploit wildlife at higher levels have led to unsustainable exploitation of some species. In the literature, there has been limited focus on the exploitation of aquatic non-fish animals for food and other purposes. Understanding the scope and potential threat of aquatic wild meat exploitation is an important first step toward appropriate inclusion on the international policy and conservation management agenda. Here, we conduct a review of the literature, and present an overview of the contemporary use of aquatic megafauna (cetaceans, sirenians, chelonians, and crocodylians) in the global tropics and subtropics, for species listed on the Appendices of the Convention on the Conservation of Migratory Species of Wild Animals (CMS). We find that consumption of aquatic megafauna is widespread in coastal regions, although to varying degrees, and that some species are likely to be at risk from overexploitation, particularly riverine megafauna. Finally, we provide recommendations for CMS in the context of the mandate of the Aquatic Wild Meat Working Group.
Introduction
Globally, especially in the tropics and subtropics, wild animals are captured or taken opportunistically and the meat, body parts, and/or eggs are consumed as food or used for traditional remedies, cultural purposes, and religious ceremonies (Alves and Albuquerque, 2017; Ingram et al., 2021). Increasingly, products are sold locally, nationally, regionally, and internationally, providing income to many communities (Coad et al., 2019; Ingram, 2020). When harvests of wildlife are unsustainable, they cause population declines and pose a threat to species’ survival (McCauley et al., 2015; Benítez-López et al., 2017; He et al., 2017; Ripple et al., 2019). Over the past few decades, a vast literature has amassed on the harvest of wild-caught fish and terrestrial animals, which includes quantitative studies of the magnitude and spatial extent of harvests in some regions, sustainability assessments, the human dimensions of wildlife use, and the impact of overexploitation on ecosystems (Worm et al., 2009; Lynch et al., 2016; Coad et al., 2019; Dobson et al., 2019; Ingram et al., 2021). While the commercial targeting of some taxa has raised serious international concerns (e.g., whaling), there has been limited focus on the exploitation of most freshwater and marine (hereafter “aquatic”) non-fish1 animals used for food and other purposes.
For both terrestrial and aquatic wildlife, the terminology used to discuss harvest (also called “takes” in the wildlife management literature, and “catches” in the fisheries literature), consumption, and trade has changed over the past few decades. In 2000, and largely focussing on terrestrial species, the International Union for Conservation of Nature (IUCN) adopted the term “wild meat” to describe the “meat and other products derived from wild animals for human consumption,” whether legal or illegal, to harmonise terminology across regions (Mainka and Trivedi, 2002). In 2017, both the International Whaling Commission (IWC) and the Convention on the Conservation of Migratory Species of Wild Animals (CMS) adopted the term “aquatic wild meat,” defined as “products obtained through all forms of take, including unregulated, legal, or illegal hunts as well as deliberate or opportunistic catches from stranded (dead or alive) and/or bycaught (also known as incidental catch) individuals” to similarly harmonise this issue for aquatic non-fish species (CMS, 2017a,b; IWC, 2018).
The consumption of aquatic animals is widespread, and is an important source of nutrition, income, and cultural identity for many communities (Robards and Reeves, 2011). Considering aquatic wild meat as all aquatic non-fish vertebrates that are used by humans for food, medicine, or handicrafts, the IUCN Red List of Threatened Species identifies 672 species of birds, 322 amphibians, 137 reptiles, and 112 mammals as aquatic species that are used or traded (IUCN, 2021). Harvest levels may be high or low, but many are unregulated and unmanaged. Growing human populations, improved animal hunting and capture methods, expanded market access, and an escalating demand for animal-sourced proteins, has increased exploitation levels and, in some areas for at least some species, unsustainable harvests are now evident (Milner-Gulland et al., 2003; Cawthorn and Hoffman, 2015; Coad et al., 2019). Overarching guidance regarding aquatic wild meat in international policy remains limited for most species. Understanding the scope and potential threat of overexploitation of aquatic wild meat species is an important first step toward developing effective international and domestic policy.
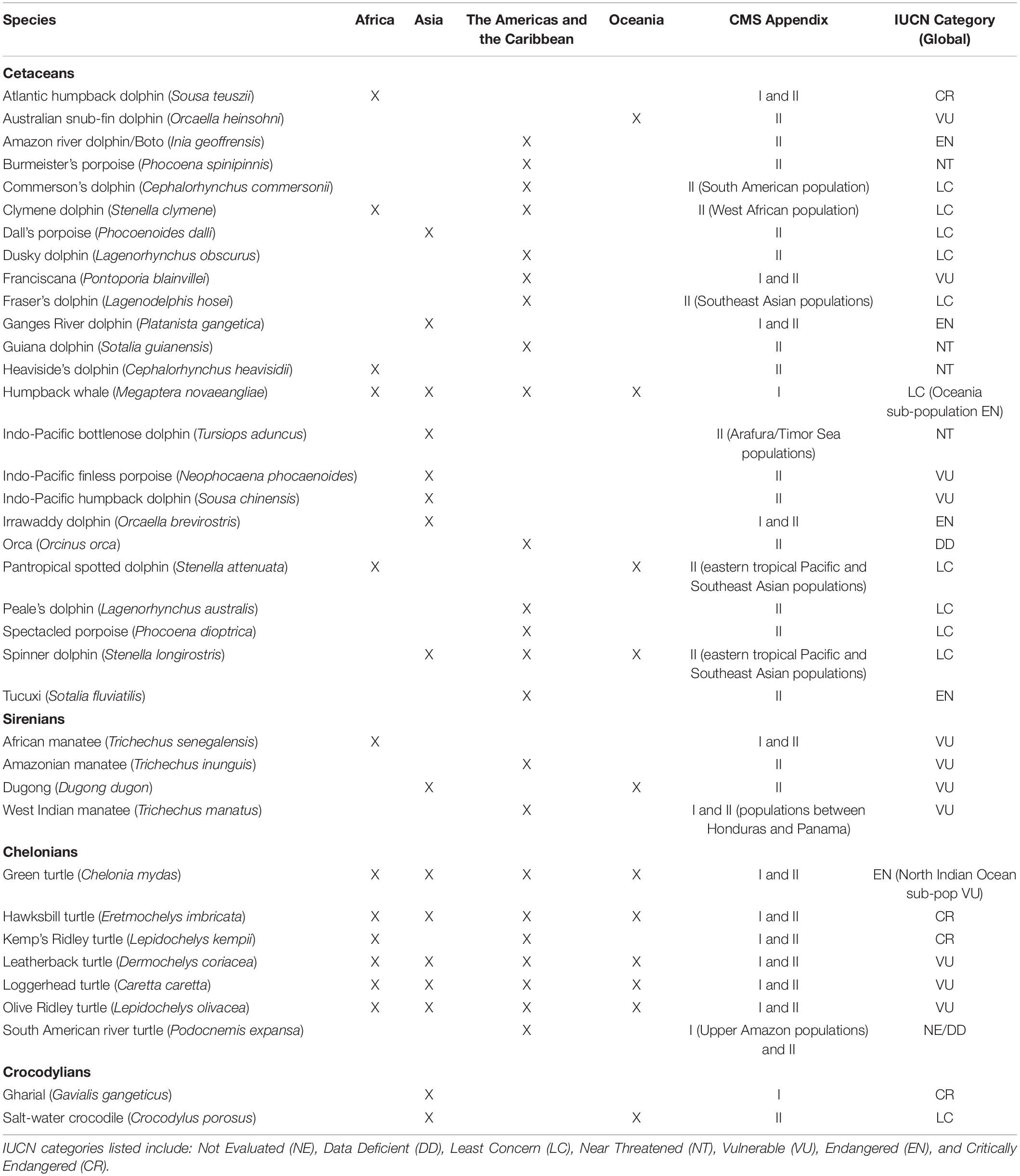
The CMS has provided some guidance on the use of migratory species, defined in the Convention text as “the entire population or any geographically separate part of the population of any species or lower taxon of wild animals, a significant proportion of whose members cyclically and predictably cross one or more national jurisdictional boundaries.” CMS provides a way for countries to cooperate to ensure species’ migrations can still continue without barriers, and to address issues such as habitat destruction and exploitation. As of 2020, 132 countries have become Parties to the CMS Convention and an additional 65 countries (non-Parties) are signatories to CMS Agreements and Memoranda of Understanding (Supplementary Material 1). In 2017, the CMS Scientific Council established an Aquatic Wild Meat Working Group to focus on the exploitation of aquatic animals (CMS, 2017b). We build here on information that was prepared for the establishment of the Working Group, and recognising the growing breadth and depth of this issue, present an overview of the use of aquatic mammals (specifically cetaceans and sirenians), turtles, and crocodylians in the tropics and subtropics (Figure 1).

Figure 1. Migratory megafauna used for aquatic wild meat, including: (A) Atlantic humpback dolphin (Sousa teuszii), Conkouati-Douli National Park, Republic of the Congo; (B) African Manatee (Trichechus senegalensis), Lagos Lagoon, Nigeria; (C) Green turtle (Chelonia mydas), Joal, Senegal; and (D) Saltwater crocodile (Crocodylus porosus) egg collection, Northern Territory, Australia. Photo credits: Tim Collins (A), Christogonus Uzoma Ejimadu (B), Pearson McGovern, African Aquatic Conservation Fund (C), and Yusuke Fukuda (D).
Many cultures have used aquatic megafauna throughout history (Robards and Reeves, 2011; Cosentino and Fisher, 2016; Porter and Lai, 2017). Here we focus on contemporary practises and the degree of conservation concern associated with them. Specifically, we examine 37 species listed on the CMS Appendices as endangered (CMS Appendix I) and/or requiring international cooperation for their conservation (CMS Appendix II and Table 1) that have been identified as being used for aquatic wild meat. This review is a first step toward increasing our understanding of aquatic wild meat use and its impacts, particularly of aquatic megafauna, and we emphasise the need for similar efforts for the hundreds of other species harvested for aquatic wild meat.
Aquatic Wild Meat Exploitation of Migratory Megafauna covered by the Convention on the Conservation of Migratory Species of Wild Animals
The Americas and the Caribbean
Cetaceans
The capture, killing, and trade of cetaceans and their products are illegal in most tropical American countries. Despite this, use for food and other purposes occurs in many countries, although the drivers and magnitude of the exploitation vary markedly between regions. In some countries (e.g., Argentina), uses are rare events and are almost exclusively opportunistic, while in others (e.g., Peru and Ecuador), there is continued use of cetaceans in spite of existing legislation, especially as bait in a number of fisheries (Van Waerebeek and Reyes, 1994; Vidal et al., 1994; Van Waerebeek et al., 1999, 2018; Avila et al., 2008; Crespo, 2009; Dawson, 2009; Flores and Da Silva, 2009; Goodall, 2009; Cosentino and Fisher, 2016; Campbell et al., 2020a,b; Castro et al., 2020). There are marked differences in the target species and methods used to capture them, both between and within countries. For example, botos (Inia geoffrensis) and tucuxis (Sotalia fluviatilis) are illegally harvested for use as bait for catfish (Calophysus macropterus) in Brazil, Peru, Colombia, and Venezuela (Flores et al., 2008; Trujillo et al., 2010; Mintzer et al., 2012; Campbell et al., 2020b) resulting in serious population declines (e.g., even in the more protected part of their range in Brazil; Martin and Da Silva, 2021), while dolphins off the coasts of Chile (Van Waerebeek et al., 1999) and Argentina are rarely recorded as taken incidentally or directly for either bait or human consumption. Common bottlenose dolphins (Tursiops truncatus) and the pantropical spotted dolphin (Stenella attenuata) have been hunted (at least 13 dolphins/year) for bait by local long-line fishers in the northern Colombian Pacific (Avila et al., 2008). In Chilean Patagonia, Peale’s dolphins (Lagenorhynchus australis) used to be hunted for crab bait, although changes in the dynamic of this fishery may have reduced their direct harvest for this purpose (Lescrauwaet and Gibbons, 1994; Goodall, 2009).
The largest exploitation of small cetaceans traditionally occurred in Peru, where they have been caught (intentionally and otherwise) in artisanal gillnets and by harpooning for decades, and sold in local markets or transported to the capital Lima (Read et al., 1988; Van Waerebeek and Reyes, 1994; Van Waerebeek et al., 1999). The use of dolphins as bait has spread to all coastal areas, with annual catches increasing since the early 2000s (Mangel et al., 2010; Tzika et al., 2010; Campbell et al., 2020a). The use of small cetaceans as bait, including dusky (Lagenorhynchus obscurus), common (Delphinus delphis), common bottlenose dolphins, as well as Burmeister’s porpoises (Phocoena spinipinnis) (e.g., in longline and gillnet shark fisheries in coastal Peru) appears prolific and has now largely replaced use for human consumption (Van Waerebeek et al., 1999, 2018; Van Waerebeek and Würsig, 2009; Mangel et al., 2010; Campbell et al., 2020a). Nonetheless, dolphin meat was still widely available in Peruvian fish markets until at least 2010, but increasingly covertly (Mangel et al., 2010; Tzika et al., 2010). A study of 942 beach-cast small cetaceans between 2000 and 2017 showed that use still continues in central and southern Peru, although the high level of stranded bycaught animals that do not show signs of butchering suggests it may occur on a significantly lower scale compared to what was observed in the 1980s and 1990s (Van Waerebeek et al., 1999, 2018).
The use of small cetaceans in other tropical American countries is less widespread. Cetacean harvests were reported for Venezuela 20 years ago (Romero et al., 2001), and exploitation for meat and bait has been more widespread than commonly thought (affecting 11 of 20 species), though it is poorly documented (Romero et al., 1997). Recent data are scarce, but Sotalia guianensis has been confirmed as a target species (Barrios-Garrido et al., 2021; Briceño et al., 2021). Recent studies in Ecuador have revealed the use of the CMS-listed Eastern Tropical Pacific (ETP) population of pantropical spotted dolphins as bait (Castro et al., 2020). In Ecuador, fishers use body parts of aquatic mammals, including dolphins, as bait in improvised Fish Aggregating Devices (FADs; Castro et al., 2020). Several species are involved in this illegal practice, caught both accidentally and via direct targeting, but the extent of harvest is unknown. CMS-listed species of concern include botos, Guiana dolphins, dusky dolphins, Burmeister’s porpoise, and common bottlenose dolphins.
Captures of several species of cetaceans have been documented throughout the Caribbean including in the Dominican Republic, St Vincent and the Grenadines (SVG), Trinidad and Tobago, St Lucia, and Dominica (Vidal et al., 1994; Bolaños-Jiménez et al., 2014). CMS-listed species harvested include Fraser’s (Lagenodelphis hosei), spinner (Stenella longirostris), common (Delphinus delphis), and clymene dolphins (S. clymene), orcas (Orcinus orca), and humpback whales (Megaptera novaeangliae). Except for SVG, direct harvesting for large whales, such as humpback whales, is scarce in tropical America (Williams-Grey et al., 2014). However, there are some recent records of opportunistic hunting, for example in 2004, in the Colombian Pacific, a humpback whale calf, which was alone and apparently lost, was killed by artisanal fishermen for consumption purposes (Avila et al., 2013). In Barrouallie in SVG, cetaceans are regularly hunted, with records of between 203 and 927 individuals harvested each year by a single whaler (Fielding and Kiszka, 2021). In 2019, reports presented to the Scientific Committee of the IWC raised concerns as it was estimated that the current levels of hunting of short-finned pilot whales (Globicephala macrorhynchus) and orcas in the waters of SVG were unsustainable (Fielding, 2018; IWC, 2019).
Sirenians
Manatees have been historically hunted throughout their range in the Americas and the Caribbean. Direct harvest of the Amazonian (Trichechus inunguis) and the West Indian manatees (T. manatus) have been recorded in at least 14 countries. Both species are mainly used for food, but also for other purposes, including remedies and traditional ceremonies (Braga Ferreira et al., 2011; Cosentino and Fisher, 2016). Although most manatees are harvested opportunistically (e.g., as bycatch), manatee hunters may still be active in Brazil (Souza et al., 2014), Colombia (Arévalo-González et al., 2010; Cruz-Antía and Gómez, 2011; Kiszka, 2014), Ecuador (Denkinger, 2010; Tirira, 2011), and Peru, where Amazonian manatee calves are also captured alive to be “grown” in ponds for food (Perea-Sicchar et al., 2011).
In the past, Amazonian manatees have experienced high hunting pressure. In the central-western Brazilian Amazon, trade in manatee hides led to the hunting of ∼113,000 animals between 1904 and 1969 (Antunes et al., 2016). This practice no longer occurs (Antunes et al., 2016), but illegal hunting for meat, largely conducted with traditional harpoons, is considered among the main contemporary threats to the species (Marmontel et al., 2016), and occurs throughout the species’ range: Brazil (Calvimontes, 2009; Franzini et al., 2013; Souza et al., 2014; Pantoja, 2015; Crema et al., 2020); Colombia (Kendall, 2013), Peru (Soto, 2007; Kendall, 2013; Kirkland et al., 2020); and Ecuador (Denkinger, 2010). Meat is consumed or sold locally (Marmontel et al., 2016), but can also be marketed in larger towns and cities (e.g., Franzini et al., 2013; Souza, 2015; El Bizri et al., 2020). Although not considered a substantial threat to the species, recent uses of Amazonian manatee for traditional remedies have been reported in the Caatinga region of Brazil by Barbosa et al. (2018). Although Amazonian manatee is not reported to occur in this region, it is plausible that these reports refer to the West Indian manatee. Changes to water levels as a result of dam construction is amplifying hunting impacts on the Amazonian manatee because migrating animals become more available to hunters in narrow passages along migration routes (Arraut et al., 2010, 2017).
West Indian manatees are also hunted opportunistically (e.g., as bycatch in fishing nets) and directly targeted throughout the Caribbean, in the Magdalena and Orinoco River systems, and the Maracaibo Lake system (Montiel-Villalobos and Barrios-Garrido, 2005; Romero and Creswell, 2005; Vail, 2005; Castelblanco-Martínez et al., 2009, 2016). They were hunted historically for oil, bones, and meat (e.g., O’Shea et al., 1988), but today hunting is exclusively for meat, a practice considered a significant threat in Brazil, Colombia, Costa Rica, Cuba, Dominican Republic, French Guiana, Guatemala, Honduras, Mexico, Suriname, Trinidad, and Venezuela (Self-Sullivan and Mignucci-Giannoni, 2012; Alvarez-Alemán et al., 2021). Killing of bycaught animals for consumption occurs in Colombia, Dominican Republic and Haiti (Kiszka, 2014). Although numbers of manatees killed by hunting in Panama are low, it still occurs (Muschett, 2008; Quintana-Rizzo and Reynolds, 2010). West Indian manatees in Colombia have been killed and sold locally for food or bait, mainly via direct hunting, for decades (Montoya-Ospina et al., 2001; Castelblanco-Martínez et al., 2009).
Chelonians
All six marine turtle species in the Americas and the Caribbean are considered threatened according to the IUCN Red List of Threatened Species (green turtle, Chelonia mydas, hawksbill turtle, Eretmochelys imbricata, Kemp ridley turtle, Lepidochelys kempii, leatherback turtle, Dermochelys coriacea, loggerhead turtle, Caretta caretta, and olive ridley turtle, Lepidochelys olivacea; IUCN, 2021; Table 1). While the threats vary by species, exploitation for meat, eggs, and carapaces (e.g., hawksbill turtles) at varying levels is ubiquitous and considered a significant threat (Amorocho et al., 2016; Nahill et al., 2020).
In the Caribbean, marine turtles have been harvested by people continuously, with commercial exploitation and trade from Cuba for 500 years. Marine turtle harvests were managed as a commercial fisheries resource in Cuba (unique for the region) until recently (Carrillo et al., 1999; Fleming, 2001), and some level of regulation of harvests occurred in other countries: Cayman Islands (Aiken et al., 2001; Bell et al., 2006), Turks and Caicos (Richardson et al., 2009), the British Virgin Islands, Anguilla and Montserrat (Richardson et al., 2006), and Nicaragua (Garland and Carthy, 2010). Contemporarily, marine turtle bycatch and direct harvest for subsistence (mainly meat and medicine) and domestic commercial purposes (mainly eggs, medicine, and hawksbill shell products) still occurs in parts of Brazil (Guebert et al., 2013), Costa Rica (Chacón-Chaverri and Eckert, 2007; Mejías-Balsalobre et al., 2021), Colombia and Venezuela (Barrios-Garrido et al., 2018), Mexico (Mancini and Koch, 2009; Senko et al., 2014), Nicaragua (Garland and Carthy, 2010; Lagueux et al., 2014, 2017; Ankersen et al., 2015), and several Caribbean islands (Richardson et al., 2006, 2009; Amorocho et al., 2016). Together, the Caribbean has one of the highest global legal marine turtle harvests. Nicaragua accounted for 22.3% of the reported global legal harvest of marine turtles in 2013, with an estimated 9,413 marine turtles harvested per year, mostly green turtles (Humber et al., 2014). However, the full extent of the global harvest is difficult to estimate because many uses by Indigenous Peoples and Local Communities (IPLCs) are not reported (but see Barrios-Garrido H.A. et al., 2020). The Turks and Caicos Islands has the largest documented legal hawksbill turtle fishery in the western Atlantic, with approximately 114–277 hawksbill turtles harvested annually, as well as 176–324 green turtles (Stringell et al., 2013).
In Colombia, Nicaragua, and Panama, use and trade in turtle meat, eggs, and handicrafts (from hawksbill shell plates) is illegal, but reasonably common. It mainly satisfies domestic consumption demand (e.g., Chacón, 2002), but with some international flow between countries in the region (e.g., Costa Rica, Ecuador, El Salvador, Guyana, Honduras, Mexico, Suriname, and Venezuela), especially for artefacts made from hawksbill shell plates (Pilcher and Williams, 2018). In Mexico, marine turtles have been exploited as a traditional activity for centuries in Baja California (Caldwell, 1963; Nabhan, 2003) and along the eastern Pacific coast (Webb and Carrillo, 2000), despite their fully protected status. Here, turtle hides are still locally cured for leather, for use as cowboy boots (Kemf et al., 2000). During the commercial meat fisheries of olive ridleys in Mexico, in the 1970s and 1980s, turtle skin for leather was a significant byproduct traded internationally (WWF, 2004). Throughout the Caribbean, current harvests on nesting beaches, for meat, oil, and eggs, are reported as contributing to declines in nesting populations or constraining recovery: Grenada and Guyana (Eckert, 2001), Dominican Republic (WWF, 2004), Belize (Meerman, 2005), and Cuba (Amorocho et al., 2016). Tortuguero, Costa Rica, hosts the largest recovered green turtle rookery in the Atlantic basin (Troëng and Rankin, 2005). In the Guianas, green turtle status is adversely affected by illegal harvest of eggs and nesting females, and by bycatch from near and off-shore fisheries (Amorocho et al., 2016). Relatively high rates of marine turtle bycatch also occur in Suriname fisheries (Amorocho et al., 2016). Prolific historical commercial exploitation devastated the abundance of nesting turtles in the British Virgin Islands (Richardson et al., 2006), Jamaica (Amorocho et al., 2016), and the Cayman Islands (Fleming, 2001). While population recoveries have occurred in recent years, the abundance of nesting turtles remains a small fraction of what existed historically (Blumenthal et al., 2021). That the unregulated, historical, large-scale commercial harvest was demonstrably unsustainable is clear. The impact on recovering populations of the widespread low level of use occurring now, and whether they constitute serious threats to turtle populations, remains largely unquantified.
In spite of protective legislation, marine turtle eggs are still collected for subsistence and commercial use (Eckert, 2001). For example, signs of illegal egg harvesting were observed on 51.1% of surveys in Tortuguero, Costa Rica (González and Harrison, 2012). However, nesting at the green turtle rookery increased by 417% between 1971 and 2003 (Troëng and Rankin, 2005). Caribbean Nicaraguans have legally harvested green turtles for more than 400 years, and continue to rely on the species as an available and inexpensive source of protein and income. Over 8,000 green turtles are harvested annually along the Miskito coast of Nicaragua (Lagueux et al., 2014, 2017), and current rates of harvest may be unsustainable (Campbell and Lagueux, 2005; Lagueux et al., 2005). How the increases in green turtle nesting at Tortuguero in Costa Rica are related to the turtle harvest in adjoining Nicaragua is largely unknown. Indeed, research is needed on harvested and source populations of green turtles in the Caribbean (Garland and Carthy, 2010).
In the Guajira Peninsula (between Colombia and Venezuela), the Wayuu indigenous people consider marine turtles the most important animal sustaining their culture, and today, there is a significant subsistence and commercial harvest of green and hawksbill turtles by the Wayuu community (Barrios-Garrido, 2012; Barrios-Garrido et al., 2017, 2018; Barrios-Garrido H.A. et al., 2020). Barrios-Garrido H.A. et al. (2020) estimated 3,402 ± 247 turtles/year were extracted in the study area (10 ports). Harvests occur in Colombia and Venezuela, where the legal framework is unclear, for both domestic and international trade (Bräutigam and Eckert, 2006; Barrios-Garrido et al., 2017). In Peru, turtle consumption has been historically important in the Pisco-San Andres area, for example in 1987, up to 110 fishing boats were recorded landing turtles in San Andrés, the highest level of marine turtle exploitation recorded in Peru (Quiñones et al., 2010). Marine turtle harvest was a legal activity until 1995, when it was completely banned, for all species, by Peruvian legislation (Morales and Vargas, 1996). However, the exploitation continues in Peru’s coastal waters, as bycaught and targeted harvesting (Quiñones et al., 2017; Alfaro-Shigueto et al., 2018). In Brazil, marine turtles were exploited historically, until it was banned in 1986 (Marcovaldi and dei Marcovaldi, 1999). Illegal harvesting still occurs in some coastal areas (Guebert et al., 2013).
Inland, the South American giant river turtle (Podocnemis expansa) is harvested for meat, eggs, and oil in the Amazon, Orinoco and Essequibo basins (Tortoise and Freshwater Turtle Specialist Group, 1996). The species has been commercially traded since at least the 1800s (Forero-Medina et al., 2021). Current harvests occur in Brazil (e.g., Lopes et al., 2012), Bolivia (Conway-Gómez, 2007), Colombia (Gómez et al., 2016), Ecuador (De la Montaña, 2013), Peru (Pineda-Catalan et al., 2012), and Venezuela (Peñaloza et al., 2013). The harvests are mainly for meat, an important source of protein for remote communities, but also for traditional remedies (Gómez et al., 2016). Trade is largely domestic, from rural to urban areas (e.g., in Peru; Pineda-Catalan et al., 2012). Despite the harvest being sustained over time, it is considered likely that wild populations have declined relative to historical abundance, but quantifying status – current abundance relative to historical abundance – is logistically challenging (Mogollones et al., 2010; Forero-Medina et al., 2021). The current Red List assessment (Least Concern/Conservation Dependent), may need to be reviewed if additional data on declines become available (Tortoise and Freshwater Turtle Specialist Group, 1996).
Although demand for chelonian meat and eggs remains strong throughout the tropical Americas and the Caribbean, conservation successes based on the implementation of national management programmes are evident. For example, the Kemp’s ridley turtle was seriously depleted and near extinction in the second half of the 20th century. After more than two decades of protection, and some management interventions, an exponential increase of adult females on nesting beaches in Mexico occurred (Webb and Carrillo, 2000; WWF, 2004; Shaver and Rubio, 2008), however, this has declined in recent years (Bevan et al., 2016). In Costa Rica, a managed olive ridley egg harvest programme was introduced at Ostional in 1987. It is a legal, commercial harvest, run by the local people through a community cooperative, which has been sustained and benefits both turtles (through increased stewardship) and local people (Campbell et al., 2007). After 35 years of sustainable operation, it provides a unique opportunity to re-examine sustainable harvest rates. There is no evidence that it has stimulated a nationwide market for eggs illegally extracted outside Ostional (Sardeshpande and MacMillan, 2019), although such possibilities are difficult to test (but see Pheasey et al., 2020; Mejías-Balsalobre et al., 2021). In Brazil, Projeto TAMAR-IBAMA (Tartarugas Marinhas) involves a marine turtle conservation programme initiated in 1980, which reduced the harvest of gravid nesting females and their eggs in all major nesting areas (Marcovaldi and dei Marcovaldi, 1999). Local participation of the fishing villages, including the employment of former egg harvesters to patrol the beaches and protect the nests, along with education programmes and ecotourism, have all contributed to the programme’s success.
Commitments to chelonian conservation generally in the Americas and Caribbean have been strengthened under several international policy instruments (although challenges still exist, e.g., Barrios-Garrido et al., 2019), for example: the banning of international trade under the Convention on International Trade in Endangered Species of Wild Fauna and Flora (CITES); bycatch reduction under the Inter-American Tropical Tuna Commission (IATTC); adaptation to climate change under the Inter-American Sea Turtle Convention (IAC); and the activities of the Permanent Commission for the South Pacific and the Protocol Concerning Specially Protected Areas and Wildlife (SPAW).
Africa
Cetaceans
There is evidence of the use of cetaceans in most countries in tropical Africa, with meat and other body parts used for human consumption, shark bait, traditional medicine, and other purposes (Van Waerebeek et al., 2003; Clapham and Van Waerebeek, 2007; Weir et al., 2010; Robards and Reeves, 2011; Weir and Pierce, 2012; Cosentino and Fisher, 2016). Dolphins are both intentionally hunted and landed as bycatch in artisanal gillnets, drift gillnets, beach seines, and other fishing gear.
The available data from West and Central Africa are limited, but recent records indicate small cetaceans are consumed as food in Benin (Sohou et al., 2013), Cameroon (Ayissi et al., 2011, 2014), Guinea (especially Atlantic humpback dolphin, Sousa teuszii, and common bottlenose dolphin; Van Waerebeek et al., 2003, 2017; Bamy et al., 2021), Guinea-Bissau (Leeney et al., 2015), Nigeria (Uwagbae and Van Waerebeek, 2010; Van Waerebeek et al., 2017), Republic of the Congo (Collins et al., 2010, 2019), Democratic Republic of the Congo (Collins et al., 2019), Senegal and The Gambia (Maigret, 1994; Murphy et al., 1997; Van Waerebeek et al., 2000, 2003; Leeney et al., 2015; Keith-Diagne et al., 2017), and Togo (Segniagbeto et al., 2014). Historically whaling was important on the Cape Verde Islands (Brito et al., 2016), and some consumption of cetacean meat remains. However, while a recent review (Segniagbeto et al., 2019) indicates instances of consumption of pilot whales and stranded melon-headed whales, and use of teeth for manufacture of local jewellery, there are no signs of any systematic utilisation on the Cape Verde Islands. This conclusion coincides with Hazevoet et al. (2010) who stated that only few bycatches or purposeful catches had come to their attention. Recent evidence also suggests that opportunistic harvest of whales occurs on Annobón (Equatorial Guinea), although rarely, and includes large cetaceans (preferentially calves), such as humpback whales (Fielding and Barrientos, 2021), and possibly São Tomé and Príncipe (Brito et al., 2010). In Ghana at least 16 cetacean species are used as aquatic wild meat and, in some years, more than one thousand individuals are landed (Ofori-Danson et al., 2003; Van Waerebeek et al., 2009, 2014; Debrah et al., 2010). In some countries, including Ghana, as demand increased for dolphin meat, for human consumption or shark bait, bycatch gradually transformed into targeted harvesting (Ofori-Danson et al., 2003). Ghanaian artisanal fishers, operating in Togolese coastal waters, are thought to promote trade and consumption of cetacean meat (Segniagbeto et al., 2014). Smoked cetacean meat from coastal Togo is traded far from the coast as wild meat in northern Togo, Burkina Faso, Niger, and Mali (Segniagbeto et al., 2014). In the western most countries, The Gambia and Senegal, dolphin meat and oil is also used in traditional remedies (Madge, 1998; Leeney et al., 2015). It is likely that cetaceans are consumed throughout the Gulf of Guinea, despite the lack of specific records (Clapham and Van Waerebeek, 2007; Collins et al., 2010; Robards and Reeves, 2011; Van Waerebeek et al., 2017). The Atlantic humpback dolphin is considered disproportionately impacted by wild meat harvest, because of its small population size (IWC, 2019), inshore habitat use, and high vulnerability to capture in small-scale coastal fisheries (Van Waerebeek et al., 2017; Bamy et al., 2021).
In Eastern Africa, there are a number of records of opportunistic use, bycatch, and intentional harvests of dolphins. In Mozambique, there is concern that increasing intentional harvest has evolved from commercial use of bycaught animals (Guissamulo and Cockcroft, 1997). Stranded cetaceans here are also used for meat (Reeve-Arnold et al., 2020). Bycatch and direct harvest, possibly for shark bait, have been reported on the Kenyan coastline (Mwango’mbe et al., 2021), and for human consumption and bait in Tanzania (Braulik et al., 2018). In Zanzibar, stranded large cetaceans are utilised as food when fresh, and when decomposed, for the extraction of oil and for use in traditional remedies. Small cetaceans are largely used as bait, rather than for human consumption or other uses (Amir et al., 2012). Direct harvests of dolphins have been reported historically around the Seychelles islands group (De Lestang, 1993), although no recent data confirm this is ongoing. Direct exploitation of small cetaceans is regularly reported from the western coast of Madagascar, mostly Indian Ocean humpback dolphin (Sousa plumbea), bottlenose dolphins (likely Tursiops truncatus), and spinner dolphins (Andrianarivelo, 2001; Razafindrakoto et al., 2004; Cerchio et al., 2009a,b; Temple et al., 2019). Between 2000 and 2018, more than 2,750 small cetaceans, comprising at least seven species, were intentionally caught or bycaught by 10 fishing communities in southern Madagascar (Andrianarivelo et al., 2019).
Sirenians
African manatees (Trichechus senegalensis), distributed exclusively in West and Central Africa, are legally protected in all 21 countries in which they occur. Domestic trade for food, traditional medicine, and other purposes has been recorded historically and continues to some degree in all countries (Reeves et al., 1988; Powell, 1996; Akoi, 2004; Dodman et al., 2008; Keith Diagne, 2014, 2015; Bachand et al., 2015; Mayaka et al., 2015, 2019; Kamla, 2019). In the past, manatee hunting was conducted by specialised hunters, but more recently also by fishers and generalist hunters. Consumption of bycaught animals also occurs widely in both coastal and inland regions, largely driven by poverty (Dodman et al., 2008; Bachand et al., 2015; Keith Diagne, 2015; Kamla, 2019). Manatee meat is also sold in markets and restaurants in urban centres, which has increased its commercial value (Thibault and Blaney, 2003; Fa et al., 2006; Mvele and Arrowood, 2013).
The eastern coast of the African mainland and nearby islands north of 25oS are the south-western extreme of the distribution range of the dugong (Dugong dugon). Historical dugong hunting with spears and nets has been recorded in Kenya and Somalia (Husar, 1975), and probably occurred throughout East Africa (Muir and Kiszka, 2012). Dugong hunting is banned in most African countries, and dugongs are now rarely hunted due to population declines. Poor enforcement enables meat to be sold from dugongs that are caught incidentally (Marsh et al., 2011) and recent evidence indicates occasional dugong hunting still occurs in Kenya, Tanzania, and Mozambique (Muir and Kiszka, 2012). Some Kenyan and Tanzanian coastal communities use dugong meat, bones, and oil for food, medicine, and ornamental purposes (Wamukoya et al., 1995; Marsh et al., 2002, 2011; Muir and Kiszka, 2012; Said et al., 2020). In Bazaruto Bay, Mozambique, which supports the most important dugong population in the region (Marsh et al., 2011), the illegal directed harvest of dugongs is estimated at 4–6 dugongs per year (Findlay et al., 2011). Bycaught animals are sold, albeit illegally, because they represent a windfall for impoverished fishers (Marsh et al., 2011). In northern Madagascar, hunting for meat is thought to have been intense historically (Durbin and Ralambo, 1994), although is considered to have declined in recent decades due to dugong population decline (Humber et al., 2015). Hunting probably still occurs along the west coast of Madagascar (Cerchio et al., 2012), Comoros, and the Department of Mayotte (French territory) (Muir and Kiszka, 2012).
Chelonians
Marine turtles are also harvested to different degrees for food and traditional remedies along the West and Central African coast, including The Gambia, Gabon, Guinea-Bissau, Sierra Leone, Ghana, Togo, Mauritania, Benin, Cape Verde, Senegal, Côte d’Ivoire, Guinea, and the Republic of Congo (CMS, 2000; Thibault and Blaney, 2003; Bal et al., 2007; Fretey et al., 2007; Catry et al., 2009; Hancock et al., 2017). Marine turtle exploitation (both illegal, and legal for Urekan people permitted with a quota) is particularly intensive on Bioko Island, Equatorial Guinea (Lepidochelys olivacea, Chelonia mydas, Dermochelys coriacea, Eretmochelys imbricata), with products sold at local and urban markets (Tomás et al., 2010), São Tomé and Príncipe (C. mydas, L. olivacea; Veríssimo et al., 2020), and Senegal (McGovern et al., 2021). For example, in São Tomé and Príncipe, 25% of rural and 32% of urban respondents in a survey had consumed marine turtle meat in the past year (Veríssimo et al., 2020). Catch of marine turtles in Nigeria is estimated to be in the thousands of individuals annually, and high numbers of turtle eggs are harvested (Lewison and Moore, 2012). Reductions in turtle exploitation in Bioko Island occurred in years when beach patrols were implemented, but this requires consistent funding (Tomás et al., 2010).
Harvest of marine turtles and their eggs is also prevalent in coastal East Africa. In Kenya, it has been estimated that 10–50 percent of nesting turtles and their eggs are harvested to supply underground markets (Nzuki, 2004). About 10 percent of turtle products available in the Tana Delta and Malindi are thought to have been supplied by foreign fishers, mostly from Somalia and Tanzania (Nzuki, 2005). In Tanzania, turtle products were being sold both openly and covertly at landing sites in Dar es Salaam (West, 2008), and two assessments (2015 and 2016) conducted in the Lindi Region confirmed a directed fishery was capturing green turtles for both local consumption and commercial trade (IOSEA, 2019). Egg collection persists in Tanzania, although on Mafia Island, nest monitoring, nest protection payments, and education programs about sea turtle conservation resulted in egg harvest declining from 100% of eggs (prior to programme initiation in 2001) to 1% (2004) and 4% (2005) (Ferraro, 2007). In Mozambique, marine turtles are caught by specialist turtle hunters and by some artisanal fishers (Williams, 2017, 2021; Pilcher and Williams, 2018). Retained turtles are most commonly consumed domestically for meat, occasionally for local remedies/cultural practises, or as bait for fishing. Trade in turtle meat is often clandestine, but turtle shells are readily available in craft markets (>200 products observed between 2017 and 2020 [exact survey period unknown]; Nahill et al., 2020). Surveys across three coastal provinces in Mozambique, although crude, suggested the directed and bycatch harvest of turtles could be as high as 800,000 turtles per year, of which ~200,000 may be consumed or traded and many are reportedly released alive (Pilcher and Williams, 2018).
Illegal domestic use and trade of marine turtles is prevalent in Madagascar, particularly along the west coast, where hawksbill and green turtles (live, meat, eggs, and plastron/ligaments) can be commercialised in other parts of the island including the capital city, Antananarivo (Muttenzer, 2007; Gough et al., 2009; Rakotondrazafy and Andrianasolo, 2012; Pilcher and Williams, 2018). Approximately 52% of fishers stated that they targeted marine turtles (Pilcher and Williams, 2018), in spite of national decrees prohibiting all exploitation (IOSEA, 2014, 2019). A new illegal trade network was uncovered in north-western Madagascar in 2012, supplying traders in Mahajanga with turtles, although the final destination was not established (IOSEA, 2014). In total, it is estimated that up to 100,000 turtles/year (mostly green turtles) are taken by Madagascan fishers in two areas alone, Toliara and Antsiranana (Pilcher and Williams, 2018). In Comoros, turtle hunting is reportedly widespread in the Moheli Marine Park (Moheli Marine Park, 2009), Grande Comore, and Anjouan (IOSEA, 2019).
The town of Āssab, Eritrea, has a well-known market for turtle products which it trades locally as well as with Yemen (IOSEA, 2014). Use of marine turtle meat and harvest of eggs is also reported from Mauritius, Seychelles, South Africa, and Sudan (IOSEA, 2019).
Asia
Cetaceans
Robards and Reeves (2011) provided the first systematic documentation of global marine mammal consumption, collating data from 1970 to 2009. It demonstrated that for some Asian cultural groups, the use of marine mammals for food, and likely for other purposes, is higher than ever before. Asia’s extensive coastline and large riverine systems are densely populated and communities rely heavily on aquatic resources. The review highlighted an increase in bycatch and showed that in some areas in Asia, bycatch has developed into deliberate catches and the harvest of small cetaceans has become commercialised. Although harming, harassing, killing (when bycaught alive), and hunting of marine mammals is illegal in most Asian countries, indigenous and subsistence use still occurs in many areas, at varying levels (Porter and Lai, 2017; Lee et al., 2019).
Both legal and illegal use and sale of CMS-listed cetaceans occurs in East Asia (here including China and Taiwan, but not Japan and South Korea). Most species are used for human consumption, but some for bait in fisheries (Mintzer et al., 2018). In Taiwan, despite the sale of bycaught cetaceans being illegal, the meat of hundreds of bycaught individuals was once sold in local markets (Sui, 2014), however, recent advances in forensic surveillance have since curtailed most trade (Lo et al., 2013). Information for China is anecdotal and limited, e.g., Jefferson and Van Waerebeek (2002) presented photographic evidence of bycaught common dolphins landed at fish markets of Putian, Fujian Province. Wang et al. (2015) presented records of 193 incidents of bycatch, stranding and injuries (mostly fisheries inflicted) of aquatic mammals nationwide in 2000 to 2006, but did not report on subsequent use of carcasses. A short study on the use of Chinese social media and national online news services to monitor cetacean trade in fish markets in China indicated that the sale of cetaceans for food, particularly finless porpoise (Neophocaena spp.), is not uncommon and that some trade occurs far inland (IWC, 2016). As elsewhere, the lack of a nationwide reporting scheme and the poorly known status of most populations of aquatic mammals in China, hampers our understanding of the impact of both targeted and incidental takes (Wang et al., 2015).
In Southeast Asia, cetaceans have been used both historically, and still today, for food and other uses (e.g., Tun, 2006; Porter and Lai, 2017; Altherr and Hodgins, 2018). In Indonesia, traditional sperm whale (Physeter macrocephalus) harvests have a long history, and are restricted to two specific villages, Lamalera and Lamakera (Leatherwood et al., 1991). These hunts have, at times, also targeted blackfish (Orcinus orca, Globicephala macrorhynchus). The hunting of small cetaceans by these villages is a relatively new practice, but it is believed to be rapidly increasing, possibly because of declining fish stocks (Cerchio et al., 2019). Data on catches from non-discriminant fisheries, such as “experimental nets” or “tiger nets,” indicate that single net sets may have bycaught thousands of cetaceans, the meat of which is subsequently used for human consumption and the pet food industry (CMS, 2015). In the Philippines, many villages have a long history of hunting cetaceans for bait but also, within the last two decades, some local communities have developed a taste for dolphin meat, leading to deliberate harvests to meet this new demand (Dolar et al., 1994; Dolar, 1999). In the southern parts of the Philippines, at least one indigenous group is known to consume dolphin meat as part of their traditional diet and to use other cetacean products for ceremonies, including wedding dowries. In Cambodia and Viet Nam, it is commonly believed that catching a dolphin brings bad luck, thus in general, dolphin exploitation is rare or non-existent (Perrin et al., 2002; Beasley and Davidson, 2007). Historically, coastal Irrawaddy dolphins (Orcaella brevirostris) were exploited for their oil in both countries (Perrin et al., 2002).
Throughout many parts of South Asia, recent data suggest cetacean meat is exploited commercially for human consumption, particularly west India, especially the Indo-Pacific finless porpoise (N. phocaenoides) and Indo-Pacific bottlenose (Tursiops aduncus; Kumarran, 2012). A few species, notably the Indo-Pacific finless porpoise, are hunted for a niche market along the west coast of India and there is an open market for cetacean meat in several locations throughout the states of Goa, Karnataka, and Kerala (Kumarran, 2012). In the town of Malpe alone, an estimated 2,000 cetaceans are caught for human consumption every year (Kumarran, 2012). In India and Bangladesh, Ganges river dolphins (Platanista gangetica) that become entangled in nylon gill-nets were sometimes killed by fishers to extract oil to be used as a fish attractant (Smith and Smith, 1998; Sinha, 2002). In Pakistan, the meat of Indian Ocean humpback, Indo-Pacific bottlenose and spinner dolphins, and finless porpoise is used for shark bait, human food, and traditional remedies (Gore et al., 2012; Kiani and Van Waerebeek, 2015). In the south coast of Sri Lanka, small cetaceans are hunted using hand-held harpoons and this practice has spread to western areas driven by the increasing demand for dolphin meat from inland and urban people (Ilangakoon, 2012).
For western Asia (Arabian region), Robards and Reeves (2011) note that cetaceans were caught and used for human consumption in Oman (dwarf sperm whale Kogia sima, common dolphin, Indo-Pacific humpback dolphin, spinner dolphin, bottlenose dolphin) and the United Arab Emirates (common dolphin) between 1970 and 2009. A recent review of finless porpoise stranding in Kuwait did not indicate the use of this species as wild meat (Nithyanandan and Bohadi, 2021). Little is known from this region on the extent of cetacean use or its sustainability.
Sirenians
Declines in dugong numbers have been linked to overexploitation both in Southeast Asia and elsewhere in Asia (Marsh et al., 1997, 2002, 2011; Perrin et al., 2002; Mustika, 2006). Although direct harvests of dugongs are now banned in most Asian countries (Marsh et al., 2011), Pilcher et al. (2017) estimated that about 16% of dugongs caught in fishing gears are consumed. These data come from a questionnaire administered in 20 countries, including 14 Asian countries; country-specific data have not been published. In some countries of Southeast Asia, dugong meat is both locally consumed and commercially traded domestically. For example, in Cambodia, dugongs are considered to bring good luck if caught, and the meat and other body parts are sold at high prices (Beasley et al., 2001). In Malaysia’s most eastern state, Sabah, community interviews indicate that substantial numbers of dugongs are bycaught and, when not discarded, may be utilised for different purposes (Jaaman et al., 2008). Dugong meat is also eaten in various parts of Indonesia, where body parts are also used for traditional remedies and the making of religious artefacts (Marsh et al., 2002, 2011). Although more recent studies are limited, dugong parts can still be openly purchased in markets in some islands (Lee and Nijman, 2015). Dugong hunting has also been documented throughout the species range in the Philippines. The meat is valued as an additional source of protein in remote areas with limited resources, and while it was once sold openly in markets, it is now sold covertly due to its current protected status (Kataoka et al., 1995; Marsh et al., 2002, 2011).
In South Asia, in west India (Gulf of Kutch), dugong oil is valued as a preservative and conditioner for wooden boats, and the meat is believed to have medicinal value (Jones, 1967; Frazier and Mundkur, 1990; Marsh et al., 2002). Historically, dugong meat was highly prised in the Gulf of Mannar (India/Sri Lanka) (Nair et al., 1975) and, in the Andaman and Nicobar Islands, dugongs are occasionally hunted, although the frequency has reduced in recent decades (Das, 1996; Marsh et al., 2002; Hines et al., 2012; D’Souza et al., 2013).
In western Asia (Arabian region), there is archaeological evidence of a 6,000-year-old dugong-hunting culture in the Arabian/Persian Gulf (Méry et al., 2009). Although officially banned by most range states in the region, some illegal hunting still occurs, at least in the Red Sea (Nasr et al., 2019). Dugongs are still caught incidentally in gillnets in both the Red Sea (Nasr et al., 2019) and the Arabian/Persian Gulf. These animals are variously released if still alive, given to researchers to study, or consumed (Nasr et al., 2019). The major drivers for deliberate captures in fishing gears or illegal hunting are poverty and declining fish stocks (Nasr et al., 2019), enabled by weak or non-existent enforcement. Although little is published on the current status of the dugong population(s) in this region, it appears to support the largest dugong population outside Australia (Marsh et al., 2011), mainly in the waters of the United Arab Emirates (UAE) and Qatar.
Chelonians
Like marine mammals, marine turtles have been used for centuries within many South and Southeast Asian countries, as food (both meat and eggs), medicine and ornamentation. Consumption of marine turtle eggs has been reported across most of South and Southeast Asia, with turtle meat also consumed in Sri Lanka, Maldives, Myanmar, Thailand, China, Philippines, and Indonesia (Yifan, 2018). Marine turtles are also used as ornamentation in Indonesia, China, Philippines, and Japan, with Indonesia supplying much of the trade with raw materials (Yifan, 2018).
In East Asia, consumption of marine turtle meat and eggs is reported, as well as a market for turtle-derived ornamentation (Yifan, 2018). In China, most marine turtle nesting beaches have been lost as a consequence of excessive historic exploitation of turtles and their eggs, as well as habitat degradation (Lin et al., 2021). China’s ongoing demand for turtle products is considered one driver encouraging turtle trade from Southeast Asia, with at-sea trade reported between Chinese vessels and fisherman in Southeast Asia, e.g., in the Philippines, Indonesia, and Malaysia (Lam et al., 2011).
Southeast Asia is a hotspot for marine turtle consumption and trade. Historically, the island of Bali, Indonesia, was one of the world’s largest markets for green turtles, and remains an important hub in Indonesia for both domestic trade and some continuing international trade (Troëng and Drews, 2004; IOSEA, 2014; Pilcher and Williams, 2018). There is evidence of ongoing high levels of harvest of turtles and their eggs in other parts of Indonesia (Dethmers and Baxter, 2011; IOSEA, 2014; Pilcher and Williams, 2018). Eight seizures occurred in Kalimantan between 2015 and 2018, with 13,200 eggs seised. The sale of eggs, turtle meat, and live turtles is common and occurs along the coasts of West Sumatra, Java, Bali, Kalimantan, Sulawesi, Maluku, and Papua (Pilcher and Williams, 2018). Open trade in markets has reportedly declined, but shifted to underground markets in some places (Gomez and Krishnasamy, 2019).
In Malaysia, consumption of turtle eggs is the main form of use, particularly in Sabah and Terengganu (Gomez and Krishnasamy, 2019; Joseph et al., 2019; Mohd Jani et al., 2020), which involves both legal and illegal trade. In most of Peninsular Malaysia, all marine turtle eggs (except leatherback turtle eggs in Terengganu and Pahang) are freely and legally traded in local markets, except in Perak and Melaka. In some places, such as Perlis and Selangor, trade in unregulated (Mohd Jani et al., 2020). In Sabah and Sarawak, it is illegal to harvest and consume sea turtle eggs, but harvest and trade, particularly with Terengganu, is reportedly common (Troëng and Drews, 2004; Chan, 2006; IOSEA, 2014; Mohd Jani et al., 2020). Increased regulation of harvests generally was implemented after a dramatic decline in leatherback turtle nests at Rantau Abang in Terengganu – from some 10,000 nests per year in 1956 to just three in 2002 (Troëng and Drews, 2004), attributed to both continuous exploitation of eggs over decades, and fisheries bycatch of adults (Chan and Liew, 1996; Eckert et al., 2012; NMFS and USFWS, 2020). However, at least on Redang Island, Terengganu, legal protection of turtle nesting beaches as well as changing attitudes toward egg consumption, are thought to have led to a decline in demand for green turtle eggs, although consumption continues (Poti et al., 2021). Population declines of leatherback turtles in Terengganu contrast with recoveries of green turtles in Sabah, following the strong enforcement of the egg protection programme in the early 1970s (Chan, 2006).
In Viet Nam, there is a commercial demand for marine turtle meat and products, observed for sale in 39 of 436 outlets surveyed (Gomez and Krishnasamy, 2019). The most widespread use involves hawksbill turtles, with shells used to make decorative items including bekko ornaments, and taxidermied whole animals. These are sold domestically, but are also seised in international trade mostly with China (IOSEA, 2014; Pilcher and Williams, 2018). In 2002, a domestic ban on turtle trade in Viet Nam markets diverted trade to at-sea exchanges (Chan et al., 2009), although they can still be found in some markets, restaurants, and shops (Hamann et al., 2006). Numerous seizures in Viet Nam, mostly hawksbill turtles, suggest that Indonesia and Malaysia remain a source of raw shell plates for tortoise shell bekko manufacture (IOSEA, 2014). Online sale of turtle products is increasing, especially in Indonesia and Viet Nam (Pilcher and Williams, 2018).
There is limited information for other countries in Southeast Asia. The Philippines report an unquantified increase in illegal harvest of marine turtles in several regions and continued high levels of egg harvesting (IOSEA, 2019), including in the Tawi-Tawi Islands (IOSEA, 2014), where historically 70% of eggs were harvested (Chan and Shepherd, 2002). Harvest of turtles and eggs is common along the coast of Myanmar (Win and Lwin, 2012), especially in remote areas (IOSEA, 2019).
In South Asia, there is limited information on the extent of turtle harvests in the northern Indian Ocean, although it does occur, as evidenced by expert elicitation (Riskas et al., 2018) and media reports of fishing vessels that have been impounded for operating illegally in other countries’ waters, which have then been found to have turtles onboard (IOSEA, 2014). In India, reports of fishermen targeting turtles for their meat are largely anecdotal (IOSEA, 2014). While some areas report extremely high egg harvests, for example nearly all Olive Ridley (Lepidochelys olivacea) nests in Tamil Nadu were reportedly harvested illegally in 2011 (IOSEA, 2014). Others report egg collection is decreasing, but the reasons why are unclear (Hasan, 2009). Anecdotal reports suggest a turtle fishery in Pakistan territorial waters may be supplying neighbouring countries with turtle meat (IOSEA, 2014). In the Maldives, turtle hunting is increasingly uncommon whereas the harvesting and consumption of eggs remains common (IOSEA, 2019). In the last half of the twentieth century, the communities at Rekawa beach, Sri Lanka, collected most of the turtle eggs laid on the beach, for sale and local consumption and the harvesting of turtles for meat was common in other parts of Sri Lanka (Ekanayake et al., 2002). A survey conducted in 2007 found consumptive use of turtle meat and eggs had significantly reduced, and noted that nesting rates were high in areas, such as Rekawa, that now had community-based protection of nests (Rajakaruna et al., 2009).
In western Asia (Arabian region), there are reports of high levels of egg collection in Saudi Arabia, and high rates of illegal harvest of animals in Iranian waters of the Oman Sea (IOSEA, 2014). Low incidence of turtle use is reported for Oman, whereas anecdotal reports suggest some level of egg collection in the UAE, but no hunting or larger scale harvesting (IOSEA, 2019).
Crocodylians
Only two crocodylian species are currently included in the Appendices of CMS, both in Asia. Today, limited gharial (Gavialis gangeticus) populations are restricted to a few large river systems in north India and lowland Nepal. The species is now extinct in Bhutan, Myanmar, Pakistan, and possibly, Bangladesh (Lang et al., 2019). Historically, gharial were hunted as sport, and occasionally taken opportunistically for skins and meat. Seasonal movements related to nesting and feeding, up and down large open rivers and their tributaries likely regularly resulted in trans-boundary movements between Pakistan, Bangladesh, Bhutan, Burma, and India by gharials resident in boundary waterways. Illegal consumption of gharial eggs sometimes occurs, but is a minor concern relative to the widespread and frequent habitat destruction/alteration (sand mining and water extraction) and mortality from net entanglement due to illegal fishing (Stevenson, 2015; Lang et al., 2019). Eggs were used in some communities along the Karnali-Girwa Rivers (Nepal–India) and Narayani-Gandak Rivers (Nepal–India) for food and use in traditional remedies (Lang et al., 2019).
Like the gharial, the population status of saltwater crocodiles (Crocodylus porosus) varies greatly between range states in Asia, effectively extinct in Thailand, Cambodia, Viet Nam, and the Seychelles, with the largest and most secure Asian wild population in West Papua and Papua Provinces in Indonesia. Subsistence use of eggs and crocodiles for food by indigenous peoples and local communities’ dates back thousands of years (Webb et al., 2010). Currently, the species is subject to legal ranching (eggs, juveniles) and wild harvest (direct hunting; Webb et al., 2010). In the state of Sarawak (Malaysia), a limited wild harvest of crocodiles was implemented recently, to address increasing levels of human-crocodile conflict, and the meat is used for domestic consumption. Cultural beliefs of indigenous people of Sarawak do not allow crocodiles to be killed and/or eaten (Tisen et al., 2013). In Myanmar, wild populations were seriously depleted by hide hunting (1950s and 1960s) and the meat was consumed by people (Platt et al., 2012). Severe depletion in Myanmar, as in Thailand, Cambodia, Vietnam and Philippines, has allowed local people to use waterways freely, which they could not do historically, complicating efforts to rebuild wild populations (Platt et al., 2012). In wildlife sanctuaries in the Ayeyarwady Delta, saltwater crocodiles are recovering, despite incidental catch in fishing nets (Than et al., 2020). A survey in Thailand (Webb and Jenkins, 1991) revealed some live saltwater crocodiles imported legally from a small government operated farm in Myanmar. Some trade may still exist at low levels (Platt et al., 2012) given the porous border.
Elsewhere in their Asian range, saltwater crocodile populations are nominally protected, with varying levels of “problem” animal control (Webb et al., 2010). Recovery of wild populations increases human-crocodile conflict on people and livestock (Brackhane et al., 2018), with many rural communities not inclined to tolerate these large predators. “Problem” crocodiles removed for public safety, legally or illegally, may be used as food, but cultural taboos may dictate whether this occurs (e.g., Corvera et al., 2017; Brackhane et al., 2018; Webb, 2020).
Oceania
Cetaceans
Contemporary use of cetaceans is reported across Polynesia, Melanesia, and Micronesia, however, often the species could not be identified (Robards and Reeves, 2011). Since 1990, the use of stranded or intentionally caught humpback whales has been reported as rare in Tonga, as has the use of bottlenose dolphins in New Caledonia. Species identification is available for the Solomon Islands, where Risso’s (Grampus griseus), Fraser’s (Lagenodelphis hosei), pantropical spotted, spinner dolphins, and Cuvier’s beaked whale (Ziphius cavirostris) are utilised. In villages on Malaita, Solomon Islands, a traditional dolphin hunt for meat and teeth occurs (100,000 dolphin teeth annually; Takekawa, 2000). Teeth are used as a traditional wedding dowry, and are also used for personal adornment (Takekawa, 2000). Attempts to stop traditional hunts in 2010, using financial compensation, were unsuccessful (Oremus et al., 2015).
The Gilbert Islands of Kiribati have a long history of marine mammal hunting (Di Piazza and Pearthree, 2001), and data suggest that some species of mesoplodont and Cuvier’s beaked whales are still targeted for human consumption (Baker et al., 2013).
Sirenians
Dugongs have high cultural value in many parts of Oceania, and traditional legal hunting is widespread in Pacific Islands range states (Marsh et al., 2011). The dugong is a cultural keystone species in the Torres Strait (Butler et al., 2012) where it has been harvested for at least 4,000 years (Crouch et al., 2007). The harvest still has significant provisioning value, although the value of dugong meat as food is less than its cultural services (Delisle et al., 2018). The Torres Strait Treaty between Australia and Papua New Guinea guarantees the rights of Islanders to their traditional way of life, including hunting dugongs (Havemann and Smith, 2007).
There is limited effective regulation of the Papua New Guinea harvest, apart from a national ban on the sale of meat, which means that some villagers still catch dugongs illegally, especially using mesh nets (Marsh et al., 2011). Throughout their range in northern Australia, native title holders are permitted to hunt dugongs in their own traditional sea country. In the Australian Torres Strait, governance mechanisms include a ban on selling dugong meat, traditional owner-only hunting rights, spatial and gear controls, and community management through 15 non−statutory, community−based turtle and dugong hunting plans (Marsh et al., 2015). These restrictions are bolstered by socioeconomic realities, for example access to regions where dugongs are abundant is financially unviable due to the high price of fuel (Marsh et al., 2015). In the Western Islands of Torres Strait, the dugong harvest in the 1990’s was estimated to be on the order of 1,000 individuals per year (Marsh et al., 1997, 2002) and concerns were expressed about its sustainability (Heinsohn et al., 2004; Marsh et al., 2004). Multiple lines of evidence, including revised population size estimates, high calving rates, and the high genetic diversity of the population indicate that the harvest in the Western and Central Torres Strait is likely sustainable at a regional scale (Marsh et al., 2015; Hagihara et al., 2018).
Outside of the Torres Strait, there is also evidence of dugong hunting. Dugong meat is highly prised in the Solomon Islands and Vanuatu (Chambers et al., 1989; Kile et al., 2000; Marsh et al., 2002, 2011). However, at least in some areas, such as the autonomous region of Bougainville, the largest island in the Solomon Archipelago, directed hunting of dugongs is now extremely rare (Kinch, 2008). In New Caledonia, dugong hunting is now prohibited in some provinces, but allowed in others with a permit, although some illegal harvests still occur (Cleguer et al., 2017). In Micronesia, dugong hunting in Palau was largely practised to obtain meat for special occasions, and had high traditional value (Marsh et al., 1995). Whilst dugong hunting is now illegal in Palau, evidence from 2003 suggests that dugong hunting may still be widespread (Matthews, 2003).
Chelonians
Marine turtles and their eggs are harvested for consumption across several regions of Oceania, except Hawaii, threatening all five species (Work et al., 2020). It is estimated that Papua New Guinea harvests 15,217 turtles per year, which is equivalent to 36.1% of the estimated global legal harvest of 2013 (Humber et al., 2014), and it is unknown if this level of take is sustainable (Work et al., 2020). In the Torres Strait, where large juvenile and adult green turtles are abundant (Fuentes et al., 2015), turtle harvests have been sustained over time, although how this is achieved biologically is unclear, hence concerns remain about the sustainability of current harvest levels (Hagihara et al., 2016). Turtles play a significant role in the customary economy of many Aboriginal and Torres Strait Islander communities in Australia, and use for personal, domestic, and non-commercial purposes is legal, although the level and impact of the harvest is unknown (IOSEA, 2019). Illegal egg harvest on the Solomon Islands has been identified as a threat to olive ridley, leatherback, and hawksbill sea turtles (Work et al., 2020), as is the estimated annual harvest of 9,473 turtles (species unknown) by spear fishers (Vuto et al., 2019).
While universally valued, there is significant variability between communities and regions about the rules and social practises surrounding turtle consumption in the Pacific Islands region (Allen, 2007). In most Polynesian islands, turtle consumption is typically restricted to high-ranking individuals (e.g., chiefs), while in Niue, meat is only consumed by males (Rudrud, 2010). On Rapa Nui (Easter Island), consumption of turtles was once common, but is now no longer practised by the islanders (Álvarez-Varas et al., 2020). In parts of Melanesia, turtle shell trade is an important part of society (Balazs, 1983; Allen, 2007; Rudrud, 2010).
Crocodylians
The largest and most secure wild populations of saltwater crocodiles in the Oceania region occur in Australia and Papua New Guinea. Within these countries, the species is subject to legal commercial ranching (eggs and juveniles), and wild harvest for commercial exports in compliance with CITES. Crocodiles are also hunted legally for subsistence by indigenous peoples (e.g., Webb, 2020). Hunting crocodiles for meat also occurs in some parts of the Solomon Islands, but this tends to involve crocodiles that have attacked people or livestock (Van der Ploeg et al., 2019). Fear of being attacked by crocodiles results in crocodiles and their nests or eggs being destroyed opportunistically by local people (Van der Ploeg et al., 2019).
Studies on saltwater crocodile movement patterns have largely been carried out in Australia (Campbell et al., 2010; Fukuda et al., 2019; Fukuda et al., 2022). Although long-distance movements of individuals out of their typical distribution range have been recorded (e.g., Marshall Islands; Manolis, 2005; Spennemann, 2020), these appear to be the exception rather than the rule. Movements of Australian saltwater crocodiles across international borders (e.g., possibly between Australia, Timor-Leste, Papua New Guinea, and Indonesia) may be linked to territorial exclusion as populations reach carrying capacity within Australia (Fukuda et al., 2011; Brackhane et al., 2018), and are not likely representative of migratory patterns.
Discussion
The harvesting of CMS-listed, non-fish migratory megafauna, for aquatic wild meat for human consumption and other purposes, is clearly widespread in the tropics and subtropics, with the extent of harvest highly variable between species and countries, and the impact on populations – whether sustainable or not– rarely known with scientific confidence. The legality of national harvests is equally variable across species and countries. The purposes of use range from subsistence by Indigenous Peoples and Local Communities (IPLCs), often in poverty, but respected under the UN Declaration of the Rights of Indigenous People (UNDRIP), to purely commercial purposes at the other extreme, and all manner of combinations within. Aquatic wild meat is clearly a significant issue that needs to be considered more seriously at national and international levels, taking into account diverse value orientations (van Vliet, 2018), including within conventions like the CMS, and some key areas are discussed below.
Conservation Concerns
Given that the extent of use relative to the size and trends (increasing, decreasing or stable) in the wild source population and rates of immigration and emigration are seldom known accurately, the impact of harvests for aquatic wild meat on the conservation status of source populations can seldom be quantified with any scientific certainty. For example, in mainland South America, it is unclear whether the harvest and trade of small cetaceans specifically for human consumption in Peru was being sustained by the wild population, but from a precautionary stance, increasing rates of harvest for bait or other purposes in Peru, Bolivia, Ecuador, Colombia, Venezuela, and Brazil (Barbosa-Filho et al., 2018; Mintzer et al., 2018; Campbell et al., 2020a; Castro et al., 2020; Briceño et al., 2021) constitute a potential threat. Similarly, the harvest of migratory small cetaceans for aquatic wild meat in the Gulf of Guinea, in particular Atlantic humpback dolphin, which meet IUCN Red List criteria for Critically Endangered (Collins et al., 2017), are assumed to be threatening despite limited information on harvest levels, population size, trends and distribution. In the south western coast of India and throughout Southeast Asia, killing, rather than releasing cetaceans by-caught in fishing gear, for aquatic wild meat is assumed to be a threat to migratory cetaceans. Indeed, high human population densities and the prevalence of fishing activities in Southeast Asia generally make it a hotspot for aquatic wild meat use, even if the harvest is opportunistic rather than targeted. Against this, traditional harvests of dolphins in the Solomon Islands appear to have been occurring for millennia.
For depleted sirenian populations, even a modest harvest for human consumption may constrain the rate of recovery of populations which are highly sensitive to changes in adult survival (Marsh et al., 2011). Most local populations cannot withstand human-induced mortality. In Senegal, The Gambia, Nigeria, Cameroon, and Democratic Republic of the Congo, more than 1,000 manatees have been killed by illegal hunting, bycatch, dams and watercraft between 2017 and 2019 (Keith-Diagne et al., 2019). Aquatic wild meat is a major motivation for this loss, impacting negatively on most populations. Against these examples, the harvest of dugong for aquatic wild meat by indigenous people across northern Australia, which contains the most abundant sirenian populations (Marsh et al., 2011), have been sustained for more than 4000 years. This highlights clearly that differences in local circumstances between areas, such as drivers of hunting and consumption, hunting technologies used, human density and other threats to animals and their habitats, and how they change over time, can influence harvest sustainability.
Capture and consumption of marine turtle adults and harvest of their eggs is ubiquitous across much of the species’ ranges, particularly in the Caribbean, Southeast Asia, off Papua New Guinea, and by Indigenous people across northern Australia. Whilst there are management programs in some places (see section “Discussion” below), and evidence of significant declines in population and nesting due to past and present exploitation in many countries (Troëng and Drews, 2004; Amorocho et al., 2016; Blumenthal et al., 2021; Lin et al., 2021), low-level harvests have been sustained for millennia in some countries, and harvests can be managed sustainably for aquatic wild meat and other products (Carrillo et al., 1999; Fleming, 2001).
Risks to riverine megafauna from harvest may be particularly high, even if opportunistic, because the risks are compounded by unique threats to riverine species, such as dams, intensive fishing, and pollution where human population density is high (He et al., 2017). In the Amazon, the tucuxi and Amazon River Dolphin are considered threatened by the IUCN Red List, are nominally protected, but are illegally harvested as bait for fishing enterprises. In India and Bangladesh, the use of Ganges River dolphins for meat and bait is less common today than in the recent past, but even small harvests may be problematic given their small population sizes (Braulik and Smith, 2019). For the sirenians, both the Amazonian and African manatees are considered sensitive to any harvest given low reproductive output and generally small local population sizes, making their use for aquatic wild meat potentially problematic. In South America, the giant South American river turtle Podocnemis expansa has been harvested for food for millennia, despite harvest levels and population responses being poorly known. Management interventions are enhancing conservation in some places (Forero-Medina et al., 2021). Riverine megafauna may suffer from a lack of management and research as they are seen as neither terrestrial species nor fish, and the case for increased research and management of riverine and freshwater megafauna is a strong one.
Finally, the use of aquatic megafauna for aquatic wild meat is likely to be far more widespread in terms of frequency and species than reported here, especially amongst IPLCs. Monitoring and reporting is limited, and because many of the species are protected by national law, or are charismatic, their use is secretive. The trans-boundary nature of harvests and associated trade of these oceanic, coastal, and riverine species requires increased international attention and cooperation. Despite the wisdom of adopting a precautionary approach in the absence of quantitative data on harvest levels, population sizes, and trends for most species used for aquatic wild meat, the case for researching in more depth the harvests that do appear sustainable is a strong one. There is a fundamental difference between assembling information on isolated population dynamics, and studying “dynamic populations” – how populations compensate to sustain uses (Webb, 2015). CMS has taken an early step, agreeing to progress the development of a sub-regional Aquatic Wild Meat Action Plan for the Gulf of Guinea for consideration by CMS COP14 (Decision 13.64, COP13; CMS, 2020). Similar urgent attention will be needed to either work through existing regional processes or developing targeted action plans for the remaining highlighted species.
Human Dimensions and Drivers of Aquatic Wild Meat Use
The drivers of aquatic wild meat consumption and trade are varied and changing. For some IPLCs, consumption of aquatic wild meat may have high socio-cultural significance (for example dugongs in the Torres Strait; Delisle et al., 2018), and is a right bestowed under UNDRIP. Food is an important driver of low-level aquatic wild meat harvests in many places (e.g., turtles for Miskito and Creole Caribbean Nicaraguans; Garland and Carthy, 2010), yet its nutritional contribution relative to alternatives is rarely quantified (Olmedo and Farnés, 2004). Some freshwater turtles eaten by indigenous people in northern Australia are exceptionally rich in iron, which is otherwise deficient in their diet (Eisemberg, 2020). Increasing market access and integration into cash-based economies changes the dynamics of aquatic wild meat consumption and trade in some areas, and as such the gradient between subsistence use and commercial trade becomes hazy (Frazier, 1980; Ingram et al., 2021). Subsistence and commercial uses merge insensibly in many of the examples of human consumption reviewed here, although some uses, for example botos and tucuxis in Amazonia, involving deliberate and illegal harvesting for bait to catch fish of higher commercial value, adds new complications (Mintzer et al., 2018).
The dynamics of aquatic wild meat harvests, use, and trade may change in response to climate change influences on food security and habitats. Declines of fish stocks and terrestrial wild meat may increase dependence on aquatic megafauna in coastal communities with few alternatives, for example cetaceans in West Africa (Leeney et al., 2015; Van Waerebeek et al., 2017). Fisheries catch potential is predicted to decline over the 21st century under all emissions scenarios, especially in the tropics (Bindoff et al., 2019), and livelihoods and food security of communities currently dependent on marine resources are predicted to be adversely affected. An interaction between declining fish stocks and use of terrestrial wild meat has occurred in West Africa (Brashares et al., 2004; Rowcliffe et al., 2005), hence increased monitoring of the extent of aquatic wild meat use in local contexts could be important.
Potential Implications for Human Health
Recognising the importance CMS Parties place on the human health consequences of species management actions, it is considered briefly here. Most research investigating the human health impacts of consuming wildlife deal with terrestrial wild meat, but some apply to aquatic wild meat. Food and nutrition, often provided to people in relative poverty by wild meat, is fundamental to their health and well-being, and a basic right (Article 25) within the UN Universal Declaration of Human Rights. However, there are clearly health risks associated with wild meat that are context-specific, and include zoonotic pathogens (viruses, bacteria, parasites) and excessive human consumption of heavy metals and pollutants. Recorded Emerging Infectious Disease (EIDs) events, which can influence economies and public health, nationally and internationally, were 60.3% zoonoses (Jones et al., 2008), with 71.8% originating in terrestrial wildlife, and increasing over time (Jones et al., 2008). Ebola virus disease (Greatorex et al., 2016), Avian Influenza A (Poovorawan et al., 2013), and Middle East Respiratory Syndrome (MERS) (Cauchemez et al., 2014) are all examples. None of these have been attributed to aquatic wild meat, but they exist side-by-side in many cultures.
Fundamental hygiene is a risk factor with all meat consumption, wild or domestic. The consumption of raw or undercooked pinniped or cetacean meat has resulted in bacterial (e.g., salmonellosis and botulism) and parasite (trichinellosis and toxoplasmosis) infections in people (Bender et al., 1972; Tryland, 2000; McLaughlin et al., 2004; Van Bressem et al., 2009; Tryland et al., 2014). Reptile meat and eggs, if not subject to inspection and hygienic treatment, can cause bacterial (Salmonella spp., Vibrio spp.) and potentially parasite infections (Spirometra, Trichinella, Gnathostoma, pentastomids) and biotoxin problems (Magnino et al., 2009; Cantlay et al., 2017). Heavy metals and pollutants in some chelonian life stages (Frías-Espericueta et al., 2006), cetaceans (Fielding and Evans, 2014), and sirenians (Marsh et al., 2002, 2011) exceed international food safety standards, and a growing list of bacterial, viral, and fungal agents in marine mammals (Waltzek et al., 2012) could be problematic. Viable substitutes for wild meat, if they can be afforded, also have a multitude of health risk implications (Tomley and Shirley, 2009), and clearly further research is needed.
Where the use of aquatic wild meat is not sustainable, clearly compromises the conservation of the species involved, and has definitive health risks that exceed those of substitute meat, strategies for reducing aquatic wild meat use and consumption may be possible through partnerships between local people, governments, public health professions, veterinarians, and both wildlife managers and ecologists.
Focused Research and Action
For most species used for aquatic wild meat reviewed here, there are vast gaps in knowledge about the extent of harvest, the source population size and trends, and the degree to which it is influenced by immigration and emigration. Added to this, there are relatively few insights into the population processes involved in aquatic wild meat harvests that have been sustained, sometimes over millennia. Hence conservation action has been precautionary, in many cases wisely implementing species protection, but tending to ignore ongoing uses or treat them as unavoidable collateral damage. This is especially so where the rights of IPLCs to use aquatic wild meat is engrained in national legislation and international agreements, such as UNDRIP.
Given many of these uses will continue, and be vulnerable to expansion for economic reasons, a strong case exists to better understand the sustainability of current uses, and be better able to apply management interventions when needed to ensure sustainability is achieved, and that unsustainable uses can be avoided. Harvests of cetaceans and sirenians are considered compromised by low reproductive rates. Crocodylians and chelonians share many life history traits, but whereas managed, sustainable uses of crocodylians are common (e.g., egg harvests), they are reasonably rare with marine turtles. The total annual legal take of wild marine turtles was thought to have decreased from 116,420 in the 1980s to 45,387 in the 2000s (Humber et al., 2014), but real levels of harvest, including illegal harvest, may be much higher (Pilcher and Williams, 2018). At some sites, successful recovery of depleted marine turtle nesting populations has been attributed to protecting nest sites from egg collection (Chan, 2006; Hamilton et al., 2015; Sardeshpande and MacMillan, 2019). At other sites, managed harvest of eggs has been sustainable and created increased stewardship of nesting animals (Campbell et al., 2007; Sardeshpande and MacMillan, 2019). Bycatch and subsequent use of turtles through industrial and small-scale fisheries also need to be addressed simultaneously. Life history parameters can be used to make crude predictions about sustainability, but factors like density-dependent adjustments are difficult to account for. It is clear that much more research is needed on the dynamics of populations where harvests have been sustained over time to understand the circumstances under which it is biologically possible.
Active management interventions, supported by local communities (community-based management), are critical to ensuring the sustainability of uses of aquatic wild meat for sustenance, maintenance of culture and tradition, and improved livelihoods. It is also important for identifying levels of use likely to be unsustainable. When a species is not used for aquatic wild meat, management action tends to be ignored. Investment in management is more likely when aquatic megafauna contributes to improved livelihoods of local people (Campbell et al., 2007; Sardeshpande and MacMillan, 2019); for example, the community turtle egg harvest at Ostional, Costa Rica (Sardeshpande and MacMillan, 2019). Public support for rebuilding wild populations of predators, such as saltwater crocodiles, has been achieved by generating economic benefits through sustainable use (Webb, 2020), and by targeting problem animals for removal and use (Fukuda et al., 2014). In northern Australia, a conservation goal of recovering a severely depleted wild population of saltwater crocodiles back to carrying capacity was achieved whilst being used sustainably to generate economic benefits in compliance with CITES.
Such case histories are unlikely to be directly transferable to most cetaceans and sirenians, given different population parameters and market values. However, traditional uses will continue and management may be needed if sustainability is compromised. Increased uses for human consumption and bait, driven by commercial incentives, are likely to compromise sustainability and need appropriate management interventions (public education, alternative food/bait substitutes, altered fishing methods and bycatch mitigation measures, increased inspections/enforcement, etc.). The drivers of consumptive use are often relative poverty, and legal protection, sustainability for the future, risk of punishment, and accessing substitutes are simply overpowered by immediate needs. Top–down legal restrictions alone are unlikely to be sufficient in many socioeconomic contexts where poverty is rife, cultural values are not understood, and management capacity is low (Barrios-Garrido H. et al., 2020; Ingram, 2020).
For both crocodylian species, it is questionable whether today, they meet the criteria for inclusion as migratory species on the CMS appendices. Historically, riverine species such as the gharial made seasonal movements up and down large, wide-open rivers, prior to the advent of numerous man-made obstructions. Now, the many barrages and dams constructed along these waterways have resulted in predominantly unidirectional movements, e.g., downstream, but not upstream, for one or more life history stages, typically the younger, smaller age cohorts. Depending on river stretches traversed, cyclical seasonal movements may cross national boundaries, e.g., seasonal movements on rivers flowing between Nepal and India. Similarly, transboundary movements of saltwater crocodiles likely occur in certain estuarine/riverine settings, but are poorly documented, even though long distance movements at sea occur (Webb and Messel, 1978). Other freshwater species, living in wetlands, riverine, and/or coastal areas at confluences of national boundaries may regularly or occasionally make daily or seasonal movements across international borders. Technically, these species pose a special challenge to the existing definitions of transboundary migrations. The result is their inclusion in the CMS listing, albeit exceptional relative to other listings.
Whilst this review focuses on megafauna that are considered migratory and threatened and are listed in CMS Appendices, there are hundreds if not thousands of other vertebrate species (including fish) used for consumption of aquatic wild meat. Many of these species occur across the tropics and subtropics: for most, sustainable uses are most likely in question. Many of these are included on the IUCN Red List, and are covered by CITES, but some are not because trans-border movements are not evident. Examples include: crocodylians (Genera: Osteolaemus, Mecistops, and Crocodylus) and otters (Genera: Aonyx, Hydrictis) in West and Central Africa; snakes in Southeast Asia (Brooks et al., 2007); and marine mammals in temperate and polar regions (Robards and Reeves, 2011). In particular, riverine and freshwater turtles are at significant risk given that ∼59% of extant turtles are considered threatened (Stanford et al., 2020; Turtle Taxonomy Working Group, 2021), including turtles in North America (Mali et al., 2014), Central and South America (e.g., Genera: Dermatemys; Tovar-De la Cruz et al., 2021), West and Central Africa (Genera: Pelusios, Pelomedusa, Cyclanrobis, Cycloderma, and Trionyx), and South, East, and Southeast Asia (Genera: Batagur, Chitra, Nilssonia). Turtle populations are declining rapidly due to habitat loss, consumption by humans for food and traditional medicines and collection for the international pet trade (Stanford et al., 2020). Consequently, the extent and magnitude of aquatic wild meat usage is a much larger issue not covered in this review, limited only to those migratory species listed in the CMS Appendices.
This has obvious implications for biodiversity conservation generally, which needs to be enabled by public and political will, effective governance, and increased technical focus on the sustainability of uses (Ingram et al., 2021). In many tropical and subtropical countries, the need is great but the capacity and will to address it is limited (Barlow et al., 2018).
Moving Forward
Based on this review of CMS-listed species, the following recommendations for research and policy are made. For research, we recommend:
1. Studies of the human dimensions of aquatic wild meat use, which are critical for designing effective conservation and management programmes that favour sustainable over unsustainable uses, including;
– contemporary and historical socio-cultural aspects of harvesting and consuming aquatic wild meat, including the role of taboos systems, to provide insights or management measures that can be respectful of IPLC cultural practices;
– the drivers of the harvest and consumption of aquatic wild meat;
– the nutritional roles aquatic wild meat provides, the strengths and weaknesses of alternatives, and the health risks from both.
2. Increased quantitative assessments of consumption and trade in aquatic wild meat to better understand demand and trade pathways;
3. Increased research into the ecological sustainability of current harvests, and the population parameters, processes, and science-based management strategies needed to overcome problems, especially in the face of climate change impacts on habitats;
4. Where bycatch is a known problem, to design and test fishing methods to minimise and ultimately eliminate bycatch mortality;
5. Increased research into the use of aquatic wild meat as bait for other commercial fisheries and the possibility of using alternative baits that can be derived sustainably;
6. Reassessment of the migratory nature of crocodylians (Genera: Gavialis, Crocodylus, Mecistops, Caiman, Melanosuchus) and freshwater chelonians and the relevance of CMS to their conservation and management, including whether or not they fit the criteria for inclusion in the Appendices; and,
7. Research of migration routes for aquatic megafauna with a view to better understanding the role of immigration and emigration in populations being used for aquatic wild meat.
In terms of policy and action, we recommend:
1. Increased efforts to assess the efficacy of existing legislation with regard to specific aquatic wild meat uses, and the degree to which enforcement capacity alone can address unsustainable uses;
2. New legislation to protect species being used unsustainably, with training and capacity building to ensure enforcement and management staff can perform tasks efficiently;
3. Implementation of environmental education programmes to raise awareness of the importance and benefits of migratory megafauna, and laws concerning them;
4. Investigating whether sustainable tourism can be implemented as a source of income for local communities, in areas where aquatic wild meat harvests and trade are unsustainable;
5. Coordination between CMS and CITES for improved regulation and sustainable management of the trade in aquatic wild meat species, where appropriate; and,
6. Encouraging the establishment of networks of appropriate experts to foster collaborative efforts to develop regional action plans for reducing unsustainable aquatic wild meat uses.
The adoption of CMS Resolution 12.15 on aquatic wild meat highlighted the importance of political cooperation to address this emerging and serious threat to aquatic species in many areas of the world. Conscious of this need, CMS has taken the early step of forming a cross-taxa Aquatic Wild Meat Working Group within the CMS Scientific Council. This Working Group was charged with producing an expert resource to contribute to international wild meat discussions (Decision 13.64, COP13; CMS, 2020). This review confirms that aquatic wild meat use has been vastly under-researched, despite its critical importance to the conservation, welfare, and sustainable management of aquatic megafauna. The use of aquatic wild meat, especially by IPLCs, has often fallen between the cracks of conservation and fisheries management agencies. This is sometimes a consequence of humanitarian obligations under UNDRIP not fitting easily into either conservation or fisheries goals. Aquatic wild meat is clearly an issue of concern that needs to be addressed openly and transparently, as an important step toward achieving sustainability for both conservation and the many communities across the tropics and subtropics that depend on it.
Author Contributions
DI and MP lead the review process, with core support from NH, HF-N, and LP. All authors contributed to reviewing the literature, writing sections of the manuscript, manuscript revision, read, and approved the submitted version.
Funding
DI was partially supported by funding from the United States Fish and Wildlife Service (AFR1732 Grant F17AP00421 Supplement 0001). IA was supported by the Universidad del Valle and the Colombian Ministry of Science, Technology and Innovation (Postdoctoral Fellowship, Call No. 848 of 2019). The article processing fee was covered by CMS.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Acknowledgments
We would like to thank Sara Kophamel for her help with the IOSEA national reports. We would also like to thank Barry Baker and Col Limpus for comments on an earlier draft, and Donna Mulvenna for assistance with referencing.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fmars.2022.837447/full#supplementary-material
Footnote
- ^ We use ‘fish’ to denote traditional fisheries products, including fishes and commercially exploited crustaceans, squids, bivalves, sea cucumbers, and gastropods, but excluding chondrichthyans (sharks, rays, and chimaeras) for policy reasons as directed by CMS.
References
Aiken, J. J., Godley, B. J., Broderick, A. C., Austin, T., Ebanks-Petrie, G., and Hays, G. C. (2001). Two hundred years after a commercial marine turtle fishery: the current status of marine turtles nesting in the Cayman Islands. Oryx 35, 145–151.
Akoi, K. (2004). Fishers and the West African manatee in the Fresco Lagoon complex, Côte D’Ivoire: common property, conflict and conservation. Ph. D. thesis. Canterbury: University of Kent.
Alfaro-Shigueto, J., Mangel, J. C., Darquea, J., Donoso, M., Baquero, A., Doherty, P., et al. (2018). Untangling the impacts of nets in the Pacific: use of rapid assessments of turtle bycatch to set conservation priorities in small-scale fisheries. Fisher. Res. 206, 185–192.
Allen, M. S. (2007). Three millennia of human and sea turtle interactions in remote Oceania. Coral Reefs 26, 959–970.
Altherr, S., and Hodgins, N. (2018). Small cetaceans, big problems: a global review of the impacts of hunting on small whales, dolphins and porpoises. Wiltshire: Whale and Dolphin Conservation.
Alvarez-Alemán, A., García Alfonso, E., Powell, J. A., Jacoby, C. A., Austin, J. D., and Frazer, T. K. (2021). Causes of Mortality for Endangered Antillean Manatees in Cuba. Front. Mar. Sci. 8:646021. doi: 10.3389/fmars.2021.646021
Álvarez-Varas, R., Barrios-Garrido, H., Skamiotis-Gomez, I., and Petitpas, R. (2020). Cultural role of sea turtles on Rapa Nui (Easter Island): Spatial and temporal contrast in the Pacific Island region. Island Stud. J. 15, 253–270.
Amir, O. A., Berggren, P., and Jiddawi, N. S. (2012). Recent records of marine mammals in Tanzanian waters. J. Cetacean Res. Manage. 12, 249–253.
Amorocho, D., Leslie, A., Fish, M., Sanjurjo, E., Amoros, S., Avila, I. C., et al. (2016). Marine Turtle Action Plan. WWF Latin America and The Caribbean: 2015-2020. Cali: WWF- Colombia, 122.
Andrianarivelo, N. (2001). Essai d’évaluation de l’importance de la pêche aux dauphins dans la région d’Anakao (sud-ouest de Madagascar). Ph. D. thesis. Madagascar: Institut Halieutique et des Sciences Marines.
Andrianarivelo, N., BertoliMarius, N., Jaohery, A., Vatosoa, O., Pepin, J., Vahoavy, R., et al. (2019). Assessment of Cetacean Diversity, Hunting, by-Catch and Stranding in Southern Madagascar. Document SC/68A/SM06 Presented at the Scientific Committee Meeting of the International Whaling Commission. Kenya.
Ankersen, T. T., Stocks, G., Paniagua, F., and Grant, S. (2015). Turtles Without Borders: The International and Domestic Law Basis for the Shared Conservation, Management, and Use of Marine turtles in Nicaragua, Costa Rica, and Panama. J. Int. Wildlife Law Policy 18, 1–62.
Antunes, A. P., Fewster, R. M., Venticinque, E. M., Peres, C. A., Levi, T., Rohe, F., et al. (2016). Empty forest or empty rivers? A century of commercial hunting in Amazonia. Sci. Adv. 2:e1600936. doi: 10.1126/sciadv.1600936
Arévalo-González, G. K., Castelblanco-Martínez, N., Sánchez-Palomino, P., and López-Arévalo, H. (2010). Uso de metodologías complementarias para la determinación del tamaño de la población de manatí (Trichechus manatus) en la Ciénaga de Pareces (Santander, Colombia). Memorias De La Conferencia Interna En Medicina Y Aprovechamiento De Fauna Silvestre, Exótica Y No Convencional 6, 4–14.
Arraut, E. M., Arraut, J. L., Marmontel, M., Mantovani, J. E., and Novo, E. M. L. M. (2017). Bottlenecks in the migration routes of Amazonian manatees and the threat of hydroelectric dams. Acta Amazon 47, 7–18.
Arraut, E. M., Marmontel, M., Mantovani, J. E., Novo, E. M. L. M., Macdonald, D. W., and Kenward, R. E. (2010). The lesser of two evils: seasonal migrations of Amazonian manatees in the western Amazon. J. Zool. 280, 247–256.
Avila, I. C., García, C., and Bastidas, J. C. (2008). A note on the use of dolphins as bait in the artisanal fisheries off Bahía Solano, Chocó, Colombia. J. Cetacean Res. Manage. 10, 179–182.
Avila, I. C., Gómez-Salazar, C., García, C., and Trujillo, F. (2013). “Interacciones entre mamíferos acuáticos y pesquerías en Colombia. Capítulo 3.2. Págs: 196- 231,” in Diagnóstico del estado de conocimiento y conservación de los mamíferos acuáticos en Colombia, eds F. Trujillo, A. Gärtner, D. Caicedo, and M. C. Diazgranados (Bogotá: Fundación Omacha, Conservación Internacional y WWF), 282.
Ayissi, I., Segniagbeto, G. H., and Van Waerebeek, K. (2014). Rediscovery of Cameroon dolphin, the Gulf of Guinea population of Sousa teuszii (Kükenthal, 1892). ISRN Biodivers. 2014, 1–6.
Ayissi, I., Van Waerebeek, K., and Segniagbeto, G. (2011). “Report on the Exploratory survey of cetaceans and their status in Cameroon,” in Document UNEP/CMS/ScC17/Inf.10. 17th Meeting CMS Scientific Council, Bergen, (Bergen: CMS).
Bachand, N., Arsenault, J., and Ravel, A. (2015). Urban household meat consumption patterns in Gabon, Central Africa, with a focus on bushmeat. Hum. Dimens. Wildlife 20, 147–158.
Baker, C. S., Hutt, A., Thompson, K., Dalebout, M. L., Robins, J., and Brownell, R. L. Jr., et al. (2013). Species identity and human consumption of beaked whales in the Gilbert Islands, Republic of Kiribati. Anim. Conserv. 16, 641–647.
Bal, G., Breheret, N., and Vanleeuwe, H. (2007). An update on sea turtle conservation activities in the Republic of Congo. Mar. Turtle Newsletter 116, 9–10.
Balazs, G. H. (1983). Marine turtles and their traditional usage in Tokelau. Atoll Res. Bull. 279:41.
Bamy, I., Djiba, A., and Van Waerebeek, K. (2021). Recent survey for delphinids at Tristao Islands, Guinea, reinforces concern for bycatches and marine bushmeat use. 2021040094. [Preprint]. doi: 10.20944/preprints202104.0094.v1
Barbosa, J. A. A., Aguiar, J. O., and Alves, R. R. D. N. (2018). Medicinal use of animals by hunters in North eastern Brazil. Ind. J. Tradit. Knowl. 17, 485–493.
Barbosa-Filho, M. L. V., Barreto, R. M. F., Siciliano, S., Seminara, C. I., and Costa-Neto, E. M. (2018). Use of cetaceans as bait in southern Bahia, Brazil, by expert fishermen that market shark fins: a lucrative trade and two threatened zoological groups. Ethnobiol. Lett. 9, 12–18.
Barlow, J., França, F., Gardner, T. A., Hicks, C. C., Lennox, G. D., et al. (2018). The future of hyperdiverse tropical ecosystems. Nature 559, 517–526. doi: 10.1038/s41586-018-0301-1
Barrios-Garrido, H. (2012). “Usos de tortugas marinas en Latinoamérica,” in Caso: indígenas Wayuu en la Península Guajira Venezolana. En Memorias de la Reunión de Especialistas sobre Tortugas Marinas en Latinoamérica - RETOMALA 18, (Huatulco: RETOMALA).
Barrios-Garrido, H. A., Montiel-Villalobos, M. G., Palmar, J., and Rodríguez-Clark, K. M. (2020). Wayuú capture of green turtles, Chelonia mydas, in the Gulf of Venezuela: a major Caribbean artisanal turtle fishery. Ocean Coastal Manage. 188:105123. doi: 10.1016/j.ocecoaman.2020.105123
Barrios-Garrido, H., Espinoza-Rodríguez, N., Rojas-Cañizales, D., Palmar, J., Wildermann, N., Montiel-Villalobos, M. G., et al. (2017). Trade of marine turtles along the Southwestern Coast of the Gulf of Venezuela. Mar. Biodivers. Records 10, 1–12.
Barrios-Garrido, H., Palmar, J., Wildermann, N., Rojas-Cañizales, D., Diedrich, A., and Hamann, M. (2018). Marine turtle presence in the traditional pharmacopoeia, cosmovision, and beliefs of Wayuú indigenous people. Chelonian Conserv. Biol. 17, 177–186. doi: 10.2744/ccb-1276.1
Barrios-Garrido, H., Shimada, T., Diedrich, A., and Hamann, M. (2020). Conservation and Enforcement Capacity index (CECi): Integrating human development, economy, and marine turtle status. J. Environ. Manage. 262:110311. doi: 10.1016/j.jenvman.2020.110311
Barrios-Garrido, H., Turris-Morales, D., and Espinoza-Rodriguez, N. E. (2021). Guiana Dolphin (Sotalia guianensis) in the Maracaibo Lake System, Venezuela: conservation, threats, and population overview. Front. Mar. Sci. 7:594021. doi: 10.3389/fmars.2020.594021
Barrios-Garrido, H., Wildermann, N., Diedrich, A., and Hamann, M. (2019). Conflicts and solutions related to marine turtle conservation initiatives in the Caribbean basin: Identifying new challenges. Ocean Coastal Manage. 171, 19–27. doi: 10.1016/j.ocecoaman.2019.01.003
Beasley, I. L., and Davidson, P. J. J. (2007). Conservation status of marine mammals in Cambodian waters, including seven new cetacean records of occurrence. Aquat. Mamm. 33, 368–379. doi: 10.1578/am.33.3.2007.368
Beasley, I., Davidson, P., Somany, P., and Samanth, L. (2001). “Abundance, distribution and conservation management of marine mammals in Cambodia’s coastal waters,” in Interim Unpublished Report, (Phnom Penh: Wildlife Conservation Society).
Bell, C. D., Blumenthal, J. M., Austin, T. J., Solomon, J. L., Ebanks-Petrie, G., Broderick, A. C., et al. (2006). Traditional Caymanian fishery may impede local marine turtle population recovery. Endangered Species Res. 2, 63–69. doi: 10.3354/esr002063
Bender, T. R., Nevius, S. E., Saslow, A. R., Clark, P. S., Gangaros, E. J., Kaplan, G. J., et al. (1972). Salmonellosis associated with whale meat in an eskimo community serologic and bacteriologic methods as adjuncts to an epidemiologic investigation. Am. J. Epidemiol. 96, 153–160. doi: 10.1093/oxfordjournals.aje.a121441
Benítez-López, A., Alkemade, J. R. M., Schipper, A. M., Ingram, D. J., Verweij, P. A., Eikelboom, J., et al. (2017). The impact of hunting on tropical mammal and bird populations. Science 356, 180–183. doi: 10.1126/science.aaj1891
Bevan, E., Wibbels, T., Najera, B. M. Z., Sarti, L., Martinez, F. I., Cuevas, J. M., et al. (2016). Estimating the historic size and current status of the Kemp’s ridley sea turtle (Lepidochelys kempii) population. Ecosphere 7:e01244.
Bindoff, N. L., Cheung, W. W. W., Kairo, J. G., Arístegui, J., Guinder, V. A., Hallberg, R., et al. (2019). “Changing Ocean, Marine Ecosystems, and Dependent Communities,” in IPCC Special Report on the Ocean and Cryosphere in a Changing Climate, [H.-O. Pörtner, D.C. Roberts, V. Masson-Delmotte, P. Zhai, M. Tignor, E. Poloczanska, K. Mintenbeck, A. Alegría, M. Nicolai, A. Okem, J. Petzold, B. Rama, and N.M. Weyer (eds.)]. In press.
Blumenthal, J. M., Hardwick, J. L., Austin, T. J., Broderick, A. C., Chin, P., Collyer, L., et al. (2021). Cayman Islands Sea Turtle Nesting Population Increases Over 22 Years of Monitoring. Front. Mar. Sci. 8:663856. doi: 10.3389/fmars.2021.663856
Bolaños-Jiménez, J., Mignucci-Giannoni, A. A., Blumenthal, J., Bogomolni, A., Casas, J. J., and Henríquez, A. (2014). Distribution, feeding habits and morphology of killer whales Orcinus orca in the Caribbean Sea. Mamm. Rev. 44, 177–189. doi: 10.1111/mam.12021
Brackhane, S., Webb, G., Xavier, F. M. E., Gusmau, M., and Pechacek, P. (2018). When conservation becomes dangerous: Human crocodile conflict in Timor-Leste. J. Wildlife Manage. 82, 1332–1344. doi: 10.1002/jwmg.21497
Braga Ferreira, P., Torres, R. A., and Garcia, J. E. (2011). Single nucleotide polymorphisms from cytochrome b gene as a useful protocol in forensic genetics against the illegal hunting of manatees: Trichechus manatus, Trichechus inunguis, Trichechus senegalensis, and Dugong dugon (Eutheria: Sirenia). Zoologia 28, 133–138. doi: 10.1590/s1984-46702011000100019
Brashares, J. S., Arcese, P., Sam, M. K., Coppolillo, P. B., Sinclair, A. R. E., and Balmford, A. (2004). Bushmeat hunting, wildlife declines, and fish supply in West Africa. Science 306, 1180–1183. doi: 10.1126/science.1102425
Braulik, G. T., and Smith, B. D. (2019). Platanista gangetica (amended version of 2017 assessment). The IUCN Red List of Threatened Species 2019: e.T41758A151913336. Gland: IUCN, doi: 10.2305/IUCN.UK.2017-3.RLTS.T41758A151913336.en
Braulik, G. T., Kasuga, M., Wittich, A., Kiszka, J. J., MacCauley, J., Gillespie, D., et al. (2018). Cetacean rapid assessment: an approach to fill knowledge gaps and target conservation across large data deficient areas. Aquat. Conserv. 28, 216–230. doi: 10.1002/aqc.2833
Bräutigam, A., and Eckert, K. L. (2006). Turning the Tide: Exploitation, Trade and Management of Marine Turtles in the Lesser Antilles, Central America, Colombia and Venezuela. Cambridge, UK: TRAFFIC International.
Briceño, Y., Sánchez, L., Trujillo, F., von Fersen, L., and Ramirez, S. (2021). Aquatic wildmeat consumption of Guiana dolphins (Sotalia guianensis) in Lake Maracaibo System, Venezuela. Front. Mar. Sci. 8:625801. doi: 10.3389/fmars.2021.625801
Brito, C., Picanco, C., and Carvalho, I. (2010). Small Cetaceans Off São Tomé (São Tomé and Príncipe, Gulf of Guinea, West Africa): Species, Sightings and Abundance, Local Human Activities and Conservation. Document SC/62/SM8, Presented to the International Whaling Commission Conference. Agadir.
Brito, C., Vieira, N., Jordão, V., and Teixeira, A. (2016). “Digging into our whaling past: Addressing the Portuguese influence in the early modern exploitation of whales in the Atlantic,” in Environmental History in the Making, Vol. II, eds C. J. de Melo, E. Vaz, and L. M. Costa Pinto (Berlin: Springer International Publishing), 33–47. doi: 10.1007/978-3-319-41139-2_3
Brooks, S. R., Reynolds, J. D., Allison, E. A., and Bunthang, T. (2007). “The exploitation of Homalopsid snakes at Tonle Sap Lake, Cambodia,” in Homalopsine Snakes; Evolutionary Experiments in Terrestrial-aquatic Transitions, ed. J. C. Murphy (Melbourne: Krieger Publishing), 31–37.
Butler, J. R. A., Tawake, A., Skewes, T., Tawake, L., and McGrath, V. (2012). Integrating traditional ecological knowledge and fisheries management in the Torres Strait, Australia: the catalytic role of turtles and dugong as cultural keystone species. Ecol. Soc. 17:34.
Caldwell, D. K. (1963). The sea turtle fishery of Baja California, Mexico. California Fish Game 49, 140–151.
Calvimontes, U. J. (2009). Etnoconocimiento, uso y conservación del manatí amazónico Trichechus inunguis en la Reserva de Desarollo Sostenible Amanã, Brasil. Lima: Universidad Nacional Agraria La Molina.
Campbell, C., and Lagueux, C. (2005). Survival probability estimates for large juvenile and adult green turtles exposed to an artisanal marine turtle fishery in the western Caribbean. Herpetologica 61, 91–103. doi: 10.1655/04-26
Campbell, E., Pasara-Polack, A., Mangel, J. C., and Alfaro-Shigueto, J. (2020a). Use of Small Cetaceans as Bait in Small-Scale Fisheries in Peru. Front. Mar. Sci. 7:534507. doi: 10.3389/fmars.2020.534507
Campbell, E., Mangel, J. C., Alfaro-Shigueto, J., Mena, J. L., Thurstan, R. H., and Godley, B. J. (2020b). Coexisting in the Peruvian Amazon: Interactions between fisheries and river dolphins. J. Nat. Conserv. 56, 1–8.
Campbell, H. A., Watts, M. E., Sullivan, S., Read, M. A., Choukroun, S., Irwin, S. R., et al. (2010). Estuarine crocodiles ride surface currents to facilitate long-distance travel. J. Anim. Ecol. 79, 955–964. doi: 10.1111/j.1365-2656.2010.01709.x
Campbell, L., Haalboom, B., and Trow, J. (2007). Sustainability of community-based conservation: Sea turtle egg harvesting in Ostional (Costa Rica) ten years later. Environ. Conserv. 34, 122–131. doi: 10.1017/s0376892907003840
Cantlay, J., Ingram, D. J., and Meredith, A. L. (2017). A review of zoonotic infection risks associated with the wild meat trade in Malaysia and their relevance to public health in Southeast Asia. EcoHealth 14, 361–388. doi: 10.1007/s10393-017-1229-x
Carrillo, E., Webb, G. J. W., and Manolis, S. C. (1999). Hawksbill turtles (Eretmochelys imbricata) in Cuba: an assessment of the historical harvest and its impacts. Chelonian Conserv. Biol. 3, 264–280.
Castelblanco-Martínez, D. N., Kendall, S., Orozco, D. L., and Arévalo-González, K. (2016). “La conservación de los manatíes Trichechus inunguis y Trichechus manatus en áreas no protegidas de Colombia,” in Conservación de grandes vertebrados en áreas no protegidas de Colombia, Brasil y Venezuela, eds E. Payán, C. A. Lasso, and C. Castaño-Uribe (Bogotá: Instituto de Investigación de Recursos Biológicos Alexander von Humboldt), 81–98.
Castelblanco-Martínez, D., Bermúdez-Romero, A., Gómez-Camelo, I., Rosas, F., Trujillo, F., and Zerda-Ordoñez, E. (2009). Seasonality of habitat use, mortality and reproduction of the Vulnerable Antillean manatee Trichechus manatus manatus in the Orinoco River, Colombia: Implications for conservation. Oryx 43, 235–242.
Castro, C., Van Waerebeek, K., Cárdenas, D., and Alava, J. J. (2020). Marine mammals used as bait for improvised fish aggregating devices in marine waters of Ecuador, eastern tropical Pacific. Endang. Species Res. 41, 289–302.
Catry, P., Barbosa, C., Paris, B., Indjai, B., Almeida, A., Limoges, B., et al. (2009). Status, Ecology, and Conservation of Sea Turtles in Guinea-Bissau. Chelonian Conserv. Biol. 8, 150–160.
Cauchemez, S., Fraser, C., Van Kerkhove, M. D., Donnelly, C. A., Riley, S., Rambaut, A., et al. (2014). Middle East respiratory syndrome coronavirus: quantification of the extent of the epidemic, surveillance biases, and transmissibility. Lancet Infect. Dis. 14, 50–56. doi: 10.1016/S1473-3099(13)70304-9
Cawthorn, D. M., and Hoffman, L. C. (2015). The bushmeat and food security nexus: A global account of the contributions, conundrums and ethical collisions. Food Res. Int. 76, 906–925. doi: 10.1016/j.foodres.2015.03.025
Cerchio, S., Andrianarivelo, N., and Andrianantenaina, B. (2012). Sightings, Incidental By-Catch, and Hunting of Dugongs (Dugong dugon) on the West Coast of Madagascar, as Inferred from Interviews with Local Fishers: Evidence for Decline in Populations due to Human Exploitation. New York, NY: Wildlife Conservation Society.
Cerchio, S., Andrianarivelo, N., Mendez, M., Razafindrakoto, Y., and Rosenbaum, H. (2009a). Coastal Dolphin Hunting in the South-West of Madagascar: Status of Populations, Human Impacts and Conservation Actions. Document SC/61/SM15 to the International Whaling Commission Scientific Committee. Madeira.
Cerchio, S., Mendez, M., Andrianarivelo, N., Razafindrakoto, Y., and Rosenbaum, H. (2009b). “Coastal dolphins in Madagascar and the MPA process: Defining critical habitat and protecting vulnerable populations,” in Presented at the First International Conference on Marine Mammal Protected Areas, March 2009b, (Maui: ICMMPA).
Cerchio, S., Yamada, T. K., and Brownell Jr. (2019). Global distribution of Omura’s (Balaenoptera omurai) and Assessment of Range-Wide Threats. Front. Mar. Sci. 6:67. doi: 10.3389/fmars.2019.00067
Chacón, D. (2002). Diagnóstico sobre el comercio de las tortugas marinas y sus derivados en el istmo centroamericano. Red Regional para la Conservación de las Tortugas Marinas en Centroamérica (RCA). San José: RCA, 247.
Chacón-Chaverri, D., and Eckert, K. L. (2007). Leatherback sea turtle nesting at Gandoca Beach in Caribbean Costa Rica: management recommendations from fifteen years of conservation. Chelonian Conserv. Biol. 6, 101–110.
Chambers, M. R., Bani, E., and Barker-Hudson, B. E. T. (1989). The status of dugong Dugong dugon in Vanuatu. Tropic Review No. 37. South Pacific: South Pacific Regional Environment Program.
Chan, E. H. (2006). Marine turtles in Malaysia: On the verge of extinction? Aquat. Ecosyst. Health Manage. 9, 175–184. doi: 10.1080/14634980600701559
Chan, E. H., and Shepherd, C. R. (2002). Marine Turtles: The Scenario in South-east Asia. Tropic. Coasts 9, 38–43.
Chan, E. H., Pilcher, N., and Hiew, K. (2009). Report of the Workshop on Regional Cooperation to Address Direct Capture of Marine turtles 1-3 June 2009, Kuala Terengganu. Penerbit UMT: Universiti Malaysia Terengganu.
Chan, E.-H., and Liew, H.-C. (1996). Decline of the leatherback population in Terengganu, Malaysia, 1956-1995. Chelonian Conserv. Biol. 2, 196–203.
Clapham, P. J., and Van Waerebeek, K. (2007). Bushmeat and bycatch: the sum of the parts. Mol. Ecol. 16, 2607–2609. doi: 10.1111/j.1365-294X.2007.03378.x
Cleguer, C., Garrigue, C., Fuentes, M. M. P. B., Everingham, Y., Hagihara, R., Hamann, M., et al. (2017). Drivers of change in the relative abundance of dugongs in New Caledonia. Wildlife Res. 44, 365–376.
CMS (2000). “Conservation Measures for Marine Turtles of the Atlantic Coast of Africa,” in CMS Technical Series Publication No.5, (Bonn: UNEP/CMS Secretariat).
CMS (2015). Report of the Third Southeast Asian Marine Mammal Symposium (SEAMAM III). UNEP / CMS Secretariat, Bonn, Germany. CMS Technical Series No. 32. Bonn: UNEP/CMS Secretariat, 643.
CMS (2017a). Resolution 12.15: Aquatic Wild Meat, Twelfth Conference of the Parties to the Convention on Migratory Species, Manila. Bonn: UNEP/CMS Secretariat.
CMS (2017b). “Report of The Aquatic Mammals Working Group on Aquatic Wild Meat UNEP/CMS/COP12/Inf.12,” in Twelfth Conference of the Parties to the Convention on Migratory Species, Manila, (Bonn: UNEP/CMS Secretariat).
CMS (2020). “Aquatic Wild Meat, within Decisions of the Conference of the Parties to CMS in effect after its 13th Meeting,” in Thirteenth Conference of the Parities to the Convention on Migratory Species, Gandhinagar, (Bonn: UNEP/CMS Secretariat).
Coad, L., Fa, J. E., Abernethy, K. A., Santamaria, C., Wilkie, D., El Bizri, H. R., et al. (2019). Towards a sustainable, participatory and inclusive wild meat sector. Bogor: CIFOR.
Collins, T., Boumba, R., Parnell, R., Vanleeuwe, H., Ngouessono, S., and Rosenbaum, H. C. (2010). The Atlantic humpback dolphin (Sousa teuszii) in Gabon and Congo: Cause for optimism or concern. SC/62/SM9. Berlin: ResearchGate.
Collins, T., Braulik, G. T., and Perrin, W. (2017). Sousa teuszii (errata version published in 2018). The IUCN Red List of Threatened Species 2017: e.T20425A123792572. Gland: IUCN.
Collins, T., Van Waerebeek, K., Carvalho, I., Boumba, R., Dilambaka, E., Mouissou, E., et al. (2019). An Assessment of Cetacean Bycatches, Strandings and Other Mortalities from Central Africa, Including Evidence of Use by People. Document SC/68A/SM/05, IWC Scientific Committee Meeting. Nairobi.
Conway-Gómez, K. (2007). Effects of human settlements on abundance of Podocnemis unifilis and P. expansa turtles in Northeastern Bolivia. Chelonian Conserv. Biol. 6, 199–205.
Corvera, M. D., Manalo, R. I., and Aquino, M. T. R. (2017). People and crocodiles sharing one environment: an analysis of local human-crocodile conflict management strategies in the Philippines. J. Anim. Sci. Res. 1, 1–6.
Cosentino, A. M., and Fisher, S. (2016). The utilization of aquatic bushmeat from small cetaceans and manatees in South America and West Africa. Front. Mar. Sci. 3:163. doi: 10.3389/fmars.2016.00163
Crema, L., Da Silva, V., and Piedade, M. (2020). Riverine people’s knowledge of the Vulnerable Amazonian manatee Trichechus inunguis in contrasting protected areas. Oryx 54, 529–538.
Crespo, E. A. (2009). “Franciscana Dolphins: Pontoporia blainvillei,” in Encyclopaedia of Marine Mammals, eds W. F. Perrin, B. Würsig, and J. G. M. Thewissen (New York, NY: Elsevier), 466–469.
Crouch, J., McNiven, I. J., David, B., Rowe, C., and Weisler, M. (2007). Berberass: marine resource specialisation and environmental change in Torres Strait during the past 4000 years. Archaeol. Oceania 42, 49–64.
Cruz-Antía, D., and Gómez, J. R. (2011). Aproximación al uso y tráfico de fauna silvestre en Puerto Carreño, Vichada, Colombia. Ambiente y Desarrollo 14, 63–94.
D’Souza, E., Patankar, V., Arthur, R., Alcoverro, T., and Kelkar, N. (2013). Long-term occupancy trends in a data-poor Dugong population in the Andaman and Nicobar Archipelago. PLoS One 8:e76181. doi: 10.1371/journal.pone.0076181
Dawson, S. M. (2009). “Cephalorhynchus Dolphins,” in Encyclopedia of Marine Mammals, eds W. F. Perrin, B. Würsig, and J. G. M. Thewissen (New York, USA: Elsevier), 191–196.
De la Montaña, E. (2013). Cacería de subsistencia de distintos grupos indígenas de la Amazonía ecuatoriana. Ecosistemas 22, 84–96.
De Lestang, J. N. (1993). “Status of marine mammals in the eastern African region,” in Report to UNEP. Regional Seas Reports and Studies Series, (Nairobi: UNEP).
Debrah, J. S., Ofori-Danson, P. K., and Van Waerebeek, K. (2010). An Update on the Catch Composition and Other Aspects of Cetacean Exploitation in Ghana. Document SC/62/SM10 Presented to the Scientific Committee of the International Whaling Commission. Agadir.
Delisle, A., Kim, M. K., Stoeckl, N., Watkin, L. F., and Marsh, H. (2018). The socio-cultural benefits and costs of the traditional hunting of dugongs Dugong dugon and green turtles Chelonia mydas in Torres Strait, Australia. Oryx 52, 250–261. doi: 10.1017/s0030605317001466
Denkinger, J. (2010). Status of the Amazonian manatee (Trichechus inunguis) in the Cuyabeno Reserve, Ecuador. Avances 2, B29–B34.
Dethmers, K. E. M., and Baxter, P. W. J. (2011). Extinction risk analysis of exploited green turtle sticks in the Indo-Pacific. Anim. Conserv. 14, 140–150.
Di Piazza, A., and Pearthree, E. (2001). An island for gardens, an island for birds and voyaging: A settlement pattern for Kiritimati and Tabuaeran, two” mystery islands” in the northern Lines, Republic of Kiribati. J. Polynesian Soc. 110, 149–170.
Dobson, A. D. M., Milner-Gulland, E. J., Ingram, D. J., and Keane, A. (2019). A framework for assessing impacts of wild meat hunting practices in the Tropics. Hum. Ecol. 47, 449–464. doi: 10.1007/s10745-019-0075-6
Dodman, T., Ndiaye, M. D. D., and Sarr, K. (2008). Conservation Strategy for the West African Manatee. Nairobi: UNEP.
Dolar, M. L. L. (1999). “Sulu-Sulawesi Cetacean Project,” in Report submitted to WWF-US, 27 August 1999, (Gland: WWF).
Dolar, M. L. L., Leatherwood, S. J., Wood, C. J., Alava, M. N. R., Hill, C. L., and Aragones, L. V. (1994). Directed fisheries for cetaceans in the Philippines. Rep. Int. Whaling Commiss. 44, 439–449.
Durbin, J. C., and Ralambo, J. A. (1994). The role of local people in the successful maintenance of protected areas in Madagascar. Environ. Conserv. 21, 115–120. doi: 10.1017/s037689290002453x
Eckert, K. L. (2001). “Status and distribution of the leatherback turtle, Dermochelys coriacea, in the wider Caribbean region,” in Proceedings from: Marine Turtle Conservation in the Wider Caribbean Region: A Dialogue for Effective Regional Management Santo Domingo, Dominican Republic 16-18 November 1999, (Santo Domingo: Regional Management Santo Domingo), 24–31.
Eckert, K., Wallace, B. P., Frazier, J. G., Eckert, S. A., and Pritchard, P. C. H. (2012). “Synopsis of the biological data on the leatherback sea turtle (Dermochelys coriacea),” in Biological Technical Publication BTP-R4015-2012, (Washington, D.C: U.S. Fish and Wildlife Service, USA).
Ekanayake, E. M. L., Ranawana, K. B., Kapurusinghe, T., Premakumara, M. G. C., and Saman, M. M. (2002). Marine Turtle Conservation in Rekawa Turtle Rookery in southern Sri Lanka. Ceylon J. Sci. 30, 79–88.
El Bizri, H. R., Morcatty, T., Ferreira, J. C., Mayor, P., Neto, C. F. A. V., Valsecchi, J., et al. (2020). Social and biological correlates of wild meat consumption and trade by rural communities in the Jutai River Basin, Central Amazonia. J. Ethnobiol. 40, 183–201. doi: 10.2993/0278-0771-40.2.183
Fa, J. E., Seymour, S., Dupain, J., Amin, R., Albrechtsen, L., and Macdonald, D. (2006). Getting to grips with the magnitude of exploitation: Bushmeat in the Cross–Sanaga rivers region, Nigeria and Cameroon. Biol. Conserv. 129, 497–510. doi: 10.1016/j.biocon.2005.11.031
Ferraro, P. (2007). A global survey of sea turtle payment incentive programs NOAA Paper Global Survey. La Jolla, CA: NOAA.
Fielding, R. (2018). The Wake of the Whale: Hunter Societies in the Caribbean and North Atlantic. Boston: Harvard University Press.
Fielding, R., and Barrientos, C. (2021). History of whaling in Annobón, Equatorial Guinea, and new evidence of its continued occurrence. J. Cetacean Res. Manage. 22, 29–37. doi: 10.47536/jcrm.v22i1.217
Fielding, R., and Evans, D. W. (2014). Mercury in Caribbean dolphins Stenella longirostris and Stenella frontalis caught for human consumption off St. Vincent, West Indies. Mar. Poll. Bull. 89, 30–34. doi: 10.1016/j.marpolbul.2014.10.040
Fielding, R., and Kiszka, J. J. (2021). Artisanal and aboriginal subsistence whaling in Saint Vincent and the Grenadines (Eastern Caribbean): history, catch characteristics, and needs for research and management. Front. Mar. Sci. 8:668597. doi: 10.3389/fmars.2021.668597
Findlay, K. P., Cockcroft, V. G., and Guissamulo, A. T. (2011). Dugong abundance and distribution in the Bazaruto Archipelago, Mozambique. Afr. J. Mar. Sci. 33, 441–462. doi: 10.2989/1814232x.2011.637347
Fleming, E. H. (2001). Swimming against the tide. Recent surveys of exploitation, trade, and management of marine turtles in the Northern Caribbean. Washington, D.C.: TRAFFIC North America, 161.
Flores, P. A. C., and Da Silva, V. M. F. (2009). “Tucuxi and Guiana Dolphin: Sotalia fluviatilis and S. guianensis,” in Encyclopaedia of Marine Mammals, eds W. F. Perrin, B. Würsig, and J. G. M. Thewissen (New York, NY: Elsevier), 1188–1192.
Flores, P. A. C., Trujillo, F., Rocha-Campos, C., Marini-Filho, O. J., Da Silva, V. M. F., Martin, A. R., et al. (2008). The Status of “piracatinga” Fishery Using Amazon Botos as Bait in South America. Document SC/60/SM17 Presented to the International Whaling Commission. Santiago.
Forero-Medina, G., Ferrara, C. R., Vogt, R. C., Fagundes, C. K., Balestra, R. A. M., Andrade, P. C. M., et al. (2021). On the future of the giant South American river turtle Podocnemis expansa. Oryx 55, 73–80.
Franzini, A. M., Castelblanco-Martínez, D. N., Rosas, F. C. W., and Da Silva, V. M. F. (2013). What do local people know about Amazonian manatees? Traditional ecological knowledge of Trichechus inunguis in the Oil Province of Urucu, AM, Brazil. Natureza Conservação 11, 75–80. doi: 10.4322/natcon.2013.012
Frazier, J. (1980). Exploitation of marine turtles in the Indian Ocean. Hum. Ecol. 8, 329–370. doi: 10.3390/genes11030278
Frazier, J. G., and Mundkur, T. (1990). The dugong, Dugong dugon (Müller) in the Gulf of Kutch, Gujarat. J. Bombay Nat. Histor. Soc. 87, 368–379.
Fretey, J., Segniagbeto, G. H., and Soumah, M. (2007). Presence of sea turtles in traditional pharmacopoeia and beliefs of West Africa. Mar. Turtle Newsletter 2007, 23–25.
Frías-Espericueta, M. G., Osuna-López, J. I., Ruiz-Telles, A., Quintero-Alvarez, J. M., López-López, G., Izaguirre-Fierro, G., et al. (2006). Heavy metals in the tissues of the sea turtle Lepidochelys olivacea from a nesting site of the northwest coast of Mexico. Bull. Environ. Contaminat. Toxicol. 77, 179–185. doi: 10.1007/s00128-006-1048-1
Fuentes, M. M. P. B., Bell, I., Hagihara, R., Hamann, M. I., Hazel, J., Huth, A., et al. (2015). Improving estimates of in-water marine turtle abundance by adjusting aerial survey counts for perception and availability biases. Exp. Mar. Biol. Ecol. 471, 77–83. doi: 10.1016/j.jembe.2015.05.003
Fukuda, Y., Manolis, C., and Appel, K. (2014). Management of human-crocodile conflict in the Northern Territory, Australia: review of crocodile attacks and removal of problem crocodiles. J. Wildlife Manage. 78, 1239–1249. doi: 10.1002/jwmg.767
Fukuda, Y., Moritz, C., Jang, N. C., Webb, G., Campbell, H., Christian, K., et al. (2022). Environmental resistance and habitat quality influence dispersal of the saltwater crocodile. Mol. Ecol. 31, 1076–1092. doi: 10.1111/mec.16310
Fukuda, Y., Webb, G., Manolis, C., Delaney, R., Letnic, M., Lindner, G., et al. (2011). Recovery of saltwater crocodiles following unregulated hunting in tidal rivers of the Northern Territory, Australia. J. Wildlife Manage. 75, 1253–1266. doi: 10.1002/jwmg.191
Fukuda, Y., Webb, G., Manolis, C., Lindner, G., and Banks, S. (2019). Translocation, genetic structure and homing ability confirm geographic barriers disrupt saltwater crocodile movement and dispersal. PLoS One 14:e0205862. doi: 10.1371/journal.pone.0205862
Garland, K. A., and Carthy, R. R. (2010). Changing taste preferences, market demands and traditions in Pearl Lagoon, Nicaragua: A community reliant on green turtles for income and nutrition. Conservat. Soc. 8, 55–72. doi: 10.4103/0972-4923.62675
Gómez, J., van Vliet, N., Restrepo, S., Daza, E., Moreno, J., Cruz-Antia, D., et al. (2016). Use and trade of bushmeat in Colombia. Relevance to rural livelihoods. CIFOR Infobrief 159:4.
Gomez, L., and Krishnasamy, K. (2019). A Rapid Assessment on the Trade in Marine Turtles in Indonesia, Malaysia and Viet Nam. Petaling Jaya: TRAFFIC.
González, C., and Harrison, E. (2012). Report on the 2011 green turtle program at Tortuguero, Costa Rica. Submitted to Sea Turtle Conservancy (Formerly Caribbean Conservation Corporation) and the Ministry of Environment, Energy and Telecommunications of Costa Rica. Costa Rica: Ministry of Environment, 66.
Goodall, R. N. P. (2009). “Peale’s Dolphin: Lagenorhynchus australis,” in Encyclopaedia of Marine Mammals, eds W. F. Perrin, B. Würsig, and J. G. M. Thewissen (New York, NY: Elsevier), 844–847.
Gore, M. A., Kiani, M. S., Ahmad, E., Hussain, B., Ormond, R. F., Siddiqui, J., et al. (2012). Occurrence of whales and dolphins in Pakistan with reference to fishers’ knowledge and impacts. J. Cetacean Res. Manage. 12, 235–247.
Gough, C., Thomas, T., Humber, F., Harris, A., Cripps, G., and Peabody, S. (2009). Vezo fishing: An introduction to the methods used by fishers in Andavadoaka, Southwest Madagascar. London, UK: Blue Ventures Conservation.
Greatorex, Z. F., Olson, S. H., Singhalath, S., Silithammavong, S., Khammavong, K., Fine, A. E., et al. (2016). Wildlife trade and human health in Lao PDR: An assessment of the zoonotic disease risk in markets. PLoS One 11:e0150666. doi: 10.1371/journal.pone.0150666
Guebert, F. M., Barletta, M., and da Costa, M. F. (2013). Threats to sea turtle populations in the Western Atlantic: Poaching and mortality in small-scale fishery gears. J. Coast. Res. 65, 42–47. doi: 10.2112/si65-008.1
Guissamulo, A. T., and Cockcroft, V. G. (1997). Dolphin and Dugong Occurrence and Fisheries Interaction in Maputo and Bazaruto bays, Mozambique. Document SC/49/SM24 Presented to the IWC Scientific Committee. London.
Hagihara, R., Cleguer, C., Preston, S., Sobtzick, S., Hamann, M., Shimada, T., et al. (2016). “Improving the estimates of abundance of dugongs and large immature and adult-sized green turtles in Western and Central Torres Strait,” in Report to the National Environmental Science Programme. Reef and Rainforest Research Centre Limited, Cairns, Australia, (Cairns: NESP), 53. doi: 10.1071/wr04015
Hagihara, R., Jones, R. E., Sobtzick, S., Cleguer, C., Garrigue, C., and Marsh, H. (2018). Compensating for geographic variation in detection probability with water depth improves abundance estimates of coastal marine megafauna. PLoS One 13:e0191476. doi: 10.1371/journal.pone.0191476
Hamann, M., Hong, N. D., Thuoc, P., and Thuhien, B. T. (2006). Distribution and abundance of marine turtles in the Socialist Republic of Viet Nam. Biodivers. Conserv. 15, 3703–3720. doi: 10.1007/s10531-005-4880-4
Hamilton, R. J., Bird, T., Gereniu, C., Pita, J., Ramohia, P. C., et al. (2015). Solomon Islands Largest Hawksbill Turtle Rookery Shows Signs of Recovery after 150 Years of Excessive Exploitation. PLoS One 10:e0121435. doi: 10.1371/journal.pone.0121435
Hancock, J. M., Furtado, S., Merino, S., Godley, B. J., and Nuno, A. (2017). Exploring drivers and deterrents of the illegal consumption and trade of marine turtle products in Cape Verde, and implications for conservation planning. Oryx 51, 428–436. doi: 10.1017/s0030605316000107
Hasan, M. M. (2009). Tourism and conservation of biodiversity: A case study of St Martins Island, Bangladesh. Dhaka: Law Social Justice and Global Development.
Havemann, P., and Smith, R. (2007). Community Based Management of Dugong and Turtle Fisheries. Australia: James Cook University.
Hazevoet, C. J., Monteiro, V., López, P., Varo, N., Torda, G., Berrow, S., et al. (2010). Recent data on whales and dolphins (Mammalia: Cetacea) from the Cape Verde Islands, including records of four taxa new to the archipelago. Zool. Caboverdiana 1, 75–99.
He, F., Zarfl, C., Bremerich, V., Henshaw, A., Darwall, W., Tockner, K., et al. (2017). Disappearing giants: a review of threats to freshwater megafauna. WIREs Water 4:e1208.
Heinsohn, R., Lacy, R. C., Lindenmayer, D. B., Marsh, H., Kwan, D., and Lawler, I. R. (2004). Unsustainable harvest of dugongs in Torres Strait and Cape York (Australia) waters: Two case studies using population viability analysis. Anim. Conserv. 7, 417–425. doi: 10.1017/s1367943004001593
Hines, E., Adulyanukosol, K., and Poochaviranon, S. (2012). “Dugongs in Asia,” in International Strategies for Manatee and Dugong Conservation, eds E. Hines, J. Reynolds, A. Mignucci-Giannoni, L. Aragones, and M. Marmontel (Gainesville: University Press of Florida).
Humber, F., Andriamahefazafy, M., Godley, B. J., and Broderick, A. C. (2015). Endangered, essential and exploited: how extant laws are not enough to protect marine megafauna in Madagascar. Mar. Policy 60, 70–83.
Humber, F., Godley, B. J., and Broderick, A. C. (2014). So excellent a fishe: A global overview of legal marine turtle fisheries. Divers. Distribut. 20, 579–590. doi: 10.1111/ddi.12183
Husar (1975). “A Review of the Literature of the Dugong (Dugong Dugon),” in Wildlife Research Report, 4, (Washington, D.C: U.S. Fish and Wildlife Service), 30.
Ilangakoon, A. (2012). A review of cetacean research and conservation in Sri Lanka. J. Cetacean Res. Manage. 12, 177–183. doi: 10.1111/j.1758-6623.2012.00160.x
Ingram, D. J. (2020). Wild meat in changing times. J. Ethnobiol. 40, 117–130. doi: 10.1371/journal.pone.0045315
Ingram, D. J., Coad, L., Milner-Gulland, E. J., Parry, L., Wilkie, D., Bakarr, M. I., et al. (2021). Wild meat is still on the menu: progress in wild meat research, policy and practice from 2002 – 2020. Annu. Rev. Environ. Resour. 4, 221–254.
IOSEA (2014). Marine Turtle MOU, Illegal Take and Trade of Marine Turtles in the IOSEA Region. MT-IOSEA/SS.7/Doc. 10.1. Nairobi: UN.
IWC (2016). Assessment of online information as a tool to improve the documentation of the availability of marine mammals for consumption and other uses in Southern China. IWC.
Jaaman, S. A., Lah-Anyi, Y. U., and Pierce, G. J. (2008). Directed fisheries for dolphins and dugongs in Sabah, East Malaysia: Past and present. Borneo Sci. 23, 1–20.
Jefferson, T. A., and Van Waerebeek, K. (2002). The taxonomic status of the nominal dolphin species Delphinus tropicalis van Bree, 1971. Mar. Mamm. Sci. 18, 787–818. doi: 10.1111/j.1748-7692.2002.tb01074.x
Jones, K. E., Patel, N. G., Levy, M. A., Storeygard, A., Balk, D., Gittleman, J. L., et al. (2008). Global trends in emerging infectious diseases. Nature 451:990. doi: 10.1038/nature06536
Jones, S. (1967). The dugong Dugong dugon (Müller), its present status in the seas round India with observations on its behaviour in captivity. Int. Zoo Yearbook 7, 215–220. doi: 10.1111/j.1748-1090.1967.tb00398.x
Joseph, J., Nishizawa, H., Alin, J. M., Othman, R., Jolis, G., Isnain, I., et al. (2019). Mass sea turtle slaughter at Pulau Tiga, Malaysia: genetic studies indicate poaching locations and its potential effects. Glob. Ecol. Conserv. 17:e00586. doi: 10.1016/j.gecco.2019.e00586
Kamla, A. T. (2019). Genetic diversity, diet, and habitat quality of the African manatee (Trichechus senegalensis) in the downstream of the Sanaga River watershed, Cameroon. Ph. D. thesis. Florida, FL: University of Florida.
Kataoka, T., Mori, T., Wakai, Y., Palma, J., Yaptinchay, A. A., de Veyra, R., et al. (1995). Dugongs of the Philippines: A report of the joint dugong research and conservation program. May 1995. Japan: Toba Aquarium and the Pawikan Conservation Project.
Keith Diagne, L. (2015). Trichechus senegalensis (errata version published in 2016). The IUCN Red List of Threatened Species 2015: e.T22104A97168578. Gland: IUCN.
Keith Diagne, L. W. (2014). Phylogenetics and Feeding Ecology of the African Manatee, Trichechus senegalensis. Ph. D. thesis. Florida, FL: University of Florida.
Keith-Diagne, L., Eniang, E., Ndjassi, C., Dimbot, J. P., Takoukam, A., Ejimadu, U., et al. (2019). “African Manatee Threat Assessments: Preliminary Results from Five Countries,” in Sirenian Symposium, World Marine Mammal Conference, Barcelona, Spain, (Barcelona: WMMC).
Keith-Diagne, L., Mullié, W., Diagne, T., Djiba, A., and Diagne, A. K. (2017). “The Senegal Stranding Network: Year-round Monitoring of Stranded Cetaceans Detects High Seasonal Mortality and Bycatch Hotspots,” in 22nd Biennial Conference on the Biology of Marine Mammals, Halifax, Nova Scotia, Canada, (Nova: Biology of Marine Mammals).
Kemf, E., Groombridge, B., Abreu, A., and Wilson, A. (2000). Wanted Alive! Marine Turtles in the Wild. Gland: WWF.
Kendall, S. (2013). Caminos para la conservación: monitoreo y manejo de la fauna acuática con la comunidad. Puerto Nariño: Fundación Natutama.
Kiani, M. S., and Van Waerebeek, K. (2015). “A Review of the Status of the Indian Ocean Humpback Dolphin (Sousa plumbea) in Pakistan,” in Advances in Marine Biology, Vol. 72, eds T. A. Jefferson and B. E. Curry (Oxford, UK: Academic Press), 201–228. doi: 10.1016/bs.amb.2015.09.002
Kile, N., Lam, M., Davis, D. C., and Donnelly, R. J. (2000). “Managing the live reef food fish trade in Solomon Islands: The role of village decision-making systems in Ontong Java, Roviana and Morovo Lagoons,” in Discussion Paper No. 2. Report to Australian Centre for International Agricultural Research, ACIAR, Canberra, (Canberra: ACIAR), 28.
Kinch, J. (2008). “An assessment of dugong Dugong dugon resources in the autonomous region of Bouganville, Papua New Guinea,” in Unpublished report prepared for Conservation International, Melanesian Centre for Biodiversity Conservation, Atherton, Queensland Australia, (Atherton: MCBC), 35.
Kirkland, M., Eisenberg, C., Bicerra, A., Bodmer, R., Mayor, P., and Axmacher, J. (2020). Sustainable wildlife extraction and the impacts of socio-economic change among the Kukama-Kukamilla people of the Pacaya-Samiria National Reserve. Peru Oryx 54, 260–269.
Kiszka, J. (2014). “Bycatch Assessment of the West Indian Manatee (Trichechus Manatus) and Other Megafauna In Artisanal Fisheries of the Caribbean,” in Report to SPAW-RAC, 41. Available online at: http://www.car-spaw-rac.org/?lang=en
Kumarran, R. P. (2012). Cetaceans and cetacean research in India. J. Cetacean Res. Manage. 12, 159–172.
Lagueux, C. J., Campbell, C. L., and Strindberg, S. (2014). Artisanal Green Turtle, Chelonia mydas, Fishery of Caribbean Nicaragua: I. Catch Rates and Trends, 1991–2011. PLoS One 9:e94667. doi: 10.1371/journal.pone.0094667
Lagueux, C. J., Campbell, C. L., and Strindberg, S. (2017). Artisanal green turtle (Chelonia mydas) fishery of Caribbean Nicaragua: II. Characterization and trends in size, sex, and maturity status of turtles killed, 1994–2011. Mar. Biol. 164:60.
Lagueux, C., Campbell, C., and Lauck, E. (2005). Management strategy for marine turtle conservation on the Caribbean Coast of Nicaragua. Species Status Report. Gland: WWF.
Lam, T., Lingxu, S., Takahashi, S., and Burgess, E. A. (2011). Market Forces: An Examination of Marine Turtle Trade in China and Japan. Hong Kong: TRAFFIC East Asia.
Lang, J., Chowfin, S., and Ross, J. P. (2019). Gavialis gangeticus (errata version published in 2019). The IUCN Red List of Threatened Species 2019: e.T8966A149227430. Gland: IUCN.
Leatherwood, S., Mcdonald, D., Prematunga, W. P., Girton, P., Ilangaakoon, A., and Mcbrearty, D. (1991). Records of the “Blackfish” (Killer, False Killer, Pygmy Killer and Melon-headed Whales) in the Indian Ocean. UNEP Mar. Mamm. Technical Rep. 3, 33–65.
Lee, P., and Nijman, V. (2015). Trade in dugong parts in Southern Bali. J. Ma. Biol. Assoc. U K. 95, 1717–1721. doi: 10.1017/s0025315415001423
Lee, S. M., Choi, Y. Y., Min, M. S., Lee, H., and Lee, M. Y. (2019). Molecular species identification of whale meat in South Korean markets. Genet. Mol. Res. 18:GMR18171. doi: 10.1098/rspb.2000.1128
Leeney, R. H., Dia, I. M., and Dia, M. (2015). Food, pharmacy, friend? Bycatch, direct take and consumption of dolphins in West Africa. Hum. Ecol. 43, 105–118. doi: 10.1007/s10745-015-9727-3
Lescrauwaet, A. C., and Gibbons, J. (1994). Mortality of small cetaceans and the crab bait fishery in the Magallanes area of Chile since 1980. Rep. Int. Whaling Commis. 15, 485–494.
Lewison, R., and Moore, J. (2012). “Improving Interview-Based Assessments of Sea Turtle and Marine Mammal Bycatch in West Africa: Putting Fishing Activity into a Socio-Economic Context,” in Project Report, San Diego State University and Southwest Fisheries, (San Diego: San Diego State University).
Lin, L., Li, S., Chen, M., Parham, J. F., and Shi, H. (2021). Sea turtle demand in China threatens the survival of wild populations. iScience 24:102517. doi: 10.1016/j.isci.2021.102517
Lo, C., Chin, L. T., Chu, C. S., Wang, Y. T., Chan, K. W., and Yang, W. C. (2013). Rapid immune colloidal gold strip for cetacean meat restraining illegal trade and consumption: implications for conservation and public health. PLoS One 8:e60704. doi: 10.1371/journal.pone.0060704
Lopes, G. P., Valsecchi, J., Vieira, T. M., do Amaral, P. V., and da Costa, E. W. M. (2012). Hunting and hunters in lowland communities in the region of the Middle Solimões, Amazonas, Brasil. Sci. Magazine UAKARI 8:7. doi: 10.31420/uakari.v8i1.120
Lynch, A. J., Cooke, S. J., Deines, A. M., Bower, S. D., Bunnell, D. B., Cowx, I. G., et al. (2016). The social, economic, and environmental importance of inland fish and fisheries. Environ. Rev. 24, 115–121. doi: 10.1111/ele.12949
Madge (1998). Therapeutic landscapes of the Jola, The Gambia, West Africa. Health Place 4, 293–311. doi: 10.1016/s1353-8292(98)00033-1
Magnino, S., Colin, P., Dei-Cas, E., Madsen, M., McLauchlin, J., Nöckler, K., et al. (2009). Biological risks associated with consumption of reptile products. Int. J. Food Microbiol. 134, 163–175. doi: 10.1016/j.ijfoodmicro.2009.07.001
Maigret, J. (1994). Marine mammals and fisheries along the West African coast. Rep. Int. Whaling Commiss. 15, 307–316.
Mainka, S. A., and Trivedi, M. (2002). Links between Biodiversity Conservation, Livelihoods and Food Security: The sustainable use of wild species for meat. Gland: IUCN, 135.
Mali, I., Vandewege, M. W., Davis, S. K., and Forstner, M. R. J. (2014). Magnitude of the freshwater turtle exports from the US: Long term trends and early effects of newly implemented harvest management regimes. PLoS One 9:e86478. doi: 10.1371/journal.pone.0086478
Mancini, A., and Koch, V. (2009). Sea turtle consumption and black market trade in Baja California Sur, Mexico. Endanger. Species Res. 7, 1–10. doi: 10.3354/esr00165
Mangel, J. C., Alfaro-Shigueto, J., Van Waerebeek, K., Cáceres, C., Bearhop, S., Witt, M. J., et al. (2010). Small cetacean captures in Peruvian artisanal fisheries: High despite protective legislation. Biol. Conservat. 143, 136–143.
Manolis, C. (2005). Long-distance movement by a saltwater crocodile. Crocodile Special. Group Newsl. 24:18.
Marcovaldi, M. A., and dei Marcovaldi, G. G. (1999). Marine turtles of Brazil: the history and structure of Projeto TAMAR-IBAMA. Biol. Conserv. 91, 35–41.
Marmontel, M., de Souza, D., and Kendall, S. (2016). Trichechus inunguis. The IUCN Red List of Threatened Species 2016: e.T22102A43793736. Gland: IUCN, doi: 10.2305/IUCN.UK.2016-2.RLTS.T22102A43793736.en
Marsh, H., Grayson, J., Grech, A., Hagihara, R., and Sobtzick, S. (2015). Re-evaluation of the sustainability of a marine mammal harvest by indigenous people using several lines of evidence. Biol. Conserv. 192, 324–330.
Marsh, H., Harris, A. N. M., and Lawler, I. R. (1997). The sustainability of the indigenous dugong fishery in Torres Strait, Australia/Papua New Guinea. Conserv. Biol. 11, 1375–1386.
Marsh, H., Lawler, I. R., Kwan, D., Delean, S., Pollock, K., and Alldredge, M. (2004). Aerial surveys and the potential biological removal technique indicate that the Torres Strait dugong fishery is unsustainable. Anim. Conserv. 7, 435–443.
Marsh, H., O’Shea, T. J., and Reynolds, J. E. (2011). The ecology and conservation of Sirenia: dugongs and manatees. Cambridge: Cambridge University Press, 521.
Marsh, H., Penrose, H., Eros, C., and Hugues, J. (2002). “Dugong: Status report and action plans for countries and territories,” in Report UNEP/DEWA/RS.02-1, (Nairobi: UNEP).
Marsh, H., Rathbun, G. B., O’Shea, T. J., and Preen, A. R. (1995). Can dugongs survive in Palau? Biol. Conserv. 72, 85–89. doi: 10.1016/0006-3207(94)00068-2
Martin, A. R., and Da Silva, V. M. F. (2021). Amazon river dolphins Inia geoffrensis are on the path to extinction in the heart of their range. Oryx 2021, 1–5.
Matthews, E. (2003). “Local knowledge about dugongs in Palau,” in PCS Report 2003-04, (Koror: Palau Conservation Society), 21.
Mayaka, T. B., Koh-Dimbot, J.-P., and Keith-Diagne, L. W. (2019). Occurrence patterns of African manatees, conflicts with humans, and local perception in the Southern Korup Area, Cameroon. Aquat. Conserv. Mar. Freshw. Ecosyst. 29, 1801–1813.
Mayaka, T., Takoukam, A. K., and Self-Sullivan, C. (2015). Using Pooled Local Expert Opinions (PLEO) to Discern Patterns in Sightings of Live and Dead Manatees (Trichechus senegalensis, Link 1785) in Lower Sanaga Basin, Cameroon. PLoS One 10:e0128579. doi: 10.1371/journal.pone.0128579
McCauley, D. J., Pinsky, M. L., Palumbi, S. R., Estes, J. A., Joyce, F. H., and Warner, R. R. (2015). Marine defaunation: animal loss in the global ocean. Science 347:1255641. doi: 10.1126/science.1255641
McGovern, P., Wade, A., Diagne, T., and Keith-Diagne, L. W. (2021). Death and hope on the coast of Central Senegal. Afr. Sea Turtle Newsl. 16, 16–20.
McLaughlin, J. B., Sobel, J., Lynn, T., Funk, E., and Middaugh, J. P. (2004). Botulism type E outbreak associated with eating a beached whale, Alaska. Emerg. Infect. Dis. 10, 1685–1687. doi: 10.3201/eid1009.040131
Meerman, J. (2005). “Compilation of Information on Biodiversity in Belize,” in Report to Inbio and Forest Department of Belize, Belize, (Belize: Forest Department of Belize).
Mejías-Balsalobre, C., Restrepo, J., Borges, G., García, R., Rojas-Cañizales, D., Barrios-Garrido, H., et al. (2021). Local community perceptions of sea turtle egg use in Tortuguero, Costa Rica. Ocean Coast. Manage. 201:105423. doi: 10.1016/j.ocecoaman.2020.105423
Méry, S., Charpentier, C., Auxiette, G., and Pelle, E. (2009). A dugong bone mound: the Neolithic ritual site on Akab in Umm al-Quwain, United Arab Emirates. Antiquity 83, 696–708. doi: 10.1017/s0003598x00098926
Milner-Gulland, E. J., Bennett, E. L., and the Samwm Group. (2003). Wild meat: the bigger picture. Trends Ecol. Evolut. 18, 351–357. doi: 10.1016/s0169-5347(03)00123-x
Mintzer, V. J., Diniz, K., and Frazer, T. K. (2018). The use of aquatic mammals for bait in global fisheries. Front. Mar. Sci. 5:191. doi: 10.3389/fmars.2018.00191
Mintzer, V. J., Martin, A. R., da Silva, V. M. F., Barbour, A. B., Lorenzen, K., and Frazer, T. K. (2012). Effect of illegal harvest on apparent survival of Amazon River dolphins Inia geoffrensis. Biol. Conserv. 158, 280–286. doi: 10.1016/j.biocon.2012.10.006
Mogollones, S. C., Rodriguez, D. J., Hernandez, O., and Barreto, G. R. (2010). A demographic study of the Arrau Turtle (Podocnemis expansa) in the Middle Orinoco River, Venezuela. Chelonian Conserv. Biol. 9, 79–89. doi: 10.2744/ccb-0778.1
Mohd Jani, J., Jamalludin, M. A., and Long, S. L. (2020). To ban or not to ban? Reviewing an ongoing dilemma on sea turtle egg trade in Terengganu, Malaysia. Front. Mar. Sci. 6:762. doi: 10.3389/fmars.2019.00762
Moheli Marine Park (2009). Les braconniers de tortues deficient les efforts de protection du PMM. Mwana Wa Nyamba Journal du Parc Marin de Moheli N0001 Octobre/novembre 2009, Comoros. Nioumachoua: Moheli Marine Park.
Montiel-Villalobos, M. G., and Barrios-Garrido, H. (2005). Observaciones sobre la distribucion y situacion actual del Manati Trichechus manatus (Sirenia: Trichechidae) en el Sistema del Lago de Maracaibo. Anartia 18, 1–12.
Montoya-Ospina, R. A., Caicedo-Herrera, C., Millan-Sanchez, S. L., Mignucci-Giannoni, A. A., and Lefebvre, L. W. (2001). Status and distribution of the West Indian manatee, Trichechus manatus manatus, in Colombia. Biol. Conserv. 102, 117–129.
Morales, V. R., and Vargas, P. (1996). Legislation protecting marine turtles in Peru. Mar. Turtle Newsl. 75, 22–23.
Muir, C. E., and Kiszka, J. J. (2012). “Eastern African dugongs,” in Sirenian conservation: issues and strategies in developing countries, eds E. M. Hines et al. (Gainesville: University Press of Florida), 84–90.
Murphy, P. F., Van Waerebeek, K., and Jallow, A. (1997). Cetaceans in Gambian Coastal Waters. Document SC/49/SM11 Presented to the IWC Scientific Committee, September 1997. Bournemouth, 8. doi: 10.13140/2.1.4468.6405
Muschett, G. (2008). Distribución y Estudios Genéticos del Manatí (Trichechus manatus) en la cuenca hidrográfica del canal de Panamá. Ph. D. thesis. Santiago: Pontificia Universidad Católica de Chile.
Mustika, P. L. K. (2006). Marine mammals in the Savu Sea (Indonesia): Indigenous knowledge, threat analysis and management options. Ph. D. thesis. Australia: James Cook University.
Muttenzer, F. (2007). Different kinds of people of the sea: Écologie, mobilité et ethnicité chez les Vezo de Madagascar. Etude de gouvernance locale des tortues marines aux îles Barren, Maintirano, Madagascar. Maintirano: RUIG-GIAN.
Mvele, C., and Arrowood, H. (2013). Rapport de vente illegale de la viande de brousse, Marché Isaac, Lambaréné. Libreville: Organisation Ecotouristique de Lac Oguemoué.
Mwango’mbe, M. G., Spilsbury, J., Trott, S., Nyunja, J., Wambiji, N., Collins, T., et al. (2021). Cetacean Research and Citizen Science in Kenya. Front. Mar. Sci. 8:642399. doi: 10.3389/fmars.2021.642399
Nabhan, G. P. (2003). Singing the Turtle to Sea: The Comcáac (Seri) Art and Science of Reptiles. Los Angeles, CA: University of California Press.
Nahill, B., von Weller, P., and Barrios-Garrido, H. (2020). “The Global Tortoiseshell Trade,” in Global Tortoiseshell Report. Available online at: https://www.tooraretowear.org
Nair, R. V., Lal Mohan, R. S., and Rao, K. S. (1975). Dugong dugong Dugon. CMFRI Bull. 26, 1–49. doi: 10.2307/3504027
Nasr, D., Shawky, A. M., and Vine, P. (2019). “Status of Red Sea Dugongs,” in Oceanographic and biological aspects of the Red Sea, eds N. Rasul and I. Stewart (Cham: Springer).
Nithyanandan, M., and Bohadi, Y. (2021). Incidental mortality events of the Indo-Pacific Finless Porpoise, Neophocaena phocaenoides in Kuwait, Northwestern Arabian Gulf. Zool. Middle East 67, 373–376.
NMFS and USFWS (2020). “Endangered Species Act status review of the leatherback turtle (Dermochelys coriacea),” in Report to the National Marine Fisheries Service Office of Protected Resources and U.S. Fish and Wildlife Service, (Washington, D.C: U.S. Fish and Wildlife Service).
Nzuki, S. (2004). “KESCOM surveying trade in turtle products,” in Marine turtle update: recent news from the WWF Africa and Madagascar marine turtle programme of Guinea islands, eds S. L. Humphrey and A. Wilson (Gland: WWF International), 15–16.
Nzuki, S. (2005). “KESCOM study exposes rampant turtle trade,” in Marine turtle update: Recent news from the WWF Africa and Madagascar marine turtle programme, eds S. L. Humphrey and A. Wilson (Gland: WWF International), 7–9.
Ofori-Danson, P. K., Van Waerebeek, K., and Debrah, S. (2003). A survey for the conservation of dolphins in Ghanaian coastal waters. J. Ghana Sci. Assoc. 5, 45–54.
Olmedo, G. G., and Farnés, O. C. (2004). “Cultural, social and nutritional value of sea turtles in Cuba,” in Research Report, (Cuba: Universidad de la Habana), 44.
Oremus, L., Leqata, J., and Scott Baker, C. (2015). Resumption of traditional drive hunting of dolphins in the Solomon Islands in 2013. R. Soc. Open Sci 2:140524. doi: 10.1098/rsos.140524
O’Shea, T. J., Correa-Viana, M., Ludlow, M. E., and Robinson, J. G. (1988). Distribution, status, and traditional significance of the West Indian manatee Trichechus manatus in Venezuela. Biol. Conserv. 46, 281–301.
Pantoja, T. M. A. (2015). O peixe-boi da Amazônia no baixo Javari, AM - Brasil. Conhecimento local, uso de habitat e conservação. Ph. D. Thesis. Belém: Universidade Federal do Pará, 202.
Peñaloza, C. L., Hernández, O., Espín, R., Crowder, L. B., and Barreto, G. R. (2013). Harvest of Endangered Sideneck River Turtles (Podocnemis spp.) in the Middle Orinoco, Venezuela. Copeia 1, 111–120.
Perea-Sicchar, C. M., Velásquez-Varela, L. J., Sánchez-Babilonia, J., Espinoza- Azan, M., Lee-Richardson, D., and Sigler, L. (2011). Manejo y rehabilitación del manatí Amazónico (Trichechus inunguis) en cautiverio en el Perú. Ciencia Amazónica 1, 104–113.
Perrin, W. F., Reeves, R. R., Dolar, M. L. L., Jefferson, T. A., Marsh, H., Wang, J. Y., et al. (2002). Report of the Second Workshop on The Biology and Conservation of Small Cetaceans and Dugongs of South East Asia. Bonn: UNEP-CMS Secretariat, 161.
Pheasey, H., Roberts, D. L., Rojas-Cañizales, D., Mejías-Balsalobre, C., Griffiths, R. A., and Williams-Guillen, K. (2020). Using GPS-enabled decoy turtle eggs to track illegal trade. Curr. Biol. 30, R1066–R1068. doi: 10.1016/j.cub.2020.08.065
Pilcher, N. J., Adulyanukosol, K., Das, H., Davis, P., Hines, E., et al. (2017). A low-cost solution for documenting distribution and abundance of endangered marine fauna and impacts from fisheries. PLoS One 12:e0190021. doi: 10.1371/journal.pone.0190021
Pilcher, N.J., and Williams, J. (2018). Assessment of the Status, Scope and Trends of the Legal and Illegal International Trade in Marine Turtles, its Conservation Impacts, Management Options and Mitigation Priorities in Mozambique. Report to the CITES Secretariat Project S-527. SSFA/2018/DKA. Geneva: CITES, 69.
Pineda-Catalan, O., Mendez, M., Gleizer, A., García-Dávila, C., Aguirre, A. A., Pinedo-Vasquez, M., et al. (2012). Conservation genetics of harvested river turtles, Podocnemis expansa and Podocnemis unifilis, in the Peruvian Amazon: All roads lead to Iquitos. Mitochondrial DNA 23, 230–238. doi: 10.3109/19401736.2012.674115
Platt, S. G., Platt, K., Ko Ko, W., Myo Myo, K., and Soe, M. W. (2012). Estuarine crocodiles in southern Myanmar. Crocodile Special. Group Newsl. 31, 18–20.
Poovorawan, Y., Pyungporn, S., Prachayangprecha, S., and Makkoch, J. (2013). Global alert to avian influenza virus infection: From H5N1 to H7N9. Pathog. Glob. Health 107, 217–223. doi: 10.1179/2047773213Y.0000000103
Porter, L., and Lai, H. (2017). Marine Mammals in Asian Societies; Trends in Consumption, Bait and Traditional Use. Front. Mar. Sci. 4:47. doi: 10.3389/fmars.2017.00047
Poti, M., Long, S. L., Rusli, M. U., Mohd Jani, J., Hugé, J., and Dahdouh-Guebas, F. (2021). Changing trends and perceptions of sea turtle egg consumption in Redang Island, Malaysia. Ecol. Soc. 26:14.
Powell, J. A. (1996). The distribution and biology of the West African Manatee (Trichechus senegalensis, Link1795) Unpublished report. Nairobi: United Nations Environmental Program.
Quiñones, J., Carman, V. G., Zeballos, J., Purca, S., and Mianzan, H. (2010). Effects of El Niño-driven environmental variability on black turtle migration to Peruvian foraging grounds. Hydrobiologia 645, 69–79. doi: 10.1007/s10750-010-0225-8
Quiñones, J., Quispe, S., and Galindo, O. (2017). Illegal capture and black market trade of sea turtles in Pisco, Peru: the never-ending story. Latin Am. J. Aquat. Res. 45, 615–621. doi: 10.3856/vol45-issue3-fulltext-11
Quintana-Rizzo, E., and Reynolds, J. E. I. (2010). “Regional management plan for the West Indian manatee (Trichechus manatus),” in CEP Tech Rep, (Kingston: United Nations Environment Programme).
Rajakaruna, R. S., Naveen, D. M., Dissanayake, J., Ekanayake, E. M. L., and Ranawana, K. B. (2009). Sea turtle conservation in Sri Lanka: Assessment of knowledge, attitude and prevalence of consumptive use of turtle products among coastal communities. Ind. Ocean Turtle Newsl. 10, 1–13.
Rakotondrazafy, A. M. N. A., and Andrianasolo, R. M. (2012). Evaluation préliminaire de la filière tortue marine dans la baie de Moramba, les zones de Marovasa Be et d’Anjajavy. Cétamada 2012:11.
Razafindrakoto, Y., Andrianarivelo, N., and Rosenbaum, H. C. (2004). Sightings, catches and other records of IndoPacific humpback dolphins in the coastal waters of Madagascar. Aquat. Mamm. 130, 103–110. doi: 10.1578/am.30.1.2004.103
Read, A. J., Van Waerebeek, K., Reyes, J. C., McKinnon, J. S., and Lehman, L. C. (1988). The exploitation of small cetaceans in Coastal Peru. Biol. Conserv. 46, 53–70. doi: 10.1016/0006-3207(88)90108-5
Reeve-Arnold, K. E., Keeping, J. A., Cockcroft, V. G., and Guissamulo, A. (2020). New strandings of True’s beaked whale, Mesoplodon mirus, in Mozambique and their destiny as marine bushmeat. Western Ind. Ocean J. Mar. Sci. 19, 167–173. doi: 10.4314/wiojms.v19i2.12
Reeves, R. R., Tuboku-Metzger, D., and Kapindi, R. A. (1988). Distribution and exploitation of manatees in Sierra Leone. Oryx 22, 75–84. doi: 10.1017/s0030605300027538
Richardson, P. B., Broderick, A. C., Campbell, L. M., Godley, B. J., and Ranger, S. (2006). Marine turtle fisheries in the UK overseas territories of the Caribbean: Domestic legislation and the requirements of multilateral agreements. J. Int. Wildlife Law Policy 9, 223–246. doi: 10.1080/13880290600764935
Richardson, P. B., Bruford, M. W., Calosso, M. C., Campbell, L. M., Clerveaux, W., Formia, A., et al. (2009). Marine turtles in the Turks and Caicos Islands: Remnant rookeries, regionally significant foraging stocks, and a major turtle fishery. Chelonian Conserv. Biol. 8, 192–207. doi: 10.2744/ccb-0871.1
Ripple, W. J., Wolf, C., Newsome, T. M., Betts, M. G., Ceballos, G., Courchamp, F., et al. (2019). Are we eating the world’s megafauna to extinction? Conserv. Lett. 2019:e12627.
Riskas, K. A., Tobin, R. C., Fuentes, M. M., and Hamann, M. (2018). Evaluating the threat of IUU fishing to sea turtles in the Indian Ocean and Southeast Asia using expert elicitation. Biol. Conserv. 217, 232–239.
Robards, M. D., and Reeves, R. R. (2011). The global extent and character of marine mammal consumption by humans: 1970–2009. Biol. Conserv. 144, 2770–2786. doi: 10.1016/j.biocon.2011.07.034
Romero, A., Agudo, A. I., Green, S. M., and Notarbartolo di Sciara, G. (2001). “Cetaceans of Venezuela: their distribution and conservation status”. NOAA Technic. Rep. NMFS 151, 1–60.
Romero, A., Agudo, I., and Green, S. M. (1997). Exploitation of cetaceans in Venezuela. Rep. Int. Whaling Commis. 47, 735–746.
Romero, A., and Creswell, J. (2005). “In the land of the mermaid: how culture, not ecology, influenced marine mammal exploitation in the southeastern Caribbean,” in Environmental Issues in Latin America and the Caribbean, eds A. Romero and S. E. West (Netherlands: Springer), 1–30. doi: 10.1111/j.1748-7692.1989.tb00210.x
Rowcliffe, J. M., Milner-Gulland, E. J., and Cowlishaw, G. (2005). Do bushmeat consumers have other fish to fry? TRENDS Ecol. Evolut. 20, 274–276. doi: 10.1016/j.tree.2005.03.007
Rudrud, R. W. (2010). Forbidden marine turtles: Traditional laws pertaining to sea turtle consumption in Polynesia (Including the Polynesian Outliers). Conserv. Soc. 8:84.
Said, S. S., Jiddawi, N., Salmin, Y. N., and Cockcroft, V. (2020). Assessment of the status, threats and management of dugongs along coastal waters of Pemba Island, Zanzibar. Int. J. Acad. Multidiscipl. Res. 4, 97–106.
Sardeshpande, M., and MacMillan, D. (2019). Sea turtles support sustainable livelihoods at Ostional, Costa Rica. Oryx 53, 81–91.
Segniagbeto, G. H., Ayissi, I., Bamy, I. L., Debrah, J., Djiba, A., Dossou-Bodjrenou, J., et al. (2019). On the Utilisation of By-caught, Hunted and Stranded Cetaceans in West Africa. Document SC/May19/AAWW/04, IWC Scientific Committee Meeting. Nairobi, 14.
Segniagbeto, G. H., Van Waerebeek, K., Bowessidjaou, J. E., Ketoh, K., Kpatcha, T., Okoumassou, K., et al. (2014). Annotated checklist and fisheries interactions of cetaceans in Togo, with evidence of Antarctic minke whale in the Gulf of Guinea. Integrat. Zool. 9, 378–390. doi: 10.1111/1749-4877.12011
Self-Sullivan, C., and Mignucci-Giannoni, A. A. (2012). “West Indian Manatees (Trichechus manatus) in the Wider Caribbean Region,” in Sirenian conservation: issues and strategies in developing countries, eds E. M. Hines et al. (Gainesville, USA: University Press of Florida), 36–46. doi: 10.1371/journal.pone.0044517
Senko, J., Mancini, A., Seminoff, J. A., and Koch, V. (2014). Bycatch and directed harvest drive high green turtle mortality at Baja California Sur, Mexico. Biol. Conserv. 169, 24–30. doi: 10.1016/j.biocon.2013.10.017
Shaver, D. J., and Rubio, S. (2008). Post-nesting movement of wild and head-started Kemp’s ridley sea turtles Lepidochelys kempii in the Gulf of Mexico. Endanger. Species Res. 4, 43–55. doi: 10.3354/esr00061
Sinha, R. K. (2002). An alternative to dolphin oil as a fish attractant in the Ganges River system: Conservation of the Ganges River dolphin. Biol. Conserv. 107, 253–257. doi: 10.1016/s0006-3207(02)00058-7
Smith, A. M., and Smith, B. D. (1998). Review of status and threats to river cetaceans and recommendations for their conservation. Environ. Rev. 6, 189–206. doi: 10.1007/s13280-014-0534-7
Sohou, Z., Dossou-Bodjrenou, J., Tchibozo, S., Chabi-Yaouré, F., Sinsin, B., and Van Waerebeek, K. (2013). Biodiversity and Status of Cetaceans in Benin, West Africa: An Initial Assessment. West Afr. J. Appl. Ecol. 21, 121–134.
Soto, A. (2007). Caza del manatí amazónico en la Reserva Nacional Pacaya Samiria. Lima: Universidad Nacional Agraria La Molina, 27.
Souza, D. A. (2015). Peixe-boi da Amazônia (Trichechus inunguis Natterer 1883): Mortalidade e uso do habitat na Reserva de Desenvolvimento Sustentável Piagaçu-Purus, Amazonas, Brasil. Ph. D. Thesis. Manaus: Universidade Federal do Amazonas, 139.
Souza, D. A., Ferreira da Silva, V. M., Fernandes da Silva, J. C., von Muhlen, E. M., Pinassi Antunes, A., and Rossoni Cardoso, F. (2014). Conservation prospects for the Amazonian manatee in the lower Purus River, Central Amazon, Brazil. Sirenews 62, 6–8.
Spennemann, D. H. R. (2020). Cruising the Currents: Observations of Extra-Limital Saltwater Crocodiles (Crocodylus porosus Schneider, 1801) in the Pacific Region. Pacific Sci. 74, 211–227.
Stanford, C. B., Iverson, J. B., Rhodin, A. G. J., van Dijk, P. P., Mittermeier, R. A., Kuchling, G., et al. (2020). Turtles and Tortoises Are in Trouble. Curr. Biol. 30, R721–R735. doi: 10.1016/j.cub.2020.04.088
Stevenson, C. J. (2015). Conservation of the Indian Gharial Gavialis gangeticus: successes and failures. Int. Zoo Yearbook 49, 150–161. doi: 10.1111/izy.12066
Stringell, T. B., Calosso, M. C., Claydon, J. A. B., Clerveaux, W., Godley, B. J., Lockhart, K. J., et al. (2013). Marine turtle harvest in a mixed small-scale fishery: Evidence for revised management measures. Ocean Coast. Manage. 82, 34–42. doi: 10.1016/j.ocecoaman.2013.05.004
Takekawa, D. (2000). Hunting method and the ecological knowledge of dolphins among the Fanalei villagers of Malaita, Solomon Islands. SPC Tradit. Mar. Resour. Manage. Knowledge Informat. Bull. 12, 3–11.
Temple, A. J., Wambiji, N., Poonian, C. N. S., Jiddawi, N., Stead, S. M., Kiszka, J. J., et al. (2019). Marine megafauna catch in southwestern Indian Ocean small-scale fisheries from landings data. Biol. Conservat. 230, 113–121.
Than, K. Z., Strine, C. T., Sritongchuay, T., Zaw, Z., and Hughes, A. C. (2020). Estimating population status and site occupancy of saltwater crocodiles Crocodylus porosus in the Ayeyarwady delta, Myanmar: Inferences from spatial modeling techniques. Glob. Ecol. Conserv. 24:e01206.
Thibault, M., and Blaney, S. (2003). The oil industry as an underlying factor in the bushmeat crisis in Central Africa. Conserv. Biol. 17, 1807–1813.
Tirira, D. G. (2011). “Libro Rojo de los mamíferos del Ecuador,” in Fundación Mamíferos y Conservación, 2nd Edn, (Quito: Pontificia Universidad Católica del Ecuador).
Tisen, O. B., Gombek, F., Ahmad, R., Gombek, F., and Kri, C. (2013). “Human-crocodile issues: Sarawak report,” in Crocodiles. Proceedings of the 22nd Working Meeting of the IUCN-SSC Crocodile Specialist Group, (Gland: IUCN), 115.
Tomás, J., Godley, B. J., Castroviejo, J., and Raga, J. A. (2010). Bioko: Critically important nesting habitat for marine turtles of West Africa. Biodivers. Conserv. 19, 2699–2714. doi: 10.1007/s10531-010-9868-z
Tomley, F. M., and Shirley, M. W. (2009). Livestock infectious diseases and zoonoses. Philos. Trans. R. Soc. Long B Biol. Sci. 364, 2637–2642. doi: 10.1098/rstb.2009.0133
Tortoise and Freshwater Turtle Specialist Group (1996). Podocnemis expansa (errata version published in 2016). Gland: IUCN.
Tovar-De la Cruz, Y. M., Castro-Perez, J. M., and Roasa-Luis, R. (2021). Involving local people in the conservation of the critically endangered freshwater turtle Dermatemys mawii in southern Mexico. Aquat. Conserv. Mar. Freshw. Ecosyst. 31, 2814–2816. doi: 10.1002/aqc.3679
Troëng, S., and Drews, C. (2004). Money talks: economic aspects of marine turtle use and conservation. Gland: WWF-International.
Troëng, S., and Rankin, E. (2005). Long-term conservation efforts contribute to positive green turtle Chelonia mydas nesting trend at Tortuguero, Costa Rica. Biol. Conserv. 121, 111–116.
Trujillo, F., Crespo, E., Van Damme, P. A., and Usma, J. S. (2010). The Action Plan for South American River Dolphins 2010 – 2020. Bogotá, D.C: WWF, 249.
Tryland, M., Nesbakken, T., Robertson, L., Grahek-Ogden, D., and Lunestad, B. T. (2014). Human pathogens in marine mammal meat–a northern perspective. Zoonoses Public Health 61, 377–394. doi: 10.1111/zph.12080
Tun, T. (2006). “Preliminary Assessment of Cetacean Catches in Coastal Waters Near Myeik and Dawei in Southeastern Myanmar,” in Report submitted to the Department of Fisheries, Myanmar, Wildlife Conservation Society and Convention on Migratory Species, (Myanmar: Department of Fisheries).
Turtle Taxonomy Working Group (2021). “Turtles of the World: Annotated Checklist and Atlas of Taxonomy, Synonymy, Distribution, and Conservation Status,” in Conservation Biology of Freshwater Turtles and Tortoises: A Compilation Project of the IUCN/SSC Tortoise and Freshwater Turtle Specialist Group, 9th Edn, eds A. G. J. Rhodin, J. B. Iverson, P. P. van Dijk, C. B. Stanford, E. V. Goode, K. A. Buhlmann, et al. (Gland: IUCN), 1–472.
Tzika, A. C., D’Amico, E., Alfaro-Shigueto, J., Mangel, J. C., Waerebeek, K., and Milinkovitch, M. C. (2010). Molecular identification of small cetacean samples from Peruvian fish markets. Conserv. Genet. 11, 2207–2218.
Uwagbae, M., and Van Waerebeek, K. (2010). Initial Evidence of Dolphin Takes in the Niger Delta Region and a Review of Nigerian Cetaceans. Document SC/62/SM1, Presented to the International Whaling Commission Scientific Committee, June 2010. Agadir, 8.
Vail, C. (2005). “Socio-economic Assessment of Marine Mammal Utilization in the Wider Caribbean Region: Captivity, Viewing and Hunting,” in Regional Workshop of Experts on the Development of the Marine Mammal Action Plan for the Wider Caribbean Region, Bridgetown, Barbados 18-21 July 2005, (Bridgetown: UNEP).
Van Bressem, M. F., Raga, J. A., Di Guardo, G., Jepson, P. D., Duignan, P. J., Siebert, U., et al. (2009). Emerging infectious diseases in cetaceans worldwide and the possible role of environmental stressors. Dis. Aquat. Organisms 86, 143–157. doi: 10.3354/dao02101
Van der Ploeg, J., Ratu, F., Viravira, J., Brien, M., Wood, C., Zama, M., et al. (2019). Human-crocodile conflict in Solomon Islands. WorldFish. Program Report: 2019-02. Penang: WorldFish.
van Vliet, N. (2018). “Bushmeat crisis” and “cultural imperialism” in wildlife management? Taking value orientations into account for a more sustainable and culturally acceptable wildmeat sector. Front. Ecol. Evolut. 6:112. doi: 10.3389/fevo.2018.00112
Van Waerebeek, K., and Reyes, J. C. (1994). Post-ban Small Cetacean Takes Off Peru: A Review. Report of the International Whaling Commission (Special Issue 15). Impington: International Whaling Commission, 503–520.
Van Waerebeek, K., and Würsig, B. (2009). “Dusky Dolphin: Lagenorhynchus obscurus,” in Encyclopedia of Marine Mammals, 2nd Edn, eds W. F. Perrin, B. Würsig, and J. G. M. Thewissen (Cambridge, USA: Academic Press), 335–338.
Van Waerebeek, K., Apaza, M., Reyes, J. C., Alfaro-Shigueto, J., Santillán, L., Barreda, E., et al. (2018). Beach Cast Small Cetaceans Bear Evidence of Continued Catches and Utilisation in Coastal Peru, 2000-2017. Document SC/67B/HIM/01 Presented to IWC Scientific Committee. Bled.
Van Waerebeek, K., Barnett, L., Camara, A., Cham, A., Diallo, M., Djiba, A., et al. (2003). Conservation of cetaceans in The Gambia and Senegal, 1999-2001, and status of the Atlantic humpback dolphin. Bonn: UNEP.
Van Waerebeek, K., Debrah, J. S., and Ofori-Danson, P. K. (2014). Cetacean Landings at the Fisheries Port of Dixcove, Ghana in 2013-14: a Preliminary Appraisal. Document SC/65b/SM17 presented to the IWC Scientific Committee, 12-24 May 2014. Bled, 4.
Van Waerebeek, K., Ndiaye, E., Djiba, A., Diallo, M., Murphy, P., Jallow, A., et al. (2000). A Survey of the Conservation Status of Cetaceans of Senegal, The Gambia and Guinea-Bissau. Bonn: UNEP.
Van Waerebeek, K., Ofori-Danson, P. K., and Debrah, J. (2009). The cetaceans of Ghana, a validated faunal checklist. West Afr. J. Appl. Ecol. 15, 61–90.
Van Waerebeek, K., Uwagbae, M., Segniagbeto, G., Bamy, I. L., and Ayissi, I. (2017). New records of Atlantic humpback dolphin (Sousa teuszii) in Guinea, Nigeria, Cameroon and Togo underscore fisheries pressure and generalised marine bushmeat demand. Revue d’Ecol. 72, 192–205.
Van Waerebeek, K., Van Bressem, M. F., Alfaro-Shigueto, J., Sanino, G. P., Montes, D., and Ontón, K. (1999). A Preliminary Analysis of Recent Captures of Small Cetaceans in Peru and Chile. Document SC/51/SM17 Presented to the IWC Scientific Committee. Grenada.
Veríssimo, D., Vieira, S., Monteiro, D., Hancock, J., and Nuno, A. (2020). Audience research as a cornerstone of demand management interventions for illegal wildlife products: demarketing sea turtle meat and eggs. Conserv. Sci. Pract. 2:e164.
Vidal, O., Van Waerebeek, K., and Findley, L. T. (1994). “Cetaceans and gillnet fisheries in Mexico, Central America and the Wider Caribbean: a preliminary review,” in Report of the International Whaling Commission, eds W. F. Perrin, G. P. Donovan, and J. Barlow (Nairobi: IWC), 221–233.
Vuto, S., Hamilton, R., Brown, C., Waldie, P., Pita, J., Peterson, N., et al. (2019). A report on turtle harvest and trade in Solomon Islands. Solomon Island: The Nature Conservancy, 34.
Waltzek, T. B., Cortés-Hinojosa, G., Wellehan, J. F. X. Jr., and Gray, G. C. (2012). Marine mammal zoonoses: a review of disease manifestations. Zoonoses Public Health 59, 521–535. doi: 10.1111/j.1863-2378.2012.01492.x
Wamukoya, G. M., Mirangi, J., and Ottichilo, W. R. (1995). “Marine Aerial Survey: Sea Turtle and Marine Mammals,” in Kenya Wildlife Service Technical Report No. 1, (Kenya: Kenya Wildlife Service), 65.
Wang, Y., Li, W., and Van Waerebeek, K. (2015). Strandings, bycatches and injuries of aquatic mammals in China, 2000-2006, as reviewed from official documents: A compelling argument for a nationwide strandings programme. Mar. Policy 51, 242–250. doi: 10.1016/j.marpol.2014.07.016
Webb, G. (2015). Principles of sustainable use. Section 4.2 in CSG Crocodilian Capacity Building Manual. Darwin: Crocodile Specialist Group.
Webb, G. J. W. (2020). “History of crocodile management in the Northern Territory of Australia: a conservation success story,” in Report for the Crocodile Farmers Association of the Northern Territory (CFANT), (Darwin: Wildlife Management International).
Webb, G. J. W., and Carrillo, C. (2000). Risk of extinction and categories of endangerment: perspectives from long-lived reptiles. Populat. Ecol. 42, 11–17.
Webb, G. J. W., and Jenkins, R. W. G. (1991). Management of crocodilians in Thailand: a review with recommendations. Canberra: Australian National Parks and Wildlife Service.
Webb, G. J. W., and Messel, H. (1978). Morphometric analysis of Crocodylus porosus from the North Coast of Arnhem Land, Northern Australia. Austral. J. Zool. 26, 1–27. doi: 10.1071/zo9780001
Webb, G. J. W., Manolis, S. C., and Brien, M. L. (2010). “Saltwater Crocodile Crocodylus porosus. Pp. 99-113 in Crocodiles,” in Status Survey and Conservation Action Plan, 3rd Edn, eds S. C. Manolis and C. Stevenson (Darwin: Crocodile Specialist Group).
Weir, C. R., and Pierce, G. J. (2012). A review of the human activities impacting cetaceans in the eastern tropical Atlantic. Mamm. Rev. 44, 258–274. doi: 10.1111/j.1365-2907.2012.00222.x
Weir, C. R., Van Waerebeek, K., Jefferson, T. A., and Collins, T. (2010). Challenges and priorities for the conservation of the Vulnerable Atlantic humpback dolphin (Sousa teuszii), with a case study from Namibe Province, Angola. SC/62/SM6. Angola: International Whaling Commission.
Williams, J. (2017). Illegal take and consumption of leatherback sea turtles (Dermochelys coriacea) in Madagascar and Mozambique. Afr. Sea Turtle Newsl. 7, 25–33.
Williams, J. L. (2021). “Working with small-scale fishers in the Southwest Indian Ocean to understand illegal take, trade, and use of sea turtles,” in Sea Turtle Research and Conservation, ed. B. Nahill (Amsterdam: Elsevier), 123–131.
Williams-Grey, A., Fuchs, A., Tossenberger, V., and Cassani, C. (2014). Whale for sale the global dead whales. United Kingdom: WDC, Whale and Dolphin Conservation.
Win, A. H., and Lwin, M. M. (2012). “Interaction between fishing activities and marine turtles in Myanmar,” in Proceedings of the 7th International Symposium on SEASTAR2000 and Asian Bio-logging Science (The 11th SEASTAR2000 workshop), (SEASTAR), 5–7.
Work, T. M., Parker, D., and Balazs, G. H. (2020). “Sea turtles in Oceania. MTSG Annual Regional Report 2020,” in Report of the IUCN-SSC Marine Turtle Specialist Group, 2020, (Gland: IUCN).
Worm, B., Hilborn, R., Baum, J. K., Branch, T. A., Collie, J. S., Costello, C., et al. (2009). Rebuilding global fisheries. Science 325, 578–584.
WWF (2004). Conserving marine turtles on a global scale. WWF International Species Programme and WWF International, Gland, Switzerland. Gland: WWF, 30.
Keywords: bushmeat, cetaceans, health implications, hunting, crocodiles, sirenians, turtles, threatened species
Citation: Ingram DJ, Prideaux M, Hodgins NK, Frisch-Nwakanma H, Avila IC, Collins T, Cosentino M, Keith-Diagne LW, Marsh H, Shirley MH, Van Waerebeek K, Djondo MK, Fukuda Y, Glaus KBJ, Jabado RW, Lang JW, Lüber S, Manolis C, Webb GJW and Porter L (2022) Widespread Use of Migratory Megafauna for Aquatic Wild Meat in the Tropics and Subtropics. Front. Mar. Sci. 9:837447. doi: 10.3389/fmars.2022.837447
Received: 16 December 2021; Accepted: 21 January 2022;
Published: 21 March 2022.
Edited by:
Maria Carolina Viana, State University of Campinas, BrazilReviewed by:
Hector Barrios-Garrido, University of Zulia, VenezuelaCristina Brito, CHAM – Center for the Humanities NOVA FCSH, Portugal
Copyright © 2022 Ingram, Prideaux, Hodgins, Frisch-Nwakanma, Avila, Collins, Cosentino, Keith-Diagne, Marsh, Shirley, Van Waerebeek, Djondo, Fukuda, Glaus, Jabado, Lang, Lüber, Manolis, Webb and Porter. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Daniel J. Ingram, ZGFuaWVsLmluZ3JhbUBzdGlyLmFjLnVr; ZGFuaWVsam9obmluZ3JhbUBnbWFpbC5jb20=
†ORCID: Daniel J. Ingram, orcid.org/0000-0001-5843-220X; Margi Prideaux, orcid.org/0000-0002-1954-9795; Nicola K. Hodgins, orcid.org/0000-0001-6591-0046; Isabel C. Avila, orcid.org/0000-0003-1389-8908; Tim Collins, orcid.org/0000-0002-7124-4876; Mel Cosentino, orcid.org/0000-0001-7837-8669; Lucy W. Keith-Diagne, orcid.org/0000-0003-0275-7966; Helene Marsh, orcid.org/0000-0003-3492-4992; Matthew H. Shirley, orcid.org/0000-0002-3095-7546; Yusuke Fukuda, orcid.org/0000-0002-8675-318X; Kerstin B. J. Glaus, orcid.org/0000-0001-9985-2243; Rima W. Jabado, orcid.org/0000-0001-6239-6723; Jeffrey W. Lang, orcid.org/0000-0001-6308-2005; Lindsay Porter, orcid.org/0000-0002-8673-9767
 Daniel J. Ingram1,2*†
Daniel J. Ingram1,2*† Heidrun Frisch-Nwakanma
Heidrun Frisch-Nwakanma Isabel C. Avila
Isabel C. Avila Tim Collins
Tim Collins Mel Cosentino
Mel Cosentino Helene Marsh
Helene Marsh Yusuke Fukuda
Yusuke Fukuda Kerstin B. J. Glaus
Kerstin B. J. Glaus Rima W. Jabado
Rima W. Jabado Jeffrey W. Lang
Jeffrey W. Lang Lindsay Porter
Lindsay Porter