- 1 Centre of Microbial and Plant Genetics, Katholieke Universiteit Leuven, Heverlee, Belgium
- 2 Institute of Molecular Biosciences, Karl-Franzens University, Graz, Austria
Plant defensins are active against plant and human pathogenic fungi (such as Candida albicans) and baker’s yeast. However, they are non-toxic to human cells, providing a possible source for treatment of fungal infections. In this study, we characterized the mode of action of the antifungal plant defensin HsAFP1 from coral bells by screening the Saccharomyces cerevisiae deletion mutant library for mutants with altered HsAFP1 sensitivity and verified the obtained genetic data by biochemical assays in S. cerevisiae and C. albicans. We identified 84 genes, which when deleted conferred at least fourfold hypersensitivity or resistance to HsAFP1. A considerable part of these genes were found to be implicated in mitochondrial functionality. In line, sodium azide, which blocks the respiratory electron transport chain, antagonized HsAFP1 antifungal activity, suggesting that a functional respiratory chain is indispensable for HsAFP1 antifungal action. Since mitochondria are the main source of cellular reactive oxygen species (ROS), we investigated the ROS-inducing nature of HsAFP1. We showed that HsAFP1 treatment of C. albicans resulted in ROS accumulation. As ROS accumulation is one of the phenotypic markers of apoptosis in yeast, we could further demonstrate that HsAFP1 induced apoptosis in C. albicans. These data provide novel mechanistic insights in the mode of action of a plant defensin.
Introduction
Plant defensins are small, basic, cysteine-rich peptides that possess antifungal and in some cases also antibacterial activity (Lay and Anderson, 2005; Aerts et al., 2008). They are postulated to be part of the innate immune system of plants and protect them against invading fungal and bacterial pathogens. Plant defensins are not only active against phytopathogenic fungi, but also against baker’s yeast Saccharomyces cerevisiae and human pathogenic fungi (such as Candida albicans). C. albicans is an opportunistic human pathogen that can cause superficial and invasive infections in immunocompromised patients (Mavor et al., 2005). Plant defensins are non-toxic to human and plant cells (Thevissen et al., 2007a; Tavares et al., 2008), highlighting their therapeutic potential as novel antimycotics. In this regard, radish defensin RsAFP2 (Terras et al., 1992) was recently proven to be prophylactically effective against murine candidiasis, and this at least to the same extent as the antimycotic fluconazole (Tavares et al., 2008).
HsAFP1 is a plant defensin, isolated from seeds of coral bells (Heuchera sanguinea; Osborn et al., 1995). HsAFP1 inhibits a wide range of fungi as well as the pathogenic yeast/fungal species C. albicans, C. krusei, and Aspergillus flavus (Thevissen et al., 2007a). We previously demonstrated that HsAFP1 specifically interacts with high affinity binding sites on the membrane of susceptible fungi and yeast species (Thevissen et al., 1997), and permeabilizes susceptible fungal/yeast cells, resulting in cell growth arrest (Thevissen et al., 1999). In contrast to HsAFP1, RsAFP2, pea defensin PsD1 (Almeida et al., 2000), and dahlia defensin DmAMP1 (Osborn et al., 1995) specifically interact with sphingolipids in the fungal envelope, being glucosylceramides (GlcCer) or mannosyl diinositolphosphoryl ceramides [M(IP)2C; Thevissen et al., 2000, 2003, 2004; de Medeiros et al., 2010]. GlcCer were also found indispensable for the antifungal activity of Medicago defensin MsDef1 (Ramamoorthy et al., 2007). The antifungal activity of HsAFP1 does not rely on the interaction with these sphingolipids since yeast mutants lacking GlcCer or M(IP)2C are as sensitive to HsAFP1 as their corresponding wild type (WT; unpublished data). The in vitro frequency of occurrence of spontaneous RsAFP2-resistant C. albicans mutants is 5–10 times higher than that of DmAMP1-resistant mutants and at least 100 times higher than that of HsAFP1-resistant C. albicans mutants (Thevissen et al., 2007a). Possibly, HsAFP1 interacts with essential fungal plasma membrane structures, resulting in low in vitro frequency of occurrence of resistant C. albicans mutants. Therefore, in view of reducing the risks of rapid emergence of resistant pathogens, HsAFP1 may offer advantages over RsAFP2 and DmAMP1 as novel antifungal agents.
In this study, we screened the haploid set of S. cerevisiae deletion mutants in non-essential genes for both hypersensitivity and resistance to HsAFP1 in order to get further insight in the mode of action of HsAFP1. Based on these genetic data, we could demonstrate the involvement of mitochondrial function in HsAFP1 antifungal action using the respiration inhibitor sodium azide and by investigating the accumulation of reactive oxygen species (ROS) in susceptible yeast species upon HsAFP1 treatment. Since mitochondrial function and the accumulation of endogenous ROS or both linked with apoptosis in yeast, we assessed the apoptosis-inducing nature of HsAFP1 in C. albicans.
Materials and Methods
Materials and Microorganisms
Yeast strains used in this study are C. albicans strain CAI4 (Fonzi and Irwin, 1993), S. cerevisiae strains W0303-1a, BY4741, and the BY4741-derived deletion mutant library (Invitrogen, Carlsbad, CA, USA). Yeast nutrient media used are YPD (10 g/l yeast extract, 20 g/l peptone, 20 g/l glucose); PDB/YPD (19.2 g/l potato dextrose broth (Difco), 2 g/l yeast extract, 4 g/l peptone, 4 g/l glucose; adjusted to pH 7.0 with 50 mM HEPES); and SC (0.8 g/l CSM, complete amino acid supplement mixture, Bio 101 Systems; 6.5 g/l YNB, yeast nitrogen base; 20 g/l glucose). HsAFP1 was purified as described previously (Osborn et al., 1995). H. sanguinea seeds were kindly provided by Kieft Seeds (Venhuizen, The Netherlands). If not mentioned otherwise, chemicals were purchased from Sigma (St. Louis, MO, USA).
Screening of a S. Cerevisiae Deletion Mutant Library for Altered HsAFP1 Sensitivity
To this end, the minimal inhibitory concentration (MIC) of HsAFP1 for the individual deletion mutants was determined in PDB/YPD and compared with the MIC of HsAFP1 for WT (Thevissen et al., 2007b). The HsAFP1-hypersensitivity (HSFs) or resistance factors (RFs) were calculated as MIC(WT)/MIC(mutant) or MIC(mutant)/MIC(WT), respectively. Strains that were at least fourfold more resistant or hypersensitive to HsAFP1 were retested.
Antifungal Activity Assay
Exponentially growing S. cerevisiae (W303-1a) in YPD (OD600 = 2.0) were incubated with 20 μg/ml HsAFP1 in the presence or absence of 0.005% sodium azide in PDB/YPD medium as described previously (Aerts et al., 2009a).
Yeast Apoptosis Assays
Exponentially growing C. albicans cultures (SC, OD600 = 2.0) were incubated with 5 μg/ml HsAFP1 or water (control) in PDB/YPD during 2 h 30 min at 30°C. Survival was determined by performing plating assays in which colony formation of 500 cells on YPD agar plates was analyzed. Apoptotic markers, including ROS levels, phosphatidylserine (PS) externalization, and DNA fragmentation of yeast cultures (n = 500 cells per measurement), were visualized as described previously (Aerts et al., 2009a).
Results
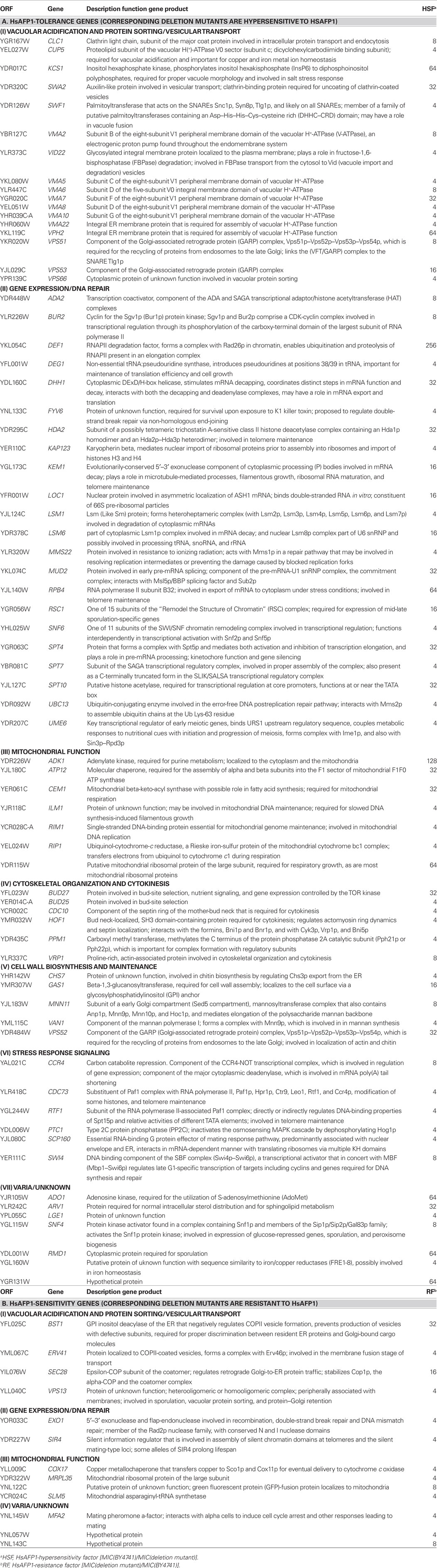
Screening of a Yeast Deletion Mutant Library for Hypersensitivity and Resistance Toward HSAFP1 Reveals a Crucial Role for Mitochondria in the Mode of Action of HsAFP1
To identify genes that may contribute to HsAFP1 tolerance or sensitivity, we screened a S. cerevisiae deletion mutant library (consisting of 4385 deletion mutants) for hypersensitivity and resistance toward HsAFP1. To this end, we determined the MIC resulting in 100% growth inhibition of HsAFP1 for all individual yeast knock-out mutants and WT yeast using twofold dilution series of HsAFP1 in liquid PDB/YPD medium. We identified 13 HsAFP1-resistant and 71 HsAFP1-hypersensitive mutants, which were at least fourfold more resistant or sensitive toward HsAFP1 as compared to WT (MIC = 20 μg/ml), and the corresponding HsAFP1-sensitivity and tolerance genes, respectively. Following major functional families of HsAFP1-sensitivity or -tolerance genes could be distinguished (according to www.yeastgenome.org), encoding gene products involved in: (i) vacuolar acidification and protein sorting/vesicular transport, (ii) gene expression/DNA repair, (iii) mitochondrial function, (iv) cytoskeletal organization and cytokinesis, (v) cell wall biosynthesis and maintenance, and (vi) stress response signaling (Table 1). Interestingly, 30% of the HsAFP1-sensitivity genes encode proteins that have a mitochondrial function, suggesting an important role for mitochondria in HsAFP1-mediated cell death. Note that the portion of disruptants in non-essential genes, which encode proteins involved in mitochondrial function, in the yeast library is approximately 7% (Dimmer et al., 2002), indicating a fourfold enrichment of genes important for proper mitochondrial function within the HsAFP1-sensitivity genes. Mitochondrial function is known to be important for the induction of apoptosis in yeast (Eisenberg et al., 2007; Fröhlich et al., 2007). In addition, among the genes subdivided in the groups (ii), (iii), and (vi), we found eight HsAFP1-tolerance or -sensitivity genes implicated in apoptosis in yeast (being DHH1, LSM1, LSM6, RIP1, SIR4, HDA2, KAP123, and SCP160). Therefore, in this study, we further investigated the involvement of functional mitochondria in the antifungal action of HsAFP1 and whether the induction of apoptosis is involved in this process.
Blocking Mitochondrial Function by Respiration Inhibitor Sodium Azide Antagonizes HsAFP1 Antifungal Capacity
Several of the identified HsAFP1-tolerance or -sensitivity genes with a mitochondrial function are directly implicated in respiratory growth, i.e., RIP1, YDR115w, CEM1, ATP12, COX17, and MRPL35. Therefore, we investigated the effect of the respiration inhibitor sodium azide on HsAFP1 antifungal action. Sodium azide blocks the electron flow at complex IV (cytochrome c oxidase) of the respiratory electron transport chain thereby preventing ATP production. We previously demonstrated that treatment of susceptible fungi with 50–100 μg/ml plant defensins results in a specific membrane permeabilization, whereas treatment with lower doses, i.e., 5–20 μg/ml, results in target-mediated membrane permeabilization (Thevissen et al., 1999). Therefore, in this study, we used rather low HsAFP1 concentrations, i.e., 5–20 μg/ml.
To assess the effect of sodium azide on HsAFP1 antifungal action, we determined survival of S. cerevisiae cultures, incubated with 20 μg/ml HsAFP1 or water in the presence or absence of 0.005% azide for different incubation times (Figure 1).
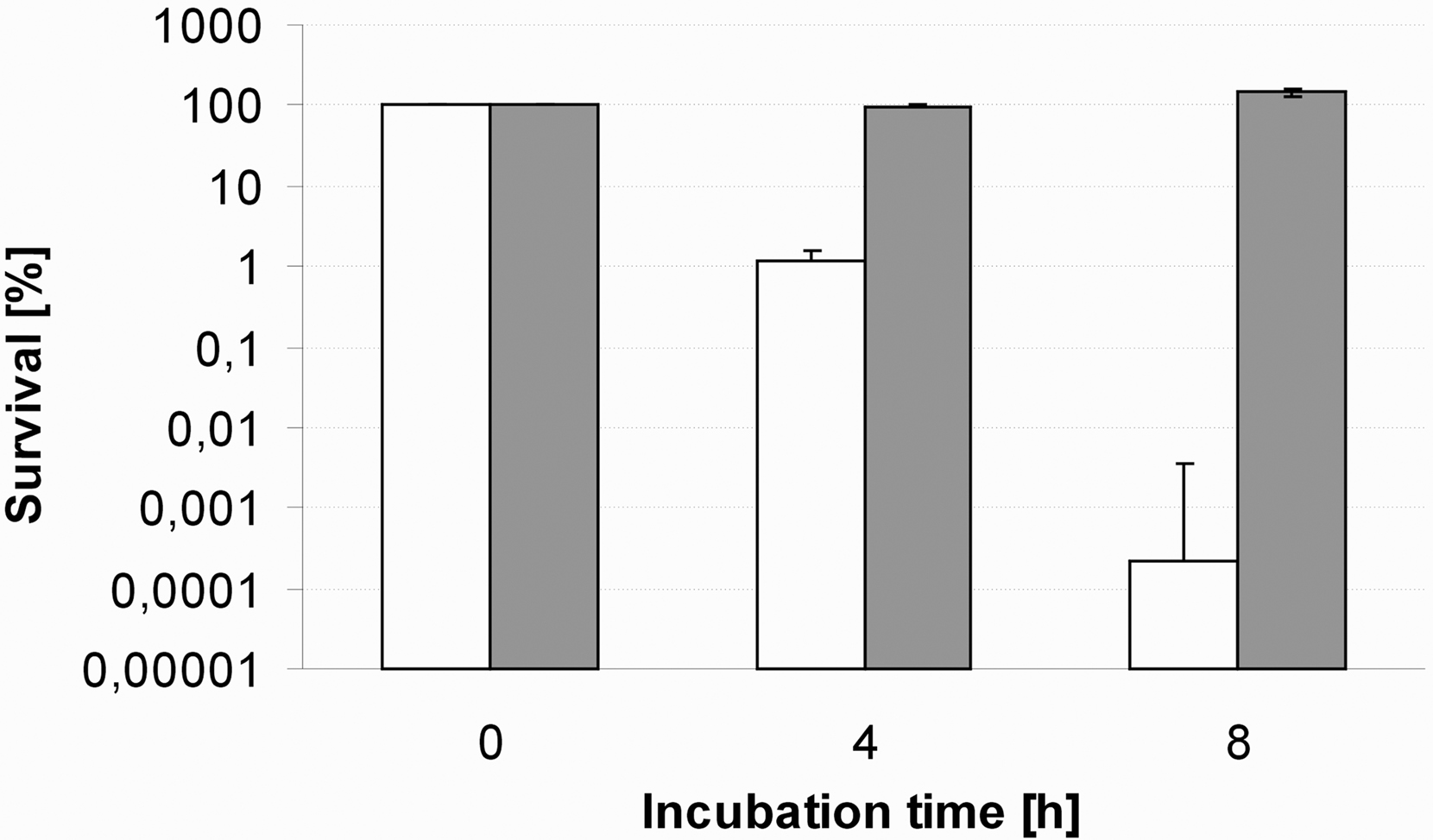
Figure 1. Effect of the respiration inhibitor sodium azide on HsAFP1 antifungal action. Exponentially growing S. cerevisiae cultures were suspended in PDB/YPD and incubated with 20 μg/ml HsAFP1 in the presence (black bars) or absence (white bars) of 0.005% azide for 0, 4, and 8 h of incubation at 30°C. Viability was analyzed by counting the number of CFUs/ml on YPD agar plates. Percentage survival was calculated as the ratio of the CFUs/ml after HsAFP1 treatment to the CFUs/ml of the corresponding control (water) treatment. Data represent mean ± SEM. This figure is a representative of three independent experiments.
Incubation of the yeast culture with 20 μg/ml HsAFP1 for 0, 4, or 8 h resulted in 100, 1, and 0.0002% survival of the culture relative to the control (water) treatment, whereas co-incubation with 0.005% sodium azide resulted in 100% survival of these cultures. Hence, sodium azide antagonizes HsAFP1 antifungal action up till 8 h of incubation. These results suggest that a functional respiratory chain is indispensable for HsAFP1 antifungal action.
HsAFP1 Induces Endogenous ROS in Susceptible Yeast
As mentioned above, the considerable part of mitochondrial related HsAFP1-sensitivity genes suggest a role for mitochondria in the HsAFP1 antifungal activity. Since mitochondria, and more specifically complexes I and III from the respiratory chain, are the main source of cellular ROS (Batandier et al., 2002), we further assessed a putative effect of HsAFP1 on endogenous ROS levels in susceptible yeast species.
We could demonstrate that the levels of endogenous ROS increased upon HsAFP1 treatment in both S. cerevisiae (data not shown) and C. albicans (Figure 2).
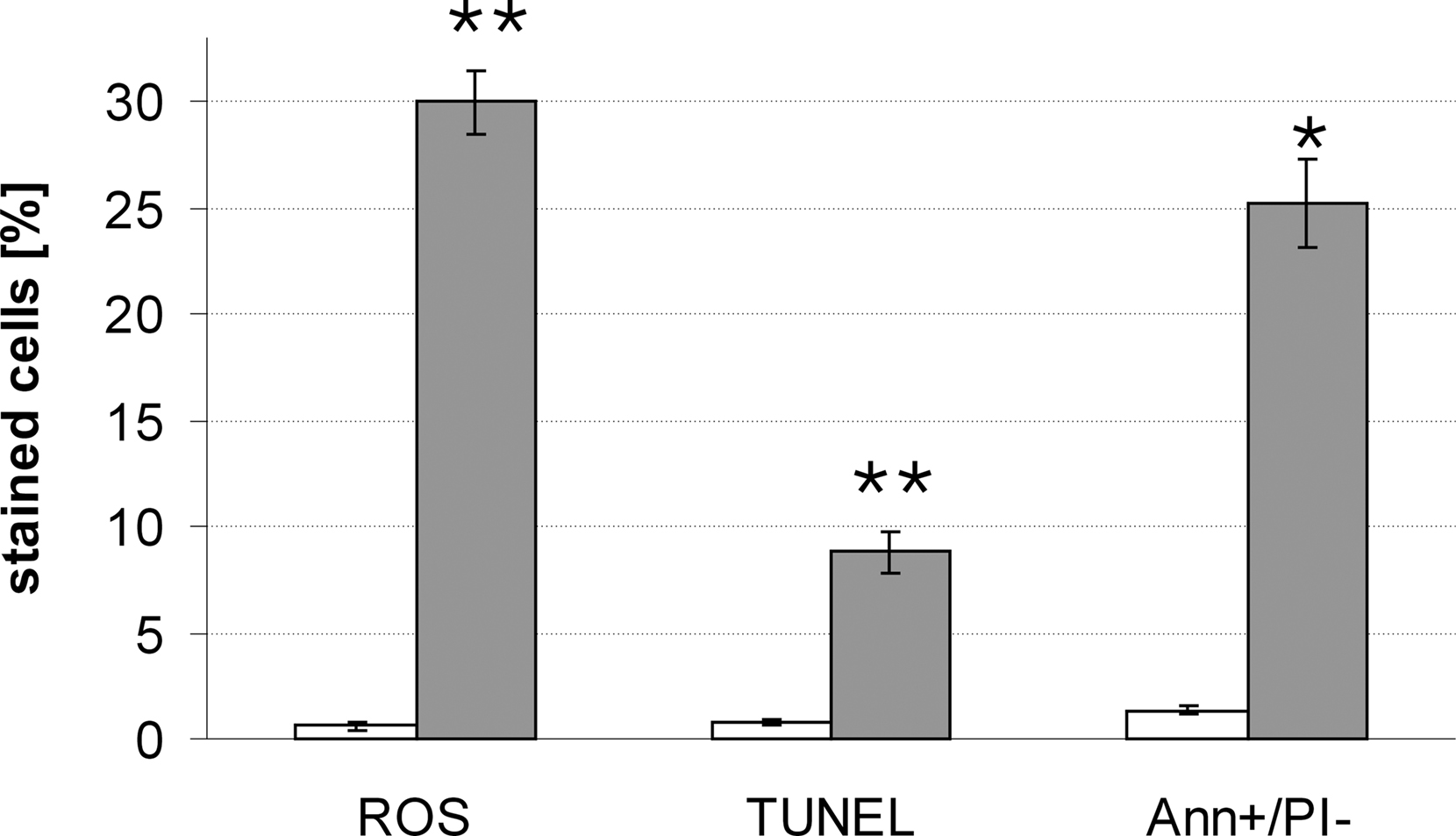
Figure 2. HsAFP1 induces apoptosis in C. albicans. Exponentially growing C. albicans cultures were treated with 5 μg/ml HsAFP1 or water for 2 h 30 min. HsAFP1-treated cells (gray bars) and control cells (white bars) were assayed for ROS accumulation (via DHE staining), DNA fragmentation (via TUNEL staining), and phosphatidylserine externalization and membrane integrity via annexinV/propidium iodide co-staining. In each experiment, 500 cells were evaluated using fluorescence microscopy (100% represents the number of cells, i.e., 500). Values are the mean of triplicate measurements. Data represent mean ± SEM. *p < 0.05; **p < 0.01.
HsAFP1 Induces Apoptosis in C. Albicans
Mitochondrial function and the accumulation of endogenous ROS are both linked with apoptosis in yeast. In addition, among the genes subdivided in the functional families (ii), (iii), and (vi), we found several HsAFP1-tolerance and -sensitivity genes implicated in apoptosis in yeast, being DHH1, LSM1, LSM6 (Mazzoni et al., 2003), RIP1 (Belhocine et al., 2004), SIR4 (Orlandi et al., 2004), HDA2 (Ahn et al., 2006), KAP123 (Büttner et al., 2007), and SCP160 (Magherini et al., 2007). In order to clarify if the HsAFP1-induced cell death is of apoptotic nature, we assessed DNA fragmentation [visualized via the deoxynucleotidyltransferase-mediated dUTP nick end labeling (TUNEL) assay] and, both PS translocation from the inner leaflet to the extracellular side of the plasma membrane and loss of membrane integrity (visualized via co-staining with FITC-labeled annexin V and propidium iodide) of C. albicans cultures treated with 5 μg/ml HsAFP1. HsAFP1-treated cultures were characterized by increased ROS levels (30.0 ± 1.5%) as compared to control cultures (0.6 ± 0.2%), increased DNA fragmentation levels (8.8 ± 1.0%) as compared to control treatment cultures (0.8 ± 0.1%), and by an excess of annexin V positive/PI negative cells (25.2 ± 2.1%) as compared to control cultures (1.36 ± 0.2%; Figure 2). The level of annexin V negative/positive, PI positive cells that represent late apoptotic and necrotic cells was 23.5 ± 3.1% after HsAFP1 treatment. In conclusion, HsAFP1-induced cell death in C. albicans is accompanied by the occurrence of typical phenotypical markers of apoptosis, thus, pointing to a HsAFP1 antifungal mechanism that involves the induction of the apoptotic machinery in C. albicans.
Discussion
In this study, we screened a S. cerevisiae deletion mutant library for altered sensitivity toward HsAFP1. In this way, we identified 71 HsAFP1-tolerance genes and 13 HsAFP1-sensitivity genes (Table 1), which we subdivided in functional families. Additional biochemical tests revealed that (i) HsAFP1 antifungal action requires a properly working yeast respiratory chain and that (ii) HsAFP1 induces apoptosis in C. albicans.
In mitochondria, respiration takes place supplying the cell with energy (ATP), meanwhile however leaking huge amounts of ROS. Indeed, the vast majority of cellular ROS (estimated at approximately 90%) can be tracked back to the mitochondria where sites I and III are the main sites of production (Batandier et al., 2002). ROS, such as hydrogen peroxide and hydroxyl radicals are produced as byproducts of aerobic respiration and cause damage to proteins, lipids, and DNA, resulting in mutation and loss of viability. In this study, we show that the respiration inhibitor sodium azide antagonizes HsAFP1 antifungal action, pointing to a functional respiratory chain as a prerequisite for HsAFP1 antifungal action. In line with this observation, we previously isolated the S. cerevisiae transposon mutant HsTnII, which is resistant toward HsAFP1 (data not shown) and characterized by respiration deficiency (Aerts et al., 2009b). All these data point to the importance of functional mitochondria and a functional respiratory chain for HsAFP1 antifungal action. Apparently, treatment of C. albicans cells with sodium azide also results in a decreased susceptibility to human β-defensin 2 (HBD2) and HBD3 (Vylkova et al., 2007), pointing toward HBD2- and HBD3-induced energy dependent C. albicans killing, as is the case for HsAFP1. However, putative induction of apoptosis by human defensins has never been reported.
The purpose of apoptosis in multicellular organisms is obvious: single cells die for the benefit of the whole organism (e.g., to eliminate dangerous, superfluous, or damaged cells). The phenomenon of yeast cells undergoing apoptosis has long been controversial, in part because of doubts of whether cell suicide could constitute an evolutionary advantage for unicellular organisms. Studies have now described yeast apoptosis during mating, aging, or exposure to killer toxins (Büttner et al., 2006). Pheromone signaling leads to the apoptotic death of cells that fail to mate, therefore depleting the population of haploid cells and favoring the survival of diploid cells that increase genetic diversity through meiotic recombination. The early death of old and damaged cells during aging and starvation enhances the chances of the rest of the population to survive and to sporulate, thus increasing the probability that the clone will survive. Apoptosis can also be induced by competing yeast strains that produce toxins in a tribal war. The death of infertile, old, or damaged yeast cells may therefore ensure the survival of a colony of yeast cells and introduces the concept of an altruistic aging and death program. In this context, we could also understand why it may be beneficial for a yeast cell in culture to undergo apoptosis in response to an antifungal defensin.
It is well documented that mitochondrial function is linked with apoptosis in S. cerevisiae. Furthermore, an excess of endogenous ROS levels is one of the phenotypical markers of apoptosis in both S. cerevisiae (Madeo et al., 1999) and in C. albicans (Phillips et al., 2003). In this study, we demonstrate that HsAFP1 induces apoptotic cell death in C. albicans. We show that, besides ROS accumulation, HsAFP1-treated C. albicans cultures exhibit other key markers of apoptosis, including exposure of PS to the outer leaflet of the plasma membrane and DNA fragmentation. All these data point to the induction of mitochondrion-dependent apoptosis by HsAFP1 in susceptible yeast. These data are in line with recently obtained results indicating that also RsAFP2, a plant defensin from radish (Terras et al., 1992), induces apoptosis in C. albicans (Aerts et al., 2009a). In contrast to HsAFP1 for which the fungal membrane target has not yet been identified, RsAFP2 has been shown to interact with GlcCer in the membrane of susceptible fungi (Thevissen et al., 2004) and cause membrane permeabilization and fungal cell death (Thevissen et al., 1999). Apart from the apoptosis-inducing potential of RsAFP2 and HsAFP1, no reports exist on the induction of apoptosis in C. albicans by other defensins. Recently, Andrés et al. (2008) demonstrated apoptotic cell death in C. albicans by lactoferrin, a protein present in mammalian mucosal secretions with antifungal and antibacterial activity. Other natural peptides/proteins that induce apoptosis in yeast are osmotin (Narasimhan et al., 2001), a truncated derivative of dermaseptin S3 (Morton et al., 2007), yeast pheromone (Pozniakovsky et al., 2005), and yeast killer toxins (Klassen and Meinhardt, 2005).
A considerable part of the identified HsAFP1-tolerance/sensitivity genes encode proteins implicated in vacuolar acidification, vacuolar protein sorting, or vesicular transport. It was recently demonstrated that yeast hampered in vacuolar protein sorting or lacking a functional vacuolar H+-ATPase shows multi-drug rather than drug-specific sensitivity (Parsons et al., 2004). Therefore, this functional group may rather represents general sickness. However, it is also reported that the vacuolar H+-ATPase in yeast is required for oxidative stress response (Kane, 2007). Consistent with such a role, vma mutants are shown to be hypersensitive to multiple forms of oxidative stress and display elevated levels of ROS even in the absence of an exogeneous oxidant (Thorpe et al., 2004; Kane, 2007; Milgrom et al., 2007). This would then suggest that an intact vacuolar H+-ATPase is required for HsAFP1 tolerance in S. cerevisiae.
The fraction of HsAFP1-tolerance/sensitivity genes implicated in gene expression/DNA repair may also represent general stress sensitivity mechanisms. In this respect, yeasts affected in genes involved in DNA synthesis and repair, transcription, and chromatin structure (including ADA/SAGA histone acetyltransferase complexes or the SWI/SNF nucleosome remodeling complex) were previously identified as hypersensitive to a variety of stresses, including oxidative and chemical stress including treatment with the antifungal miconazole (Thorpe et al., 2004; Thevissen et al., 2007b). Several HsAFP1-tolerance genes (DHH1, CCR4, LSM1, LSM6, and KEM1) have a function in post-translational mRNA regulation and mRNA decay. These data highlight the importance of de novo transcription in response to environmental stress, and may indicate that posttranslational gene regulation plays a role in the cellular stress response against HsAFP1.
We further identified HsAFP1-tolerance genes implicated in different mitogen-activated protein kinase (MAPK) pathways. CDC73, CCR4, RTF1, and SWI4 are HsAFP1-tolerance genes that are implicated in the MAPK cell integrity pathway. The cell integrity pathway plays a key role in maintaining the cell wall integrity in distinct environmental conditions. This pathway is induced in periods of polarized growth and responds to heat, hypo-osmotic shock, cell wall damage, and oxidative stress (Martin et al., 2005). Swi4p is a transcriptional activator in the cell integrity pathway (Gustin et al., 1998). Cdc73p, Ccd4p, and Rtf1p are constituents of the Paf1 complex that is required for full expression of many cell wall biosynthetic genes in the cell integrity pathway (Porter et al., 2002). In this respect, it has to be noted that we also identified several HsAFP1-tolerance genes involved in cell wall maintenance and architecture (CHS7, VPS52, MNN11, VAN1, and GAS1). Apparently, the MAPK cell integrity pathway is also involved in protection of S. cerevisiae to caffeine (Kuranda et al., 2006), caspofungin (Reinoso-Martin et al., 2003), and farnesol (Fairn et al., 2007), and in protection of F. graminearum to RsAFP2 and the medicago MsDef1 (Ramamoorthy et al., 2007). Furthermore, we identified one HsAFP1-tolerance gene, PTC1, implicated in the osmosensing high osmolarity glycerol (HOG) MAPK pathway. The HOG pathway responds to osmotic stress. In addition, this MAPK pathway has also been shown to respond to heat shock, oxidative stress, and citric acid (Martin et al., 2005). Remarkably, although Ptc1p negatively regulates the HOG MAPK pathway (Warmka et al., 2001), PTC1 deletion confers HsAFP1 hypersensitivity. Finally, we identified SCP160 as a HsAFP1-tolerance gene. Scp160p is an RNA-binding G protein effector of the MAPK mating response pathway (Guo et al., 2003). All these findings suggest the involvement of different MAPK stress response pathways in yeast tolerance toward HsAFP1.
Finally, we identified five HsAFP1-tolerance genes implicated in cytoskeletal organization and cytokinesis. Interestingly, decreased actin turnover was previously shown to result in increased mitochondrial ROS production and apoptosis (Gourlay and Ayscough, 2006). It was recently proposed that actin residues can act as oxidative stress sensors that further regulate cell death in yeast (Farah and Amberg, 2007). Only one study reports on a role for actin cytoskeleton alterations in the mechanism of action of an apoptosis-inducing antifungal compound. In this respect, the antifungal drug jasplakinolide has been shown to cause a block in actin dynamics (Ayscough, 2000) and to induce ROS and apoptosis in S. cerevisiae (Gourlay et al., 2004). Whether HsAFP1 induces changes in the actin cytoskeleton organization and whether this process results directly in elevated ROS levels and subsequent apoptosis, needs to be investigated further.
In conclusion, we identified 71 HsAFP1-tolerance genes and 13 HsAFP1-sensitivity genes, which we subdivided in different groups according to their function. In this way, we were able to deduce part of the HsAFP1 antifungal action mechanism. Indeed, we demonstrated that proper mitochondrial respiration is required for HsAFP1 antifungal action. Moreover, HsAFP1 induces ROS accumulation and apoptosis in susceptible yeast species. Whether disturbance of the actin cytoskeleton is involved in HsAFP1-induced ROS accumulation and apoptosis, needs to be investigated further. Regarding HsAFP1-tolerance mechanisms in yeast, we hypothesize that MAPK signaling pathways are involved.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
This work was supported by FWO-Vlaanderen (research project G.0440.07 to Bruno P. A. Cammue and travel grant V3/5-CVW.D5 to An M. Aerts). We are grateful for the postdoctoral fellowships to An M. Aerts (Research Council) and to Karin Thevissen (Industrial Research Found), both from KU Leuven. Heuchera sanguinea seeds were kindly provided by Kieft Seeds (Venhuizen, The Netherlands).
References
Aerts, A. M., Carmona-Gutierrez, D., Lefevre, S., Govaert, G., François, I. E., Madeo, F., Santos, R., Cammue, B. P., and Thevissen, K. (2009a). The antifungal plant defensin RsAFP2 from radish induces apoptosis in a metacaspase independent way in Candida albicans. FEBS Lett. 583, 2513–2516.
Aerts, A. M., Zabrocki, P., Govaert, G., Mathys, J., Carmona-Gutierrez, D., Madeo, F., Winderickx, J., Cammue, B. P., and Thevissen, K. (2009b). Mitochondrial dysfunction leads to reduced chronological lifespan and increased apoptosis in yeast. FEBS Lett. 583, 113–117.
Aerts, A. M., François, I. E., Cammue, B. P., and Thevissen, K. (2008). The mode of antifungal action of plant, insect and human defensins. Cell. Mol. Life Sci. 65, 2069–2079.
Ahn, S. H., Diaz, R. L., Grunstein, M., and Allis, C. D. (2006). Histone H2B deacetylation at lysine 11 is required for yeast apoptosis induced by phosphorylation of H2B at serine 10. Mol. Cell 24, 211–220.
Almeida, M. S., Cabral, K. M., Zingali, R. B., and Kurtenbach, E. (2000). Characterization of two novel defense peptides from pea (Pisum sativum) seeds. Arch. Biochem. Biophys. 378, 278–286.
Andrés, M. T., Viejo-Díaz, M., and Fierro, J. F. (2008). Human lactoferrin induces apoptosis-like cell death in Candida albicans: critical role of K+-channel-mediated K+-efflux. Antimicrob. Agents Chemother. 52, 4081–4088.
Ayscough, K. R. (2000). Endocytosis and the development of cell polarity in yeast require a dynamic F-actin cytoskeleton. Curr. Biol. 10, 1587–1590.
Batandier, C., Fontaine, E., Kériel, C., and Leverve, X. M. (2002). Determination of mitochondrial reactive oxygen species: methodological aspects. J. Cell. Mol. Med. 6, 175–187.
Belhocine, S., Mbithe, C., Dimitrova, I., Kampranis, S. C., and Makris, A. M. (2004). Yeast mutants resistant to Bax lethality reveal distinct vacuolar and mitochondrial alterations. Cell Death Differ. 11, 946–948.
Büttner, S., Eisenberg, T., Carmona-Gutierrez, D., Ruli, D., Knauer, H., Ruckenstuhl, C., Sigrist, C., Wissing, S., Kollroser, M., Fröhlich, K. U., Sigrist, S., and Madeo, F. (2007). Endonuclease G regulates budding yeast life and death. Mol. Cell 25, 233–246.
Büttner, S., Eisenberg, T., Herker, E., Carmona-Gutierrez, D., Kroemer, G., and Madeo, F. J. (2006). Why yeast cells can undergo apoptosis: death in times of peace, love, and war. J. Cell Biol. 175, 521–525.
de Medeiros, L. N., Angeli, R., Sarzedas, C. G., Barreto-Bergter, E., Valente, A. P., Kurtenbach, E., and Almeida, F. C. (2010). Backbone dynamics of the antifungal Psd1 pea defensin and its correlation with membrane interaction by NMR spectroscopy. Biochim. Biophys. Acta 1798, 105–113.
Dimmer, K. S., Fritz, S., Fuchs, F., Messerschmitt, M., Weinbach, N., Neupert, W., and Westermann, B. (2002). Genetic basis of mitochondrial function and morphology in Saccharomyces cerevisiae. Mol. Biol. Cell 13, 847–853.
Eisenberg, T., Büttner, S., Kroemer, G., and Madeo, F. (2007). The mitochondrial pathway in yeast apoptosis. Apoptosis 12, 1011–1023.
Fairn, G. D., Macdonald, K., and McMaster, C. R. (2007). A chemogenomic screen in Saccharomyces cerevisiae uncovers a primary role for the mitochondria in farnesol toxicity and its regulation by the Pkc1 pathway. J. Biol. Chem. 282, 4868–4874.
Farah, M. E., and Amberg, D. C. (2007). Conserved actin cysteine residues are oxidative stress sensors that can regulate cell death in yeast. Mol. Biol. Cell 18, 1359–1365.
Fonzi, W. A., and Irwin, M. Y. (1993). Isogenic strain construction and gene mapping in Candida albicans. Genetics 134, 717–728.
Fröhlich, K. U., Fussi, H., and Ruckenstuhl, C. (2007). Yeast apoptosis – from genes to pathways. Semin. Cancer Biol. 17, 112–121.
Gourlay, C. W., and Ayscough, K. R. (2006). Actin-induced hyperactivation of the Ras signaling pathway leads to apoptosis in Saccharomyces cerevisiae. Mol. Cell. Biol. 26, 6487–6501.
Gourlay, C. W., Carpp, L. N., Timpson, P., Winder, S. J., and Ayscough, K. R. (2004). A role for the actin cytoskeleton in cell death and aging in yeast. J. Cell Biol. 164, 803–809.
Guo, M., Aston, C., Burchett, S. A., Dyke, C., Fields, S., Rajarao, S. J., Uetz, P., Wang, Y., Young, K., and Dohlman, H. G. (2003). The yeast G protein alpha subunit Gpa1 transmits a signal through an RNA binding effector protein Scp160. Mol. Cell 12, 517–524.
Gustin, M. C., Albertyn, J., Alexander, M., and Davenport, K. (1998). MAP kinase pathways in the yeast Saccharomyces cerevisiae. Microbiol. Mol. Biol. Rev. 62, 1264–1300.
Kane, P. M. (2007). The long physiological reach of the yeast vacuolar H+-ATPase. J. Bioenerg. Biomembr. 39, 415–421.
Klassen, R., and Meinhardt, F. (2005). Induction of DNA damage and apoptosis in Saccharomyces cerevisiae by a yeast killer toxin. Cell. Microbiol. 7, 393–401.
Kuranda, K., Leberre, V., Sokol, S., Palamarczyk, G., and Francois, J. (2006). Investigating the caffeine effects in the yeast Saccharomyces cerevisiae brings new insights into the connection between TOR, PKC and Ras/cAMP signaling pathways. Mol. Microbiol. 61, 1147–1166.
Lay, F. T., and Anderson, M. A. (2005). Defensins – components of the innate immune system in plants. Curr. Protein Pept. Sci. 6, 85–101.
Madeo, F., Frohlich, E., Ligr, M., Grey, M., Sigrist, S. J., Wolf, D. H., and Frohlich, K. U. (1999). Oxygen stress: a regulator of apoptosis in yeast. J. Cell Biol. 145, 757–767.
Magherini, F., Tani, C., Gamberi, T., Caselli, A., Bianchi, L., Bini, L., and Modesti, A. (2007). Protein expression profiles in Saccharomyces cerevisiae during apoptosis induced by H2O2. Proteomics 7, 1434–1445.
Martin, H., Flandez, M., Nombelaand, C., and Molina, M. (2005). Protein phosphatases in MAPK signaling: we keep learning from yeast. Mol. Microbiol. 58, 6–16.
Mavor, A. L., Thewes, S., and Hube, B. (2005). Systemic fungal infections caused by Candida species: epidemiology, infection process and virulence attributes. Curr. Drug Targets 6, 863–874.
Mazzoni, C., Mancini, P., Verdone, L., Madeo, F., Serafini, A., Herker, E., and Falcone, C. (2003). A truncated form of KlLsm4p and the absence of factors involved in mRNA decapping trigger apoptosis in yeast. Mol. Biol. Cell 14, 721–729.
Milgrom, E., Diab, H., Middleton, F., and Kane, P. M. (2007). Loss of vacuolar proton-translocating ATPase activity in yeast results in chronic oxidative stress. J. Biol. Chem. 282, 7125–7136.
Morton, C. O., Dos Santos, S. C., and Coote, P. (2007). An amphibian-derived, cationic, alpha-helical antimicrobial peptide kills yeast by caspase-independent but AIF-dependent programmed cell death. Mol. Microbiol. 65, 494–507.
Narasimhan, M. L., Damsz, B., Coca, M. A., Ibeas, J. I., Yun, D. J., Pardo, J. M., Hasegawa, P. M., and Bressan, R. A. (2001). A plant defense response effector induces microbial apoptosis. Mol. Cell 8, 921–930.
Orlandi, I., Bettiga, M., Alberghina, L., and Vai, M. (2004). Transcriptional profiling of ubp10 null mutant reveals altered subtelomeric gene expression and insurgence of oxidative stress response. J. Biol. Chem. 279, 6414–6425.
Osborn, R. W., De Samblanx, G. W., Thevissen, K., Goderis, I., Torrekens, S., Van Leuven, F., Attenborough, S., Rees, S. B., and Broekaert, W. F. (1995). Isolation and characterisation of plant defensins from seeds of Asteraceae, Fabaceae, Hippocastanaceae and Saxifragaceae. FEBS Lett. 368, 257–262.
Parsons, A. B., Brost, R. L., Ding, H., Li, Z., Zhang, C., Sheikh, B., Brown, G. W., Kane, P. M., Hughes, T. R., and Boone, C. (2004). Integration of chemical-genetic and genetic interaction data links bioactive compounds to cellular target pathways. Nat. Biotechnol. 22, 62–69.
Phillips, A. J., Sudbery, I., and Ramsdale, M. (2003). Apoptosis induced by environmental stresses and amphotericin B in Candida albicans. Proc. Natl. Acad. Sci. U.S.A. 100, 14327–14332.
Porter, S. E., Washburn, T. M., Chang, M., and Jaehning, J. A. (2002). The yeast pafl-rNA polymerase II complex is required for full expression of a subset of cell cycle-regulated genes. Eukaryot. Cell 1, 830–842.
Pozniakovsky, A. I., Knorre, D. A., Markova, O. V., Hyman, A. A., Skulachev, V. P., and Severin, F. F. (2005). Role of mitochondria in the pheromone- and amiodarone-induced programmed death of yeast. J. Cell Biol. 168, 257–269.
Ramamoorthy, V., Zhao, X., Snyder, A. K., Xu, J. R., and Shah, D. M. (2007). Two mitogen-activated protein kinase signaling cascades mediate basal resistance to antifungal plant defensins in Fusarium graminearum. Cell. Microbiol. 9, 1491–1506.
Reinoso-Martin, C., Schuller, C., Schuetzer-Muehlbauer, M., and Kuchler, K. (2003). The yeast protein kinase C cell integrity pathway mediates tolerance to the antifungal drug caspofungin through activation of Slt2p mitogen-activated protein kinase signaling. Eukaryot. Cell 2, 1200–1210.
Tavares, P. M., Thevissen, K., Cammue, B. P., François, I. E., Barreto-Bergter, E., Taborda, C. P., Marques, A. F., Rodrigues, M. L., and Nimrichter, L. (2008). In vitro activity of the antifungal pant defensin RsAFP2 against Candida isolates and its in vivo efficacy in prophylactic murine models of candidiasis. Antimicrob. Agents Chemother. 52, 4522–4525.
Terras, F. R., Schoofs, H. M., De Bolle, M. F., Van Leuven, F., Rees, S. B., Vanderleyden, J., Cammue, B. P., and Broekaert, W. F. (1992). Analysis of two novel classes of plant antifungal proteins from radish (Raphanus sativus L.) seeds. J. Biol. Chem. 267, 15301–15309.
Thevissen, K., Cammue, B. P., Lemaire, K., Winderickx, J., Dickson, R. C., Lester, R. L., Ferket, K. K., Van Even, F., Parret, A. H., and Broekaert, W. F. (2000). A gene encoding a sphingolipid biosynthesis enzyme determines the sensitivity of Saccharomyces cerevisiae to an antifungal plant defensin from dahlia (Dahlia merckii). Proc. Natl. Acad. Sci. U.S.A. 97, 9531–9536.
Thevissen, K., François, I. E., Takemoto, J. Y., Ferket, K. K., Meert, E. M., and Cammue, B. P. (2003). DmAMP1, an antifungal plant defensin from dahlia (Dahlia merckii), interacts with sphingolipids from Saccharomyces cerevisiae. FEMS Microbiol. Lett. 226, 169–173.
Thevissen, K., Kristensen, H. H., Thomma, B. P., Cammue, B. P., and François, I. E. (2007a). Therapeutic potential of antifungal plant and insect defensins. Drug Discov. Today 12, 966–971.
Thevissen, K., Ayscough, K. R., Aerts, A. M., Du, W., De Brucker, K., Meert, E. M., Ausma, J., Borgers, M., Cammue, B. P., and François, I. E. (2007b). Miconazole induces changes in actin cytoskeleton prior to reactive oxygen species induction in yeast. J. Biol. Chem. 282, 21592–21597.
Thevissen, K., Osborn, R. W., Acland, D. P., and Broekaert, W. F. (1997). Specific, high affinity binding sites for an antifungal plant defensin on Neurospora crassa hyphae and microsomal membranes. J. Biol. Chem. 272, 32176–32181.
Thevissen, K., Terras, F. R., and Broekaert, W. F. (1999). Permeabilization of fungal membranes by plant defensins inhibits fungal growth. Appl. Environ. Microbiol. 65, 5451–5458.
Thevissen, K., Warnecke, D. C., François, I. E., Leipelt, M., Heinz, E., Ott, C., Zahringer, U., Thomma, B. P., Ferket, K. K., and Cammue, B. P. (2004). Defensins from insects and plants interact with fungal glucosylceramides. J. Biol. Chem. 279, 3900–3905.
Thorpe, G. W., Fong, C. S., Alic, N., Higgins, V. J., and Dawes, I. W. (2004). Cells have distinct mechanisms to maintain protection against different reactive oxygen species: oxidative-stress-response genes. Proc. Natl. Acad. Sci. U.S.A. 101, 6564–6569.
Vylkova, S., Nayyar, N., Li, W., and Edgerton, M. (2007). Human beta-defensins kill Candida albicans in an energy-dependent and salt-sensitive manner without causing membrane disruption. Antimicrob. Agents Chemother. 51, 154–161.
Keywords: plant defensin, Candida albicans, Saccharomyces cerevisiae, mitochondria, apoptosis, mode of action
Citation: Aerts AM, Bammens L, Govaert G, Carmona-Gutierrez D, Madeo F, Cammue BPA and Thevissen K (2011) The antifungal plant defensin HsAFP1 from Heuchera sanguinea induces apoptosis in Candida albicans. Front. Microbio. 2:47. doi: 10.3389/fmicb.2011.00047
Received: 10 February 2011;
Accepted: 02 March 2011;
Published online: 16 March 2011.
Edited by:
Maurizio Del Poeta, Medical University of South Carolina, USAReviewed by:
Maurizio Del Poeta, Medical University of South Carolina, USAAna Traven, Monash Univerisity, Australia
Copyright: © 2011 Aerts, Bammens, Govaert, Carmona-Gutierrez, Madeo, Cammue and Thevissen. This is an open-access article subject to an exclusive license agreement between the authors and Frontiers Media SA, which permits unrestricted use, distribution, and reproduction in any medium, provided the original authors and source are credited.
*Correspondence: Karin Thevissen, Centre of Microbial and Plant Genetics, Kasteelpark Arenberg 20, B-3001 Heverlee, Belgium. e-mail:a2FyaW4udGhldmlzc2VuQGJpdy5rdWxldXZlbi5iZQ==
 An M. Aerts1
An M. Aerts1