Abstract
Bacterial persisters are phenotypic variants with extraordinary tolerances toward antibiotics. Persister survival has been attributed to inhibition of essential cell functions during antibiotic stress, followed by reversal of the process and resumption of growth upon removal of the antibiotic. Metabolism plays a critical role in this process, since it participates in the entry, maintenance, and exit from the persister phenotype. Here, we review the experimental evidence that demonstrates the importance of metabolism to persistence, highlight the successes and potential of targeting metabolism in the search for anti-persister therapies, and discuss the current methods and challenges to understand persister physiology.
Introduction
Bacterial cultures contain a small subpopulation of cells that cannot readily be killed by antibiotics (Bigger, 1944). These cells have been named persisters, and their existence can be detected from antibiotic kill curves, where the first, rapid killing regime represents the death of normal cells and the second, slower killing regime indicates the presence of persisters (Balaban et al., 2004; Kint et al., 2012). Further, when these survivors are cultured, they produce populations with antibiotic sensitivities identical to those of the original culture. This establishes persistence as a phenotypic trait, unique from antibiotic resistance where genetic determinants allow growth at higher antibiotic concentrations. Persisters are an important health concern because they are enriched in biofilms and thought to underlie the proclivity of biofilm infections to relapse following the conclusion of antibiotic therapy (Lewis, 2008, 2010). Persisters have proven to be difficult to analyze due to their transient nature, low abundance, and similarity to the viable but non-culturable (VBNC) cell-type (Roostalu et al., 2008; Orman and Brynildsen, 2013b). However, strong evidence, in the form of genetic- and microscopy-based data (Balaban et al., 2004; Lewis, 2010; Maisonneuve et al., 2013), exists to support that, while under antibiotic stress, persister tolerances are derived from inactivity of essential cell functions. While this is not always the case, as demonstrated in a study of isoniazid (a prodrug requiring activation) (Wakamoto et al., 2013), and dormancy is not essential for persistence prior to antibiotic stress (Orman and Brynildsen, 2013a), prolonged survival to the majority of antibiotics, in the absence of genetic mutations, requires inactivity of the antibiotic's primary target. To achieve and maintain this state, as well as reverse the process to repopulate environments, coordinated metabolic action is needed. Namely, metabolism would participate in cessation of essential functions, be needed to maintain culturability (e.g., sustain a minimal adenylate charge: [ATP + 0.5ADP]/[ATP + ADP + AMP] (Chapman et al., 1971), repair/resynthesize damaged proteins (Nystrom and Gustavsson, 1998)), and reactivate the cell during reawakening (Figure 1). We refer to this process as the persister metabolic program and summarize the accumulated evidence substantiating the importance of metabolism to the persister phenotype as well as current methods and challenges to studying the metabolism of these rare and transient phenotypic variants.
Figure 1
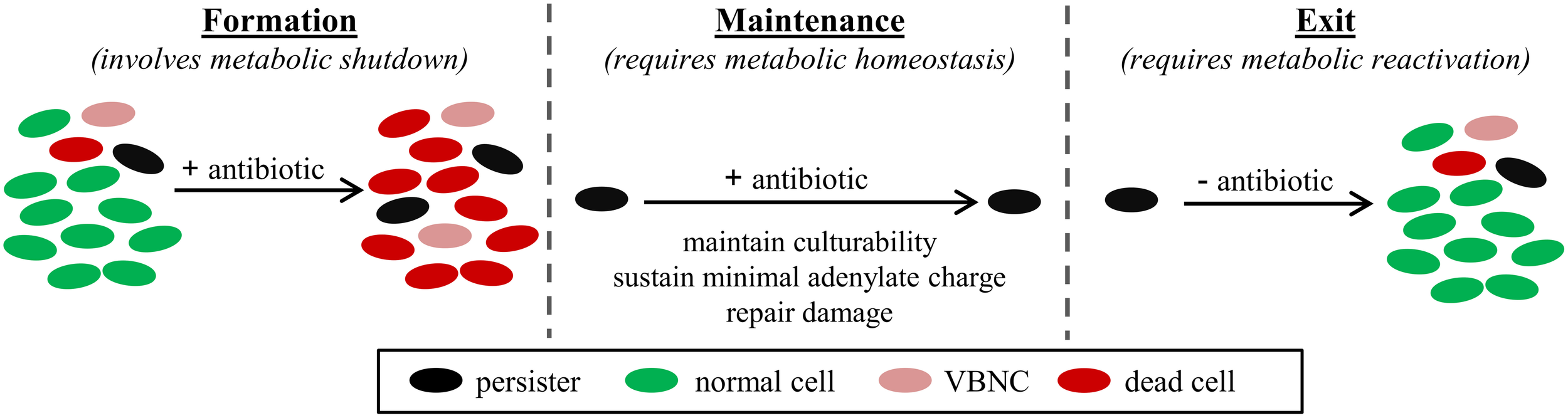
Persister metabolic program. Persisters can be pre-existing in a bacterial population (Balaban et al., 2004), formed in response to stress, such as stresses that activate the stringent response (Amato et al., 2013), and induced during antibiotic treatment (Dörr et al., 2009; Orman and Brynildsen, 2013a). Maintenance of the persister state for the duration of the antibiotic treatment requires temporary inhibition of essential cell functions; however, persisters must remain culturable, which requires a minimal adenylate charge to be sustained (Chapman et al., 1971) and damage to be repaired (Nystrom and Gustavsson, 1998). Upon removal of the antibiotic, persisters exit their tolerant state and give rise to a bacterial population of identical antibiotic susceptibility as the original population (Balaban et al., 2004).
Genomic studies identify metabolic genes as important to the persister phenotype
Perturbations to genes that encode enzymes or regulators of metabolism have frequently been found to alter persister levels (Table 1). In one of the initial genomic screens for persistence, a library was generated through digestion of the Escherichia coli chromosome, ligation of the fragments into plasmids, and transformation of the library into E. coli (Spoering et al., 2006). Upon successive rounds of ampicillin (AMP) treatment and culturing of survivors, a plasmid carrying glpD, encoding G3P-dehydrogenase that converts glycerol-3-phosphate (G3P) to dihydroxyacetone-phosphate (DHAP), was found to increase the abundance of persisters. Further analysis identified additional enzymes in G3P metabolism important for persistence to AMP, ofloxacin (OFL), and ciprofloxacin (CIP) (Table 1). The importance of G3P to E. coli persistence was further supported by a transposon mutant screen where glpD mutants were found to increase persistence after successive rounds of selection on LB-AMP agar (Girgis et al., 2012). This effect was attributed to elevated levels of methylglyoxal, a toxic compound derived from DHAP. Interestingly, these observations, where GlpD inactivation increased persistence, were opposite to those of Spoering and colleagues. However, we note that G3P is a highly connected metabolite, given its proximity to central metabolism, interaction with the quinone pool, and use as a precursor for phospholipid biosynthesis. Therefore, different assay conditions may explain the variable impacts on persistence.
Table 1
| Organism | Geneα | Gene function | Metabolic role | Mutation | Antibioticβ | Culture conditionsγ | Persistence outcome | References |
|---|---|---|---|---|---|---|---|---|
| E. coli | argE | Acetylornithine deacetylase | AA metabolism | Mutation | TIC, OFL | Biofilm | Increase | Bernier et al., 2013 |
| E. coli | argH | Argininosuccinate lyase | AA metabolism | Deletion | TIC, OFL | Biofilm | Increase | Bernier et al., 2013 |
| E. coli | aroE | Shikimate 5-dehydrogenase | AA metabolism | Mutation | TIC, OFL | Biofilm | Increase | Bernier et al., 2013 |
| E. coli | atpA | Fo/F1 ATP synthase subunit α | Energy production | Mutation | AMP | Plates | Increase | Girgis et al., 2012 |
| E. coli | atpF | Fo/F1 ATP synthase subunit B | Energy production | Mutation | AMP | Plates | Increase | Girgis et al., 2012 |
| E. coli | cysD | Sulfate adenylyltransferase subunit 2 | AA metabolism | Deletion | TIC | STAT | Increase | Bernier et al., 2013 |
| Deletion | TIC, OFL | Biofilm | Increase | Bernier et al., 2013 | ||||
| E. coli | dksA | Transcription regulation | Stringent response regulation | Deletion | OFL, CIP, STM, AMP | STAT, EXP | Decrease | Hansen et al., 2008 |
| E. coli | galU | Glucose-1-phosphate uridylyltransferase | Glycogen metabolism | Mutation | AMP | Plates | Increase | Girgis et al., 2012 |
| E. coli | glpABC | Anaerobic G3P dehydrogenase | G3P metabolism | Deletion | CIP | STAT | Decrease | Spoering et al., 2006 |
| E. coli | glpD | G3P dehydrogenase | G3P metabolism | Mutation | AMP | Plates | Increase | Girgis et al., 2012 |
| Deletion | CIP | STAT | Decrease | Spoering et al., 2006 | ||||
| Over-expression | AMP, OFL | EXP | Increase | Spoering et al., 2006 | ||||
| E. coli | hipA | Serine/threonine kinase | Stringent response regulation | Mutation | AMP, CYC, PHM | EXP, Plates | Increase | Moyed and Bertrand, 1983 |
| E. coli | hipB | Transcriptional repressor, antitoxin | Stringent response regulation | Mutation | AMP | Plates | Increase | Girgis et al., 2012 |
| E. coli | hisG | ATP phosphoribosyl-transferase | AA metabolism | Deletion | TIC, OFL | Biofilm | Increase | Bernier et al., 2013 |
| E. coli | ilvA | Threonine dehydratase | AA metabolism | Deletion | TIC, OFL | Biofilm | Increase | Bernier et al., 2013 |
| E. coli | ilvC | Ketol-acid reductoisomerase | AA metabolism | Mutation | TIC, OFL | Biofilm | Increase | Bernier et al., 2013 |
| E. coli | livJ | Leucine/isoleucine/valine transporter | AA transport | Mutation | AMP | Plates | Increase | Girgis et al., 2012 |
| E. coli | leuB | 3-Isopropylmalate dehydrogenase | AA metabolism | Mutation | TIC, OFL | Biofilm | Increase | Bernier et al., 2013 |
| E. coli | leuC | Isopropylmalate isomerase large subunit | AA metabolism | Mutation | TIC, OFL | Biofilm | Increase | Bernier et al., 2013 |
| Deletion | TIC | STAT | Increase | Bernier et al., 2013 | ||||
| Deletion | TIC, OFL | Biofilm | Increase | Bernier et al., 2013 | ||||
| E. coli | lysA | Diaminopimelate decarboxylase, PLP-binding | AA metabolism | Deletion | TIC | STAT | Increase | Bernier et al., 2013 |
| Deletion | TIC, OFL | Biofilm | Increase | Bernier et al., 2013 | ||||
| E. coli | metA | Homoserine O-succinyltransferase | AA metabolism | Deletion | TIC, OFL | Biofilm | Increase | Bernier et al., 2013 |
| E. coli | pheA | Fused chorismate mutase P/prephenate dehydratase | AA metabolism | Deletion | TIC, OFL | Biofilm | Increase | Bernier et al., 2013 |
| E. coli | phoU | Pho operon repressor | Phosphate metabolism | Mutation | AMP, NOR, PZA, AMP/GEN | STAT, EXP | Decrease | Li and Zhang, 2007 |
| Deletion | AMP, NOR | STAT, EXP | Decrease | Li and Zhang, 2007 | ||||
| E. coli | proA | γ-Glutamyl phosphate reductase | AA metabolism | Mutation | TIC, OFL | Biofilm | Increase | Bernier et al., 2013 |
| E. coli | proC | Pyrroline-5-carboxylate reductase | AA metabolism | Deletion | TIC, OFL | Biofilm | Increase | Bernier et al., 2013 |
| E. coli | sucB | Dihydrolipoamide acetyltransferase | Energy production | Deletion | AMP, GEN | STAT, EXP | Decrease | Ma et al., 2010 |
| E. coli | tktA | Transketolase | Energy production | Mutation | AMP | Plates | Increase | Girgis et al., 2012 |
| E. coli | trpA | Tryptophan synthase subunit α | AA metabolism | Deletion | TIC | Biofilm | Increase | Bernier et al., 2013 |
| E. coli | tyrA | Fused chorismate mutase T/prephenate dehydratase | AA metabolism | Deletion | TIC, OFL | Biofilm | Increase | Bernier et al., 2013 |
| E. coli | ubiF | 2-Octaprenyl-3-methyl-6-methoxy-1,4-benzoquinone hydroxylase | Energy production | Deletion | AMP, GEN | STAT, EXP | Decrease | Ma et al., 2010 |
| E. coli | ygfA | 5-Formyltetrahydrofolate cyclo-ligase | Coenzyme biosynthesis | Deletion | OFL, CIP, STM, AMP | STAT, EXP | Decrease | Hansen et al., 2008 |
| P. aeruginosa | PA14_13680 | Putative short-chain dehydrogenase | Unknown | Mutation | OFL | STAT | Increase | De Groote et al., 2009 |
| P. aeruginosa | PA14_17880 | Acetyl-CoA acetyltransferase | Fatty acid and phospholipid metabolism | Mutation | OFL | STAT | Decrease | De Groote et al., 2009 |
| P. aeruginosa | PA4115 | Lysine decarboxylase | AA metabolism | Mutation | CB | EXP, Plates | Increase | Manuel et al., 2010 |
| Deletion | CB, TIC | EXP | Increase | Manuel et al., 2010 | ||||
| P. aeruginosa | pheA | Prephenate dehydratase | AA metabolism | Mutation | OFL | STAT | Increase | De Groote et al., 2009 |
| P. aeruginosa | spuC | Putrescine aminotransferase | Polyamine metabolism | Mutation | OFL | STAT | Decrease | De Groote et al., 2009 |
| P. aeruginosa | ycgM | Putative fumarylaceto-acetate hydrolase family protein | Secondary metabolite biosynthesis, transport, and catabolism | Mutation | OFL | STAT | Increase | De Groote et al., 2009 |
| S. mutans | fruA | Fructan hydrolase | Carbohydrate metabolism | Fragment over-expression | OFL | STAT | Decrease | Leung and Lévesque, 2012 |
| S. mutans | pfl | Pyruvate formate-lyase | Energy production | Fragment over-expression | OFL | STAT | Increase | Leung and Lévesque, 2012 |
| S. mutans | scrA/scrB | Sucrose-specific IIABC PTS component/Sucrose-6-phosphate hydrolase | Carbohydrate metabolism | Promoter over-expression | OFL | STAT | Increase | Leung and Lévesque, 2012 |
| S. mutans | scrR | Sucrose-PTS operon repressor | Carbohydrate metabolism | Deletion | OFL | STAT | Increase | Leung and Lévesque, 2012 |
| S. mutans | SMU.1278 | Putative phosphoglycolate phosphatase | Unknown | Fragment over-expression | OFL | STAT | Increase | Leung and Lévesque, 2012 |
Metabolism-associated genes identified through genomic studies to influence persistence.
αGenes listed in this table are those associated with metabolism that were found to influence persistence in studies that included a genomic screen. Genes not directly connected to metabolism are not listed here, and metabolic genes identified in non-genomic studies are also not presented. βAntibiotic abbreviations: ampicillin (AMP), carbenicillin (CB), ciprofloxacin (CIP), cycloserine (CYC), gentamicin (GEN), norfloxacin (NOR), ofloxacin (OFL), phosphomycin (PHM), pyrazinamide (PZA), streptomycin (STM), ticarcillin (TIC). γCulture conditions describe the bacterial growth state at the time of antibiotic exposure: exponential (EXP), stationary (STAT), agar plates (Plates), biofilm.
Beyond G3P, genomic studies have found that mutations perturbing amino acid (AA) metabolism significantly influence persistence (Table 1). Screening of an E. coli transposon library for persistence to ticarcillin (TIC) or OFL identified 18 mutants with increased persister levels, and of those, 16 mapped to genes involved in AA biosynthesis (Bernier et al., 2013). Pseudomonas aeruginosa screens have also uncovered disruptions in AA metabolism as important to persistence. Mutation of PA4115, a putative lysine decarboxylase, was found to increase persistence to carbenicillin (CB) (Manuel et al., 2010), whereas mutation of pheA, which is also involved in AA metabolism, was found to increase persistence to OFL (De Groote et al., 2009). These studies suggest that AA metabolism is a critical mediator of persistence, and as one would expect, the stringent response, a major metabolic regulatory system controlled by ppGpp and its transcriptional partner DksA, also mediates persistence (Korch et al., 2003; Viducic et al., 2006; Fung et al., 2010; Nguyen et al., 2011; Amato et al., 2013; Maisonneuve et al., 2013). This influence was also detected in a screen where ΔdksA was found to produce far fewer persisters toward OFL (Hansen et al., 2008).
The third major metabolic system that has been shown to impact persistence is energy metabolism. A screen of an E. coli transposon library found that deactivation of phoU reduced persistence (Li and Zhang, 2007). PhoU is a negative regulator of the phosphate operon, and its inactivation led to a hyperactive metabolic state. In a screen of the Keio collection for AMP persistence, ΔsucB and ΔubiF were found to produce lower persister levels (Ma et al., 2010). SucB participates in the TCA cycle, whereas UbiF is an enzyme in ubiquinone biosynthesis, and deactivation of either of these genes leads to deficient energy production. Interestingly, these studies point to both metabolic hyperactivity and inhibition as methods to reduce persistence. One interpretation of these results could be that metabolic hyperactivity reduces entry into the persister state, whereas inhibition of energy production prevents exit from the phenotype. Regardless, energy generation appears to be a critical process to the persister metabolic program.
Collectively, these studies have provided a wealth of evidence on the importance of metabolism to bacterial persistence, even though they have sampled only a fraction of the mutational landscape. The details of how each genetic perturbation affects entry into, maintenance of, or exit from the persister state largely remains to be elucidated; however, it is clear that G3P, AA metabolism, and energy production are all important to defining persistence in a bacterial population.
Persister levels depend on the nutritional environment
In addition to genetic evidence, the importance of metabolism to persistence has been supported by the impact of nutrient availability on persister levels. The most comprehensive investigation in this regard explored how the absence of AAs, glucose, ammonium, phosphate, and nucleobases altered persistence to AMP, OFL, and gentamicin (GEN) in E. coli (Fung et al., 2010). This study concluded that AA deprivation often increases persistence, mirroring the results from genomic screens that found mutations in AA metabolism to enhance persistence (Table 1). In a study of persister awakening, the number of E. coli persisters to AMP and norfloxacin (NOR) were found to be higher when the same stationary-phase culture was inoculated into media unable to support rapid growth resumption (minimal glycerol) in comparison to media with rapid regrowth (LB and minimal glucose) (Joers et al., 2010). Similarly, E. coli biofilms have been reported to exhibit higher tolerance to OFL or TIC in fresh media lacking glucose, in comparison to controls with glucose (Bernier et al., 2013). Further support derives from the numerous studies that have shown that nutrient-limited stationary phase and biofilm cultures produce higher persister levels than their exponentially growing counterparts (Spoering and Lewis, 2001; Keren et al., 2004a; Lechner et al., 2012; Bernier et al., 2013). However, it is important to note that high density phenotypes such as quorum signaling may also contribute to persistence in such populations (Möker et al., 2010; Vega et al., 2012).
Taken together, these studies demonstrate that the nutritional environment directly influences persistence, suggesting a central role for metabolism in the persistence phenotype. Further, the mechanisms by which these nutritional stresses enhance persistence have been investigated, and ppGpp has been found to be a key mediator of this process.
ppGpp, the metabolite controller of persistence
ppGpp and the transcriptional regulator DksA are global regulators of metabolism (Traxler et al., 2006; Dalebroux and Swanson, 2012) that are critical mediators of persistence (Korch et al., 2003; Hansen et al., 2008; Amato et al., 2013; Bokinsky et al., 2013; Germain et al., 2013; Maisonneuve et al., 2013). In E. coli, AA limitation stimulates the ribosome-associated RelA to synthesize ppGpp, whereas various stress conditions, such as carbon (Xiao et al., 1991) and fatty acid starvation (Seyfzadeh et al., 1993), stimulate ppGpp synthesis from the cytoplasmic SpoT, which also encodes the sole ppGpp hydrolase. In conjunction with DksA, ppGpp interacts with RNA polymerase to inhibit transcription from stable ribosomal RNA promoters, while simultaneously upregulating transcription of AA biosynthesis and stress response genes (Potrykus and Cashel, 2008; Dalebroux and Swanson, 2012). ppGpp was initially associated with persistence through hipA7, a toxin mutant that required ppGpp for its elevated persister phenotype (Korch et al., 2003). Recent work on the native HipA has also shown that its impact on persistence requires ppGpp (Bokinsky et al., 2013; Germain et al., 2013). ppGpp can also increase persistence through its inhibition of exopolyphosphatase (ppx), a modulator of the antitoxin degrading Lon protease (Maisonneuve et al., 2013). Additionally, we have demonstrated that RelA, SpoT, and DksA mediate persister formation in response to carbon source transitions (Amato et al., 2013). In particular, we found that the ppGpp biochemical network can act as a metabolic toxin–antitoxin module, where ppGpp is the metabolite toxin and SpoT is its enzymatic antitoxin. We demonstrated that increased ppGpp levels resulted in growth arrest and increased persistence, which could be reverted by SpoT coexpression, and using a mathematical model, we showed that the ppGpp biochemical network can exhibit bistability, where one subpopulation corresponds to normal cells (low ppGpp) and the other to persisters (high ppGpp). Interestingly, RelA–SpoT also demonstrate the prototypical conditional essentiality of a classical toxin–antitoxin system, where the toxin (relA) can be deleted, but the antitoxin (spoT) can only be removed in a strain without the toxin. In addition to E. coli, the stringent response has been shown to impact persistence in other organisms as well. In P. aeruginosa, RelA, SpoT, and DksA have all been found to impact persistence (Viducic et al., 2006; Nguyen et al., 2011), whereas in Mycobacterium tuberculosis, ppGpp was required for long term survival in an in vitro starvation and murine model (Primm et al., 2000; Dahl et al., 2003). Further, the mycobacterial stringent response was shown to exhibit bistability (Ghosh et al., 2011), supporting the assertion that ppGpp is a possible source of phenotypic heterogeneity. In addition, in Staphylococcus aureus, ppGpp has been shown to mediate antibiotic tolerance in response to cell envelope stress (Geiger et al., 2014).
These studies demonstrate the importance of the stringent response to persistence and highlight a prevalent mechanism by which metabolic stress can induce persistence. Considering this evidence supporting a central role for ppGpp in persistence, it is attractive to propose that an inhibitor of ppGpp synthesis, such as Relacin (Wexselblatt et al., 2012), or an activator of ppGpp hydrolysis could be effective therapeutics against persisters (Amato et al., 2013; Maisonneuve et al., 2013).
Persister metabolism as a source of elimination strategies
To date, only a limited number of methods to kill persisters have been discovered, and interestingly, persister metabolism plays a vital role in each approach. For example, the first method, which we co-developed, used metabolites to stimulate proton motive force (pmf) generation in persisters, enabling aminoglycoside (AG) transport and their subsequent killing of E. coli and S. aureus persisters (Allison et al., 2011b). The participation of persister metabolism was confirmed with genetic mutants and chemical inhibitors, and subsequent studies have found the method to also be effective against P. aeruginosa persisters (Barraud et al., 2013). Another method was identified by Kim and colleagues, who screened a chemical library and found that a chemical named C10 promoted fluoroquinolone killing of E. coli persisters by stimulating their reversion to a replicating state (Kim et al., 2011). In another study, the quorum-sensing (QS) inhibitor BF8 facilitated elimination of P. aeruginosa persisters when combined with CIP or tobramycin (TOB) (Pan et al., 2012). However, upon further analysis, it was discovered that the effect of BF8 was likely due to reactivation of metabolism rather than inhibition of QS. Interestingly, BF8 has also been found to reduce E. coli persister levels when combined with OFL, tetracycline (TET), TOB, or GEN (Pan et al., 2013). Recently, another method to eliminate S. aureus persisters was discovered by leveraging knowledge that energy levels are low in persisters (Conlon et al., 2013). Specifically, ADEP4, which renders the ClpP protease ATP-independent, led to non-specific protein degradation and death in energy-depleted persisters. Taken together, these studies show that targeting persister metabolism holds great potential for the elimination of these dangerous bacteria and that greater knowledge of persister metabolism will facilitate the discovery of novel therapeutic strategies.
Methods to measure persister metabolism
Given the potential of persister metabolism to yield anti-persister therapeutics, enhanced metabolic knowledge of these phenotypic variants is desirable. However, direct measurement of metabolites in persisters or assessment of their metabolic activities using conventional approaches, such as mass spectrometry and formazan-based colorimetric assays, are not currently possible due to isolation difficulties (Roostalu et al., 2008; Kint et al., 2012; Orman and Brynildsen, 2013a,b). Although several methods can provide persister-enriched samples (Keren et al., 2004b; Shah et al., 2006), such samples still contain many more other cell-types, such as normal cells and VBNCs, than persisters, and thus are of limited utility for metabolic measurements (Orman and Brynildsen, 2013b). Indeed, the major limitation to segregating persisters from a heterogeneous population is their similarity to VBNCs, which are often more highly abundant. Both VBNCs and persisters stain as live cells, harbor metabolic activity, and are non-growing under antibiotic stress. The only present difference between these subpopulations is that persisters resume growth on standard media, though we note that some VBNCs can regain culturability on non-standard media (Oliver, 2005) suggesting a role for the post-antibiotic environment in defining those cells that survive. Given these technical limitations, we have developed two methods to quantify persister metabolism. The first uses fluorescence activated cell sorting (FACS), a fluorescent measure of metabolic activity, and persistence assays to evaluate the metabolic status of persisters (Orman and Brynildsen, 2013a). This study, which provided the first direct measurement of persister metabolism, demonstrated that E. coli persisters largely contained low cellular reductase activity prior to antibiotic stress, confirming previous assumptions about the metabolic activity of the persister state. The second method leveraged the phenomenon by which specific metabolites enabled AG killing of persisters (Allison et al., 2011b). AG potentiation offered a rapid way to measure the breadth of persister metabolic activities (Orman and Brynildsen, 2013b), since the phenomenon relies on persister catabolism of nutrients for pmf generation. Persister metabolic activities are inferred from culturability on standard media, the distinguishing feature between VBNCs and persisters, thereby allowing investigation of persister metabolism even in the presence of VBNCs. This method enabled identification of nutrients metabolized by persisters to different antibiotics (AMP, OFL) during distinct growth stages (exponential, stationary), and thus allowed quantification of heterogeneity in persister metabolism. From these investigations, we demonstrated that glycerol and glucose are the most ubiquitously used carbon sources by various types of persisters, suggesting that the enzymes required for their catabolism are broadly available in persisters.
Challenges in the study of persister metabolism
The technical hurdles associated with isolation of persisters have hindered understanding of the persister metabolic program and other aspects of persister physiology, including their transcriptome and proteome content. However, FACS offers a technical opportunity to discriminate between VBNC and persister phenotypes. For instance, mixed populations of VBNCs and persisters can be segregated from antibiotic-treated cultures using FACS (Roostalu et al., 2008; Orman and Brynildsen, 2013b), and since VBNCs are much more abundant than persisters in these samples, VBNC physiology can be quantified and potential biomarkers to discriminate between these two cell-types can be found. Nevertheless, any distinguishing features may be condition-specific, since numerous mechanisms can contribute to persister formation (Dhar and McKinney, 2007; Allison et al., 2011a; Balaban, 2011). Indeed, activation of particular pathways will depend on the environment and antibiotic used (Li and Zhang, 2007; Luidalepp et al., 2011), and different formation mechanisms may be active in different growth stages, giving rise to persister heterogeneity, where multiple, distinct persister subpopulations, each with its own unique antibiotic tolerances, coexist in a bacterial culture (Allison et al., 2011a). As a result of heterogeneity, any isolation technique may only capture a fraction of the persisters present, yielding a limited sample of the persister population. Single-cell analysis techniques offer means to interrogate individual cells (Iino et al., 2012, 2013); however, the identification of persisters before they exit their non-replicative state is not presently possible. Perhaps a viable path forward is to study model persisters generated following the over-expression of genes that have been shown to increase persister levels (Korch and Hill, 2006; Vázquez-Laslop et al., 2006). Quantifying metabolic changes in these model systems may provide insight into the physiology and metabolic capabilities of different types of persisters (Bokinsky et al., 2013).
Conclusion
Persisters embody a medically important bacterial phenotype that relies on metabolism to establish and maintain a dormant, tolerant state during antibiotic stress, and exit that state upon removal of antibiotics (Figure 1). Considerable experimental evidence has accumulated substantiating the importance of metabolism to persistence, and the participation of metabolism in current persister eradication methods provides a convincing argument that enhanced knowledge of the persister metabolic program will accelerate the discovery of additional elimination strategies. However, isolation difficulties impede progress in the understanding of persister physiology, including metabolism. Two potential paths forward are to improve isolation techniques by studying the differences between persisters and VBNCs and to use model persisters to define the breadth and landscape of the persister metabolic program.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Statements
Acknowledgments
Support was provided by the Department of the Army under award number W81XWH-12-2-0138 and the NIAID of the NIH under award number R21AI105342. The content is solely the responsibility of the authors and does not necessarily represent the official views of the funding agencies.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
1
AllisonK. R.BrynildsenM. P.CollinsJ. J. (2011a). Heterogeneous bacterial persisters and engineering approaches to eliminate them. Curr. Opin. Microbiol. 14, 593–598. 10.1016/j.mib.2011.09.002
2
AllisonK. R.BrynildsenM. P.CollinsJ. J. (2011b). Metabolite-enabled eradication of bacterial persisters by aminoglycosides. Nature473, 216–220. 10.1038/nature10069
3
AmatoS. M.OrmanM. A.BrynildsenM. P. (2013). Metabolic control of persister formation in Escherichia coli. Mol. Cell50, 475–487. 10.1016/j.molcel.2013.04.002
4
BalabanN. Q. (2011). Persistence: mechanisms for triggering and enhancing phenotypic variability. Curr. Opin. Genet. Dev. 21, 768–775. 10.1016/j.gde.2011.10.001
5
BalabanN. Q.MerrinJ.ChaitR.KowalikL.LeiblerS. (2004). Bacterial persistence as a phenotypic switch. Science305, 1622–1625. 10.1126/science.1099390
6
BarraudN.BusonA.JarolimekW.RiceS. A. (2013). Mannitol enhances antibiotic sensitivity of persister bacteria in Pseudomonas aeruginosa biofilms. PLoS ONE8:e84220. 10.1371/journal.pone.0084220
7
BernierS. P.LebeauxD.DefrancescoA. S.ValomonA.SoubigouG.CoppéeJ.-Y.et al. (2013). Starvation, together with the SOS response, mediates high biofilm-specific tolerance to the fluoroquinolone ofloxacin. PLoS Genet. 9:e1003144. 10.1371/journal.pgen.1003144
8
BiggerJ. W. (1944). The bactericidal action of penicillin on Staphylococcus pyogenes. Ir. J. Med. Sci. 19, 553–568. 10.1007/BF02948386
9
BokinskyG.BaidooE. E.AkellaS.BurdH.WeaverD.Alonso-GutierrezJ.et al. (2013). HipA-triggered growth arrest and β-lactam tolerance in Escherichia coli are mediated by RelA-dependent ppGpp synthesis. J. Bacteriol. 195, 3173–3182. 10.1128/JB.02210-12
10
ChapmanA. G.FallL.AtkinsonD. E. (1971). Adenylate energy charge in Escherichia coli during growth and starvation. J. Bacteriol. 108, 1072–1086.
11
ConlonB. P.NakayasuE. S.FleckL. E.LafleurM. D.IsabellaV. M.ColemanK.et al. (2013). Activated ClpP kills persisters and eradicates a chronic biofilm infection. Nature503, 365–370. 10.1038/nature12790
12
DahlJ. L.KrausC. N.BoshoffH. I. M.DoanB.FoleyK.AvarbockD.et al. (2003). The role of RelMtb-mediated adaptation to stationary phase in long-term persistence of Mycobacterium tuberculosis in mice. Proc. Natl. Acad. Sci. U.S.A. 100, 10026–10031. 10.1073/pnas.1631248100
13
DalebrouxZ. D.SwansonM. S. (2012). ppGpp: magic beyond RNA polymerase. Nat. Rev. Microbiol. 10, 203–212. 10.1038/nrmicro2720
14
De GrooteV. N.VerstraetenN.FauvartM.KintC. I.VerbeeckA. M.BeullensS.et al. (2009). Novel persistence genes in Pseudomonas aeruginosa identified by high-throughput screening. FEMS Microbiol. Lett. 297, 73–79. 10.1111/j.1574-6968.2009.01657.x
15
DharN.McKinneyJ. D. (2007). Microbial phenotypic heterogeneity and antibiotic tolerance. Curr. Opin. Microbiol. 10, 30–38. 10.1016/j.mib.2006.12.007
16
DörrT.LewisK.VulićM. (2009). SOS response induces persistence to fluoroquinolones in Escherichia coli. PLoS Genet. 5:e1000760. 10.1371/journal.pgen.1000760
17
FungD. K. C.ChanE. W. C.ChinM. L.ChanR. C. Y. (2010). Delineation of a bacterial starvation stress response network which can mediate antibiotic tolerance development. Antimicrob. Agents Chemother. 54, 1082–1093. 10.1128/AAC.01218-09
18
GeigerT.KastleB.GrataniF.GoerkeC.WolzC. (2014). Two small (p)ppGpp synthases in Staphylococcus aureus mediate tolerance against cell envelope stress conditions. J. Bacteriol. 196, 894–902. 10.1128/JB.01201-13
19
GermainE.Castro-RoaD.ZenkinN.GerdesK. (2013). Molecular mechanism of bacterial persistence by HipA. Mol. Cell52, 248–254. 10.1016/j.molcel.2013.08.045
20
GhoshS.SurekaK.GhoshB.BoseI.BasuJ.KunduM. (2011). Phenotypic heterogeneity in mycobacterial stringent response. BMC Syst. Biol. 5:18. 10.1186/1752-0509-5-18
21
GirgisH. S.HarrisK.TavazoieS. (2012). Large mutational target size for rapid emergence of bacterial persistence. Proc. Natl. Acad. Sci. U.S.A. 109, 12740–12745. 10.1073/pnas.1205124109
22
HansenS.LewisK.VulicM. (2008). Role of global regulators and nucleotide metabolism in antibiotic tolerance in Escherichia coli. Antimicrob. Agents Chemother. 52, 2718–2726. 10.1128/AAC.00144-08
23
IinoR.HayamaK.AmezawaH.SakakiharaS.KimS. H.MatsumonoY.et al. (2012). A single-cell drug efflux assay in bacteria by using a directly accessible femtoliter droplet array. Lab Chip12, 3923–3929. 10.1039/c2lc40394c
24
IinoR.MatsumotoY.NishinoK.YamaguchiA.NojiH. (2013). Design of a large-scale femtoliter droplet array for single-cell analysis of drug-tolerant and drug-resistant bacteria. Front. Microbiol. 4:300. 10.3389/fmicb.2013.00300
25
JoersA.KaldaluN.TensonT. (2010). The frequency of persisters in Escherichia coli reflects the kinetics of awakening from dormancy. J. Bacteriol. 192, 3379–3384. 10.1128/JB.00056-10
26
KerenI.KaldaluN.SpoeringA.WangY. P.LewisK. (2004a). Persister cells and tolerance to antimicrobials. FEMS Microbiol. Lett. 230, 13–18. 10.1016/S0378-1097(03)00856-5
27
KerenI.ShahD.SpoeringA.KaldaluN.LewisK. (2004b). Specialized persister cells and the mechanism of multidrug tolerance in Escherichia coli. J. Bacteriol. 186, 8172–8180. 10.1128/JB.186.24.8172-8180.2004
28
KimJ. S.HeoP.YangT. J.LeeK. S.ChoD. H.KimB. T.et al. (2011). Selective killing of bacterial persisters by a single chemical compound without affecting normal antibiotic-sensitive cells. Antimicrob. Agents Chemother. 55, 5380–5383. 10.1128/AAC.00708-11
29
KintC. I.VerstraetenN.FauvartM.MichielsJ. (2012). New-found fundamentals of bacterial persistence. Trends Microbiol. 20, 577–585. 10.1016/j.tim.2012.08.009
30
KorchS. B.HendersonT. A.HillT. M. (2003). Characterization of the hipA7 allele of Escherichia coli and evidence that high persistence is governed by (p)ppGpp synthesis. Mol. Microbiol. 50, 1199–1213. 10.1046/j.1365-2958.2003.03779.x
31
KorchS. B.HillT. M. (2006). Ectopic overexpression of wild-type and mutant hipA genes in Escherichia coli: effects on macromolecular synthesis and persister formation. J. Bacteriol. 188, 3826–3836. 10.1128/JB.01740-05
32
LechnerS.LewisK.BertramR. (2012). Staphylococcus aureus persisters tolerant to bactericidal antibiotics. J. Mol. Microbiol. Biotechnol. 22, 235–244. 10.1159/000342449
33
LeungV.LévesqueC. M. (2012). A stress-inducible quorum-sensing peptide mediates the formation of persister cells with noninherited multidrug tolerance. J. Bacteriol. 194, 2265–2274. 10.1128/JB.06707-11
34
LewisK. (2008). Multidrug tolerance of biofilms and persister cells. Curr. Top. Microbiol. Immunol. 322, 107–131. 10.1007/978-3-540-75418-3_6
35
LewisK. (2010). Persister Cells. Annu. Rev. Microbiol. 64, 357–372. 10.1146/annurev.micro.112408.134306
36
LiY.ZhangY. (2007). PhoU is a persistence switch involved in persister formation and tolerance to multiple antibiotics and stresses in Escherichia coli. Antimicrob. Agents Chemother. 51, 2092–2099. 10.1128/AAC.00052-07
37
LuidaleppH.JoersA.KaldaluN.TensonT. (2011). Age of inoculum strongly influences persister frequency and can mask effects of mutations implicated in altered persistence. J. Bacteriol. 193, 3598–3605. 10.1128/JB.00085-11
38
MaC.SimS.ShiW.DuL.XingD.ZhangY. (2010). Energy production genes sucB and ubiF are involved in persister survival and tolerance to multiple antibiotics and stresses in Escherichia coli. FEMS Microbiol. Lett. 303, 33–40. 10.1111/j.1574-6968.2009.01857.x
39
MaisonneuveE.Castro-CamargoM.GerdesK. (2013). (p)ppGpp controls bacterial persistence by stochastic induction of toxin-antitoxin activity. Cell154, 1140–1150. 10.1016/j.cell.2013.07.048
40
ManuelJ.ZhanelG. G.De KievitT. (2010). Cadaverine suppresses persistence to carboxypenicillins in Pseudomonas aeruginosa PAO1. Antimicrob. Agents Chemother. 54, 5173–5179. 10.1128/AAC.01751-09
41
MökerN.DeanC. R.TaoJ. (2010). Pseudomonas aeruginosa increases formation of multidrug-tolerant persister cells in response to quorum-sensing signaling molecules. J. Bacteriol. 192, 1946–1955. 10.1128/JB.01231-09
42
MoyedH. S.BertrandK. P. (1983). hipA, a newly recognized gene of Escherichia coli K-12 that affects frequency of persistence after inhibition of murein synthesis. J. Bacteriol. 155, 768–775.
43
NguyenD.Joshi-DatarA.LepineF.BauerleE.OlakanmiO.BeerK.et al. (2011). Active starvation responses mediate antibiotic tolerance in biofilms and nutrient-limited bacteria. Science334, 982–986. 10.1126/science.1211037
44
NystromT.GustavssonN. (1998). Maintenance energy requirement: what is required for stasis survival of Escherichia coli?Biochim. Biophys. Acta1365, 225–231. 10.1016/S0005-2728(98)00072-3
45
OliverJ. D. (2005). The viable but nonculturable state in bacteria. J. Microbiol. 43, 93–100.
46
OrmanM. A.BrynildsenM. P. (2013a). Dormancy is not necessary or sufficient for bacterial persistence. Antimicrob. Agents Chemother. 57, 3230–3239. 10.1128/AAC.00243-13
47
OrmanM. A.BrynildsenM. P. (2013b). Establishment of a method to rapidly assay bacterial persister metabolism. Antimicrob. Agents Chemother. 57, 4398–4409. 10.1128/AAC.00372-13
48
PanJ. C.BaharA. A.SyedH.RenD. C. (2012). Reverting antibiotic tolerance of Pseudomonas aeruginosa PAO1 persister cells by (Z)-4-bromo-5-(bromomethylene)-3-methylfuran-2(5H)-one. PLoS ONE7:e45778. 10.1371/journal.pone.0045778
49
PanJ. C.XieX.TianW.BaharA. A.LinN.SongF. C.et al. (2013). (Z)-4-Bromo-5-(bromomethylene)-3-methylfuran-2(5H)-one sensitizes Escherichia coli persister cells to antibiotics. Appl. Microbiol. Biotechnol. 97, 9145–9154. 10.1007/s00253-013-5185-2
50
PotrykusK.CashelM. (2008). (p)ppGpp: still magical?Annu. Rev. Microbiol. 62, 35–51. 10.1146/annurev.micro.62.081307.162903
51
PrimmT. P.AndersenS. J.MizrahiV.AvarbockD.RubinH.BarryC. E.3rd. (2000). The stringent response of Mycobacterium tuberculosis is required for long-term survival. J. Bacteriol. 182, 4889–4898. 10.1128/JB.182.17.4889-4898.2000
52
RoostaluJ.JõersA.LuidaleppH.KaldaluN.TensonT. (2008). Cell division in Escherichia coli cultures monitored at single cell resolution. BMC Microbiol. 8:68. 10.1186/1471-2180-8-68
53
SeyfzadehM.KeenerJ.NomuraM. (1993). spoT-dependent accumulation of guanosine tetraphosphate in response to fatty acid starvation in Escherichia coli. Proc. Natl. Acad. Sci. U.S.A. 90, 11004–11008. 10.1073/pnas.90.23.11004
54
ShahD.ZhangZ. G.KhodurskyA.KaldaluN.KurgK.LewisK. (2006). Persisters: a distinct physiological state of E. coli.BMC Microbiol. 6:53. 10.1186/1471-2180-6-53
55
SpoeringA. L.LewisK. (2001). Biofilms and planktonic cells of Pseudomonas aeruginosa have similar resistance to killing by antimicrobials. J. Bacteriol. 183, 6746–6751. 10.1128/JB.183.23.6746-6751.2001
56
SpoeringA. L.VulicM.LewisK. (2006). GlpD and PlsB participate in persister cell formation in Escherichia coli. J. Bacteriol. 188, 5136–5144. 10.1128/JB.00369-06
57
TraxlerM. F.ChangD.-E.ConwayT. (2006). Guanosine 3',5'-bispyrophosphate coordinates global gene expression during glucose-lactose diauxie in Escherichia coli. Proc. Natl. Acad. Sci. U.S.A. 103, 2374–2379. 10.1073/pnas.0510995103
58
Vázquez-LaslopN.LeeH.NeyfakhA. A. (2006). Increased persistence in Escherichia coli caused by controlled expression of toxins or other unrelated proteins. J. Bacteriol. 188, 3494–3497. 10.1128/JB.188.10.3494-3497.2006
59
VegaN. M.AllisonK. R.KhalilA. S.CollinsJ. J. (2012). Signaling-mediated bacterial persister formation. Nat. Chem. Biol. 8, 431–433. 10.1038/nchembio.915
60
ViducicD.OnoT.MurakamiK.SusilowatiH.KayamaS.HirotaK.et al. (2006). Functional analysis of spoT, relA and dksA genes on quinolone tolerance in Pseudomonas aeruginosa under nongrowing condition. Microbiol. Immunol. 50, 349–357. 10.1111/j.1348-0421.2006.tb03793.x
61
WakamotoY.DharN.ChaitR.SchneiderK.Signorino-GeloF.LeiblerS.et al. (2013). Dynamic persistence of antibiotic-stressed mycobacteria. Science339, 91–95. 10.1126/science.1229858
62
WexselblattE.Oppenheimer-ShaananY.KaspyI.LondonN.Schueler-FurmanO.YavinE.et al. (2012). Relacin, a novel antibacterial agent targeting the stringent response. PLoS Pathog. 8:e1002925. 10.1371/journal.ppat.1002925
63
XiaoH.KalmanM.IkeharaK.ZemelS.GlaserG.CashelM. (1991). Residual guanosine 3',5'-bispyrophosphate synthetic activity of relA null mutants can be eliminated by spoT null mutations. J. Biol. Chem. 266, 5980–5990.
Summary
Keywords
bacterial persistence, metabolism, antibiotic tolerance, ppGpp, nutrient environment
Citation
Amato SM, Fazen CH, Henry TC, Mok WWK, Orman MA, Sandvik EL, Volzing KG and Brynildsen MP (2014) The role of metabolism in bacterial persistence. Front. Microbiol. 5:70. doi: 10.3389/fmicb.2014.00070
Received
21 December 2013
Accepted
09 February 2014
Published
03 March 2014
Volume
5 - 2014
Edited by
Sebastien P. Faucher, McGill University, Canada
Reviewed by
Martin I. Voskuil, University of Colorado Denver, USA; Jill Zeilstra-Ryalls, Bowling Green State University, USA; Ray A. Larsen, Bowling Green State University, USA
Copyright
© 2014 Amato, Fazen, Henry, Mok, Orman, Sandvik, Volzing and Brynildsen.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Mark P. Brynildsen, Hoyt Laboratory, Department of Chemical and Biological Engineering, Princeton University, William Street, Princeton, NJ 08544, USA e-mail: mbrynild@princeton.edu
†These authors have contributed equally to this work and are listed in alphabetical order.
This article was submitted to Microbial Physiology and Metabolism, a section of the journal Frontiers in Microbiology.
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.