- 1College of Animal Science and Technology, Zhejiang A&F University, Lin'an, China
- 2Zhejiang Provincial Key Laboratory of Preventive Veterinary Medicine, Zhejiang University Institute of Preventive Veterinary Medicine, Hangzhou, China
Listeria monocytogenes, a food-borne pathogen, has the capacity to maintain intracellular pH (pHi) homeostasis in acidic environments, but the underlying mechanisms remain elusive. Here, we report a simple microplate-based fluorescent method to determine pHi of listerial cells that were prelabeled with the fluorescent dye carboxyfluorescein diacetate N-succinimidyl ester and subjected to acid stress. We found that L. monocytogenes responds differently among strains toward organic and inorganic acids to maintain pHi homeostasis. The capacity of L. monocytogenes to maintain pHi at extracellular pH 4.5 (pHex) was compromised in the presence of acetic acid and lactic acid, but not by hydrochloric acid and citric acid. Organic acids exhibited more inhibitory effects than hydrochloric acid at certain pH conditions. Furthermore, the virulent stains L. monocytogenes EGDe, 850658 and 10403S was more resistant to acidic stress than the avirulent M7 which showed a defect in maintaining pHi homeostasis. Deletion of sigB, a stress-responsive alternative sigma factor from 10403S, markedly altered intracellular pHi homeostasis, and showed a significant growth and survival defect under acidic conditions. Thus, this work provides new insights into bacterial survival mechanism to acidic stresses.
Introduction
Listeria monocytogenes is a Gram-positive foodborne pathogen that is responsible for severe and often life-threatening disease with high mortality (Vazquez-Boland et al., 2001; Corr and O'neill, 2009). L monocytogenes grows optimally in the pH ranging from 6.0 to 7.0 (Tessema et al., 2012). However, acidic environments are the common conditions encountered by listeria outside (e.g., acidic foods and soil) or inside the host (e.g., stomach and phagosomes of macrophages) (Cotter and Hill, 2003; Gray et al., 2006). This may have enabled L. monocytogenes to evolve a capability to grow over a wide range of pH from 4.3 to 9.4 (Te Giffel and Zwietering, 1999).
Organic acids are natural antimicrobials that have been widely used in the food industry to inhibit growth of important microbial pathogens such as Listeria monocytogenes and Escherichia coli (Carpenter and Broadbent, 2009; Otto et al., 2011). Protonated organic acids diffuse across cell membranes more freely than inorganic molecules, thus decreasing pHi of the cell due to the dissociated protons (Young and Foegeding, 1993; Tessema et al., 2012). However, L. monocytogenes apparently adapts a resistance to acidic stress through multiple mechanisms. For example, glutamate decarboxylase (GAD), which consumes intracellular protons by converting glutamate to γ-aminobutyrate (Cotter et al., 2001a; Karatzas et al., 2012), has been suggested as an alternative acid resistance system of L. monocytogenes for its survival in low pH foods (Cotter et al., 2001b). Nevertheless, ammonia produced through arginine deiminase (ADI) and agmatine deiminase (AgDI) systems was found to neutralize intracellular protons by releasing NH+4 to elevate cytoplasmic pH, thereby protecting L. monocytogenes from lethal acidic stresses aroused from extracellular environments (Ryan et al., 2009; Chen et al., 2011a; Cheng et al., 2013a,b).
L. monocytogenes could maintain its intracellular pH (pHi) within a narrow range of 7.6–8.0 when exposed to extracellular pH (pHex) beyond the range (Siegumfeldt et al., 1999; Budde and Jakobsen, 2000) by an unknown mechanism. Earlier reports showed that pHi of individual bacterial cells could be measured by fluorescent ratio imaging (FRIM) using a special microscope backed up by a particular software such as Metamorph (Budde and Jakobsen, 2000; Kastbjerg et al., 2009). In FRIM, the bacterial cells were labeled with the fluorescent probe 5-(6)-carboxyfluorescein diacetate N-succinimidyl ester (cFDA-SE). cFDA-SE is a non-fluorescence precursor that diffuses across the cell membrane. Once inside the cell, it is hydrolyzed by the intracellular esterases and converted into a fluorescent compound which exhibits varying fluorescence intensity dependent on pH only when excited at 490 nm, but not at 435 nm. Thus, the ratio of the emitted fluorescence from two excitations at 490 nm and 435 nm (R490/435) reflects the pHi that could be calculated (Budde and Jakobsen, 2000; Fang et al., 2006; Kastbjerg et al., 2009; Smigic et al., 2009). Pan et al. (2011) examined the pHi changes of cFDA-SE labeled lactic acid bacteria cells to chitosan treatment on the cuvette-based fluorometry where no curve-fitting was performed to quantify the intracellular pH (Pan et al., 2011).
Here, we report a more effective and simple high-throughput method to determine dynamic changes of pHi of different L. monocytogenes strains under different acidic conditions. This method was then used to examine the role of SigB in intracellular pH homeostasis upon acidic stress.
Materials and Methods
Bacterial Strains and Culture Conditions
Listeria monocytogenes lineage II (EGDe Glaser et al., 2001 and 10403S) and lineage III (M7 Chen et al., 2011b and 850658) strains were retrieved from glycerol stocks maintained at −80°C, and cultured in Brain Heart Infusion broth (BHI) (Oxoid, Hampshire, England) at 37°C. BHI broth media were adjusted with the stock solutions of hydrochloric acid (HA), acetic acid (AA), citric acid (CA), lactic acid (LA) and sodium hydroxide (NaOH) to the pH as indicated. All the pH-adjusted media were freshly made, sterilized by filtration through 0.22 μm polyethersulfone membrane filters (Millipore, Boston, USA). All chemicals were obtained from Sangon Biotech (Shanghai, China), Invitrogen (California, USA), or Sigma (St. Louis, USA) at the highest purity available.
Fluorescent Staining of L. monocytogenes Cells
Cell labeling was performed as described previously (Budde and Jakobsen, 2000). Briefly, L. monocytogenes strains were grown overnight at 37°C in BHI broth at pH 7.0 with shaking, and harvested by centrifugation at 5000 × g for 3 min and re-suspended to a final OD600 nm of 0.6 in sterile cold 10 mM potassium phosphate buffer (pH 7.4). The cells were stained with 10 μM 5-(6)-carboxyfluorescein diacetate N-succinimidyl ester (cFDA-SE, Invitrogen) and incubated at 37°C for 30 min. The cell suspension was centrifuged for 5 min at 10,000 g, resuspended in 50 mM potassium phosphate buffer (pH 6.0) containing 10 mM glucose, and energized at 30°C for 30 min. Subsequently, the cell suspension was centrifuged at 10,000 × g for 5 min and resuspended in 50 mM potassium phosphate buffer (pH 6.0) containing 10 mM glucose. The labeled bacteria were used immediately for the following pHi determination.
Intracellular pH Calibration Under Stresses by Organic and Inorganic Acids
In order to equilibrate the intracellular pH (pHi) and external pH (pHex) of listerial cells, ethanol (63%, v/v) was added to the stained cells to permeabilize for 30 min at 30°C(Budde and Jakobsen, 2000). Subsequently, the bacterial cells were harvested by centrifugation at 10,000 × g for 5 min and re-suspended in BHI medium with pH ranging from 5.5 to 8.0 (in 0.5 increments), adjusted by using HA, AA, CA, and LA, respectively. Fluorescence was measured by using the microplate fluorometric reader (Biotek Synergy H1, Winooski, USA). Fluorecent ratio490/435 was obtained by dividing fluorescence at 490 nm by that at 435 nm. The calibration curve was plotted by polynomial fitting between Ratio490/435 and pHi of the equilibrated cells corresponding to the pH ranging from 5.5 to 8.0, respectively. All data are reported as the mean of two independent experiments, each in triplicate wells.
Real-Time Measurement of Bacterial Intracellular pH Under Stresses by Organic and Inorganic Acids
To evaluate pHi dynamics of L. monocytogenes strains under stresses by different acids, the labeled cells were re-suspended in BHI broth, adjusted to pH 3.5, 4.5, and 5.5 with HA, AA, CA and LA, respectively, and incubated for 60 min at 37°C. The fluorescence intensity at 490 nm and 435 nm were respectively collected every 5 min, and the corresponding pHi values were determined according to the Ratio490/435 vs. pHi calibration curves of each strain under acidic environments as described above. The data are reported as the mean of two independent experiments, each in triplicate wells.
Growth of L. monocytogenes Under Organic and Inorganic Acidic Conditions
L. monocytogenes strains were grown overnight at 37°C in BHI broth at pH 7.0 with shaking. The cultures were collected by centrifugation at 5000 × g at 4°C, washed in PBS (10 mM, pH 7.4) and adjusted to 0.6 at OD600 nm. The bacteria were then diluted 1:50 in fresh BHI broth (pre-adjusted to pH 4.5 or 5.5 with HA, AA, CA, and LA, respectively), pipetted into microplate wells (each strain-treatment in triplicate wells) and incubated in a microplate reader at 37°C for 14 h for automatic measurement of kinetic growth at OD600 nm and 1-h interval.
Bacterial Survival in Lethal Acid Conditions
Overnight-grown L. monocytogenes strains 10403S, EGDe, 850658 and M7 were harvested by centrifugation at 5000 × g for 10 min at 4°C, and then washed once in PBS (10 mM, pH 7.4). The bacterial pellets were re-suspended in BHI broth (pre-adjusted to pH 3.5 by using HA, AA, CA and LA, respectively) and incubated for 60 min at 37°C. Similar experiments were employed for 30 min survival in the synthetic human gastric fluid [8.3 g proteose peptone (Oxoid), 3.5 g D-glucose, 2.05 g NaCl, 0.6 g KH2PO4, 0.11 g CaCl2, 0.37 g KCl, 0.05 g bile salts (Sigma), 0.1 g lysozyme and 13.3 mg pepsin (Sigma), all L−1; adjusted to pH 2.5 with HCl] as described previously (Cotter et al., 2001a; Cheng et al., 2013b). The survival bacterial cells were plated onto BHI agar after appropriate dilutions. The plates were incubated at 37°C for 24 h and survival rates are reported as the mean of three independent experiments, each performed in duplicate.
Construction of sigB Deletion Mutant
A homologous recombination strategy with SOE-PCR procedure was used for in-frame deletion of the full-length sigB (780 bp) from L. monocytogenes 10403S according to the protocol as described previously (Monk et al., 2008; Cheng et al., 2013b). The DNA fragments containing homologous arms upstream and downstream of sigB were obtained by PCR amplification using the SOE primers listed in Table 1. Transformants were screened as described previously (Monk et al., 2008; Cheng et al., 2013b). The resulting knockout mutant was verified by sequencing and designated as ΔsigB (Figure S1).
Statistical Analysis
All data were analyzed using the two-tailed Student's t-test with P < 0.05 as statistically significant or P < 0.01 as of marked statistical significance.
Results
cFDA-SE is a Stable Fluorescent Indicator to Measure Listerial Intracellular pH
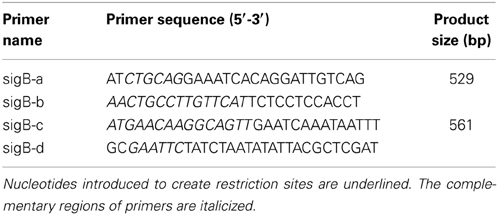
We sought to determine whether sigB was required for intracellular pH homeostasis of L. monocytogenes. To this end, it is critical to develop an accurate method to probe the intracellular pH of the bacterium. Therefore, calibration curves (Ratio490/435 vs. pHi) were plotted using ethanol-treated cells of L. monocytogenes under different acids (HA, AA, CA, and LA) in BHI broth for each strain (EGDe, 10403S, 850658, and M7) as indicated (Figure 1). Experimental data for each curve were found to be best fitted by a third degree polynomial equation with correlation indexes over 0.95. This indicates that the method developed in this study by using cFDA-SE as a fluorescent indicator to measure listerial pHi is stable and applicable to a wide range of strains. Thus, this method was further used in the following studies to determine pHi kinetics at various conditions to reveal acidic resistance of L. monocytogenes.
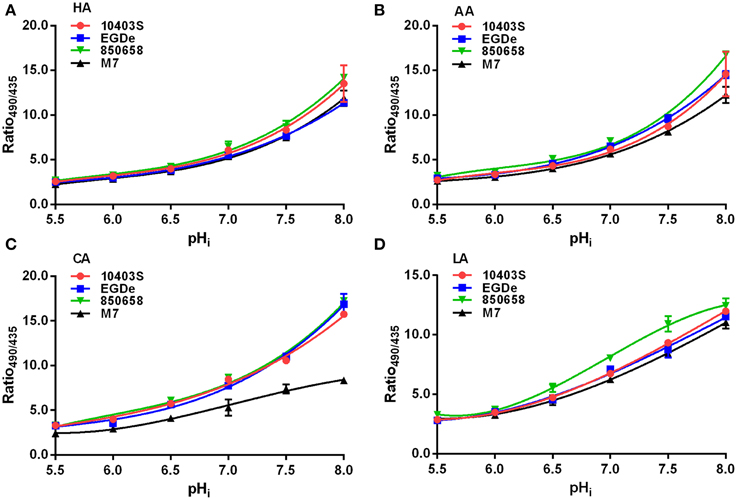
Figure 1. L. monocytogenes intracellular pH (pHi) determination. L. monocytogenes (10403S, EGDe, 850658, and M7) strains were exposed to organic and inorganic acids HA (A), AA (B), CA (C), and LA (D). The cells were equilibrated to pHex by incubating cell preparations with ethanol and resuspending in BHI medium at certain pHs as indicated. The cells were then stained with the fluorescence dye cFDA-SE and measured in a microplate reader at 490 nm and 435 nm respectively. The pHi was plotted against Ratio490/435. HA, Hydrochloric acid; AA, acetic acid; CA, citric acid; LA, lactic acid. Values are expressed as mean ± SD of two independent experiments, each in triplicate wells.
The Capability of L. monocytogenes to Maintain Intracellular pH Homeostasis Varies with Strains, Proton Donors and Extracellular pH
L. monocytogenes strains (virulent EGDe, 10403S, 850658, and avirulent M7) exhibited drastic variations in pHi kinetics in response to different acids. Under pH 5.5 conditions, the pHi of EGDe, 10403S, and 850658 strains increased rapidly after a sharp decline in the first 5 min, and maintained a steady state afterwards. However, the avirulent M7 failed to maintain its original intracellular pH when exposed to the four acids tested (Figure 2). The pHi at specific time point of M7 were significantly lower than the other three strains under the same acidic conditions (Figures 2, 3). This indicates that the capability of L. monocytogenes to maintain intracellular pH homeostasis varied among strains at certain pH conditions. Interestingly, all listerial strains failed to maintain pHi homeostasis at pHex 4.5 to the proton donor AA and LA, which was in contrast to HA and CA (Figure 3), indicating a lethal stress at this pH state induced by AA and LA. These suggest that organic and weak acids alleviate intracellular pH more effectively than inorganic and strong acids. In the case of pH 3.5, an unfavorable condition to all strains, the pHi kinetics of M7 descended more slowly than other strains (Figures 4A,C,D), indicating that the M7 strain might be more resistance to HA and CA than other virulent strains at pH 3.5, although this is unlikely to happen in natural or host environments.
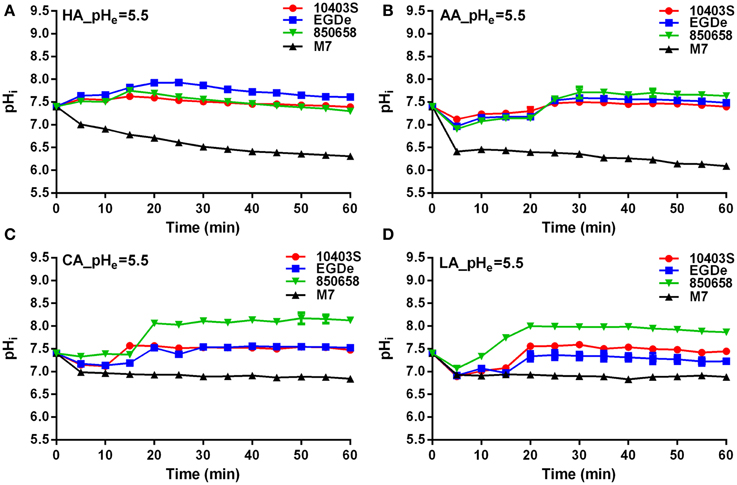
Figure 2. Kinetics of intracellular pH (pHi) of L. monocytogenes strains (10403S, EGDe, 850658, and M7) exposed to organic and inorganic acids at pH 5.5. L. monocytogenes strains were labeled and incubated for 60 min at 37°C in BHI broth with pH of 5.5 pre-adjusted by using HA (A), AA (B), CA (C), and LA (D), respectively. The fluorescence intensities at 490 and 435 nm were collected every 5 min, and the corresponding pHi values were determined according to the Ratio490/435 vs. pHi calibration curves (Figure 1). HA, hydrochloric acid; AA, acetic acid; CA, citric acid; LA, lactic acid. Values are expressed as mean ± SD of two independent experiments, each in triplicate wells.
Figure 3. Kinetics of intracellular pH (pHi) of L. monocytogenes strains (10403S, EGDe, 850658, and M7) exposed to organic and inorganic acids at pH 4.5. L. monocytogenes strains were labeled and incubated for 60 min at 37°C in BHI broth with pH of 4.5 pre-adjusted by using HA (A), AA (B), CA (C), and LA (D), respectively. The fluorescence intensities at 490 and 435 nm were respectively collected every 5 min, and the corresponding pHi values were determined according to the Ratio490/435 vs. pHi calibration curves (Figure 1). HA, hydrochloric acid; AA, acetic acid; CA, citric acid; LA, lactic acid. Values are expressed as mean ± SD of three replicates.
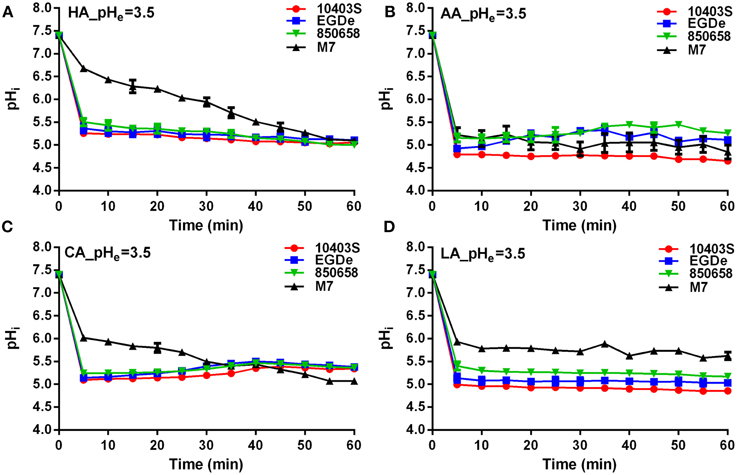
Figure 4. Kinetics of intracellular pH (pHi) of L. monocytogenes strains (10403S, EGDe, 850658, and M7) exposed to the organic and inorganic acids at pH 3.5. L. monocytogenes strains were labeled and incubated for 60 min at 37°C in BHI broth with pH of 3.5 pre-adjusted by using HA (A), AA (B), CA (C), and LA (D), respectively. The fluorescence intensities at 490 and 435 nm were respectively collected every 5 min, and the corresponding pHi values were determined according to the Ratio490/435 vs. pHi calibration curves (Figure 1). HA, hydrochloric acid; AA, acetic acid; CA, citric acid; LA, lactic acid. Values are expressed as mean ± SD of three replicates.
Growth and Survival of L. monocytogenes at Acidic Conditions Varied Among Strains
In the BHI broth pre-adjusted to pH 5.5 by organic or inorganic acids, the growth ability of the virulent strain 10403S was nearly equal to 850658, slightly higher than EGDe (the growth order: 10403S=850658≥EGDe>>M7) (Figure 5). The avirulent strain M7 of L. monocytogenes showed much slower growth. In the case of pH 4.5 HA, the growth order is 10403S>EGDe=850658>>M7 (Figure 6A). Under the pH 4.5 CA, M7 almost stopped growing, but the other three strains still showed a slow yet detectable growth (Figure 6C). All strains stopped growing when exposed to AA and LA at pH 4.5 (Figures 6B,D). These results indicate that organic acids exhibited much more inhibitory effects to listerial cells than hydrochloric acid at certain pH conditions. Furthermore, M7 was more sensitive to any kind of acids compared to other four strains, which was consistent to previous pHi kinetics (Figure 2).
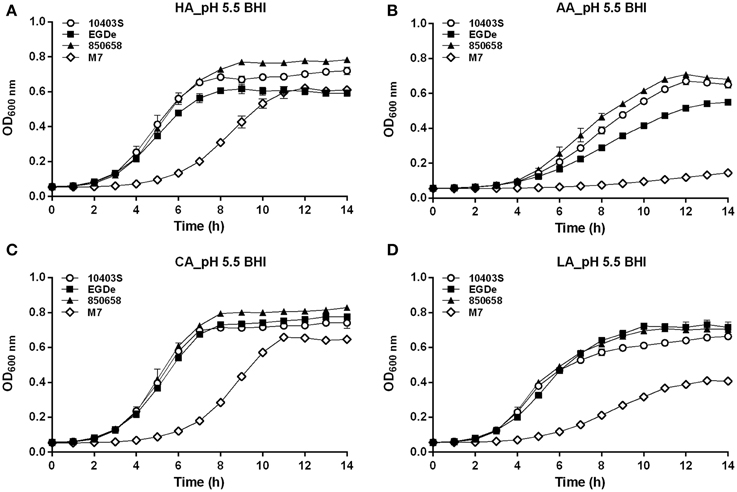
Figure 5. Growth of L. monocytogenes strains (10403S, EGDe, 850658, and M7) exposed to organic and inorganic acids at pH 5.5. L. monocytogenes strains were grown overnight at 37°C in BHI broth at pH 7.0. The cultures were collected, washed and the initial OD600 nm adjusted to 0.6. The bacteria were then incubated at 37°C for 14 h in fresh BHI broth with pH of 5.5 pre-adjusted by using HA (A), AA (B), CA (C), and LA (D), respectively. The kinetic growth OD600 nm was then measured with 1 h interval. All experiments were performed in triplicate. HA, hydrochloric acid; AA, acetic acid; CA, citric acid; LA, lactic acid. Values are expressed as mean ± SD of three replicates.
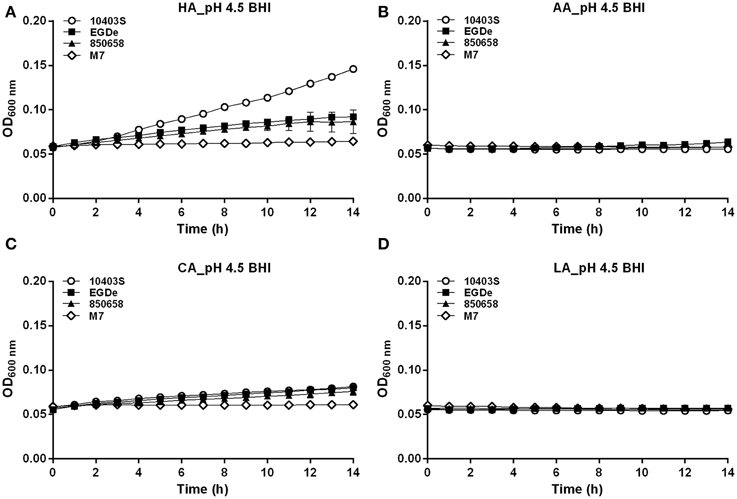
Figure 6. Growth of L. monocytogenes strains (10403S, EGDe, 850658, and M7) exposed to organic and inorganic acids at pH 4.5. L. monocytogenes strains were grown overnight at 37°C in BHI broth at pH 7.0. The cultures were collected, washed and the initial OD600 nm adjusted to 0.6. The bacteria were then incubated at 37°C for 14 h in fresh BHI broth with pH of 4.5 pre-adjusted by using HA (A), AA (B), CA (C) and LA (D), respectively. The OD600 nm was then measured at 1 h interval. All experiments were performed in triplicate. HA, hydrochloric acid; AA, acetic acid; CA, citric acid; LA, lactic acid. Values are expressed as mean ± SD of three replicates.
To further determine the acid tolerance of four different L. monocytogenes strains in the lethal acid conditions, the strain 10403S, EGDe, 850658, and M7 were exposed to HA, AA, CA, and LA at pH 3.5 and to synthetic gastric fluid at pH 2.5, respectively. The survival rate of the virulent strain 850658 at lethal acidic conditions was the highest for HA, CA, LA, and gastric fluid as compared to strains 10403S and EGDe, whereas M7 exhibited poorest survival (Figures 7A,B).
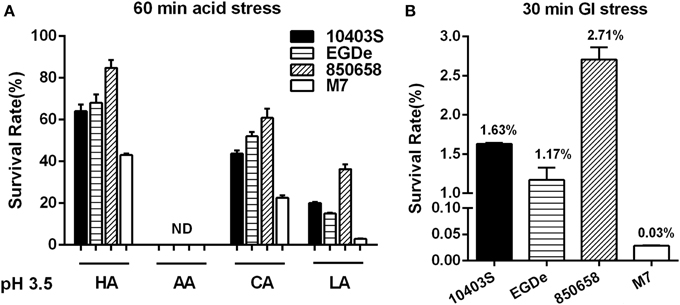
Figure 7. Survival of L. monocytogenes strains (10403S, EGDe, 850658, and M7) in organic and inorganic acids at pH 3.5 (A) and in synthetic human gastric fluid at pH 2.5 (B). Overnight-grown L. monocytogenes strains were harvested, washed and then incubated in BHI broth (pre-adjusted to pH 3.5 by using HA, AA, CA, and LA, respectively) for 60 min and in synthetic human gastric fluid (pH 2.5) for 30 min at 37°C. Values are expressed as mean ± SD of three independent experiments, each performed in duplicate.
SigB Contributes to pHi Homeostasis of L. moncytogenes at Acidic Conditions
SigB was previously shown to contribute to acid tolerance response in L. monocytogenes (Wiedmann et al., 1998). We hypothesized that SigB is involved in maintaining L. moncytogenes intracellular pH. Thus, the pHi dynamic of L. moncytogenes sigB deletion mutant was characterized by using the established method as described above. The pHi of 10403S increased and then maintained stable following initial decrease in 5 min when exposed to pHex of 4.5, while sigB deletion mutant also showed immediate initial decline but maintained at significant lower pHi than its parent strain from minutes 15 (P < 0.05, Figure 8A). At pHex 3.5, both the mutant and parent strains exhibited initial decline, and then maintained a lower level between 5 and 5.5 with the pHi of the parent strain staying higher with statistical difference at P < 0.05 (Figure 8A). In addition, the growth of L. monocytogenes was compromised in the absence of sigB in the sub-lethal pH of 4.8 with a marked difference starting from hour 4 to hour 12 (P < 0.05), but not in the neutral pH (Figure 8B). However, deletion of sigB exhibited a markedly decrease in survival compared to that of its parent strain in pH 2.5 BHI or in synthetic gastric fluid (Figure 8C).
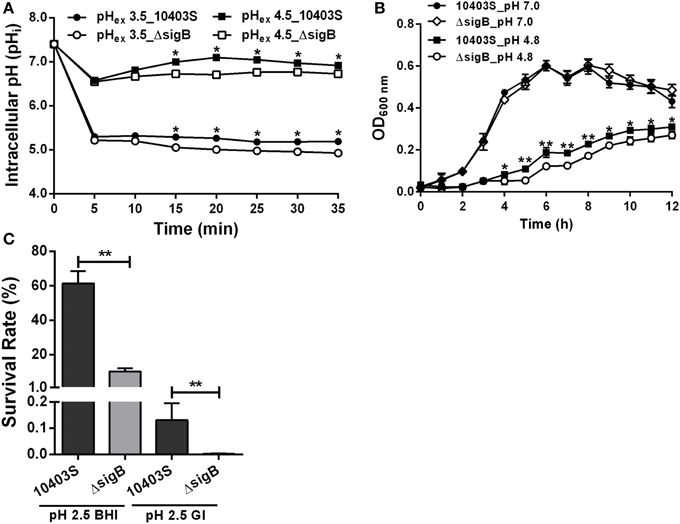
Figure 8. Profiles of intracellular pH (pHi) (A), growth (B) and survival (C) of L. monocytogenes sigB deletion mutant. HA, hydrochloric acid; AA, acetic acid; CA, citric acid; LA, lactic acid. All data are expressed as mean ± SD of three replicates. *P < 0.05 and **P < 0.01 for comparisons between the wild-type and mutant strains.
Discussion
L. monocytogenes survives or even grows in a wide range of environmental conditions (Begley et al., 2010). Tolerance to low pH is important for listeria to survive because listeria encounters acidic conditions in natural and food processing environments, and in host stomach and cellular phagosome as well (O'driscoll et al., 1996). L. monocytogenes resists acidic stresses by up-regulating expression of specific proteins that alter cell membrane structure, increasing the bacterial ability to maintain intracellular pH (Phan-Thanh, 1998; Otto et al., 2011). SigB, a sigma factor found in Gram-positive bacteria, plays a key role in acid tolerance (Wiedmann et al., 1998; Raengpradub et al., 2008; Oliver et al., 2010; Smith et al., 2013). However, different L. monocytogenes strains exhibit varying abilities of acid tolerance under acidic environments, which might contribute to varying pathogenicity among strains (Conte et al., 2000; Chen et al., 2011a). This could be seen from the strain M7, an avirulent strain (Chen et al., 2011b) that was found to be more sensitive to acidic stresses than the other virulent strains in terms of growth, survival or maintenance of intracellular homeostasis.
Here, we developed a simple and high-throughput approach to measure dynamic pHi changes of L. monocytogenes under acidic conditions by using the fluorescent dye cFDA-SE. As discussed previously, the pH range applicable to cFDA-SE dye was from 5.0 to 8.0 based on the fluorescence ratio-imaging method (FRIM) (Breeuwer et al., 1996; Budde and Jakobsen, 2000; Shabala et al., 2002; Giulitti et al., 2011). Particularly, cFDA-SE fluorescence is sensitive to pH ranging from 6.0 to 9.0. It is not sensitive enough for the FRIM to distinguish the Ratio490/435 between different pH gradients below 5.5 (Shabala et al., 2002). Nevertheless, the FRIM-based technique could still be extended to measure pHi of 5.0 under the lethal acidic stress with pHex of 3.5 (Shabala et al., 2002; Kastbjerg et al., 2009). Therefore, we believe that cFDA-SE is applicable at pH 5.0 and can be used to measure pHi even under the lethal acidic stress with pHex of 3.5, as used in this study. However, the microplate reader based method is easier and more applicable for high-throughput measurement than FRIM.
It was shown previously that protonated organic acids cross cell membrane more freely than inorganic acid molecules (Young and Foegeding, 1993; Ferreira et al., 2003). Once the disassociated protons enter inside cells, pHi of the cell decreases (Bearson et al., 1997). Phan-Thanh and Montagne previously showed that when acetic acid was used to create an extracellular pH of 3.5, intracellular pH was lower than that of HCl (internal pH of 3.34 with acetic acid compared to pH of 4.22 with HCl) (Phan-Thanh, 1998). This indicates that the dissociated organic anions inside kill cells if they are not expelled or consumed. Accumulation of anions could induce cell burst if increasing osmolality and pressure persist (Carpenter and Broadbent, 2009; Otto et al., 2011). The pHi of L. monocytogenes exposed to organic acids (acetic acid and lactic acid) is lower than that of cells exposed to HCl at the same external pH (Figures 2–4). The capacity to maintain pHi homeostasis was correlated to bacterial growth and survival at acidic conditions. Therefore, we conclude that the weak acid could be used as an alternative food preservative to prevent the growth of L. monocytogenes and extend food shelf-life as shown previously (Le Marc et al., 2002; Lues and Theron, 2012).
Under pH 5.5, the virulent strains 10403S, EGDe and 850658 exhibited higher capacity to maintain pHi homeostasis than the avirulent M7. Similar pHi kinetic changes were also found at pH 4.5. Christensen and Hutkins (1992) reported that listeria cells remained viable as long as the ΔpH could be balanced. Our results showed that pHex 3.5 is close to the limit of pHi homeostasis for listeria, which is consistent with the determined minimum pHex for listerial growth (Phan-Thanh et al., 2000; Le Marc et al., 2002; Shabala et al., 2006). Nevertheless, L. monocytogenes tends to have a buffering capacity in the cytosol around pH 5.5, which delays further pHi decrease (Shabala et al., 2006). However, this buffering capacity is a short-term protection and listeria requires proton pumps to keep long-term acid tolerance (Shabala et al., 2006). Shabala et al. measured a pHi of ≤ 5 after 2 h for L. monocytogenes incubated at pHex 3.0, and cells remained viable as these organisms recovered immediately and remained constant at pHi 7.3 when returning to pHex 6.0 (Shabala et al., 2002). The ability of listeria to maintain pHi homeostasis is critical for many cellular processes, such as DNA transcription, protein synthesis and enzyme activities in acidified environments (Kastbjerg et al., 2009).
SigB functions as a central regulator toward stress responses mainly through regulating expression of effector proteins (Smith et al., 2013; Ribeiro et al., 2014). When exposed to stresses, the cells respond through a regulatory cascade with the activation of σB followed by transcription of σB-regulated genes involved in resistance to temperature, osmotic, chemical and pH stresses (Van Schaik and Abee, 2005; Palmer et al., 2011). However, whether SigB is involved in intracellular pH regulations is still unknown. We demonstrated that deletion of sigB markedly compromised intracellular pH homeostasis, and led to a significantly impaired growth and survival when the mutant strain was exposed to acidic conditions (Figure 8). Further work is still required to illustrate the mechanisms underlying the σB mediated pHi homeostasis.
In summary, this study demonstrates that the microplate-based fluorometry is simple and high-throughput to measure dynamic changes of listerial pHi in response to acid stresses. The method should be applicable to other bacterial species or even mutant strains involved in regulation of acid stress. We have found that L. monocytogenes responds differently toward organic and inorganic acids to maintain pHi homeostasis. We further show that SigB plays an important role in maintaining intracellular pH homeostasis, thus providing an insight to reveal the underlying mechanisms of this central regulator in acid stress regulations in L. monocytogenes.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
This study was supported by National Natural Science Foundation of China (Nos. 31470179, 31402215), Zhejiang Provincial Natural Science Foundation (No. LQ14C010011) and ZAFU talents starting program (Nos. 2014FR073, 2011FR025), Scientific Research Foundation for the Returned Overseas Chinese Scholars, State Education Ministry to HS and the Graduate Inovation Project (No. 3122013240260). We thank Dr. Martin Wiedmann at Cornell University and John Bowman at University of Tasmania for kindly providing the shuttle plasmid pKSV7 and L. monocytogenes 850658, respectively. We thank anonymous reviewers and Wei Li at Michigan State University for critically reading the manuscript.
Supplementary Material
The Supplementary Material for this article can be found online at: http://www.frontiersin.org/journal/10.3389/fmicb.2015.00015/abstract
References
Bearson, S., Bearson, B., and Foster, J. W. (1997). Acid stress responses in enterobacteria. FEMS Microbiol. Lett. 147, 173–180. doi: 10.1111/j.1574-6968.1997.tb10238.x
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Begley, M., Cotter, P. D., Hill, C., and Ross, R. P. (2010). Glutamate decarboxylase-mediated nisin resistance in Listeria monocytogenes. Appl. Environ. Microbiol. 76, 6541–6546. doi: 10.1128/AEM.00203-10
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Breeuwer, P., Drocourt, J., Rombouts, F. M., and Abee, T. (1996). A novel method for continuous determination of the intracellular pH in bacteria with the internally conjugated fluorescent probe 5 (and 6-)-Carboxyfluorescein succinimidyl ester. Appl. Environ. Microbiol. 62, 178–183.
Budde, B. B., and Jakobsen, M. (2000). Real-time measurements of the interaction between single cells of Listeria monocytogenes and Nisin on a solid surface. Appl. Environ. Microbiol. 66, 3586–3591. doi: 10.1128/AEM.66.8.3586-3591.2000
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Carpenter, C. E., and Broadbent, J. R. (2009). External concentration of organic acid anions and pH: key independent variables for studying how organic acids inhibit growth of bacteria in mildly acidic foods. J. Food Sci. 74, R12–R15. doi: 10.1111/j.1750-3841.2008.00994.x
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Chen, J., Cheng, C., Xia, Y., Zhao, H., Fang, C., Shan, Y., et al. (2011a). Lmo0036, an ornithine and putrescine carbamoyltransferase in Listeria monocytogenes, participates in arginine deiminase and agmatine deiminase pathways and mediates acid tolerance. Microbiology 157, 3150–3161. doi: 10.1099/mic.0.049619-0
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Chen, J., Xia, Y., Cheng, C., Fang, C., Shan, Y., Jin, G., et al. (2011b). Genome sequence of the nonpathogenic Listeria monocytogenes serovar 4a strain M7. J. Bacteriol. 193, 5019–5020. doi: 10.1128/JB.05501-11
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Cheng, C., Chen, J., Fang, C., Xia, Y., Shan, Y., Liu, Y., et al. (2013a). Listeria monocytogenes aguA1, but not aguA2, encodes a functional agmatine deiminase: biochemical characterization of its catalytic properties and roles in acid tolerance. J. Biol. Chem. 288, 26606–26615. doi: 10.1074/jbc.M113.477380
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Cheng, C., Chen, J., Shan, Y., Fang, C., Liu, Y., Xia, Y., et al. (2013b). Listeria monocytogenes ArcA contributes to acid tolerance. J. Med. Microbiol. 62, 813–821. doi: 10.1099/jmm.0.055145-0
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Christensen, D. P., and Hutkins, R. W. (1992). Collapse of the proton motive force in Listeria monocytogenes caused by a bacteriocin produced by Pediococcus acidilactici. Appl. Environ. Microbiol. 58, 3312–3315.
Conte, M. P., Petrone, G., Di Biase, A. M., Ammendolia, M. G., Superti, F., and Seganti, L. (2000). Acid tolerance in Listeria monocytogenes influences invasiveness of enterocyte-like cells and macrophage-like cells. Microb. Pathog. 29, 137–144. doi: 10.1006/mpat.2000.0379
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Corr, S. C., and O'neill, L. A. (2009). Listeria monocytogenes infection in the face of innate immunity. Cell. Microbiol. 11, 703–709. doi: 10.1111/j.1462-5822.2009.01294.x
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Cotter, P. D., Gahan, C. G., and Hill, C. (2001a). A glutamate decarboxylase system protects Listeria monocytogenes in gastric fluid. Mol. Microbiol. 40, 465–475. doi: 10.1046/j.1365-2958.2001.02398.x
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Cotter, P. D., and Hill, C. (2003). Surviving the acid test: responses of gram-positive bacteria to low pH. Microbiol. Mol. Biol. Rev. 67, 429–453. doi: 10.1128/MMBR.67.3.429-453.2003
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Cotter, P. D., O'Reilly, K., and Hill, C. (2001b). Role of the glutamate decarboxylase acid resistance system in the survival of Listeria monocytogenes LO28 in low pH foods. J. Food Prot. 64, 1362–1368.
Fang, W., Budde, B. B., and Siegumfeldt, H. (2006). Leucocins 4010 from Leuconostoc carnosum cause a matrix related decrease in intracellular pH of Listeria monocytogenes. FEMS Microbiol. Lett. 258, 208–213. doi: 10.1111/j.1574-6968.2006.00219.x
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Ferreira, A., Sue, D., O'byrne, C. P., and Boor, K. J. (2003). Role of Listeria monocytogenes B in survival of lethal acidic conditions and in the acquired acid tolerance response. Appl. Environ. Microbiol. 69, 2692–2698. doi: 10.1128/AEM.69.5.2692-2698.2003
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Giulitti, S., Cinquemani, C., Quaranta, A., and Spilimbergo, S. (2011). Real time intracellular pH dynamics in Listeria innocua under CO2 and N2O pressure. J. Supercrit. Fluids 58, 385–390. doi: 10.1016/j.supflu.2011.07.012
Glaser, P., Frangeul, L., Buchrieser, C., Rusniok, C., Amend, A., Baquero, F., et al. (2001). Comparative genomics of Listeria species. Science 294, 849–852. doi: 10.1126/science.1063447
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Gray, M. J., Freitag, N. E., and Boor, K. J. (2006). How the bacterial pathogen Listeria monocytogenes mediates the switch from environmental Dr. Jekyll to pathogenic Mr. Hyde. Infect. Immun. 74, 2505–2512. doi: 10.1128/IAI.74.5.2505-2512.2006
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Karatzas, K. A., Suur, L., and O'byrne, C. P. (2012). Characterization of the intracellular glutamate decarboxylase system: analysis of its function, transcription, and role in the acid resistance of various strains of Listeria monocytogenes. Appl. Environ. Microbiol. 78, 3571–3579. doi: 10.1128/AEM.00227-12
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Kastbjerg, V. G., Nielsen, D. S., Arneborg, N., and Gram, L. (2009). Response of Listeria monocytogenes to disinfection stress at the single-cell and population levels as monitored by intracellular pH measurements and viable-cell counts. Appl. Environ. Microbiol. 75, 4550–4556. doi: 10.1128/AEM.02625-08
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Le Marc, Y., Huchet, V., Bourgeois, C. M., Guyonnet, J. P., Mafart, P., and Thuault, D. (2002). Modelling the growth kinetics of Listeria as a function of temperature, pH and organic acid concentration. Int. J. Food Microbiol. 73, 219–237. doi: 10.1016/S0168-1605(01)00640-7
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Lues, J. F., and Theron, M. M. (2012). Comparing organic acids and salt derivatives as antimicrobials against selected poultry-borne Listeria monocytogenes strains in vitro. Foodborne Pathog. Dis. 9, 1126–1129. doi: 10.1089/fpd.2012.1220
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Monk, I. R., Gahan, C. G., and Hill, C. (2008). Tools for functional postgenomic analysis of Listeria monocytogenes. Appl. Environ. Microbiol. 74, 3921–3934. doi: 10.1128/AEM.00314-08
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
O'driscoll, B., Gahan, C. G., and Hill, C. (1996). Adaptive acid tolerance response in Listeria monocytogenes: isolation of an acid-tolerant mutant which demonstrates increased virulence. Appl. Environ. Microbiol. 62, 1693–1698.
Oliver, H. F., Orsi, R. H., Wiedmann, M., and Boor, K. J. (2010). Listeria monocytogenes {sigma}B has a small core regulon and a conserved role in virulence but makes differential contributions to stress tolerance across a diverse collection of strains. Appl. Environ. Microbiol. 76, 4216–4232. doi: 10.1128/AEM.00031-10
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Otto, R. A., Beamer, S., Jaczynski, J., and Matak, K. E. (2011). The effect of using citric or acetic acid on survival of Listeria monocytogenes during fish protein recovery by isoelectric solubilization and precipitation process. J. Food Sci. 76, M579–M583. doi: 10.1111/j.1750-3841.2011.02340.x
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Palmer, M. E., Chaturongakul, S., Wiedmann, M., and Boor, K. J. (2011). The Listeria monocytogenes sigmaB regulon and its virulence-associated functions are inhibited by a small molecule. MBio 2:e00241-11. doi: 10.1128/mBio.00241-11
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Pan, C., Rezaei, H., and Soor, A. (2011). Chitosan disrupts membrane permeability of lactic acid bacteria. J. Exp. Microbiol. Immunol. 15, 7–14.
Phan-Thanh, L. (1998). Physiological and biochemical aspects of the acid survival of Listeria monocytogenes. J. Gen. Appl. Microbiol. 44, 183–191. doi: 10.2323/jgam.44.183
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Phan-Thanh, L., Mahouin, F., and Alige, S. (2000). Acid responses of Listeria monocytogenes. Int. J. Food Microbiol. 55, 121–126. doi: 10.1016/S0168-1605(00)00167-7
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Raengpradub, S., Wiedmann, M., and Boor, K. J. (2008). Comparative analysis of the sigma B-dependent stress responses in Listeria monocytogenes and Listeria innocua strains exposed to selected stress conditions. Appl. Environ. Microbiol. 74, 158–171. doi: 10.1128/AEM.00951-07
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Ribeiro, V. B., Mujahid, S., Orsi, R. H., Bergholz, T. M., Wiedmann, M., Boor, K. J., et al. (2014). Contributions of sigma(B) and PrfA to Listeria monocytogenes salt stress tinder food relevant conditions. Int. J. Food Microbiol. 177, 98–108. doi: 10.1016/j.ijfoodmicro.2014.02.018
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Ryan, S., Begley, M., Gahan, C. G., and Hill, C. (2009). Molecular characterization of the arginine deiminase system in Listeria monocytogenes: regulation and role in acid tolerance. Environ. Microbiol. 11, 432–445. doi: 10.1111/j.1462-2920.2008.01782.x
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Shabala, L., Budde, B., Ross, T., Siegumfeldt, H., and McMeekin, T. (2002). Responses of Listeria monocytogenes to acid stress and glucose availability monitored by measurements of intracellular pH and viable counts. Int. J. Food Microbiol. 75, 89–97. doi: 10.1016/S0168-1605(01)00740-1
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Shabala, L., McMeekin, T., Budde, B. B., and Siegumfeldt, H. (2006). Listeria innocua and Lactobacillus delbrueckii subsp. bulgaricus employ different strategies to cope with acid stress. Int. J. Food Microbiol. 110, 1–7. doi: 10.1016/j.ijfoodmicro.2006.01.026
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Siegumfeldt, H., Rechinger, K. B., and Jakobsen, M. (1999). Use of fluorescence ratio imaging for intracellular pH determination of individual bacterial cells in mixed cultures. Microbiology 145(Pt 7), 1703–1709. doi: 10.1099/13500872-145-7-1703
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Smigic, N., Rajkovic, A., Nielsen, D. S., Siegumfeldt, H., Uyttendaele, M., Devlieghere, F., et al. (2009). Intracellular pH as an indicator of viability and resuscitation of Campylobacter jejuni after decontamination with lactic acid. Int. J. Food Microbiol. 135, 136–143. doi: 10.1016/j.ijfoodmicro.2009.07.023
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Smith, J. L., Liu, Y. H., and Paoli, G. C. (2013). How does Listeria monocytogenes combat acid conditions? Can. J. Microbiol. 59, 141–152. doi: 10.1139/cjm-2012-0392
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
te Giffel, M. C., and Zwietering, M. H. (1999). Validation of predictive models describing the growth of Listeria monocytogenes. Int. J. Food Microbiol. 46, 135–149. doi: 10.1016/S0168-1605(98)00189-5
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Tessema, G. T., Moretro, T., Snipen, L., Heir, E., Holck, A., Naterstad, K., et al. (2012). Microarray-based transcriptome of Listeria monocytogenes adapted to sublethal concentrations of acetic acid, lactic acid, and hydrochloric acid. Can. J. Microbiol. 58, 1112–1123. doi: 10.1139/w2012-091
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Van Schaik, W., and Abee, T. (2005). The role of sigma(B) in the stress response of Gram-positive bacteria - targets for food preservation and safety. Curr. Opin. Biotechnol. 16, 218–224. doi: 10.1016/j.copbio.2005.01.008
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Vazquez-Boland, J. A., Kuhn, M., Berche, P., Chakraborty, T., Dominguez-Bernal, G., Goebel, W., et al. (2001). Listeria pathogenesis and molecular virulence determinants. Clin. Microbiol. Rev. 14, 584–640. doi: 10.1128/CMR.14.3.584-640.2001
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Wiedmann, M., Arvik, T. J., Hurley, R. J., and Boor, K. J. (1998). General stress transcription factor sigmaB and its role in acid tolerance and virulence of Listeria monocytogenes. J. Bacteriol. 180, 3650–3656.
Keywords: Listeria monocytogenes, acid tolerance, intracellular pH, SigB, pH homeostasis
Citation: Cheng C, Yang Y, Dong Z, Wang X, Fang C, Yang M, Sun J, Xiao L, Fang W and Song H (2015) Listeria monocytogenes varies among strains to maintain intracellular pH homeostasis under stresses by different acids as analyzed by a high-throughput microplate-based fluorometry. Front. Microbiol. 6:15. doi: 10.3389/fmicb.2015.00015
Received: 02 December 2014; Accepted: 07 January 2015;
Published online: 23 January 2015.
Edited by:
Yi-Cheng Sun, Chinese Academy of Medical Sciences and Peking Union Medical College, ChinaReviewed by:
Claudia Guldimann, Cornell University, USAChaomin Sun, Chinese Academy of Science, China
Jianrong Li, The Ohio State University, USA
Copyright © 2015 Cheng, Yang, Dong, Wang, Fang, Yang, Sun, Xiao, Fang and Song. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Houhui Song, College of Animal Science and Technology, Zhejiang A&F University, 88 Huanbei Road, Lin'an, Zhejiang 311300, China e-mail:c29uZ2hoQHphZnUuZWR1LmNu
†These authors have contributed equally to this work.
 Changyong Cheng
Changyong Cheng Yongchun Yang
Yongchun Yang Zhimei Dong
Zhimei Dong Xiaowen Wang
Xiaowen Wang Chun Fang
Chun Fang Menghua Yang
Menghua Yang Jing Sun
Jing Sun Liya Xiao
Liya Xiao Weihuan Fang
Weihuan Fang Houhui Song
Houhui Song