- 1Division of Infectious Diseases, Department of Medicine, Taipei Veterans General Hospital, Taipei, Taiwan
- 2School of Medicine, National Yang-Ming University, Taipei, Taiwan
Klebsiella pneumoniae is an important cause of community-onset pneumonia in Asian countries and South Africa. We investigated the clinical characteristics of K. pneumoniae causing community-onset pneumonia, and the associated microbiological features between K. pneumoniae isolates from pneumonia and those from the nasopharynx in Taiwan. This study was conducted at the Taipei Veterans General Hospital during July, 2012 to February, 2014. The clinical characteristics in patients with community-onset K. pneumoniae pneumonia were analyzed. K. pneumoniae isolates from the nasopharynx of adults attending otorhinolaryngology outpatient clinics were collected to compare their microbiological features with those from pneumonia. Capsular genotypes, antimicrobial susceptibility, and multilocus sequence type (MLST) were determined among these strains. Ninety-one patients with community-onset K. pneumoniae pneumonia were enrolled. We found a high mortality (29.7%) among these patients. Capsular types K1, K2, K5, K20, K54, and K57 accounted for ∼70% of the K. pneumoniae isolates causing pneumonia, and ∼70% of all the K. pneumoniae strains isolated from the nasopharynx of patients in outpatient clinics. The MLST profiles further demonstrated the genetic relatedness between most pneumonia isolates and those from the nasopharynx. In conclusion, our results show that community-onset pneumonia caused by K. pneumoniae was associated with high mortality and could have a reservoir in the nasopharynx. To tackle this high-mortality disease, the distribution of capsular types in the nasopharynx might have implications for future vaccine development.
Introduction
Klebsiella pneumoniae is recognized 100 years ago as a potential cause of community-acquired pneumonia (CAP). However, over the past few decades, K. pneumoniae has been a rare cause of CAP in North America, Europe, and Australia (Shon et al., 2013). By contrast, it is an important cause of lower respiratory tract infection (LRTI) in Asian countries and South Africa and is associated with high mortality (Wang et al., 2005; Yu et al., 2007; Song et al., 2008; Lin et al., 2010a,b). In one prospective surveillance study from eight Asian countries, Streptococcus pneumoniae (29.2%) was the most common bacterial pathogen, followed by K. pneumoniae (15.4%; Song et al., 2008). In Cambodia, K. pneumoniae accounted for 8% of community-acquired LRTI (Rammaert et al., 2012). A previous study in an island in the Indian Ocean 2,500 km east of the South African coast demonstrated that S. pneumoniae and K. pneumoniae (42 and 22%, respectively) were the most frequent microbial agents in CAP patients admitted to intensive care units (Paganin et al., 2004).
In Western countries, although K. pneumoniae is a rare cause of CAP, one recent study highlighted that CAP caused by K. pneumoniae was associated with increased risk of cardiovascular events following this infection (Griffin et al., 2013). Recently, the emergence of fatal bacteremic CAP due to K. pneumoniae was reported in France (Decre et al., 2011; Rafat et al., 2013). This suggests that the re-emergence of severe K. pneumoniae pulmonary infections in Western countries cannot be overlooked.
Klebsiella pneumoniae, especially the virulent capsular types K1 and K2, has emerged as the major cause of community-acquired liver abscess in the past three decades in East Asia, especially Taiwan (Fung et al., 2002; Fang et al., 2004; Lin et al., 2013). However, the clinical features and microbiological characteristics of K. pneumoniae causing community-onset pneumonia have rarely been addressed (Lin et al., 2010a,b). The pathogenesis of K. pneumoniae causing community-onset pneumonia is unknown and nasopharyngeal colonization may precede pneumonia.
In the current study, we investigated the clinical features of community-onset pneumonia caused by K. pneumoniae in our hospital. We further compared the microbiological characteristics between K. pneumoniae strains from the nasopharynx in adult patients attending otorhinolaryngology outpatient clinics and those from community-onset pneumonia.
Materials and Methods
Study Design and Patients
A retrospective study was carried out in the Taipei Veterans General Hospital from July, 2012 to February, 2014. All consecutive adult patients (aged > 20 years) admitted with community-onset pneumonia due to K. pneumoniae were included. Community-onset pneumonia refers to both CAP and healthcare-associated pneumonia (HCAP). Polymicrobial infections were excluded in this study. CAP, HCAP, and hospital-acquired pneumonia were diagnosed based on the criteria previously described (Maruyama et al., 2013). HCAP included patients with pneumonia and any of the following: (1) hospitalization for 2 days during the preceding 90 days; (2) residence in a nursing home or extended care facility; (3) home infusion therapy (including antibiotics); (4) chronic dialysis (including haemodialysis and peritoneal dialysis) during the preceding 30 days; or (5) home wound care. Taiwan is endemic for K. pneumoniae liver abscess and cases of liver abscess with septic lung metastasis were also excluded.
We also consecutively collected K. pneumoniae isolates from the nasopharyngeal region in adult patients (aged > 20 years) attending otorhinolaryngology outpatient clinics from July, 2013 to February, 2014 to determine their clinical and microbiological characteristics. The main diagnosis of patients attending otorhinolaryngology outpatient clinics during this study period were acute sinusitis, chronic sinusitis, and allergic rhinitis. The protocol was approved by the hospital’s institutional review board.
Data Collection and Outcome Measures
The following data were collected on admission to hospital: age, sex, comorbid illnesses, clinical symptoms, concomitant bacteremia, and antimicrobial treatment. The Pneumonia Severity Index (PSI) was determined in all patients (Fine et al., 1997). In this observational study, we focused on the following endpoints: 28-day and in-hospital mortality, receiving mechanical ventilation during hospitalization and requiring intensive care.
Microbiological Evaluation
The Vitek 2 automated system (bioMérieux, Marcy l’Etoile, France) was used for bacterial identification and antimicrobial susceptibility testing. The following antimicrobial agents were assayed: ampicillin, cefazolin, cefuroxime, cefoxitin, ceftriaxone, ceftazidime, cefepime, piperacillin–tazobactam, ertapenem, imipenem, amikacin, gentamicin, ciprofloxacin, levofloxacin, trimethoprim–sulfamethoxazole, and tigecycline. To determine the capsular genotypes of K. pneumoniae, we undertook cps genotyping by the polymerase chain reaction detection of K-serotype-specific alleles at wzy loci, including serotypes K1, K2, K5, K20, K54, and K57, as described previously (Fang et al., 2007). Other capsular types were determined by a simple and useful capsular genotyping method for K. pneumoniae based on wzc sequences as described previously (Pan et al., 2013). rmpA (regulator of the mucoid phenotype), a gene known as an extracapsular polysaccharide synthesis regulator, can positively control the mucoid phenotype of K. pneumoniae. The presence of rmpA was proposed as an important virulence factor (Yu et al., 2006). One recent study showed that plasmid rmpA genes could be co-inherited together with the adjacent virulence genes carried by a large plasmid in K. pneumoniae (Hsu et al., 2011). Plasmid rmpA and rmpA2 genes were determined as described previously (Hsu et al., 2011).
Multi-locus sequence type (MLST) was performed on all isolates according to the protocol described on the K. pneumoniae MLST website (http://www.pasteur.fr/recherche/genopole/PF8/mlst/). MLST results were analyzed using the international K. pneumoniae MLST database created in 2005 at the Pasteur Institute in Paris, France (Diancourt et al., 2005).
Statistical Analysis
The χ2 test or Fisher’s exact test were used in the comparison of categorical data, and the Student’s t-test or Mann–Whitney U test were used for the comparison of continuous variables. Univariate and multivariate logistic regression analyses were performed to predict 28-day mortality. Variables that showed a significant difference (P < 0.1) in the univariate analysis were included in the forward likelihood ratio, stepwise multivariate logistic regression model to determine if any of them were independently related to outcome. Analysis was performed using SPSS version 19.0 (SPSS Inc., Chicago, IL, USA) with P < 0.05 considered statistically significant.
Results
Clinical Characteristics of Patients with Community-Onset K. Pneumoniae Pneumonia and Risk Factors for Mortality
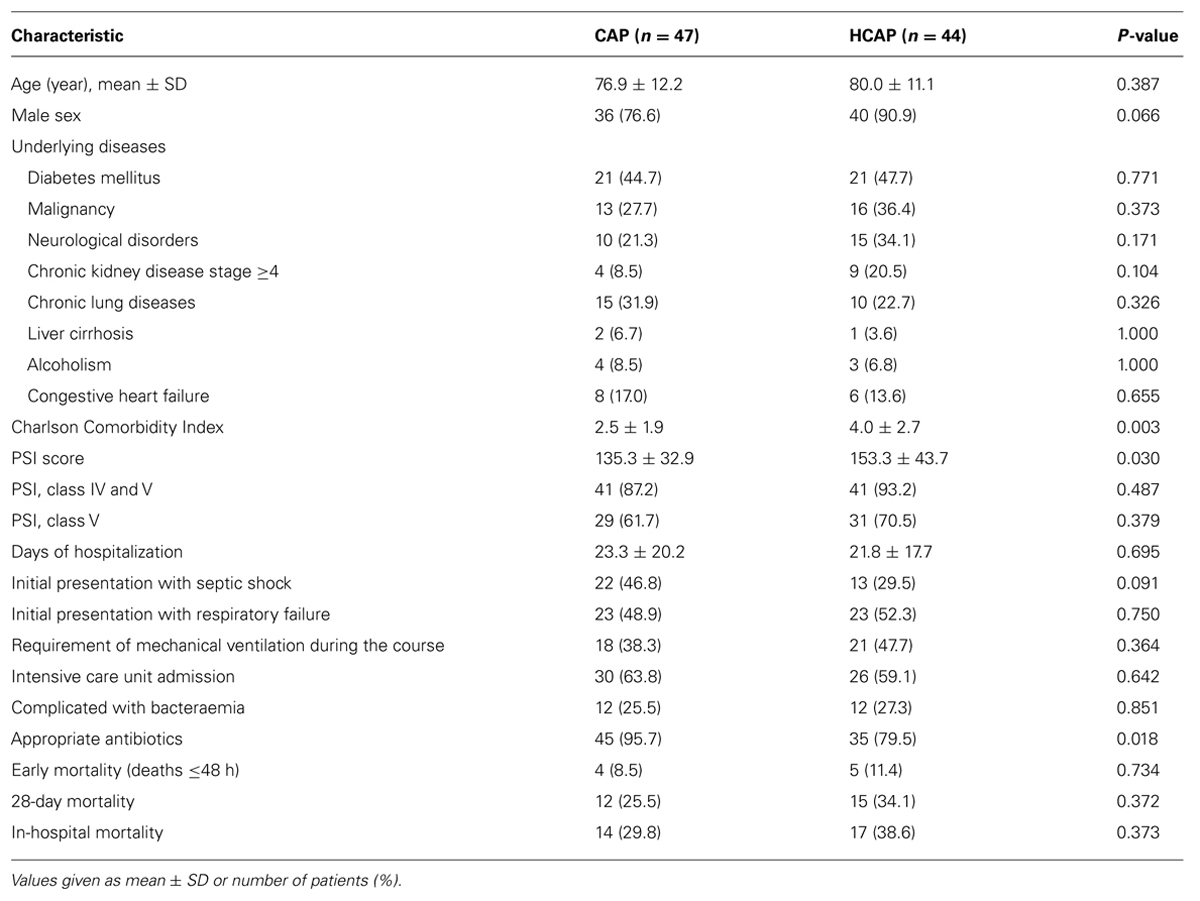
During the study period, a total of 191 consecutive patients with monomicrobial K. pneumoniae pneumonia were identified. Of these patients, 91 (47.6%) were classified as having community-onset infection, and the remaining 100 (52.6%) were classified as having hospital-acquired infection. Among the 91 patients diagnosed with community-onset K. pneumoniae pneumonia, there was a male predominance (n = 76) and the mean age was 77.9 ± 11.7 years. CAP accounted for 51.6% (n = 47) of the patients. None of the cases had concomitant distant abscesses. The 28-day mortality was 29.7% (27 patients). The overall in-hospital mortality was 34.1% (31 patients). Table 1 lists the clinical features of CAP and HCAP. HCAP patients had a significantly higher Charlson Comorbidity Index and PSI scores than CAP patients. CAP patients had a higher level of appropriate antibiotic use. Both the 28-day mortality and in-hospital mortality were not different statistically between CAP and HCAP.
Microbiological Characteristics of K. Pneumoniae from Patients with Community-Onset Pneumonia
Table 2 shows the distribution of capsular type among K. pneumoniae isolates. Capsular types K1, K2, K5, K20, K54, and K57 accounted for 67% (61/91) of all the K. pneumoniae isolates. During the same collection period for K. pneumoniae isolates from outpatients, the rate of capsular types K1, K2, K5, K20, K54, and K57 was consistent (72%, 31/43). Plasmid rmpA and rmpA2 genes were identified in 65 and 63 isolates, respectively. All but two strains belonging to the six capsular types (K1, K2, K5, K20, K54, and K57) carried the plasmid rmpA or rmpA2 genes. Seventy-one of the 91 (78%) isolates had the wild-type antibiotic susceptibility (susceptible to several classes of antibiotics, except for intrinsic resistance to ampicillin). MLST profiles were further determined in the isolates with capsular types K1, K2, K5, K20, K54, and K57 (Table 3).
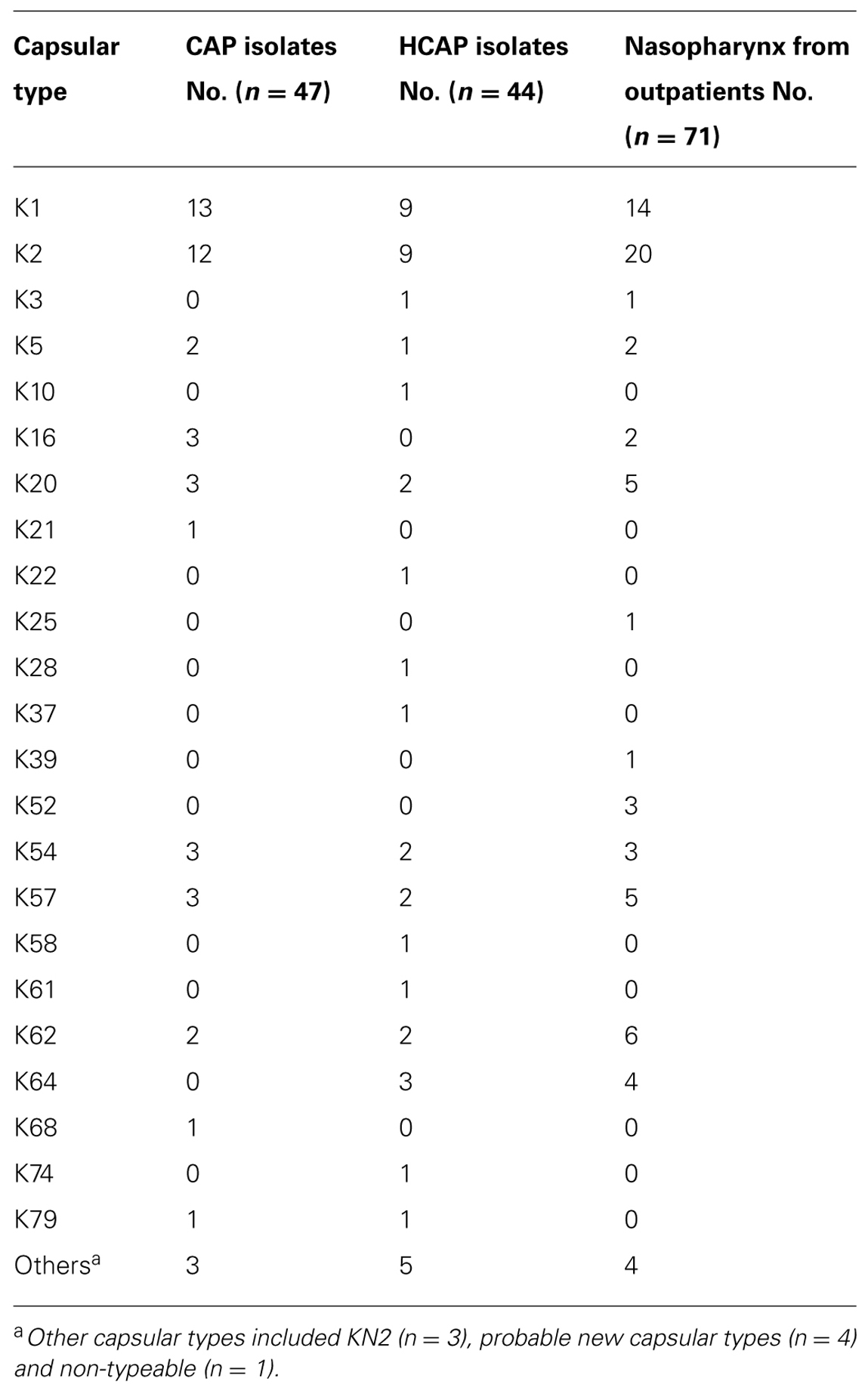
TABLE 2. Capsular type distribution among K. pneumoniae isolates collected from patients with community-onset pneumonia and nasopharynx from outpatients.
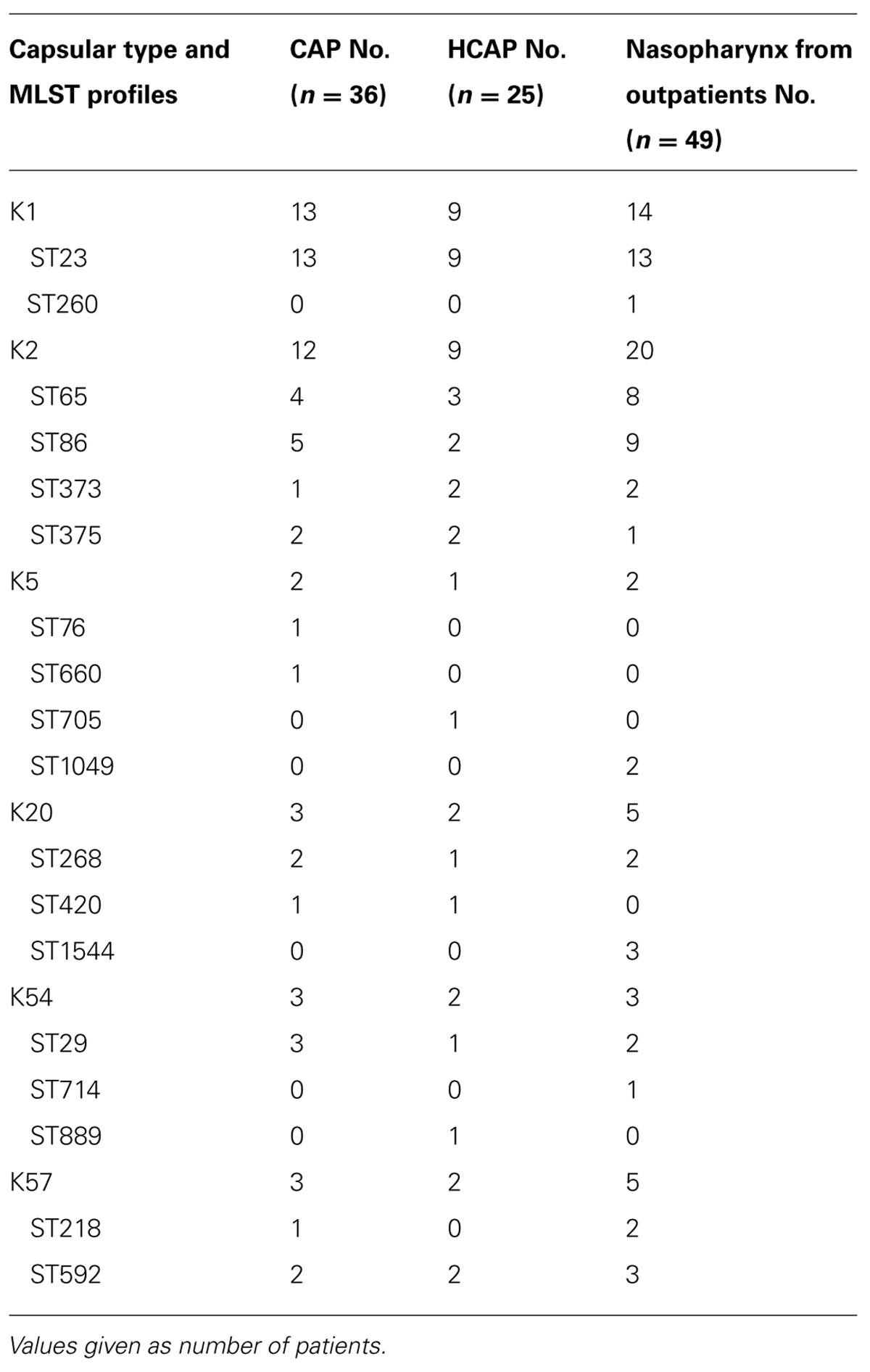
TABLE 3. MLST profiles among K. pneumoniae isolates collected from patients with community-onset pneumonia and nasopharynx from outpatients.
Capsular Type of K. Pneumoniae Isolated From Nasopharynx in Outpatients
Seventy-one isolates from the nasopharyngeal region collected consecutively in otorhinolaryngology outpatient clinics were available for capsular genotyping. Of the 71 patients (45 were male), the mean age was 55.3 ± 15.6 years. Most patients (57/71, 80.2%) were healthy and no underlying diseases were documented. Diabetes mellitus (6/14, 42.9%) and malignancy (6/14, 42.9%) were the most common underlying diseases among the other 14 patients. Most of them did not receive any antibiotic in the visit. K. pneumoniae accounted for 11.5% (39/340) of all bacteria isolated from nasopharyngeal region from November, 2013 to February, 2014.
The detailed capsular types are shown in Table 2. Capsular types K1, K2, K5, K20, K54, and K57 accounted for ∼70% (n = 49) of all the K. pneumoniae isolates. Plasmid rmpA and rmpA2 genes were identified in 55 and 52 isolates, respectively. All K. pneumoniae isolates that belonged to these six capsular types carried plasmid rmpA or rmpA2 genes. Sixty-eight K. pneumoniae isolates showed the wild-type antibiotic susceptibility. Among the other three isolates, one showed an extended-spectrum β-lactamase phenotype, and the other two showed resistance to fluoroquinolones. The detailed MLST profiles in the capsular types K1, K2, K5, K20, K54, and K57 isolates are shown in Table 3. ST23, ST65, ST86, ST373, ST375, ST705, ST268, ST29, ST218, and ST592 were found in isolates from both pneumonia and the nasopharynx.
Comparison of Microbiological Characteristics Among K. Pneumoniae Isolates Collected From Patients with Community-Onset Pneumonia and Outpatients
Regarding the microbiological characteristics (Table 4), the proportion of capsular type K1, K2, K5, K20, K54, and K57, wild-type antibiotic susceptibility and presence of plasmid rmpA/A2 were significantly higher in isolates from CAP than those from HCAP. In other words, 8.5% of CAP, 36.4% of HCAP, and 4.2% of outpatients isolates showed multidrug resistance. The characteristics were similar between isolates from CAP and outpatients. However, the proportion of wild-type antibiotic susceptibility and presence of plasmid rmpA/A2 were significantly higher in isolates from outpatients than those from HCAP.
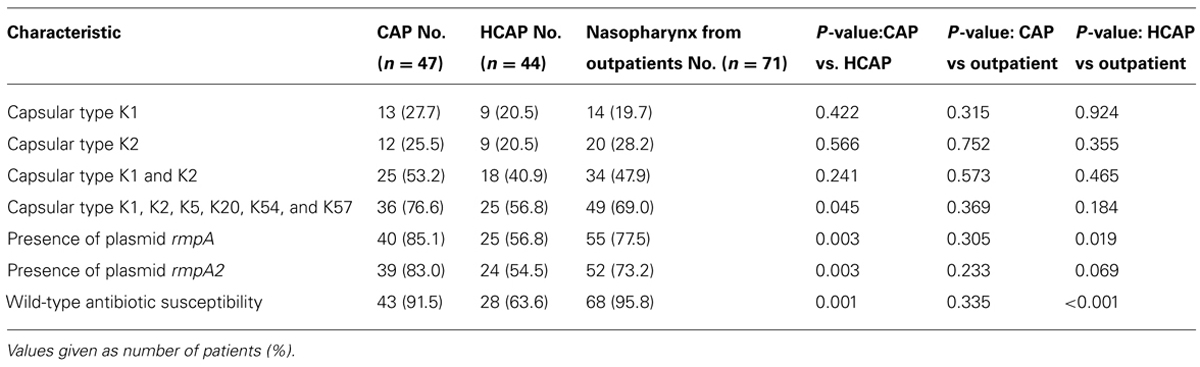
TABLE 4. Comparison of microbiological characteristics among K. pneumoniae isolates collected from patients with community-onset pneumonia and nasopharynx from outpatients.
Risk Factors for Mortality
Univariate analyses of the factors associated with 28-day mortality are shown in Table 5. The microbiological characteristics of K. pneumoniae were not associated with mortality. The logistic regression model showed that malignancy [odds ratio (OR) 4.08, 95% confidence interval (CI) 1.32–12.66, P = 0.015) and PSI scores (OR 1.017, 95% CI 1.001–1.034, P = 0.042) were independent risk factors for 28-day mortality. Initial presentation with respiratory failure (OR 3.38, 95% CI 0.99–11.44, P = 0.051) showed borderline significance in predicting mortality.
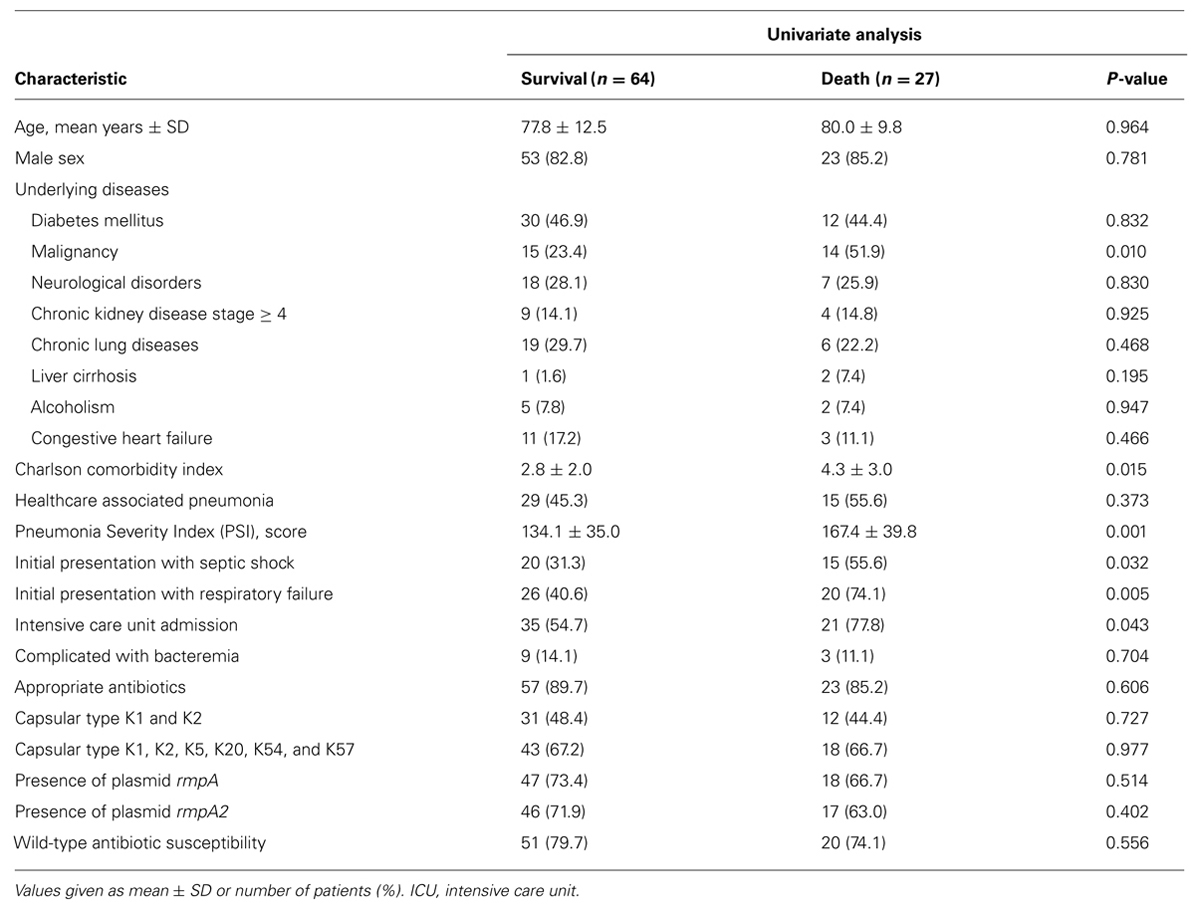
TABLE 5. Factors associated with 28-day mortality in adult patients with community-acquired pneumonia (CAP) caused by K. pneumoniae.
Discussion
The present study demonstrated the clinical features of community-onset K. pneumoniae pneumonia as well as the distribution of capsular types and MLST of K. pneumoniae in a medical center in Taiwan. We found a high mortality (29.7%) among these patients. Capsular types K1, K2, K5, K20, K54, and K57 accounted for ∼70% of all the K. pneumoniae isolates from patients with pneumonia, and ∼70% of K. pneumoniae isolates from the nasopharynx in adults attending otorhinolaryngology outpatient clinics. The MLST profiles further demonstrated several clones common in both pneumonia isolates and those from the nasopharynx.
Only one study has investigated a series of K. pneumoniae LRTIs and documented the high fatality rate (15/40, 37.5%) in Cambodia (Rammaert et al., 2012). The current study demonstrated community-onset pneumonia with K. pneumoniae had high mortality (29.7%) despite being in a developed country with well-equipped healthcare facilities. Generally, the 30-day mortality rate of CAP is 7.3% in Asia (Song et al., 2008). The mortality rate of overall CAP is 8.3% in Taiwan (Lauderdale et al., 2005). The high mortality caused by K. pneumoniae suggested that the virulent strains played an important role in pneumonia. Our previous studies showed high mortality in bacteremic CAP caused by K. pneumoniae (Lin et al., 2010b). The current study demonstrated that community-onset K. pneumoniae pneumonia is a critical problem in the modern era in endemic areas.
The capsule is a major virulence factor of K. pneumoniae, and some capsular types are related to the invasive syndromes (Chung et al., 2012; Siu et al., 2012). The distribution of capsular types in each K. pneumoniae-related disease could be crucial for disease control and prevention. We determined the capsular types of K. pneumoniae from community-onset pneumonia. We found that capsular types K1, K2, K5, K20, K54, and K57 were prevalent in community-onset pneumonia, which have been reported as the major types for liver abscess in Taiwan (Fang et al., 2007). Most of the applications of MLST for K. pneumoniae were for drug-resistant strains and only a few studies have applied MLST to virulent strains (capsular type K1 or K2; Siu et al., 2011; Liao et al., 2014). In the current study, we analyzed the MLST among the virulent capsular types (K1, K2, K5, K20, K54, and K57) from pneumonia, and we found several ST types were prevalent in pneumonia isolates. These virulent clones and the associated increased epidemic potential may be associated with the spread in the community in endemic areas. In western countries, investigations about capsular types of K. pneumoniae have rarely been reported. One large-scale study conducted in Europe and North America several decades ago has found that K2 represented the most common types (8.9%) among bacteremic isolates (Cryz et al., 1986). This suggest that capsular K2 type K. pneumoniae may be the global threat in this invasive disease.
Patients with HCAP had more exposure to healthcare environments or procedures, and the isolates may have the multidrug resistant problems. However, the virulence factors between CAP and HCAP caused by K. pneumoniae have never been compared. In this study, HCAP patients had a significantly higher Charlson Comorbidity Index and PSI score than CAP patients had. However, the number of capsular type K1, K2, K5, K20, K54, and K57, and wild-type antibiotic susceptibility strains were more common in CAP than HCAP. Overall, most of the isolates in this study showed wild-type antibiotic susceptibility. Our previous study on community-onset K. pneumonia bacteremia also displayed the prevalent wild-type antibiotic susceptibility isolates (Wu et al., 2012). Although the virulent but less drug-resistant isolates were rarely identified in western countries, they were indeed widespread in Taiwan and Asia countries. It is the remarkable features of K. pneumoniae isolates in Taiwan. These virulent capsular types are considered to be closely associated with community-acquired invasive disease or pathogenicity, and are rarely identified in nosocomial strains (Turton et al., 2010; Lin et al., 2014). We found that capsular type K1/K2, traditionally known as the most virulent strains of K. pneumoniae types, accounted for 40% of cases of HCAP. We cannot ignore the possible risk of progression to nosocomial spread of these invasive strains, which might result in severe therapeutic problems in immunocompromised hosts.
In our previous study, we suggested that the route of entry in liver abscess is from the gastrointestinal tract (Fung et al., 2012; Lin et al., 2013). We also indicated that none of 49 patients with bacteremic CAP caused by K. pneumoniae showed development of K. pneumoniae liver abscess concomitantly (Lin et al., 2010b). The pathogenesis of pneumonia and liver abscess may be different and the gastrointestinal tract carriage may not be related to pneumonia. Pharyngeal carriage of K. pneumoniae was considered to potentially play a role in the pathogenesis of K. pneumoniae pneumonia in recent studies from Asia (Farida et al., 2013; Dao et al., 2014). Farida et al. have shown that the carriage rate of K. pneumoniae in healthy individuals in Semarang, Indonesia, exceeds that of S. pneumoniae in adults (Farida et al., 2013). Almost all K. pneumoniae strains have the wild-type antibiotic susceptibility to common antimicrobial agents; similar to those found in CAP patients in Semarang, Indonesia (Farida et al., 2013). In our findings, most patients from outpatient departments were healthy and had no underlying diseases. Therefore, the findings may represent the colonization status and distribution of capsular types in the general population. Consistent with the recent study conducted by Farida et al. (2013), nearly all the strains from the nasopharynx in our study showed wild-type antibiotic susceptibility. The high prevalence of virulent capsular types (K1, K2, K5, K20, K54, and K57) in the community setting may correspond to the isolates from community-onset pneumonia. The MLST profiles further demonstrated the genetic relatedness between pneumonia isolates and those from the nasopharynx. Several ST types, such as ST23, ST65, and ST86, were prevalent in both pneumonia and the nasopharynx. These findings are a first step in elucidating potential pathways leading to community-onset pneumonia with K. pneumoniae.
Our study was limited by its retrospective design. However, our data have important clinical implications because of the lack of capsular types from pneumonia and the nasopharyngeal region reported in the literature. The other limitation was that we could not explore the association between infection and colonization precisely because of the different groups studied. A future prospective study would give us the opportunity to detect colonization and infection by the same K. pneumoniae strain and the associated clinical features. Lastly, while most the patients from outpatient departments had no underlying diseases and did not receive antibiotics, they did have upper respiratory tract infections. Collecting nasopharyngeal samples from non-ill healthy adults could be the best method to investigate the nasopharyngeal carriage. Despite this limitation, we give an insight that community-onset pneumonia could have a reservoir in the nasopharynx, rather than being spread from specific sources.
In conclusion, we found a high mortality (29.7%) among community-onset K. pneumoniae pneumonia. Capsular types K1, K2, K5, K20, K54, and K57 accounted for ∼70% of all the K. pneumoniae isolates from patients with pneumonia, and ∼70% of K. pneumoniae isolates from the nasopharynx in adults attending otorhinolaryngology outpatient clinics. These six capsular types and wild-type antibiotic susceptibility strains were more common in CAP than HCAP. The MLST profiles further demonstrated the genetic relatedness between most pneumonia isolates and those from the nasopharynx. The similar microbiological characteristics imply that K. pneumoniae isolates in the nasopharynx play a role in the pathogenesis of pneumonia. The distribution of capsular types in the nasopharynx might have implications for future vaccine development.
Author Contributions
YTL contributed to study concept and design, and data analysis and interpretation; YTL and YPW contributed to data acquisition and manuscript drafting; FDW contributed to critical manuscript revision; and CPF contributed to the supervision of this investigation.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
This study was partly supported by grants from the Ministry of Science and Technology, Taiwan, Republic of China (MOST 103-2314-B-075-078-MY2), Taipei Veterans General Hospital (V103B-016 and V103C-127), and Szu-Yuan Research Foundation of Internal Medicine.
References
Chung, D. R., Park, M. H., Kim, S. H., Ko, K. S., Kang, C. I., Peck, K. R.,et al. (2012). Prevalence and molecular characterization of serotype K1 Klebsiella pneumoniae strains from various clinical specimen sources in 11 Asian countries. J. Infect. 64, 622–625. doi: 10.1016/j.jinf.2012.02.007
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Cryz, S. J. Jr, Mortimer, P. M., Mansfield, V., and Germanier, R. (1986). Seroepidemiology of Klebsiella bacteremic isolates, and implications for vaccine development. J. Clin. Microbiol. 23, 687–690.
Dao, T. T., Liebenthal, D., Tran, T. K., Ngoc Thi Vu, B., Ngoc Thi Nguyen, D., Thi Tran, H. K.,et al. (2014). Klebsiella pneumoniae oropharyngeal carriage in rural and urban Vietnam and the effect of alcohol consumption. PLoS ONE 9:e91999. doi: 10.1371/journal.pone.0091999
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Decre, D., Verdet, C., Emirian, A., Le Gourrierec, T., Petit, J. C., Offenstadt, G.,et al. (2011). Emerging severe and fatal infections due to Klebsiella pneumoniae in two university hospitals in France. J. Clin. Microbiol. 49, 3012–3014. doi: 10.1128/jcm.00676-11
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Diancourt, L., Passet, V., Verhoef, J., Grimont, P. A., and Brisse, S. (2005). Multilocus sequence typing of Klebsiella pneumoniae nosocomial isolates. J. Clin. Microbiol. 43, 4178–4182. doi: 10.1128/jcm.43.8.4178-4182.2005
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Fang, C. T., Chuang, Y. P., Shun, C. T., Chang, S. C., and Wang, J. T. (2004). A novel virulence gene in Klebsiella pneumoniae strains causing primary liver abscess and septic metastatic complications. J. Exp. Med. 199, 697–705. doi: 10.1084/jem.20030857
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Fang, C. T., Lai, S. Y., Yi, W. C., Hsueh, P. R., Liu, K. L., and Chang, S. C. (2007). Klebsiella pneumoniae genotype K1: an emerging pathogen that causes septic ocular or central nervous system complications from pyogenic liver abscess. Clin. Infect. Dis. 45, 284–293. doi: 10.1086/519262
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Farida, H., Severin, J. A., Gasem, M. H., Keuter, M., Van Den Broek, P., Hermans, P. W.,et al. (2013). Nasopharyngeal carriage of Klebsiella pneumoniae and other Gram-negative bacilli in pneumonia-prone age groups in Semarang, Indonesia. J. Clin. Microbiol. 51, 1614–1616. doi: 10.1128/jcm.00589-13
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Fine, M. J., Auble, T. E., Yealy, D. M., Hanusa, B. H., Weissfeld, L. A., Singer, D. E.,et al. (1997). A prediction rule to identify low-risk patients with community-acquired pneumonia. N. Engl. J. Med. 336, 243–250. doi: 10.1056/nejm199701233360402
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Fung, C. P., Chang, F. Y., Lee, S. C., Hu, B. S., Kuo, B. I., Liu, C. Y.,et al. (2002). A global emerging disease of Klebsiella pneumoniae liver abscess: is serotype K1 an important factor for complicated endophthalmitis? Gut 50, 420–424. doi: 10.1136/gut.50.3.420
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Fung, C. P., Lin, Y. T., Lin, J. C., Chen, T. L., Yeh, K. M., Chang, F. Y.,et al. (2012). Klebsiella pneumoniae in gastrointestinal tract and pyogenic liver abscess. Emerg. Infect. Dis. 18, 1322–1325. doi: 10.3201/eid1808.111053
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Griffin, A. T., Wiemken, T. L., and Arnold, F. W. (2013). Risk factors for cardiovascular events in hospitalized patients with community-acquired pneumonia. Int. J. Infect. Dis. 17, e1125–e1129. doi: 10.1016/j.ijid.2013.07.005
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Hsu, C. R., Lin, T. L., Chen, Y. C., Chou, H. C., and Wang, J. T. (2011). The role of Klebsiella pneumoniae rmpA in capsular polysaccharide synthesis and virulence revisited. Microbiology 157, 3446–3457. doi: 10.1099/mic.0.050336-0
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Lauderdale, T. L., Chang, F. Y., Ben, R. J., Yin, H. C., Ni, Y. H., Tsai, J. W.,et al. (2005). Etiology of community acquired pneumonia among adult patients requiring hospitalization in Taiwan. Respir. Med. 99, 1079–1086. doi: 10.1016/j.rmed.2005.02.026
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Liao, C. H., Huang, Y. T., Chang, C. Y., Hsu, H. S., and Hsueh, P. R. (2014). Capsular serotypes and multilocus sequence types of bacteremic Klebsiella pneumoniae isolates associated with different types of infections. Eur. J. Clin. Microbiol. Infect. Dis. 33, 365–369. doi: 10.1007/s10096-013-1964-z
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Lin, Y. T., Chen, T. L., Siu, L. K., Hsu, S. F., and Fung, C. P. (2010a). Clinical and microbiological characteristics of community-acquired thoracic empyema or complicated parapneumonic effusion caused by Klebsiella pneumoniae in Taiwan. Eur. J. Clin. Microbiol. Infect. Dis. 29, 1003–1010. doi: 10.1007/s10096-010-0961-8
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Lin, Y. T., Jeng, Y. Y., Chen, T. L., and Fung, C. P. (2010b). Bacteremic community-acquired pneumonia due to Klebsiella pneumoniae: clinical and microbiological characteristics in Taiwan, 2001-2008. BMC. Infect. Dis. 10:307. doi: 10.1186/1471-2334-10-307
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Lin, Y. T., Liu, C. J., Yeh, Y. C., Chen, T. J., and Fung, C. P. (2013). Ampicillin and amoxicillin use and the risk of Klebsiella pneumoniae liver abscess in Taiwan. J. Infect. Dis. 208, 211–217. doi: 10.1093/infdis/jit157
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Lin, Y. T., Wang, F. D., Chan, Y. J., Fu, Y. C., and Fung, C. P. (2014). Clinical and microbiological characteristics of tigecycline non-susceptible Klebsiella pneumoniae bacteremia in Taiwan. BMC. Infect. Dis. 14:1. doi: 10.1186/1471-2334-14-1
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Maruyama, T., Fujisawa, T., Okuno, M., Toyoshima, H., Tsutsui, K., Maeda, H.,et al. (2013). A new strategy for healthcare-associated pneumonia: a 2-year prospective multicenter cohort study using risk factors for multidrug-resistant pathogens to select initial empiric therapy. Clin. Infect. Dis. 57, 1373–1383. doi: 10.1093/cid/cit571
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Paganin, F., Lilienthal, F., Bourdin, A., Lugagne, N., Tixier, F., Genin, R.,et al. (2004). Severe community-acquired pneumonia: assessment of microbial aetiology as mortality factor. Eur. Respir. J. 24, 779–785. doi: 10.1183/09031936.04.00119503
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Pan, Y. J., Lin, T. L., Chen, Y. H., Hsu, C. R., Hsieh, P. F., Wu, M. C.,et al. (2013). Capsular types of Klebsiella pneumoniae revisited by wzc sequencing. PLoS ONE 8:e80670. doi: 10.1371/journal.pone.0080670
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Rafat, C., Fihman, V., and Ricard, J. D. (2013). A 51-year-old man presenting with shock and lower-lobe consolidation with interlobar bulging fissure. Chest 143, 1167–1169. doi: 10.1378/chest.12-2007
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Rammaert, B., Goyet, S., Beaute, J., Hem, S., Te, V., Try, P. L.,et al. (2012). Klebsiella pneumoniae related community-acquired acute lower respiratory infections in Cambodia: clinical characteristics and treatment. BMC. Infect. Dis. 12:3. doi: 10.1186/1471-2334-12-3
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Shon, A. S., Bajwa, R. P., and Russo, T. A. (2013). Hypervirulent (hypermucoviscous) Klebsiella pneumoniae: a new and dangerous breed. Virulence 4, 107–118. doi: 10.4161/viru.22718
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Siu, L. K., Fung, C. P., Chang, F. Y., Lee, N., Yeh, K. M., Koh, T. H.,et al. (2011). Molecular typing and virulence analysis of serotype K1 Klebsiella pneumoniae strains isolated from liver abscess patients and stool samples from noninfectious subjects in Hong Kong, Singapore, and Taiwan. J. Clin. Microbiol. 49, 3761–3765. doi: 10.1128/jcm.00977-11
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Siu, L. K., Yeh, K. M., Lin, J. C., Fung, C. P., and Chang, F. -Y. (2012). Klebsiella pneumoniae liver abscess: a new invasive syndrome. Lancet Infect. Dis. 12, 881–887. doi: 10.1016/S1473-3099(12)70205-0
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Song, J. H., Oh, W. S., Kang, C. I., Chung, D. R., Peck, K. R., Ko, K. S.,et al. (2008). Epidemiology and clinical outcomes of community-acquired pneumonia in adult patients in Asian countries: a prospective study by the Asian network for surveillance of resistant pathogens. Int. J. Antimicrob. Agents 31, 107–114. doi: 10.1016/j.ijantimicag.2007.09.014
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Turton, J. F., Perry, C., Elgohari, S., and Hampton, C. V. (2010). PCR characterization and typing of Klebsiella pneumoniae using capsular type-specific, variable number tandem repeat and virulence gene targets. J. Med. Microbiol. 59, 541–547. doi: 10.1099/jmm.0.015198-0
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Wang, J. L., Chen, K. Y., Fang, C. T., Hsueh, P. R., Yang, P. C., and Chang, S. C. (2005). Changing bacteriology of adult community-acquired lung abscess in Taiwan: Klebsiella pneumoniae versus anaerobes. Clin. Infect. Dis. 40, 915–922. doi: 10.1086/428574
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Wu, H. S., Wang, F. D., Tseng, C. P., Wu, T. H., Lin, Y. T., and Fung, C. P. (2012). Characteristics of healthcare-associated and community-acquired Klebsiella pneumoniae bacteremia in Taiwan. J. Infect. 64, 162–168. doi: 10.1016/j.jinf.2011.11.005
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Yu, V. L., Hansen, D. S., Ko, W. C., Sagnimeni, A., Klugman, K. P., Von Gottberg, A.,et al. (2007). Virulence characteristics of Klebsiella and clinical manifestations of K. pneumoniae bloodstream infections. Emerg. Infect. Dis. 13, 986–993. doi: 10.3201/eid1307.070187
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Yu, W. L., Ko, W. C., Cheng, K. C., Lee, H. C., Ke, D. S., Lee, C. C.,et al. (2006). Association between rmpA and magA genes and clinical syndromes caused by Klebsiella pneumoniae in Taiwan. Clin. Infect. Dis. 42, 1351–1358. doi: 10.1086/503420
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Keywords: capsular types, Klebsiella pneumoniae, MLST, nasopharynx, pneumonia
Citation: Lin Y-T, Wang Y-P, Wang F-D and Fung C-P (2015) Community-onset Klebsiella pneumoniae pneumonia in Taiwan: clinical features of the disease and associated microbiological characteristics of isolates from pneumonia and nasopharynx. Front. Microbiol. 6:122. doi: 10.3389/fmicb.2015.00122
Received: 25 December 2014; Accepted: 30 January 2015;
Published online: 18 February 2015.
Edited by:
Evangelos Giamarellos-Bourboulis, Medical School of University of Athens, GreeceReviewed by:
Alan S. Cross, University of Maryland School of Medicine, USAGeorge Dimopoulos, University Hospital Attikon, Greece
Copyright © 2015 Lin, Wang, Wang and Fung. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Yi-Tsung Lin, Division of Infectious Diseases, Department of Medicine, Taipei Veterans General Hospital, Number 201, Section 2, Shih-Pai Road, Beitou, Taipei 112, Taiwan e-mail:eXRsaW44QHZnaHRwZS5nb3YudHc=
 Yi-Tsung Lin
Yi-Tsung Lin Yu-Ping Wang
Yu-Ping Wang Fu-Der Wang1,2
Fu-Der Wang1,2 Chang-Phone Fung
Chang-Phone Fung